Abstract
The developmental neuropathology examination in juvenile toxicity studies depends on the nature of the product candidate, its intended use, and the exposure scenario (eg, dose, duration, and route). Expectations for sampling, processing, and evaluating neural tissues differ for developmental neurotoxicity studies (DNTS) for chemicals and juvenile animal studies (JAS) for pediatric pharmaceuticals. Juvenile toxicity studies typically include macroscopic observations, brain weights, and light microscopic evaluation of routine hematoxylin and eosin (H&E)-stained sections from major neural tissues (brain, spinal cord, and sciatic nerve) as neuropathology endpoints. The DNTS is a focused evaluation of the nervous system, so the study design incorporates perfusion fixation, plastic embedding of at least one nerve, quantitative analysis of selected brain regions, and sometimes special neurohistological stains. In contrast, the JAS examines multiple systems, so neural tissues undergo conventional tissue processing (eg, immersion fixation, paraffin embedding, H&E staining only). An “expanded neurohistopathology” (or “expanded neuropathology”) approach may be performed for JAS if warranted, typically by light microscopic evaluation of more neural tissues (usually additional sections of brain, ganglia, and/or more nerves) or/and special neurohistological stains, to investigate specific questions (eg, a more detailed exploration of a potential neuroactive effect) or to fulfill regulatory requests.
Keywords
Introduction
The developing nervous system is sensitive to many toxicants. Specification of the central nervous system (CNS [brain and spinal cord]) begins very early in gestation with formation of the neural plate and neural tube (between gestational days [GD] 15 and 30 in humans and GD 8.5 and 11 in rats). Subsequently, the expansion of the CNS and peripheral nervous system (PNS [ganglia and nerves]) continues through the end of adolescence (18 years of age in humans and postnatal day [PND] 70 to 90 in rats) and beyond. Neural development involves many processes that are differentially vulnerable to perturbation by toxicants including such structural phenomena as neural tube formation (“neurulation’), generation of neurons (“neuronogenesis”), synapses (“synaptogenesis”), various glial cell classes (“gliogenesis”), and myelin (“myelinogenesis”), as well as progressive maturation of neurobehavioral function. 1,2 Neural function (behavior and cognition) is an integrated activity mediated by the brain as a whole, indicating that optimal function relies on full integrity of the nervous system. Thus, even small neurodevelopmental deviations may lead to neural dysfunction and risk of disease. 3 Differences in neuroanatomic structure and physiological processes (eg, xenobiotic pharmacokinetics and pharmacodynamics) between immature and mature animals 4 indicate that safety testing to explore the presence and extent of neurotoxicity is an essential element in developing new products to which the immature nervous system might be exposed. Indeed, juvenile animals often exhibit more severe toxicities and/or novel CNS effects compared to adult animals. 5
Neuropathology evaluation is a core component in juvenile toxicity testing of new products. Regulatory guidance regarding the neuropathology examination in juvenile toxicity testing depends on the nature of the product candidate, its intended use, and the scenario (eg, dose, duration, frequency, and route) in which developing humans might be exposed. Two classes of juvenile toxicity studies with fairly specific neuropathology guidance—developmental neurotoxicity studies (DNTS) for chemicals 6,7 and juvenile animal studies (JAS) for pediatric pharmaceuticals 8 -10 —have overlapping but not identical expectations for sampling, processing, and evaluating neural tissues due to their different objectives. This article describes principal neurobiological factors and regulatory guidance to consider in determining how to design and perform the neuropathology evaluation for these two types of juvenile toxicity studies.
Neurobiological Factors Influencing the Design of Juvenile Toxicity Studies
The susceptibility of the developing nervous system to xenobiotics often serves as the basis for performing a juvenile toxicity study. Biological considerations that may focus attention on the neuropathology evaluation of such studies include many neural-specific factors as well as more general parameters related to developmental toxicity.
Neurobiological factors that often underpin the decision to perform a juvenile toxicity study typically are related to developmental aspects of the CNS, especially the brain. The evolution of the vertebrate brain is divided into discrete stages based on the waxing and waning of morphological landmarks (or more recently biochemical and/or molecular signals) over time. These events follow a consistent sequence across species, but the timing of events in a given species varies with the lengths of both gestation and the life span (Table 1). 1,2,11 -14 Formation of the neuraxis and specification of its cephalic end (ie, the brain progenitor) occur during the embryonic period. Generation, migration, and differentiation of neurons and glia are pronounced during the fetal, neonatal, infant, and early childhood periods (Figure 1), although the extents of each process differ among various brain domains at any given point in time. 2,11,15,16 Synaptogenesis is extensive throughout the fetal, neonatal, infant, childhood, and adolescent stages of development and to a modestly reduced degree in adults. 2,11 Myelination begins slowly in the midfetal period (ie, the last trimester of gestation in humans and in the first postnatal week in rats) and progressively increases throughout the adolescent period (Figure 1). 2,17 -19 Therefore, the brain as a whole is susceptible to neurotoxic agents for an extended time during prenatal and postnatal stages of brain development, while specific sites and cells in the CNS and PNS possess more limited critical periods of vulnerability. Exposures in human adolescents (12-18 years of age) generally do not require further safety testing in juvenile animals because dosing for repeat-dose toxicity studies in both rodents and nonrodents typically is initiated in young adult animals during neurodevelopmental stages equivalent to human adolescence (Table 1). For pharmaceuticals to be used in pre-term newborn infants, term newborn infants (birth-27 days of age), infants (28 days-23 months), or children (2-11 years), a JAS is likely to be included since safety data derived from adults are often not predictive of possible responses in pediatric patients, 20 where exposure occurs during the brain growth spurt.
