Abstract
Degenerative lesions specific to the basal nuclei have not been described as a background finding in Beagle dogs. This report comprises a documentation of seven cases. In the context of a nonclinical safety studies, the authors suggest documenting the lesion descriptively as degeneration neuropil, basal nuclei, bilateral as it is characterized by (1) vacuolation, neuropil; (2) gliosis (astro- and/or microgliosis); and (3) demyelination. This novel lesion is considered a potential new background change for several reasons: (1) It occurred in animals from test item–treated and also vehicle-treated groups; (2) no dose dependency was observed; (3) in one of six affected test item–treated dogs, the given compound was shown not to penetrate the blood-brain barrier; and (4) statistical comparison between the proportions of affected dogs in the treatment and control groups did not yield a statistically significant difference. The etiology remains unknown and is subject to further investigations.
Keywords
Spontaneous encephalopathies are a well-described entity in dogs, may be acquired or genetically inherited, and may be caused by infections, trauma, toxic-metabolic disturbances, alterations in blood supply, and nutrient deficiencies. 4
Specifically in Beagle dogs, descriptions of encephalopathies are rare and comprise mainly type II fibrinoid leukodystrophy (Alexander disease) 8 or cortical atrophy in Lafora disease. 1 In laboratory Beagle dogs, spontaneous degenerative lesions in the central nervous system are uncommon 9 with the exception of spontaneously occurring axonal dystrophy. 7
Bilateral vacuolation of the caudate nucleus was recently discovered during histopathologic examination in individual Beagle dogs (Marshall BioResources, North Rose, New York) that had been used during conventional nonclinical toxicity testing. The dogs had been treated either with vehicle or compounds with or without the ability to penetrate the blood-brain barrier (Table 1). This report comprises a documentation of seven cases pertaining to six studies and includes animal information (age, sex), information on the designated dose group (low dose, mid dose, high dose, or control), and histomorphologic features of the lesion. Moreover, a term for documentation of the lesion in nonclinical studies is proposed. Due to the fact that the lesion was only discovered during histopathologic examination, this report is limited to its morphological characterization. Conclusions on a potential functional relevance of this finding could thus not be drawn.
Demographics of affected dogs, information on the vehicle and blood-brain barrier penetrance of a given compound, and special pathology procedures.
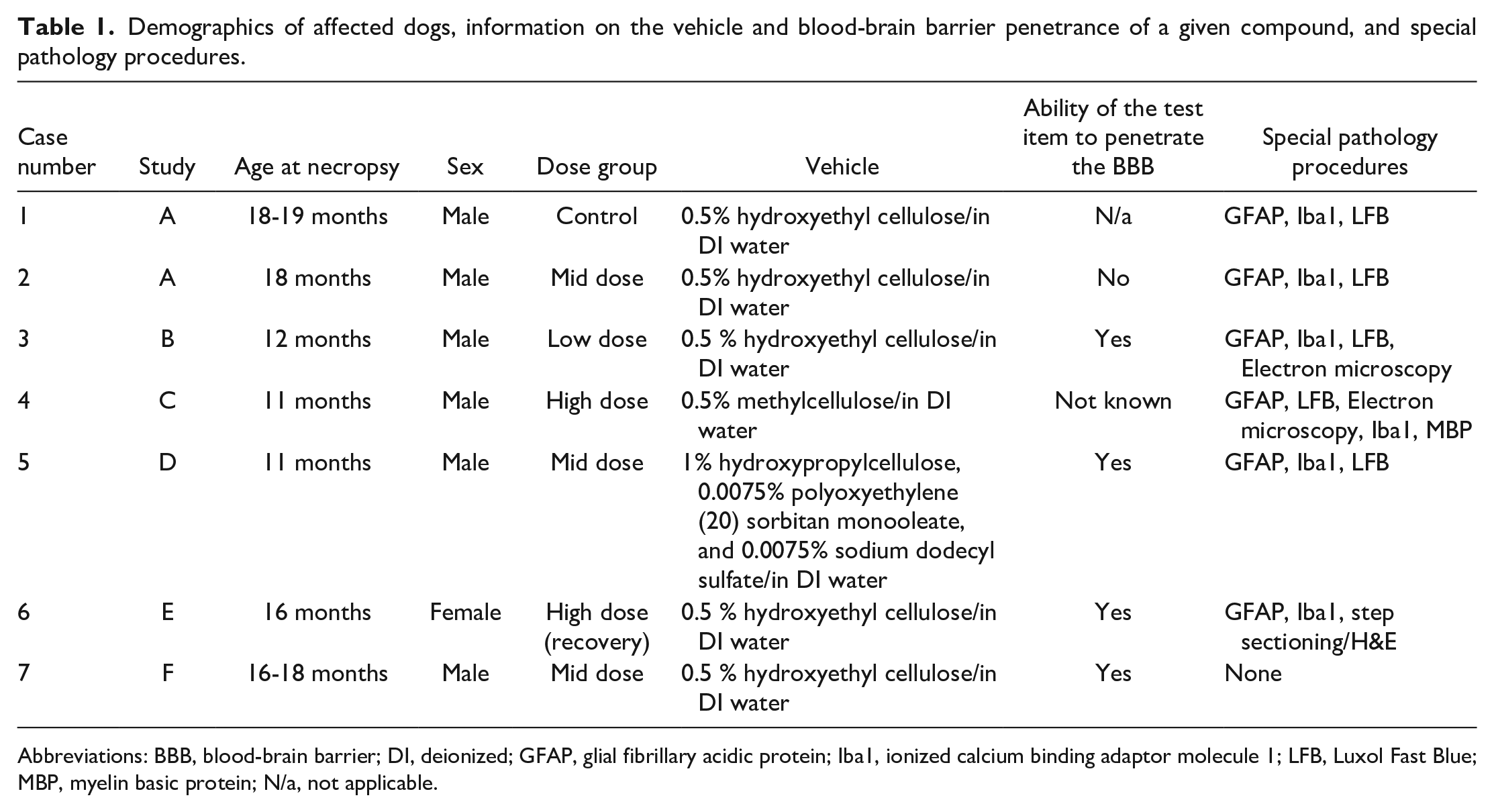
Abbreviations: BBB, blood-brain barrier; DI, deionized; GFAP, glial fibrillary acidic protein; Iba1, ionized calcium binding adaptor molecule 1; LFB, Luxol Fast Blue; MBP, myelin basic protein; N/a, not applicable.
