Abstract
The choroid plexus (CP) produces cerebrospinal fluid and has epithelial, interstitial, and vascular compartments. The CP is a potential site of toxicity, and recognizing the normal microanatomy in different animal models is important for the pathologist. In preclinical studies with beagle dog and New Zealand white rabbits, we observed variable numbers of adipocytes in the CP interstitium of control and xenobiotic-treated animals. The adipocytes were unilocular and consistent morphologically with white adipose tissue. There was a striking variability in the number of adipocytes; however, the presence of adipocytes was not associated with other microscopic findings that would suggest a pathologic process. The morphology of adipocytes was reminiscent of what is observed normally in the interstitium of other tissues like skeletal muscle, bone marrow, and the subcutis. Therefore, we propose that the interstitial adipocytes not be recorded as a finding in preclinical studies unless the adipocytes are altered spontaneously (ie, lipoma) or after xenobiotic treatment.
The beagle dog is a common species used in general toxicology studies and New Zealand white (NZW) rabbits are frequently the animal model of choice in vaccination and reproductive toxicology studies for the safety assessment of xenobiotics. While the anatomy, physiology, and histology are well studied in these species, heightened attention to a specific potential target organ or tissue may reveal normal variation or background findings for which awareness is helpful in assessing a potential xenobiotic-related effect. In this communication, we describe interstitial adipocytes in the choroid plexus (CP) of both the beagle dog and NZW rabbit.
The CP is responsible for cerebrospinal fluid (CSF) production in the central nervous system (CNS) and is a critical component of the blood-CSF barrier. 1,2 In the human, the CP contains over 100 million epithelial cells and has a total surface area of approximately 213 cm2. 3 The CP consists of epithelial, interstitial, and vascular zones, which as a unit support fetal brain development, modulate the hormonal-CNS axis, and regulate xenobiotic movement and immune cell trafficking between the blood and CSF. 1 The microanatomy of the CP in preclinical species and man is similar; however, to our knowledge, a systematic cross-species review has not been published and is beyond the scope of this brief communication. 3,4 Species differences have been reported. For example, fat droplets have been described as normal intracytoplasmic features of the rabbit CP epithelium but were considered abnormal in the cat CP epithelium. 3
In the CP interstitium, collagen fibrils, macrophages, fibroblasts, leukocytes, melanophores, mast cells, adipocytes, and nerve fibers have been described in various species. 3,5 Any of these cell populations could be a target of xenobiotic toxicity. Adipocytes in the rabbit CP interstitium were mentioned briefly by Barthold et al, but to our knowledge, adipocytes within the CP interstitium of dogs have not been described. 5 PEGylated xenobiotics, methylcellulose, and xenobiotics that induce phospholipidosis have produced changes in the CP interstitium. 1 PEGylated xenobiotic-related findings in the rat, dog, and primate have been observed in both the CP epithelial and interstitial compartments. In the CP interstitium, affected cell populations include macrophages and endothelial cells which have increased cytoplasm that contains clear vacuoles. 1,6 –8 Vacuolation can also be observed in the CP interstitium (macrophages) after treatment with phospholipidosis-inducing xenobiotics and has been reported in rat, dog, and monkey. 9,10 There are no published reports of xenobiotic-related lesions in the rabbit CP; however, this may simply be a reflection of the infrequent use of the rabbit for general toxicology studies.
A standard subchronic toxicity study in beagle dogs and reproductive toxicity study in NZW rabbits were conducted in 2018 at the CRL-Ashland site. The Institutional Animal Care and Use Committee of Charles River Laboratories approved all experimental protocols. For the rabbit study, male and female NZW rabbits, 21 days of age at initiation of dosing, were administered vehicle or test article via subcutaneous or intravenous injection once every 5 days from Post Natal Day (PND) 21 through PND 96. The dose volume was 2 mL/kg for all groups. Complete postmortem examinations were performed on all animals found dead, euthanized in extremis, or at the scheduled necropsy. Animals were euthanized by an intravenous injection of sodium pentobarbital. At the time of necropsy, brain was collected and placed in 10% neutral-buffered formalin fixative. Trimming was done as follows: Section 1 (forebrain)—From the right hemisphere, a transverse section (3- to 5-mm thick) was obtained perpendicular to the ventral surface through the optic chiasm; Section 2 (midbrain)—From the right hemisphere, a transverse section (3- to 5-mm thick) perpendicular to the brain stem at the caudal end of the interthalamic adhesion; Section 3 (hindbrain)—From the right hemisphere, a transverse section (3- to 5-mm thick) perpendicular to the brain stem through the middle of the cerebellum and medulla; and Section 4 (medulla)—A transverse section in the middle of the medulla. All sections were embedded rostral face down. Microscopic examination was performed on routinely prepared hematoxylin-eosin-stained paraffin sections of brain from all animals euthanized at the scheduled necropsy.
