Abstract
Sharing pathology data is critical for educational and scientific purposes. Since most pharmaceutical or (agro)chemical companies outsource nonclinical safety assessment studies to contract research organizations (CROs), the pathology data of those studies are not owned by the investigator but is the legal property of the respective company sponsoring the work. Although some companies have installed policies that govern sharing of pathology data, many companies generally do not allow the external use of data by either the CRO-based study pathologist or the sponsor pathologist. Policies for governing the external use of data vary significantly. In this article, we present an overview of the different approaches taken across different companies (CROs, pharmaceutical/chemical companies, or other institutes) for sharing pathology material for educational and/or scientific purposes. The results of a survey and interviews with legal departments of different companies will be presented (anonymously) and discussed. In addition, the importance of sharing pathology data is addressed, as well as the challenges and opportunities this presents. Suggestions will be provided regarding what material should be made available and what will be needed to achieve agreement for this to happen.
Keywords
Introduction
Toxicologic pathology is a scientific discipline that evaluates and interprets morphological or molecular alterations in animal tissues that are associated with the effects of xenobiotics, ie, mostly industrial chemicals and pharmaceuticals. This discipline plays an essential role in the characterization of hazards for human health, safety assessment in drug development, and contributes to regulatory decisions on product approval, safety classification and labeling, or the mandatory use of protective equipment at the workplace. Scientists working in this field typically undergo comprehensive training in veterinary pathology and subsequently gain experience by evaluating tissues from a wide variety of nonclinical and environmental safety studies in a variety of species. Adequate training material is needed to gain experience or maintain qualifications, yet it is recognized that material for scientific or educational purposes is increasingly difficult to obtain. One of the primary reasons for this is the ownership, perceived risk for competitive advantage, and protection of such material. Safety studies are typically sponsored by industrial corporations and are increasingly outsourced to contract research organizations (CROs), creating a dichotomy between those who generate the data and those who own the data. The CROs are bound to confidentiality by master service agreements (MSAs), and industry corporations commonly fear that the unique properties of a test item, its unique mechanism, and/or its profile of side-effects may be given away to competitors who could use this information for their benefit. Therefore, using pathology data from (nonclinical and environmental) safety studies for scientific investigations or educational purposes has become increasingly difficult. Although some industrial corporations have installed policies for the use of pathology data, those policies vary considerably among companies.
We advocate that anonymized pathology data from preclinical safety studies should be made available as scientific or educational material, taking the following aspects into consideration. First, pathology data and images of morphologic changes can easily be anonymized so that no relation to a specific study, sponsor, or test article is possible. However, for some test articles, recognition of a certain compound class cannot be avoided based on known class effects. Second, the variety of xenobiotics to which humans are exposed has increased massively, which requires that toxicologic pathologists share their knowledge (eg, on rare incidental findings, on genetic drifts in background findings in laboratory animals, on the pathogenesis of specific lesions, or on species differences or compound class effects). Third, many experienced toxicologic pathologists will retire over the next decade, resulting in a need to train pathologists early in their career. This inevitably means that high-quality and up-to-date training material must be available. Fourth, the field of toxicologic pathology is rapidly evolving, with many artificial intelligence (AI)-based algorithms being developed for advanced evaluation of pathology images. Such algorithms can only be trained if abundant high-quality pathology material is available.
With an awareness of the above limitations, a working group was formed with pathologists from different industrial sectors (pharmaceutical corporations, biotechnology corporations, [agro]chemical corporations, CROs) and from different international toxicologic pathology societies (Society of Toxicologic Pathology [STP], British Society of Toxicological Pathology [BSTP], French Society of Toxicologic Pathology [SFTP], European Society of Toxicological Pathology [ESTP], Dutch Society of Toxicologic Pathology [Dutch-STP]). The working group conducted a survey across the industry and across publicly funded research institutes. The following aspects were reviewed and discussed:
Common practice on how pathology material is shared for scientific or educational purposes.
Common practice regarding for what specific purposes study material is shared.
Arguments as to why the sharing of pathology data is important for the scientific discipline of toxicologic pathology.
The type(s) of pathology data that is (are) required for scientific or educational purposes.
The risks that are associated with the sharing of pathology data.
The use of pathology data as historical control data (HCD).
Requirements that would need to be fulfilled for the sharing of pathology data.
Specific examples where pathology data have been shared for educational or scientific purposes.
Materials and Methods
Survey
To capture the current practice on sharing and using pathology data for scientific and educational purposes, two different questionnaires were prepared: one for CROs and one for non-CROs (ie, any industry corporation in the biotechnology, pharmaceutical or [agro]chemical sector, or any publicly funded research institute). Both questionnaires addressed the type of pathology data that is shared, the conditions under which pathology data is shared, and how shared pathology data is used. Definitions used in the questionnaire were as follows:
Pathology data (including tabulated formats): pathology data include (a) microscopic diagnoses, (b) microscopic/macroscopic photographs, (c) whole slide images (WSIs), (d) macroscopic observations, (e) organ weights, (f) other relevant in-life endpoints (in-life observations, clinical pathology, sperm parameters, pharmacokinetic/toxicokinetic [PK/TK] data, immunophenotyping, reproductive parameters).
Anonymous: (a) no traceability to study/study number, (b) no traceability to sponsor, and/or (c) no specific information is given of the test item, but very general information of the test item can be given (such as class or therapeutic area) when relevant.
Scientific exchange/consultation with peers/experts: consulting experts outside your own, and the sponsor’s, organizations.
HCD: a data set consisting of findings/data from control or vehicle control animals from different studies and/or sources and/or sponsors. These HCD are used for the determination of background incidences and to help to distinguish background from test item-related findings/data.
The questionnaires were distributed via different societies for toxicology pathology (STPs), namely STP-India (STP-I), USA-STP (STP), French-STP, Hungarian-STP, Dutch-STP, British-STP (BSTP), Chinese-STPs (CSOT, CSPS), Japan-STP (JSTP), Latin American-STP (LASTP), Korean-STP (KSTP), and European-STP (ESTP). In addition, questionnaires were distributed through the European College of Veterinary Pathologists (ECVP), the European Society of Veterinary Pathology (ESVP), and the International Academy of Toxicologic Pathology (IATP).
Forty questionnaires with responses were returned to the working group, representing 13 different CROs and 23 non-CROs.
Data Analysis
For the analysis of the responses, the CRO survey data were split into two categories: 4 responses were classified as “Large CRO” (ie, an organization with globally more than 15 pathologists) and 9 responses were classified as “Small CRO” (ie, an organization with fewer than 15 pathologists). According to the responses received, the majority of CROs were generating pathology data in safety assessment, a minority of CROs also generated pathology data in the area of discovery, investigative, or translational pathology.
For the analysis of the responses, the non-CRO survey data were split into 11 responses from industry corporations (ie, biotechnology, pharmaceutical, or [agro]chemical corporations), and 12 responses from publicly funded institutes (ie, academia, diagnostic institutes, or animal disease/surveillance institutes). The majority of responding pathologists worked in pharmaceutical corporations or academia; fewer pathologists worked in the areas of animal disease and/or diagnostics, and only a single representative of the agrochemical industry responded to the survey.
Results
General Approach to Sharing of Pathology Data
The first questions in the questionnaire explored if toxicologic pathologists from the different organizations are allowed to share pathology data with colleagues outside of their organization, if they experience difficulties in sharing pathology data, and—in cases when they do experience difficulties—what these difficulties are. In addition, it was asked what procedures their organization had established for the sharing of pathology data.
On the question of whether pathologists experience difficulties in sharing pathology data, more than 50% responded with YES, both in the CRO and in the non-CRO cohort (Figure 1). Differences between the CRO and the non-CRO sector were negligible. Within the CRO sector, pathologists working for Large CROs experienced more difficulties than those working for Small CROs. Within the non-CRO sector, pathologists working for pharmaceutical corporations experienced more difficulties than those working for other industry sectors.