All affected animals were used in studies conducted in an AAALAC (Association for Assessment and Accreditation of Laboratory Animal Care International)–accredited laboratory in full accordance with German animal welfare legislation and Boehringer Ingelheim principles for animal use. Six of the seven affected dogs (males; young adult; cases 1-5 and 7 [Table 1]) were designated to main study control or treatment groups of standard nonclinical safety studies. One young adult female was allocated in a recovery group (case 6 [Table 1]). There were no clinical signs or macroscopic lesions that could clearly be attributed to the vacuolation of the caudate nucleus in neither of the cases.
At necropsy, the time interval between exsanguination and start of brain fixation was approximately 30 minutes. Entire whole brains were immersion-fixed in a commercial formulation of neutral buffered 4% formaldehyde (Sigma-Aldrich, MerckKGaA, Darmstadt, Germany) on a rocking platform shaker at room temperature. After approximately 24 hours, the fixative was exchanged to fresh neutral buffered 4% formaldehyde and left to fix for a minimum of 48 hours before trimming of the brains. As a matter of course, brains were at no time placed in ethanol. Subsequently, brains were trimmed to seven sections meeting current best practice recommendations2,3,6 for general toxicity studies and routinely processed to paraffin blocks. Consistent with standard toxicological pathology practice, histopathologic evaluation was initially performed unilaterally on approximately 4-µm-thick paraffin sections, routinely stained with H&E (Axio Imager A2; Zeiss, Oberkochen, Germany). In the cases described here, examination was expanded to the contralateral side. In addition, in case 6 step sectioning and subsequent histopathologic examination on H&E-stained slides was performed. For immunohistochemical characterization of the lesion, antibodies directed against GFAP (glial fibrillary acidic protein; catalog no. Z0334; Dako, Agilent Technologies Inc., Santa Clara, USA) and Iba1 (ionized calcium binding adaptor molecule 1; catalog no. pab72473; Enzo Life Science GmbH, Lörrach, Germany) were used in six of seven cases. The LFB (Luxol Fast Blue) histochemistry method was used to demonstrate myelin in five cases. Case 4 was exemplarily stained with an antibody directed against myelin basic protein (MBP; order no. MAV386; EMD Millipore, MerckKGaA, Darmstadt, Germany). From one case, formalin-fixed wet tissues from the caudate nucleus lesion area and caudate nucleus area from an unaffected animal were further processed for ultrastructural investigation according to standard electron microscopy laboratory techniques. Ultrathin sections were investigated on a transmission electron microscope (912 AB; Zeiss).
Statistical comparison of proportions of affected dogs was performed between treatment and control groups of the facility’s dog cohort since the occurrence of the first case. To this end, a total number 158 male dogs in control groups and 487 male dogs in treatment groups were retrospectively analyzed. The proportion of animals affected was compared between treatment and control groups using a generalized linear model including a logit link function and a binomial assumption.