For the beagle dog study, male and female dogs, at least 5 months of age at initiation of dosing, were administered vehicle or test article via oral gavage daily for 28 consecutive days. The dose volume was 5 mL/kg for all groups. Animals were euthanized by an intravenous injection of sodium pentobarbital and exsanguinated. At the time of necropsy, brain was collected and placed in 10% neutral-buffered formalin fixative. Sectioning was performed per recommendations by Bolon et al for nonrodent brains. 11 Microscopic examination was performed on routinely prepared hematoxylin-eosin-stained paraffin sections of brain from all animals euthanized at the scheduled necropsies.
In both studies, controls and treated animals had variable amounts of CP that were visible in sections. Lateral ventricular CP and fourth ventricular CP were most commonly present in the beagle dog and NZW rabbit sections evaluated. When adipocytes were noted in the CP, adipocytes were usually few in number and occurred in both ventricles. However, in some animals, adipocytes were more frequent (Figure 1A–D). The NZW rabbit with the largest number of adipocytes (Figure 1A–B) had the highest terminal body weight (3622 g) in the study by approximately 500 g (study range = 2793-3622 g), which suggests that higher body weights may be associated with interstitial CP adipocytes in the rabbit. In the beagle dog study, the dog with the highest number of interstitial CP adipocytes (Figure 1B and C) had a terminal body weight of 7.1 kg. The body weight range for the study was 5.8 to 8.8 kg and the incidence of interstitial CP adipocytes was low (2/24). Therefore, the influence of body weight on the incidence of interstitial CP adipocytes in the beagle dog study was unclear.
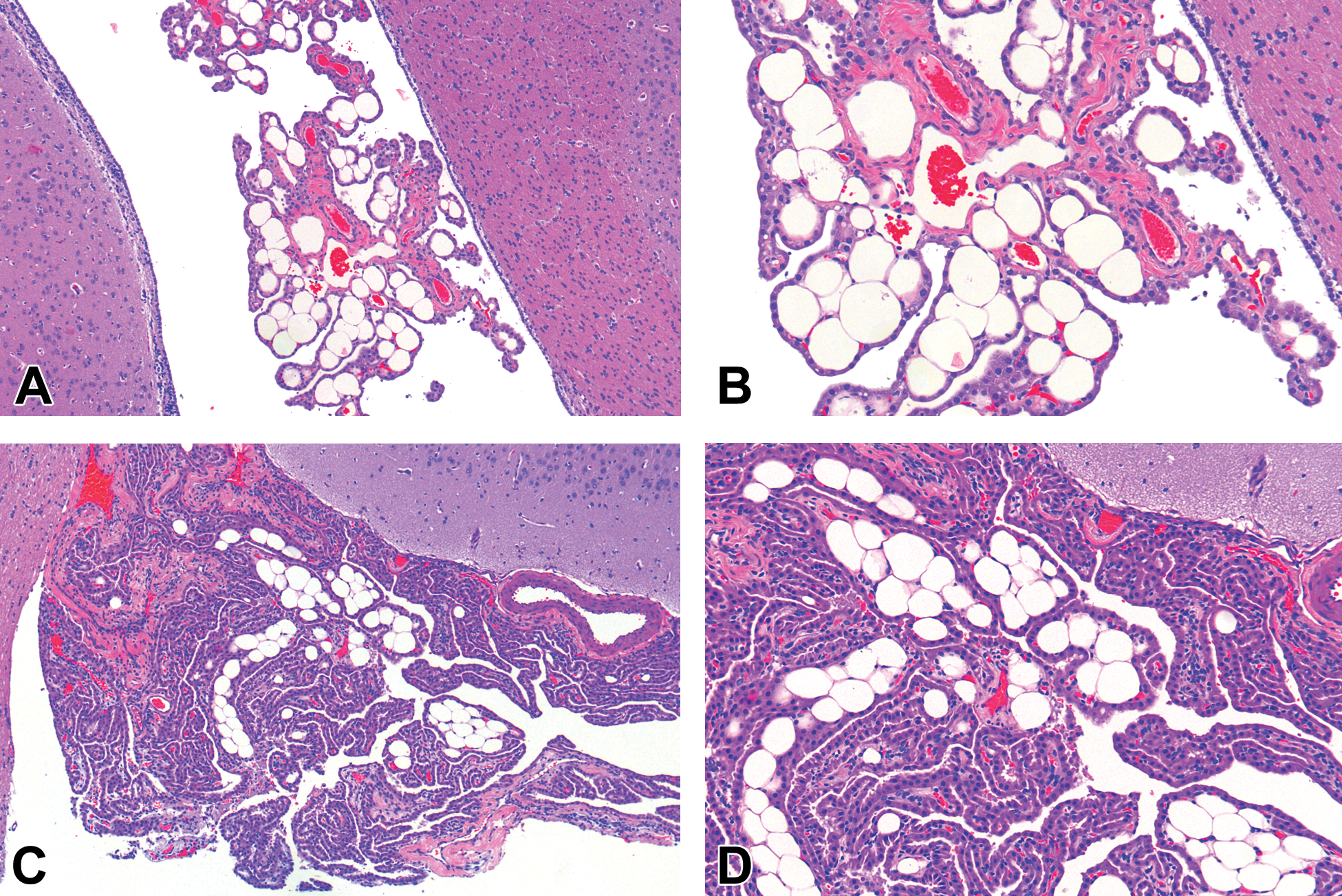
New Zealand white (NZW) rabbit: Numerous adipocytes within the lateral ventricle choroid plexus (CP) interstitium (A, B). Beagle Dog: Interstitial adipocytes within the lateral ventricular CP interstitium (C, D).