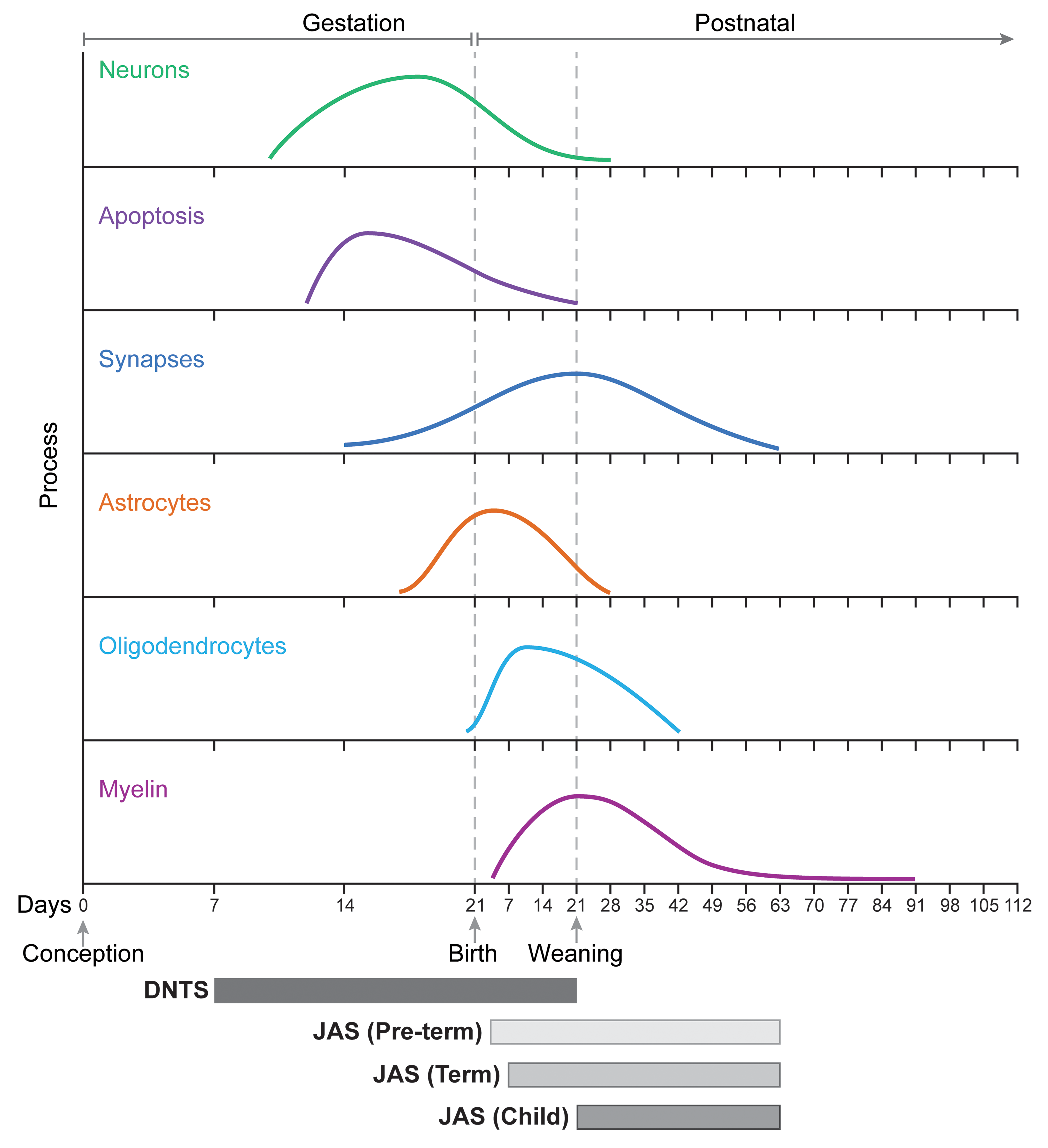
Schematic diagram showing the relationship of key neurodevelopmental events in the rat brain relative to the timing of test article administration for two categories of juvenile toxicity studies. The X-axis shows prenatal (to the left of the “birth” line) and postnatal times in days, with each tick representing one week. In the brain, generation of neurons (top curve) followed by apoptosis of excess neurons precedes production of glia (astrocytes and oligodendrocytes). Formation of synapses and myelin are later events. Chemical exposure during a conventional developmental neurotoxicity study (DNTS) typically involves maternal treatment from gestational day (GD) 6 through postnatal day (PND) 21. Juvenile animal studies (JAS) to develop pediatric pharmaceuticals vary in the timing of exposure. Common designs include “JAS (Term),” in which administration begins on PND 7 (the stage of rat brain development equivalent to a full-term human infant), and “JAS (Child),” in which treatment begins on PND 22 (the day after weaning, which is the rat brain equivalent of a 2-year-old human toddler). An unusual variant, “JAS (Pre-term),” involves exposure beginning on PND 4 (which is the rat brain equivalent of a pre-term human infant). Most JAS administer the pharmaceutical candidate until PND 63 to 70 (the rat brain equivalent of a late adolescent or early young adult). The timing (onset, peak, and end) of the neurodevelopmental events was extrapolated from the published literature. 2,11 –13,16,17,19 The figure was crafted by Mr Timothy Vojt.
