Abstract
A critical event in embryo development is the proper formation of the vascular system, of which the hepatobiliary system plays a pivotal role. This has led researchers to use transgenic mice to identify the critical steps involved in developmental disorders associated with the hepatobiliary vascular system. Vascular development is dependent upon normal vasculogenesis, angiogenesis, and the transformation of vessels into their adult counterparts. Any alteration in vascular development has the potential to cause deformities or embryonic death. Numerous publications describe specific stages of vascular development relating to various organs, but a single resource detailing the stage-by-stage development of the vasculature pertaining to the hepatobiliary system has not been available. This comprehensive histology atlas provides hematoxylin & eosin and immunohistochemical-stained sections of the developing mouse blood and lymphatic vasculature with emphasis on the hepatobiliary system between embryonic days (E) 11.5–18.5 and the early postnatal period. Additionally, this atlas includes a 3-dimensional video representation of the E18.5 mouse venous vasculature. One of the most noteworthy findings of this atlas is the identification of the portal sinus within the mouse, which has been erroneously misinterpreted as the ductus venosus in previous publications. Although the primary purpose of this atlas is to identify normal hepatobiliary vascular development, potential embryonic abnormalities are also described.
Keywords
Introduction
Normal development of the vascular system is essential for individual organ development. Without normal vasculogenesis, organogenesis would not occur and embryonic death would ensue. The largest internal organ, the liver, depends heavily on proper vasculogenesis to initiate development and growth during embryo and adult life stages (Zorn 2008). It is essential that the liver be capable of performing important functions such as producing the main source of hematopoietic cells during embryonic life, metabolizing nutrients, detoxifying harmful substances, producing blood clotting proteins, and many other essential functions (Crawford, Foley, and Elmore 2010). Previously, the development of the hepatic blood and lymphatic vasculature has not been studied in a systematic and detailed fashion. In humans, the initial ability to study the microanatomy of the liver depended on the availability of material and proper interpretation/analysis, which were both challenging. It was not until the development of ultrasound imaging that a more detailed analysis of the hepatic vasculature was possible; even so, there remain limits to the analysis of very small caliber vessels in adult and embryonic animals. With the advent of genetically engineered animal models, especially the mouse, it is now possible to assess the impact of specific genetic changes on hepatic vascular development.
Resources of general rodent embryonic development that can be utilized for investigative studies include, but are not limited to, M. H. Kaufman’s (1999) detailed histology atlas of mouse development, Sulik and Bream’s online scanning electron micrograph tutorial (http://www.med.unc.edu/embryo_images/), and the online Edinburgh mouse atlas of 3-dimensional (3-D) digital images (http://www.emouseatlas.org). Additionally, The Jackson Laboratory provides an online Mammalian Phenotype Browser that includes abnormalities of all murine organ systems (http://www.informatics.jax.org). The list of liver vasculature abnormalities provided by this website has been compiled in Table 1.
Abnormal Hepatic Vascular Phenotypes Found in Transgenic Mice.

Note. Main categories of abnormal hepatic vascular phenotypes are indicated in bold and subcategories are italicized. The number of genotypes with a certain hepatic vascular phenotype are indicated. One phenotype may have several different abnormalities. These data were collected from the Mouse Genome Database (MGD), Mouse Genome Informatics website, The Jackson Laboratory, Bar Harbor, Marine (http://www.informatics.jax.org), retrieved July 2015. (Search: Phenotypes & Mutant Alleles; Phenotypes, Alleles & Disease Models Query; Anatomical Systems Affected by Phenotypes; liver/biliary system; Full mammalian Phenotype [MP] Ontology; liver/biliary system phenotype; abnormal hepatobiliary system morphology; abnormal liver morphology; abnormal liver vasculature morphology; etc.).
The goal of this atlas is to describe, using low- and high-magnification color images, the developing embryonic mouse hepatobiliary vascular system. Both hematoxylin & eosin (H&E) and immunohistochemical (IHC) stains were used. Furthermore, select H&E images have been used to construct a 3-D rendering of the hepatobiliary blood vascular system at embryonic day (E) 18.5. Detailed documentation regarding organogenesis of the liver, gallbladder, or bile ducts is not highlighted in this atlas. The main morphological changes that occur during mouse gestation with regard to the hepatobiliary vasculature are summarized in Table 2. However, due to the potential for strain differences, a similarly matched control group should always be used when comparing vascular development; littermates are ideal. The findings reported in this atlas are based on the CD1 mouse strain; developmental differences may be noted when comparing other strains. Abnormalities may also vary depending on the strain. Only selected abnormalities were chosen for review based on the prevalence, level of severity, and/or the relative significance to human medicine.
The digital images and video can be viewed online at https://niehsimages.epl-inc.com with the username “ToxPathLiver” and the password “embryolivers.”
Main Morphological Changes during Mouse Gestation.
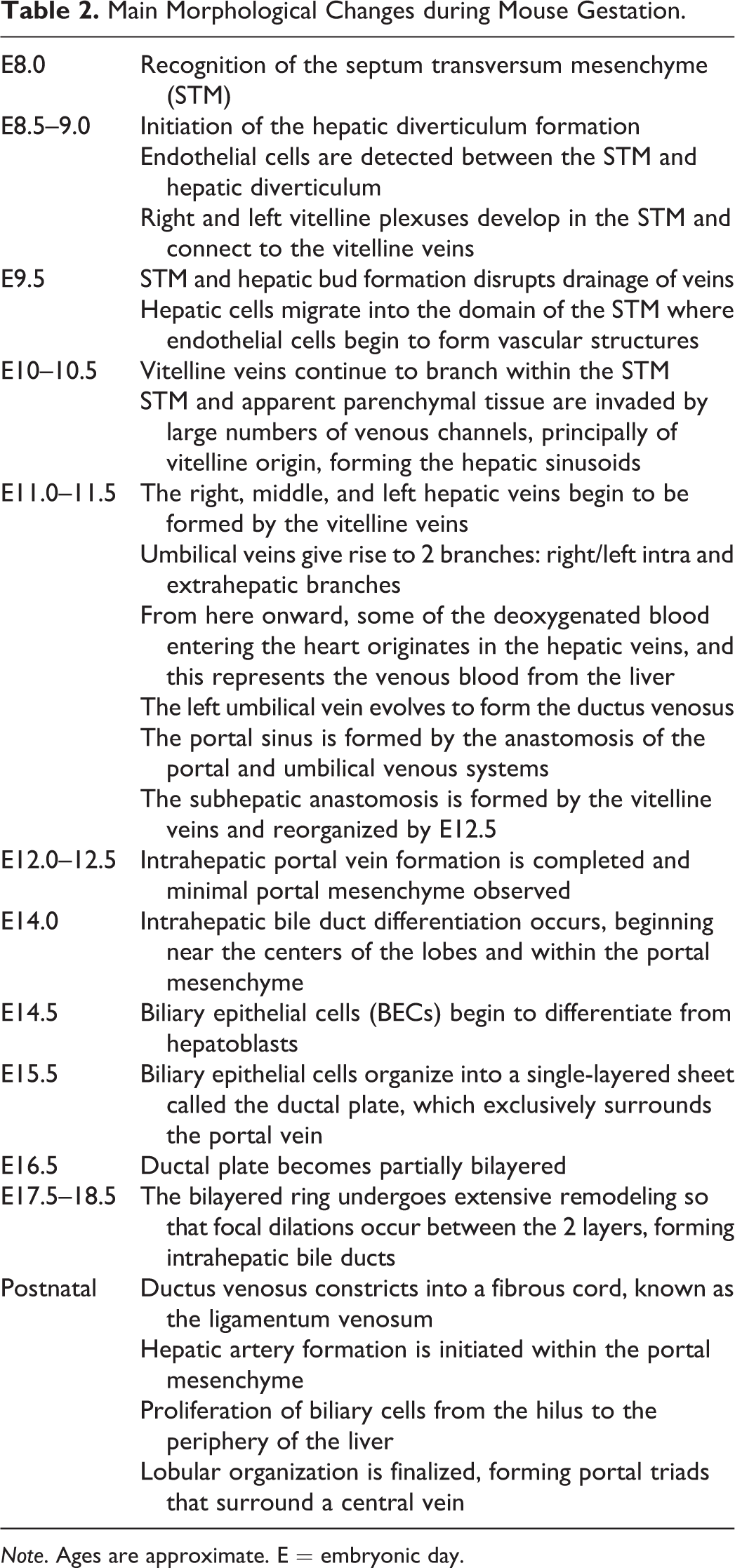
Note. Ages are approximate. E = embryonic day.
Material and Method
Animals
CD-1 IGS mice/Crl: CD1 (ICR) timed pregnant dams (Charles River Laboratories, Raleigh, NC) were used. All animal procedures used in this study were approved by the National Institute of Environmental Health Sciences Animal Care and Use Committee.
Staging
The morning on which the vaginal plug was found was referred to as E0.5 (in the literature, it is also described as 0.5 days postconception). Since considerable variation occurs in the timing of ovulation and conception and in the developmental status of individual embryos, even within the same litter, special care was taken to match both the external and the internal features of each embryo to the known developmental landmarks (Kaufman 1999).
Collection of the Embryos
Collection was carried out on the mornings of the designated days (E9.5–E18.5). Pregnant mice were euthanized by carbon dioxide. Individual embryos were isolated under the dissection microscope from the uterus, and extraembryonic membranes were immersed in cold 0.1 M phosphate buffered saline and then transferred to fixative.
Fixation
Embryos were fixed in Bouin’s solution (Poly Scientific, Bay Shore, NY) or 10% neutral buffered formalin (NBF; IMEB Inc., San Marcos, CA). Tissue fixed in 10% NBF was used for immunohistochemistry. Fixation time for both fixatives was dependent on the embryonic age: E9.5 to E11.5—2 hr, E12.5 to E16.5—4 hr, and E17.5 to E18.5—72 hr. Following fixation with Bouin’s solution, embryos were rinsed in 70% ethanol saturated with lithium carbonate (Sigma-Aldrich, St. Louis, MO) for three 30-min washes and then processed for paraffin embedding. Embryos younger than E13.5 were embedded in 1% agar before submission to minimize handling during paraffin embedding. For each time point, embryos were embedded on their backs, sides, or heads for sectioning in the respective frontal (coronal), sagittal (longitudinal), or transverse (horizontal) plane. Serial 6-µm sections through the entire embryo were placed on charged slides (A. Daigger & Company, Vernon Hills, IL) and routinely stained with H&E for histopathology review.
Scanning
Digital images were captured from H&E and IHC-stained slides scanned on the Aperio ScanScope XT instrument (Vista, CA) using ImageScope software, (v9.0, Aperio). If a digitally scanned image required rotation, the selected region of interest was captured using the extraction feature in ImageScope. White balance correction and image resizing were completed using Adobe Photoshop (Adobe Photoshop Creative Cloud 2014.0.0; Adobe Systems Incorporated, San Jose, CA). Image resolution was set at 300 dpi to fit the publisher’s requirements.
Immunohistochemistry
Indirect IHC staining was performed on formalin-fixed, paraffin-embedded embryos. The tissues were deparaffinized in xylene and rehydrated through a graded series of ethanol. Endogenous peroxidase was blocked by immersing slides in 3% hydrogen peroxide for 15 min. A summary of the immunohistochemistry reagents is provided in Table 3. Detailed protocols are provided on the National Institute of Environmental Health Sciences (NIEHS) immunohistochemistry website (http://www.niehs.nih.gov/research/atniehs/labs/lep/path-support/immuno/protocols.cfm).
Immunohistochemical Stain Summary.

Note. CK = cytokeratin; SMA = smooth muscle actin; LYVE1 = lymphatic vessel endothelial hyaluronan receptor 1.
aAbcam, Cambridge, MA.
bBiocare Medical, Concord, CA.
cEMD Millipore, Billerica, MA.
dVector Laboratories, Burlingame, CA.
3-D Imaging
A 3-D reconstruction was produced from serial sectioned, H&E-stained slides using Adobe After Effects. A detailed description of the technique was described elsewhere (Livingston and Tuong 2014). Briefly, sequential individual images (pixel-based, JPEG2000) were imported into Adobe After Effects and manually aligned with each other. The venous vascular system was isolated from the parenchyma of the liver by rotoscoping (Livingston et al. 2014) and colored. Color keying was used to digitally clear background and allow visualization of the vessels in isolation. The animation was created within Adobe After Effects.
