Abstract
Following implantation of patient-derived xenograft (PDX) breast carcinomas from three separate individuals, 33/51 female NOD.Cg-Prkdc scid Il2rg tm1Wjl /SzJ (NSG) mice presented with progressive, unilateral to bilateral, ascending hindlimb paresis to paralysis. Mice were mildly dehydrated, in thin to poor body condition, with reduced to absent hindlimb withdrawal reflex and deep pain sensation. Microscopically, there was variable axonal swelling, vacuolation, and dilation of myelin sheaths within the ventral spinal cord and spinal nerve roots of the thoracolumbar and sacral spinal cord, as well as within corresponding sciatic nerves. Results of PCR screening of PDX samples obtained at necropsy and pooled environmental swabs from the racks housing affected animals were positive for lactate dehydrogenase–elevating virus (LDV). LDV is transmitted through animal-animal contact or commonly as a contaminant of biologic materials of mouse origin. Infection is associated with progressive degenerative myelopathy and neuropathy in strains of mice harboring endogenous retrovirus (AKR, C58), or in immunosuppressed strains (NOD-SCID, Foxn1nu), and can interfere with normal immune responses and alter engraftment and growth of xenograft tumors in immunosuppressed mice. This is the first reported series of LDV-induced poliomyelitis in NSG mice and should be recognized as a potentially significant confounder to biomedical studies utilizing immunodeficient xenograft models.
Keywords
Introduction
Lactate dehydrogenase–elevating virus (LDV) is an enveloped single-stranded RNA virus that belongs to the family Arteriviridae. Other members of this family include equine infectious arteritis virus (EIA), porcine reproductive and respiratory syndrome virus (PRRSV), and simian hemorrhagic fever virus (SHFV). 1 LDV has become recognized as an important pathogen in laboratory mice as a contaminant of biologic materials of mouse origin, including culture reagents, xenograft tumor implants, cell cultures, and basement membrane matrix; however, animal-animal contact, generally through fighting, is also an important mode of transmission.2,3 In most immunocompetent mouse strains, infection results in lifelong viremia and the disease is asymptomatic, but causes acutely elevated serum lactate dehydrogenase (LDH) levels shortly after infection.2,4,5 Following exposure, viral replication occurs in a subpopulation of F4/80-positive macrophages that are responsible for clearing LDH from the circulation. 6 The cytopathic effect of LDV viral replication in these cells results in loss of LDH clearance from the circulation and clinically elevated serum LDH levels.
Based on tissue tropism and clinical signs, LDV is categorized into non-neuropathic (LDV-P) and neuropathic strains (LDV-V and LDV-vx). The non-neuropathic strain is present in wild mouse populations at a prevalence rate of 1% to 2% in North and Central America 7 and elicits a robust anti-LDV antibody response following infection. While most mice infected with LDV are asymptomatic, certain strains of mice (C58, AKR, C3H/Fg, PL/J) are susceptible to LDV-induced poliomyelitis due to infection with neuropathic LDV.4,8,9 Neuropathic strains of LDV cause age-related poliomyelitis under specific circumstances requiring a combination of immunosuppression, strain genetics, and presence of murine leukemia virus (MuLV). In the C58 and AKR strains for example, homozygosity for the permissive allele for N-tropic viral replication (Fv-1 n/n) allows for replication of endogenous ecotropic MuLV in glial cells of the spinal cord, which through an unknown mechanism renders anterior horn neurons susceptible to entry and cytolytic infection with neuropathic strains of LDV. 6
Altered immune function plays an important role in development of poliomyelitis, whether experimentally induced or as a result of host genetics. Infection with neuropathic LDV has been reported to cause paralytic disease in immunocompromised mice, including NOD-SCID and nude (FoxN1) mice, due to severe defects in cell-mediated immune function and lack of an antibody response to neuropathic LDV.6,10 Immunocompromised mice are an important tool in carcinogenesis studies utilizing implantation of cell-based or patient-derived xenografts (PDX), and contamination of cell culture materials or xenograft samples with LDV represents an important cause of confounding and experimental failure, which can result in substantial loss of animals, valuable biologic samples, and research data. Therefore, recognition of LDV as an important cause of paresis and paralysis in immunocompromised mice is critical in maintaining and safeguarding high-quality research results and animal welfare. In the following, we document the first reported series of degenerative myelopathy and neuropathy in NOD.Cg-Prkdc scid Il2rgtm1Wjl/SzJ (NSG) mice due to infection with neuropathic LDV resulting from transplantation of PDX.
