Abstract
Objective
There have been case reports of adverse events of hypoglossal nerve stimulator (HNS) implantation not seen in previous clinical trials, including pneumothorax and pleural effusion. The purpose of this study was to determine the rates of these complications and potential risk factors.
Study Design
Retrospective case-control study during 2014 to 2021.
Setting
Twenty-five health care organizations across the United States.
Methods
The TriNetX Research Network was queried by using disease codes to retrospectively identify patients with obstructive sleep apnea who underwent HNS implantation. Rates of pneumothorax, pleural effusion, other complications, and need for revision/replacement or explant were determined.
Results
We identified 1813 patients from 25 health care organizations who underwent HNS implantation. The average age was 60 years, and there were 68% males and 32% females. Of the cohort, 2.4% (n = 44 from 7 implant centers) experienced a pneumothorax, and 0.6% (n = 11) were diagnosed with a pleural effusion on the day of surgery. Patients who experienced pneumothorax were more likely to have a history of chronic lower respiratory diseases when compared with those who did not (odds ratio, 2.2; 95% CI, 1.1-4.1; P = .02).
Conclusion
The incidence of intraoperative pneumothorax and pleural effusion during HNS implantation may be greater than initially thought. Patients with chronic lower respiratory diseases may be at increased risk. This should be communicated with patients during the informed consent process.
Obstructive sleep apnea (OSA) is a chronic condition caused by collapse of upper airway tissue during sleep. 1 OSA affects approximately 15% to 30% of males and 10% to 15% of females in the United States,2,3 and prevalence continues to rise as a result of increasing rates of obesity. 4 Aside from snoring and daytime sleepiness, OSA is associated with increased cardiovascular morbidity and mortality. 5 In addition to behavioral modifications such as exercise and weight loss, 6 changes in sleep position, 7 and avoidance of alcohol, 8 the first-line treatment for OSA is continuous positive airway pressure (CPAP). 9 CPAP is effective, although adherence is limited to 60% to 70%. 9 Therefore, many patients require alternative management strategies.
Hypoglossal nerve stimulation is a surgical treatment for patients whose treatment with CPAP has failed, whose body mass index is <35 kg/m2, and whose anatomy is favorable as determined by a drug-induced sleep endoscopy.10,11 In 2014, the Inspire System was approved by the US Food and Drug Administration (FDA) based on efficacy and a low incidence of adverse events in the STAR trial (Stimulation Therapy for Apnea Reduction). 11 It is composed of a stimulating lead that activates the hypoglossal nerve, a respiratory sensing lead located between the intercostal muscles, and a pulse generator, which together function to coordinate protrusion of the tongue on inspiration and relieve airway obstruction. 11 A hypoglossal nerve stimulator (HNS) was found to result in improved outcomes as compared with other surgical management strategies for OSA in select patients. 12
As hypoglossal nerve stimulation becomes more common, monitoring safety outside of those patients enrolled within the controlled setting of a clinical trial is important due to potential variations in implantation and management by different specialists. 13 There have been case reports of adverse events not seen in previous clinical trials, including pneumothorax and pleural effusion.14,15 The purpose of this study was to utilize a database with a large sample size of patients who underwent HNS implantation to report rates of these complications and potential risk factors.
Methods
Data were collected on January 24, 2022, from the TriNetX Research Network, which provided access to electronic medical records from approximately 85 million individuals from >50 health care organizations (HCOs) across the United States. 16 TriNetX is compliant with the HIPAA (Health Insurance Portability and Accountability Act). Any data displayed on the TriNetX platform in aggregate form or any patient-level data provided in a data set generated by the TriNetX platform are deidentified per the deidentification standard defined in section 164.514(a) of the HIPAA privacy rule. This study utilizes deidentified patient records with no individually identifiable data and was exempted by the Penn State Institutional Review Board review (STUDY00018629).
The database was queried with diagnosis and procedure codes to identify patients with OSA who underwent HNS implantation between 2014 and 2021. Outcomes included pneumothorax, pleural effusion, infection, hematoma/seroma, pain, disorders of the hypoglossal or facial nerves, as well as need for implant revision/replacement or removal. For patients who had a pneumothorax, an additional outcome was placement of a chest tube. The diagnosis and procedure codes utilized to execute this search strategy are listed in Supplement 1 (available online). For complications experienced by 1 to 9 patients, TriNetX rounds to 10 to protect patient confidentiality.