Overviews of General Vascular Development and Early Embryo Hepatic Blood Circulation
The following sections provide overviews on vasculogenesis/angiogenesis, arteriovenous differentiation, lymphatic vascular development, and early embryo hepatic blood circulation. Prior to exploring the chronological order of the hepatobiliary vascular development, it is helpful to have background information on these topics. Selected hepatobiliary structural or hematopoietic events are discussed briefly in these sections when it pertains to an appropriate stage of vascular development.
Vasculogenesis and Angiogenesis
One of the most important events that occurs in the developing mammalian embryo is the formation of the vascular system. Without the proper construction of this system, the embryo will be unequipped to carry out future developmental milestones, thus leading to embryonic death. For this reason, scientists, dating back to the early 20th century, have heavily investigated the vascular system. Recent monumental discoveries have been attributed to advances in technology (IHC, imaging, etc.) and the usage of transgenic mice to map the processes pertaining to the development of this system (Sato and Loughna 2002).
The earliest sign of vascular development is apparent during gastrulation (Sato and Loughna 2002). This phase occurs at E6.0 directly after a 2-day postimplantation period that drastically changes the appearance of the embryo. The purpose of gastrulation is to form 3 germ layers, the ectoderm, mesoderm, and endoderm, which are responsible for organogenesis at a later time (Tam and Behringer 1997). Prior to organ formation, the vascular network must be arranged by 2 subsequent processes known as vasculogenesis and angiogenesis (Sato and Loughna 2002).
The initial vascular development is characterized by the differentiation of angioblasts, a resident mesodermal cell, into endothelial cells (Dumont et al. 1995). The latter forms the entirety of a vascular network known as the primary capillary plexus. Within the plexus, endothelial cells group together to form clusters, known as blood islands, which create a uniform honeycomb-like network of capillaries. The completion of the capillary plexus is the end product of vasculogenesis (Sato and Loughna 2002).
Angiogenesis is the next phase of this critical process. It involves sprouting and nonsprouting processes, both of which necessitate endothelial cell movement into new tissue territory. Sprouting is the less complicated of the 2 processes and requires endothelial cells from the primary capillary plexus to penetrate nearby tissue by elongating their shape to create new vessels. The process of nonsprouting is less understood but is defined as the invasion of an established vascular channel by surrounding cells which causes the division of the vessel, thus increasing the capillary network. The final step in angiogenesis is the incorporation of smooth muscle cells into the vascular channels to provide rigidity and contractility to the vessel wall (Sato and Loughna 2002).
It is essential during angiogenesis that the growth of new blood vessels is tightly regulated (Breier et al. 1992). A reduction in endothelial cells can result in undervascularization and, if severe, can lead to embryo death (Sato and Loughna 2002). This is prevented by vascular endothelial growth factor (VEGF), a proangiogenic, soluble glycoprotein, expressed prior to gastrulation to control the endothelial population (Maharaj and D’Amore 2007). VEGF consists of 7 unique family members heterogeneously expressed in embryonic and adult tissues. It stimulates endothelial cells by means of 3 known receptors: Flt-1 (VEFGR-1), Flk-1 (VEFGR-2), and Flt-4 (VEGFR-3). The primary 2 receptors expressed on blood vascular endothelial cells are Flt-1 and Flk-1 (Hoeben et al. 2004). VEGFR-3 is expressed initially on venous endothelial cells and later becomes solely expressed on lymphatic endothelial cells (Sato and Loughna 2002). Any variation in this crucial glycoprotein/receptor relationship can lead to abnormalities, which will be discussed later at specific developmental stages.
Arteriovenous Differentiation
In the adult stages, arteries and veins can be differentiated by anatomic and hemodynamic differences. During early embryonic development, both vessel types are tubular structures lined by endothelial cells and smooth muscle (H. U. Wang, Chen, and Anderson 1998). This structural similarity makes it difficult to distinguish the 2 vessels during early embryo development. For this reason, factors initiating venous versus arterial differentiation during vasculogenesis have been researched (Sato and Loughna 2002). In late embryonic development, knowing the anatomic location of an embryo vessel can help with identification.
To help differentiate arteries from veins during early embryonic development, endothelial cell markers are utilized. The first markers discovered for arteriovenous differentiation were ephrinB2 and EphB4, expressed by arterial and venous endothelial cells, respectively (Fuller et al. 2003; Swift and Weinstein 2009). During embryogenesis, endothelial cells express Eph, a tyrosine kinase receptor that is activated by ephrin ligands. This expression, along with a variety of molecular factors, supports vessel formation and differentiation (Swift and Weinstein 2009). Although ephrinB2 and EphB4 are commonly used arteriovenous markers, the expression of these markers during early stages of angiogenesis is weak and detection can be challenging. A number of other markers are now used, including arterial markers Cx37, Cx40, Hey1, Nrpl, and Dll4, and venous markers endomucin, Nrp2, Flt4, CoupTFII, and APJ (Chong et al. 2011).
Arteriovenous differentiation progresses through a series of stages that initially begins with the expression of a limited amount of arterial markers, as early as E8. Therefore, using IHC markers to detect arteriovenous differentiation prior to E8 may be futile. As the embryo matures, however, the expression of these markers increases. Once arterial specification occurs, venous expression is subsequently initiated. At E8.25, weak venous signals can be noted, but it is not until E8.5 that venous markers are strongly present in the vitelline vein angioblasts (Chong et al. 2011).
In the liver, the vitelline veins are the first vessels to infiltrate the organ. When the vitelline vein matures into its adult counterpart, the portal vein, the mesenchyme surrounding the vessel plays a key role in vasculogenesis. Interactions between the venous vasculature and portal mesenchyme drive the differentiation of hepatoblasts into biliary epithelial cells (BECs), which mature into bile ducts that initiate intrahepatic arterial formation.
Lymphatic Vascular Development
For many years, the scientific interest of researching lymphatic development was minimal. It was only recently, with the use of lymphatic markers, that scientists began to extensively investigate the embryologic development of the lymphatic vasculature (Oliver 2004). During the first 2 decades of the 20th century, 2 contradicting models of lymphatic development were proposed. The first and most popular model to date was developed by Florence Sabin (1902) who proposed that lymphatic sacs are formed by the budding of endothelial cells from veins. On the opposing side, Huntington and McClure (1910) believed that lymphatic sacs formed separately from veins and established connections later in development. Although many questions still remain unanswered, scientists have been able to use Sabin’s theory to develop a 4-step process for lymphatic vascular development that occurs secondary to blood vascular development; these steps include competence, bias, specification, and differentiation (Oliver 2004).
The first step, competence, requires that the endothelial cells of the venous system be able to respond to an inducing signal (Oliver 2004). This signal is primarily generated by lymphatic vessel endothelial hyaluronan receptor 1 (LYVE1) but can be initiated by another gene if the embryo lacks LYVE1. Evidence confirming venous endothelial cell competence can be observed at E9–9.5 on the entire anterior cardinal vein. At E9.5, expression of the lymphatic transcription factor, prospero-related homeobox 1 (Prox1) is observed on a subpopulation of endothelial cells on one side of the anterior cardinal vein (Wigle et al. 2002). This specific segregation of cells confirms that lymphatic bias (step 2) is occurring. This step indicates that the select endothelial cells are now programmed to become lymphatic vessels (Oliver 2004). They will begin budding to form lymph sacs at E10–10.5. Two jugular lymphatic sacs are the initial sacs to be formed, which give rise to the first branches of the lymphatic vascular network. Shortly after, the retroperitoneal lymphatic sac forms near the primitive caudal vena cava and provides lymphatic branching to the abdominal viscera (Oliver 2004). During specification (step 3), budding increases markedly. Additional lymphatic markers associated with maturation are also observed; these include VEGFR3, Nrp2, and podoplanin (Oliver 2004). By E12.5, no arteriovenous markers are observed on these vessels, only lymphatic markers. Differentiation (step 4) is the final stage where lymphatic endothelial cells spread throughout the body to begin forming lymphatic capillaries and collecting vessels, at approximately E14.5 (Oliver 2004; van der Putte 1975).
Once the lymphatic vessels have matured, the fetus will be equipped with a blind-ended network of capillaries and collecting vessels that transport lymph contents to the thoracic duct to drain into the subclavian veins. A mature lymph vessel will have a layer of endothelial cells, smooth muscle, and adventitia (Schulte-Merker, Sabine, and Petrova 2011). Unlike the hepatic blood vascular system, the lymphatic system lacks fenestrations and instead has loose intercellular junctions that allow fluid to enter, but not leave, due to semilunar valves that prevent backflow (Oliver 2004; Schulte-Merker, Sabine, and Petrova 2011). For the untrained eye, it is difficult to correctly identify the lymphatic vessels within the developing mouse liver using H&E staining. Any abnormality in the development or function of this system can result in minimal to severe consequences.
Early Embryo Hepatic Blood Circulation
The blood circulatory system in the mammalian fetus is considerably different than that of the adult (Figure 1A–C). From the placenta, highly oxygenated blood enters the umbilical vein and travels to the liver. The umbilical veins (left and right branches) are first noticed at E8–8.5 running on the lateral sides of the vitelline veins (Figure 1A and B). Originally, the umbilical veins are symmetric in diameter, but as the embryo ages, they become asymmetric; a change that is first noticed at E10.5–11.0. By 11.5, right and left extrahepatic and intrahepatic branches of the paired umbilical veins have formed (Figure 1C). Approximately 24 hr later, all portions of the right umbilical vein will regress along with the left extrahepatic portion. Thus, the intrahepatic branch of the left umbilical vein is the only portion of the vessel that remains intact. The ductus venosus, a narrow shunt, is formed from a portion of the left intrahepatic umbilical vein.
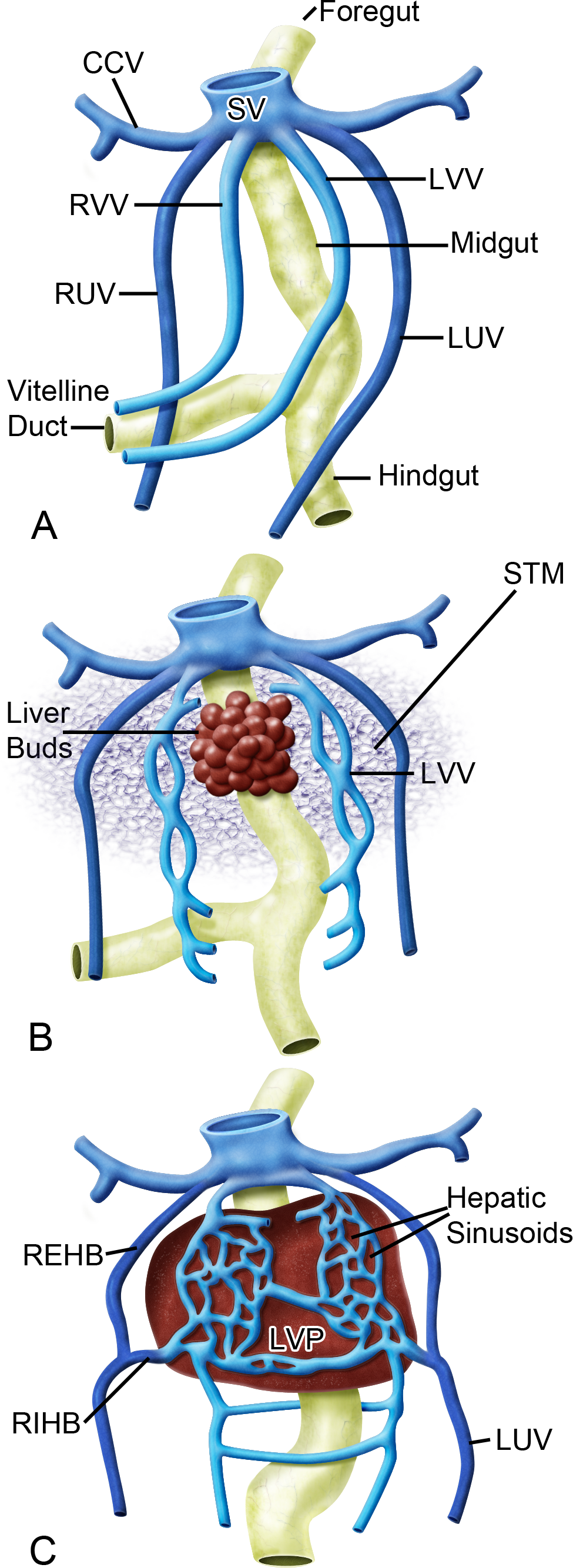
Development of venous drainage within the liver. Prior to development of the septum transversum and liver, the vitelline veins drain the primitive gut. The umbilical veins carry oxygenated blood from the placenta and run on the lateral sides of the vitelline veins (A). Later in development, the liver bud can be seen developing at the foregut–midgut junction (B). As the liver grows, changes to the vascular system occur. The vitelline veins enter and ramify within the septum transversum, forming the hepatic sinusoids. The 2 umbilical veins give rise to both an intra- and extrahepatic branch (C). Adapted with permission from Crawford, Foley, and Elmore (2010, 877, figure 2). CCV = common cardinal vein; LUV = left umbilical vein; LVP = liver parenchyma; LVV = left vitelline vein; REHB = right extrahepatic branch of umbilical vein; RIHB = right intrahepatic branch of umbilical vein; RUV = right umbilical vein; RVV = right vitelline vein; STM = septum transversum mesenchyme; SV = sinus venosus.