On routine histopathologic examination, a circumscribed bilateral symmetric pallor was observed subgrossly at the (dorso)lateral aspect of the caudate nucleus (Figures 1A and B and 2A). At higher magnification, there was variable and, in most cases, marked neuropil vacuolation, centering in but not being confined to white matter tracts (Figure 2B). Increased cellularity was interpreted as astro- and microgliosis. In more advanced and larger lesions, gemistocytic astrocytes characterized by a brightly eosinophilic cytoplasm with a peripherally placed nucleus and astrocyte-like processes were seen (Figure 2C). Neuronal degeneration could not be unequivocally identified in six of the seven cases. On step sectioning of the lesion in the recovery female (case 6), an extensive area of coagulative necrosis was discovered (not shown).
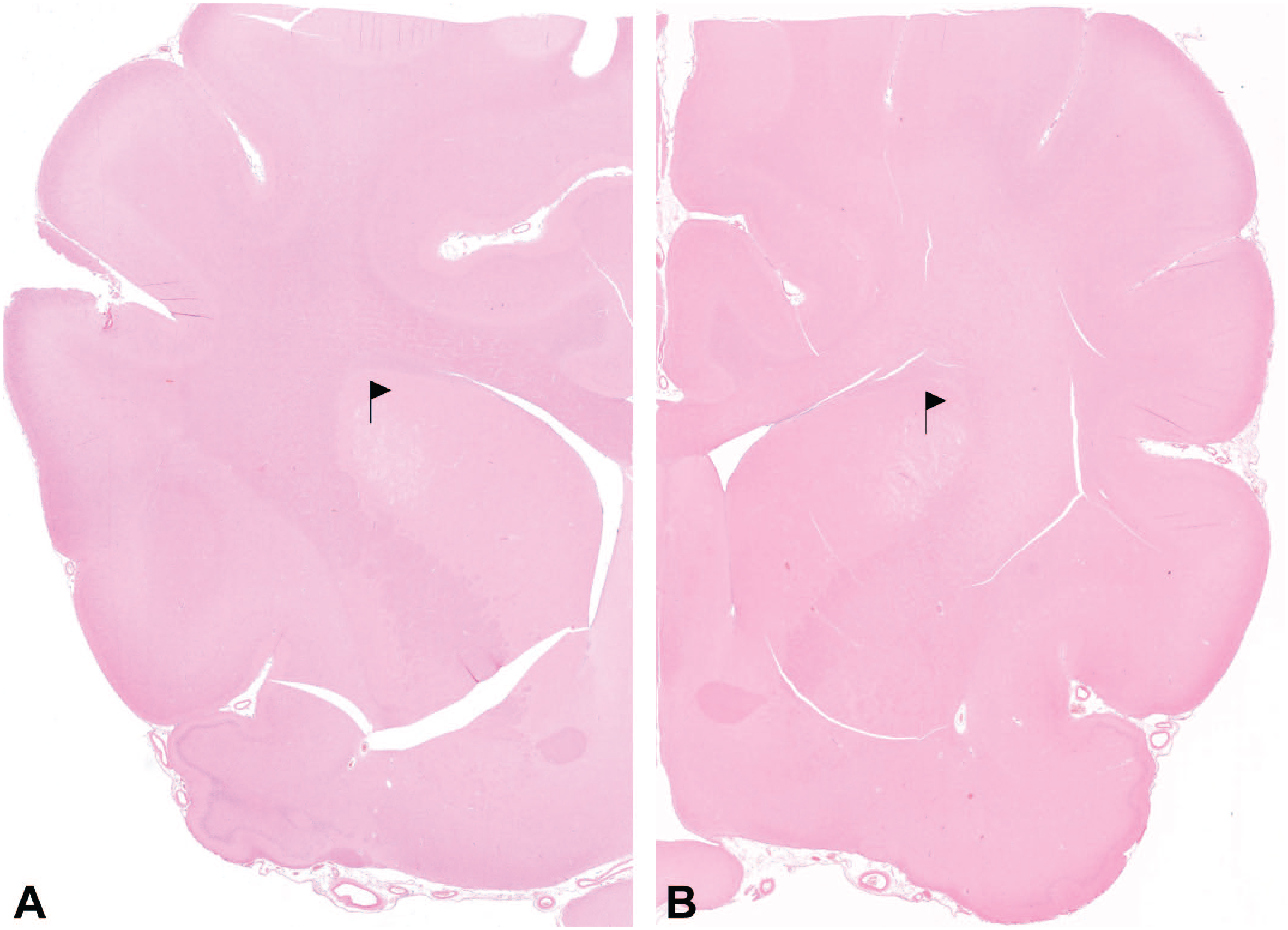
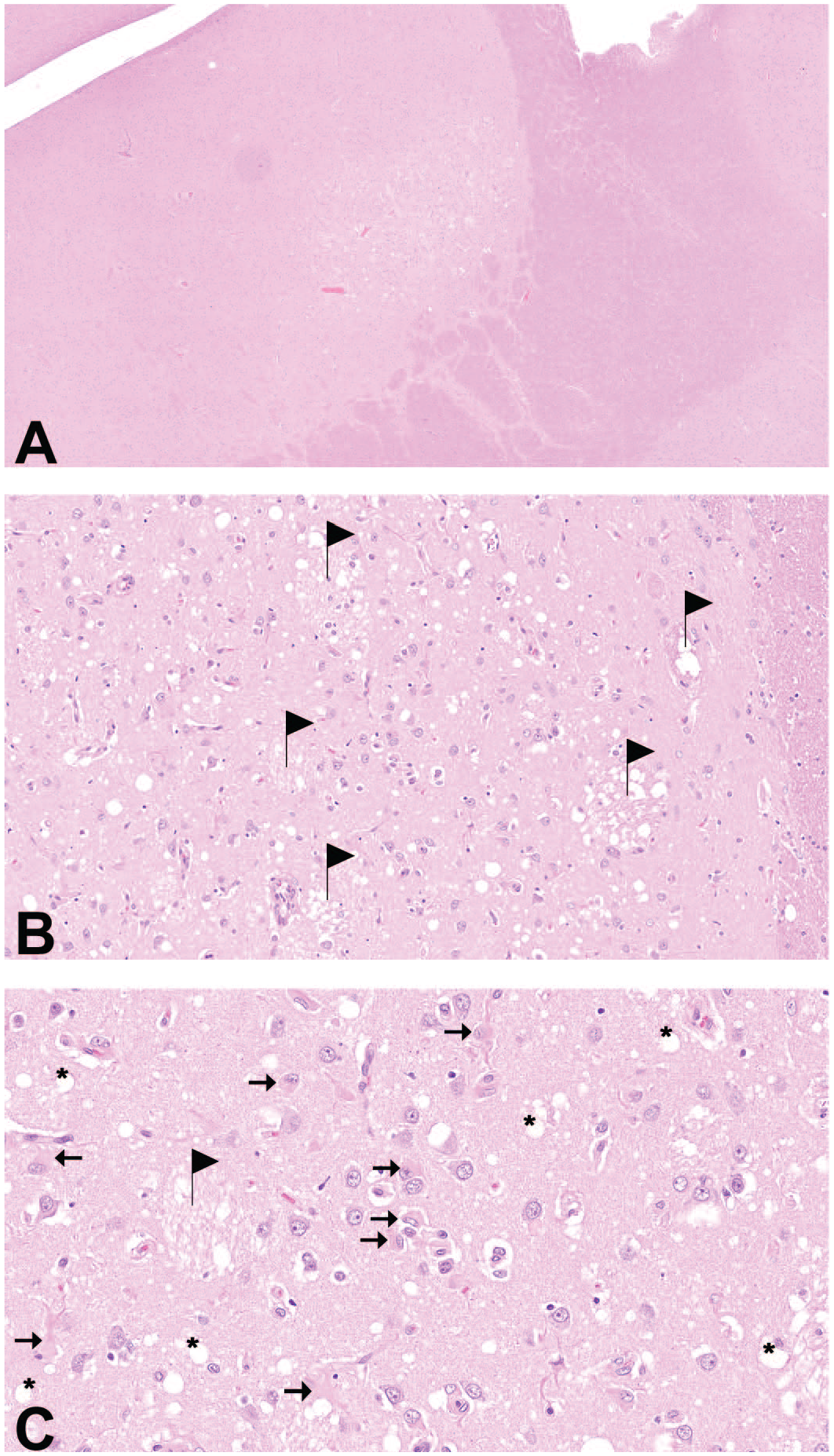
Case 1: A, overview of the circumscribed vacuolar lesion, HE, 2.5×; B, vacuoles are present in the neuropil and white matter tracts (flags) with evidence of gliosis, HE, 20×; C, gemistocytic astrocytes (arrows), vacuoles in the neuropil (asterisk) and vacuoles in white matter tract (flag), HE, 40×.
