Abstract
2′-methoxyethyl (MOE) antisense oligonucleotides (ASOs) tested in multidose intrathecal nonhuman primate (NHP) toxicity studies have consistently revealed the presence of single large vacuoles in pyramidal neurons of the hippocampus in the absence of any cellular response. Termed “macrovesicular,” these vacuoles were characterized by immunohistochemistry and transmission electron microscopy which showed that these vacuoles are dilated lysosomes in neurons containing accumulated ASO. Additionally, two NHP studies were conducted to investigate the role of tissue fixation on their histogenesis. In Fixation Study 1, 6 doses of 5 mg 2′-MOE ASO with a full phosphorothioate backbone were administered by lumbar puncture over 5 weeks; in Fixation Study 2, 5 doses of 35 mg 2′-MOE ASO with a mixed phosphorothioate/phosphodiester backbone were administered over 12 weeks. At necropsy in each study, brain slices were either immersion fixed in neutral buffered 10% formalin or Carnoy’s fixative; frozen at −80 °C; or perfusion fixed with modified Karnovsky’s fixative. Fixed samples were processed to paraffin, sectioned, and stained with hematoxylin and eosin (H&E) and compared with H&E cryosections prepared from the frozen tissue of the same brain. The presence of vacuoles in fixed brain tissue but never in fresh frozen tissue showed that they arose during postmortem tissue fixation, and as such represent a processing artifact that is not relevant to human safety assessment of intrathecally administered 2′-MOE ASOs.
Introduction
Antisense oligonucleotides (ASOs) are an emerging class of biotherapeutic designed to treat diseases of known genetic origin by modifying gene expression in various ways. Antisense oligonucleotides show particular promise in the treatment of certain neurological conditions; however, following systemic (intravenous) administration, they do not appreciably cross the blood–brain barrier into the central nervous system (CNS). Therefore, the treatment of CNS-associated diseases requires direct administration of the ASO into the cerebrospinal fluid (CSF) via intrathecal (IT) administration to achieve spinal cord and/or brain tissue concentrations that are sufficient to elicit the intended therapeutic effect.
In the associated nonclinical development programs for these ASOs, the nonrodent species of choice is the nonhuman primate (NHP) as they are most representative of humans with regard to tissue distribution, metabolism, and sensitivity to class-related effects, and are widely accepted by regulatory agencies as the nonrodent species for nonclinical toxicity testing. Histopathological evaluation of the CNS from NHP studies for 7 different 2′-methoxyethyl (2′-MOE) ASO programs regularly revealed the presence of single, large, clear, “macrovesicular” cytoplasmic vacuoles in neurons of the hippocampus (Figure 1), suggesting this is a class effect unrelated to a specific gene target or activity in the NHP. Similar cytoplasmic vacuolation has been previously observed in the proximal tubular epithelium of the kidney in monkeys administered 2′-MOE ASOs by the subcutaneous route. 1,2 In these cases, the vacuoles were frequently seen in association with basophilic granules which were shown by immunohistochemistry (IHC) to be accumulated ASO, consistent with the kidney being the main route of excretion, while transmission electron microscopy (TEM) revealed granules free in the cytoplasm and contained within vacuoles (possibly endosomal/lysosomal). 3 Investigation into the mechanism of development of the renal vacuolation suggested that it was a postmortem artifact caused by an osmotic effect arising from immersion of the tissue in aqueous fixative.
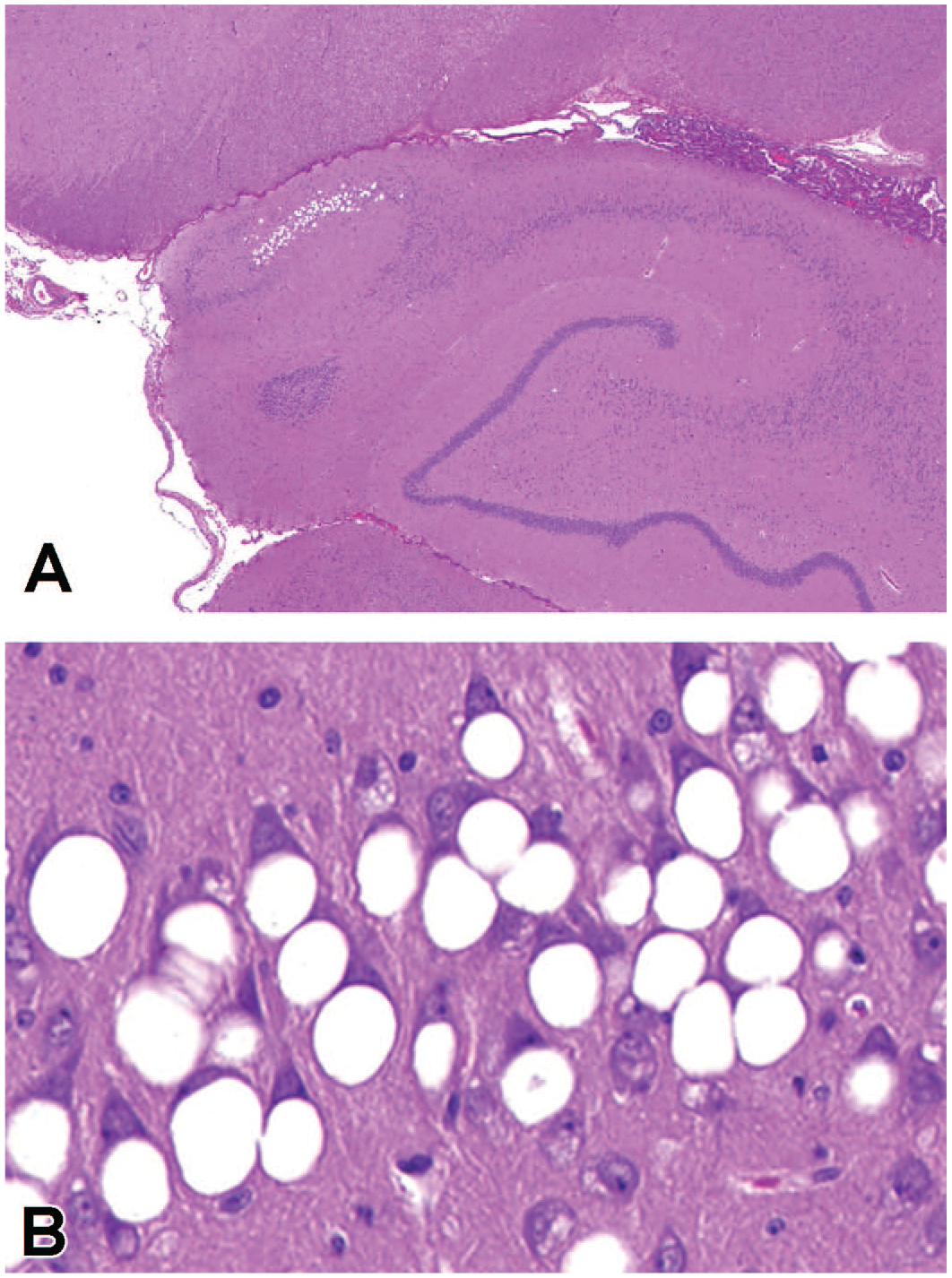
Hippocampus of a nonhuman primate (NHP) which was administered 5 doses of 35 mg antisense oligonucleotide (ASO) via lumbar puncture over 13 weeks in a toxicity study. Large vacuoles are observed in pyramidal neurons of the cornu ammonis region 1 (CA1) (A). At higher magnification, single, large, clear, “macrovesicular” vacuoles in neurons frequently displace the cell nucleus to the cell periphery (B). Hematoxylin and eosin (H&E). A, Original objective ×4; B, original objective ×40.
The hypothesis that a similar mechanism was responsible for the origin of the macrovesicular neuronal vacuolation observed in the hippocampus was investigated in studies examining the effects of tissue fixation on the brains of NHPs administered ASOs by the IT route. Two investigative studies were performed using 3 different ASOs which were either active or inactive in the NHP to determine if the vacuolation was related to activity on the gene target, and comparing the results of 3 different fixation methods with fresh frozen tissue. Neuronal vacuolation was observed following fixation by all 3 methods, but never in the fresh frozen brain tissue. These results suggest that macrovesicular neuronal vacuolation in the hippocampus is also an artifact arising during postmortem fixation and is related to the physicochemical properties of the ASO independent of its activity.
Materials and Methods
Intrathecal Antisense Oligonucleotide Toxicity Studies in the Nonhuman Primate
All toxicity studies and the investigative fixation studies were conducted in accordance with institutional principles and government regulations at facilities accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, and where appropriate, the United States or German Animal Welfare Acts. Furthermore, the toxicity studies were conducted in compliance with the relevant International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use guidelines, European Economic Community directives, and in accordance with German, US Food and Drug Administration, and Organisation for Economic Co-operation and Development principles of Good Laboratory Practice (GLP). All studies were preapproved in advance by the relevant Institutional Animal Care and Use Committee (or equivalent body in Germany).
Eleven toxicity studies ranging from 13 to 53 weeks in duration of dosing, with a 12- to 26-week recovery period, were conducted using cynomolgus monkeys (Macaca fascicularis) of Asian (Chinese or Vietnamese) origin (Table 1). Studies of shorter duration did not routinely include full histologic examination of the brain and spinal cord and were therefore not included in this review. Animals were 2 to 3 years old at study start, with body weight ranges of 2 to 8 kg in males and 2 to 6 kg in females. Animals were pair-housed in stainless steel cages with wooden chips on the flooring, and provided with environmental enrichment (eg, mirrors, colored plastic balls or tools, foraging devices). They were fed a certified lab diet for primates (LabDiet 5048, PMI Nutrition International, LLC) twice daily, supplemented with fresh fruit and vegetables. Ad libitum tap water was provided. Water was purified by reverse osmosis and ultraviolet irradiation, or by automated filter system. Housing was in climate-controlled rooms with a minimum of 8 air changes/hour, maintained at an ambient temperature of 19 °C to 25 °C and 40% to 70% relative humidity. There was artificial lighting under a 12 hours light/12 hours dark cycle.
