Abstract
Göttingen minipigs are increasingly used as an alternative large animal model in nonclinical toxicology studies, and proliferative lesions in this species are rare. Here, we report four cases of cardiac rhabdomyoma in Göttingen minipigs, an incidental and benign mass in the heart. Three cases lacked gross observations and had a microscopic nodule in either the left ventricle or interventricular septum. The last case had a large, firm, raised nodule on a left ventricular papillary muscle noted at necropsy, with additional microscopic intramural masses in the left ventricular wall. In all cases, microscopic evaluation revealed well-circumscribed, expansile nodules composed of bundles of large, highly vacuolated, ovoid to polygonal cells with variable cytoplasmic processes radiating from a centrally located nucleus. Cells displayed patchy accumulation of intracytoplasmic, PAS-positive material and haphazardly arranged cytoplasmic cross-striations. There was no evidence of cardiac insufficiency or other data to suggest the masses were clinically meaningful. Cardiac rhabdomyomas have been reported in meat-hybrid swine, with a breed predisposition in red wattle. This lesion is well established in guinea pigs, but documentation in other laboratory species used in toxicologic studies is limited to two beagle dogs. To our knowledge, this is the first report of spontaneous cardiac rhabdomyoma in Göttingen minipigs.
Keywords
Introduction
Cardiac rhabdomyomas (CRs) are relatively rare in domestic animals and have been detected most frequently in meat-hybrid swine with a breed predisposition in red wattle and red wattle crosses. 1 Spontaneous cases have also been reported in sheep, cattle, and dogs. 2 As there are no reports of CRs associated with heart failure in swine, these growths are generally regarded as incidental findings.1,3,4 However, they have rarely been identified in animals that died with no ante-mortem clinical signs or identifiable concurrent disease at necropsy, so the possibility of sudden death from myocardial conduction disruption cannot be excluded.1,5 In addition, CRs have been found in neonatal pigs dying of other causes (e.g., viral diarrheal disease) and in fetal piglets from euthanized ill sows, supporting their congenital origin. 1 Grossly, CRs are well-demarcated, often multiple, intramural nodules and are most common in the ventricular wall. Similar to human cases, these nodules are histologically characterized by large, highly vacuolated cells that stain strongly with periodic acid-Schiff (PAS), consistent with intracytoplasmic glycogen accumulation.2,4
Except for guinea pigs, reports of CR are sparse in laboratory species used in toxicologic studies. While macroscopic CRs have been reported in guinea pigs, the more typical presentation is multiple, microscopic nodules within the ventricular wall with no obvious gross lesions. These microscopic foci retain the characteristic histologic features of CR cells and are often termed rhabdomyomatosis.6 -10 Spontaneous CRs have not been reported in any other laboratory rodent, including mice, rats, or hamsters. Reports of CRs in large animal species are limited to two isolated case reports in laboratory beagle dogs,11,12 and there are no reports in commonly used non-human primate species, including marmosets, rhesus macaques, and cynomolgus macaques.
In contrast, CR is the most common congenital cardiac tumor in humans and accounts for 40-60% of all pediatric heart tumors. It is considered a benign, non-neoplastic hamartoma in humans and occurs most frequently within the ventricles, though any cardiac location is possible.13,14 Clinical signs depend on tumor location and size, but range from asymptomatic to causing arrhythmias, valvular obstruction, decreased cardiac output, and death.13,15,16 Partial to complete spontaneous regression occurs in 33-50% of cases during the first four years of life, supporting a non-neoplastic etiology. 16 CRs in humans are strongly associated with tuberous sclerosis complex (TSC), a genetic disorder characterized by the development of multiorgan hamartomas, most commonly in the brain, kidney, heart, and lung.16,17 Development of TSC is associated with inactivating mutations in one of two tumor suppressor genes: tuberous sclerosis-1 (TSC1) hamartin gene or tuberous sclerosis-2 (TSC2) tuberin gene.16,18 Between 50-60% of patients with TSC have CR, and approximately 80% of children with CR have TSC.16,17,19,20 Unlike in humans, no congenital tumor syndrome resembling TSC has been linked with CRs in swine.
