Abstract
Cardiac exposure to ionizing radiation can damage both the microvasculature and coronary arteries, as well as increase the long-term risk of heart disease, myocardial fibrosis, and conduction abnormalities. Therapeutic agents capable of promoting recovery from radiation injury to the heart are limited. Growth hormone is linked to improved cardiac function following injury. Here, we leveraged a cynomolgus macaque model to determine the long-term outcomes of recombinant human growth hormone (rhGH) therapy on the heart following low-dose ionizing radiation. Macaques were exposed to 2 Gy radiation, treated with rhGH for one month, and assessed after 2 years. Overall, plasma lipid profile, cardiac function, and coronary artery disease were similar between rhGH and placebo treated animals. However, a subgroup of rhGH-treated animals exhibited more extensive atherosclerotic plaques in the coronary arteries. Together, these findings indicate that transient human growth hormone therapy subsequent to a single low dose of ionizing radiation involving the heart does not result in long-term changes to plasma cholesterol but may promote exacerbated coronary artery disease in a subset of individuals.
Introduction
Therapeutic radiation therapy of the thorax in which the heart and vascular structures are included in the radiation field increases the risk of detrimental cardiovascular outcomes, often manifesting years after exposure.10,22 Cardiac exposure to ionizing radiation damages the vascular endothelium and increases the risk of heart disease, myocardial fibrosis, heart failure, and conduction disease.4,36 Direct damage to the heart and vessels is further compounded by radiation-induced changes to plasma lipids, an important atherogenic risk factor. For example, radiotherapy has been linked to elevated plasma triglycerides (TG). 27 Modern radiotherapy protocols have been successful in reducing radiation exposure to nontarget tissues; however, even very low exposure to irradiation is associated with increased inflammation and enhanced collagen deposition, 24 and therapeutics capable of promoting recovery from radiation injury remain limited.
Growth hormone (GH), best known for its namesake role in promoting childhood growth, is a multifunctional protein hormone with roles in lipolysis, electrolyte balance, immune function, muscle homeostasis, and metabolism.5,29 In particular, its roles in immune function and lipid metabolism render growth hormone an intriguing cardioprotective candidate, given the significance of plasma lipids as a risk factor for cardiovascular disease. Specifically, lipid based atherogenic and cardiovascular risk factors include elevated TG and low-density lipoprotein cholesterol (LDLC), and reduced high-density lipoprotein cholesterol (HDLC). 2 Growth hormone deficiency is associated with increased total cholesterol (TC) and LDLC, increased TG, and reduced HDLC, 15 while GH therapy rescued HDLC and reduced TC, TG, LDLC in obese rats fed a high fat diet. 17 Likewise, obese individuals treated with a 6-month course of recombinant human GH (rhGH) experienced reduced LDLC and TC, and increased HDLC.32,39 In addition to its role in lipid metabolism, rhGH therapy was associated with improved cardiac function and recovery subsequent to experimental myocardial infarction, including improved left ventricular ejection fraction, and reduced infarct scar.3,19
Several studies have provided further evidence for the therapeutic potential of growth hormone in ameliorating radiation-induced injury. Growth hormone therapy following a dose of 7.5 Gy radiation in mice improved survival over 35 days and stimulated hematopoiesis and leukocyte production, which may promote healing. 7 Similarly, rhGH treatment promoted recovery of leukocytes following sublethal 2 Gy irradiation in the same nonhuman primate (NHP) subjects reported here. 7 However, studies of the impact of rhGH therapy on the heart following sublethal radiation are lacking. Here, we investigated the long-term outcomes of one month of rhGH treatment in macaques subjected to sublethal radiation. Specifically, we examined plasma lipids, cardiac function, myocardial collagen deposition, and coronary artery atherosclerotic extent and severity.
Methods
Study Subjects
For irradiation studies, n = 20 adult male cynomolgus macaques (Macaca fasicularis) were purchased from the Institut Pertanian Bogor (Bogor, Indonesia) and socially housed in indoor pens at Wake Forest University Primate Center. All animals were in good physical health and free of antibodies against simian retroviruses. Animals were maintained on standard Monkey Chow (Purina). Beginning 13-20 months prior to irradiation, animals were fed 120 calories/kg body weight per day of an atherogenic diet containing 0.2 mg of cholesterol per calorie (Purina). This diet was designed to approximate the cholesterol (188 mg/day) and saturated fat (40% of calories) content consumed by the average North American person. Study subjects remained on this diet for an average of 53 months until the conclusion of the study. Two animals were excluded from the study: one died unexpectedly after irradiation due to encephalitis, and one developed diabetes mellitus.
Nonirradiated control cases were obtained from the Wake Forest University Cardiovascular Pathology Archives to assess myocardial fibrosis. Nonirradiated control cases for echocardiographic data (n = 24) were obtained from animals on another study and were matched for body weight. Nonirradiated control cases for plasma lipids were matched for the duration of dietary cholesterol exposure calculated as cholesterol days (Table 1). For each diet, the number of days on diet was multiplied by the amount of cholesterol in that diet. The results from each diet were summed to obtain the total cholesterol days.
Study population.
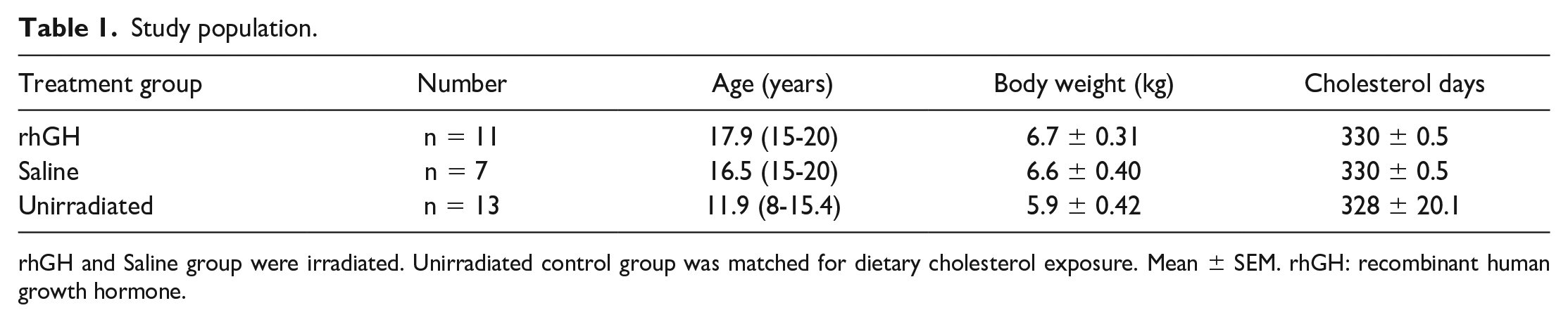
rhGH and Saline group were irradiated. Unirradiated control group was matched for dietary cholesterol exposure. Mean ± SEM. rhGH: recombinant human growth hormone.
