Abstract
Background
There are increasing reports of early externally mounted pericardial Trifecta bioprosthesis failure. We compared the hemodynamic performance of Trifecta and Carpentier–Edwards Perimount Magna Ease valves to determine the failure mechanism.
Methods
We retrospectively included 270 consecutive patients (age: 73.4 ± 8.2 years; 57.5% male; mean follow-up: 48.0 ± 20.3 months) who underwent aortic valve replacement from 2014 to 2021 at a single center and compared the Trifecta (N = 137) and Carpentier–Edwards Perimount Magna Ease valve (N = 133) patients.
Results
The prosthetic valve major aortic regurgitation incidence was higher for the Trifecta than that for the Carpentier–Edwards Perimount Magna Ease valve (6.3% vs. 0%, P < 0.009). Among the Trifecta failures, 33% developed structural valve deterioration, but all requiring redo aortic valve replacement developed major prosthetic valve aortic regurgitation. Freedom at 5 years from redo aortic valve replacement due to structural valve deterioration was significantly lower for Trifecta (89.4% vs. 100%, P = 0.003). The reoperation hazards were determined for Trifecta (vs. Carpentier–Edwards Perimount Magna Ease): 11.6 (1.47–90.9; P = 0.02), prosthetic valve aortic regurgitation: 2.38 (1.70–3.32; P < 0.01), structural valve deterioration: 20.82 (4.08–106.2; P < 0.01), 5-year mean transprosthetic pressure gradient: 1.14 per 1-point increase (1.03–1.24; P = 0.007), and urgent surgery: 10.1 (2.59–39.0; P = 0.001). The Cox regression analysis identified that prosthetic valve aortic regurgitation solely contributed to redo aortic valve replacement (hazard ratio: 2.38; confidence intervals: 1.70–3.32).
Conclusions
Significantly, more early failures occurred with the Trifecta valve than the Carpentier–Edwards Perimount Magna Ease valve but the Trifecta showed reasonable mean transprosthetic pressure gradient over time. Prosthetic valve aortic regurgitation and calcific structural valve deterioration synergistically contributed to Trifecta valve failure alternatively.
Keywords
Introduction
Aortic bioprostheses are the most common prostheses used for aortic valve replacement (AVR) in the current era because they have the advantage of avoiding anticoagulation in a predominantly older adult population because bleeding complications due to anticoagulation cannot be neglected. However, their drawback is greater susceptibility to structural valve deterioration (SVD) over time. 1 The unique Trifecta design with valve leaflets manufactured from a single bovine pericardial sheet externally mounted onto a flexible titanium stent enables a superior effective orifice area over internally mounted bioprostheses, especially for smaller valve sizes.2,3 In addition, the Trifecta valve eliminated alignment stitches on the top of the leaflets, which is thought to be a plausible cause of early valve failure in externally mounted bioprostheses. 4 One study reported favorable midterm 6-year outcomes of 710 Trifecta valve patients implanted between 2007 and 2009. The rate of freedom from reoperation due to SVD was 97%. 5 However, there have been an increasing number of reports regarding early Trifecta valve failure,6,7 which raises concerns about its durability. The Trifecta bioprosthesis appears to be associated with a greater rate of SVD and a higher need for repeat AVR for SVD than those of some other bioprostheses. 8 This study compared the hemodynamic performance of Trifecta valves with that of new or old-generation internally mounted valves to determine the mechanism of failure.
Materials and methods
Study design
We retrospectively reviewed consecutive patients who underwent AVR between 2014 and 2021 at a single center. Of these patients, the Trifecta valve (St Jude Medical Inc., St Paul, MN, USA), Carpentier–Edwards Perimount Magna Ease (CEP) valve (Edwards Lifesciences, Inc., Irvine, CA, USA) were compared preoperatively and postoperatively.
Data selection
We participated in the national database registry, Japan Cardiovascular Surgery Database and had the right to use our own data out of the database.
Ethics
The local ethics committee granted the permission for the use of patients’ clinical data for the present observational retrospective study (EC/IRB file number: 2022-3).
Inclusion criteria
Urgent cases, redo operations, AVR concomitant with other cardiac operations, and all causes of aortic valve disease were included. Patients who received an initial aortic mechanical valve and underwent redo AVR with either Trifecta or CEP within the observational period were included in the study, regardless of the date of the mechanical valve implant.
Exclusion criteria
AVR cases using mechanical prostheses were excluded. Patients who underwent initial AVR with either Trifecta or CEP valve >10 years prior to the observation period and had redo AVR within the observation period were not considered as valve-failure events and were not enrolled in the present study.