Recently, the use of pigs and minipigs in large animal toxicologic studies has increased due to similarity with humans in their drug metabolism and integumentary, cardiovascular, and reproductive systems. 21 Minipigs are particularly useful as they are more handleable, easier to house, and require less test article than full-sized pigs. 22 The Göttingen minipig is the preferred breed for Good Laboratory Practice (GLP) studies, as this purpose-bred breed has the highest specific pathogen-free standards and defined microbiota of commercially available minipigs.22,23
Reports of tumors in the Göttingen minipig (and minipigs in general) are uncommon. Although rare, lymphoma is the most frequently diagnosed neoplasia in minipigs, as well as the most common malignancy in all pig breeds.23,24 Here, we provide the first report of spontaneous CR in Göttingen minipigs, which is also the first report of this condition in minipigs as a whole.
Materials and Methods
All in vivo animal procedures were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited facility under an Institutional Animal Care and Use Committee–approved protocol. Standard procedures and conditions for animal care, housing, access to water and food, environment, and room maintenance were used. All other procedures were performed in accordance with laboratory standard operating procedures and/or established laboratory best practices.
Animals
Case 1 was a 7.5-month-old female Göttingen minipig at termination and was in the high-dose treatment group of a three-and-a-half-month toxicity study with transdermal application of the test article. Cases 2 and 3 were 9-month-old male Göttingen minipigs in the 56-day recovery vehicle control and mid-dose groups, respectively, of a 93-day subcutaneous injection toxicity study. Case 4 was an 8-month-old male Göttingen minipig in the mid-dose treatment group of a 24- to 28-week oral gavage toxicity study. All pigs were sourced from a commercial supplier (Marshall Farms). Euthanasia and necropsy were performed according to SOP guidelines.
For cases 1, 2, and 3, the heart was a protocol-directed tissue for organ weight collection, macroscopic examination, and microscopic evaluation. For case 4, the heart was macroscopically evaluated but not weighed per protocol, and was not a protocol-directed tissue for microscopic examination; the gross observation of nodules in the heart prompted histologic collection and microscopic evaluation. Based on the cardiac finding, hearts from all animals in the study (control and treated) were evaluated microscopically as a potential target organ.
Histology Processing and Special Staining
Cardiac tissue was fixed in 10% neutral-buffered formalin, embedded in paraffin, and sectioned at 4 to 5 μm. Tissue sections were placed onto glass microscope slides and stained with hematoxylin and eosin (H&E), periodic acid-Schiff (PAS) reaction, or phosphotungstic acid hematoxylin (PTAH).
Results
No clinical abnormalities were observed in any case animals during their respective studies (Table 1). A single, intramural nodule was present microscopically in the left ventricle of case 1 and 2 and in the interventricular septum of case 3. Case 4 had a firm, red, raised nodule measuring 1.7 cm × 1.0 cm × 0.7 cm noted grossly on the papillary muscle of the left ventricle at scheduled necropsy. During microscopic evaluation, an additional nodule was present in an adjacent papillary muscle (multifocal). No animals demonstrated evidence of heart failure or systemic manifestations of cardiac dysfunction (e.g., lacked pulmonary edema). No additional masses were observed in other organs, and no clinically significant cardiac findings or cardiac-related lesions were present in remaining treated or control animals from these studies.
Summary of clinical features and gross findings.
