Abstract
Background
Enhanced Recovery After Surgery (ERAS) applies multimodal, perioperative, and evidence-based practices to decrease postoperative morbi-mortality, the length of hospital stay, and hospitalization costs. Implementing enhanced recovery after thoracic surgery (ERATS) in low- and middle-income countries (LMIC) is problematic. This randomized controlled trial evaluated the feasibility and effectiveness of an ERATS protocol adapted to LMIC conditions in Tunisia.
Materials and Methods
We conducted this randomized controlled trial between December 2015 and August 2017 in the Thoracic and Cardiovascular Surgery Department at Habib Bourguiba University Hospital of Sfax, Tunisia.
Results
One hundred patients undergoing thoracic surgery were randomly allocated to the ERATS group or Control group. During the postoperative phase, 13 patients (13%) were excluded secondary. These complication rates were lower in the ERATS group: lack of reexpansion (14.63% vs 16.10%: p = 0.72), pleural effusion (0% vs 10.86%, p = 0.05), and prolonged air leak (17.07% vs 30.43%, p = 0.14). The pain level decreased significantly in the ERATS group from postoperative H3 (p = 0.006). This difference was significant at H6 (p = 0.001), H24 (p = 0.05), H48 (p = 0.01), discharge (p = 0.002), and after 15 days (p = 0.01), with a decreased analgesic consumption. The length of hospital stay was shorter in the ERAS group (median six days vs seven days, p = 0.17).
Conclusion
This study provides an adapted ERATS protocol, applicable regardless of the surgical approach or the type of resection and suitable for LMIC hospital's conditions. This protocol can improve the postoperative outcomes of thoracic surgery.
Background
The World Health Assembly Resolution 68.15, based on the Lancet commission “Global surgery 2030,” 1 recommends an improvement of surgery and anesthesia in low- and middle-income countries (LMIC) to strengthen the healthcare system. 2 The implementation of Enhanced Recovery After Surgery (ERAS) was proposed as a key component of this strategy.3,4
Enhanced Recovery After Surgery is the application of multimodal, perioperative, and evidence-based practices to decrease postoperative morbi-mortality and the length of hospital stay and hospitalization costs. 5 In thoracic surgery, the ERAS Society and the European Society of Thoracic Surgeons (ESTS) guidelines for lung surgery describe 45 enhanced recovery items based on the best available and eligible articles. 6