The choice of valve was at the discretion of the surgeon. We gradually switched over to the Trifecta™ Valve with Glide™ Technology (Trifecta GT) in October 2016. Before this, first-generation Trifecta valves were used. Regarding operative techniques, median sternotomy was performed, and everting mattress sutures with pledgets were placed at the supra-annular position to secure the valves. We checked the operative gradient with transesophageal echocardiography (TEE) when coming off the cardiopulmonary bypass. With that, we were left with almost no or trivial transvalvular or paravalvular leakage (PVL) in the operating room. What if there was significant leakage found by TEE, we usually employed a second pump run to fix the leakage. Within the Trifecta group, patients receiving redo AVR were compared with non-redo AVR patients. Echocardiography was performed in the short-term within 1 month of post-implant and periodically thereafter for 5 years. Five-year echocardiography was defined as either right before the reintervention or at the end of the follow-up period of 5 years. Univariate and multivariate analyses were performed to investigate the factors associated with redo AVR among the patients in the Trifecta and CEP groups.
Definition of SVD
SVD is an acquired intrinsic bioprosthetic valve abnormality characterized by deterioration of the leaflets, resulting in thickening, calcification, and tearing of the prosthetic valve materials with eventual associated hemodynamic dysfunction, manifested as stenosis or regurgitation. 9 Our definition of SVD was based on the universal definitions proposed by Rodriguez–Gabella et al., 10 which were modified to reflect the availability of our retrospective data. Accordingly, SVD was defined as an increase in the mean transprosthetic pressure gradient (mTPG) by >20 mmHg with a concomitant decrease in the effective orifice area by >0.6 cm2 during follow-up. 10 The cases showing a high mTPG from initial echocardiography postoperatively were considered not to have SVD but as having a patient prosthesis mismatch (PPM). Hence, SVD occurs over a certain period, while PPM occurs immediately post-implant in patients who receive an inappropriately sized valve, given the body surface area (BSA) of particular patients. Clinical valve abnormalities that did not result from deterioration of the valve tissue were not included in the definition of SVD. These include PPM, device malposition, PVL, and prosthetic valve endocarditis (PVE). 9 In general, SVD encompasses calcific SVD and prosthetic aortic regurgitation (AR) due to tearing of the prosthetic valve. However, for the purpose of distinguishing the mode of failure precisely, we modified the definition so that prosthetic AR was excluded from SVD.
Statistical analysis
SPSS version 22.0 (IBM Japan, Ltd, Tokyo, Japan) was used for data analysis. Continuous variables were summarized as the mean ± standard deviation. The Shapiro–Wilk test was used to assess normality. Categorical data are expressed as frequencies and percentages. A paired t-test was used to compare the pressure gradients of a particular valve over time. An unpaired t-test was used to compare two independent groups when the dependent variables were normally distributed. If they were not normally distributed, a Mann–Whitney U-test was performed to compare two independent groups when the dependent variables were either ordinal or continuous. Fisher's exact test was used to compare categorical variables between the valve groups. The univariate and multivariate analyses were conducted for redo AVR among the patients in the Trifecta and CEP groups. To avoid bias, a comparison between the two groups was performed to determine whether there were any differences in the variables. Variables that were significantly different in this comparison were then subjected to univariate analysis. For the Cox regression analysis, variables with a P-value of <0.05 in the univariate analysis were entered into the final multivariate analysis. The forward stepwise selection was performed for the multivariate analysis. The results are presented as hazard ratios (HRs) with corresponding 95% confidence intervals (CIs). Freedom from reoperation was compared by performing Kaplan–Meier survival analysis and log-rank tests. A two-tailed P-value of <0.05 was considered to be indicative of statistical significance.
Results
A total of 270 consecutive patients (Trifecta valve (N = 137) and CEP valve (N = 133)) admitted to our institution between 2014 and 2021 were reviewed. Fifteen patients, who underwent initial AVR with CEP valve >10 years ago prior to the observation period and had redo AVR within the observation period, were not enrolled in the study. In total, 11.6% (16/137) and 18.8% (25/133) of the patients in the Trifecta and CEP groups, respectively, were aged 65 years or younger. No patients in the CEP group underwent redo AVR except one case complicated by PVE. Nine patients in the Trifecta group underwent redo AVR (6.5%, 9/134), including one who underwent transcatheter AVR (TAVR). Of these, three patients were complicated by PVE and PVL, which was a consequence of the endocarditis process. Therefore, six patients in the Trifecta group developed prosthesis failure due to the new onset of severe prosthetic AR because of a cusp tear, which required redo AVR (4.47%). Among these six patients, only one received the Trifecta GT; the remaining five patients received a first-generation Trifecta valve at the initial AVR. Three patients in their 60s received surgical redo AVR, two patients in their 70s had anatomical issues that prevented them from receiving TAVR, and they instead received surgical redo AVR, and one patient in their 70s underwent valve-in-valve TAVR. This AR disorder was characterized by a partial cusp tear along with the stent post, as shown in Figure 1. Cusp tears included 3 of the right coronary cusp (RCC) tears, 1 of non-coronary cusp (NCC) tear, 1 each of RCC and NCC stent post-tear, and 1 of left coronary cusp tear. Two patients in the Trifecta group developed pannus formation on the prosthesis, which was confirmed by the last echocardiography that exhibited a low pressure gradient (6 and 11 mmHg). The average duration from initial AVR with a Trifecta valve to redo AVR was 46.8 months. The preoperative characteristics, operative data, and outcomes of the two groups are summarized in Table 1. Heart failure symptoms based on the New York Heart Association classification were worse in the Trifecta group than in the CEP group, but the difference was not significant. The mTPG at 1 month was lower in the Trifecta than the CEP group (7.7 ± 3.1 vs. 9.3 ± 2.8 mmHg; P < 0.05), but the 5-year results were similar. There were no significant differences in total operating time, pump time, or concomitant procedures between the groups. Thirty-day mortality was similar among the groups (3.6 vs. 3.0, 0%). Other comorbidities were similar among the groups, except for prolonged ventilation (5.8% vs. 0.8, 0% P < 0.05). The incidence of SVD was greater in the Trifecta group, although the difference was not statistically significant (Trifecta vs. CEP: 3.9% (5/127) versus 0.9% (1/112)). The rate of prosthetic valve AR was significantly greater in the Trifecta group than in the CEP group (6.3% vs. 0% for major AR P < 0.009).

