Abstract
Sinusoidal obstruction syndrome (SOS) is a unique form of liver injury that occurs after exposure to chemotherapeutic drugs and toxins. The diagnosis of SOS in humans remains a challenge as the clinical criteria have low specificity and there are no reliable noninvasive biomarkers. The mechanism of injury is believed to be damage to liver endothelial cells, primarily sinusoidal endothelial cells (SECs), which leads to sinusoidal dilation, central venous fibrosis, and/or nodular regeneration. Nonclinical data suggest that this uncommon liver toxicity can be recapitulated in cynomolgus monkeys, and it is critical that pathologists are familiar with its characteristic clinicopathologic features. Elevations in liver enzymes, in particular aspartate aminotransferase, associated with isolated thrombocytopenia, should raise the suspicion of SEC injury for specific drug classes. Characterization of liver microscopic findings in monkeys benefits from the use of appropriate stains, such as reticulin stain, and VEGFR2 and CD34 immunohistochemical (IHC) stains. CD41 IHC demonstrates platelet accumulation in injured sinusoids, the likely cause of thrombocytopenia commonly reported in SOS. In conclusion, this report provides a comprehensive characterization of the pathology findings of drug-induced SOS in monkeys with the objectives of ensuring appropriate nonclinical recognition of the liability and informing clinical development strategy and monitoring.
Keywords
Introduction
Sinusoidal obstruction syndrome (SOS), previously known as veno-occlusive disease, is a distinctive and potentially life-threatening form of liver injury that is thought to begin with an injury to sinusoidal and/or central venous endothelial cells. 1,2 Sinusoidal obstruction syndrome occurs almost exclusively after drug or toxin exposure and is a common complication in cancer patients receiving conditioning myeloablative chemoirradiation prior to hematopoietic stem cell transplantation (HSCT) 3,4 and in people following consumption of Jamaican bush tea containing Senecio or Crotalaria plant extracts. 5 -7 Drug-induced SOS is also described in cancer patients in nontransplant settings following the use of chemotherapeutic agents, such as oxaliplatin or calicheamicin-based antibody-drug conjugates (ADCs). 8 -10
Gemtuzumab ozogamicin (GO, Mylotarg) and Inotuzumab ozogamicin (InO, Besponsa) are ADCs comprised of anti-CD33 and anti-CD22 antibody, respectively, conjugated to a calicheamicin derivative. Both GO and InO have a black box warning for hepatotoxicity/SOS 10 -12 with an overall estimated incidence of SOS in 9% of people following GO treatment 13 and in 1.5% of people following InO treatment. 12 The rates of SOS for both drugs are higher in the HSCT setting than in the nontransplant setting. 14,15 In patients receiving neoadjuvant chemotherapy containing oxaliplatin for colorectal liver metastases, histopathology evaluation of therapeutic surgical liver resections showed high-grade lesions of SOS in 59% of patients in nontumoral tissue. 16
Clinically, patients with SOS present with painful hepatomegaly, ascites, and weight gain, often associated with jaundice. 17 There is a critical lack of sensitive and/or specific biomarkers, and the diagnosis is usually based on clinical signs and routine laboratory tests. 3 Notably, although hepatic biopsy is considered the gold standard for the diagnosis of SOS, it is often not recommended due to potential clinical complications of hemorrhage due to associated thrombocytopenia. 12,18,19 Histopathologic features of SOS as described in patients include sinusoidal dilation, atrophy or dissociation of hepatic plates, central venous intimal fibrosis, and nodular regeneration. 9,20 These changes are described as distributed haphazardly across the liver, predominantly within centrilobular or midzonal regions. 9 Additional features described in humans include perisinusoidal fibrosis and centrilobular hemorrhage.
Evaluation of the safety of new medicines usually requires toxicity studies in relevant animal species prior to initiation of clinical trials, and standard endpoints in these studies include clinical observations, clinical pathology, and histopathology. A recent prospective exploratory study in monkeys dosed with a nonbinding antibody-calicheamicin conjugate demonstrated the relevance of the monkey for the identification of SOS induced by GO and InO. 21 However, due to specific microscopic features requiring nonroutine stains, the histopathologic diagnosis of drug-induced SOS in nonclinical studies is challenging and it is our experience that the condition may be overlooked or inaccurately diagnosed by pathologists. It is therefore critical that both pathologists and clinicians are aware and well informed of this type of liver injury to ensure appropriate identification of the liability in nonclinical species, comprehension of the translational relevance, and accurate and prompt recognition of the condition in patients.
In this report, we present an example of SOS in monkeys dosed with an ADC carrying a potent DNA-crosslinking cytotoxic agent. The objective of this article is to provide a comprehensive microscopic characterization of SOS in monkeys, highlight diagnostic challenges, present major associated clinical pathology changes, and discuss clinical translation of this underrecognized toxicity.
Materials and Methods
Test Article
ADC-008 is an ADC that consists of a targeting human immunoglobulin G1 monoclonal antibody linked to a potent DNA-crosslinking cytotoxic agent via a dipeptide cleavable linker. The target is not expressed in liver sinusoidal endothelial cells (SECs) in monkeys or humans. The test article ADC-008 was formulated in 20 mM histidine hydrochloride buffer. The vehicle control article consisted of the formulation buffer diluted in PBS.
Animals
Cynomolgus monkeys (Macaca fascicularis), 2.2 to 3.3 years of age and weighing between 2.2 and 2.7 kg at dosing initiation, were obtained from Charles River Laboratories. All procedures performed on animals in this study were conducted in accordance with established guidelines and regulations and were reviewed and approved by AbbVie’s Institutional Animal Care and Use Committee. The Contract Research Organization care facilities (Charles River Laboratories) that supported this work are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Experimental Design
Cynomolgus monkeys (N = 5/sex/group) received 2 intravenous administrations on days 1 and 22 (Q3W ×2) of either the vehicle control or ADC-008 at 0.01, 0.03, or 0.1 mg/kg/dose. Animals were necropsied on day 29 (N = 3/sex/group) or allowed to recover for 8 weeks and necropsied on day 78 (N = 2/sex/group). The test article ADC-008 and vehicle control were administered at a dose volume of 1 mL/kg over a duration of approximately 30 minutes.