The implementation of enhanced recovery after thoracic surgery (ERATS) in LMIC poses a dual challenge:
Even in high-income countries, ERATS faces many barriers to be applied.
7
Limited resources and poor infrastructure are additional crucial problems in LMIC.
8
Based on mainly operating lung cancer, ERATS guidelines focus on lobectomy.
7
The high incidence of infectious thoracic diseases in LMIC
9
may affect perioperative practices and results there.
Thus, extrapolating the feasibility and outcomes of these guidelines is problematic.
This randomized controlled trial evaluated the feasibility and effectiveness of an ERATS protocol adapted to LMIC conditions in Tunisia. 10
Materials and methods
Study design and subjects’ selection
The study protocol was designed under Clinical Trials requirements and registered under the identifier: NCT02831140 (Figure 1). 11 We conducted this randomized controlled trial between December 2015 and August 2017 in the Thoracic and Cardiovascular Surgery Department at Habib Bourguiba University Hospital of Sfax, Tunisia.
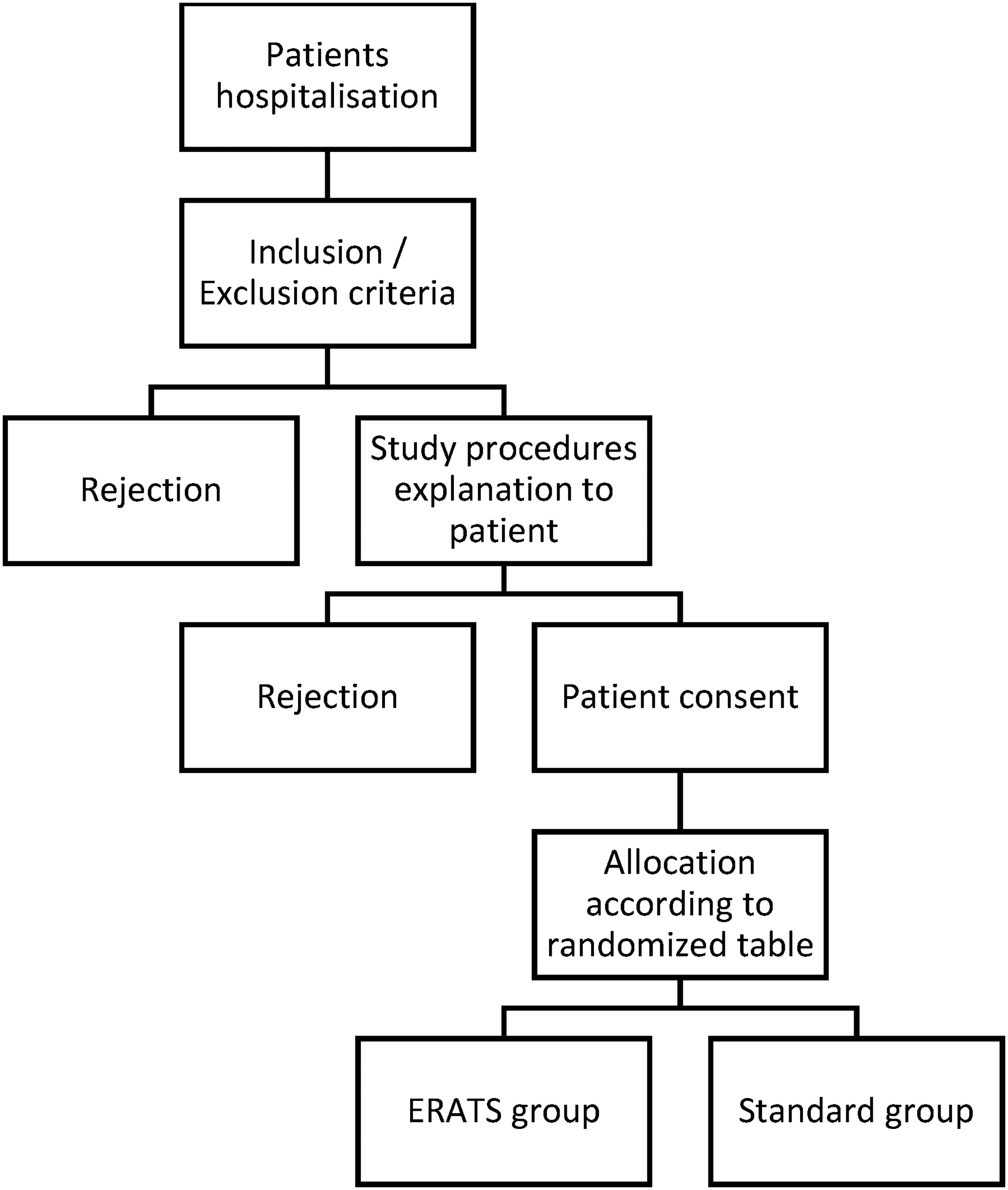
Study design and subjects’ selection.
We calculated the required subjects with Package epi R 0.9-30 software based on Muehling et al. pilot study outcomes. 12 Then, using Kendall and Babington Smith's table, 100 eligible patients have been randomly allocated to the “ERATS group” or “Control group.” After their consent, all programmed patients to thoracic surgery during the study period, aged between 18 and 80 years, were included and allocated to one study group according to randomized table order.
The exclusion criteria were:
A patient with Postural Status > 2, unable to walk, or requiring an intensive care unit (ICU) hospitalization. Soft tissue and lymph node biopsy thoracic procedures. Emergency thoracic surgery procedures
Intervention description
Common arm
At the preoperative, all patients had to stop smoking for two weeks at least with a balanced alimentation (Figure 2). Preoperative evaluation and decision-making were established according to ERS/ESTS guidelines. Fasting before the surgery was 6 hours.
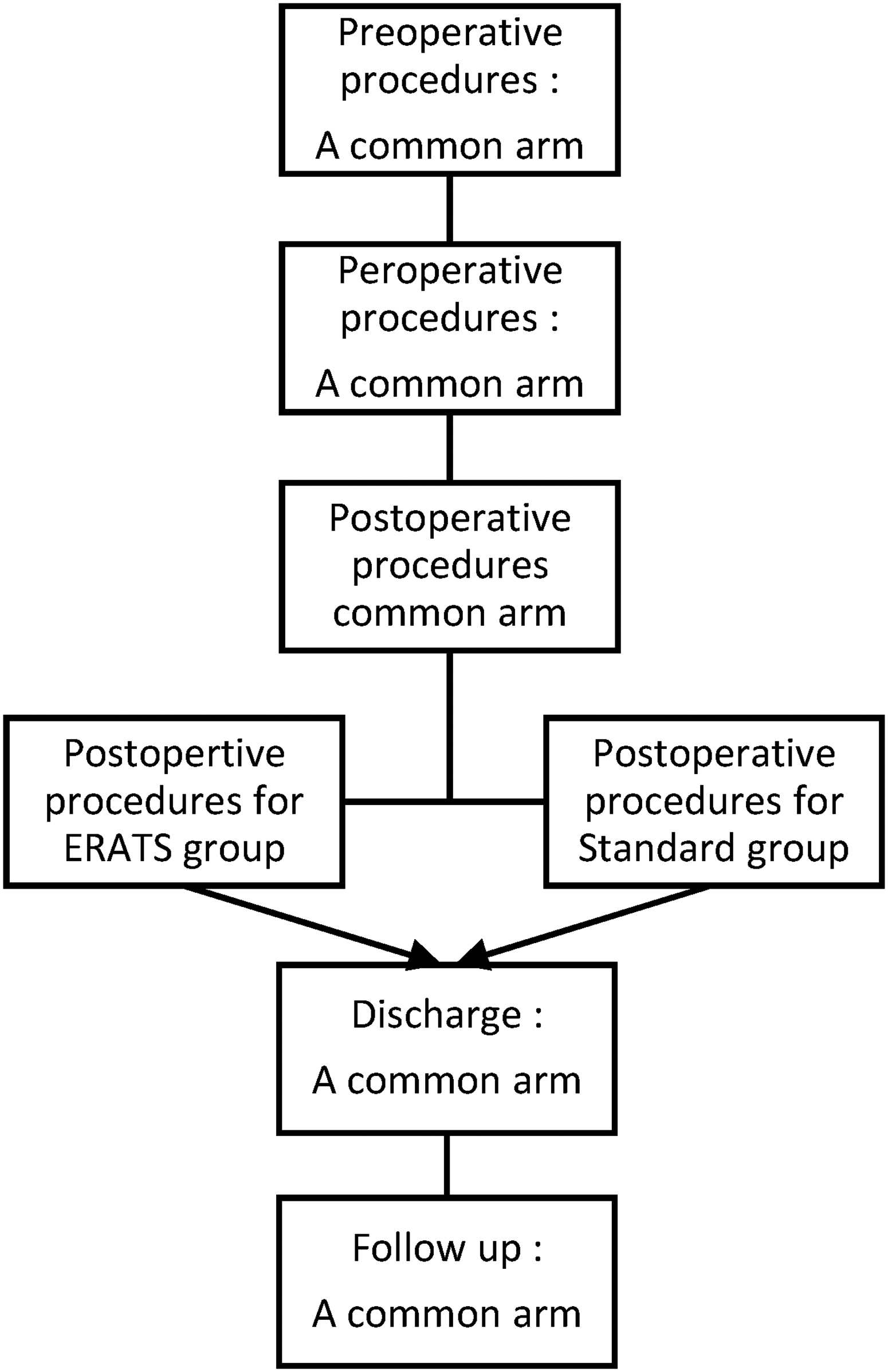
Study arms design.