Representative photographs of explanted Trifecta valves. Arrows indicate a tear around the commissure post.
Comparison of baseline characteristics, operative data, and postoperative outcomes between the Trifecta and Perimount Magna Ease valves.
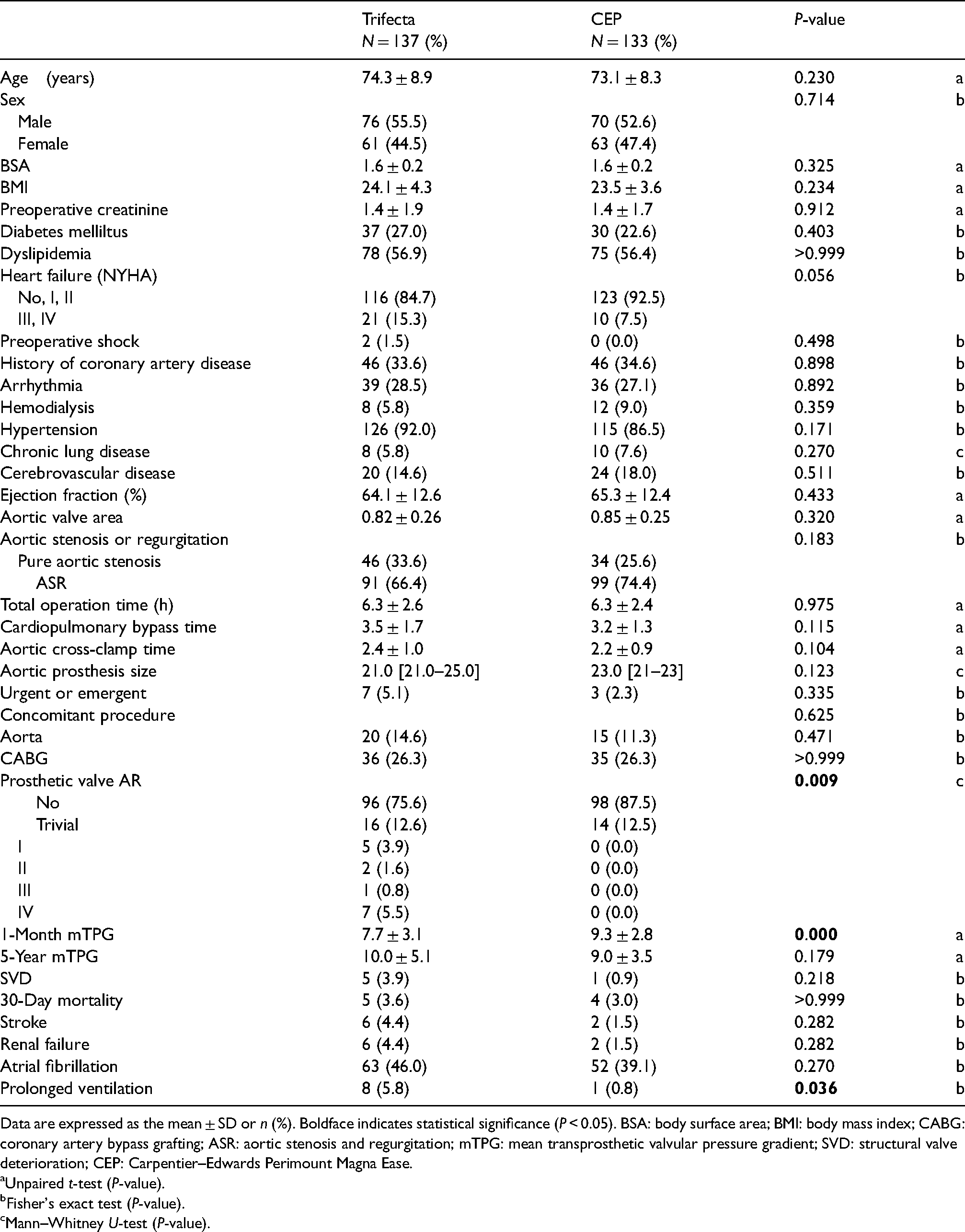
Data are expressed as the mean ± SD or n (%). Boldface indicates statistical significance (P < 0.05). BSA: body surface area; BMI: body mass index; CABG: coronary artery bypass grafting; ASR: aortic stenosis and regurgitation; mTPG: mean transprosthetic valvular pressure gradient; SVD: structural valve deterioration; CEP: Carpentier–Edwards Perimount Magna Ease.
