Abstract
Thymic lymphoid hyperplasia is a common age-related finding, which occurs particularly in female CD-1 mice. The main differential diagnoses are malignant lymphoma and thymoma. A systematic investigation of control groups from two carcinogenicity studies was performed including measurements of thymic size, and the immunohistochemistry (IHC) markers pan-Cytokeratin (pan-CK) for thymic epithelial cells; CD3 and CD45R/B220 for T and B lymphocytes, respectively; CD31 for endothelial cells; and F4/80 for macrophages. Thymoma can be differentiated by increased numbers of proliferating epithelial cells demonstrated by pan-CK IHC staining. Differentiation between lymphoid hyperplasia and lymphoma is more challenging as a mixture of B and T lymphocytes can be present in both findings. The present investigation showed that the thymic perivascular space is the compartment where the increased numbers of lymphocytes in hyperplasia are localized and not the medulla, as previously thought. The lymphoepithelial compartment is atrophic to the same extent in thymi diagnosed with age-related involution or lymphoid hyperplasia. Both diagnoses are thus related to variations in lymphoid cellularity of the nonepithelial perivascular space, which is continuous with the perithymic tissue. Likewise, lymphomas have a predilection to colonize the perivascular space and to spare the lymphoepithelial compartment.
Keywords
Introduction
The thymus is a lymphoepithelial organ, consisting of the two compartments cortex and medulla that are supported by different types of thymic epithelial cells. 1 In hematoxylin and eosin (H&E)-stained sections, the epithelial cells are not clearly visible, but staining with pan-Cytokeratin (pan-CK) reveals networks of abundant epithelial cells throughout the cortex and medulla. The thymic vasculature, which enters the thymus from the interlobular septa, includes a blood-thymus barrier in the lymphoepithelial cortex. Along the thymic vasculature, the perivascular space forms a compartment of its own, which is devoid of epithelial cells. It is not visible in young animals because it is very narrow except for the region adjacent to the postcapillary venules at the border between cortex and medulla. 2
The lymphoepithelial thymus undergoes age-related involution in all species. In toxicology studies, the thymus of mice has variable morphologies depending on the duration of the study, ranging from a highly populated cortex and medulla in study durations up to 3 months, to a marked decrease in thymic lymphoid cellularity in long-term studies such as the 2-year carcinogenicity study. In the latter, reduced thymic lymphoid cellularity can principally result either from physiological age-related involution or from thymic atrophy due to stressful conditions such as chronic disease or treatment with a test item. Distinction of age-related involution and atrophy is not always possible and there may be an overlap of the two conditions. 3 Although trimming of the thymus follows a standardized protocol, 4 the inherent variability is a challenge in the determination what is normal for age in old mice.
The CD-1 strain of mice is widely used in toxicity studies including carcinogenicity studies; hence, background data of proliferative findings are readily available to help in the interpretation of study findings. Females of this strain tend to develop lymphoid hyperplasia which is the most common spontaneous finding in the thymus above 6 months of age.5,6 The cause of thymic lymphoid hyperplasia, however, is not well understood.
Lymphoid hyperplasia in the thymus can reach unusually large sizes, can mimic neoplasia, and therefore the main differential diagnoses of high-grade hyperplasia are malignant lymphoma and thymoma with a high proportion of lymphocytes. Malignant lymphoma is a systemic tumor derived from the hematolymphoid system, which may originate from the thymus or alternatively infiltrates into various lymphoid and nonlymphoid organs including colonization of the thymus. 7 Thymomas are tumors originating from the thymic epithelial cells and support variable amounts of lymphoid cells.
Recently, a substantial number of thymus-proliferative lesions diagnosed as lymphoid hyperplasia by the study pathologist were submitted to the Registry of Industrial Toxicology Animal-data (RITA), an industry-sponsored toxicology/pathology project for the maintenance of a historical control database for proliferative lesions observed in long-term carcinogenicity studies in rodents. 8 Most of these cases were re-diagnosed as either lymphoma or thymoma by the pathologists participating in the RITA panel meeting and diagnosed as lymphoid hyperplasia again after immunohistochemistry (IHC) staining and evaluation. As this problem already occurred in previous studies and was followed up in ongoing discussions, there was a mutual agreement that a more thorough and systematic investigation was needed. To further investigate the distribution of epithelial cells and B and T lymphocytes within the different compartments of the thymus, all thymi from control mice of a second carcinogenicity study were investigated by IHC. While tumors of the lymphoreticular system in mice have been intensively investigated and classified, only limited literature is available on the immunohistochemical characterization of lymphoid hyperplasia in the thymus of mice. The aim of this study was to improve the understanding of thymic lesions that include an increase of lymphocytes. Moreover, there was a perceived need for guidance on diagnostic criteria of these proliferative and often large focal or diffuse lesions in the thymus of CD-1 mice. In addition, incidences of proliferative lesions of the thymus from the RITA database are given for different mouse strains and compared with the current studies and the available literature.
Materials and Methods
Study Type and Animal Husbandry
Control animals from two carcinogenicity studies conducted over 104 weeks were evaluated. The animals were group-housed and conventionally kept according to the standard procedures established in the respective laboratory of study conduct (study A: Labcorp Early Development Laboratories Inc./former Covance Laboratories Ltd., Harrogate, North Yorkshire, UK or study B: Bayer AG, Wuppertal, Research and Development, Toxicology, Wuppertal, Germany). The studies were conducted in accordance with the requirements of the Animals (Scientific Procedures) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe).
For study A, thymi from 115 males and 117 females (two control groups present in study A) and for study B, thymi from 44 males and 49 females (only one control group included in this study) were evaluated.
Animal Strain and Breeder
CD-1 mice, Crl:CD-1(ICR)BR were used for both studies. Animals were provided by Charles River Laboratories, Margate, UK (study A) or Charles River Laboratories, Sulzfeld, Germany (study B).
Histology
Fixation of tissues was in 4% buffered formaldehyde. Typically fixation times can be variable in carcinogenicity studies, as decedents are not processed immediately and stay in the fixative until after the scheduled termination of the study. After formalin fixation, trimming of the thymus was performed according to the standardized RITA trimming guidelines. 4 Accordingly, one lobe of the thymus was cut along the longitudinal axis to have the maximum longitudinal diameter in section. In aging animals, standardized trimming may be challenging given the variability in organ size. In some animals, there may be no thymic tissue present due to marked atrophy or involution. An additional section may be processed in animals with gross lesions, if necessary. The thymus was embedded in paraffin, cut at an approximate thickness of 4 µm and stained with H&E.
Immunohistochemistry
Thymus tissue sections were stained by IHC according to a standardized protocol. For study A, thymi from 32 control animals (9 males and 23 females, Table 1) were stained on a Ventana Discovery XT immunostainer (Roche Diagnostics, Rotkreuz, Switzerland) with an automated protocol. For study B, thymi from all control animals were stained with a manual IHC protocol. Wherever possible, serial sections of 4 μm thickness were stained for CD3, CD45R/B220, and pan-CK. F4/80 was performed in study A only. Selected cases (study A) or all specimens (study B) were stained for CD31.
Diagnosis of the 32 thymus cases from study A by the study pathologist, the RITA panel before and after IHC assessment; number of cases given.
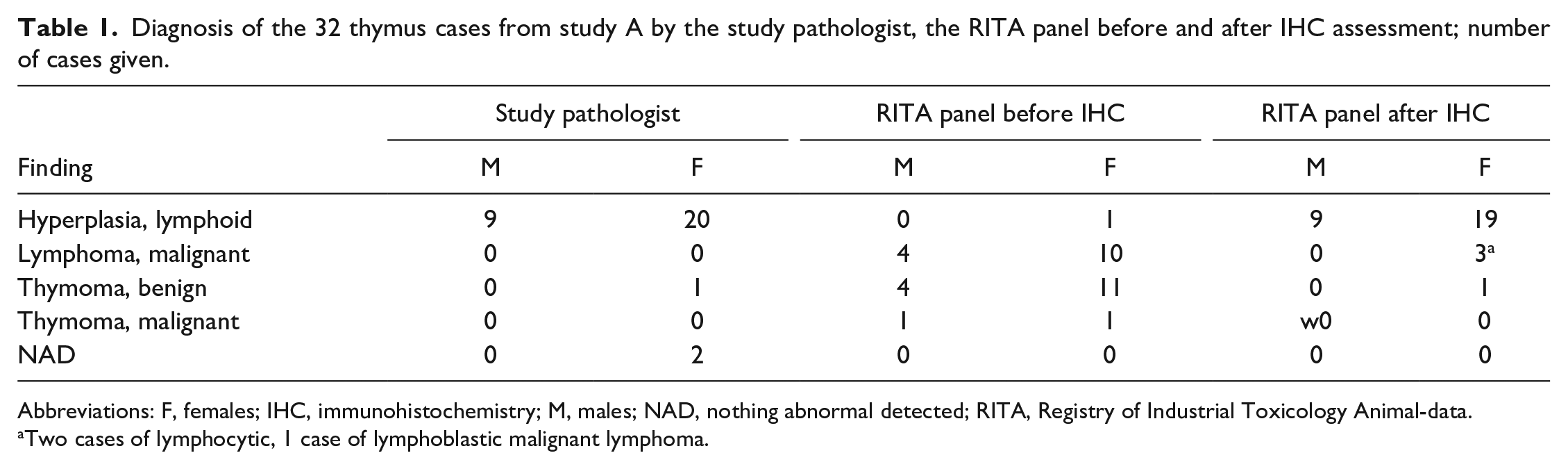
Abbreviations: F, females; IHC, immunohistochemistry; M, males; NAD, nothing abnormal detected; RITA, Registry of Industrial Toxicology Animal-data.
Two cases of lymphocytic, 1 case of lymphoblastic malignant lymphoma.
IHC was performed with the following primary antibodies: anti-CD3 rabbit polyclonal antibody (Dako, Santa Clara, CA, USA, A0452), anti-CD45R/B220 rat monoclonal (BD Pharmingen, Franklin Lakes, NJ, USA, 553084, clone RA3-6B2), anti-pan-CK rabbit polyclonal (Biorbyt, Cambridge, UK, orb10399, study A), anti-pan-CK mouse monoclonal (Sigma, Darmstadt, Germany, C1801, clone PCK-26, study B), anti-F4/80 rat monoclonal (AbD Serotec, Oxford, UK, MCA497GA, clone Cl:A3-1) and anti-CD31 rabbit polyclonal (Thermo Fisher Scientific, Waltham, MA, USA, RB-10333-R7, prediluted ready-to-use). Further details on IHC protocols for both studies on dilutions, incubation time, secondary antibodies, and detection kits are given in Tables 2 and 3. Slides were counterstained with hematoxylin.
Details for IHC staining of study A.

Abbreviations: CK, cytokeratin; IHC, immunohistochemistry.
Details for IHC staining of study B.
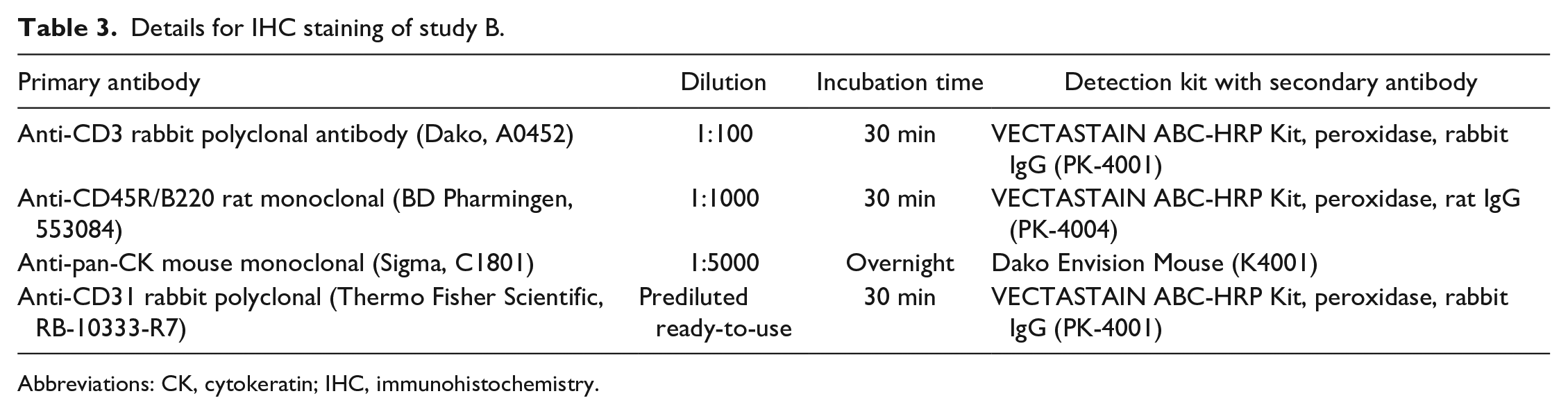
Abbreviations: CK, cytokeratin; IHC, immunohistochemistry.
Variability in fixation duration as well as incipient autolytic changes in some of the decedent animals may have influenced the results of immunohistochemical investigations in individual animals.
Slide Scanning and Size Measurement
All H&E and IHC slides were digitized with an Aperio ScanScopeAT (Wetzlar, Germany, study A) or a ZEISS Axio Scan.Z1 (Jena, Germany, study B) slide scanner.
From the longitudinal central cut of the thymus lobe, the maximum transverse diameter was measured from both studies on H&E slides. For study A, scanned slides from H&E-stained thymi were measured from control group 1 only (49 males and 52 females) with the Aperio ImageScope 12.4.3 software from Leica Biosystems (Wetzlar, Germany). For study B, scanned slides from H&E-stained thymi from all control animals (44 males and 49 females) were measured using the measurement tool in the ZEN 2.3 (blue edition) Carl Zeiss Microscopy GmbH (Jena, Germany).
Based on IHC slide scans, cortex, medulla, and perivascular space were investigated. IHC slide scans were evaluated semi-quantitatively based on the number of CD3, CD45R/B220, and pan-CK positive-stained cells and unstained cells in the thymus sections.
Whenever possible serial sections were used so that the H&E and IHC figures from higher magnifications could show the same area within the thymus tissue, to allow direct comparison of the different stains.
Diagnostics
Histopathological investigation was performed by an experienced pathologist, followed by a peer review by a second pathologist in both studies. Finally, the control groups of both studies were reviewed in the panel session of the RITA database working group. 8 The WHO terminology further developed to the INHAND terminology was used as a basis for the histopathological diagnosis.7,9 This terminology sets standard guidelines for studies and the same criteria are used by the RITA database. Due to a perceived need for a refinement of criteria for the proliferative changes in the thymus, the RITA database working group requested additional immunohistochemical investigations of the thymus from 32 selected control animals of study A. Final results are based on H&E and IHC staining. Table 1 (study A) shows the diagnoses of the study pathologist and the RITA panel before and after IHC assessment. To further investigate the distribution of epithelial cells and B and T lymphocytes within the different compartments of the thymus, all thymi of control mice from study B were investigated by IHC.
Incidence of Lymphoid Hyperplasia, Malignant Lymphoma, and Thymoma From the RITA Database
The incidence of lymphoid hyperplasia, malignant lymphoma, and thymoma from different mouse strains is given in Tables 4–8. Data included 101 two-year mouse carcinogenicity studies. The number of organs investigated, organs with the respective lesions, percentage of lesions, and the minimum and maximum percentage per study are given in the tables.
Incidence of lymphoid hyperplasia in different mouse strains from the RITA database (data from 51 two-year carcinogenicity studies; last change of data entry June 16, 2021).