Key Developmental Stages of Mammals Relevant to Developmental Neuropathology Evaluation.a
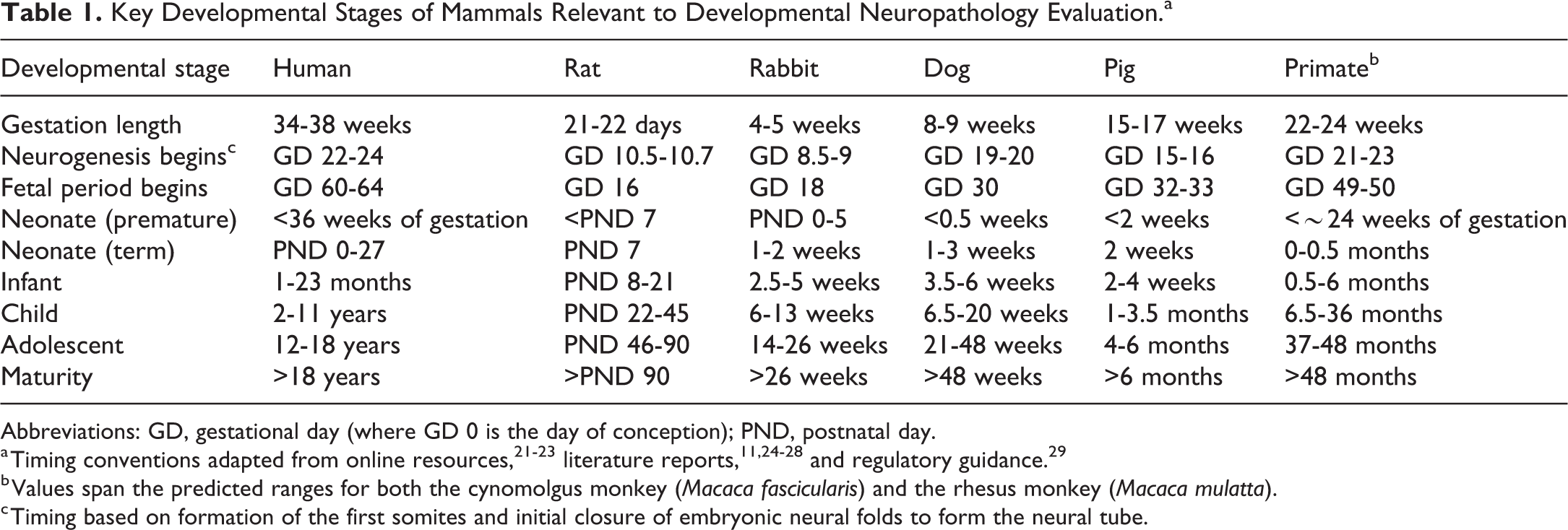
Abbreviations: GD, gestational day (where GD 0 is the day of conception); PND, postnatal day.
a Timing conventions adapted from online resources, 21 –23 literature reports, 11,24 –28 and regulatory guidance. 29
b Values span the predicted ranges for both the cynomolgus monkey (Macaca fascicularis) and the rhesus monkey (Macaca mulatta).
c Timing based on formation of the first somites and initial closure of embryonic neural folds to form the neural tube.
A juvenile toxicity study is likely warranted if damage is anticipated in one or many developing organs, 9,10,30 of which the nervous system is a principal target. 31 The CNS (and particularly the brain) of developing animals and humans is known to be targeted by certain small molecule classes including ethanol, 32,33 anesthetics (eg, isoflurane, ketamine, and other N-methyl-D-aspartate receptor antagonists), 11,34 antiepileptics (eg, phenytoin, sodium valproate, vigabatrin), 11,35 -37 antineoplastic chemotherapies, 38,39 retinoids, 36 and stimulants (eg, caffeine, methamphetamine) 36,40 as well as many environmental contaminants (chemicals and metals) 41 -43 and various endogenous metabolites. 33 Neurodevelopmental effects of such agents depend on the developmental age of exposure. During gestation, neurotoxic embryonic exposures before or during organogenesis (ie, the first trimester in humans, prior to GD 16 in rats) usually produce embryonic lethality or grossly visible malformations such as neural tube defects or microencephaly. In contrast, neurotoxic exposures during fetal (ie, the last two trimesters in humans, from GD 16 to approximately PND 7 in rats) or postnatal phases of neural development usually elicit microscopic defects such as neuronal heterotopias, 44 reduced neuronal 44 and/or synapse 43,45 numbers, altered myelination, 37 and/or functional (neurobehavioral) deficits. 46,47
Neuropathology Endpoints in Juvenile Toxicity Testing
The design of the neuropathology evaluation is influenced by regulatory guidance regarding major endpoints to examine, which is determined in large part by the predicted exposure scenario. In this respect, the divergence of the neuropathology evaluations for DNTS and JAS is due to their differing objectives. A DNTS to assess chemical safety following in utero and early postnatal exposure is designed to specifically provide a detailed analysis of the nervous system (emphasizing the CNS) as the principal target system. In contrast, a JAS to assess pediatric pharmaceutical safety after postnatal exposure is designed to screen many potential target systems, of which the nervous system represents just one. The distinct goals of these two studies are reflected in the neuropathology endpoints that generally are performed when conducting such studies. The two subsections below briefly compare and contrast the data expectations for these two types of juvenile toxicity studies (Tables 2 and 3).
Selected Developmental Neuropathology Endpoints for Juvenile Toxicity Testing.

Abbreviations: DNTS, developmental neurotoxicity study; JAS, juvenile animal study.
a The special stains for highlighting myelin (LFB) and specific groups of neurons (CV) are often combined on a single section.
Regulatory Guidance-Recommended Neuropathology Endpoints for Typical Juvenile Toxicity Studies.