Clinical Pathology
Blood was collected for hematology in EDTA tubes and for clinical chemistry in serum separator tubes. Hematology and clinical chemistry samples were collected at 2 baseline time points (days 9 and 2) and during the dosing phase on days 8, 15, and 22, and prior to necropsy on day 29. Additional hematology and clinical chemistry collections were performed during the recovery period on days 36, 43, 50, 64, and prior to necropsy on day 78. Individual animal data from the dosing and recovery periods were compared with respective mean baseline data. In addition, group mean values (males and females combined) for individual parameters for ADC-008-treated groups were compared with concurrent mean vehicle control values when appropriate for the purpose of graphical representation of the changes.
Histopathology and Histochemistry
Animals were fasted overnight before scheduled necropsies. Animals were euthanized by an intravenous injection of a commercially available veterinary euthanasia solution followed by exsanguination. A comprehensive set of tissues was collected and evaluated microscopically. Only microscopic findings related to the liver and bone marrow will be detailed in this article. Samples of liver and bone marrow (sternum) were fixed in 10% neutral buffered formalin, processed to paraffin blocks, and the sections were stained with hematoxylin and eosin (H&E) for examination. In addition, 5 µm-thick liver sections from a subset of necropsied monkeys (including terminal and recovery animals from the control group and the group dosed at 0.1 mg/kg/dose) were stained per manufacturer’s instructions using Reticulin staining kit (catalog number 9168) from Newcomer Supply.
Immunohistochemistry
Antibodies against VEGFR2 for endothelial cells, CD34 for liver sinusoidal capillarization, 22 and CD41 for platelets were used for immunohistochemical (IHC) staining of samples from a subset of necropsied monkeys (including terminal and recovery animals from the control group and the group dosed at 0.1 mg/kg/dose). Five µm-thick, formalin-fixed, paraffin-embedded liver sections were immunohistochemically stained on BOND RX Automated Research Stainer (Leica Biosystems). The pertinent details of the IHC assays are provided in Supplemental Table 1. Slides were counterstained with hematoxylin, dehydrated in graded alcohol, cleared with xylene, and coverslipped.
Results
ADC-008 Was Clinically Well Tolerated in Monkeys
ADC-008 was well tolerated up to 0.1 mg/kg/dose, and all animals survived to scheduled necropsy at the end of the dosing and recovery phases. Outside of skin findings characterized by localized or generalized black discoloration, there were no notable clinical signs in any animal throughout the study at any dose level. At the high dose of 0.1 mg/kg/dose, there was mild body weight loss (up to 200 grams) in individual animals during the recovery period that was accompanied by transiently reduced appetite. There were no ADC-008-related effects on body weight and food consumption at 0.01 and 0.03 mg/kg/dose.
Clinical Pathology Findings Consisted of Increases in Aspartate Aminotransferase and Transient Thrombocytopenia
ADC-008-related effects on liver aminotransferases at 0.1 mg/kg/dose consisted of minimal increases in mean aspartate aminotransferase (AST) to 1.8-fold above mean baseline values on day 29 (Figure 1A). Noteworthy increases in alanine aminotransferase (ALT) were not observed at any dose level in the study (Figure 1B). The trend in higher mean ALT values during the recovery period at 0.1 mg/kg/dose as compared to controls was attributed to biological variability rather than an effect of ADC-008. One recovery phase animal at 0.1 mg/kg/dose also had a transient mild increase in bilirubin (2.5-fold) on days 43 and 50 as compared to mean baseline value. There were no ADC-008-related changes in alkaline phosphatase, gamma-glutamyltransferase, or coagulation parameters (fibrinogen, prothrombin time, activated partial thromboplastin time) at any dose level.
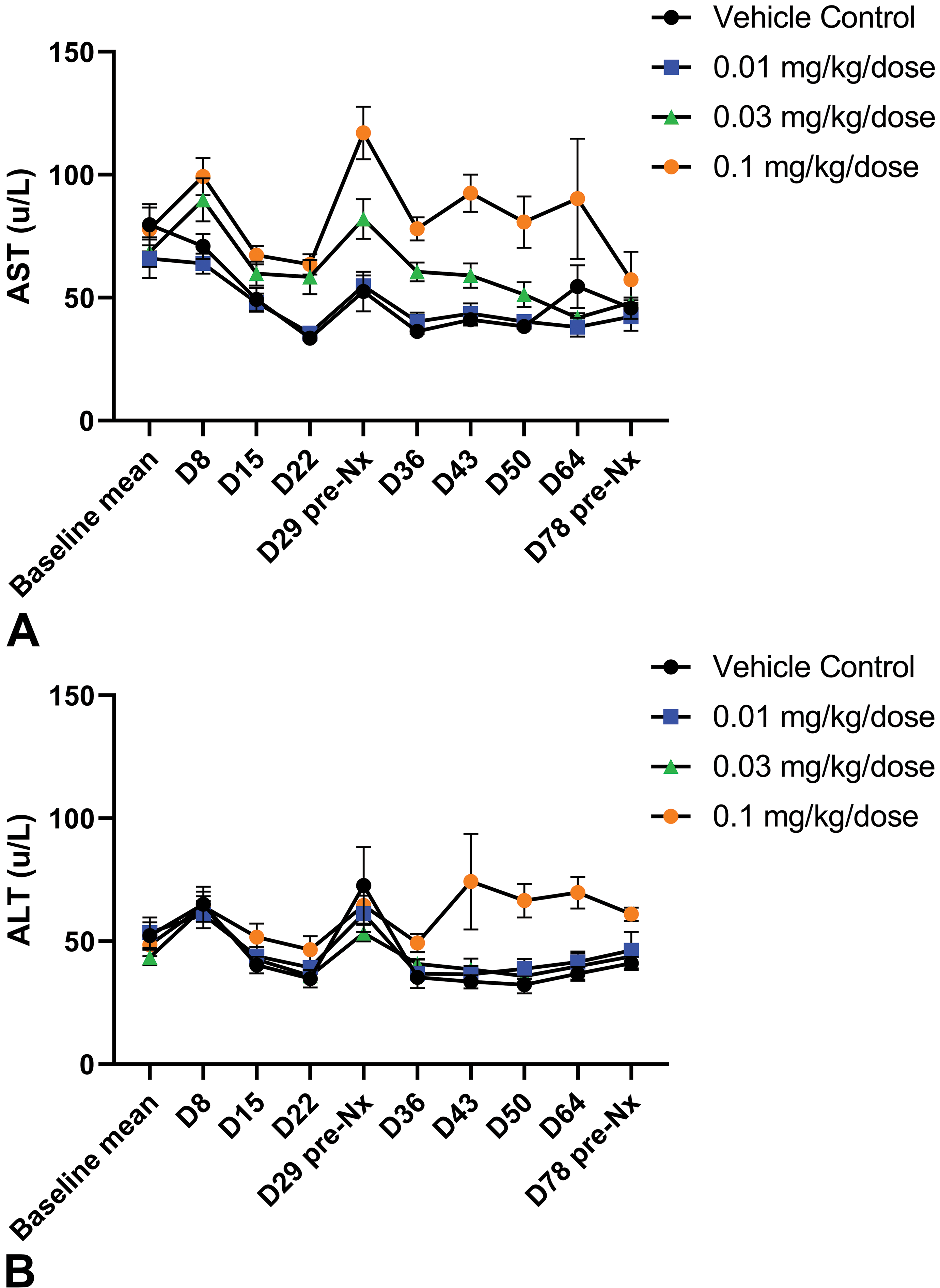
A, Dose-dependent increase in aspartate aminotransferase (AST) following ADC-008 administration. AST values at all study collection time points at all doses presented as mean ± SEM (N = 10/group/time point dosing phase; N = 6/group/time point recovery phase). The initial value at day 0 represents the mean value of 2 baseline measurements for each animal. Note peak AST value at 0.1 mg/kg/dose occurs at end of dosing phase, day 29, and persists through the recovery period. A lesser increase at 0.03 mg/kg/dose follows the same temporal pattern. B, No effect on alanine aminotransferase (ALT) following ADC administration. ALT values at all study collection time points at all doses presented as mean ± SEM (N = 10/group/time point dosing phase; N = 6/group/time point recovery phase). The initial value at day 0 represents the mean value of 2 baseline measurements for each animal. Note that ALT values for all dose groups remain close to baseline values.