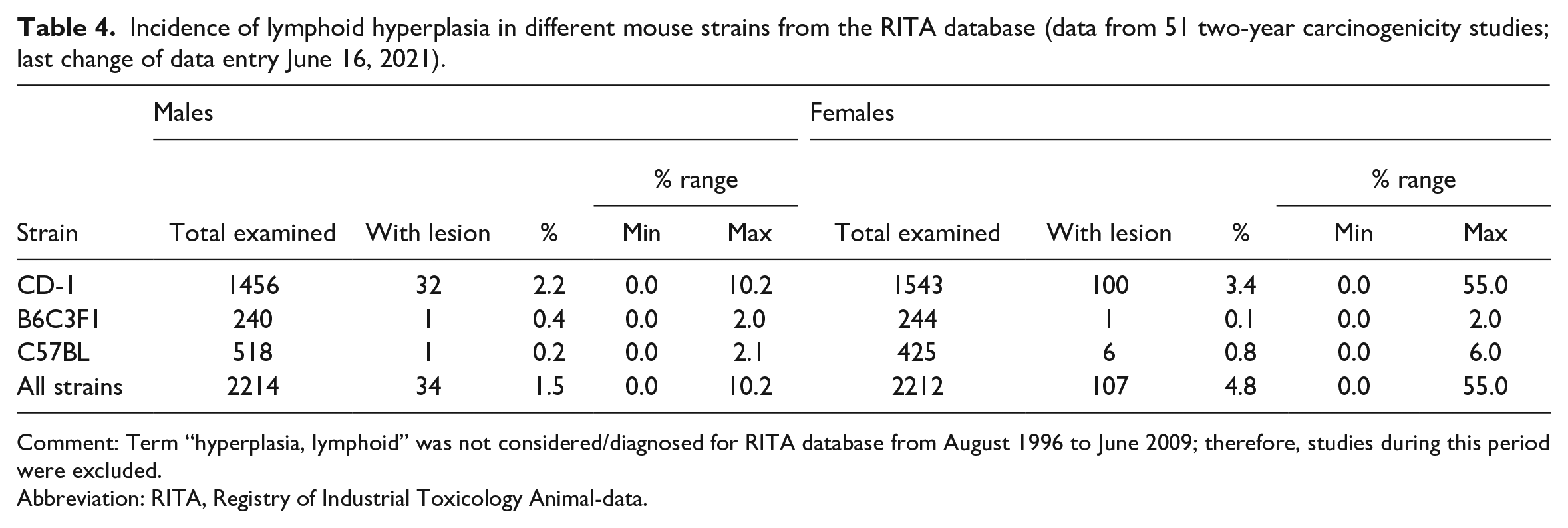
Comment: Term “hyperplasia, lymphoid” was not considered/diagnosed for RITA database from August 1996 to June 2009; therefore, studies during this period were excluded.
Abbreviation: RITA, Registry of Industrial Toxicology Animal-data.
Incidence of malignant lymphoma (that involves the thymus, multiple organs affected) in different mouse strains from the RITA database (data from 101 two-year carcinogenicity studies; last change of data entry June 16, 2021).
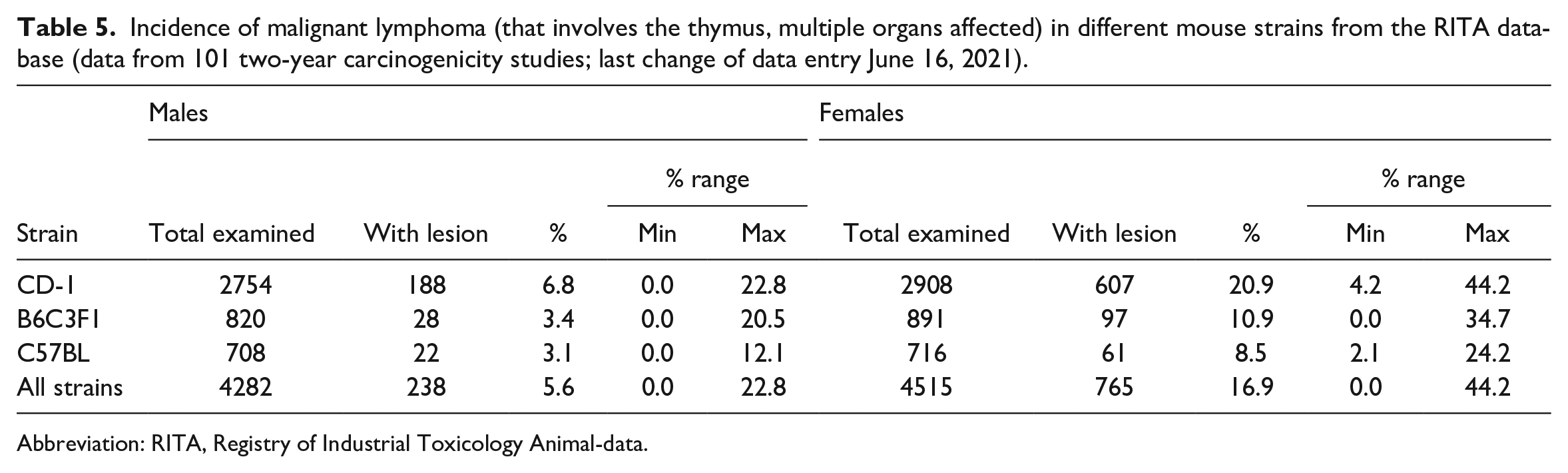
Abbreviation: RITA, Registry of Industrial Toxicology Animal-data.
Number of animals with different modifiers for thymic lymphoma in CD-1 mice (only thymus affected); total numbers shown.
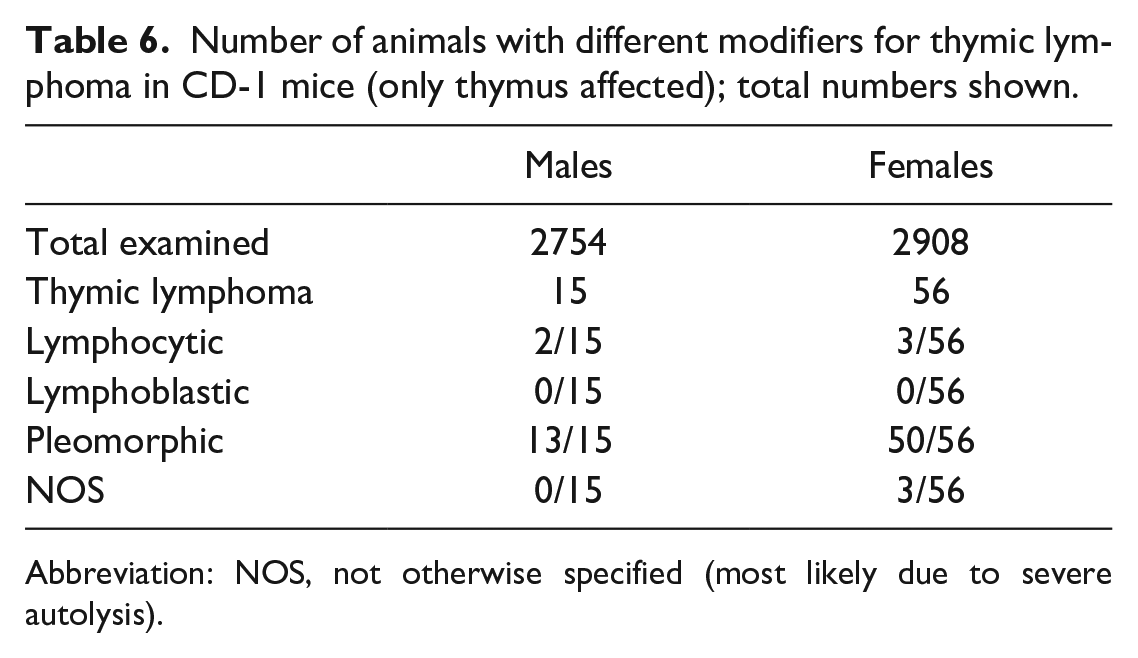
Abbreviation: NOS, not otherwise specified (most likely due to severe autolysis).
Incidence of benign thymoma in different mouse strains from the RITA database (data from 101 two-year carcinogenicity studies; last change of data entry June 16, 2021).
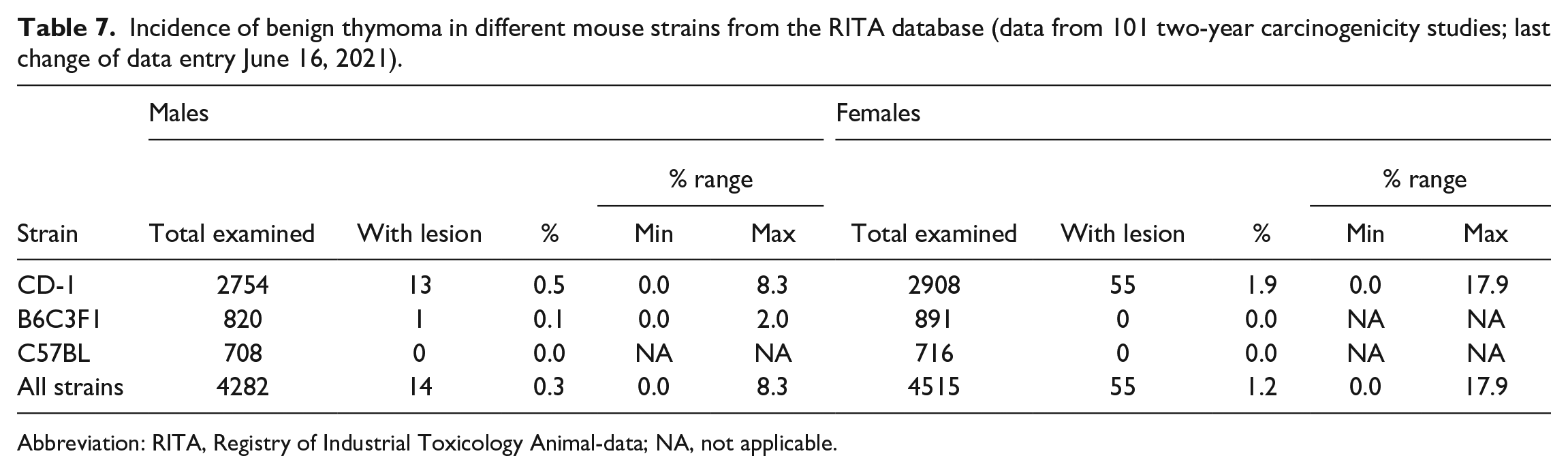
Abbreviation: RITA, Registry of Industrial Toxicology Animal-data; NA, not applicable.
Incidence of malignant thymoma in different mouse strains from the RITA database (data from 101 two-year carcinogenicity studies; last change of data entry June 16, 2021).
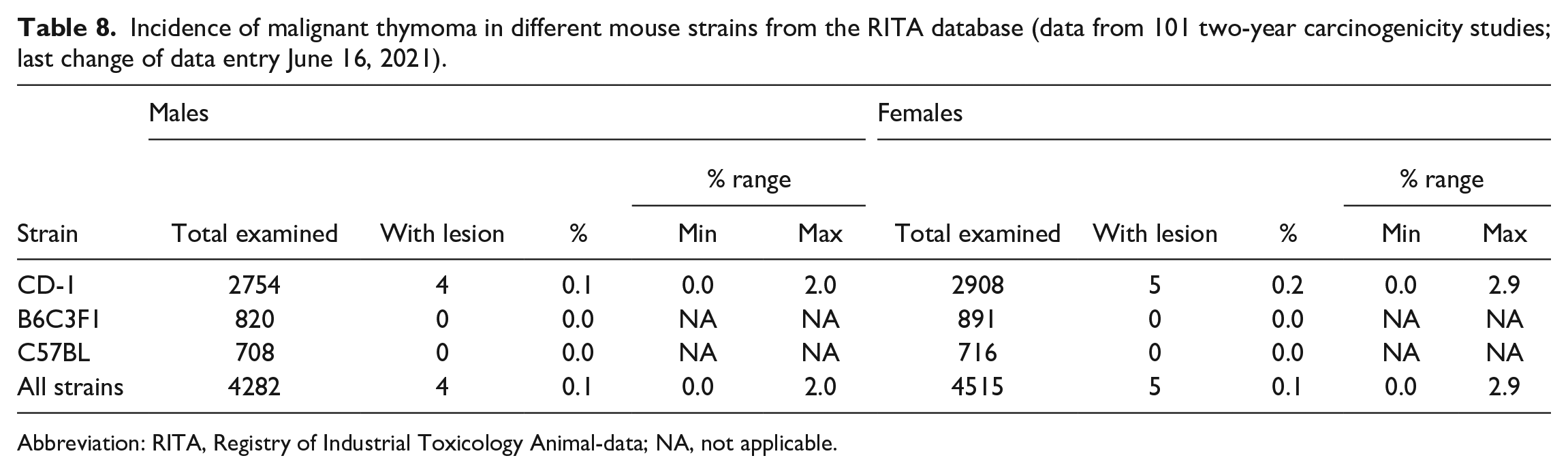
Abbreviation: RITA, Registry of Industrial Toxicology Animal-data; NA, not applicable.
Results
Atrophic/Involuted Thymus
Measurements of the transverse diameter from atrophic/involuted thymi of study A (Figure 1) revealed a range mostly between 600 and 2000 µm in males and 800 to 2500 µm in females. Measurement of the transverse diameter of atrophic thymic lobes from study B (Figure 2) was mostly in a range between 500 and 1000 µm in males and females but in some of the males the diameter was below 500 µm. This suggests that atrophy/age-related involution in study A was not as pronounced as in study B (most likely related to the different origin of the animals and the different environment).
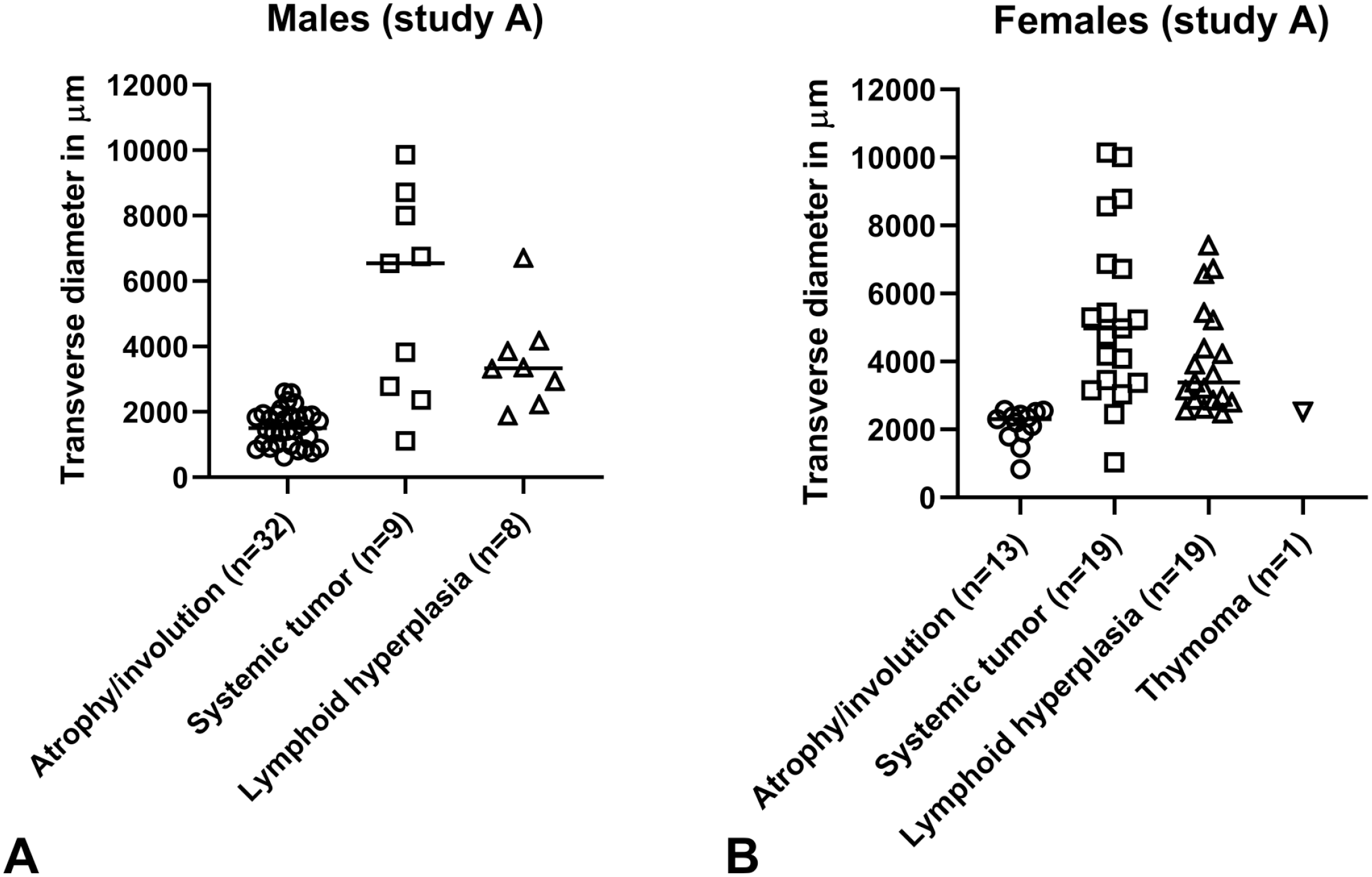
(A, B) Maximum transverse diameter from study A. (A) Males. (B) Females. The individual maximum transverse diameter of the thymic lobe (individual values and median in micrometer) are shown. Atrophy/involution, lymphoid hyperplasia, systemic tumor, and thymoma are shown. In some cases, low diameters are associated nevertheless with lymphoid hyperplasia and higher diameters with atrophy. The final decision was based on the histopathological finding and not made by size. For most animals, however, there was concordance between size and histopathological finding. Higher values were noted for study A.