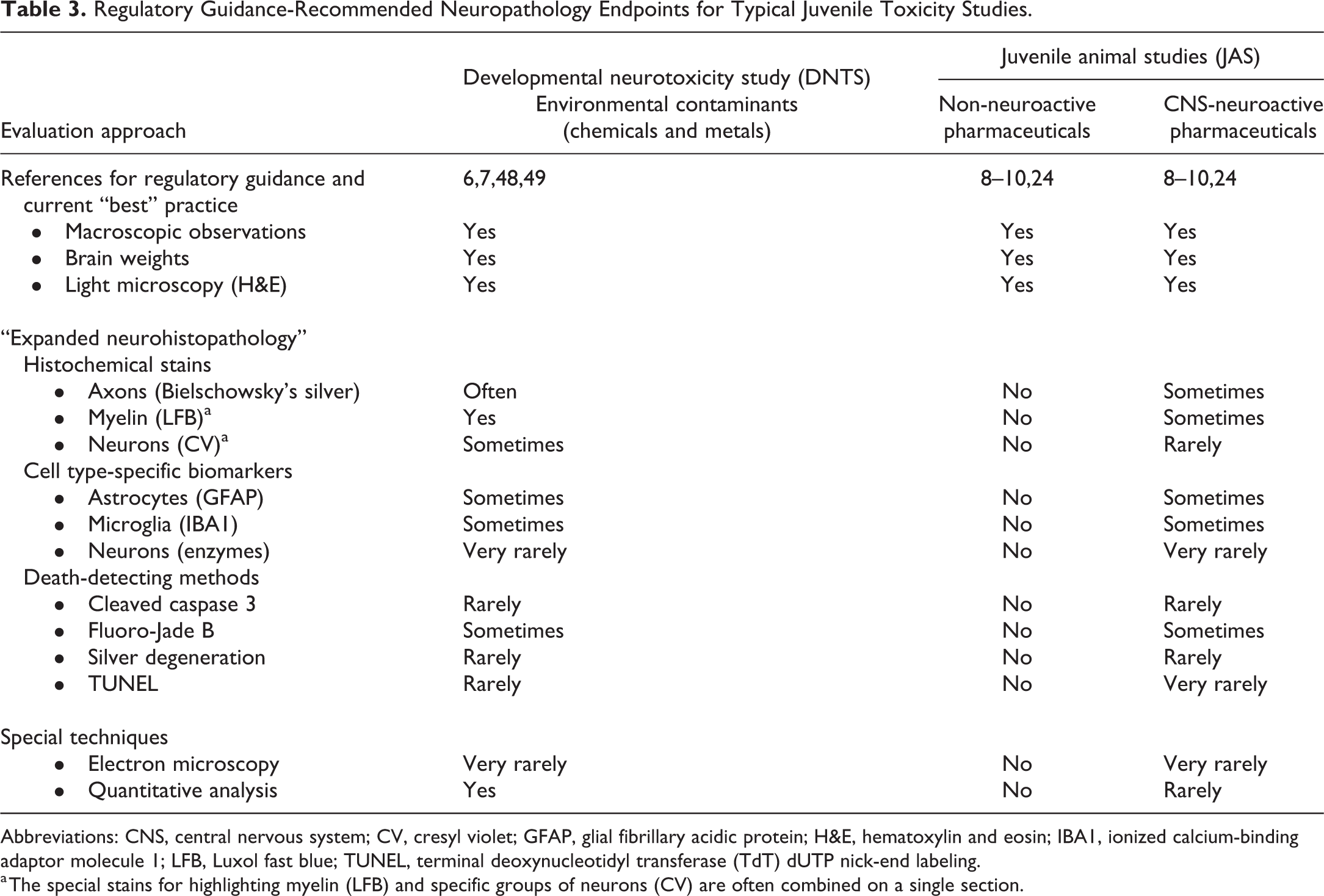
Abbreviations: CNS, central nervous system; CV, cresyl violet; GFAP, glial fibrillary acidic protein; H&E, hematoxylin and eosin; IBA1, ionized calcium-binding adaptor molecule 1; LFB, Luxol fast blue; TUNEL, terminal deoxynucleotidyl transferase (TdT) dUTP nick-end labeling.
a The special stains for highlighting myelin (LFB) and specific groups of neurons (CV) are often combined on a single section.
Developmental Neurotoxicity Studies for Chemicals
Considerations for rodent DNTS have been produced by the US Environmental Protection Agency (EPA) 6 and the Organisation for Economic Co-operation and Development (OECD). 7,50 Working groups of toxicologic pathologists and toxicologists have created “best practice” recommendations 48,49 for implementing the neuropathology evaluation as well as integrating the neuropathology data with associated neurobehavioral (cognitive, motor, and sensory) data acquired during a DNTS. 51,52 Study procedures for immature (pre-weaning) animals may differ from those of mature animals, so positive control test substances often are included in the DNTS design to ensure that the experimental methods are capable of detecting xenobiotic-induced changes in neural structure or function. 53,54
In routine DNTS performed in rats, indirect exposure of developing animals occurs during gestation (by maternal treatment) and after birth (usually by continued maternal treatment leading to lactational exposure of the nursing offspring). The duration of exposure in rat DNTS extends from GD 6 to either PND 10 (if following the original EPA guidance 6 ) or PND 21 (if using the OECD guidance 7 ); most current DNTS dose through PND 21 both to produce exposure for a larger portion of neural development and because necropsy procedures for the early time point (carried out the day following the last dose) are simpler in PND 22 rats versus PND 11 rats. 48,55 The late time point for a rat DNTS typically is performed between PND 75 and 90.