The GFAP immunohistochemistry performed in six of seven cases demonstrated an increase in number, size, and staining intensity of astrocytes (astrocyte hyperplasia/astrogliosis and astrocyte hypertrophy, Figure 3A). Astrocyte somata and filament-rich cell processes appeared thickened, plump, and swollen with markedly increased immunoreactivity compared with the slenderly branched processes in a control dog (Figure 3B). By Iba1 immunohistochemistry, a subtle increase in size and staining intensity of microglial cells was seen, which was hardly evident in H&E (Figure 3C). Some microglia appeared to be transforming toward the rod-shaped reactive phenotype displaying a more apparent perinuclear cytoplasmic rim. Cell processes, to a markedly lesser degree to what was observed in astrocytes, were thickened and more immunoreactive compared with a control dog (Figure 3D). A minimal increase in the number of microglia in the lesion was suspected considering the increased density of microglial processes. However, this impression may also have been due to an elongation and thickening of the latter. The MBP immunohistochemistry mirrored the lesion pallor that was seen in H&E at low magnification (Figure 4A). At higher magnification, a paucity of immunoreactivity was observed within the vacuolated neuropil with myelin sheaths having lost their integrity (myelin loss/demyelination). Myelin sheaths appeared altered displaying discontinuity and irregular staining intensity (Figure 4B). The LFB stain likewise showed decreased myelin density (not shown).
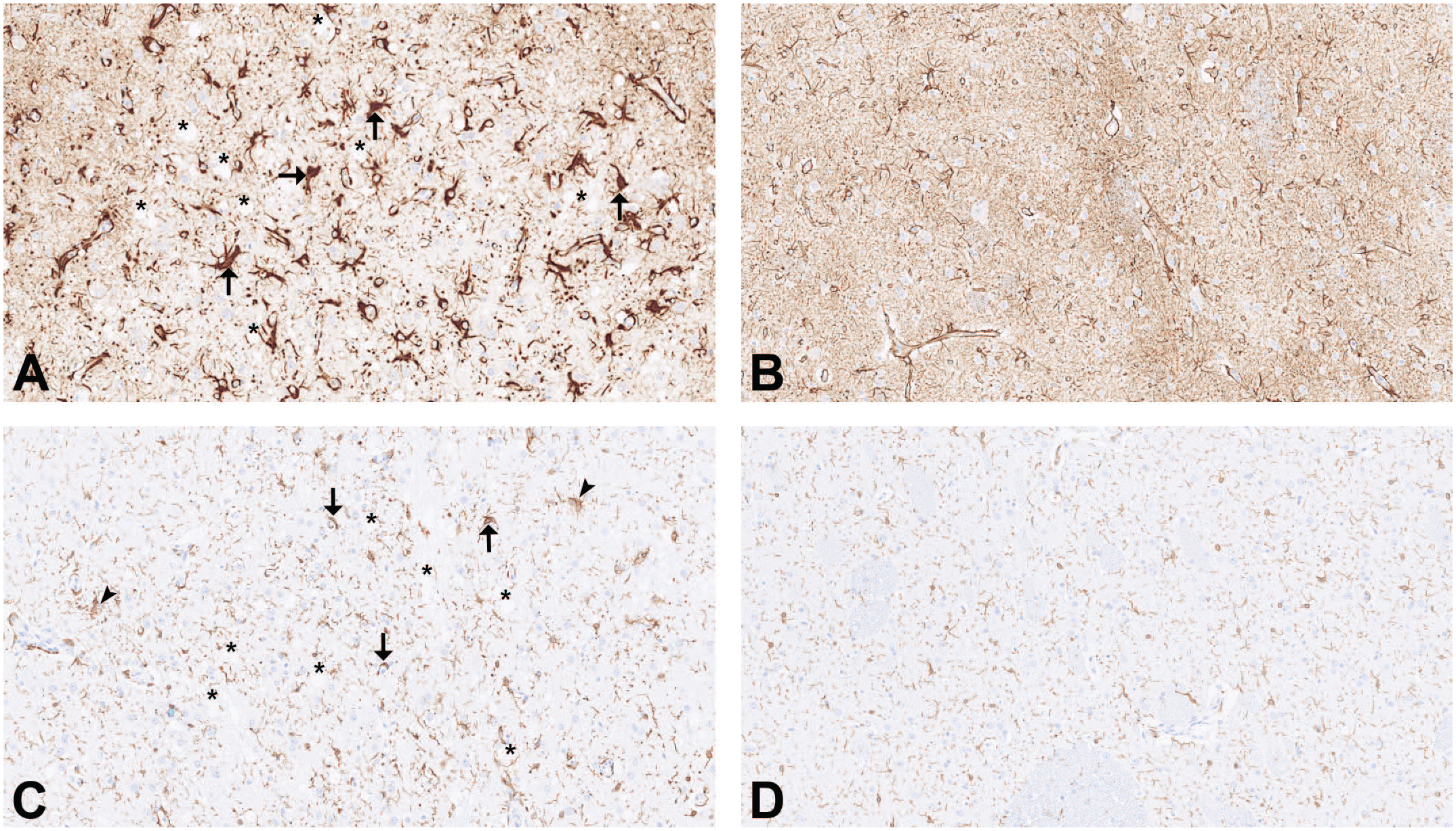
Case 1: A, increased staining of enlarged astrocyte cell bodies and processes (arrows), vacuoles in neuropil (asterisks), GFAP IHC, 20×; B, unaffected animal from the same study, GFAP IHC, 20×; C, subtle staining increase of microglia/microgliosis, rod-shaped activated microglia (arrows), areas of increased density of microglial processes (arrowheads), vacuoles in neuropil (asterisks); Iba1 IHC, 20×; D, unaffected animal from the same study, Iba1 IHC, 20×.