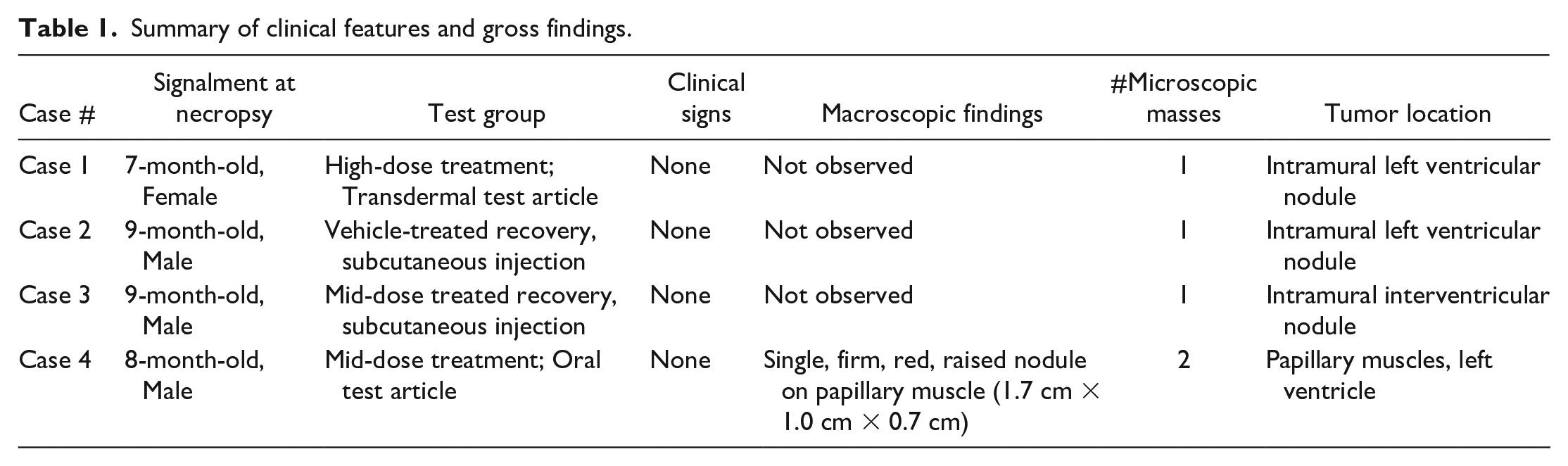
Microscopically, lesions were round, well-demarcated, non-encapsulated, moderately cellular nodules within the myocardium of the left ventricular wall (cases 1 and 2), the interventricular septum (case 3), and papillary muscles (case 4) (Figure 1A). Nodules were composed of bundles of large, oval to polygonal cells with variably distinct cell borders and vacuolated to deeply eosinophilic cytoplasm, supported by a subtle fibrovascular stroma (Figure 1B). Cytoplasmic vacuoles were variably sized and often clear (devoid of content) on H&E-stained sections; occasional vacuoles contained fibrillar to flocculent eosinophilic material. Cells had large, round to oval, predominantly centrally located nuclei with coarsely stippled chromatin and one to two prominent nucleoli. Nuclei were frequently surrounded by a slight perinuclear clear zone with finely granular basophilia of the adjacent cytoplasm (Figure 1C). Many cells had eosinophilic tendrils of cytoplasm that radiated out from a central nucleus and interdigitated around cytoplasmic vacuoles (Spider cells, Figure 1D). Mild to moderate anisokaryosis was present with a lack of mitotic figures.
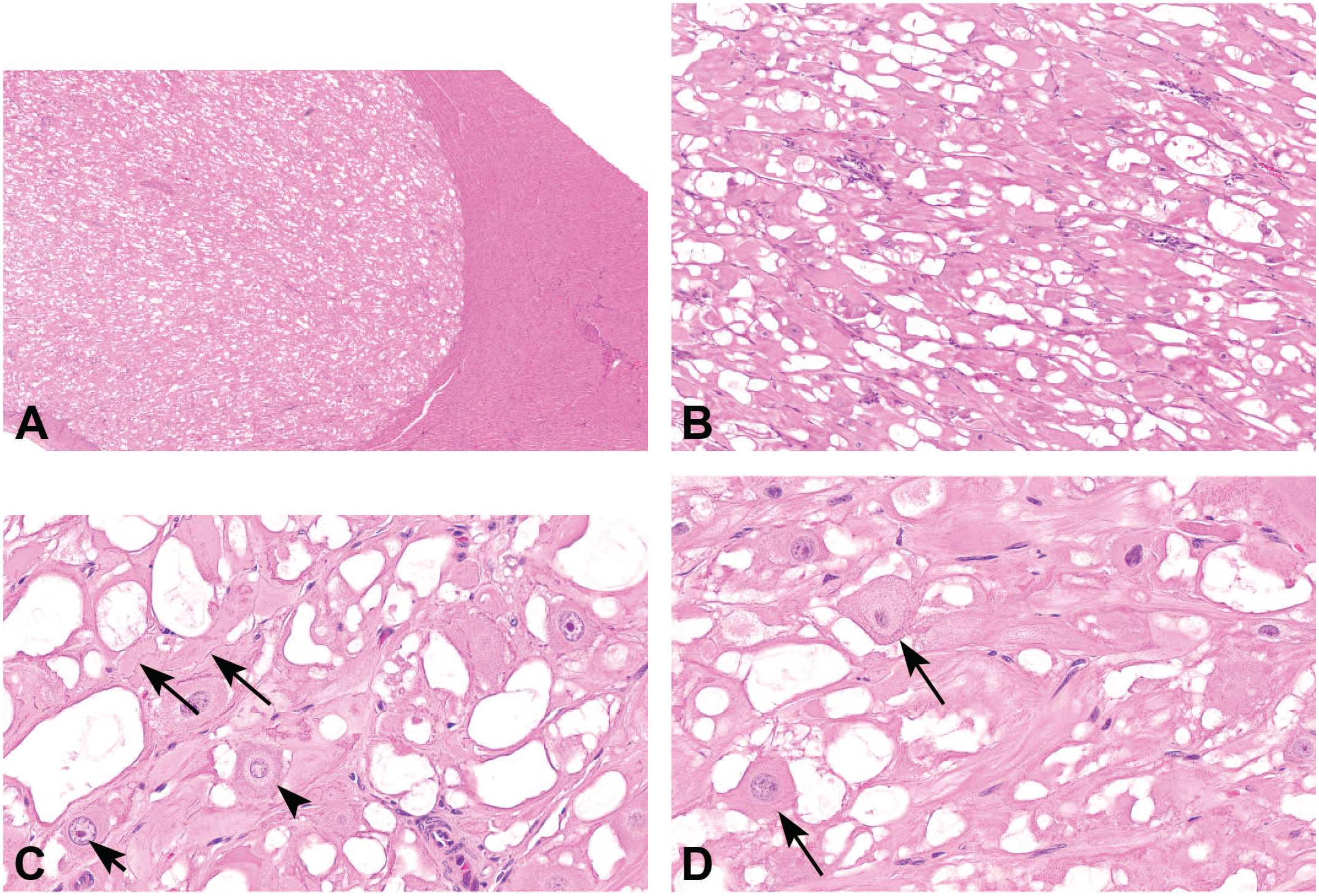
Microscopic features of cardiac rhabdomyoma, hematoxylin and eosin. (A) Cardiac mass is well-demarcated, non-encapsulated, intraventricular, and nodular, original objective 2X. (B) Mass composed of bundles of large, variably