All animals were housed and studied at Wake Forest University under approved Institutional Care and Use Committee guidelines, under accreditation by the Association for the Assessment and Accreditation of Laboratory Animal Care and conformed to the recommendations of the Guide for the Care and Use of Laboratory Animals.
Irradiation
Procedures for irradiation were previously described. 7 Briefly, a Varian 2100 EX dual energy linear accelerator was used to deliver a single fraction whole body dose of 2 Gy using right and left lateral fields of 6 MV X-rays at a mid-line dose rate of 0.69 Gy/min. This dose is approximately equal to a single fraction of the dose used for localized radiation treatment of cancer and was sufficient to induce a transient hematopoietic depletion. Animals were evaluated twice daily for signs of illness.
Diagnostics
Complete blood counts and serum chemistry were monitored daily for the first 5 days following irradiation, 3 times weekly during the remainder of the treatment period, then monthly for a year as previously described. 7 All animals received prophylactic antibiotic treatment with enrofloxacin (5mg/kg) until clinical pathology parameters returned to the normal range. Supportive fluid therapy, analgesics (ketoprofen), and symptomatic care were provided as needed based on physical exam, clinical signs, and clinical pathology abnormalities. Pulse wave velocity and cardiac ultrasound were performed approximately 2.5 years after irradiation to assess cardiovascular function.
Treatment
rhGH was purchased from Genentech (South San Francisco, CA), reconstituted in saline, and stored at 4°C until use according to the manufacturer’s instructions. Beginning the day of irradiation, animals received 5 µg/kg/day of rhGH (rhGH treatment group, n = 11) or 0.9% saline (Placebo group, n = 7) by subcutaneous injection into the flank once daily for 30 days.
Pathology
At the conclusion of the study, animals were humanely euthanized with an overdose of sodium pentobarbital, and the cardiovascular system was perfused with cold saline. The heart chambers were perfused with 10% NBF and the coronary arteries were dissected. Cardiac tissues and coronary arteries were fixed in 10% neutral buffered formalin, subjected to routine processing, embedded in paraffin, cut into 5-µm thick sections and stained with hematoxylin and eosin.
Morphometric analysis of coronary artery plaque size was obtained for the right coronary artery (RCA), left anterior descending artery (LAD), and left circumflex artery (LCX). Five serial tissue blocks for each artery were cut perpendicular to the long axis of the artery at approximately 5-mm intervals. Arterial sections were stained with Verhoeff’s and van Gieson’s, imaged, and the cross-sectional area of the plaque lesion measured. Epicardial coronary arteries were evaluated for the extent of atherosclerosis expressed as the mean cross-sectional area of the intima in millimeters squared, and severity utilizing the American Heart Association grading scale (AHA grading, Supplemental Table S1).34,35
Left ventricle cardiac sections were stained with picrosirius red (PSR). Twenty randomly chosen fields per section were analyzed by light microscopy (×400) to determine the average collagen volume fraction. 25 Briefly, PSR-stained interstitial collagen fibers were highlighted and a pixel count obtained for the amount collagen represented in each field. Collagen volume fraction was expressed as a percentage area of interstitial collagen excluding perivascular areas. 20
Statistics
Data analysis was performed using GraphPad Prism 10.1.1 (GraphPad Software, San Diego, CA) software. Shapiro-Wilk and Komogorov-Smirnov tests were used to test for normality. A Student’s t test was used to analyze differences when samples followed a normal distribution, and a Mann-Whitney test was used for pair-wise comparisons for samples that were not normally distributed. All results are expressed as the mean ± SEM. P values less than .05 were considered statistically significant and are denoted as *P < .05; **P < .01; ***P < .001; ****P < .0001.
Results
Lipid Profile is Not Altered by rhGH Treatment in Irradiated Cynomolgus Macaques
Two years following irradiation and rhGH or placebo treatment, the plasma lipids of irradiated and nonirradiated control macaques were measured to identify the long-term impact of rhGH treatment post irradiation. A dose of 2-Gy irradiation was used as it is approximately equal to a single fraction of the dose used for localized radiation treatment of cancer and was sufficient to induce a transient hematopoietic depletion. 7 We observed a statistically significant increase in plasma mean TG levels in cynomolgus macaques (P ≤ .0001) who received a single 2-Gy whole-body dose irradiation compared with nonirradiated controls (Figure 1A). In contrast, we observed a decrease in low-density lipoprotein-very low-density lipoprotein cholesterol (LDL-VLDLC) (P = .0010); total plasma cholesterol (P = .1428) and HDLC (P = .8366) levels were unaltered between the irradiated and unirradiated control animals (Figure 1A).
