Abstract
The 2021 annual National Toxicology Program (NTP) Satellite Symposium, entitled “Pathology Potpourri,” was the 20th anniversary of the symposia and held virtually on June 25th, in advance of the Society of Toxicologic Pathology’s 40th annual meeting. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers’ talks along with select images that were presented to the audience for voting and discussion. Various lesions and topics covered during the symposium included differentiation of canine oligodendroglioma, astrocytoma, and undefined glioma with presentation of the National Cancer Institute’s updated diagnostic terminology for canine glioma; differentiation of polycystic kidney, dilated tubules and cystic tubules with a discussion of human polycystic kidney disease; a review of various rodent nervous system background lesions in control animals from NTP studies with a focus on incidence rates and potential rat strain differences; vehicle/excipient-related renal lesions in cynomolgus monkeys with a discussion on the various cyclodextrins and their bioavailability, toxicity, and tumorigenicity; examples of rodent endometrial tumors including intestinal differentiation in an endometrial adenocarcinoma that has not previously been reported in rats; a review of various rodent adrenal cortex lesions including those that represented diagnostic challenges with multiple processes such as vacuolation, degeneration, necrosis, hyperplasia, and hypertrophy; and finally, a discussion of diagnostic criteria for uterine adenomyosis, atypical hyperplasia, and adenocarcinoma in the rat.
Keywords
An Update on Canine Glioma Diagnostic Criteria and Terminology
Dr Gregory Krane (Charles River Laboratories [CRL]) provided an update on canine glioma and shared some of his research on this topic as a National Institute of Environmental Health Sciences (NIEHS)/National Toxicology Program (NTP) toxicologic pathology postdoctoral fellow and as a North Carolina State University College of Veterinary Medicine (NCSU CVM) graduate student. He presented 3 cases of spontaneous intracranial canine glioma from pet dogs from the NCSU CVM pathology archives. Diagnoses for these cases were achieved via majority diagnosis by a panel of 5 pathologists (4 American College of Veterinary Pathologists [ACVP] board-certified veterinary anatomic pathologists and 1 physician neuropathologist). Dr Krane acknowledges Drs David Malarkey (NIEHS/NTP), Andrew Miller (Cornell University College of Veterinary Medicine), C. Ryan Miller (University of Alabama at Birmingham: School of Medicine), and Deb Tokarz (Experimental Pathology Laboratories, Inc [EPL]) for their participation in the abovementioned pathology review panel, Heather Jensen (NIEHS) and Dr Kyathanahalli Janardhan (Integrated Laboratory Systems) for support with immunohistochemistry, Dr Keith Shockley (NIEHS/NTP) for statistical support, Carly O’Dea (CRL) for image analysis, Eli Ney (NIEHS/NTP) and Beth Mahler (NIEHS/NTP) for assistance with optimizing manuscript images, as well as his principal investigator, Dr Chris Mariani (NCSU CVM).
The voting choices for all of Dr Krane’s cases were identical: low- or high-grade astrocytoma, low- or high-grade oligodendroglioma, low- or high-grade undefined glioma, low- or high-grade oligoastrocytoma/mixed glioma, and other. Images were presented from hematoxylin and eosin (H&E)-stained slides, as well as slides immunolabeled for oligonucleotide transcription factor 2 (Olig2), glial fibrillary acidic protein (GFAP), and 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase). Voting results for all cases are presented in Table 1.
Voting Choices and Results for Krane Cases 1, 2, and 3.a

a Voting choices were identical for all 3 cases. The voting results are listed, and the diagnosis as chosen by the pathology working group described in the narrative is designated in bold.
Case 1 (Figure 1A-E) was a well-demarcated, unencapsulated, expansile mass within the cerebral cortex. Sheets of round cells had distinct borders and perinuclear clearing, forming a honeycomb or fried-egg appearance. Nuclei were round with hyperchromatic chromatin. Cellular and nuclear pleomorphism were moderate, and mitotic figures were present. There were regional areas of necrosis, and glomeruloid microvascular proliferation was present. Tumor cells diffusely immunolabeled for Olig2 (nuclear) and CNPase (cytoplasmic), and they lacked immunolabeling for GFAP. The diagnosis was high-grade oligodendroglioma.