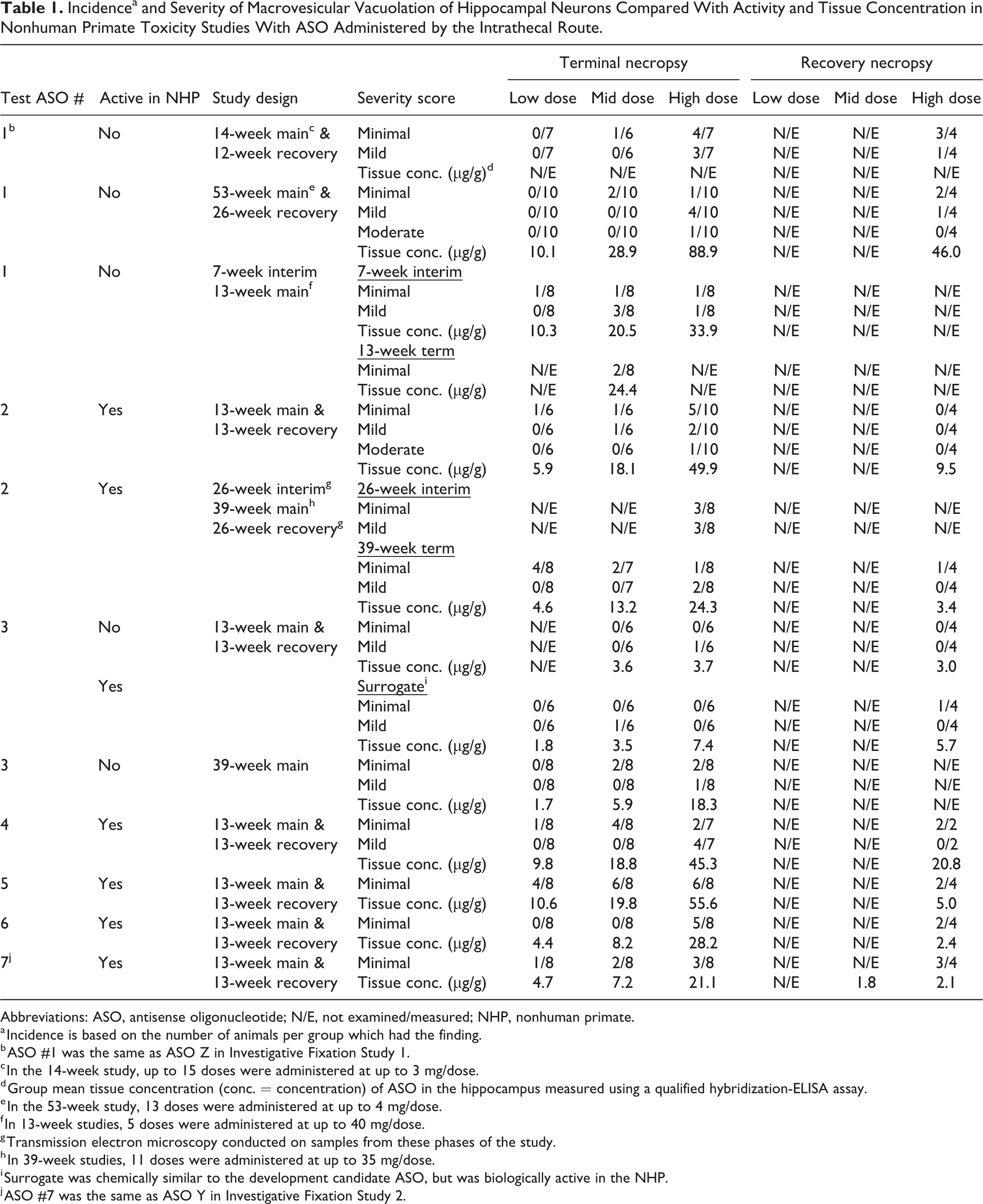
Incidencea and Severity of Macrovesicular Vacuolation of Hippocampal Neurons Compared With Activity and Tissue Concentration in Nonhuman Primate Toxicity Studies With ASO Administered by the Intrathecal Route.
Abbreviations: ASO, antisense oligonucleotide; N/E, not examined/measured; NHP, nonhuman primate.
a Incidence is based on the number of animals per group which had the finding.
b ASO #1 was the same as ASO Z in Investigative Fixation Study 1.
c In the 14-week study, up to 15 doses were administered at up to 3 mg/dose.
d Group mean tissue concentration (conc. = concentration) of ASO in the hippocampus measured using a qualified hybridization-ELISA assay.
e In the 53-week study, 13 doses were administered at up to 4 mg/dose.
f In 13-week studies, 5 doses were administered at up to 40 mg/dose.
g Transmission electron microscopy conducted on samples from these phases of the study.
h In 39-week studies, 11 doses were administered at up to 35 mg/dose.
i Surrogate was chemically similar to the development candidate ASO, but was biologically active in the NHP.
j ASO #7 was the same as ASO Y in Investigative Fixation Study 2.
In the 13-week studies, the ASO test item or vehicle reference item, artificial cerebrospinal fluid (aCSF; Harvard Apparatus or R & D Systems) was administered by intrathecal injection on study days 1, 14, and 28, and then every 4 weeks for a total of 5 doses, up to a maximum of 40 mg/dose. Some animals then underwent a 13-week dosing-free recovery phase. In the 14-week study, the low- and mid-dose groups were administered the ASO on days 1, 8, 15, 22, 29, 43, 57, 71, 85, and 99 (total 10 doses), while the high-dose group was dosed on days 1, 8, 15, 22, 29, 36, 43, 50, 57, 64, 71, 78, 85, 92, and 99 (total 15 doses; 3 mg/dose). There was a subsequent 12-week recovery period for the high-dose group. In the 39-week studies, doses were administered on study days 1, 15, 29, and then every 4 weeks for a total of 11 doses, up to a maximum of 35 mg/dose. This was followed by a 26-week recovery phase for some animals. In the 53-week study, animals were dosed once weekly on days 1, 8, 15, 22, and 29, and then once every 6 weeks on days 71, 113, 155, 197, 239, 281, 323, and 365 (total 13 doses; maximum 4 mg/dose). This was followed by a 26-week recovery phase.
For IT ASO administration, the NHPs were fasted, then anesthetized using ketamine (intramuscular, 2.5-10 mg/kg) and (dex)medetomidine (Dexdomitor; Zoetis; intramuscular, 0.02-0.06 mg/kg), supplemented with isoflurane (Isoflurane USP; Fresenius Kabi) when necessary. In preparation for the lumbar puncture, the puncture site was typically shaved, cleaned, and disinfected using a sterile antiseptic solution (Chloraprep, BD), or Kodan Spray (Schülke & Mayr GmbH), and/or Betaisodona (Mundipharma AG). A lumbar puncture at L3-L4, L4-5, or L5-6 was performed using a 25 or 50 mm long 25G Pencan spinal needle (B. Braun). Between 0.7 and 1.2 mL of CSF was withdrawn, and then the ASO test article (1 mL) was infused over 1 minute as a slow bolus, followed by a slow bolus 0.25 mL aCSF flush. Intramuscular atipamezole (Antisedan; Zoetis) was used as an anesthetic reversal agent following lumbar puncture.
The ASO test article was provided preformulated or as stock concentrate (50-75 mg/mL, to be diluted with aCSF), and stored at 1 °C to 8 °C, protected from light, prior to use. On the day of dosing, formulations were held at room temperature (RT) for 30 minutes prior to dosing.
During the course of each study, in-life assessments were performed including cage-side observations and food evaluation (daily); morbidity and mortality (twice daily); clinical signs and body weights (every 2 weeks). Physical and neurological examinations were performed once during the prestudy period, and on each dosing day, before and after dosing. They were also conducted in the last week of the recovery phase. The neurobehavioral and neurological examinations included assessments of general attitude/mentation, motor function, postural reactions, and cranial and spinal nerve function. Ophthalmic examinations were conducted once during the prestudy period and during the last week of the main and recovery phases. Electrocardiology and blood pressure measurements were made once during the prestudy period and on the first and last dosing day, predose and within 23 to 25 hours post-dose.
Samples of blood were collected for clinical pathology (hematology, coagulation, and clinical chemistry) prior to study start, 2 days after the last dose, and at the end of the recovery phase. Blood was also collected for bioanalytical (ASO concentration) and toxicokinetic analyses at multiple time points. Cerebrospinal fluid was collected for cell count, clinical chemistry, bioanalytical, and toxicokinetic analyses (prestudy and prior to terminal and recovery necropsy). Urine was collected for urinalysis prestudy, 2 days after the last dose, and at the end of the recovery phase.
Results of the in-life and clinical pathology analyses are not provided based on the focus of the paper, although it is important to note there were no neurobehavioral/neurologic signs associated with the neuronal vacuolation nor changes in hematology, coagulation, clinical chemistry, urinalysis, CSF cell count, or CSF clinical chemistry parameters indicative of neuronal toxicity.
One week after the last dose, animals were euthanized by intravenous sodium pentobarbital (Euthanyl; Bimeda-MTC) injection after sedation by intramuscular injection of ketamine hydrochloride (Ketalean; Bimeda-MTC), and submitted for necropsy. A complete necropsy examination was performed, which included evaluation of the carcass and musculoskeletal system; all external surfaces and orifices; cranial cavity and external surfaces of the brain; and thoracic, abdominal, and pelvic cavities with their associated organs and tissues. Organ weights were measured, and tissue samples collected for subsequent histopathological examination, as specified in the study protocols. The brain and spinal cord were each harvested intact; the brain was dealt with as described below, while sections of the spinal cord were collected from the cervical (C1-3), thoracic (T6-8), and lumbar (L1-2, L3-S1) regions, and immersion fixed in neutral buffered 10% formalin (NBF) for histopathological evaluation, or frozen (−80 °C) for bioanalytical and pharmacodynamic evaluations.