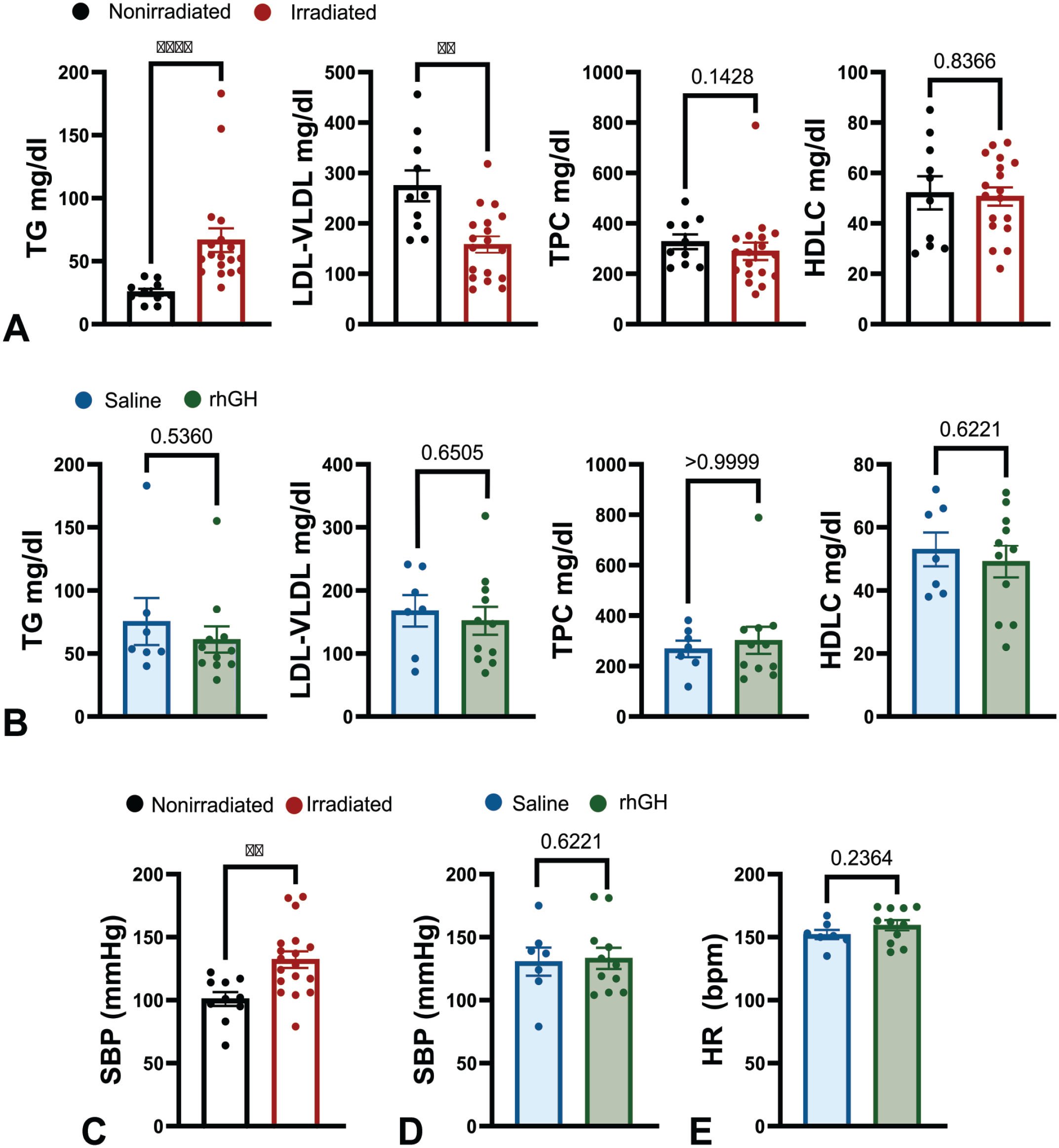
Lipid profile. Each point represents an individual animal and data are expressed as mean ± SEM. TG: triglyceride. LDL-VLDLC: low-density lipoprotein-very low-density lipoprotein cholesterol. TPC: total plasma cholesterol. HDLC: high-density lipoprotein cholesterol. rhGH: human growth hormone. SBP: systolic blood pressure. HR: heart rate.
Growth hormone deficiency has been linked to lipid profile anomalies, including elevated LDLC and triglycerides, and reduced HDLC. 15 Likewise, treatment with rhGH is linked with decreased TC, LDLC, TG and increased HLDC.13,27,39 To investigate if rhGH therapy rescues irradiation-induced elevations in TG and low-density lipoprotein-very low-density lipoprotein cholesterol (LDL-VLDL) plasma lipids, we next compared the lipid profile of irradiated cynomolgus macaques treated with rhGH or saline control. We observed no differences in mean TPC (P > .9999), HDLC (P = .6221), LDL-VLDC (P = .6505), or mean TG (P = .5360) in irradiated animals treated with rhGH or saline (Figure 1B).
We previously found that while blood pressure (BP) is positively correlated with age, irradiation had no effect on BP. 1 To test the hypothesis that growth hormone would not result in long-term negative outcomes on blood pressure, we compared systolic blood pressure of irradiated subjects treated with rhGH or saline, as well as nonirradiated controls. Systolic blood pressure was elevated in irradiated cynomolgus macaques compared with nonirradiated control animals with comparable dietary cholesterol exposure (P = .0035, Figure 1C), however since the mean age of controls used in this study was younger than the irradiated group (Table 1), it is likely that this difference is age related. As expected, we observed no difference in BP (P = .8521) or heart rate (P = .2364) in irradiated animals treated with rhGH or saline (Figure 1D and E).
rhGH Does Not Reduce Left Ventricular Myocardial Fibrosis or Cardiac Function in Irradiated Macaques
Moderate dose irradiation is reported to induce myocardial fibrosis in rhesus macaques fed an atherogenic diet. 11 GHRH agonists, which function to induce GH release, have been shown to reduce fibrosis in a swine model of myocardial infarction. 3 To investigate how rhGH therapy impacts irradiation-induced myocardial fibrosis, we stained sections of the cardiac left ventricle (LV) with PSR, which stains collagen and polarizes for quantification. However, we observed similar collagen fractions in rhGH and saline treated animals (P = .1447, Figure 2A).