Statistical analysis was performed with embedded software on the TriNetX platform to compare demographics and clinical characteristics between those who experienced pneumothorax (cases) and patients who did not (controls). Odds ratios and 95% CIs were calculated for categorical variables, and t tests were performed for quantitative variables to compare differences between means. Statistical significance was defined as P < .05.
Results
We identified 1813 patients who underwent HNS implantation at 25 HCOs across the United States, with demographics described in Table 1 .
Description of Sample (1813 Patients).

Of the cohort, 2.4% (n = 44) experienced a pneumothorax, and 0.6% (n = 11) were diagnosed with a pleural effusion on the day of surgery. Of note, 7 of 25 (28.0%) implant sites accounted for all 44 pneumothoraces. There were no significant differences in age, sex, race, ethnicity, height, or weight between patients who experienced pneumothorax and those who did not. Individuals with pneumothorax were more likely to have a history of chronic lower respiratory diseases than those who did not ( Table 2 ). All of the pneumothoraces were managed without the need for a chest tube. Of note, 26.8% (n = 486) of HNS implantations occurred following FDA approval of the 2-incision approach in March 2021. Of this cohort, <10 patients experienced a pneumothorax.
Comparison of Patients Who Experienced Pneumothorax vs Those Who Did Not.
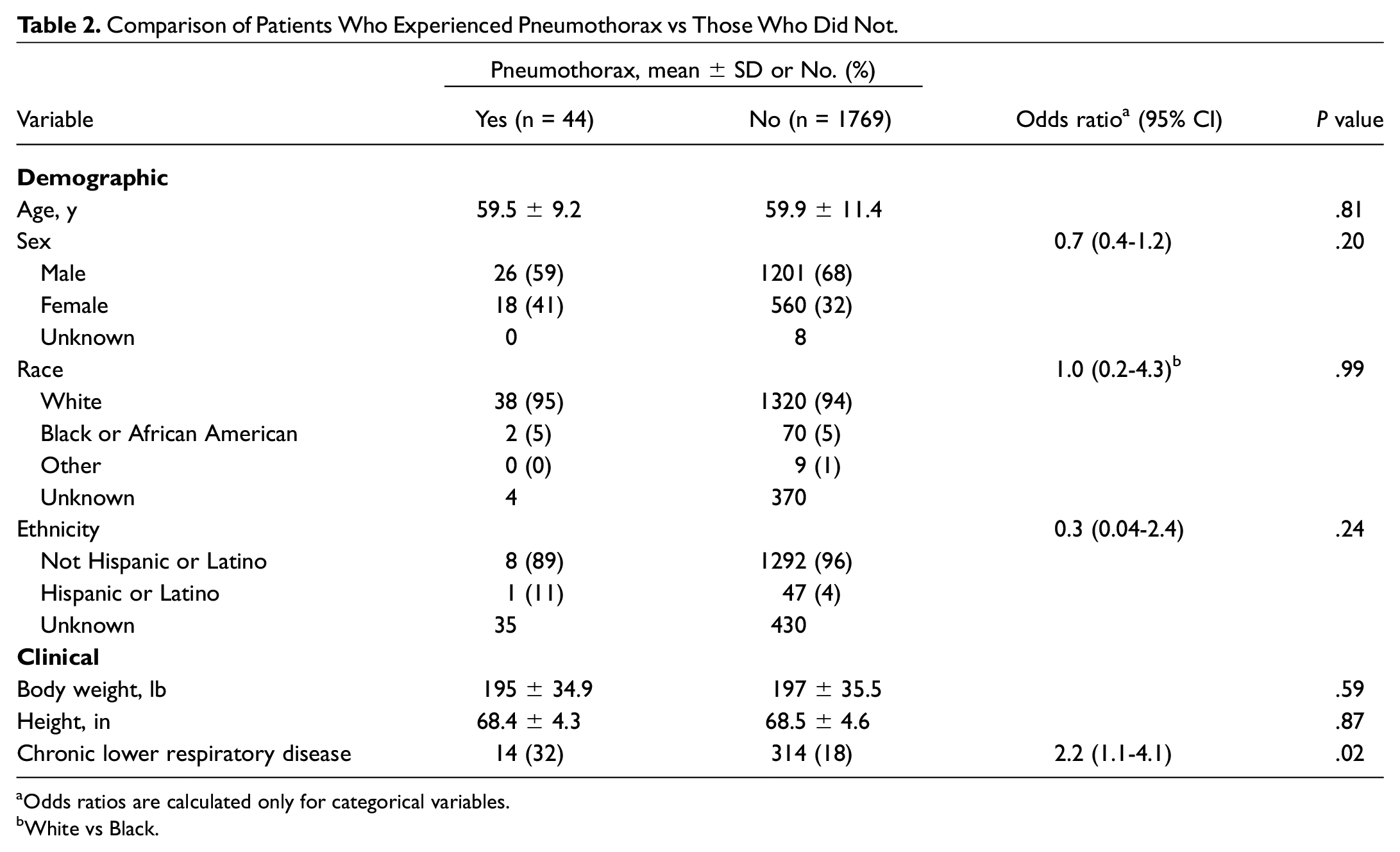
Odds ratios are calculated only for categorical variables.