In all cases, adipocytes were unilocular and consistent morphologically with white adipose tissue. There was variability in the number of adipocytes between animals and in different choroid plexi; however, the presence of adipocytes was never associated with other microscopic findings that would suggest a pathologic process. The morphology of adipocytes was reminiscent of what is observed normally in the interstitium of other tissues like skeletal muscle, bone marrow, and skin subcutis. In these NZW rabbit and beagle dog studies, the incidence of CP interstitial adipocytes was 8.3% (dog: 2/24) and 50% (rabbit: 6/12). The CRL-Ashland historical reference ranges for NZW rabbits and beagle dogs do not have this observation recorded, most likely because pathologists have considered it normal microanatomy (beagle dog), or in the case of the NZW rabbit, the number of animals in the historical control database for brain was low (26 accessions).
Documenting the normal microanatomy in support of establishing terminology for abnormalities in the rodent and nonrodent model is a critical exercise for the pathologist. To our knowledge, interstitial CP adipocytes are not observed in the rat and mouse as an outcome of xenobiotic treatment and they are not described in the published INHAND guide. 9 INHAND working groups are presently establishing terminology for nonrodents with the purpose of extending the rodent nomenclature in support of differences observed between rodent and nonrodent models. While a few adipocytes observed in the CP could be easily overlooked or discounted in a beagle dog or NZW rabbit study, the larger number of adipocytes observed in a few animals, especially if found solely in the high-dose group, could mislead the pathologist’s interpretation of a potential test article–related effect in the CP. For the CP, interstitial adipocytes seem to vary considerably and, based on the morphologic characteristics of the finding, should be considered normal microanatomy and not recorded in the microscopic raw data. That said, if the interstitial adipocytes are morphologically altered based on concurrent controls, especially in a dose-responsive manner, the pathologist should consider reporting the alteration (increased cellularity/size) in the microscopic raw data.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