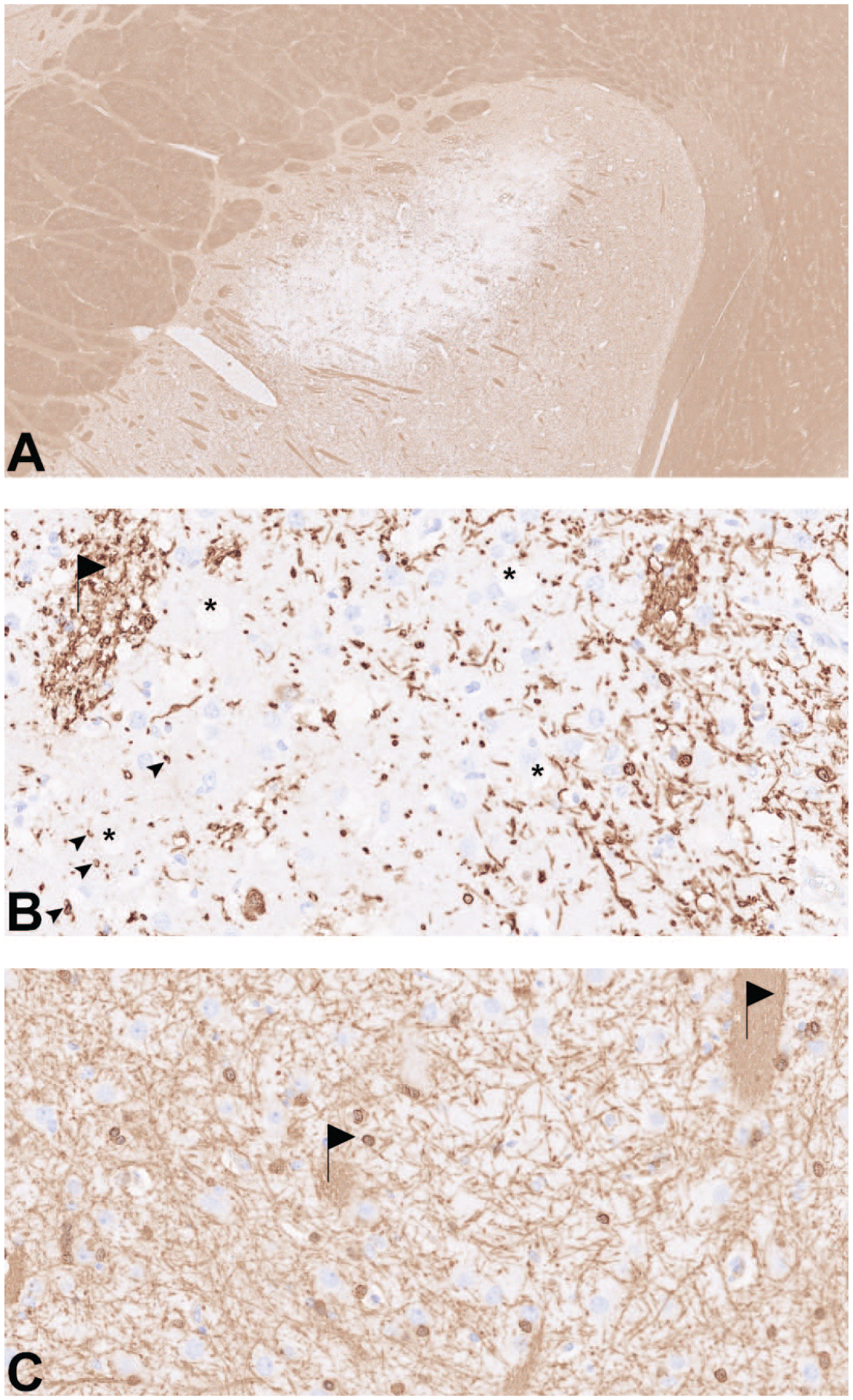
Case 4: A, decreased myelin stain delineates the lesion, MBP 2.5×; B, intralesional myelin loss and myelin alteration (arrowheads), vacuolated white matter tracts (flag), vacuoles in neuropil (asterisks), MBP IHC, 40×; C, unaltered myelin stain, white matter tracts (flags), caudate nucleus of an unaffected dog; MBP IHC, 40×.