White vs Black.
Infection occurred in 0.9% (n = 17) in the first 3 months. Less common complications (<10 patients) included hematoma/seroma, pain due to implant, and disorders of the hypoglossal or facial nerves. Overall, 1.4% (n = 25 from 12 implant centers) of the total cohort underwent revision/replacement, and 1.2% (n = 22 from 10 implant centers) had their stimulators explanted. The time from implant to revision/replacement was 1.8 ± 1.3 years (mean ± SD). Those who underwent removal had their stimulators for 1.8 ± 1.4 years. Of note, none of the 44 patients with intraoperative pneumothorax required future revision/replacement or removal of their HNS.
Discussion
During placement of the respiratory lead in HNS implantation, a bent malleable retractor is advanced between the external and internal intercostal muscles, and a tunnel is created via blunt dissection. 17 If advanced at an unfavorable angle or if there is overdissection, the lung pleura is vulnerable to injury and pneumothorax development. The rate of intraoperative pneumothorax in our study was >2%, which may indicate that it is an underreported complication.
The original STAR trial was a multicenter prospective study conducted to demonstrate the efficacy and safety of HNS. It included 126 patients who underwent HNS implantation and reported nonserious procedure-related adverse events, such as postoperative discomfort, temporary tongue weakness, intubation effects, headache, and mild infection. 11 There were no cases of iatrogenic pneumothorax. In 2018, Arteaga et al published the first case report of pneumothorax in a patient undergoing HNS implantation, which occurred during the creation of a tunnel for chest lead insertion. 15 More recently, a study of 1849 patients enrolled in the Adherence and Outcome of Upper Airway Stimulation for OSA International Registry (ADHERE) found an intraoperative serious adverse event rate of 0.43%, with 2 cases of hematoma, 1 infection, and 1 pneumothorax. 18 The ADHERE is a prospective comprehensive database of surgical outcomes, patient-reported outcomes, and complications of HNS.13,18 It is designed to analyze outcomes outside of the clinical trial setting; however, not all implant centers are enrolled in this database, and it is composed of higher-volume surgeons. A 5-year analysis of adverse events voluntarily reported to the FDA MAUDE database (Manufacturer and User Facility Device Experience) noted complications that were not described in other clinical trials, including 5 cases of pneumothorax and 1 pleural effusion. 14 Yet, that study was unable to determine the frequency or risk of these complications due to the nature of the database.
Interestingly, we noted that a quarter of implant sites accounted for the entirety of the pneumothoraces experienced by patients in our study. In conjunction with the overall increased rate of pneumothorax as compared with the ADHERE analysis of higher-volume centers, 18 this may be an indication that the incidence of pneumothorax in HNS could be dependent on hospital case volume and surgeon experience. There may be variability in practice and technical error as more surgeons begin to offer this treatment to their patients. Another potential contributing factor is that the data obtained from the TriNetX Research Network are predominantly from academic HCOs. It has been reported that the overall incidence of iatrogenic pneumothorax in thoracic procedures is significantly higher in teaching than nonteaching hospitals. 19 With regard to HNS implantation, future studies are warranted to determine whether experience and case volume have an impact on complication rates.