Unpaired t-test (P-value).
Fisher's exact test (P-value).
Mann–Whitney U-test (P-value).
Redo AVR patients versus non-redo AVR patients within the Trifecta group
Results from a comparative analysis are presented in Table 2. All six patients who underwent redo AVR were complicated by major prosthetic valve AR (>III). None of the patients in the non-redo AVR group developed major prosthetic valve AR. Of the six patients, two were complicated by calcific SVD (33%). The 5-year mTPG was higher in the early failure group than in the non-redo AVR group (16.6 ± 9.0 vs. 9.6 ± 4.5 mmHg P = 0.001), although the 1-month mTPGs were similar between the groups (7.8 ± 3.6 vs. 7.7 ± 3.1 mmHg). The incidence of SVD was significantly higher in the redo AVR group than in the non-redo AVR group (2/6, 33.3%; 3/119, 2.5%; P = 0.018).
Comparison between the early failure and non-failure patients within the Trifecta group.
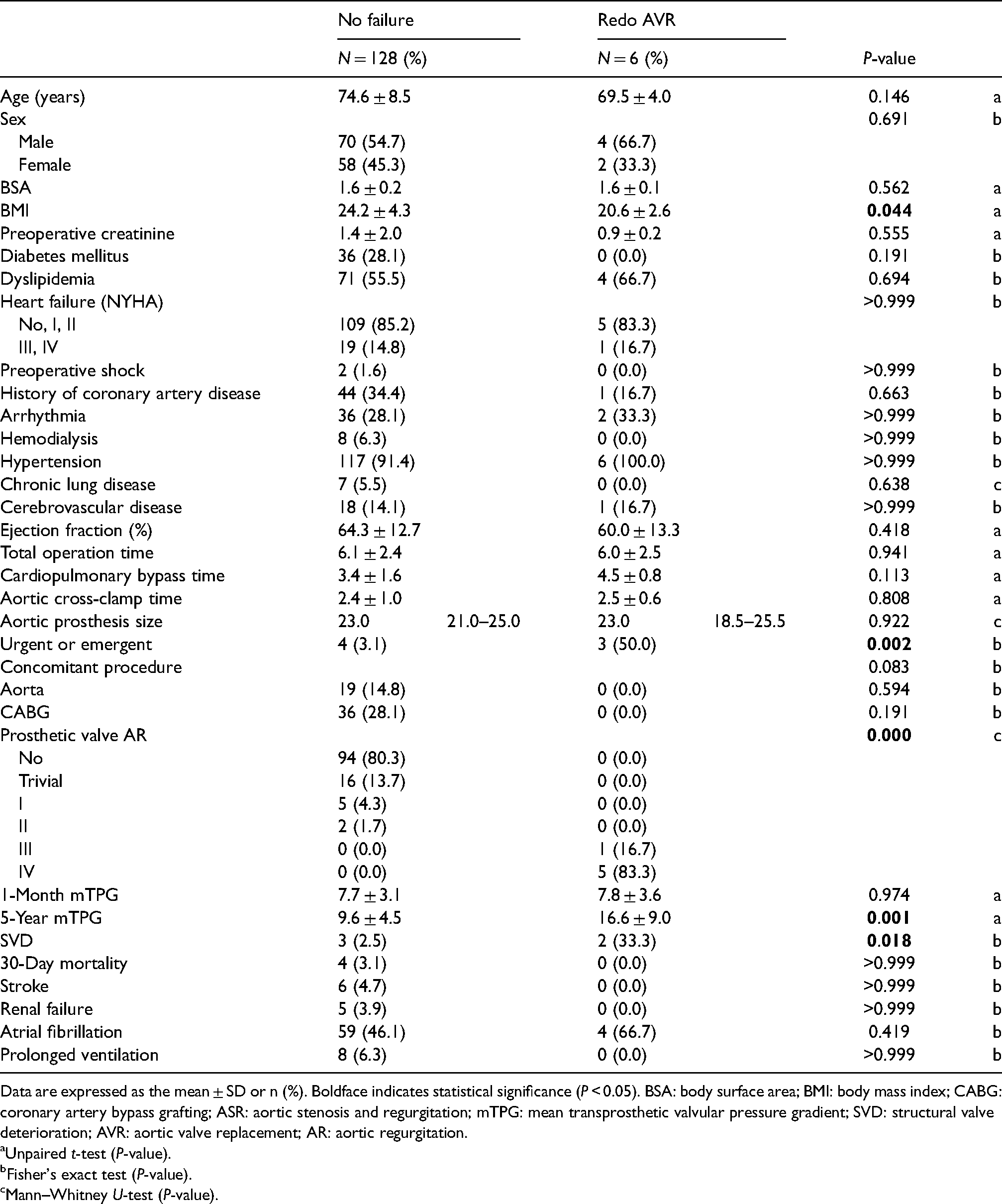
Data are expressed as the mean ± SD or n (%). Boldface indicates statistical significance (P < 0.05). BSA: body surface area; BMI: body mass index; CABG: coronary artery bypass grafting; ASR: aortic stenosis and regurgitation; mTPG: mean transprosthetic valvular pressure gradient; SVD: structural valve deterioration; AVR: aortic valve replacement; AR: aortic regurgitation.