In addition, ADC-008-related hematology changes included mild to moderate transient decreases in platelet counts at ≥0.03 mg/kg/dose, beginning on day 15 at 0.1 mg/kg/dose and on day 22 at 0.03 mg/kg/dose, with partial reversibility during the recovery period (Figure 2). The decreases in platelet counts were accompanied by mildly increased mean platelet volume and platelet distribution width at 0.1 mg/kg/dose beginning day 22 (data not shown). On an individual animal basis, platelet counts decreased to as low as 52 × 103/µL on day 22 while on a mean group basis, the nadir occurred during the recovery period on day 36. Histologic examination of sternal bone marrow did not demonstrate any significant ADC-008-related effect on hematopoietic cellularity, and the megakaryocyte density and morphology appeared within normal ranges at all dose levels (data not shown).
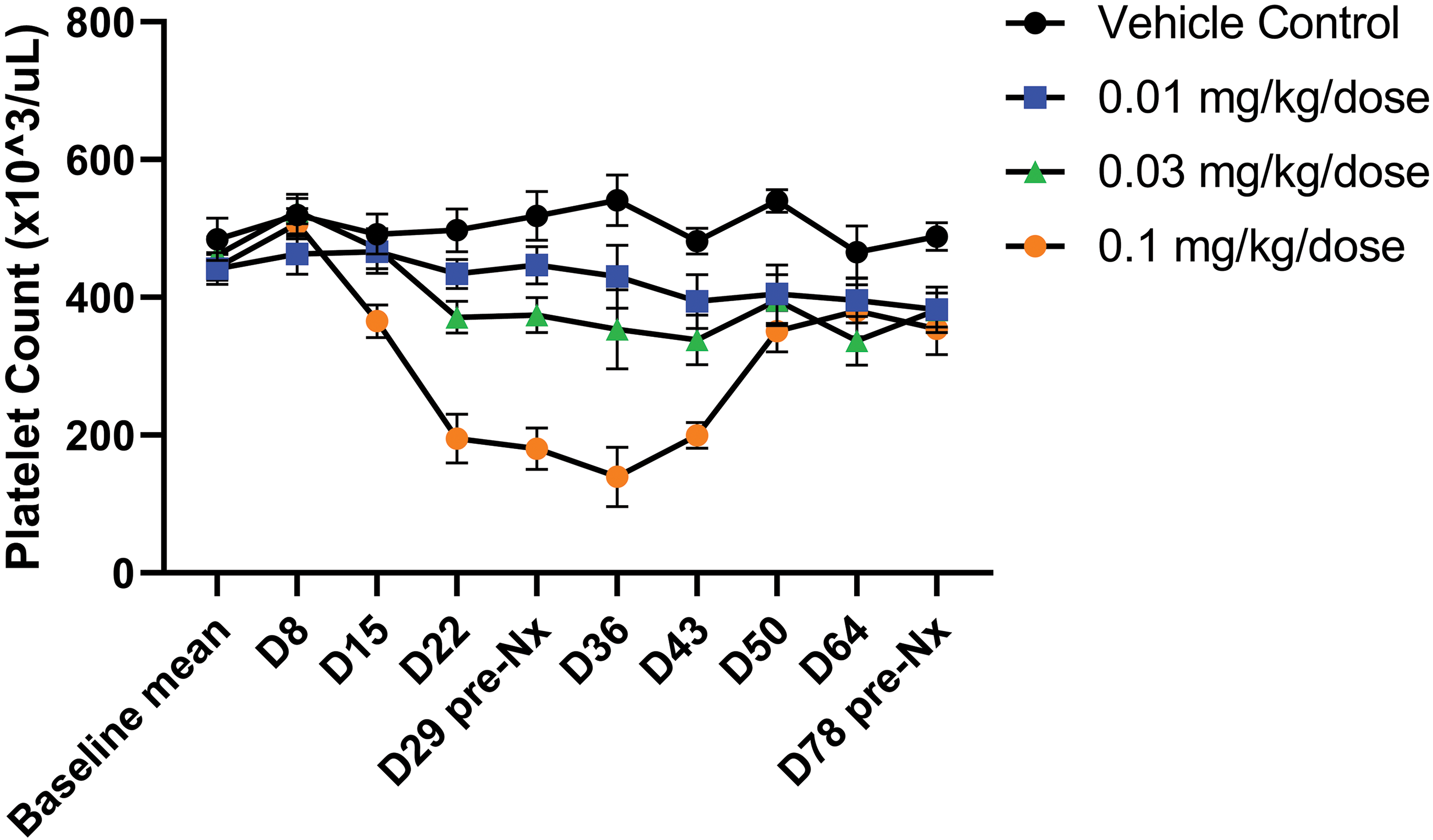
Transient dose-dependent thrombocytopenia following ADC-008 administration. Platelet counts at all study collection time points at all doses presented as mean ± SEM (N = 10/group/time point dosing phase; N = 6/group/time point recovery phase). The initial value at day 0 represents the mean value of 2 baseline measurements for each animal. Note delayed effects after the first dose, with platelet decreases occurring toward the end of the first cycle and only minimally more pronounced platelet decreases after the second test article administration.