The vitelline veins also play a pivotal role in hepatic circulation. In early embryo development, these veins, which are divided into proximal, middle, and distal segments, are responsible for transporting deoxygenated blood from the yolk sac (Washington Smoak 2007). The paired left and right veins are first recognized at E8, making them the first vessels of the venous system to be formed in the developing embryo (Kaufman 2008). At E8.5–9, the middle segment begins forming a venous plexus within the septum transversum mesenchyme (STM; Figure 1B). The plexus ultimately unites with the hepatic sinusoids to form an even larger plexus at approximately E11.5 (Figure 1C). By E12–12.5, the distal segment of the right vitelline vein gives rise to the portal vein, which branches within the liver to drain poorly oxygenated blood into the sinusoids. Prior to exiting the liver, blood from the sinusoids enters the hepatic veins, which are also formed by the right vitelline vein. From there, blood enters the posthepatic caudal vena cava, which is formed by a portion of the proximal segment of the right vitelline vein (Crawford, Foley, and Elmore 2010; Kaufman and Bard 1999; Washington Smoak 2007).
With the differentiation and maturation of the umbilical and vitelline veins, these afferent vessels now provide the liver with 2 pathways of entry for blood flow. Blood can travel into the vascular channels of the liver via branches of the portal vein or it can flow into the ductus venosus to provide highly oxygenated umbilical blood to the heart. Regardless of the route of entry, all efferent vessels (ductus venosus and hepatic veins) will converge in the caudal vena cava to transport blood out of the liver and to the heart (Bort and Zaret 2009).
Development at Various Embryonic Stages
Early Vascular Development
E8.5–10.5
At E8.5–9, prior to the initiation of liver formation, the left and right vitelline veins are forming a venous plexus within the STM (the cranial mesenchyme that forms portions of the diaphragm and ventral mesentery of the foregut; Kaufman and Bard 1999). At E9–9.5, liver formation is initiated from cells along the ventral aspect of the distal foregut and spreads into the STM (Crawford, Foley, and Elmore 2010). The liver primordium then gives rise to a liver bud (or diverticulum) that continues to grow and mature throughout gestation, to form the lobular organization present in the adult liver.
Within the liver bud, epithelial cells proliferate and elongate into a sheet of cells that form hepatic cords (Figure 2A and B), which are separated from the STM by a basement membrane. During growth of the liver bud, the hepatic cords disrupt the basement membrane allowing hepatoblasts to enter the STM. The invasion of hepatoblasts disrupts the drainage of the vitelline venous plexus, at E9.5 (Kaufman and Bard 1999). The hepatoblasts are now closely associated with endothelial cells from the venous plexus, which initiates vasculogenesis within the liver (Matsumoto et al. 2001).
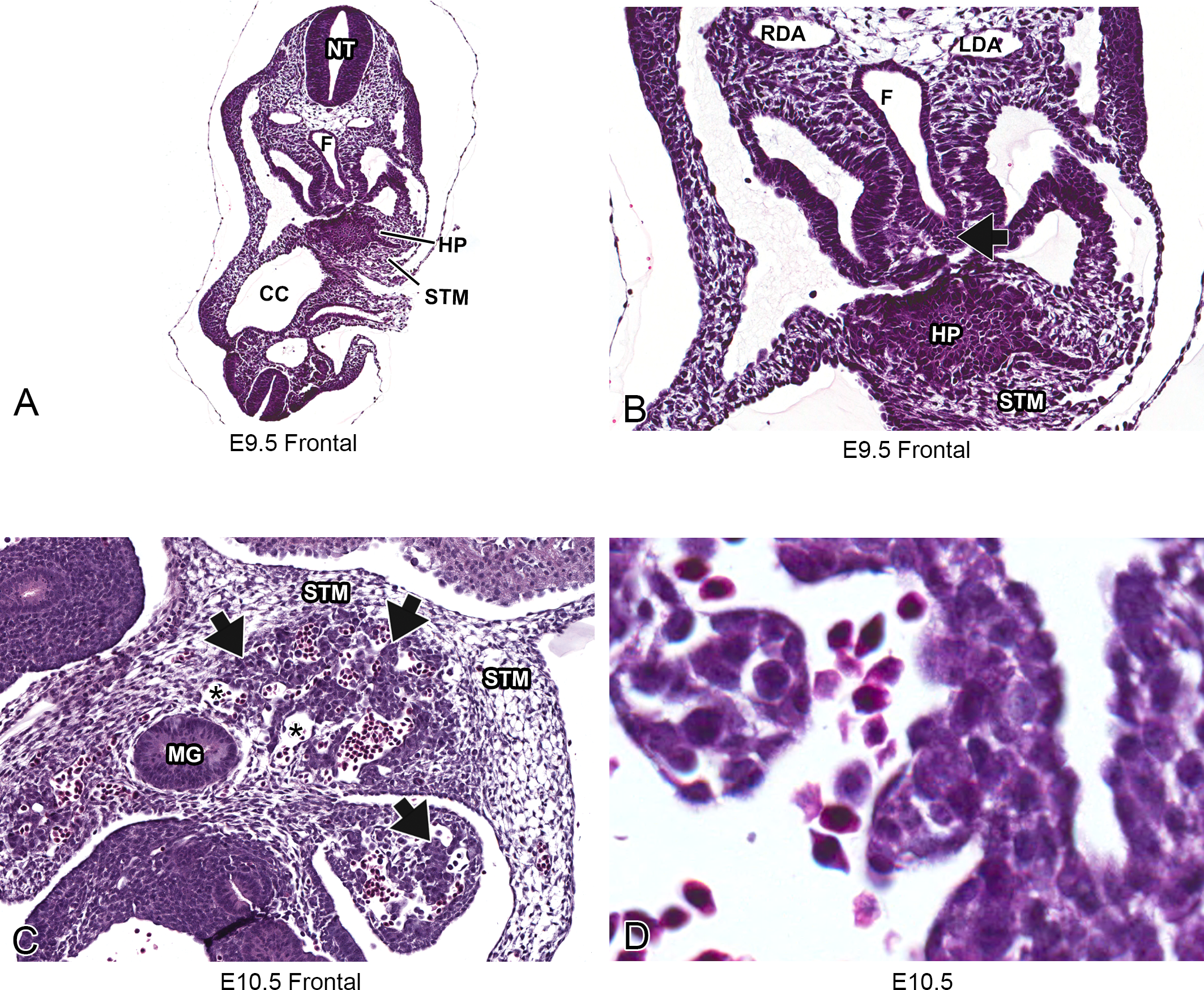
Representative images of embryonic day (E) 9.5 and E10.5 mouse liver. Low-magnification, hematoxylin & eosin-stained frontal sections at E9.5 (A and B) and E10.5 (C). High-magnification, H&E-stained frontal section at E10.5 (D). Histologically, formation of the liver begins around E9.5 within the STM when endodermal cells arising from the ventral surface of the caudal foregut form a diverticulum (A, B, and arrow). By E10.5, the vitelline venous plexus, formed by the vitelline veins, is branching throughout the STM. Hepatic sinusoids are recognized histologically as blood-filled, capillary-like vessels (asterisks) that are located between the hepatic cords (arrows) throughout the liver (C). At high-magnification, nucleated red blood cells can be seen within the sinusoids (D). CC = coelomic cavity; F = foregut; HP = hepatic primordium; LDA = left dorsal aorta; MG = midgut; NT = neural tube; RDA = right dorsal aorta; STM = septum transversum mesenchyme.
At E10–10.5, the vitelline venous plexus continues to branch within the STM and into the liver parenchyma (Crawford, Foley, and Elmore 2010). Large numbers of venous channels originating from the vitelline venous plexus are apparent at this stage. These channels, known as hepatic sinusoids, are recognized histologically as blood-filled, capillary-like vessels located between the hepatic cords throughout the liver (Figure 2C and D). The hepatic sinusoids comprise the majority of the volume of the liver.
Hepatic Vascular Development in Mid- to Late Gestation (E11.5–18.5)
E11.5
A significant event occurring between E11.5 and E12.5 is the formation of the ductus venosus. This newly constructed vessel arises from the left umbilical vein (Figure 3A) and forms the only intrahepatic shunt in the embryo. The ductus venosus is commonly misinterpreted as a large dilated extension of the umbilical vein, which shunts all oxygenated blood directly to the heart, bypassing the liver parenchyma entirely (Kaufman 2008). This definition has been erroneously used due to the lack of relevant data published on the anatomy of the mouse embryo liver vasculature. We report that the ductus venosus is in fact a narrow, branchless, tubular structure that steeply ascends cranially toward the caudal vena cava (Figure 3B). Although the ductus venosus originates from the left umbilical vein, these 2 vessels have no direct connection from this stage on. The vessels are interrupted by a large intrahepatic structure, called the portal sinus, which transports blood from the left umbilical vein to the ductus venosus (Figure 3A and B) as well as throughout the liver parenchyma via the portal system. Oxygenated blood from the left umbilical vein enters the portal sinus and is transported to the left intrahepatic portal vein and ductus venosus (Mavrides et al. 2001).
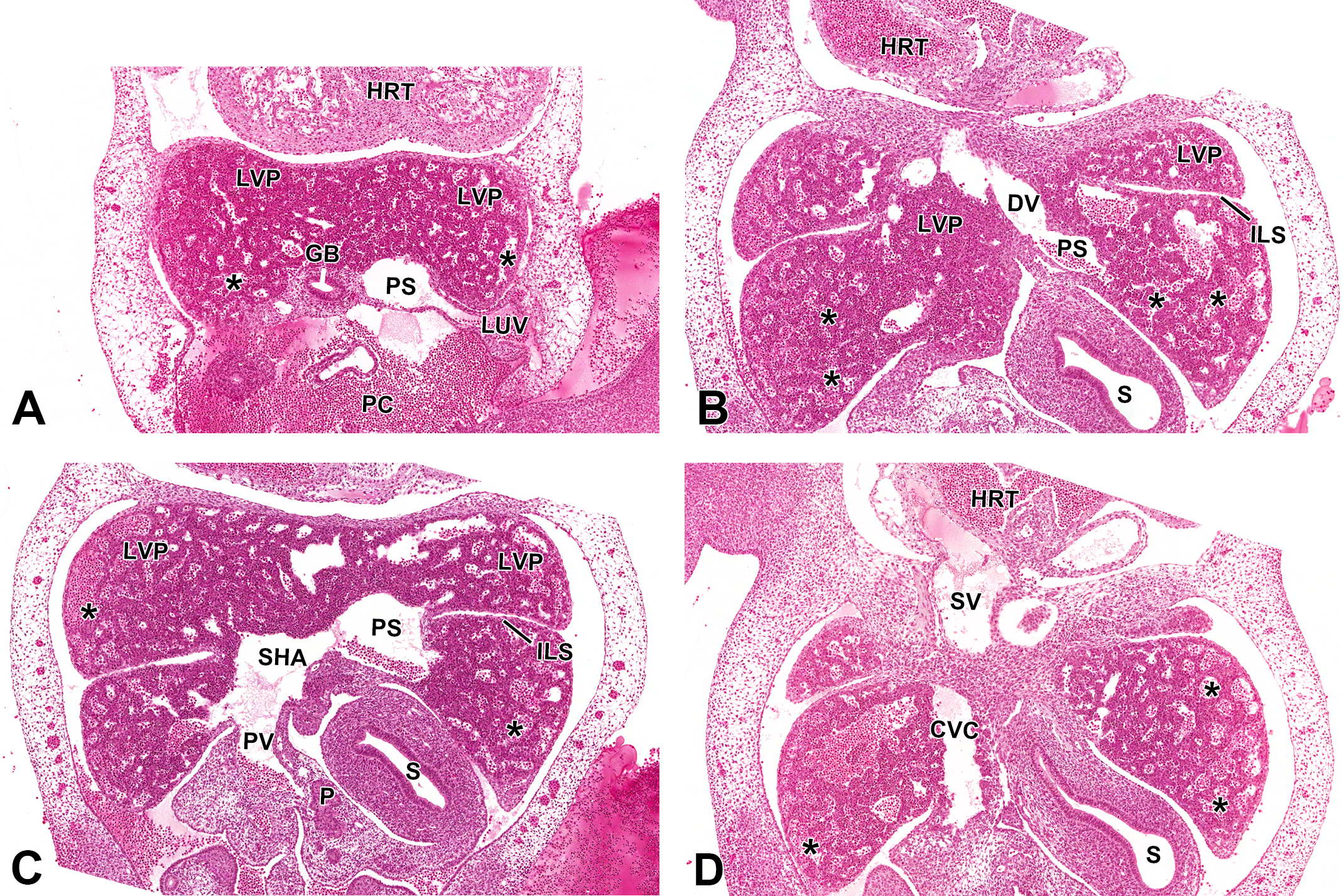
Representative images of the embryonic day (E) 11.5 mouse liver vasculature. Hematoxylin & eosin-stained (A, B, C, and D) frontal sections of the liver vasculature at low magnification from ventral to dorsal view, in a single embryo. The left umbilical vein enters from the left side of the liver and connects to the portal sinus (A). Once blood enters the portal sinus, it is distributed to the ductus venosus (B). Blood can also enter the liver through the portal vein, which at this stage, forms a confluence with the subhepatic anastomosis (C). Afferent blood flow then can enter the newly formed hepatic sinusoids (asterisks) or the majority will enter the portal sinus. The subhepatic anastomosis and portal vein should not be confused with the caudal vena cava, which at this stage appears to be intimately associated with the liver (D). CVC = caudal vena cava; DV = ductus venosus; GB = gall bladder; HRT = heart; ILS = interlobar space; LUV = left umbilical vein; LVP = liver parenchyma; P = pancreas; PC = peritoneal cavity; PS = portal sinus; PV = portal vein; S = stomach; SHA = subhepatic anastomosis; SV = sinus venosus.