Fresh brains were sliced coronally at 4 mm using a metal brain matrix (Model MBM-2500C; ASI Instruments Inc), and then separated into left and right hemispheres by a sagittal section through the midline longitudinal fissure. The slices from the left hemibrain were immersion fixed in NBF at RT, while slices from the right hemibrain were frozen on dry ice and stored at −80 °C ± 10 °C for subsequent bioanalytical analysis using a qualified hybridization-ELISA assay, and pharmacodynamic analysis using polymerase chain reaction for mRNA, and immunoassay, for example, Singulex Erenna (Millipore Sigma) for protein analysis. The NBF was a commercial formulation containing 3.7% formaldehyde with 1.0% to 1.5% methanol as a stabilizer (Cochimbec Inc or Pioneer Research Chemicals Ltd). Following fixation for 2 to 7 days, 7 brain section blocks from 5 levels were selected from the left hemibrain for microscopic evaluation based on the STP position paper: recommended practices for sampling and processing the nervous system. 4 Brains were processed using a 23-hour protocol (through ascending alcohols and xylene) and then embedded in paraffin, sectioned, mounted on glass slides (Fisherbrand Superfrost Plus, Thermo Fisher Scientific, or using chrome alum gelatin as an adhesive), and stained with hematoxylin and eosin (H&E) for histopathological examination.
In addition, paraffin sections of hippocampus were stained with Fluoro-Jade B or C to identify any neuronal degeneration. 5,6 In both methods, the staining was conducted in the dark. Following treatment with 0.06% potassium permanganate solution for 15 to 30 minutes, slides were rinsed in deionized water and placed in a freshly prepared working solution of 0.001% Fluoro-Jade B or C in 0.1% acetic acid for 30 or 40 minutes, respectively. Slides were then rinsed with 3 changes of deionized water, and dried in an oven or slide dryer at 37 °C, before clearing by immersion in xylene and mounting in a nonaqueous, nonfluorescent mounting medium such as DPX (Sigma-Aldrich, Inc). Slide examination was performed using a fluorescence microscope with a filter suitable for visualizing fluorescein isothiocyanate.
All slides were examined by a board-certified (Diplomate, American College of Veterinary Pathologists, DACVP) veterinary pathologist and the findings subjected to peer review by a second board-certified (DACVP) veterinary pathologist. Examination was conducted in a nonblinded fashion as is common practice in GLP toxicity studies. Microscopic findings were recorded in an electronic database using either Pristima or Provantis using a nominal six-point grading scale (within normal limits, minimal, mild, moderate, marked, severe), although in reality the highest two grades were never applied in these studies. Grading criteria for hippocampal neuronal macrovesicular vacuolation were uniformly applied across all studies and were based solely upon the number of affected neurons, thus: within normal limits (0); minimal (1-30); mild (30-60); moderate (60-120); marked (120-180); severe (>180). Consistency of grading across studies was also confirmed by peer review.
Immunohistochemistry
Hippocampal sections from selected studies were further examined using non-GLP IHC techniques to confirm the identity of the vacuolated cells observed by H&E. Thus, neuronal nuclear protein (NeuN) staining was used to identify neurons; myelin basic protein (MBP) staining was used to identify oligodendrocytes; glial fibrillary acidic protein (GFAP) staining was used to identify astrocytes; and connexin 43 (Cx43) was used as a secondary marker for microglia. To confirm lack of a cellular response to the vacuolation, immunohistochemical staining for the microglial marker ionized calcium binding adaptor molecule-1 (Iba-1) was performed, along with GFAP and Cx43. Immunohistochemistry was also used to detect ASO, and lysosome-associated membrane protein-2 (LAMP2), an endolysosomal marker, was employed to help identify the nature of the macrovesicular vacuoles.
For each tissue marker, formalin-fixed paraffin-embedded, 4 to 5 µm thick serial hippocampal tissue sections mounted on Millennia 1000 Adhesion Slides (StatLab) were deparaffinized, rehydrated, and stained using a Ventana Discovery Ultra immunostainer (Roche Diagnostics). For the NeuN, MBP, GFAP, Iba-1, Cx43, and ASO staining, antigen pretreatment was performed using 0.1% trypsin (T8003 Sigma; 5 mL 0.5% aqueous trypsin, 5 mL 1% CaCl2, and 20 mL water) and samples were incubated at 37 °C for 16 minutes. Slides were blocked using an endogenous biotin blocking kit (Roche 760-050) for 8 minutes at RT. Nonspecific antibody binding was blocked with normal goat serum at a 1:20 dilution (Jackson Immunoresearch, 005-000-121) for 4 minutes at RT. The primary antibody was applied to slides at an optimized dilution (Discovery antibody diluent; Roche 760-108) and slides were incubated at 37 °C for 1 hour. (The optimized dilution of antibody was that which gave the best signal-to-noise ratio while also minimizing nonspecific background staining.) After two, 30-minute buffer washes (Reaction Buffer, Ventana 950-300), the slides were incubated with the biotinylated or horseradish peroxidase (HRP) goat anti-rabbit secondary antibody at 37 °C for 1 hour, then developed with DAB Map Kit (Roche 760-124) at 37 °C for 1 hour, and finally counterstained with hematoxylin.
The antibodies used were chicken polyclonal NeuN primary (1:800; Millipore ABN91) with a biotinylated goat-anti-chicken secondary antibody (Jackson Immunoresearch, 103-065-155); rabbit monoclonal MBP primary (1:3200; Abcam 218011) with an UltraMAP goat anti-rabbit HRP secondary antibody (Roche 760-4315); rabbit polyclonal GFAP primary (1:800; Dako Z00334) with a biotinylated goat-anti-rabbit secondary antibody (Jackson Immunoresearch, 111-065-003); rabbit polyclonal Iba-1 primary (1:3200; Wako 019-19741) with an UltraMAP goat anti-rabbit HRP secondary antibody (Roche 760-4315); rabbit polyclonal Cx43 primary (1:100; Cell Signaling 3512S) with a biotinylated goat-anti-rabbit secondary antibody (Jackson Immunoresearch, 111-065-003); and proprietary polyclonal rabbit anti-ASO primary antibody directed against the phosphorothioate backbone developed in-house (1:5000; Ionis) with a biotinylated goat-anti-rabbit secondary antibody (1:250; Jackson Immunoresearch, 111-065-003). Neutral buffered formalin fixed paraffin sections of liver from one control animal and one animal treated with ASO were used as negative and positive control tissues, respectively, for the ASO IHC, since the liver contained high concentrations of ASO as revealed by bioanalysis (data not shown).
For LAMP2, sections were subjected to antigen retrieval using either Proteinase K (Dako S3020) applied undiluted for 4 minutes at RT or ULTRA Cell Conditioning Solution (CC1; Ventana, 950-224) for 64 minutes at RT, followed by a rabbit monoclonal (H4B4) primary anti-LAMP2 antibody at 0.5 µg/mL (1:200; Abcam, ab25631) for 60 minutes at RT. The signal was detected with polymer-based secondary antibody Discovery OmniMap goat anti-Rb HRP (Roche 760-4311) and Discovery ChromoMap DAB kit (Roche 760-159). Neutral buffered formalin fixed paraffin sections of liver from one control animal were used as positive control tissue for the LAMP2 IHC.
Transmission Electron Microscopy
In the 39-week study conducted with ASO #2 (Table 1), samples of the hippocampus were collected at the interim necropsy (at 26 weeks) and the recovery necropsy (26 weeks after the 39-week dosing period), trimmed into approximately 1 × 1 × 2 mm pieces, and immersed in Karnovsky’s fixative with 0.2M cacodylate buffer (pH 7.4) at +5 °C ± 3 °C prior to transfer to 0.1M cacodylate sucrose storage buffer (5.0% sucrose wt/vol) for approximately 1 week. (The osmotic effect of sucrose aids the preservation of cytological detail post-fixation.) Tissues were then post-fixed in 1.0% osmium tetroxide solution for 90 minutes at ambient temperature, and dehydrated stepwise in increasing concentrations of ethanol (30%, 50%, 80%, 95%, 100%). Samples were processed and embedded in epoxy resin. Six epoxy blocks were prepared from each tissue, sectioned at one micron, stained with toluidine blue, and examined by light microscopy to identify representative areas of interest. Based on review of toluidine blue sections, selected blocks of hippocampus were trimmed for ultrathin sections (approximately 50 to 70 nm thickness), placed onto copper grids, and stained with uranyl acetate (2% at 60 °C), followed by Reynold’s lead citrate (at 60 °C). Image capture was conducted with a Philips CM 120 STEM (FEI) with a BioSprint side-mounted camera and an AMT Image Capture Engine using version 7.0.0.130 software (Advanced Microscopy Techniques, Corp). All TEM procedures were GLP compliant.
Investigative Fixation Studies 1 and 2
Two studies (Fixation Studies 1 and 2) were conducted to investigate the hypothesis that the macrovesicular vacuolation observed in the hippocampal neurons was an artifact arising from tissue fixation. In the first study, a single ASO was examined, but in the second, two different ASOs were used, one of which was active in the NHP while the other was inactive. This was to determine whether the vacuolation was related to activity on the gene target or simply due to the physicochemical properties of the ASO.
In Fixation Study 1, using 22-gauge noncoring Huber needles, catheters were placed into the IT lumbar space of 12 cynomolgus monkeys (6 male, 6 female) which were assigned to 2 groups (Group 1 and Group 2; 3/sex/group) based on the method of brain fixation intended at necropsy. All animals were administered once weekly IT bolus doses of ASO “Z” (the same as ASO #1 in Table 1), a 2′-MOE ASO with a full phosphorothioate backbone (not active in the NHP), at 5 mg/dose for a total of 6 doses (30 mg total dose). Each dose was administered as a slow bolus in a volume of 1.25 mL and flushed from the catheter system with a 0.50 mL slow bolus of artificial CSF. Doses were administered to all animals on days 1, 8, 15, 22, 29, and 36. In-life assessments consisted of food consumption (daily); body weights (weekly); morbidity and mortality checks (twice daily); clinical observations (twice daily); physical examinations (prior to surgery, prior to initiation of dosing, and prior to necropsy); and neurological examinations (prior to surgery, pre- and post-dosing on days 1 and 36). On day 50 (14 days after the 6th dose), all animals were euthanized, and samples were taken for postmortem examination.
At the time of necropsy, the brain from the Group 1 animals was sectioned in a metal brain matrix at 4 mm coronal slice thickness. Each coronal slice was further divided sagittally into right and left hemispheres. The odd numbered slices from the left hemisphere were immersed in RT NBF, a protein cross-linking fixative, for a minimum of 48 hours for histopathological evaluation. The even numbered slices from the left hemisphere were immersed in RT Carnoy’s solution (a protein precipitating fixative made up of 60% ethanol, 30% chloroform, 10% glacial acetic acid) for histopathological evaluation. All slices from the right hemisphere were frozen on dry ice and stored at −80 °C or below) prior to histopathological and bioanalytical evaluation.
