Abstract
Atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) are cardiac hormones located in atria granules. Both peptides respond to cardiac pressure and volume dynamics and accordingly serve as translation biomarkers for the clinical treatment of heart failure. Serum ANP and BNP play central secretary roles in blood pressure and cardiac output regulation and have proven utility as differential biomarkers of cardiovascular proficiency and drug-induced maladaptation, yet both peptides are impervious to exercise-induced hypertrophy. We employed immunoelectron microscopy to examine the effects of 28 days of chronic swim exercise or administration of a PPARγ agonist on atrial granules and their stored natriuretic peptides in Sprague Dawley rats. Chronic swimming and drug treatment both resulted in a 15% increase in heart weight compared with controls, with no treatment effects on perinuclear granule area in the left atria (LAs). Drug treatment resulted in larger size granules with greater BNP density in the right atria. Comparing swimming and PPARγ agonist treatment effects on ANP:BNP granule density ratios between atrial chambers revealed a shift toward a greater proportion of ANP than BNP in LAs of swim-trained rats. These data suggest a distinction in the population of ANP and BNP after chronic swim or PPARγ that makes it a novel metric for the differentiation of pathological and physiological hypertrophy.
Keywords
Introduction
Natriuretic peptides are cardiac hormones that serve essential roles in maintaining systemic hemodynamics. Two of these peptides, atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP), are contained in granules in the atria of the heart and are released in response to pressure- or volume-related cardiac stretch. Both ANP and BNP and their more stable cleavage products, NT-proANP and NT-proBNP, respectively, serve as effective biomarkers in the evaluation and management of heart failure. Recent evidence has provided information on the translational utility of these biomarkers in the drug development space, where they have promise for the identification of compounds with potential cardiac liabilities.1,2
Natriuretic peptides are synthesized in the rough surface endoplasmic reticulum of the cardiomyocyte perinuclear (PN) region and transported to the Golgi apparatus where they are packaged into atrial granules and prepared for exocytosis. ANP reacts to vascular distension by providing a response that results in plasma volume reduction.3,4 Atrial ANP granule density has shown promise as a predictor for cardiovascular events because it is sensitive to acute and chronic physiologic challenges, including (1) blood volume expansion, 5 (2) high sodium diet, 6 (3) temperature, 7 and (4) beta-adrenoceptor blockade compounds. 6 ANP is synthesized and secreted predominately in the atria in response to atrial stretch. 8 Atrial BNP is synthesized in small amounts; however, during conditions such as heart failure there is a shift to more BNP synthesis in the ventricles. 9 This provides protection against elevated filling pressure 10 especially in association with pulmonary hypertension and right ventricular decompensation.11,12 Both peptides were initially immunolabeled and identified, localized, and quantified in the rat and other species by Jamieson and Palade. 13
In humans, during wall stress or changes in oxygen tension as occurs in both pathologic (ie, heart failure) and physiologic (ie, exercise) settings, both ANP and BNP are released in an attempt to address volume or pressure overload. 14 Natriuretic peptides respond to myocardial physiologic overload by providing functional and metabolic protective effects that include regulation of sodium reabsorption and water balance. 15 They also regulate blood volume and pressure by directly increasing venous compliance, which in turn decreases central venous pressure and cardiac output. Furthermore, by vasodilating arteries, they reduce both systemic vascular resistance and arterial pressure. 16 Exercise training, including swimming, is known to transiently increase natriuretic peptide concentrations in circulation. 17 In the case of swimming, immersion in water alone has been reported to increase plasma concentrations of natriuretic peptides due to a shift of fluid from intracellular compartments to the intravascular compartment, thereby increasing plasma volume and cardiac output without increased energy expenditure according to the Frank-Starling law. 18
Chronic treatment with a peroxisome proliferator–activated receptor γ (PPARγ) agonist causes left-ventricular hypertrophy in rats.19,20 Recently, we compared the effects of exercise and PPARγ agonism on cardiac hypertrophy in male Sprague Dawley rats and found that even though both treatments resulted in a 15% increase in heart weight, NT-proANP and NT-proBNP serum levels were persistently elevated only in concert with drug-induced cardiac hypertrophy. 1 In contrast, chronic exercise in rats failed to result in persistent measurable increases in circulating NT-proANP and NT-proBNP concentrations. Exercise,21,22 and drug overload,23-25 in part through the differential expression and secretion of natriuretic peptides, points out a unique distribution in peptide population resulting from different stresses that cause cardiac hypertrophy. In summary, serum natriuretic peptides serve as differential translational biomarkers between drug- and exercise-induced pathological and physiological cardiac hypertrophy. 1 Furthermore, the utility of two-faced immunolabeling of atria and ventricle ANP and BNP can identify population differences in natriuretic peptides to highlight differences between physiological and pathological cardiac stress.
The aim of the present study was to identify the distribution of ANP and BNP granules associated with drug-induced and exercise-induced cardiac hypertrophy in rats. We used immunoelectron ultramicroscopy to characterize the morphology of ANP and BNP and peptide granules (PGs) in the atria of chronic swim-trained and drug treatment subgroups. This study supports the utility of localization and quantification of immunolabeled natriuretic peptides as tissue biomarkers of physiologic- and pathologic-induced cardiac hypertrophy.