Until now, limited information has been reported describing the portal sinus in mice. In human fetuses, it is described as a large L-shaped venous confluence that connects the left and right lobes via the right and left intrahepatic portal veins and is situated at the terminal end of the umbilical vein (Mavrides et al. 2001). A uniform term for this structure has still not been established by the scientific community. Various human studies referred to this structure as a portal sinus, umbilical sinus, sinus intermedius, or umbilical recess (Collardeau-Frachon and Scoazec 2008; Mavrides et al. 2001; Meyer and Lind 1966). We refer to this structure as the portal sinus due to the presence of portal mesenchyme and BECs surrounding the structure at later stages of development. Histologically at E11.5, this structure, along with the left portal vein, umbilical vein, and ductus venosus, is indistinguishable with regard to their cellular composition. Therefore, anatomic location and size within the liver is the best way to distinguish these vessels.
Transverse sections are ideal for observing the embryonic vascular architecture in later stages of development; however, frontal sections are more useful in early stages of development due to orientation of the vasculature (Figure 3A–D). It is important to note the use of serial sectioning in studying the hepatobiliary vasculature, as it is not possible to observe all vessels on a single histologic section. At this stage, large sinusoids can be observed throughout the liver (Figures 3C–D, asterisks). As the intrahepatic vitelline veins transform, various branches begin anastomosing throughout the parenchyma. The largest of these, known as the subhepatic anastomosis (Figure 3C), forms caudolateral to the portal sinus and conveys blood from the incoming portal vein to the sinusoidal plexuses that are dispersed within the parenchyma. By E12, the subhepatic anastomosis undergoes remodeling to form organized branches of the portal venous system.
At this stage in embryo development, no biliary or lymphatic formation has occurred within the liver. No IHC stains were performed at this stage due to conclusive evidence from multiple resources, indicating that the earliest development of these 2 systems, within the liver, is not until E14 (Crawford, Foley, and Elmore 2010; Lemaigre 2003; Oliver 2004). Although there is no lymphatic or biliary involvement within the liver at this stage, abnormal formation of either system can have a strong impact on the development and functionality of the liver.
Abnormal Development (E11.5)
The lymphatic transcription factor, Prox1, is essential for the initiation of a subpopulation of venous endothelial cells to begin differentiation into lymphatic endothelial cells, at E11.5 (Oliver 2004). Without Prox1 expression, the embryo will be unable to properly form enough lymphatic endothelial cells and will therefore lack lymphatic vessels that are required for later embryo and adult life (Wigle et al. 2002). A study comparing Prox1+/− to Prox1−/− mice showed that although both had no differences at E10.5, by E11.5, a substantial decrease in the number of Prox1-positive endothelial cells was present in Prox1−/− embryos (Wigle and Oliver 1999). Additionally, at E12.5, only a few endothelial cells were found undergoing lymphatic budding (Wigle and Oliver 1999). When lymphatic endothelial cells fail to bud, it results in developmental failure of lymph sacs, which subsequently inhibits lymphatic capillary networks and collecting vessels from forming (Oliver 2004). Using immunohistochemistry, Wigle and Oliver (1999) revealed that the Prox1−/− embryos lacked any sign of lymphatic vasculature development in all visceral organs; this in turn resulted in lethality of all embryos at approximately E14.5.
E12.5
By E12.5, the formation of the ductus venosus and portal sinus is complete and final transformations for the portal venous system are underway (Figure 4A–D). At the caudal aspect of the liver, the distal segment of the right vitelline vein has extended into the liver parenchyma via right and left branches that travel into the right and left lobes, respectively. Upon exiting the portal vein, blood then enters the hepatic sinusoids (Figure 4C). For the remainder of embryonic life, blood will enter the liver from 2 different pathways. All oxygenated blood from the left umbilical vein will enter the left portal sinus and distribute to the ductus venosus or intrahepatic branches of the left portal veins primarily supplying the left side of the liver. Deoxygenated blood from the extrahepatic portal vein almost exclusively supplies the right hepatic lobes. Rudolph and colleagues (1971) have documented a similar pattern of blood flow in human fetuses.
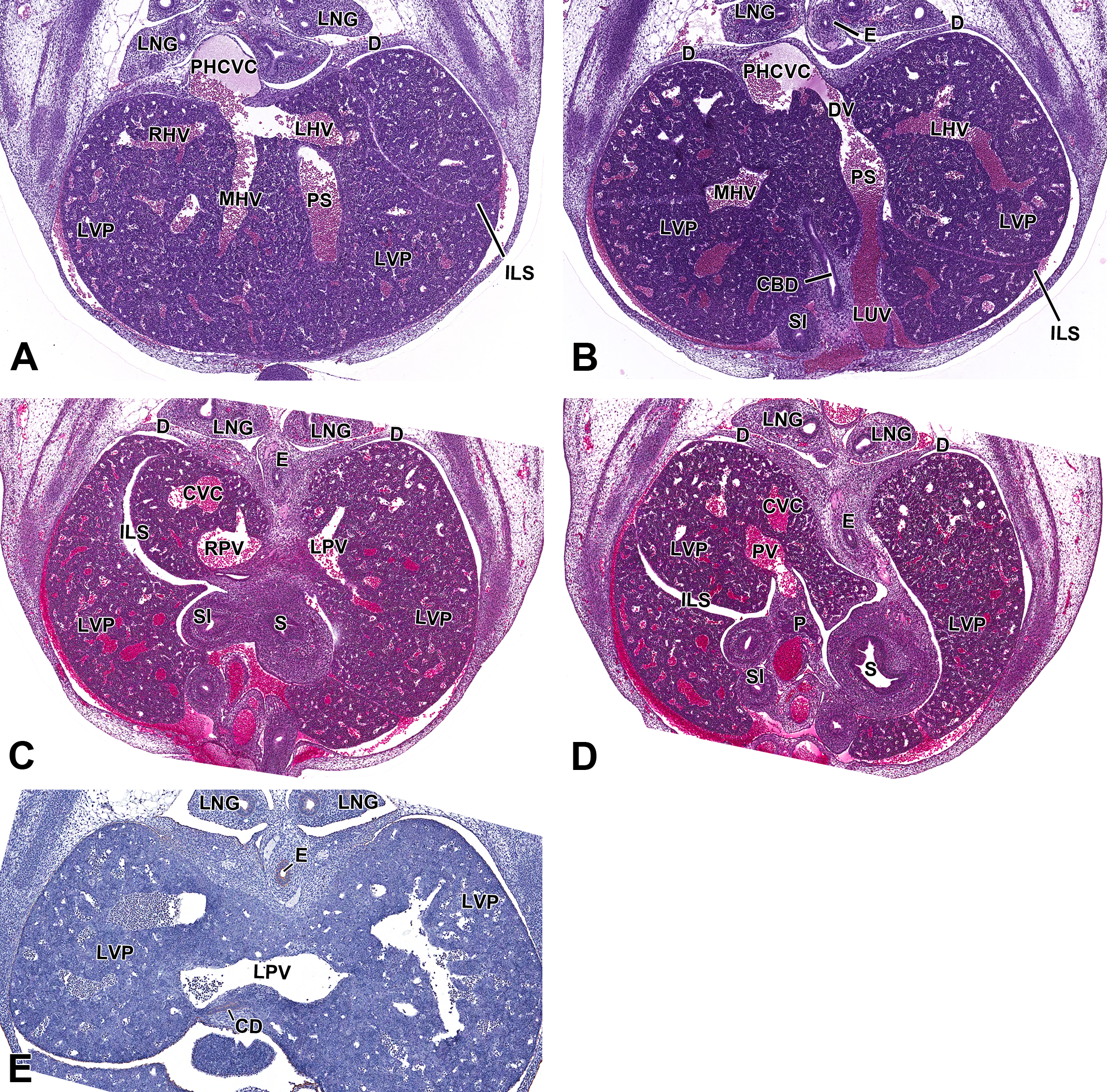
Representative images of the embryonic day (E) 12.5 mouse liver vasculature. Hematoxylin & eosin-stained (A, B, C, and D) transverse sections of the liver vasculature at low magnification from cranial to caudal view, in a single embryo, highlighting the major vessels in the developing liver. Low-magnification cytokeratin-19 IHC (E) transverse section. In the most cranial view of the 4 serial sections, the left and middle hepatic veins converge together before exiting the liver through the posthepatic caudal vena cava (A). Slightly more caudal, the left umbilical vein enters the liver and drains into the portal sinus which connects to the posthepatic caudal vena cava via the ductus venosus (B). The left portal vein runs perpendicular and caudal to the portal sinus. Cranial to the entrance of the portal vein, the left portal vein supplies the left side (C). On the right side of the liver, the portal vein is observed entering the organ and expanding into the parenchyma (D). At E12.5, there is no biliary epithelial cell differentiation occurring (E). CBD = common bile duct; CD = collecting duct; CVC = caudal vena cava; D = diaphragm; DV = ductus venosus; E = esophagus; ILS = interlobar space; LHV = left hepatic vein; LNG = lung; LPV = left portal vein; LUV = left umbilical vein; LVP = liver parenchyma; MHV = middle hepatic vein; P = pancreas; PHCVC = posthepatic caudal vena cava; PS = portal sinus; PV = portal vein; RHV = right hepatic vein; S = stomach; SI = small intestine.
As the distal segment of the right vitelline vein undergoes changes, the proximal segment of the vein does as well. This segment gives rise to 3 branches collectively known as the hepatic veins. The right, middle, and left hepatic veins drain blood from the hepatic sinusoids and transport it to the posthepatic caudal vena cava where it then navigates to the heart (Figure 4A). The right and middle hepatic veins are collectively responsible for draining the right side of the liver (Figure 4A) while the largest of the 3 veins, the left hepatic, drains the entire left lobe (Figure 4B). In mouse embryos, the ductus venosus runs in between the left and the middle hepatic veins. This is helpful histologically when identifying the different hepatic veins or for determining orientation within the liver. The convergence of the efferent veins appears histologically as a large subdiaphragmatic vestibulum (post hepatic caudal vena cava; PHCVC) located cranially in the liver (Figure 4A).
At this stage, biliary and lymphatic development within the liver is absent. Lack of biliary formation was confirmed by IHC using the biomarker, cytokeratin-19 (CK-19), a protein, which in the liver is unique to BECs. Our results indicate no biliary development adjacent to the left portal vein (Figure 4E). This location was chosen due to the high concentration of biliary precursor cells present within the portal mesenchyme surrounding this vessel at later stages. Other histological sections could also be used as long as the section is from the hilar region of the liver and contains a portal vein or a portal sinus, which is where biliary differentiation first occurs.