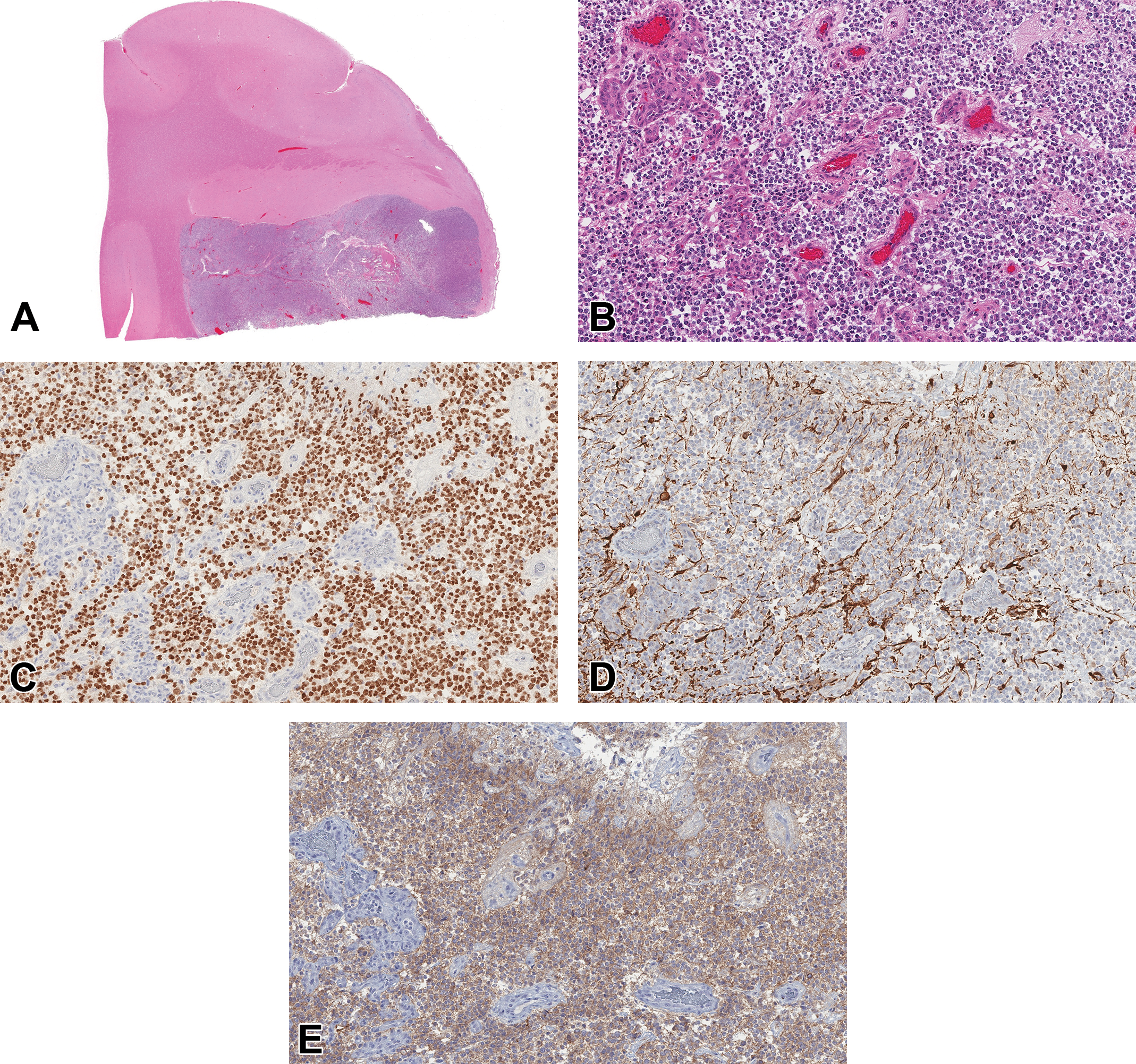
(A-E) High-grade oligodendroglioma, canine, brain. A, A subgross image shows a well-demarcated, expansile mass in the cerebral cortex (hematoxylin and eosin [H&E]). B, Sheets of round cells with perinuclear clearing form a honeycomb appearance. There are areas of glomeruloid microvascular proliferation, justifying a high-grade designation (H&E). C, There is diffuse nuclear immunolabeling for oligonucleotide 2 (Olig2). D, Cells lack immunolabeling for glial fibrillary acidic protein (GFAP). E, There is diffuse cytoplasmic immunolabeling for 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase).