Ultrastructurally, myelinated axons appeared to have been pushed apart by optically empty spaces (Figure 5A). These were interpreted as dilated myelin sheaths most likely corresponding to vacuolated neuropil observed in light microscopy. There was ballooning, distancing, and rupture of myelin sheaths, which was interpreted as distension and disintegration of myelin. Between the sheaths, accumulations of not further defined material were observed. Cell/axonal organelles, for example, mitochondria, were hardly discernible in axons with altered myelin sheaths (Figure 5A). In the control dog, myelinated axons were closely packed and featuring electron-dense, compact, concentric rings of myelin and clearly visible cell organelles (Figure 5B).
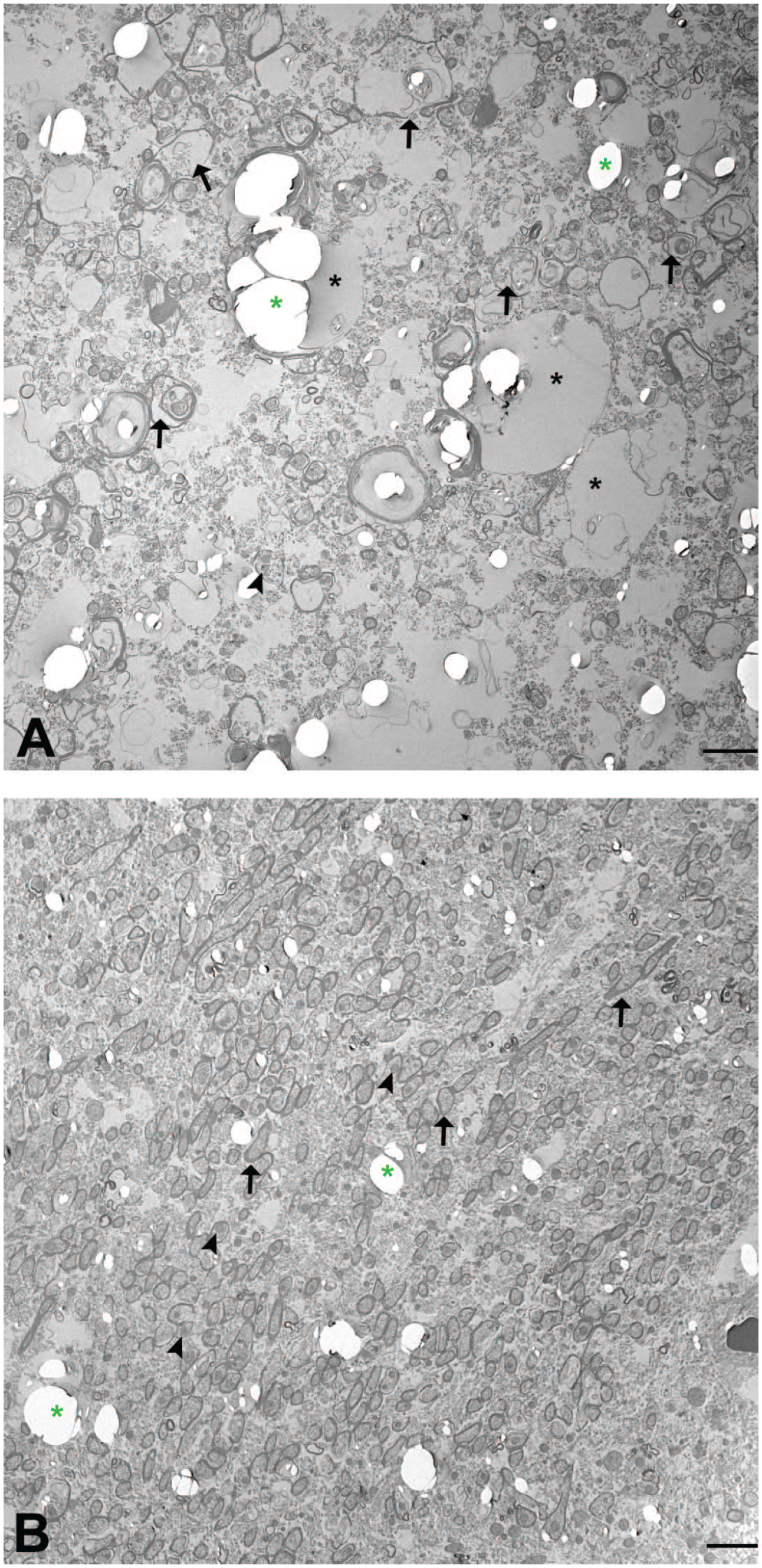
A, case 3, ballooning, distancing, and rupture of myelin sheaths with material accumulation between sheaths (arrows), poorly discernible cell organelles for example, mitochondria (arrowheads), dilated myelin sheaths presumably corresponding to neuropil vacuoles in light microscopy (black asterisks), processing artifacts (green asterisks), electron microscopy, bar = 2500 nm; B, unaltered/compact myelin sheaths (arrows), clearly discernible cell organelles, for example, mitochondria (arrowheads), unaffected animal from the same study, electron microscopy; bar = 2500 nm.