Liver Microscopic Findings Were Characterized by Sinusoidal Dilation, Hepatic Plate Atrophy, and/or Nodular Regeneration on H&E-Stained Sections
There were no ADC-008-related changes in liver weights at any dose level at the end of the dosing and recovery phases. There were no macroscopic or histopathologic findings in the liver at the examination of H&E-stained slides at the end of the dosing phase. Grossly, at recovery necropsy, there were multiple black pinpoint foci in the liver of 2 animals at 0.1 mg/kg/dose, which correlated with adverse histopathology findings observed in 3 of 4 recovery phase animals. Microscopic findings in the liver of these animals included sinusoidal dilation (Figure 3A and B), hepatic plate atrophy (Figure 3C), and nodular regeneration (Figure 3D). Sinusoidal dilation was characterized by irregular expansion of the sinusoidal vascular space, often occurring in midzonal areas and occasionally bridging across multiple lobules. Hepatocytes in these areas were atrophic and hepatic plates were discontinuous. This finding was limited to scattered multifocal areas or widespread across the liver. Notably, there was no overt associated hepatocellular degeneration/necrosis or inflammatory response in any of the affected livers. Hepatocellular atrophy was also observed in the absence of sinusoidal dilation (Figure 3C) and was characterized by irregular bands and clusters of small hepatocytes with nuclear crowding. Occasionally, several plates of atrophic hepatocytes in midzonal areas surrounded an area of nodular regeneration forming a region of compressed trabeculae. Nodular regeneration was characterized by small nodular foci of enlarged hepatocytes arranged in double-cell plates and was often an indistinct architectural change on H&E-stained slides, requiring reticulin staining (see below) for complete appreciation of this change. Importantly, and in contrast to cirrhosis, there was no fibrosis surrounding these areas of nodular regeneration. Central venous intimal fibrosis, as described in humans with SOS, was not a consistent feature in monkeys.
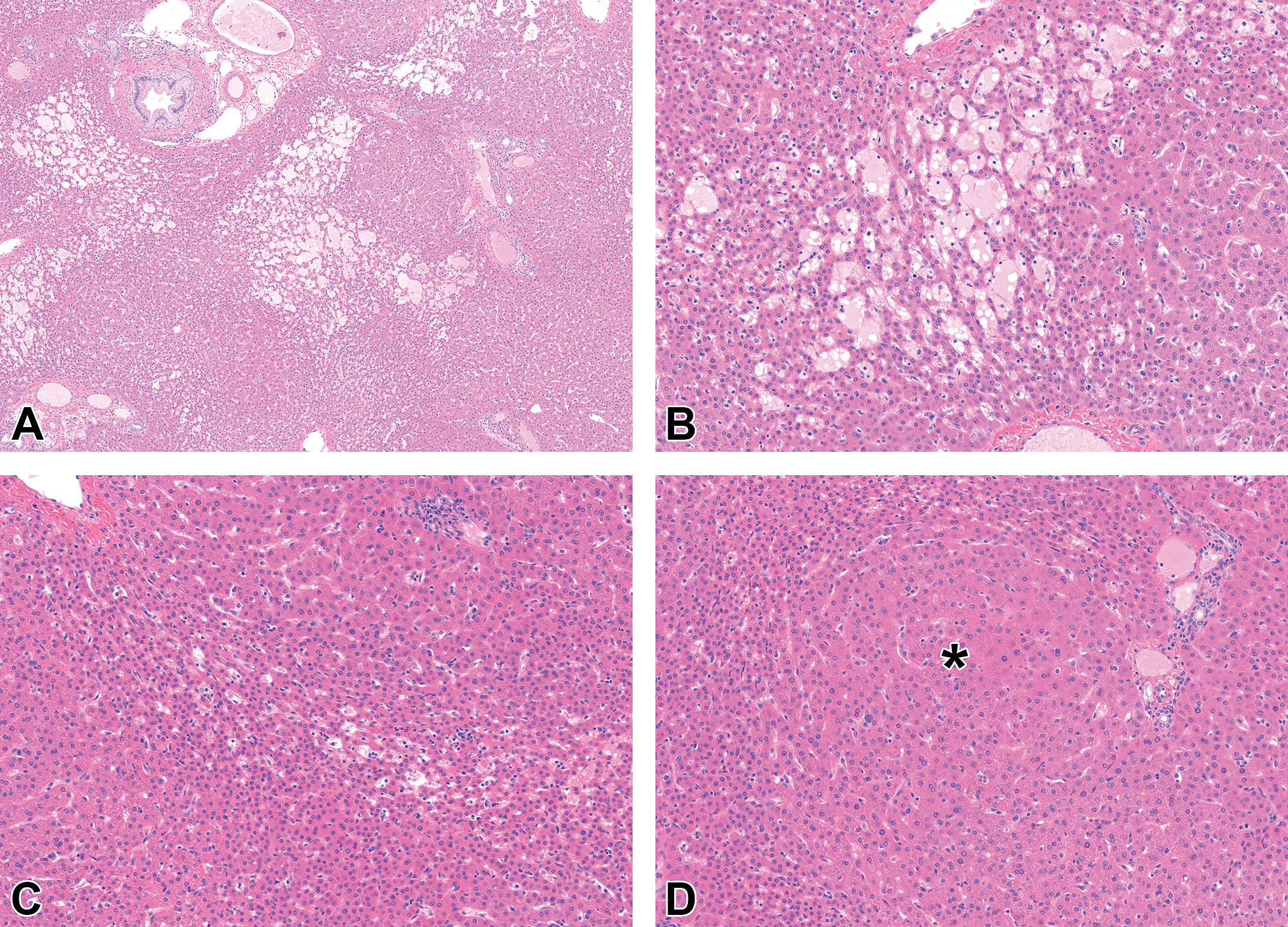
Characteristic light microscopic findings of sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, recovery phase. Hematoxylin and eosin (H&E) stain. (A) Low-power magnification reveals irregular areas of sinusoidal dilation. (B) Area of sinusoidal dilation. The affected sinusoids are variably dilated, filled with homogeneous eosinophilic material (plasma) or less commonly red blood cells (congestion—not shown), and separated by thin cords of atrophic hepatocytes. (C) Midzonal band of atrophic hepatocellular plates composed of cells with scant cytoplasm. (D) Nodular regeneration. Nodule of regenerative double cell-thick hepatocellular plates (*) limited at the periphery by atrophic plates.