Case 2 (Figure 1F-J) was a well-demarcated, unencapsulated, infiltrative mass within the cerebral cortex. Bundles, streams, and whorls of elongated cells had indistinct borders and small amounts of fibrillar, eosinophilic cytoplasm. Nuclei were spindle-shaped with condensed chromatin. Cellular and nuclear pleomorphism were moderate, and mitoses were not detected. There were large areas of regional necrosis which was often admixed with hemorrhage. The majority of tumor cells immunolabeled for Olig2 (nuclear), and they diffusely immunolabeled for GFAP (cytoplasmic). They lacked immunolabeling for CNPase. The diagnosis was high-grade astrocytoma.
(Continued). F-J, High-grade astrocytoma, canine, brain. F, A subgross image shows a well-demarcated, infiltrative mass in the cerebral cortex. There are regions of geographic necrosis associated with the hemorrhage, and this necrosis justifies a high-grade designation H&E. G, Bundles and streams of spindle-shaped cells exhibit high nuclear and cytoplasmic pleomorphism (H&E). H, The majority of cells exhibit nuclear immunolabeling for Olig2. I, There is diffuse cytoplasmic immunolabeling to GFAP. J, Cells lack immunolabeling to CNPase.
(Continued). K-M, High-grade undefined glioma, canine, and brain. K, Most regions of the mass contain streams of spindle cells with low amounts of fibrillar, eosinophilic cytoplasm. L, Approximately 30% of the mass contains round cells with perinuclear clearing. M, Higher magnification illustrating the perinuclear clearing (H&E).
Case 3 was a well-demarcated, unencapsulated mass infiltrating the cerebral cortex. Most regions of the mass (Figure 1K) were similar to those described in case 2, although large portions of the mass (Figure 1L and M), estimated 30% of total, were similar to those described in case 1. Cellular and nuclear pleomorphism were marked. Necrosis and microvascular proliferation were absent, and mitoses were present. Approximately half of the tumor cells were immunolabeled for Olig2 (nuclear). They lacked immunolabeling for GFAP and CNPase. The diagnosis was high-grade undefined glioma.
Approximately 25,000 humans and 2000 dogs in the United States are diagnosed with glioma annually. 1,2 Canine glioma occurs at a median age of 8 years and represents 35% of intracranial tumors (second only to meningioma); brachycephalic breeds such as the boxer, Boston terrier, and English and French bulldog are overrepresented. 3,4 Until recently, diagnostic guidelines for canine glioma were adapted from the World Health Organization (WHO) guidelines for the diagnosis of human glioma. 2 In an effort to harmonize diagnostic criteria for canine glioma and to advance the dog as a translational model for human glioma, the National Cancer Institute (NCI) convened a comparative glioma pathology board composed of physician and veterinarian neuropathologists and recently released an updated set of consensus diagnostic guidelines for canine glioma. 5,6
The NCI board consolidated the numerous diagnoses of the WHO into the broad subtypes of oligodendroglioma, astrocytoma, and undefined glioma (previously known as mixed glioma or oligoastrocytoma). Each subtype can then be further characterized as high- or low-grade status. Oligodendroglioma and astrocytoma retain their classic morphological features as described in the literature. 3,5 An undefined tumor contains both oligodendroglial and astrocytic morphologies, with the minority representing at least 20% of the total population. If a tumor has regions of geographic necrosis (excluding single-cell necrosis), glomeruloid microvascular proliferation, any mitoses, or overt cellular or nuclear pleomorphism or atypia, it is a high-grade tumor, and tumors lacking these features are low-grade tumors. 5 In case 1, more attendees chose low-grade oligodendroglioma than high-grade oligodendroglioma (41% vs 31%, respectively; Table 1), leading Dr Krane to highlight the presence of microvascular proliferation in that case (Figure 1B) to justify its high-grade designation.