Statistical comparison of proportions of affected dogs between treatment and control groups did not yield a statistical significance at the 5% level, indicating no relationship of treatment with a test item and occurrence of the lesion. However, due to the low number of cases in control groups (n = 1), the statistical power of this calculation was considered low.
Taking into account the lesion characteristics of one high dose recovery female (case 6, central area of coagulative necrosis) and the overrepresentation of cases in studies with compounds that penetrate the blood-brain barrier, it cannot be ruled out completely that the treatment with a test item may provoke background lesion exacerbation. Background lesion exacerbation poses a great challenge on the toxicological pathologist to decide on test item-relationship and adversity. 5 It is, therefore, of importance that all lesions, including background lesions, are documented and thoroughly characterized to enable the study pathologist to differentiate between mere background and test item–related exacerbation. Accordingly, the authors recommend the following when this lesion is encountered by a study pathologist in a nonclinical safety study: (1) examination of the contralateral side of the appropriate section including the caudate nucleus to confirm the bilateral occurrence, (2) inclusion of GFAP and Iba1 immunohistochemistry and a myelin stain (MBP or LFB) as a minimum means to further characterize the lesion, and (3) recording the lesion in a descriptive way using the following term: degeneration neuropil, basal nuclei, bilateral. Aiming at international harmonization of lesion recording and thus enabling collection of data for this finding, the authors furthermore suggest this term to be introduced into the current INHAND nomenclature for the dog. 9
The etiology of this change is unknown at this time. In general, genetic imbalances leading to toxic-metabolic disturbances in the brain, intoxications, alterations in blood supply, nutrient deficiencies, or viral infection can be discussed as possible causes. Further investigations regarding pathogenesis are ongoing and include an extended panel of immunohistochemical stains, transcriptomics, proteomics, pedigree analyses, and whole genome sequencing. An international joint effort driven by pharmaceutical companies and contract research organizations (CROs) is currently in progress with the goal to gather a larger set of affected animals, to substantiate the interpretation as background lesion, and to elucidate further the yet unknown etiology.
Degeneration neuropil, basal nuclei, bilateral in the Beagle dog is regarded a potential background lesion for several reasons: (1) It occurred in animals from test item–treated and also vehicle-treated groups; (2) no dose dependency was observed; (3) in one of six affected test item–treated dogs, the given compound was shown not to penetrate the blood-brain barrier; and (4) statistical comparison of proportions of affected dogs between treatment and control groups did not yield a statistically significant result.
It is of utmost importance that toxicological pathologists are aware of this novel finding in Beagle dogs including the fact that it may occur spontaneously: With one exceptional study comprising two cases, there was always only one animal per study affected (with a total of 32 or 10 animals/study); thus, the significance lies in the likelihood that it may be seen in a test item–treated animal as a chance event. Due to its degenerative character, it would be considered as being adverse in nature in the context of nonclinical safety studies and difficult to monitor in clinical trials, if at all. Thus, it may significantly impact the benefit risk assessment and stall or terminate the development of a test item, if interpreted as a test item–related effect.
Footnotes
Acknowledgements
The authors would like to thank histology laboratory, electron microscopy laboratory technical, in-life and scientific staff, especially Mrs S. Kasten, D. Schrodi, M. Ruf, and L. Herkommer as well B.W. Igl for statistical investigations. B. Bolon, R. Garman, D. Rao, and I. Pardo are thanked for their scientific advice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