Research Involving Animals
The University of Rhode Island and Institutional Animal Care and Use Committee reviewed and approved all experimental procedures in accordance with federal and state guidelines. Male Sprague Dawley rats (Charles River Laboratories, Raleigh, North Carolina), aged 8 to 10 weeks and weighing 240 to 290 g were acclimated for one week prior to the experimental study. Rats were caged in a temperature-controlled environment (22 ± 2°C) of 12-hour light-dark cycles and fed Purina Certified Rodent pellets #5002-9 and reverse osmosis filtered water. Atrial tissue samples were taken from active control (AC), swimming (S) and drug-treated (DT) Sprague Dawley rats that underwent four weeks of daily swim exercise or PPARγ agonist administration as previously described. 1
Materials and Methods
Swimming and PPARγ Agonist Protocols
In the study by Dunn et al (2017), rats were randomly assigned to four groups: (1) AC, (2) S, and (3) DT, and (4) control (C). The AC consisted of 10 rats, while the S and DT groups consisted of 11 rats each. Control group rats, which did not undergo any swim training and were not handled except for blood collection, were not included in this study. We used sub-samples from AC, S, and DT animals for immunolabeling and qualitative tissue analysis. Animals in the AC group swam twice daily for four weeks with each swim session lasting two minutes and separated by at least four hours. The 28-day swim training regimen (group S) followed similar protocols by Hickson et al 26 and Kaplan et al, 27 culminating in two 90-minute sessions daily separated by at least 4 hours. Animals in the DT group were administered a PPARγ agonist, previously reported to expand plasma volume in concert with cardiac hypertrophy in Sprague Dawley rats. 28 PPARγ agonist dosage was given once daily by oral gavage at 100 mg/kg for 4 weeks. A more detailed description of the swim and drug treatment protocols is reported in Dunn et al (2017). 1
Electron Microscopic Immunolabeling of ANP and BNP
ANP and BNP hormones are packaged within atrial secretory granules localized in PN, myofibrillar (mf), and subsarcolemmal (SS) regions of cardiomyocytes. We used transmission electron microscopy and immunogold labeling of atrial tissue from rats used to identify serum levels of NT-proANP and NT-proBNP as potential biomarkers for differentiating drug-induced pathological hypertrophy and physiological hypertrophy caused by swim exercise. 1 Immunogold labeling enables identification, localization, and quantification of ultrastructures, 29 including atrial natriuretic granules and their respective ANP and BNP.30,31
Small portions of left atrium (LA) and right atrium (RA) and both ventricles were excised and placed into fixative containing 1% paraformaldehyde and 2% glutaraldehyde in 0.15 M sodium cacodylate buffer at 4°C. Atrial and ventricle samples were immediately prepared into 1 mm3 blocks and allowed to fix in refrigeration for 3 hours. Following several buffer rinses, tissue samples were dehydrated in an ascending ethanol series up to 75%, filtrated with LR White acrylic resin, hard grade (London Resin Co. Stansted, England, Dissolved Janurary 7, 2020) and polymerized in filled gelatin capsules at 50°C overnight. We selected the PN area of cardiomyocytes containing a clearly distinct region with densely populated mitochondria, PGs, and endoplasmic reticulum for quantitative analysis of atrial granules and their secretory natriuretic peptides. Semi-thin sections (1 µm) were prepared using a Reichert Ultracut S microtome, stained with methylene blue-azure II, and evaluated for regions of longitudinal muscle fibers. For immunogold labeling, ultrathin sections (75 nm) from PN areas of myocytes were prepared and collected on 300-mesh nickel grids that had been pretreated with Coat-Quick G (EMS, Hatfield, Pennsylvania) for better section adhesion. For double immunogold labeling, each side of the grid was labeled separately. Sections were pretreated by floating on filtered droplets of blocking solution, phosphate buffered saline (PBS) with 0.4% bovine serum albumin (BSA), and 2% normal goat serum for 30 minutes in a humid chamber. Grids were carefully transferred to the surface of droplets of rabbit anti-rat ANP antibody diluted 1:500 in PBS/BSA 0.2% buffer (Phoenix Pharmaceuticals, Belmont, California) and incubated overnight at 4°C. Following rinses on droplets of PBS/BSA 6 × 5 minutes each, the sections were placed on droplets of 10-nm gold-labeled goat anti-rabbit IgG antibody (EMS/Aurion, Hatfield, Pennsylvania), diluted 1:30, and incubated for two hours at room temperature and used to label ANP. Grids were rinsed on droplets of buffer followed by distilled water, then blotted and air-dried.
Using a similar protocol on the opposite side of the grid completed immunogold labeling of BNP. Following pretreatment, the grids were incubated on primary antibody rabbit anti-rat BNP-32 serum in PBS/BSA at 1:500 dilution (Phoenix Pharmaceuticals, Belmont, California), rinsed and incubated with a 6-nm gold-labeled goat anti-rabbit IgG secondary antibody. Following buffer and distilled water rinses, sections were lightly stained for contrast with aqueous 2% uranyl acetate for 5 minutes and rinsed with distilled water. Some sections were utilized as controls in which the primary antibody was omitted, or normal rabbit serum was substituted for the primary antibody.
Labeled sections were examined using a Morgagni 268 transmission electron microscope, and images were collected with an AMT Advantage CCD camera system. Particle analysis of gold-labeled ANP and BNP was performed at a direct magnification of 56,000X and saved as tiff files.
Approximately, 10 sections from the LA and RA were used for image analysis, using two animals from each group. A mean of 36.8 granules/atria (N = 442) and 1121 ANP (N = 13,453) and 818 BNP (N = 9800) peptide counts/atria from the LA and RA were captured for data analysis. Image analysis was performed in iVision scientific image processing software (BioVision Technologies, Exton, Pennsylvania). Labeled particles were threshold with area parameters set to distinguish between large and small labels. Only particles within granules were counted.