Immunohistochemistry is often utilized to help characterize canine gliomas. The NCI glioma pathology board recommends a panel of Olig2, GFAP, CNPase, and Ki-67 to help assess canine gliomas. Olig2 is a transcription factor necessary for the differentiation of oligodendrocytes, and it is reported to be a sensitive marker for all subtypes of glioma. 3,5,7 Glial fibrillary acidic protein is an intermediate filament that is reported to be a useful marker for astrocytoma, although it cannot rule out oligodendroglioma. 3,5,7 CNPase is an enzyme that regulates the distribution of cytoplasmic tubules in myelin-producing cells. 8,9 It is reported to be helpful in establishing the diagnosis of oligodendroglioma; however, it can be sensitive to fixation and lack of immunolabeling does not rule out oligodendroglioma. 5 Ki-67 labels cells in all active phases of the cell cycle, although it is absent in noncycling cells. 10 Ki-67 labeling index (LI), which is a more sensitive indicator of active cell growth than mitotic rate, represents the percentage of tumor cells immunolabeled for Ki-67 antigen. 11 In human glioma, increased Ki-67 LI has been linked to decreased survival and increased histological grade. 12 -14
Dr Krane shared some data from his graduate work investigating immunohistochemical (IHC) profiles in a cohort of 73 canine patients with glioma. In this research, with regard to manual detection of immunolabeling, astrocytoma was more likely to immunolabel to GFAP than did oligodendroglioma, and oligodendroglioma was more likely to immunolabel to CNPase than did astrocytoma. In this study, tumors were also subject to area fraction assessment, which measures the percentage of tumor that immunolabels to a particular marker. These fractions were determined by computerized image analysis. The area fraction for Olig2 was greater for oligodendroglioma than it was for astrocytoma. The area fraction for GFAP was greater for astrocytoma than it was for oligodendroglioma, and the GFAP area fraction was also greater in low-grade tumors than it was in high-grade tumors. Although these data reinforce that the IHC profiles can be useful to diagnose glioma subtypes, future work is indicated to assess whether they predict prognosis or response to particular therapies in the dog.
The NCI consensus guidelines reported moderate overall interpathologist agreement with regard to subtype and grade among its board members utilizing a kappa scoring system. 5 Kappa statistics estimate the degree of agreement compared to what would be predicted based on chance alone. 15 The kappa statistic can range from +1 (complete agreement) to 0 (random chance) to −1 (complete disagreement). 16 The kappa statistic can represent poor (<0), slight (0.01-0.20), fair (0.21-0.40), moderate (0.41-0.60), substantial (0.61-0.80), and almost perfect agreement (0.81-1.0). 16 Dr Krane shared data from his graduate work exploring the extent of interpathologist agreement for caning glioma utilizing the NCI system by the aforementioned pathologist panel examining 85 cases previously diagnosed with glioma. Although an agreement for oligodendroglioma was higher than was the agreement for astrocytoma, both were within the moderate category of kappa statistics. There was no difference in agreement between low- and high-grade tumors, which also were within the moderate kappa statistic category. This shows that the NCI system results in reproducible diagnoses. Dr Krane discussed the aspects reported to affect interpathologist agreement in human glioma, such as pathologist experience and glioma subtype. Although 20% to 30% of human gliomas are often reclassified after specialist review, only 8.8% of cases result in disagreements significant enough to markedly affect prognosis and therapy. 17,18
Discussion topics raised by Dr Krane focused on how standardization of these diagnostic criteria can allow for multi-institutional studies utilizing canine glioma as a comparative oncology model for human glioma. The intact immune status, spontaneous nature of disease, and similar environmental exposures to humans for canine glioma create advantages in the utilization of the dog above induced rodent models for glioma research. Toxicologic pathologists should be prepared to utilize the NCI system if tasked with evaluating the efficacy or safety of a glioma therapeutic using spontaneous canine glioma as a disease model.
In response to the presentation, an audience member asked if there are any known causes of glioma in the dog. Beyond breed predispositions, Dr Krane was not aware of any published causes of glioma in the dog, whether it be an induced model or an environmental exposure, the latter of which would have a significant impact on humans from comparative oncology and public health perspective. Another audience member asked about the image analysis methods used to distinguish tumor cell immunolabeling from background/stromal immunolabeling, particularly for GFAP. The methods used to train the computer to filter tumor cell immunolabeling from background, along with data summarized in this article that is currently being prepared for publication, as well as in-depth discussions of glioma across different species, can be found in Dr Krane’s NCSU dissertation for those interested in further details. 19
More work is indicated to assess the impact of the NCI canine glioma diagnostic system on canine comparative oncology. The field of human glioma has been revolutionized by the use of molecular pathology to help characterize glioma, determine prognosis, and choose therapies. Such research will be the wave of the future for canine glioma and should advance diagnostic, comparative, and toxicologic pathology, improving the health of both dogs and humans alike.