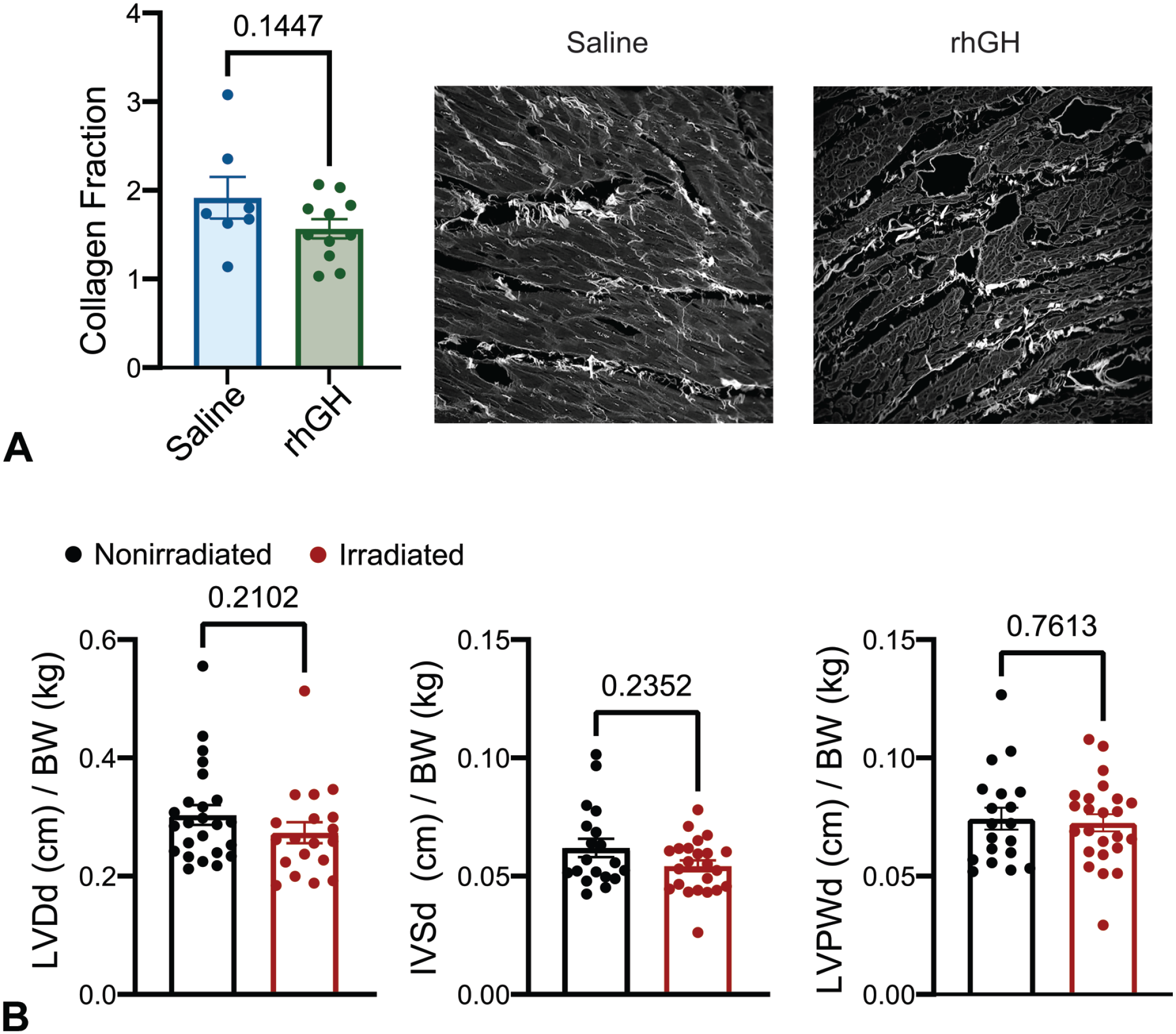
Cardiac structure. (A) Collagen fraction in irradiated macaques treated with saline or rhGH. Each point represents an individual animal and data are expressed as mean ± SEM. Representative confocal photomicrographs of picrosirius red-stained sections of the left ventricle highlighting collagen deposition in white. (B) Echocardiograph structural measurements of nonirradiated and irradiated macaques normalized to body weight (BW). Left ventricular diameter in diastole (LVDd), interventricular septum in diastole (IVSd) and left ventricular posterior wall in diastole (LVPWd).
Because rhGH treatment is associated with improved cardiac function subsequent to myocardial infarction, 19 we postulated that rhGH might also ameliorate the potentially damaging effects of irradiation on cardiac function. To test this, we measured ejection fraction (EF), fractional shortening (FS), left ventricular diameter in diastole (LVDd), interventricular septal thickness in diastole (IVSd), and left ventricular posterior wall thickness in diastole (LVPWd) of nonirradiated or irradiated macaques treated with rhGH or saline. All parameters of cardiac function in irradiated NHPs were similar between rhGH and saline treated animals (Figure 2B, Table 2). We identified a cohort of weight matched unirradiated macaques (n = 24) for comparison but we observed no significant differences between irradiated and nonirradiated controls in EF, FS, and cardiac structural measurements, suggesting that a single dose of 2 Gy does not impair cardiac function in cynomolgus macaques 2 years following irradiation. Taken together, these findings indicate that rhGH does not alter cardiovascular risk factors, including lipid profile or blood pressure, or cardiac function.
Cardiac functional measurements.