Reticulin Staining Was Useful for Identifying Liver Architectural Alterations of SOS in Monkeys
To better characterize the architectural alterations in the livers of recovery phase animals, a silver histochemical stain was performed to highlight the reticulin fibers of the hepatocellular plate scaffold. The reticulin stain readily highlighted areas of nodular regeneration in recovery phase animals as well as atrophic hepatocellular plates peripheral to the nodules (Figure 4E and F). The use of this stain allowed underappreciated changes on H&E-stained slides to be readily differentiated from control animals (Figure 4A and B). Reticulin staining also helped to demonstrate atrophic hepatocytes in areas of sinusoidal dilation, which were difficult to appreciate on H&E (Figure 4C and D). Due to the significant benefit of this method, the reticulin stain was subsequently performed on liver tissue from dosing phase animals, revealing previously unrecognized architectural alterations consistent with hepatocellular atrophy (data not shown).
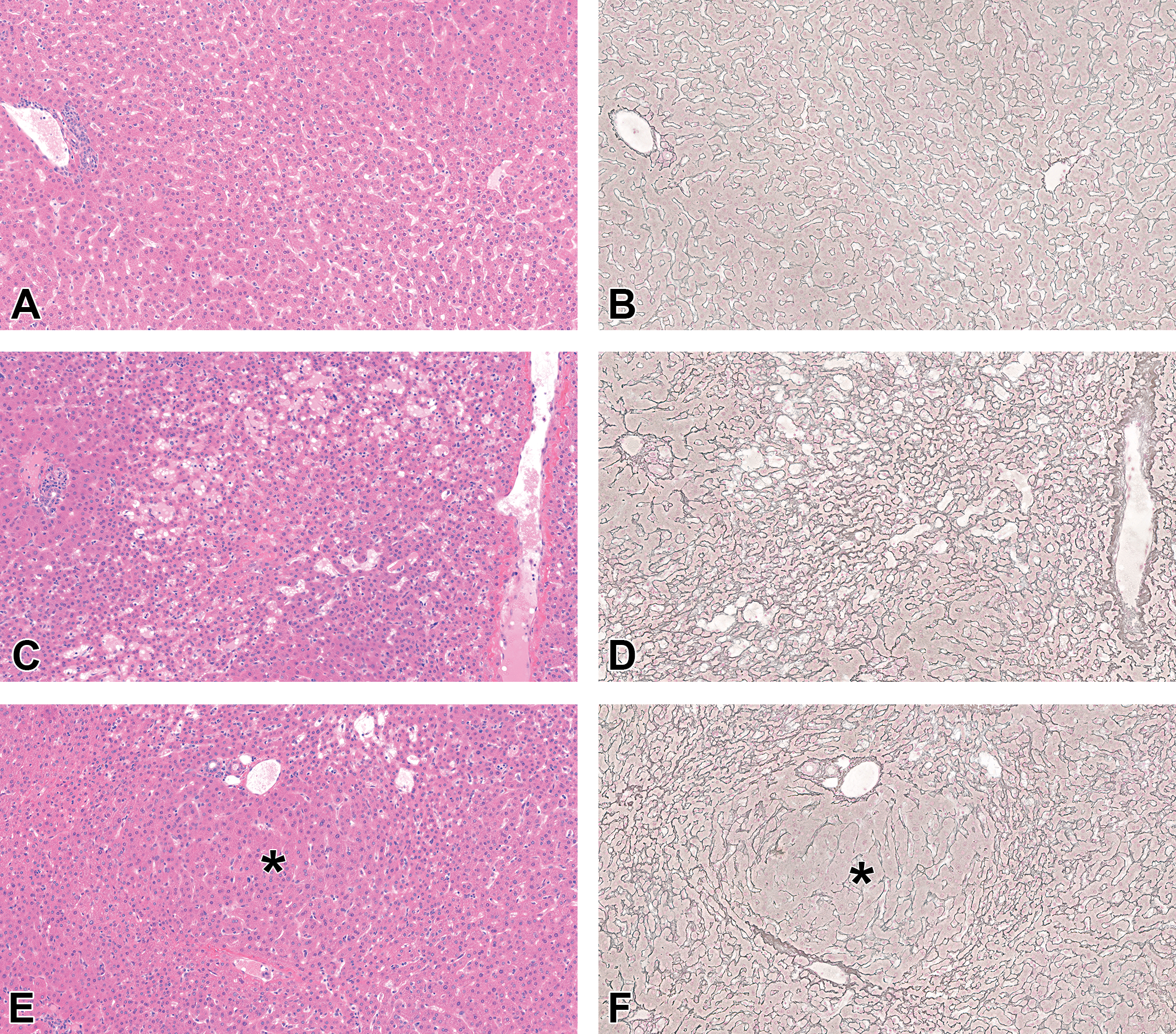
The reticulin stain is most useful to demonstrate the parenchymal alterations of sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, recovery phase. (A, C, E) Hematoxylin and eosin (H&E) stain. (B, D, F) Silver stain. (A, B) Control monkey. Note the evenly sized hepatocellular plates nicely outlined by the silver stain for reticulin fibers. (C, D) ADC-008-dosed monkey. Locally extensive area of hepatocellular plate atrophy associated with mild sinusoidal dilation. The silver stain (D) demonstrates the extent and magnitude of the plate atrophy that is only partially appreciated at evaluation of H&E-stained section (C). (E, F) ADC-008-dosed monkey. Nodular regeneration. The silver stain (F) clearly demonstrates a regenerative nodule (*) composed of double-cell hepatocellular plates limited at the periphery by atrophic hepatocellular plates. The matching H&E-stained section (E) shows parenchymal alterations and variation in hepatocellular plate thickness without clear identification of a regenerative nodule (*).