The main DNTS objective, to specifically investigate neural tissues as primary target organs for test substances, is reflected in regulatory guidance 6,7 that the neuropathology sampling and evaluation go beyond the neural tissue analysis expected in general toxicity testing. Neuropathology evaluations for DNTS mirror those performed for neurotoxicity testing of adult animals, 56,57 with some modifications. At necropsy, neural organs are observed to detect any macroscopic abnormalities. Brain weights may be obtained for unfixed organs at necropsy or for preserved organs once fixation is complete; postfixation weighing is often chosen to minimize potential microscopic artifacts (eg, “dark neurons” in certain cerebrocortical, hippocampal, and cerebellar regions 58 ) that develop when unfixed neural tissue is handled for weighing. Either approach is acceptable as long as the brains from the control animals (concurrent and historical) and test article-exposed animals have undergone comparable fixation conditions at the time of weighing. Since DNTS are performed specifically to evaluate neural tissues as potential target organs, fixation of neural tissues by intravascular perfusion is preferred to ensure rapid preservation of deep regions of lipid-rich CNS organs. Perfusion fixation is required at the late (young adult) time point and is encouraged for the early time point. However, regulatory guidance permits immersion fixation at the early time point because perfusion via small-diameter blood vessels is more difficult technically; this immersion option is used most often if the early necropsy is performed at PND 11. Buffered (∼pH 7.4) aldehydes are preferred fixatives for rat DNTS, so the usual fixative choices are standard neutral buffered 10% formalin (NBF, which contains 3.7% formaldehyde with ∼1% methanol as a stabilizing agent) or methanol-free formaldehyde (MFF, ∼3.7% formaldehyde made from paraformaldehyde pellets or powder); NBF is less expensive, while MFF removes methanol (a potential lipid-mobilizing solvent) as a potential source of processing-related artifacts in heavily myelinated structures (white matter of the brain and spinal cord). These myelin artifacts are particularly prominent when animals are perfused with NBF and the brain is removed immediately afterward rather than following a 1-day period of additional fixation in situ by immersion in fresh NBF.
Recommendations for tissue sampling, processing, and evaluation of the CNS and PNS are generally similar for DNTS 48,49 and adult neurotoxicity studies, 59,60 since neural organs are the specific focus for these studies. Currently recognized best practices for nervous system sampling for DNTS include preparation and examination of multiple brain levels (minimally 6-8 full coronal sections in rats 48,49,59,61 or coronal hemi-sections in non-rodents 59 ), 3 spinal cord divisions (cervical, thoracic, and lumbar), 49 at least 2 dorsal root ganglia (DRG, associated with the cervical and lumbar spinal cord divisions at minimum), 49 trigeminal (cranial nerve V) ganglia, and 1 or more nerves (sciatic nerve at minimum). 49 Spinal cord and nerve typically should be evaluated in longitudinal (or oblique) and transverse planes, 49,59,62 although some sponsors assess several (usually 3-6) transversely oriented sections per tissue instead. Bilaterally symmetrical tissues (eg, trigeminal ganglia, nerves) usually should be collected bilaterally but may be processed and evaluated unilaterally. 60 Bilateral collection is recommended so that a backup sample is retained in case it is needed, while unilateral evaluation is suitable since xenobiotics generally are expected to be distributed to bilaterally symmetrical organs to a similar degree. Tissues typically are embedded routinely in paraffin, 49 although regulatory guidance recommends plastic (epoxy resin) embedding of nerve. 6,7 Sections of these tissues are stained with hematoxylin and eosin (H&E) for the basic evaluation, 49 while toluidine blue is typically utilized instead for plastic-embedded sections of nerve. Additional sampling (usually H&E-stained sections from several more coronal and/or sagittal levels of the brain 63 ) and/or including one or more advanced techniques (Table 2) 49,59 may be useful to ensure that the neuropathology evaluation has been sufficient. Currently, advanced methods incorporated in DNTS based on existing regulatory guidance typically include one or more special neurohistological methods (“stains”) to identify cell type-specific markers (eg, for neuronal damage or reactive glial cells) and quantitative analysis of key brain features. 6,7,64
Special neurohistological methods used during DNTS include histochemical stains and cell type-specific immunohistochemical (IHC) markers. Common histochemical stains are used to demonstrate axonal integrity (eg, Bielschowsky’s silver), myelin quality (eg, Luxol fast blue [LFB]), and/or neuronal number (eg, cresyl violet). Standard IHC methods are employed to detect specific populations of reactive glial cells (eg, anti-glial fibrillary acidic protein [GFAP] for astrocytes, anti-ionized calcium-binding adaptor molecule 1 [IBA1] for microglia) may be performed on CNS tissues to highlight glial responses to parenchymal injury. Typically, retention of robust IHC labeling requires that fixation be limited to 48 hours. Special neurohistological stains to demonstrate cell death (eg, silver degeneration stains on frozen sections, 65 Fluoro-Jade B on paraffin sections 66 ) may be used, but interpretation often is challenging at the early DNTS time point due to ongoing programmed cell death of neurons (which continues in the rat CNS until weaning). 11
Another special neurohistological consideration is the regulatory recommendation 6,7 that at least one nerve should be embedded in plastic to provide improved resolution of any toxicant-induced changes in nerve fibers. 6 Plastic embedding typically employs postfixation in 1% osmium tetroxide to enhance myelin preservation, which improves the cellular details visible in toluidine blue-stained thin (1-µm-thick) sections of nerve. 60 Experience has shown that hard plastic sections (eg, epoxy resin) offer better resolution of fine cell architecture than soft plastic sections (eg, glycol methacrylate). 60 Guidance for DNTS does indicate that paraffin embedding is acceptable for nerves. 6,7
Regulatory guidance that quantitative analysis of selected brain regions be included in DNTS 6,7 is designed to provide objective measurements of xenobiotic-induced changes in dimensions of critical neural tissues that undergo extensive structural evolution over the entire course of test substance exposure. Regulatory guidance 6,7 and recognized “best practice” recommendations 48,49 focus on quantitative analysis (linear and/or areal morphometry) of the cerebral cortex, hippocampus, and cerebellum as critical sites of neuron and synapse generation (with well-known functions that are measured during the neurobehavioral arm of a DNTS); morphometry of the striatum and corpus callosum (a major white matter tract) often are added as well. 48 Such quantitative measurements may be obtained at the macroscopic level (eg, from images of the brain taken at necropsy or after fixation), microscopic level (from highly homologous paraffin-embedded sections), or both. 18,48,49 The quantitative analysis is performed in the control and high-dose groups initially, adding other dose groups only if warranted by discovery of altered (usually reduced) dimensions in one or more brain regions in the high-dose animals. 48,52 To limit processing artifacts as a confounding factor when interpreting quantitative measurements, all tissues intended for potential morphometric analysis should be processed to the block stage for all dose groups at the same time. The analysis itself is performed in coded (“blinded”) fashion to minimize observer bias.