When compared with patients who did not experience pneumothorax, those who did were more than twice as likely to have a preexisting diagnosis of chronic lower respiratory disease. Respiratory disease is a known risk factor for secondary spontaneous pneumothorax 20 and may have predisposed these patients to an iatrogenic pneumothorax in the setting of surgery.
The management of pneumothorax is dependent on its size as well as the hemodynamic stability of the patient. 21 Small pleural defects can be treated intraoperatively by inserting a red rubber catheter into the pleural cavity and closing with suture. The lungs are then hyperinflated to evacuate the pneumothorax, followed by removal of the catheter. 22 Larger defects or those that do not resolve may necessitate placement of a chest tube. Since none of the pneumothoraces experienced by patients in this study required a chest tube, it can be inferred that they were small. It is also unlikely that this complication had a major impact on the future use of the HNS given that none of these patients required revision or explant. Even though our study did not identify the need for a chest tube and the case by Arteaga et al was also managed with observation, 15 it should be noted that 2 of the 5 pneumothoraces reported to the MAUDE database required chest tube placement and hospitalization. However, none resulted in long-term patient harm. 14
In the traditional 3-incision approach for HNS implantation, the respiratory lead is placed via its own incision at the fifth intercostal space. Since receiving FDA approval in 2021, the standard technique has transitioned to a 2-incision approach. In this method, the respiratory sensing lead is placed transpectorally in the second intercostal space, deep to the implantable pulse generator. 23 Kent et al and Sagalow et al found no significant differences in rates of adverse events comparing the 2- and 3-incision approaches.23,24
There have been reports of pneumothoraces occurring during both techniques.15,23,24 In our study, <10 of 486 HNS implantations performed after FDA approval were complicated by a pneumothorax. While we cannot be certain of the surgical approach from our data, we hypothesize that some of these adverse events may have occurred via the 2-incision approach. Although there are indications that the 2-incision approach is noninferior in terms of efficacy and complication rates, future large-scale comparisons of adverse events are necessary as practice shifts. There are other HNS devices under active clinical trial that are designed without respiratory sensing leads, which may eliminate the risk of pneumothorax entirely.
Pneumothorax is not listed as a complication on the Inspire website, and most patients are likely unaware of this as a potential adverse outcome of surgery. Therefore, head and neck surgeons should communicate the risk of pneumothorax during the informed consent process.
The TriNetX Research Network is a direct translation of electronic medical records and does not rely on voluntary reporting of adverse events. This enabled determination of the frequency of pneumothorax as well as exploration of potential risk factors. Our study of 1813 individuals from 25 HCOs represents an additional large analysis of patients who underwent HNS implantation.
This study is not without limitations. We were unable to determine how each pneumothorax affected the course of surgery, including changes in vital signs and operative management. Since the TriNetX Research Network is based on diagnosis and procedure codes, it was not possible to analyze rates of specific complications that do not have corresponding codes. In addition, if a pneumothorax was noted at any point during or after surgery but was not coded as an official diagnosis, these adverse events would not have been included in our analysis. Therefore, it is possible that we may have underreported the true rate of pneumothorax.
Conclusion
HNS implantation is a safe treatment for OSA, as the overall rate of complications is low. However, the risk of pneumothorax may be greater than initially thought and should be communicated with patients during the informed consent process. Otolaryngologists should take care to prevent this complication, which may be more likely in patients with chronic lower respiratory diseases.
Supplemental Material
sj-docx-1-oto-10.1177_01945998221122696 – Supplemental material for Iatrogenic Pneumothorax During Hypoglossal Nerve Stimulator Implantation: A Large Database Analysis
Supplemental material, sj-docx-1-oto-10.1177_01945998221122696 for Iatrogenic Pneumothorax During Hypoglossal Nerve Stimulator Implantation: A Large Database Analysis by F. Jeffrey Lorenz and Neerav Goyal in Otolaryngology–Head and Neck Surgery
Footnotes
This article was accepted for presentation at the 2022 AAO-HNSF Annual Meeting & OTO Experience; September 10-14, 2022; Philadelphia, Pennsylvania. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Contributions
Disclosures
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