At the preoperative, long-acting benzodiazepine drugs were not used within anesthesia. Mini-invasive surgical approaches were performed if possible (Video-Assisted Thoracic Surgery: VATS). For open surgical approaches, conservation of posterior chest wall muscles techniques was applied.
Locoregional analgesia techniques were used in addition to systematic drugs at the postoperative. Chest physiotherapy was started in the sixth hour and was performed daily. All patients were discharged after chest tube removal and recontrolled on day 15 postdischarge with a clinical examination and a chest X-ray.
Enhanced recovery after thoracic surgery group
At the postoperative, the ERATS group underwent early exercises. Within the first hour, the patient had to: set a half-bed position, deep breathing, cough, and move his arms and legs. In the second hour, after curbing vagal malaise, the patient had to perform relaxation movements: moving up the upper extremities accompanied by deep inspiration with a 2-s inspiratory apnea, followed by moving down the upper extremities accompanied by deep expiration. The amplitude of movements was increased progressively. Then, he stood up carefully near his bed. The patient started an early alimentation between the first and the second hour, beginning with liquids and then a regular diet if there was no vomiting. In the third hour, he had to start early mobilization by strolling for about 20–30 min under the control of the medical team to avoid his fall down or all other incidents. Thoracic surgery residents always performed this protocol.
Standard group
At the postoperative, the first get-up and alimentation permissions were in 24 h. The patient had to stay in bed, and we prohibited mobilization and food. Alimentation was maintained by filling with glucose serum for the first 24 h.
Outcomes and statistical analysis
The primary outcome measures were postoperative respiratory complications. Secondary outcome measures were: pain score (measured by the Visual Analog scale of pain Intensity VAS), drainage length, and postoperative hospital stay.
We used SPSS 20.0 software for data assessment. Observed numbers (N) and rates (%) expressed qualitative variables. Frequencies were compared by the chi 2 test of Pearson or the Fisher test when appropriate. Quantitative variables’ normality was assessed by Skewness & Kurtosis coefficients and the Shapiro–Wilk test. Then, data were expressed by the arithmetic mean (m) and the standard deviation (SD) for the Gaussian distribution or the median (M) and range (R[ ]) for the non-Gaussian distribution. Averages were compared by the Student's or Mann–Whitney tests, if appropriate. A p-value <0.05 was deemed significant.
Results
Patients’ profile and perioperative conditions
Among 100 included patients, 13 (13%) were excluded secondary during the postoperative: six noncooperative patients (refusing to continue the care procedure or not applying one or more elements from the postoperative protocol), five patients were transferred to the ICU; one case of angina stroke and one exploratory thoracotomy. There was no significant difference between the number of eliminated patients in the ERATS group and the Standard group (p = 0.13). Thus, 87 patients were definitively maintained until the study's endpoint: 46 patients (52.9%) in the Standard protocol versus 41 patients (47.1%) in the ERATS protocol.
Patients’ profile is summarized in Table 1. The mean age in both groups was 47.15 years (SD: 18.4; Range: 18–80), and the sex ratio was 1.48. The intellectual level of most patients was in secondary school (29.9%). No significant difference between ERATS and Standard groups was noted in these parameters.
Patients’ profile.
