Abstract
Objective
To determine the diagnostic value of measuring squamous cell carcinoma antigen (SCC-Ag) and cancer antigen 15-3 (CA15-3) concentrations in fine-needle aspiration (FNA) samples for the detection of squamous cell carcinoma (SCC) metastases in cervical lymph nodes.
Study Design
A prospective study with patients consecutively included between November 2018 and May 2021.
Setting
A tertiary head and neck oncologic center.
Methods
Out of 138 patients, SCC-Ag concentrations were analyzed in 168 FNA cervical lymph node samples and CA15-3 in 152 samples. Results were compared with FNA cytology (FNAC) or definitive histology to establish sensitivity and specificity rates.
Results
For the detection of cervical SCC lymph node metastases, SCC-Ag measurement had an 89.4% sensitivity and 79.3% specificity at a cutoff concentration of 0.1 µg/L. Measurement of CA15-3 concentration in addition to SCC-Ag concentration did not lead to improved accuracy for the detection of SCC. In histology-confirmed cases, FNAC had an 80.0% sensitivity and 100% specificity, as opposed to 93.3% and 57.1%, respectively, for SCC-Ag.
Conclusion
Measurement of SCC-Ag concentration for detection of SCC lymph node metastases has a sensitivity at least comparable to FNAC and could be used as a relatively cheap screening tool in samples with nondiagnostic or indeterminate FNAC results or when multiple lymph nodes are sampled. However, SCC-Ag in FNA samples has a lower specificity than FNAC assessed by pathologists experienced in head and neck oncology. Addition of CA15-3 measurement did not lead to improved accuracy.
Over 60% of patients with head and neck squamous cell carcinoma (SCC) present with advanced-stage disease, which is associated with a poor prognosis. 1 Therefore, early and rapid detection of lymph node metastases is of importance to improve clinical outcome.
We recently demonstrated in a retrospective pilot study a 96% sensitivity and 74% specificity for the detection of head and neck SCC lymph node metastases by measuring the SCC antigen (SCC-Ag) concentration in fine-needle aspiration (FNA) samples in a population of patients with a suspicious neck mass. 2 This objective measurement of biomarker concentration in FNA samples could serve as an adjunct tool with or without cytologic examination to detect SCC lymph node metastases.
High SCC-Ag concentrations can be seen in FNA samples of what later prove to be branchial cleft cysts, leading to false-positive SCC-Ag results. 2 This is a misdiagnosis that has also been reported in cytology results. 3 Since mucins are typically present on the apical surface of glandular or luminal epithelial cells, we hypothesized that measurement of cancer antigen 15-3 (CA15-3) as the soluble form of mucin 1 may reduce false-positive SCC-Ag results.4,5 In this prospective study based on a group of consecutive patients presenting with a neck mass suspicious for regional SCC metastases, we aimed to confirm our earlier findings of the feasibility of using SCC-Ag concentration for the detection of SCC lymph node metastases and to evaluate the potential additional value of CA15-3 concentration to increase the specificity.
Materials and Methods
Data Collection and Measurement
Between November 2018 and May 2021 at the University Medical Center Groningen, a total of 174 ultrasound-guided FNA samples were prospectively collected from 142 consecutive patients with masses in the neck that were suspicious for a malignancy. After preparation of the FNA samples for cytology, the residual material of the FNA samples (needle washout) was collected in 25 mL of Cytolyt (Hologic), separated from cytologic workflow, and stored at −20°C until SCC-Ag and CA15-3 measurements were performed, roughly every 6 months rather than immediately for practical reasons. The SCC-Ag analyses were conducted on a fully automated immunoassay analyzer (Architect; Abbott) with the SCC-Ag assay, with an interassay coefficient of variation <5% and an analytic sensitivity of 0.1 µg/L. CA15-3 measurements were performed with the CA15-3 assay on an automated immunoassay analyzer (Alinity; Abbott) with an interassay coefficient of variation <3% and an analytic sensitivity of 0.1 U/mL.