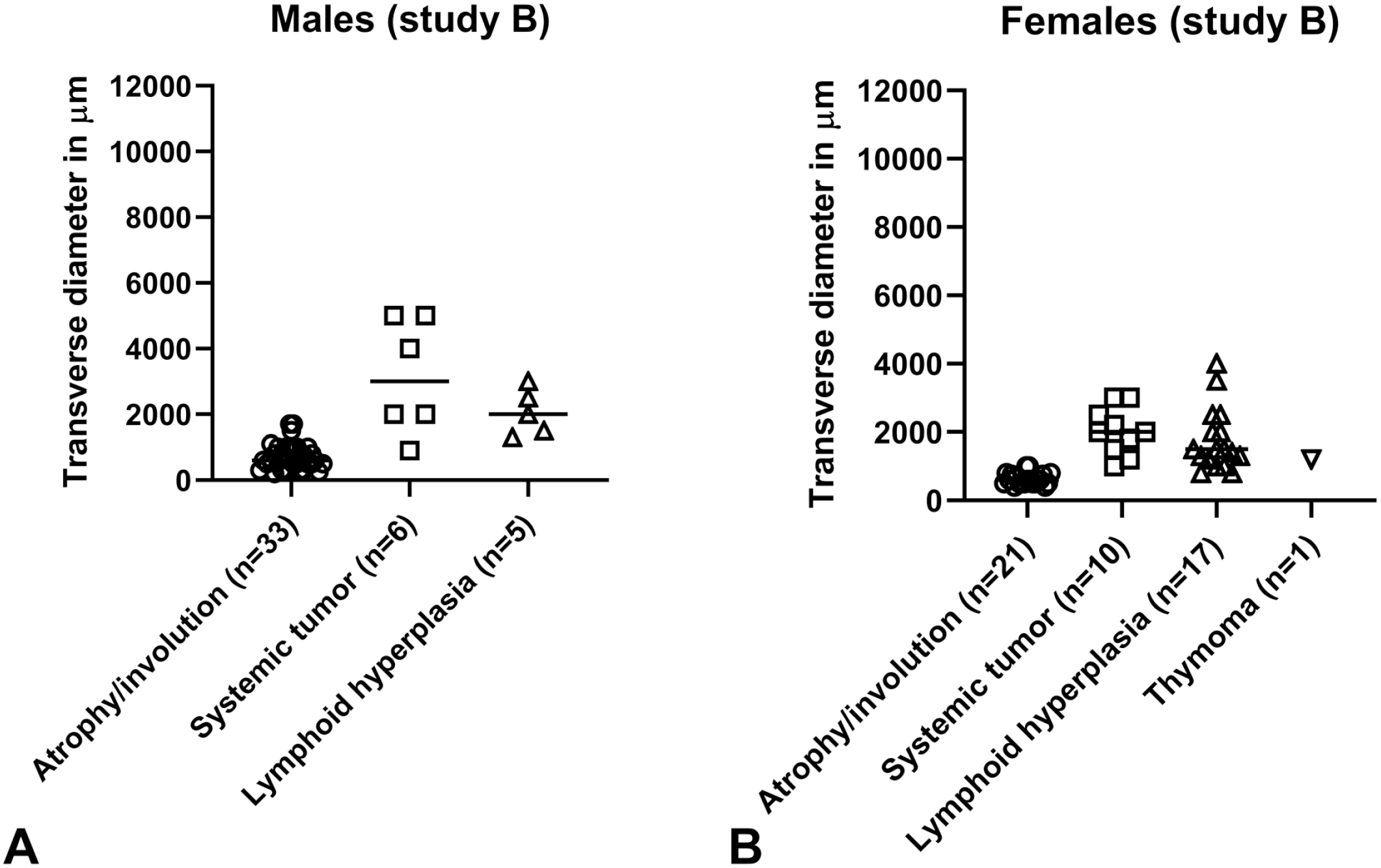
(A, B) Maximum transverse diameter from study B. (A) Males. (B) Females. The individual maximum transverse diameter of the thymic lobe (individual values and median in micrometer) is shown.
Atrophy/age-related involution was a common background finding in both studies, particularly in males. Characteristically, the organ was decreased in size and the border between the cortex and medulla was not clearly discernible in H&E-stained sections. The H&E-stained sections in Figures 3A and 4A show the severely atrophic thymus of a male and the moderately atrophic thymus of a female from study B.
Staining of epithelial cells with pan-CK IHC showed a collapse of medullary epithelia, which gave a streak-like appearance (Figures 3B and 4B). The cortical compartment was reduced in width but still possessed a network of epithelial cells surrounding the thymocytes (Figure 3B). Regardless whether the thymus showed slight or more pronounced atrophy, the lymphoepithelial compartment consisting of cortex and medulla was markedly atrophic when assessed by pan-CK IHC.
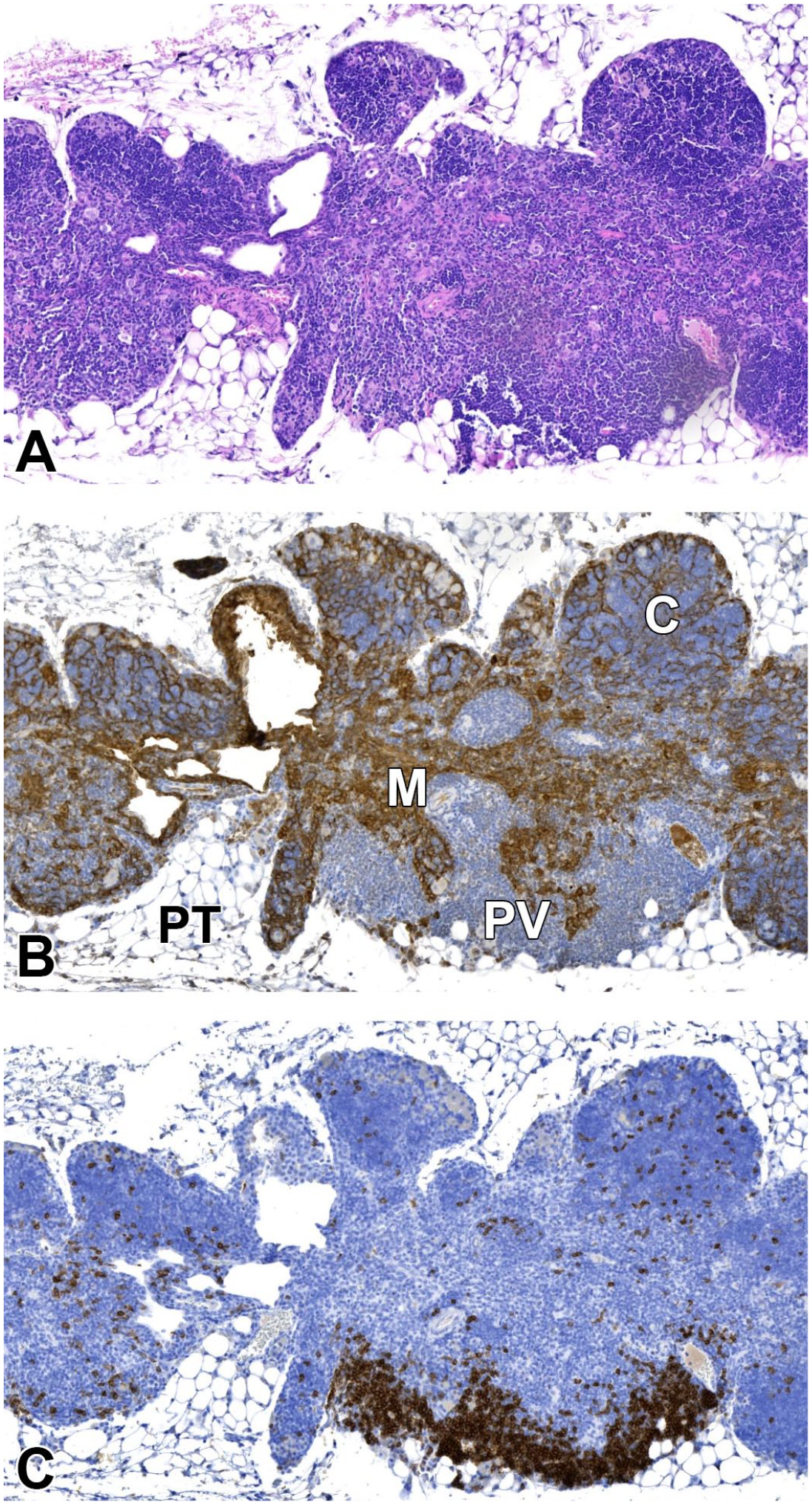
(A-C) Involuted thymus in an old male mouse. (A) With the H&E stain, only some areas with densely packed lymphoid cells can be identified as cortex. Other larger areas appear as medulla in the routine stain. (B) Pan-CK IHC shows the collapsed medullary area (M), cortex (C), and dilated perivascular space (PV) containing lymphocytes. (C) Demonstration of B lymphocytes with CD45R/B220 IHC shows scattered B lymphocytes in the lymphoepithelial cortex (C) and accumulation of B lymphocytes in particular in the PV. Perithymic tissue (PT) is unaffected. Original scan 4X. CK indicates cytokeratin; H&E, hematoxylin and eosin; IHC, immunohistochemistry.
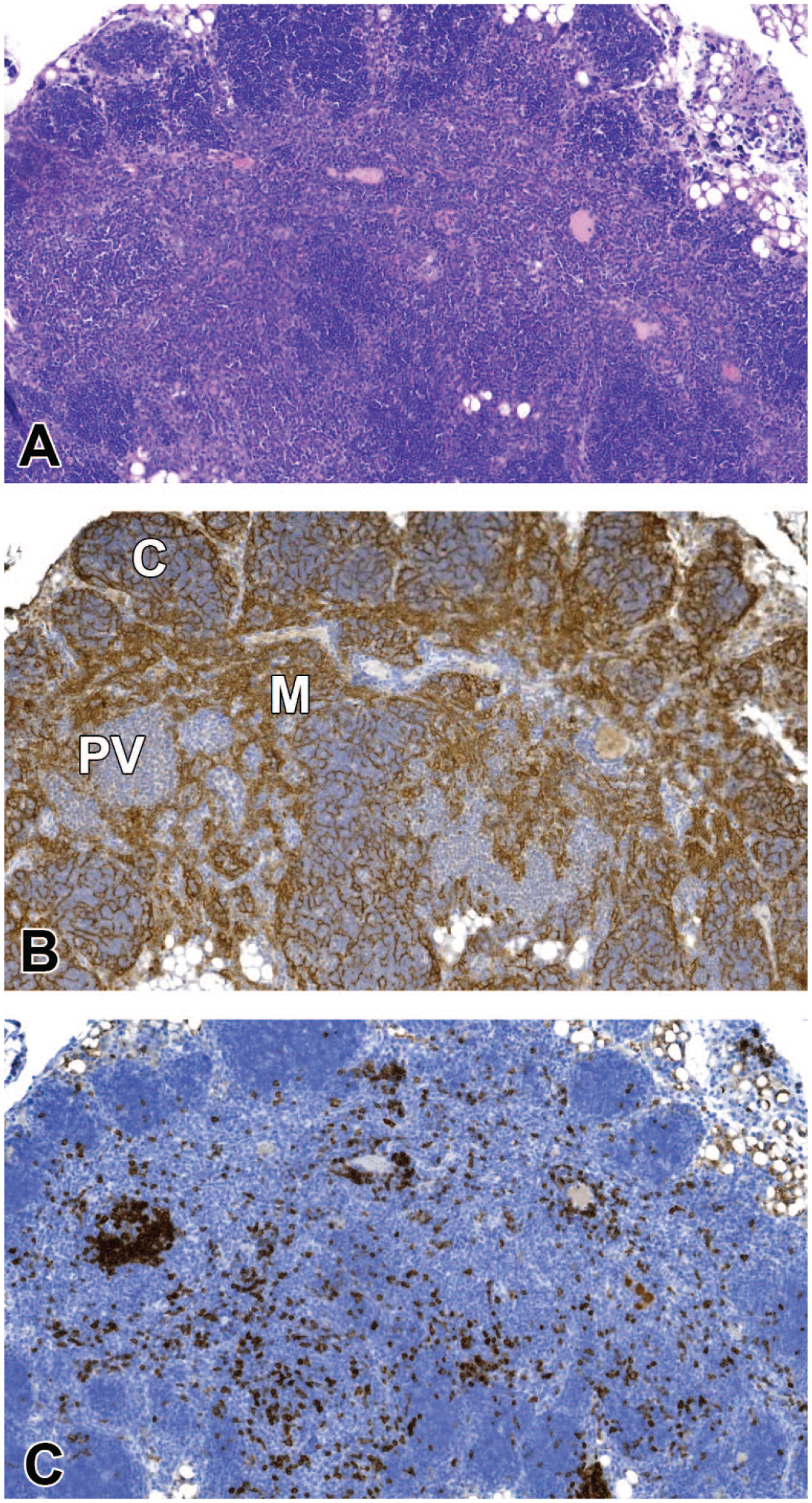
(A-C) Involuted thymus in an old female mouse. (A) H&E stain shows a partly indistinct border between cortex and medulla and distinct boundaries of the organ with the perithymic tissue. The medullary region seems wider than the cortex. (B) Pan-CK IHC shows that the medulla (M) is very small and the paler area in the center is made up of collapsed epithelial structures and a perivascular space (PV) devoid of epithelial cells. The cortex (C) has retained a network of epithelial cells around the lymphocytes and corresponds to the region which can be identified as cortex in the H&E-stained section. (C) With CD45R/B220 IHC, B lymphocytes are found in small quantities throughout the thymus but mostly in the PV. Original scan 4X. CK indicates cytokeratin; H&E, hematoxylin and eosin; IHC, immunohistochemistry.
With markers for T and B lymphocytes, respectively, it could be shown that in the lymphoepithelial cortex as well as in between the CD3-positive T-cell precursors (thymocytes), there was always a considerable number of CD45R/B220-positive B lymphocytes present. Lymphoid cells were sparse in the collapsed lymphoepithelial medulla. In the perivascular space, immunohistochemical staining with CD3 and CD45R/B220 showed mixed populations of T and B lymphocytes, including aggregates of B lymphocytes (Figures 3C and 4C).
Lower grades of atrophy seen with H&E staining had a wider perivascular space populated by lymphocytes (Figure 4A). The indistinct border between cortex and medulla, which is typical in atrophic thymi and obvious in the H&E-stained section, could be explained by the numerous lymphoid cells in the perivascular spaces in this region. Thus, surprisingly, the investigators scored rather the cellularity of the perivascular space than the age-related decline of the lymphoepithelial compartment when grading age-related atrophy.
The schematic drawings (Figure 5) demonstrate the changes in the thymus in transition from young (Figure 5A) to old age (Figure 5B) more clearly than it would be possible in histological sections. Along with the atrophy of the lymphoepithelial compartment, the perivascular compartment, which is continuous with the perithymic tissue (yellow), increases in size and is filled mainly with fat cells indicated by the white circles. Lymphoid cells (purple, Figure 5B) start to accumulate in the perithymic tissue of aging mice and further distend the perivascular space. The vasculature enters and leaves the thymus in the perivascular space of the septae and includes as a specialized segment for recirculation, the postcapillary venules with their high endothelium at the border between cortex and medulla. This is shown schematically in Figure 6.
