Abstract
Respiratory diseases are one of the leading causes of death and disability around the world. Mice are commonly used as models of human respiratory disease. Phenotypic analysis of mice with spontaneous, congenital, inherited, or treatment-related respiratory tract abnormalities requires investigators to discriminate normal anatomic features of the respiratory system from those that have been altered by disease. Many publications describe individual aspects of normal respiratory tract development, primarily focusing on morphogenesis of the trachea and lung. However, a single reference providing detailed low- and high-magnification, high-resolution images of routine hematoxylin and eosin (H&E)-stained sections depicting all major structures of the entire developing murine respiratory system does not exist. The purpose of this atlas is to correct this deficiency by establishing one concise reference of high-resolution color photomicrographs from whole-slide scans of H&E-stained tissue sections. The atlas has detailed descriptions and well-annotated images of the developing mouse upper and lower respiratory tracts emphasizing embryonic days (E) 9.0 to 18.5 and major early postnatal events. The selected images illustrate the main structures and events at key developmental stages and thus should help investigators both confirm the chronological age of mouse embryos and distinguish normal morphology as well as structural (cellular and organ) abnormalities.
Introduction
The respiratory system has many vital functions including intake of oxygen, removal of carbon dioxide (CO2), protection from airborne environmental irritants and pathogens, and facilitating the senses of smell and taste. In order to fulfill these functions, the respiratory system operates as a complex interconnected network of air-filled passages. As with all organs and tissues, and especially complex ones, the respiratory system has the potential to be malformed during embryonic development. In humans, tracheoesophageal fistula (an abnormal connection [fistula] between the lumens of the esophagus and trachea) is one of the most common congenital respiratory anomalies, occurring in approximately 1 in 2500 to 1 in 3500 births.146,152 Such fistulas reflect abnormal division of the caudal foregut due to displacement of the site of tracheoesophageal septum formation, leading to a persistent connection between the trachea and esophagus. 146 Congenital pulmonary airway malformation (CPAM, also called congenital cystic adenomatous malformation of the lung [CCAM]) has an incidence of 1 in 1000 to 1 in 35,000 births.55,97 Infants with large CPAM lesions can experience a compression of the fetal esophagus with impaired swallowing of amniotic fluid, thereby resulting in excessive accumulation of amniotic fluid within the amniotic sac (termed polyhydramnios). 55
Aside from congenital defects, respiratory tract infections are the leading cause of morbidity and mortality from infectious diseases worldwide. 193 The following four respiratory diseases are the most common causes of illness and death globally: acute lower respiratory tract infections, which are responsible for 4 million deaths a year and are the leading cause of death among children under 5 years of age 184 ; tuberculosis, which infected 1.7 billion people in 2018 (23% of the world’s population) and thus was the leading lethal infectious disease in the world 25 ; chronic obstructive pulmonary disease (COPD) as the third leading cause of death18,50; and asthma, which affects 262 million people (approximately 3.5% of the world’s population). 163 As seen with the recent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, the emergence of lethal pathogens that attack the respiratory system stresses the importance of respiratory research.
Genetically engineered mice (GEM) are commonly used to recapitulate diseases and anomalies of the human respiratory tract. 174 GEM frequently die in utero or soon after birth due to lethal mutations leading to morphological defects. 178 Therefore, scientists studying translational medicine should develop a strong understanding of the normal murine respiratory system at each stage of life. Currently, phenotypic evaluation of the embryonic mouse respiratory system is a challenging task due to limited resources. Commonly used references include Kaufman’s 88 and Theiler’s 164 detailed, descriptive anatomical atlases of mouse development, Sulik et al.’s 155 scanning electron micrographs, Petiet and colleagues’ 131 high-resolution magnetic resonance histology atlas of the embryonic and neonatal mouse, and the Edinburgh 3-dimensional mouse embryo atlas project (eMAP). 183 While excellent, these resources mostly display images that are either not histologic representations, are shown at low magnification, and/or are black-and-white depictions with variably complete annotations. The purpose of this current atlas is to provide well-annotated color, high-magnification, and high-resolution images of conventional hematoxylin and eosin (H&E)-stained tissue sections for pathologists and biomedical scientists to use as a resource in identifying normal structures within the mouse respiratory system during embryonic and early postnatal development.
Tables of major morphological events of the upper and lower respiratory tracts are provided in Tables 1 and 2, respectively, as an overview of key events in murine respiratory system development. These milestones are further described and illustrated through many well-annotated microscopic images. An overview of normal adult mouse upper and lower anatomy is provided in Supplemental Section 1. The most common developmental defects are separated by upper and lower respiratory tracts in Tables 3 and 4, respectively, using phenotype data provided in The Jackson Laboratory’s Mouse Genome Informatics website. 172 Additional information on proliferative and nonproliferative lesions of the upper and lower airways can be found in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) publication that outlines standardized nomenclature for classifying microscopic lesions of the respiratory tract and olfactory organs in rodents.135,139 A comprehensive list of upper and lower respiratory tract abbreviations used in image annotation is provided in Supplemental Table 1.
Key developmental events in upper respiratory tract development.

Key developmental events in lower respiratory tract development.

Most common abnormal upper respiratory and laryngeal phenotypes in mice.

Main categories of upper respiratory tract phenotypes are indicated in bold text while subcategories are italicized and further indented. These data were compiled from the Mouse Genome Database, Mouse Genome Informatics website, hosted by The Jackson Laboratory, Bar Harbor, ME (http://www.informatics.jax.org/vocab/mp_ontology/MP:0002132); last accessed November 7, 2023.
Most common abnormal lower respiratory phenotypes in mice.

Main categories of lower respiratory tract phenotypes are indicated in bold text while subcategories are italicized and further indented. These data were compiled from the Mouse Genome Database, Mouse Genome Informatics website, hosted by The Jackson Laboratory, Bar Harbor, ME (http://www.informatics.jax.org/vocab/mp_ontology/MP:0002132); last accessed November 7, 2023.
Materials and Methods
Animals
Young adult, male and nulliparous female CD-1® IGS/Crl: CD1(ICR) mice (Charles River Laboratories, Raleigh, NC) 141 were obtained and quarantined for one week, after which they were mated overnight commencing at the start of the 12-hour dark cycle. This mouse model was used based on availability, large litter size, and the common use of this outbred stock for developmental toxicity testing in mice. Differences in developmental events do occur in the respiratory system among various mouse strains, 165 but this outbred stock was considered to be appropriate for this project as events in time follow the normal (i.e., “average”) course of respiratory development in mice.
Dams were group-housed (2-3/cage) in Green Line IVC Sealsafe PLUS mouse cages (Tecniplast, West Chester, PA) on autoclaved Sani-Chip hardwood bedding (PJ Murphy Forest Products Corp, Montville, NJ) and given reverse osmosis/deionized water and pelleted rodent chow (NIH-31; Envigo Corp, Indianapolis, IN) ad libitum. Environmental conditions were maintained at 22 ± 2°C and 45 ± 10% relative humidity. A constant light cycle (12 hours of light, 12 hours of dark) was maintained before and after breeding. Mice were provided with Enviro-dri® Eco-bedding (Shepherd Specialty Papers, Morrisville, NC) and nestlets for nesting purposes.
All animal procedures used in this study were approved in advance by the U.S. National Institute of Environmental Health Sciences (NIEHS) Institutional Animal Care and Use Committee (IACUC) and conducted in accordance with current US federal regulations 120 and the US National Research Council’s Guide for the Care and Use of Laboratory Animals. 121 Mice were maintained in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-approved animal facility where colony health surveillance reports and in-house quality assurance data confirmed that mice were free of known pathogens.
Time Points Evaluated
This atlas focuses on major respiratory tract developmental events from embryonic day (E) 9 (E9.0) to E18.5, with E9.0 being the start of foregut tube differentiation into the laryngotracheal groove and the initial appearance of the olfactory placode. In addition, postnatal day (P)1 to P30 were evaluated since P30 represents the approximate day when the adult respiratory tract conformation is attained.7,21,162
Embryonic Staging
The morning on which the vaginal copulatory plug was detected was designated as E0.5 (often described in the literature as 0.5 days post-conception [dpc]). During overnight breeding of mice, considerable variation occurs in the exact timing of ovulation and conception and in the developmental status of individual embryos within a given litter (i.e., calculated “developmental age” based on mating chronology ≠ observed “developmental stage” based on organ anatomy), with the age difference between the oldest and youngest embryos in a mouse litter reported to range from 10 hours 88 to 16 hours 143 to as much as 24 hours. 165 Therefore, in the current project, special care was taken early in gestation (E13.5 and earlier) to match both the external and internal features of each embryo to known developmental landmarks in staging embryos. 88 It is important to keep in mind that in order to view a specific anatomical landmark, each section may reveal a different structure than expected due to oblique cuts; therefore, serial sections may be beneficial. Corresponding Theiler stages (TS), another morphology-based system widely used for staging mouse embryos, 164 can also be utilized to ensure that control and experimental animals (whether engineered or mutant or treated) share the same developmental stage.
Terminology
The “embryo” classification scheme allows for a standardized staging system for human embryos, and distinguishing between these stages may occasionally be of critical importance. By this system, an “embryo” is the in utero entity in which all organ primordia are initially forming (weeks 1-8 following fertilization in humans,144,171 approximately E8-E14.5 in mice12,89), while a “fetus” is the in utero organism in which all primordia have formed and now are undergoing extensive organ expansion and remodeling (weeks 9-37 following fertilization in humans,144,171 about E15 to P7 for equivalent developmental stages in mice12,89). Since the mouse has a much shorter gestation period (19-20 days), the distinction between “embryo” versus “fetus” is less important in this species, whereas the developmental age after conception is critically important. For this reason, the term “embryo” is used in this atlas in referring to all prenatal stages of murine development between fertilization and birth, with the stage of development indicated by the gestational age (with mating occurring at E0 and conception designated as taking place at approximately 0.5 days after mating). 89
Anatomic terms follow the nomenclature convention for quadruped animals as stated in the Nomina Anatomica Veterinaria. 80 At first mention, corresponding medical terms are included in parentheses to ensure understanding among investigators. In some cases, medical terms have been used to annotate features in photomicrographs to both accord with terminology used in key mouse embryonic atlases88,89,131,164,171 (thus facilitating cross-comparison among respiratory tract structures across atlases) and to permit assignment of unique abbreviations to distinct features.
Selection of Control Specimens
Selection of appropriate control specimens (especially for times before E15.0) is defined in one of two fashions. The preferred means is to identify “developmental stage-matched” control mice using key macroscopic or microscopic features of the embryo rather than to choose “age-matched” controls based on the gestational day at which the animal is collected. This strategy is essential because the difference in developmental stage between the oldest and youngest embryos in a mouse litter of a given embryonic age varies from 10 to 24 hours11,110,143,165; exposure to a toxicant may amplify the apparent difference between developmental stage and embryonic age by delaying the rate at which treated embryos reach particular developmental milestones. 89 This factor is especially critical if the pathology assessment will include acquisition of quantitative data (e.g., morphometric or stereological measurements). While an obvious “best practice”, control mice should also share the same genetic background as the experimental animals because the average developmental stage for one mouse strain may vary by as much as 0.5 days from that of other mouse strains of the same embryologic age. 165 For transgenic experiments, the genotypes of the embryos are typically determined via limb snips (E11.5-E13.5) or tail snips (E14.0-E18.5), and the wild-type littermates are used as controls; yolk sac or amniotic sac (up to E11.5) also may be used as a specimen for genotyping. Normal developmental variation of embryos within a litter should still be considered during histopathological evaluation.
Tissue Collection, Handling, and Processing
Embryo collection was carried out on the mornings of the designated days, between E9.0 and E18.5. Pregnant mice were euthanized by carbon dioxide inhalation according to the NIEHS standard operating procedure 5 for euthanasia of rodents. Using a dissecting stereomicroscope (Leica MZ16; Leica, Vista, CA), individual embryos and selected tissues were isolated and immersed in ice-cold 0.1M phosphate-buffered saline (1× PBS, pH 7.4). Near-term (E17.5 and E18.5) embryos were euthanized by hypothermia, 5 after which umbilical vessels were cut between the placenta and embryo to rapidly halt oxygen uptake.
The E18.5 embryos were blanched by immersion for 20 seconds in boiling water followed by 20 seconds in ice water prior to fixation to aid removal of the epidermis, thereby improving fixative penetration. 7 Following blanching, the end of a cotton swab was used to slowly and gently rub the epidermis, thus peeling away/removing the impermeable outer skin layer. For this method, one embryo was blanched at a time.
Embryos were fixed by immersion in either neutral buffered 10% formalin (NBF) containing ~1% methanol as a stabilizer (IMEB Inc., San Marcos, CA) or 4% methanol-free formaldehyde (MFF, known colloquially as “paraformaldehyde”; Affymetrix) at 4°C. Fixation duration depended on the embryonic age/stage (i.e., size). Examples of fixation times for embryos of different ages are as follows: E9.0 to E11.5, ~12 hours; E12.5 to E14.5, ~24 hours; E15.5 to E16.5, ~48 hours; and E17.5 to E18.5, ~72 hours. Embryos were then processed by dehydration in a graded series of ethanol (starting at 70%), clearing in xylene, and embedding in paraffin. Embryos younger than E12.5 were embedded in 1% agar before processing into paraffin to minimize direct handling and facilitate specimen orientation during embedding.
For each time point, embryos were embedded on their backs, sides, or heads for sectioning in the frontal (coronal), sagittal (longitudinal), or transverse (horizontal) planes, respectively. Serial 5- to 6-µm-thick sections through the entire embryo were placed on Superfrost Plus and ColorFrost Plus Microslides (A. Daigger & Company, Vernon Hills, IL). Every fifth slide was routinely stained with H&E to permit histopathologic evaluation and whole-slide scanning. Some color variation among the H&E stained slides occurs due to tissue processing at different times. The histological processing schedule for the embryos utilized for this atlas is provided in Supplemental Table 2.
Section Scanning
Bright-field whole-slide scanning was completed on all stained slides with a ScanScope AT2 instrument (Leica). Regions of interest were digitally captured as screen shots using ImageScope software (v12.4.3.5008; Leica). If an image required rotation, the selected region of interest was captured using the extraction feature in ImageScope. Global white balance correction and image resizing were completed where warranted using Adobe Photoshop (v2014.0.0 or later; Adobe, San Jose, CA). Image resolution was set at 300 dpi to fit the publisher’s requirements for acceptable image resolution in the journal. All magnifications indicated in figure legends are the scanned image magnifications, although some images may have been cropped slightly to remove distracting white borders at the margins.
Morphologic Evaluation
Embryos were evaluated macroscopically at necropsy (prior to blanching or immersion in fixative). Coloration of the embryo was noted (pink, pale, gray, etc.), and determinations of heartbeat and circulation were made. Subsequently, the evolution of normal respiratory tract structures was assessed in tissue sections using a bright-field microscope and/or whole-slide scanned images. The atlas was assembled from representative microscopic images, annotated to identify salient features of the evolving organization of upper and lower respiratory tract structures.
Overview of Early Respiratory Tract Development (E9.0-E10.5)
The vertebrate respiratory tract arises through differential development, as the formation of upper respiratory and lower respiratory airways develop from varying components of the germ layers. The upper respiratory tract (nasal passages including sinuses) involves anatomic evolution of the ectoderm (outermost layer of germ tissue) and superficial mesoderm (middle layer of germ tissue) associated with the rostral pole of the embryo, while the lower respiratory tract (larnyx, trachea, bronchi, and lungs) reflects extensions from the primitive foregut endoderm (inner layer of germ tissue) and deep mesoderm.
Upper Respiratory Tract
The nasal (or olfactory) placodes appear bilaterally as thickenings of the ectoderm (epithelium) on the rostral pole of the embryo ventral to the telencephalon (primordial forebrain) around E9.0. 168 As embryonic development proceeds, the lateral edges of the frontonasal region (primordial face) rotate toward the central axis, bringing the nasal placodes into proximity and establishing the snout (nose) and philtrum (midline indentation) of the upper lip (Figure 1). Central depressions in the nasal placodes are termed the nasal pits. The pit openings are the progenitors of the nares (nostrils) while the invaginations will become the nasal passages. The epithelium lining the nasal passages will differentiate into respiratory epithelium rostrally and olfactory (sensory) neuroepithelium caudally.
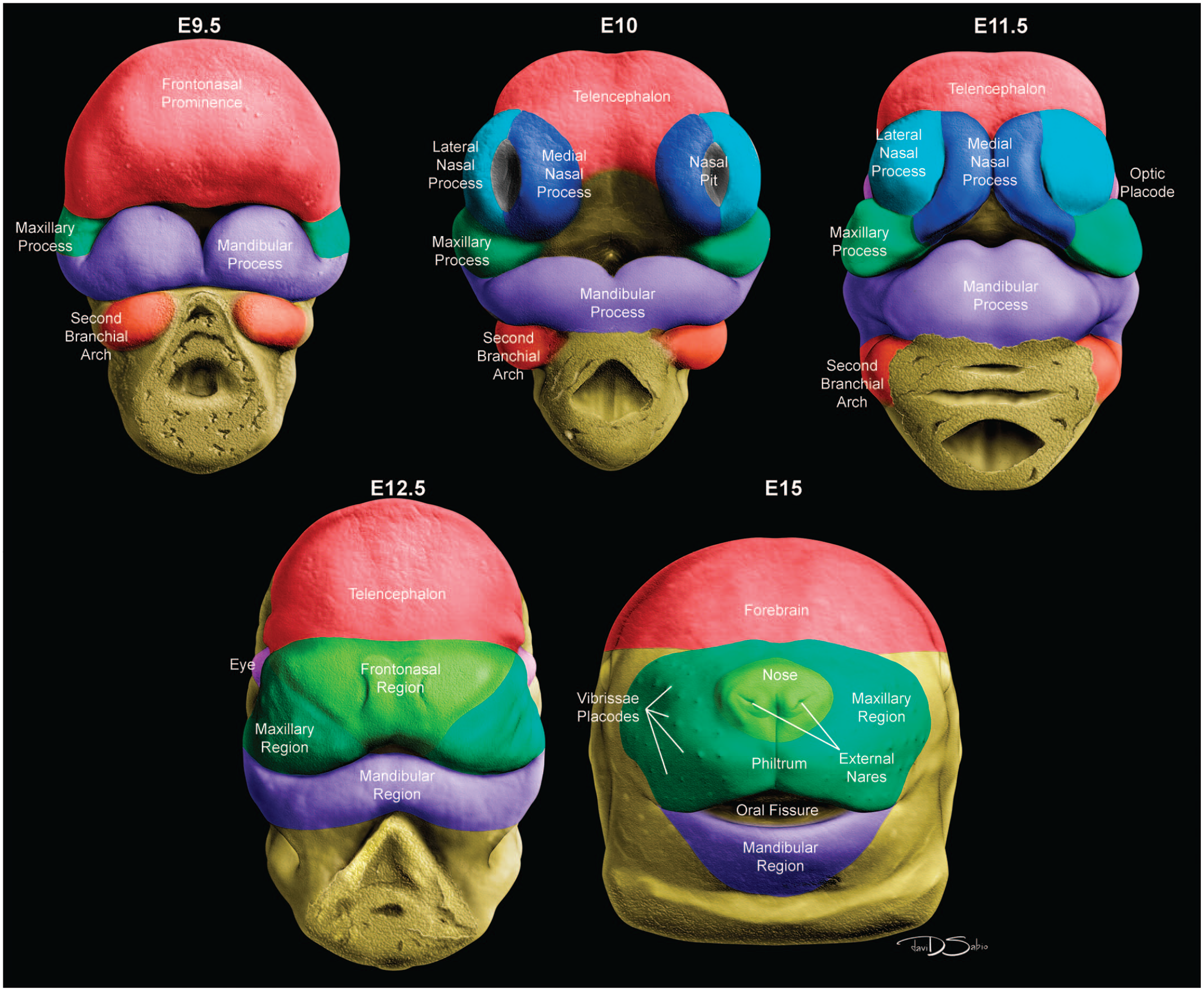
Diagrammatic representation of the formation of the nares and five facial processes of the embryonic mouse between E9.5 and E15. At E9.5, the face is characterized by a prominent frontonasal prominence (or process) as well as the paired maxillary processes, mandibular processes prior to fusion, and second branchial arches. At E10, growth of the maxillary processes causes the nasal pits to migrate medially and transform into elongated slits while the medial nasal processes grow ventrolaterally; at this stage, the medial and lateral nasal processes begin to fuse, forming the nares. At E11.5, growth of the maxillary processes continues to shift the nasal pits medially, the medial nasal processes mediofrontally, and the lateral nasal processes rostrally. The distal ends of the maxillary and medial nasal processes begin to merge while fusion of the lateral and medial nasal processes permits the nasal pits to evolve into nasal chambers and eventually nasal ducts. At E12.5, the maxillary and medial nasal processes have expanded mediofrontally and fused to produce the frontonasal region, completing the formation of the upper lip. At E15, formation of the right and left nasal passages of the external nares occurs by fusion of the nasal septum and palatal shelves. The oral fissure (stomodeum) is prominent, and vibrissae placodes begin to form.
Junctional Area
The pharynx serves as a junction that passes materials to both the respiratory tract (as a connection between the nasal passages and trachea) and the digestive tract (as a connection between the oral cavity and esophagus). This complex cavity is located in the cranial cervical region, ventral to the caudal aspect of the basal skull. The pharynx comprises three elements: the nasopharynx, which is located rostrodorsally and transmits air from the nasal passages; the laryngopharynx, an intermediate and ventral domain that receives air from the nasopharynx; and the oropharynx, which is located dorsocaudally and accepts consumed fluids and solids from the oral cavity. In adults, the larynx is the main respiratory tract component in this region and is classified variously as a component of the upper or lower respiratory tract in the literature. In this atlas, the larynx is discussed as a junctional area due to its interaction with both the upper and lower respiratory tracts.
Between E10.0 and E10.5, the primitive laryngopharynx exists within a segment of the foregut at the level of the fourth branchial arch (Figure 2). The caudal laryngopharynx eventually develops into the vocal folds with the cranial region becoming the supraglottis (upper part of the larynx). 106 The ventral laryngopharynx serves as the origin of the trachea and lung buds, while the dorsal laryngopharynx expands into the esophagus. 107 Also at this time, the laryngotracheal groove is at the cranial border of the fourth branchial arch, and the primitive pharyngeal floor is at the caudal border, just cranial to the tracheoesophageal septum.70,107
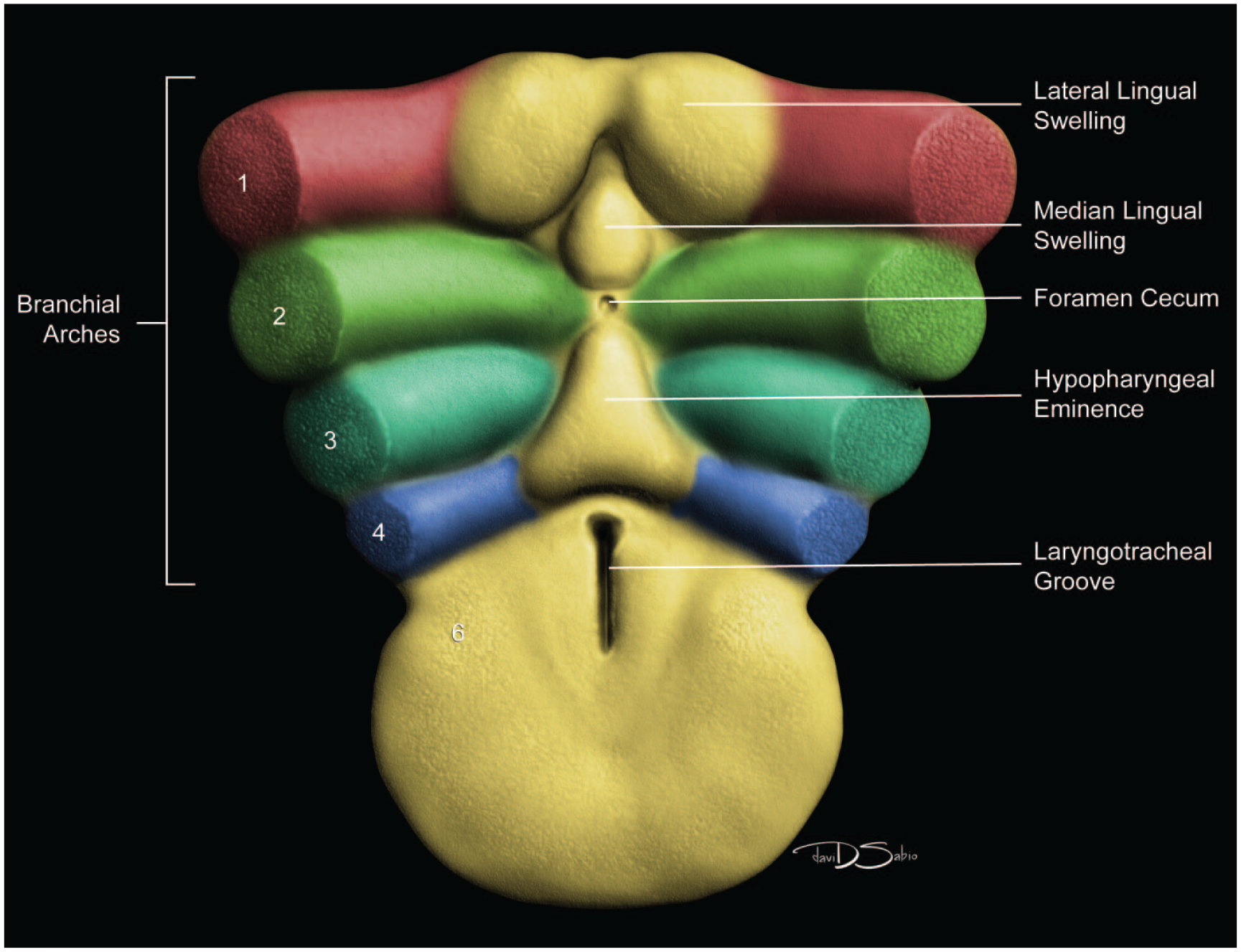
Diagrammatic representation of the branchial arches and laryngotracheal groove of the mouse embryo between E9.0 and E9.5. This image depicts branchial arches 1 to 4 and 6. A mid-sagittal slit, the laryngotracheal groove, appearing at the border of the fourth pharyngeal pouch is also indicated. The branchial arches are derived from all three germ layers and help develop specific areas of the face, neck, and pharynx. The first pharyngeal pouch lies between branchial arches one and two, the second between arches two and three, the third between arches three and four, and the fourth between arches four and six. The fifth arch either disappears during embryonal development or is nonexistent. The proximal region of the laryngotracheal groove develops into the larynx and trachea while the distal region gives rise to the right and left primary lung buds, which later form the right and left main bronchi. The median lingual swelling, foramen cecum of the tongue, and hypopharyngeal eminence are shown for orientation.
Lower Respiratory Tract
Dorsoventral patterning of the foregut tube results in separation of the future esophagus and trachea (Figure 3). 81 The separation is thought to be due to fusion of the lateral ridges of the foregut endoderm and mesoderm. 81 The opposing walls of the foregut migrate toward the middle of the lumen until the two walls make contact. 134 Separation between the esophageal and tracheal primordia occurs as these walls join at a ventral position of the forgut and extend dorsally over time. This partitioning, along with rearrangement and differentiation of epithelial cells, results in formation of the two separate esophageal and tracheal tubes. 134 Tracheoesophageal separation is divided into five sequential processes: craniocaudal (anterior-posterior) patterning (E7.0-E8.5), dorsoventral patterning (E8.5-E10.5), tube separation (E9.5-E12.5), tube elongation (E10.5-E14.5), and tube diameter expansion and elongation (E14.5-E18.5).92,93,150,192 The tube elongation and diameter expansion processes are elaborated further in the sections below.
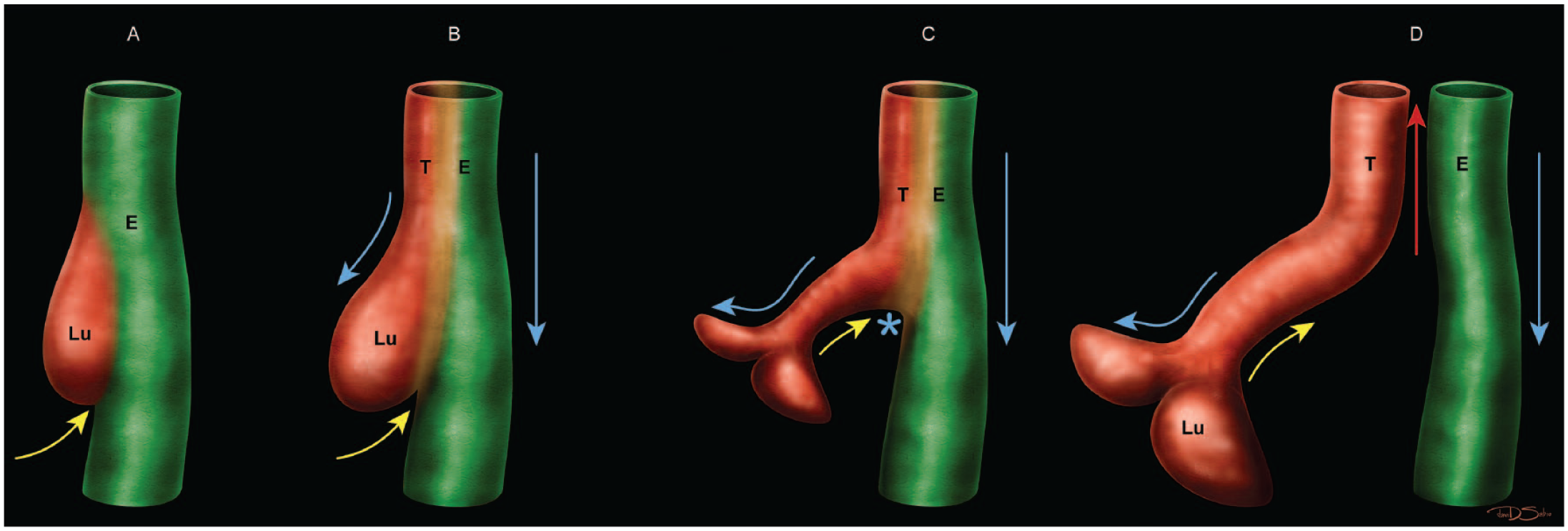
Diagrammatic representation of the “splitting and extension model” of lung and tracheal development in which dorsoventral patterning of the endodermal foregut tube results in separation of the future esophagus and trachea. Panel A depicts the first wave of “splitting” (separation) beginning when the epithelial saddle emerges at E9.5 between the primordial lung (Lu) and esophagus (E). The yellow arrow represents the epithelial saddle-like structure elevating. Panels B and C demonstrate the saddle expanding, bifurcating, and “extending” distally, resulting in the lung separating from the esophagus. The blue arrows represent the lung and esophagus elongating distally. The blue asterisk in panel C indicates the location where the first wave of splitting ends. Panel D shows the second wave of splitting, where the saddle continues moving cranially until the separation of the trachea (T) and esophagus is complete (as indicated by the red arrow).
The first process of tracheoesophageal separation (cranio-caudal patterning) begins between E7.0 and E8.5 when the primitive gut tube segregates along the craniocaudal axis into the foregut, midgut, and hindgut. At E7.5, the blind-ended foregut pocket invaginates toward the rostral end of the embryo.99,147,192 Subsequently, the lateral endoderm folds toward the ventral midline in a cranial-to-caudal direction.99,147,192
For the second process, dorsoventral patterning of the foregut results in the separation of the future esophagus dorsally and trachea ventrally (Figure 3). Dorsoventral patterning begins after the initial (or “embryonic”) stage of lung development and is recognized by formation of two (left and right) primary lung buds, which emerge at the distal end of the trachea at E9.5. Subsequently, the trachea appears at the cranioventral foregut endoderm dorsal to the lung buds. 92 The primitive trachea and primary lung buds begin to elongate distally. 93 An epithelial saddle-like structure, consisting of cells of the primitive lung and esophagus, forms at the distal end of the cranial foregut at E9.5 and shifts dorsally to divide the lung from the esophagus (Figure 3). 191
The third process, tube separation, occurs between E9.0 and E9.5 when a sagittal slit, known as the laryngotracheal groove, appears at the level of the fourth branchial arch (Figure 2).18,88,106,175 The proximal region of the laryngotracheal groove develops into the larynx and trachea while the distal region gives rise to the left and right primary lung buds, which later form the left and right main bronchi.88,175 Each primary lung bud later divides to form secondary lung buds supplied by large bronchial branches; in mice, the left primary lung bud forms one bronchial branch while the right primary bud forms four bronchial branches. 76 The secondary lung buds supplied by these bronchial branches ultimately differentiate into the distinct five lung lobes (one left and four right) of the adult. 21
The lung arises from two germ layers: The gut endoderm gives rise to the lung epithelium, whereas the splanchnic mesoderm gives rise to the lung connective tissues. The second (or “pseudoglandular”) stage of lung development, detailed further below, begins at E9.5 and is characterized by the development and branching of the bronchial tree within the lung buds.73,100 These airways arise as extensions from the trachea (a derivative of the ventral endodermal foregut) that protrude into the surrounding mesenchyme of the laryngotracheal groove cranially and the midgut caudally to initiate formation of the primary buds of the left and right lungs (Figure 4).21,88,111 The intricate tree of bronchi and bronchioles are generated by branching morphogenesis. 98 The lung buds continue to expand and elongate into the pericardioperitoneal canals at E10.0 where they are bounded by the trachea cranially and the ventral gastric dilation region caudally. 88 The pericardioperitoneal canals open caudally between the primitive pericardial and peritoneal cavities and will eventually form the two pleural cavities surrounding the lung. These canals are the first component of the diaphragm, which divides the thoracic and abdominal cavities. 154
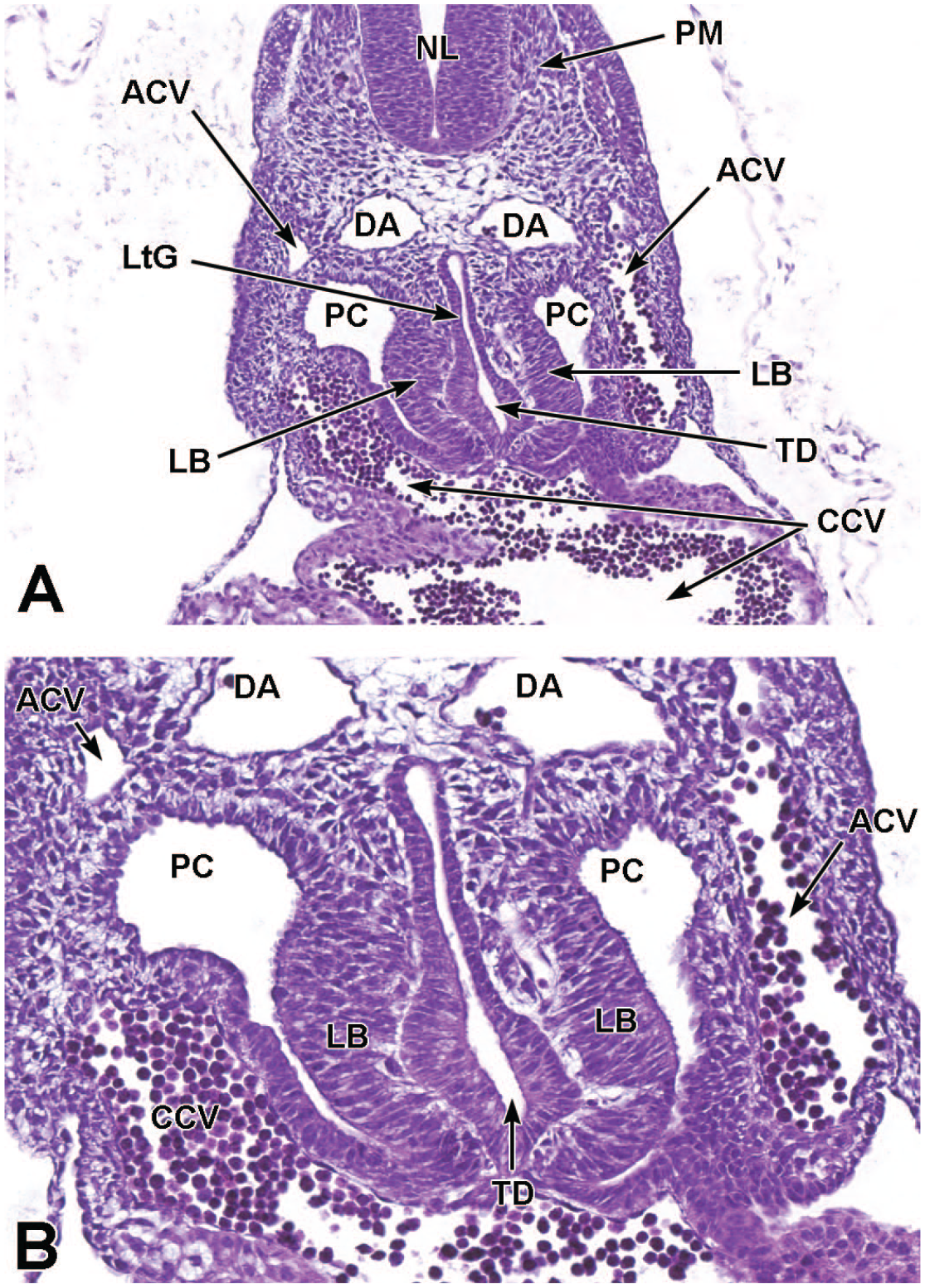
Transverse images illustrating the laryngotracheal groove and developing lung buds at E9.5. Panels A (10×) and B (20×) illustrate the laryngotracheal groove (LtG) elongating within the endodermal foregut and early differentiation of the right and left lung buds (LB) as they begin to arise from the cranial ventral foregut endoderm. The neural lumen (NL); condensation of paraxial mesoderm (PM) for the cervical myotome; right and left anterior cardinal (internal jugular) veins (ACV); right and left dorsal aortas (DA); right and left pericardioperitoneal canals (PC); common cardinal vein (CCV); and tracheal diverticulum (TD) are labeled for orientation. (Note: extra tissue on panel A located on the right side of the field was removed for optimal viewing of anatomic features.) H&E.
Upper Respiratory Tract Development (E9.5-E18.5)
The upper respiratory tract is comprised of major airways in the head (nasal passages and sinuses; Figure 5) and the various apertures (external nares of the snout and choanae [internal nares] of the nasopharynx) that permit air to enter and exit these channels. A biologically significant satellite function served by these channels is olfaction, which is served by the main olfactory mucosa (a specialized sensory neuroepithelium that arises principally from the ectoderm of the olfactory placode with a lesser contribution from neural crest cells) 86 and a series of ancillary olfactory subsystems. The respiratory and olfactory organs evolve in tandem, as described below. Figure 5 is a representation of the adult murine nasal cavity as a structural reference.
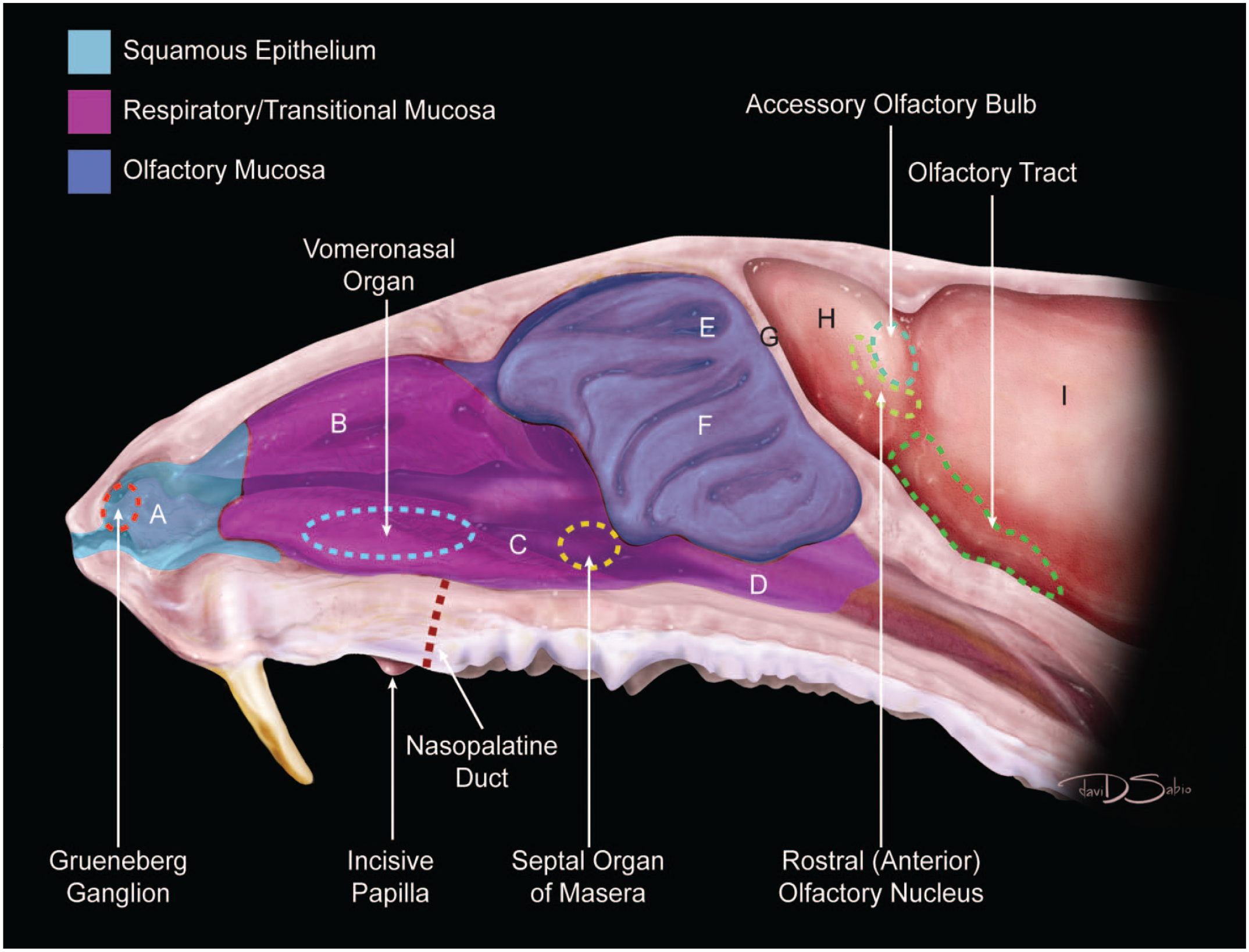
Diagrammatic representation of the nasal cavity of the adult mouse. Squamous epithelium is found in the nasal vestibule (A). Respiratory/transitional mucosa lines the nasoturbinates (B), maxilloturbinates (C), and nasopharynx (D). Olfactory mucosa is located within the olfactory recess (E) and ethmoturbinates (F). The cribiform plate (G), olfactory bulb (H), and brain (I), as well as the locations of key ancillary sensory organs—Grueneberg ganglion, vomeronasal organ, septal organ of Masera —and other recognizable anatomic landmarks such as the incisive papilla and nasopalatine duct (also called the incisive canal or nasopharyngeal canal) and elements of the olfactory brain (rostral [anterior] olfactory nucleus, accessory olfactory bulb, and olfactory tract) are also indicated.
Craniofacial and Nares Development
In mice, facial development begins with the formation of five facial processes (also called prominences): the frontonasal process, paired maxillary processes, and paired mandibular processes, all of which are located around the stomodeum (primitive mouth) at E9.5 (Figure 1).19,84,158 The frontonasal process is located rostrally, the maxillary processes are found laterally, and the mandibular processes are positioned caudally.19,84 Neural crest cells (Table 5), which originate from the interface between the outer surface epithelium and inner neuroepithelium covering the sides of the developing rostral neural tube, support the facial processes by migrating in the ventrolateral direction from the brain to expand the evolving facial processes.19,161 The mesenchyme of the five facial processes originates from the developing rostral neural tube. 19 The maxillary processes develop into the upper lip, the maxillary bone, and the secondary palate. 100 The mandibular processes give rise to the mandible and rostral two-thirds of the tongue. 100 The frontonasal process develops into the nose, the upper lip, and the primary palate. 100 The frontonasal process is divided into the medial and lateral processes through the formation of the nasal pits. 19
Contribution of neural crest cells to the developing respiratory system.

The development of the upper lip begins when the stomodeum is defined rostrally by the primitive forebrain and caudally by the first branchial arch (Figure 2) at E9.0. 84 The unpaired frontonasal process is located rostral to the stomodeum and consists of mesenchymal cells originating from the fore- and midbrain neural crest. 84 The stomodeum is confined laterally by the pair of maxillary processes and caudally by the pair of mandibular processes. 84 At E10.0, the nasal placodes are formed due to the raised, circular thickening of surface ectoderm on the ventrolateral region of the frontonasal process. 84 This process continues to develop around the nasal placode, resulting in the formation of the nasal pits and the lateral and medial nasal processes (Figure 1). 84 At E10.5, growth within the maxillary processes causes the nasal pits to be pushed medially and the medial nasal processes to grow ventrolaterally. 84 This movement causes the nasal pits to shift in shape from round orifices to elongated slits. 84 The medial and lateral nasal processes begin to fuse together at E11.0, forming the nares (nostrils; Figure 1). 84 The growth of the medial nasal and maxillary processes causes the lateral processes to move rostrally, causing the distal regions of the maxillary and medial nasal processes to fuse and form the upper lip. 84
The maxillary processes continue to push the nasal pits and medial nasal processes mediofrontally between E11.5 and E12.0 (Figure 1). 84 These developmental processes cause the nasal pits to form into paired nose chambers, and eventually nasal ducts, once the fusion between the medial and lateral processes is complete. 84 Between E15.0 and E15.5, the nasal septum fuses with the palatal shelves, resulting in the formation of the left and right nasal passages. 88 The left and right passages each have an external naris (Figure 1) where the air enters to pass caudally through the nasal cavity to reach the choana (also termed the internal naris or caudal nasal aperture). Passage through the choana directs inspired air into the nasopharynx and ultimately into the larynx. 88
Nasal Septum and Sinuses
In the adult mouse, a delicate cartilaginous nasal septum separates two symmetrical nasal compartments. The nasal septum is a planar structure, and the nasal cavities and associated nasal sinuses are formed and supported by the development of the curved rostral (anterior), paraseptal, and parietotectal cartilages. In adult mice, there are four types of epithelia found in the nasal cavity: squamous, transitional, respiratory, and olfactory (Figure 5). 4 Squamous stratified epithelium lines the nasal vestibule. A ciliated pseudostratified cuboidal-columnar respiratory epithelium lines the turbinates, nasal septum, and paranasal sinuses of the nasal chamber. A pseudostratified olfactory neuroepithelium lines the entire dorsocaudal region of the nasal chamber as well as some parts of the nasal septum and turbinates. The transition zones between the squamous and respiratory epithelium are lined by a nonciliated cuboidal transitional epithelium.
The septum is a component of the nasal capsule (the cartilage case that encloses and stabilizes the embryonic nasal cavity) and is a neural crest cell-derived tissue that has been defined as playing a critical role in the growth of the face and skull, with influence on the migration and condensation of mesenchymal cells (Table 5).34,126 The ventral region of the primitive nasal septum becomes apparent between E12.5 and E13.0 (Figure 6). 88 At E14.0, the anatomical structures may be distinguished as discrete entities, and the nasal septum can be identified as clusters of packed mesenchymal cells. At E13.0, the primitive nasal septum has a similar width to the rostral tongue. 88 The nasal septum grows down toward the rising palatal shelves at E14.0. 88 The morphogenesis and molecular mechanisms of secondary palate development have been fully described, including scanning electron micrographs and histology. 19 The rostral region of the nasal septum fuses with the primary palate at E14.5 while caudally the ventral part of the nasal septum is located above the palatal shelves. 88 Between E15.5 and E16.0, the cartilaginous nasal septum fuses in the midline with the rostral half of the palatal shelves and the primary palate (Figure 7). 88 The lateral sides of the lower region of the soft tissue within the nasal septum fuse with the soft tissues over the maxillary bones.
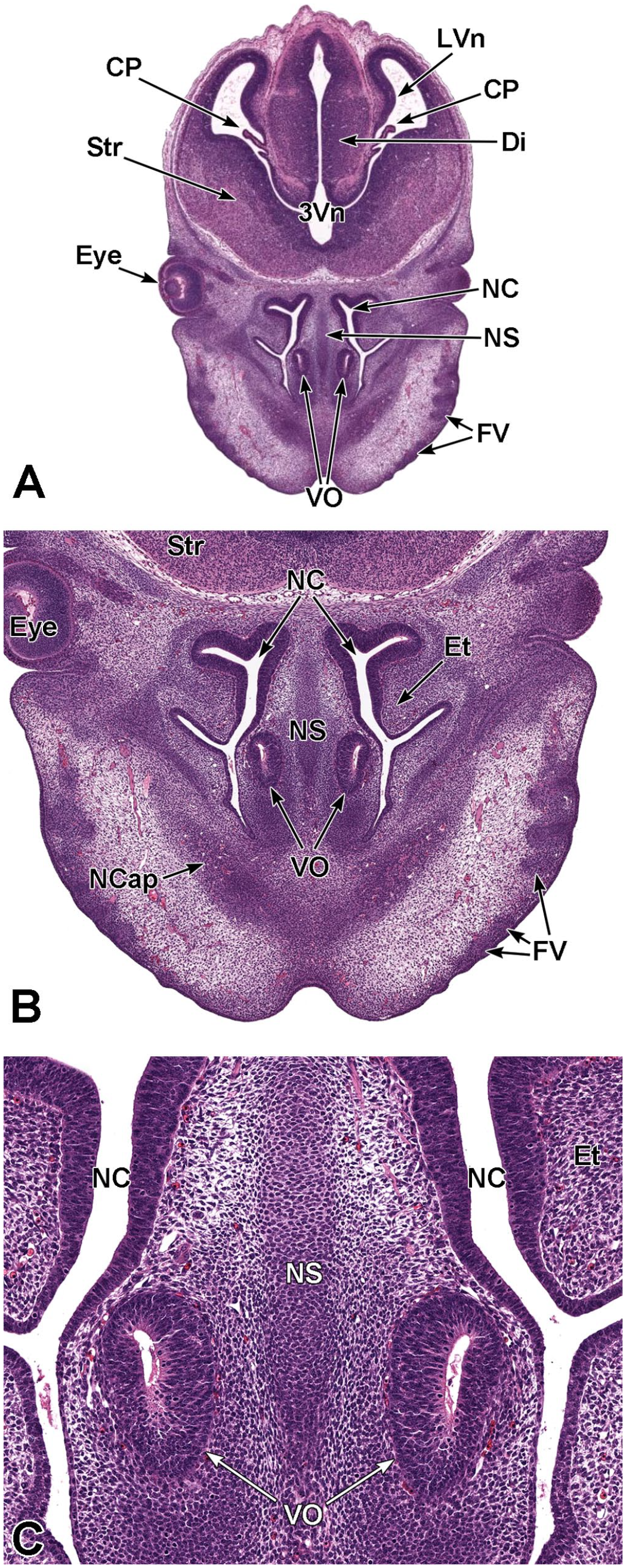
Transverse images illustrating the primitive nasal cavity, turbinates, septum, and associated structures at E13.5. Panels A (1×), B (3.2×), and C (10.5×) demonstrate the locations of the primitive nasal cavity (NC), nasal capsule (NCap), cartilage primordium of the nasal septum (NS), and the vomeronasal organ (VO). The expansion of the ethmoturbinates (Et) on the lateral nasal cavity wall is also shown. Other major landmarks include the third ventricle (3Vn); primordium of dorsal horn of the lateral ventricle (LVn); choroid plexus (CP); diencephalon (Di); striatum (Str); eye (Eye); and follicles of vibrissae (FV). (Note: Extra tissue at the bottom of panel A was removed for optimal viewing of anatomic features.) H&E.