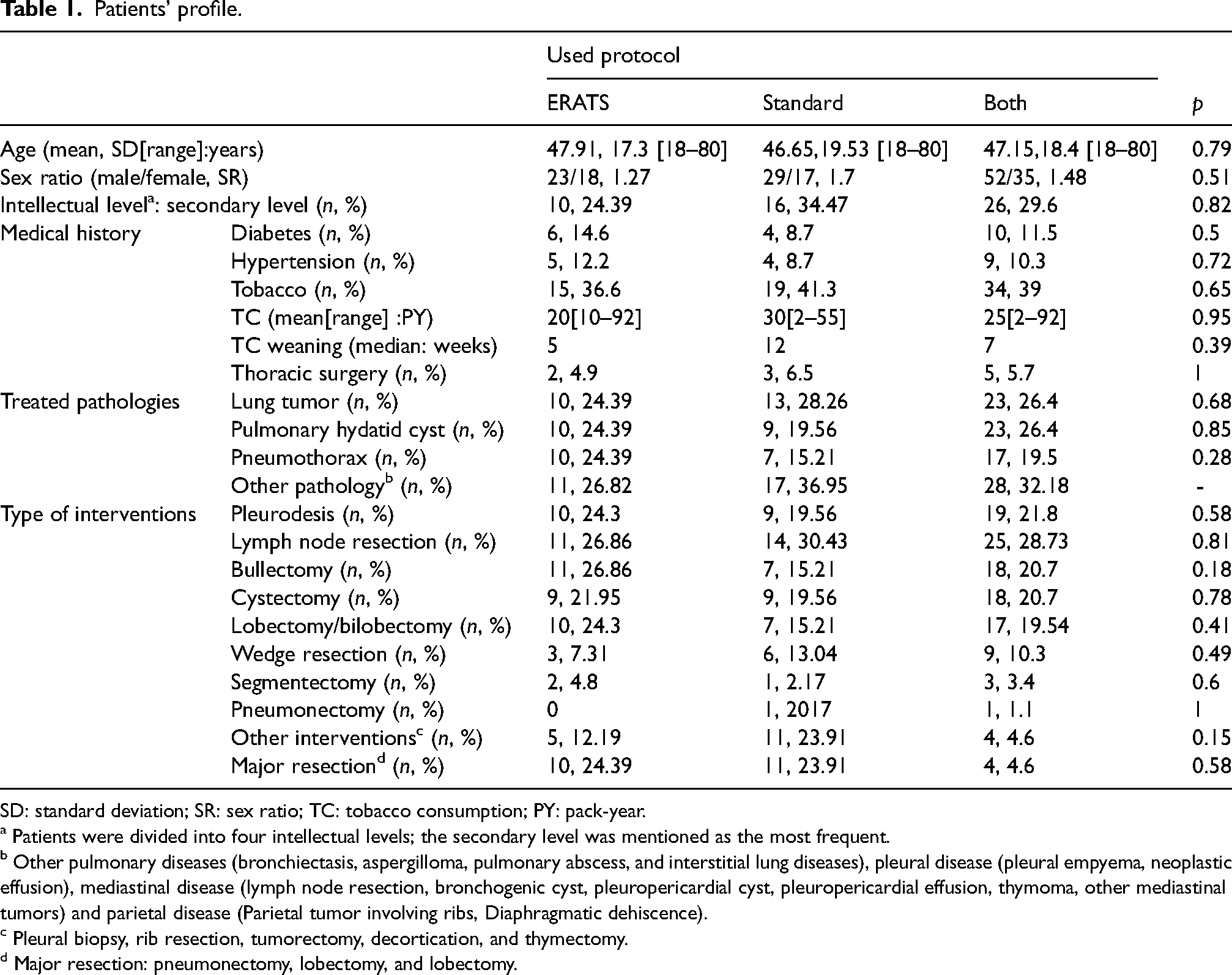
SD: standard deviation; SR: sex ratio; TC: tobacco consumption; PY: pack-year.
a Patients were divided into four intellectual levels; the secondary level was mentioned as the most frequent.
b Other pulmonary diseases (bronchiectasis, aspergilloma, pulmonary abscess, and interstitial lung diseases), pleural disease (pleural empyema, neoplastic effusion), mediastinal disease (lymph node resection, bronchogenic cyst, pleuropericardial cyst, pleuropericardial effusion, thymoma, other mediastinal tumors) and parietal disease (Parietal tumor involving ribs, Diaphragmatic dehiscence).
c Pleural biopsy, rib resection, tumorectomy, decortication, and thymectomy.
d Major resection: pneumonectomy, lobectomy, and lobectomy.
Also, patients ‘medical history, number, pathology, type of surgical procedure, and perioperative conditions’ were homogeneous between the two groups. Table 2 resumes assessed variables.
Perioperative conditions.