Collected data consisted of demographics and relevant medical history. In case of SCC lymph node metastasis, data were also collected for the following: radiologic information on lymph node size, smoking status including pack years, alcohol intake, and human papillomavirus status of oropharyngeal primary tumors for corresponding lymph node metastases. Cytology and histology results were collected. Histology was considered the gold standard (n = 36); however, if it was not present, the cytology diagnosis was used (n = 132).
In 6 samples of 4 patients, there was no definitive diagnosis after a nondiagnostic FNAC. These samples were excluded. This led to a total of 168 FNA samples from 138 unique patients. Multiple samples were taken in 23 patients, leading to 30 extra samples. Since the samples in these patients were often taken at different moments and in some cases led to a different diagnosis, patient data were analyzed as if there were 168 unique patients. In 16 cases insufficient material was left for a subsequent CA15-3 analysis, which was measured in 152 samples of 125 unique patients.
Patient Groups
For the evaluation of SCC-Ag concentration in the detection of SCC lymph node metastases in the neck region, FNA samples were divided into 4 groups based on cytologic diagnosis to better distinguish SCC-Ag concentration profiles: lymph nodes with SCC (ie, including cutaneous and esophageal SCC), other carcinomas, cystic lesions (including branchial cleft cysts), and other diagnosis.
This study was assessed by the Institutional Review Board (METc 2018/347) of the University Medical Center Groningen, and no approval was required per Dutch Medical Research Law (Wet medisch-wetenschappelijk onderzoek met mensen).
Statistical Analysis
Sensitivity and specificity rates were established through receiver operating characteristic curves. The Kruskal-Wallis test for nonparametric data was used to compare >2 groups. Two groups were compared via the Mann-Whitney U test for nonparametric data with post hoc Bonferroni corrections depending on the number of tests that were performed to compare all groups. Spearman rank correlation was used for comparing 2 continuous variables. P values <.05 were considered statistically significant. Statistical analyses were performed with SPSS version 23 for Windows (IBM Corp).
Results
Out of 168 patients, 111 were male (66%) and 57 were female (34%). The median age was 66 years (range, 31-90; Table 1 ). Primary tumor characteristics of the regional SCC lymph node metastases are described in Table 2 . The group of other carcinomas consisted of adenocarcinomas not otherwise specified (n = 5), Merkel cell carcinoma (n = 1), and epithelial-myoepithelial carcinoma (n = 1). The “other diagnosis” group mostly consisted of benign diagnoses, such as no malignancy, inflammation, or reactive lymph node. However, there were also non-Hodgkin lymphomas (n = 6), Warthin tumors (n = 2), a melanoma (n = 1), and a myofibroblastic tumor (n = 1).
Patient and Fine-Needle Aspiration Lymph Node Sample Characteristics.
Corresponding Primary Tumor Characteristics of SCC Lymph Node Metastases.

Abbreviations: OPSCC, oropharyngeal squamous cell carcinoma; SCC, squamous cell carcinoma.