The brain of the Group 2 animals was fixed in situ at necropsy by perfusion with modified Karnovsky’s fixative (2% paraformaldehyde/2.5% glutaraldehyde), another protein crosslinking fixative. Following perfusion, the brain was removed and held in toto in NBF for approximately 24 hours at ambient temperature. The brain was then sectioned in a metal brain matrix at 4 mm coronal slice thickness. Each coronal slice was further divided sagittally and all the slices were then immersed in NBF for subsequent histopathological evaluation.
Following fixation, all coronal sections of the left hemisphere from all animals were trimmed, processed, and embedded in paraffin, stained with H&E and examined microscopically. The appropriate coronal sections of the frozen right hemisphere from NHPs in Group 1 were trimmed to isolate the hippocampus, which was then cryosectioned, stained with H&E, and examined microscopically.
In Fixation Study 2, two 2′-MOE ASOs, “X” or “Y,” each having a mixed phosphorothioate/phosphodiester backbone were evaluated. These ASOs were selected to determine whether the vacuolation effect was related to any specific gene target or activity in the test species; hence ASO “X” (not represented in Table 1) was inactive in the NHP, while ASO “Y” (the same as ASO #7 in Table 1) was active. For ASO “X,” a total of 6 female cynomolgus monkeys were assigned, while for ASO “Y” there were 8. All animals were administered the ASO by the intrathecal route at a dose of 35 mg on days 1, 14, 28, 56, and 84 (175 mg total dose). The ASO was injected as a slow bolus over 1 minute at the L4-5 lumbar space via an atraumatic percutaneous needle (Pencan Paed pencil-point needles). The dose volume was 1 mL followed by a slow bolus 0.25 mL flush of artificial CSF. In-life assessments consisted of: food consumption (daily); body weights (weekly); morbidity and mortality checks (twice daily); clinical observations (cage-side—daily; detailed—weekly); physical examinations (prior to initiation of dosing and prior to necropsy); and neurological examinations (prior to initiation of dosing and post-dosing on dosing days). Necropsy was on day 91, 7 days after the last dose, with animals from each ASO group being equally split (3/3 ASO “X”; 4/4 ASO “Y”) for the purposes of brain collection, fixation, trimming, and staining similar to groups 1 and 2 in Fixation Study 1.
Although both Fixation Studies were designed to test a hypothesis—that macrovesicular vacuolation of neurons in the hippocampus is an artifact arising during postmortem fixation—and in theory would have warranted the H&E slides being examined in a blinded fashion, in reality it was not feasible to mask the unfixed, frozen sections which were readily identifiable compared to any of the paraffin sections prepared from fixed tissue.
For both Fixation Studies, the slides were examined and peer reviewed as described previously for the GLP toxicity studies.
Results
Intrathecal Antisense Oligonucleotide Toxicity Studies in the Nonhuman Primate
Table 1 shows the common occurrence of the macrovesicular vacuolation in hippocampal neurons across studies, independent of the mRNA target of the ASO. These vacuoles are usually found in the cytoplasm of the pyramidal neurons of the inferior hippocampus in a focal area of the cornu ammonis (CA) 1-3 region. By light microscopy, this alteration is characterized by a single large (up to 40 µm diameter), clear, apparently empty vacuole expanding the cytoplasm and sometimes displacing the cell nucleus to the periphery (Figure 1). Based on these characteristics, they have been termed “macrovesicular” as a descriptive qualifier. The size of the vacuoles usually enables them to be detected at a low magnification, for example, ×4 objective (Figure 1A), but in order to accurately count them for the purpose of grading, a higher magnification is used, for example, ×20 or ×40 (Figure 1B). There is typically a dose-related increase in incidence and/or severity of the finding, which most commonly is graded minimal to mild, and correlates with the concentration of ASO measured in the hippocampus, although hippocampal tissue concentrations associated with neuronal macrovesicular vacuolation at the end of each main study period varied by ASO (Table 1). In main study animals, the minimum group mean tissue concentration at which macrovesicular neuronal vacuolation was recorded ranged from 3.5 µg/g (ASO #3, 13-week main, surrogate group) to 28.9 µg/g (ASO #1, 53-week main).
Where studies have included a recovery arm, there is a clear trend to reversibility of this vacuolation based on decreased incidence and/or severity, corresponding with diminished ASO concentrations (Table 1). Not unlike main study results, the tissue concentration at which neuronal vacuolation was still observed in recovery animals also varied by ASO, and ranged from a group mean of 2.1 µg/g (ASO #7, 13-week main and 13-week recovery) to 46.0 µg/g (ASO #1, 53-week main and 26-week recovery).
Similar macrovesicular vacuolation has not been observed in any other tissue, for example, kidney or liver, in any of these studies in spite of measurable ASO accumulation in non-neural organs.
Immunohistochemistry
The vacuolated cells stained strongly with NeuN (Figure 2A), a common nuclear marker for neurons, confirming that the vacuolated cells were neurons. Myelin basic protein (Figure 2B) and GFAP (Figure 2C) did not stain the vacuolated cells, thus indicating they were neither oligodendrocytes nor astrocytes, respectively.
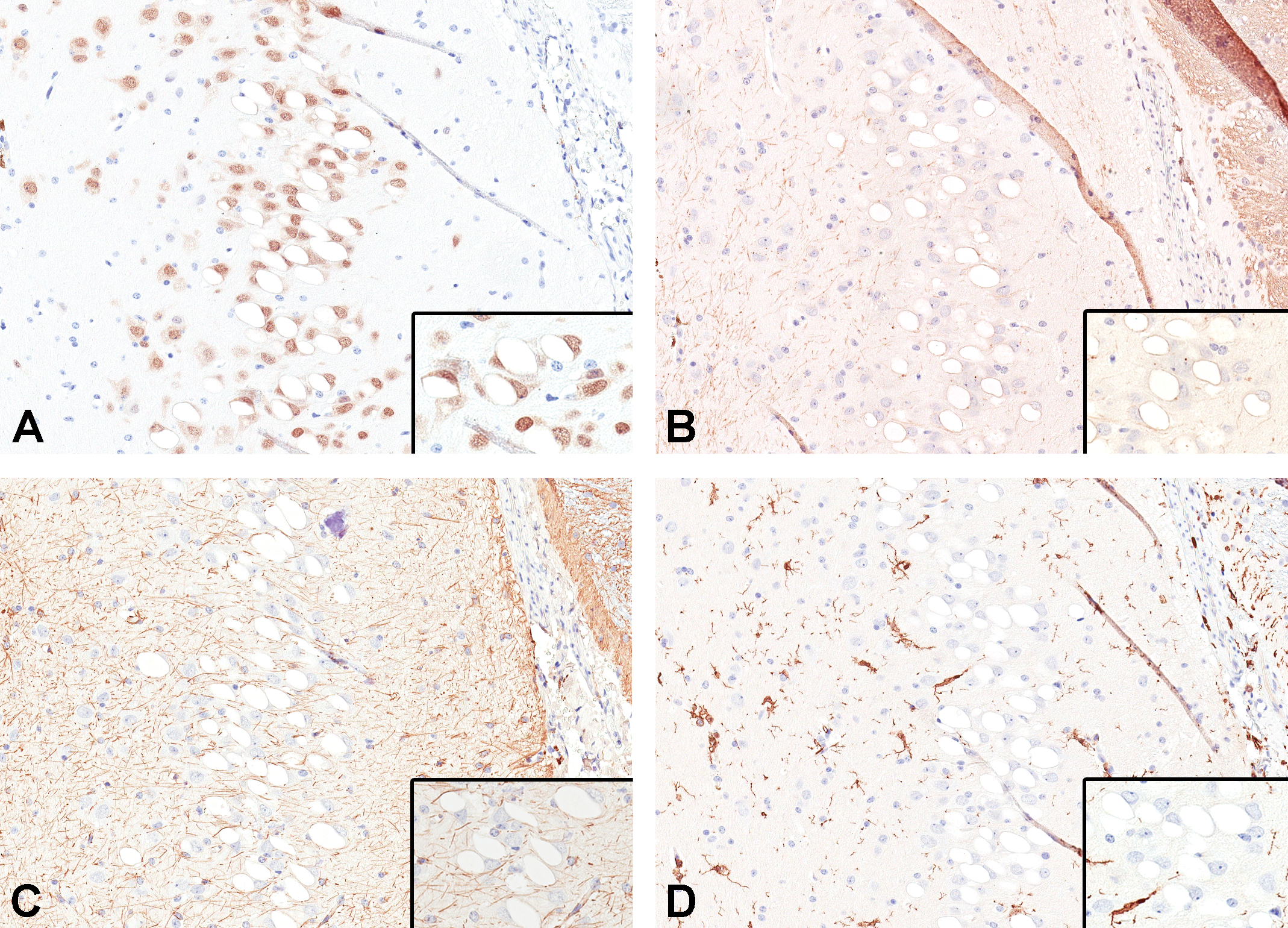
Immunohistochemical characterization of the vacuolated cells in the hippocampal CA1 region of a nonhuman primate (NHP) in a toxicity study confirmed their identity as neurons. (A) Neuronal nuclear protein (NeuN) shows positive staining of neuron cell nuclei. (B) Myelin basic protein (MBP) is negative indicating the vacuolated cells are not oligodendrocytes. (C) Glial fibrillary acidic protein (GFAP) is negative indicating the vacuolated cells are not astrocytes. The GFAP along with (D) ionized calcium binding adaptor molecule 1 (Iba-1) show no increased staining around the vacuolated neurons indicating there is no cellular response from astrocytes or microglia, respectively. A-D, original objective ×20.
The GFAP (Figure 2C), Cx43 (no staining—not shown), and Iba-1 (Figure 2D) staining demonstrated neither an increase in the number of astrocytes or microglia nor the presence of astrocytic/microglial processes around the vacuolated neurons, indicating the absence of a cellular response. The lack of a cellular response is consistent with the absence of any neuronal degeneration associated with the vacuolation (confirmed by the lack of Fluoro-Jade B/C staining—results not shown).