Immunohistochemistry Reveals Endothelial Cell Loss and Replacement by CD34-Positive Cells
In humans, the underlying cause of SOS is an injury to endothelial cells, primarily SECs. 2 To evaluate the SECs in monkeys in this study, IHC staining for VEGFR2 (endothelial cell marker) and CD34 (marker of endothelial cell precursors likely derived from bone marrow) was performed. VEGFR2 staining showed a continuous positive signal in SECs lining hepatic plates in control animal liver tissue throughout the hepatic lobule (Figure 5A). In contrast, livers from recovery phase animals demonstrated multifocal loss of VEGFR2 staining, consistent with endothelial cell loss, particularly in areas of sinusoidal dilation (Figure 5C and D). CD34-positive endothelial cells in control animals were limited to the portal tract and centrilobular vasculature (Figure 5B). In recovery animals, CD34-positive endothelial cells were seen lining hepatic plates multifocally throughout the hepatic lobule, consistent with a regenerative response to SEC loss in these areas (Figure 5E and F). These characteristic IHC staining patterns were also present in animals at the end of dosing phase in the absence of significant hepatic architectural alteration (data not shown).
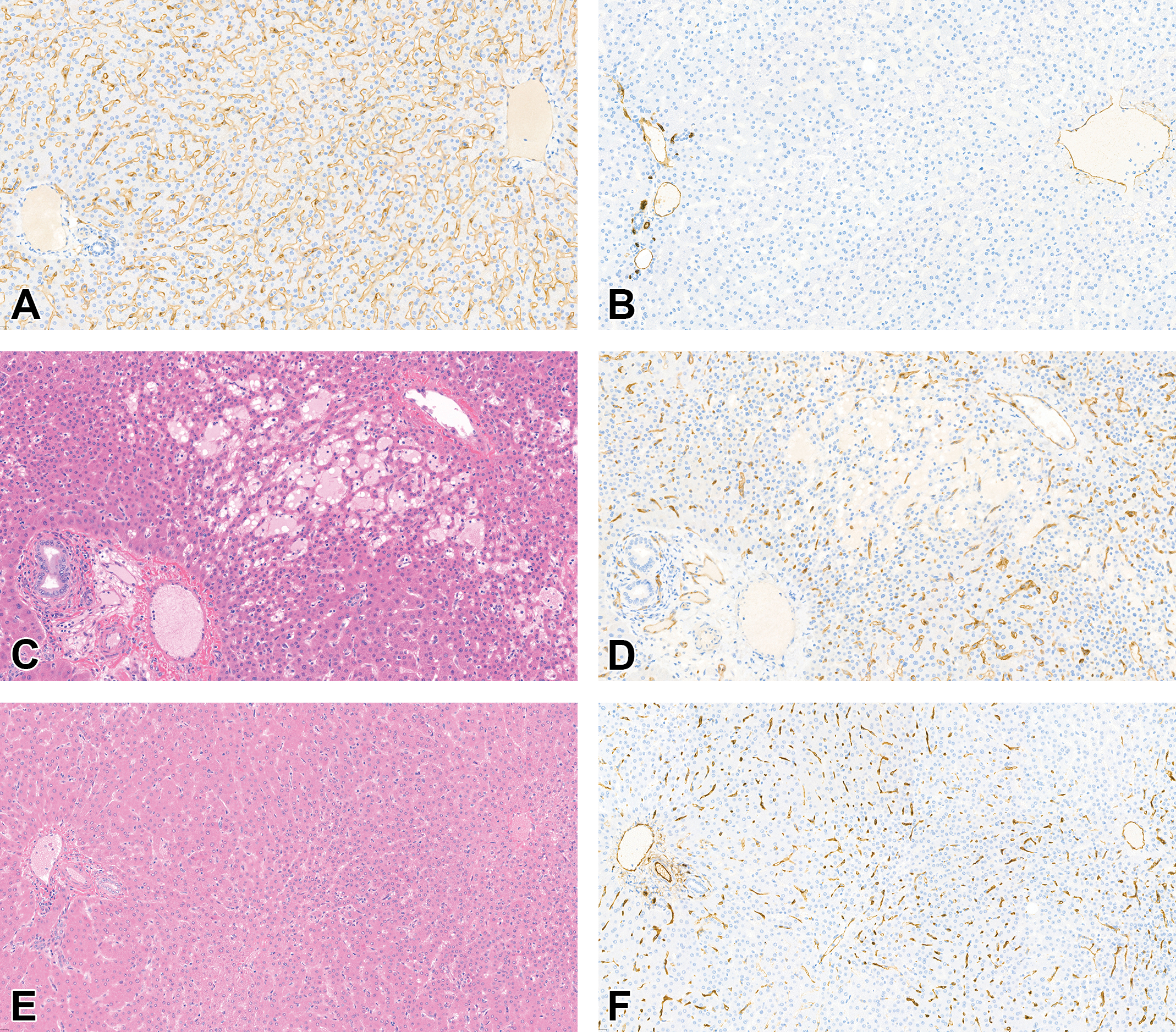
VEGFR2 and CD34 immunohistochemistry (IHC) demonstrate alterations of sinusoidal endothelial cells (SECs) in sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, recovery phase. (A, D) VEGFR2 IHC. (B, F) CD34 IHC. (C, E) Hematoxylin and eosin (H&E) stain. (A, C, D) VEGFR2 IHC shows delicate and continuous staining of SECs lining hepatocellular plates in control monkey (A) while there is staining disruption and loss of VEGFR2 immunoreactivity in an area of sinusoidal dilation in ADC-008-dosed monkey, which is indicative of loss of endothelial cells (D). The area of sinusoidal dilation is confirmed at evaluation of the matching H&E-stained section (C). (B, E, F) CD34 IHC only stains endothelial cells associated with or in close proximity of the portal triads and central veins in control monkey (B) while there is moderate to marked CD34 immunostaining of SECs throughout the liver lobules in areas of parenchymal alterations in ADC-008-dosed monkey, which is indicative of sinusoidal capillarization (F). The matching H&E-stained section demonstrates alterations in hepatocellular plate thickness (E).