vacuolated to deeply eosinophilic, branching cells, original objective 10X. (C) Cardiac rhabdomyoma cells with prominent nucleoli (solid arrow) or perinuclear clearing with adjacent increased basophilic cytoplasmic granularity (arrowhead). Cells contain low numbers of thin basophilic strands suggestive of disorganized cross-striations (dashed arrow), original objective 40X. (D) Spider cells with eosinophilic cytoplasmic streams radiating out from a centrally located nucleus (arrows), original objective 40X. To access the whole-slide image provided with this figure, scan the QR code on cover page 3 or visit slide 1 , slide 2 .
Special histochemical stains demonstrated patchy accumulation of PAS-positive material within CR cells, with lack of staining in adjacent normal cardiac myocytes (Figure 2A). PAS-positive material, consistent with glycogen, occasionally accumulated within vacuoles (Figure 2B), but was generally intracytoplasmic and displaced toward the cellular periphery or rarely clustered around the nucleus. PTAH histochemical stain highlighted muscle cross-striations in normal, unaffected cardiac myocytes as dense accumulations of regularly-arranged, PTAH-positive filaments (upper right corner of Figure 2C), whereas CR cells in Figures 2C and 2D demonstrated a comparatively low density of cross-striations with PTAH that were haphazardly arranged and occasionally encircled vacuoles or irregularly partitioned segments of cytoplasm.