Analysis of Neck Mass Samples
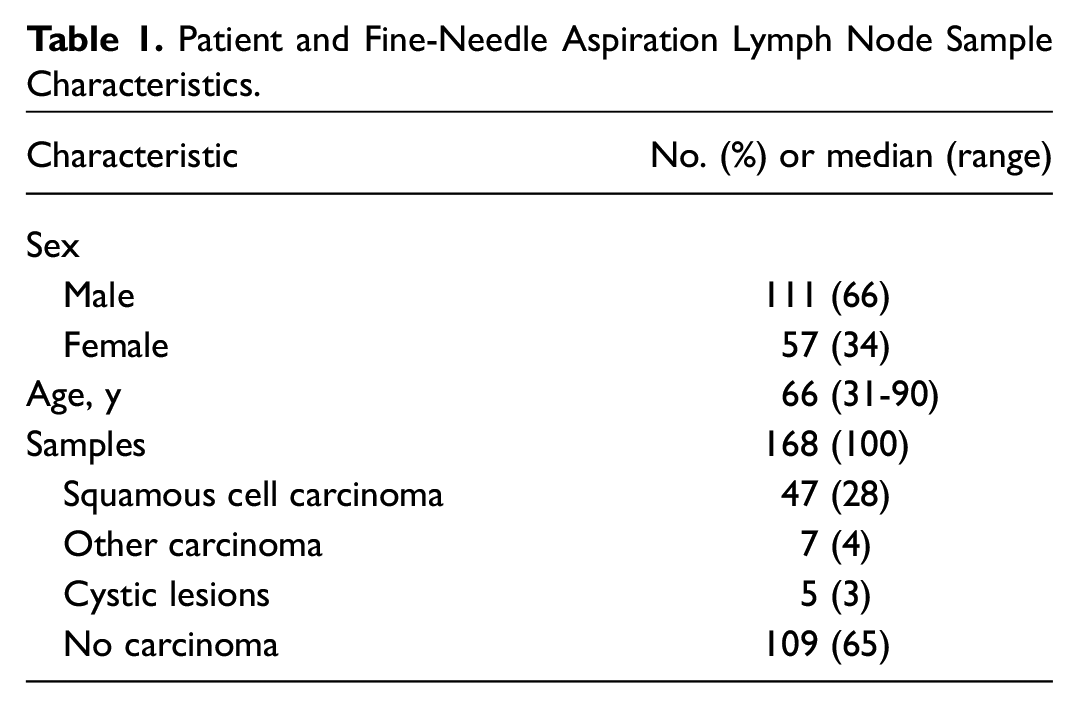
The SCC-Ag concentration in the various groups was not normally distributed. In samples of SCC lymph node metastases, the median concentration was 4.4 µg/L (range, 0.0-1547.5). In samples of other carcinomas, the median was 0.0 µg/L (range, 0.0-8.0); cystic lesions, 28.2 µg/L (range, 2.5-77.5); and other diagnosis, 0.0 µg/L (range, 0.0-0.5; Figure 1 ). The groups of other diagnosis and other carcinoma had significantly lower SCC-Ag concentrations as compared with SCC (P < .001 and P = .027, respectively) and cystic lesions (P < .001 and P = .046), while the SCC and cystic lesion groups did not significantly differ from each other. At the cutoff concentration ≥0.1 µg/L, SCC-Ag had a sensitivity of 89.4% and a specificity of 79.3% for the detection of SCC lymph node metastases. Tumor site of the primary SCC and p16 status in oropharyngeal SCC did not influence SCC-Ag concentrations (P = .81 and P = .18). Neither did the size of the lymph nodes with SCC metastases influence SCC-Ag concentrations (P = .43).
Distribution of SCC-Ag concentrations in fine-needle aspiration samples (logarithmic scale). Horizontal line, median; SCC, squamous cell carcinoma; SCC-Ag, SCC antigen.