Materials and Methods
Husbandry and Animal Care
Animals evaluated for this study were part of a colony used for breast cancer research at the University of Michigan. Cohorts of 6- to 8-week-old female NOD.Cg-Prkdc scid Il2rg tm1Wjl /SzJ (NSG, Strain #:005557) mice were obtained from The Jackson Laboratory (Bar Harbor, ME) prior to the initiation of each experiment. Mice were housed in autoclaved ventilated cages (Allentown Caging, Allentown, NJ) with autoclaved Pure-o’Cel paper square bedding (The Andersons, Maumee, OH) on a 12:12-h photocycle and were offered free access to irradiated rodent chow (LabDiet 5LOD; PMI Nutrition International, Brentwood, MO) and acidified deionized water. The mice were housed in temperature- and humidity-controlled rooms in compliance with the recommendations of the Guide for the Care and Use of Laboratory Animals. The health of the colony was evaluated quarterly by using dirty-bedding transfer to sentinel mice and environmental testing. All excluded flora Swiss Webster sentinels (Taconic Biosciences, Rensselaer, NY) were negative on both serology and PCR testing for the following pathogens for the study duration: mouse hepatitis virus (MHV), mouse parvovirus (MPV), minute virus of mice (MVM), epizootic diarrhea of infant mice, and ectromelia virus. In addition, excluded flora Swiss Webster sentinels were negative by serology screening for Sendai virus, pneumonia virus of mice (PVM), Theiler’s murine encephalomyelitis virus (TMEV), reovirus type 3, lymphocytic choriomeningitis virus (LCMV), mouse adenovirus, polyoma virus, and Mycoplasma pulmonis for the duration of the study. Finally, PCR screening for Corynebacterium bovis (C. bovis) and murine pinworms (Aspiculuris and Syphacia) was also negative for the study duration, and environmental PCR was negative for fur mites (Myobia, Mycoptes, Radfordia spp.).
Patient-derived human breast carcinoma samples were obtained from three different female individuals (PDX A, B, and C). The tumor specimens were disinfected with 70% ethanol then stored in a solution with 90% fetal bovine serum (FBS) and 10% DMSO (dimethylsulfoxide) in liquid nitrogen. PDX tumors were not subcultured or passaged in vitro, and basement membrane matrix was not used. Prior to implantation, PDX samples were rinsed with 20% FBS and phosphate-buffered saline (PBS) and were then surgically implanted into the subcutaneous space at the level of right 4th mammary fat pad of three cohorts of recipient mice. Growth of PDX tumors was monitored for each set of experiments and serially passaged in additional recipient mice to maintain each tumor line. Once each PDX was able to be stably passaged successfully through mice, they were surgically xenografted into recipient mice under anesthesia using aseptic technique; PDX A was transplanted into a group of 43 mice (passage 4), PDX B into a group of three mice (passage 3), and PDX C into a group of five mice (passage 5). All mouse experiments were performed in accordance with protocols approved by the University of Michigan Institutional Animal Care and Use Committee.
Necropsy and Histopathology
Clinically affected animals were humanely euthanized with CO2 asphyxiation. Routine diagnostic necropsy was performed on sixteen affected animals (ten from PDX A, one from PDX B, and three from PDX C groups), with collection of a standard set of tissues for fixation, including multiple sections of cervical, thoracic, lumbar, and sacral vertebral segments containing spinal cord, and fresh xenograft tumor tissue was collected for PCR testing from nine animals (six from group PDX A, one from PDX B, and two from PDX C). Tissues for histology were immersion-fixed in 10% neutral-buffered formalin (NBF) for 24 hours. Following fixation, vertebral segments were decalcified in Immunocal (StatLab, McKinney, TX) overnight prior to trimming. All tissues were routinely trimmed, cassetted, and processed to paraffin blocks, and 4 μm sections prepared and stained with hematoxylin and eosin for microscopic examination.