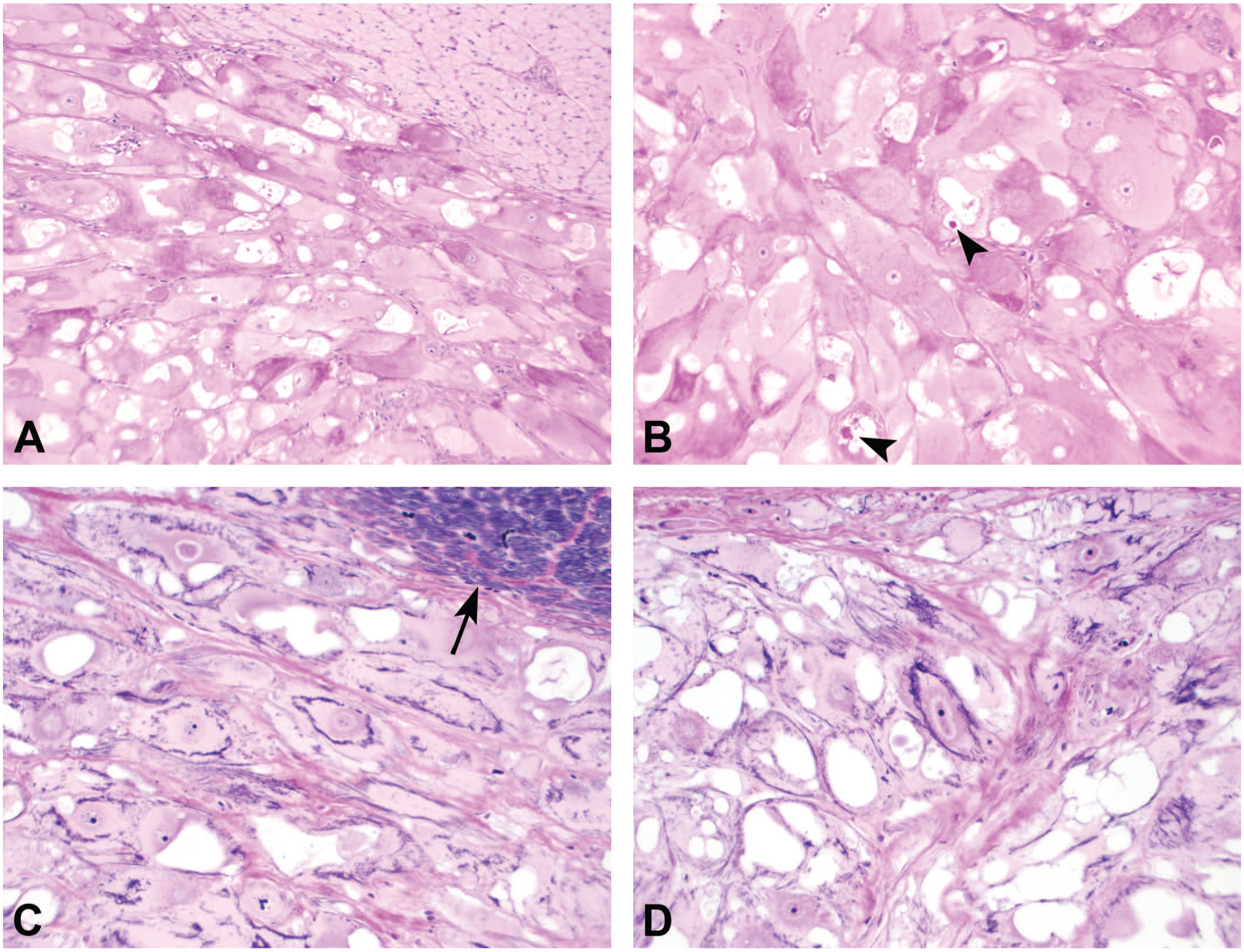
PAS and PTAH histochemical stains. (A) CR cells have patchy accumulation of PAS-positive material, consistent with cytoplasmic glycogen, dispersed toward the cellular periphery and often compressed by PAS-negative vacuoles. Adjacent normal cardiac myocytes (upper right corner) are PAS-negative, original objective 10X, PAS stain. (B) Intracytoplasmic vacuoles occasionally contain PAS-positive material (arrowheads), original objective 20X, PAS stain. (C and D) There is a low density of haphazardly arranged cytoplasmic cross-striations in CR cells compared with normal cardiac myocytes (arrow), original objective 20X, PTAH. CR indicates cardiac rhabdomyoma; PAS, periodic acid-Schiff; PTAH, phosphotungstic acid hematoxylin.