Platelet Accumulation in Sinusoids Correlates With Thrombocytopenia in Monkeys
Isolated thrombocytopenia is described in humans with SOS. 23,24 To investigate the potential for platelet sequestration in the liver following SEC injury in monkeys, IHC staining for platelet marker CD41 was performed on dosing and recovery phase animals. Despite lack of liver findings on H&E-stained slides or significant architectural alterations, dosing phase animals did demonstrate platelet accumulation in the sinusoids, as detected by CD41 IHC (Figure 6D), coinciding with areas of SEC injury evidenced by loss of VEGFR2 staining and presence of CD34-positive cells. This platelet sequestration is temporally concurrent with decreases in platelet counts in this study. As expected, few circulating platelets can be appreciated in the sinusoids of control animals (Figure 6B).
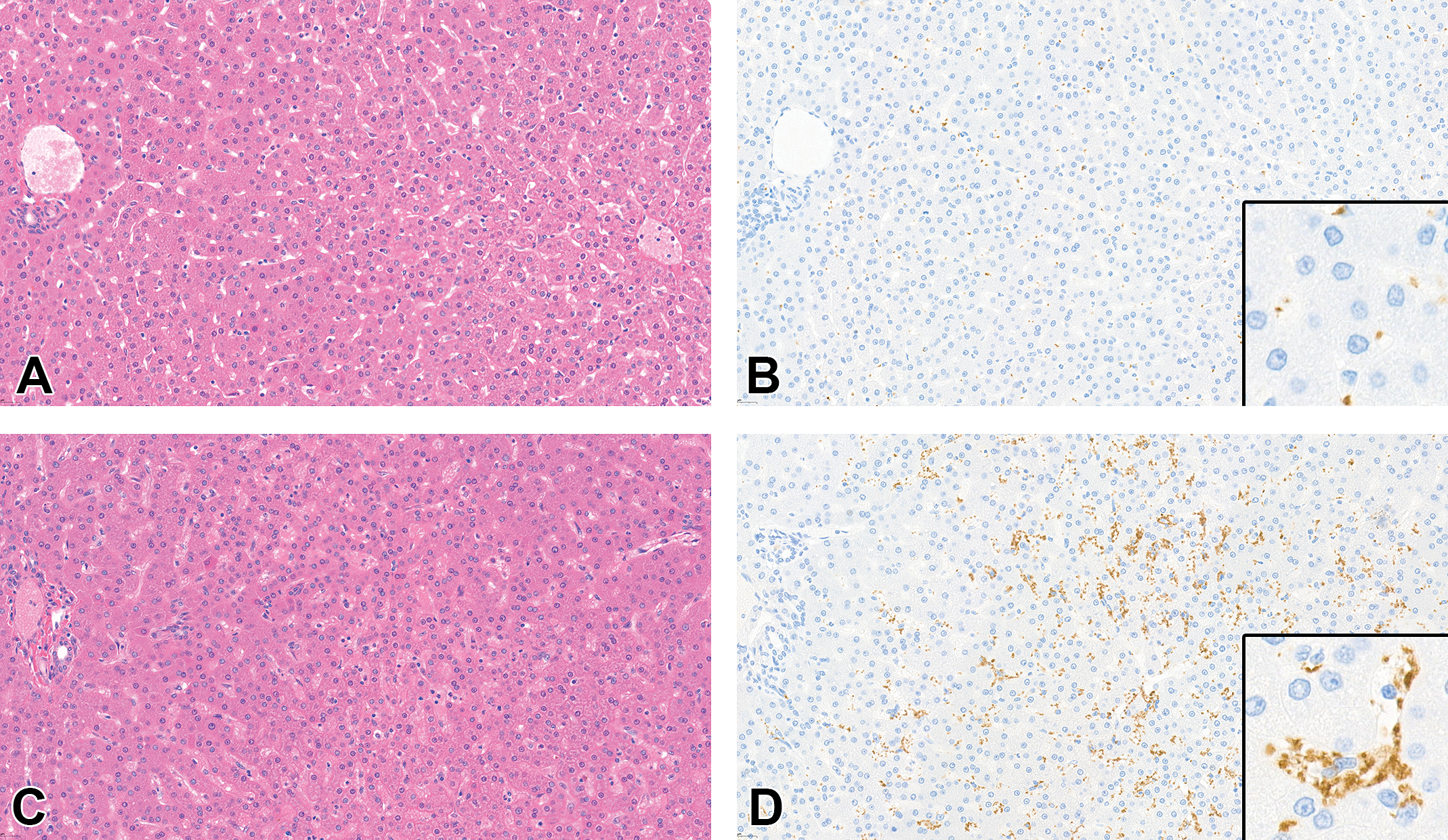
CD41 immunohistochemistry (IHC) demonstrates sinusoidal accumulation of platelets in sinusoidal obstruction syndrome (SOS), liver, cynomolgus monkey, dosing phase. (A, C) Hematoxylin and eosin (H&E) stain. (B, D) CD41 IHC. (A, B) Normal hepatic architecture (A) and few circulating CD41-positive platelets within sinusoids (C, inset) in a control monkey. (C, D) Hepatocellular atrophy and sinusoidal dilation are unremarkable on H&E sections in a dosing phase monkey (C); however, CD41 IHC demonstrates markedly elevated numbers of platelets accumulated in sinusoids (D), with occasional clumping of platelets (inset).
Discussion
Hepatic SOS is an uncommon form of drug-induced liver injury that may be observed both in human patients and in nonclinical animal species during toxicology studies performed to support drug development. We recently observed liver findings consistent with SOS in cynomolgus monkeys dosed with an ADC carrying a potent cytotoxic payload. This article provides the first comprehensive description of the pathology findings associated with this liver condition in monkeys. Although there is published literature on experimental rodent models of SOS, the research has focused on the investigation of mechanism and exploration of treatment or prevention strategies for SOS, 25 with less emphasis on microscopic characterization of the pathology. As SOS is observed almost exclusively after drug or toxin exposure, it is critical to ensure accurate recognition of the liability in toxicity studies in nonclinical species to evaluate human risk, raise awareness of clinicians and, if appropriate, design a suitable monitoring strategy in clinical trials.