A PCR rodent infectious agent (PRIA) screen (Charles River Laboratories, Wilmington MA) was performed on fecal pellets (n = 7 from cages housing affected mice) for pathogens including LCMV, Hantavirus Hantaan (HANT), mouse adenovirus (MAV1 and 2), MHV, ectromelia virus (mousepox), MPVs, MVM, mouse rotavirus, murine polyomavirus, PVM, reovirus, Sendai virus, TMEV, Mycoplasma pulmonis, C. bovis, fur mites, and pinworms (Aspiculuris and Syphacia). Environmental screening for LDV was also performed by PCR on pooled exhaust plenum swabs from the cage rack housing clinically symptomatic animals, and from the cage rack housing asymptomatic animals that were in the same animal room. LDV PCR was also performed on the current lot of FBS used for tumor passage. While not specific for neuropathic strains, PCR testing detects both variants of LDV. Finally, fresh tissue samples of each of the aforementioned xenograft tumors collected from the three PDX groups (n = 9) were submitted for LDV PCR testing.
Results
Thirty-three out of 51 total animals that were previously xenografted with one of the three different human breast carcinoma tumors developed neurologic symptoms approximately 3 to 4 weeks post-implantation. Thirty of 43 animals (70%) receiving PDX A, one of three animals (33%) receiving PDX B, and two of five animals transplanted with PDX C (40%) developed clinical disease characterized by progressive unilateral to bilateral hindlimb paresis and paralysis (Figure 1), with moderate muscle atrophy and overall poor body condition. In all sixteen clinically affected animals collected for necropsy examination, histologically there was variable vacuolation, swelling of axons (spheroids), and dilation of myelin sheaths predominantly affecting the white matter of the ventral to ventrolateral portions of the spinal cord (Figure 2A), preferentially within the thoracolumbar and sacral cord segments. Lesions extended unilaterally or bilaterally into the adjacent spinal nerve roots and spinal nerves in some sections (Figure 2B-D), and distally within the associated peripheral (sciatic) nerves (Figure 2E). In more severe cases, there was vacuolation in the adjacent gray matter of the spinal cord, and lesions extended proximally into the cervicothoracic region and distally within sacral segments. In associated skeletal muscle of the affected hindlimb(s), there was minimal to mild multifocal myofiber atrophy, characterized by decreased individual myofiber size, alterations in myofiber tincture, and occasional crowding, rowing, and centralization of myofiber nuclei (Figure 2F).
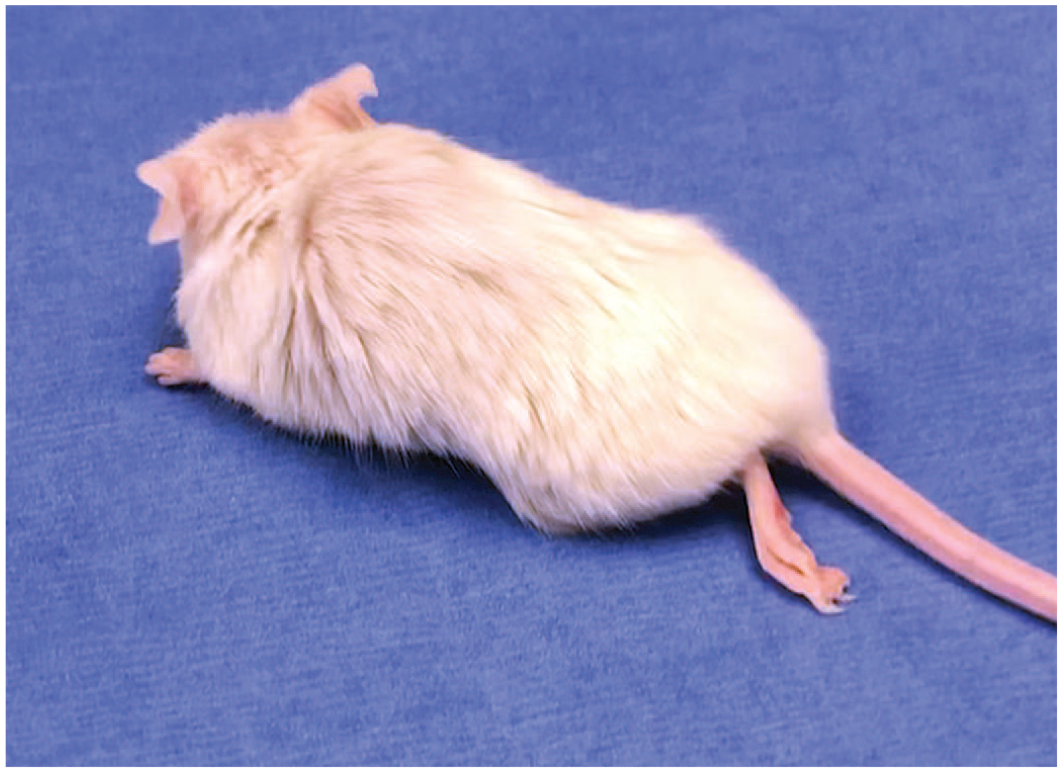
Unilateral hindlimb paresis/paralysis, NSG mouse. Affected mouse with unkempt appearance, roughened haircoat, and unilateral hindlimb paresis/paralysis evidenced by extension of the left hindlimb.