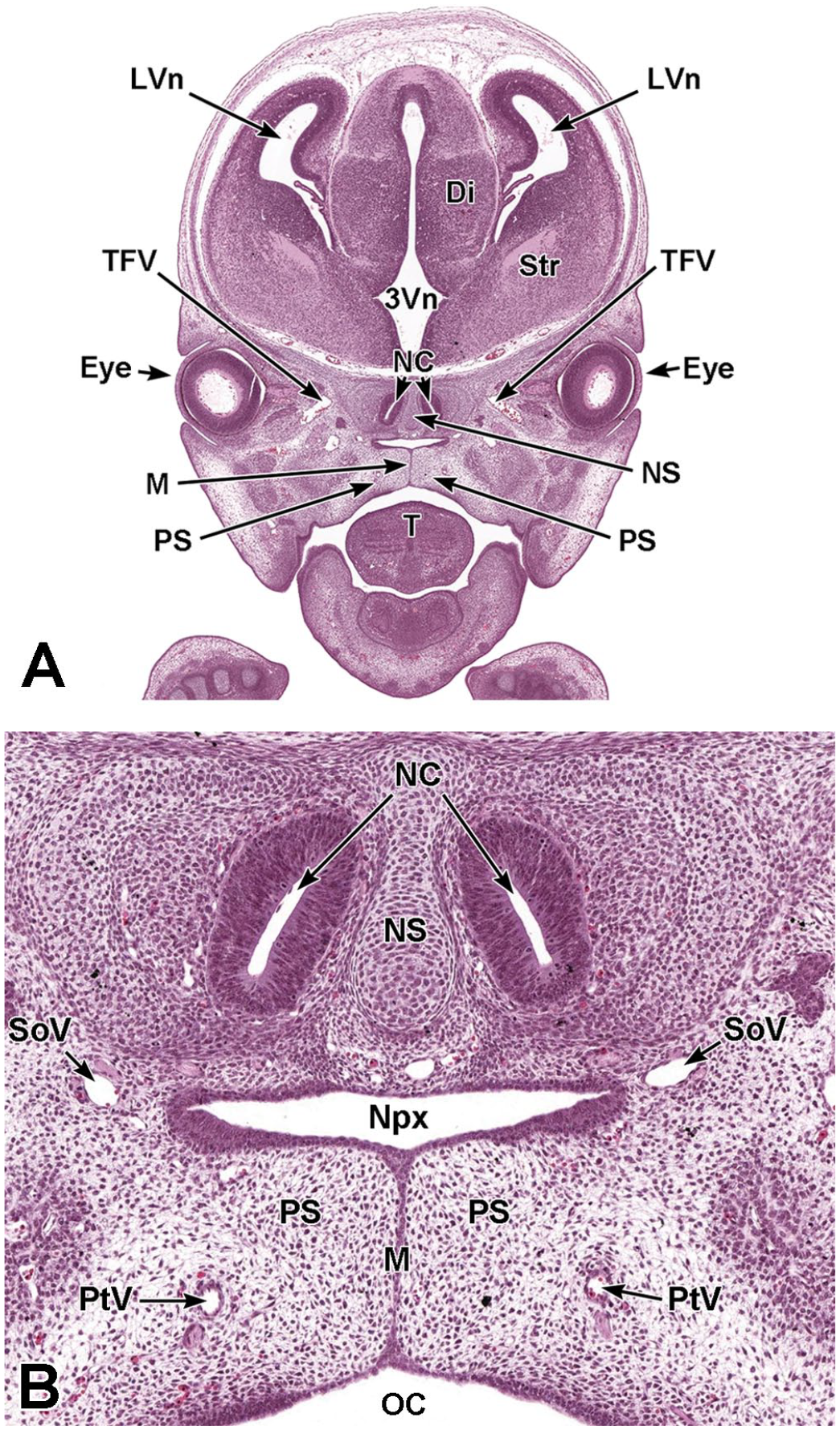
Coronal images illustrating nasal cavity formation at E15.5. Panels A (1.4×) and B (8.1×) illustrate the complete separation of the nasopharynx (Npx) from the oral cavity (OC) by the start of nasal septum (NS) fusion at the site of midline (M) apposition of the right and left and palatal shelves (PS). The right and left lateral ventricles (LVn); third ventricle (3Vn); diencephalon (Di); striatum (Str); transverse facial veins (TFV); right and left eyes (Eye); nasal cavity (NC); tongue (T); supraorbital veins (SoV); and right and left palatine vessels (PtV) are labeled for orientation. H&E.
The development of the nasal cavities and sinuses depends on craniofacial mesenchyme differentiation and morphogenesis of the facial primordia. At E12.0, the nasomaxillary complex consists of a uniform mass of undifferentiated cells. At E16.0, the parietotectal cartilage of the nasal capsule and ethmoturbinates and the hyaloid cartilage of the nasal septum are well organized cartilage plates (Figure 8). By E18.0, differentiated cartilaginous matrix is present in these structures. A nasal cross-section at E18.5 will reveal the brightly stained hyaline cartilage of the nasal septum, rostral paraseptal cartilage, and parietotectal cartilage of the nasal capsule (Figure 9).
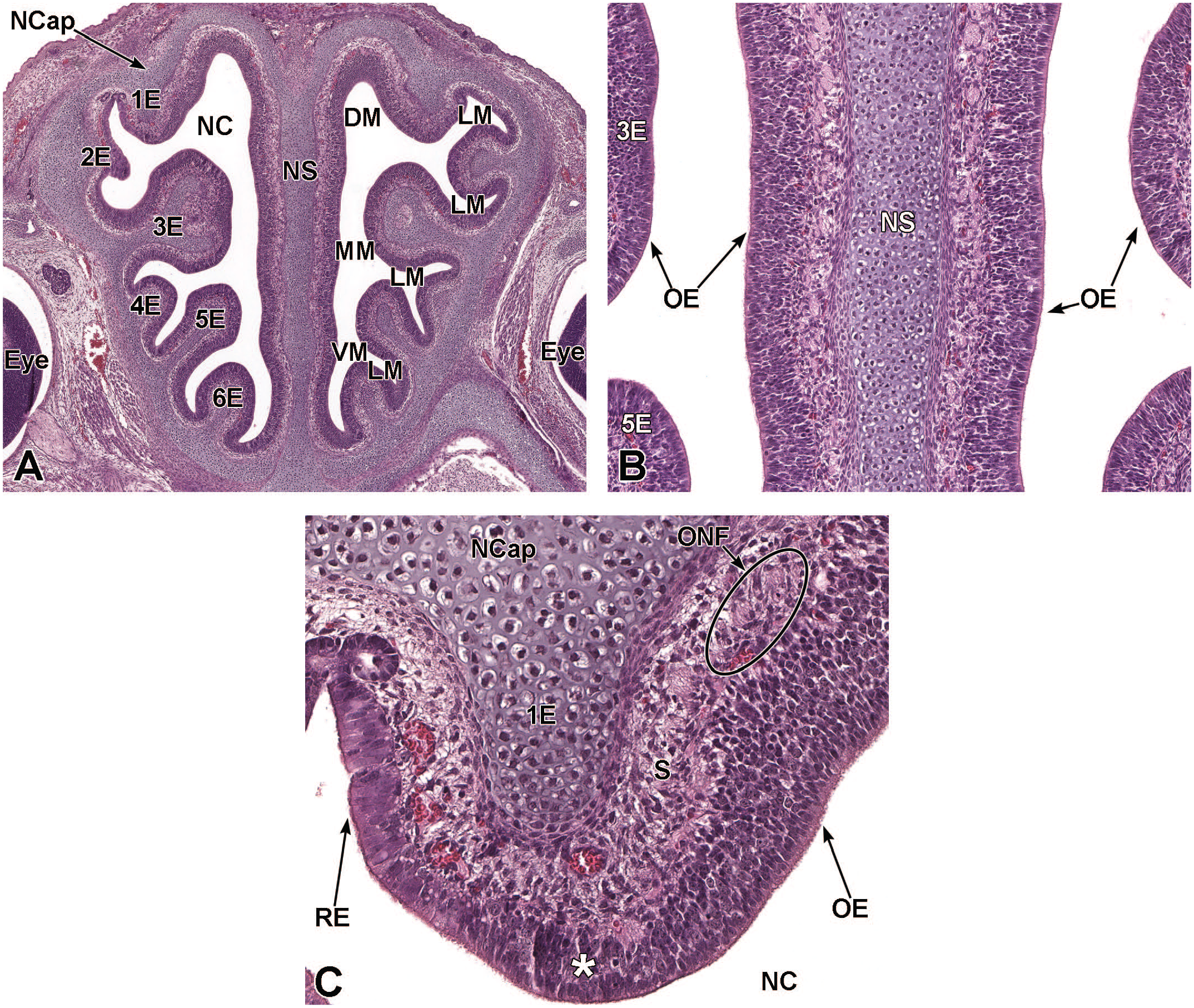
Coronal images of the developing nasal capsule, nasal septum, respiratory epithelium, and olfactory epithelium at E16.5. Panel A (5×) shows the hyaloid cartilage of the nasal septum (NS), as well as the parietotectal cartilage of the nasal capsule (NCap) and 6 ethmoturbinates (1E-6E). The narrow passages of the dorsal meatus (DM), middle meatus (MM), ventral meatus (VM), and lateral meatuses (LM) within the nasal cavity (NC) are indicated. Panel B (20×) is at the level of the middle ethmoturbinates (3E-5E) and illustrates the developing olfactory epithelium (OE) on either side of the nasal septum. Panel C (40×) is at the location of the first ethmoturbinate (1E) showing the developing olfactory and respiratory epithelium (RE). The transition between the respiratory and olfactory epithelium is indicated with an asterisk. The retinas of the right and left eyes (Eye); olfactory nerve fibers (ONF); and submucosa (S) are labeled for orientation. H&E.
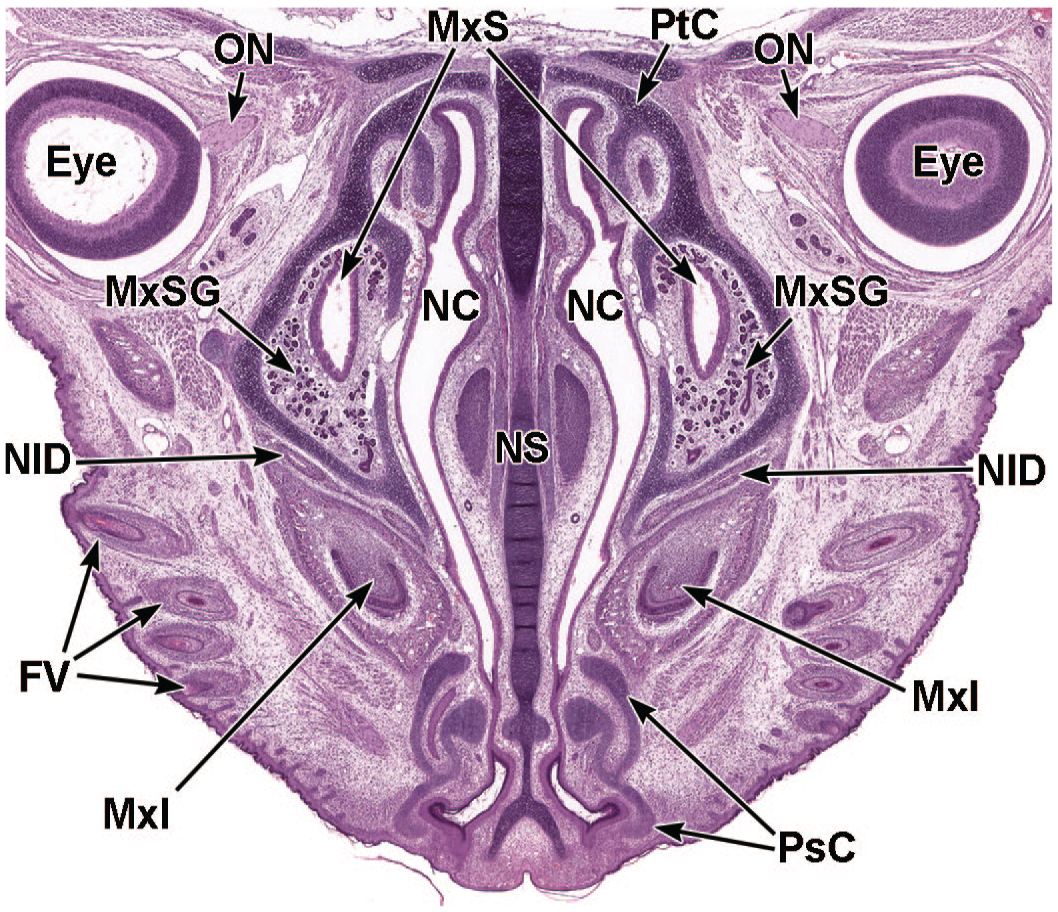
Coronal image illustrating the different cartilages of the nasal cavity at E18.5. This image (3.6×) shows the hyaline cartilage of the nasal septum (NS), the cranial paraseptal cartilage (PsC), and the parietotectal cartilage (PtC) of the nasal capsule and ethmoturbinates. The optic nerves (ON) corresponding to the right and left eyes (Eye); maxillary sinuses (MxS) with maxillary sinus glands (MxSG); nasal cavity (NC); nasolacrimal ducts (NlD); maxillary incisors (MxI); and follicles of vibrissae (FV) are labeled for orientation. H&E.
Palate
The nasopalatine duct (incisive foramen or anterior palatine foramen or nasopharyngeal duct) is an interosseus conduit through the rostral maxilla that connects the oral and nasal cavities, with the conduit opening located between the soft and hard palates. The primary palate is located rostral to the nasopalatine duct and is formed by the caudal expansion of the frontonasal process. 189 The secondary palate is located caudal to the nasopalatine duct and is formed by the fusion of the palatal shelves, which originate from the maxillary processes. 189 The development of the secondary palate involves vertical growth of the shelves initially followed in time with inward rotation (elevation) of their medial edges leading to eventual fusion at the midline.
Palatal outgrowths are first detectable by E11.5.19,189 Between E12.5 and E13.0, rugae (surface wrinkles) run across the full width of the palatal shelves. 88 The secondary palate begins to develop from the lateral edges of the maxillary processes at E12.5 while the palatal shelves remain in the vertical position.27,100,164 These shelves are derived from neural crest cells from the rostral neural tube and are surrounded by pharyngeal ectoderm-derived epithelium.26,100 By E13.0, the palatal shelves are separated by formation of the tongue, which bulges upward to fill the space between the shelves. 88 Also at this time, the caudal two thirds of the medial region of the palatal shelves are directed vertically while the rostral one third has a rounded shape and is farther from the dorsal and lateral regions of the tongue. 88 The tongue flattens and clears the path for the vertically oriented palatal shelves to rotate into the horizontal position (Figure 10).19,161 The medial borders of the caudal region of the palatal shelves continue to grow vertically and elevate above the dorsal tongue surface, achieving a horizontal position around E14.0.88,100 By E14.5, the edges of the palatal shelves rest adjacent to each other, thereby forming the secondary palate that separates the oral cavity below from the nasopharyngeal cavity above. 61
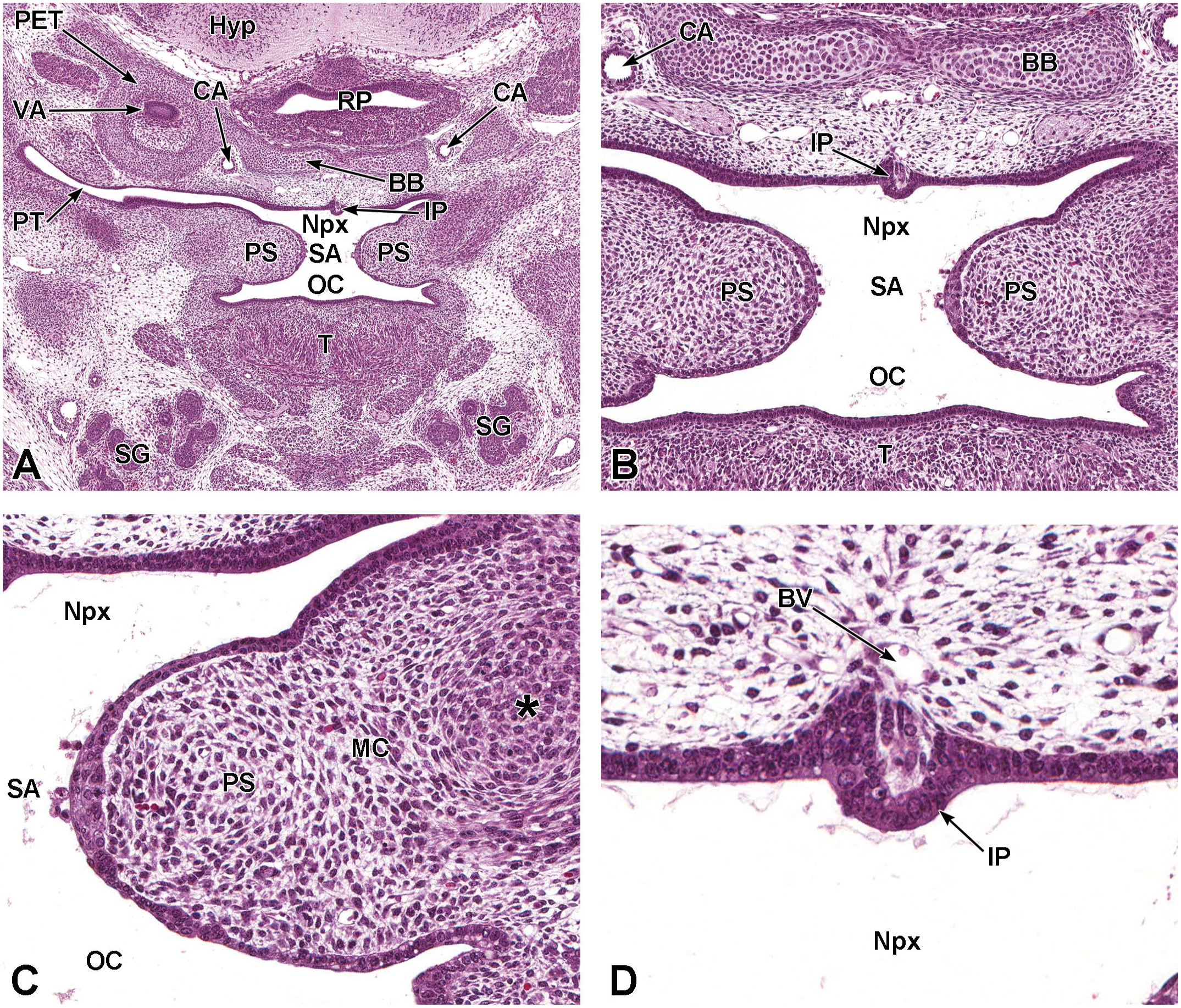
Coronal images of the palatal shelves and incisive papilla at E13.5. Panels A (8×) and B (20×) depict the right and left palatal shelves (PS) prior to fusion. The space between the palatal shelves is the future site of apposition (SA) and lies above the oral cavity (OC) and below the nasopharynx (Npx). The incisive papilla (IP) is a cartilaginous projection on the hard palate, caudal to the upper incisors. Panel C (40×) depicts one of the two palatal shelves, which is lined by epithelial cells and consisting of loosely arranged mesenchymal cells (MC) on the medial aspect and more condensed mesenchymal cells (asterisk) on the lateral aspect. Panel D (40×) is a higher magnification of the incisive papilla, which is lined by cuboidal epithelium. The caudal region of the hypothalamus (Hyp); Rathke’s pouch (RP [pituitary gland precursor]); cartilage primordium of the petrous temporal bone (PET) encircling a small portion of the vestibular apparatus (VA); cartilage primordium of the basisphenoid bone (BB); the right and left internal carotid arteries (CA); pharyngo-tympanic (Eustachian) tube (PT); root of the tongue (T); the right and left salivary glands (SG); and blood vessel (BV) are labeled for orientation. H&E.
Upon elevation, the palatal shelves continue to grow in the horizontal direction, resulting in contact between the medial edge epithelium of the two shelves.19,189 The opposing medial edge epithelia make initial contact approximately a third of the way along the palate (relative to the nares), after which fusion continues both rostrally and caudally to form the midline epithelial seam (Figure 7). 189 During fusion of the shelves, a “Y”-shaped hole in the cranial palate near the nasopalatine duct represents the boundary between the primary and secondary palates 189 ; this gap becomes smaller after the formation of the midline epithelial seam and disappears by E16.5. 189 By E15.5, the midline epithelial seam has degenerated, resulting in intermingled growth of the mesenchyme across the midline and completion of palate closure and separation of the oropharynx and nasopharynx.88,189 At E16.0, nine horizontal rugae on the ventral surface of the palate become visible; the primary and secondary palate meet at the nasopalatine duct, and the primary palate fuses with the rostral borders of the palatal shelves near the region of the initial site of fusion. 88 After E16.5, the nerves and blood vessels are located in the medial region near the oral side of the palatal shelves. 63 Palatal fusion is complete by E17.0. 19
Nasal Turbinates
In the adult mouse, bilaterally symmetrical nasal turbinates (also called nasal conchae) extend horizontally along the lateral walls of the nasal passage and are divided into the nasal (naso-), maxillary, and ethmoid turbinates.28,65 Each turbinate has a central bony trabeculum.28,65 The nasal and maxillary turbinates are simple and slightly scrolled and are lined by respiratory epithelium, whereas the ethmoid turbinates form complex scrolls and are lined by olfactory neuroepithelium.4,65 A layer of mesenchyme, the lamina propria, lies between the epithelium and underlying trabecula. 187 Turbinate scrolling subdivides the two halves of the nasal cavity into dorsal, middle, ventral, and lateral meatuses, where each meatus is an air passage that channels a discrete stream of air. 72 The air streams exhibit variable turbulence based on the complexity of the turbinates; the more intricate scrolling of the ethmoid turbinates enhances contact of the air and its dispersed odorants with the olfactory neuroepithelium.
Turbinate development begins at E12.5 as epithelial shelves on the lateral nasal cavity walls near the rostral region of the nasopharynx. 187 The process begins with epithelial budding followed by mesenchymal proliferation and condensation and then chondrocyte differentiation. 187 The timing of this process varies between turbinates but begins at around E12.5-E14.5. 187 Between E14.0 and E15.0, the mesenchymal shelves of the turbinates contain a cartilaginous center that begins to ossify to form the turbinate bones, and the surrounding epithelium begins to fold, causing the mucosal surface area to increase (Figure 11). The nasal turbinate chondrocytes continue to undergo hypertrophy throughout early postnatal development and are replaced by bone by P7, 89 which is described further below in the postnatal development section.
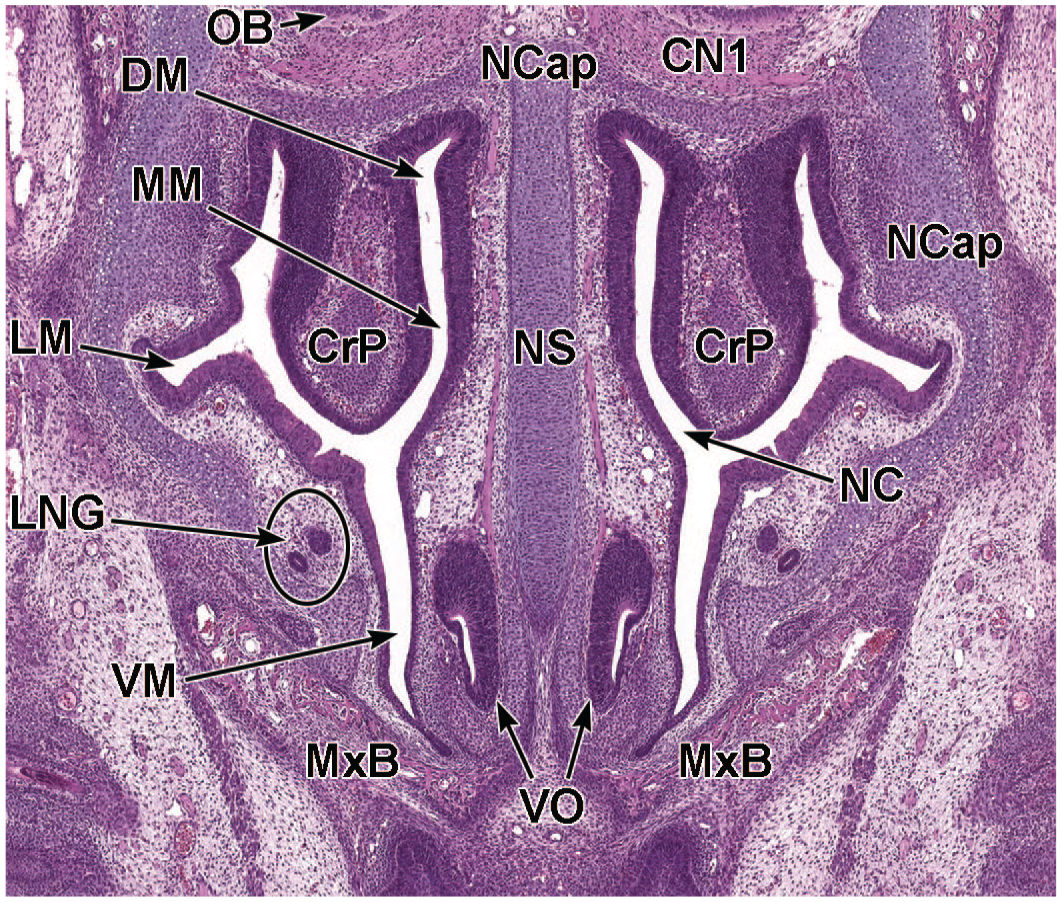
Transverse image of nasal turbinate ossification and folding at E15.5. This image (7.5×) illustrates the mesenchymal shelves of the turbinates, characterized by cartilaginous primordia (CrP) that will eventually ossify to form bone. The nasal cavity (NC) with dorsal meatus (DM), middle meatus (MM), lateral meatus (LM), and ventral meatus (VM), as well as the hyaline cartilages forming the nasal septum (NS) and nasal capsule (NCap) that frame the nasal passages, are shown. Lateral nasal glands (LNG); the vomeronasal organ (VO); and the right and left developing maxillary bones (MxB) are labeled for orientation. The olfactory nerve (cranial nerve 1 [CN1]) is also denoted, as is the location of the edge of the olfactory bulb (OB). H&E.
Nasopharyngeal Duct
In the adult mouse, the nasopharyngeal duct is the passage that connects the nasal cavity to the nasopharynx. This quadrilateral tube is visible ventral to the caudal nasal septum and dorsal to the secondary palate. It is separated from the chambers containing the ethmoid turbinate scrolls by thin walls of soft tissue lined by ciliated respiratory epithelium covering loose mesenchyme. The duct is visible by mid-gestation (E12.5-E15.0). Formation of nasal-associated lymphoid tissues (NALT, also called nasopharyngeal- or nasopharynx-associated lymphoid tissue) bilaterally in the lamina propria of this region occurs postnatally.
Nasal Mucosa
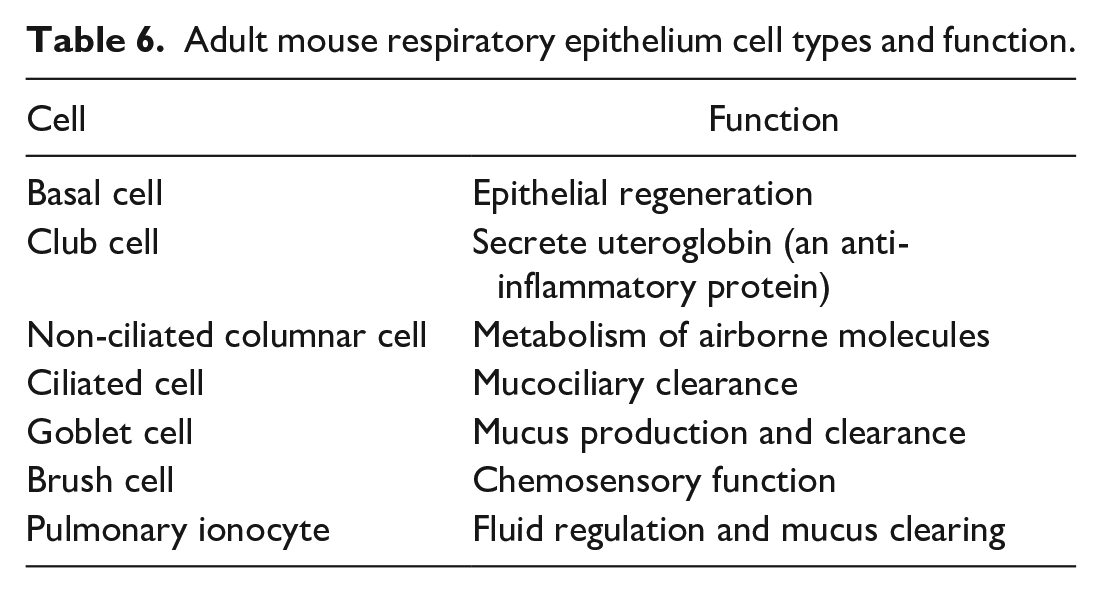
Various parts of the nasal passages are lined by different types of epithelium. The nature of the epithelium is dictated by the region-specific function of that portion of the nasal cavity. Table 6 describes the adult mouse respiratory epithelium cell types and functions.
Adult mouse respiratory epithelium cell types and function.
Stratified squamous epithelium
The nasal atrium and vestibule are small domains just inside the external naris that are characterized by a thin layer of keratinized, stratified squamous epithelium. 4 The function of this epithelial type is to protect the nasal passages from inspired objects (e.g., grit particles, hair fragments) and high levels of chemicals in ambient air.
Respiratory epithelium
In the adult mouse, this epithelium lines about half of the mouse airway, covering most of the nasoturbinates, maxilloturbinates, nasal septum, segments of the rostral and ventral ethmoid turbinates, and the nasal sinuses.66,132 This layer is one to two cells thick and is comprised mainly of ciliated, simple cuboidal to pseudocolumnar cells intermingled with various cells possessing specialized functions. The main function of this epithelial type is to move nasal fluids and mucus caudally through the nasal passages into the pharynx, where it may be expectorated or swallowed.
The development of the murine respiratory epithelium has not been well-studied. Respiratory epithelium begins development at about E12.5 (Figure 12) and, at later stages, can be seen lining the nasal cavity (Figures 8 and 13-15). By E18.5, pseudocolumnar ciliated respiratory epithelium can be identified lining the lateral and ventral meatuses of the ethmoturbinates (Figure 16). 72
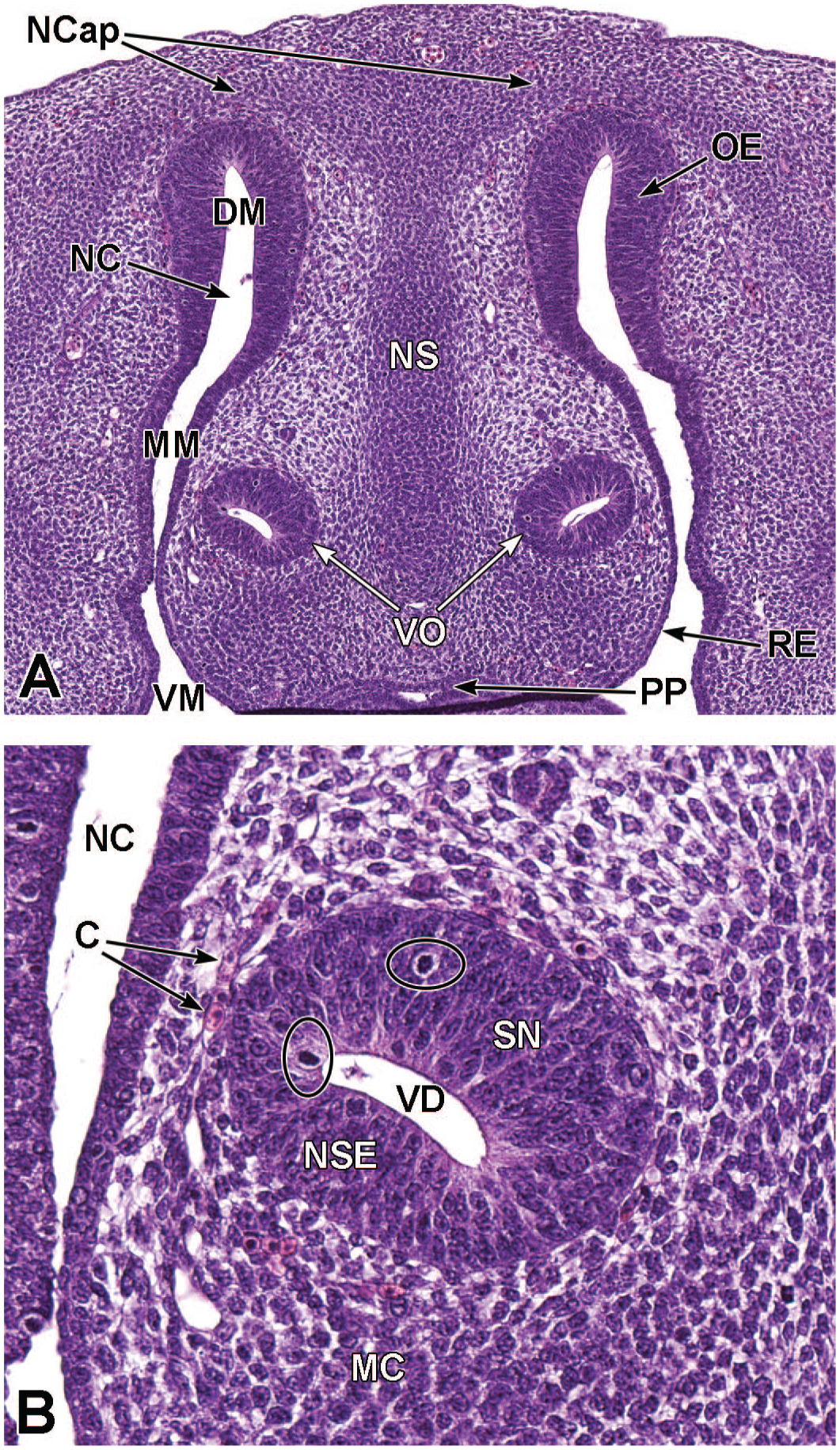
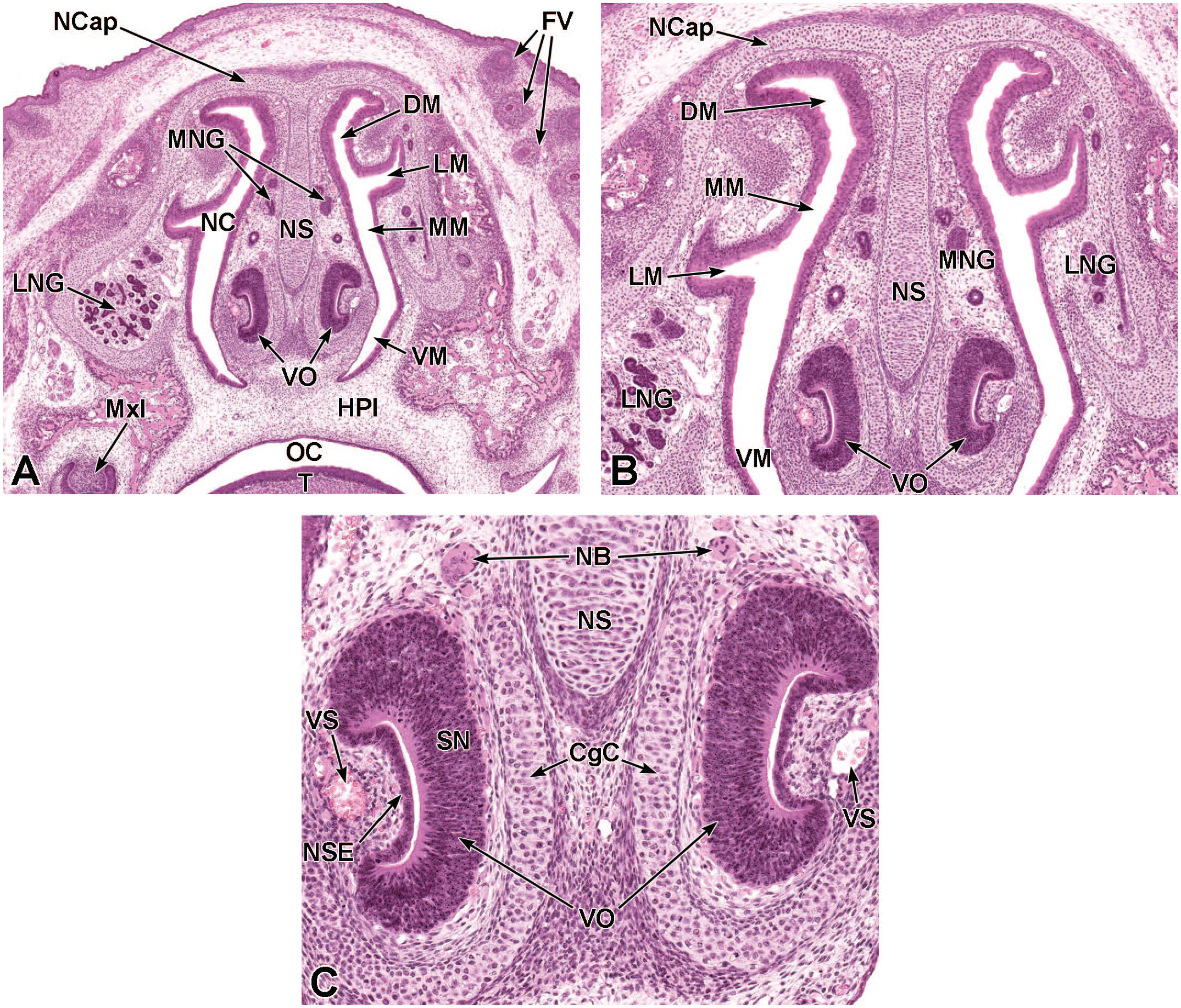
Transverse images of the developing nasal cavity at E12.5. Panel A (10×) shows that within the nasal cavity (NC), olfactory epithelium (OE) lines the dorsal meatus (DM) while respiratory epithelium (RE) lines the middle and ventral meatuses (MM and VM, respectively). As the vomeronasal organ (VO) splits, it becomes located on either side of the nasal septum (NS). Panel B (59x) illustrates the central vomeronasal duct (VD), pseudostratified sensory neuroepithelium (SN) lining the medial wall, and non-sensory ciliated epithelium (NSE) lining the lateral wall. Mitotic figures are indicated by circles. The VO is surrounded by a thin layer of connective tissue containing capillaries (C) and a thick layer of mesenchymal cells (MC). The nasal capsule (NCap) and primary palate (PP) are labeled for orientation. H&E.
Transverse images of the developing respiratory and olfactory epithelium at E14.5. Panel A (4.6×) depicts the location of the olfactory epithelium (OE) lining the medial and lateral walls of the caudal nasal cavity (NC) as well as the respiratory epithelium (RE) lining the medial and lateral walls of the rostral nasal cavity. Panel B (18.9×) shows the contrast between the thick, pseudostratified olfactory epithelium; the thin, pseudocolumnar respiratory epithelium; and the transition between the two (indicated by asterisks). The hyaline cartilage of the nasal septum (NS); nares (N); and submucosa (S) are labeled for orientation. (Note: Extraneous tissue was removed from the lower left field of panel A for optimal viewing of anatomic features.) H&E.
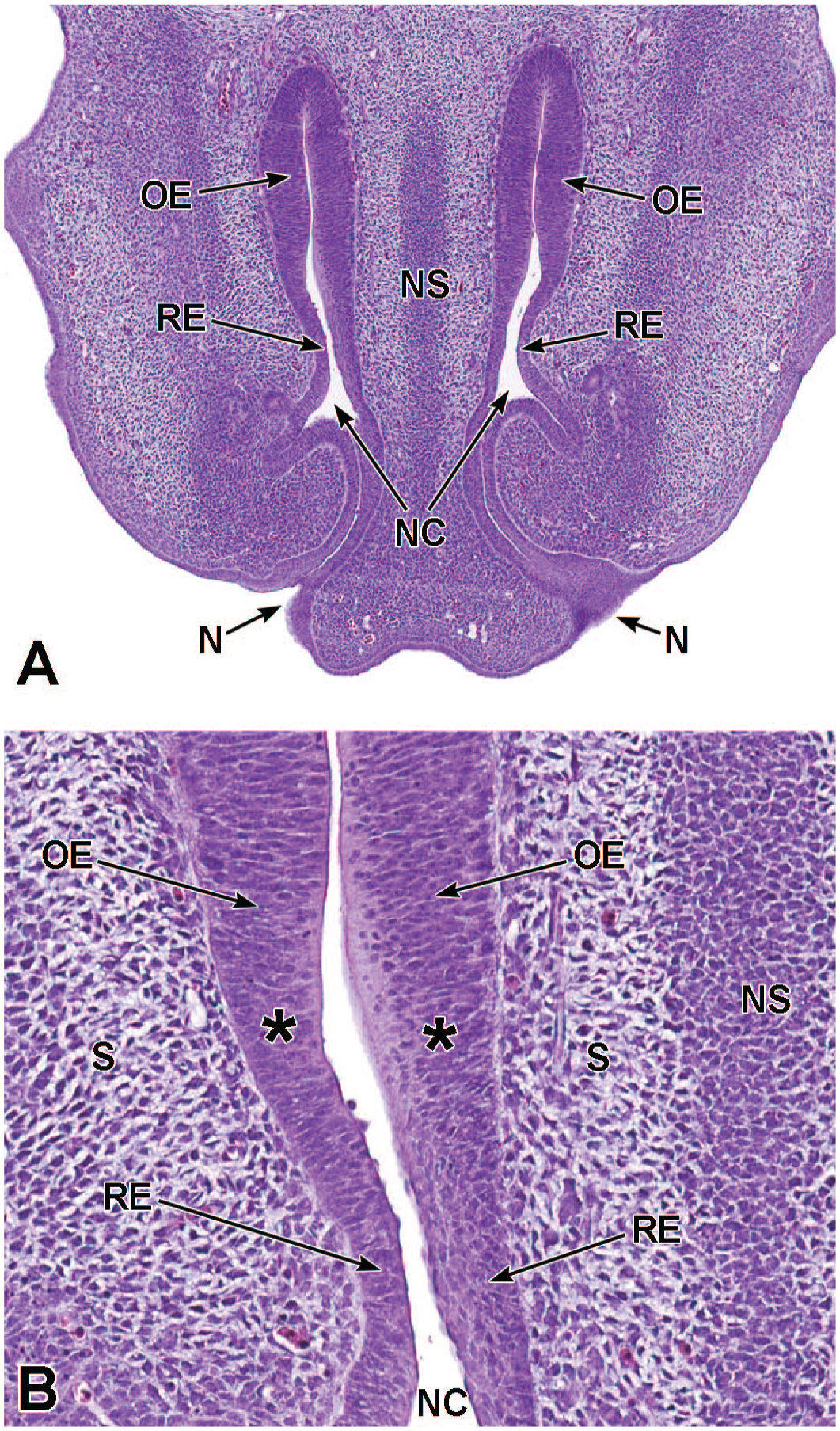
Transverse images of the developing nasal cavity at E15.5. Panel A (10×) shows the olfactory epithelium (OE) lining the dorsal and lateral meatuses (DM and LM, respectively), the respiratory epithelium (RE) lining the lining the middle and ventral meatuses (MM and VM, respectively), as well as the expanding vomeronasal organ (VO) occupying a large portion of the rostroventral nasal septum (NS). Panel B (40×) shows the VO consisting of thin non-sensory epithelium (NSE) along the lateral wall and thicker pseudostratified sensory neuroepithelium (SN) lining the medial wall; the organ is partially surrounded by a cartilaginous capsule (CgC). The connective tissue surrounding the VO consists of capillaries (C) and a vascular sinus (VS) on the lateral aspect. The nasal cavity (NC); nasal capsule (NCap); right and left maxillary bones (MxB); and lateral nasal glands (LNG) are labeled for orientation. Transitions from OE to RE are indicated by asterisks. H&E.
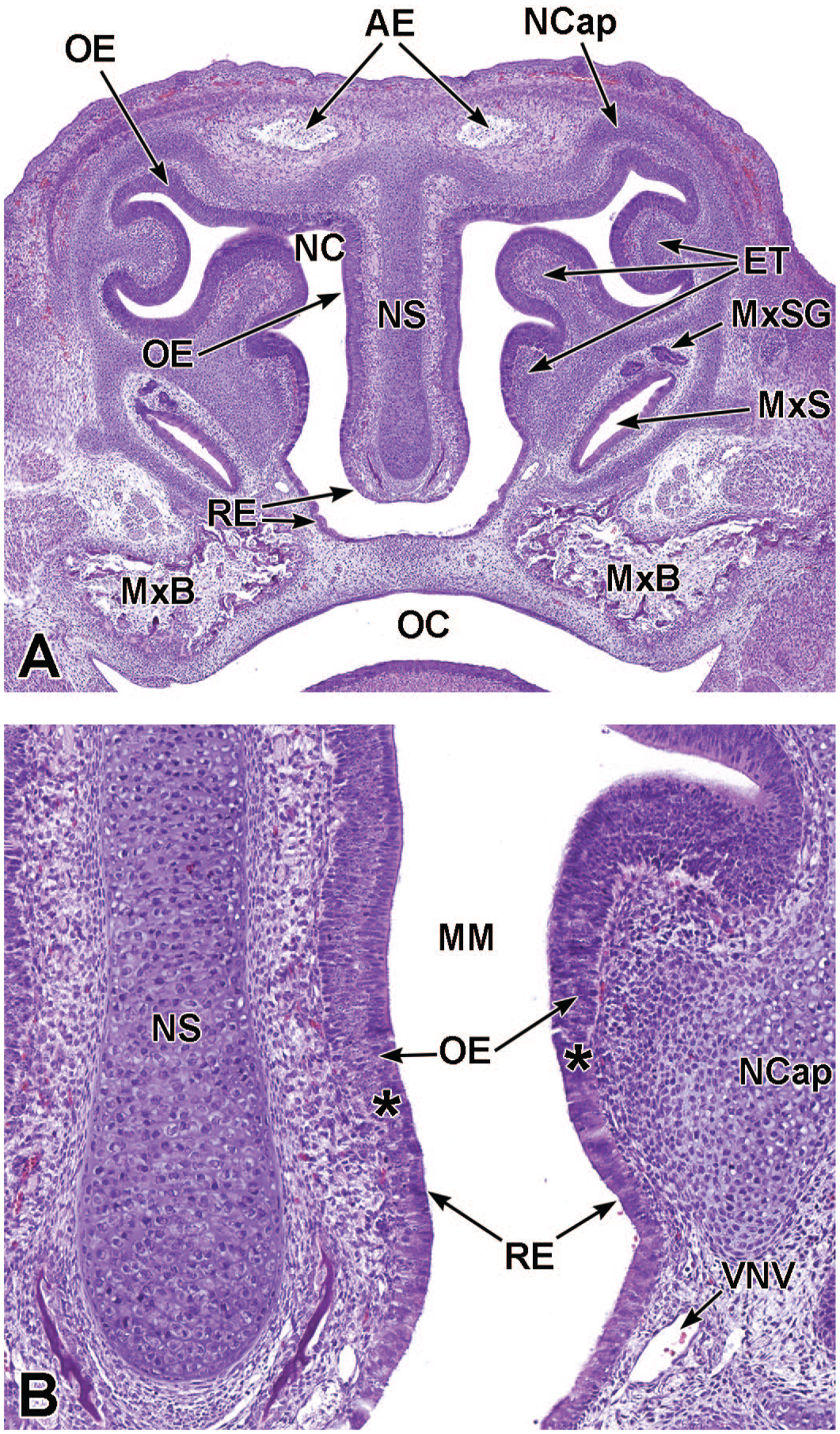
Coronal images of the caudal nasal cavity at E17.5. Panel A (5.2×) shows the hyaline cartilage of the nasal septum (NS), respiratory epithelium (RE), and olfactory epithelium (OE) lining the nasal cavity (NC), parietotectal cartilage of the nasal capsule (NCap) and ethmoturbinates (Et), and the maxillary sinus glands (MxSG) of the maxillary sinuses (MxS). Panel B (20×) shows the transition (asterisks) of OE to RE in the region of the middle meatus (MM). The rostral (anterior) extremities (AE) of the right and left olfactory bulbs; early ossification of the right and left maxillary bones (MxB); oral cavity (OC); and ventral nasal vein (VNV) are labeled for orientation. H&E.