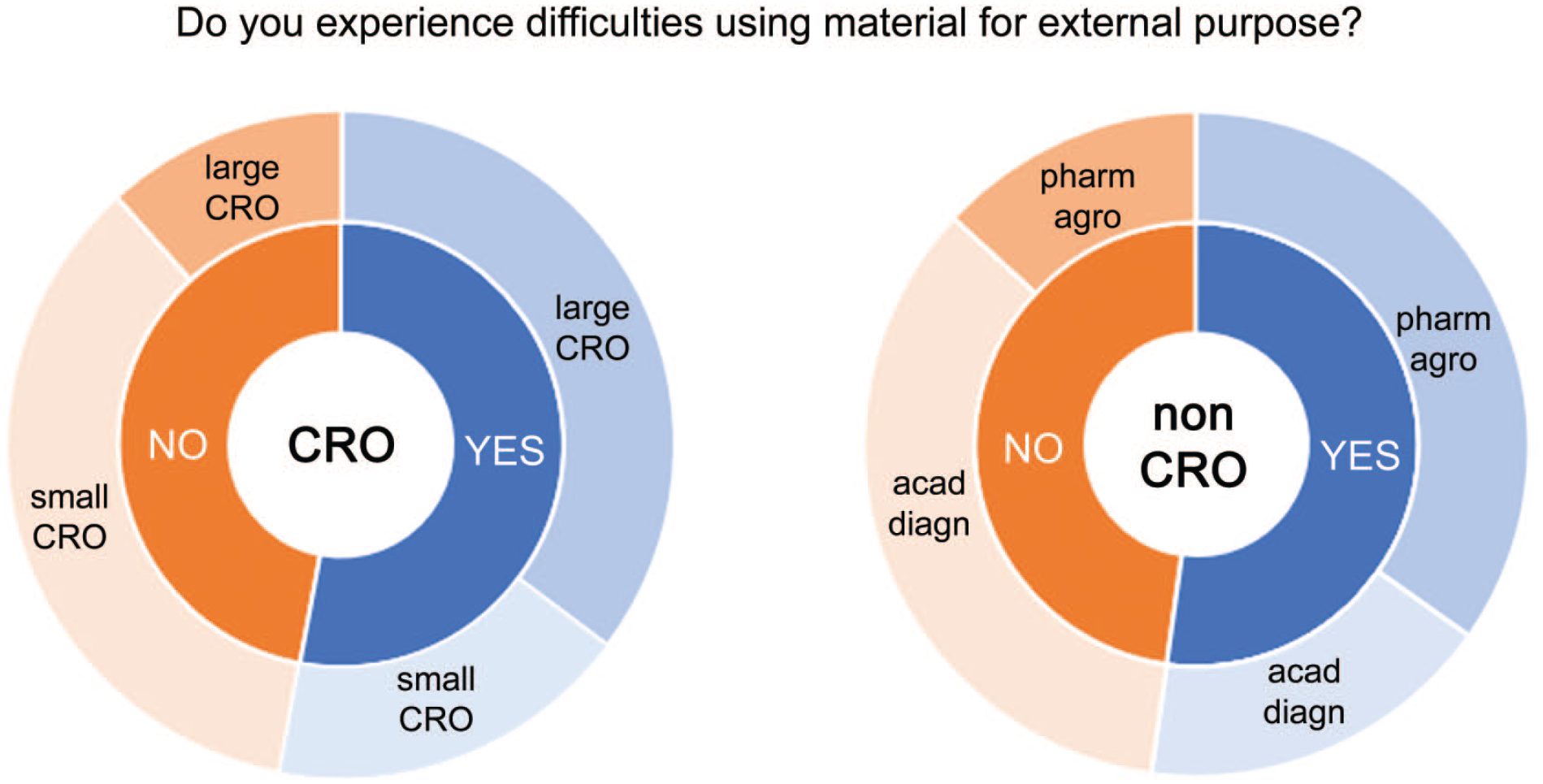
Responses for the question “Do you experience difficulties using material for external purpose?” Answers were YES or NO (inner circle) and were split between Large and Small CROs (left) and between non-CROs (right), ie, pharmaceutical/(agro)chemical corporations (pharm/agro), and academic/diagnostic (aca/diagn) institutes (outer circle).
Pathology Data From Vehicle Control Groups
The questionnaire explored if the sharing of pathology data from vehicle control groups is handled differently than pathology data from test article-treated animals. Most respondents indicated that the sharing of pathology data from vehicle control groups was allowed. A positive response was higher in CROs compared with non-CROs (Figure 2). Some organizations allow the sharing of such data without any restrictions (nonconditional approval), whereas others request that the data are anonymized (ie, without any link to the study or sponsor), or that sponsor approval is obtained with or without anonymization.
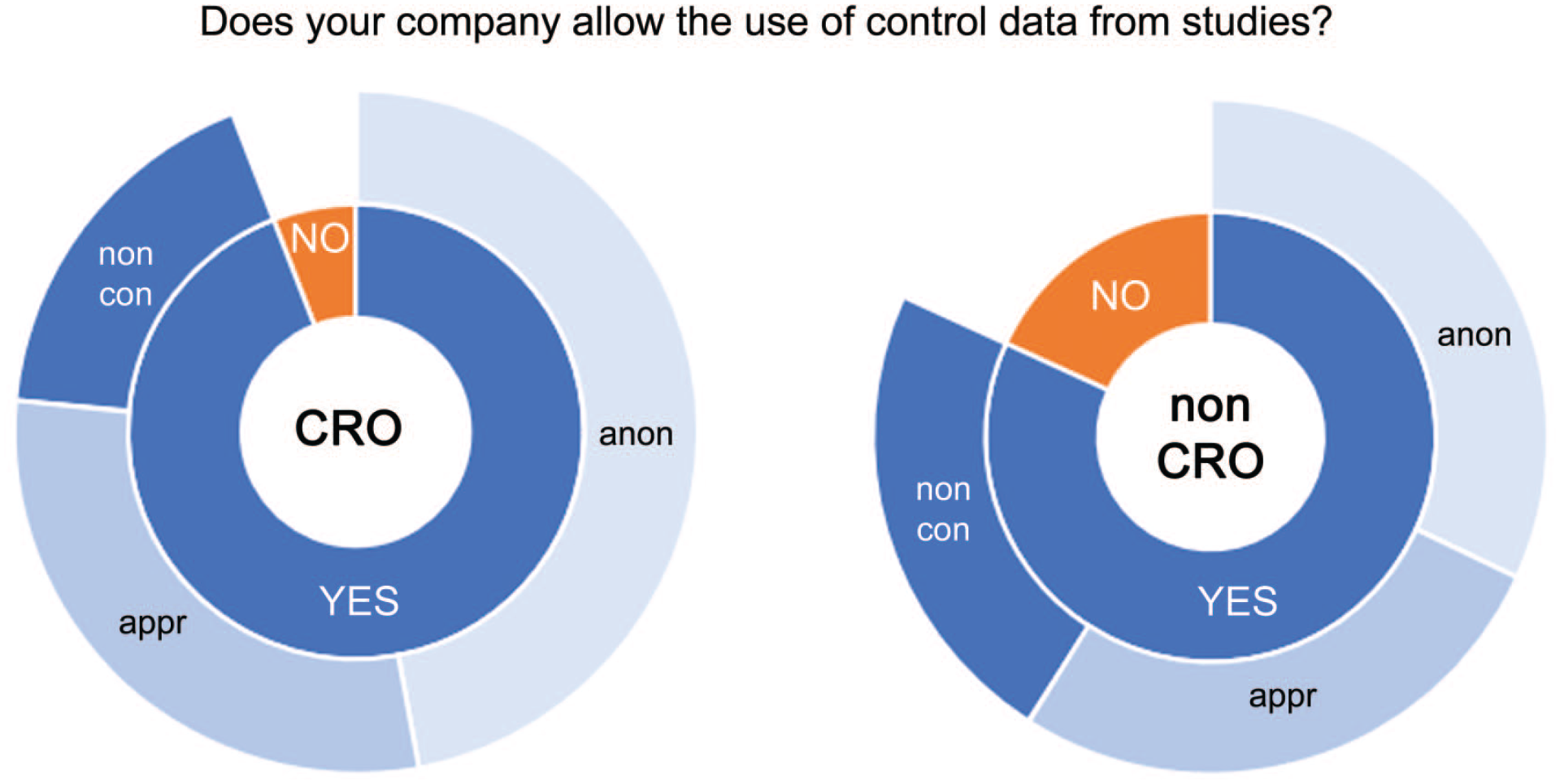
Responses of CRO (left) and non-CRO (right) for the question “Does your company allow use of control data from studies?” Answers were YES or NO (inner circle) and were split between nonconditional approval (noncon), approval (appr), and anonymization (anon) (outer circle).