In practice, acquisition of highly homologous brain sections is technically difficult even in Good Laboratory Practice-compliant histology laboratories with detailed standard operating procedures. Accordingly, the quantitative data are deemed acceptable if measurements can be acquired from 6 (of 10) animals/sex/group. 48,52 Conceptually, other methods might be used for the DNTS quantitative analysis to try to increase the number of animals per group from which quantitative measurements may be obtained. One option is to embed multiple brains in one block so that all brains are sectioned at essentially the same level (Figure 2). This approach facilitates the coded analysis since brains from all groups may be randomly positioned in a block and analyzed before the position code is broken. A potential disadvantage of this approach for the early DNTS time point (especially PND 11) is that the variation in brain size among immature rats 48,55 may prevent all brains in a single section from having a high degree of homology; in such cases, adjacent sections may be evaluated to find appropriately homologous sections, as is practiced for individually embedded brains. 48 A second option for quantitative analysis is stereology. 67 -69 The systematic uniform random sampling provided by routine stereological preparations obviates the need for highly homologous sections. 70 The main disadvantage of stereology for this application is that the processing time for each animal is much longer, 64 thereby increasing the time needed to complete the analysis.
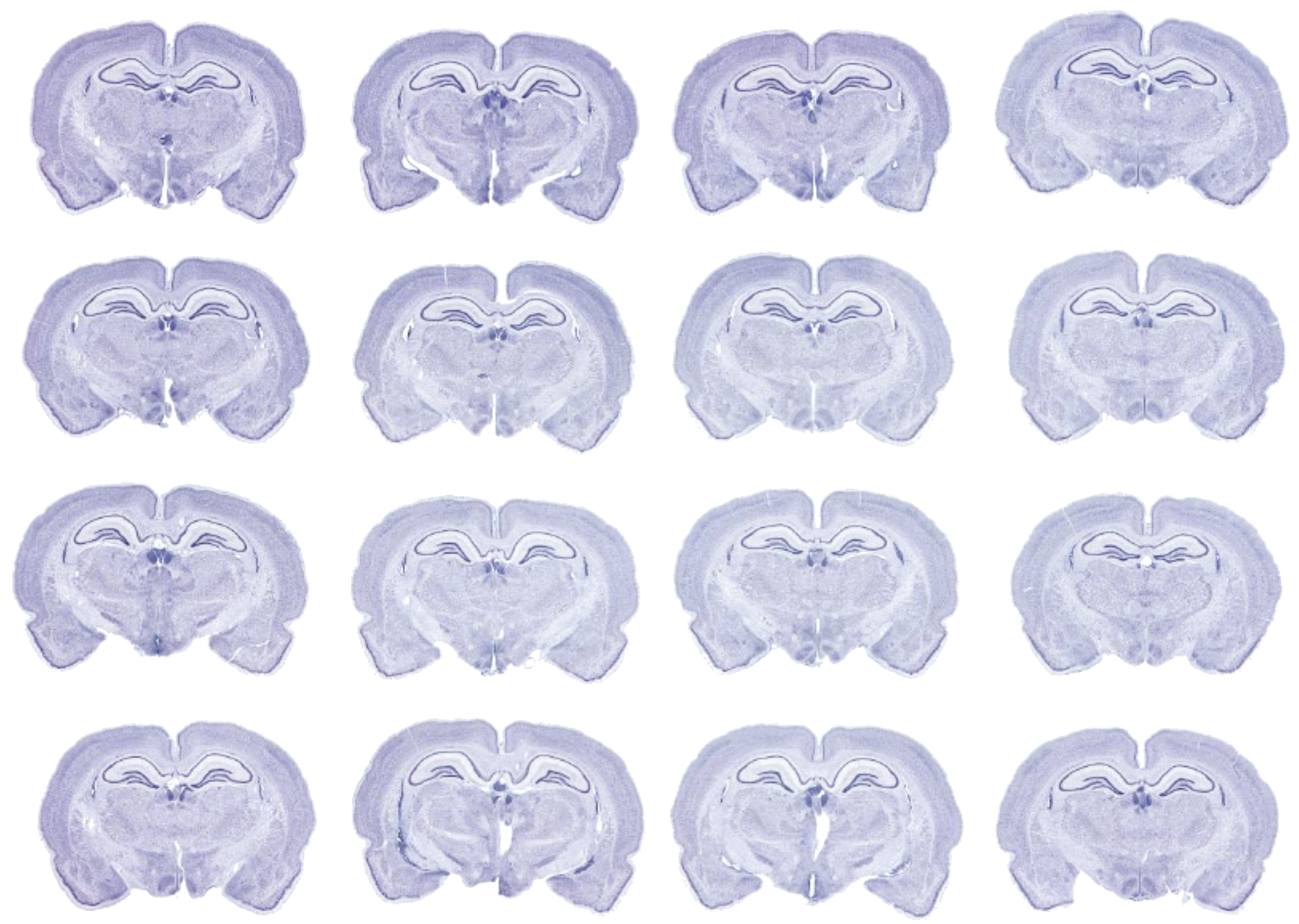
Image of a Multibrain preparation showing multiple young adult rat brains coembedded and simultaneously sectioned to produce highly homologous coronal brain sections to permit morphometric measurements on the hippocampus or on other neuroanatomic structures at this level of the brain. This anatomic orientation conforms to current STP “best practice” recommendations for the quantitative analysis portion of conventional developmental neurotoxicity studies (DNTS) in rats. 48,49 Nissl stain (to highlight neuronal populations). The image is provided by and reproduced with the permission of NeuroScience Associates, Knoxville, Tennessee (https://www.neuroscienceassociates.com).