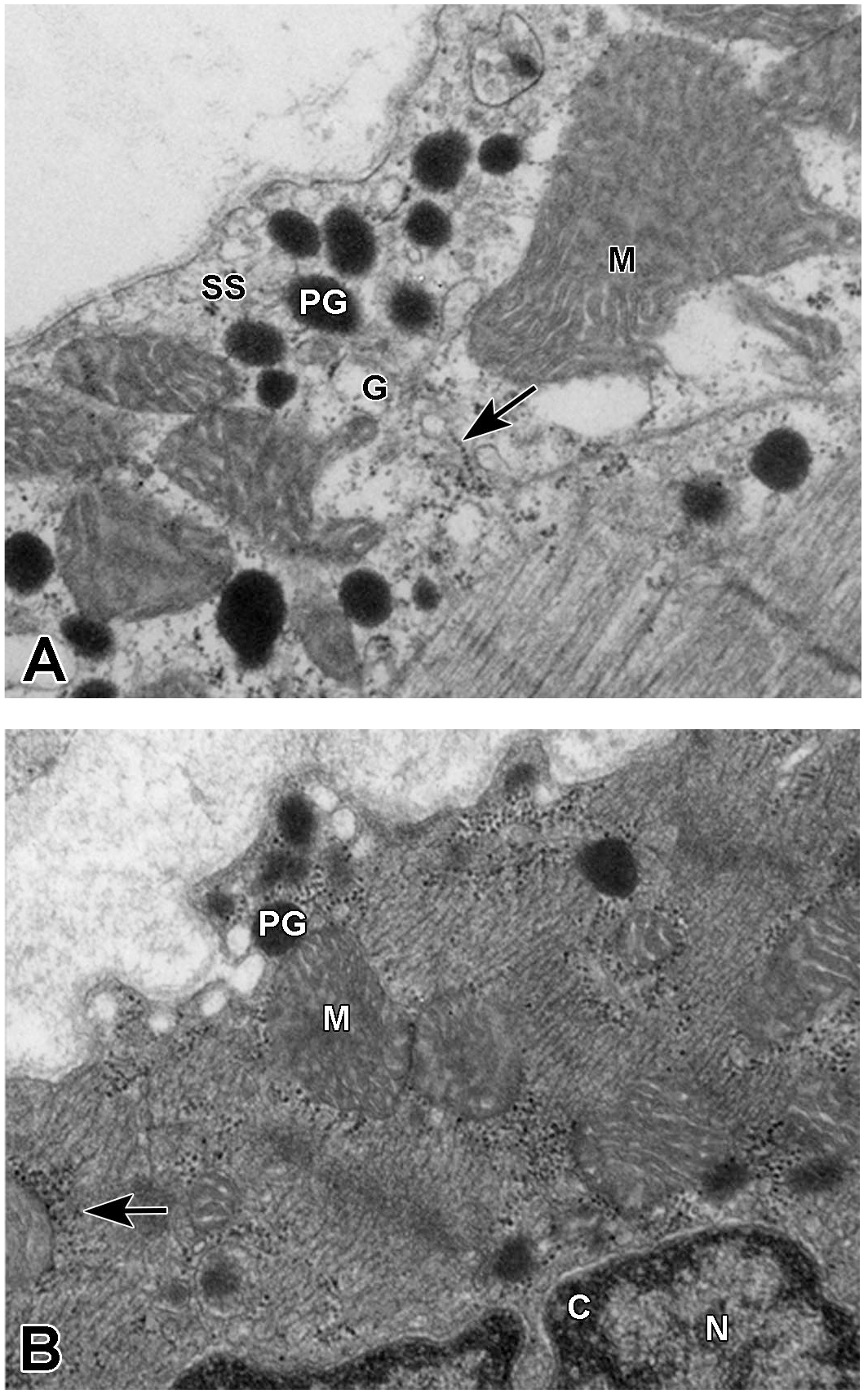
Additional atria and ventricle samples were processed using osmium tetroxide as a second fixative and Spurrs rather than LR White embedment. The double fixative samples were less sensitive for immunolabeling; however, these samples offered greater fixation of ultrastructure and were used for qualitative observation of ultrastructure (Figure 2A and 2B).
Electron micrographs from active control (A) and drug-treated (B) rats show SS regions of non-immunolabeled right atrial granules that were prepared using a double fixative and embedded in Spurrs. (A) Dense PGs and mitochondria (M) appear throughout the SS and Golgi (G) regions of an active control rat. Golgi apparatus appears to be emitting ribosomes (arrow). Direct magnification = 36,000X. (B) Densely populated ribosomes (arrow) are adjacent to mitochondria (M) and PGs of a drug-treated rat. Chromatin (C) surrounds the periphery of the nucleus (N). Direct magnification = 36,000X. PGs indicate peptide granules; SS, subsarcolemma.
Statistical Analysis
Mean, median, standard deviation, standard error of the mean, and skewness scores were all calculated using GraphPad Prism 6. Granule size frequency was determined using skewness. Skewness is an assessment of asymmetry in the data set and we used this to determine the frequency of which sized granule sizes are present for a particular intervention. We interpreted our data set similarly to what was described in the report by Picard et al. 32 A value of 0 indicates normally or randomly distributed population of small and larger granules; a value of >0 indicates increase in presence of small granules; and a value of <0 indicates increase in presence of larger granules. A one-way analysis of variance with a Tukey post hoc test was used to test for significant group differences in granule area and peptide counts/unit granule area.