Unpaired t-test (P-value).
Fisher's exact test (P-value).
Mann–Whitney U-test (P-value).
Longitudinal hemodynamics of prostheses measured by echocardiography
The mTPG measured by echocardiography was followed over time (Table 3). The follow-up period was 5 years. The follow-up rate was 89% (122/137) for the Trifecta group and 89.4% (119/133) for the CEP group. The mTPG was greater for the CEP group than for the Trifecta group at 1-month post-implant (9.3 ± 3.1 vs. 8.0 ± 3.3 mmHg, P = 0.011). The 5-year mTPGs were similar between the groups. There was a gradual increase in the gradient for the Trifecta valve over time, but the absolute difference was small (8.0 ± 3.3 to 10.0 ± 5.1 mmHg; P < 0.05).
Mean transprosthetic gradient trends after implant over time.

TF: Trifecta; CEP: Carpentier–Edwards Perimount Magna Ease.
Unpaired t-test (P-value).
Fisher's exact test (P-value).
Mann–Whitney U-test (P-value).
Freedom from redo AVR
The average observation period was 41.2 ± 20.3 for the Trifecta group and 53.8 ± 19.1 months for the CEP group. The estimated freedom from redo AVR overall at 60 months was 87.4% ± 4.4% in the Trifecta group and 99.2% ± 0.8% in the CEP group as shown by the Kaplan–Meier curve in Figure 2A. The freedom from redo AVR at 5 years due to SVD, excluding PVE patients, was 89.4% ± 4.3% in the Trifecta group and 100% in the CEP group (Figure 2B) (log-rank test; P = 0.003).
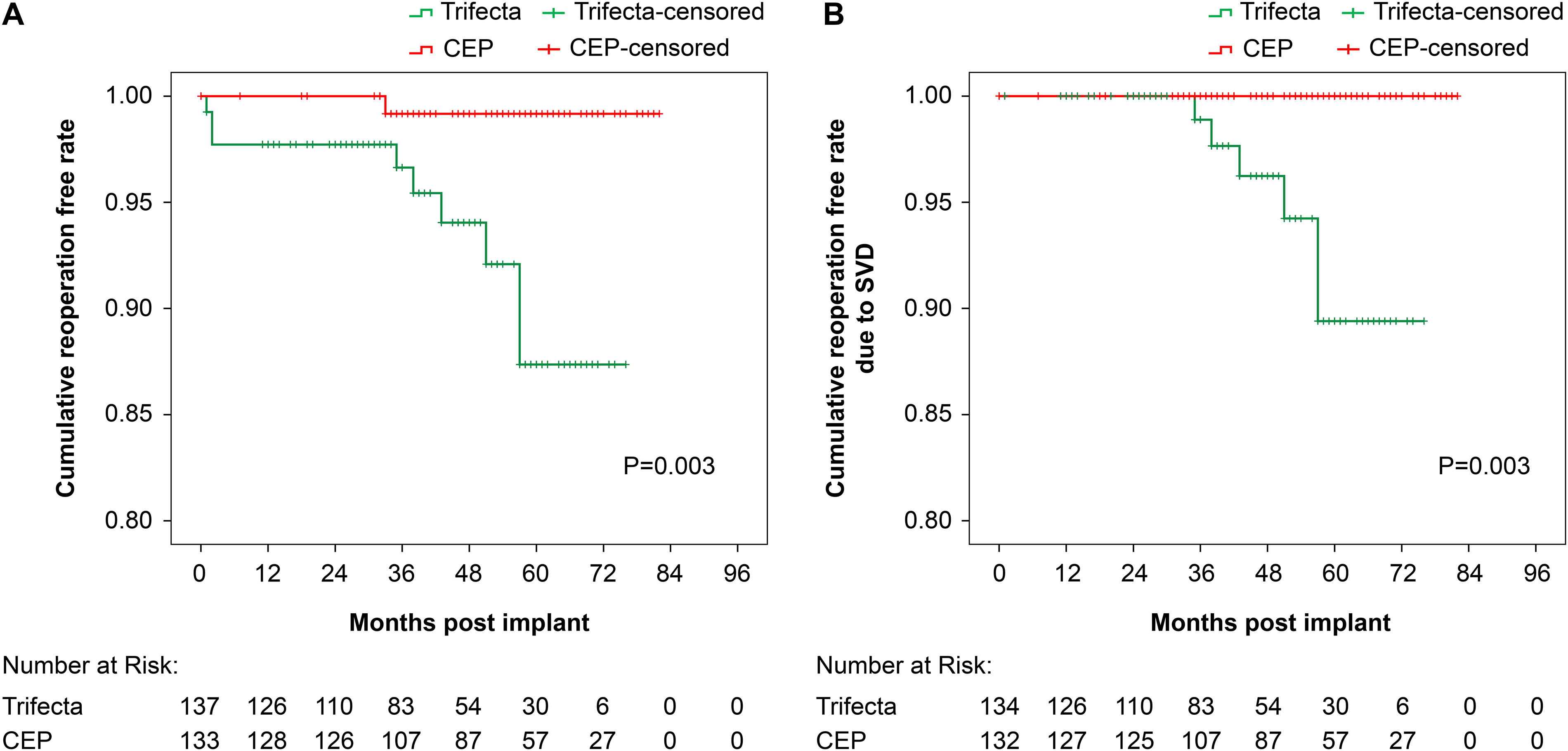
Kaplan–Meier 5-year freedom from reoperation for the Trifecta and Perimount Magna Ease valves. (A) All reoperation cohorts; (B) reoperation due to structural valve deterioration (SVD).