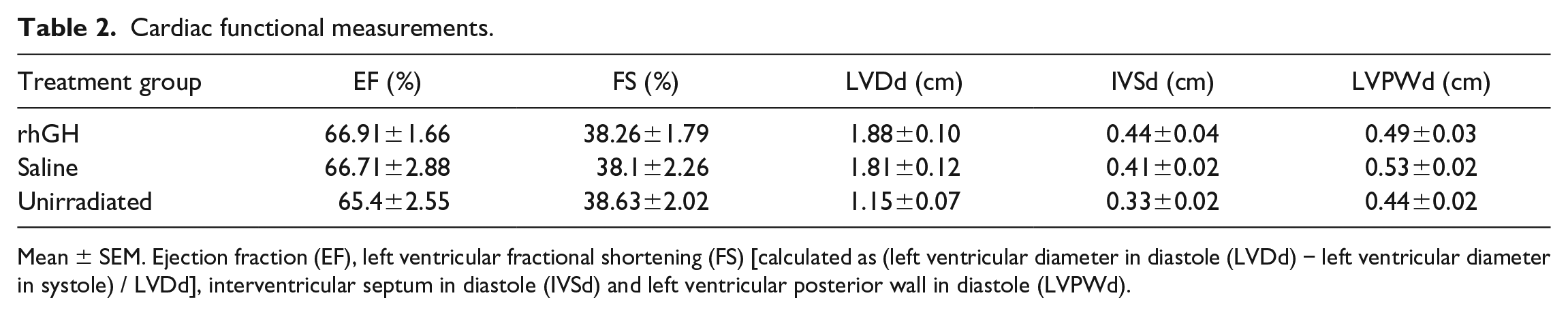
Mean ± SEM. Ejection fraction (EF), left ventricular fractional shortening (FS) [calculated as (left ventricular diameter in diastole (LVDd) − left ventricular diameter in systole) / LVDd], interventricular septum in diastole (IVSd) and left ventricular posterior wall in diastole (LVPWd).
Coronary Artery Atherosclerosis is Similar in Irradiated Macaques Treated With rhGH or Saline
Because irradiation is associated with an increased risk for coronary artery disease, 37 we investigated the therapeutic potential of rhGH in ameliorating coronary artery damage. We first measured the extent of atherosclerotic plaque formation expressed as mean cross-sectional area of the intimal layers of the left anterior descending (LAD), left circumflex (LCX) and right (RCA) coronary arteries. Epicardial coronary artery intimal area (IA) trended higher in animals treated with rhGH but did not reach statistical significance (Figure 3A; LAD P = .2957, LCX P = .3873, RCA P = .3170). We next evaluated the severity of coronary vessel atherosclerosis using the AHA scoring guidelines34,35 and overall found no difference in AHA atherosclerosis lesion scores between rhGH and saline treated animals (Figure 3B; LAD P = .4767, LCX P = .9095, RCA P = .2505).
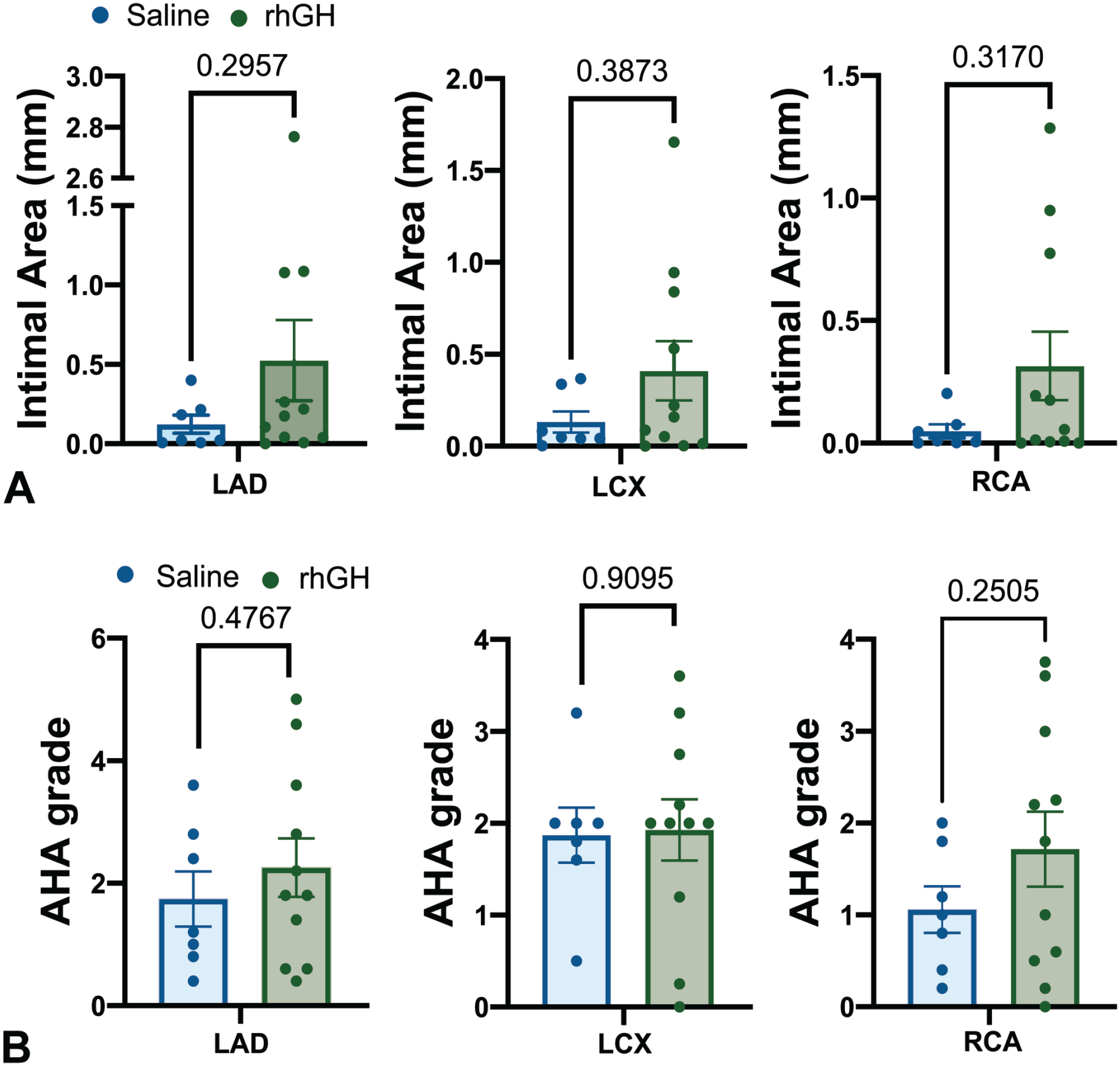
Coronary artery intimal area and AHA atherosclerosis severity grade. Histological sections of the left anterior descending coronary artery (LAD), left circumflex coronary artery (LCX), and right coronary artery (RCA) were evaluated for (A) intimal area or (B) graded for atherosclerotic lesion severity. Each point represents the mean of 5 serial histological sections of each artery and data are expressed as treatment group mean ± SEM.