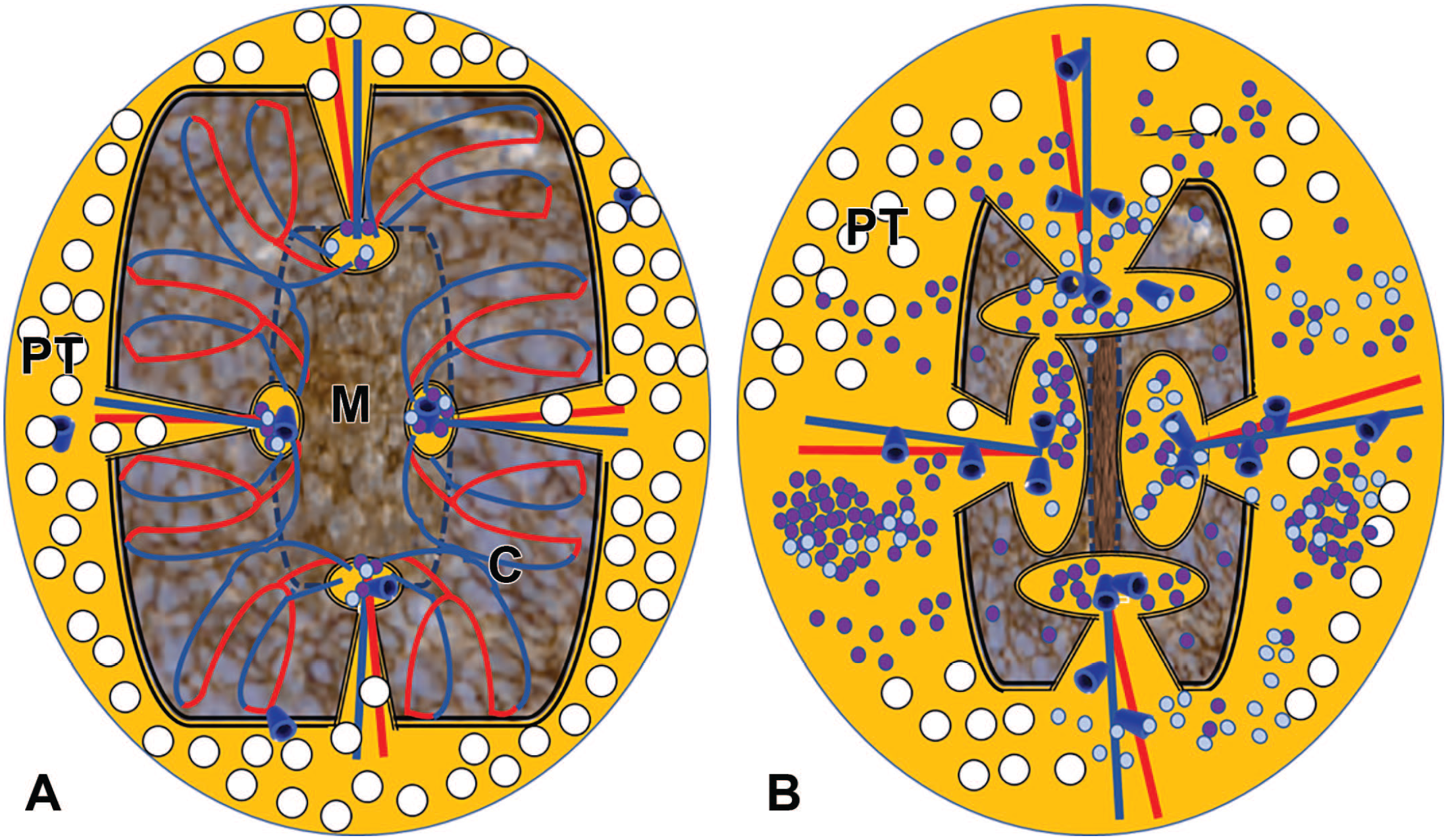
(A, B) Schematic drawings of the thymus in young and old mice. (A) In the young thymus, the lymphoepithelial compartment (brown color) consisting of cortex (C) and medulla (M) makes up most of the organ. The perithymic tissue (PT, yellow color and fat cells represented by white round circles) is small and extends as perivascular space with the interlobular vasculature into the thymus. At the corticomedullary junction, the perivascular space is slightly dilated as lymphoid cells migrate through the space adjacent to the postcapillary venules (arteries: red, veins: blue). (B) In the old thymus, the lymphoepithelial compartment (brown) has collapsed and is reduced in size, whereas the perithymic tissue with the perivascular space has expanded. Besides the fat cells (white), increasing numbers of lymphoid cells (purple) populate the perivascular space and in large lesions also the perithymic tissue.
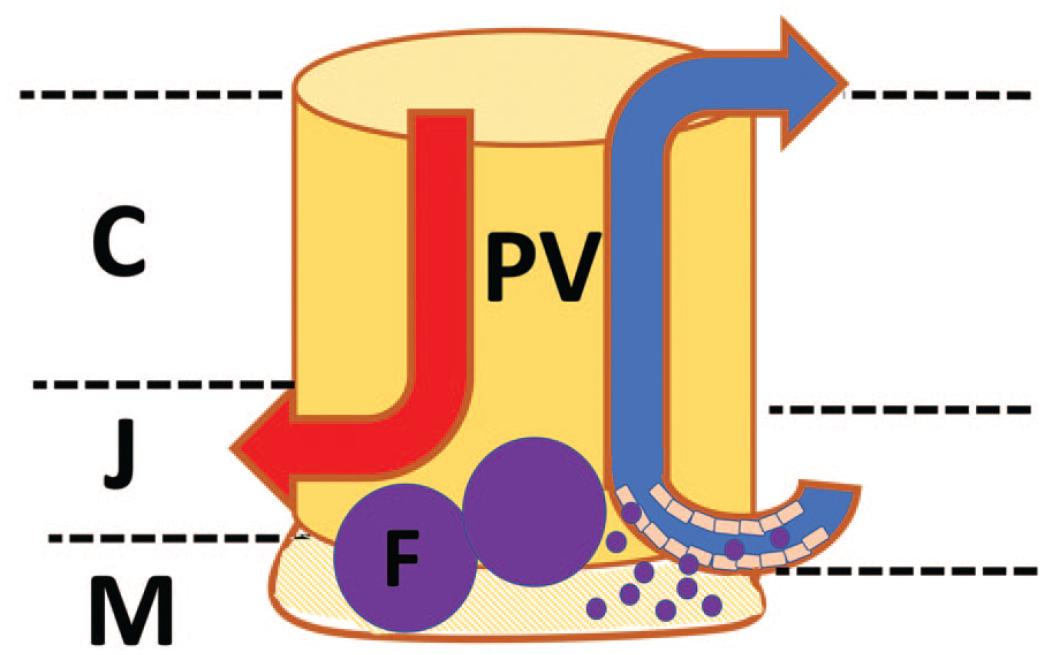
Schematic drawing of perivascular space. Perivascular space (PV) containing an artery (red) and vein (blue) and in the junctional area (J) between cortex (C) and medulla (M) a postcapillary venule. The perivascular space is separated by basement membranes (brown) from the lymphoepithelial compartment and from the vessel. It can protrude in the direction of the medulla when filling with lymphocytes coming in from the postcapillary venule. An area of protrusion is marked by hatching lines in the same color. In the protruding area, follicular structures (F) may be found which are still separated by the basement membrane from the medullary lymphoepithelial compartment but seem to be localized in the medulla if no cytokeratin stain is available.
Distribution of Proliferative Findings in the Thymus and Possible Association With Inflammation or Lymphoid Hyperplasia in Other Lymphoid Organs
The size of the thymus might be a hallmark of differentiation of malignant lymphoma versus lymphoid hyperplasia. Thus, grossly detected enlargements and diagnoses of lymphoid hyperplasia and lymphoma are shown for both studies in Table 9. Gross enlargement of the thymus makes a tumor more likely. However, it also may occur with lymphoid hyperplasia.
Overview of proliferative findings in the thymus; total numbers of all males or females per study shown.
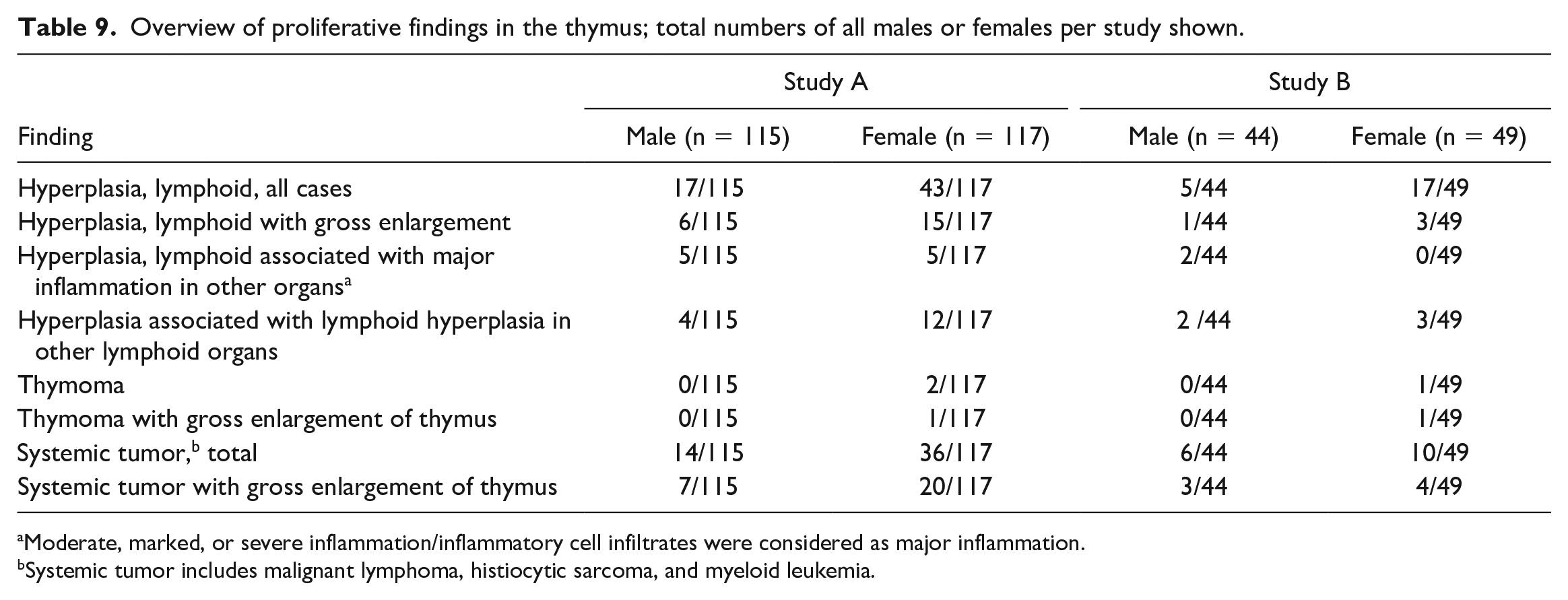
Moderate, marked, or severe inflammation/inflammatory cell infiltrates were considered as major inflammation.
Systemic tumor includes malignant lymphoma, histiocytic sarcoma, and myeloid leukemia.
As lymphoid hyperplasia may also result from a reaction to some inflammatory stimulus in other organs, results including major inflammation are also given in Table 9. Major inflammation is defined as at least moderate inflammation in one organ or tissue. Skin sores were the most frequent type of inflammation. Lymphoid hyperplasia was particularly frequent in female mice and was not associated with major inflammation.
We also investigated whether thymic hyperplasia was associated with hyperplasia in other lymphoid organs and concluded that there was no association. Most of the cases with thymic lymphoid hyperplasia had no other lymphoid organs diagnosed with lymphoid hyperplasia (Table 9). There was no difference in the incidence and severity of hyperplasia in other lymphoid organs between cases with or without thymic lymphoid hyperplasia (data not shown).
Lymphoid Hyperplasia
The transverse diameter of an affected thymic lobe was mostly between 1800 and 5500 µm in study A (Figure 1) and mostly between 1500 and 3000 µm in study B (Figure 2). These measurements suggest that lymphoid hyperplasia in some animals from study A was more pronounced than in study B, again most likely due to the different origin of the animals and the different environment. The measurements show the clear difference in the appearance of the thymus when comparing male and female CD-1 mice, with the higher values in females. In some of the animals, gross enlargement and nodularity were observed.
The normal anatomy of the borders within the lymphoepithelial thymus is different in young and old mice. In young mice with a large lymphoepithelial thymus, the border of the thymus is the border of the lymphoepithelial cortical compartment. There is a thin rim of connective tissue (connective tissue capsule) marking the limits of the organ with the perithymic tissue. In older animals with a regressing lymphoepithelial thymus and an increase of lymphocytes in the perivascular compartment, the border of the thymus is not as clearly defined as the outer part of the thymus comprises the perivascular space that is continuous with the perithymic tissue and what is left of the border of the lymphoepithelial cortical compartment (Figure 5). Nevertheless, the thymus has still a clear contour even when it is enlarged due to the increased cellularity of the perivascular space in lymphoid hyperplasia. Obvious blurring by lymphocytes lying outside the contour and populating the mediastinum in a larger area would be regarded as growth beyond the border of the thymus.
It was difficult to separate cortex and medulla in H&E-stained sections of the thymus with lymphoid hyperplasia. Normal structure was often not retained or was only present in some areas. IHC staining revealed that lymphoid hyperplasia could be separated mainly into two categories:
T-lymphocyte predominant/rich with rare to numerous B cell–rich follicle-like structures and either remnants of epithelial structures or slightly increased epithelial cells; normal or atrophic cortical structures were present in some areas (T-lymphocyte predominant type).
No predominant lymphocyte type with few to numerous B cell–rich follicle-like structures and either remnants of epithelial structures or slightly increased epithelial cells (mixed type).
There was a mixture of small, mostly uniform, and well-differentiated T and B lymphocytes with a prevalence of T lymphocytes (T-lymphocyte predominant type, Figure 7) or no predominant lymphocyte type (mixed type, Figure 8) with aggregates of mainly B lymphocytes (positive for CD45R/B220). In rare cases, there was a small number of lymphocytes outside the well-defined border of the thymus but no further infiltrative growth into the adjacent fat tissue. All cases from study A, which were investigated by F4/80 IHC for the presence of macrophages, were negative.
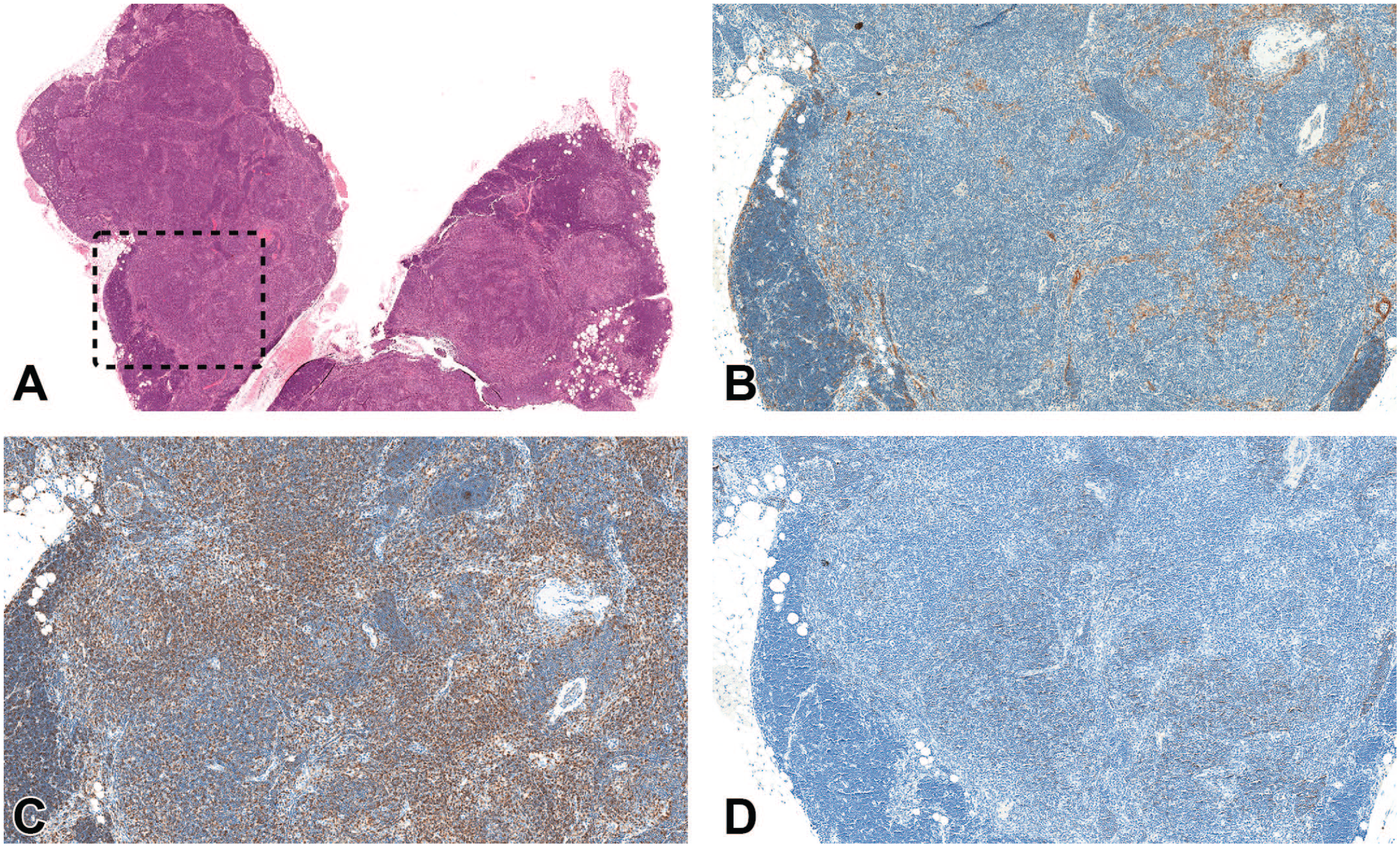
(A-D) Lymphoid hyperplasia in a female mouse (terminal sacrifice after 730 days). (A) H&E stain shows lymphoid hyperplasia. Both lobes are affected, only a few areas with normal cortex are present. (B-D) Show a higher magnification of the dashed box area. (B) Pan-CK IHC shows mainly remnants of epithelial tubules and cords with a minor increase of epithelial cells. (C) Most of the lymphocytes are stained for CD3 (T cells). (D) Only few lymphocytes are positive for CD45R/B220 (B cells). The minor increase of epithelial cells was not considered enough for a diagnosis of benign thymoma. Original scan 1.5X (A), 5X (B-D). CK indicates cytokeratin; H&E, hematoxylin and eosin; IHC, immunohistochemistry.