Staining for the presence of ASO was positive in the cytoplasm of vacuolated and nonvacuolated neurons (Figure 3A), with ASO present along the inner surface of the vacuoles and fine stippling in the cytoplasm indicating accumulation of ASO. Positive staining of the vacuole border with LAMP2 indicated that they were of lysosomal origin (Figure 3B). Stippled LAMP2 positive staining consistent with lysosomes was also present in the cytoplasm of neighboring nonvacuolated neurons.
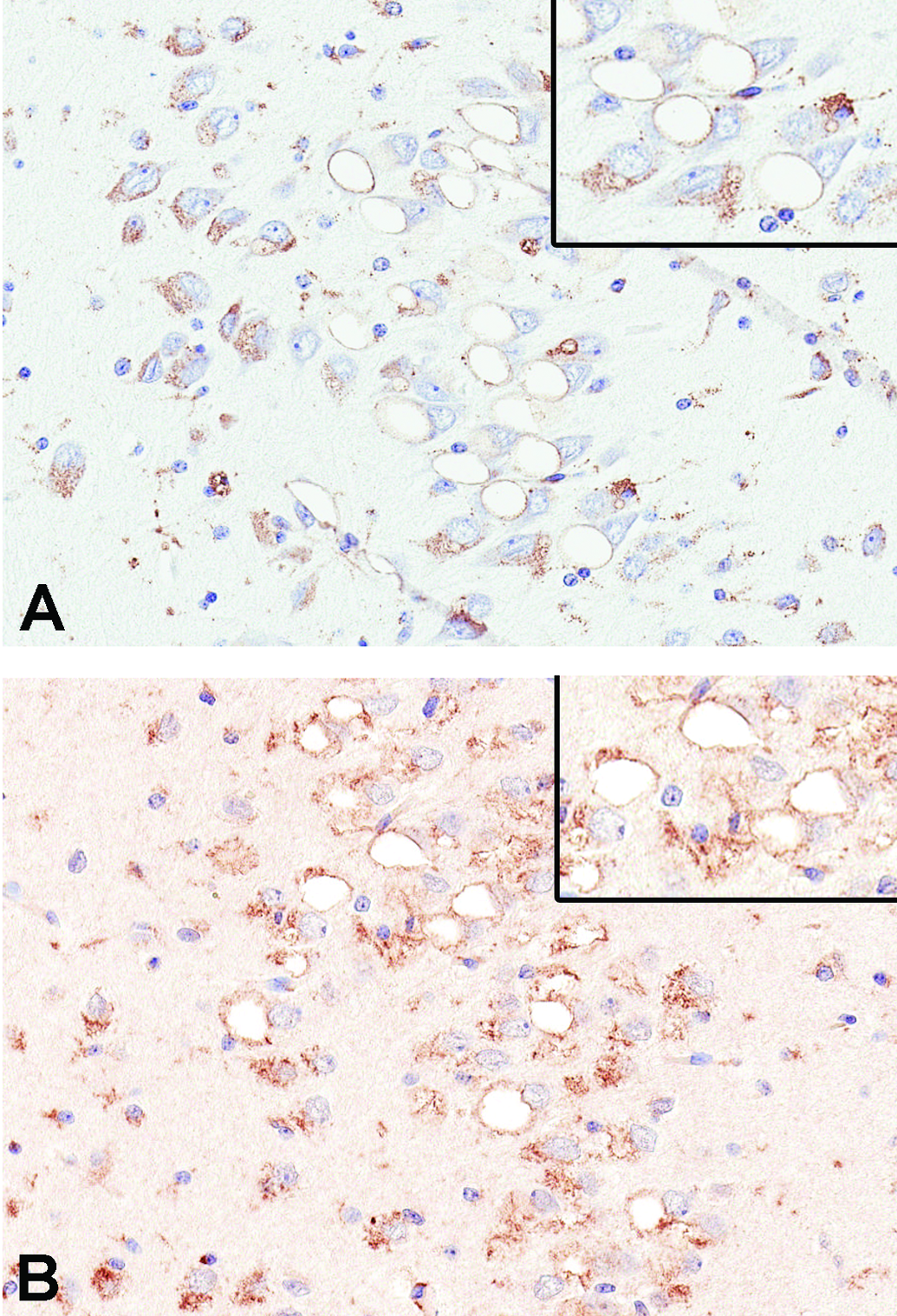
Immunohistochemical staining of the neuronal vacuoles of an NHP in a toxicity study indicated they arose from lysosomes which contained ASO. A, Positive staining by an anti-ASO antibody reveals the presence of ASO intimately associated with the vacuole limiting membrane. Finely stippled staining in unaffected neurons indicates the presence of ASO in them. B, Positive staining for lysosome-associated membrane protein 2 (LAMP2) of the outline of the vacuoles shows that they are dilated lysosomes. The presence of finely stippled cytoplasmic staining in unaffected neurons indicates the normal presence of lysosomes. A & B, original objective ×20.
Electron Microscopy
In the hippocampus, solitary, large, circular intracytoplasmic vacuoles were characterized ultrastructurally as having a single-layered limiting membrane and primarily a clear space centrally, with small quantities of variably electron-dense material localized primarily to the peripheral margin (Figure 4). This peripheral material was further characterized by a mixture of thin laminations of stacked and/or reticulated membranous structures, often oriented perpendicular to the outer limiting membrane, and interposed with smaller vesicles that contained lipid, fine flocculent/granular material, and/or empty myelinoid membranes. These large intracytoplasmic vacuoles were considered consistent with secondary lysosomes 7 and corresponded with the macrovesicular vacuolation of neurons observed by light microscopy. Some of the material within the enlarged lysosomes of neurons in the hippocampus may have represented accumulation of ASO, with/without lipidic material.
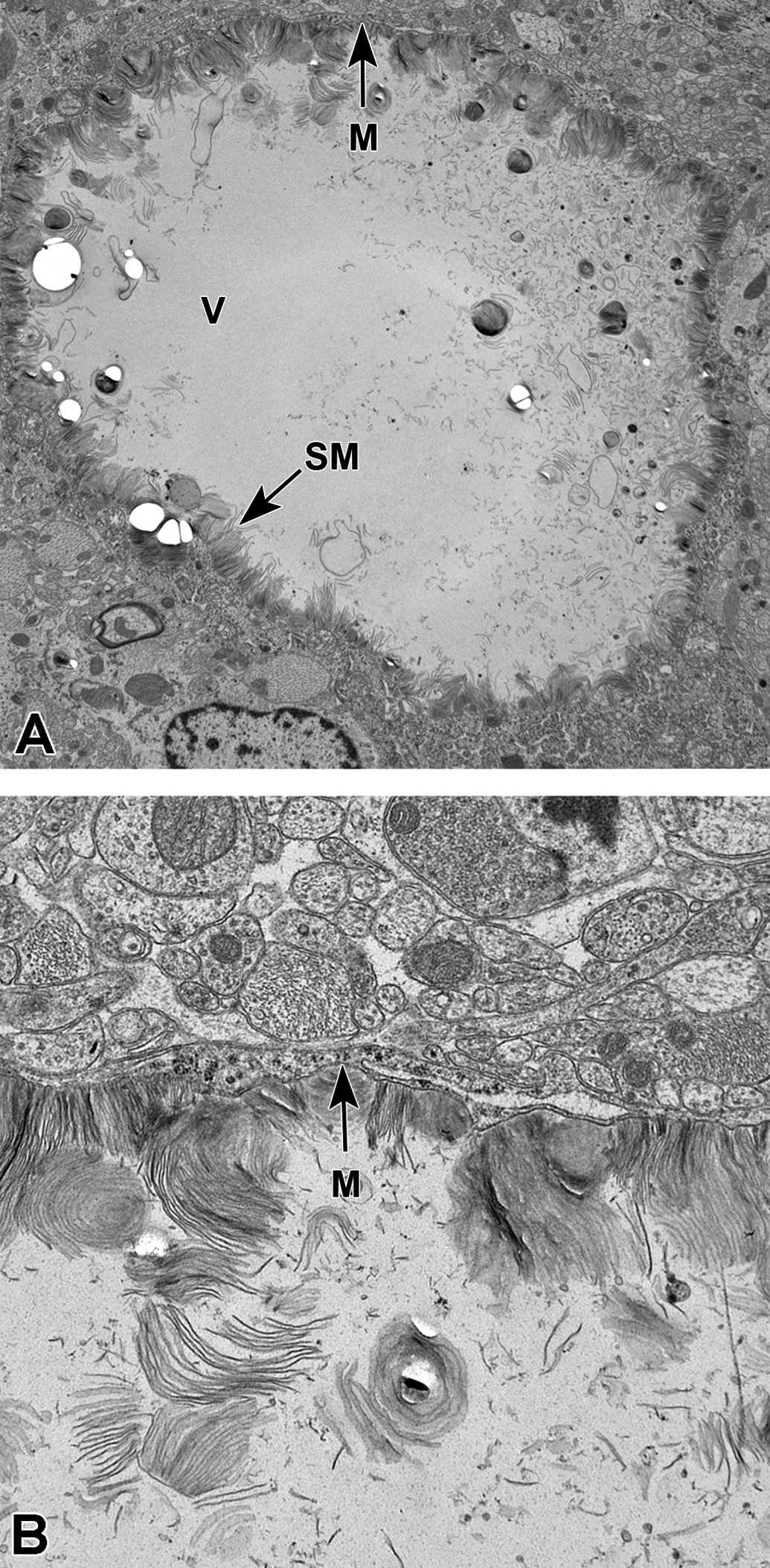
Transmission electron microscopy of a vacuole in an affected neuron from a nonhuman primate (NHP) in a toxicity study. A, Note the single, large, roughly circular, intracytoplasmic vacuole (V) lined by a single limiting membrane (M), consistent with a secondary lysosome. The vacuole contains clear space centrally and small quantities of variably electron-dense thin laminations of stacked membranous (SM) material along the peripheral margin. The nucleus (N) is present at the bottom of the image. Original magnification ×2000. B, At higher magnification, the single-limiting membrane (M) circumscribing the vacuole is clearly evident. Original magnification ×4000.