Cox regression analysis for redo AVR
Univariate analysis revealed that the Trifecta valve (HR: 11.62; CI: 1.47–90.9; P = 0.02), prosthetic valve AR (HR 2.38; CI: 1.70–3.32; P < 0.01), 5-year mTPG (HR: 1.14 per 1-point increase; CI: 1.03–1.24; P < 0.01), SVD (HR 20.8; CI: 4.08–106; P < 0.01) and urgent surgery (HR: 10; CI: 2.6–39; P < 0.01) contributed to redo AVR (Table 4). However, multivariate analysis identified prosthetic valve AR as the sole strong contributor to redo AVR (HR: 2.38; CI: 1.70–3.32; P < 0.05).
Cox regression analysis for redo AVR.
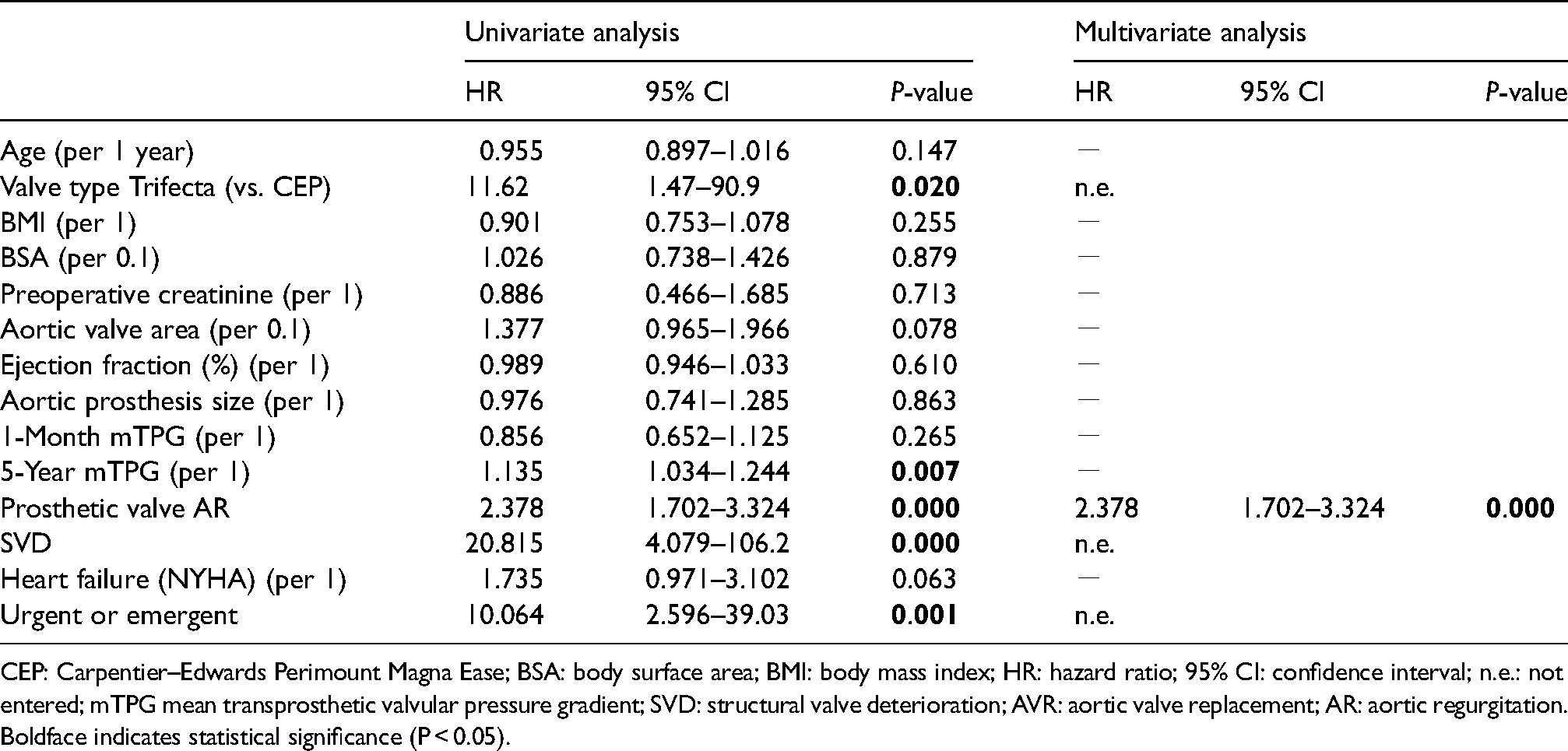
CEP: Carpentier–Edwards Perimount Magna Ease; BSA: body surface area; BMI: body mass index; HR: hazard ratio; 95% CI: confidence interval; n.e.: not entered; mTPG mean transprosthetic valvular pressure gradient; SVD: structural valve deterioration; AVR: aortic valve replacement; AR: aortic regurgitation.