Results
Ultrastructure of Atrial PGs
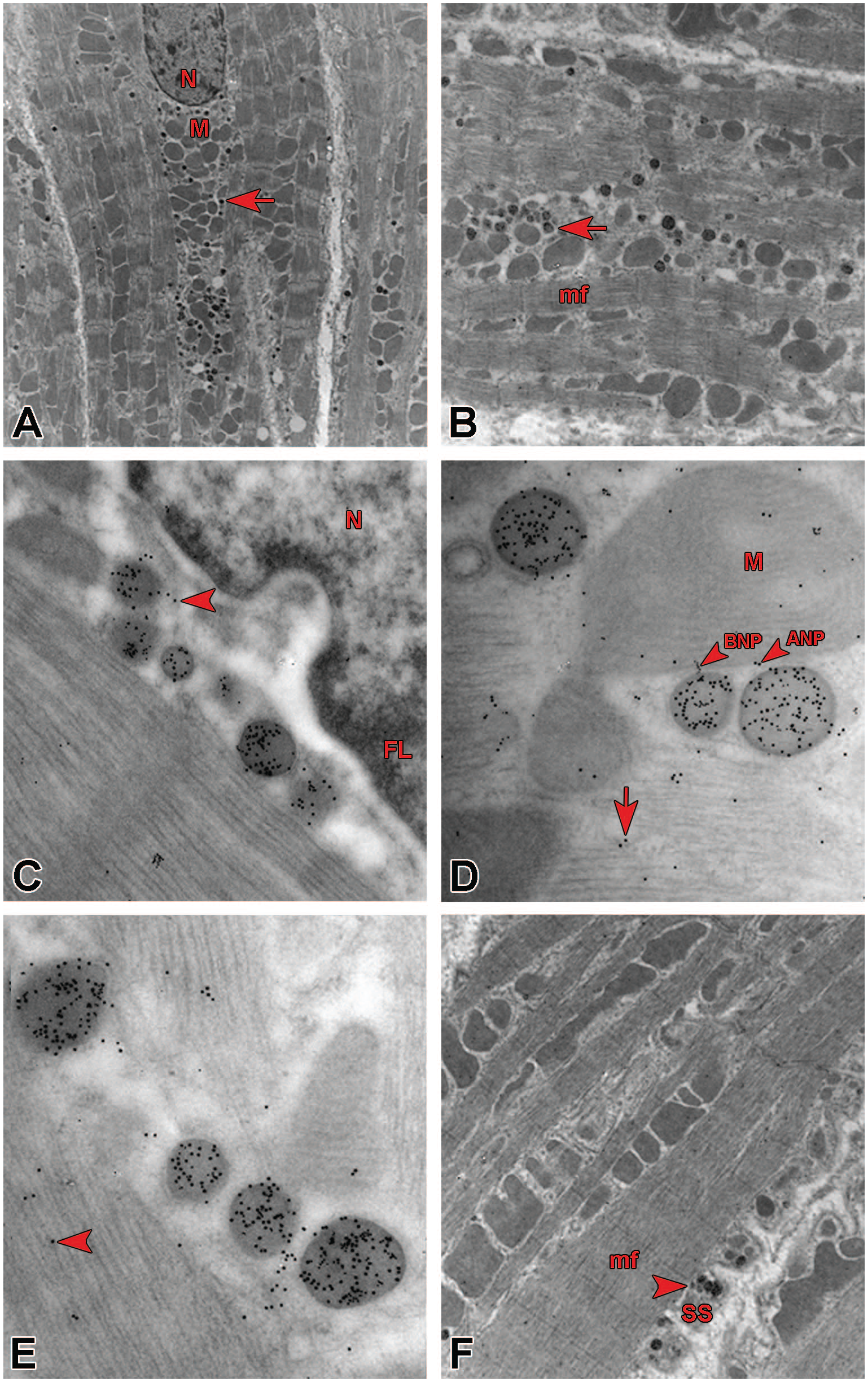
Electron microscopic qualitative examination of unlabeled SS, mf, and PN regions of Spurrs-embedded atria (Figure 2A and 2B) showed evidence of darkly stained PGs and ribosomes (arrow; Figure 2A) released from Golgi apparatus (G; Figure 2A). Mitochondria (M) and nuclei with chromatin (C) borders (Figure 2B) appeared throughout the myocytes. Electron microscopic qualitative examination of mf, PN, and SS regions of the atria showed presence of immunolabeled PGs, mitochondria, Golgi apparatus, and ribosomes in both atria (Figure 1). Evidence of heavily populated immunolabeled ANP and BNP granules (arrows) was seen in the PN (Figure 1A-1C), mf (Figure 1D and 1E), and SS (Figure 1F) regions. Double-immunolabeled 10-nm ANP and 6-nm BNP are visible in Figure 1C and 1D (arrows). At low magnification (Figure 1A, 1B, and 1F), the natriuretic PGs (arrows) appear as small round dense ultrastructures surrounded by mitochondrial (M) clusters. At higher magnifications (Figure 1C-1E), the labeled ANP and BNP are visible as distinct peptides undergoing granule transport within their respective cardiomyocyte regions. Immunolabeled ANP and BNP electron micrographs of ventricles from AC and DT rats (Figure 3A and 3B) showed evidence of free ANP and BNP throughout the ventricles, including along the sarcolemma membrane (SS; Panel C) with no evidence of natriuretic granules, consistent with previous reports. 13
(A) Electron micrograph of the left atrium nuclear region from an active control rat containing immunogold-labeled natriuretic peptide granules (arrow) within clusters of mitochondria (M) surrounding the nucleus (N). Direct magnification = 5600X. (B) Enlarged mf area from Figure 1A. Immunolabeled ANP and BNP (arrow) appear as small dense clusters within mf granules. (C) Nuclear (N) region of the left atrium from an exercise-trained rat shows transport of ANP and BNP (arrowhead). Peptides can be seen within and outside the FL border of the nucleus. The arrowhead points to a granule membrane which disappears to allow peptide transport. Direct magnification = 56,000X. (D) Right atrium of a drug-treated rat. The lower right mf area shows transport of 10-nm ANP and 6-nm BNP (arrowhead) between granules and a mitochondria (M). Some peptides appear on myofilaments (arrow). Direct magnification = 56,000X. (E) mf granule transport (arrowhead) of peptides is taking place in the mf region of the right atrium of an active control rat. Direct magnification = 56,000X. (F) Enlarged area of the SS region of the right atrium mf from the same animal from panel A. Natriuretic ANP and BNP inside granules (arrowhead) housed within SS folds presumably undergoing exocytosis appear within folded regions of the sarcolemma (arrowhead). Direct magnification = 8900X. ANP indicates atrial natriuretic peptide; BNP, brain natriuretic peptide; mf, myofibril; FL, fibrous lamina; SS, subsarcolemma.

Electron micrographs of right ventricles show immunolabeled-free natriuretic peptides. (A) mf region of right ventricle of an active control rat populated with immunolabeled free ANP and BNP (arrows). Peptides appear within mitochondria (M) and in mf and subsarcolemma regions. Direct magnification = 10,500X. (B) mf region of right ventricle of an active control rat populated with free ANP- and BNP-labeled peptides. ANP- and BNP-labeled peptides also appear to be present within the mfs (arrows). Direct magnification = 56,000X. ANP indicates atrial natriuretic peptide; BNP, brain natriuretic peptide; mf, myofibril.