Abnormal Development (E12.5)
At this critical stage in embryonic development, abnormal vitelline vein transformation can cause irreversible damage to the liver. Adrenomedullin (ADM), a multifunctional peptide that is primarily responsible for decreasing blood pressure and blood vessel resistance by vasodilation in the adult (Hinson, Kapas, and Smith 2000), causes embryonic death if absent in mouse embryos (Shindo et al. 2001). In 1993, scientists first discovered this protein in pheochromocytoma cells (Kitamura et al. 1993). Further analysis revealed the detection of the peptide in many visceral organs, including the adrenal medulla, heart, lung, and liver (Washimine et al. 1994). During these initial investigations, scientists noted an elevated amount of ADM in plasma samples. Additional studies concluded that blood vascular endothelial cells actively produce and secrete ADM at a higher rate than any other cells in the body (Sugo et al. 1994). Accordingly, if an embryo does not express ADM during embryo blood vessel development, catastrophic consequences rapidly develop; this was confirmed by an ADM null mutation study in embryonic mice (Shindo et al. 2001).
In ADM−/− embryos, Shindo and colleagues (2001) noted edema and severe subdermal, hepatic and pulmonary hemorrhage. These findings were absent in the wild-type mice. Using an IHC marker for blood vascular endothelial cells (PECAM-1), abnormal development of the vitelline veins and hepatic capillaries was observed as early as E12.5 (Shindo et al. 2001). The venous and capillary endothelial cells, which are normally flat, appeared cuboidal in shape and became partially detached from the basement membrane. This caused gaps in the vasculature allowing fluids to escape through the defective membrane, leading to hemorrhage and edema, by E13.5–14 (Shindo et al. 2001). A vessel deformity of this magnitude was not compatible with life and embryonic lethality ensued by E15 (Shindo et al. 2001). These results show that not only is ADM essential for blood pressure regulation in adult vessels, but its presence within embryo endothelial cells is also required to properly form vessels, especially within the liver.
E13.5
By E13.5, intrahepatic bile duct (IHBD) differentiation begins at the center of the liver lobes. This is a pivotal stage in the development of the liver for multiple reasons. The liver uses IHBDs to facilitate the transport of bile, which is produced by the hepatocytes, from the canaliculi to the hepatic ducts and stored in the gallbladder (Bort and Zaret 2009). This is a very important and essential function in the adult liver that is continuously being utilized by the body. In the embryo; however, the IHBDs play a key role in the development of the arterial vasculature within the liver. The proper formation of the hepatic artery is entirely dependent upon the presence of IHBDs within the portal mesenchyme. The initiation of IHBD formation begins with the differentiation of a subset of bi-potential hepatoblasts, surrounding the portal veins and sinus, into hepatocytes or biliary precursor cells. This early stage of differentiation is contingent upon the presence of portal mesenchyme. Within the mesenchyme, biliary-specific cytokines are produced, which regulate the development of the surrounding hepatoblasts into BECs (Lemaigre 2003). As these cells mature, they become strongly immunoreactive to CK-8, CK-18, and CK-19, which can be visualized with IHC to document the progression of IHBD development (Desmet, Van Eyken, and Sciot 1990). This process begins at the liver hilus around the intrahepatic portal vein branches and the portal sinus. At E13.5, a small number of biliary precursor cells adjacent to the portal vessels express faint CK-19-positive staining (Figure 5A). Although levels of CK-19 detection at this stage are low, numerous BECs are present around the portal sinus, while only a few are present around the major portal vein branch. This noticeable difference may suggest that BEC differentiation along the portal sinus is initiated slightly earlier in development than those BECs neighboring the more caudal portal vein branches.
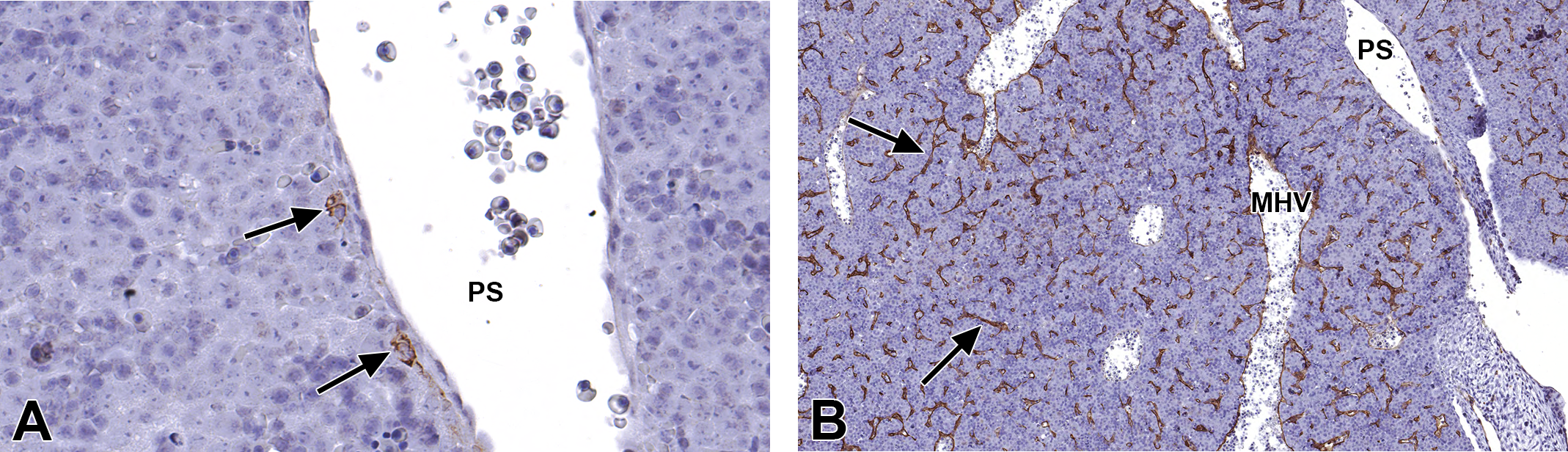
Representative images of the embryonic day (E) 13.5 mouse liver vasculature. High-magnification cytokeratin-19 (CK-19) (A) and Lyve-1 (B) immunohistochemical (IHC)-stained transverse sections. A small number of biliary precursor cells, adjacent to the portal sinus, express CK-19-positive staining (A, arrows). Throughout the entire liver, Lyve-1 IHC staining occurs in normal hepatic sinusoid epithelial cells (B, arrows). No indication of hepatic lymphatic vessel formation is apparent at this stage. MHV = middle hepatic vein; PS = portal sinus.
Due to the confirmed presence of major embryonic lymphatic sacs at this stage of development (Oliver 2004), we performed IHC staining using LYVE1 to determine whether lymphatic formation had begun in the E13.5 liver. It should be noted that LYVE1 is not exclusive to lymphatic endothelial cells (Carreira et al. 2001). Our findings demonstrate the presence of LYVE1 in normal hepatic venous and sinusoidal endothelial cells, thus making the identification of individual lymphatic vessels difficult (Figure 5B). If present, key histologic features to look for would be extremely small, darkly stained, LYVE1-positive endothelial cells, lining a thin-walled vessel. Structures resembling that description were only found in postnatal animals.
Abnormal Development (E13.5)
Initiation of proper BEC differentiation, in the E13.5 embryo, is a vital step for IHBD formation in later stages of development. Abnormal BECs can lead to devastating consequences that end in lethality. Alagille syndrome, a disease characterized by paucity of the IHBDs due to incomplete formation of the ductal plates, is an autosomal dominant, multisystemic disorder caused by mutations in the human JAG1 gene, which encodes a Notch ligand, Jagged1 (Li et al. 1997). Further investigation of Alagille syndrome by Hofmann and colleagues (2010) revealed that a specific deletion of Jag1 in SM22α-expressing cells within the portal vein mesenchyme caused paucity of IHBDs as well as jaundice and liver failure in mice. This was confirmed through the use of Jag1-smooth muscle (J1SMKO) and Jag1-endothelial cell (J1ECKO) knockout mice. Viable pups were birthed by both lines; however, J1SMKO pups experienced jaundice and growth retardation and most died within the first 2 days. Furthermore, upon histologic examination, the livers revealed bile deposits, necrosis, hemorrhage, and incompletely formed IHBDs surrounding the portal vein mesenchyme (Hofmann et al. 2010). These features were absent in the J1ECKO pups.
Similarly, Kodama and colleagues (2004) studied the relationship between Notch2 and Hes1 ligands, both of which are expressed exclusively in BECs in proximity to the Jagged1-positive cells in the portal mesenchyme. They found that Hes1 and Notch2 are upregulated by BECs and without these, mice lacked IHBD formation (Kodama et al. 2004). Their findings suggest that Notch signaling is essential in the E13.5 mouse in order to properly form the IHBDs needed to survive into adulthood.
E14.5
Beyond E14.5, the architecture of the great vessels of the liver is established and remains unchanged (Figure 6A–D). Vessels growth continues, but no substantial structural changes occur until postnatal development. The parenchyma surrounding the portal veins begins to undergo significant change as the number of BECs increases.
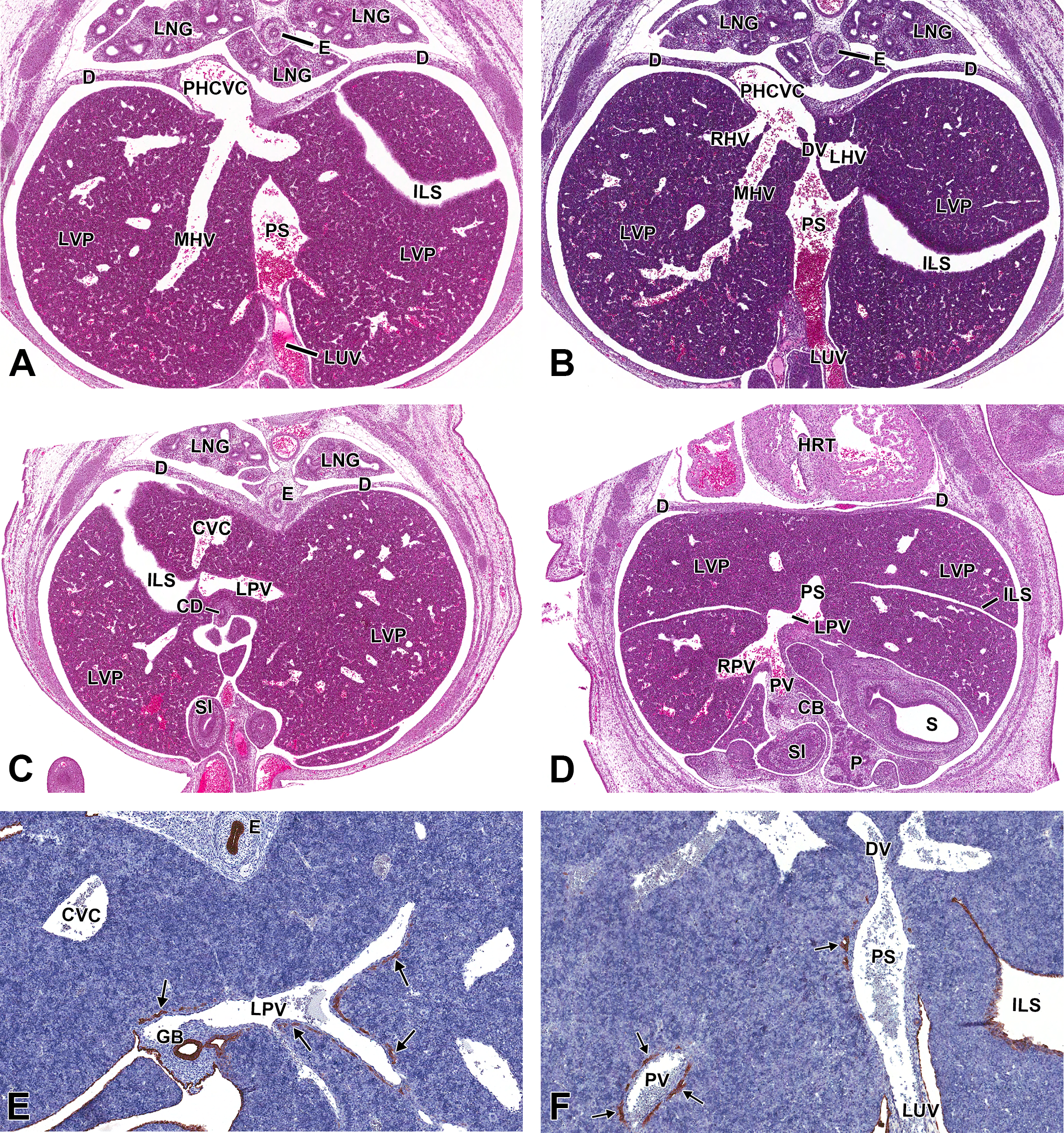
Representative images of the embryonic day (E) 14.5 mouse liver vasculature. Hematoxylin & eosin-stained (A, B, C, and D) sections of the liver vasculature at low magnification from cranial to caudal view, in a single embryo (A-C), highlighting the major vessels in the developing liver. Images A-C are transverse sections and Image D is a frontal section. Low-magnification cytokeratin-19 (E, F) immunohistochemical-stained, transverse sections. The primary blood supply to the liver is provided by the left umbilical vein which first drains into the portal sinus (A). Venous blood in the portal sinus has 2 options: it can enter the ductus venosus or enter tributary branches of the portal sinus which will eventually drain into the hepatic veins; ultimately blood from the ductus venosus and the 3 hepatic veins will enter the posthepatic caudal vena cava and travel to the heart (B). Caudal to the portal sinus, the left portal vein is observed traveling horizontally to connect to the portal sinus and supply blood to the left side of the liver (C). The origin of the portal vein can be observed, as it enters the liver on the right side (D). At this stage, biliary precursor cells surrounding the portal mesenchyme have successfully differentiated into BECs (E, arrows). These cytokeratin-19-positive cells are almost completely surrounding the portal vein in a linear arrangement (E). Additionally, BECs are segmentally present around the portal sinus, whereas the ductus venosus lacks portal mesenchyme and therefore has no BEC differentiation occurring (F). BEC = biliary epithelial cells; CBD = common bile duct; CD = collecting duct; CVC = caudal vena cava; D = diaphragm; DV = ductus venosus; E = esophagus; GB = gall bladder; HRT = heart; ILS = interlobar space; LHV = left hepatic vein; LPV = left portal vein; LNG = lung; LUV = left umbilical vein; LVP = liver parenchyma; MHV = middle hepatic vein; P = pancreas; PHCVC = posthepatic caudal vena cava; PS = portal sinus; PV = portal vein; RHV = right hepatic vein; RPV = right portal vein; S = stomach; SI = small intestine.