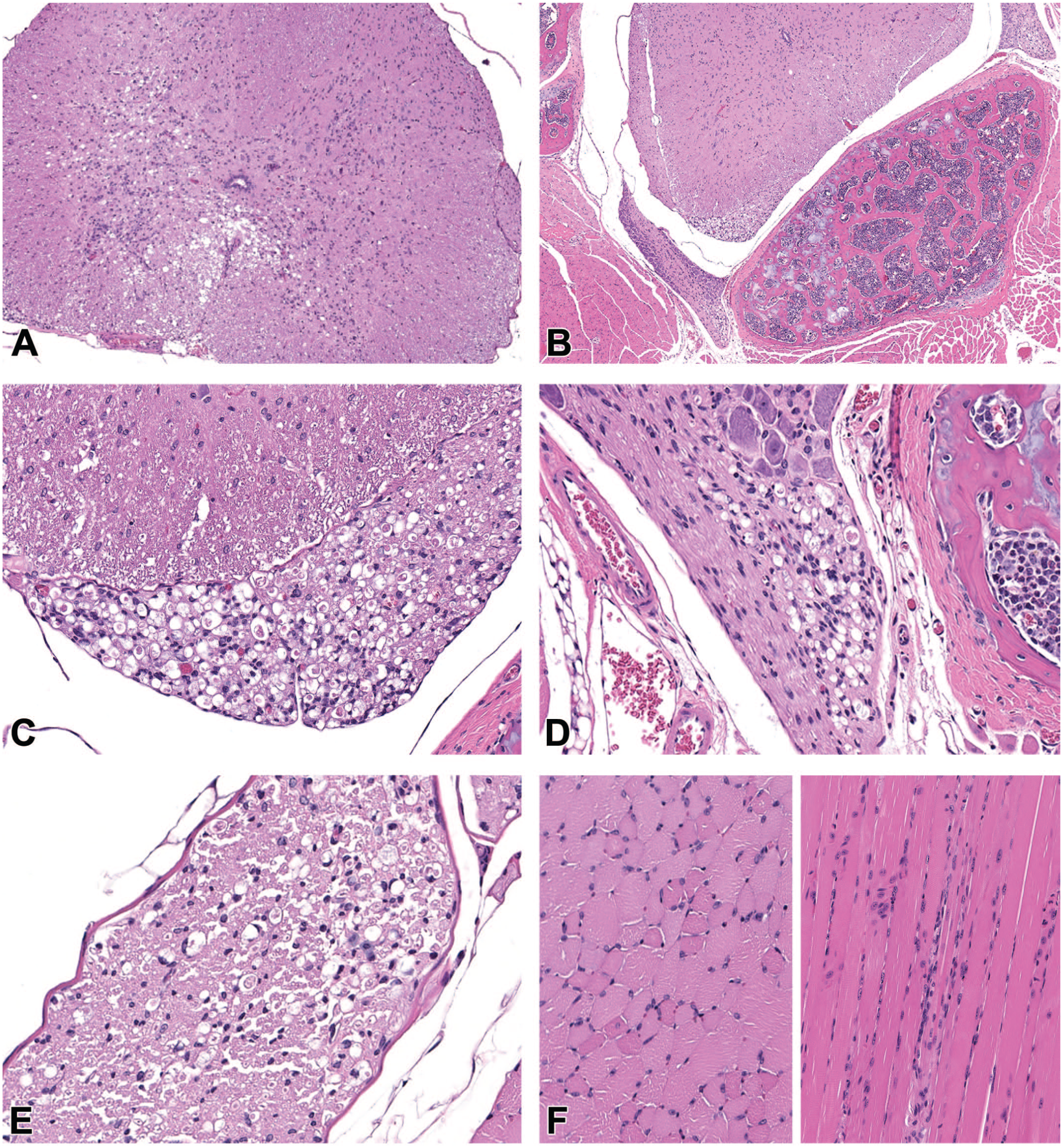
Degenerative myelopathy, thoracolumbar spinal cord, NSG mouse. (A) There is locally extensive vacuolation present ventrally and laterally within the white matter of the spinal cord. H&E, 10X. (B) In other sections, there is vacuolation extending within the ventral spinal nerve root (arrow) and spinal nerve (arrowhead). H&E, 4X. (C) Higher magnification of the lesion in the spinal nerve root in Figure 2B, showing dilation of myelin sheaths, vacuolation, and axonal swelling (spheroids, arrowhead) in the ventral nerve root. H&E, 20X. (D) Higher magnification of lesion at arrowhead in Figure 2B, demonstrating vacuolation, spheroid formation, and dilation of myelin sheaths within the ventral spinal nerve. H&E, 20X. (E) Dilation of myelin sheaths and spheroid formation in the associated sciatic nerve. H&E, 20X. (F) Minimal to mild scattered skeletal muscle myofiber atrophy of the affected hindlimb (left cross-section, right longitudinal), characterized by shrinkage of individual myofibers, increased eosinophilia, and crowding and rowing (arrows) and rare centralization (arrowhead) of myofiber nuclei. H&E, 20X.
Each PDX tumor sample (n = 9) collected from clinically affected animals at the time of necropsy confirmed the presence of LDV by PCR. No other pathogens were detected from fecal pellets on the submitted PRIA screen. In addition, environmental testing of the affected room confirmed the presence of LDV from pooled plenum swabs of cage racks housing clinically affected animals, whereas swabs of cage racks housing clinically unaffected animals within the same room as well as other areas of the same facility were negative for LDV by PCR. FBS samples were also negative for LDV by PCR.
Discussion
Infection of susceptible strains of mice with neuropathic LDV causes a progressive, ascending paralysis as a result of axonal degeneration in the motor neuron tracts of the thoracolumbar spinal cord. In severe cases, lesions may be seen throughout the ventral white matter, as well as within the more proximal thoracic and even cervical spinal cord. As with other viral central nervous system (CNS) diseases, the severity and distribution of disease following neuropathic LDV infection varies depending on host strain, genotype, and immune status. In immunocompetent animals susceptible to LDV infection, a mild inflammatory response may accompany neurodegenerative changes, whereas in immunodeficient animals, inflammatory lesions are absent. 6 For this reason, the disease in this case series of NSG mice manifested as a degenerative myelopathy and peripheral neuropathy, rather than a poliomyelitis with an inflammatory component.