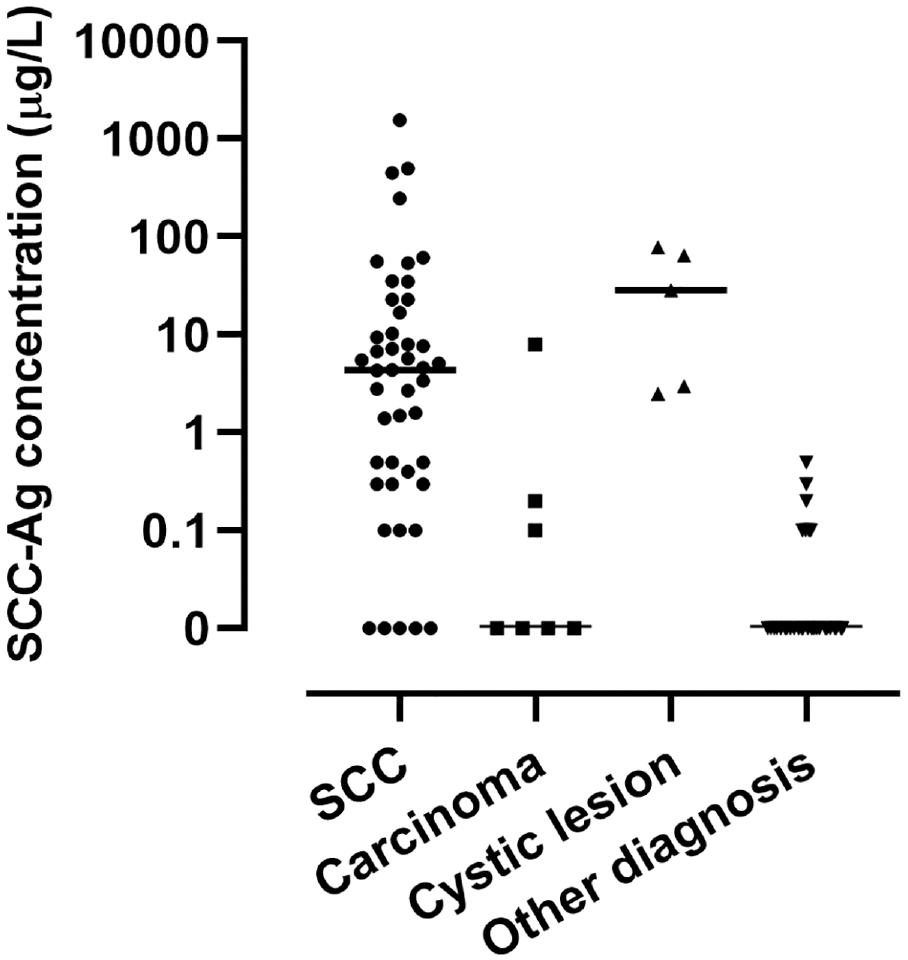
The median CA15-3 concentration in samples of SCC lymph node metastases was 0.0 U/mL (range, 0.0-9.7); other carcinomas, 0.0 U/mL (range, 0.0-1.1); cystic lesions, 0.8 U/mL (range, 0.0-9.4); and other diagnosis, 0.0 U/mL (range, 0.0-0.3; Figure 2 ). The CA15-3 concentration in SCC lymph node metastases was not different from the concentration in cystic lesions (P = .63).
Distribution of CA15-3 concentrations in fine-needle aspiration samples. Horizontal line, median; CA, cancer antigen; SCC, squamous cell carcinoma.
CA15-3 concentration measurement could be performed in 34 of the 47 SCC lymph node metastases. Within this group, the SCC-Ag sensitivity and specificity were 94.1% and 79.7%, respectively. Combining the 2 tests for the detection of SCC lymph node metastases, by (1) taking an SCC-Ag cutoff concentration ≥0.1 µg/L, and by (2) setting the CA15-3 cutoff concentration at <9.4 U/mL to filter out the cystic lesion with the highest CA15-3 concentration resulted in an 91.2% sensitivity and 80.5% specificity.
Analysis in Histology-Confirmed Samples
There were 36 histology-confirmed samples, of which 15 were SCC lymph node metastases. When only histology-confirmed FNAC samples were analyzed, the sensitivity for the detection of SCC lymph node metastases was 93.3% while the specificity was 57.1% at an SCC-Ag concentration ≥0.1 µg/L. FNAC had a sensitivity of 80.0% and specificity of 100% for the detection of SCC lymph node metastases.
In 4 cases, FNAC initially had an indeterminate or incorrect result. These cases were all SCC and had SCC-Ag concentrations of 0.1, 0.3, 6.8, and 34.7 µg/L.