At E14.5, biliary precursor cells begin to differentiate into BECs that will later form the IHBDs. To confirm this early stage of differentiation, we used a CK-19 IHC stain that identifies BECs. Our results show that the biliary precursor cells within the portal mesenchyme, surrounding the portal vein, have successfully differentiated into BECs by E14.5. The brown staining, CK-19-positive BECs are almost completely surrounding the portal vein in a linear arrangement (Figure 6E). Furthermore, differentiation is also occurring in the portal mesenchyme surrounding the portal sinus. At this stage, the more cranially located portal sinus has fewer differentiating BECs in comparison to the portal vein. Similarly, the portal sinus BECs are CK-19 positive and segmentally organized in a linear arrangement around the sinus (Figure 6F).
BEC differentiation is an important step that is required to initiate hepatic arterial development; furthermore, it is also important in terms of differentiating the portal sinus from the ductus venosus. The most significant evidence supporting the existence of the portal sinus and its independence from the ductus venosus is made apparent by evaluating the presence of BECs surrounding the 2 vessels. At E14.5, CK-19-positive BECs segmentally surround the portal sinus, thus confirming that portal vein mesenchyme is encompassing the vessel. The umbilical vein-derived ductus venosus, however, lacks portal mesenchyme and therefore has no BECs surrounding the shunt (Figure 6F). The positively stained cells can be identified in proximity to the ductus venosus, at the apex of the portal sinus, but as the sinus connects to the narrowed shunt, the BECs discontinue and are nonexistent in the tissue immediately bordering the shunt.
Abnormal Development (E14.5)
Although the architecture of the great vessels remains unchanged at this stage, inactivation of certain genes can have a substantial impact on embryonic vasculature. von Hippel-Lindau (VHL) disease is a hereditary disorder caused by VHL tumor suppressor gene inactivation. In humans, this disease affects 1 in 36,000 individuals and causes highly vascularized tumors or fluid-filled cysts to be formed in various parts of the body (Frantzen et al. 2015). Vhl gene inactivation also leads to early embryonic death between E10.5 and E12.5 in mice due to defective placental vasculogenesis (Gnarra et al. 1997). In mice that died prior to E12.5, Gnarra and colleagues (1997) reported lesions only in the placenta with no other abnormalities to the fetus. This report led other scientists to develop a specific time-induced mutation that would allow the mouse embryos to live longer, thus allowing scientists to visualize the formation of the vascular tumors and cysts that are present in humans.
Hong and colleagues (2006) created VHLf/d/CreER™ mutated mice to evaluate the effects of Vhl inactivation in mice at mid-gestation. Tamoxifen, a cancer treatment drug, induces Cre recombinase (CreER™) to inactivate Vhl at a later stage in embryologic development. By inducing the gene inactivation at E10.5, the VHLf/d/CreER embryos exhibited signs of hemorrhage as early as E13.5, which progressed to severe hemorrhage at E14.5 (Hong et al. 2006). Between E14.5 and E15.5, all embryos died while the littermate controls displayed normal development (Hong et al. 2006). Histopathology revealed dilated blood vessels, vascular cell leakage, and focal necrosis in the liver (Hong et al. 2006). The disruption and malformation of the blood vascular system caused by Vhl inactivation causes the liver to become the most susceptible organ, leading to death in homozygous mice or vascular tumors in human adults (Haase et al. 2001).
E15.5
At E15.5, the vascular architecture remains the same as previous stages. The great vessels continue to increase in size corresponding to the growth of the embryo; however, no significant structural changes take place.
At this stage, BECs maintain a similar linear arrangement as seen in previous stages. BEC proliferation is observed within the portal mesenchyme surrounding the portal sinus and intrahepatic branches of the portal vein. Unlike previous stages, BECs now encompass the major branches of the intrahepatic portal vein. These BECs form a single-layered periportal sheet termed the ductal plate (Crawford, Foley, and Elmore 2010). As the embryo develops, the ductal plate will continue to evolve until the BECs constructing the ductal plate become surrounded by portal mesenchyme and begin functioning as IHBDs.
Abnormal Development (E15.5)
Often in embryologic development, an abnormality of any type can cause minimal to severe changes that may resolve postnatally or become life-threatening anomalies. The aryl hydrocarbon receptor (AHR) mutation is 1 example of a developmental problem that causes significant abnormalities in the late-gestation fetus but has the potential to be resolved postpartum (Harstad et al. 2006). The AHR is a chemical/ligand-dependent protein that acts as a transcription factor for synthetic and environmental chemicals in the adult (Denison and Nagy 2003). In the fetus, the primary role of AHR in the liver is to facilitate proper vascular formation and growth of the liver (Walisser et al. 2005). Scientists have performed numerous studies linking a decrease in liver size and patency of the ductus venosus to a mutation in the AHR locus (Harstad et al. 2006; Walisser et al. 2005; Schmidt et al. 1996).
In Ahr−/− embryos, signs of periportal hepatic necrosis are present as early as E15.5 with lesions increasing in frequency until 2 days postpartum (Harstad et al. 2006). The cause of the necrosis is most likely related to a decrease in peripheral blood flow through the hepatic sinusoids of the liver, which leads to abnormal morphology and decreased organ weight (Harstad et al. 2006). The underlying mechanism explaining the inciting cause of the altered hepatic blood flow is still unknown.
AHR activation is also responsible for signaling the closure of the ductus venosus (Harstad et al. 2006). In Ahr−/− embryos, the ductus venosus fails to close properly and remains patent throughout life (Walisser et al. 2005). During late gestation, a noteworthy dilation of the ductus venosus is evident in Ahr−/− mice as compared to wild-type mice; this can potentially be linked to an increase in portal blood pressure that can further enable patency of the ductus venosus (Harstad et al. 2006). Thus, failure of AHR activation can lead to vascular and morphologic liver abnormalities that affect the fetus during embryonic development and throughout life.
E16.5
At E16.5, the venous vasculature, in comparison to the size of the liver, appears smaller than it did previously (Figure 7A–D). The liver has greatly increased in size and hepatocytes at this age have an increased abundance of cytoplasm making the vasculature at this stage appear smaller (Crawford, Foley, and Elmore 2010).
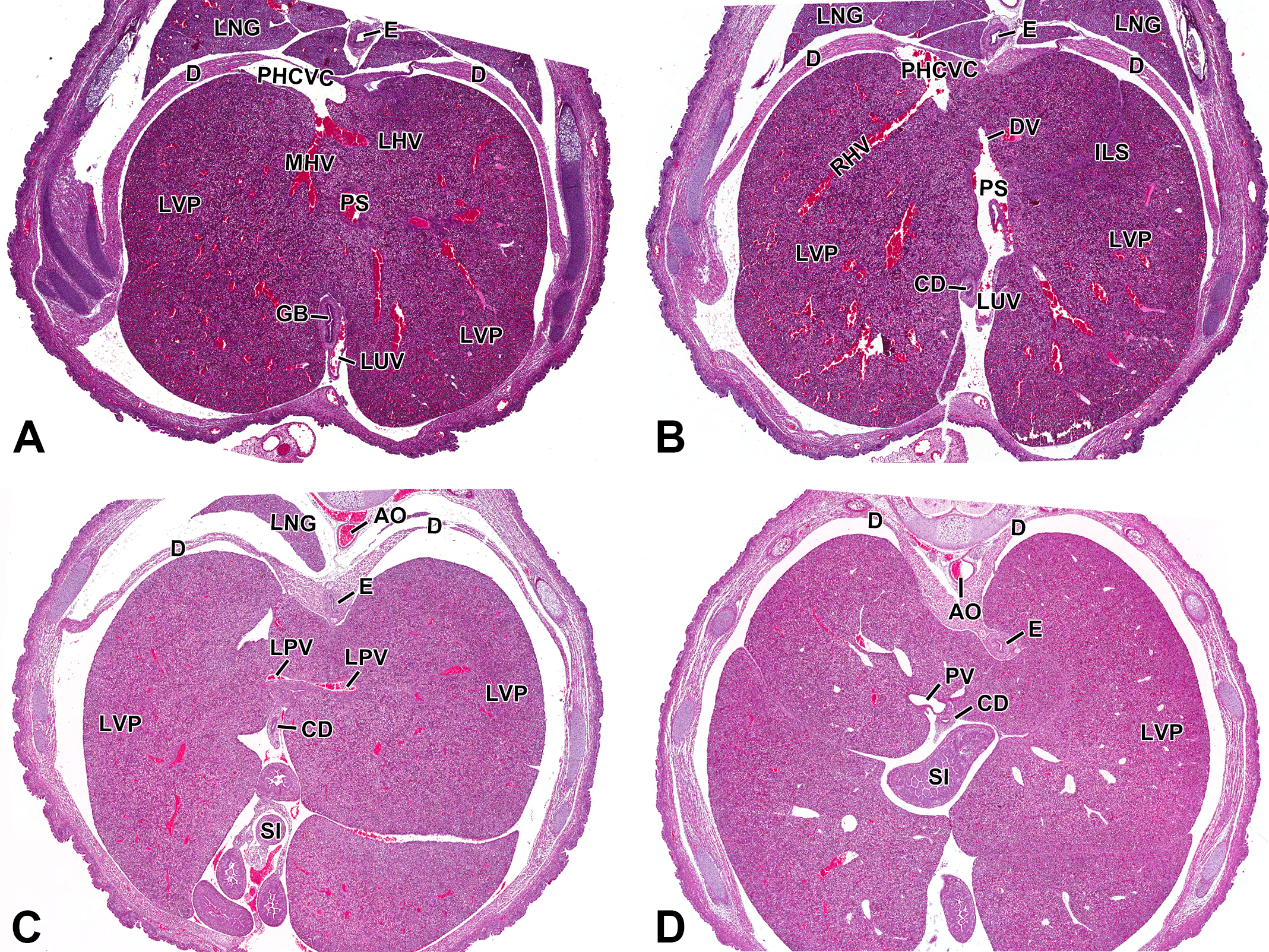
Representative images of the embryonic day (E) 16.5 mouse liver vasculature. Hematoxylin & eosin-stained (A, B, C, and D) transverse sections of the liver vasculature at low magnification, from cranial to caudal view, highlighting the major vessels in the developing liver. Major vessels are identical to previous stages, with the exception that the vessels appear smaller in comparison to the size of the liver. This is due to the substantial increase in organ size at this stage in development (A–D). AO = aorta; CD = collecting duct; D = diaphragm; DV = ductus venosus; E = esophagus; GB = gall bladder; ILS = interlobar space; LHV = left hepatic vein; LPV = left portal vein; LNG = lung; LUV = left umbilical vein; LVP = liver parenchyma; MHV = middle hepatic vein; PHCVC = posthepatic caudal vena cava; PS = portal sinus; PV = portal vein; RHV = right hepatic vein; SI = small intestine.