Vehicle control pathology data are used for the following purposes: education (internal and/or external), webinars, publications, or scientific exchange (data not shown). In more than half of the responding organizations, no distinction was made as to the purpose of data sharing. In others, data sharing was restricted to educational material and/or webinars. There was no clear difference between the answers from Small CROs and Large CROs. There was also no clear difference in the answers from the pharmaceutical/chemical corporations compared with those from academia/publicly funded research institutes or animal disease/diagnostic institutes.
Pathology Data From Test Article-Treated Animals
Concerning sharing of pathology data from test article-treated animals, more CROs answered with NO compared with the sharing of data from vehicle control groups, but still a majority of CROs allowed the sharing of such data with sponsor approval (Figure 3). For all CROs, except one, education, presentations, webinars, publications, and scientific exchange were all acceptable purposes for the sharing of data. In a single CRO, allowance was limited to education and/or webinars only. Again, no clear difference in the responses was found between Small CROs and Large CROs. For non-CROs, the conditions for the sharing of data from test article-treated animals were very similar to those for vehicle control groups. Approval from management was required in 45% of non-CROs, which included all pharmaceutical/chemical companies, and anonymization was required in 23% of non-CROs. As with control materials, education, presentations, webinars, publications, and/or scientific exchange were acceptable purposes for pathology data sharing. Some respondents stated that the use of such data was limited to education and/or webinars.
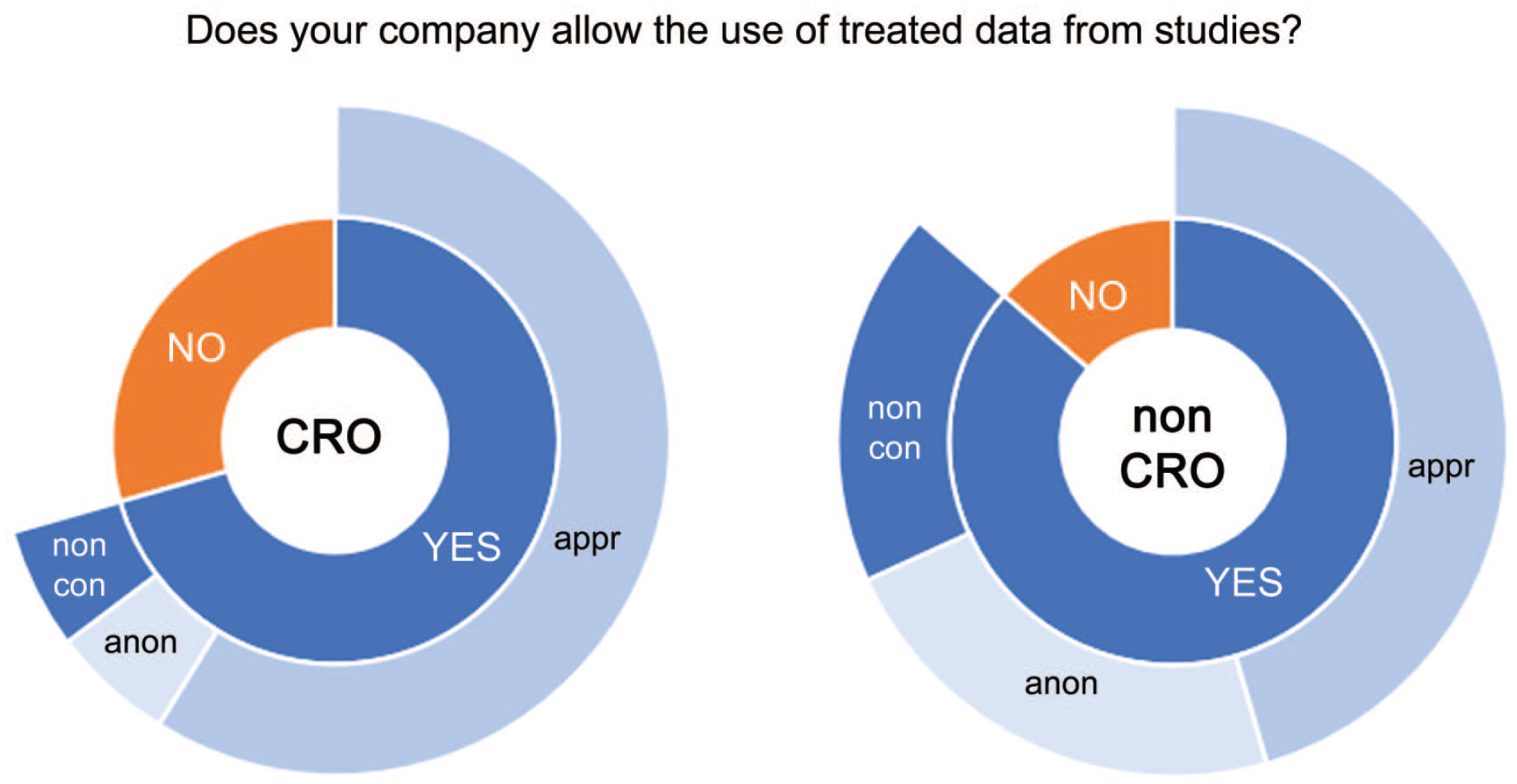
Responses of CRO (left) and non-CRO (right) for the question “Does your company allow use of treated data from studies?” Answers were YES or NO (inner circle) and were split between nonconditional approval (noncon), approval (appr), and anonymization (anon) (outer circle).
The questionnaire also explored if the sharing of pathology data from Good Laboratory Practice (GLP) studies is handled differently to pathology data from non-GLP studies (data not shown). In the majority of CROs, and in all non-CROs, no difference was noted in the handling of pathology data from GLP studies vs non-GLP studies. For both GLP and non-GLP control data, Large CROs allowed sharing when the data were anonymized. In Small CROs, sponsor approval was needed. In the majority of non-CROs, the sharing of GLP and non-GLP control data required approval from management. In the case of GLP and non-GLP data from treated animals, the majority of CROs required sponsor approval, and approval from management was a requirement in most non-CROs. The few organizations that managed data from GLP and non-GLP studies differently stated that non-GLP data could be used without any conditions while using data from GLP studies required that the data be anonymized.
Furthermore, the questionnaire explored if organizations allowed generation of new slides from tissue blocks (eg, for specific staining or embedding of new samples from formalin-fixed tissues [data not shown]). For non-GLP studies, two third of CROs and almost all non-CROs accepted this practice, mostly with the approval of the sponsor (CRO) or management (non-CRO). No approval was needed in academia. For GLP studies, only half of the CROs and 75% of the non-CROs accepted the processing of new slides or new blocks. In most of these cases, approval from the sponsor or management was needed. Half of the CRO responders and 30% of the non-CRO responders did not allow new slides or blocks to be generated (it should be noted that some of the participating non-CROs do not perform GLP studies).
Another question was: “When approval is needed from your company, what condition(s) allow(s) the use of the pathology data?” The answers were quite variable. In many CROs, a case-by-case approach was taken (ie, approval was mainly driven by the sponsor and needed to be documented in a written agreement or the study protocol). In some CROs, additional approval from management and/or their internal legal department was needed. In some of the academic institutes, no approval was needed. In non-CROs from industry, management and/or legal approval was required.