Investigative Fixation Studies 1 and 2
In Fixation Study 1, the brain fixed by NBF immersion revealed minimal macrovesicular neuronal vacuolation (Figure 5A) in the hippocampus in 3/6 monkeys (Table 2). No additional histological alterations were present in the brain. In the brain fixed by the alternative methods evaluated—immersion fixation in Carnoy’s solution (Figure 5B), perfusion fixation with modified Karnovsky’s solution (Figure 5C), or freezing at −80 °C or below (Figure 5D)—no vacuolation of the hippocampus or any other histological alterations were observed.
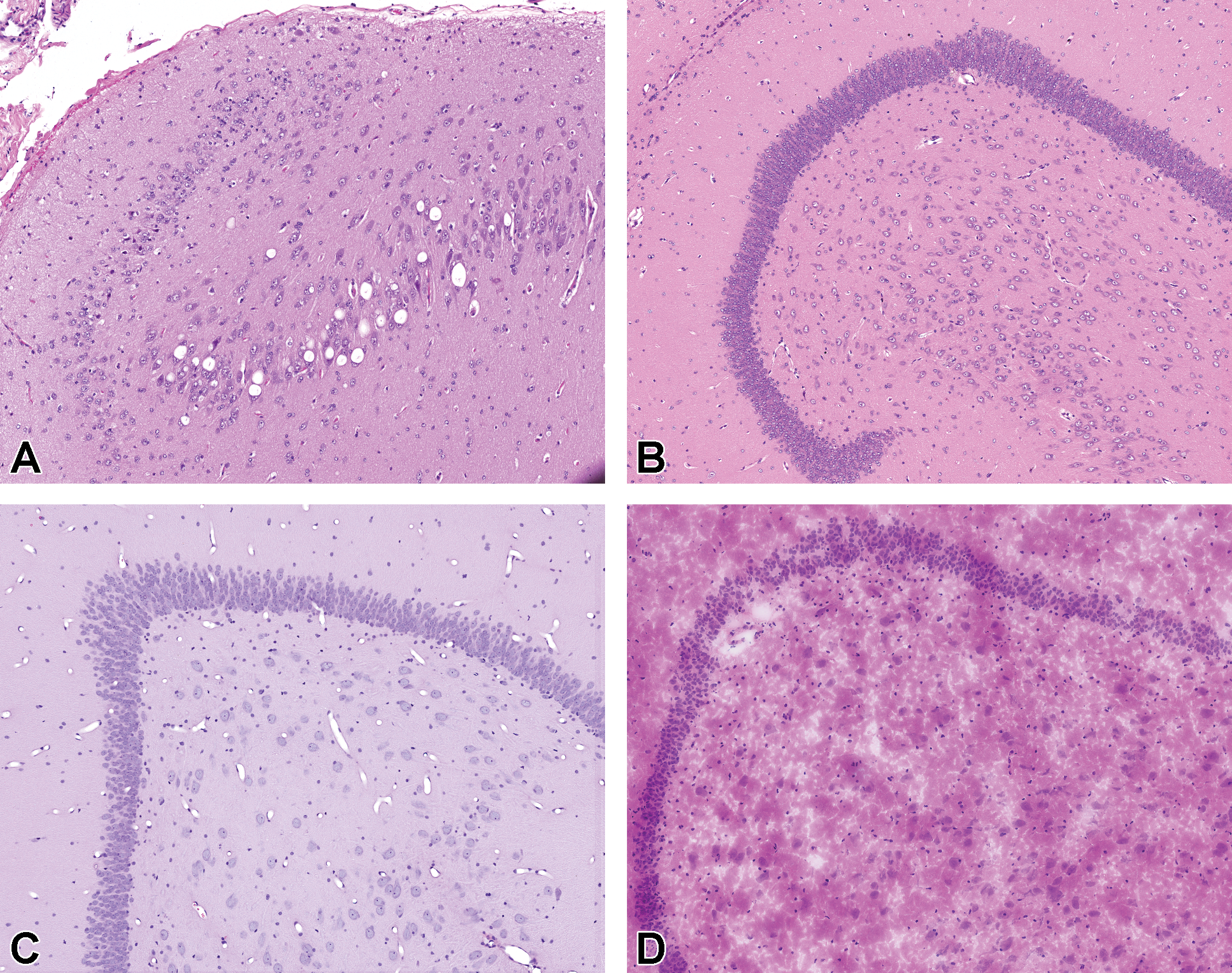
Results from Fixation Study 1 investigating the effects of fixation on vacuolation of hippocampal neurons, in which nonhuman primates (NHPs) were administered 6 weekly doses of 5 mg antisense oligonucleotide (ASO) via the intrathecal route. (A) Hippocampus immersion fixed in neutral buffered formalin (NBF) shows macrovesicular vacuolation in neurons. By comparison, no vacuolation was observed in hippocampus immersion fixed in Carnoy’s (B), perfusion fixed with modified Karnovsky’s (C), or in unfixed frozen tissue examined by cryosection (D). Hematoxylin and eosin (H&E). A-D, Original objective ×20.
Incidence and Severity of Macrovesicular Neuronal Vacuolation in the Hippocampus (Fixation Study 1).
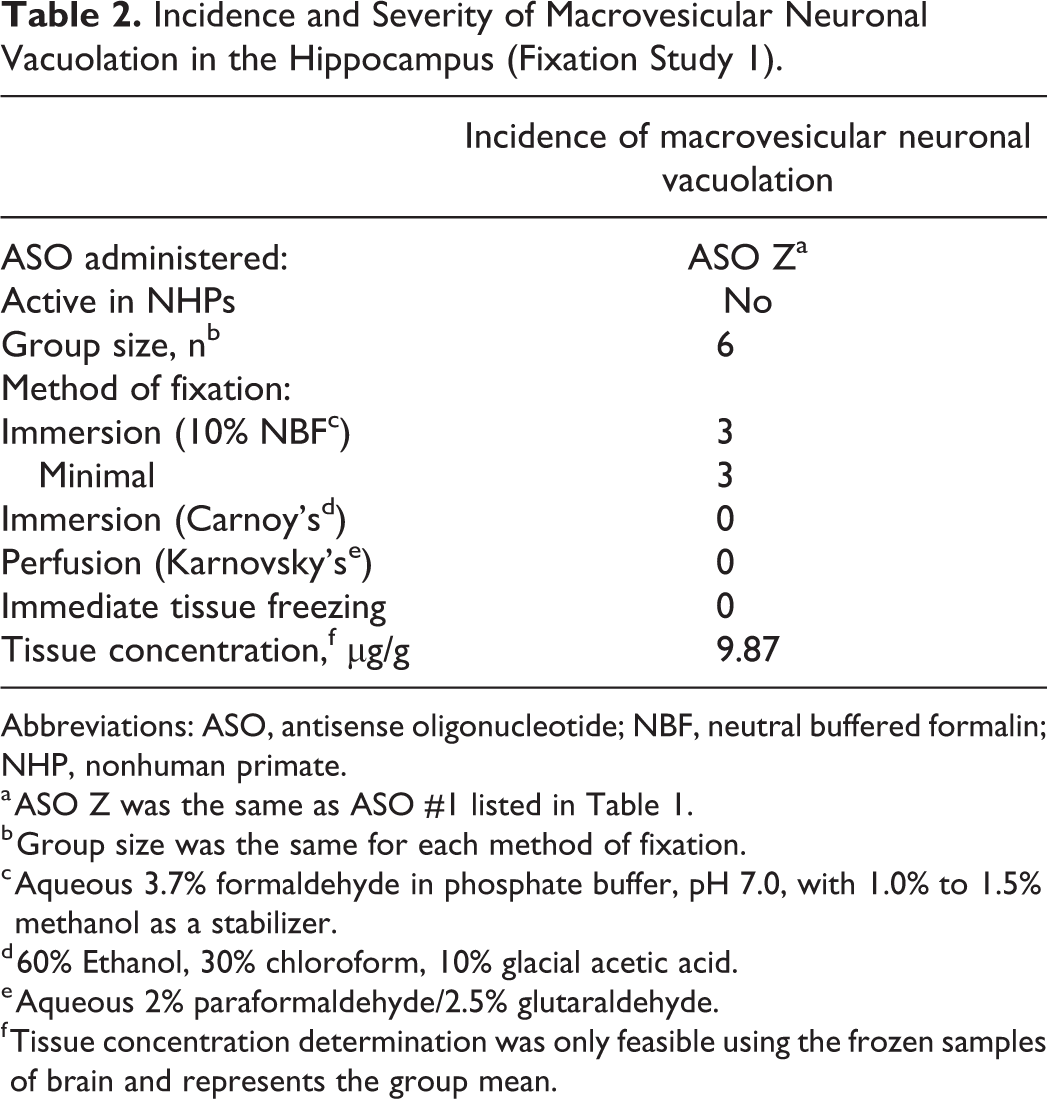
Abbreviations: ASO, antisense oligonucleotide; NBF, neutral buffered formalin; NHP, nonhuman primate.
a ASO Z was the same as ASO #1 listed in Table 1.
b Group size was the same for each method of fixation.
c Aqueous 3.7% formaldehyde in phosphate buffer, pH 7.0, with 1.0% to 1.5% methanol as a stabilizer.
d 60% Ethanol, 30% chloroform, 10% glacial acetic acid.
e Aqueous 2% paraformaldehyde/2.5% glutaraldehyde.
f Tissue concentration determination was only feasible using the frozen samples of brain and represents the group mean.
In Fixation Study 2, macrovesicular neuronal vacuolation was observed in brain samples subjected to fixation by both cross-linking and tissue precipitation methods but not in tissues frozen at the time of collection (Table 3). It was most consistently present in the pyramidal neurons of the hippocampus, but in a few instances was additionally observed in neurons of the cerebral cortex and/or pons. The incidence was greatest in the brains fixed by perfusion (with Karnovsky’s) where 7 out of 7 were affected (Figure 6A). The next most commonly affected were the brain samples fixed by immersion in Carnoy’s solution (5/7; Figure 6B), and least often affected were the immersion NBF-fixed brain samples (3/7; Figure 6C). Notably, as before, macrovesicular neuronal vacuolation was not observed in cryosections from the hippocampus of the brain samples that were frozen at the time of collection (thus not exposed to any fixative; Figure 6D).