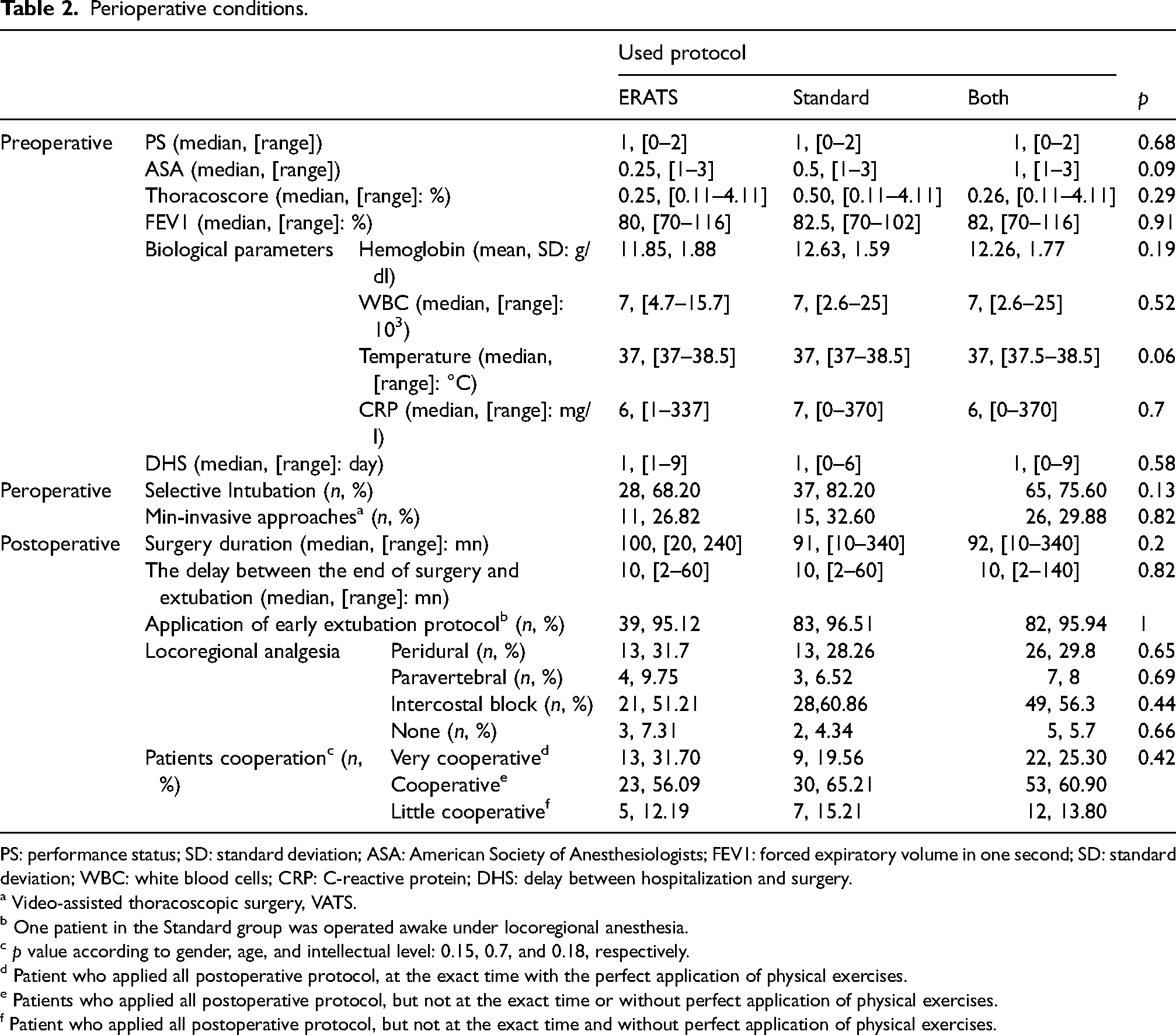
PS: performance status; SD: standard deviation; ASA: American Society of Anesthesiologists; FEV1: forced expiratory volume in one second; SD: standard deviation; WBC: white blood cells; CRP: C-reactive protein; DHS: delay between hospitalization and surgery.
a Video-assisted thoracoscopic surgery, VATS.
b One patient in the Standard group was operated awake under locoregional anesthesia.
c p value according to gender, age, and intellectual level: 0.15, 0.7, and 0.18, respectively.
d Patient who applied all postoperative protocol, at the exact time with the perfect application of physical exercises.
e Patients who applied all postoperative protocol, but not at the exact time or without perfect application of physical exercises.
f Patient who applied all postoperative protocol, but not at the exact time and without perfect application of physical exercises.
Judgment criteria results
Pneumonia, lack of reexpansion, prolonged air leak, and pleural effusion were the main complications, and they were more frequent in the Standard group (28.26% vs. 19.51% in the ERATS group, p = 0.41). Table 3 summarizes different rates with p-values. Even if there was no significant statistical difference when comparing these rates between the two groups, complications were always more frequent in the standard one (except pneumonia). Additionally, this difference was almost statistically significant regarding postoperative pleural effusion (p = 0.05).
Judgment criteria results.