Historical Control Data
The survey also evaluated if pathology data are used as HCD. For CROs, the majority of the responses was YES, with an even distribution across Large CROs and Small CROs (Figure 4). For non-CROs, the majority was also YES. Responses from pharmaceutical and (agro)chemical corporations were mainly YES, whereas the responses from academic/diagnostic institutes were evenly distributed between YES and NO.
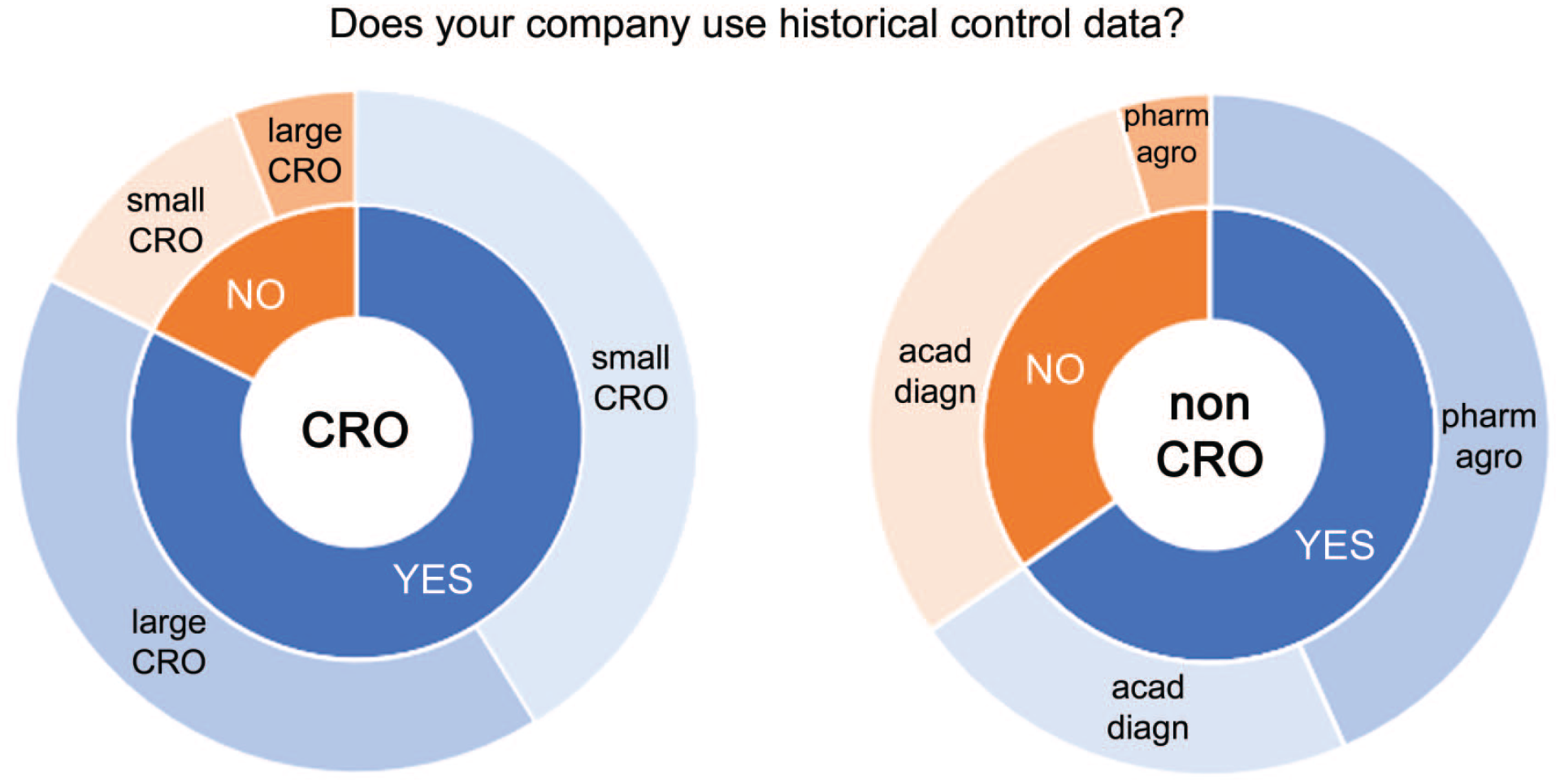
Responses of CRO (left) and non-CRO (right) for the question “Does your company use historical control data?” Answers were YES or NO (inner circle) and were split between Large CROs and Small CROs (left) and between non-CRO (right), ie, pharmaceutical and (agro)chemical corporations (pharm/agro) and academic/diagnostic institutes (acad/diagn) (outer circle).
Additional questions addressed the type of pathology data used as HCD. The CROs and pharmaceutical/chemical companies all use HCD from all available sources (ie, pooled control data from own company or own CRO, from company data for work performed at a CRO, control data from other CROs/companies, and/or data from suppliers, the literature, the Registry of Industrial Toxicology Animal data [RITA] database). Academic institutes do not typically use HCD. Generally, pharmaceutical/chemical companies have multiple available sources, whereas academia is limited to internal data.
Archived Materials
Typically, study materials are archived once the study is completed. The survey evaluated if retrieval of material from the archive for educational or scientific purposes is supported (Figure 5). In CROs, archive retrieval was not allowed without the approval of the sponsor (Figure 5A). There was no difference between Large CROs and Small CROs; there was also no difference between raw material/slides/blocks and photograph/scans of archived material. A slightly smaller number of non-CROs, compared with CROs, did not allow retrieval of archived material without approval. There was no difference between academia/institutes and industry (pharmaceutical/chemical). Surprisingly, there was a notable difference between raw material/slides/blocks and photograph/scans of archived material (Figure 5B). In 65% of the non-CROs, photographs/scans of archived material could be used without approval. Most of the 65% were representatives of academia/diagnostic institutes.
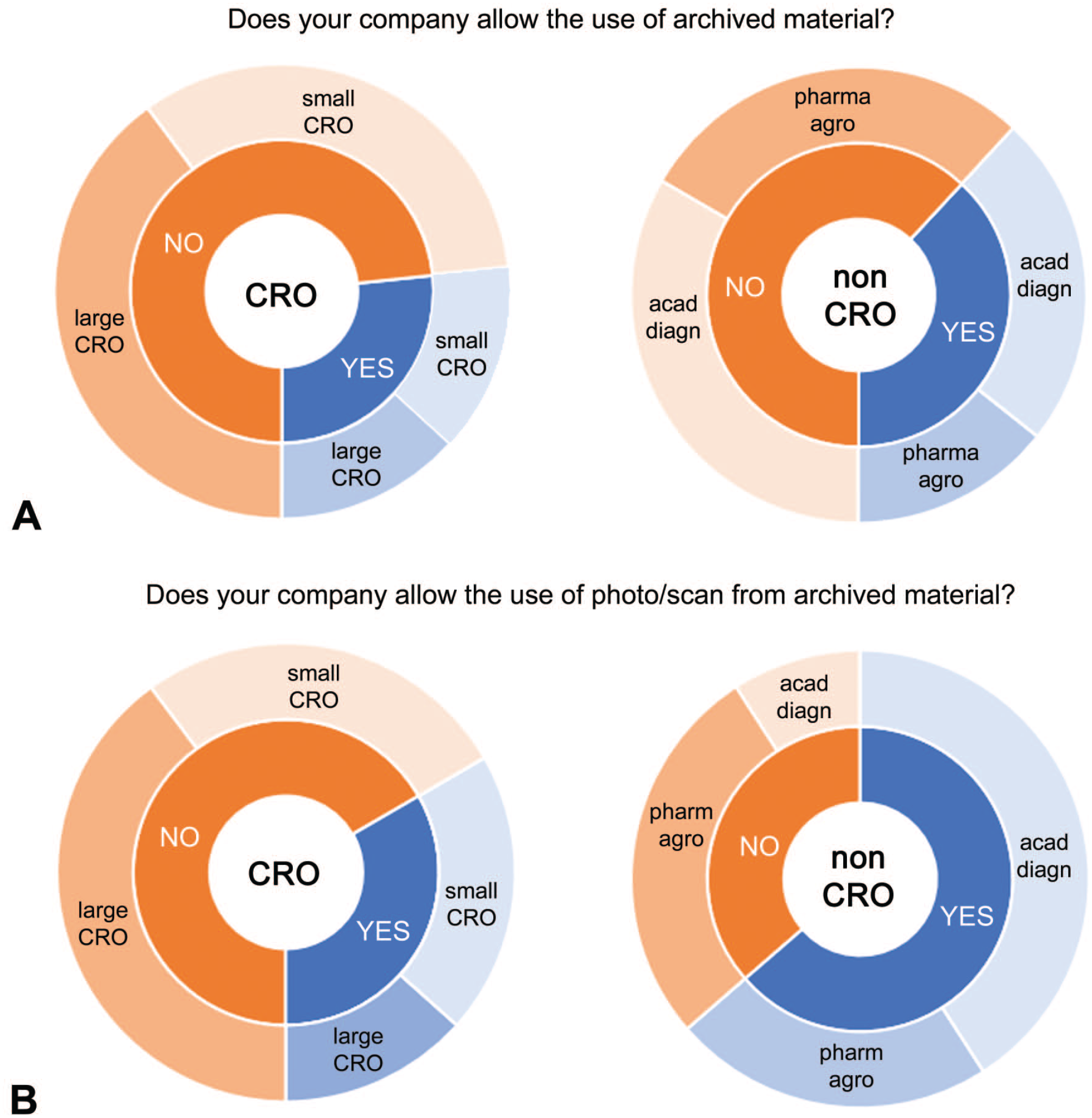
Responses of CROs (left) and non-CROs (right) for the questions (A) “Does your company allow use of archived material without approval?” and (B) “Does your organization allow use of photo/scan from archived material?” Answers were YES or NO (inner circle) and were split between Large and Small CROs and between pharmaceutical and (agro)chemical corporations (pharm/agro) and academic/diagnostic institutes (acad/diagn).