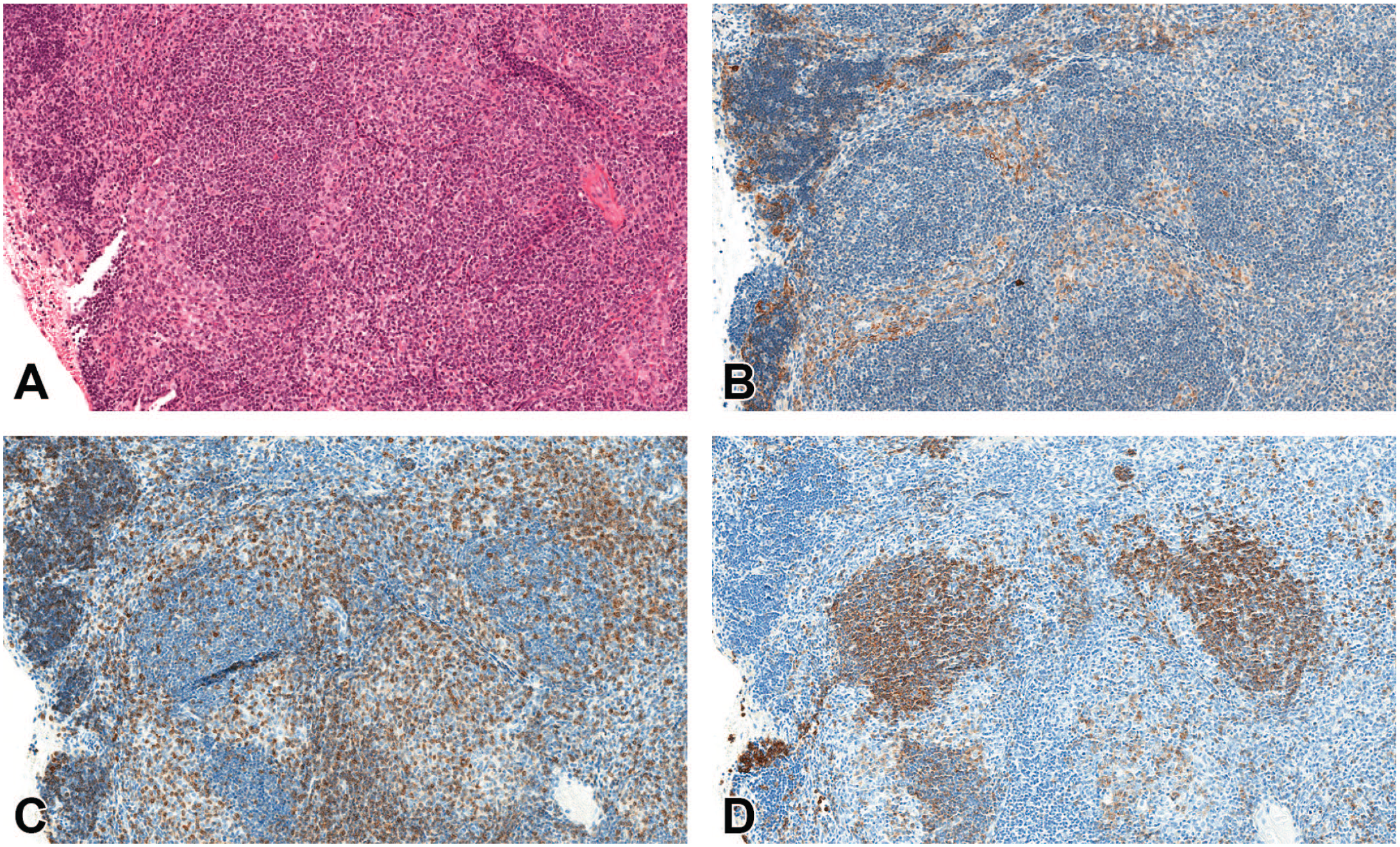
(A-D) Lymphoid hyperplasia in a male mouse (terminal sacrifice after 733 days). (A) H&E stain shows a mixture of small, mostly uniform, and well-differentiated lymphocytes. (B) Remnants of epithelial structures with a minor increase of epithelial cells (pan-CK). (C) No predominant T cell type present, follicle-like structures negative for CD3. (D) Numerous B cell–rich follicle-like structures present in CD45R/B220 IHC. The minor increase of epithelial cells was not considered enough for a diagnosis of benign thymoma. Original scan 10X. CK indicates cytokeratin; H&E, hematoxylin and eosin; IHC, immunohistochemistry.
As in the involuted/atrophic thymi of the old mice in Figures 3 and 4, H&E stain (Figure 9) and pan-CK IHC on lymphoid hyperplasia (Figures 7B, 8B, 10A, and 11A) showed a marked atrophy of the lymphoepithelial compartment with collapse of the medullary epithelial cells and a reduction of the lymphoepithelial cortical compartment. Either epithelial remnants from normal thymus or minor focal/multifocal areas of epithelial hyperplasia (tubules and cords) were common in all cases of lymphoid hyperplasia. With CD3, CD45R/B220, and pan-CK IHC, it became clear that the T and B lymphocytes mentioned above were populating the perivascular space. The perivascular space was distended by the mass of cells, which was much higher than in the atrophic thymi. The follicle-like structures (Figures 8, 12, and 13) were different from organized lymphoid follicles present in lymph nodes, not only by the absence of germinal centers but also by their mixed cell composition, often with a prevalence of B lymphocytes (Figure 8D) and presence of prominent postcapillary venules (Figure 13D). They were often abundant and enlarged and resembled high endothelial venules of T-cell areas in lymph nodes.
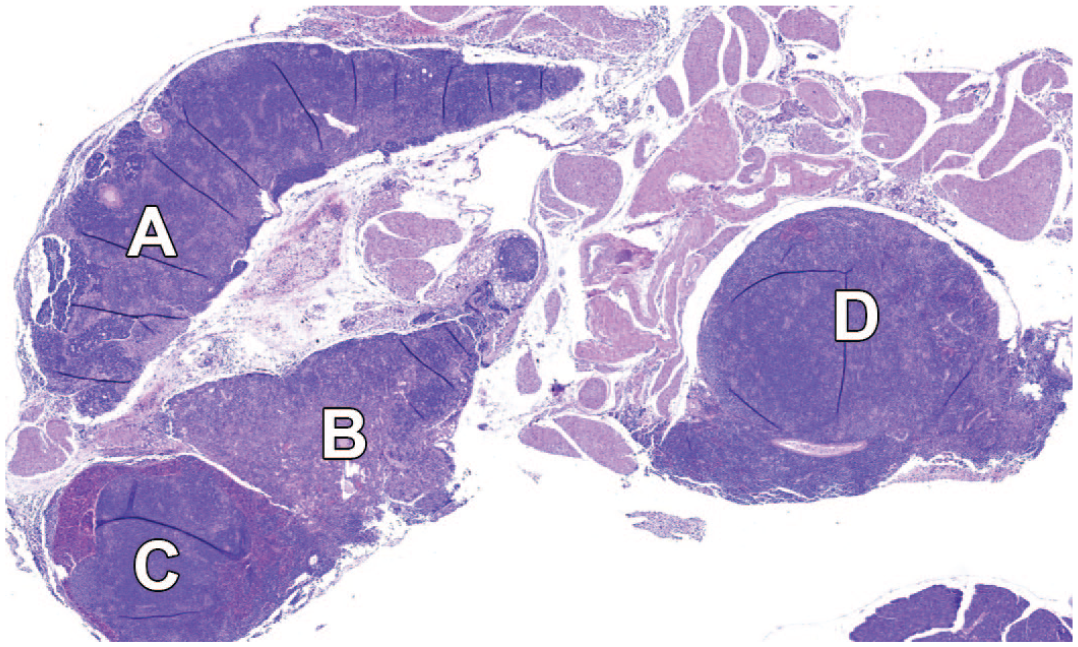
Lymphoid hyperplasia in a female mouse (sacrifice after 603 days). The thymic lobes are enlarged and show different regions in the H&E stain, which are labeled with A, B, C, and D. The regions are followed up with immunohistochemical stains in subsequent figures. Original scan 2X. H&E indicates hematoxylin and eosin.
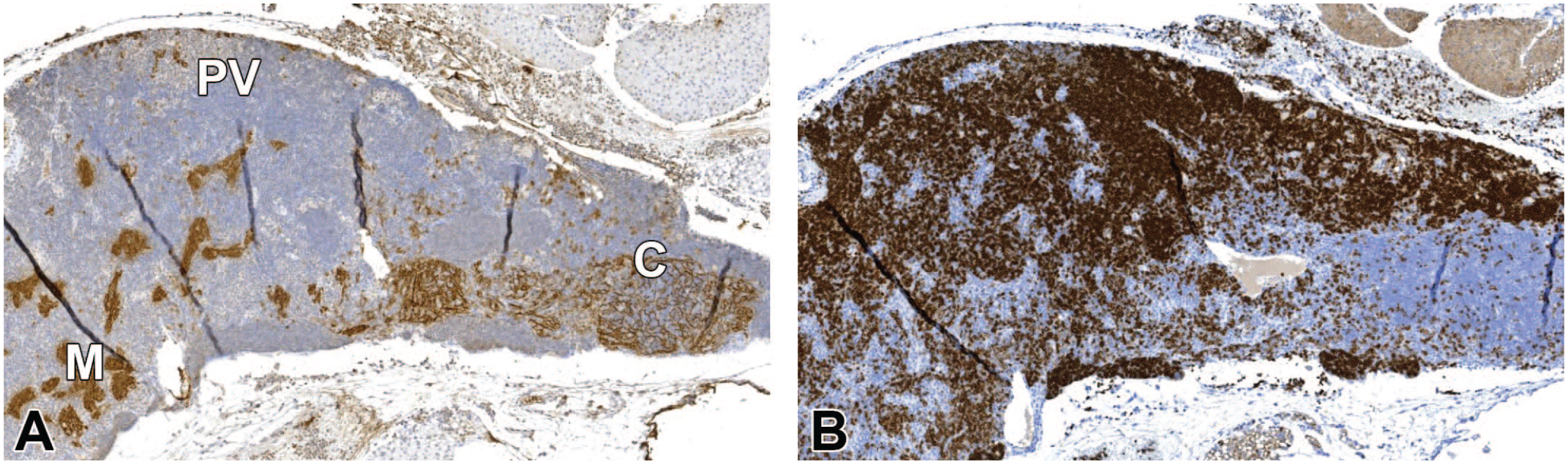
(A, B) Region A from Figure 9. (A) Medullary areas (M) are completely collapsed. Only a minor area with retained cortex (C) seen with the pan-CK IHC. (B) In the area with retained cortex, (C) (A), most lymphocytes are negative for CD45R/B220. In regions which belong to the perivascular space, (PV) (A), the population of lymphocytes consists mostly of B lymphocytes. In the upper right part of (B), a few CD45R/B220-positive cells are present in the perithymic tissue. Original scan 3X. CK indicates cytokeratin; IHC, immunohistochemistry.
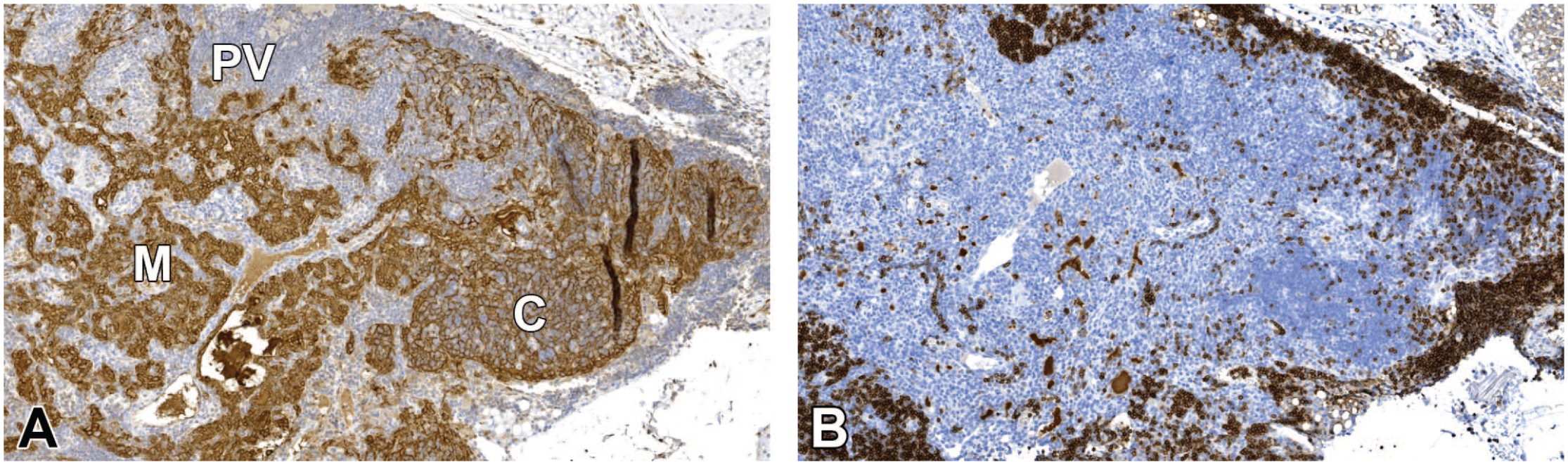
(A, B) Region B from Figure 9. (A) Medullary areas (M) are completely collapsed (pan-CK IHC). Epithelial cells were increased indicating hyperplasia of epithelial cells in the area with retained cortex (C). (B) Most lymphocytes were negative for CD45R/B220 in the retained cortex, (Figure 11A) (C). Lymphocytes in the perivascular space, (Figure 11A) (PV), and the rim of cells outside the cortex in the perithymic tissue were mostly B lymphocytes. Original scan 4X. CK indicates cytokeratin; IHC, immunohistochemistry.
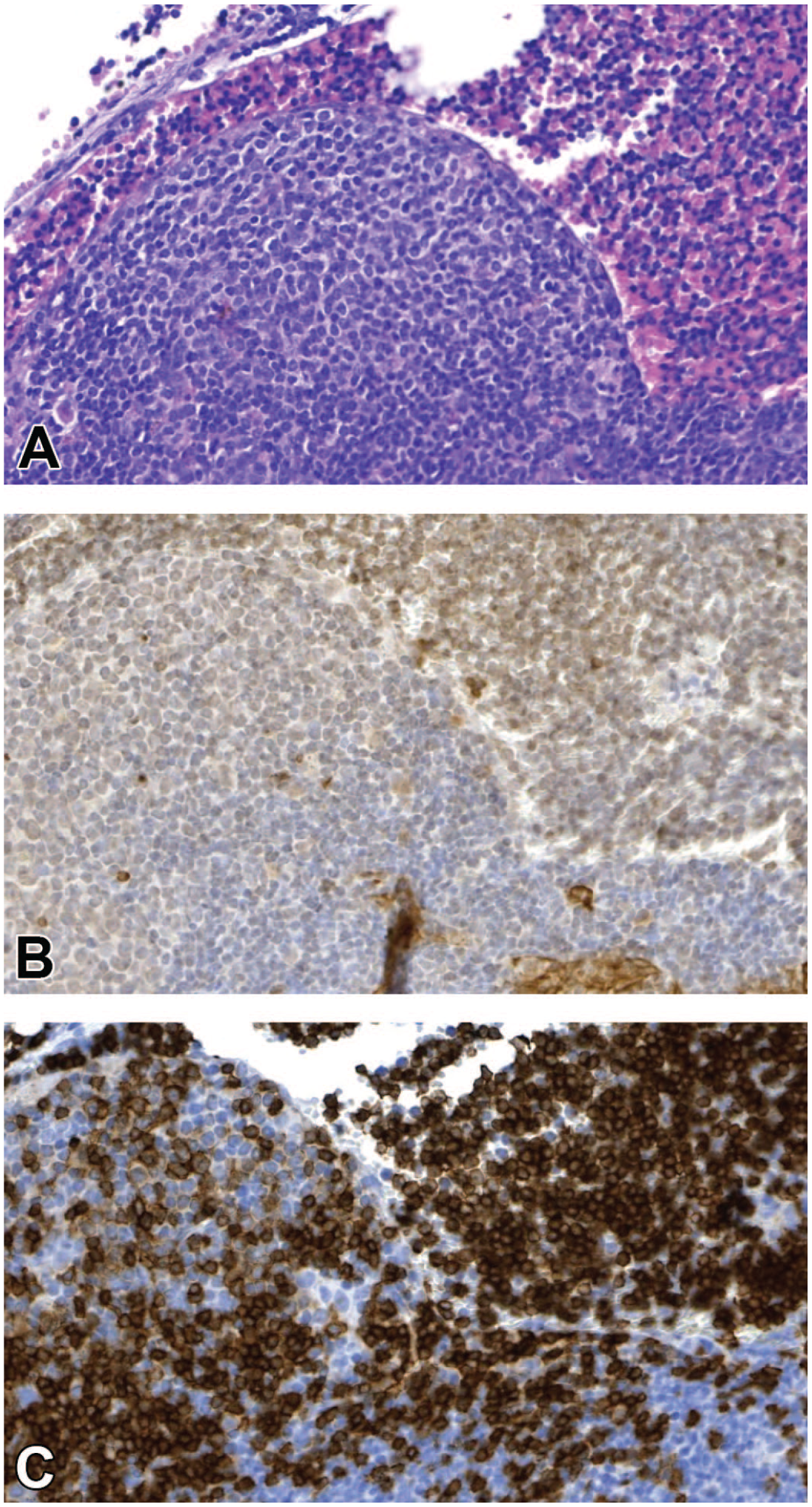
(A-C) Region C from Figure 9. (A) In the H&E stain, the region does not look typical for thymus and has a sinus-like structure filled with erythrocytes and lymphocytes at the margin. (B) Pan-CK IHC shows some epithelial cells. (C) Most lymphocytes are B lymphocytes (CD45R/B220 IHC) including those present in the sinus-like area. Moderate numbers of T lymphocytes were present in addition (CD3 IHC not shown). Original scan 10X. CK indicates cytokeratin; H&E, hematoxylin and eosin; IHC, immunohistochemistry.