Discussion
In this study we aimed to prospectively confirm the diagnostic value of SCC-Ag concentration measurement for the detection of SCC lymph node metastases. Additionally, we wanted to establish the value of additional CA15-3 concentration measurement to increase specificity of the SCC-Ag measurement. We found an 89.4% sensitivity and 79.3% specificity to detect SCC lymph node metastases at an SCC-Ag cutoff concentration ≥0.1 µg/L. Although the addition of CA15-3 could slightly increase specificity, it did not improve accuracy.
In our previous retrospective study, we reported a sensitivity of 95.8% and specificity of 74.4% for the detection of SCC lymph node metastases. 2 In the current prospective study, there was a slightly lower sensitivity of 89.4% and a slightly higher specificity of 79.3% for the detection of SCC lymph node metastases. This in comparison with FNAC, for which we found an 80.0% sensitivity and 100% specificity for the detection of SCC lymph node metastases in only histology-confirmed samples. According to the literature, a sensitivity of 82% to 92% and specificity of 97% to 100% can be reached for the detection of head and neck SCC lymph node metastases with FNAC, while FNAC correctly gives a specific diagnosis to lesions in lymph nodes in 84%.6-8
We have demonstrated that the use of FNA samples for the detection of SCC lymph node metastases with SCC-Ag is technically feasible and can be used as an objective, cheap, and easily applicable technique with a sensitivity at least comparable to FNAC. It can be measured in the same FNA sample as the one used for cytology and therefore does not require extra samples to be taken. In our institute, the cost of the measurement of SCC-Ag is a mere 8.5% the cost of FNAC, and results can be available within 2 hours. Moreover, additional FNA samples from multiple enlarged lymph nodes can be taken in each patient for screening and staging purposes. The largest lymph node could be assessed by cytologic examination per standard of care, and if diagnosed as SCC, additional (eg, contralateral) lymph nodes can be evaluated through SCC-Ag, without a great increase in costs or time-consuming cytologic assessments. The measurement of SCC-Ag could therefore serve as a supportive screening tool in patients with cytologically proven cervical SCC lymph node metastasis for staging purposes.
Furthermore, the measurement of SCC-Ag concentrations in FNA samples can be used as an additional diagnostic modality in smaller institutes without experienced teams specialized in the assessment of FNAC or management of SCC in the head and neck region. A finding of elevated SCC-Ag levels in an FNA sample of a suspicious neck mass might help the pathologist or prompt evaluation by a head and neck oncologist. In lower-resource settings without a well-equipped laboratory, SCC-Ag measurement can be performed by enzyme-linked immunosorbent assay.
An interesting subgroup for SCC-Ag is the FNA samples with nondiagnostic or indeterminate results after cytologic analysis, which occur in 2.6% to 9.6% of cases.9-11 Apart from the 6 excluded samples with a nondiagnostic FNAC result, there were cases present in this study with an indeterminate or incorrect initial diagnosis, of which 4 were SCC lymph node metastases. The 4 SCC cases that had a false-negative FNAC result initially had a diagnosis other than carcinoma (2 cases with a diagnosis of no malignancy and 2 in which no classifying diagnosis could be determined), while they did have a positive SCC-Ag test result. In 1 case it took 5 months until SCC was eventually diagnosed. If an SCC-Ag test had been performed due to discrepancy between clinical suspicion and FNAC result, the positive SCC-Ag result could have indicated that the patient required a closer and timelier follow-up and could have led to a sooner definitive diagnosis and start of treatment.
Larger studies are required to accurately determine the sensitivity and specificity and fully elucidate the clinical value of SCC-Ag for the detection of SCC lymph node metastases, before it can potentially play a role in decision making or be performed as screening prior to or instead of cytologic examination.