rhGH Exacerbates Atherosclerosis in a Subset of Irradiated Macaques
Intriguingly, the results of the overall effects of rhGH on coronary artery atherosclerosis extent and severity suggested that animals clustered into one of two groups; those that developed low-grade plaques (n = 6), and those that developed large, complicated plaques (n = 5) (Figure 4A). To further investigate this phenotype, we categorized the rhGH-treated animals as those having high-grade plaques, defined in this study as those having an AHA score of 3 or higher, or low grade, defined as having an AHA score of 0-2. To account for cholesterol as a risk factor, we matched rhGH animals in the high- or low-grade plaque groups with saline treated animals with similar plasma cholesterol levels (Table 3, Figure 4B). Unsurprisingly, higher grade plaques were associated with higher average TPC regardless of treatment (Figure 4B). This appeared to be due to elevated LDL-VLDL since TG and HDLC were similar in animals with small or large plaques (Figure 4C). We next compared the atherosclerotic plaque extent and found that among animals with higher grade atherosclerotic plaques, the intimal area of all three coronary arteries was larger in rhGH-treated animals compared with saline treated animals with comparable terminal TPC values (Figure 4D; LAD P = .0303; LCX P = .0084; RCA P = .0173). Coronary artery IELA trended higher in rhGH-treated animals with high-grade atherosclerotic plaques but did not reach significance (Figure 4E; LAD P = .1257; LCX P = .1263; RCA P = .2222). Taken together, these data suggest that rhGH overall does not impact coronary artery atherosclerosis extent nor severity subsequent to low-dose whole body irradiation, however rhGH therapy may exacerbate coronary artery atherosclerosis in a subset of individuals independent of plasma lipid profile.
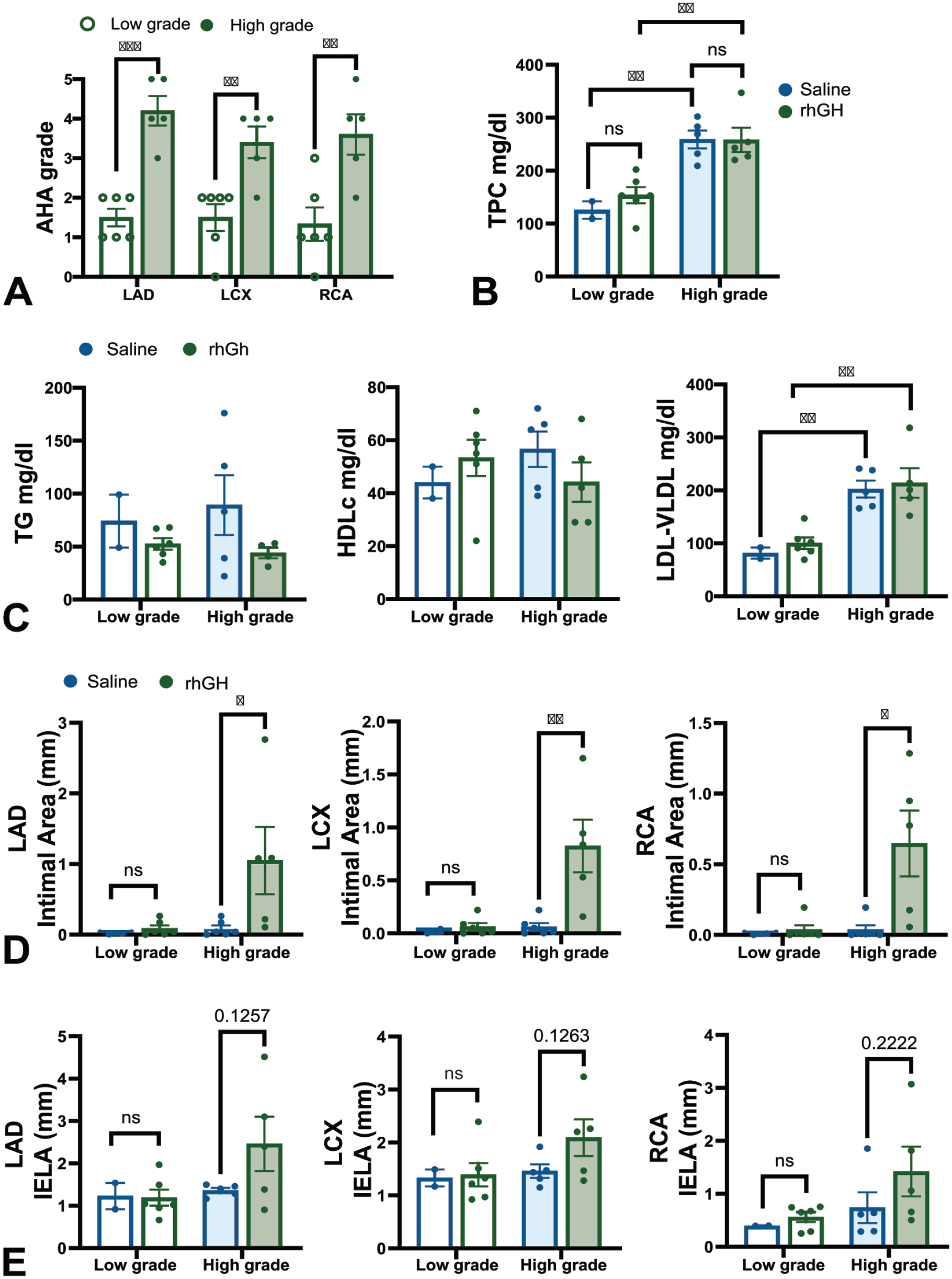
rhGH promotes atherosclerosis in a subset of irradiated individuals. (A) High- and low-grade atherosclerotic plaques in rhGH-treated animals. (B) Total plasma cholesterol (TPC) of irradiated macaques treated with rhGH compared with saline controls with similar TPC levels. (C) Lipid profile of irradiated macaques treated with rhGH compared with saline controls with similar TPC levels. (D) Intimal area and (E) internal elastic lamina area (IELA) of coronary arteries from irradiated macaques treated with rhGH compared with saline controls with similar TPC levels. Each point represents the mean of 5 serial histological sections of each artery for each animal and data are expressed as treatment group mean ± SEM. Abbreviations: Triglyceride (TG); low-density lipoprotein-very low-density lipoprotein (LDL-VLDL); high-density lipoprotein cholesterol (HDLC); left anterior descending coronary artery (LAD), left circumflex coronary artery (LCX); right coronary artery (RCA).