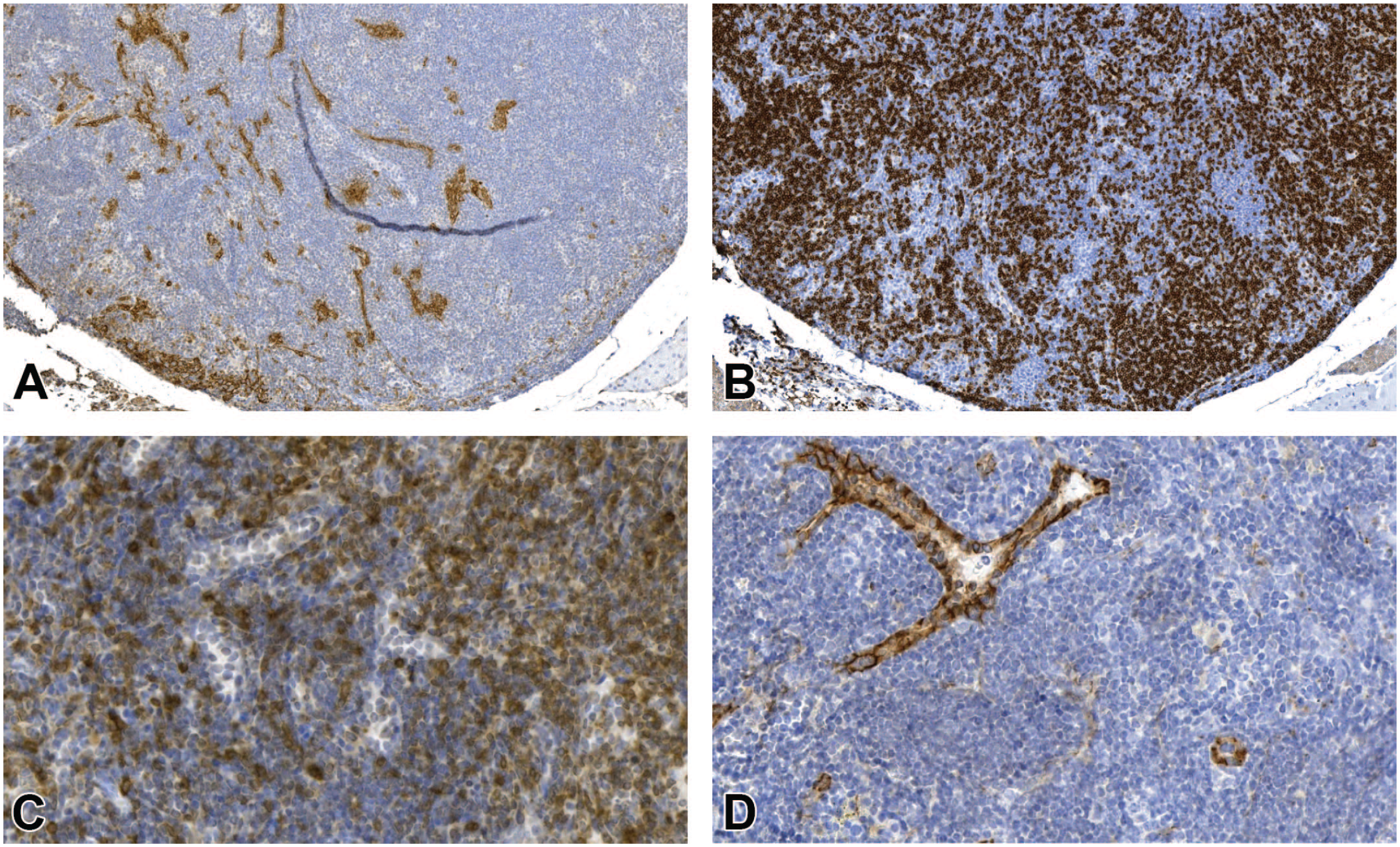
(A-D) Region D (bulging nodular structure) from Figure 9. (A) Demonstration of pan-CK shows the presence of some epithelial cells. (B) B lymphocytes (CD45R/B220) are abundant. (C) B lymphocytes are intermingled with T lymphocytes (CD3). Nodular, bulging areas give the impression of a follicle but are atypical due to the absence of germinal centers. (D) Within the lesion, prominent venules resembling high endothelial venules (HEV) are present which are also not typical for B-cell follicles (CD31). Original scan 4X (A, B), 10X (C, D). CK indicates cytokeratin.
As there was no increased proliferation of epithelial cells in combination with lymphoid cells, the investigators diagnosed lymphoid hyperplasia and excluded the diagnosis of thymoma. In conclusion, the difference between atrophic and hyperplastic thymi was essentially in the perivascular space which was highly populated by T and B lymphocytes in lymphoid hyperplasia.
Malignant Lymphoma
Gross enlargement of the thymus made a diagnosis of malignant lymphoma more likely but was not regarded as indicative of systemic neoplasia, as lymphoid hyperplasia could also lead to gross enlargement (Figures 1 and 2, Table 9). Malignant lymphoma was diagnosed in H&E-stained sections by cell type, pronounced infiltrate beyond the boundaries of the thymus, and presence of infiltrates of the same cell type in other organs.
Lymphocytic lymphoma of T-cell origin was characterized by small and uniform lymphocytes with narrow cytoplasm and condensed nuclei. Rarely, B cell–positive follicle-like structures were present. The mitotic index was low. A high degree of spread or infiltrative growth into the adjacent tissues was present in one case (Figure 14). No generalization throughout the hematopoietic system was present and lymphoma was limited to the thymus.
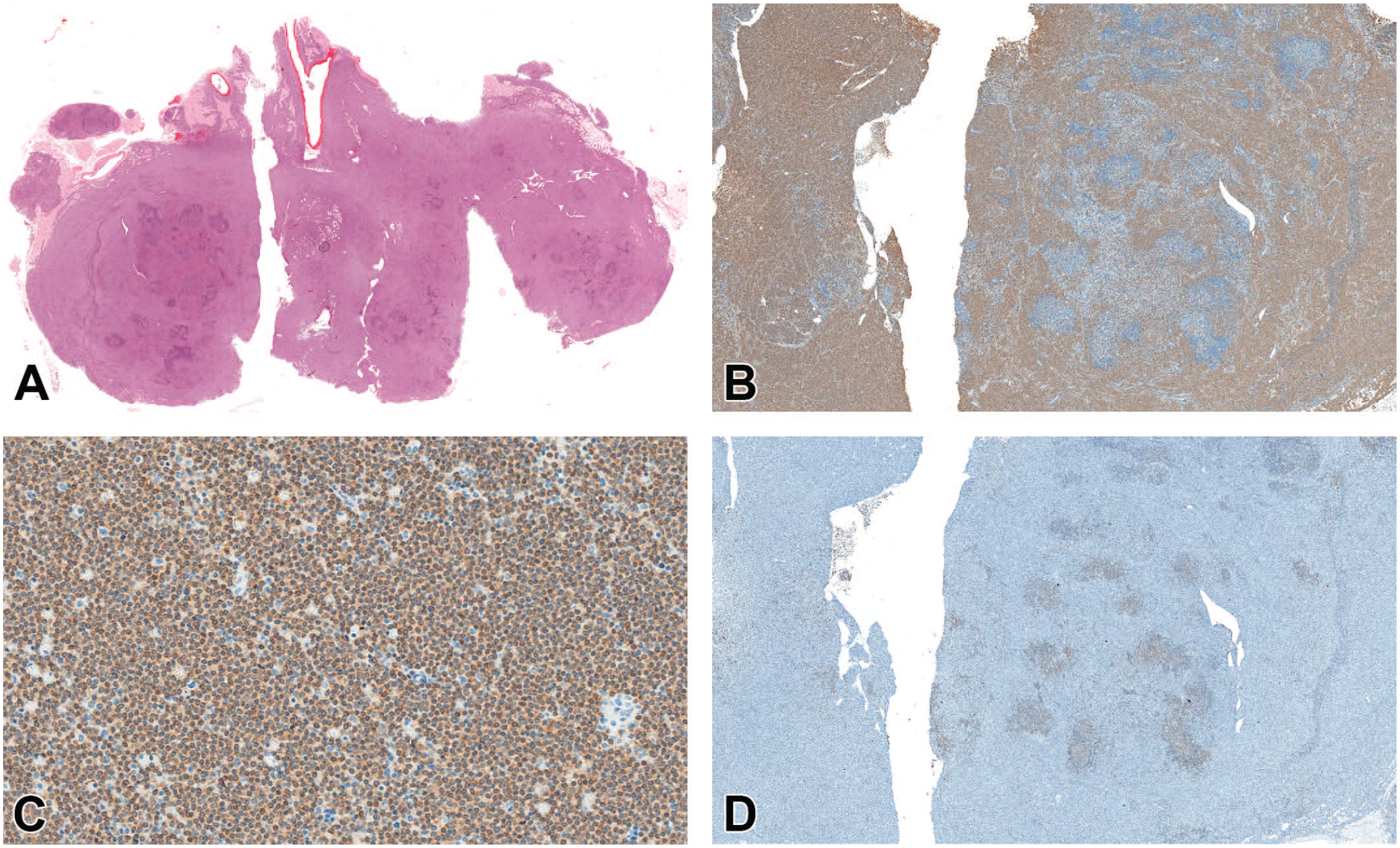
(A-D) Malignant lymphocytic lymphoma in a female mouse (terminal sacrifice after 731 days). (A) Overview of H&E stain shows infiltrative growth of malignant lymphocytic lymphoma into the adjacent tissues. (B) Overview of CD3 IHC shows diffuse staining for T lymphocytes. (C) Higher magnification of (B). Diffuse staining of small and uniform T lymphocytes for CD3. (D) Only rare follicle-like structures positive for CD45R/B220 present. Original scan 1X (A), 2X (B, D), 10X (C). H&E indicates hematoxylin and eosin; IHC, immunohistochemistry.
Lymphoblastic lymphoma was characterized by medium-sized lymphoblasts with some apoptotic and mitotic cells. However, the typical starry sky appearance was not present. Epithelial cells were slightly increased with cortical remnants in some areas (Figure 15). In the animal shown, the spleen was also affected by lymphoblastic lymphoma.
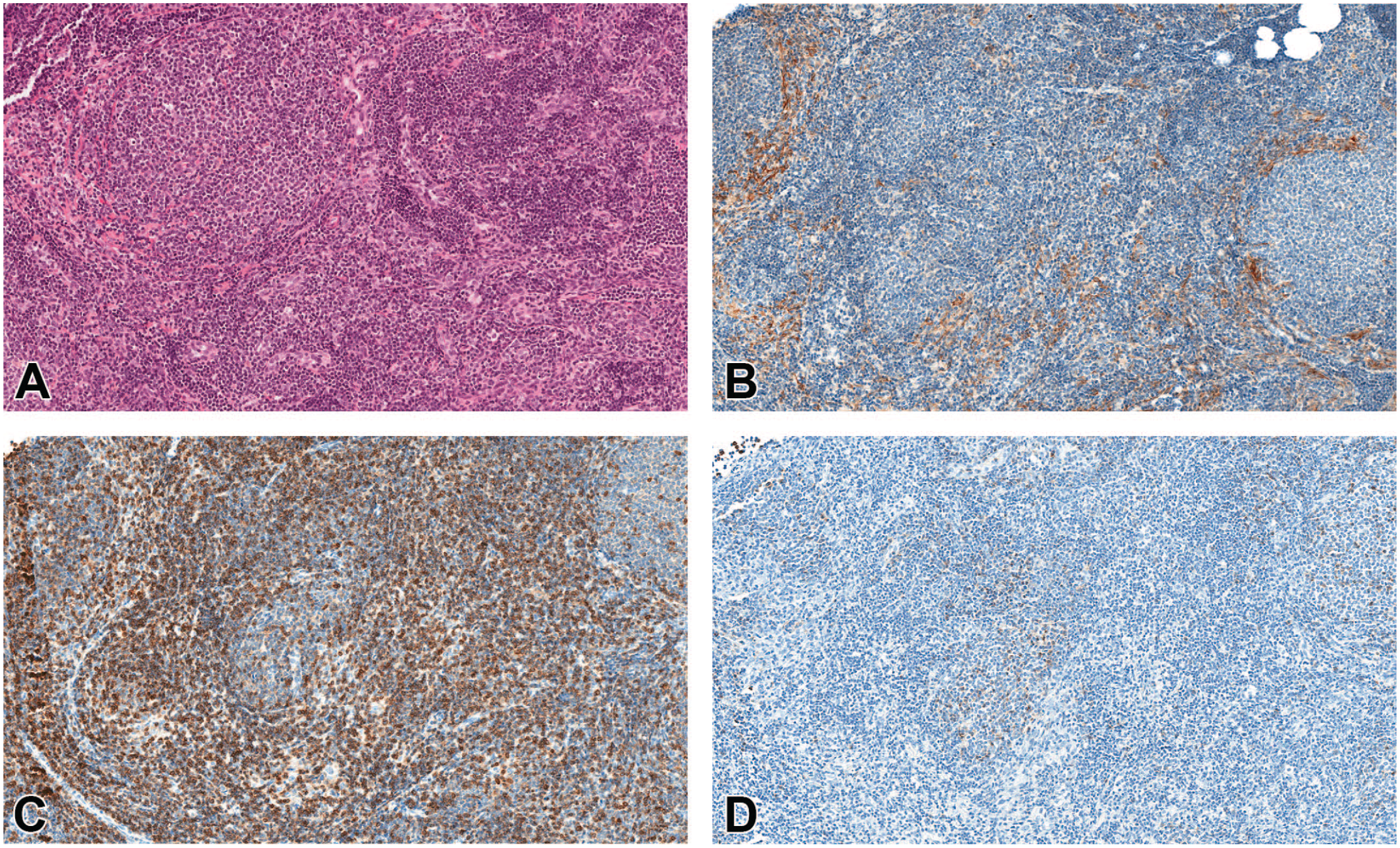
(A-D) Malignant lymphoblastic lymphoma in a female mouse (sacrifice after 669 days). (A) The H&E stain shows medium-sized lymphoblasts. (B) The tumor has grown around islands of epithelial cells, which are remnants of the lymphoepithelial thymus (pan-CK). (C) Most of the cells are positive for CD3; therefore, malignant lymphoma of T-cell origin was diagnosed. (D) There was almost no positivity for CD45R/B220. The spleen from this animal was also affected by lymphoblastic lymphoma. Original scan 10X. CK indicates cytokeratin; H&E, hematoxylin and eosin.
Pleomorphic lymphoma was characterized by the presence of several different lymphoid cell types of variable size and shape with increased and variable amounts of cytoplasm and a cohesive pattern (cell borders not discernible). In the example case, the tumor has grown around minimal focal epithelial hyperplasia or remnants of the lymphoepithelial thymus. Most of the cells were positive for CD45R/B220 characteristic of B-cell lymphoma (Figure 16). B cells were diffusely intermingled with small amounts of CD3-positive T lymphocytes. The tumor was also present in many other organs.