Incidence and Severity of Macrovesicular Neuronal Vacuolation in the Hippocampus (Fixation Study 2).
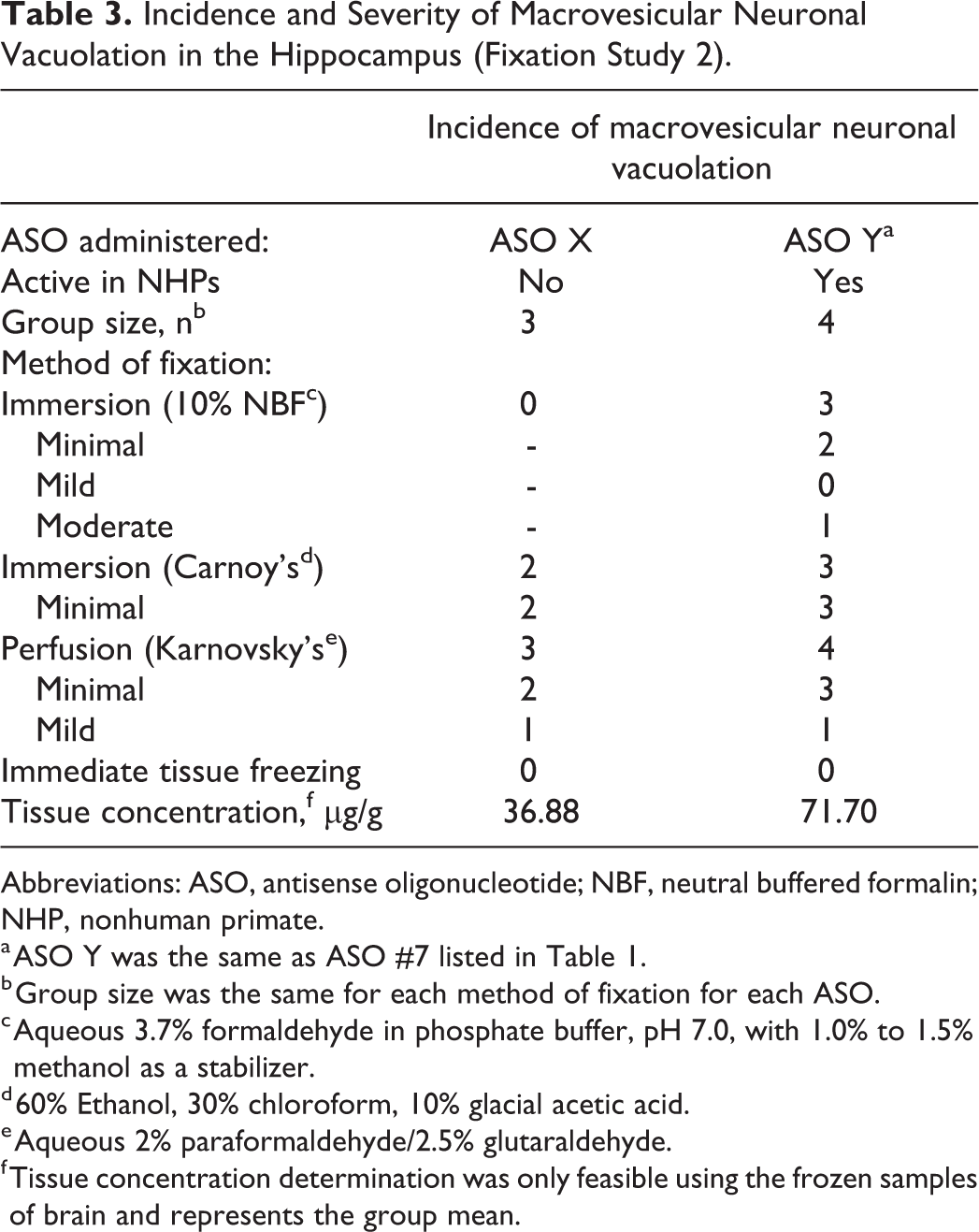
Abbreviations: ASO, antisense oligonucleotide; NBF, neutral buffered formalin; NHP, nonhuman primate.
a ASO Y was the same as ASO #7 listed in Table 1.
b Group size was the same for each method of fixation for each ASO.
c Aqueous 3.7% formaldehyde in phosphate buffer, pH 7.0, with 1.0% to 1.5% methanol as a stabilizer.
d 60% Ethanol, 30% chloroform, 10% glacial acetic acid.
e Aqueous 2% paraformaldehyde/2.5% glutaraldehyde.
f Tissue concentration determination was only feasible using the frozen samples of brain and represents the group mean.
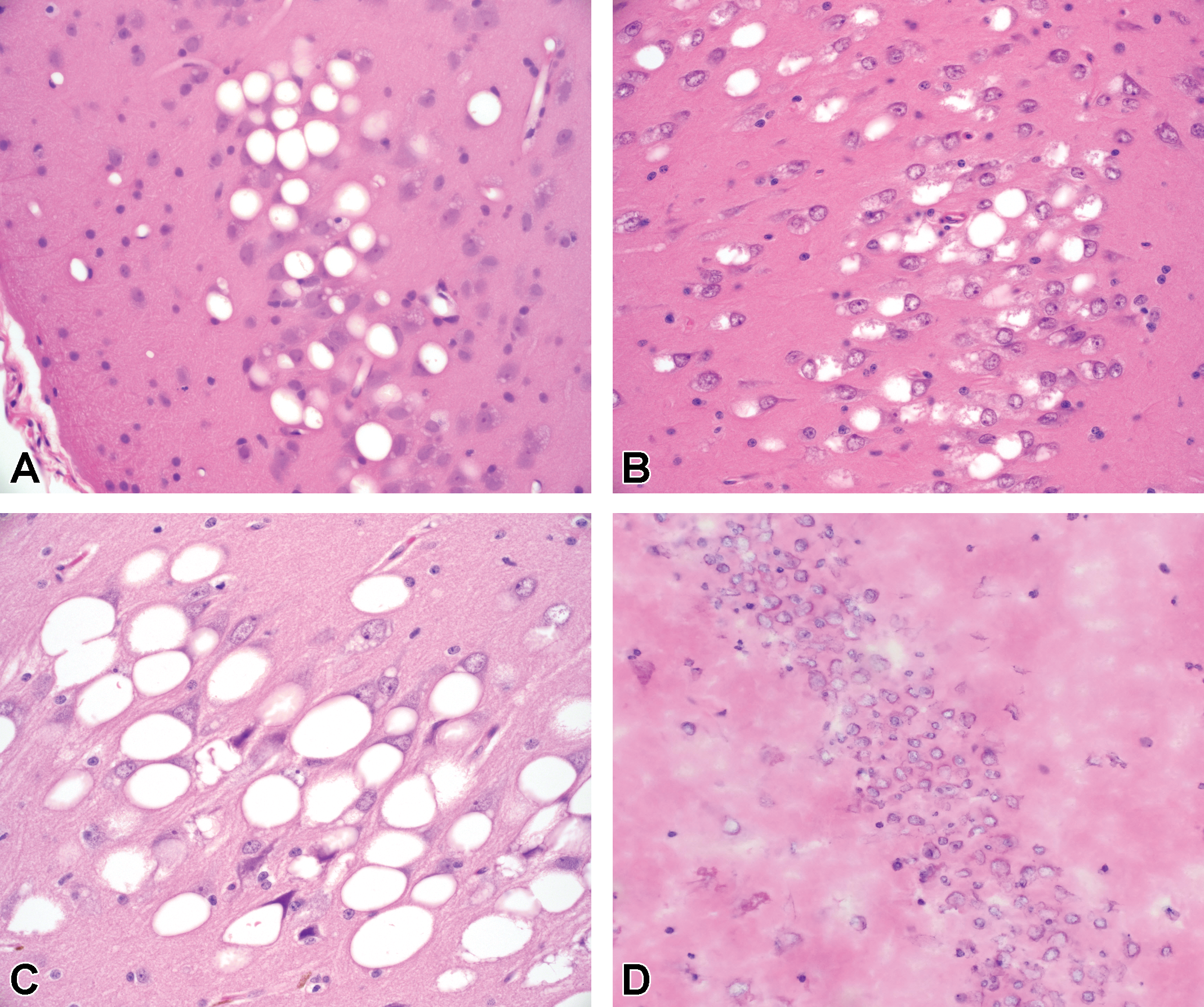
Results from Fixation Study 2 investigating the effects of fixation on vacuolation of hippocampal neurons, in which nonhuman primates (NHPs) were administered 5 doses of 35 mg antisense oligonucleotide (ASO) over 13-weeks via the intrathecal route. Macrovesicular neuronal vacuolation is clearly seen in hippocampus perfusion fixed with modified Karnovsky’s (A), immersion fixed in Carnoy’s (B), and immersion fixed in neutral buffered formalin (NBF) (C), but is absent in unfixed frozen tissue examined by cryosection (D). Images C and D represent tissue from contralateral hemispheres containing the same hippocampal neuron field of the same monkey. Hematoxylin and eosin (H&E). A-D, Original objective ×40.
Discussion
Results of the histopathological evaluation of the CNS from NHP IT studies of 13 to 53 weeks in duration for 7 different 2′-MOE ASO programs revealed the presence of single, large, clear, cytoplasmic vacuoles in neurons of the hippocampus, suggesting this is a class effect unrelated to a specific gene target or activity in the NHP.
The severity of the macrovesicular neuronal vacuolation is typically minimal to mild and does not appear to be notably more severe following 39 weeks of dosing compared to 13 weeks. Significantly, our results also show a trend to reversibility (based on reduced incidence/severity) following a typical 13- or 26-week recovery period, corresponding with decreases in drug concentration in the hippocampus (Table 1).
In one study (ASO #2 13-week study with 13-week recovery), full recovery (no neuronal macrovesicular vacuolation) was noted in all 4 animals even when the group mean hippocampal tissue concentration of 9.5 µg/g remained higher than the low dose in the main study, 5.9 µg/g, which did have minimal vacuolation in 1 of 6 animals. There are number of possible explanations for this apparent inconsistency, especially for vacuolation graded as “minimal,” including comparison of groups of different sizes (ie, smaller recovery group size than main); variation in individual animal response to the ASO; redistribution of the ASO within the tissue; changes in the character of the ASO over time; variable sampling and analysis of the tissue for both histopathology and bioanalysis. Of these explanations, there is clearly some variation in individual response to the ASOs as evidenced by the incidence of macrovesicular vacuolation in high-dose groups where it is rare to observe it in all treated NHPs (Table 1). Although variation in hippocampal tissue sampling cannot be entirely ruled out, the use of a brain trimming matrix would be expected to minimize any variation and contribute significantly to the overall consistency of brain tissue sections.