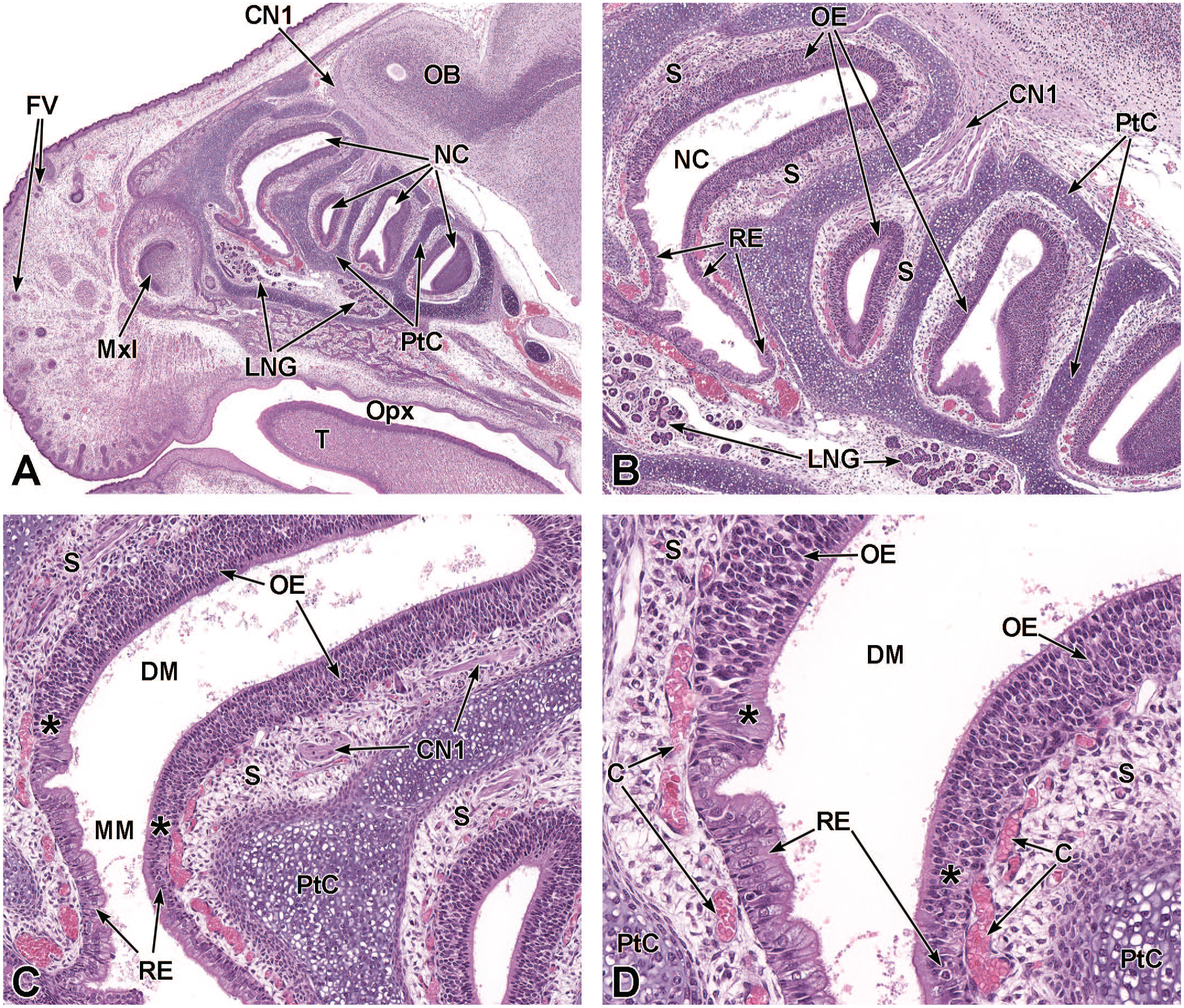
Sagittal images of the ethmoturbinates at E18.5. Panel A (4×) shows the nasal cavity (NC) and parietotectal cartilage (PtC) supporting the turbinates at low magnification for orientation. Panel B (10×) shows the locations of the thick pseudostratified olfactory epithelium (OE) and shorter pseudocolumnar respiratory epithelium (RE) lining the nasal cavity. Panel C (20×) demonstrates the transition (asterisks) from OE to RE at the point where the dorsal meatus (DM) meets the middle meatus (MM). Panel D (40×) highlights the ciliated RE and pseudostratified OE as well as the blood-filled capillaries (C) amid the loose connective tissue of the submucosa (S) lining an ethmoturbinate. The olfactory nerve (cranial nerve 1 [CN1]); olfactory bulb (OB); maxillary incisor (MxI); lateral nasal glands (LNG); oropharynx (Opx); tongue (T); and follicles of vibrissae (FV) are also indicated. H&E.
Olfactory neuroepithelium
In the adult mouse, olfactory neuroepithelium (often designated as olfactory epithelium) lines the ethmoid turbinates, extending along the dorsal wall of the rostral nasal cavity to line the dorsal meatus. This pseudostratified mucosal layer consists of specialized bipolar olfactory neurons, sustentacular (glial-like support) cells, basal (stem) cells, and the necks of mucous (Bowman’s) glands. 72 The lamina propria contains numerous Bowman’s glands, which exhibit a branched tubuloalveolar organization; many capillaries and lymphatic vessels83,72; and myriad nerve fibers (derived from olfactory neurons) that pass through the cribriform plate as the olfactory nerve (cranial nerve I) to innervate the main olfactory bulbs of the brain. In-depth reviews of the olfactory neuroepithelial anatomy based on an exhaustive light and electron microscopy study of murine olfactory neuroepithelium development are available elsewhere.141,165
Olfactory neuroepithelium originates from both the olfactory placode (ectoderm) and neural crest cells (Table 5). Its initial presence at approximately E10.0 is inferred based on its location, with visible evidence of its differentiation pathway evident by approximately E10.5 as gradual thickening of the nasal mucosa in the dorsal meatus and over the nascent ethmoid turbinates. Olfactory neuroepithelium development continues at E11.0 when some epithelial cells (i.e., nascent neurons) develop dendritic processes that extend to the epithelial surface, ending in irregular swellings. 39 At this stage, these dendrites are more numerous in the deeper parts of the deep (or olfactory) portion of the nasal pit. 165 At E12.0, these dendrites occur most frequently in the epithelium of the recesses in the most caudal region of the nasal cavity. 38 At this time, nuclei differentiation and layering becomes apparent within the olfactory neuroepithelium. 165 Centrioles, mitochondria, small coated vesicles, and microtubules can be seen oriented longitudinally within the terminal swellings of the dendrites using transmission electron microscopy. 38 Cilia begin to develop on the surface of the terminal swellings at E12.0 and continue developing until E16.0. 38 After E12.0, olfactory dendrites and receptor cell bodies continue to proliferate. 38 The developing olfactory epithelium can be identified lining the nasal cavity at E12.5 (Figure 12), E14.5 (Figure 13), E15.5 (Figure 14), E16.5 (Figure 8), E17.5 (Figure 15), and E18.5 (Figure 16). At E17.0, neuroepithelial cell bodies hosting the dendrites contain proliferating granular and agranular endoplasmic reticulum. 38 The nuclei of these cells are concentrated in the middle regions of the pseudostratified epithelium. In contrast, cells with nuclei located at the surface of the olfactory neuroepithelium develop into sustentacular cells. 38 Basal cells within the basal cell layer are indistinguishable from the surrounding cells until 2 weeks after birth at which time they develop dense membranes. 38 Also at E17.0, Bowman’s glands are distinguishable as small subepithelial clumps of cells with vesicular nuclei growing from the base of the olfactory neuroepithelium into the underlying mesenchyme. 39 At this point, the glands appear small and tubular-shaped, but they increase in size and number during the first week after birth. 39
Olfactory Subsystems
The main olfactory sense organ is the olfactory neuroepithelium located in the caudodorsal nasal cavity, lining the dorsal meatus and ethmoid turbinates. The three principal olfactory subsystems in rodents are the vomeronasal organ, Grueneberg (also Grüneberg or Grünenberg) ganglion, and the septal organ of Masera (SOM). The vomeronasal organ and SOM occur in other mammals, including primates, whereas the Grueneberg ganglion (GG) is specific to rodents. Figure 17 is a diagrammatic illustration of the olfactory neural tracts arising within the nasal cavity of the mouse.
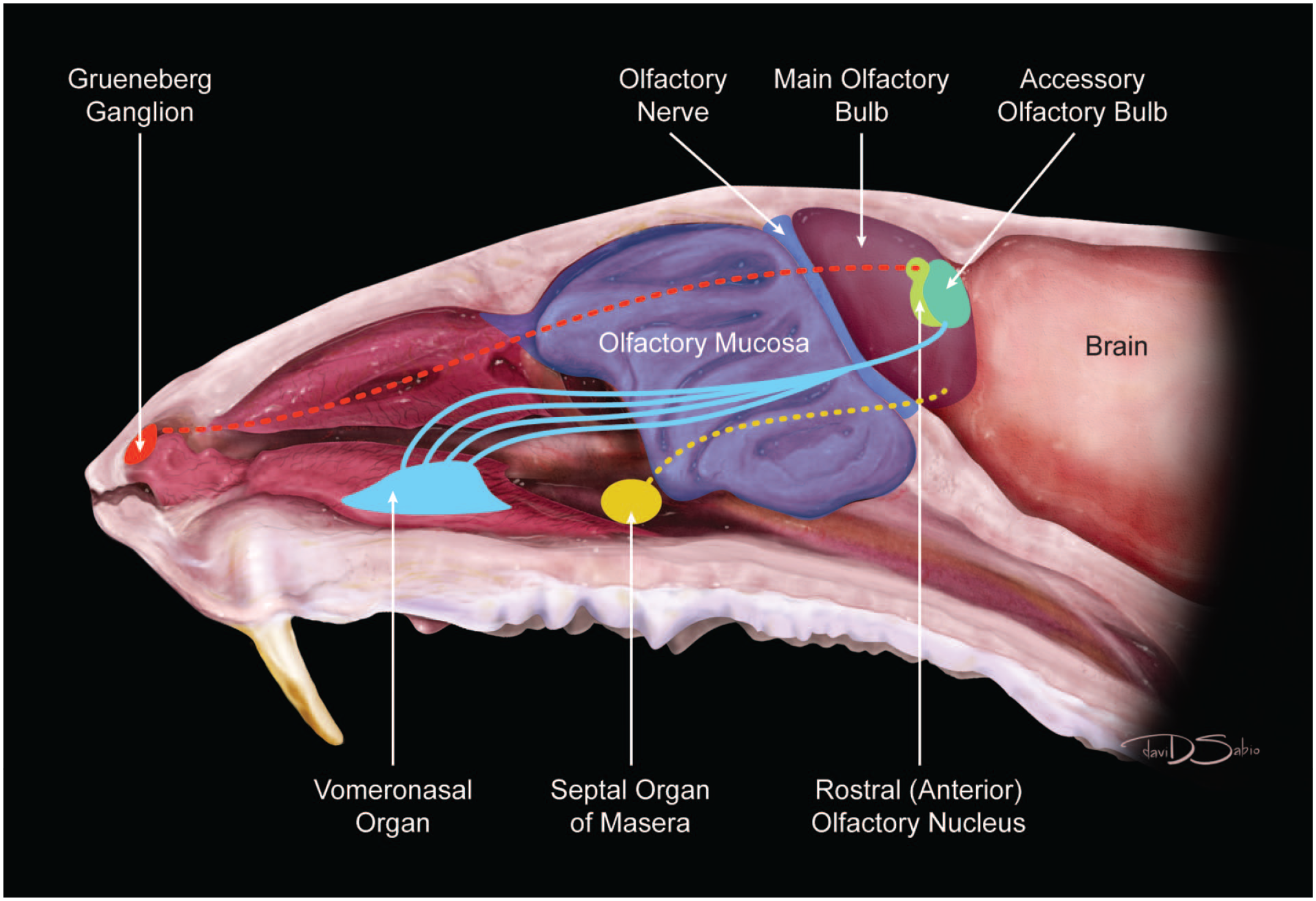
Diagrammatic illustration of the olfactory neural tracts arising from the nasal cavity of the mouse. Sensory neurons of the olfactory mucosa lining the ethmoturbinates as well as the neurons of the septal organ of Masera connect to the main olfactory bulb. Sensory neurons of the Grueneberg ganglion project to the rostral (anterior) olfactory nucleus. The vomeronasal organ contains many neural connections to the accessory olfactory bulb.
Vomeronasal organ
The function of the bilaterally symmetrical vomeronasal organ (VO, also called Jacobson’s organ) is to detect pheromones and other semiochemicals (i.e., substances [“pheromones”] released by an organism that affect the activities of other individuals) as an aid in the regulation of innate social and sexual behaviors. 123 The lumen of the vomeronasal duct communicates rostrally with the nasopalatine duct (also called the incisive canal), thereby connecting the VO to the nasal cavity and allowing sampling of chemical stimuli from the external world (Figure 5). Vomeronasal sensory neurons in the VO reside in a neuroepithelium and project axons to the accessory olfactory bulbs, embedded along the caudodorsal margins of the main olfactory bulbs (Figure 17), where they form synapses with principal neurons known as mitral cells. In the adult mouse, the paired, crescent-shaped diverticula of the VO can be viewed in the ventral nasal septum in cross-section. Sensory neuroepithelium lines the medial wall while non-sensory ciliated columnar epithelium lines the lateral wall of the VO diverticula.
Vomeronasal olfactory neurogenesis occurs in two phases: primary and established neurogenesis.87,79,181 Primary neurogenesis begins between E10.0 and E10.5 when the olfactory (nasal) placode invaginates to form the nasal pit (Figure 18). 87 Between E10.5 and E11.5, migratory neurons travel from the nasal pit toward the brain. 87 These neurons within this migratory mass eventually move from the nasal pit to the accessory olfactory bulb. 87 Between E11.0 and E11.5, the primordia of the VO appear as thickenings (known as the vomeronasal placode) in the epithelium of the medial wall of the nasal pit (Figure 19); these thickenings grow caudally and dorsally while continuing to invaginate to form the vomeronasal groove.39,56,72,87 The tissue of the groove fuses to form an epithelial-lined tube that opens rostrally into the main nasal pit. 39 This groove forms the primitive VO, which eventually becomes separated from the nasal pit except for a ductular connection. 87 At this time, axons derived from primitive VO neurons are present at the base of the vomeronasal epithelium and extend into the cartilage of the nasal septum. 39 At E12.0, nerve fibers can be seen at the base of the vomeronasal epithelium and extending along the nasal septum cartilage. 39 Also at this time, the neuroepithelium lining the medial and ventral regions of the VO have dendritic processes that terminate at the epithelial surface. 39 This neuroepithelium eventually develops into the sensory epithelium within the VO. 39
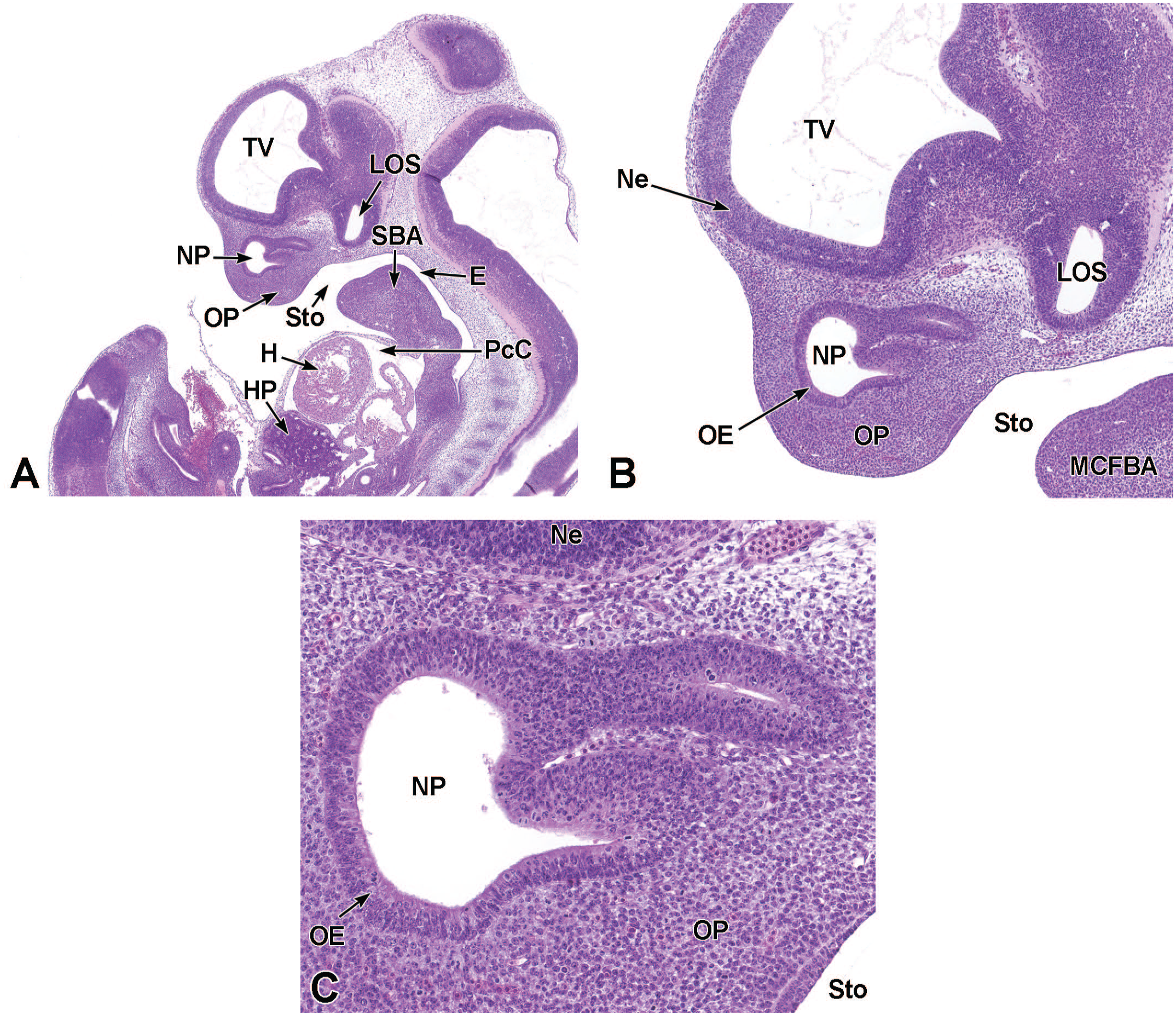
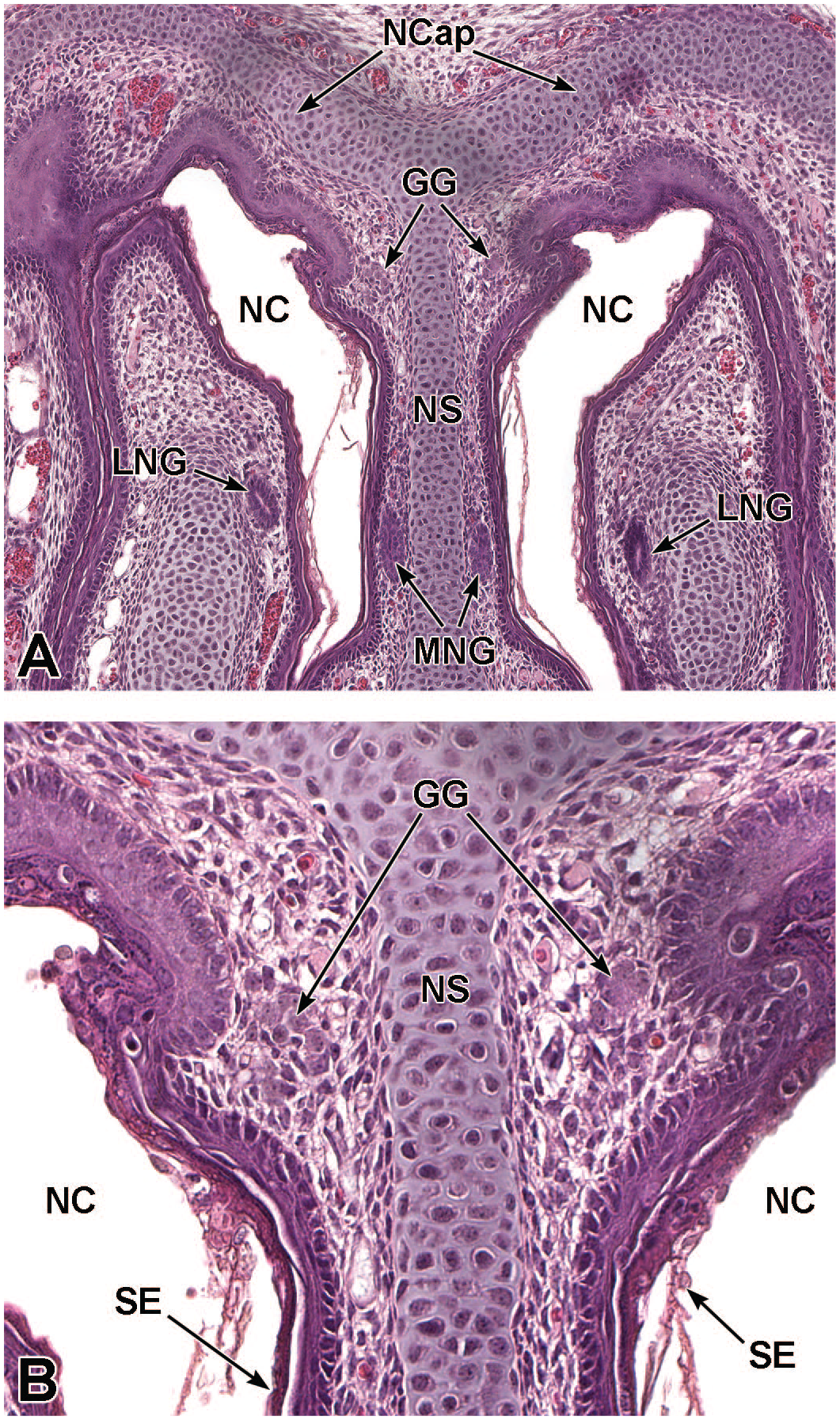
Sagittal images illustrating early development of the olfactory region of the nasal cavity at E11.5. Panel A (4×) demonstrates the invagination of the olfactory (nasal) placode (OP) to form the nasal pit (NP). Panels B (10×) and C (30×) show initial formation of the pseudostratified olfactory epithelium (OE) lining the nasal pit. The telencephalic vesicle (TV) of the forebrain with lining neuroepithelium (Ne); lumen of the optic stalk (LOS); oral cavity (stomodeum [Sto]); second branchial arch (SBA); mandibular component of the first branchial arch (MCFBA); esophagus (E); pericardial cavity (PcC); heart (H); and hepatic primordium (HP) are labeled for orientation. (Note: A small bit of artifact tissue to the left of the OP was removed from panels A and B for optimal viewing of anatomic features.) H&E.
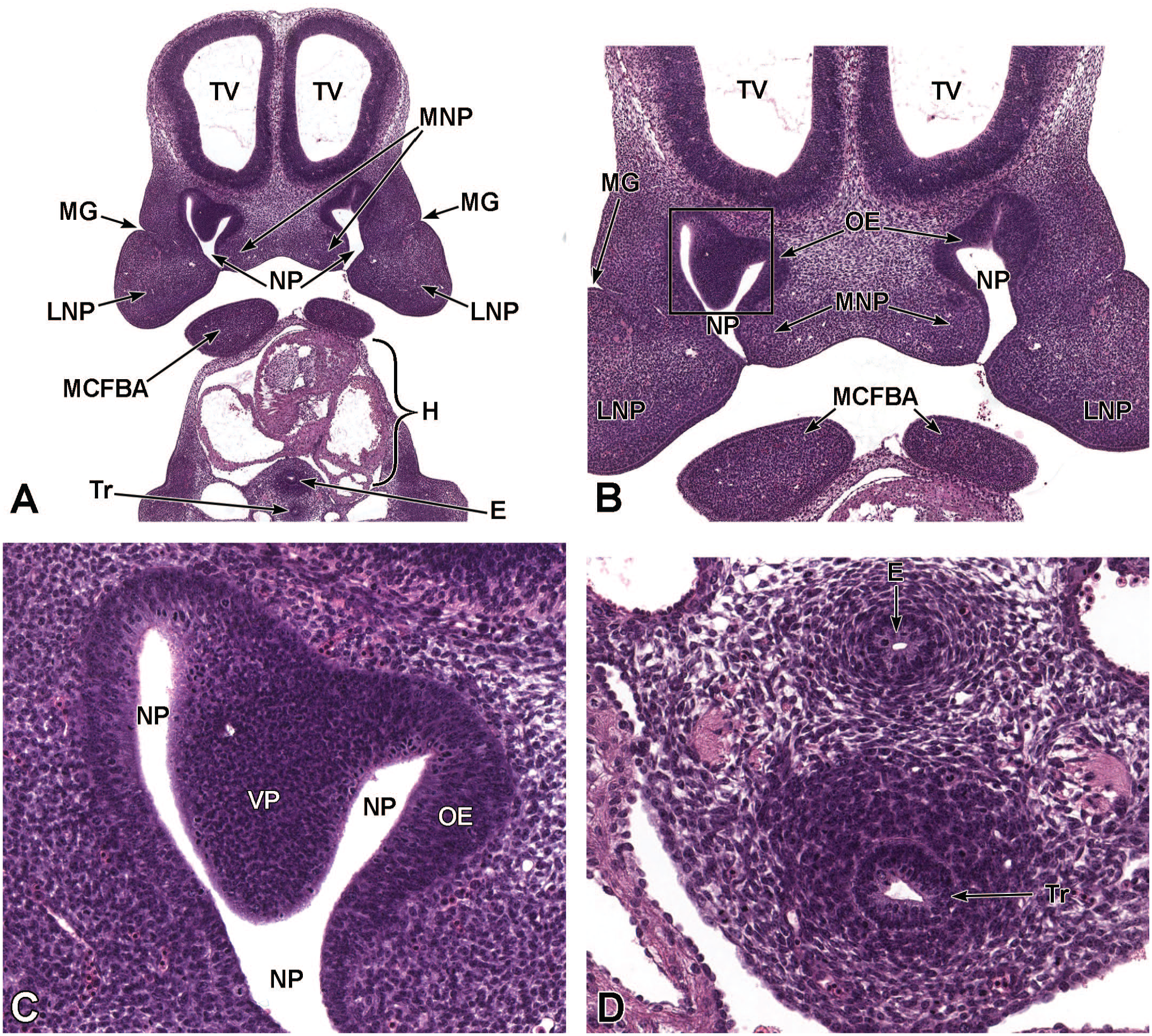
Frontal images that illustrate early development of the olfactory region of the nasal cavity at E11.5. Panel A (6.5×) shows the location of the nasal pit (NP) just ventral to the forebrain. Panels B (10×) and C (40×) demonstrate the primitive vomeronasal placode (VP) thickening in the medial walls of the nasal pit (square in panel B) and the formation of pseudostratified olfactory epithelium (OE) in the region of the VP and lining the nasal pit. Panel D (21×) depicts the trachea (Tr) and esophagus (E), both surrounded by circular dense mesenchymal tissue and encompassed by outer loose mesenchymal tissue. The right and left telencephalic vesicles (TV); maxillo-nasal grooves (MG); medial nasal processes (MNP); lateral nasal processes (LNP); mandibular component of the first branchial arch (MCFBA); and heart (H) are labeled for orientation. H&E.
Established neurogenesis begins at E12.5 when the VO separates from the main olfactory epithelium. 87 At this time, the vomeronasal groove is located along the medial region of the olfactory neuroepithelium 79 but continues to grow into a tubular-shaped structure that causes the primitive VO to separate from the deep (olfactory) portion of the nasal pit. 87 The medial region of the VO consists of pseudostratified sensory neuroepithelium while the lateral side of the VO is lined by cuboidal, non-sensory, ciliated epithelium that serves to circulate fluid and suspended pheromone molecules within the organ (Figure 12).39,87 The VO neuroepithelium consists of three cell types: apical progenitor cells near the lumen, basal (neurogenic) progenitor cells near the basement membrane, and postmitotic vomeronasal sensory neurons in the intermediate zone.39,87,160 At E13.5, the VO resembles a kidney with a crescent-shaped lumen (Figure 6).87,190 Also at this time, the VO is connected to the nasal and oral cavities via intersection of the VO ducts with the nasopalatine ducts (Figure 5). 87 The nasopalatine ducts begin just caudal to the incisive papilla with the two oral openings located on the upper palate, on the border between the soft and hard palates, near the caudal part of the VO (Figure 5). 103 Highly vascularized connective tissue is recognizable under the pseudostratified sensory epithelium at this time.79,87 By E15.0, the prominent VO occupies a large amount of the rostroventral nasal septum on both sides of the midline (Figure 14). 88
Between E15.0 and birth, the VO continues to grow. 190 At E16.0, the VO is located within the soft tissues adjacent to the cartilage of the nasal septum. 88 A cartilaginous capsule can be seen partially surrounding the VO at E15.5 and E16.5 (Figure 20). At E16.5, simple cuboidal epithelium lines the nasopalatine ducts. The sensory neuroepithelium continues to thicken while the non-sensory epithelium thins (Figure 20). 190 Structural VO development is completed by birth (Figure 21), but the organ only becomes fully functional during postnatal development. 190 During late embryonic development and throughout adult life, the VO contains two types of sensory neurons that express G protein-coupled receptors. These two neuron classes are confined to distinct regions of the VO where they bind various ligands and modulate innate behaviors. 87
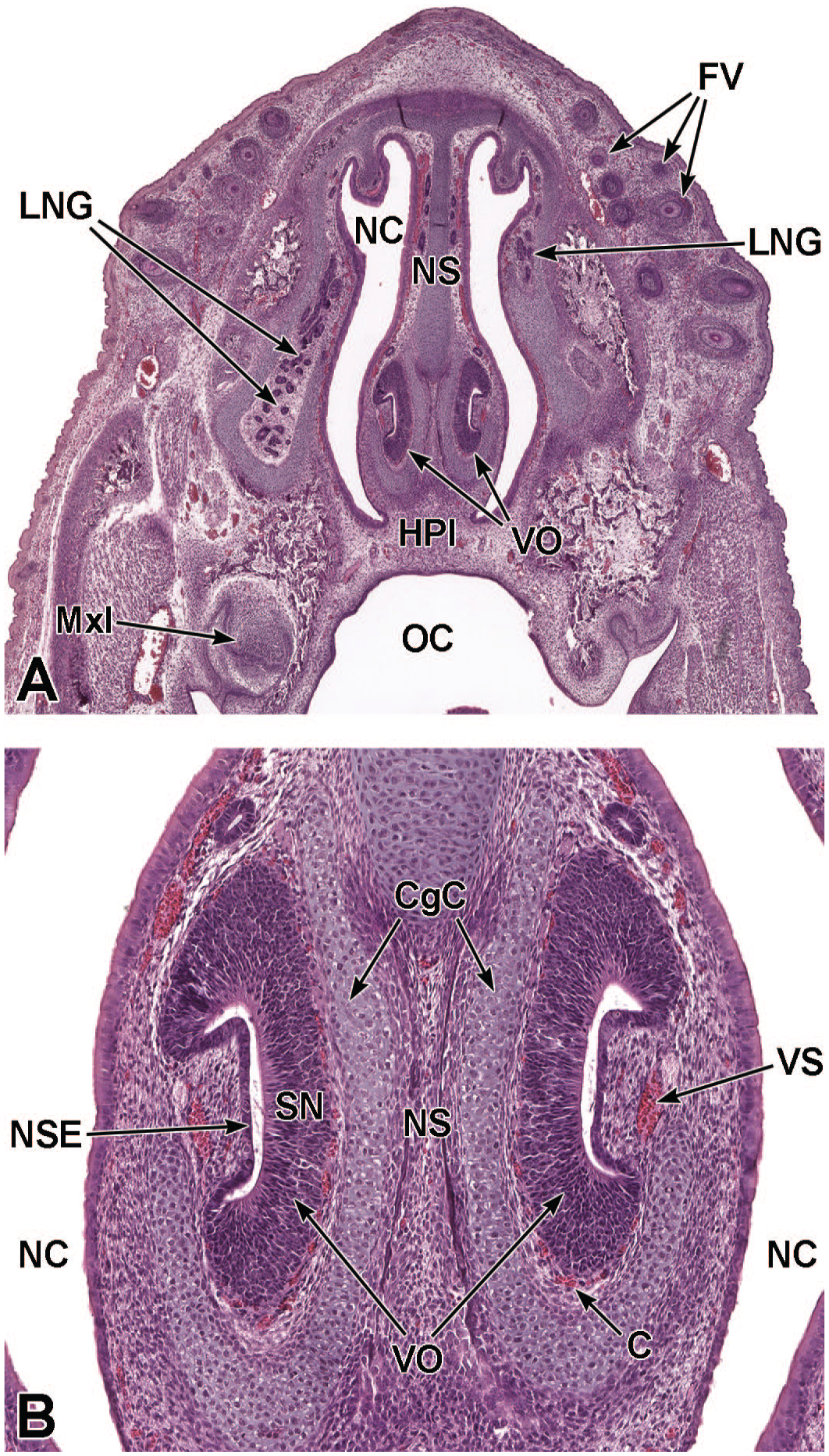
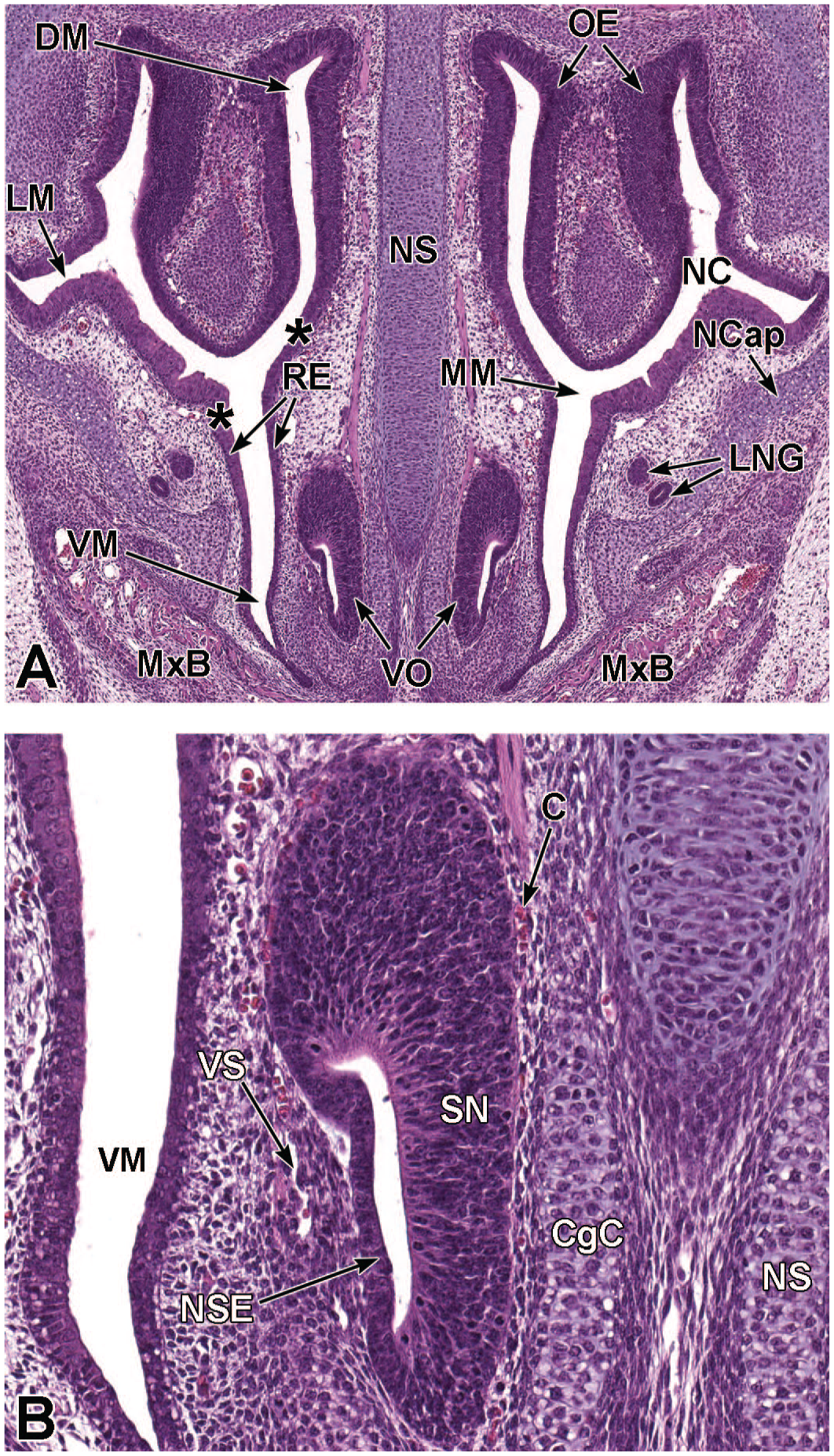
Transverse images of the paired vomeronasal organ and ducts at E16.5. Panel A (4×) depicts the vomeronasal organ (VO) residing within the tissues adjacent to the cartilage of the nasal septum (NS). Panel B (18.5×) illustrates the thick pseudostratified sensory neuroepithelium (SN) on the medial wall and thin non-sensory epithelium (NSE) on the lateral wall of the VO. Capillaries (C) and a lateral vascular sinus (VS) can be seen in the connective tissue surrounding the VO. Note the prominent cartilaginous capsule (CgC) partially encapsulating the paired organs on the caudal and medial sides. The primordia of follicles of vibrissae (FV); lateral nasal glands (LNG) associated with the lateral walls of the nasal cavity (NC); hard palate (HPl); oral cavity (OC); and maxillary incisor (MxI) are labeled for orientation. H&E.
Transverse images of the vomeronasal organ at E18.5. Panels A (3.5×), B (6×), and C (15.5×) show the paired, crescent-shaped vomeronasal organs (VO) at the base of the nasal septum (NS). Panel C highlights the thick pseudostratified sensory neuroepithelium (SN) on the dorsal, medial, and ventral walls; the thin non-sensory epithelium (NSE) on the lateral wall; and the clearly defined narrow central ducts of the VO. A prominent cartilaginous capsule (CgC) partially encircles the VO, and vascular sinuses (VS) are present in the lateral margins. The parietotectal cartilage of the nasal capsule (NCap); follicles of vibrissae (FV); dorsal (DM), lateral (LM), middle (MM), and ventral (VM) meatuses of the nasal cavity (NC); lateral nasal glands (LNG), medial nasal glands (MNG); hard palate (HPl); oral cavity (OC); maxillary incisor (MxI); tongue (T); and nerve bundles (NB) are labeled for orientation. H&E.
Grueneberg ganglion
The GG is used for both chemo- and thermo-sensing to avoid danger and identify safe food sources. 16 This neuronal structure is located dorsally at the rostral end of the nasal cavity close to the nares, lining both sides of the nasal septum and situated among large blood vessels. 16 Unlike the VO and SOM (described in the following section), cells of the GG do not reside in a neuroepithelium but rather are embedded in connective tissue and encircled by the septum, the nasal roof, and a thin overlying epithelial layer. 46 The sensory neurons of the GG have axons that project to the rostral (anterior) olfactory nucleus of the olfactory bulb (Figure 17). 48
At E14.0, the GG becomes visible as focal oval thickenings within the rostral epithelium (Figure 22). 47 The GG consists of two cell types: the ciliated neuronal cells (also called GG neurons) and glia-like cells (also called ensheathing or satellite cells) that surround the GG neurons. 47 Between E15.0 and E16.0, the GG neurons travel from the epithelium to the underlying connective tissue where they appear in clusters (Figure 23). 47 At this time, the epithelial layer above the GG decreases in thickness. 47
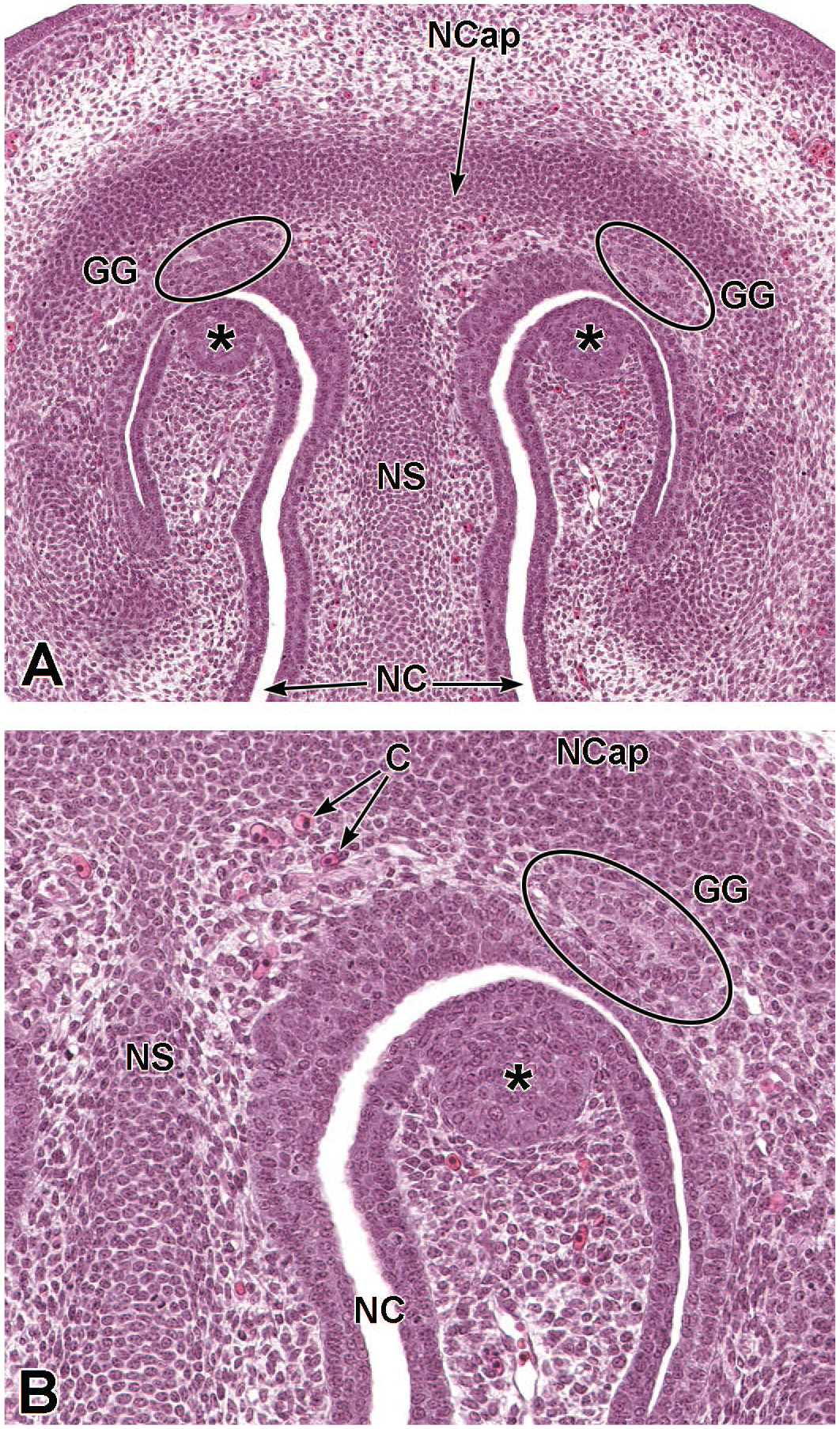
Transverse images of the Grueneberg ganglion at E14.5. Ovals outline the location of the Grueneberg ganglion (GG) in Panels A (20×) and B (40×). GG are an ancillary collection of sensory neurons located near the nares, as focal oval thickenings adjacent to the epithelium. The bilateral round epithelial foci (asterisks) are primordia that will contribute to future expansion of the turbinate apex. The nasal capsule (NCap); nasal septum (NS); nasal cavity (NC); and capillaries (C) are labeled for orientation. H&E.
Transverse images of the Grueneberg ganglia at E16.5. Panels A (20×) and B (60×) show the paired Grueneberg ganglia (GG), which appear bilaterally near the nares as a cluster of neurons and support cells in the connective tissue adjacent to the nasal septum (NS). The parietotectal cartilage of the nasal capsule (NCap); nasal cavity (NC); superficial sections of the medial nasal glands (MNG); lateral nasal glands (LNG); and squamous epithelium (SE) of the nasal vestibule are labeled for orientation. H&E.
While recognizable at E15.5, the primitive GG then assumes an arrow shape at E16.0. 54 Axons emerging from the GG cells can be detected and exist in clusters at E16.0. 54 Between E16.0 and E18.5, the GG undergoes rapid growth (Figure 23) and reaches a peak number of GG cells just after birth. 54 Structural GG development is completed at the time of birth and does not continue to grow (in size or cell numbers) as aging continues. 54 Instead, the number of GG cells decreases during postnatal development. 54 At E16.0, axons leave the GG and run along the roof of the nose, through the cribriform plate, and along the dorsal border of the main olfactory bulb. 47 The axons from GG neurons separate into smaller bundles, innervating up to 10 olfactory bulb glomeruli that envelop the rostral region of the accessory olfactory bulb (located on the dorsocaudal part of the olfactory bulb). 47
Septal organ of Masera
In the adult mouse, the SOM is a chemosensory organ that is reported to have major signal transduction pathways and odorant response properties similar to those of the main olfactory neuroepithelium. 109 It has been suggested that this chemosensory organ may serve an alerting function by sensing odors in the environment during quiet respiration, when the non-turbulent air stream does not deeply penetrate the complex scrolls of the ethmoid turbinates to reach the main olfactory epithelium. It has also been suggested that it may detect low-volatility compounds in urine, which can reach both the SOM and the VO but not the main olfactory epithelium.112,124,167,185
The SOM develops from the nasal placode and appears bilaterally as small islands of olfactory neuroepithelium near the base of the caudal nasal septum at the entrance to the nasopharynx.167,179 The SOM is distinguishable from the olfactory neuroepithelium in other portions of the nasal passages due to its location and its thickened olfactory-like epithelium, which is separated from the main olfactory neuroepithelium by the surrounding respiratory epithelium.
The SOM neuroepithelium is comprised of ciliated sensory neurons, supporting cells, and basal cells. The axons of SOM neurons project to the main olfactory bulb (Figure 17).109,179 At E16.0, the main olfactory neuroepithelium separates from the respiratory epithelium, and cells begin to assemble in the primitive septal organ region, near the caudoventral region of the nasal septum (Figure 24). 167 Sensory cells, immunopositive for protein gene product 9.5 (PGP 9.5, a neuron-specific marker) in the rat, are seen throughout the neuroepithelium. 124 Just before birth, the SOM increases in thickness due to aggregation of sensory neurons, although stratification of sensory and supporting cells has not yet occurred at this stage of development. 124
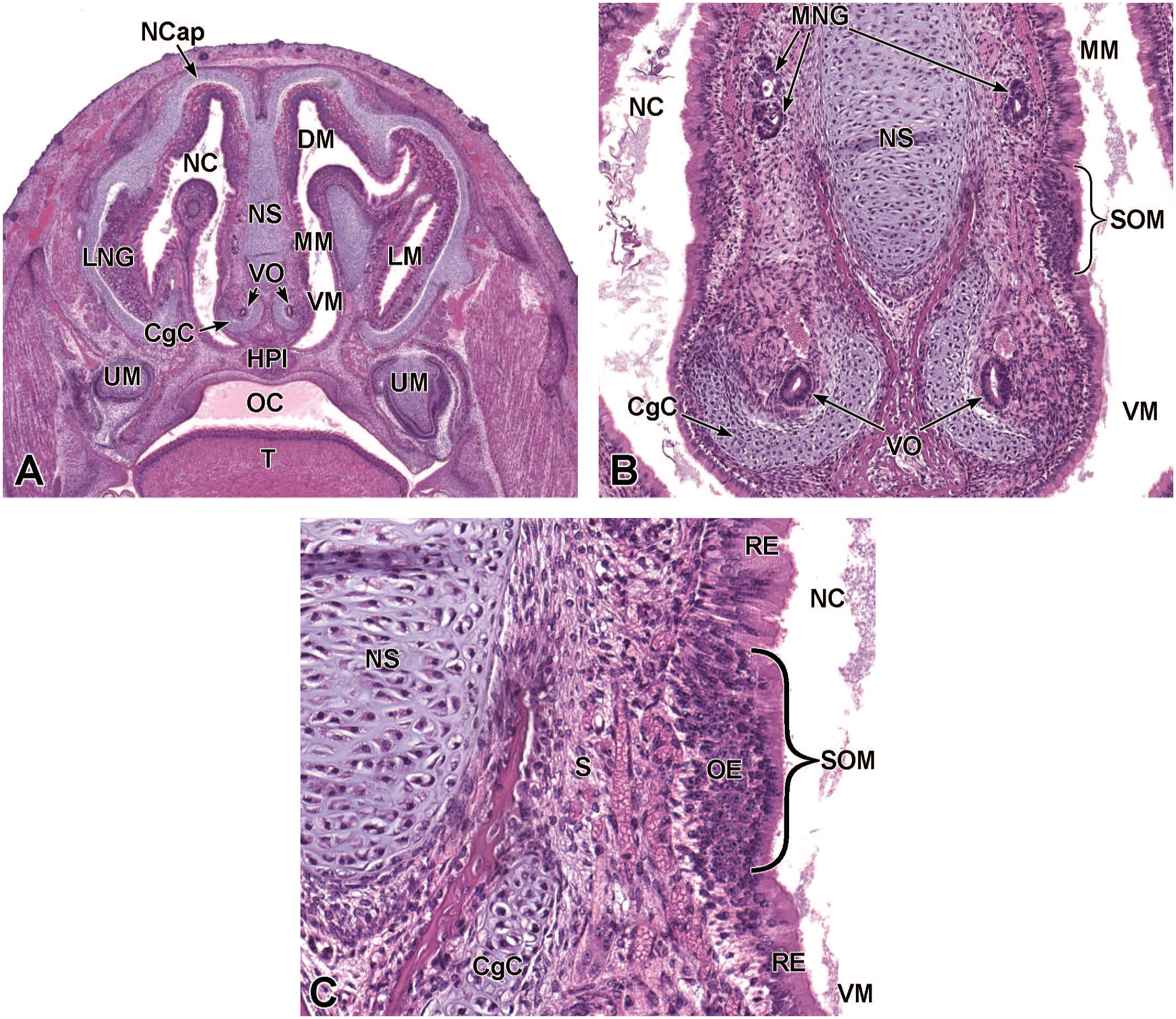
Transverse images of the septal organ of Masera at E17.5. Panels A (2.5×), B (10×), and C (24×) show the caudal portion of the nasal cavity (NC), where the septal organ of Masera (SOM) can be found bilaterally near the base of the nasal septum (NS). Since the panel B section is not exactly symmetrical, only the left SOM is clearly visible. The SOM (bracket) appears as small islands of thick pseudostratified olfactory epithelium (OE) covered by respiratory epithelium (RE) and separated from the main OE fields, which cover the caudodorsal portions of the nasal passages. The nasal capsule (NCap); vomeronasal organs (VO) and surrounding cartilaginous capsule (CgC); dorsal, middle, ventral, and lateral meatuses (DM, MM, VM, and LM, respectively); hard palate (HPl); roots of the right and left upper molars (UM); oral cavity (OC); tongue (T); lateral nasal glands (LNG); medial nasal glands (MNG); and nasal submucosa (S) are labeled for orientation. H&E.