Discussion
Interpretation of Survey Responses
As is often the case with surveys, it should be noted that the interpretation of some of the questions may have differed between responders. For example, some pathologists may not have experienced any difficulties because the external use of pathology material is not permitted, whereas other pathologists do not experience difficulties because their company is very supportive of data sharing. In contrast, the cause of any difficulties may mean that procedures are complex and time-consuming, but it may also indicate that procedures are unclear. The survey was meant to give a global indication of the common practices and procedures within different organizations sharing pathology data externally (ie, outside of the organization). It was remarkable that the experience of pathologists could be completely different, not only between different organizations but also within the same organization at different sites. This strongly indicates that there is enormous variability in handling pathology data for external purposes between organizations but also within organizations.
The Use of Pathology Data as Historical Control Data
It is well acknowledged by pathologists from industry and regulatory agencies that HCD can be crucial for the interpretation of animal pathology data. Most publications involving HCD focus on rodent carcinogenicity studies where HCD can aid in the interpretation of tumor data, especially in studies with rare tumors or marginally increased incidences of a specific tumor type. 3 In Europe, a centralized database system known as the RITA was founded in 1988, aiming to provide standardized and valid HCD for carcinogenic risk assessment.
The HCD can also assist with the interpretation of general toxicity studies in animals. Such studies might be confounded by various factors such as animal strain, animal supplier, housing conditions, route of exposure/administration, or diet.2,7 It is also well known that over time genetic drift occurs within inbred strains of animals; this may result in changes in the type or incidence of background (spontaneous) pathology.
The HCD can be particularly valuable for studies conducted in nonrodent animal species. One obvious example is the Cynomolgus monkey, in which the spontaneous background pathology differs significantly between animals originating from Asia and animals originating from Mauritius, 1 and the animal numbers per group and sex in nonclinical studies are low.
In recent years, the idea of replacing concurrent control groups in preclinical safety studies with a group of virtually created animals, known as a virtual control group, has gained increasing attention.4,5,6,9 Although this concept is still in its infancy, the potential to significantly reduce the number of animals in a study (estimated reduction is up to 25%) is generally considered an important initiative fitting within the area of the three R’s (Reduction, Refinement, Replacement). 8 To enable the development of this concept and finally its implementation, a large set of data is needed, including pathology data. Yet, virtual control groups should not be confused with HCD, as virtual control data are curated and managed in a different, far more sophisticated, manner.
Artificial Intelligence
Pathology data are also required to train deep learning networks for automated image analysis based on AI. There are many AI approaches under development, from mere differentiation between “normal” and “abnormal” images to automated quantification of specific cell populations or tissue compartments (eg, ovarian follicle counts, spermatogenic staging, or quantitative evaluation of hepatocellular cell hypertrophy). The goal of these AI approaches is to enhance the consistency and accuracy of studies by reducing the interobserver variability and to enhance the efficiency of the pathology evaluation. To train these AI models, a large data set of annotated WSI data is needed.
Using Archived Materials
In safety studies, the archiving of data is required by the principles of GLP. In most companies, archiving occurs just before, or during, finalization of the study, and material will be archived as defined by GLP regulations. Archiving periods differ between specimens but also between countries. The circumstances under which archived material may be removed from the archive are not specifically addressed by GLP regulations, but it is required that such a process is regulated by policies or standard operating procedures within an organization. In some organizations (mostly CROs), the retrieval of archived material is typically limited to audits from regulating authorities or sponsors. Nevertheless, exceptions can be made if GLP rules are followed. This typically means that any retrieval from an archive should be approved and documented. Such documentation generally includes the name of the person retrieving the material, the study director, essential information of the material to be retrieved, the reason(s) for retrieval, and the duration of retrieval approval. Since archiving procedures in GLP organizations are mainly dictated by GLP regulations, the procedures are not very different between CROs and non-CROs.
Property of Data and Legal Considerations
Many nonclinical and environmental safety studies, in which toxicologic pathologists generate data, are conducted at CROs. The relationship between a CRO and a sponsor is governed by a contract such as an MSA. Although MSAs can be sponsor-specific, in some CROs, most of these MSAs do not currently contain any specific wording about the sharing of images or data for educational or scientific purposes. In such cases, MSAs often contain a paragraph on “samples” which means biological samples associated with the service. The MSAs regulate the retention, return, or disposal of such samples, but typically do not regulate their use for any purpose outside of the actual study. Authorization to use such samples would need to be given by the sponsor. Without such authorization, liability concerns would arise that relate to copyright and intellectual property protection. Even the sharing of an image without metadata (ie, a photograph of a microscopic observation) could raise concerns and should be authorized by some kind of contract. In contrast, other CROs do include wording in their MSAs with the sponsor that allow the use of control material (eg, for building an HCD base) and material for internal education and/or the development/advancement of (new) techniques, but with the condition that the material cannot be traced back to the study. As already mentioned, the content of MSAs can be sponsor-specific and a general sentence covering the use of study material (except that of controls) outside the study without permission might be a scenario used or requested by some sponsors. In addition, the MSA can be left intentionally vague allowing more specific wording in the study-specific protocol, adding more flexibility.
We propose that certain scenarios that require the sharing of images or data outside the purpose of the study are described, and that contractual wording is found that supports the flexible use of this material under such scenarios.
Despite the lack of specific contractual wording, it is already common practice for pathology data from vehicle control groups to be used for generating HCD. These databases are mostly composed of control data pooled from different studies and, in the case of CROs, from different sponsors. The use of such data can be supported by a statement in the study protocol, mentioning that material from control animals may be used for purposes other than the study in question or can be included in the MSA as already mentioned. Since most sponsors rely on having up-to-date HCD for a wide range of parameters (not just microscopic findings), it appears to be tacitly understood within the industry that these data are used and have never been a cause of concern. A logical reason is that the HCD are anonymized, and therefore tracing to study numbers or sponsors is not possible.
This highlights the differences among companies in procedures and policies between the sharing of HCD and the sharing of data from control material (eg, images of pathology findings) from a particular study if the data are anonymized.