The neuropathology evaluation for a rodent DNTS is quite laborious relative to the design used in general toxicity studies in rats. 64 This increased labor is a direct consequence of the special processing needs (eg, perfusion fixation, plastic embedding); increased sampling (more CNS and PNS tissues, usually with special neurohistological techniques for several tissues); and time-consuming quantitative analysis of selected brain regions. The justification for this effort is that the DNTS is designed to provide a highly detailed assessment of the nervous system as a potential or likely target organ.
Juvenile Animal Studies for Pediatric Pharmaceuticals
With the advent of the Best Pharmaceuticals for Children Act in 2002 and the Pediatric Research Equity Act in 2003, 71 JAS in the United States are often required to support the pediatric use of new pharmaceutical products. This requirement encompasses not only small molecule drugs but biologics unless an evidence-based justification shows that a JAS would not yield relevant data (eg, the biologic is not pharmacologically active in rodents). Current globally standardized guidance for JAS 9,10 recommends a weight-of-evidence approach in considering whether or not a nonclinical juvenile toxicity study is needed to support clinical trials. Multiple elements contribute to such decisions, but two factors are particularly important: the age of the intended patient population and the potential of a given test article or structurally related agents to induce developmental toxicity in one or more organs that continue to develop postnatally. Given that developmental processes associated with the immature CNS differentially affect the pharmacokinetics and pharmacodynamics of xenobiotics, 72 -75 the neurotoxic potential of a pharmaceutical candidate is a major consideration in making such weight-of-evidence decisions. 5,31
Guidance for pharmaceutical development by regulatory agencies (eg, the US Food and Drug Administration [FDA] 8 ) or global consortia to standardize safety assessment methods for new medical products (eg, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 76 ) suggest that several CNS organs (multiple regions of the brain and several divisions of the spinal cord) and one PNS component (usually the sciatic or sometimes the tibial nerve) should be evaluated during toxicity studies. 77 Unlike guidance for DNTS for chemicals, the S11 guidance for JAS 9,10 does not specify the functional (ie, neurobehavioral) or structural (ie, neuropathology) assessments that should be performed. Therefore, the design of the neuropathology evaluation for a JAS will vary depending on many factors in the weight-of-evidence assessment and the specific study objectives. 9,24
In most cases, JAS are performed in rats, although occasionally a second study in a nonrodent species (dog or rarely nonhuman primate) may be performed. 5,31 The preference for rats is founded in well-recognized methods for assessing and integrating anatomic and neurobehavioral data in this species as well as the robust historical control data for such endpoints in this species. 9,10 In a typical JAS in rats, test article exposure begins after birth by direct treatment of the offspring. The duration of exposure in a rat JAS varies depending on the age of the intended pediatric population (Figure 1). Initiation of dosing on PND 7 (the stage of nervous system development in the rat that is equivalent to a full-term human infant) continuing to PND 63 or 70 (the rat equivalent of an adult human) will provide coverage for all phases of postnatal neural development in a human pediatric population. Treatment of pre-term human infants may be modeled in a JAS by initiating rat dosing at PND 4, but this study design is seldom used because most pharmaceuticals are not intended for administration to pre-term infants.
In rat JAS with non-neuroactive or minimally CNS-neuroactive test articles, the neuropathology evaluation typically is comparable to that performed for a general toxicity study in adult animals. At necropsy, CNS and PNS tissues are removed and assessed macroscopically and then fixed by immersion in NBF. Brains may be weighed prior to (ie, at necropsy) or after fixation. Neural tissues are sampled as described above for the DNTS neuropathology design. In JAS using rats, the basic neural tissue list will include 6 to 7 full coronal brain sections, 3 spinal cord divisions (cervical, thoracic, and lumbar), and 1 or more nerves (sciatic and/or tibial nerve at minimum). 59 Other neural tissues (eg, DRGs, trigeminal ganglia, additional nerves) should be considered for collection at necropsy but may be archived without evaluation unless a test article-mediated effect needs to be investigated further (eg, DRG toxicity produced by antineoplastic chemotherapies 78 and some viral gene therapy vectors 79 ). Spinal cord and nerve should be evaluated in both longitudinal and transverse planes. 60 The existing S11 guidance for JAS 9,10 does not specify tissue processing conditions for routine neuropathology evaluation. Accordingly, typical JAS use current “best practices” adopted from the neuropathology portion of general toxicity studies in adult animals—embedding in paraffin and staining with H&E only. 59
In rat JAS for test articles with predicted or known neuroactive effects, the extent of the neuropathology evaluation may be increased. The S11 guidance for JAS 9,10 refers to this augmented analysis as an “expanded neurohistopathology evaluation.” (Synonyms for this concept encountered by the authors include “expanded neuropathology” and in rare cases “enhanced neuropathology.”) In JAS, “expanded neurohistopathology” is varied to fit the particular questions raised by the mechanism of the test article or the potential sites of neuroactivity/neurotoxicity. 9,10,31 As in JAS for minimally neuroactive test articles, CNS and PNS tissues are removed at necropsy for macroscopic evaluation, brains are weighed, and all samples are fixed by immersion in NBF. Although the CNS may be a primary target tissue in JAS for neuroactive test article, perfusion fixation is not employed since this technique may affect the organ weights and tissue architecture of highly vascular organs (eg, gonad, lung, spleen) that are also important potential target organs for JAS. The expanded neurohistopathology design typically will include one or both of the following expansions:
additional neural sites beyond the typical neural tissue list (brain, spinal cord [cervical and lumbar divisions], sciatic nerve)—common choices are:
ˆ a few more brain sections (often 1 to 5 over the recommended 6-7 sections assessed in the baseline evaluation), 63
ˆ DRG sections (cervical and lumbar ± thoracic divisions), with spinal nerve roots, 60
ˆ trigeminal ganglia, 60 and/or
ˆ other nerves (usually sciatic nerve tributaries: tibial and fibular nerves). 60
special neurohistological stains (as described above for DNTS) 59 —in the authors’ experience, the most common choices for JAS are anti-GFAP (to show astrocytic reactions), anti-IBA1 (to reveal microglial reactions), and sometimes LFB (to view myelin integrity).