Glands in the Nasal Passages
The submucosal glands of the respiratory system are essential in regulating airway homeostasis. 114 These glands secrete mucus into the nasal cavity, trapping airborne pathogens and environmental irritants (chemical and physical), and warming/humidifying the air. 114 The murine submucosal glands in the nasal passages are located in the medial and lateral nasal cavity walls.37,45,114 Adult murine submucosal glands consist of sacs of serous cells that form acini and secrete a watery-like secretion containing bactericidal enzymes. 114 The acini connect to mucous cells that secrete a thicker gel-like substance containing glycosylated proteins. 114 The secretions travel into a collecting duct composed of non-ciliated columnar epithelial cells, which then connects to a ciliated duct with beating cilia on the apical surfaces. 114 This ciliated duct secretes the mucus into the airway lumen. 114 May and Tucker 114 have provided a thorough analysis of the development of the lateral, medial, and maxillary nasal glands.
The lateral nasal glands are located in the submucosa of the lateral walls of the caudal nasal chamber and contain long ducts that open into the airways near the nasal vestibule (Figures 11, 14, 16, and 11, 14, 16, 20, 21, 23, 24). 114 During embryonic development, 13 lateral nasal glands (1 Steno’s gland and 12 other lateral nasal glands) form that extend branches and end buds during development. 114 Lateral nasal gland 1 (Steno’s gland) is the largest of the nasal glands and is located below the wall of the maxillary sinus. 114 The elongated excretory duct (Steno’s duct) for this gland appears between E12.0 and E12.5 as a bud in the rostral respiratory epithelium on the septal region of the nasal vestibule. 114 The bud forms by invagination of the pseudostratified respiratory epithelium into the underlying mesenchyme. 114 The duct elongates over the dorsal meatus and extends distally through the mesenchyme of the middle concha at E13.5. 114 At E14.5, the distal region of the duct contacts the mesenchyme below the respiratory epithelium of the maxillary sinus cavity, causing the duct to stop elongating and the Steno’s gland to branch distally for the next few days. 114 Acinar cell differentiation in Steno’s gland can be detected at E16.5, and additional gland branching as well as mucus production by acinar cells is evident by E17.0. 114 Lateral nasal glands 2 to 7 develop as buds from the caudal respiratory epithelium from the rostral region of the middle concha while lateral nasal glands 8 to 13 develop from the lateral walls of the dorsal meatus. 114 All the lateral nasal glands except the Steno’s gland bud through invagination of cells from the caudal respiratory epithelium into the mesenchyme. 114
The medial nasal glands are located within the submucosa of the medial wall on each side of the nasal septum, rostral to the VO (Figures 21, 23, and 24). 114 Mice have 4 medial nasal glands that contain serous acini and that extend branches and end buds during development. 90 Medial nasal glands 1 to 3 contain “light” and “dark” acinar cells as seen with transmission electron microscopy. 90 These medial glands begin to develop between E14.0 and E14.5. 114 The “light” cells contain a dense secretory product packaged in supranuclear granules while the “dark” cells contain mucous vesicles. 60 Medial nasal gland 4, which is morphologically similar to serous salivary glands, 90 develops from the rostral nasal septum at a slightly later time compared to the medial nasal glands 1 to 3. 114 Medial nasal gland 4 begins to develop at E17.5 as an elongated duct. 114
Maxillary sinus glands are located on the rostral and caudal walls of the maxillary sinus (Figures 9 and 15). 114 Their short branches are located within the mesenchyme near the surface epithelium of the sinus. 114 These glands appear at E15.0 and begin branching by E15.5. 114
Junctional Area Development (E9.5-E18.5)
Two portions of the pharynx are intimately related to respiratory tract development. The nasopharynx (also called the rhinopharynx) receives bilateral airstreams from the left and right nasal passages and moves the airstreams farther into the pharynx. The laryngopharynx (or hypopharynx) receives the air from the nasopharynx and transfers it into the larynx and then on to the trachea. This section discusses key events in the anatomic evolution of these structures.
Pharyngeal Development
In the adult mouse, the pharynx is a muscular tubular passage that connects the oral cavity and nasal cavity with the esophagus and larynx, respectively. It is divided into the nasopharynx (passage above the soft palate), oropharynx (space into which the oral cavity opens), and laryngopharynx (connection with larynx and esophagus). The muscles of the pharynx are integrally connected to the laryngeal cartilages and the hyoid bone. The pharynx provides sphincteric protection of the lower respiratory tract by adduction of the vocal cords when the oral cavity contains ingested fluids or solids. Ciliated respiratory epithelium with mucous goblet cells lines the nasopharynx while non-keratinized stratified squamous epithelium lines the oropharynx and laryngopharynx. 67
The pharynx (or pharyngeal apparatus) originates from the fourth branchial arch (Figure 2). There are six pairs of branchial arches (alternatively pharyngeal arches or visceral arches). The arches are separated by clefts on both surfaces, and the cavities formed inside the pharynx between adjacent branchial arches are called pharyngeal pouches.23,51,62 The lumen of the pharynx is a portion of the respiratory tract while the pharyngeal apparatus serves as the origin for such cranial and cervical structures as regional endocrine glands, lymphoid tissues, muscles, nerves, skeletal (bone and cartilage) elements, and vessels. 23
At E11.0, the primordial thyroid gland disconnects from the floor of the pharynx. 88 At E11.5, the thyroid gland begins differentiating from a thickening in the endoderm in the floor of the pharynx. 88 Primitive palatal shelves and nares are seen at the roof of the pharynx beginning at E11.5 (Figure 25 as example at E12.5). 88 At approximately E13.0, the entrance into the esophagus from the pharynx is clearly defined (Figure 26). 88 At this time, the primitive oropharynx, which has not yet divided into the oral and nasal components, is continuous with the external naris via the primitive internal (caudal) naris. 88 The thymic primordium has separated from the pharynx at E13.0. 88 At this time, the palatal shelves have assumed a vertical position (Figure 10) and are preparing to begin their elevation into the horizontal position (to partition the nasal passages from the oral cavity), which occurs between E14.0 and E14.5.88,161 At E14.0, the nasopharynx separates from the oropharynx.9,88 This event is concurrent with downward extension of the nasal septum toward the elevating palatal shelves, resulting in the separation of the nasopharynx into the left and right nasal cavities. 88 Between E15.5 and E16.0, the palatal shelves have closed, and the nasopharynx is completely separated from the oropharynx (Figure 27).19,88 At E16.0, conchae within the nasopharynx are delineated, with the dorsal region lined by olfactory neuroepithelium. 88 The caudal region of the oropharynx is continuous with the entry to the lower respiratory tract (larynx) and the esophagus (Figure 28). 88 At E17.0, elongation of the face is accompanied by increased differentiation in the nasopharynx. 88 By E18.5, the nasopharynx is positioned in continuity with the oropharynx (Figure 29). 88
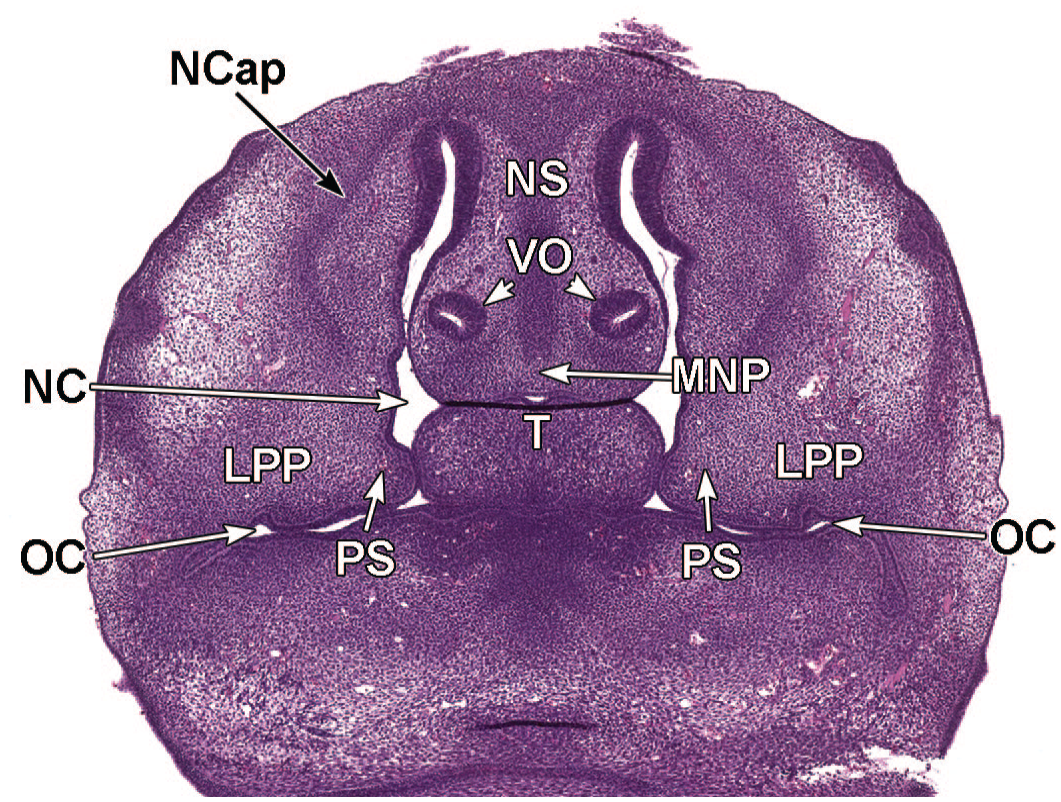
Transverse image of the primitive palatal shelves at E12.5. This image (6.5×) demonstrates the location of the right and left palatal shelves (PS) on either side of the tongue (T). The nasal capsule (NCap); nasal septum (NS); vomeronasal organs (VO); nasal cavity (NC); medial nasal process (MNP); lateral palatine processes (LPP); and oral cavity (OC) are labeled for orientation. H&E.
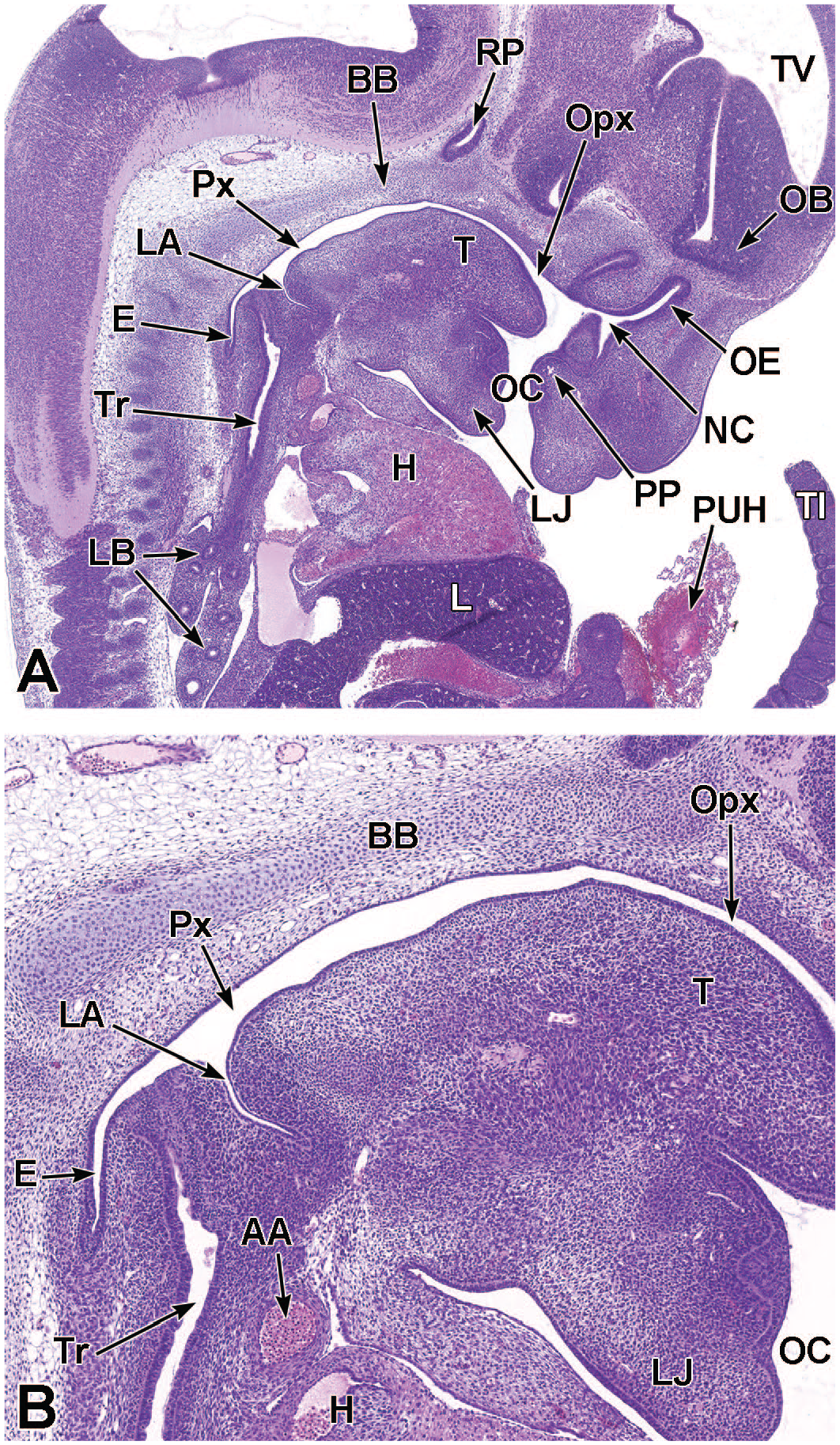
Sagittal images depicting the continuity of upper and lower respiratory tracts and digestive tract at E12.5. Panels A (4×) and B (10×) show the connectivity among the nasal cavity (NC), oral cavity (OC), pharynx (Px), laryngeal aditus (LA [entrance to the laryngeal cavity]), trachea (Tr), and esophagus (E). The cartilage anlage of the basisphenoid bone (BB); Rathke’s pouch (RP [pituitary gland precursor]); telencephalic vesicle (TV); olfactory bulb (OB) primordium; olfactory epithelium (OE) lining the nasal cavity; primary palate (PP); ventral extremity of the lower jaw (LJ); oropharynx (Opx); tongue (T); lung buds (LB); aortic arch (AA) of the heart (H); liver (L); physiological umbilical herniation (PUH); and tail (Tl) are shown for orientation. H&E.
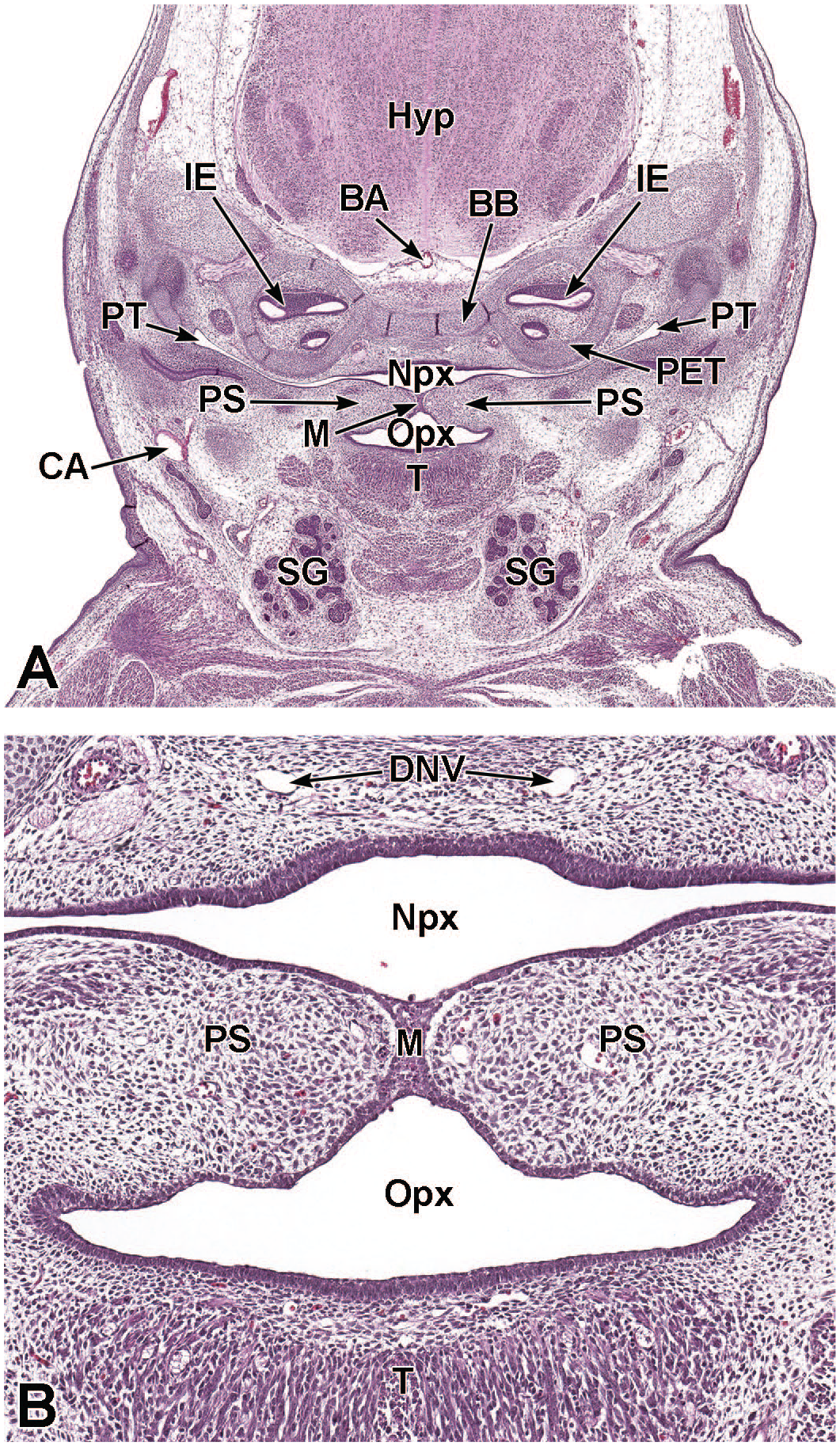
Coronal images of palatal shelf fusion to form the palate at E15.5. Panels A (4×) and B (20×) show the site of midline (M) apposition in the right and left palatal shelves (PS), resulting in separation of the nasopharynx (Npx) and oropharynx (Opx). The caudal hypothalamus (Hyp); basilar artery (BA); cartilage primordia of the basisphenoid bone (BB) and petrous temporal bone (PET); inner ears (IE); pharyngo-tympanic (Eustachian) tubes (PT); external carotid artery (CA); salivary glands (SG); root of the tongue (T); and dorsal nasal veins (DNV) are labeled for orientation. H&E.
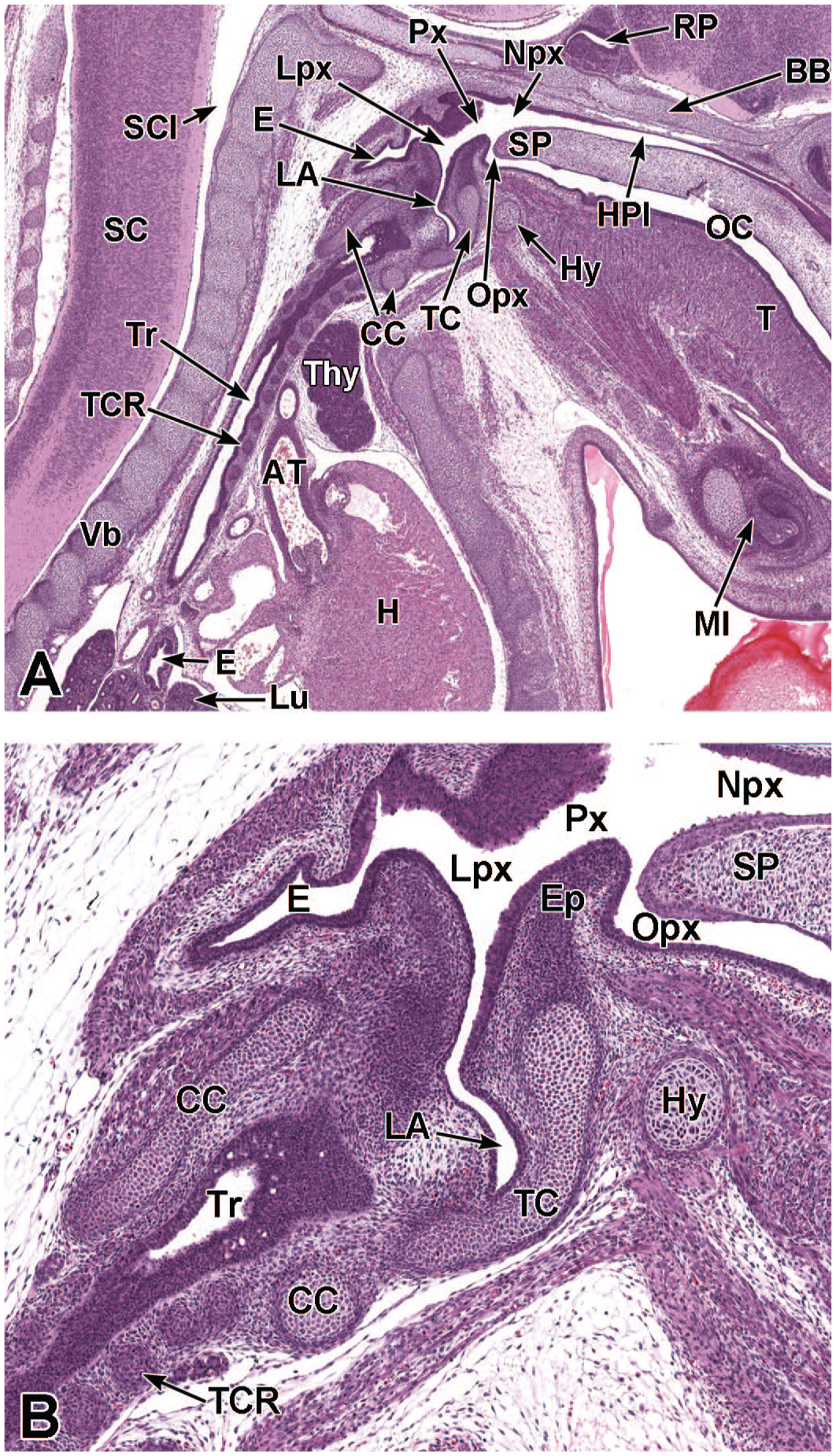
Sagittal images showing the continuity of the pharynx with the entrances to the lower respiratory and digestive tracts at E16.5. Panels A (2x) and B (6x) depict the separate nasopharynx (Npx), oropharynx (Opx), pharynx (Px), and laryngopharynx (Lpx) intersecting caudal to the soft palate (SP). Key structures located in or near the larynx include the laryngeal aditus (LA [entrance to the laryngeal cavity]), epiglottis (Ep), cricoid (CC) and thyroid (TC) cartilages, trachea (Tr), esophagus (E), and the cartilage primordium of the hyoid bone (Hy). The basisphenoid bone (BB); Rathke’s pouch (RP [pituitary gland precursor]); hard palate (HPl); oral cavity (OC); tongue (T); mandibular incisor (MI); thymus (Thy); tracheal cartilage rings (TCR); aortic trunk (AT) of the heart (H); lungs (Lu); vertebrae (Vb); spinal cord (SC); and spinal canal (SCl) are labeled for orientation. H&E.
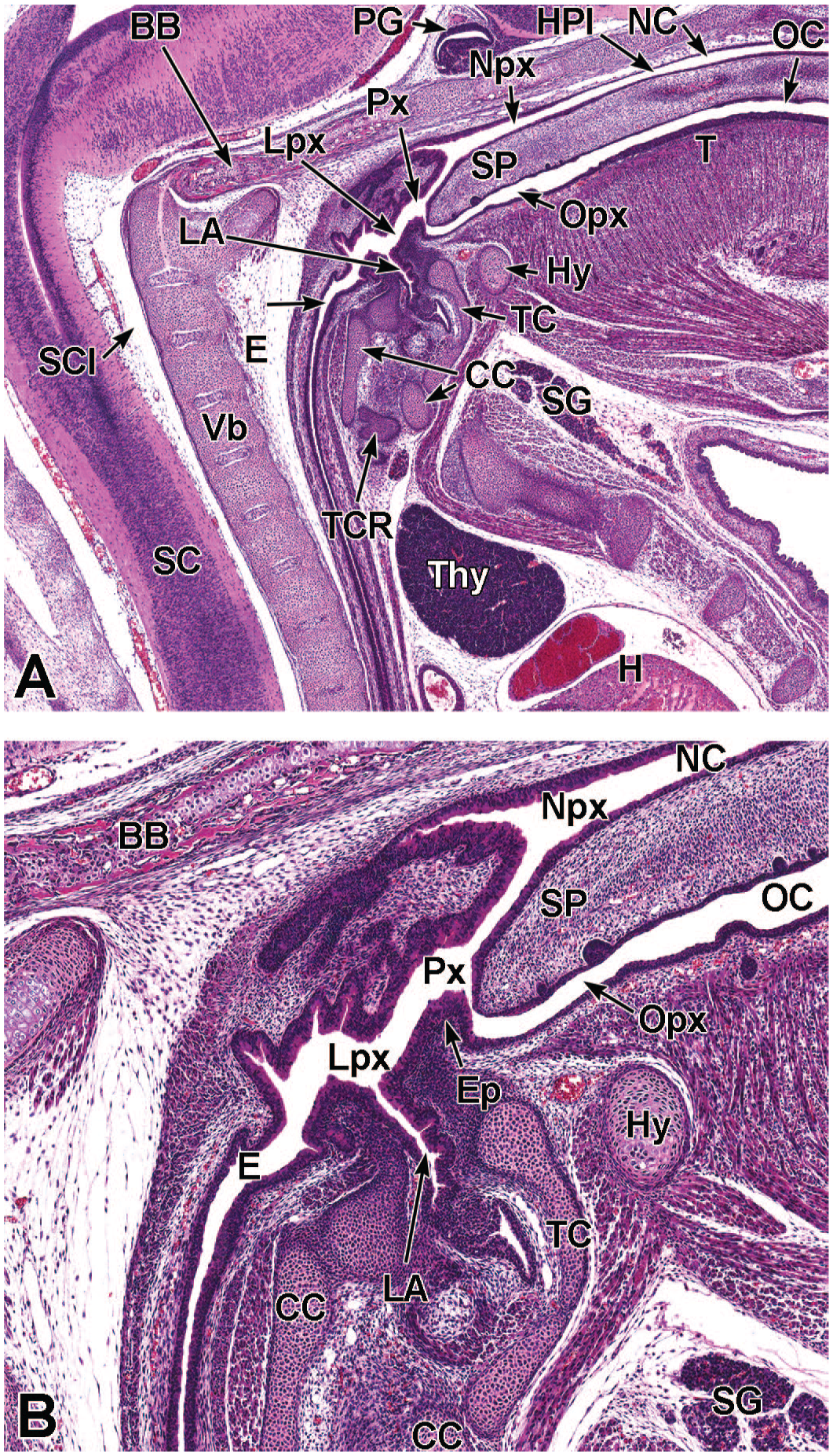
Sagittal images showing the continuity of the respiratory and digestive tracts at E18.5. Panels A (4×) and B (10×) depict the connectivity of the nasal cavity (NC), nasopharynx (Npx), oral cavity (OC), oropharynx (Opx), pharynx (Px), laryngopharynx (Lpx), laryngeal aditus (LA [entrance to the laryngeal cavity]), and esophagus (E). The cartilage primordium of the hyoid bone (Hy), cricoid (CC) and thyroid (TC) cartilages, and epiglottis (Ep) can be identified in the laryngeal region. The basisphenoid bone (BB); pituitary gland (PG); hard palate (HPl); soft palate (SP); tongue (T); salivary gland (SG); tracheal cartilage rings (TCR); thymus (Thy); heart (H); spinal cord (SC); spinal canal (SCl); and vertebrae (Vb) are labeled for orientation. H&E.
Laryngeal and Vocal Fold Development
The larynx typically is considered to be part of the upper respiratory tract. This “vocal (or voice) box” is comprised of an elaborate mixture of cartilages, connective tissue, muscles, nerves, and surface projections, including vocal folds (“vocal cords”).140,159 The rostrodorsal aspect of the laryngeal cavity opens into the pharynx while the caudal opening of the laryngeal cavity connects to the trachea. The laryngeal aditus is an opening that connects the pharynx with the larynx. In the adult mouse, the functions of the larynx are phonation, protecting the lower airways (especially during swallowing), and facilitating respiration. The larynx originates from the endodermal lining and adjacent mesenchyme of the embryonic foregut between the fourth and sixth branchial arches (Figure 2).
In the adult mouse, vocal folds are ridges of tissue located at the distal aspect of the larynx. They are comprised of paired vocal ligaments associated with paired cricoarytenoid, thyroarytenoid, cricothyroid, and vocalis muscles. 159 The mucosa covering the folds consists of stratified squamous epithelium. In addition to phonation, vocal folds protect the lower airways from occlusion (“choking”) by material lodged in the pharynx and regulate the flow of air into the trachea and lungs. They will open during inhalation, completely close during swallowing, and variably close during phonation. Five morphogenic events drive murine embryonic larynx and vocal fold development (Table 7). 108
Morphogenic events in larynx and vocal fold development. 160

Development of the larynx and vocal folds begins at E10.5 with apposition of the lateral walls of the laryngotracheal groove, forming the laryngopharynx with the elimination of the ventral lumen (Figure 30). The vocal folds connect onto the trachea via the infraglottic region of the laryngopharynx (Figure 30). The epithelial vocal fold cells are located in the primitive laryngopharynx. 107 At E11.0, the laryngopharynx is bilaterally compressed and elongated by the laryngeal mesoderm, resulting in formation of a transverse ridge on the floor of the foregut. 70 Each side of the laryngopharynx from the fourth to the sixth branchial arches features wedge-shaped swellings that eventually become the laryngeal cartilages and associated skeletal muscle (Figure 2). 70 The restructuring of the lateral wall swellings is caused by proliferation and differentiation of vocal fold basal progenitors (epithelial cells) and cartilage (mesenchymal cells). 106 Evolving positioning of the lateral walls of the primitive laryngopharynx between E11.5 and E12.0 causes the two layers of the vocal fold epithelial cells to fuse together, thus eliminating the ventral laryngopharynx lumen and creating the epithelial lamina (Figure 31).70,106,107 The epithelial lamina first forms the laryngeal region of the tracheoesophageal septum (E13.5) and eventually becomes resorbed to open the laryngotracheal tube (E13.5-E18.5), 106 thus joining the upper and lower airways. The pseudostratified columnar epithelium of the foregut differentiates into a single layer of cuboidal cells, which is unique to the vocal folds. 107 At E11.5, the epithelial lamina temporarily occludes the ventral laryngopharynx.70,106 Between E13.0 and E14.0, the epithelial lamina continues to partition the laryngopharynx except for the ventral vestibular recess (or laryngeal cecum, a pouch-like protrusion of the pharynx) and the dorsal pharyngoepiglottic duct (Figure 32). 70
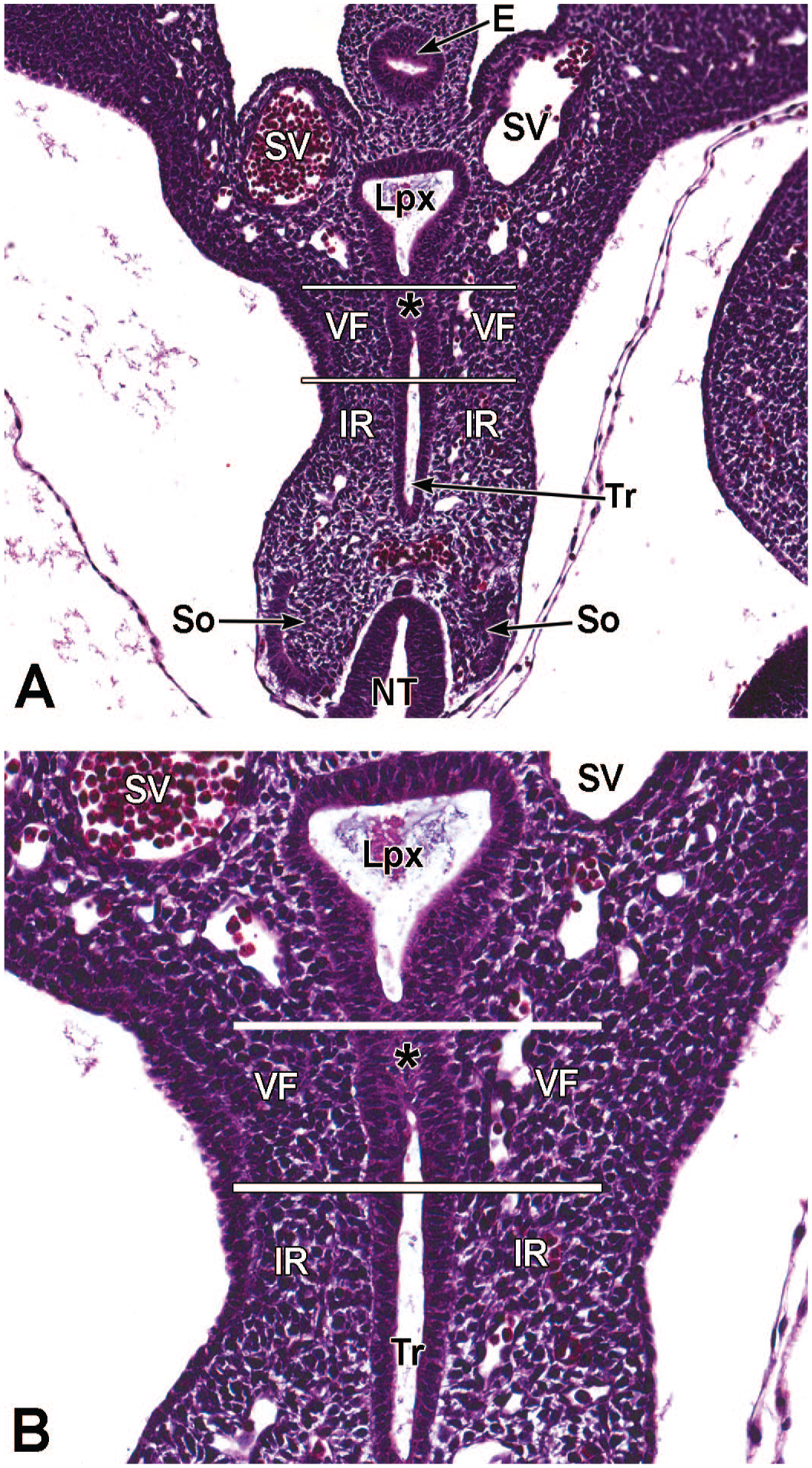
Transverse images of the initial apposition of the lateral walls of the laryngotracheal groove at E10.5. Panels A (20×) and B (40×) illustrate the initial site (asterisk) at which the opposing lateral walls of the laryngotracheal groove meet to form the laryngopharynx (Lpx) and trachea (Tr). Closure of the central portion of the groove ultimately results in the formation of distinct esophageal and tracheal lumens. The Lpx connects with the esophagus (E) caudally. The vocal folds (VF; denoted by lines) are associated with the trachea via the infraglottic region (IR). The right and left subcardinal veins (SV); neural tube (NT [nascent spinal cord]); and somites (So) are labeled for orientation. H&E.
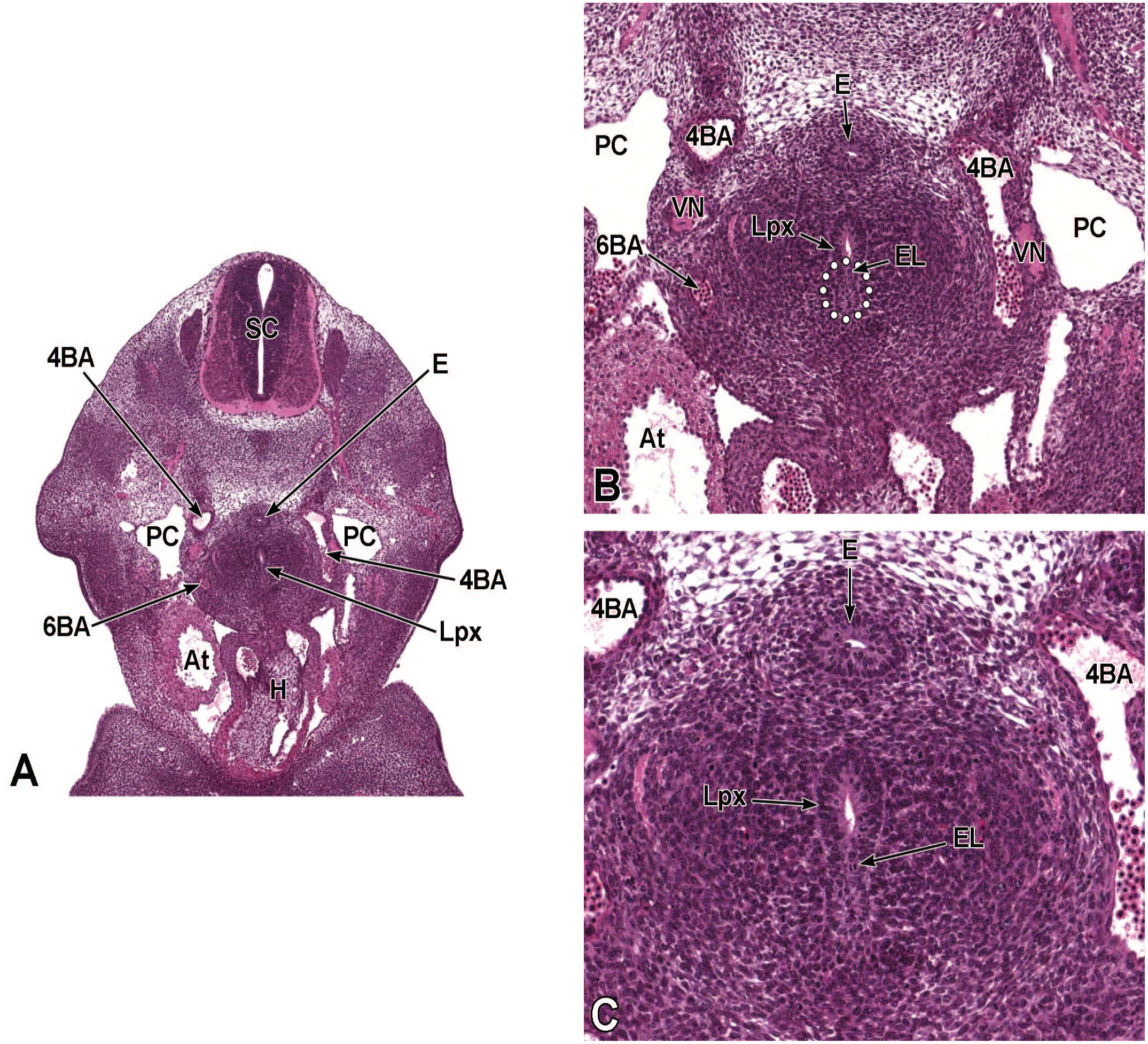
Transverse images demonstrating the continued apposition of the walls of the primitive laryngopharynx and formation of the epithelial lamina at E11.5. Panels A (4×), B (10.5×), and C (19×) illustrate the continued fusion of the vocal fold epithelial cells of the primitive laryngopharynx (Lpx), eliminating the ventral laryngopharynx lumen (dotted oval), while forming the epithelial lamina (EL). The spinal cord (SC); fourth branchial arch artery (4BA); esophagus (E); the right and left vagus nerves (VN); the right and left pericardioperitoneal canals (PC); sixth branchial arch artery (6BA; future pulmonary artery); and atrium (At) of the heart (H) are labeled for orientation. H&E.
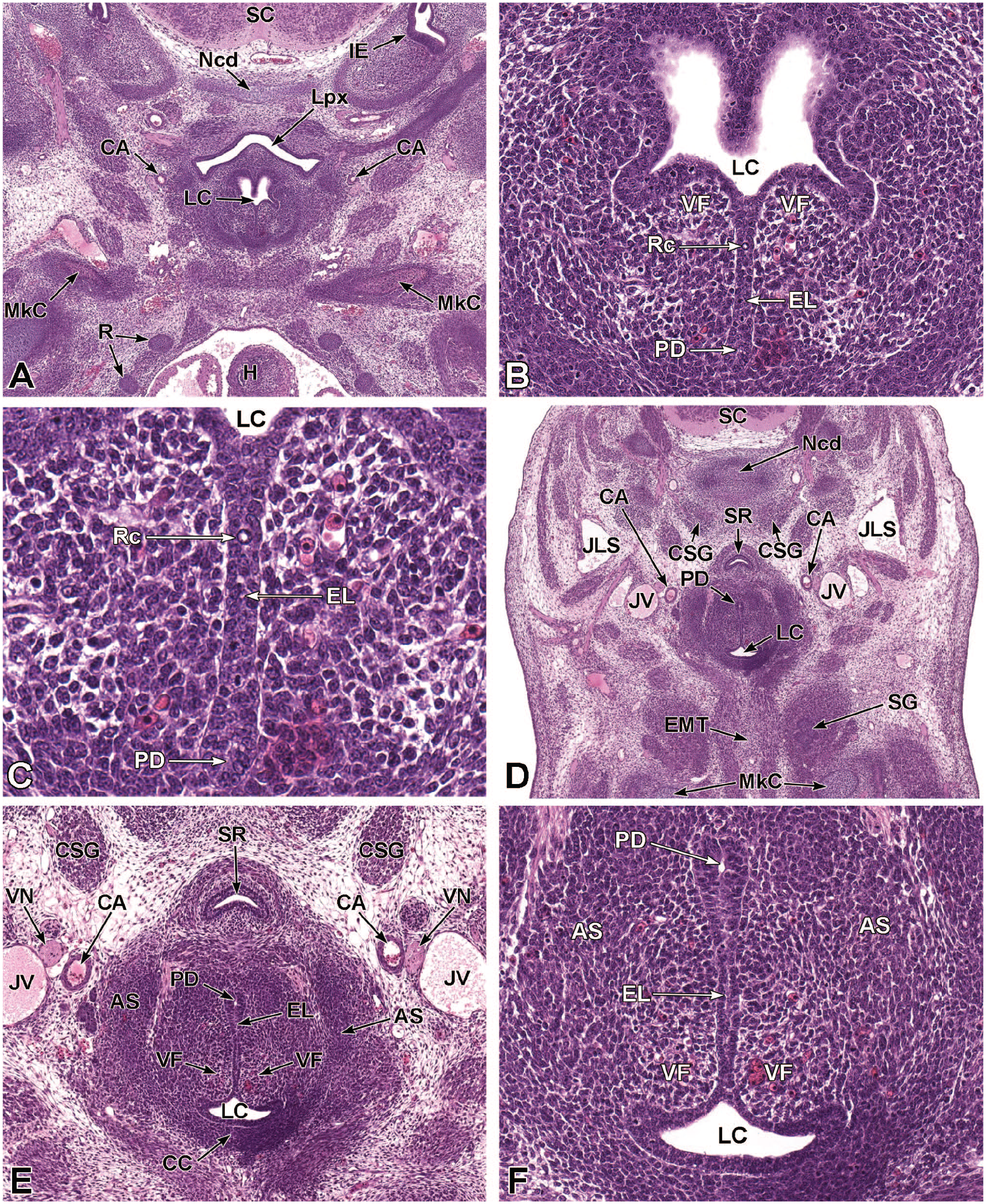
Frontal and transverse images of the laryngeal recanalization at E13.5. Panels A (4×), B (40×), C (80×), D (3.5×), E (8.5×), and F (13.5×) depict the initiation of epithelial lamina (EL) recanalization (Rc) as well as vocal fold (VF) separation. Panels A to C are frontal sections in the region of the laryngopharynx (Lpx) while panels D to F are more caudally located transverse sections in the supraglottic region (SR); the Rc can clearly be seen in panels B and C. Proliferation of cells in the epithelial lamina expands the lamina from one to two layers. The cranially located laryngeal cecum (LC) is a dilated extension from the laryngeal groove, producing a ventral vestibular recess or pouch-like protrusion of the pharynx. The caudally located pharyngoepiglottic duct (PD, which leads to the trachea) also forms at this stage. Panels D to F depict the epithelial lamina and vocal folds just before the Rc process and show the arytenoid swellings (AS) surrounding the epithelial lamina and the initial separation of the VF. The upper cervical region of the spinal cord (SC); notochord (Ncd [located in the vertebral bodies]); inner ear (IE); the right and left common carotid arteries (CA); precartilage primordium of Meckel’s cartilage (MkC); ribs (R); heart (H); internal jugular vein (JV); the right and left vagus nerves (VN); jugular lymph sac (JLS); salivary glands (SG); extrinsic muscle of the tongue (EMT); cervical sympathetic ganglia (CSG); and cricoid cartilage (CC) are labeled for orientation. H&E.
Between E13.5 and E18.5, the laryngeal cartilages and muscles develop, the vocal fold epithelium becomes stratified, and the epithelial lamina is recanalized.3,107 Recanalization prevents the airways from being obstructed and is necessary for further respiratory tract development to occur. 106 Recanalization begins with the formation of the laryngeal cecum and pharyngoepiglottic duct, which extend into the laryngopharynx and apply pressure to the epithelial walls (Figures 32 and 33). 106 The laryngeal cecum, ventral to the epithelial lamina, dilates from the laryngeal groove. 107 Ventral to the epithelial lamina is the pharyngoepiglottic duct, which leads to the trachea. 107 The expansion of these cavities causes the vestibular recess to unite with the pharyngoepiglottic duct, causing the vocal folds to move apart.106,107 Separation of the vocal folds occurs simultaneously with laryngeal cartilage chondrification and formation of the intrinsic laryngeal muscles. 107 Also at E13.5, the epithelial basal progenitors present within the epithelial lamina change shape and proliferate from one to two layers of cells. 107 Elongated cells make up the basal layer while irregularly round cells lie on the top of the basal layer and form the apical layer (Figure 32). 107 The proliferation of progenitor cells is thought to be required for epithelial lamina recanalization as well as the conversion from simple to multilayered or stratified epithelium. 106
Recanalization continues at E14.5 and E15.5 when the epithelial lamina proceeds to separate and the surrounding arytenoid, cricoid, and thyroid cartilages are visible (Figures 33 and 34). 107 The epithelial cells encircling the separating vocal folds begin to stratify. 107 At E16.5, the vestibular recess and pharyngoepiglottic duct continue to dilate, and the epithelial lamina slowly disintegrates to open the laryngotracheal tube by E18.5. 108 Between E17.5 and E18.5, rhythmic contraction of the intrinsic laryngeal muscles move the vocal folds to close and open the glottis to allow amniotic fluid to flow bidirectionally (Figure 35). 106 By E18.5, the vocal folds are completely separated, and the mesenchymal structures are fully developed (Figure 35). 106 Larynx and vocal fold development continues after birth (as described below).
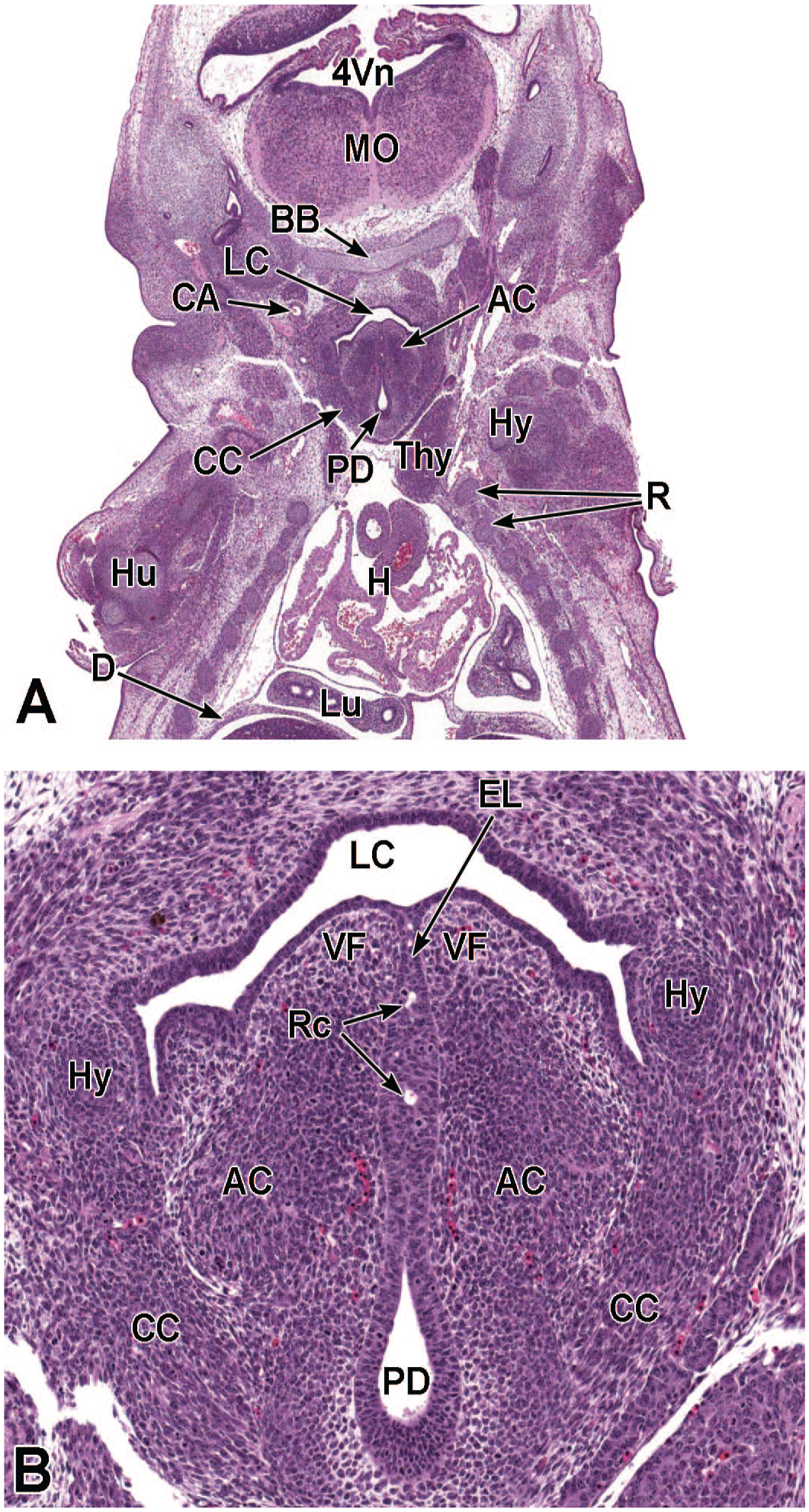
Frontal images of continued laryngeal recanalization at E14.5. Panels A (2×) and B (12×) represent the continued recanalization (Rc) of the epithelial lamina (EL) and vocal fold (VF) separation in the supraglottic region, revealing the nascent lumen that will connect the laryngeal cecum (LC) and pharyngoepiglottic duct (PD). The cartilage primordia of the arytenoid (AC) and cricoid (CC) cartilages and hyoid bone (Hy) are also shown. The fourth ventricle (4Vn); medulla oblongata (MO [caudal brainstem]); cartilage primordium of the basisphenoid bone (BB); carotid artery (CA); humerus (Hu); ribs (R); thymus (Thy); heart (H); lungs (Lu); and diaphragm (D) are labeled for orientation. H&E.