Other viral causes of hindlimb paralysis in laboratory mice include MHV (neuropathic coronavirus), TMEV (cardiovirus), reovirus, polyomavirus, and LCMV (arenavirus). Immunodeficient strains are also very susceptible to these pathogens, particularly polytropic MHV, for which necrotizing encephalomyelitis with demyelination follows infection with neurovirulent strains. 11 TMEV causes neuronal necrosis, inflammation, and demyelination in the ventral horn of the spinal cord and various portions of the brain secondary to the immune response to the virus, but natural infection is rare and disease is generally only reproducible through experimental intracranial injection. 12 In neonates, reovirus infection may cause paralysis secondary to encephalomyelitis, diarrhea, and runting; adult mice are resistant to natural infection, but develop flaccid paralysis with experimental infection due to necrosis of ventral motor neurons. 13
The immunophenotype of immunocompromised mouse strains that are commonly used in cancer research, NOD-SCID FoxN1 nu/nu (nude), NSG, makes them extremely susceptible to infection with neuropathic LDV. The effects of LDV on serum LDH were first observed by Riley et al. 14 in a variety of stocks and strains used for passage of mouse cell lines and tumors, including ICR, C57BL/6, and AKR mice. It was later found that age-related poliomyelitis induced by LDV, generally occurring in older susceptible animals, could be induced in young animals after treatment with immunosuppressive drugs or whole-body irradiation. 15 Finally, the first recognition of LDV-induced poliomyelitis in immunosuppressed mice was observed in MuLV-infected ICR-SCID mice injected with LDV-contaminated basement membrane matrix. 6 Immunodeficient strains are used commonly in cancer research and given the popularity and widespread use of these models in biomedical and translational research, the effects of LDV infection can be devastating to researchers. It is critical to recognize the clinical and pathologic manifestations of LDV infection and understand how this disease may confound tumor xenograft studies and result in loss of time, resources, animals, and critical research data. The paralytic effects of the virus, for example, should not be confused with manifestations of metastatic disease or paraneoplastic syndrome; this is particularly critical when studying xenograft tumors that under natural biologic conditions metastasize commonly to the vertebral column (eg, metastatic prostate or breast cancers). 16 In addition, LDV infection may affect successful engraftment, normal growth, biologic behavior, and response to therapy of implanted tumors.17,18