Atrial ANP and BNP Granule Area and Skewness Scores
Swim training and drug treatment had no effect on the LA mean granule area when compared with AC animals (Figure 4A). The mean granule area in the RA of the PPARγ agonist–treated animals (DT) was significantly greater compared with S animals (Figure 4B). Skewness scores function to compare granule size symmetry and shifts in size distributions across groups and across atria (Table 1). LA and RA skewness scores for AC animals were similar and highly skewed toward a greater percentage of smaller size granules. In contrast to the LA, the RA of DT rats reflected a fairly symmetrical distribution of PGs compared with S and AC animals. RA skewness scores of swim-trained rats, however, further elevated the asymmetry of granule distributions compared with AC and DT animals. The contrasting effects of exercise and drug treatments in both atria suggest unique mechanisms controlling PN granule size and peptide synthesis.
Peptide granule area (nm2) skewness score.
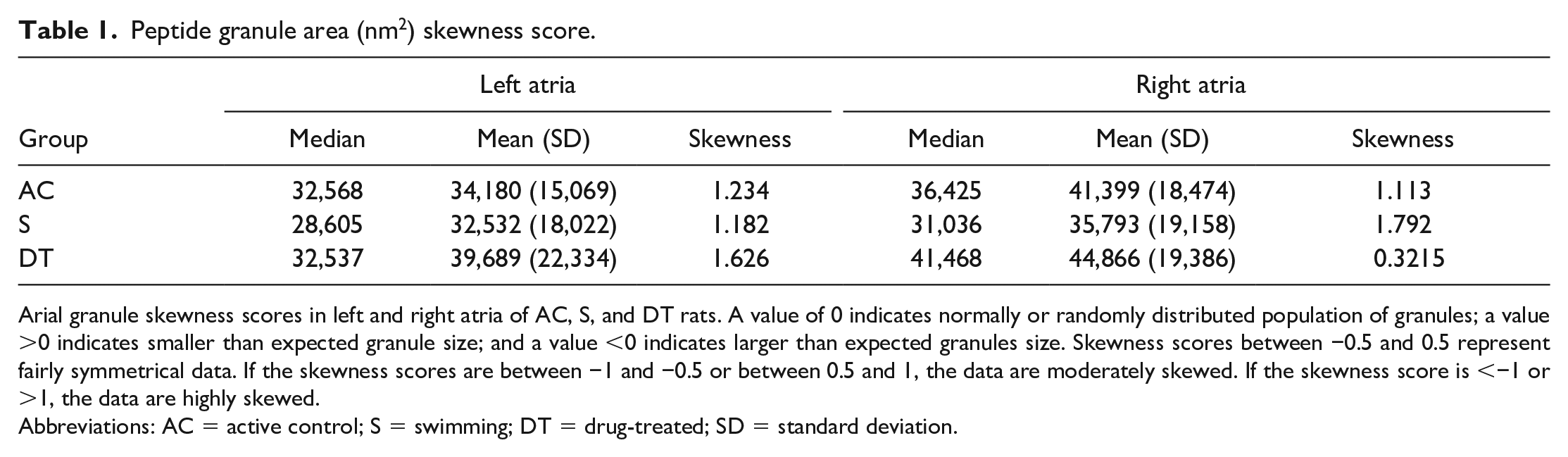
Arial granule skewness scores in left and right atria of AC, S, and DT rats. A value of 0 indicates normally or randomly distributed population of granules; a value >0 indicates smaller than expected granule size; and a value <0 indicates larger than expected granules size. Skewness scores between −0.5 and 0.5 represent fairly symmetrical data. If the skewness scores are between −1 and −0.5 or between 0.5 and 1, the data are moderately skewed. If the skewness score is <−1 or >1, the data are highly skewed.
Abbreviations: AC = active control; S = swimming; DT = drug-treated; SD = standard deviation.
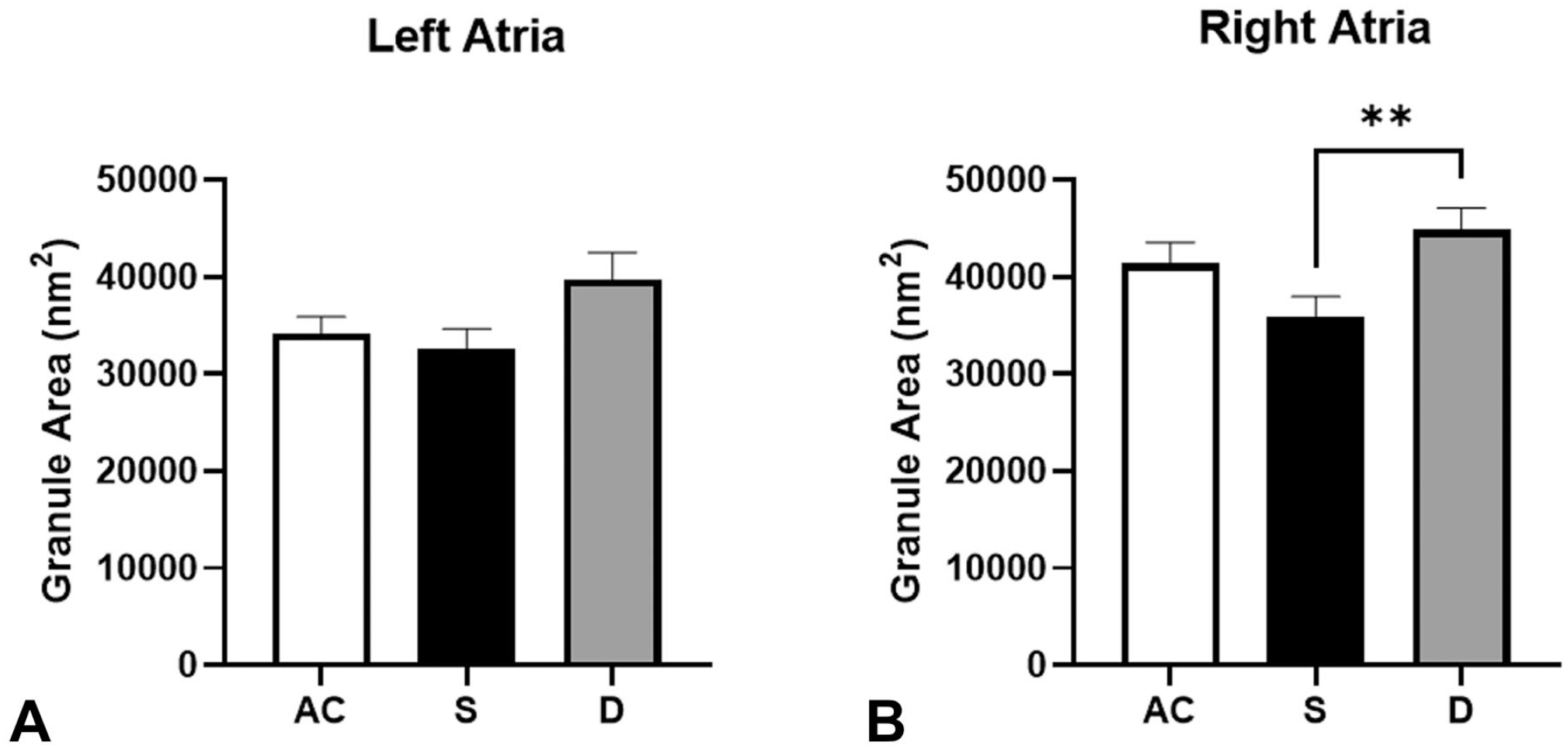