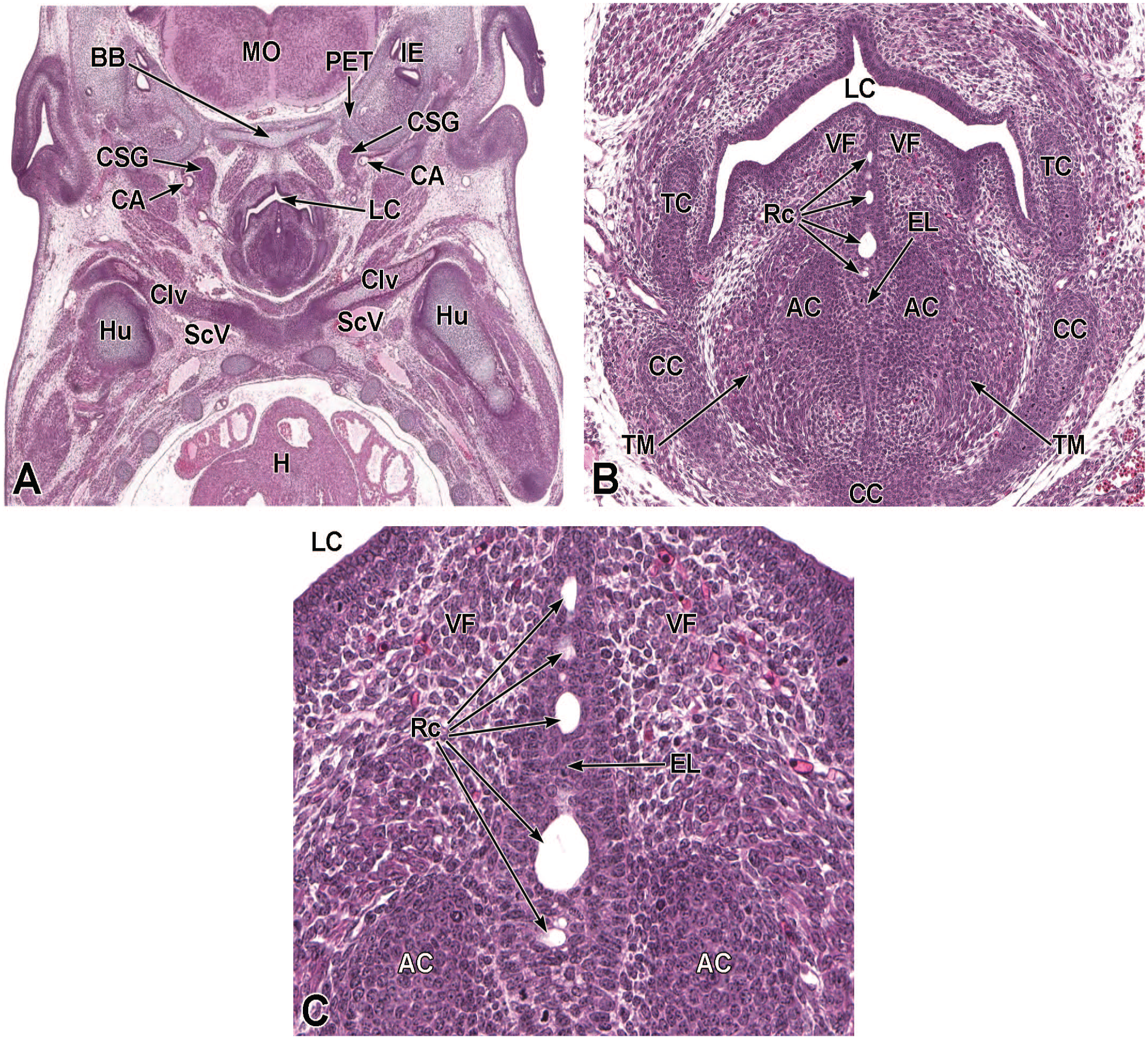
Frontal images demonstrating continued laryngeal recanalization at E15.5. Panels A (2.5×), B (20×), and C (60×) depict the laryngeal cecum (LC) and ongoing recanalization (Rc) of the epithelial lamina (EL), which will eventually result in the laryngopharynx uniting with the pharyngoepiglottic duct (not present at this cranial level). The medulla oblongata (MO); inner ear (IE) within the cartilage primordium of the petrous temporal bone (PET); cartilage primordium of the basioccipital bone (BB); cranial cervical sympathetic ganglion (CSG); right and left common carotid arteries (CA); arytenoid (AC), cricoid (CC), and thyroid (TC) cartilages; vocal folds (VF); thyroarytenoid muscles (TM); cartilage primordium of the clavicle (Clv); subclavian vein (ScV); cartilage primordia of the shafts of the right and left humerus (Hu); and heart (H) are shown for orientation. H&E.
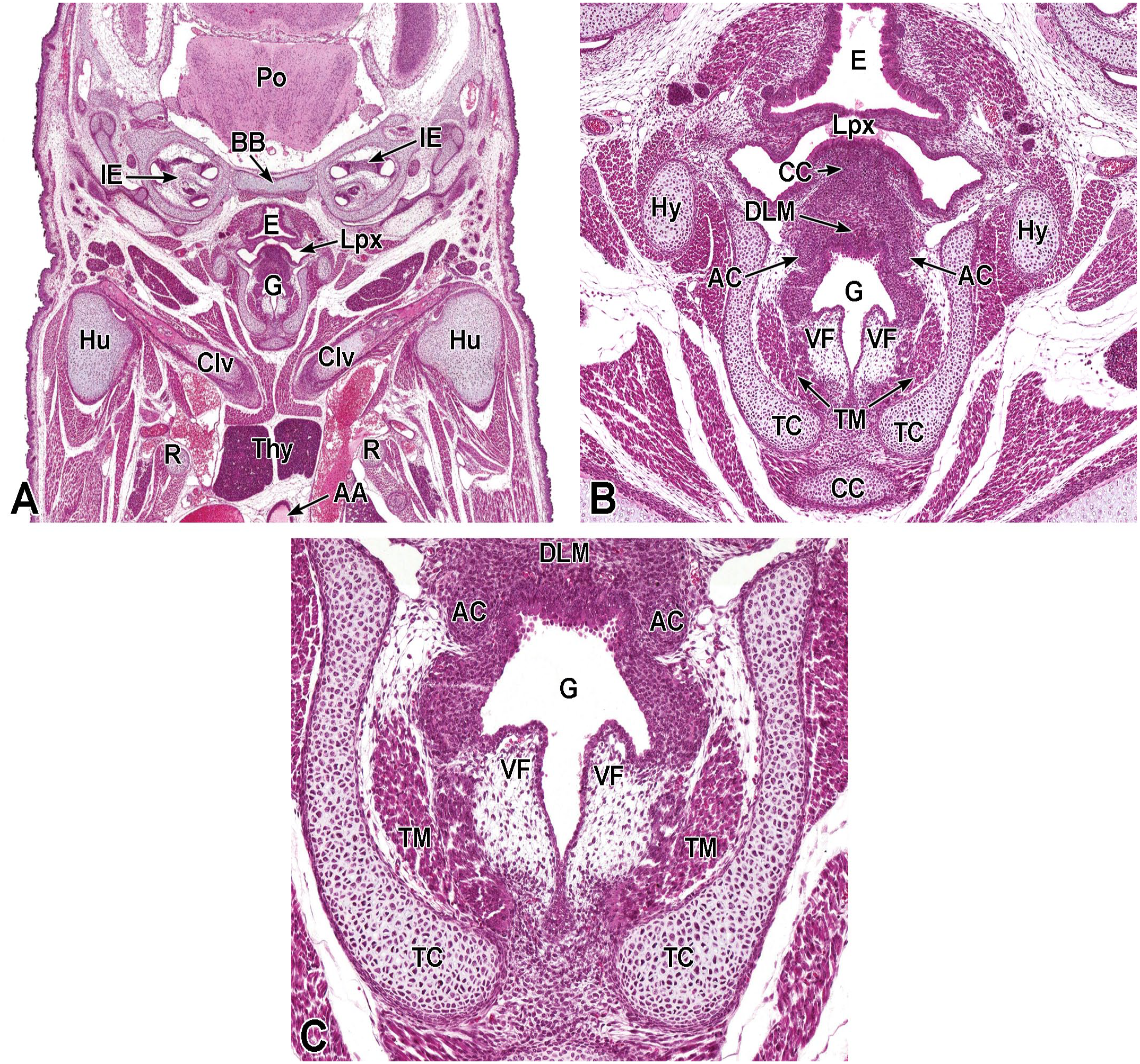
Frontal images of complete vocal fold separation in the larynx at E17.5. Panels A (1.5×), B (10×), and C (20×) depict the completely separated vocal folds (VF) and the diaphanous nature of the VF tissue at this stage. The pons (Po [rostral brainstem]); cartilage primordium of the hypophyseal fossa (sella turcica) of the basisphenoid bone (BB); right and left inner ears (IE); esophagus (E); laryngopharynx (Lpx); glottis (G); clavicle (Clv), humerus (Hu), rib (R); thymus (Thy); aortic arch (AA) of the heart; arytenoid (AC), cricoid (CC), and thyroid (TC) cartilages; dorsal laryngeal muscles (DLM); hyoid bone (Hy); and thyroarytenoid muscles (TM) are labeled for orientation. H&E.
Lower Respiratory Tract Development (E9.5-E18.5)
The lower respiratory tract includes the major cervical (larynx and trachea) and thoracic (lung [bronchial and alveolar]) airways. The larynx, trachea, and bronchi (including bronchioles) serve as air-tight channels for air passage while the lung parenchyma (via the alveoli) are the site of gas exchange between the atmosphere and circulation.
Trachea
In the adult mouse, the trachea is a wide air channel that extends distally from the larynx to branch into the carina (bifurcation point), the first airway branch point that serves as the origin of the left and right main bronchi. The membranous trachea wall is composed of adventitia; regularly spaced, C-shaped (open dorsally), hyaline cartilage rings; smooth muscle; annular ligaments (connecting the tips of the cartilage rings); and mucosa with submucosa. The tracheal mucosa consists of pseudostratified epithelium with ciliated and occasional secretory cells, scattered clusters of neuroendocrine cells, and basal cells. Submucosal glands are comprised of intermingled mucous and serous cells. Occasional small clusters of leukocytes (mainly lymphocytes with a few plasma cells) may be seen in the tracheal submucosa of adults but not embryos as these cells develop in lymphoid organs after birth before migrating to this location once respiration has begun.
The trachea and esophagus separate beginning about E9.5. 92 After separation of the trachea and the esophagus anlagen, the trachea continues to develop in two steps: tube length elongation (E10.5-E14.5) and diameter expansion and further elongation (E.14.5-E18.5). 93 As early as E11.0, blood vessels can be identified along the outer wall of the trachea (Figure 36). 111 At E11.5, columnar epithelial cells surround the tracheal lumen while the trachea walls feature multiple layers of outer concentric rings of mesenchymal cells and blood vessels (Figure 36). Tracheal smooth muscle differentiation near the dorsal tracheal splanchnic mesenchyme (mesoderm) begins at E11.5. 93 At E12.0, the trachea is surrounded by an expanded splanchnic mesenchyme, which will differentiate over time into the cartilage rings, ligaments, connective tissues, and smooth muscle of the wall (Figure 37). 88 At E13.0, the diameter of the tracheal lumen continues to expand while differentiation continues for both the epithelial cell lining and the surrounding splanchnic mesenchyme. 93 At E13.5, the trachea is lined by pseudostratified columnar epithelium with interspersed mitotic figures (Figure 38). The cartilaginous rings of the trachea have not formed at this time. By E14.5, chondrogenesis begins to support the ventral and lateral sides of the tracheal tube and main bronchi (Figure 39 as an E15.5 example).88,93 Between E14.5 and E16.5, about 60% of cells have an apical surface exposed to the lumen, and about 40% of cells occupy a basal position and do not reach the lumen. 93 At E14.5, the numbers of proliferating cells peak in both the mesenchyme and the epithelium. 93 At E15.0, the trachea is aligned along the midline before later shifting to an offset position to the right of the midline and ventral to the esophagus. 88
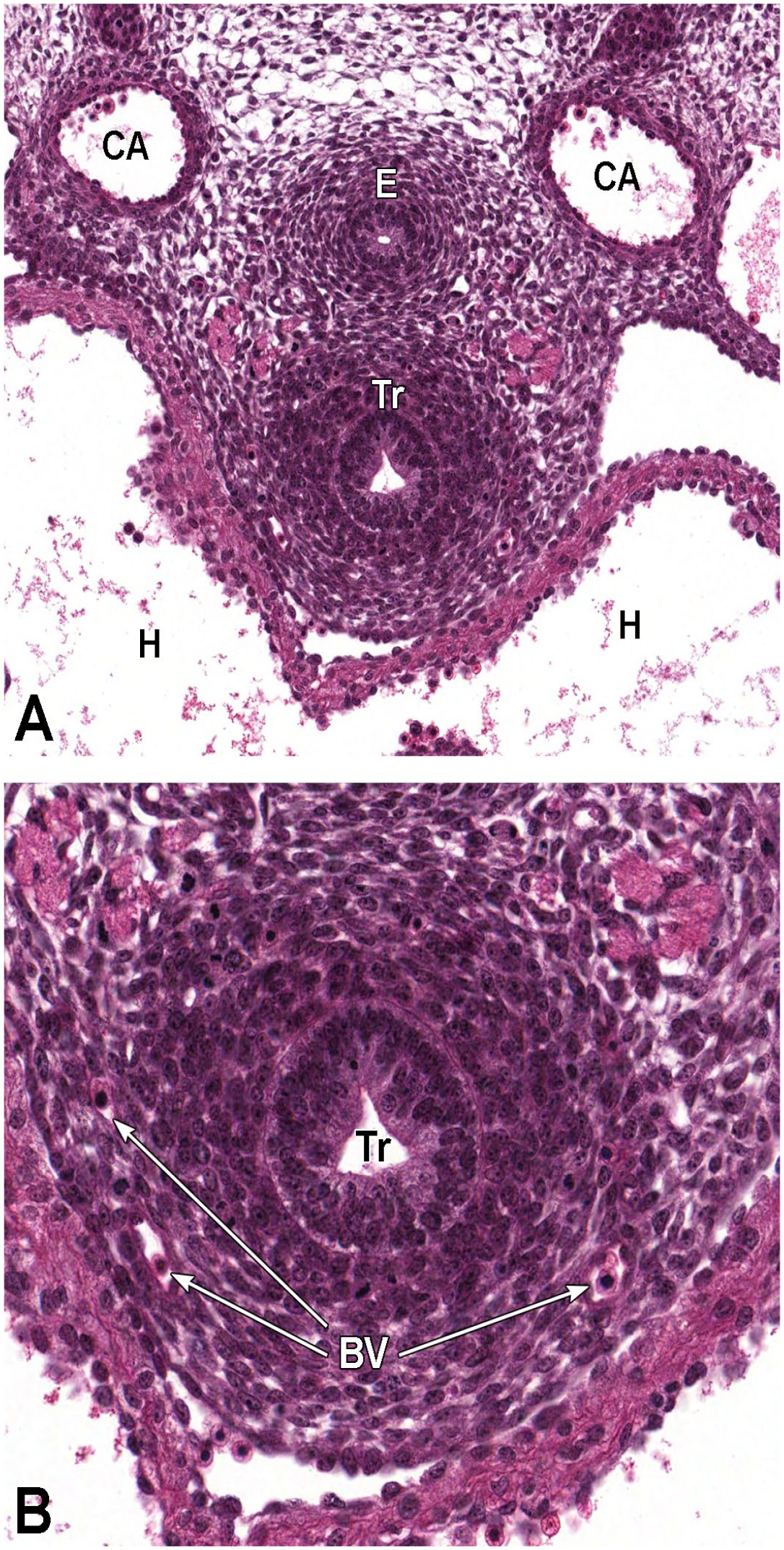
Transverse images of the trachea at E11.5. Panels A (15×) and B (30×) show the trachea with columnar epithelial cells surrounding the lumen. The walls are composed of concentric rings of mesenchymal cells with blood vessels (BV). The esophagus (E), carotid arteries (CA) and heart (H) are shown for orientation. H&E.
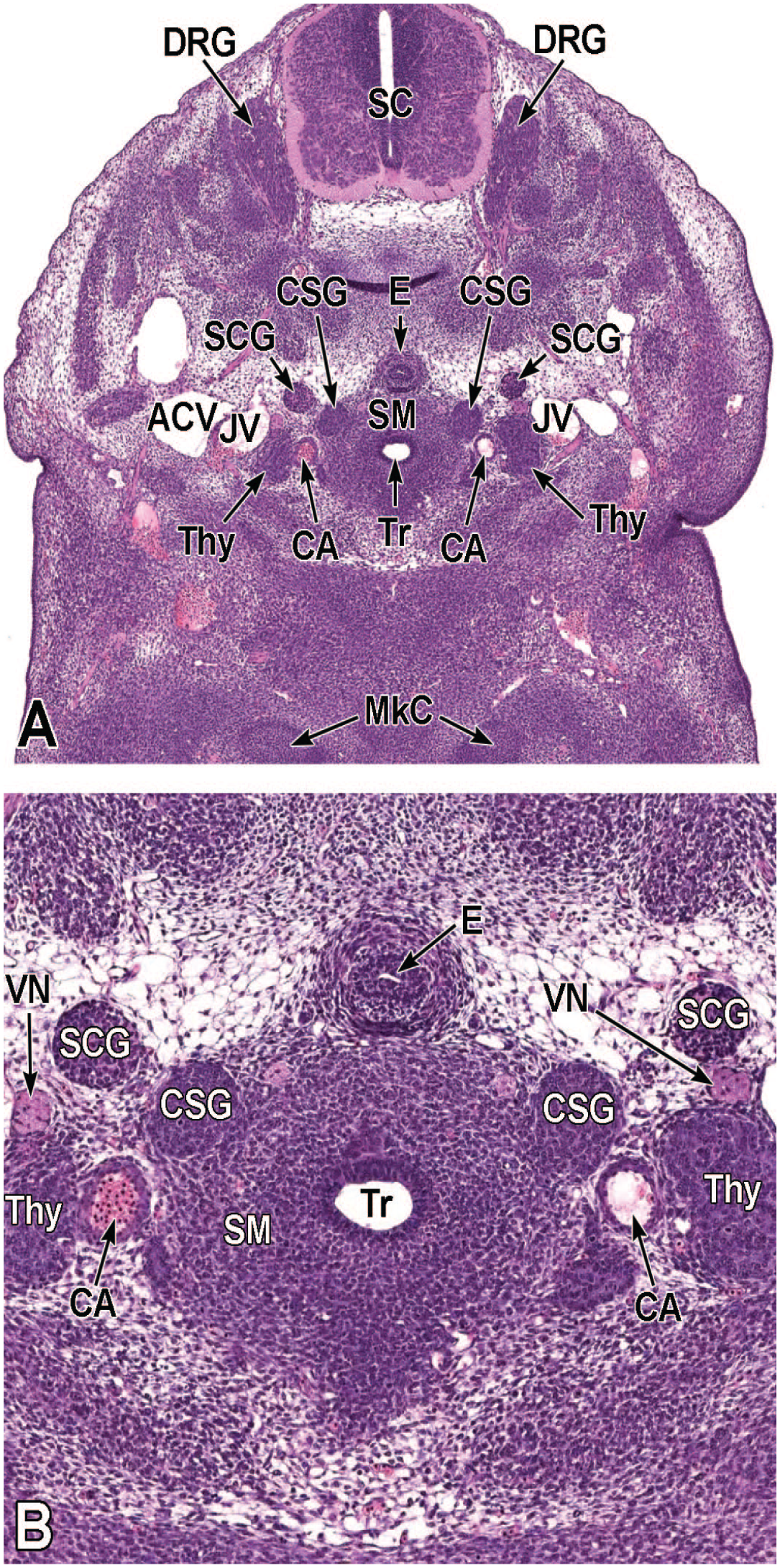
Transverse images of the expanded splanchnic mesenchyme surrounding the trachea at E12.5. Panels A (3.5×) and B (10×) illustrate the splanchnic mesenchyme (SM) surrounding the trachea (Tr), which is located between the right and left carotid arteries (CA). The SM will eventually form the tracheal cartilage rings, ligaments, and smooth muscle of the tracheal wall. Note the fold artifact (dark line in panel A) in the tissue above the esophagus (E). The spinal cord (SC); dorsal root ganglia (DRG); cervical sympathetic ganglia (CSG); sympathetic chain ganglia (SCG); the right and left lobes of the primordial thymus (Thy); anterior (rostral) cardinal vein (ACV); internal jugular veins (JV); right and left vagus nerves (VN); and Meckel’s cartilage (MkC) are labeled for orientation. H&E.
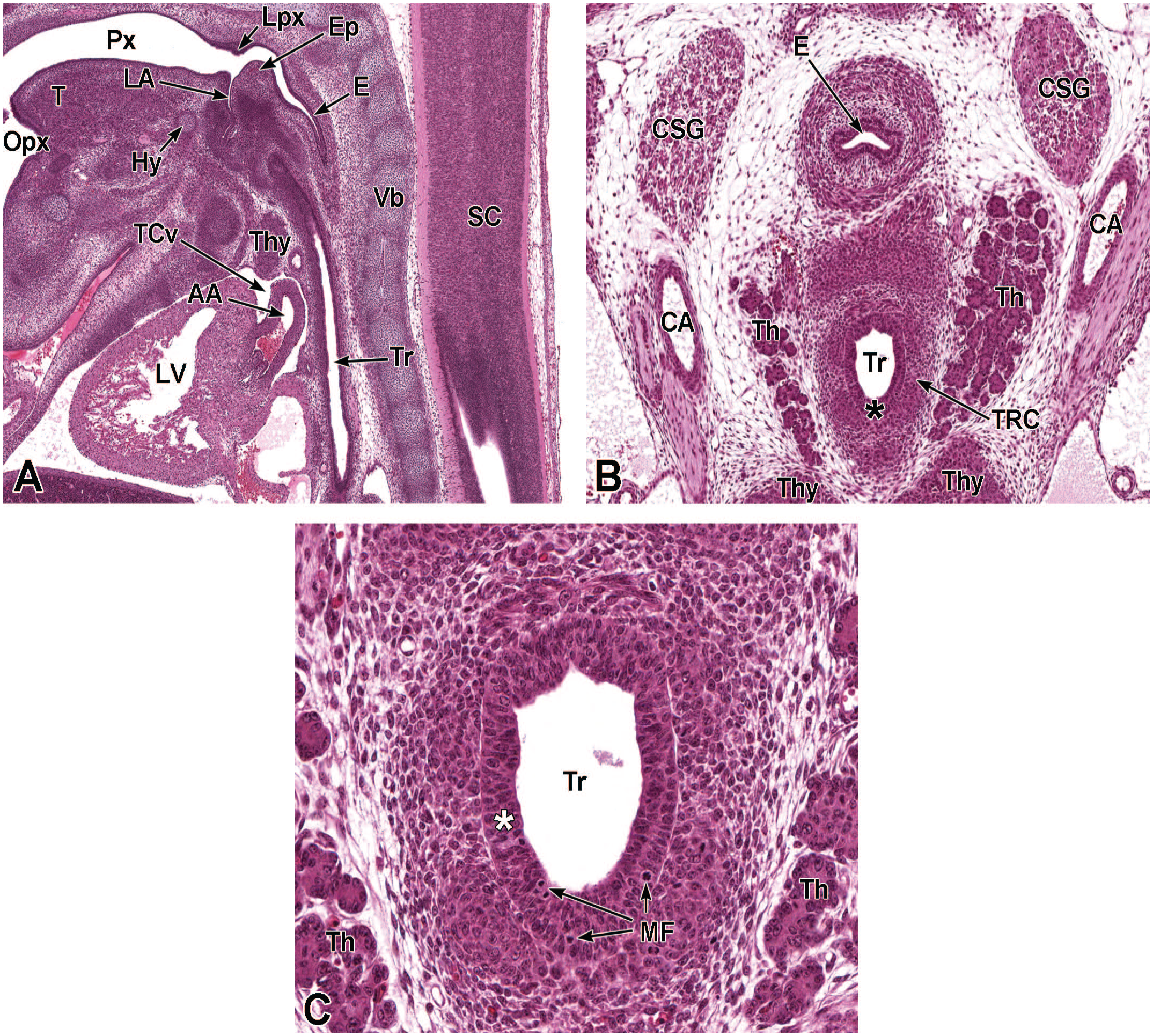
Sagittal and frontal images of the trachea at E13.5. Panel A (sagittal section, 3×), B (7.8×, frontal section), and C (13.5×, frontal section) show the elongating trachea (Tr) and its expanding lumen. At this stage, the trachea is lined by pseudostratified columnar epithelium (asterisks) containing multiple mitotic figures (MF) while the lamina propria and submucosa have not yet developed. The oropharynx (Opx), pharynx (Px), and laryngopharynx (Lpx); laryngeal aditus (LA); epiglottis (Ep); esophagus (E); tongue (T); hyoid bone (Hy); vertebrae (Vb); spinal cord (SC); thoracic cavity (TCv); thymus (Thy); thyroid gland (Th); left ventricle (LV) and aortic arch (AA) of the heart; the right and left cervical sympathetic ganglia (CSG); and the right and left carotid arteries (CA) are labeled for orientation. H&E.
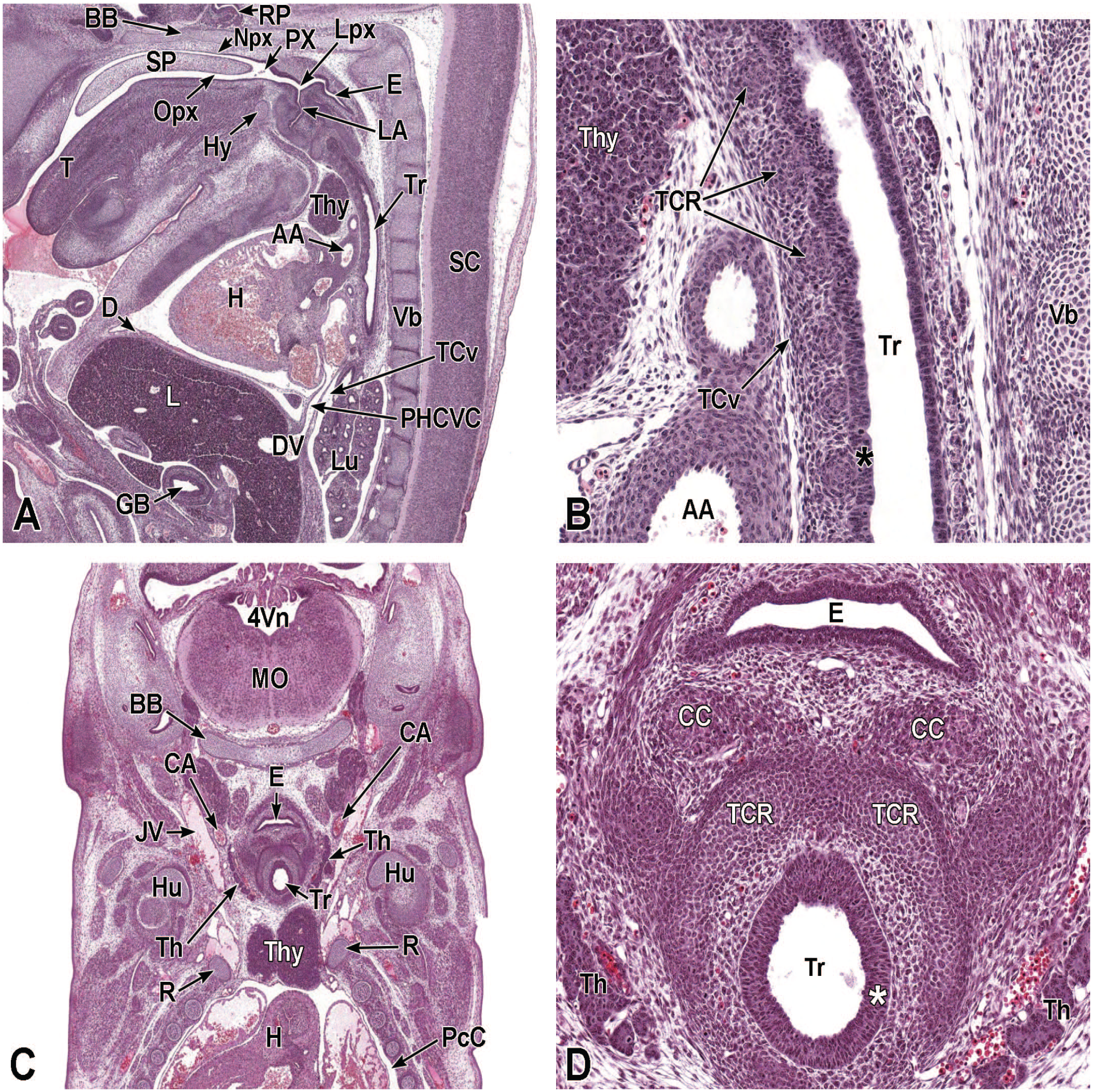
Sagittal and frontal images illustrating tracheal chondrogenesis at E15.5. Panels A (1.8×, sagittal section), B (13.5×, sagittal section), C (2×, frontal section), and D (11.5×, frontal section) show the formation of the tracheal cartilage rings (TCR) surrounding the trachea (Tr). Panels C and D also highlight the cartilage condensations of the cricoid cartilage (CC) and the widening tracheal lumen. The trachea is lined by pseudostratified columnar epithelium (asterisks). The nasopharynx (Npx), soft palate (SP), oropharynx (Opx), pharynx (Px), laryngopharynx (Lpx), laryngeal aditus (LA [entrance to the laryngeal cavity]) and its contiguity (not visible in this section) with the trachea and eventually the lungs (Lu), and entrance to the esophagus (E) form a connected set of passages intersecting at the pharynx. The cartilage primordium of the basisphenoid bone (BB); residual lumen of Rathke’s pouch (RP) within the pituitary primordium; tongue (T); hyoid bone (Hy); thymus (Thy); aortic arch (AA) and pericardial cavity (PcC) of the heart (H); vertebrae (Vb); spinal cord (SC); thoracic cavity (TCv); diaphragm (D); liver (L); gall bladder (GB); ductus venosus (DV); post-hepatic caudal vena cava (PHCVC); fourth ventricle (4Vn); medulla oblongata (MO); right and left lobes of the thyroid gland (Th); right and left common carotid arteries (CA); internal jugular vein (JV); humerus (Hu); and first rib (R) are labeled for orientation. H&E.
Between E16.5 and E18.5, the proportion of luminal cells (i.e., those with an apical surface exposed to the lumen) increases to about 80%, and the apical surface area increases 1.5 times. 93 By E16.0, the C-shaped tracheal cartilage rings are easily identifiable (Figure 40). 88 The ends of the cartilage rings are joined by annular ligaments and smooth muscle, which can contract and expand to control the tracheal diameter. 100 At E16.5, the tracheal lumen is lined with pseudostratified columnar epithelium with scattered patches of ciliated cells and nonciliated goblet cells (Figure 40). At E18.0, the trachea may be seen to extend distally along the midline of the entire cervical region to reach the thoracic inlet, where the carina is located just distal to the arch of the aorta. At E18.5, the tracheal lumen is lined by typical respiratory (ciliated, pseudostratified columnar) epithelium containing a single layer of irregularly shaped and differently sized columnar luminal cells overlaying a layer of flat basal cells (Figure 41). 93 The tracheal hyaline cartilage rings are composed of chondrocytes and are surrounded by a thin layer of perichondrium (Figure 42).
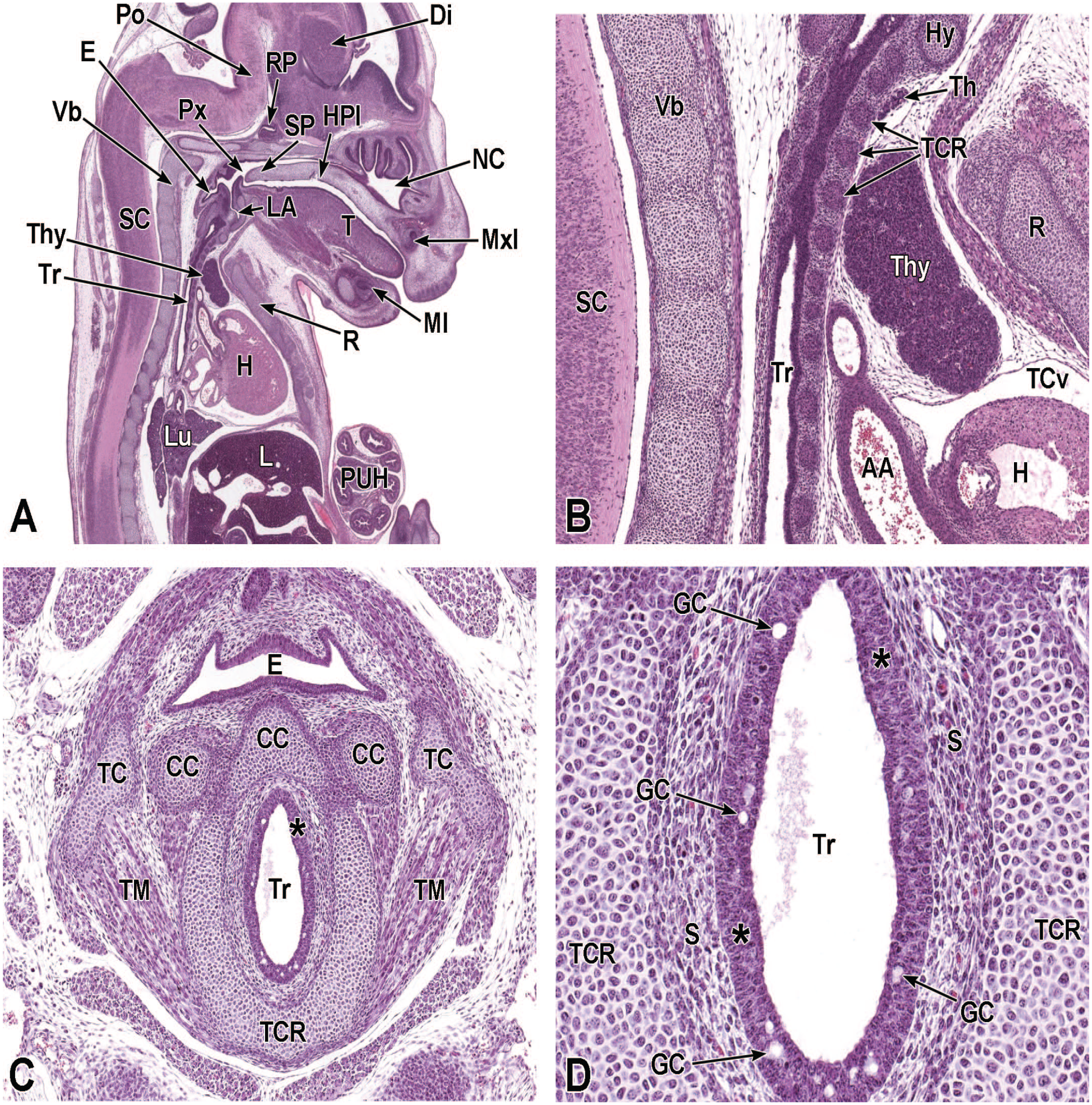
Sagittal and frontal images of tracheal development at E16.5. Panels A (1.0×, sagittal section), B (5.2×, sagittal section), C (7×, frontal section), and D (22×, frontal section) of the proximal trachea (Tr) highlight the distinct tracheal cartilage rings (TCR) that maintain patency of the lumen. The C-shaped TCR are formed of hyaline cartilage and are incomplete dorsally. The trachea lumen is lined by pseudostratified columnar epithelium (asterisks) with scattered patches of ciliated cells as well as non-ciliated goblet cells (GC), which contribute to the mucus layer covering the mucosa. The spinal cord (SC); vertebrae (Vb); pons (Po); diencephalon (Di); Rathke’s pouch (RP); esophagus (E); pharynx (Px); soft palate (SP); hard palate (HPl); nasal cavity (NC); tongue (T); laryngeal aditus (LA); maxillary incisor (MxI) and mandibular incisor (MI); rib (R); lungs (Lu); liver (L); physiological umbilical herniation (PUH); hyoid bone (Hy); thyroid gland (Th); thymus (Thy); thoracic cavity (TCv); proximal region of the aortic arch (AA) of the heart (H); cricoid (CC) and thyroid (TC) cartilages; and thyroarytenoid muscle (TM) and submucosa (S) are labeled for orientation. (Note: Extraneous tissue was removed from area adjacent to the PUH in panel A for optimal viewing of anatomic features.) H&E.
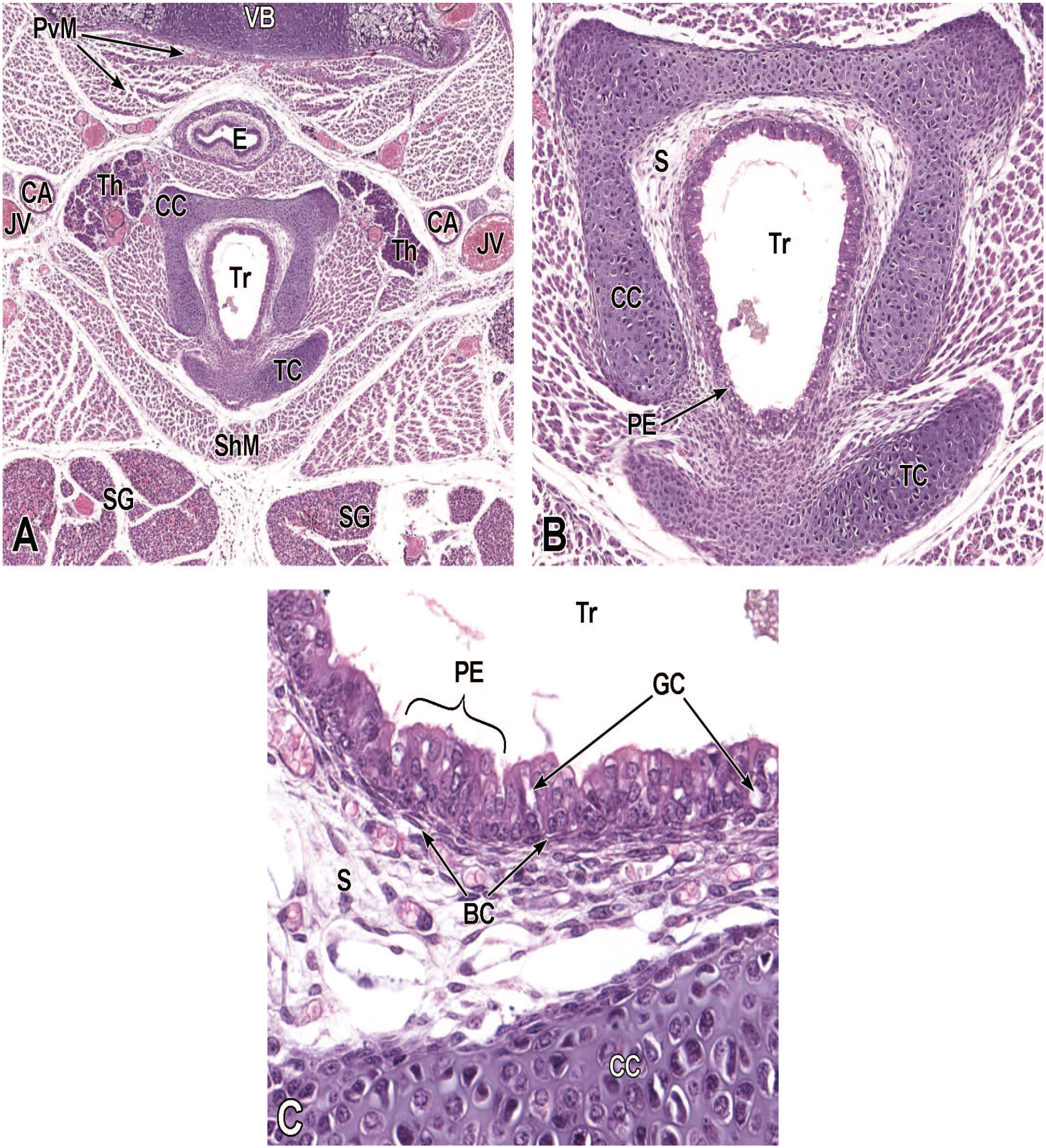
Transverse images highlighting the cranial trachea at E18.5. Panels A (4×) and B (11×) depict the well-defined cricoid (CC) and thyroid (TC) cartilages partially surrounding the trachea (Tr). Panel C (40×) illustrates the tracheal mucosa, which is composed of pseudostratified columnar epithelium (PE) with scattered goblet cells (GC) resting over a layer of flat basal cells (BC) located just above the submucosa (S); the bracket in panel C denotes a patch of ciliated epithelium. A cervical vertebral body (VB); prevertebral muscles (PvM) of the neck; esophagus (E); the right and left common carotid arteries (CA); the right and left internal jugular veins (JV); the right and left lobes of the thyroid gland (Th); sternohyoid muscle (ShM); and salivary glands (SG) are labeled for orientation. H&E.
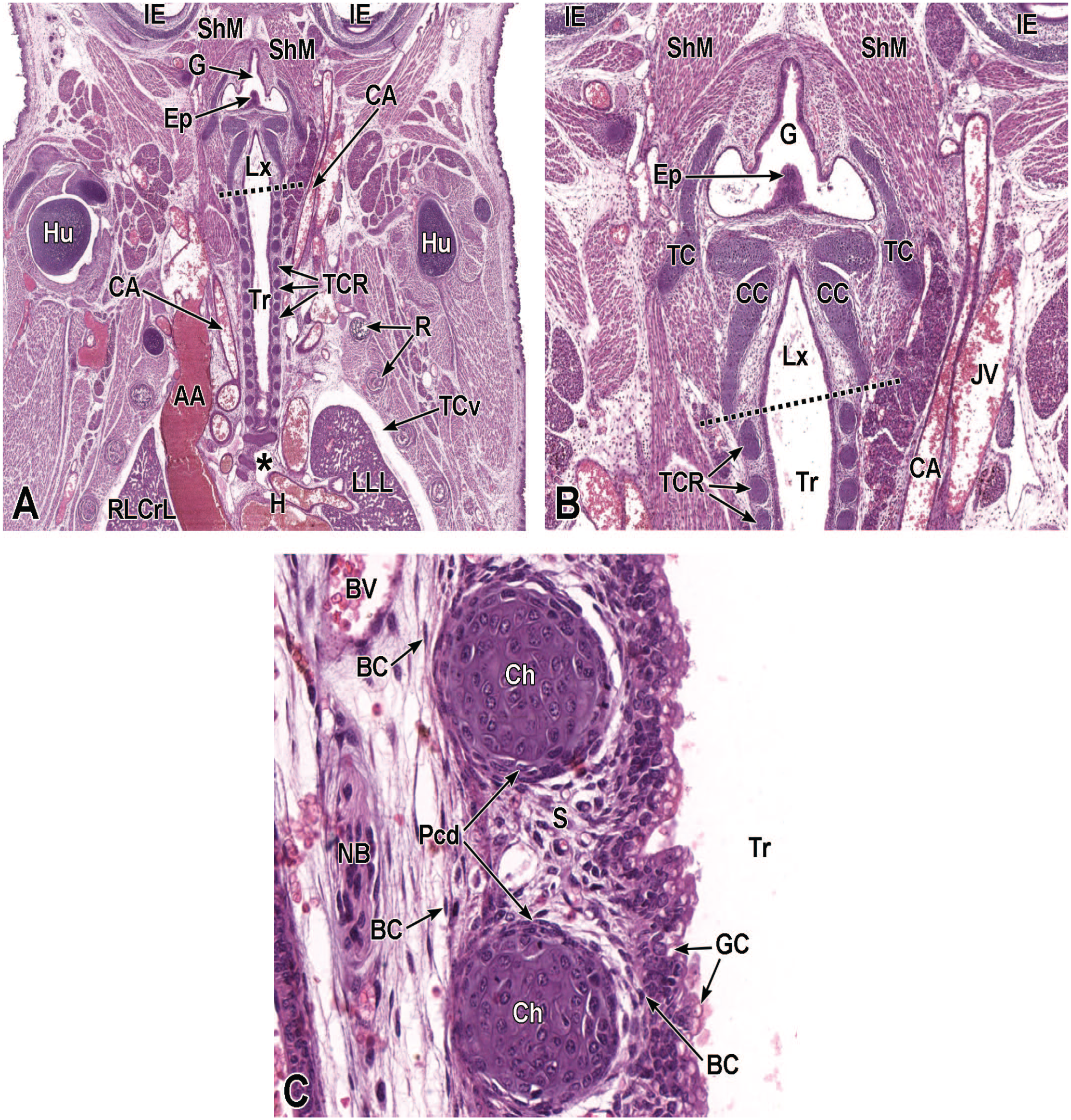
Frontal images of the trachea at E18.5. Panel A (3×) shows the entire trachea (Tr) surrounded laterally by sections of C-shaped hyaline cartilage rings (TCR), extending distally along the midline of the ventral cervical region. The start of the tracheal bifurcation into the right main bronchus (asterisk) is evident at the distal end of the trachea. Panel B (4×) shows the cricoid (CC) and thyroid (TC) cartilages of the larynx (Lx); the dashed line shows the border between the Lx and Tr. Panel C (32×) depicts the hyaline TCRs composed of dense chondrocytes (Ch) surrounded by a thin layer of perichondrium (Pcd). The tracheal lumen is lined by pseudostratified, ciliated columnar epithelium interspersed with non-ciliated secretory (club) cells and scattered mucus-secreting goblet cells (GC), all resting on a deep layer of flat basal cells (BC). The underlying submucosa (S) contains a rich supply of blood vessels (BV) and nerve bundles (NB). The right and left inner ears (IE); glottis (G) and epiglottis (Ep); carotid arteries (CA); internal jugular vein (JV); the right and left humerus (Hu); ribs (R); aortic arch (AA) of the heart (H); thoracic cavity (TCv); right lung cranial lobe RLCrL and left lung lobe (LLL); and sternohyoid muscles (ShM) are labeled for orientation. H&E.
Glands in the Lower Respiratory Tract
The submucosal glands of the lower respiratory tract also participate in regulating airway homeostasis. 114 The murine tracheobronchial submucosal glands are located within the submucosal walls (between the cartilage rings and smooth muscle).37,45,114 The cranial region of the trachea has a high volume of submucosal glands, which are located between the cricoid cartilage of the larynx and first tracheal cartilage ring until the sixth or seventh cartilage rings.114,136 Adult murine tracheobronchial submucosal glands produce a combination of mucous and serous secretions comparable to those produced by nasal submucosal glands. 114
Conducting Airways and Lungs
In the adult mouse, the conducting airways (bronchi and bronchioles) form an intricate tree. Bronchi participate in conducting and warming inspired air while bronchioles conduct air to the alveoli. Unlike many other mammalian species, mice lack terminal respiratory bronchioles that can engage in gas exchange; the short alveolar ducts that connect terminal bronchioles to alveoli may participate in gas exchange. 127 The larger main bronchi possess C-shaped cartilage rings comparable to those of the trachea, but the smaller secondary bronchi lack such rings. The lining of larger bronchi may possess folds that can flatten if the airways expand during intense exertion, but the lining of smaller conducting airways are always flat. Secondary bronchi are lined with a pseudostratified columnar ciliated epithelium, while the tertiary bronchi are lined by a simple thickened cuboidal epithelium.
The lung parenchyma is composed of intricate alveolar fields characterized by multiple differentiated cell types, each with specialized activities that in concert maintain normal lung function and architecture. 71 Lung development (as defined by morphological criteria) after initial specification of the embryo axis encompasses four distinct phases, designated as the pseudoglandular (E9.5-16.5), canalicular (E16.5-17.5), saccular (E17.5-P5), and alveolar (P5-P30) stages. Lung organogenesis has been shown to be instructed by a coordination of genetic, epigenetic, and environmental factors with a coordinated mesenchymal-to-epithelial crosstalk.2,105,177
During the pseudoglandular stage, branching morphogenesis leads to initial formation of the conducting (bronchi and proximal bronchioles) and gas exchange (alveolar ducts) airways. 29 The earliest distinct stage of lung differentiation can be seen at around E9.5 as evagination of the primary lung bud from the ventral foregut (Figure 43). The left and right buds form simple tubes of endodermal epithelium internally, a surrounding layer of splanchnic mesodermal mesenchyme, and an external lining of coelomic epithelium.88,95 The laryngotracheal groove in the foregut indents to form the lumen of the tracheal diverticulum. The distal tracheal diverticulum then divides to form the left and right main bronchi around which the two primary lung buds will elongate and expand. 88 The epithelium of the laryngotracheal groove ranges from cuboidal ventrally to pseudostratified columnar dorsally (Figure 43). At E10.0, the lung buds are slightly larger, extending laterally into the pericardioperitoneal canals and caudally along the ventral aspect of the foregut gastric dilatation. 88 At E10.5, the primary lung buds are caudal to the common atrial chamber of the heart and remain near the esophageal region of the foregut 88 The bronchi are lined by simple cuboidal or columnar epithelium at this time (Figure 44).