In immunocompetent strains, infection with LDV can modify the host immune response. Immunomodulatory effects of LDV infection include nonspecific polyclonal activation of B cells, immunoglobulin secretion, acute lymphopenia, chronic lymphocytosis, and abnormal interferon and natural killer (NK) cell responses.2-4,6,19,20 Reported impacts of LDV infection on other mouse models include increased susceptibility to LPS-induced septic shock,21,22 suppression of immune-mediated islet cell injury in non-obese diabetic (NOD) mice by interfering with function of 1a-positive macrophages, 23 and antibody-mediated thrombocytopenia, 24 among others.
While LDV has the ability to evade the host immune system, causing lifelong viremia in infected mice, rapid elevation in anti-LDV antibodies is typically achieved within two weeks of infection, and development of neutralizing antibody titers by 3 to 4 weeks post-infection in immunocompetent mice. 1 However, serology testing for LDV in immunocompetent mice is less than ideal due to the interference of virus-antibody complexes. 25 CD4+ and CD8+ lymphocyte responses are critical in neuroprotection through induction of viremia.1,4-6,26,27 Nude (Foxn1nu null) mice, which lack all T cells, including CD4+ and CD8 + lymphocytes, are unable to mount an adaptive immune response against LDV. Not surprisingly, it has been shown that NOD-SCID mice, which lack functional B and T lymphocytes, produce little to no anti-LDV antibody, and these animals fail to generate a specific IgG2a anti-LDV immune response. 28 Similarly, NSG mice, as used in this study, lack functional T, B, and NK cells, have altered innate cell function, and as a result are incapable of producing the T-cell-dependent anti-LDV antibody necessary to prevent neuropathic disease. 29
In the current study, while incidence of paralytic disease was fairly high (65%), some mice remained asymptomatic. One possibility for this finding may be due to the reported differences in virulence between neuropathic and non-neuropathic strains of LDV. It has been shown that the non-neuropathic (LDV-P) strain can rapidly displace the neuropathic strains (LDV-C, LDV-v), limiting clinical manifestations of neurologic disease. 30 In this way, the non-neuropathic LDV strain can outcompete and replace the neuropathic strain in the pool of LDV, resulting in coexistence of both strains in the host, but with the non-neuropathic strain predominating. In addition, neuropathic strains of LDV have a reduced capacity for establishment of persistent infection, making it less likely to be transmitted by animal-animal contact. 30 Alternatively, if the virus is not equally disseminated throughout the tumor sample, there is a possibility that specific areas sampled may be virus free and go undetected.