Lipid profile of rhGH-treated high- versus low-grade atherosclerotic plaques.
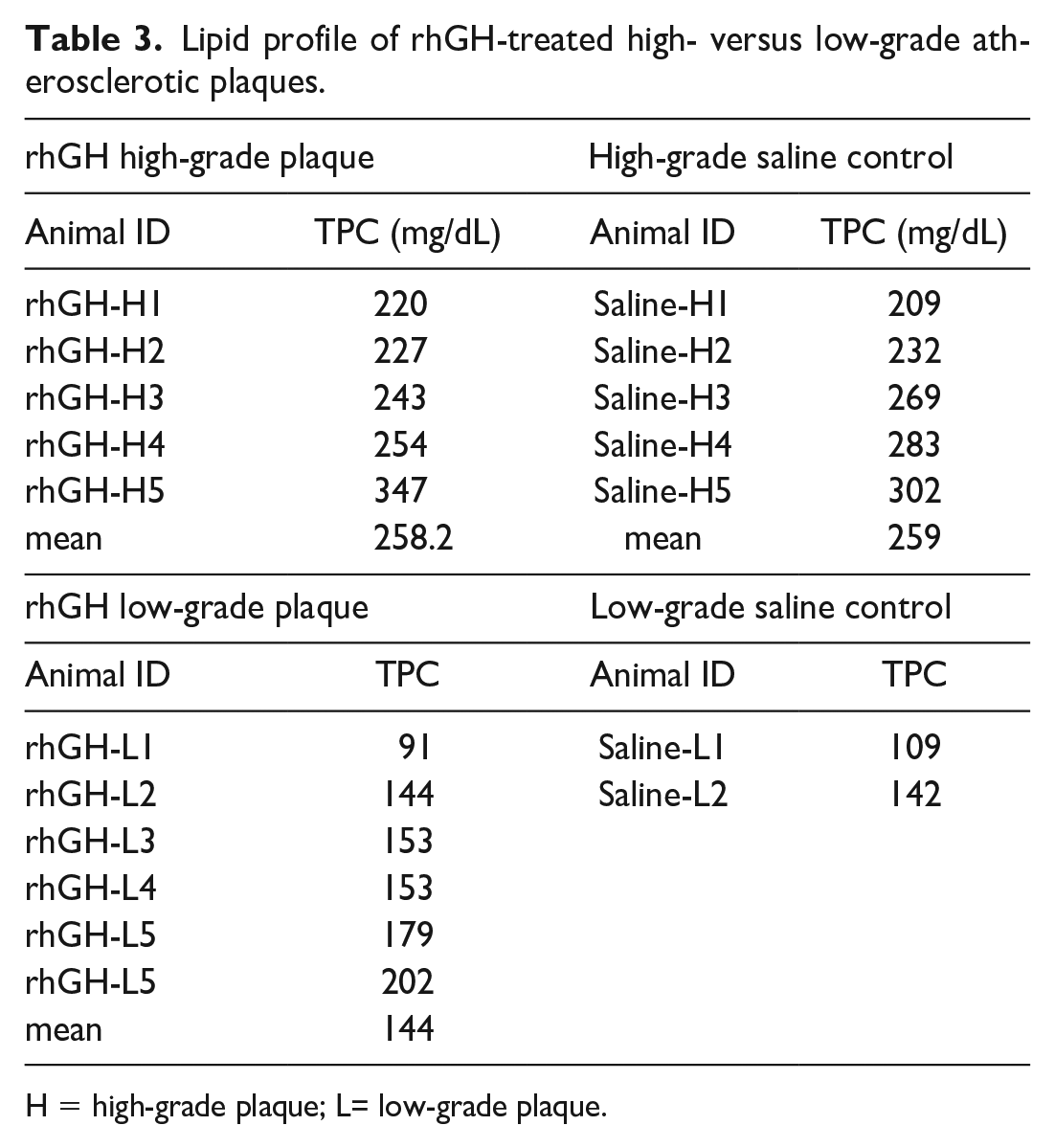
H = high-grade plaque; L= low-grade plaque.
Discussion/Conclusion
We previously reported a dose dependent effect of the long-term impacts of radiation exposure where 6.5 to 8.4 Gy (LD50/ -90) resulted in severe hematopoietic injury and cardiac damage characterized by increased myocardial fibrosis, reduced left ventricular diameter, and elevated circulating markers of cardiac injury,11,12 while 2-Gy irradiation led to hematopoietic insult to white blood cell populations which was partially rescued by treatment with rhGH. 7 Here, we report that post-irradiation rhGH treatment 2 years prior has no impact on cardiac function or myocardial fibrosis but is associated with alterations in atherosclerotic extent in a subset of animals. In addition, consistent with previous reports,11,27 we find altered lipid profiles in irradiated animals compared with nonirradiated controls.
While total cholesterol levels were similar between irradiated and nonirradiated control animals, radiation exposure was associated with different components of atherogenic plasma lipid risk factors. Animals exposed to low-dose radiation had higher plasma levels of TG while nonirradiated animals had higher LDL-VLDL cholesterol, both of which are features of atherogenic dyslipidemia, in addition to low levels of HDLC. 14 Furthermore, altered lipid profiles in irradiated rhesus macaques have been associated with increased incidence of diabetes. 11 Only one animal in our study developed evidence of diabetes and was subsequently excluded from our study.