Mean granule surface area of (A) left atria and (B) right atria from AC (79 granules measure in left atria and 74 granules measure in right atria), S (72 granules measure in left atria and 78 granules measure in right atria), and DT (63 granules measure in left atria and 75 granules measure in right atria) animals. Values are represented as means ± SEM. Pairwise comparisons were determined using one-way ANOVA with Tukey post hoc test. ANOVA indicates analysis of variance; SEM, standard error of the mean; AC, active control; S, swimming; DT, drug-treated.
Atrial PG Density
Rats administered the PPARγ agonist reported a lowering of ANP granule density in both atria (Figure 5A and 5C) compared with AC rats and a lowering effect on ANP density in the LA compared with S group rats (Figure 5A). A significantly lower ANP density was observed in the RA in swim-trained rats relative to the AC and DT animals (Figure 5C). Swim training and drug treatment had no effect on LA BNP density compared with ACs (Figure 5B). In contrast, swim training lowered BNP granule densities in the RA compared with DT and AC rats (Figure 5D).
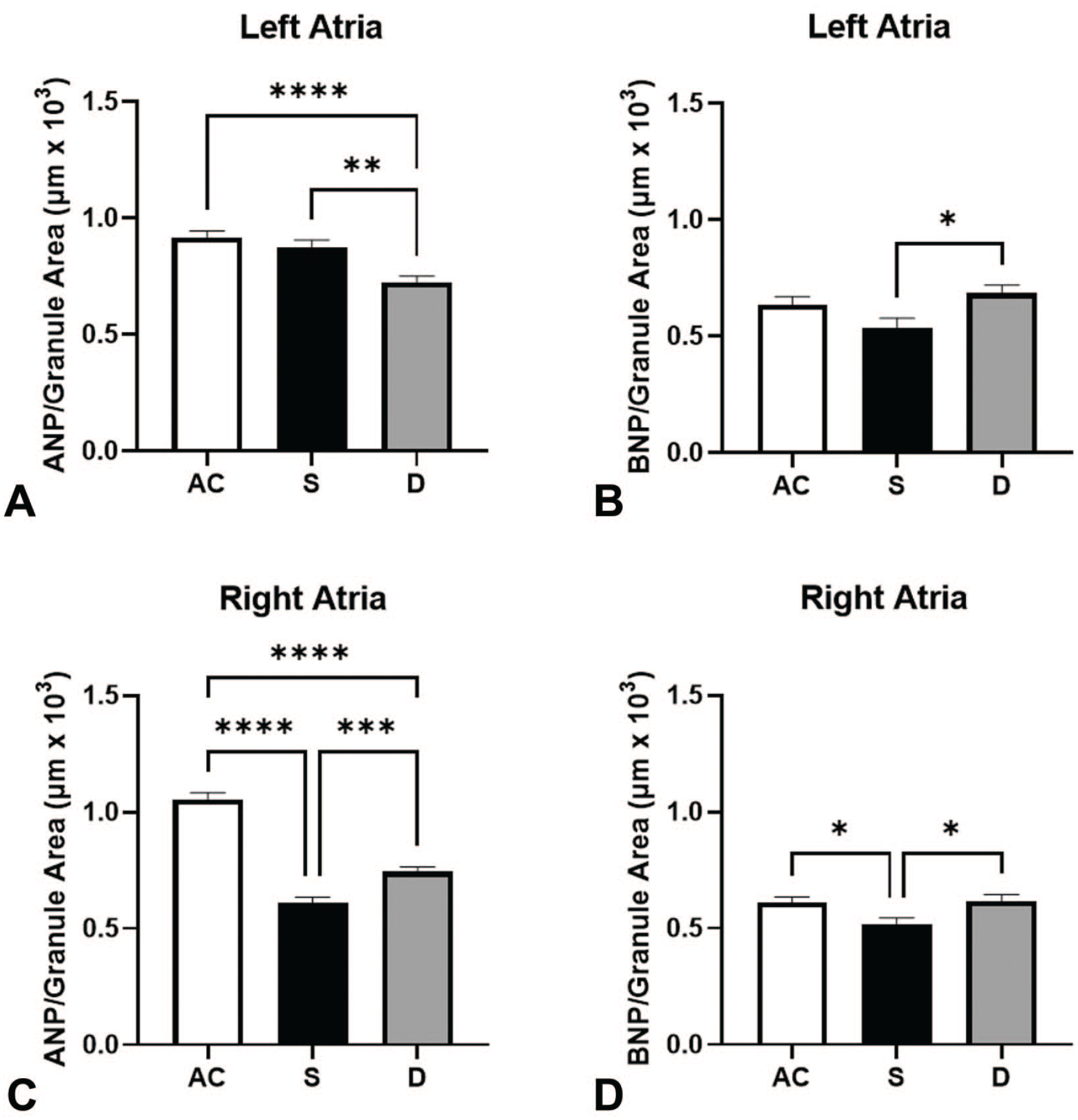
The ANP and BNP density per unit area in (A and B) left atria and (C and D) right atria. AC (79 granules measure in left atria and 74 granules measure in right atria), S (72 granules measure in left atria and 78 granules measure in right atria), and DT (63 granules measure in left atria and 75 granules measure in right atria) animals. Values are represented as means ± SEM. Pairwise comparisons were determined using one-way ANOVA with Tukey post hoc test. ANOVA indicates analysis of variance; ANP, atrial natriuretic peptide; BNP, brain natriuretic peptide; SEM, standard error of the mean; AC, active control; S, swimming; DT, drug-treated.