Chemically Induced Cystic Kidneys in a Rat
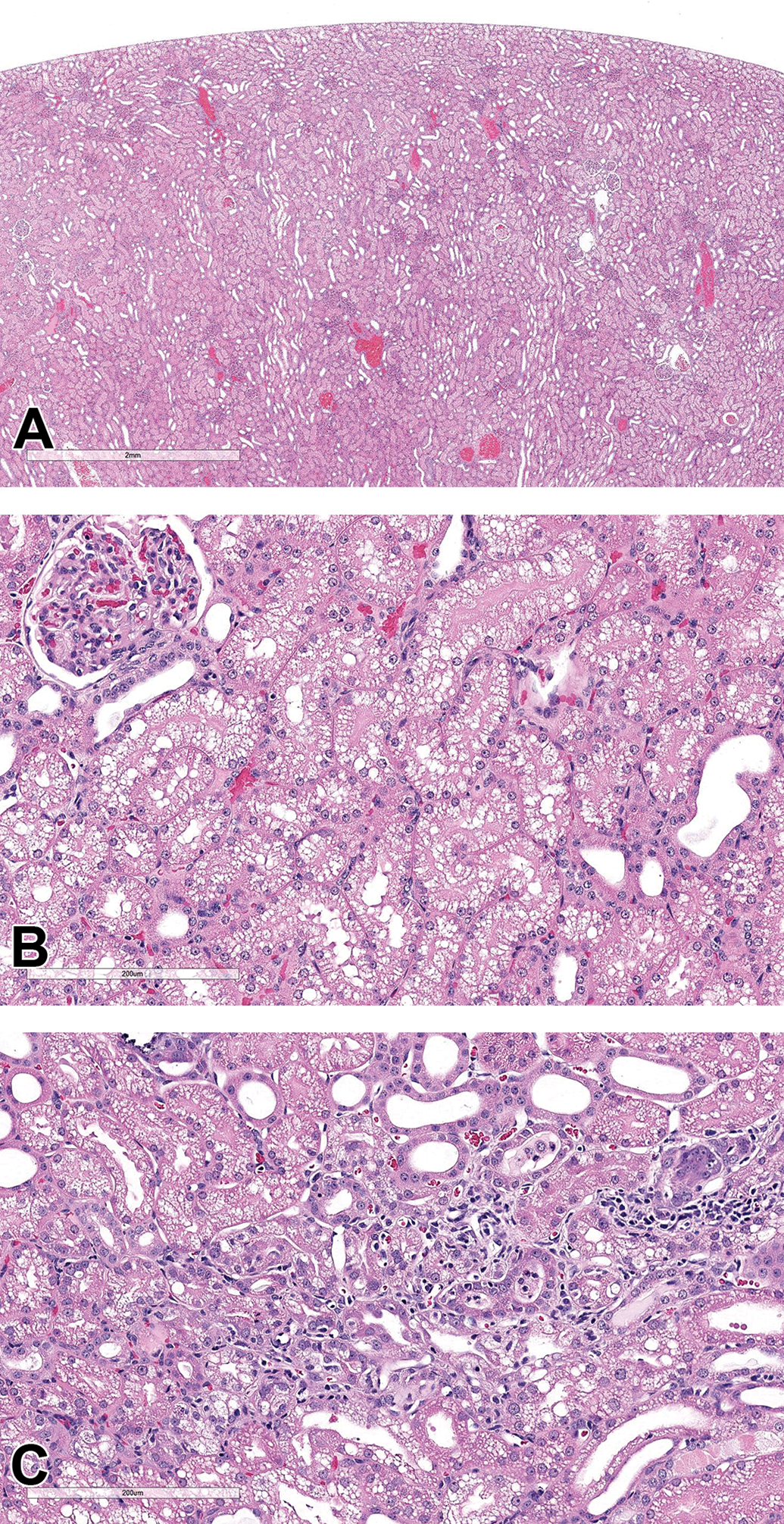
Dr Jerrold M. Ward (Global VetPathology) presented a case of chemically induced cystic kidneys in a 19-day old rat. The case was from a study by Nakazawa Tomomi and her colleagues at Bozo Research Center, Shizuoka, Japan. 20 Four-day-old (postnatal day, PND) male and female Sprague-Dawley [Crl: CD(SD)] rats were given p-cumylphenol (PCP). PCP is commonly used for the production of plastics, surfactants, fungicides, and preservatives. In this study, 15 animals per sex were treated orally with 300 mg/kg/d of PCP in olive oil once a day for up to 18 days from PND 4 and were sacrificed on PNDs 8, 12, 19, and 22 and after a 7-day recovery period. On PNDs 8 and 12, slight dilatation of the collecting ducts was frequently observed in the medulla, and slight papillary necrosis was also noted in some cases. These dilated collecting ducts were lined with slightly hyperplastic epithelial cells. On PNDs 19 and 22, multiple large cystic changes arising from the collecting ducts in the outer medulla were seen in all animals in the treated groups.