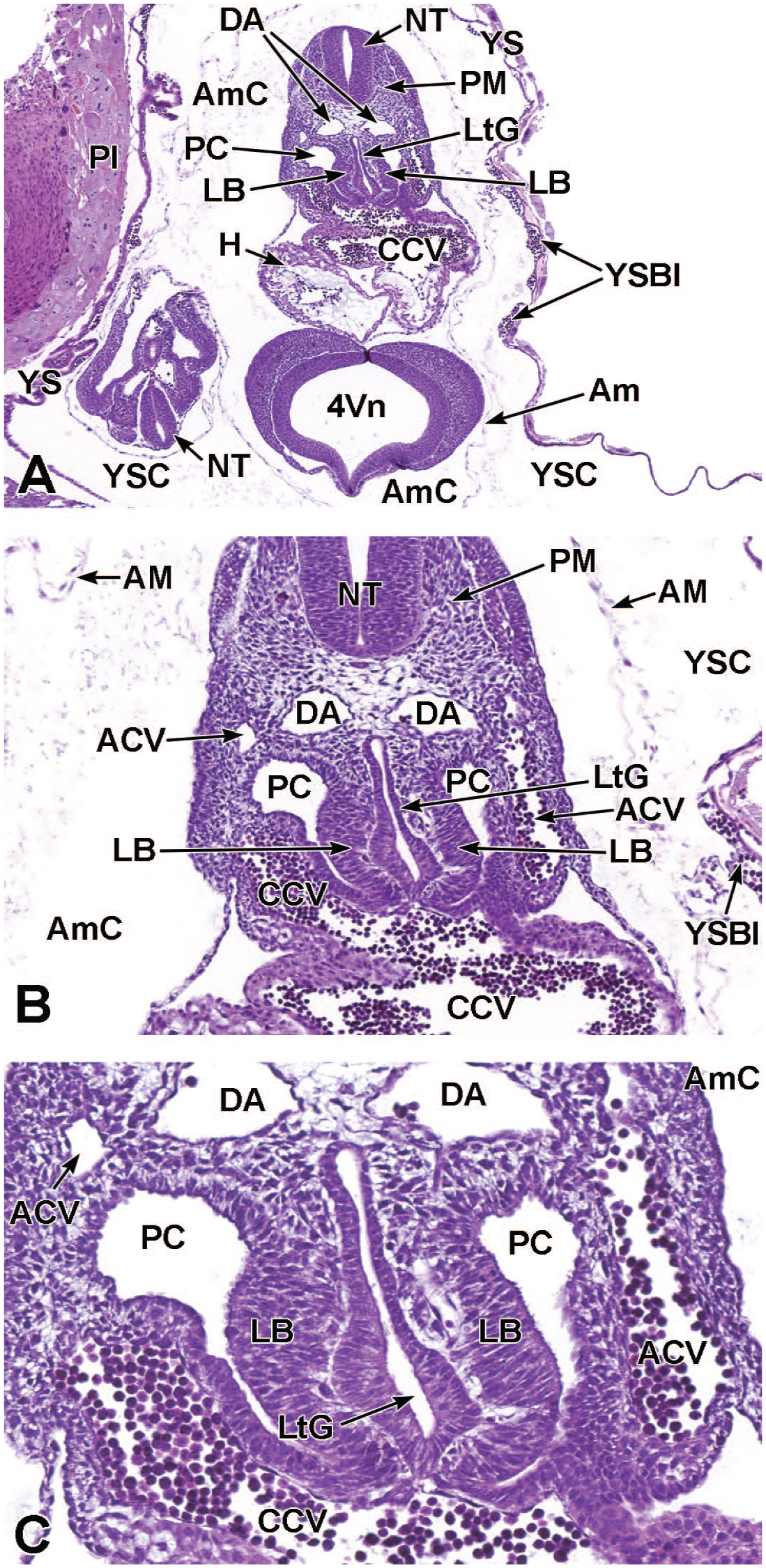
Transverse images of the laryngotracheal groove and early lung buds at E9.5. Panels A (4×), B (10×), and C (20×) demonstrate the right and left lung buds (LB) forming within the splanchnic mesodermal mesenchyme adjacent to the laryngotracheal groove (LtG). Panel C shows that the epithelium lining the LtG varies in conformation from cuboidal ventrally to pseudostratified columnar dorsally. Condensation of paraxial mesoderm (PM) to form a cervical myotome; the left and right dorsal aortas (DA); common cardinal vein (CCV); heart (H); yolk sac (YS); yolk sac cavity (YSC); yolk sac blood islands (YSBI); amnion (Am); amnionic cavity (AmC); fourth ventricle (4Vn); neural tube (NT [at two spinal cord levels]); placenta (Pl); the right and left pericardioperitoneal canals (PC); and the right and left anterior (rostral) cardinal veins (ACV) are labeled for orientation. H&E.
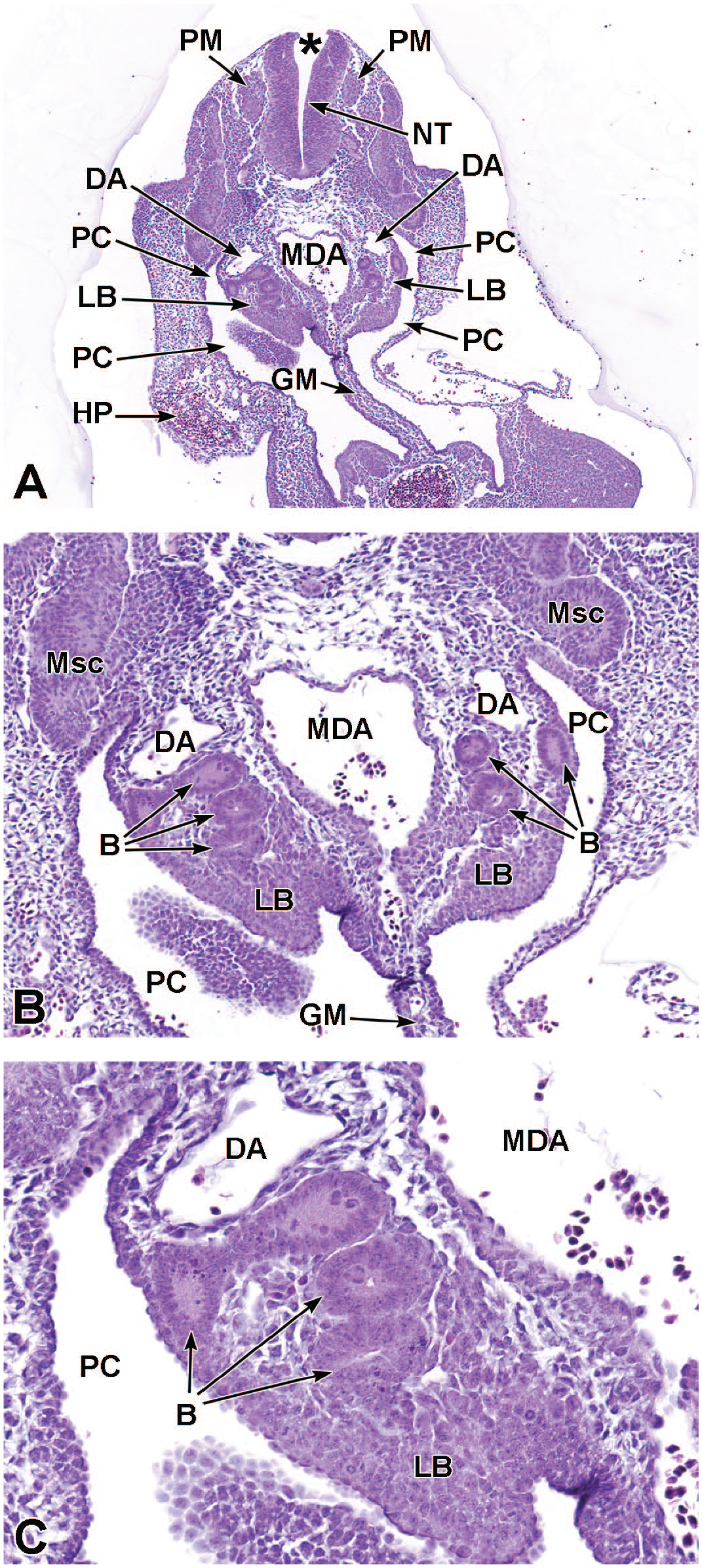
Transverse images for the pseudoglandular stage of lung development at E10.5. Panels A (4×) and B (10×) show the lung buds (LB) extending into the right and left pericardioperitoneal canals (PC). The LB are surrounded by splanchnopleuric mesenchyme and reside on either side of the midline dorsal aorta (MDA). Panel C (20×) highlights the nascent bronchi (B), which are lined by simple cuboidal or columnar epithelium. The neural tube (NT); bilateral condensations of paraxial mesoderm (PM); the right and left dorsal aorta (DA); gut mesentery (GM); hepatic primordium (HP); and condensed mesenchyme (Msc) are labeled for orientation. The asterisk denotes an artifactual separation of the roof of the neural tube. H&E.
At E11.0 to 11.5, the primary lung buds are still small, bulge bilaterally into the pericardioperitoneal canals, and have obtained a more caudal position (Figure 45). 88
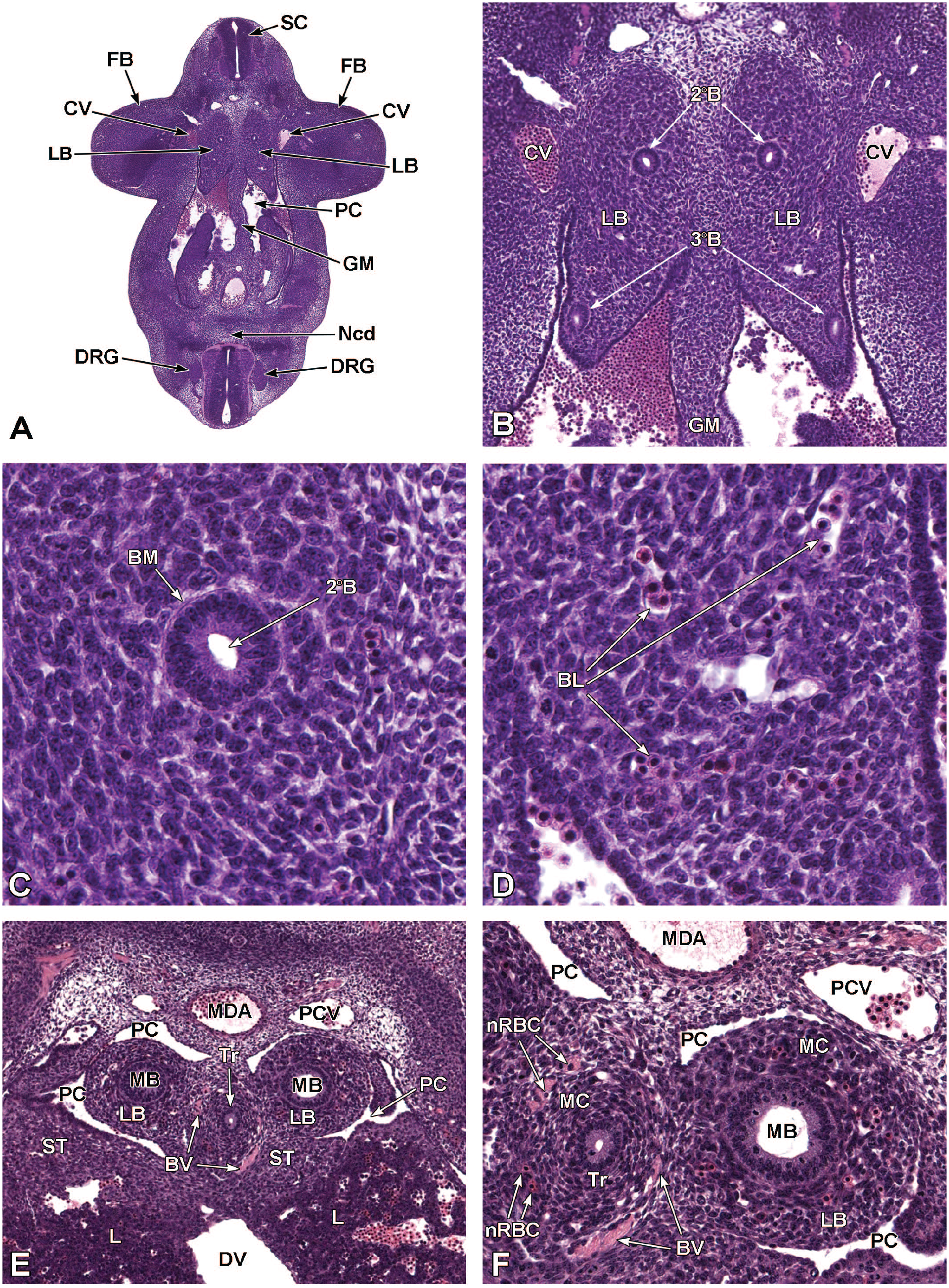
Frontal and transverse sections of the pseudoglandular stage of lung development at E11.5. Panels A (2.5×), B (10×), C (40×), and D (40×) are frontal sections that illustrate the lung buds as they expand into the pericardioperitoneal canal (PC). There are secondary (lobar) bronchi (2°B) and tertiary bronchi (3°B) within the lung buds (LB). Panel C highlights the cuboidal to low columnar epithelium, dense mesenchyme, clearly identified basement membrane (BM), and lack of a basal cell layer characteristic of early bronchi. Panel D demonstrates the high density of blood lakes (BL) filled with nucleated erythrocytes within the mesenchyme. Panels E (18.5x) and F (22×) are transverse sections that depict the trachea (Tr) and left and right lung buds (LB) with their main bronchi (MB, which represent the two initial branches from the distal trachea). The trachea is characterized by columnar epithelial cells surrounding a lumen with multiple layers of outer concentric rings of mesenchymal cells (MC). Blood vessels (BV) can be seen along the outer wall of the trachea and nucleated red blood cells (nRBC) are evident within the LB mesenchyme. The spinal cord (SC); right and left cardinal veins (CV); right and left forelimb buds (FB); gut mesentery (GM); dorsal root ganglia (DRG); notochord (Ncd); midline dorsal aorta (MDA); dorsal (posterior) cardinal veins (PCV); the right and left pericardioperitoneal canals (PC); septum transversum (ST [a precursor for the diaphragm]); liver (L); and ductus venosus (DV) are labeled for orientation. H&E.
At E11.5, the two main bronchi branch from the bifurcated trachea. At this stage, the main bronchi have produced secondary (lobar) bronchi (Figure 45) 88 ; formation of the lobar bronchi is concurrent with evolution of the secondary lung buds, which represent the precursors of the separate lung lobes. Lung buds continue to develop and expand into the pericardioperitoneal canal at E12.0 (Figure 46). Thereafter, the bronchi within the lung buds multiply, expand, and continue to develop (Figure 47). 88 At E13.0, the lumens of the right and left main bronchi have enlarged and are patent throughout their length (Figure 48). The right lung has differentiated into four distinct lobes: cranial, medial, and caudal lobes located within the right thorax and an accessory lobe that originates on the right side but is primarily located to the left of the midline. The left lung has developed as a single lobe and appears smaller than the right (Figure 48). 88 The bronchi are composed of pseudostratified epithelium at this time. At E14.0, mesenchymal condensations of pre-cartilage are present intermittently along the main bronchi, and apically constricted columnar cells predominate in the lung buds.53,88 The lung lobes are fully differentiated and contain numerous primary, secondary, and tertiary bronchi (Figure 49). 88 At E15.0, the right main bronchus (which serves four lobes) is larger and has a luminal diameter that is greater than that of the left main bronchus. It bifurcates into secondary bronchi just before before entering the various lobes of the right lung (Figure 49). The left main bronchus does not bifurcate before or after entering the left lung. The bronchioles continue to branch and become dispersed throughout the lungs with the differentiation of tertiary bronchi and bronchioles. 88 The conducting airways are lined proximally by ciliated respiratory epithelium and distally by cuboidal epithelium that nearly occludes the lumen (a “pseudoglandular” appearance resembling the structure of large exocrine glands). Alveoli have yet to develop.
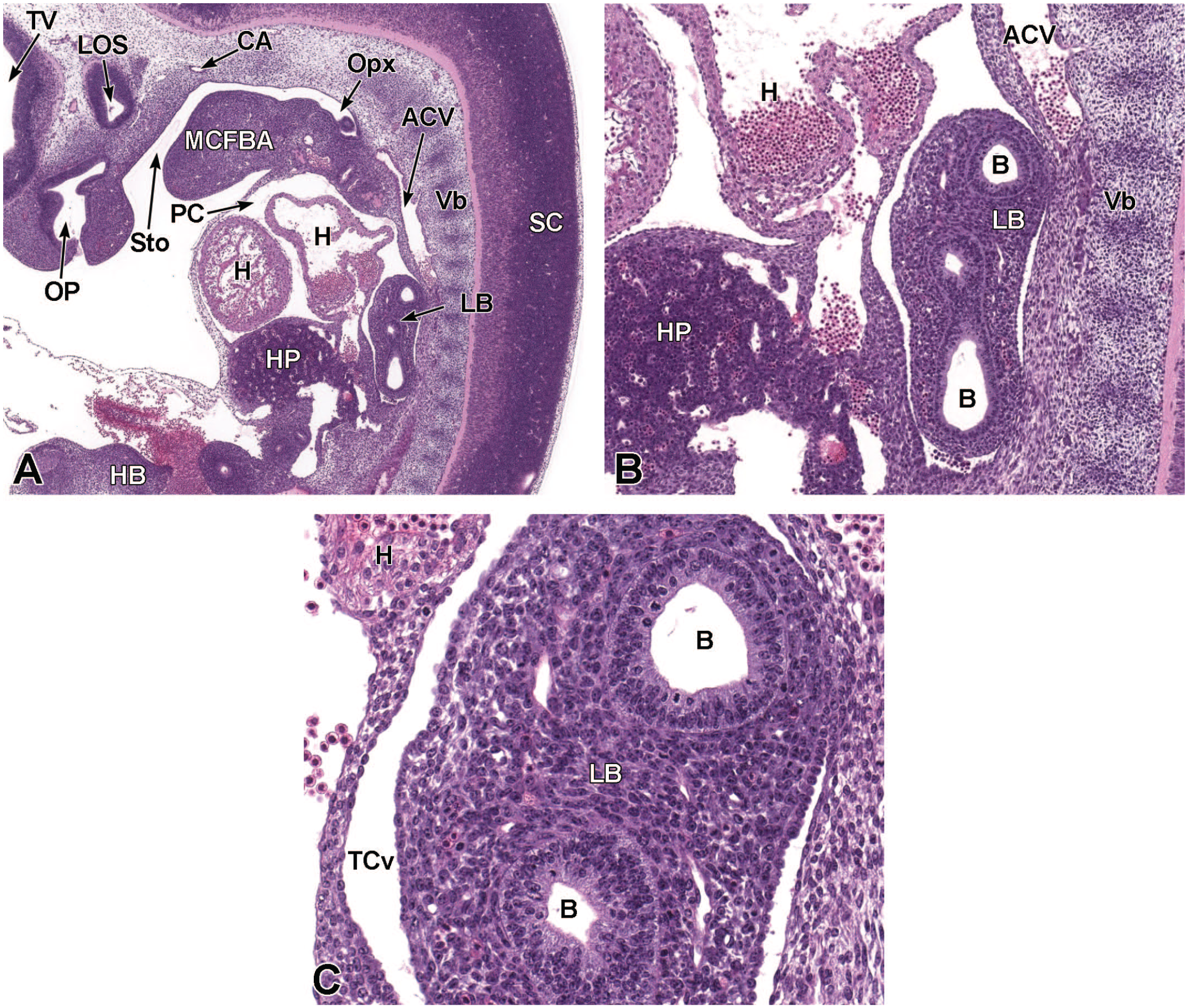
Sagittal images of the pseudoglandular stage of lung development at E12.0. The pseudoglandular appearance results from prominent primary and secondary (lobar) bronchi (B) multiplying in the lung buds (LB). Panels A (3×) and B (8.5×) depict the position of the LB in the pericardioperitoneal canal (PC). Panel C (23.5×) shows that the primary and secondary bronchi have multilayered epithelium surrounded by dense mesenchyme. The olfactory (nasal) placode (OP); lumen of the optic stalk (LOS); internal carotid artery (CA); stomodeum (Sto); oropharynx (Opx); mandibular component of the first branchial arch (MCFBA); anterior (rostral) cardinal vein (ACV); vertebrae (Vb); spinal cord (SC); heart (H), hepatic primordium (HP); hind limb bud (HB); thoracic cavity (TCv); and telencephalic vesicle (TV) are labeled for orientation. H&E.
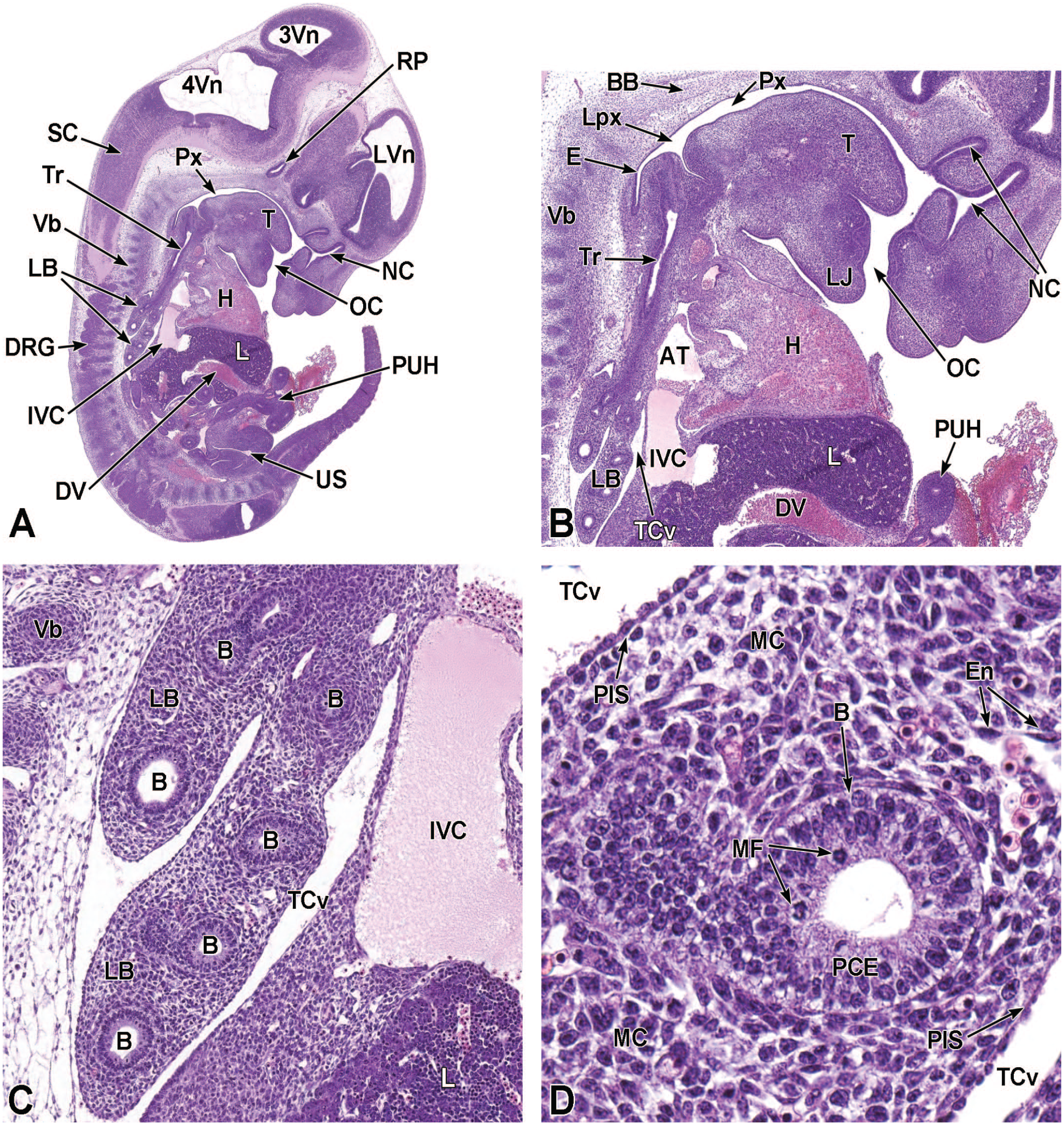
Sagittal images illustrating the pseudoglandular stage of lung development at E12.5. Panels A (1.2×), B (3×), and C (10×) illustrate the right and left lung buds (LB) arising from the caudal end of the trachea (Tr). Continued branching morphogensis during this stage forms the primary, secondary, and tertiary bronchi (B). Panel D (40×) shows a developing bronchus lined by pseudostratified ciliated epithelium (PCE) containing mitotic figures (MF). The surrounding tissue is composed of mesenchymal cells (MC) with many vascular structures lined with endothelial cells (En) and filled with nucleated erythrocytes. The lung bud pleural surfaces (PlS) are covered by a primitive pleural lining. The lateral, third, and fourth ventricles (LVn, 3Vn, and 4Vn, respectively) of the brain; Rathke’s pouch (RP); laryngopharynx (Lpx); pharynx (Px); esophagus (E); tongue (T); nasal cavity (NC); oral cavity (OC); heart (H); thoracic cavity (TCv); post-hepatic part of the inferior (caudal) vena cava (IVC); liver (L); ductus venosus (DV); physiological umbilical herniation (PUH); urogenital sinus (US); dorsal root ganglia (DRG); vertebrae (Vb); spinal cord (SC); cartilage anlage of the basisphenoid bone (BB); lower jaw (LJ); and lumen of the heart atrium (At) are labeled for orientation. H&E.
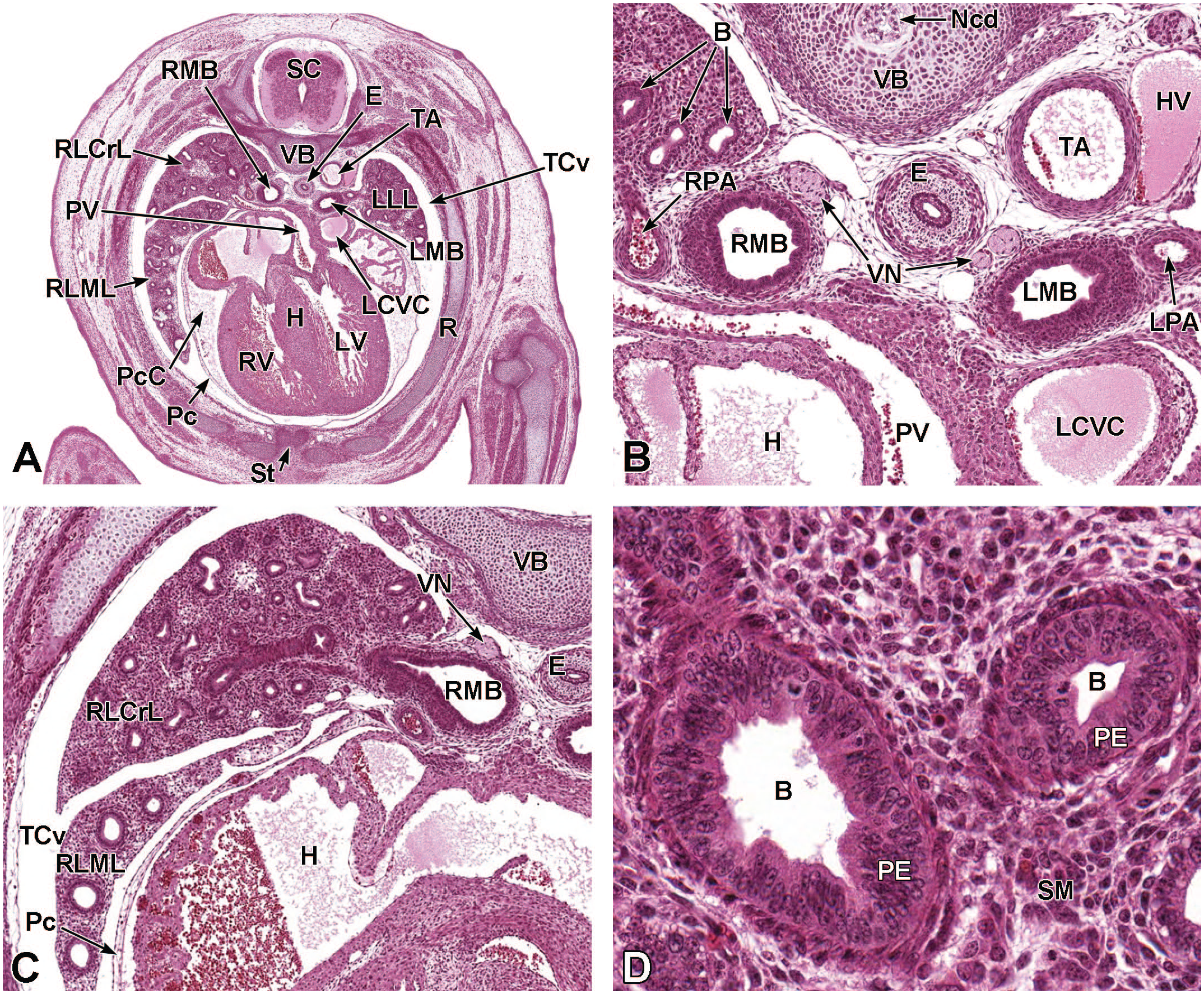
Transverse images illustrating the pseudoglandular stage of lung development at E13.5. Panel A (2×) shows the location and size of the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), left lung lobe (LLL), and right and left main bronchi (RMB and LMB, respectively) located on either side of the esophagus (E). Panel B (10×) shows the expanding lumens of the right and left main bronchi as well as the pulmonary vein (PV) extending from the RLCrL to the left ventricle (LV) of the heart (H). Panel C (6×, different section) depicts the right main bronchus entering the RLCrL. Panel D (40×) demonstrates two bronchi (B) lined by pseudostratified epithelium (PE) and surrounded by splanchnic mesoderm (SM). The spinal cord (SC); vertebral body (VB); thoracic aorta (TA); hemiazygos vein (HV); thoracic cavity (TCv); rib (R); left cranial vena cava (LCVC); pericardium (Pc); pericardial cavity (PcC); right ventricle (RV) of the heart; xyphoid process of the sternum (St); notochord (Ncd); the right and left pulmonary arteries (RPA and LPA, respectively); and right and left vagus nerves (VN) are labeled for orientation. H&E.
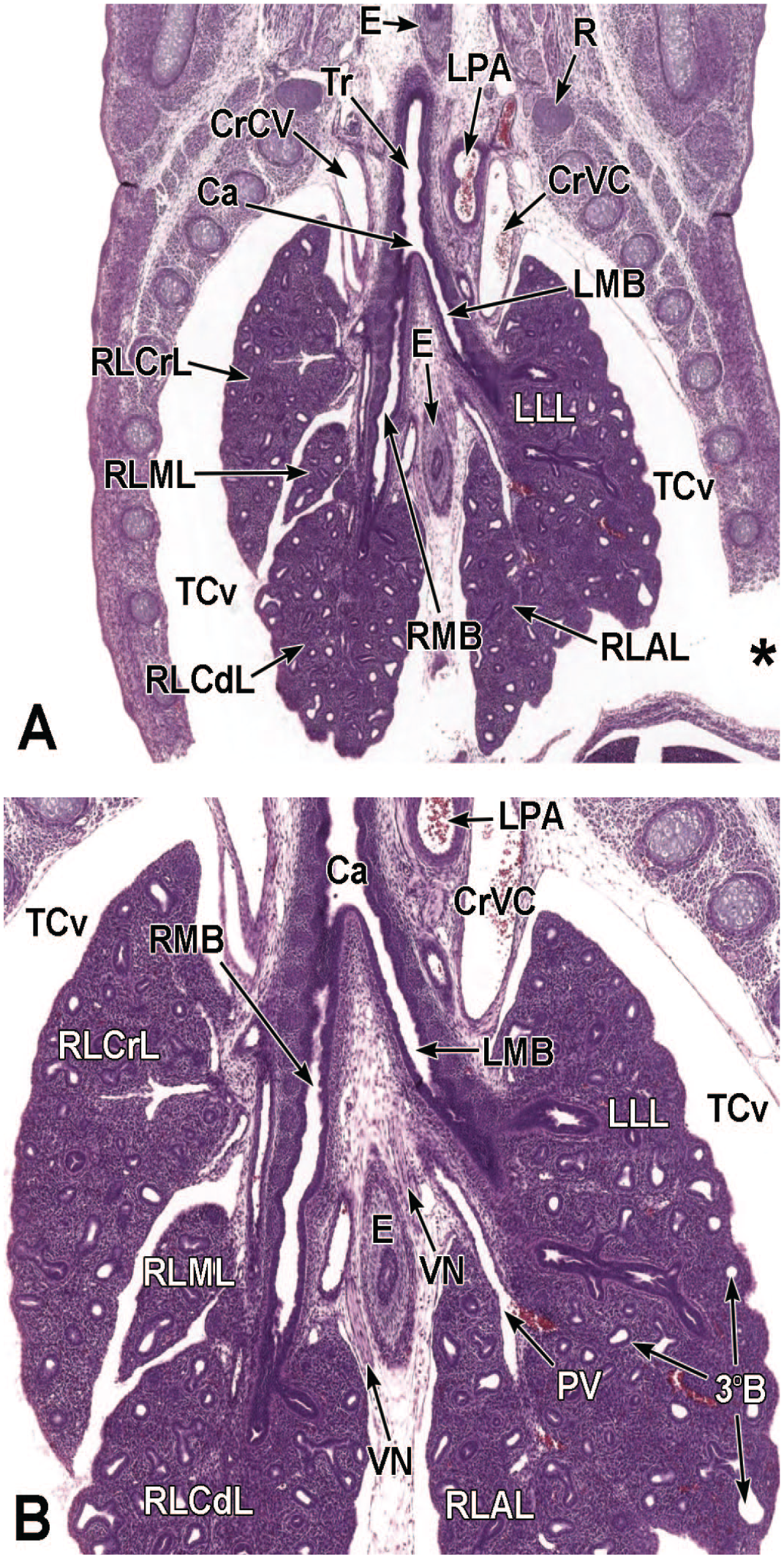
Frontal images of the pseudoglandular stage of lung development at E14.5. Panels A (2.5×) and B (4×) show that the trachea (Tr) bifurcates at the carina (Ca) to produce the right and left main bronchi (RMB and LMB, respectively). The right main bronchus can be seen entering the right lung caudal lobe (RLCdL) while the left main bronchus is entering the left lung lobe (LLL). As the main bronchi enter their respective lung lobes, further bifurcation forms the lobar (secondary) and tertiary bronchi (3°B). The esophagus (E); first rib (R); left pulmonary artery (LPA); thoracic cavity (TCv); the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), and right lung accessory lobe (RLAL); pulmonary vein (PV); cranial vena cava (CrVC); and right and left vagus nerves (VN) are labeled for orientation. Note the artifactual separation (asterisk) in the region of the left thorax. H&E.
At E16.5, the canalicular stage begins, and the histological appearance of the lungs changes considerably. Between E15.5 and E17.0, the volume of the thoracic cavity increases, but the volume occupied by lung tissue does not increase at the same rate. 88 The higher volume of the thoracic cavity allows the lungs to eventually expand. During this stage, the conducting airways in the lung continue to develop as the bronchi and bronchioles expand and lengthen (Figure 50) while alveolar ducts form and narrow.88,176 Club (formerly Clara) cells and ciliated cells are now present throughout the conducting airways, marking a distinction between bronchiolar and alveolar cell lineages.169,176 By this stage, vascular endothelial growth factor (VEGF) production has completely shifted from mesenchyme surrounding the airways to the epithelium lining the terminal air sacs (primitive alveoli). 111 This shift results in a stronger interaction between the epithelium and endothelium and causes the differentiation of the epithelial progenitor cells into alveolar type I (ATI) and alveolar type II (ATII) cells.30,111 The squamous ATI cells line most of the inner surface of the alveoli and contribute to the thin air-blood barrier, thus providing the first epithelium specialized for efficient gas exchange. The cuboidal ATII cells are dispersed intermittently in the alveoli where they secrete surfactant and serve as stem cells for replenishing ATI cells.13,111 Pulmonary lipofibroblast cells emerge during this stage as important contributors to alveolar septal development and surfactant production.30,115 Once the ATI cells and capillary endothelial cells closely contact each other, they generate a common basement membrane constituted by the fusion of endothelial- and epithelial-derived basement membranes. 111 This process is explained further in the “Lung Vasculogenesis and Angiogenesis During Gestation” section below.
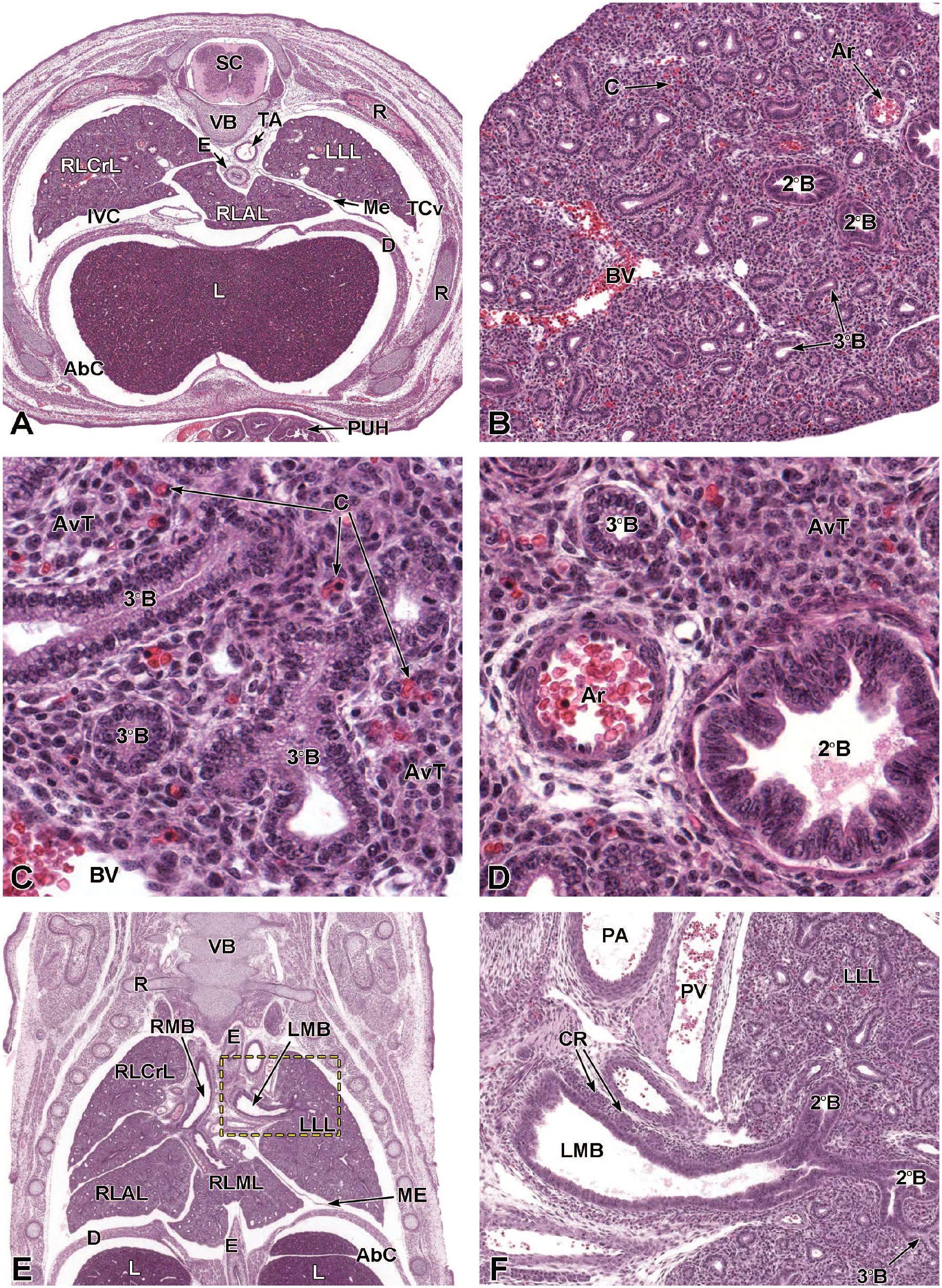
Transverse and frontal images illustrating the canalicular stage of lung development at E16.5. Panel A (2×, transverse) depicts the location and size of the right lung cranial lobe (RLCrL), right lung accessory lobe (RLAL), and left lung lobe (LLL). At this stage, the cell types within the bronchi and the stronger interaction between the epithelium and endothelium result in differentiation of the epithelial progenitor cells into alveolar type I and type II cells. However, distinct alveoli are still not visible at this stage. Panels B (10×, transverse), C (40×, transverse), and D (40×, transverse) show secondary (lobar) bronchi (2°B) lined by pseudostratified columnar, ciliated epithelium (note the lack of goblet cells at this stage) and tertiary bronchi (3°B) lined by simple cuboidal epithelium surrounded by nascent alveolar tissue (AvT). The artery (Ar) in panel D is an elastic pulmonary artery with a tunica media comprised of concentric elastic laminae mixed with smooth muscle. Panel E (2×, frontal) depicts the left lung lobe, right lung cranial lobe, right lung middle lobe (RLML), right lung accessory lobe, and right and left main bronchi (RMB and LMB, respectively). Panel F (10×, frontal view of the field in the dashed box in panel E) illustrates the left main [primary] bronchus, which is encapsulated by cartilage rings (CR), branching into and secondary and tertiary bronchi. The pulmonary artery (PA) and pulmonary vein (PV) are descending into the left lung. The spinal cord (SC); vertebral body (VB); thoracic aorta (TA); esophagus (E); mediastinum (Me); diaphragm (D); origin of post-hepatic component of inferior (caudal) vena cava (IVC); rib (R [osseous part dorsally and cartilaginous part ventrally in panel A]); physiological umbilical herniation (PUH); liver (L); abdominal cavity (AbC); thoracic cavity (TCv); blood vessel (BV); and capillaries (C) are labeled for orientation. H&E.
At E17.5, the saccular stage begins (Figure 51) and continues through the early postnatal period to approximately P5. The transition from the canalicular stage to the saccular stage is imperative for survival after birth. This stage is characterized by primary saccule (also called alveolar sacs, primary septa, and primitive alveoli) formation (including a patent central lumen), surfactant production, capillary system growth and expansion, and mesenchymal thinning in preparation for efficient gas exchange.29,111 The lung volume expands between E17.5 and E18.0 when the alveoli begin to develop in earnest. The lung airways are patent from the carina to the alveoli, albeit filled with ingested amniotic fluid and bronchial secretions. 174 Between E17.5 and E18.0, the cells in the lungs are identical to those seen at birth except for the presence of undifferentiated ATI cells. 88 These cells differentiate from cuboidal to squamous (flattened) conformation between E18.5 and E19.0, allowing for future gas exchange to occur between the alveolar spaces and the lung capillaries once breathing begins at birth.74,88 The difference in developmental stages among mice of a given litter can result in some mice with primary saccules bordered by cuboidal cells, whereas more developmentally mature mice may contain primary saccules lined by squamous cells. Around E18.0, development of the gas exchange interface is increased, which reflects an upswing in differentiation of both alveolar ducts and primary saccules (Figure 52). The saccular stage continues to P5, after which the alveolar stage begins and continues to around P30. The alveolar and saccular stages of lung development are further described below in the Postnatal Development section.
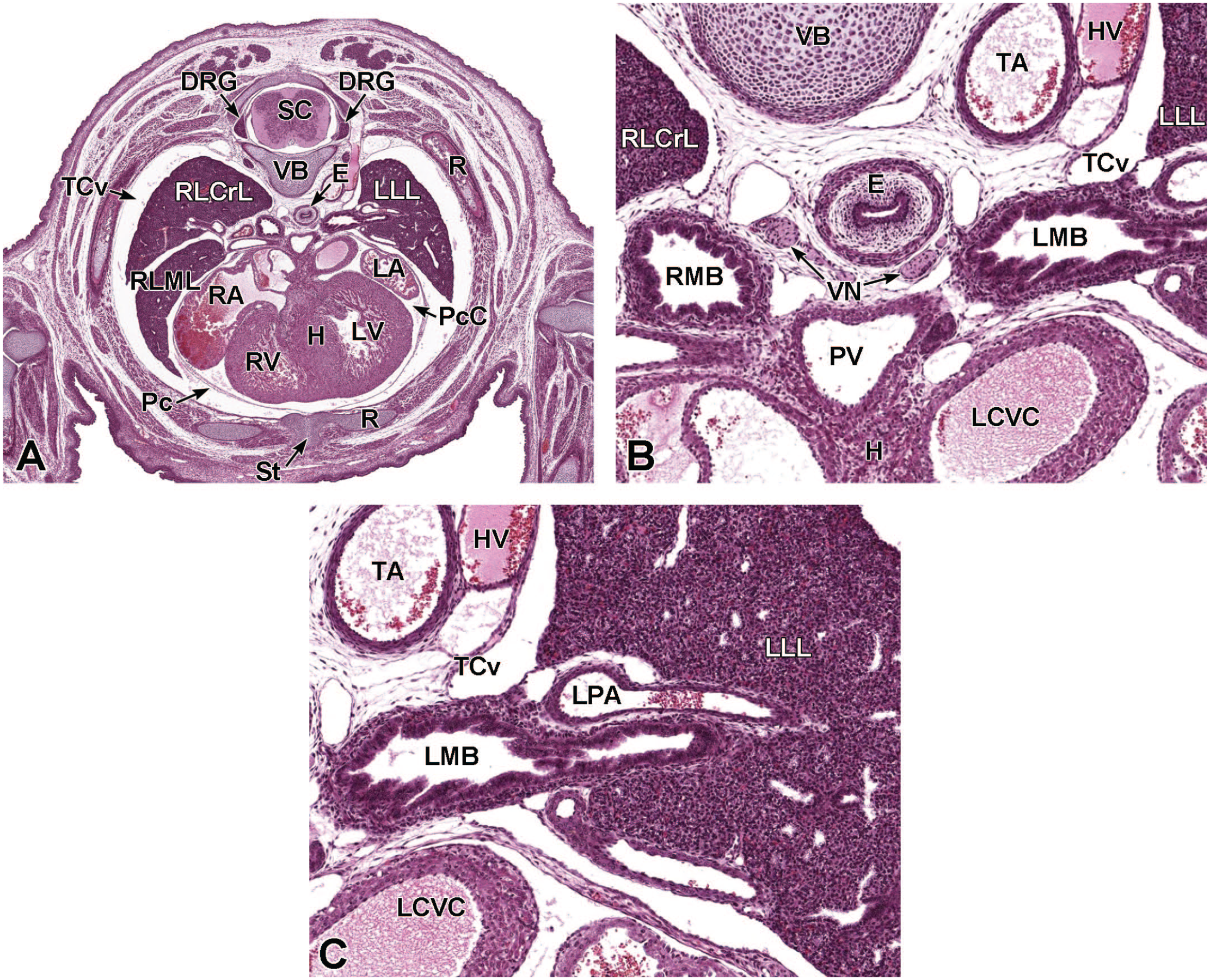
Transverse images illustrating the saccular stage of lung development at E17.5. Panel A (2×) shows the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), and the left lung lobe (LLL). Panel B (8.7×) illustrates the right and left main bronchi (RMB and LMB, respectively) on either side of the esophagus (E); panel C (10×) depicts the left main bronchus and the left pulmonary artery (LPA) entering the LLL. The alveolar primary saccules have not yet formed at this stage. The spinal cord (SC); dorsal root ganglia (DRG); vertebral body (VB); the right and left ventricles (RV and LV, respectively) and right and left atria (RA and LA, respectively) of the heart (H), pericardium (Pc), pericardial cavity (PcC), and left cranial vena cava (LCVC); thoracic cavity (TCv); xyphoid process of the sternum (St); rib (R); thoracic aorta (TA); hemiazygos vein (HV); pulmonary vein (PV); and right and left vagus nerves (VN) are labeled for orientation. H&E.