The presence of these vacuoles is not associated with neuronal degeneration/necrosis based on examination of H&E-stained sections and supported by negative Fluoro-Jade B/C staining, a method used to specifically identify neuronal degeneration. Nor is there any associated gliosis or inflammation based on H&E and confirmed by negative immunohistochemical staining for GFAP for astrocytes, and Iba-1 for microglia. The lack of any cellular response is consistent with the contention that the vacuolation arises postmortem as a processing artifact related to tissue fixation.
The neuronal vacuoles observed in the NHP toxicity studies have also been characterized by TEM which confirmed that the vacuoles were within neurons and showed they were dilated lysosomes containing thin laminations of stacked and/or reticulated membranous structures with granular electron-dense material along the peripheral margin. The character of the granular electron-dense material was consistent in location with ASO previously shown by IHC to be along the inner surface of the vacuoles (Figure 3A). Immunogold-TEM could have been employed in an effort to confirm the nature of the electron-dense material as being ASO, but was not pursued at the time based on strong histologic evidence from the IHC.
Cellular Uptake and Trafficking
The identification of the macrovesicular vacuoles as being dilated lysosomes is consistent with our current understanding of the cellular uptake and trafficking of ASOs. 8,9 The predominant mechanism of cellular uptake appears to be endocytosis following binding to cell surface proteins such as caveolin and clathrin. Uptake of ASO can occur in most tissues, but following systemic administration the highest levels are found in the liver, kidney, bone marrow, adipocytes (cell cytoplasm not lipid), and lymph nodes. 8 Sequestration is also evident in cells of the reticulo-endothelial system. 10 Following uptake, the ASO is present in an endosome which enters either a “productive” pathway whereby it exerts its intended pharmacologic effect by acting on the target RNA in the cytosol or nucleus, or else it enters a “nonproductive” pathway resulting in sequestration in lysosomes where it is hydrolyzed and metabolized 11 without associated toxicity. It should be noted that ASOs hydrolyze much more slowly than normal RNA due to the presence of phosphorothioate rather than phosphodiester linkages. This makes the ASOs more resistant to digestion by lysosomal esterases and allows them to accumulate. Thus, it would appear that the vacuoles we are seeing are most likely linked to the nonproductive pathway accumulation of ASO in lysosomes.
Fixation Effects
The results of the investigations on the influence of fixation on the occurrence of vacuoles in the hippocampus (Fixation Studies 1 and 2) are consistent with previously described changes in the kidney relative to the occurrence of vacuoles in the renal proximal tubular epithelium. 1,2 In the kidney investigation, formalin immersed tissue was compared to paraformaldehyde/glutaraldehyde (modified Karnovsky’s) perfused and frozen tissue. Vacuolation occurred in the proximal tubular epithelium of formalin fixed tissue in the same location as oligonucleotide and was characterized by the presence of single to multiple large, clear, cytoplasmic vacuoles. This demonstrated that the formation of vacuoles was dependent on the presence of oligonucleotide within the cell. Similarly, our IHC has demonstrated that oligonucleotide was distributed to the hippocampus and colocalized with the vacuolation seen in the hippocampal neurons of formalin fixed brain sections.
In the first of the two investigative fixation studies (Fixation Study 1), no vacuolation was found in the hippocampus with the rapidly penetrating Carnoy’s fixative, perfusion with Karnovsky’s, or frozen tissue preparations. However, in the second investigative fixation study (Fixation Study 2), vacuolation in the neurons of the hippocampus occurred with all 3 chemical fixation methods—immersion in formalin, perfusion with Karnovsky’s, and immersion in Carnoy’s fixative. This apparent discrepancy with the first study may simply be the consequence of differences in study design; in particular the total dose of ASO administered over 7 weeks in the first study was 30 mg, whereas in the second study a notably higher total dose of 175 mg was administered over 13 weeks. This is borne out by the difference in tissue concentration of the ASOs in the two fixation studies (Tables 2 and 3) which was approximately 3 to 7 times higher in the hippocampus of Fixation Study 2 than Study 1. Furthermore, in Fixation Study 2, the difference in tissue concentration between ASO X and ASO Y is reflected in the difference in incidence of the vacuolation (Table 3). These observations are consistent with the dose–responsive occurrence of vacuoles seen in repeated dose NHP toxicity studies. Most significantly, though, vacuolation was not observed in either study in the cryosections prepared from any of the frozen, unfixed, hippocampal tissue.
The presence of vacuoles in the hippocampus of monkeys treated with ASOs is consistent with the mechanism elucidated for concentration-dependent vacuoles in the renal proximal tubule where oligonucleotide taken up into the endosomal/lysosomal system is eluted during the fixation/processing of these tissues leaving a dilated vacuolar remnant seen by light microscopy. 1 Concurrent vacuolation of the renal tubule epithelium was not observed in our toxicology studies due to the lower tissue concentrations achieved from IT administration (data not shown) compared with much higher concentrations in the renal cortex resulting from systemic administration. 1
This mechanism is related to the physicochemical properties of ASOs rather than their gene modifying activity. Antisense oligonucleotides are small, hydrophilic, soluble molecules that could be readily cleared from their intracellular location if not adequately cross-linked by fixation. Given that they also become sequestered in high concentrations within the lysosomal compartment, then a simultaneous osmotic effect can occur leading to visible dilation of the lysosome. Similar osmotic vacuolation in the renal tubule epithelium occurs with other hydrophilic compounds such as sugars 12 and pegylated proteins. 13
In the case of ASO-related renal vacuolation, it was found to be abolished by employing more rapid fixation by perfusion with modified Karnovsky’s fixative. 1 This was replicated in our first investigative study but not in our second, where, for reasons unknown, perfusion with Karnovsky’s fixative actually led to a higher incidence of neuronal vacuolation than did immersion in NBF. However, what is most significant from both investigative studies is that fresh frozen tissue contained no neuronal vacuolation, supporting the hypothesis of the macrovesicular vacuolation occurring as an artifact of postmortem tissue fixation.
The direct implication of this macrovesicular vacuolation being an artifact of tissue preservation is that it does not represent an adverse toxicological finding because it occurs postmortem and is not present in vivo. The absence of any associated neurobehavioral/neurological signs or tissue response observed in H&E, Fluoro-Jade B/C, GFAP, and Iba-1 staining supports that proposition.
Why the Hippocampus?
Why the macrovesicular vacuolation consistently occurs in the pyramidal neurons of the hippocampus, and very rarely in any other location of the CNS, remains unknown. Interestingly, the levels of ASO measured in this region are not always the highest in the brain and are usually lower than those measured within the lumbar spinal cord where this vacuolation of neurons has not been observed. Nor has this vacuolation been observed in rodent species following administration into the CNS, either by the intracerebroventricular route in mice or lumbar intrathecal route in rats. In those studies, the ASO in the brain as a whole can reach similar concentrations to that observed in the NHP hippocampus (unpublished internal data), but since it hasn’t been measured in specific rodent brain regions, it’s impossible to make a direct comparison.
Clearly the reason for the predilection of the hippocampal neurons to developing this vacuolation could warrant further investigation, as would the elucidation of the exact mechanism by which the vacuolation occurs, but these are beyond the scope of this focused investigation of fixation methods.
Other CNS Vacuolation Artifacts
It is worth noting that this observation of neuronal vacuolation as a postmortem artifact is not unique in the CNS where postmortem vacuolation of neurons and/or the neuropil may be observed following immersion fixation in formalin. 14 –17 Similar vacuolation has been observed in brain samples from rats treated with the ampakine CX717, 18 and the N-methyl-D-aspartate (NMDA) antagonist MK-801. 19 In both cases, those samples fixed in formalin revealed cytoplasmic vacuoles, whereas samples flash frozen and cryosectioned did not. In the case of CX717, investigations revealed that a postmortem exothermic reaction between the ampakine and formalin resulted in gas pockets in the parenchyma and astrocyte vacuolation. In the latter case, the vacuolation caused by NMDA antagonists was considered to be a reaction to aldehyde fixation of perturbed but living neurons resulting in artifactual distortion of multiple intracellular compartments.
Conclusions
Macrovesicular vacuolation of pyramidal neurons is routinely observed in H&E-stained sections of formalin-fixed, paraffin embedded hippocampus from intrathecally administered, repeat-dose, subchronic, and chronic NHP ASO toxicity studies and has been attributed to ASO treatment. However, no evidence of associated neuronal degeneration or cellular response is seen, confirmed by the results of Fluoro-Jade B/C staining and IHC for GFAP and Iba-1. Immunohistochemistry for LAMP2 and ASO, and examination by TEM, has demonstrated the vacuoles are dilated lysosomes containing ASO. Further investigation showed that they only occurred in hippocampus subjected to chemical fixation prior to processing and preparation of H&E-stained paraffin sections. Unfixed brain samples which were frozen, cryosectioned, and stained with H&E showed no macrovesicular neuronal vacuolation. These collective observations, and the lack of any associated clinical, neurological, or neurobehavioral signs, suggest that these vacuoles are a postmortem artifact arising from fixation of the tissue and are presumed not to be present in neurons of the hippocampus in vivo.
It is recognized that there are limited nonclinical and clinical options by which to monitor for potential treatment-related neuronal vacuolation, should it exist. However, results of this investigation indicate the neuronal macrovesicular vacuolation is a fixation artifact, so doesn’t represent a true lesion and therefore a hazard. Coupled with the lack of any correlative clinical signs in nonhuman primates or in clinical studies, this investigative work supports the conclusion that the hippocampal neuronal macrovesicular vacuolation does not represent a meaningful risk to patients being treated with IT 2′-MOE ASOs and has no significant relevance for patient benefit/risk analysis.
Footnotes
Acknowledgments
The authors would like to thank Bea Debrosse-Sera at Ionis for the histology preparations for the investigative fixation studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