Growth hormone therapy has shown some protective potential in lipid metabolism. Obese rats treated with GH for 4 weeks had reduced plasma TC, TG, and LDLC, and higher HDLC than nontreated rats. 17 In human studies, obese adolescents treated with rhGH for 6 months had reduced TC and LDLC, and higher HDLC.32,39 However, in our study, we observed no differences in the plasma lipid profile between irradiated animals treated with rhGH or saline.
A key limitation of this study is the age of the cholesterol-matched unirradiated control animals. Because diet plays such an integral role in plasma lipid levels, we prioritized matching dietary cholesterol exposure resulting in only partial age matching, with nonirradiated controls being younger on average than irradiated study subjects. This imperfect age matching precluded interpretation of blood pressure comparisons between irradiated and nonirradiated animals. We recently observed a positive correlation between age and blood pressure, but no association between irradiation and blood pressure. 1 In this study, irradiated macaques had higher systolic blood pressure on average, but were also older than nonirradiated controls, thus it is likely this observation represents an age-related finding rather than a consequence of irradiation. Nonetheless, we observed no differences in rhGH-treated animals compared with saline controls, indicating that rhGH had no long-term impact on blood pressure in irradiated animals.
In this study, we found no differences in cardiac fibrosis or cardiac function between nonirradiated and irradiated animals treated or not with rhGH, indicating a dose of 2 Gy was insufficient to cause increased myocardial collagen deposition. This contrasts with an earlier study in which we observed increased myocardial collagen deposition in macaques receiving at least 6.5-Gy irradiation. 11 A case-control study of breast cancer patients undergoing radiation treatment, with a mean cardiac dose of 2.5 Gy for the group, found that the risk of heart failure increased with cardiac radiation dose at about 7% per Gy. 30 Growth hormone is reported to improve cardiac function following ischemic heart disease, 19 although in another study rhGH therapy did not improve cardiac function in spite of reducing fibrosis. 3 This study used a dose of 2 Gy, which is comparable to a single fraction of the dose used for localized radiation treatment of cancer, and focused on a single time point 2 years after irradiation. It is likely that a higher radiation dose and/or longer post-irradiation time period may have resulted in greater cardiac damage, as observed in our previous study which examined cardiac morphology and function 5.6 to 9.7 years after 6.5-Gy radiation exposure. 11 Human studies report heart failure several years following irradiation. In the survivors of Hiroshima and Nagasaki, most of whom received doses below 0.2 Gy, there was an estimated excess relative risk of 14% per Gy for cardiac mortality. 31
Cardiomyocytes are relatively resistant to the direct effects of radiation, and much of the detrimental effects of radiation therapy are due to damage to the cardiac blood supply, with moderate-to-high doses of irradiation inducing vascular endothelial activation and inflammation of the microvasculature and coronary arteries.4,9 Cardiac exposure to irradiation in hypercholesterolemic ApoE-/- mice resulted in enhanced development of coronary artery atherosclerosis. 13 Atherosclerosis is a chronic inflammatory disease, requiring both inflammation and cholesterol deposition. 23 Here, we found no differences overall in atherosclerotic extent nor severity between irradiated animals treated with rhGH or saline alone. However, we observed clustering of rhGH-treated animals into two subgroups: those having low-grade, uncomplicated atherosclerotic plaques or those having high-grade, complicated atherosclerotic plaques. Those with high-grade plaques had atherogenic lipid profiles, with higher average TPC and LDL-VLDL. Importantly, rhGH-treated animals with high-grade plaques had greater intimal area compared with saline treated animals with similar plasma TPC values, suggesting a subgroup of irradiated animals might be more sensitive to growth hormone, although the cause remains to be determined. Existing literature on the influence of GH on vascular function is conflicted. Both GH deficiency6,8 and excess 28 are associated with increased vascular intimal thickness, which is partially rescued by GH supplementation or inhibition, respectively. The extent of atherosclerotic plaque development is influenced by several factors, including inflammation and ROS production, cholesterol accumulation, and endothelial cell dysfunction.33,35 rhGH had no effect on atherosclerotic development in this study compared with vehicle, thus a factor not yet identified may be at play. rhGH has been reported to improve endothelial dysfunction associated with GH deficiency, 21 and we observed no differences in lipid profile between rhGH and saline treated animals in this study. However, GH has been shown to prime macrophages and monocytes for ROS production, 38 whereas inhibiting GH through administration of GHRH antagonists was found to have an anti-inflammatory effect in macrophages, 16 suggesting rhGH might enhance the extent of existing atherosclerotic plaques. In addition, vascular cell adhesion molecule 1(VCAM1), which is enriched in coronary atherosclerotic plaques, 26 was found to be induced in cultured endothelial cells upon stimulation with serum from healthy GH treated individuals. 18 We did not measure VCAM1 in this study, thus it remains to be determined whether a transient increase in VCAM1 subsequent to irradiation-induced vascular damage might exacerbate existing atherosclerotic lesions.
Taken together, these findings indicate rhGH does not have a lasting impact on cardiac function, collagen deposition, or lipid profile, supporting the safety of rhGH therapy at the dose and duration used in this study. Additional studies would be required to assess the therapeutic potential of rhGH in chronic cardiac damage associated with higher irradiation doses.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241277454 – Supplemental material for Exogenous Growth Hormone Exacerbates Post-Irradiation Atherosclerosis in Susceptible Epicardial Coronary Arteries
Supplemental material, sj-docx-1-tpx-10.1177_01926233241277454 for Exogenous Growth Hormone Exacerbates Post-Irradiation Atherosclerosis in Susceptible Epicardial Coronary Arteries by Krystal J. Vail, J. Daniel Bourland, Gregory O. Dugan, Benny J. Chen, Thomas B. Clarkson, J. Mark Cline and Giselle C. Meléndez in Toxicologic Pathology
Footnotes
Acknowledgements
We gratefully acknowledge the Cline Laboratory staff and the Wake Forest University Histopathology laboratory for technical support. We thank Brittany Macha for thoughtful feedback and proofreading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NIAID U19 AI67798 (Duke), U01 AI150578 and T35OD10946-5 (WFUSM), and K01OD036106-0 (TNPRC).
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