Concerning images of pathology findings, a few companies have indicated they support the use of images by including specific text in the protocol or MSA indicating that any images may be used for illustrative and/or educational purposes and that these will not be used to generate new data or interpretation, and they will not be included in the report or archived.
Scenarios Regarding the Use of Pathology Data for Educational or Scientific Purpose
As already stated, the policies within the different industries and CROs vary considerably. Some industries stimulate the sharing of pathology data (sometimes in cooperation with CROs) for educational and scientific purposes, whereas, in other companies, the pathology group is more or less treated as an external CRO with strict rules governing the use of pathology data for external use. However, in most cases, requests to share pathology data for external purposes are often declined. The potential reasons for a negative decision are manyfold, including the fear of providing a strategic advantage to competitors, uncertainty about liability, lack of clarity regarding what is specifically shared, the time and effort required to follow internal procedures, the interpretation of regulations (GLP) may be different between different companies and countries, or companies prefer to publish their own data.
While those reasons are understandable, most block scientific development and professional educational training. It seems that clear contractual wording on data sharing, and a better understanding what data is shared, with whom, and for what purpose, would help mitigate many of the risks that currently prevent the sharing of such data. In general, the working group estimated that there would be no risk for companies/sponsors in using control material for educational or scientific purposes when the material cannot be traced back to the study and company/sponsor. Furthermore, pathology data from treated animals are also considered to be of no risk if the data are appropriately anonymized, depending on the class of compound. However, in cases in which a sponsor/company is working on a first-in-class approach, inducing unusual, and so far unpublished morphologic lesions, a clear risk would be associated even with anonymized pictures as the origin of the compound and/or class may potentially be deduced. It must be highlighted that for educational purposes, published or well-known morphologic lesions associated with specific compound classes are of interest, and that sharing images of lesions in treated animals is considered to pose no risk for companies/sponsors if appropriately anonymized. We have collected some examples, including figures (Figures 6-10; further referenced in Table 1) for nonpathologist to visualize what can be shared. Sharing such pathology data would considerably benefit the scientific discipline of toxicologic pathology as well as the training and education of toxicologic pathologists.
Example scenarios of sharing anonymized pathology data and potential risk.

No = No risk of identifying test article and/or company/sponsor (except easily identifiable features of certain compound classes).
Low = Magnitude is depending on what is shared can be mitigated by reducing the amount of data shared or generalizing it further (such as the type or class of the test article). With a negligible risk if the test article is part of a well-known class and there are more similar test articles (from different companies) and a slightly higher risk if the test article is first/only in class or with only few other test articles or few companies producing that class of test articles or if the test article produces very specific observations.
Only for Historical Control Data.
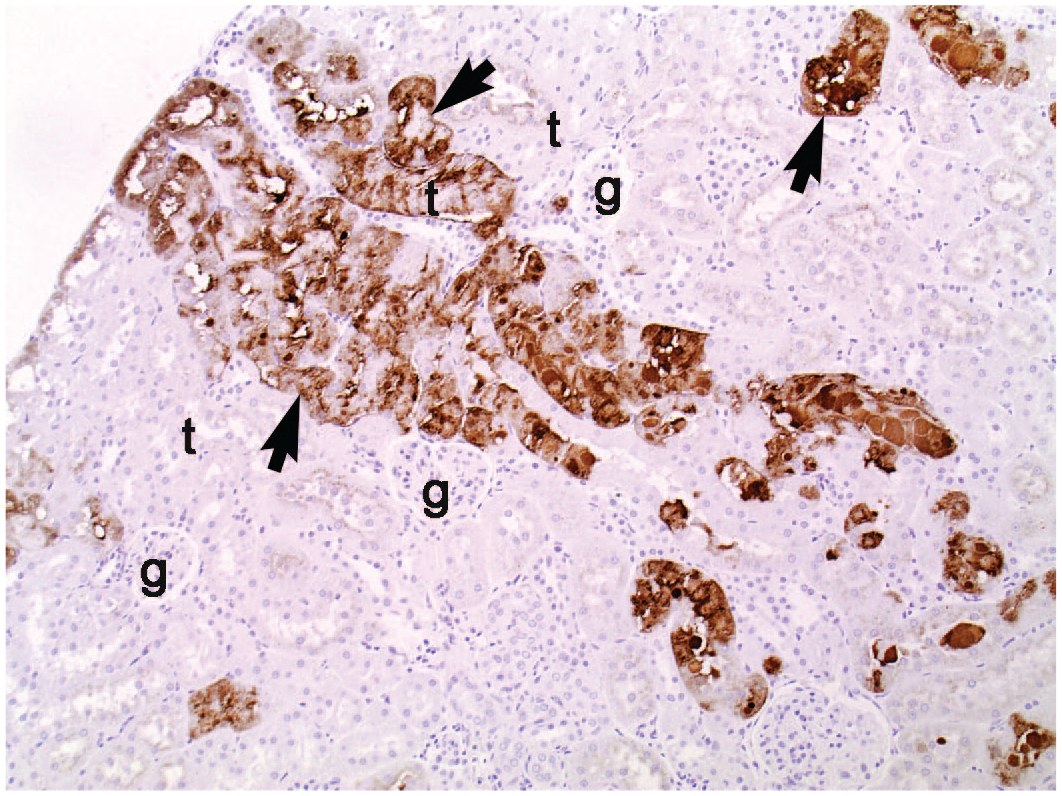
Positive α-2u globulin immunostaining of hyaline droplets (arrows) in the proximal tubules (t) of a kidney from an untreated male Wistar rat. g, glomeruli. Original magnification 10×.
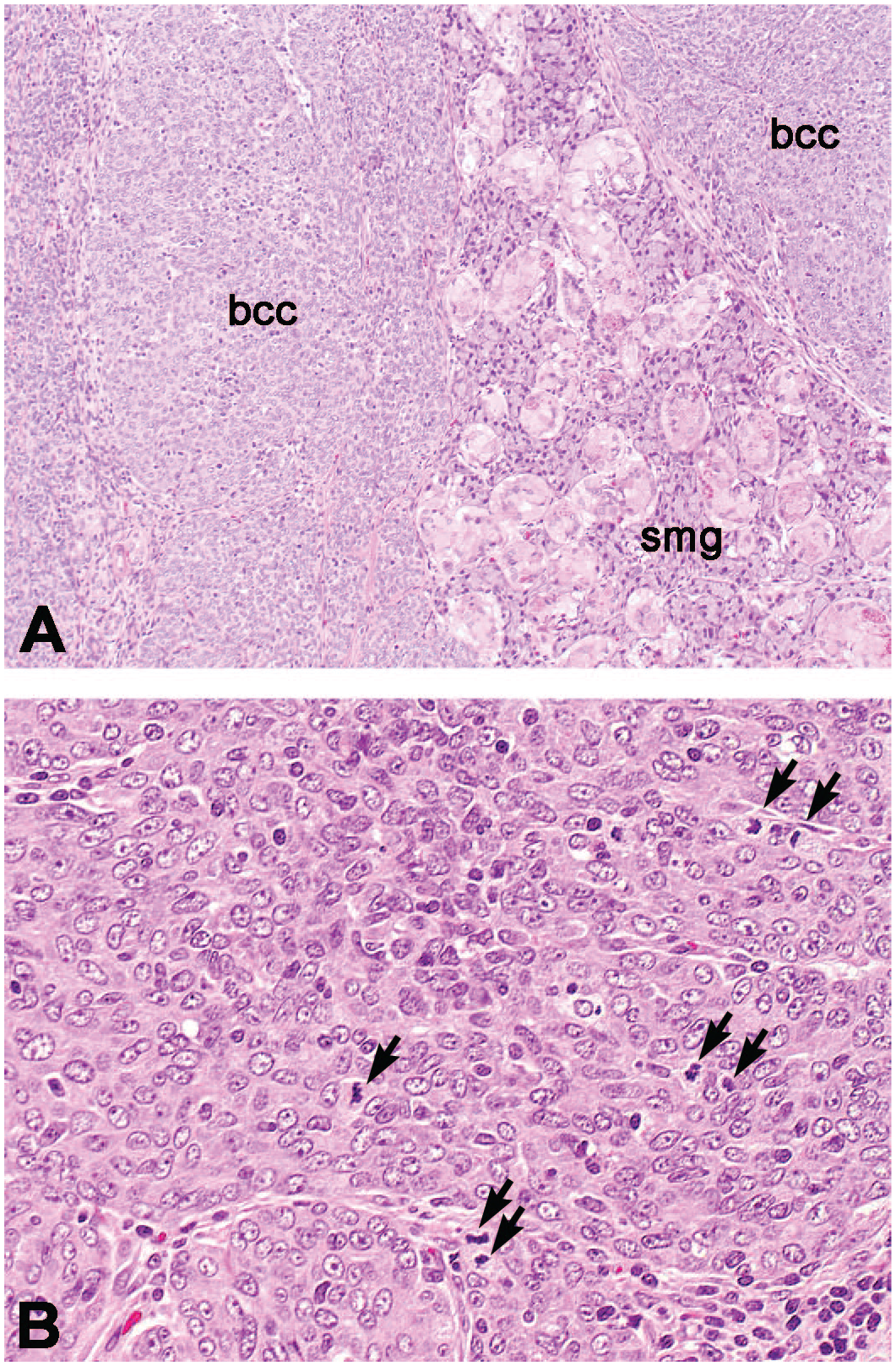
(A) Male rat (age 20 weeks) with a tan and hard mass of 15 × 12 × 10 mm in the submandibular salivary gland (smg) which was microscopically diagnosed as a basal cell carcinoma (bcc) and considered spontaneous in nature. (B) The tumor shows many mitotic figures (arrows). HE staining. Original magnification 10× (A) and 20× (B).