The animal presented by Dr Ward was sacrificed at 19 days. The kidney had large cystic tubules, noted as collecting ducts by the authors, which were not seen in the outer cortex (Figure 2A and B). The epithelium lining of the dilated tubules appeared hyperplastic but only rare mitotic figures were found. Proliferating cell nuclear antigen was increased in tubule nuclei. 20 No tubular casts, inflammatory cells, or other evidence of tissue damage were seen.
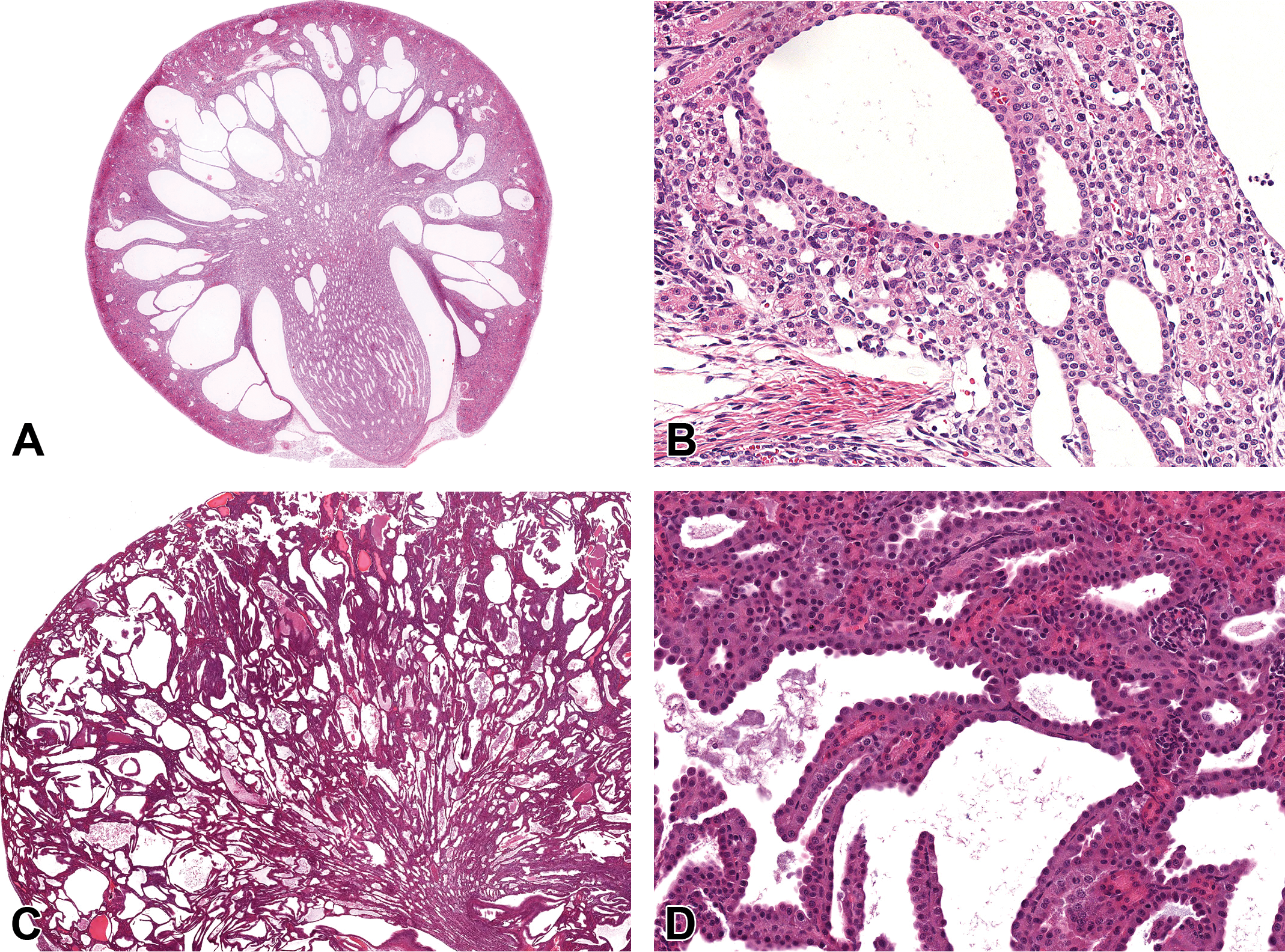
A, Polycystic kidney from a 19-day-old male Sprague Dawley [Crl: CD(SD)] rat. Dilatation of collecting ducts and less so of tubules in the cortex. High magnification (B) shows dilation of tubules. C, Cystic kidney from a 3-week-old
The voting choices and results were congenital lesion (15%), cysts (8%), dilatation of tubules (15%), dysplasia (5%), end-stage renal disease (0%), nephropathy (2%), and polycystic kidney (PCK; 55%). The diagnosis was PCK, also known as polycystic kidney disease (PKD), based on the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) kidney nomenclature guidance as follows. Polycystic kidney nor PKD was in the INHAND renal nomenclature, but PKD or other congenital cysts were noted in the differential diagnosis discussion regarding dilation, tubule. 22 Based on consideration of PCK in other species, Dr Ken Frazier, Chairman of the INHAND Urinary Tract Nomenclature Committee, and Dr Jerry Ward (presenter) suggested that PCK in rodents be considered when there are more than 6 cysts; the size of the lumina with multiple dilation is such that the nephrons cannot return to normal function (PCK is irreversible and progressive); there is no evidence of obstructive nephropathy or retrograde nephropathy; the primary diagnosis is not the result of other severe lesions such as chronic progressive nephropathy (CPN) or obstruction from crystalluria, hydronephrosis, or adjacent neoplasia; and the lesion is consistent among animals in the same dose group. Of course, evidence of genetic etiology or exposure in neonatal animals assists in the diagnosis.