Microscopic findings of SOS in monkeys dosed with ADC-008 were primarily characterized by sinusoidal dilation, hepatic plate atrophy, and/or nodular regeneration, associated with overall minimal increases in aminotransferases (primarily AST) and mild to moderate thrombocytopenia. The liver microscopic features described in monkeys are morphologically similar to those reported in human patients with SOS. 9,26 Although SOS is a well-recognized entity, the histopathology diagnosis of the condition remains challenging, especially in nonclinical species, due to its uncommon occurrence, lack of prominent histopathologic features, need for histochemical and/or IHC stains to facilitate the diagnosis, and temporal variability in its manifestation. The findings in monkeys mainly demonstrated irregular hepatocellular plates and sinusoids without any appreciable hepatocyte death or inflammatory response. In the absence of overt sinusoidal dilation, the changes might not be readily appreciated at the examination of H&E-stained slides and could be overlooked. It is therefore critical that the evaluation of suspected cases of hepatic SOS (eg, known class effect of therapeutic agent/modality; observation of isolated thrombocytopenia concurrent with AST increases) is complemented by appropriate special stains. The silver stain for reticulin fibers was undoubtedly the single most useful stain to further characterize the pathology as it provided a detailed assessment of the architecture of the hepatic plates, including detection of sinusoidal dilation, hepatic plate atrophy with a demonstration of collapse of reticulin network, and regenerative nodules with a demonstration of widened double-cell thick plates. Reticulin staining enabled the identification of hepatic plate alterations in dosing phase animals that were not readily identified on H&E-stained slides, although toxicologically meaningful.
Additional stains of diagnostic utility included IHC for VEGFR2 and CD34. Sinusoidal obstruction syndrome is known to be caused by toxic injury to SECs and phenotypic characterization of endothelial cells provided further information on the extent of the damage and confirmed the suspected pathogenesis. VEGFR2 IHC demonstrated altered staining in SOS samples, with loss or disruption of VEGFR2 staining in areas of sinusoidal dilation and hepatic plate atrophy, respectively. Previous investigations in monkeys have demonstrated that the loss of VEGFR2 immunoreactivity is due to the loss of SECs confirming the central role of SEC injury in the development of SOS pathology. 21 Loss of endothelial cells was also confirmed in a study of oxaliplatin in humans where the loss of CD31 staining was observed in areas of sinusoidal dilatation and EM confirmed degeneration and detachment of endothelial cells. 27 We suggest that loss of endothelial lining with aggregation of platelets results in altered local microvascular perfusion in the liver, ultimately causing hepatic cord atrophy and architectural derangement. Additionally, in our study, CD34 IHC demonstrated strong CD34 expression in SECs multifocally in the affected liver parenchyma, while SECs in control monkeys were CD34 negative. CD34 expression in SECs is believed to indicate recovery of previous endothelial damage through the recruitment of bone marrow–derived CD34-positive endothelial progenitor cells, as has been shown in other vascular injury settings. 28,29 However, CD34 expression is indicative of potential liver functional impairment as it is associated with sinusoidal capillarization due to morphological alteration of endothelial cells (ie, development of a basement membrane and loss of fenestrations). 30 Based on the benefit of phenotypic characterization of endothelial cells in monkeys regarding functional significance of the liver findings, we recommend that VEGFR2 and CD34 IHC be considered for the diagnostic work-up of suspected SOS cases not only in nonclinical species but also in patients.
Clinical pathology findings in monkeys dosed with ADC-008 included increases in liver aminotransferases and thrombocytopenia. The effects on liver enzymes were mainly characterized by minimal increases in AST without parallel changes in ALT. Transiently elevated bilirubin was seen in a single recovery animal. This is consistent with previously reported clinical pathology findings in monkeys with sinusoidal injury 21 and is indicative of the poor sensitivity of routine laboratory tests for the diagnosis of SOS. In human patients, hyperbilirubinemia is the only laboratory finding included in the diagnostic criteria of SOS, 3,17 which may be inadequate as increases in serum bilirubin likely occur late in the progression of the pathology. Mild to moderate dose-dependent thrombocytopenia was observed in monkeys dosed with ADC-008 at 0.1 mg/kg/dose (to −89% vs individual baseline) and 0.03 mg/kg/dose (to −58%). Decreases in platelet counts were first noted on day 15 or 22 and partially resolved over the recovery period. Histologic evaluation of sternal bone marrow at the end of the dosing phase did not demonstrate any alterations in hematopoietic cellularity or changes in megakaryocyte density and morphology in ADC-008-dosed animals, indicating that the thrombocytopenia was likely not due to decreased production of platelets by the bone marrow. CD41 IHC for platelets performed on liver samples from selected monkeys demonstrated platelet accumulation within sinusoids in animals with thrombocytopenia necropsied at the end of the dosing phase. These observations are consistent with previous reports indicating that the thrombocytopenia in SOS is due to platelet sequestration in the liver secondary to SEC injury. 21,31,32 Although thrombocytopenia could be a sensitive marker of the initial sinusoidal injury, it is not clear that platelet accumulation is a consistent feature of progressive and/or resolving SOS liver alterations. Peripheral thrombocytopenia with rapid consumption of transfused platelets is frequently observed in human patients with SOS and has recently been added as a diagnostic criterion for SOS in adult and pediatric patients resulting in improved early detection of the condition. 24,33,34
In conclusion, this report has provided a comprehensive characterization of clinical pathology and histopathology findings of SOS in monkeys and highlighted diagnostic challenges. As SOS is primarily a drug-induced injury, it is critical that the liability is appropriately recognized by toxicologic pathologists in nonclinical toxicity studies to appropriately inform drug development strategy and clinical monitoring. Despite refined diagnostic criteria and imaging techniques, SOS is also underrecognized in human patients in clinical trials and should be considered in the differential diagnosis of patients with increases in liver aminotransferases and isolated thrombocytopenia when the liability has been identified in nonclinical toxicology studies.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211037932 - Anatomic and Clinical Pathology Characterization of Drug-Induced Sinusoidal Obstruction Syndrome (Veno-Occlusive Disease) in Cynomolgus Macaques
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211037932 for Anatomic and Clinical Pathology Characterization of Drug-Induced Sinusoidal Obstruction Syndrome (Veno-Occlusive Disease) in Cynomolgus Macaques by Rebecca Kohnken, Hadi Falahatpisheh, Kyathanahalli S. Janardhan and Magali Guffroy in Toxicologic Pathology
Footnotes
Acknowledgments
The authors gratefully acknowledge the histology laboratory staff for preparation and scanning of study materials. Charles River Laboratories staff and study personnel are also gratefully acknowledged for the execution of the described study. AbbVie sponsored and funded the study; contributed to the design; participated in the collection, analysis, and interpretation of data, and in writing, reviewing, and approving the final publication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are or were employees of AbbVie and may own AbbVie stock. HF is currently an employee of Samumed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AbbVie sponsored and funded the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