The single-layered ductal plate formed at E15.5 now becomes partially bilayered by duplication of the cuboidal ductal plate BECs within the portal mesenchyme. These original cells are thus referred to as the portal ductal plate layer, while the newly formed outer layer is termed the second or lobular ductal plate layer (Roskams and Desmet 2008). The cells forming the ductal plates strongly amplify the expression of CK-19 while also expressing CK-8 and CK-18. Hepatoblasts that have differentiated into hepatocytes, rather than BECs, strongly express CK-8 and CK-18 but lack CK-19 expression (Desmet, Van Eyken, and Sciot 1990). Once the bilayered ductal plates are formed, the BECs constructing the plates gradually gain the shape and function of cholangiocytes.
Abnormal Development (E16.5)
Similar to earlier stages of development, vascular deformities are possible at E16.5 even though the major vessels remain structurally unchanged. If inactivated, 1 protein that causes blood vascular deformities is the Ah receptor nuclear translocator (ARNT) protein, which is a member of the Per-Arnt-Sim protein family (F. W. Wang et al. 2006). ARNT is an essential component of the xenobiotic responsive element binding form of the AHR (Reyes, Reisz-Porszasz, and Hankinson 1992). Yim and colleagues showed that, in Arnt Δ EC mutant mice, the Arnt gene found in endothelial cells was inactivated (2006). Of the 13% of Arnt Δ EC embryos that survived beyond early gestation, all exhibited abnormal vascular development, thus indicating the important role of ARNT during embryo development. At E16.5, embryos expressed signs of hepatic vascular deformities such as poor formation of a blood vascular network throughout the liver, dilation of vascular channels, multiple abnormal central veins, and necrosis of the liver due to a lack of microvessels (Yim et al. 2006). These findings are different from the abnormalities observed in AHR-mutated mice, which develop hepatic necrosis and a patent ductus venosus due to vascular defects beginning at E15.5 (Harstad et al. 2006).
E17.5–E18.5
As the embryo approaches the final days in utero, venous vasculature branching continues expanding throughout the liver parenchyma until reaching a functional size that will support life and prepare for the upcoming postnatal changes (Figure 8A–D). To help visualize the final organizational pattern of the embryonic mouse hepatic venous vasculature, we constructed a 3-D video of these structures from H&E-stained, transverse, serial sections of an E18.5 liver (https://niehsimages.epl-inc.com). This video provides viewers with a labeled and color-coded diagram identifying the major afferent (left umbilical vein, right vitelline vein, portal sinus, and portal vein and branches) and efferent (left, right, and middle hepatic veins; ductus venosus; and caudal vena cava) vessels discussed in this atlas. Creating a 3-D video of the hepatic venous vasculature further demonstrates the relative shape, size, and orientation of the vasculature and provides a better understanding of vessel integration throughout the organ. A representative image of the 3-D video is provided (Figure 8E); however, visualization of all the vessels in a single image is limited due to the complexity of the vasculature at this stage.
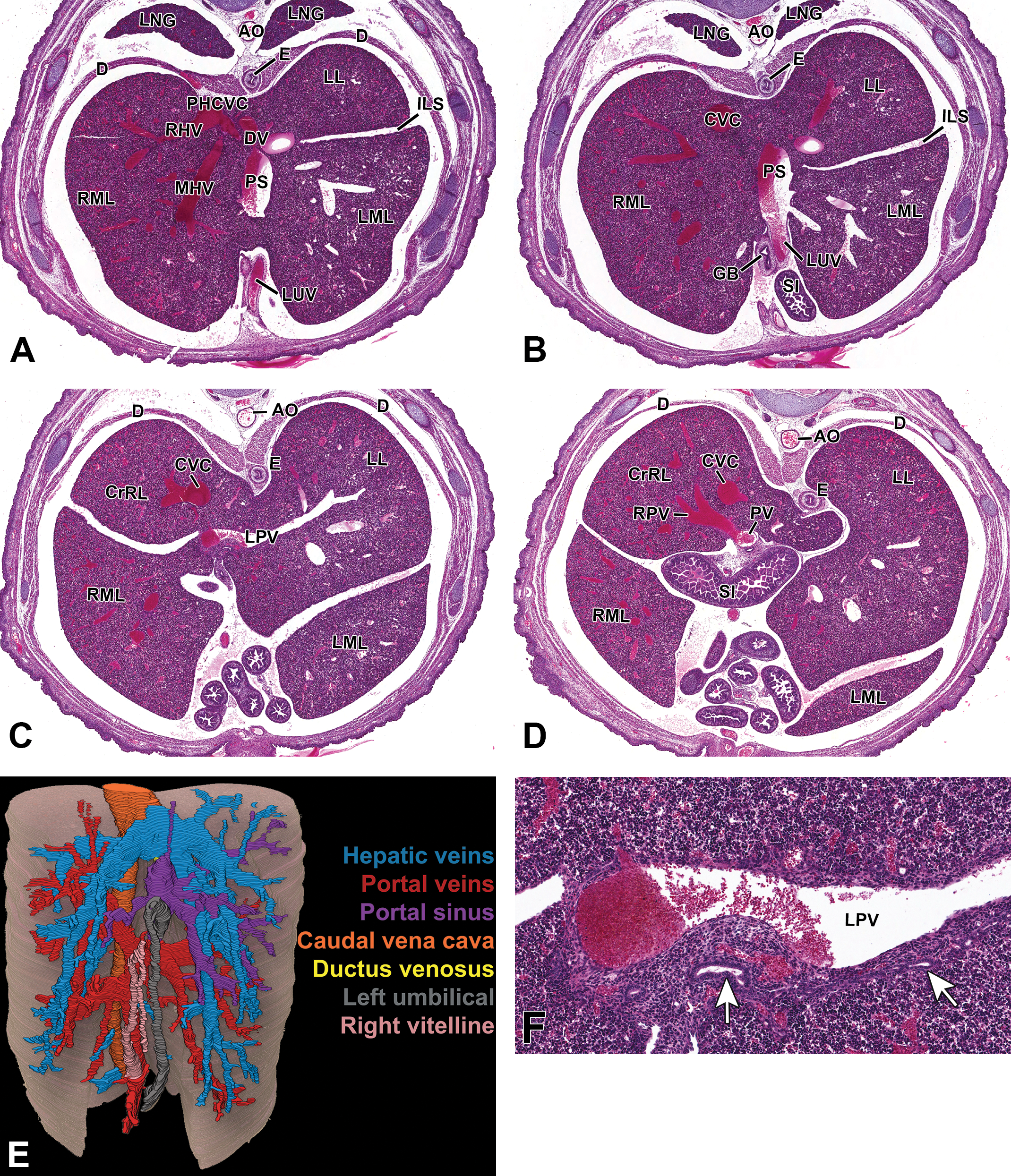
Representative images of the embryonic day (E) 18.5 mouse liver vasculature. Hematoxylin & eosin (H&E)-stained (A, B, C, and D) transverse sections of the liver vasculature at low magnification from cranial to caudal view, in a single embryo, highlighting the major vessels in the developing liver. Three-dimensional (3-D), H&E-stained serial sections of the major vessels in the developing liver (E). High-magnification, H&E-stained (F) transverse section emphasizing primitive intrahepatic bile ducts. Major vessels are identical to previous stages. The expansive venous vasculature within the liver parenchyma is appreciated through color-coded 3-D imaging (E). At this stage, primitive intrahepatic bile ducts can be identified within the portal mesenchyme using H&E staining (F, arrows). AO = aorta; CrRL = cranial right lobe; CVC = caudal vena cava; D = diaphragm; DV = ductus venosus; E = esophagus; GB = gall bladder; ILS = interlobar space; LL = left lobe; LML = left middle lobe; LNG = lung; LPV = left portal vein; LUV = left umbilical vein; MHV = middle hepatic vein; PHCVC = posthepatic caudal vena cava; PS = portal sinus; PV = portal vein; RHV = right hepatic vein; RML = right middle lobe; RPV = right portal vein; SI = small intestine.
Although the vasculature structure is uninterrupted in late gestation, the ductal plates undergo extensive remodeling, as early as E17.5. Once again, this remodeling process begins at the liver hilus with the earliest formed periportal ductal plates and, with time, reaches the most peripheral ductal plates. Segmentally, various tubular, focal dilations form between the 2 layers of ductal plates and will eventually become the lumen of the IHBDs. The early cholangiocytes that form the bilayered ductal plates and encircle the newly formed dilations create the cellular architecture of the postnatal IHBD. The portal mesenchyme, which previously made exclusive contact with the portal ductal plate layer, now begins migrating and encircling the primitive circular bile ducts, separating the lobular ductal plate layer from the neighboring hepatocytes. The remnant ductal plate cells not destined for duct formation quickly begin to regress by apoptosis (Lemaigre 2003). By E18.5, numerous individual duct-like structures are visible on high-magnification, H&E-stained tissue surrounding the portal vessels within the liver (Figure 8F).
Abnormal Development (E17.5–18.5)
Multiple congenital diseases in humans are caused by a genetic abnormality related to the malformation of the ductal plates during embryo development (Desmet 1998). In mice, the inactivation of the hepatocyte nuclear factor (Hnf6) gene, expressed only in the BEC (not the hepatic artery), results in a ductal plate malformation (DPM; Clotman et al. 2002). A DPM causes abnormal formation of the bile ducts and failure of ductal plate regression at birth (Clotman et al. 2003). Since hepatic artery formation is dependent upon proper formation of the biliary system, a malformation in the ductal plate leads to the abnormal development of the artery. Clotman and colleagues (2003) also showed that, during postnatal investigation of Hnf−/− mice, the portal veins were surrounded by a discontinuous bilayered structure formed by the BEC with moderate portal fibrosis. Additionally, the hepatic arteries closest to the DPM and hilum of the liver were hyperplastic and deranged, while peripheral portal tracts lacked hepatic arteries completely.
Postnatal Development
The adult liver is recognized for having a unique lobular vascular pattern. Histologically, each liver lobule consists of 6 afferent portal vein braches, shaped in a hexagon configuration, all of which connect, via numerous hepatic sinusoids, to a single efferent central vein. The central veins, also known as terminal branches of the hepatic veins, are linear vessels that receive blood from the afferent vessels and lack portal mesenchyme. Adjacent to every portal vein branch is a hepatic artery and IHBD that together, along with nerves and lymphatic vessels, form the commonly known portal tracts (or portal triads). Formation of portal tracts is initiated soon after birth, upon closure of the ductus venosus.
At approximately postnatal day (PND) 2, the most significant postnatal vascular change occurs: the closure of the ductus venosus. After birth, the umbilical vessels are closed, which consequently terminates blood flow to the ductus venosus. Without the highly pressurized and oxygenated blood flow, the intrahepatic shunt begins to regress. After 48 hr of life, this narrow vessel constricts into a fibrous cord incapable of transporting blood, termed the ligamentum venosum (Washington Smoak 2007). Histologically, the ligamentum venosum can be identified as a thin, linear band of fibrous tissue that is positioned between the posthepatic caudal vena cava and the portal sinus. Transverse sections of PND3 mice are best for observing the newly transformed ligamentum venosum (Figure 9A). As the umbilical vein and ductus venosus become inactive components of the vasculature system, the portal sinus takes on a new role. Rather than being a confluence for highly oxygenated umbilical blood, the sinus with its tributaries now forms the distal aspects of the left portal vein and continues to supply the cranial portions of the left side of the liver.