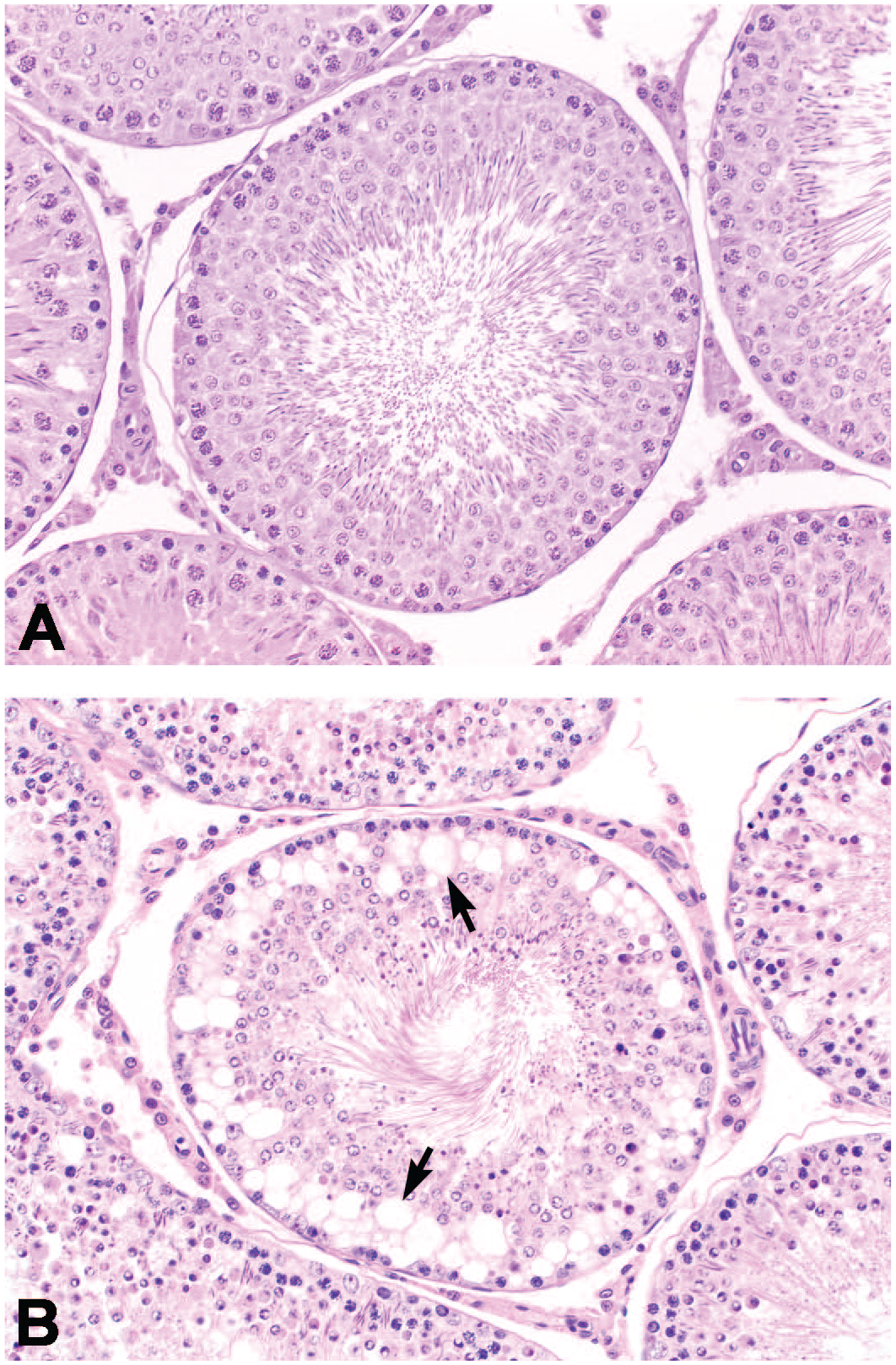
Testis of a control Wistar rat (A) and a Wistar rat treated for 3 days with a testicular toxicant (B) showing marked vacuolation of germ/Sertoli cells located at the base of the seminiferous tubule (arrows). HE staining, Original magnification 20×.