Boldface indicates statistical significance (P < 0.05).
Discussion
The 5-year rate of redo AVR due to SVD was 4% for the Trifecta valve, which was close to the 4%–6% rates previously reported. 7 Among the six Trifecta failure cases requiring reintervention in our study, only one received Trifecta GT; the remaining five patients received a first-generation Trifecta valve at the initial AVR. It has been reported that the rate of reintervention in patients who received the first-generation Trifecta valve is more than double that of those who received the Trifecta GT at the initial AVR, at 1.86% and 0.56%, respectively. 11 Early failure of the first-generation Trifecta valve prompted improvement in the design of Trifecta GT, including new anticalcification technology and the Glide Technology for ease of implantation and minimal distortion during implantation. Despite these changes, they reported the early failure of Trifecta GT with cusp tear. 12 Furthermore, Trifecta GT valves were naturally implanted at later dates; therefore, their lower reintervention rate may reflect the shorter duration of observation. If standardized for time, the true-reintervention rate may differ. 11 When patients required reintervention for a failing Trifecta valve, we provided a redo surgical AVR or valve-in-valve TAVR option. The 10-year durability of the TAVR valve in tissue valve is now being guaranteed but not for 15 years. Therefore, for patients younger than 70 years, we usually offer surgical redo AVR over the TAVR option given their life expectancy; patients receiving valve-in-valve TAVR following a failing Trifecta valve may require a third surgery. For patients older than 70 years, we offer both options. If the patient has any anatomical issues that would prevent them from receiving the TAVR, we offer the surgical redo AVR option. The 5-year rate of redo AVR for the CEP group was 0%. Kaplan–Meier curve analysis gave similar findings for the estimated freedom from redo AVR due to SVD at 5 years. Other studies have found that the Trifecta valve failed significantly earlier than the CEP valve due to SVD over 5–7 years.1,13 In general, externally mounted pericardial bioprostheses such as Trifecta valve show superior hemodynamics, but they are known to cause leaflet tears, leading to early valve failure. 14 In vitro experiments comparing internally and externally mounted tissue valves identified the reason for the failure of an externally mounted prosthesis as leaflet tears around the commissure posts after 400 billion cycles of mechanical wear tests. 15 A single pericardial leaflet wrapped around stent frames repeatedly experienced leaflet to stent contact, which seemed to cause tissue abrasion and ultimately tears or holes in the leaflet at commissure posts of the Trifecta valve. In clinical settings, there have been reports of early Trifecta valve failures caused by cusp tears which lead to prosthetic valve AR.4,16,17 On the other hand, Yongue et al. 3 reported that the main mode of Trifecta valve failure was calcific SVD rather than prosthetic valve AR. Consequently, the mode that contributes to the valve failure remains unknown.
Low transprosthetic valvular gradient and relatively low rate of SVD in Trifecta valve
We observed that the 5-year mTPG was higher in the redo AVR than in the non-redo AVR patients within the Trifecta group (Table 2). However, the 5-year mTPG was low and similar between the Trifecta and CEP valve groups (Table 3). Good hemodynamic performance and durability of the Trifecta valve in relatively old patients with small BSA in which very low mean gradients at discharge that were maintained through 7 years of follow-up have been reported in the past. 8 Biancari et al. 13 and Fukuhara et al. 6 reported that they implanted an average of 22.7–23 mm Trifecta valves in patients with BSAs of 1.9–2.0, whereas we implanted an average of 23 mm Trifecta valves in Asian patients with a BSA of 1.6 ± 0.2. As a result, low mTPG was maintained for over 5 years. The reason for this was that we used the ultrasonic surgical aspirator for the aortic annulus debridement, which enabled us to place a larger valve. 18 For bioprostheses, it is crucial to expose to a low-pressure environment from the durability standpoint.