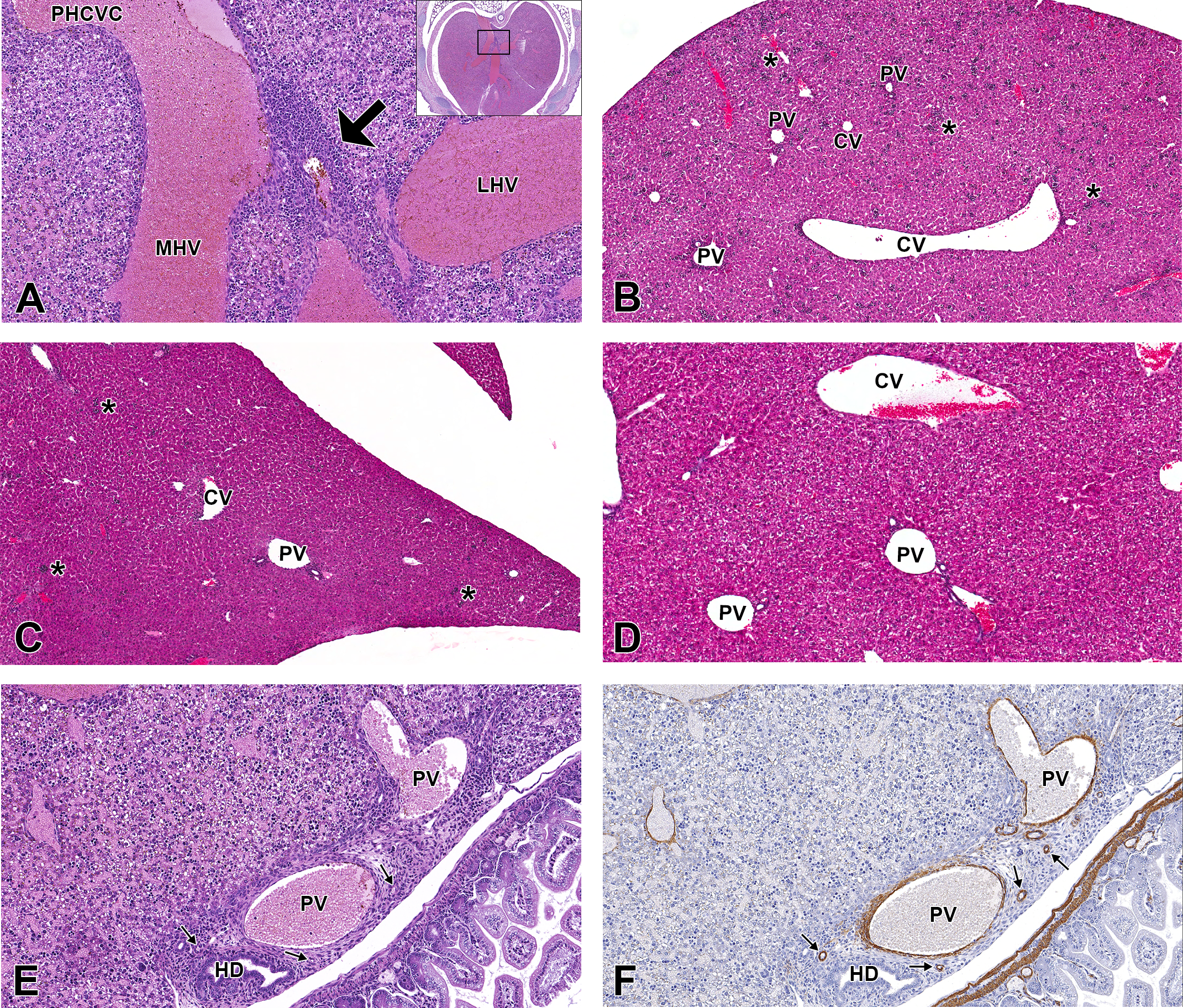
Representative images of the postnatal mouse liver vasculature. High-magnification, hematoxylin & eosin (H&E)-stained, transverse sections at postnatal day (PND) 3 (A and E), and low-magnification, H&E-stained, frontal sections at PND5 (B), PND10 (C), and PND21 (D). High-magnification, smooth muscle actin immunohistochemical-stained, transverse sections at PND3 (F), PND5 (G), PND10 (H), and PND21 (I). Lyve-1 IHC-stained, frontal sections at PND5 (J), PND10 (K), and PND21 (L) at high magnification. Soon after birth, the ductus venosus constricts into a fibrous cord that is identified as a thin, linear band of fibrous tissue (arrow) positioned between the PHCVC and the portal sinus (A). As the postnatal fetus matures, the liver vasculature begins to arrange into a noticeable lobular formation, consisting of 6 portal triads surrounding a central vein (B–D). The hematopoetic population (asterisks) within the liver also decreases as the mouse matures. At the liver hilus, multiple, small circular to elongate, arteries are observed in the connective tissue adjacent to the portal vein and common bile duct (E and F, arrows). At PND5, hepatic arteries are visible within the parenchyma (G, arrows), and as postnatal development continues, the arteries proliferate from the hilus to the capsular region of the liver (H and I). Lymphatics within the liver also change dramatically during postnatal development. At PND5, the endothelial cells of the hepatic sinusoids continue to strongly express Lyve-1, and there is no indication of lymphatic vessel development (J). As postnatal maturation continues, this expression dramatically decreases at PND10 (K) and PND21 (L) until it is faintly observed throughout the sinusoids. At PND21, the first distinct lymphatic vessels are observed (L, arrow heads). CV = central vein; HA = hepatic artery; HV = hepatic vein; IHBD = intrahepatic bile duct; LHV = left hepatic vein; MHV = middle hepatic vein; PHCVC = posthepatic caudal vena cava; PS = portal sinus; PV = portal vein.
After closure of the ductus venosus, the remaining vasculature continues to undergo substantial remodeling until PND21. Terminal branches of the portal vein that have been present and for the most part unchanged since E12.5 continue to transport deoxygenated blood from the extrahepatic portal vein. Due to the lack of oxygenated blood flow into the liver, the hepatic arteries, which deliver 25% of the organ’s blood supply during adulthood, must be formed (Bort and Zaret 2009). Hepatic artery formation, however, is not initiated until IHBDs are completely formed and incorporated into the portal mesenchyme. Starting at the liver hilus, IHBDs continue to proliferate alongside portal vein branches traveling peripherally throughout the parenchyma. Once an IHBD is established within the portal mesenchyme, hepatic artery formation can be initiated, which also begins at the hilus and migrates toward the periphery to form portal triads (Figure 9B–D).
Histologically, at E18.5, no hepatic artery formation is observed; it is not until PND3 that multiple, small, circular to elongate, arterial vessels are observed in the connective tissue adjacent to the portal vein and common bile duct (Figure 9E). To confirm arterial origin of these vessels, we used IHC biomarker, smooth muscle actin (SMA), which targets smooth muscle cells in the arterial wall. Unlike arteries, venous vessels lack a thick muscular wall; therefore, SMA can be used to differentiate arteries from veins at this stage of development. When interpreting SMA IHC, positively stained smooth muscle cells in hepatic arterial walls stain intensely and are much thicker than intrahepatic veins (Figure 9F). Using the same PND3 embryo as used for the H&E stain (Figure 9E), we confirm that the vessels identified in the connective tissue are arterial. Although these arterial vessels are present in the connective tissue adjacent to the portal vein, no arterial formation is observed within the parenchyma of the liver at PND3. At PND5, hepatic arteries can be found in the liver parenchyma neighboring portal vein branches near the hilus (Figure 9G). The arteries continue to proliferate toward the periphery, as the postnatal mouse matures (Figure 9H and I).
During embryo development, the role of lymphatics within the liver is presumed to be insignificant. At birth, the endothelial cells of the hepatic sinusoids continue to strongly express LYVE1. As postnatal maturation continues, this expression dramatically decreases until PND21 where faint staining is present throughout the sinusoids (Figure 9J–L). At PND5 and PND10, no distinct lymphatic vessels are observed by LYVE1 staining (Figure 9J and K). However, at PND21, the definitive portal triad is observed. Contained within the portal mesenchyme is a branch of the portal vein, hepatic artery, and a small thin-walled spherical vessel which exhibits positive LYVE1 staining (Figure 9L). Due to the substantial decrease in sinusoidal endothelial cell expression for LYVE1 and the location of the vessel within the portal triad, we can confidently report these as lymphatic vessels.
Discussion
This histology atlas was created to provide scientists and pathologists with a resource documenting the development of the normal mouse hepatobiliary hemolymphatic vasculature covering E11.5–18.5 and early postnatal development. Selected abnormalities that occur during each stage of development have also been identified to assist scientists working with abnormal phenotypes. When evaluating embryonic hepatobiliary hemolymphatic development, it is important to be aware of gestational, age-specific changes that are occurring throughout the stages of maturation. We have identified and confirmed these using H&E and IHC-stained images of the embryonic and postnatal liver. Additionally, the creation of a 3-D video of the venous vasculature at E18.5 using H&E-stained, transverse, serial sections, aids in the conceptualization of the complex vascular architecture.
There are a significant number of anatomic and cellular changes that begin during early embryonic life and continue weeks into early postnatal development. One of the most noteworthy findings of this atlas is the identification of the portal sinus within the mouse. This structure is defined in the medical literature and with advanced imaging techniques more is now known regarding the precise shape and function of this vessel in humans (Mavrides et al. 2001; Paris, Cabaret, and Grall 2004). For decades, the portal sinus has been erroneously misinterpreted as the ductus venosus in the mouse; however, these 2 vessels are functionally and cellularly independent of one another. The ductus venosus is a narrow, branchless, tubular shunt connecting the portal sinus and posthepatic caudal vena cava; whereas, the sinus is a large portal venous confluence with tributaries that transport oxygenated blood from the left umbilical vein to the ductus venosus. During early postnatal development when the ductus venosus closes and becomes the ligamentum venosum and the left umbilical vein is transformed into the ligamentum teres, the portal sinus becomes integrated into the distal left portal vein, primarily supplying the left side of the liver.
Furthermore, this atlas describes the developmental timeline of the hepatic artery and the IHBDs. We report that hepatic artery development is contingent upon the proper formation of the biliary ductal plate. Adjacent to the portal mesenchyme, bilayered ductal plates transform into IHBDs that become incorporated into the mesenchyme. As the IHBDs proliferate, they migrate from the hilus to the periphery. Once mature IHBDs are established near the hilus, hepatic artery formation is initiated. At PND3, arteries are observed in the connective tissue adjacent to the portal vein at the hilus; however, no arteries are observed entering the liver or observed within the parenchyma. Other arteries in the abdomen, such as the mesenteric and renal arteries, are well defined at this stage of development, but no similar-sized arteries can be found in the liver. Histologically at PND5, hepatic arteries can be visualized within the liver parenchyma. This suggests that the blood supply to the liver during early postnatal development is solely dependent on portal blood flow. As the postnatal mouse matures, the hepatic arteries proliferate toward the periphery of the liver to form portal triads. The end result is a mature liver, organized in a lobular formation consisting of a central vein surrounded by 6 portal triads, each of which contain a branch of the portal vein, hepatic artery, IHBD, and other structures such as nerves and lymphatics.
Unlike the biliary system, development of the intrahepatic lymphatics has a presumably less significant role in the developing hepatobiliary system and occurs at a much later stage in the mouse. We report that during embryonic development, lymphatic growth within the liver is not a substantial process. True lymphatic vessels have a very thin vessel wall and express LYVE1-positive staining. Our findings indicate these vessels, present in portal triads, are not histologically visible within the liver until weeks after birth. As postnatal mice mature, LYVE1 staining of the sinusoidal endothelial cells decreases and staining of the lymphatic vessels becomes more apparent. Lymphatic vessels can be identified histologically at PND21 using H&E and LYVE1 IHC-stained tissue sections. Other markers are available for the detection of lymphatic vessels, such as Prox1; however, this protein is important in liver development and is ubiquitously expressed making detection of microscopically small lymphatic vessels challenging.
When reviewing embryonic development, it is important to remember that strain differences as well as individual animal variations occur. The events and changes described in this atlas have been cited in the literature but may differ slightly depending on the species, strain, or individual animal so comparison to concurrent controls, preferably littermates, is critical. As genetically modified mice become increasingly utilized in the scientific field, it is crucial that researchers have a resource for normal anatomic and cellular morphology when assessing phenotypic defects in the liver vasculature. We hope the information provided in this atlas will serve as a useful guide to pathologists and scientists evaluating the embryonic and early postnatal mouse hepatobiliary hemolymphatic vascular system.
In the future, we hope to further investigate the venous vascular architecture of the E11.5 mouse. We anticipate that this video will help to further visualize the complexity of the vasculature at this critical stage of intrahepatic venous vascular development.
Footnotes
Acknowledgments
The authors wish to acknowledge the following Special Technique Group members of the NIEHS Imaging Core: Norris Flagler (NIEHS), Beth Mahler (EPL), and Elizabeth Ney (NIEHS) for their image scanning and photographic assistance and, David Sabio (ELP) for schematic illustrations, David Olsen (NIEHS) for SMA IHC staining.
Author Contribution
Authors contributed to conception or design (OS, JF, DL, JC, SE); data acquisition, analysis, or interpretation (OS, JF, DL, JC, SE); and drafting the manuscript (OS). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the Intramural Research Program of the National Institutes of Health (NIH), National Institute of Environmental Health Sciences (NIEHS).