Discussion
These 4 cases of incidental, intracardiac masses in Göttingen minipigs had macroscopic and microscopic features of CR, a condition reported in humans and veterinary species. These masses are characterized by well-demarcated nodules within the myocardium composed of large, variably vacuolated cells with abundant, intracytoplasmic, PAS-positive material (glycogen) and few haphazardly arranged cross-striations with PTAH staining.2,5,13,14 Based on the tumor composition of large, polygonal cells with abundant eosinophilic granular cytoplasm, differentials of oncocytoma and granular cell tumor were considered. However, these tumors of epithelial or neuroendocrine/neuroectodermal origin, respectively, were ultimately excluded based on the myocardial location and presence of PTAH-positive cross-striations, indicative of striated muscle origin in this case.25 -28 Similarly, the accumulation of highly vacuolated, PAS-positive cells in the heart in CR was distinguished from the differential of glycogen storage disease by their formation of discrete well-formed nodules, absence of histologic changes in other organs (namely skeletal muscle, liver and/or central nervous system), and the absence of clinical signs in all affected animals.29,30
In humans, CRs are the most common pediatric heart tumor and are often detected in utero via fetal echocardiography. 14 Although the CRs presented here were first detected at post-mortem examination, all animals were young and less than one year of age. This correlates well with previous reports of CRs occurring in stillborn and neonatal red wattle piglets and suggests CRs occur as congenital lesions in Göttingen minipigs. 1 While CRs in humans are highly associated with TSC, a genetic disorder characterized by benign tumors forming in multiple organs,13,16,20 no extra-cardiac tumors were detected grossly or microscopically in the affected minipigs described here.
A notable feature of human CRs is their tendency to spontaneously regress during the first few years of life. Perinatal development of CRs is documented in pigs however, due to infrequent in utero/in vivo monitoring, it is unknown whether porcine CRs exhibit a similar pattern of in utero growth followed by spontaneous regression in early life. 1 It has been proposed that this abnormal growth pattern is related to fetal exposure to high levels of maternal progesterone, followed by its abrupt removal at birth.31,32 Considering this, it is not surprising that guinea pigs are the only reported rodent species to spontaneously develop CRs, as they have a markedly longer gestational period (59-72 days) than mice (19-21 days), rats (21-23 days), or hamsters (16-22 days).
Although these intracardiac nodules have been commonly termed CRs and/or rhabdomyomatosis, the histogenesis of CRs remains unclear. Additional alternative nomenclature has included cardiac hamartoma, dysplasia of fetal cardiac tissue, and myocardial or Purkinje cell neoplasm.2,4,13,33 Previous ultrastructural studies of CRs demonstrated features specific to cardiac myocytes (versus Purkinje cells), including T-tubules, a sarcoplasmic reticulum, and intercalated disks. 4 However, the abundant intracytoplasmic glycogen, low density of unorganized myofibrils, and reports of cytoplasmic expression of Purkinje fiber marker Protein Gene Product 9.5 (PGP 9.5) suggests Purkinje fiber origin.4,33 While the uncertain histogenesis of CRs has contributed to the inconsistent terminology applied to this mass, the term we have applied and that is currently in use in humans and in swine is that of cardiac rhabdomyoma, which is considered to be a benign, non-neoplastic hamartoma. Hamartomas are defined as benign, non-neoplastic growths composed of an abnormal mixture of cells and tissues found in their normal anatomic location, which is consistent with the mixture of Purkinje fiber and cardiac myocyte features observed in CRs. In contrast, neoplasms are defined as an abnormal proliferation of cells that are nonresponsive to normal physiologic growth controls, which is inconsistent with CRs because of their lack of mitotic activity, their incidental and congenital nature, and their tendency to spontaneously regress in humans. Based on their similarity to the human condition, we also consider CRs in pigs to be a benign non-neoplastic hamartoma.
These four cases constitute the first report of cardiac rhabdomyoma in Göttingen minipigs. The low incidence of spontaneous lesions in minipigs is likely influenced by their short history as a toxicologic model, the relatively young age of study animals, and the tendency to use minipigs in short-term studies, all of which combine to limit the potential development and thus presence of tumors. 23 The limited number of animals affected in multiple studies, the occurrence of CRs in vehicle-treated as well as test article–treated animals, the absence of a dose-responsive incidence, the absence of cardiac lesions in other treated animals, and the morphologic similarity to spontaneous CRs reported in domestic red wattle pigs are strongly suggestive that CRs occur as a rare, spontaneous, incidental, and potentially congenital background lesion in minipigs. As Göttingen minipigs are increasingly used in toxicologic testing and as models of human disease, greater understanding and recognition of background lesions in this breed is essential to ensure accurate interpretation of toxicologic findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