The structure of the expanded neurohistopathology evaluation for JAS is flexible by intent, and many designs may be appropriate when supported by a sufficient weight of evidence and well-crafted scientific rationale.
Certain neuropathology endpoints as implemented for the DNTS are ill-suited for typical JAS. In the authors’ experience, neuropathology findings in JAS are observed much more often in the CNS rather than the PNS. Accordingly, plastic embedding of a nerve typically is not warranted for JAS except in those infrequent instances when in-life neurological signs and/or light microscopic evaluation of nerves suggest that the PNS is impacted. Quantitative analysis as undertaken in DNTS typically is not performed or warranted for JAS since the key structures probed by this method (eg, cerebral cortex, hippocampus, and cerebellum) have attained approximately adult contours and sizes before direct exposure of the developing animals is initiated.
The neuropathology data set acquired during a JAS will be integrated in the study report with the results of neurobehavioral (cognitive, motor, and sensory) testing. A recent FDA review notes that neurobehavioral and neuropathology data from JAS for CNS-active drugs are often included in the labels for pediatric pharmaceuticals, and that neurobehavioral tests are more commonly affected relative to typical neuropathology endpoints. 31
Summary
Neuropathology evaluation is a primary component of assessing neural development in animal toxicity studies. 48,51,52 Structure-based endpoints are particularly valuable for developmental neuropathology screening due to their reproducibility within and across laboratories. 80
The neuropathology evaluations for DNTS and JAS are similar but not identical. Design differences between these studies reflect their distinct study objectives and divergent exposure scenarios. In a conventional DNTS, the CNS (and sometimes the PNS) is the specific focus of a detailed hazard identification and characterization effort. In contrast, for a typical JAS, the CNS and PNS are just two among many potentially affected systems that must be assessed. These fundamentally dissimilar objectives require different study designs and approaches to implementing the neuropathology evaluation.
Both DNTS and JAS include conventional endpoints like macroscopic observations, brain weights, and light microscopic evaluation of H&E-stained sections as the basis for the neuropathology evaluation. Tissue processing for DNTS typically utilizes tissue perfusion to provide optimal preservation of neural target organs and sometimes employs specialty fixatives (eg, MFF or combinations of formaldehyde and glutaraldehyde) to better stabilize cell microarchitecture and myelin lipids. Such specialized processing conditions generally are not needed for JAS. Similarly, DNTS typically includes a greater number of neuropathology endpoints (eg, sampling additional neural organs, serial sections processed using special neurohistological stains, quantitative analysis of selected brain regions) as initial elements of the neuropathology evaluation. This substantial neuropathology expansion for DNTS over the conventional neuropathology evaluation as performed in JAS is warranted since the focus of DNTS is to specifically examine the nervous system in detail. In contrast, expanded neurohistopathology for JAS typically is performed only if warranted and is limited to evaluating more neural tissues (especially additional brain sections and occasionally more nerves or ganglia) and/or supplementing the H&E-stained sections with serial sections processed with selected neurohistological stains.
The rationale for inclusion of quantitative analysis for DNTS but not JAS is two-fold. First, in terms of neurobiological relevance, the timing of exposure for these two studies encompasses different neurodevelopment processes (Figure 1). Exposure for DNTS extends from the start of organogenesis (GD 6 in rats, the preferred test species) to either the end of the human third-trimester equivalent (ie, birth in humans or approximately PND 7 in rats) or to the end of the human infancy equivalent (2 years in humans and PND 21 in rats). In contrast, dosing for JAS may be initiated at various ages depending on the intended patient population, typically beginning at either the human full-term birth equivalent (PND 7 in rats) to cover all of childhood or at the human toddler equivalent (PND 22 in rats). For JAS, where dosing starts at PND 22, the neurodevelopmental events that might be detected by quantitative analysis (eg, large decreases in neuron numbers or synapse-rich parenchymal fields) are receding or completed by the time of necropsy (commonly PND 56 or later in rats). The second reason for not performing quantitative morphometric analysis of brain regions as a routine element of the JAS neuropathology evaluation is that neural effects associated with typical JAS dosing regimens are more sensitive to detection by neurobehavioral tests rather than structural evaluation. 31,81 On rare occasions, quantitative analysis of specific neural structures may be considered for a JAS to answer a specific question.
The many interacting factors that must be considered in designing juvenile toxicity studies indicate that prudent sponsors will consult with regulatory authorities in advance when designing the appropriate neurotoxicity (including neuropathology) endpoints to include in the study protocol. 82 This proactive approach will be especially helpful to sponsors performing JAS as the design of such studies varies considerably compared to the relatively more prescriptive guidance offered regarding the design of the DNTS neuropathology evaluation.
Footnotes
Acknowledgments
Author Contributions
The analyses, conclusions, and opinions expressed in this article are solely those of the authors. All authors participated in the discussions involved with formulation and organization of this article.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