We hypothesized that the additional measurement of CA15-3 concentration after SCC-Ag measurement could help reduce false-positive results by better distinguishing SCC lymph node metastases from cystic lesions. Even though a slight increase in mucin 1 expression was reported in head and neck SCC lymph node metastases, we hypothesized the CA15-3 concentration to be significantly higher in branchial cleft cysts as compared with SCC lymph node metastases. 12 Although the median CA15-3 concentration was higher in cystic lesions (0.8 U/mL) than in SCC lymph node metastases (0.0 U/mL), the difference was not significant, and the addition of CA15-3 measurement to increase specificity did not lead to clinically desirable results. To filter out 1 cystic lesion, the sensitivity dropped from 94.1% to 91.2% for only a slight increase in specificity from 79.7% to 80.5%. False-positive cases in the “other diagnosis” group that could not be filtered out by CA15-3 measurement were 2 Warthin tumors, 4 lymph nodes with inflammation, and 1 metastasis of a myofibroblastic tumor.
In our analysis of histology-confirmed samples, the sensitivity rate of SCC-Ag rose to 93.3% and specificity dropped to 57.1% for the detection of SCC lymph node metastases. The increase in sensitivity can be explained by the fact that histologic results of some metastases of unknown primary tumors were not available and had an SCC-Ag concentration of 0.0 µg/L. These patients had residual or recurrent SCC metastases, and earlier neck dissections (eg, contralateral side) showed that the lymph node metastases of these unknown primary tumors had been p16 negative. The loss in specificity, however, occurs because biopsy or surgery is usually performed after a diagnosis is confirmed by FNAC. Therefore, there is a selection bias for lesions, which consequently leads to more positive SCC-Ag findings, more false positives, and a lower specificity. This hampers the direct comparison of SCC-Ag and FNAC results in histology-confirmed samples.
In line with our earlier findings, p16 status did not lead to differences in SCC-Ag concentrations. The SCC-Ag serum concentration is used as a prognostic marker in cervical cancer.13,14 Due to the short follow-up time of 0 to 2.5 years since the start of this study, we were not able to evaluate the prognostic value in lymph node samples. However, since great variations in SCC-Ag concentration were seen in patients with a second FNA of their SCC lymph node metastasis after a relatively short follow-up, we suspect the concentration to be dependent on the amount of harvested material rather than lymph node size or other tumor characteristics.
In the present study, SCC-Ag and CA15-3 concentrations were measured in FNA samples. To our knowledge, this method of measuring SCC-specific protein levels in FNA samples has not been studied previously other than in our retrospective study. We have proven that the measurement of SCC-Ag protein concentrations in FNA samples is technically possible and feasible for diagnostic purposes in the detection of SCC lymph node metastases. The choice for SCC-Ag and CA15-3 was based on assumptions rather than earlier findings. More research into proteins specific for SCC or other lesions (eg, melanoma, thyroid gland carcinoma, malignant lymphoma) that may be measured in FNA lymph node samples may therefore lead to better results or the possibility to differentiate among lesions when a panel of tumor markers sensitive for the various causes of neck masses is measured.
Conclusion
The measurement of SCC-Ag concentration as a tumor marker in FNA samples is a feasible option in the diagnosis of SCC in cervical lymph node metastases. Although SCC-Ag measurement is not as accurate as FNAC, its sensitivity is at least comparable, and it is a cheap and supportive method to screen for SCC. CA15-3 has no added value in the detection of SCC lymph node metastases. Future studies could therefore investigate the diagnostic value of SCC-Ag in nondiagnostic and indeterminate FNAC results and examine the concentration of other tumor markers even more specific for SCC in FNA samples.
Footnotes
Acknowledgements
We kindly thank the administrators of the ultrasound section of the Department of Radiology for their assistance and the cytologic technicians, especially T. Ruitenbeek, pathologic technician at the Department of Pathology, for saving the aspiration samples in Cytolyt. We also thank M. Scholten, technician at the Laboratory of Binding Analysis, Department of Laboratory Medicine, for performing the SCC-Ag and CA15-3 measurements.
Authors’ Note
Bernard F.A.M. van der Laan’s current affiliation is the Department of Otorhinolaryngology–Head and Neck Surgery, Haaglanden Medical Center, The Hague, the Netherlands.