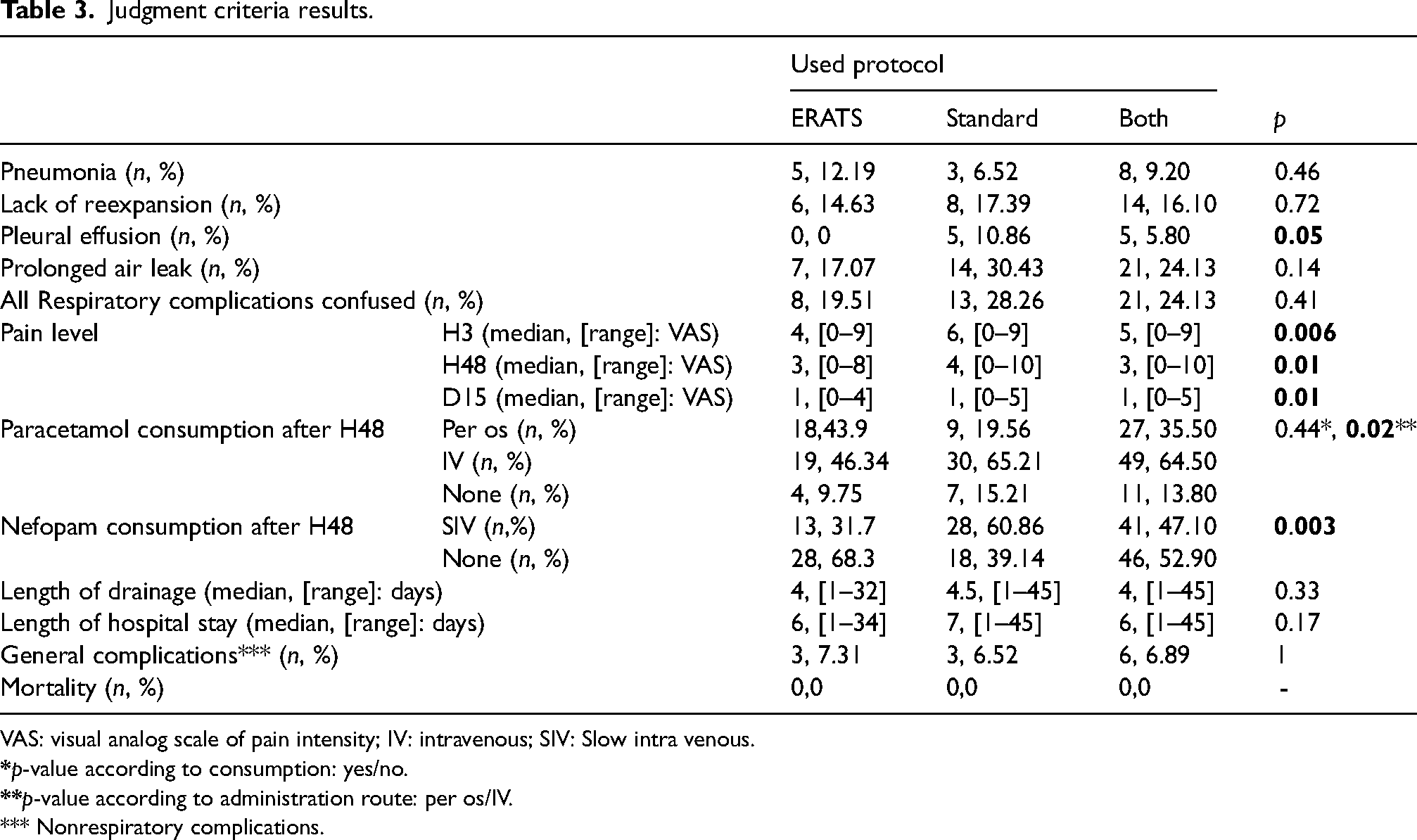
VAS: visual analog scale of pain intensity; IV: intravenous; SIV: Slow intra venous.
*** Nonrespiratory complications.
It is essential to mention that for patients who were operated on for infectious pathology (like an infected hydatid cyst), signs of pneumonia were not considered a postoperative complication considering its preoperative status.
However, it is remarkable that the difference in pain level between ERATS and Standard group becomes statistically significant from H3. At postoperative H1, patients had the same VAS in the two groups with a median of 7. From postoperative H3, VAS was decreased in the ERATS group with a significant p-value for the rest of the postoperative course. Figure 3 shows the median range and p-values at every evaluation.
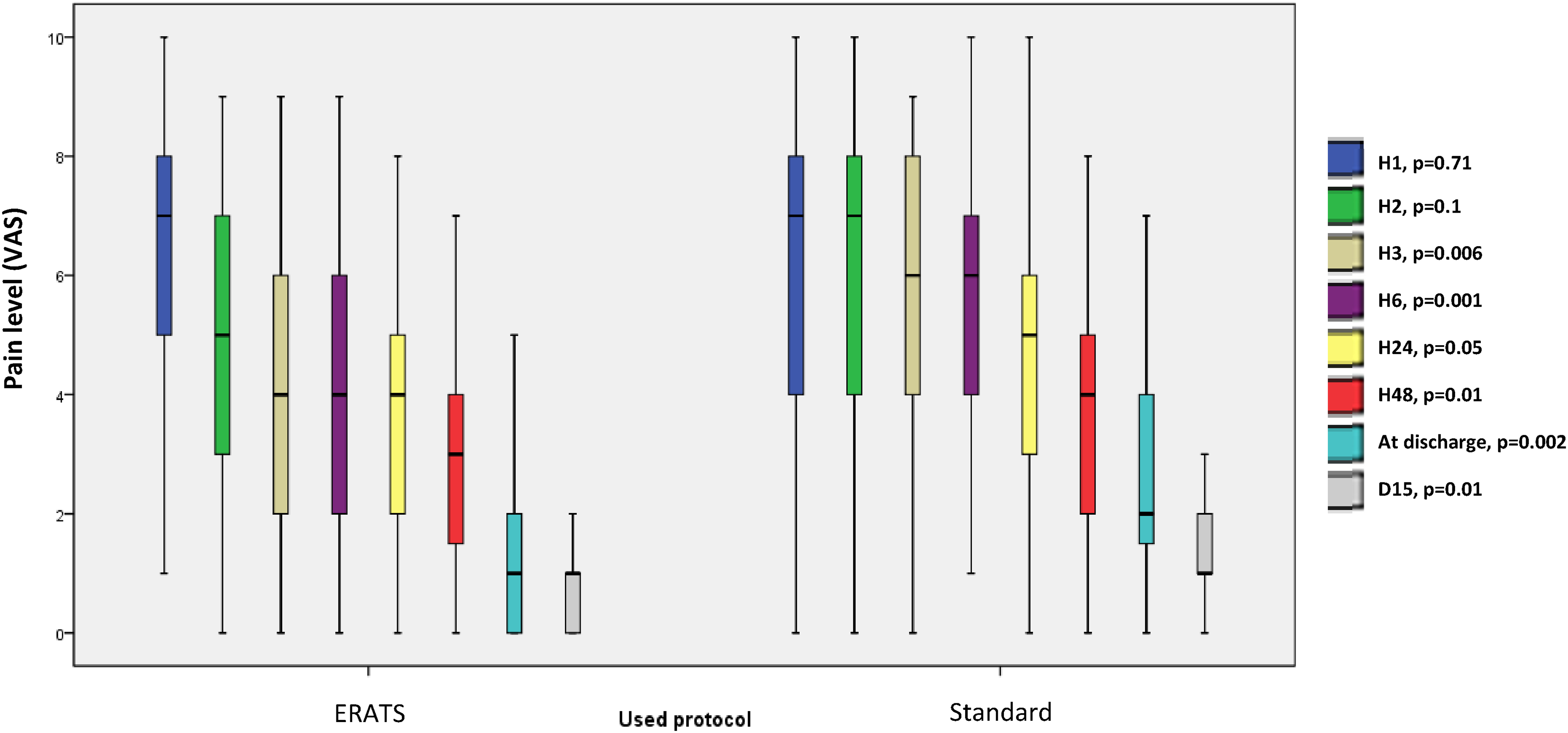
Pain level in ERATS and standard groups. VAS: visual analog scale of pain intensity; ERATS: enhanced recovery after thoracic surgery.
It is also essential to notice a nonsignificant difference between the ERATS and the Standard groups in the use of locoregional analgesia. However, for systematic analgesia after H48 in the Standard group, the IV paracetamol administration route was significantly more used (p = 0.02), and Nefopam consumption was significantly higher (p = 0.003).
Finally, there was no significant difference between the two groups in terms of length of drainage (p = 0.33), postoperative hospital stay (p = 0.17), or general postoperative complications (including ketosis, ulnar nerve paresis, cutaneous necrosis, and local incision infection (p = 1).
In this study, no mortality was noticed.
Discussion
Enhanced recovery after thoracic surgery feasibility in LMIC
The first objective of this trial was to prove the feasibility of ERATS in LMIC. There was no significant difference between the ages, gender, and intellectual levels of patients in terms of cooperation between the two groups. Das-Neves-Pereira et al. study 13 found that the rate of ERATS protocol achievement was lower in males. It could be concluded that the current ERATS protocol may be applied regardless of the age, gender, and intellectual level of the patient.