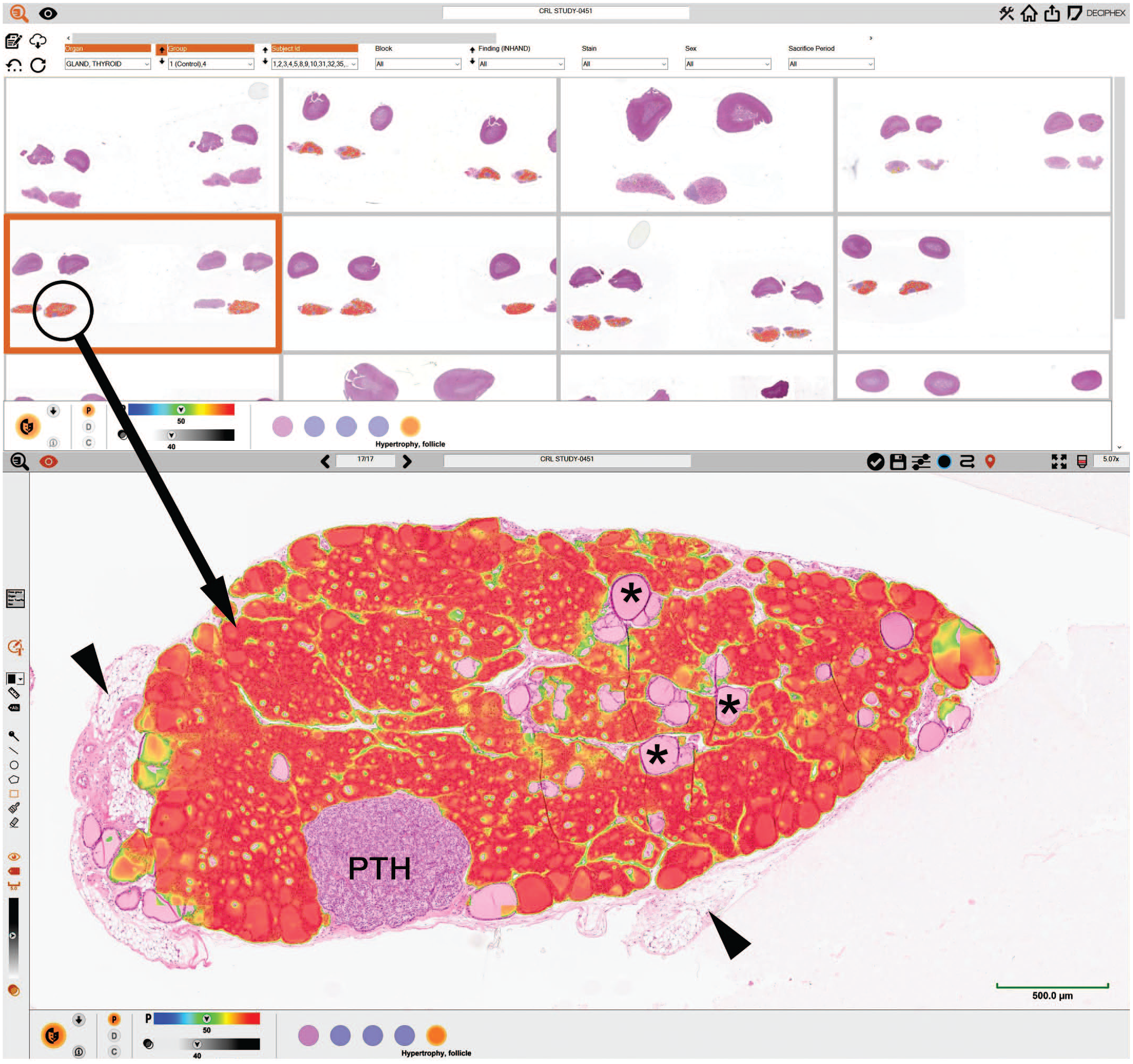
Example of the ongoing development of automatic detection of follicular cell hypertrophy in the thyroid gland, using artificial intelligence tools. In these images, a lesion-specific probability mask is applied over the whole slide image which highlights potential areas of follicular cell hypertrophy (red/orange color). In the high magnification of one thyroid gland (circled in the upper figure), the segmentation of the hypertrophic follicular glands is shown in red/orange, whereas the normal follicles (asterisks), parathyroid gland (PTH), and adjacent adipose tissue (arrow heads) are not selected. The overlay indicates the follicular hypertrophy detection probability (green/blue: low probability; orange/red: high probability). Digital pathology workflow using Patholytix from Deciphex (Deciphex Ltd, Ireland) and integrated AI-based decision support tool Foresight, co-developed in partnership with Charles River (Charles River Laboratories International, Inc, Wilmington, Massachusetts).
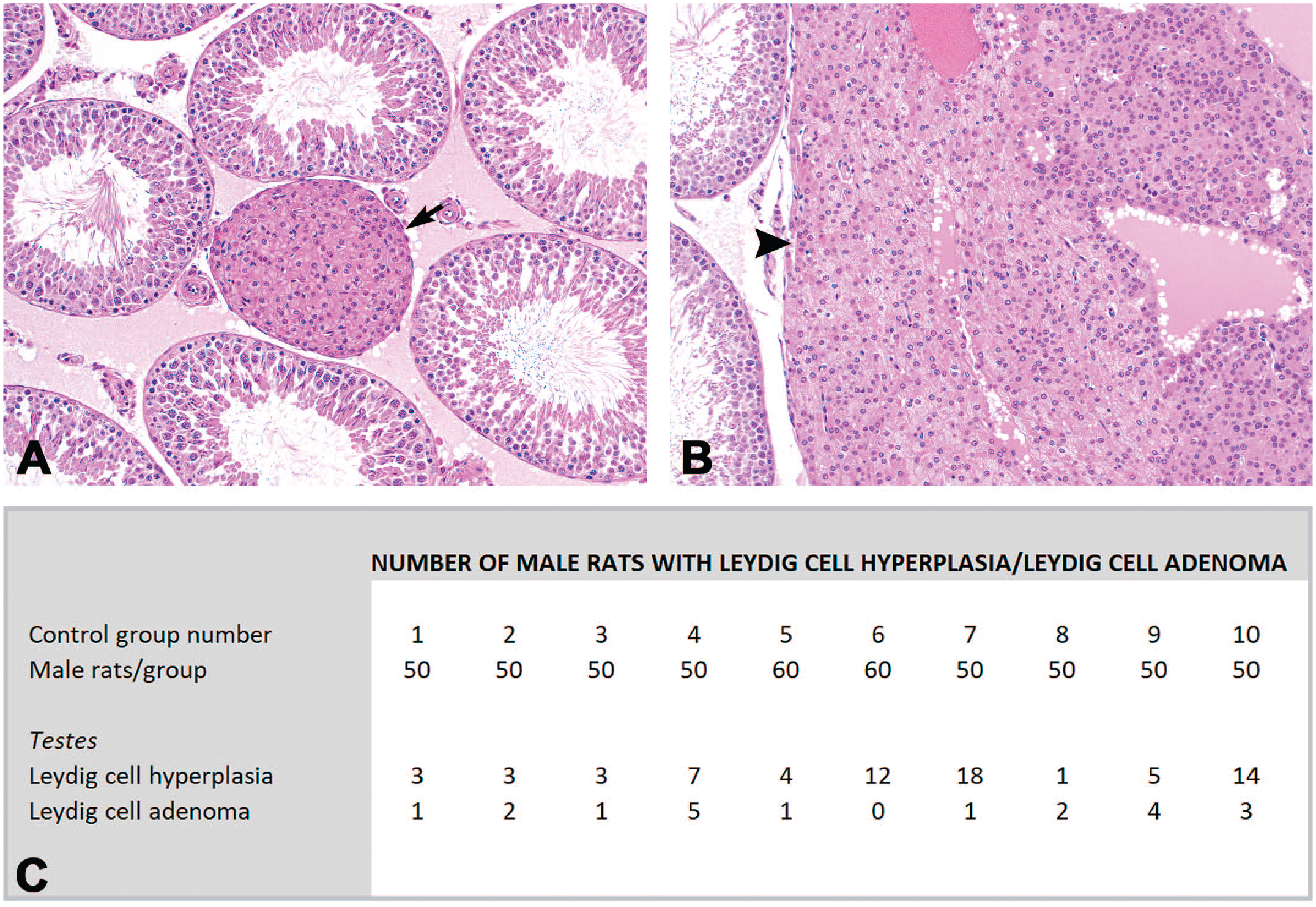
Incidence of Leydig cell hyperplasias (A, arrow) and Leydig cell adenomas (B, arrow head) in Wistar rats. Incidences (given in C), are part from a historical control data set which is based on 10 control groups (from 7 carcinogenicity studies) performed at Charles River Laboratories, ‘s-Hertogenbosch (formerly NOTOX B.V. and WIL Research) in the period between 1989 and 2011.
Concluding Remarks
In the present article, the authors aimed:
To demonstrate the importance of sharing pathology data for education and defined scientific purposes in the field of toxicologic pathology.
To provide arguments for the mutual benefits for industries/institutes and toxicology pathology societies resulting from pathology data sharing.
To provide information on the variation in policies within the different companies and institutes. While some companies permit data sharing, a number of them are hesitant or decline requests to share pathology data for external purposes.
To share scenarios and examples of pathology data sharing that, to the opinion of the authors, will be of low or no risk for the industry/institute. These scenarios mainly involve the anonymization of the data. It was advocated that the risk for control data would be absent, and for test article-related data, the risk would generally be low, with rare examples of tangible risk for the sponsor being dependent upon the level of anonymization.
Although the authors’ representation, together with the survey responders, covered a variety of industrial sectors and publicly funded research institutes, this article does not claim to give a complete overview of the common practices of pathology data sharing. The intention of the article was to create awareness of the importance of sharing pathology data and to identify possibilities without risk (or with low risk), which assist in advancing future science and education in toxicologic pathology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
This opinion article represents the views of the authors and has been approved by the ESTP Executive Council. It does not constitute an official position of the European Society of Toxicologic Pathology, the Society of Toxicologic Pathology, or British Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective agencies and organizations.