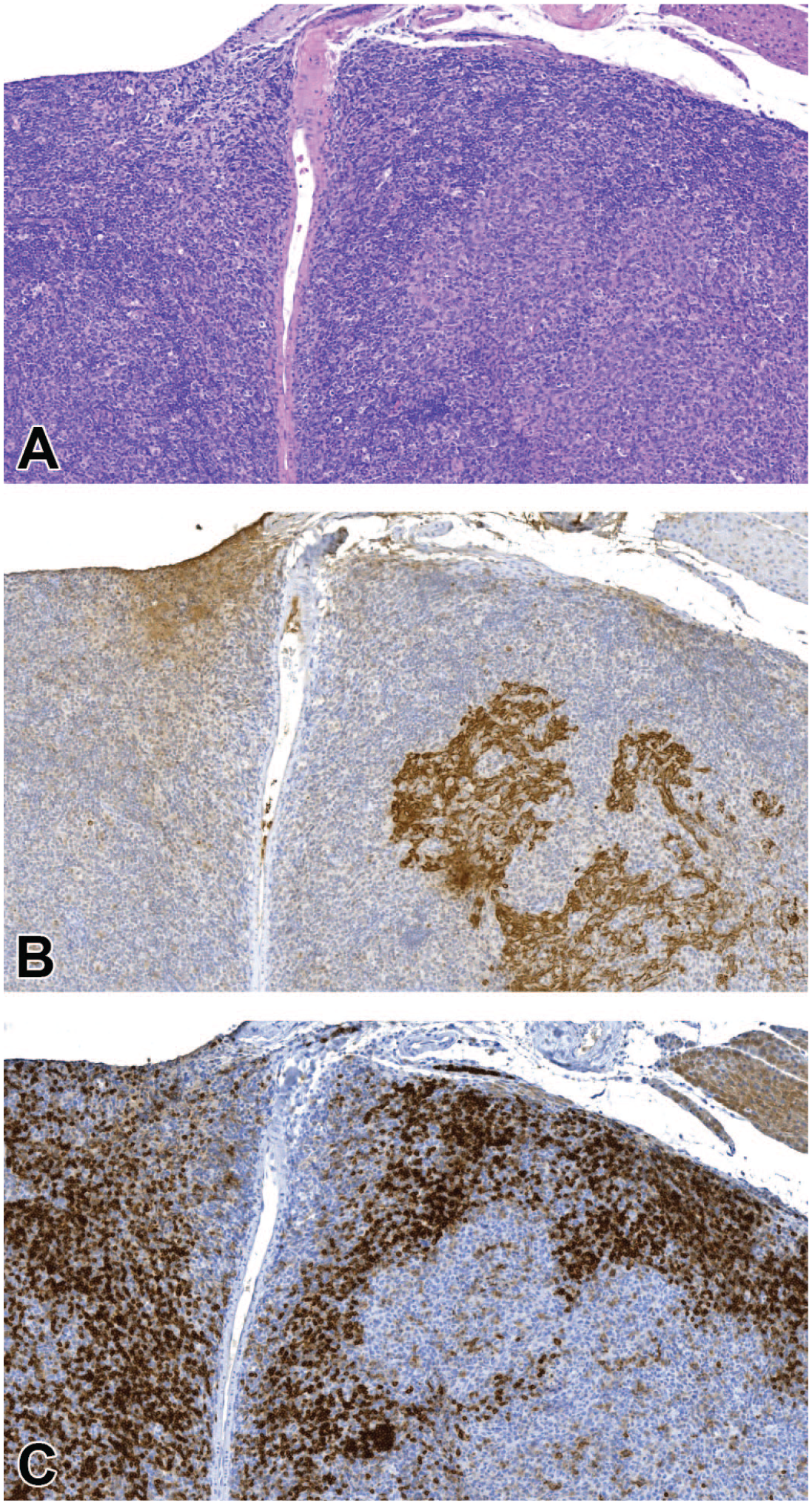
(A-C) Malignant pleomorphic B-cell lymphoma in a female mouse (sacrifice day after 655 days). (A) The H&E stain shows a bulging mass. (B) The tumor has grown around islands of epithelial cells, which are either minimal focal epithelial hyperplasia or remnants of the lymphoepithelial thymus (pan-CK). (C) Most of the cells were positive for CD45R/B220 suggesting a diagnosis of B-cell lymphoma. Original scan 4X. CK indicates cytokeratin; H&E, hematoxylin and eosin.
It was shown for both studies that lymphomas were mainly present in the perivascular compartment including the perithymic tissue (pan-CK IHC). The apparent growth beyond the thymic border in malignant lymphoma was often more diffuse and abundant compared with lymphoid hyperplasia where it usually was not a prominent feature (Figures 14 and 16).
Benign Thymoma
In the cases that were stained by IHC from studies A and B, there was one female in each study diagnosed with benign thymoma after IHC assessment.
In the case from study A, pan-CK-positive epithelial cells and CD3-positive lymphocytes were moderately increased (Figure 17). Few neoplastic epithelial cells showed mitotic figures; there was solid growth at the thymus border and centrally located within the thymus. The tumor was well circumscribed with few, mainly T lymphocytes infiltrating the surrounding adipose tissue.
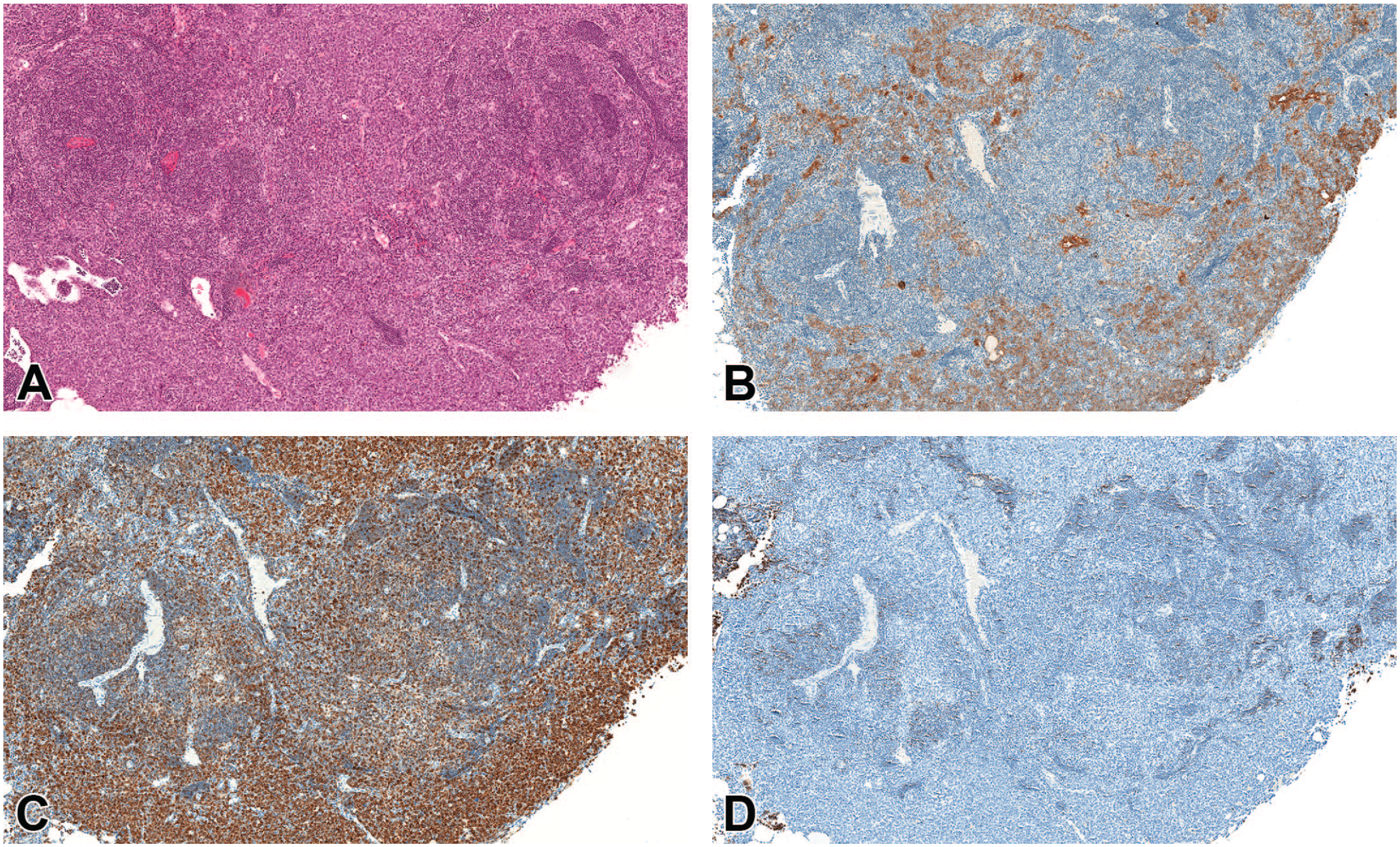
(A-D) Benign thymoma in a female mouse (sacrifice after 708 days). (A) The H&E stain shows solid growth within the thymus. Epithelial structures cannot clearly identified by the H&E stain. (B) Neoplastic epithelial cells with slight amounts of mitotic figures (pan-CK). (C) Moderate increase of CD3-positive lymphocytes. (D) Only few CD45R/B220-positive lymphocytes present. Original scan 5X. CK indicates cytokeratin; H&E, hematoxylin and eosin.
One additional female from study A was diagnosed with benign thymoma without IHC staining. This thymoma was characterized by solid areas of neoplastic epithelial-like cells centrally located within the thymus and moderate amounts of neoplastic lymphocytes (Figure 18). The tumor was well demarcated from the surrounding tissue.
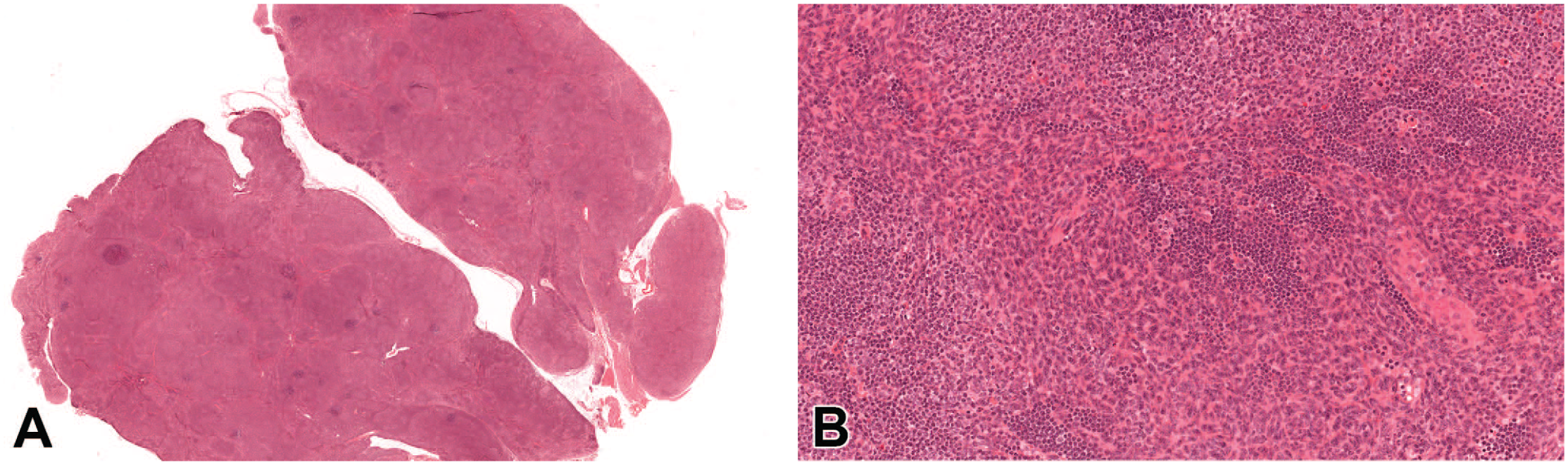
(A, B) Benign thymoma of a female mouse (sacrifice after 253 days). (A) The H&E stain shows solid growth in both thymic lobes. (B) The H&E stain shows a higher magnification, solid areas of neoplastic epithelial-like cells are present. No IHC was performed for this animal. Original scan 1X (A), 10X (B). H&E indicates hematoxylin and eosin; IHC, immunohistochemistry.
In the benign thymoma from study B, it was difficult to see the demarcation between tumor tissue and normal thymic cortex on H&E stain. Neoplastic epithelial cells were clearly visible in the pan-CK stain and appeared to grow beyond the compartment borders into the perivascular space and toward the cortex. They formed sheets and microcysts with mainly CD3-positive and some CD45R/B220-positive lymphocytes in between (Figure 19). As the criterion for malignant thymoma is marked local invasion or metastasis, 7 growth into the perivascular space detectable by IHC was regarded to be compatible with benign thymoma.
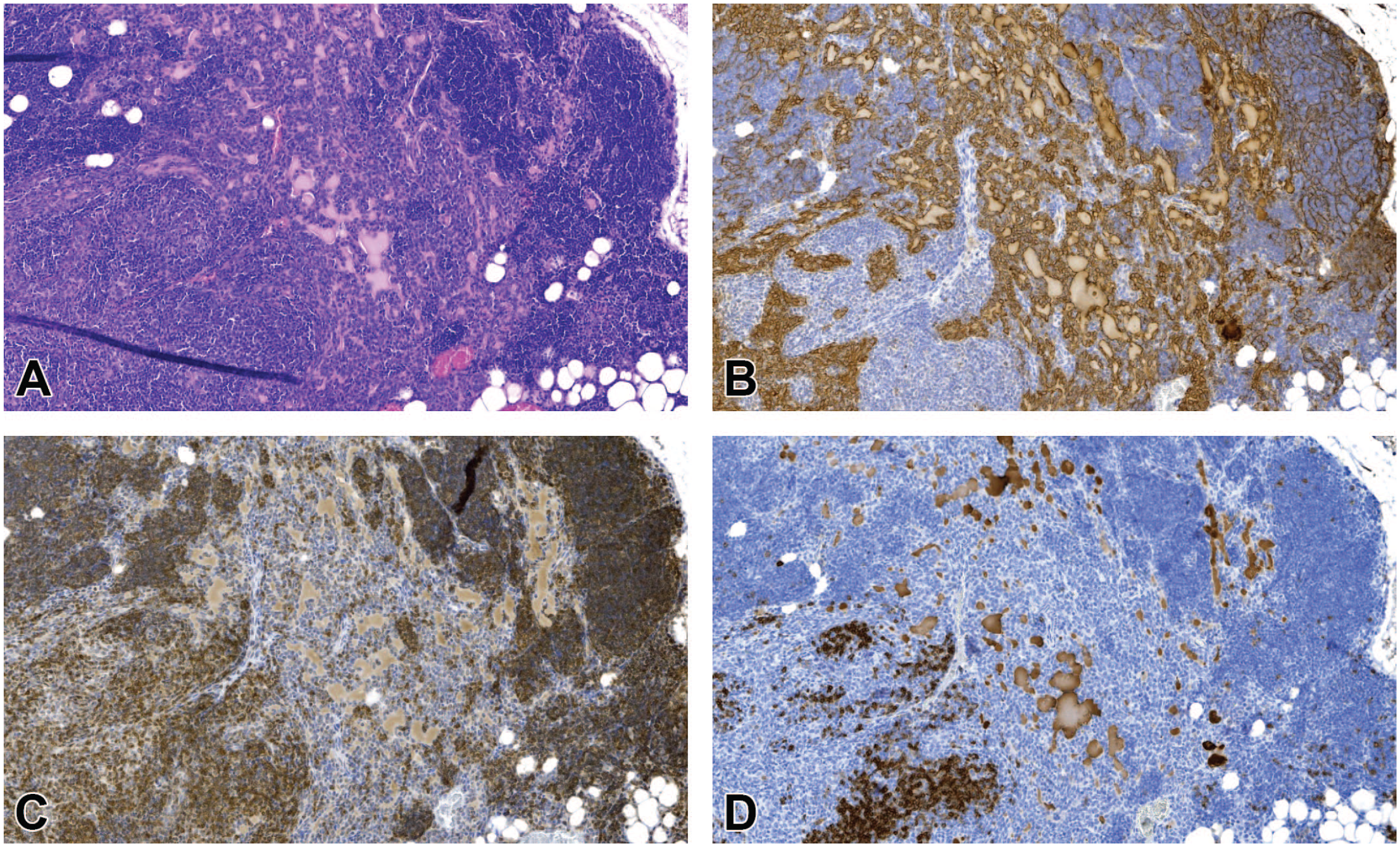
(A-D) Benign thymoma in a female mouse (sacrifice after 731 days). (A) The H&E stain showed no clear demarcation of tumor from the thymic cortex. (B) Neoplastic epithelial cells are clearly visible and form sheets and microcysts (pan-CK). (C) Mainly CD3-positive lymphocytes present. (D) Only few CD45R/B220-positive lymphocytes are in between the pan-CK–positive areas. The tumor is growing beyond compartment borders into the perivascular space and toward the cortex. Original scan 4X. CK indicates cytokeratin; H&E, hematoxylin and eosin.