Atrial PG Density Peptide Ratios
ANP:BNP granule density ratios (Figure 6) were computed from the ANP and BNP granule density values shown in Figure 5. When comparing the within-group ANP:BNP granule density ratios (Figure 6) in the atria of AC rats (LAANP:BNP = 1.436; RAANP:BNP = 1.304) and DT rats (LAANP:BNP = 0.996; RAANP:BNP = 0.994), the respective chamber ratios of ANP:BNP densities were lower in both atria of the DT rats. Consequently, the ANP:BNP granule density ratio in RA of AC rats was greater than the S and DT rats (Figure 6—middle column; 1.304 > 0.994 and 1.022). Comparing the drug and exercise treatment effects on ANP:BNP ratios in the LA versus the RA chambers, the LA:RAANP:BNP ratio recorded from the swim-trained rats was greater than the DT rats, due in part to the interactive effects of PPARγ agonist and swim exercise on left and right atrial chambers (Figure 6).
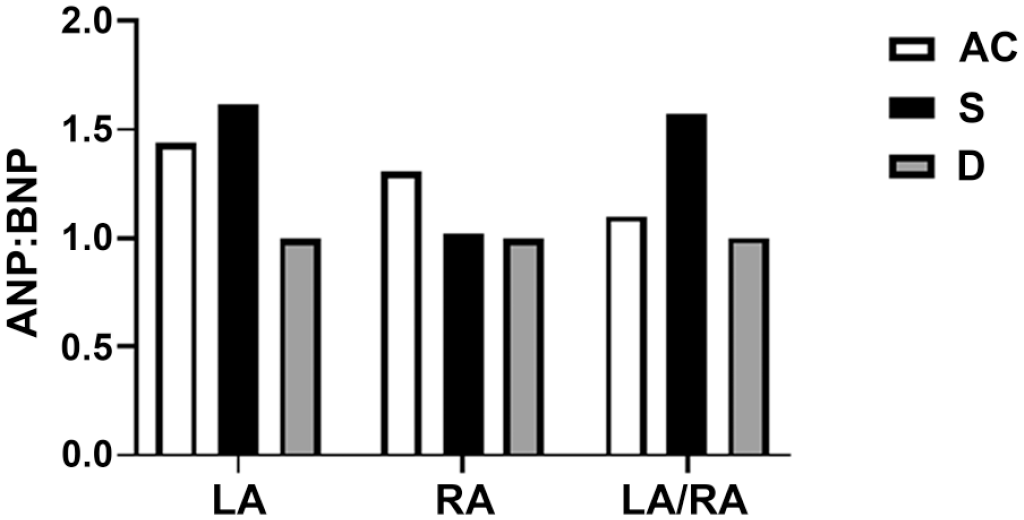
Swim and drug effects on perinuclear ANP:BNP granular density ratios in the LA and RA. The right column calculates the ANP:BNP granular peptide ratios as a relationship between the LA and the RA (LA:RAANP:BNP). Values are represented as means. Due to the low number of observations (N = 2 rats/group), SEM was not calculated and statistical analysis could not be performed. ANP indicates atrial natriuretic peptide; BNP, brain natriuretic peptide; SEM, standard error of the mean; LA, left atrium; RA, right atrium.
Discussion
The main objective of this study was to examine the effects of PPARγ agonism and exercise training on the populations of ANP and BNP within atrial secretory PGs to discriminate between physiological and pathological hypertrophy. The absence of granules in the ventricles of healthy animals is consistent with previous reports13; however, the presence of secretory granules has been reported in ventricular myocytes of spontaneously hypertensive rats 33 and patients with dilated cardiomyopathy. 10 The lack of secretory granules in the ventricles of the DT rats suggests that the administration of a PPARγ agonist for 28 days did not cause atrial or diastolic dysfunction significant enough to be considered cardiomyopathic. This finding is consistent with previous reports of rats administered a PPARα/γ dual agonist for up to 28 days in which heart weights were increased by as much as 75% while cardiac output continued to increase. 20
The unique effects of exercise and PPARγ agonist on the size and peptide populations within granules are a central finding of our study. During cardiac stress induced by drug treatment, we observed a decrease in ANP density and an increase in BNP density within the left atrial granules associated with a noticeable shift in LA granule size distribution toward smaller sized PN granules, compared with swim-trained rats. Larger granules caused by granule-to-granule fusion reveal older granules,23,34 in contrast to smaller size granules in rats with experimentally induced congestive heart failure (CHF), that spend shorter durations with less time to fuse prior to exocytosis.23,34,35 The smaller granules in CHF rats imply CHF accelerated peptide synthesis and release 23 and sodium retention. 24 The moderately greater shift toward smaller size granules in the LA of our DT rats was predictive of enhanced peptide synthesis, delivery, and exocytosis as evidenced by significant increases in serum concentrations of natriuretic peptides in PPARγ agonist–treated rats relative to AC and swim-trained rats. 1 In contrast to reduced mean granule surface area in the LA and RA of CHF rats induced by artrio-venous (A-V) fistula, 23 PPARγ agonist treatment moderately increased mean granule surface area in the RA compared with ACs and had no statistical effect on mean granule surface area in the LA. Most notably, the PPARγ agonist treatment effect resulting in a fairly symmetrical distribution of granule surface area in the RA, compared with an asymmetrical size distribution of granules in the LA, suggests that swim and PPARγ agonist treatment may have contrasting yet synergist effects on the left and right atrial granule size distributions in response to their distinctive atrial stretch responses to treatment, explaining in part the discrete temporal differences in circulating NT-proANP and NT-proBNP observed previously. 1
Atrial granule ANP:BNP density ratio is reflective of species, heart chamber anatomy, 36 and atrial stretch response to cardiac pacing protocol37-39 offering insight into the progression of pathological and physiological effects on cardiac morphology.24,40 Compensated CHF induced by A-V fistula reported no effect on RAANP:BNP ratio in sham and compensated animals in contrast to the lowering effect on ANP:BNP ratio in decompensated rats, implying that ANP and BNP operate through common mechanisms in response to atrial stretch, 24 yet RAANP:BNP density ratio discriminates compensated and decompensated heart failure. Our PPARγ agonist-treated and swim-trained rats lowered the ANP:BNP ratio in the RA compared with AC animals. In the LA, drug treatment had a comparable ANP:BNP ratio lowering effect and swim exercise had an elevating effect on the ANP:BNP ratio. The contrasting treatment effects on atria peptide ratios are reflected in the LA:RA/ANP:BNP ratio. The LA:RA/ANP:BNP ratio in our swim-trained rats was significantly greater than the PPARγ agonist–treated rats, implying that exercise may provide a protective effect by enhancing the ANP:BNP ratio in the LA.