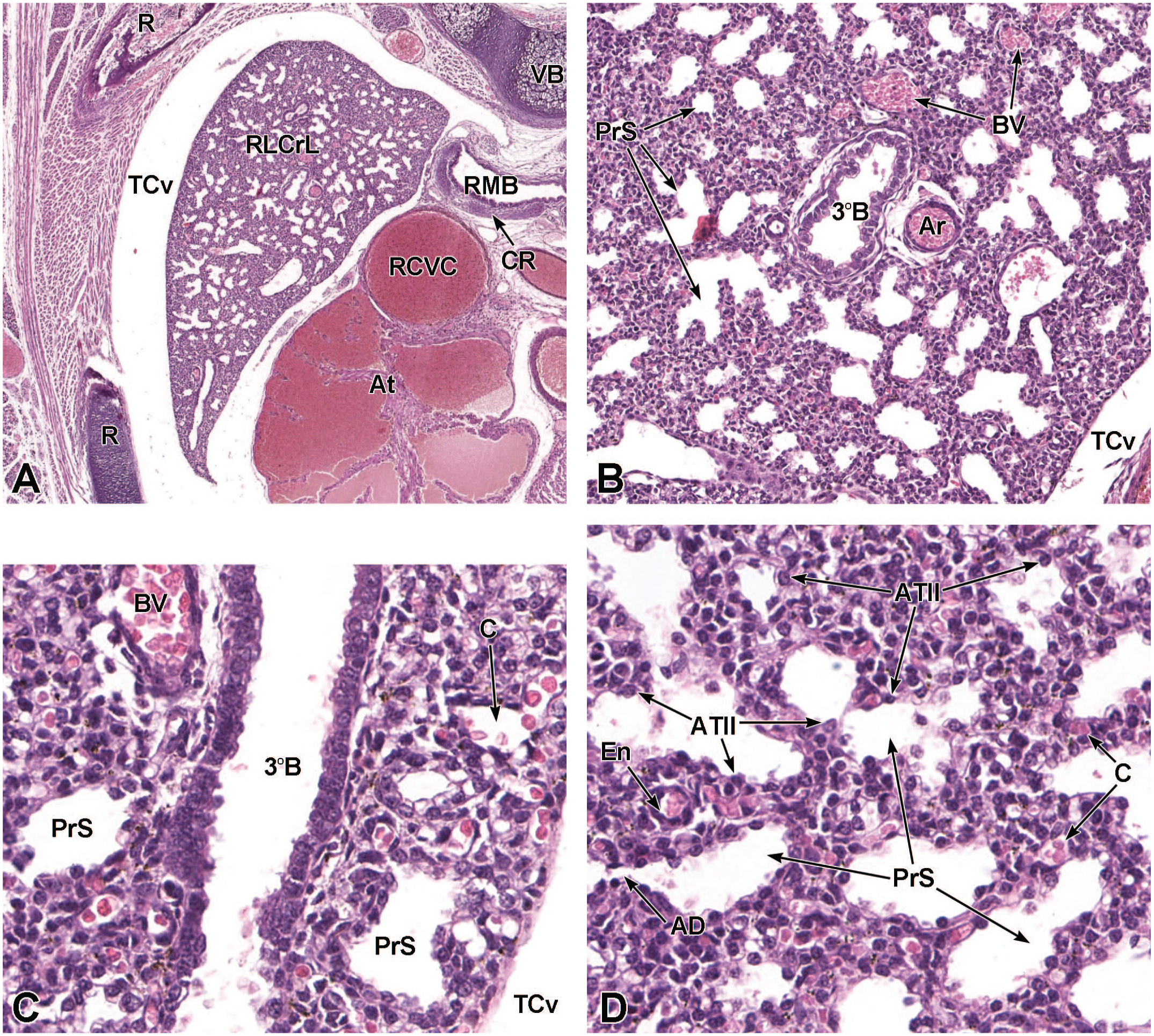
Transverse images of the saccular stage of lung development at E18.5. Panel A (3.5×) shows the right lung cranial lobe (RLCrL) and the right main bronchus (RMB) surrounded by cartilage rings (CR). Panel B (14.5×) depicts a tertiary bronchus (3°B) with a thick cuboidal epithelial lining surrounded by primary saccules (PrS), an artery (Ar), and blood vessels (BV). Primary saccules (also called alveolar sacs and primitive alveoli) are larger than alveoli and function as the main gas exchange units at birth. Panel C (40×) illustrates a longitudinal section of a tertiary bronchus lined by thick cuboidal epithelium. Panel D (36×) shows that only a small portion of the cuboidal alveolar type 2 (ATII) cells line the primary saccules. Capillaries (C) lined by endothelial cells (En) align in close proximity to the primitive alveolar epithelium. Alveolar ducts (AD) are first present at this stage. The vertebral body (VB); ribs (R); thoracic cavity (TCv); right cranial vena cava (RCVC) and atrium (At) of the heart are labeled for orientation. H&E.
Lung Vasculogenesis and Angiogenesis During Gestation
Formation of the vascular niche in the developing lung begins with vasculogenesis and angiogenesis. Vasculogenesis is the differentiation of precursor cells (hemangioblasts and angioblasts) into endothelial cells and blood vessels with the subsequent de novo formation of a primitive vascular network. 29 Angiogenesis, defined as the growth of new vessels from preexisting vasculature, follows with endothelial cell differentiation into capillaries. 22 Using a variety of resources, including vascular casts and electron microscopy of murine lungs, deMello and colleagues determined that two processes are involved in pulmonary blood vessel formation: Central vessels are formed by angiogenesis as extensions of larger vessels tracking along the lobar bronchial trees while peripheral (parenchymal) vessels are formed by vasculogenesis via blood lakes in the mesenchyme. 41
Vasculogenesis and angiogenesis begin in the lung mesenchyme at around E9.5 during the pseudoglandular stage.29,41 At this time, the lung vasculature is initiated as a part of the splanchnic plexus surrounding the esophagus and conducting airways; these early vessels contain primitive (yolk sac-derived) nucleated erythrocytes. 41 The mesenchyme around the lung buds becomes sparse in density and begins to express VEGF, after which intercellular spaces form (Figure 53).41,111 In response to VEGF, hemangioblasts (a subset of mesenchymal cells that serve as precursors for both hematopoietic and endothelial cells 20 ) begin to form blood lakes 41 within the mesenchyme near the distal end of the epithelial buds (Figure 53).111,186 Blood lakes will become sinusoidal spaces (where sinusoids are tubular channels, slightly larger than and lacking the connective tissue scaffold characteristic of true capillaries) that consist of an outer discontinuous layer of thin endothelial cells and an inner layer of hematopoietic cells.1,111 In addition, during this stage, the primitive lungs show an increase in intercellular spaces lined with flattened, spindle-shaped cells. 41 Two vascular tubes, the right and left pulmonary arteries, can be seen running adjacent to the trachea while smaller vessels can be seen branching from these larger vessels via angiogenesis.58,111
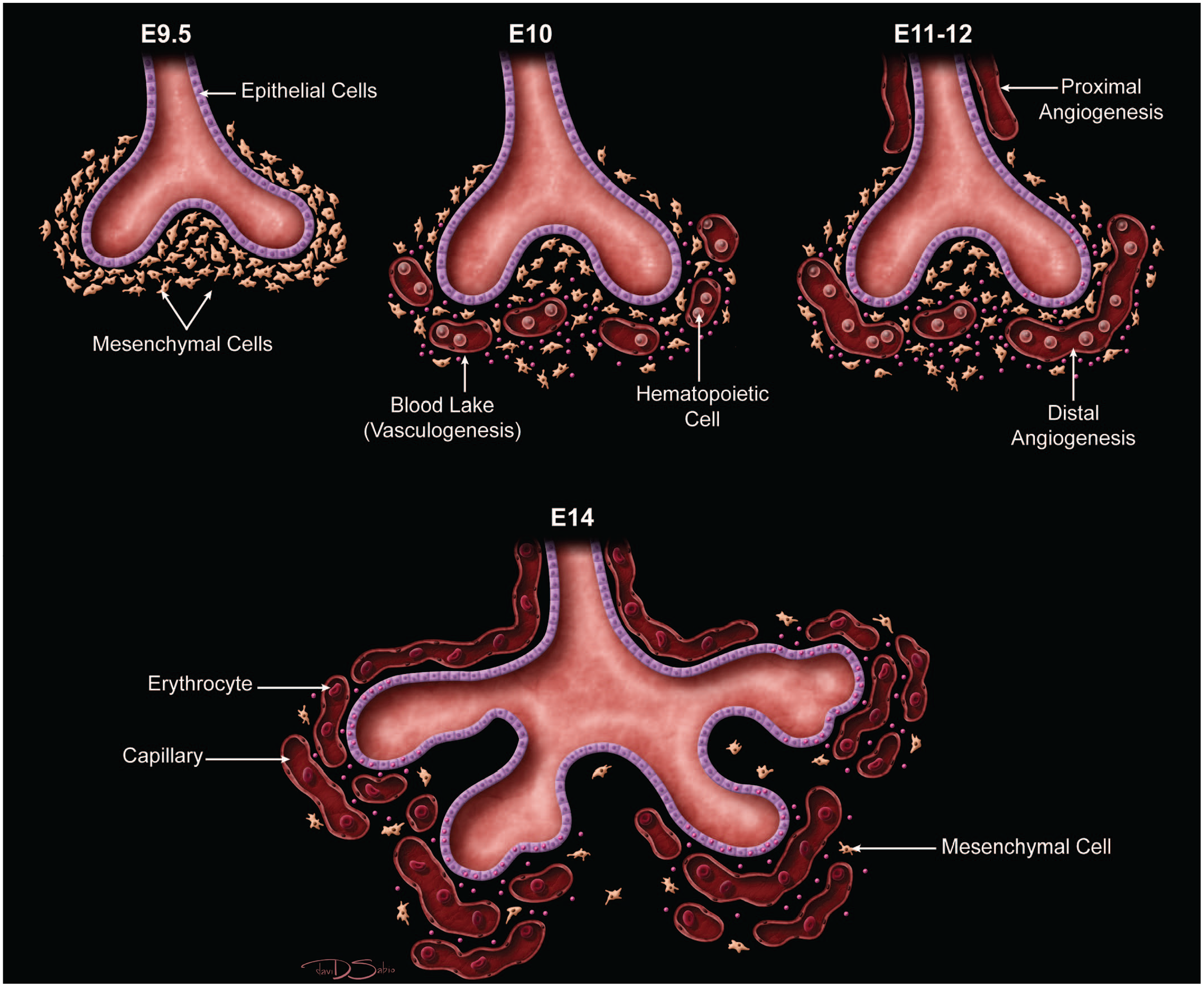
Diagrammatic representation of branching morphogenesis during the pseudoglandular stage of lung development. At E9.5, mesenchyme surrounding the lung buds becomes sparse. By E10.0, hematopoietic precursors begin to form blood lakes within the mesenchyme at the distal ends of the lung buds. By E11.0, distal angiogenesis leads to increased density of blood lakes in the mesenchyme. The epithelial cells within the blood lakes proliferate to form the primitive capillary plexuses around the lung buds. Proximal angiogenesis results in visible blood vessels running along the sides of the trachea. At E14.0, the peripheral mesenchyme exhibits an increase in the density of blood lakes. The lumens of the capillary plexuses contain erythrocytes, indicating a viable connection between peripheral and central pulmonary blood vessels.
At E10.5, the pulmonary arteries enter the lung buds, and capillaries surround the primitive bronchi (Figure 54).58,129 Angiogenesis begins at E11.0 when the central pulmonary arteries and veins begin to form.1,41 When branching morphogenesis of airways occurs during the pseudoglandular stage, the mesenchymal cells undergo apoptosis, and the density of the mesenchyme between the epithelium and endothelial cells becomes sparse (Figures 53 and 54).41,111 Expansion of the capillary networks that surround the most peripheral airways (alveolar primordia) in the lung buds occurs by distal angiogenesis beginning between E11.0 and E12.0; the increasing density of the blood lakes in the mesenchyme and the proliferation of epithelial cells in proximity to the blood lakes result in the formation of primitive, polygonal capillary plexuses around the terminal epithelial buds (Figure 53).41,58,111,129 Lumens of the capillary plexuses are filled with erythrocytes, indicating a direct connection between these capillaries and the embryonic circulation.111,129 Also during this time, proximal angiogenesis occurs when the blood vessels running along the sides of the trachea can be identified as vascular tubes. 111 Between E13.0 and E14.0, the earliest connection between the peripheral and central pulmonary blood vessels is present. 41 The pulmonary vein (Figure 48) has canalized in the midline of the body and returns oxygen-rich blood from the lungs into the left atrium. 148 The central conducting airways are associated with prominent arteries, and the peripheral mesenchyme has an increased density of blood lakes. 41
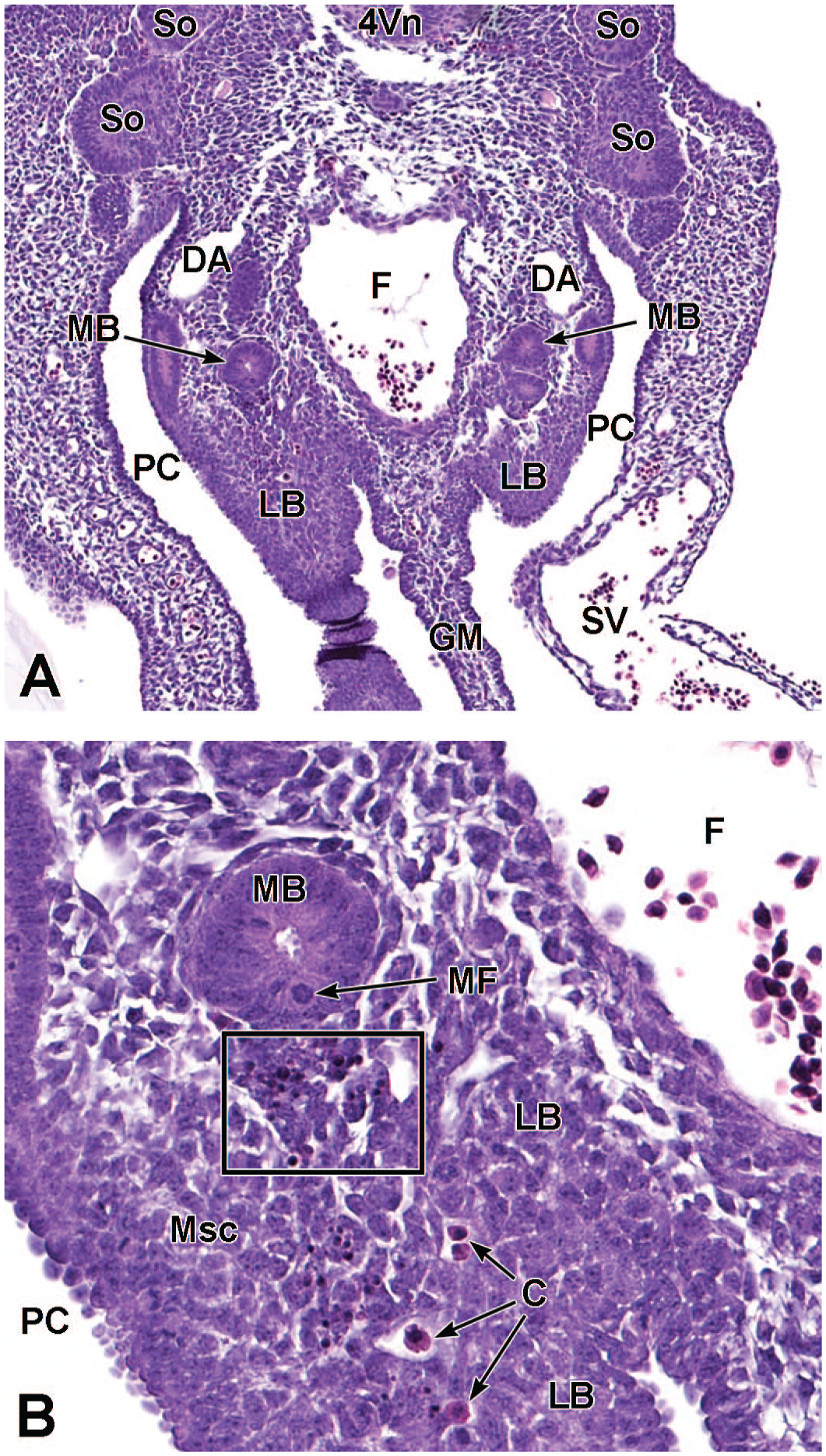
Transverse images of the capillaries surrounding primitive bronchi at E10.5. Panel A (8.5×) depicts the right and left lung buds (LB) with their right and left main bronchi (MB) on either side of the esophageal region of the foregut (F). Erythrocytes within the foregut region are artifact. Panel B (30×) shows a main bronchus lined by epithelium and surrounded by a condensed mesenchyme (Msc) containing capillaries (C); cell fragmentation consistent with physiological apoptosis (programmed cell death) is indicated by a box. Note the presence of mitotic figures (MF) within the main bronchus epithelium. The tip of the fourth ventricle (4Vn); mesenchymal condensation of somites (So), right and left dorsal aorta (DA); right and left pericardioperitoneal canals (PC); subcardinal vein (SV); and gut mesentery (GM) are labeled for orientation. H&E.
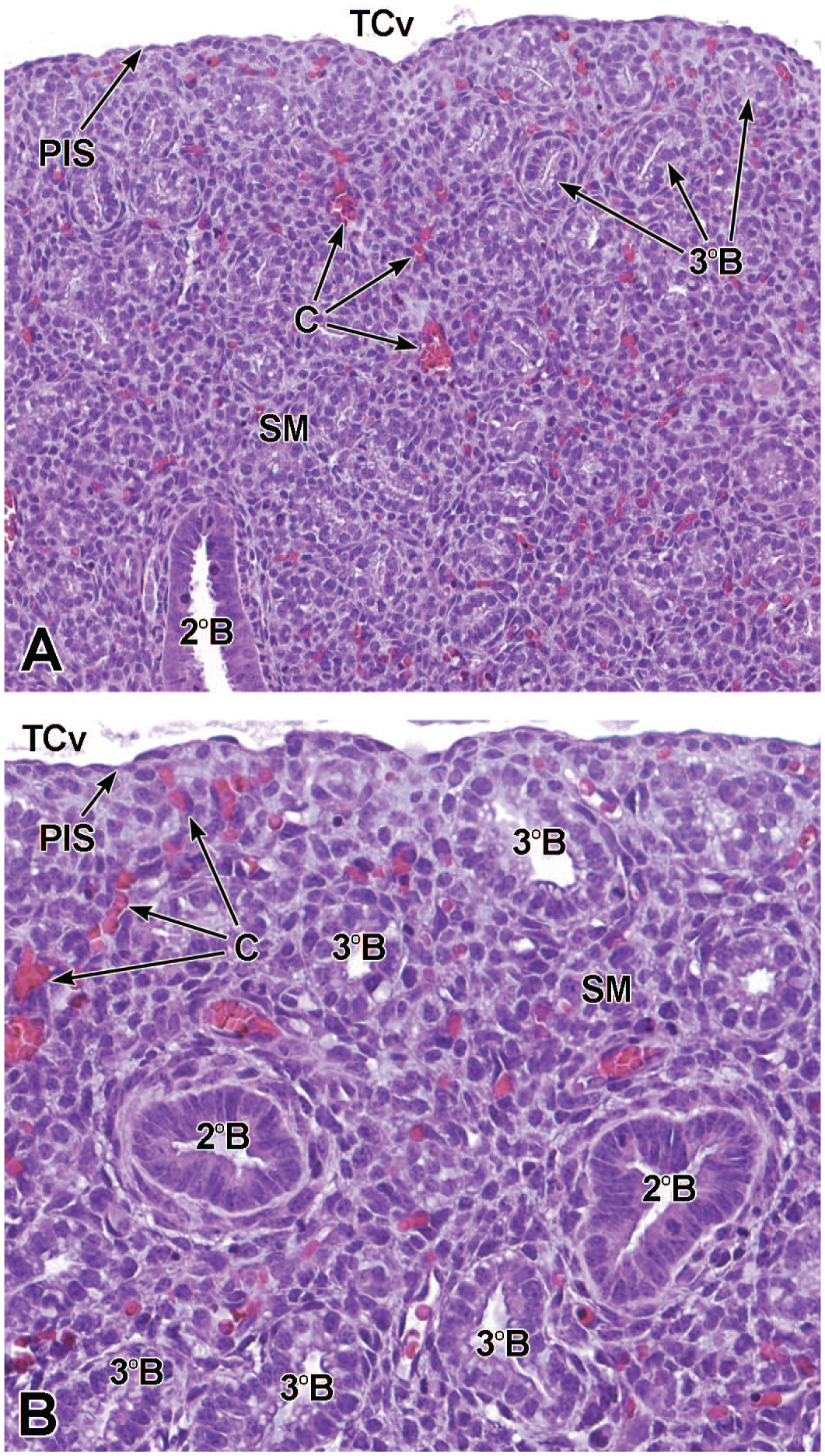
Transverse images of vascularization during the canalicular stage of lung development at E16.5. Panels A (16×) and B (29×) demonstrate a shift in expansion of capillaries (C) in the lung splanchnic mesenchyme (SM) toward the airway epithelium, causing the epithelium to thin over time. The secondary (2°B) and tertiary (3°B) bronchi are dispersed throughout the lung. The thoracic cavity (TCv) and pleural surface (PlS) are labeled for orientation. H&E.
During the canalicular stage, capillary vascularization continues in association with the distal airways. The VEGF gradient continues to induce the capillaries within the mesenchyme to adopt positions adjacent to the airway epithelium (Figure 55).30,111 Eventually, VEGF production transitions completely from the mesenchyme to the epithelium. The close proximity of the capillaries to the epithelium at this stage causes a thinning of the epithelium and differentiation over time of epithelial progenitor cells into ATI and ATII cells.30,111
Postnatal Development
Developmental events during the first week of postnatal life in rodents are approximately equal to those of later in utero development in humans. This section highlights substantial changes in respiratory tract components that occur after birth in mice. Respiratory tract parts that have assumed the adult configuration by birth are not discussed further.
Upper Respiratory Tract
Nasal Turbinates
At birth, the nasal turbinates make up most of the upper half of the murine nasal cavity and are lined extensively by olfactory neuroepithelium, a specialized sensory epithelium that lines the caudodorsal aspect of the nasal cavity (Figure 4).88,149 From the mid-embryonic period (approximately E13.0) and continuing into adulthood, progenitor cells produced in the olfactory neuroepithelium move to a basal position, thus replenishing the stem cell population.70,151 These basal cells continue division and differentiation throughout life to replenish the overlying olfactory neuroepithelium, providing new sustentacular cells in the apical layer and olfactory receptor neurons in the middle layers (a process called “vertical expansion”). Using fibroblast growth factor (FGF)-20, Yang and colleagues have identified a population of olfactory progenitor cells that also facilitate horizontal expansion of the olfactory neuroepithelium. 70 Such horizontal expansion is likely important in restoring olfactory neuroepithelial integrity following mucosal necrosis, which is a response induced by certain blood-borne and inhaled toxicants.59,135
From early postnatal development to approximately P7, chondrocytes within the parietotectal cartilage of the nasal capsule and ethmoturbinates, and within the hyaloid cartilage of the nasal septum, continue to undergo hypertrophy (Figure 56) before being replaced by ossified bone (Figure 57). 187 The extreme ventral aspect of the nasal septum will remain hyaloid cartilage (Figure 57), while the dorsal aspect will ossify (Figure 58). Once ossification is complete, the turbinates continue to grow and fold into scrolls until the final conformation is attained at approximately P30 (Figure 59). 187
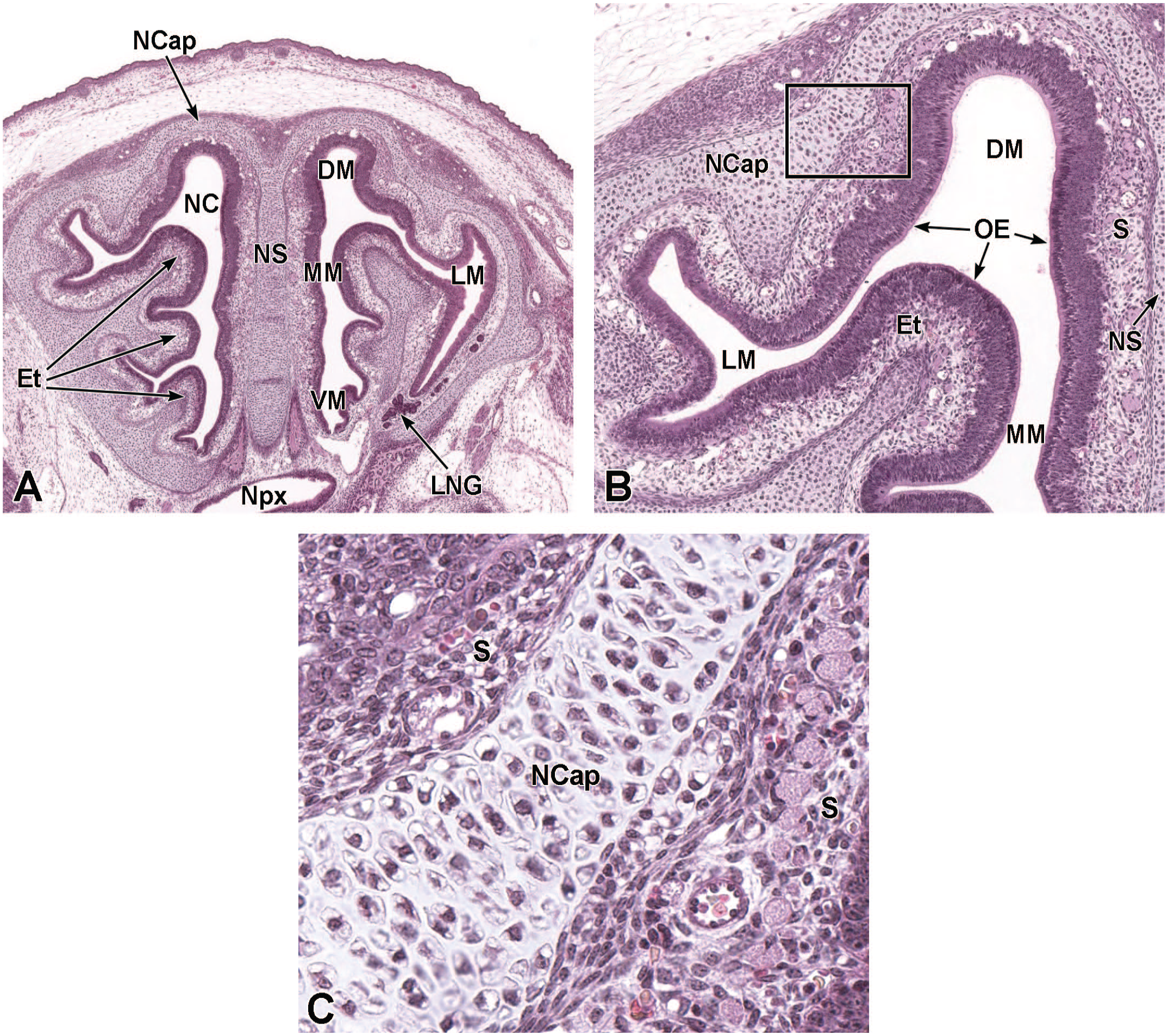
Coronal images of nasal skeletal evolution at E18.5. Panels A (3.5×), B (9.5×), and C (40×) depict chondrocyte hypertrophy within the parietotectal cartilage of the nasal capsule (NCap) and ethmoturbinates (Et), and the hyaloid cartilage of nasal capsule, before being replaced by ossified bone. The extreme ventral aspect of the nasal septum (NS) will remain hyaloid cartilage postnatally. Panels B and C (higher magnification of boxed region in panel B) illustrate the elliptical chondrocytes within the cartilage matrix of the nasal capsule and ethmoturbinates. The olfactory epithelium (OE) lining the dorsal (DM), middle (MM), ventral (VM), and lateral (LM) meatuses of the nasal cavity (NC); the underlying well-vascularized submucosa (S) containing numerous nerve bundles; lateral nasal glands (LNG); and the nasopharynx (Npx) are labeled for orientation. H&E.
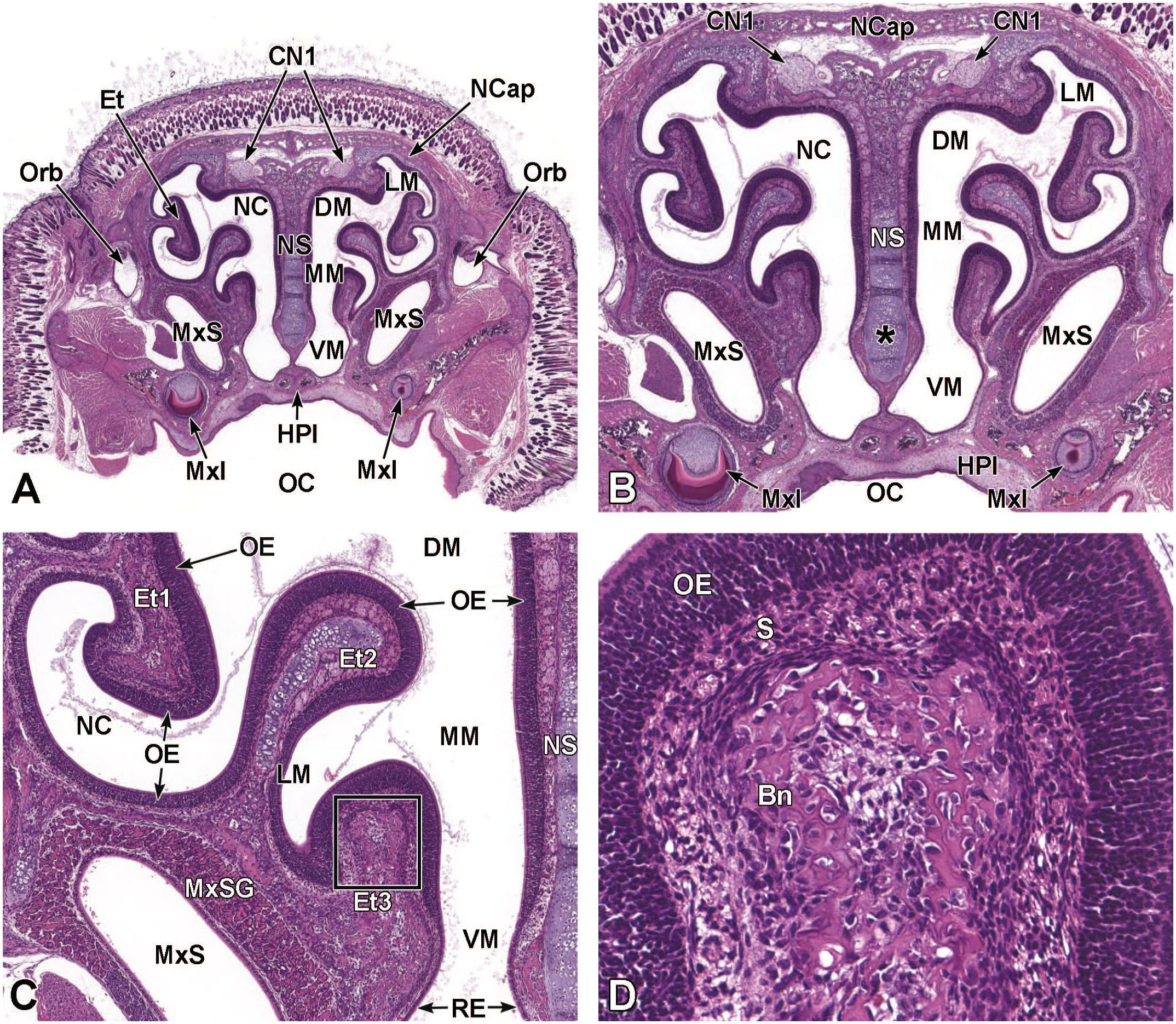
Coronal images of nasal skeleton ossification at P7. Panels A (1.4×), B (2.5×), C (5.5×), and D (29×) illustrate the increased ossification (bone [Bn] formation) of the parietotectal cartilage of the nasal capsule (NCap) and ethmoturbinates (Et) and the hyaloid cartilage within the dorsal aspect of the nasal septum (NS). The central part of the ventral aspect of the nasal septum (asterisk) remains cartilaginous. Panels C and D (a higher magnification of Et3 in the box in panel C) show variable ossification among ethmotubinates, with Et1 and Et3 exhibiting ossified foci while Et2 has none. The nasal cavity (NC); ocular orbits (Orb); maxillary sinuses (MxS); hard palate (HPl); oral cavity (OC); right and left maxillary incisors (MxI); cranial nerve I (CNI); olfactory epithelium (OE) lining the dorsal (DM), lateral (LM), and middle (MM) meatuses and respiratory epithelium (RE) lining the ventral meatus (VM); maxillary sinus glands (MxSG); and the submucosa (S) are labeled for orientation. H&E.
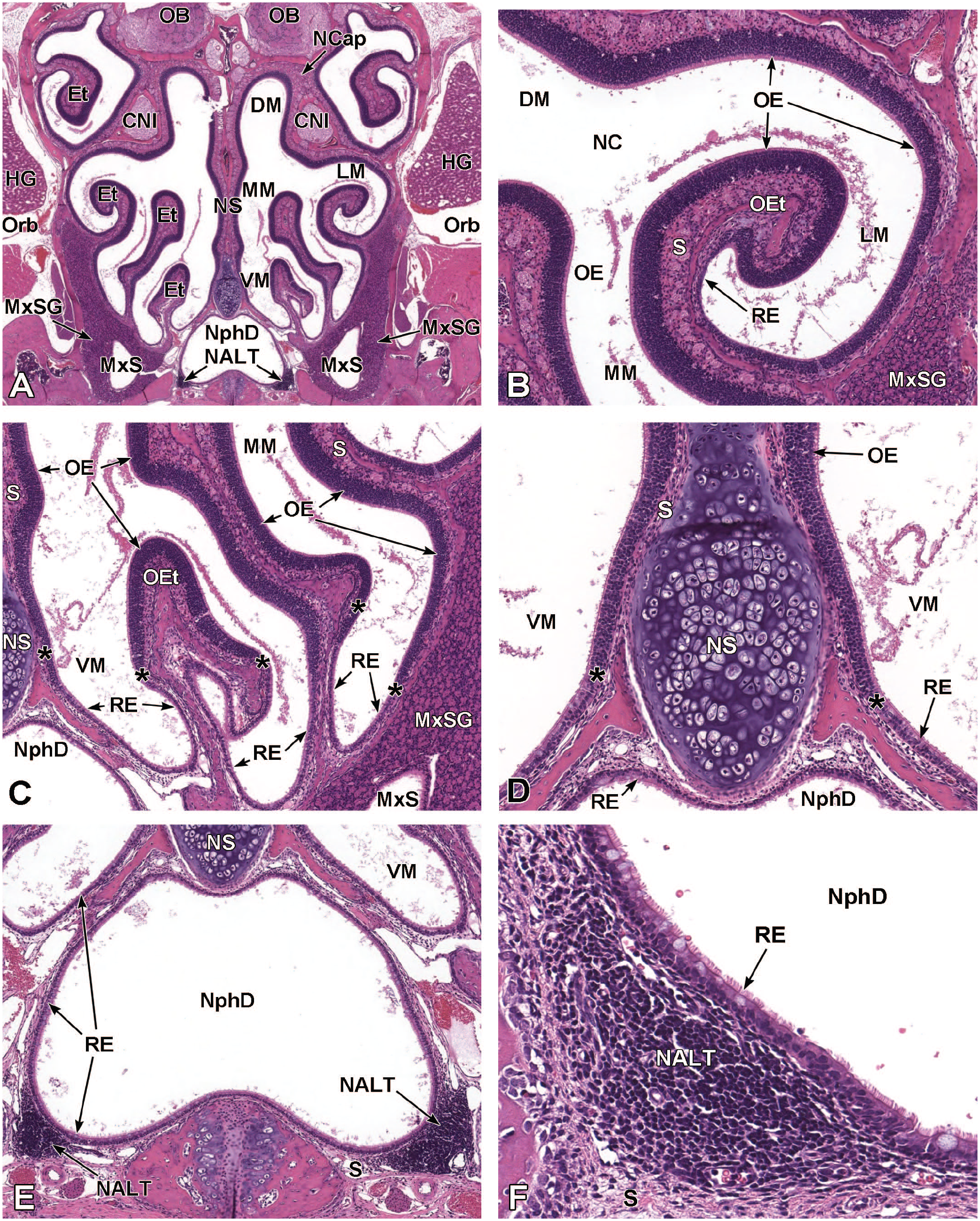
Frontal images of the nasal skeleton ossification, turbinate evolution, and lymphoid tissue development at P7. Panel A (2×) illustrates the ossification of the parietotectal cartilage of the nasal capsule (NCap) and ethmoturbinates (Et) and ossification of the hyaloid cartilage in the dorsal nasal septum (NS). Panels B (8.5×) and C (7×) show ossified ethmoturbinates (OEt) lined by olfactory epithelium (OE) interrupted by patches of respiratory epithelium (RE), a common incidental finding over the lateral meatus (LM) but the expected location for RE in the ventral meatus (VM); transition zones at the borders between the OE and RE are marked by asterisks. Panel D (14×) depicts a lack of ossification in the ventral nasal septum as well as more OE to RE transitions. Panels E (8×) and F (30×) illustrate the densely ciliated RE lining the nasopharyngeal duct (NphD) while nasal-associated lymphoid tissue (NALT) is evident as bilateral triangular aggregates of dense lymphoid tissue (B and T cells) with follicle-associated epithelium equipped with epithelial M cells. The submucosa (S) surrounding the NALT is enriched with lymphatic vessels, veins, and arterioles. The olfactory bulbs (OB); condensations of right and left cranial nerves I (CNI); dorsal (DM) and middle (MM) meatuses; nasal cavity (NC); the right and left Harderian glands (HG); ocular orbits (Orb); maxillary sinus glands (MxSG), and right and left maxillary sinuses (MxS) are labeled for orientation. H&E.
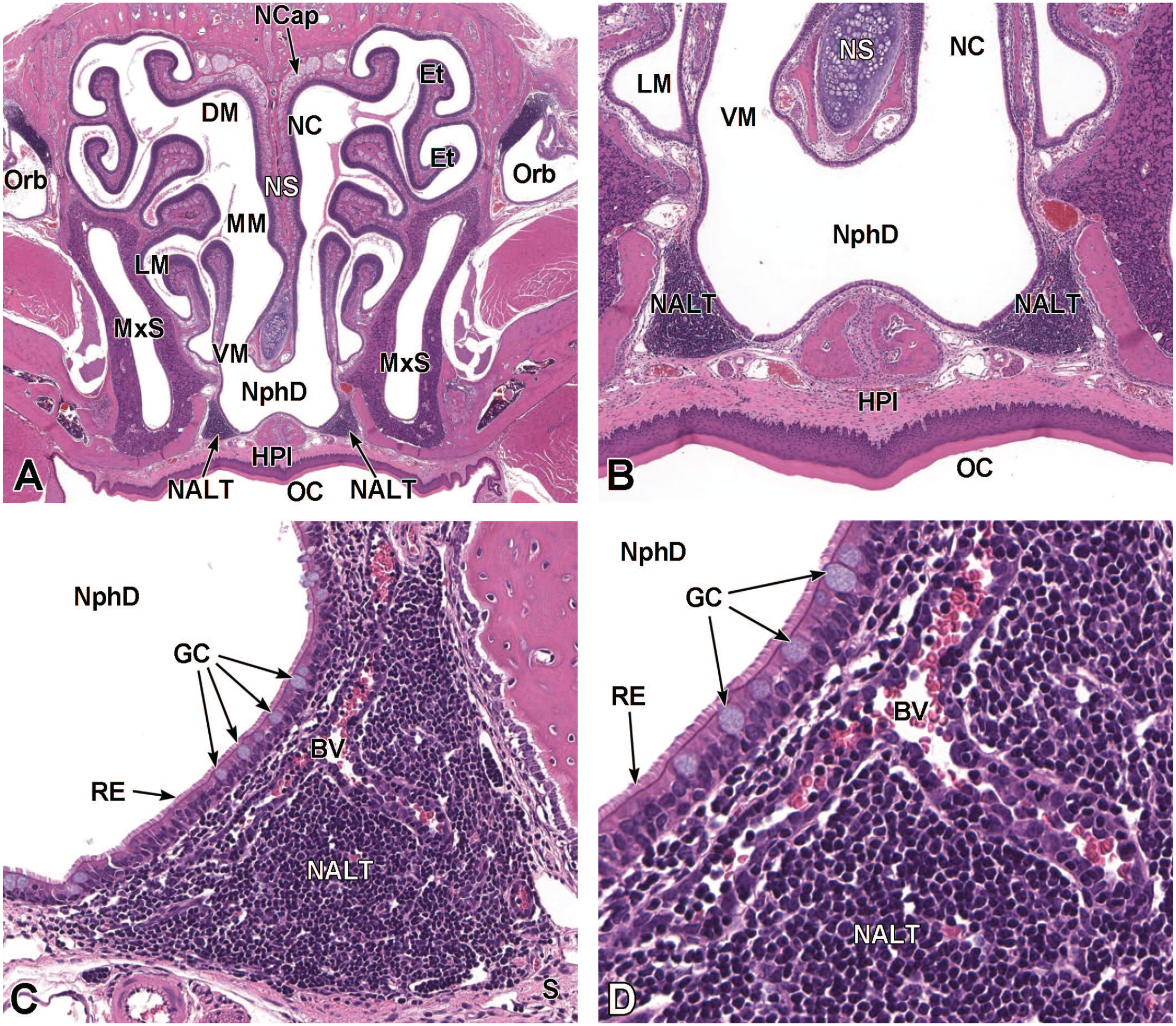
Coronal images of fully formed turbinates and nasal-associated lymphoid tissue at P30. Panels A (1.7×) and B (5×) illustrate the expansion of the ethmoturbinates (Et) within the caudal nasal cavity (NC) and the location of the nasal-associated lymphoid tissue (NALT) along the ventrolateral margin of the nasopharyngeal duct (NphD). Panels C (20×) and D (40×) highlight the dense lymphoid tissue, ciliated respiratory epithelium (RE) with scattered goblet cells (GC), and blood vessels (BV), characteristic of NALT and the adjacent submucosa (S). The nasal capsule (NCap); nasal septum (NS); ocular orbits (Orb); dorsal (DM), middle (MM), lateral (LM), and ventral (VM) meatuses; maxillary sinuses (MxS); hard palate (HPl); and oral cavity (OC) are labeled for orientation. H&E.
Vomeronasal Organ
At birth, the paired VOs are crescent-shaped sacs at the base of the rostral nasal septum (Figure 21) and contain all the adult structural elements, albeit in immature form. Figure 5 demonstrates a diagrammatic representation of the nasal cavity, including the location of the VO of the adult mouse. The VOs become morphologically mature and fully functional during postnatal development (Figures 60-62).14,190 Garrosa and colleagues proposed that, in the rat, initiation of VO secretory activity occurs on P1, cytoarchitectural maturity occurs by P2, and complete organ maturity occurs after P3. 57 It has been shown that, in the mouse, ciliated sensory neurons within the VO project axons to mitral cells within the accessory olfactory bulb and that these synaptic circuits are functional at birth. 77 In the adult mouse, the bilateral nasopalatine ducts extend from the nasal cavity, just caudal to the external nares, and thus are subject to immediate exposure to inhaled stimuli such as pheromones. They extend past the VOs, to the oral cavity, at the junction of the hard and soft palates (Figure 61).33,103
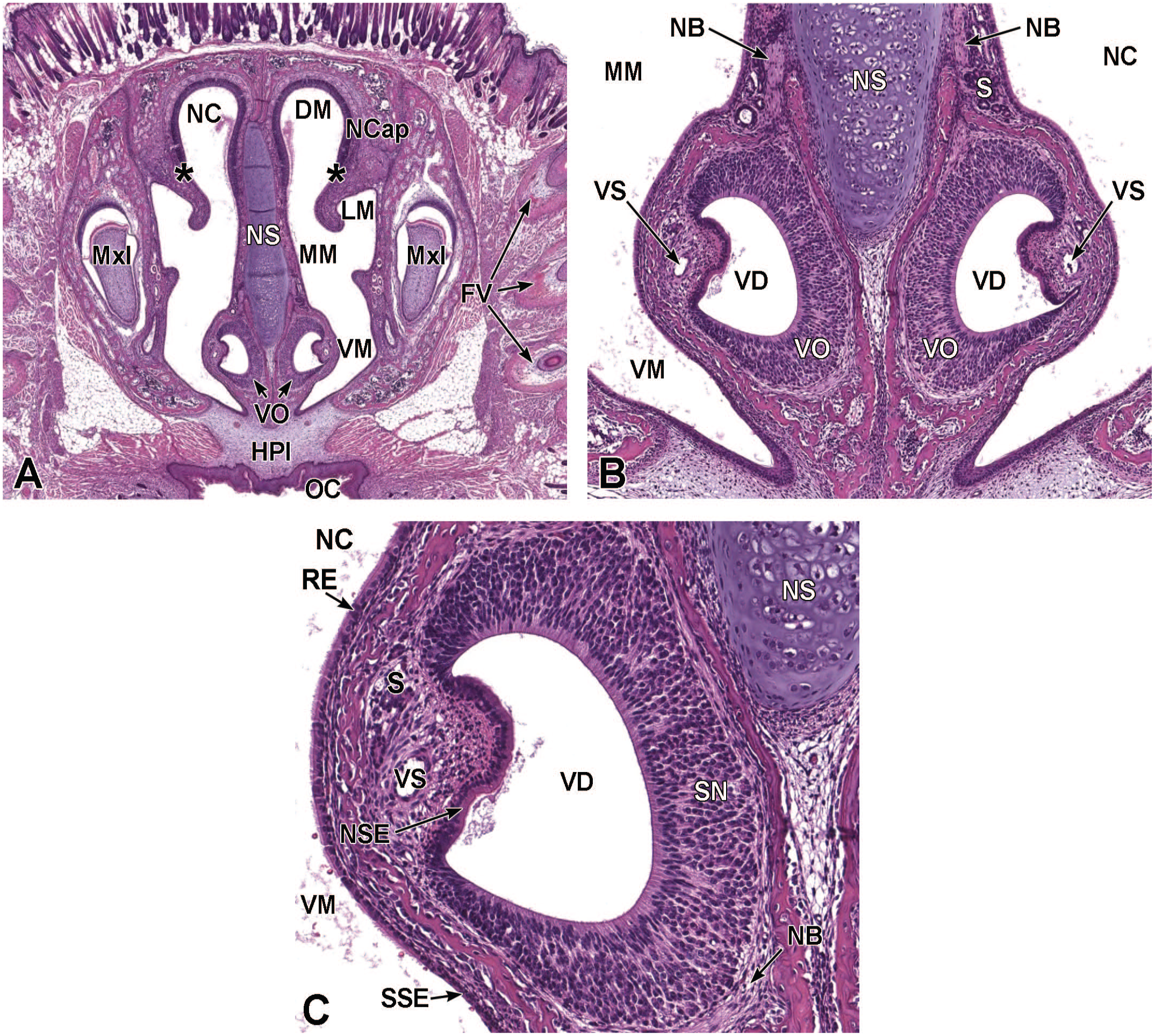
Transverse images of the fully functional vomeronasal organ during postnatal development at P7. Panel A (2.5×) depicts the vomeronasal organ (VO) on either side of the ventral aspect of the nasal septum (NS). Panels B (8.5×) and C (17.5×) illustrate the vomeronasal duct (VD), pseudostratified sensory neuroepithelium (SN) in the dorsal, medial, and ventral walls, and non-sensory ciliated epithelium (NSE) along the lateral wall of the VO. Panel C illustrates the rich supply of nerve bundles (NB) surrounding the VO and a characteristic vascular sinus (VS) in the lateral submucosa (S), adjacent to the NSE. Note the transition from RE to stratified squamous epithelium (SSE) along the nasal septum. The nasal cavity (NC); nasal capsule (NCap); dorsal (DM), middle (MM), lateral (LM), and ventral (VM) meatuses; maxillary incisors (MxI); follicles of vibrissae (FV); hard palate (HPl); and oral cavity (OC) are labeled for orientation. The transitions from olfactory to respiratory epithelium are indicated by asterisks. H&E.
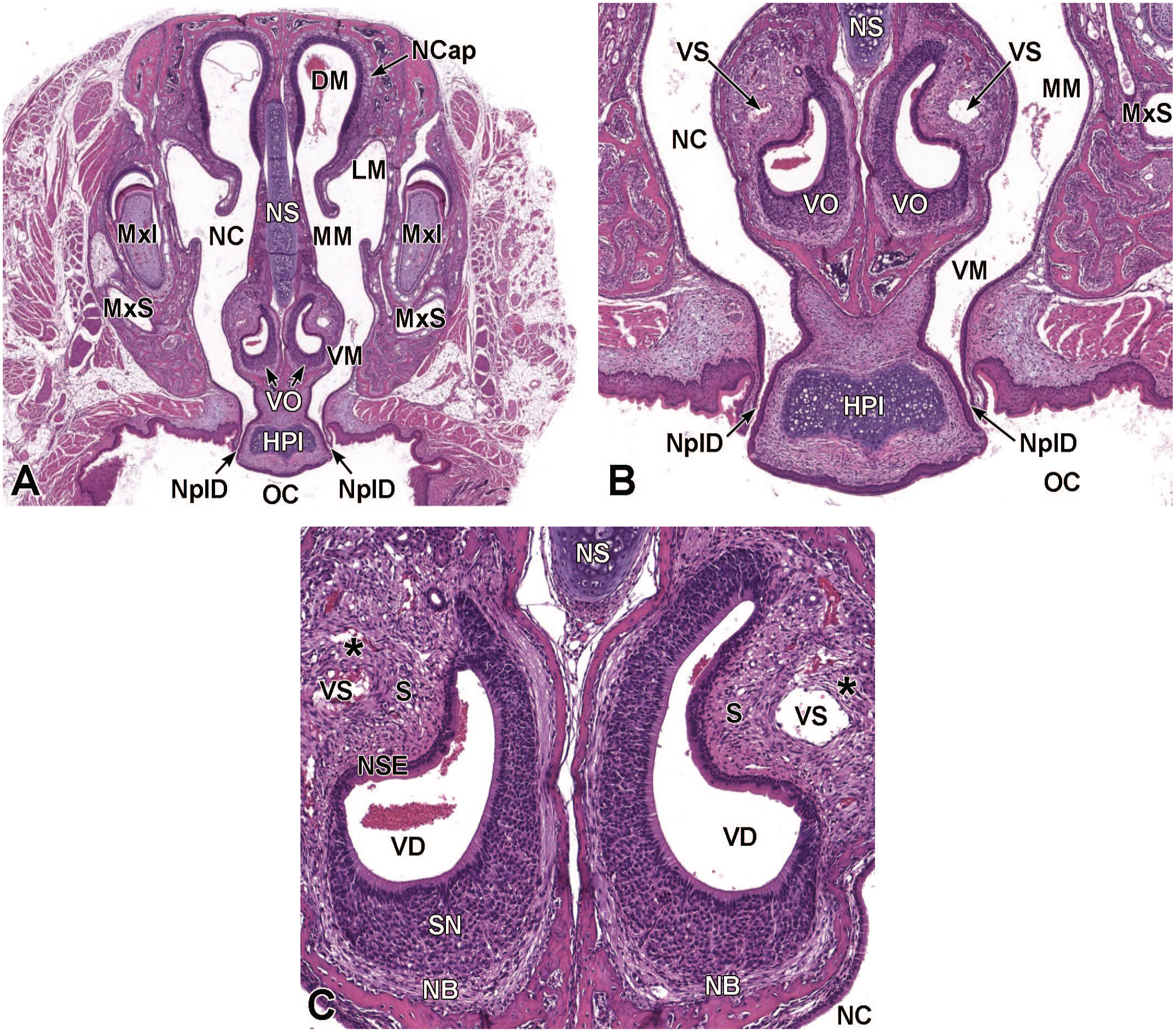
Transverse images of the fully functional vomeronasal organ at P15. Panels A (2×) and B (5×) depict the vomeronasal organ (VO) at the ventral aspect of the nasal septum (NS). Panel C (11.5×) illustrates the central vomeronasal duct (VD) surrounded by nerve bundles (NB) with pseudostratified sensory neuroepithelium (SN) lining the dorsal, medial and ventral walls, and non-sensory ciliated epithelium (NSE) lining the lateral wall of the VO. There is a single prominent venous sinus (VS), surrounded by thick smooth muscle layers (asterisks), present in the adjacent submucosa (S), near the NSE. Note that the erythrocytes within the VO lumens are an artifact. The nasal cavity (NC); nasal capsule (NCap); dorsal (DM), middle (MM), lateral (LM), and ventral (VM) meatuses; maxillary incisors (MxI); maxillary sinuses (MxS); oral openings of the nasopalatine duct (NplD) at the junction between the hard and soft palates; oral cavity (OC); and hard palate (HPl) are labeled for orientation. H&E.