Incidence of Lymphoid Hyperplasia, Malignant Lymphoma, and Thymoma in the RITA Database
The incidence of lymphoid hyperplasia, malignant lymphoma, and thymoma from different mouse strains is given in Tables 4–8.
Lymphoid hyperplasia was most common in CD-1 mice and in general more often observed in female mice (Table 4). For the other mouse strains, this was not a common finding. In one of the studies, lymphoid hyperplasia of the thymus in CD-1 mice was observed in up to 10.2% of the males and 55.0% of the females.
Malignant lymphoma was a common finding in mice, and in many cases it also involved the thymus (Table 5), but it can also be limited to the thymus only (thymic lymphoma). Females were more often affected than males. It occurred more or less at the same incidence in B6C3F1 and C57BL mice. The highest incidence was seen in CD-1 mice (6.8% in males, 20.9% in females). For all systemic malignant lymphoma in CD-1 mice, the thymus was involved in 62.7% (males) and 75.4% (females) of the cases (data not shown).
In CD-1 mice, thymic lymphoma (tumor only present in thymus) was observed in 15 males (0.5%) and 56 females (1.9%, Table 6). In B6C3F1 and C57BL mice each, there was 1 female with thymic lymphoma (0.1% each, data not shown). Thymic lymphomas in CD-1 mice were mostly classified as pleomorphic (13 cases in males, 50 cases in females). The other eight cases were either lymphocytic (2 males, 3 females) or not otherwise specified (3 females). The two cases in B6C3F1 and C57BL mice had the modifier lymphocytic.
As shown in Tables 7 and 8, thymomas were rare in mice. Only a limited number of cases were captured. Most cases of benign thymoma (Table 7) were observed in CD-1 mice with an overall incidence of 0.5% in males and 1.9% in females. The maximum observed occurrence of benign thymoma per study was 8.3% in males and 17.9% in females (CD-1 mice). Only one case of benign thymoma was diagnosed in B6C3F1 mice. Malignant thymomas were extremely rare in mice (Table 8) and only nine cases in CD-1 mice were observed (0.1% of males and 0.2% females).
Discussion
Differentiation of Lymphoid Hyperplasia, Malignant Lymphoma, and Thymoma
To our knowledge, this is the first report characterizing a substantial number of thymic lesions by applying pan-CK, CD3, and CD45R/B220 IHC on serial sections in aging mice and showing the distribution of epithelial components and T and B lymphocytes. It was possible to confirm that most of these thymic proliferative lesions were lymphoid hyperplasia as previously described 6 and did not represent malignant lymphomas or thymomas.
In all of these cases of lymphoid hyperplasia, there was a mixture of small and large lymphocytes, mostly well-differentiated T and B lymphocytes, which infiltrated the perivascular space in most of the animals. There was no indication for an expansion of a unique or uniform lymphocyte type, neither T nor B lymphocytes, which suggests lymphoid hyperplasia rather than neoplasia. Hyperplastic lesions are usually not as large as tumor masses seen in thymic lymphomas, even when they are nodular. 5 This has been confirmed in the present investigation by measurements of the transverse thymus diameter. However, in lymphoid hyperplasia, gross enlargement of the thymus at necropsy seems to be a common observation. Therefore, size is an unreliable criterion.
B-cell lymphomas may include variable numbers of T lymphocytes, and, the most common type in mice, pleomorphic lymphoma, frequently displays several cell types, shows a nodular appearance, and although originating most often from the B-cell lineage may include variable proportions of T lymphocytes. 7 Moreover, lymphoma could be seen in the perivascular space already populated by mixtures of lymphocytes. Thus, even mixed lymphocyte populations are an unreliable criterion for differentiation. Overall, there is not a single attribute by which lymphoid hyperplasia can be differentiated from lymphoma. Lymphomas tend to be larger, have more pronounced growth toward the perithymic tissue, and may have a prevalence of either B or T cells throughout the lesion. Regarding the criterion “growth beyond compartments,” this should be used with caution as lymphocytes settling in the perivascular space may extend to the perithymic tissue in lymphoid hyperplasia and in lymphoma. Presence of prominent postcapillary venules with high endothelium is evidence of active recirculation of peripheral lymphocytes, which is more indicative of lymphoid hyperplasia than lymphoma. However, as postcapillary venules are part of the normal perivascular space, their presence does not preclude a lymphoma. In general, early-stage and single-site lymphomas may be difficult to differentiate from lymphoid hyperplasia. Clearest evidence would be infiltrates of the same cell type in multiple organs. In theory, differentiation could be based on clonality investigations as lymphoid hyperplasia is generally considered a polyclonal, reactive change while a tumor should have a clonal origin. 7 However, it is technically difficult to show clonality in routine studies.
Regarding the differentiation from thymoma, which is a benign tumor originating from the thymic epithelial cells and containing variable amounts of lymphocytes, 7 it is easiest to make a decision based on the number and distribution of epithelial cells in the lesion by pan-CK IHC. The recommendation based on the present investigation would be not to diagnose thymoma in CD-1 mice without pan-CK IHC and diagnose lymphoid hyperplasia if there is any doubt.
Age-Related Involution, Lymphoid Hyperplasia, and INHAND Nomenclature
Published terminology regarding the INHAND nomenclature recommended the term “Involution, age-related” for aging atrophy, which is characterized by gradual loss of lymphocytes and decrease in organ size. Stress-related atrophy, which can be found in diseased/moribund animals, is termed as “Cellularity, decreased, lymphocyte.” 7 Increases in lymphocytes in the perivascular space of atrophic thymi accompanied by a loss of corticomedullary distinction should not be diagnosed in routine studies as a separate finding as this is part of “Involution, age-related” based on the results of the present investigation. Of note, grades of age-related involution do not primarily describe the loss of the lymphoepithelial compartment but rather the variable numbers of lymphocytes within the perivascular space (Figure 5B).
For lymphoid hyperplasia, the INHAND nomenclature recommends “Cellularity, Increased, Lymphocyte” as a descriptive term. B-cell accumulation in the medulla is described as a feature of “Cellularity, Increased, Lymphocyte.” 7 The finding should mention B-cell accumulation in the perivascular space instead of the medulla which is scarcely populated by lymphocytes according to our IHC results. It was surprising that the lymphoepithelial compartment in hyperplastic thymi was as atrophic as in those with “Involution, age-related.” The diagnosis of involution/atrophy or lymphoid hyperplasia is thus based on the cellularity of the perivascular space. This is not evident when only H&E slides are evaluated.
Thymic involution is characterized by a collapse of the lymphoepithelial compartment and expansion of the perivascular space. Thymic lymphoid hyperplasia, more frequently seen in females, was related to a marked increase in lymphocytes in the perivascular space with a high proportion of B cells. The nodular or follicle-like structures formed by lymphocytes pushing the perivascular space forward and toward medulla give H&E-stained sections the impression that this is a lesion of the medulla. Figure 6 illustrates as a schematic drawing the extension and protrusion of the perivascular space (yellow) toward the medulla (yellow, hatching lines) therefore pushing the border of the compartment with the basement membrane (brown) forward. Nevertheless, the compartments are still separated. The basement membranes shown in the schematic drawing have not been investigated in the present work. Depending on the three-dimensional orientation of a real specimen, these structures are difficult to demonstrate in sections. However, the perivascular space can be followed from the outside to the border between cortex and medulla in atrophic thymi. Figure 3A shows some compression of the lymphoepithelial compartment by the populated perivascular space. Expansive follicular structures are absent in this example of an atrophic thymus.
It is interesting to compare changes in the thymic perivascular space with other species. In humans, the perivascular space is small at birth and increases in size with age while the lymphoepithelial thymus undergoes involution. 10 According to this research, the perivascular space is filled with lymphocytes of a peripheral phenotype in youth up to an age of 15 years. Thereafter, lymphocytes decline and are substituted by adipose tissue. Because of the invariable presence of peripheral lymphoid cells in the perivascular space of humans, it has been hypothesized that the thymus is a chimeric lymphoid organ consisting of a primary lymphoid organ, which is the lymphoepithelial part, and a peripheral lymphoid organ represented by the perivascular space. 11 In human autoimmune disease, formation of lymphoid follicular structures with germinal centers is described in the perivascular space at the border between cortex and medulla.12-14 In mice, a peripheral lymphoid organ should not be postulated as the lymphoid tissue in the perivascular space, different from the human situation, has no zonal organization of T and B lymphocytes but rather an intermingling of T and B cells. However, clearly the traffic of lymphoid cells in the thymus includes more than entrance of precursor cells and release of mature T cells. At least in older animals, recirculation of lymphocytes plays a major role. In lupus-prone BWF1 mice, follicle-like B-cell accumulations have been observed in the perivascular space of the thymus with the onset of autoimmune disease along with an increase of thymic B cells, plasma cells, and follicular helper T cells. 15 Likewise, nonobese diabetic mouse models of autoimmune type I diabetes have been associated with a giant perivascular space containing T cells, clusters of B cells, and upregulation of cell migration–related molecules in the perivascular space.16,17 In the CD-1 mouse, thymic lymphoid hyperplasia, however, has not been associated with any immune-related disease. There is some evidence that immune cell subpopulations of natural killer cells, T-helper cells and cytotoxic T cells in female CD-1 mice undergo significant changes when comparing 2- to 6-month-old animals to animals at 7 to 8 months of age in immunophenotyping investigations. 18 In other experimental species, B-cell follicles have been reported to occur as a presumably normal constituent of the thymus including the healthy ferret and healthy beagle dog.19,20
The thymic vasculature forms a blood-thymus barrier in the cortex but is incomplete in the postcapillary venules at the corticomedullary junction and in the medulla and therefore macromolecules can access the thymus here.21,22 Postcapillary venules are entry sites of both homing progenitor cells and recirculating lymphocytes including B cells and support the egress of mature thymocytes.23-26 Postcapillary venules with a high endothelium are thus part of normal thymic morphology and function. In aging mice and in autoimmune disease, they are involved in lymphocyte migration resulting in increased lymphoid cellularity of the perivascular space. Hence, the venules are more abundant and well developed in thymic lymphoid hyperplasia of mice, whereas they are small in young animals. The cause of the changes remains unclear as several stimulatory players could potentially play a role such as local dendritic cells observed in the perivascular space and thymic medulla in humans, increases in B lymphocytes in the thymic cortex of aging mice with lower autoimmune regulator (AIRE) expression, or even stimulatory molecules and modifying cell types coming in with the circulation.27,28 Lymphoid hyperplasia is generally thought to represent a reactive change in secondary lymphoid organs such as the spleen. The descriptive term “Cellularity, increased, lymphocyte” reflects that lymphocyte increase is regarded as a consequence of recirculation rather than local proliferation. 7 In the thymus of mice, there is no good understanding what the stimulus might be and whether local proliferation may play a role in addition to recirculation. In both studies of the present investigation, there was no obvious correlation to increases in lymphocytes in other lymphoid organs or to major inflammation. Thus, the nature of a possible reactive change could not be determined by considering pathological findings in the totality of organs of a given animal. Unfortunately, functional parameters are not available in carcinogenicity studies. Laboratory parameters are often taken from subgroups or additional animals and immune parameters are not investigated given the wide variation that can be expected in long-term studies. Potentially, the perivascular space can form a lymphoid organ apart from the lymphoepithelial thymus as a primary lymphoid organ. However, the perivascular space is not in the same category of secondary lymphoid organs as lymph nodes or spleen. It may rather have a function in the interaction of primary and secondary lymphoid organs and the prevention of harm to the organism by autoimmune reactions or to present antigen to the thymocytes in a controlled manner.
Awareness that the perivascular space is a compartment on its own and potentially generates a specific microenvironment may thus be helpful in the understanding of lymphoid hyperplasia in aging mice and a starting point for further investigations.
Incidence of Lymphoid Hyperplasia, Malignant Lymphoma, and Thymoma
Lymphoid hyperplasia commonly occurs in older CD-1 mice (older than 60 weeks), more frequently in females and it has been described as the most common spontaneous finding in this organ (183 out of 1658 animals, 11%). 6 In study A, the first case of lymphoid hyperplasia in control animals occurred at 49 weeks of age, and indeed, in 70% of the cases, lymphoid hyperplasia in the thymus was observed in females. In addition to the 28 cases of thymic lymphoid hyperplasia investigated by IHC from study A, there were a further 32 animals with lymphoid hyperplasia in the thymus (not investigated by IHC) resulting in an incidence of 25.9%, which was about twice as many as were described by Bradley et al. 6 In study B, the first case of hyperplasia occurred in week 50 of the study but most hyperplasias were observed beyond week 72. The incidence of lymphoid hyperplasia was in the same range (22.5%) as for study A. Incidences in CD-1 mice from the RITA database are 2.2% in males and 6.5% in females with the highest incidence of 55.0% in an individual study. Differences in the incidences of various neoplasms, and this might include preneoplastic/hyperplastic lesions, may depend upon the source of animals, sex, genetic variation, diet, age at death, environmental conditions, and histological threshold criteria used by the pathologist. 29
In mice, neoplasms of the thymus were commonly T-cell lymphomas of thymic lymphocyte origin. 30 Lymphoma arising in the thymus usually also involved other lymphoid organs, mainly the spleen and lymph nodes, which was also seen in our studies. 6 Lymphocytic lymphoma tends to arise in the thymus or from circulating T lymphocytes and spreads to involve the spleen. Lymphoblastic lymphoma most often arises in the thymus or bone marrow and spreads to the spleen, lymph nodes, and other organs.3,6,31 Spreading to the spleen was also observed in the case of lymphoblastic lymphoma reported in this investigation.
Thymic lymphomas, exclusively the lymphocytic type, were described in five out of 891 control males (0.6%) and eleven out of 890 control females (1.2%). 32 In study A, we only saw thymic lymphomas in females with a frequency of 2.5%. Incidences of CD-1 mice from the RITA database were in the same range (0.5% in males, 1.9% in females) with a maximum occurrence of 20.0% in females within a study. For the other references, thymic lymphomas were not specified regarding their modifier or cell type. However, lymphomas in females were also more frequent.6,33,34 Willard-Mack et al. 7 mentioned that malignant lymphoma is usually the lymphoblastic type when only the thymus is involved. This cannot be confirmed for CD-1 mice by the RITA database as no case of lymphoblastic thymic lymphoma was recorded.
Thymomas are very rare in CD-1 mice. They were characterized by a neoplastic epithelial component with or without neoplastic lymphocytes.6,30 Tubules and epithelial cords showed a solid growth centrally located within the lobes of the thymus. 9 In our study, significant amounts of proliferating epithelial cells were shown by H&E in one case and by pan-CK IHC in two cases which is consistent with thymoma. Bradley et al. 6 described three cases out of 1658 control animals (0.18%) from 104-week carcinogenicity studies and four out of 608 animals (0.65%) from 80-week carcinogenicity studies. Baldrick and Reeve 33 found two cases of benign thymoma out of 292 controls (0.7%) and both were seen in females. In our studies, we found three cases of benign thymoma out of 325 controls (0.9%, all three in females) which is comparable with incidences reported in the literature. Incidences from the RITA database were in the same range in males (0.5%) and slightly higher in females (1.9%) with the highest incidence of 17.9% in females for one single study. The reason might be the variability among studies, which is observed for most tumor entities collected in the database. However, similarities of thymoma to lymphoid hyperplasia might have biased the panel decision in some cases when pan-CK IHC was not available. Very low incidences were reported in an early investigation by Maita et al. 32 with only one thymoma out of 1781 control animals (0.06%). Very old historical data are generally less acceptable for comparison and should be used with care due to changes in various parameters including, but not limited to, changes in environmental conditions, genetic changes within a given strain, and changes in diagnostic criteria.
In summary, our findings in CD-1 mice indicate that most of the proliferative lesions in the thymus (in the absence of any other systemic lymphoid neoplasms) are consistent with lymphoid hyperplasia especially in the absence of a uniform, clonal lymphocytic, or lymphoblastic cell population and the presence of follicle-like structures. Lymphoma should only be diagnosed in the case where a uniform lymphocyte population is seen on H&E-stained slides or a uniform expansion of T or B lymphocytes was shown by IHC. Thymoma should be considered in case of a clear proliferation of epithelial cells on H&E-stained sections or proliferating epithelial cells are shown by pan-CK IHC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