Similarly, the incidence of SVD was higher in the redo AVR patients than in the non-redo AVR patients within the Trifecta group; however, the comparison between the Trifecta and CEP valve groups did not show a significant difference, suggesting that the mTPG and incidence of SVD were relatively low in all cohorts of our study. Many original articles with a high volume of Trifecta cases revealed that most of the failed Trifecta valves were complicated by SVD or high mTPG at 5–7 years, postoperatively (Table 5). One study reported that the average increase in mTPG between the initial echocardiography after the first AVR and reintervention was 31 ± 17 mmHg, which is considered to be SVD, suggesting that all Trifecta failure cases requiring redo AVR were complicated by calcific SVD. 7 However, in our study, the rate of SVD in the failed Trifecta patients was only 33% and their mTPG remained low over time, although it was increased from 7.8 ± 3.6 before discharge to 16.6 ± 9 mmHg on the last echocardiographic assessment.
Original articles with high-volume Trifecta valve patients.

CEP: Carpentier–Edwards Perimount Magna Ease; TF: Trifecta; SVD: structural valve deterioration.
High rate of prosthetic valve Ar in Trifecta valve patients
On the other hand, the rate of major prosthetic valve AR (greater than III) was consistently greater in the Trifecta group than in the CEP group in all comparisons either within the Trifecta groups or among the groups (Tables 1 and 2). Therefore, univariate analysis revealed that prosthetic valve AR and 5-year mTPG and SVD contributed to redo AVR; however, Cox regression analysis showed that prosthetic valve AR was the sole strong contributor to redo AVR. Fukuhara et al. 6 reported that there was no prosthetic valve AR in the failed CEP valves, whereas the prosthetic AR component was involved in >50% of failed Trifecta cases, which led to repeat AVR within 7 years. It may be premature to conclude that prosthetic valve AR is the sole contributor to redo AVR as the incidence of SVD occurs around 7 years generally for any kind of bioprosthesis. 9 Given our follow-up duration, further follow-up is required to elucidate the cause of failure. It is worth mentioning that the important features of Trifecta valve failure are different from others as patients tend to develop cardiogenic shock due to the acute onset of wide-open AR. Therefore, the requirement of an emergency surgery was commonly seen, and consequently, the outcomes were worse. 1 Univariate analysis identified emergent surgery as a factor associated with redo AVR for Trifecta valve in our study (Table 4).
Prosthetic valve Ar and calcific SVD synergistically contributed to the Trifecta valve failure
A fair number of original articles with high-volume Trifecta patients have revealed the presence of a 5- to 7-year high prosthetic gradients on echocardiography and prosthetic valve AR (Table 5). The only report from the Cleveland Clinic 3 affirmed that the main mode of Trifecta valve failure was calcific SVD rather than prosthetic valve AR due to cusp tear within 5 years post-implant. However, the incidence of prosthetic valve AR in Trifecta patients was twice as much as that of CEP cases overall in their study. 3 Thus, we believe that some AR component contributed to the SVD. The majority of those original articles showed that most of the failed Trifecta valves exhibited high mTPG at 5–7 years post-implant and the prosthetic AR component was involved in >50% of failed Trifecta patients, which led to the repeat AVR within 5–7 years (Table 5). In addition to other bioprostheses, some percentage of Trifecta valves usually develop SVD around 7 years after implant. 6 However, prosthetic valve AR occurs relatively early up to 3–5 years after implant in Trifecta patients.3,16,17 Therefore, there was some time delay between those two events. Given this delay, we speculated that SVD had started with prosthetic valve AR, which was exacerbated by calcific SVD later with the development of leaflet thickening that can affect leaflet coaptation. As a result, prosthetic valve AR and calcific SVD synergistically contributed to early Trifecta valve failure in phases.
Limitations
This was basically a retrospective, observational study with limited statistical power to elucidate the mechanisms of how and why Trifecta valve failed earlier than the Perimount valve. A future follow-up study with a large-scale protocol is needed for clarification. Although we included all patients receiving either a Trifecta valve or Perimount valve between 2014 and 2021, only 10%–20% were aged 65 years or younger. In general, SVD occurs earlier in the process post-implant for young patients. 20 Therefore, if we had a greater fraction of young patients receiving bioprosthesis in our study population, SVD would have occurred earlier and there would have been less time delay between the prosthetic valve AR and calcific SVD events. However, it was clear that those two factors contributed to Trifecta valve failure synergistically.
Conclusion
The Trifecta valve failed significantly earlier than the CEP valve, although its hemodynamic performance showed a reasonable transprosthetic pressure gradient over time. Prosthetic valve AR and calcific SVD synergistically contributed to early Trifecta valve failure alternatively.
Footnotes
Acknowledgement
Author thank the statistical support provided by Yosuke Sasaki.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The local ethics committee granted permission for the use of patients’ clinical data for the present observational retrospective study (EC/IRB file number: 2022-3).
Informed consent
All patients provided signed informed consent to allow clinical and administrative data storage and utilization for scientific purposes.