The used ERATS protocol was adapted to our institution's local conditions. Rare studies have assessed the feasibility of the ERAS protocol in LMIC in thoracic surgery,14,15 with an adapted protocol to comply with the overall ESTS recommendations.
In our study, after their consent, all operated patients were informed about the concept of ERATS. Preadmission information and education are highly recommended by the ESTS guidelines, particularly for pain control. 16 The evidence level of the association between smoking and postoperative complications is high. 6 Four-weeks-delay is reasonable for the ERAS society. 6 This study did not respect this delay, but smoking cessation was an element of the common arm. All patients had a balanced alimentation one day before the surgery. “Balanced alimentation” was not well defined and controlled. European Society of Thoracic Surgeon recommends screening patients for nutrition status preoperatively 6 with an oral nutritive supplementation for malnourished patients. 17
Long-acting benzodiazepines were not used in anesthesia for all patients. The anesthesia protocol and drugs were not assessed in this study. Indeed, benzodiazepines’ hypnotic effect could make postoperative oral alimentation and mobilization difficulties. European Society of Thoracic Surgeon recommendations focus on short-acting drugs that allow early extubation. 6 Selective intubation was the most frequent, while other ERATS analyses used selective intubation in all cases.18,19 This is due to the inclusion of thoracic approaches in this survey, which did not require a double-lumen tracheal tube. Posterolateral-thoracotomy with the mini-muscle-sparing technique was the most frequent surgical approach used in the ERATS and Standard groups. In some analyses, 19 only the ERATS group underwent VATS, which could be a bias. The mini-invasive approach and mini-muscle-sparing thoracotomy are recommended by ESTS guidelines. 6 In our study, using VATS in the common arm was aimed to neutralize VATS bias on postoperative outcomes between the two groups.