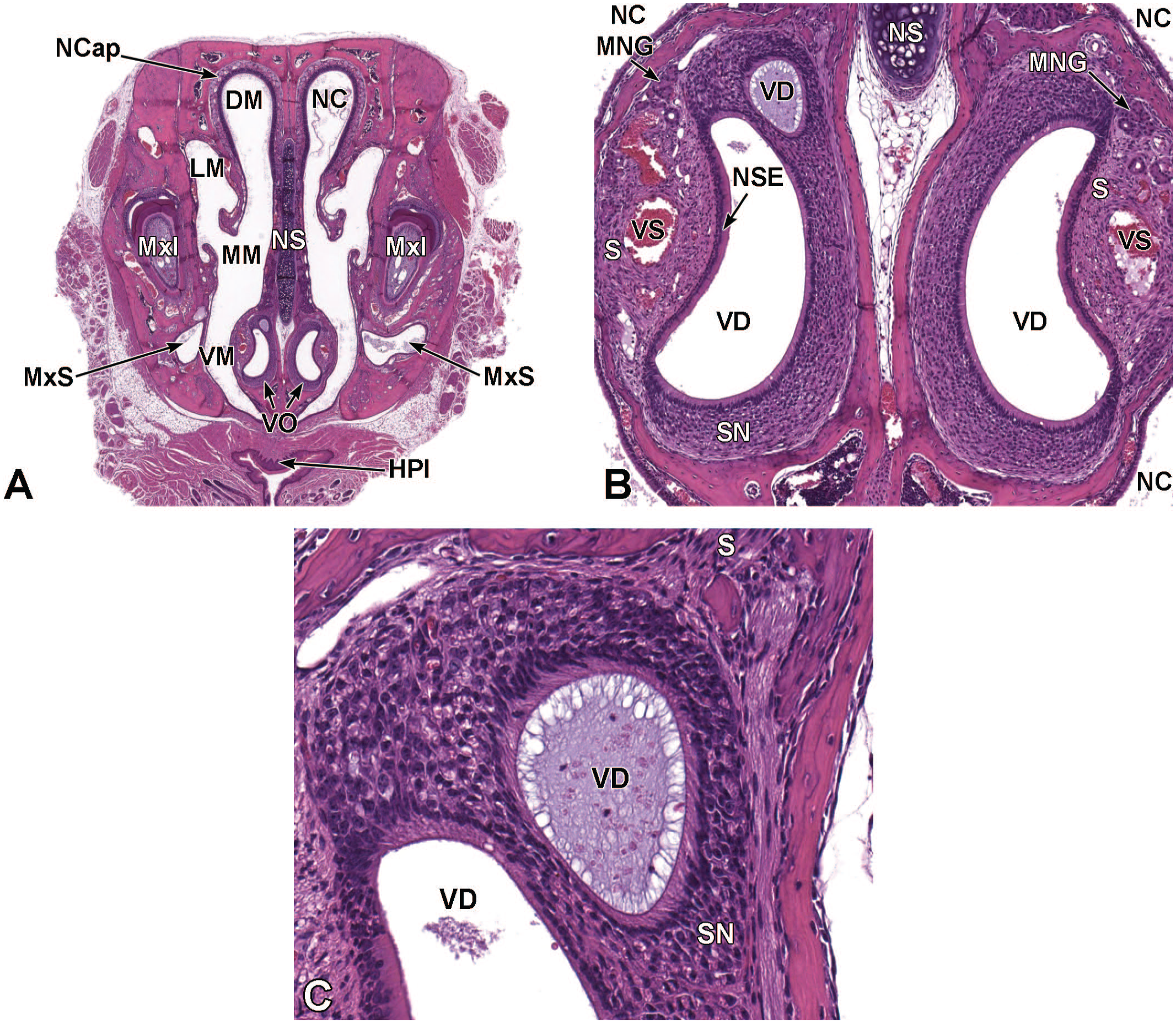
Transverse images of the fully functional vomeronasal organ at P30. Panels A (1.6×) and B (9.5×) depict the vomeronasal organ (VO) in the ventral nasal septum (NS) medial to the ventral meatus (VM). Panel B shows the central vomeronasal duct (VD), pseudostratified sensory neuroepithelium (SN) along the dorsal, medial, and ventral walls, as well as the non-sensory ciliated epithelium (NSE) lining the lateral wall of the VO. In panel B, the development of the irregularly shaped venous sinuses (VS) in the submucosa (S) on the lateral aspect of the VO is apparent. These sinuses are surrounded by smooth muscle and serve as pressure pumps to regulate pheromone entry from the external environment. Panels B and C (30x) depict the VD within the SN with pale basophilic (proteinaceous) intraluminal flocculent material, as it enters the larger VD. This VD is only present in one VO due to sectioning that is slightly oblique. The nasal capsule (NCap); nasal cavity (NC); maxillary incisors (MxI); maxillary sinuses (MxS): dorsal (DM), middle (MM), and lateral (LM) meatuses; medial nasal glands (MNG); and hard palate (HPl) are labeled for orientation. H&E.
The lumen of the VO communicates rostrally with the nasopalatine ducts (Figure 61). There are striated muscles surrounding the rostral aspect of the nasopalatine ducts, most likely allowing voluntary contraction or expansion of the duct opening to optimize detection of external stimuli. 103 On the last day of gestation, the nasopalatine ducts appear as solid tubes lined with stratified columnar epithelial cells. At P1, the nasopalatine ducts extend more medially than before birth and may or may not be patent, possibly depending on the mouse strain or timing of initial exposure to external chemical stimuli.33,77 The nasopalatine ducts become patent after P1 (Figure 61), but the epithelial lining may remain in an immature form throughout the normal prepubertal period. 33 During this period, pyknotic cells (representing physiological programmed cell death), desquamated cells, and sloughed keratin are associated with the epithelial surfaces of the nasopalatine ducts, and these dead cells and cell fragments may still be present within the duct lumens up to P15. At P30, the nasopalatine ducts appear larger in size and contain an intralumenal proteinaceous material, which is clearly visible within the vomeronasal duct at the dorsal aspect of the vomeronasal organ (Figure 62). These nasopalatine ducts will reach adult form around sexual maturity at which time little to no intraluminal cellular debris will remain. 33
Grueneberg ganglion
In juvenile and adult mice, the GG are bilateral filiform aggregates of ciliated sensory neurons located in the rostral nose near the lateral nasal (Steno’s) gland (Figure 17). Development of the GG appears to be complete at birth except that sensory neurons transition from a clustered morphology to one that is more threadlike in shape, and neuron numbers undergo a minor decrease as adulthood approaches.47,54 These olfactory sensory neurons may function in detecting pheromones, controlling odor-driven food choices, and act as thermosensors in mediating cold-evoked neural responses. 15
Septal organ of Masera
During postnatal development, the SOM separates from the main olfactory neuroepithelium at P7. At this time, it has almost the same histological features as the adult SOM with olfactory sensory neurons, supporting cells, and Bowman’s glands. 124 The SOM continues to increase in size during postnatal development.
Nasal-Associated Lymphoid Tissue
The NALT plays an important role in both antigen recognition and immune activation after intranasal exposure. 104 In the adult mouse, the NALT occurs as bilaterally symmetrical aggregates of organized lymphoid tissue at various locations in the submucosa lining the nasal passages (including sinuses) and nasopharynx. In rodents, NALT is seen most often as an organized, circular or triangular (in cross-section) lymphoid structure found on both sides of the nasopharyngeal duct dorsal to the cartilaginous soft palate. 96 Common locations include the nasopharyngeal duct and nearby nasopharynx; these elements are analogous to the lymphoid tissues (i.e., nasopharyngeal tonsil [adenoid], lingual tonsils, tubal tonsils, and lateral pharyngeal bands, collectively referred to as “Waldeyer’s ring”) in humans. 69
Development of NALT in mice is an exclusively postnatal event. The first evidence of NALT formation can be seen around P7 (Figure 58) as high endothelial venules with associated T and B lymphocytes beneath the mucosa. 52 The NALT continues to increase in size with increasing age into adulthood, reaching its full extent at 5 to 8 weeks after birth (Figure 59). 52
Larynx and Vocal Folds
Postnatal development of the mouse larynx is necessary for the transition at weaning (typically day 21 for mice) from a diet of milk to solid food. The anatomic restructuring allows laryngeal closure and prevents aspiration of particulate matter. The restructuring is also critical for acoustic communication.
The adult larynx is composed of the hyoid bone, an elaborate frame of cartilages, skeletal muscles, nerves, connective tissue, and mucosa. The flanged circle composed of the cricoid cartilage, C-shaped thyroid cartilage, and intervening paired epiglottic cartilages makes up the core of the laryngeal scaffold. Other cartilages including the paired arytenoid cartilages and attached corniculate processes are instrumental in normal laryngeal function. Although little research has been done to investigate postnatal laryngeal development in mice, laryngeal cartilages exhibit both size and shape variations during the postnatal period.3,140,166 Using 3-dimensional reconstruction and geometric morphometrics, Riede and colleagues determined that postnatal development of the mouse laryngeal cartilages is characterized by developmental age-specific differences in the shape of the thyroid, arytenoid, and epiglottic cartilages but not the cricoid cartilage, showing that these four laryngeal cartilages do not develop uniformly after birth. 140
Before birth, the vocal folds have completely separated (Figure 63) and are firmly attached to the thyroid cartilage (ventrally) and arytenoid cartilages (dorsally), and the epithelial lamina has been fully recanalized. After birth, the laryngeal cavity further elongates to accommodate vocal fold restructuring. Maturation of vocal fold mucosa occurs as epithelial cells continue their process of stratification and evolve from a bilayer during development to a multi-layer (3 or 4 cells deep) in the adult mouse. 107 The basal cells adjacent to the lamina propria change from columnar to cuboidal in shape, whereas the cuboidal, apical, and suprabasal cells that line the lumen become more elongated. 107 The postnatal maturation process takes approximately 6 weeks, and its completion coincides with sexual maturation. 107
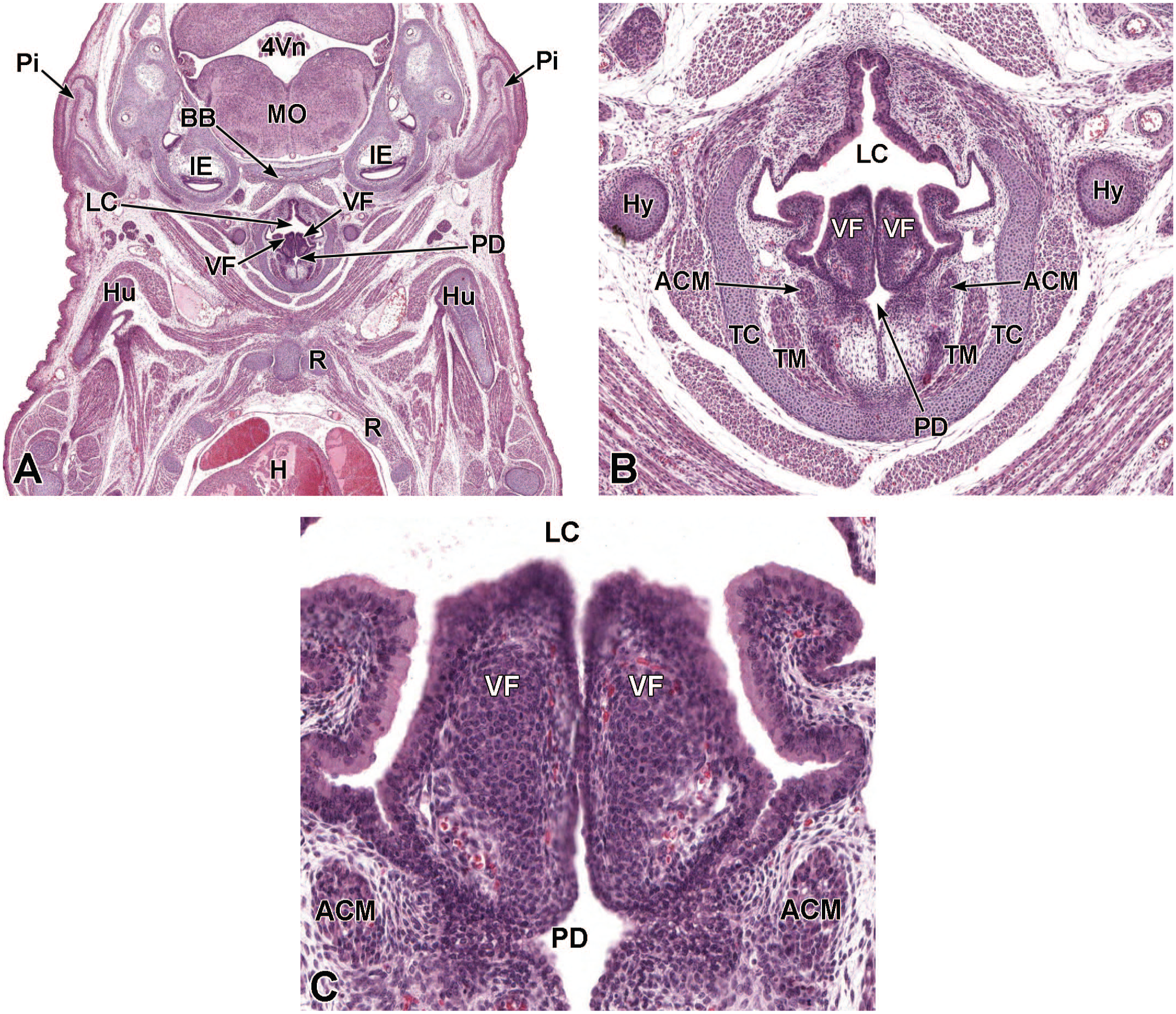
Frontal images of the vocal folds at E18.5. Panels A (1.5×), B (6×), and C (36×) are sections taken at the level of the laryngeal cecum (LC), which has dilated from the laryngeal groove, and where it has united with the pharyngoglottic duct (PD) that leads to the trachea. Panel C is a high magnification of the vocal fold (VF) lining epithelium as it continues to stratify after epithelial lamina separation and begins to evolve from a bilayer to a multi-layer as development continues. The fourth ventricle (4Vn); medulla oblongata (MO); basisphenoid bone (BB); left and right pinnae (Pi) and inner ears (IE); hyoid bones (Hy); alar cricoarytenoid muscles (ACM); thyroid cartilage (TC): lateral thyroarytenoid muscles (TM); the right and left humerus (Hu); ribs (R); and heart (H) are labeled for orientation. H&E.
Trachea
At birth, the trachea is surrounded laterally and ventrally by 15 to 18 C-shaped hyaline cartilage rings separated by fibroelastic tissue (Figure 42). The dorsal openings of the rings are joined by smooth muscle. Pseudostratified epithelium lines the lumen and is composed of a variety of cells including pseudostratified ciliated columnar epithelium, nonciliated secretory (club), mucus-secreting goblet, and basal (stem) epithelial cells as well as neuroendocrine cells. Cholinergic chemosensory cells are also scattered within the epithelium throughout the length of the trachea, first appearing at E18.0 and then continuing to expand in number after birth. 130 The trachea continues to grow in length and diameter during postnatal development along with an associated increase in the stratification and thickness of the mucosa, including basal cells. 142 Submucosal glands develop postnatally and are found only in the proximal trachea, generally between the cricoid cartilage of the larynx and the first tracheal ring but sometimes extending as far caudally as the sixth and seventh tracheal ring. 113 The proximal glands have fully developed by P21, but the more distal glands develop later between P21 and P42. 113
Lungs
After birth, the volume of the thoracic cavity expands to a greater degree than the lungs to allow for the rapid expansion of the lungs during initial inspiration. 88 The lungs increase in size after birth as a result of the increasing size of airway diameters and the growing number of fully differentiated (and air-inflated) alveoli. 118 Changes within the lung parenchyma at this stage of development include enlargement of alveolar sacs, more efficient removal of airway fluid, increased intra-alveolar oxygen tension, and enhanced pulmonary circulation. 111
The saccular stage of lung development continues until approximately P5 as the surface area for gas exchange in the lungs expands significantly. On the day of birth, true alveoli are absent. Instead, primary saccules are present and surround the terminal bronchioles (Figures 52 and 64). These saccules appear larger than alveoli and function as the gas exchange units during this stage despite their relatively thick (low cuboidal) epithelial linings (Figure 64). 6 As these saccules come together, they form thick primary septa. The saccules are smooth-walled spaces lined by low cuboidal epithelium initially, and the thick primary septa that separate them contain interstitial cells as well as capillary networks interspersed amid central layers of connective tissue (Figure 64). Only a small portion of the primary saccules are lined with cuboidal ATII cells at this stage. Between P2 and P4, the primary saccules continue to enlarge, and the smooth walls begin to be modified by the formation of secondary crests seen histologically as triangular-shaped elevations (Figures 65 and 66). 6 These crests are formed as myofibroblasts secrete elastin within small areas in the primary septa. 111 The secondary crests are ridges that contain capillaries and stromal cells that protrude into the lumens of the primary saccules and divide the saccules into multiple parts (forming grape-like clusters), resulting in the formation of secondary saccules and eventually the formation of individual alveoli (Figures 65 and 66).6,100 The stromal cells within the secondary crests consist of myofibroblasts and fibroblasts that produce elastin and collagen to provide elasticity and structure to the lung. 100 This change is most dramatic in the first 10 days of postnatal life. 133
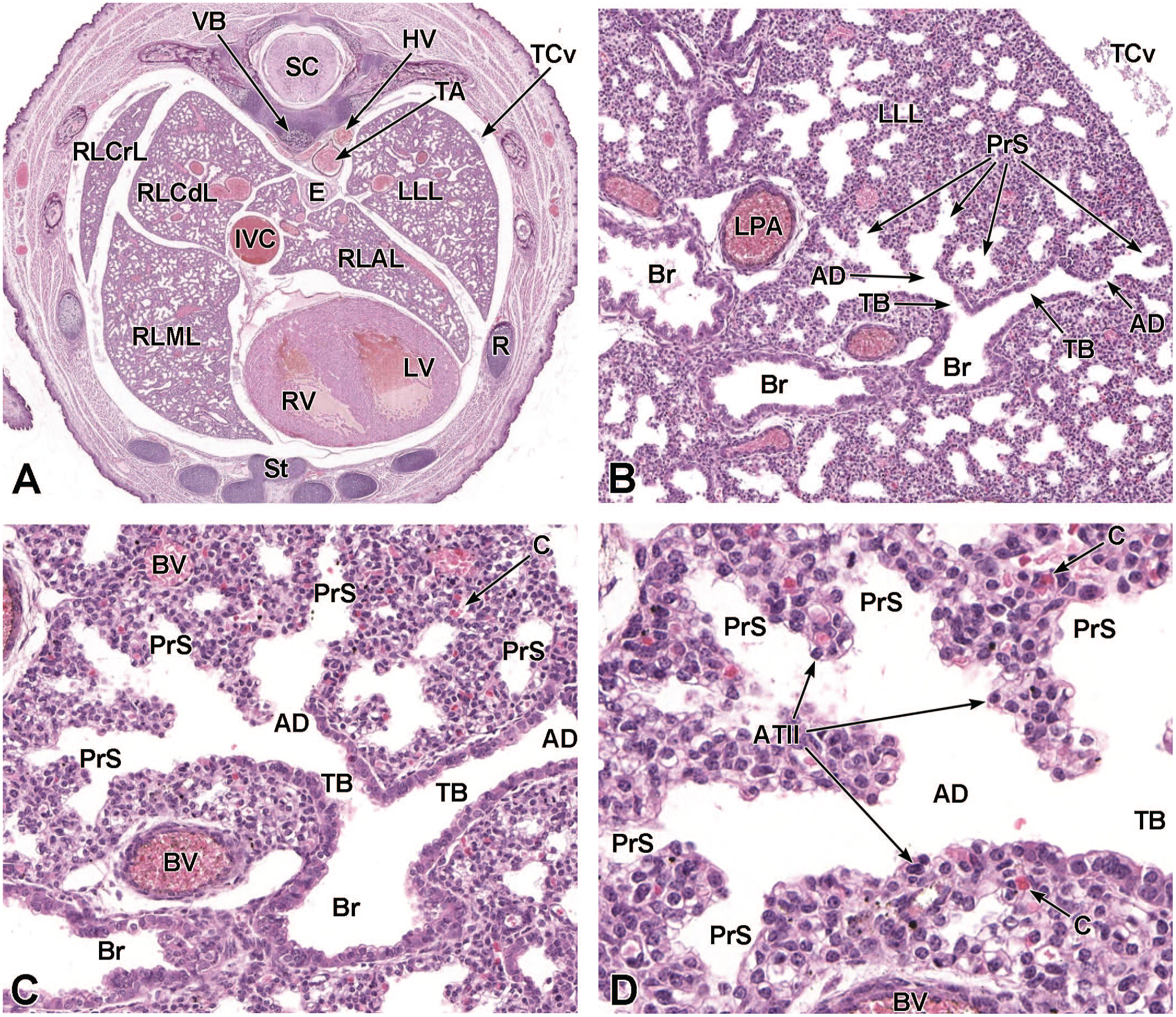
Transverse images of the saccular stage of lung development at E18.5. Panel A (1.5×) depicts the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), right lung caudal lobe (RLCdL), right lung accessory lobe (RLAL), and left lung lobe (LLL). Panels B (9.5×), C (19.5×), and D (40×) illustrate fields of primary saccules (PrS), also called primary septa, surrounding conducting (Br) and terminal (TB) bronchioles, and interspersed with capillaries (C) and larger blood vessels (BV). Bronchi branch progressively into bronchioles, terminal bronchioles, alveolar ducts (AD), and finally primary saccules lined by a small numbers of cuboidal alveolar type 2 (ATII) cells. A branch of the left pulmonary artery (LPA) can be seen within the left lung lobe in Figure B. The spinal cord (SC); vertebral body (VB); esophagus (E); hemiazygos vein (HV); thoracic aorta (TA); thoracic cavity (TCv); rib (R); the right and left ventricles (RV and LV, respectively) of the heart; xyphoid process of the sternum (St); and inferior (caudal) vena cava (IVC) are labeled for orientation. H&E.
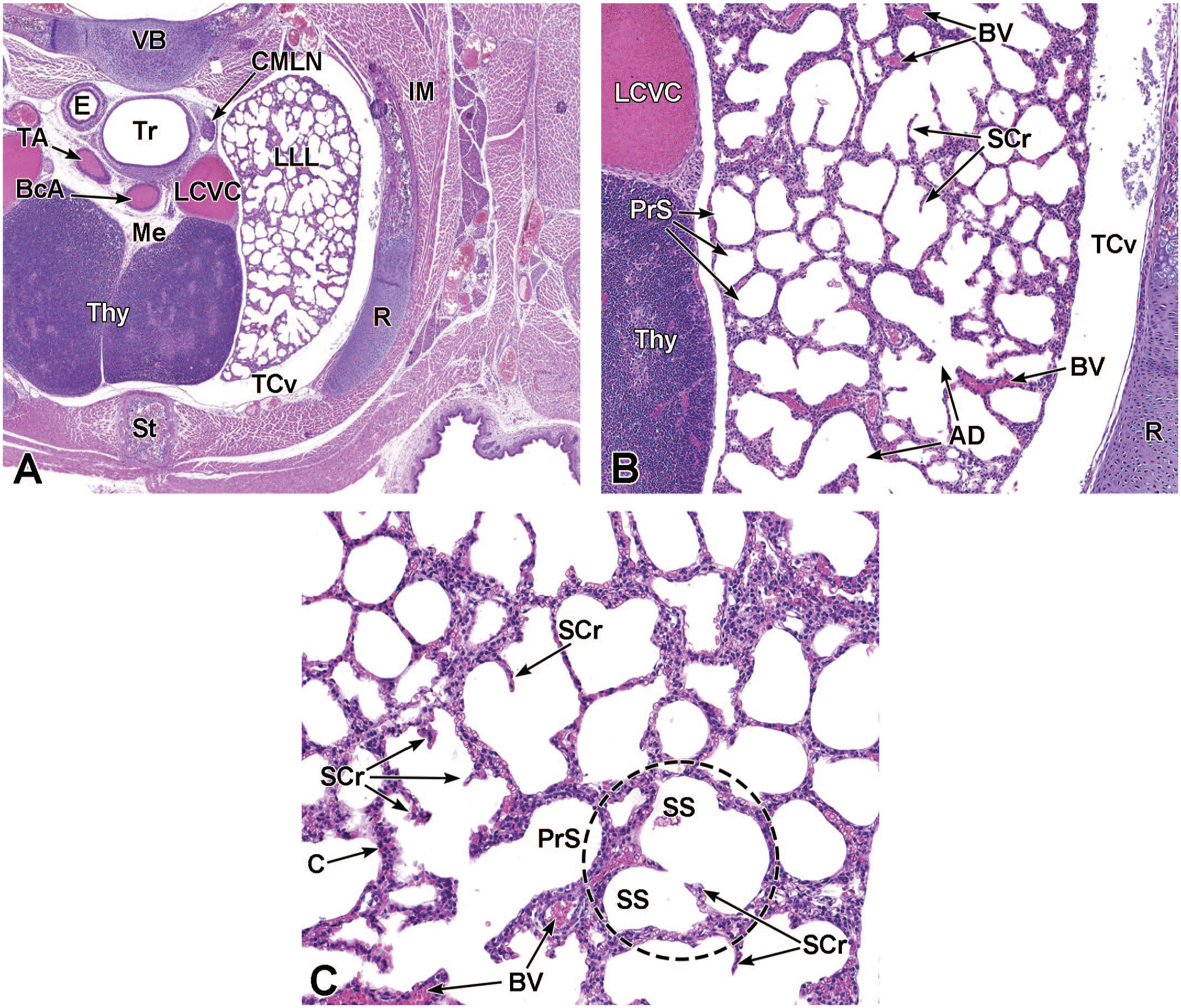
Transverse images the saccular stage of lung development at P2. Panels A (2×), B (7×), and C (12×) use the well-inflated left lung lobe (LLL) to illustrate the intimate connections between the alveolar ducts (AD) and primary saccules (PrS) with formation of the secondary crests (SCr) that serve to define the imminent advent of the secondary saccules. The triangle-shaped secondary crests consist of elevated leaflets of stromal cells and capillaries that divide the lumens of the primary saccules into grape-like clusters termed secondary saccules (SS); the dashed circle in panel C demonstrates the subdivision of a primary saccule into two secondary saccules. The vertebral body (VB); esophagus (E); trachea (Tr); cranial mediastinal lymph node (CMLN); thoracic aorta (TA); brachiocephalic artery (BcA); left cranial vena cava (LCVC); thymus (Thy); mediastinum (Me); intercostal muscle (IM); rib (R); thoracic cavity (TCv); xyphoid process of the sternum (St); capillaries (C); and blood vessels (BV) are labeled for orientation. H&E.
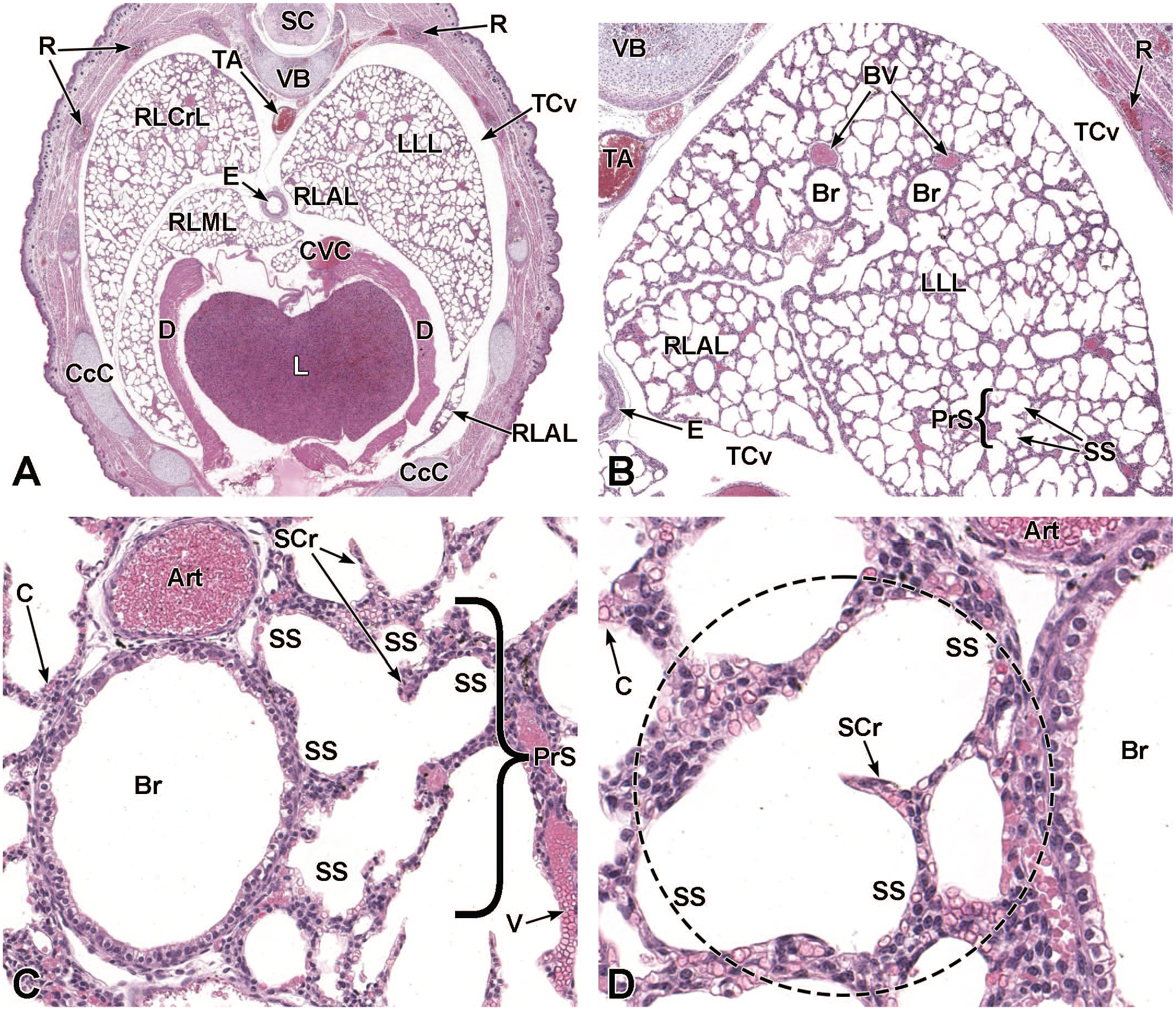
Transverse images depicting the saccular stage of lung development at P3. Panel A (1.3×) shows well-inflated right lung cranial lobe (RLCrL), right lung middle lobe (RLML), left lung lobe (LLL), and right lung accessory lobe (RLAL) within the caudal thorax. Panels B (3.5×), C (19×), and D (38.5×) depict enlargement of the primary saccules (PrS, denoted by the brackets in panels B and C and circled area in panel D), as well as increased numbers of secondary crests (SCr) that form multiple secondary saccules (SS). The secondary crests are composed of stromal cells and capillaries as depicted in panel D. The spinal cord (SC); vertebral body (VB); thoracic aorta (TA); esophagus (E); thoracic cavity (TCv); caudal vena cava (CVC); diaphragm (D); costochondral cartilage (CcC) of the ribs (R); liver (L); bronchiole (Br); arteriole (Art); capillary (C), venule (V); and other blood vessels (BV) are labeled for orientation. H&E.
The saccules undergo extensive remodeling of both the capillary and epithelial elements in the saccular walls to establish the more efficient thin-walled gas exchange units within the alveoli. The capillary networks that cover the saccules invaginate into the septa to create double capillary layers. 111 Endothelial cell proliferation and heterogeneity, angiogenesis, VEGF expression, and capillary surface area all increase during this time while capillaries align in close proximity to the alveolar epithelium.8,75,111 The mesenchymal cells within the septa will decrease in number via apoptosis, resulting in a decreased distance and reduced tissue mass between the capillary endothelium and the adjacent alveolar epithelial surface, allowing for more efficient gas exchange to occur in the primary saccules. 111 The ATI and ATII cells transition into fully differentiated squamous ATI pneumocytes (numerous) and cuboidal ATII pneumocytes (fewer).6,176 The squamous ATI cells facilitate gas exchange between the alveolar spaces and blood and also maintain the permeability barrier function of the alveolar wall.153,180 The cuboidal ATII cells lose their glycogen and begin to produce the lamellated intracytoplasmic inclusion granules that contain a surfactant. 188 Besides secreting a surfactant, these ATII cells also metabolize xenobiotics, provide for the transepithelial movement of water, and serve as progenitor cells to repopulate the alveolar epithelium (both ATI and ATII cells) after an injury.24,80 The ATI and ATII cells share their basement membranes with the basement membranes produced by the underlying capillary endothelium. 80 At the end of the saccular stage on approximately P5, the lung parenchyma consists of saccules with thick, immature septa that contain a double-layered capillary network.
The alveolar stage of lung development begins around P5 and ends around P30. This timing contrasts with humans where this final stage begins in utero as early as 36 weeks of gestation (i.e., nearing term) and ends at around 8 years of age. It is during this stage that pulmonary circulation increases, oxygen tension increases, and the majority of the alveolar surface needed for gas exchange is developed. During this time, the interstitial tissue in alveolar walls decreases significantly in amount and density. Thin alveolar walls consist of a single layer of endothelium, an intermediate basement membrane, and a monolayer of squamous ATI cells with scattered cuboidal ATII cells. 111 During the process of “alveolarization,” secondary septa (or secondary crests) arising from primary septa subdivide the alveolar region and create a greater number of alveoli that are all smaller in size (Figures 67 and 68); this process dramatically increases the surface area available for efficient gas exchange.13,111 In the adult mouse, the alveolar membrane (i.e., the gas exchange surface) is composed of approximately 93% to 97% ATI pneumocytes; 3% to 5% ATII pneumocytes; occasional brush cells; 1% ionocytes; and capillary endothelial cells.111,137 Brush cells are elements with abundant apical microvilli that act as cholinergic chemosensory epithelial cells, transmitting signals from the airway lumen to the immune and nervous systems. 170 Ionocytes are a recently discovered cell type found at multiple levels of the respiratory tract that are involved in fluid regulation (by active chloride removal with associated fluid movement from the airway) at the epithelial surface, affecting airway surface physiology including mucous viscosity. 117
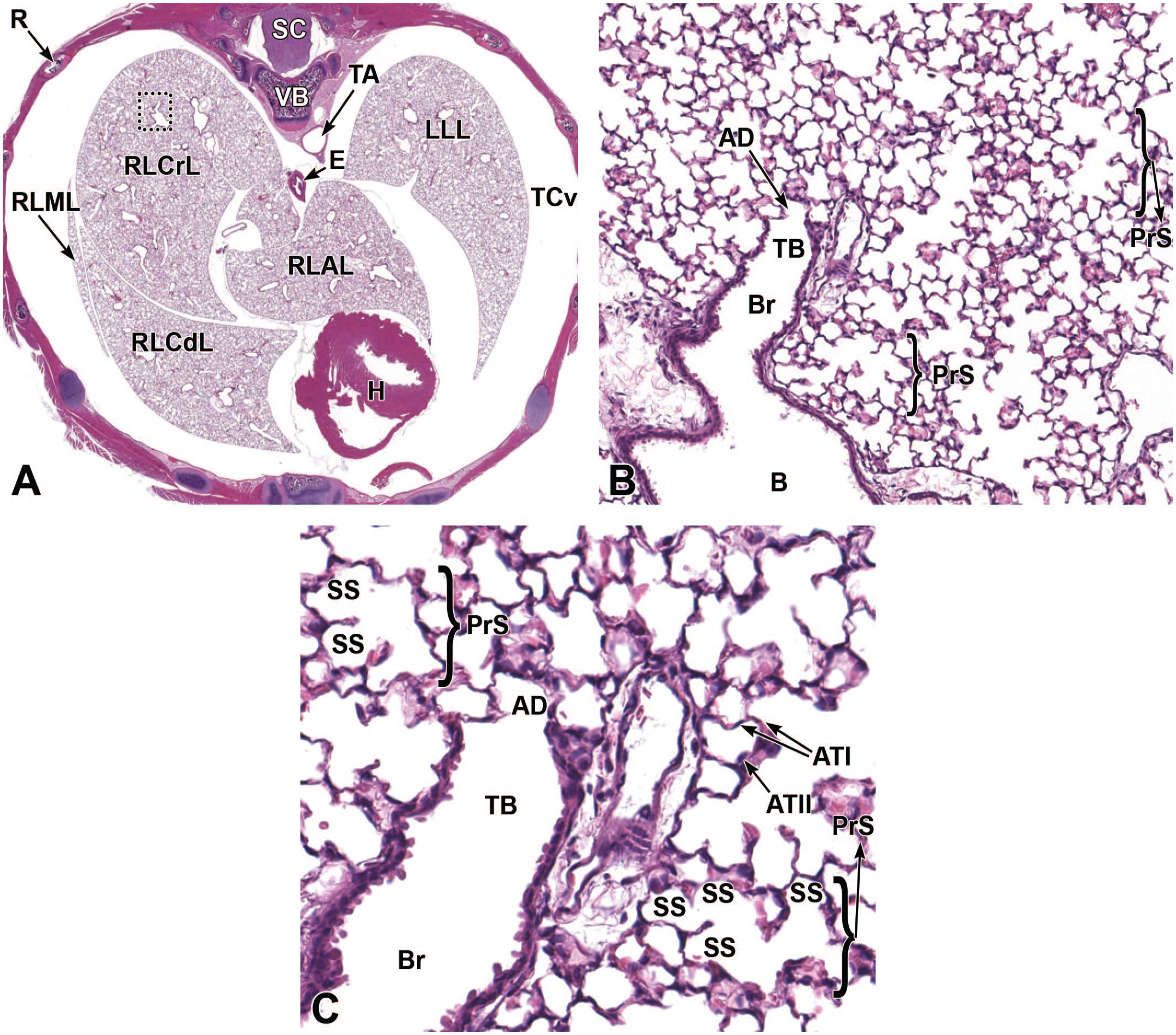
Transverse images of the alveolar stage of lung development at P5. Panel A (1×) illustrates the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), right lung caudal lobe (RLCdL), right lung accessory lobe (RLAL), and left lung lobe (LLL). Panel B (17×, depicting the field in the dashed box in panel A) shows the bronchi (B) leading to bronchioles (Br), terminal bronchioles (TB), alveolar ducts (AD), and primary saccules (PrS [denoted by brackets]). Panel C (40×) illustrates subdivision of the primary saccules into secondary saccules (SS). The saccules are lined with squamous alveolar type I (ATI) cells and cuboidal surfactant-secreting alveolar type II (ATII) cells. At this stage, the interstitial tissue within the alveolar walls is decreased in amount and density. The spinal cord (SC); vertebral body (VB); thoracic aorta (TA); esophagus (E); thoracic cavity (TCv); heart (H); and rib (R) are labeled for orientation. H&E.
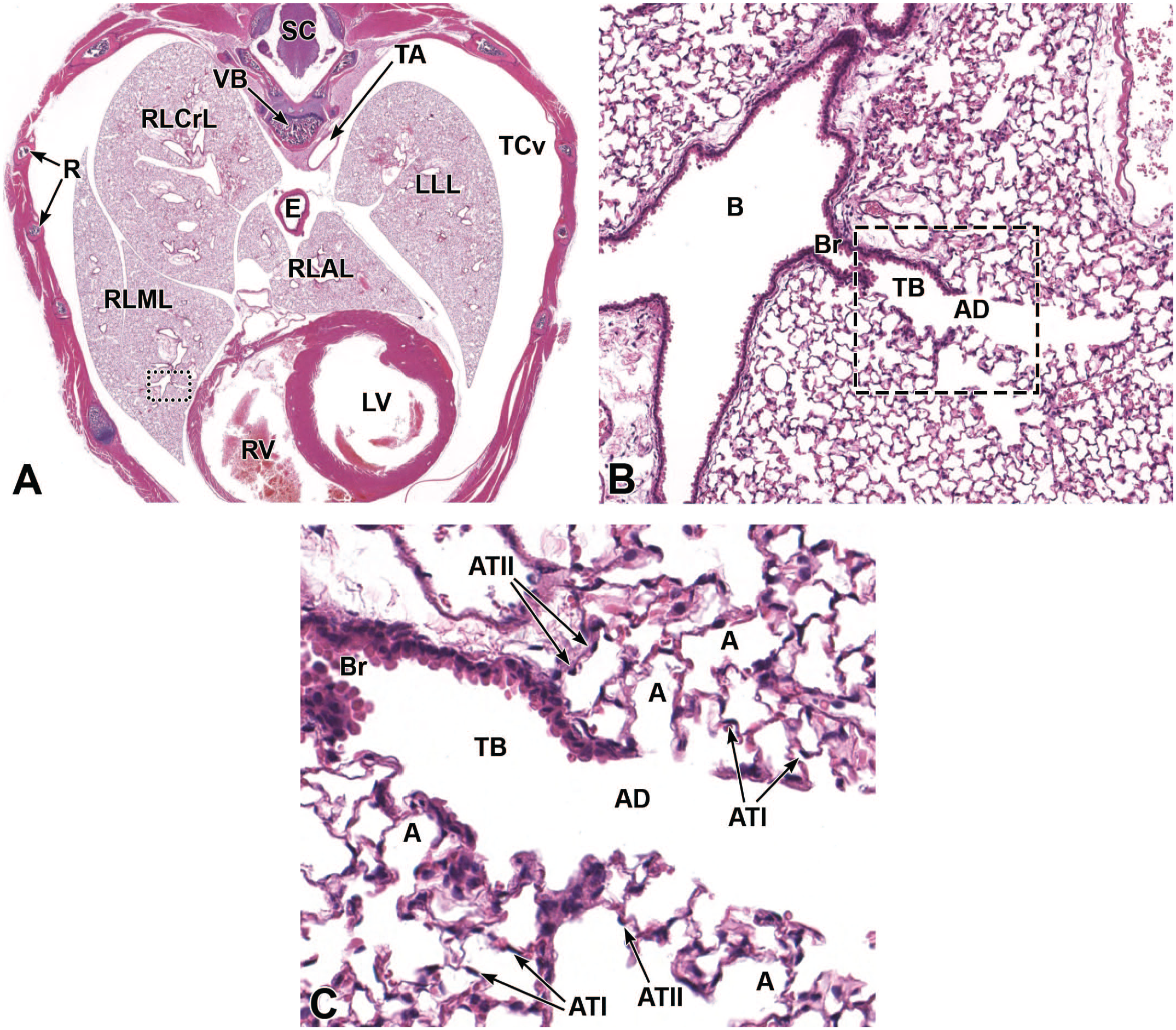
Transverse images of the alveolar stage of lung development at P30. Panel A (1×) depicts the right lung cranial lobe (RLCrL), right lung middle lobe (RLML), right lung accessory lobe (RLAL), and left lung lobe (LLL). Panel B (13×, field in the dashed box in panel A) shows the bronchi (B) leading to a bronchiole (Br), terminal bronchiole (TB), and alveolar duct (AD). Panel C (40×, field in the dashed box in panel B) illustrates the alveolar duct opening into the fully formed alveoli (A) that are lined with squamous alveolar type I (ATI) and surfactant-secreting cuboidal alveolar type II (ATII) cells. The spinal cord (SC); vertebral body (VB); thoracic aorta (TA); esophagus (E); thoracic cavity (TCv); the right (RV) and left (LV) ventricles of the heart; and ribs (R) are labeled for orientation. H&E.
Microvascular maturation in alveolar walls occurs in parallel with secondary septation. The outer layer of the double capillary network within the primary septa folds to support the new secondary septa, creating a new set of double-layered capillaries.13,111 Subsequently, the septa thin and the double-layered capillary networks fuse to form a single-layered capillary network held within a thin septum (Figure 69). 119 The airway fluid is cleared, alveolar sacs increase in size, and increases in oxygen and blood circulation cause the alveolar capillary surface areas to expand. 111 Retinoic acid is produced by alveolar capillary endothelial cells, which causes elastin to be synthesized in myofibroblasts as a factor for regulating capillary morphogenesis. 111
Diagrammatic representation of microvascular maturation during the alveolar stage of lung development. The top image shows a primary septum of the primitive alveoli with squamous alveolar type I cells (which line most of the inner surface of the alveoli) and fewer cuboidal alveolar type II cells (which secrete surfactant and serve as stem cells). The mesenchymal pericyte cells interact with the endothelial cells of the alveolar capillaries and alveolar type II cells to modulate alveolarization. The middle image shows the formation of the secondary septa and double capillary layer, demonstrating that the outer layer of the double capillaries within primary septa fold to create a new set of double-layered capillaries. The bottom image shows that as the septum thins, the double capillary layer will fuse to form a single-layered capillary network within the septum.
The basement membrane in alveolar septa is dense and amorphous with discontinuities beneath the ATII pneumocytes, allowing penetration of cytoplasmic processes into the interstitium. The sparse interstitial tissue contains an extracellular network of banded collagen fibrils forming fibers. 94 The most abundant interstitial cells are fibroblasts, including myofibroblasts with contractile properties that can dynamically alter septa shape to reflect shifting physiological needs for gas exchange. Elastin fibers, alveolar macrophages, and pericytes are also present. 111
Abnormal Respiratory Tract Development
Structural defects affecting the respiratory tract, whether directly or indirectly, are among the most common human congenital anomalies. Congenital deviation (curving or tortuosity) of the nasal septum affects up to 60% of neonates and 90% of adults. 138 Palatoschisis (cleft palate), a skeletal defect that secondarily leads to incomplete closure of the nasal passage floor, has a reported incidence of 0.3 to 1 in 1000 births.125,145
Mice exhibit a similar range of defects affecting the upper and lower respiratory tracts. The most frequent abnormalities in the upper respiratory tract of mice (Table 3) include gross malformations of the nose and nasal septum and microscopic anomalies of various nasal epithelial types. The most common defects in the lower respiratory tract of mice (Table 4) include macroscopic tracheal malformations, abnormal positioning and/or size of various lung lobes, altered bronchial tree branching, and divergent conformation of pulmonary alveoli and/or vasculature. Neoplasia is a regular phenotype in the lungs of mice but is rare in other parts of the respiratory tract. Incidences of these findings may be increased following exposure to toxic agents either in utero (by maternal encounters with teratogens) or after birth. Supplemental Table 3 contains descriptions of common transcription factors and molecular signals that contribute to, or are responsible for, abnormal respiratory tract phenotypes in mouse models.
Lethal developmental phenotypes affecting the respiratory system proper commonly manifest at delivery (i.e., in 1 to 2 hours or less of birth). Such conditions reflect an inability to shift from transplacental (maternal-based) gas exchange in utero to independent intake of oxygen. Respiratory insufficiency is easily diagnosed in instances where neonates possess small32,64 or no91,116 lungs. Respiratory complications secondary to cleft palate may be lethal, albeit less immediately, including aspiration pneumonia (via inhaled milk while nursing) and severe rhinitis (which may penetrate the cribriform plate and induce meningitis). These causes of lethality will be visible during necropsy and/or the microscopic evaluation.
An important consideration when investigating mouse models of respiratory disease is that some causes of respiratory failure result from abnormalities located outside the respiratory tract. For example, lethal respiratory insufficiency may arise from subtle lesions like neuronal reductions in brainstem autonomic centers that control the respiratory rate,10,44,78 abnormal synaptic transmission at neuromuscular junctions,40,182 skeletal malformations (of axial bones like the ribs and vertebrae) that constrain expansion of the thorax,85,102 and skeletal muscle paralysis.68,128 These defects may (brainstem neuron numbers, skeletal malformations) or may not (functional deficits affecting intercostal muscles) be detectable by structural analyses, but in general, special techniques beyond routine microscopic evaluation will be necessary to confirm their existence.
Conclusions
The increasing use of mouse models (especially engineered gene targeted [knockout] and transgenic strains) in genomic studies has provided extensive insights into the genetics, anatomy, physiology, and etiology of human diseases. With the high worldwide incidence of respiratory disease (asthma, COPD, pneumonia, and so on) coupled with a recent pandemic caused by the rampant global spread of SARS-CoV-2, it is imperative that researchers possess a variety of comprehensive resources to investigate respiratory diseases. This atlas provides biomedical scientists and pathologists an in-depth overview of the normal microscopic features of the murine respiratory tract during each stage of development. This atlas should allow investigators to identify developmental abnormalities that may be encountered when studying mouse models of respiratory disease, thereby enhancing our ability to understand and treat severe respiratory diseases in humans.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241252114 – Supplemental material for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30
Supplemental material, sj-docx-1-tpx-10.1177_01926233241252114 for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30 by Vanessa R. Parslow, Susan A. Elmore, Robert Z. Cochran, Brad Bolon, Beth Mahler, David Sabio and Beth A. Lubeck in Toxicologic Pathology
Supplemental Material
sj-docx-2-tpx-10.1177_01926233241252114 – Supplemental material for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30
Supplemental material, sj-docx-2-tpx-10.1177_01926233241252114 for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30 by Vanessa R. Parslow, Susan A. Elmore, Robert Z. Cochran, Brad Bolon, Beth Mahler, David Sabio and Beth A. Lubeck in Toxicologic Pathology
Supplemental Material
sj-docx-3-tpx-10.1177_01926233241252114 – Supplemental material for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30
Supplemental material, sj-docx-3-tpx-10.1177_01926233241252114 for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30 by Vanessa R. Parslow, Susan A. Elmore, Robert Z. Cochran, Brad Bolon, Beth Mahler, David Sabio and Beth A. Lubeck in Toxicologic Pathology
Supplemental Material
sj-docx-4-tpx-10.1177_01926233241252114 – Supplemental material for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30
Supplemental material, sj-docx-4-tpx-10.1177_01926233241252114 for Histology Atlas of the Developing Mouse Respiratory System From Prenatal Day 9.0 Through Postnatal Day 30 by Vanessa R. Parslow, Susan A. Elmore, Robert Z. Cochran, Brad Bolon, Beth Mahler, David Sabio and Beth A. Lubeck in Toxicologic Pathology
Footnotes
Acknowledgements
The authors wish to thank Drs. Priyanka Thakur and Sonika Patiel (NIEHS) for their careful review and edits.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the U.S. National Institutes of Health (NIH), specifically the U.S. National Institute of Environmental Health Sciences (NIEHS).
Author’s Note
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