A second objective of our study was to evaluate effects of 28 days of swim exercise on alterations in atrial granule peptide population, corresponding with cardiac hypertrophy. A potential mechanism to explain cardiac pressure and volume overload leading to decompensated hypertrophy includes the inability of peptide expression and granule release to keep pace with increasing load.1,2 In contrast, aerobic exercise in mice augments PN granule production, natriuretic peptide expression, and secretory activity in the RA. 22 The differential changes in PG mean surface area and granule size distribution likely reflect the physiological differences between exercise and PPARγ agonism–induced cardiac hypertrophy, demonstrating that the effects of exercise, at least after four weeks, stimulate more rapid turnover of natriuretic PGs in the RA, while PPARγ agonism affects both LA and RA granule characteristics. The lack of evidence of myocardial and collagen degeneration in swim-trained rats that exhibited 15% cardiac hypertrophy 1 is suggestive of an exercise-inductive protective effect reflective in the peptide and granule morphology of the swim-trained rats. Skewness scores of the swim animals had opposite atrial shifts relative to the DT animals. Prolonged exercise training on the functional adaptation of the LA and RA primarily affects the RA, reflective of increased venous return, suggesting that a longer period of training may have led to physiologic effects on the LA as well. 21 An exercise-induced increase in circulating ANP appears to be transient, resolving shortly after the cessation of exercise and does not result in a chronically increased circulating concentration of natriuretic peptides, consistent with our findings in swim-trained rats.41,42 Evidence of increased natriuretic peptide secretion in swim-trained rats without measurable increases in circulating concentrations may appear to offer conflicting results, but could be explained by the timing of blood sampling or the sensitivity of the assay used. 1
Our results support the premise that atrial ANP and BNP granule density and size distributions are potential biomarkers of physiological and pathological hypertrophy, providing granule peptide density changes characteristic of augmented natriuretic peptide secretion and exocytosis effects in swim-trained rats, without measurable increases in circulating concentrations. 1
This study addressed the need to identify the characteristics of atrial ANP and BNP release and clearance in the setting of physiological or pathological hypertrophy.1,2,20 PPARγ agonist compound delivery or adaptive swim training, both resulting in 15% cardiac hypertrophy, was identified by distinct treatment effects on PN granule morphology and peptide densities within and across LA and RA. It appears that ANPs and ANP:BNP ratios work in synergy across atria, playing a mechanistic role that characterize atrial granule peptide population, and provide novel insight into the unique effects of physiologic and pathologic hypertrophy, reflective in part by serum NT-proANP and NT-proBNP biomarkers.1,2,20,43 Unfortunately, our study was limited in that we were not able to measure cardiac function. Accordingly, progress toward testing natriuretic prohormones as biomarkers for compound utility aimed at sustaining cardiac output in inactive and active patients with CHF can be applied in translational and clinical settings.44-48
Footnotes
Acknowledgements
We would like to thank Dr Michael E. Dunn for his initial contribution showing the clinical utility of NT-proANP as a biomarker of pathological hypertrophy that sets the foundation for the present article. We would like to also thank Dr James Agostinucci for his assistance in formatting the electron microscopy images for this article. Special thanks to Carol Ayala for her three-year commitment in respect to all technical support regarding transmission electron microscopy, in particular the immunohistochemistry labeling of atrial and ventricle natriuretic peptides. The authors acknowledge Reilly Loomis for his valuable input regarding the immunohistochemistry section of the article. Special appreciation is given to URI professor Angela Slitt, PhD, and her team of graduate students. We acknowledge Denise Vaz, Laura Huntley, and Lelah Sciarra for their technical assistance and expert training of the University of Rhode Island students regarding animal care. We acknowledge the following University of Rhode Island students who successfully performed the swim training and drug delivery procedures throughout the three-month pilot and research studies: Kaitlin Abbate, Dana Demers, Ryan Johnson, Angela Phongsavan, Stephen Sladen, Erik Simpanen, Christopher DeSantos, and Cara Finan.
Author Contribution
KA, TGM, and ACC contributed to the conception and design of the experiments and drafting the manuscript. KA, TGM, ACC, FJV, and SKE contributed to the data collection, analysis, and interpretation. All authors (KA, TGM ACC, FJV, and SKE) critically revised the manuscript and gave the final approval. All authors agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Predictive Safety Testing Consortium of the Critical Path Institute, FDA Grant (U01 FD003865-05).