Sources of LDV infection in traditional animal facilities include biologics of mouse origin, such as serum or basement membrane matrix (eg, Matrigel) which are contaminated with the virus, or potentially from other mice introduced to the facility which are asymptomatic; in some cases, this may include wild mice that gain access to research animal colonies. In an animal containment barrier setting, the most likely mode of infection is through introduction of contaminated tumors, cells, serum, or mouse biological material into recipient mice, generally during allograftment or xenograftment, rather than animal-animal contact (fighting) as in natural infection. 3 For example, LDV may remain undetected in animal colonies used to harvest and produce basement membrane matrix due to asymptomatic carriers, and therefore may be distributed unknowingly to recipient colonies despite implementation of sentinel animal screening. Traditional live-animal sentinel health monitoring programs (with or without dirty-bedding transfers) used to detect pathogens in animal facilities are inefficient for detecting LDV, and detection requires testing of the contaminated sample or affected animal rather than sentinels.3,31 This is in part due to the fact that there is poor transmission of the virus via fecal transfers. Infected mice still shed virus into the environment; however, environmental surveillance via individually ventilated cage (IVC) rack exhaust air dust collection media has been reported to detect the presence of LDV within a given colony. 32 In this study, environmental sampling indicated that only the rack housing LDV-infected animals tested positive while other racks housing NSG and other immunodeficient strains were negative, suggesting LDV is not enzootic in these colonies. Serial passage of PDX tumors is necessary to ensure these samples are stable and can be maintained in recipient mice over time; therefore, rather than contracting the disease from the environment or infected mice as occurs in natural infection, it is thought that infection occurred at some point through the process of passage in recipient animals. Sharing of PDX tumors is commonplace and it is often difficult to determine the impact sample handling at prior institutions may have on LDV or other pathogen contamination of the PDX. It is extremely difficult to retrospectively determine the source of contamination after multiple serial passages in vivo, without evidence of a common source of infection such as shared cell culture materials used in passaging. FBS was the only material reported by the laboratory to be used during the passage of PDXs and PCR for LDV on the current lot used was negative. Therefore, environmental testing in combination with screening of xenograft tumors, infected animals, and biologic materials can yield important and useful data pertaining to incidence and persistence of infection within a given animal colony. Indeed, routine screening of xenograft tumors previously passaged in mice from other colonies outside an animal facility may be warranted to prevent exposure to susceptible populations.
Once detected in an animal population, clearing LDV from infected xenografts and other biologic materials is challenging for various reasons. Clinical signs of LDV-associated poliomyelitis in mice may manifest late in the disease process, before which tumors may have undergone further passage in additional animals, or have been cultured extensively, causing contamination of shared cell culture materials. In addition, each passaged xenograft is composed of a complex tumor consisting of heterogeneous cell populations from both human and mouse origin that may not be equally distributed across samples post-harvest. Furthermore, it remains unclear what the distribution of LDV is in the original tumor and how that distributes into each individual tumor piece in the next passage. Successful eradication of the virus from valuable xenograft tumors has been accomplished by transplantation of infected tumors into nonmurine models, such as nude (Foxn1 nu null) rats, in which the virus cannot replicate; however, some xenografts cannot survive transplantation in other species regardless of immune status and may even change biologic behavior or phenotype. 3 Disassociation and subculture of xenograft tumors in vitro is another method used to remove LDV from tumor cell populations although many early PDX tumors cannot be propagated in vitro. In vitro propagation of PDX may also remove constituents of the microenvironment which could impact the utility of these animal models as more relevant heterogeneous models of cancer. Finally, removal of LDV from xenograft tumors has been reported through fluorescence-activated cell sorting (FACS) of disassociated LDV-negative human tumor cells from LDV-positive mouse stromal cells and may be less likely to alter the tumor phenotype than other reported methods. 3
In summary, we describe the first reported series of LDV-induced poliomyelitis, manifest histologically as degenerative myelopathy and neuropathy, in NSG mice. This case series in NSG mice highlights the importance of screening for LDV in all products derived or passaged in rodents. As a complete history of xenograft and allograft tumors is not always available, researchers should be aware of the contamination status of these samples through routine screening of any xenograft or allograft tumors which have been passaged in mice or cultured using biologic materials of mouse origin. Diagnostic tools in combination with environmental testing are available to detect the virus and should be incorporated into institutional health surveillance programs. Moreover, it is critical to recognize the manifestation of this disease and not to confuse clinical signs of LDV infection with paraneoplastic syndromes or effects of experimental manipulation.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221091747 – Supplemental material for Degenerative Myelopathy and Neuropathy in NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) Mice Caused by Lactate Dehydrogenase–Elevating Virus (LDV)
Supplemental material, sj-docx-1-tpx-10.1177_01926233221091747 for Degenerative Myelopathy and Neuropathy in NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) Mice Caused by Lactate Dehydrogenase–Elevating Virus (LDV) by P. Yang, Z. T. Freeman, R. C. Dysko and M. J. Hoenerhoff in Toxicologic Pathology
Footnotes
Acknowledgements
The authors would like to thank the In Vivo Animal Core of the Unit for Laboratory Animal Medicine at the University of Michigan Medical School for necropsy and histology expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