The rate of early extubation as an element of the common arm was 95.94%. European Society of Thoracic Surgeon guidelines recommend short-acting anesthesia agents to permit early extubation. 6 Only five cases (5.70%) did not have locoregional anesthesia, with no significant difference between the two groups. Locoregional analgesia is highly proven and recommended. 6 The intercostal block could be beneficial in the short term after surgery. 20 Early mobilization was an essential element of ERATS at the postoperative. Despite the low evidence level between early exercise and postoperative pulmonary complications, 21 this parameter is highly recommended by the ESTS guideline. Early enteral alimentation was the second main element of the ERATS procedure, starting from the second post at the postoperative. Das-Neves-Pereira et al. study 13 started alimentation within the first hour. European Society of Thoracic Surgeon guidelines extrapolate this behavior from nonthoracic studies. It is recommended to start oral alimentation as soon as possible. 6 The primary discharge criterion was chest tube removal. Before, external suction was temporarily stopped in addition to removing the urinary probe to allow patient mobilization. Chest tube removal conditions have differed between ERATS analyses.18,19,22 Chest drain early removal has become a condition that makes patients “wireless” with decreased pain, infection, and hospital stay. 23 European Society of Thoracic Surgeon guidelines recommend avoiding external suction, using a digital drainage system, and accepting early chest tube removal even with high pleural fluid output. 6
It is important to compare our ERATS protocol with others from LMIC. In the Mohan et al. study performed in India, 14 the ERATS protocol was applied with sufficient specialist and paramedical staff, which could not reflect actual conditions in other LMCI hospitals. In the Abrao et al. study performed in Brazil, 15 some relatively sophisticated techniques were used (Acupuncture and electrostimulation with Acu-TENS), which are unavailable in all LMCI hospitals. In our study, the central part of the ERATS protocol was performed with no additional sophisticated devices by thoracic surgery residents to replace enough paramedical staff.
Adapted ERATS effectiveness
The ERATS effect had significantly modified at least one major and one secondary judgment criteria: pleural effusion and postoperative pain level. ERATS studies assessed: only one selected pathology, like Xia et al. who published a meta-analysis about lung cancer 1 and Malik et al. retrospective study about pneumothorax, 2 or only one selected type of technique, like Holbek et al. systematic review about VATS, 3 or only one selected type of resection, like Asteriou et al. prospective study about sublobar resection. 4 Prospective studies to assess the ERATS effect regardless of the type of pathology, resection, or surgical approach are rare. 5
The study's strength is the inclusion of new thoracic pathologies in ERATS protocol, such as lung hydatid cysts and aspergilloma, often treated in LMIC hospitals.
In this study, the better results in the ERATS group were found in terms of respiratory complications separately (except pneumonia), confused respiratory complications, land length of both, drainage duration, and hospital stay, but without being statically significant (except pleural effusion). A retrospective review including 2886 patients operated on for lung cancer throw thoracotomy 6 found a significant decrease in postoperative complications and the length of hospital stay. Xia et al. meta-analysis 1 found that in eight studies, including 660 patients, the retention time of chest tube was significantly shorter with ERATS. A prospective noncontrolled study, including 422 patients operated on for lung cancer, 7 found a significant inverse relationship between ERATS protocol compliance and postoperative complications. In LMIC, Mohan et al. found a significantly better effect with ERATS regarding postoperative complications, chest tube duration, and hospital stay 8 after operating tubercular empyema. Abaro et al. found similar results regarding chest tube duration and hospital stay 5 after operating heterogeneous thoracic pathologies.
We conclude that complications often decrease under the ERATS protocol. The reason that could explain the nonsignificant difference in our study is the application of some ERATS procedures in the common arm (like mini-invasive approaches or early chest physiotherapy).
VAS levels were decreased significantly in the ERATS group from H3 and even after 15 days with a decreased consumption of some analgesic drugs. This parameter needed to have been better assessed in other ERATS trials. In a study that included 99 patients, 24 Vandenbos et al. found that respiratory rehabilitation following pulmonary resection for cancer decreases postoperative pain levels significantly and increases quality-of-life scores. This effect is still significant in patients undergoing thoracotomy and mini-invasive approaches. The decreased pain level with the ERATS protocol proves the benefit of this procedure even after patient discharge.
The rate of mortality was 0%. The mortality rate was always null or around 1% but without being significantly different after an ERATS protocol.1,4,6,7,9–11 The inclusion criteria could explain this in these studies.
We excluded the hospital costs of our study. In the Xia et al. analysis (eight studies), 9 The difference in hospital stay was significantly lower in the ERATS group, which is related to a decreased hospital cost. Even in LMIC, ERATS significantly decreased hospital costs. 5
Study limits
Even though this trial was prospective and randomized to include different thoracic pathologies, there were some limitations:
Because of the trial design, we compared heterogeneous thoracic pathologies. Dividing these pathologies into homogeneous categories led to a low effective sample with a low evidence level of results. We could not measure the effectiveness of applying some ERATS procedures by thoracic surgery residents, which could be a bias. The ERATS protocol needs more standardization of some behavior: psychotherapy support, preoperative alimentation, and postoperative analgesia.
Conclusion
The ERATS protocol is a simple procedure that could be adapted to LMIC hospital's conditions to improve postoperative outcomes in thoracic surgery. This study provides an adapted ERATS protocol, applicable regardless of the surgical approach or the type of resection. It could be reproduced as a multicentric analysis in LMIC.
Footnotes
Authors’ Note
Clinicaltrials.gov. All details were registered under the identifier: NCT02831140.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics committee approval: by “Habib Bourguiba University Hospital Ethics Committee.”
Informed consent
Informed consent was signed by all included patients.
