Abstract
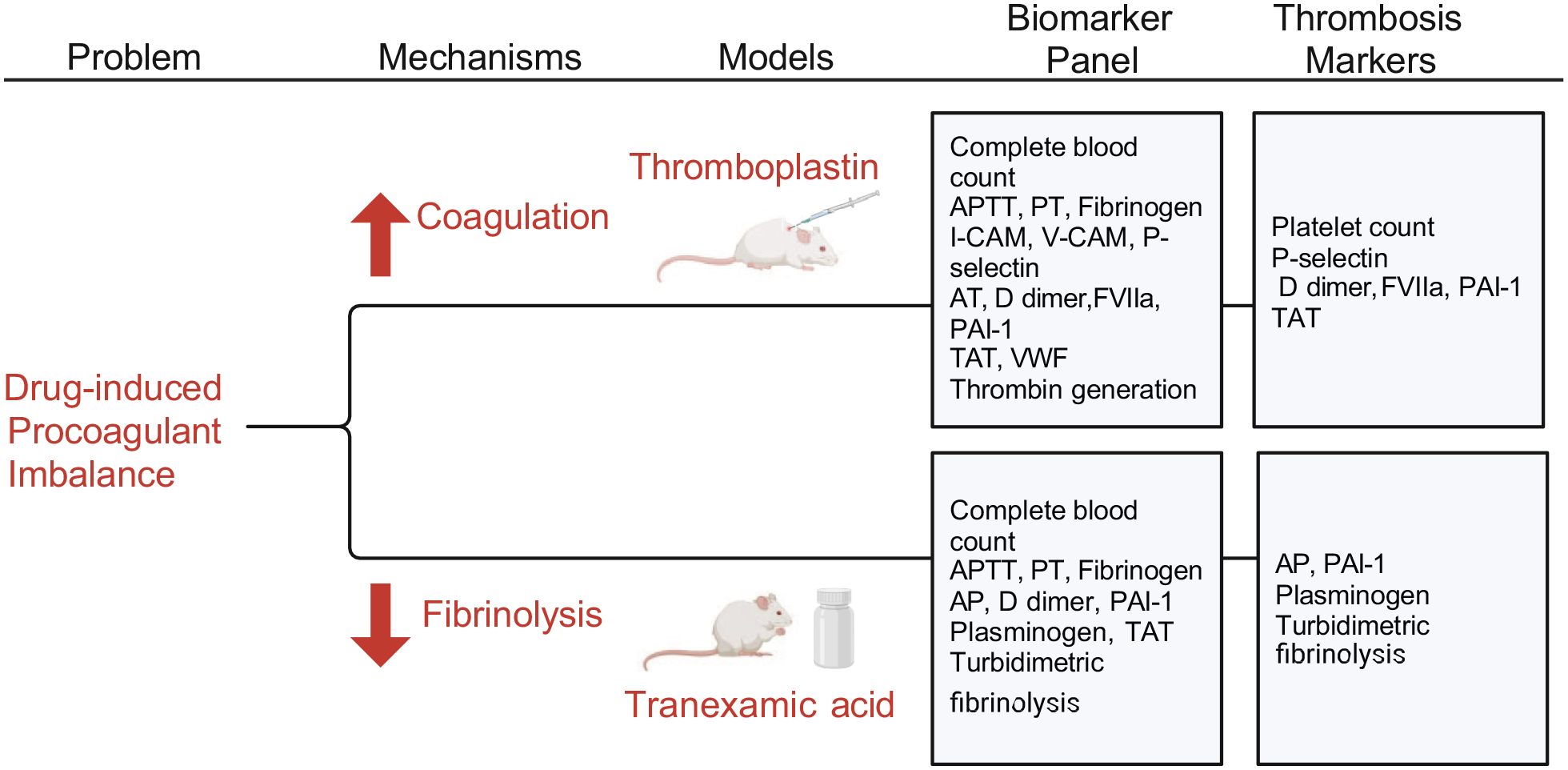
Thrombotic complications including myocardial infarction, stroke, venous thrombosis and pulmonary thromboembolism are common causes of drug attrition often discovered at late stages of drug development. Current nonclinical safety assessments include screening tests that detect hemorrhagic complications but do not identify conditions signaling a risk of thrombosis. Our study aimed to identify sensitive tests for detecting prothrombotic imbalance, without overt thrombosis, for use in early nonclinical drug safety assessments in rodents. Sprague Dawley rats were administered different doses of thromboplastin or tranexamic acid to induce variable intensity hypercoagulable or hypofibrinolytic states, respectively. A panel of functional and quantitative assays measuring hemostatic proteins and pathways were evaluated, in concert with traditional coagulation screening tests and blood cell counts. Profound changes were observed with different patterns of test abnormalities for the different stimuli. Measurements of D-dimer and thrombin antithrombin complex concentrations, plasminogen activator inhibitor-1 and Factor VIIa activity were among the most sensitive tests of hypercoagulability. In contrast, hypofibrinolysis was best characterized in a kinetic, turbidimetric assay. Traditional coagulation screening tests were relatively insensitive, and no single test defined the cause of prothrombotic imbalance. Our results demonstrate that customized biomarker panels can detect early drug-induced prothrombotic states in rats arising from distinct mechanisms.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