The mechanism of kidney cyst formation is probably related to the toxic effects of the chemical on normal rat nephron differentiation 23 from the first day of treatment at PND 4. 20 The authors stated that the toxic effects also included cell debris (probably from necrosis) and neutrophil infiltration in the medullary tubules and interstitium. 20 Neonatal rats appear to be more sensitive than adult rats to renal toxins and to the development of PCK. 24
Markedly dilated renal tubules may be observed in other renal conditions including chronic nephropathy induced by chemicals, such as β-myrcene,
25
and to some degree in aging nephropathy. Polycystic kidney is also observed in rats,
26
mice,
21,27
and humans
28
with spontaneous or induced specific genetic mutations. A variety of genetically engineered mice show PCKs from an early age with diffuse cortical and medullary cysts (Figure 2C and D), sometimes involving cilia development.
27
Genes involved include those found in human PKDs (eg,
In summary, a case of greatly dilated renal tubules in a rat exposed to a renal toxin at PND 4 was diagnosed as PCK and compared to the genetic disease in humans and to genetically engineered rats and mice.
Nervous System Findings in NTP Control Mice and Rats
Dr Debra Tokarz (EPL) presented 2 cases of brain findings from control rats used in studies conducted by the NTP/NIEHS, along with a summary of historical control findings in the nervous system for mice and rats from NTP/NIEHS studies over a 10-year period. Dr Tokarz acknowledges contributions from Dr Ramesh Kovi (EPL), assistance in data collection from Cari Martini, Frances Liu, Julie Berke, Jamie Moose, and Jennifer Fostel (NTP/NIEHS), and imaging help from Emily Singletary, Maureen Puccini, and David Sabio (EPL).
Case 1 (Figure 3A and B) was from a female Sprague Dawley rat from the vehicle control group of a 26-week toxicity bioassay with feed administration. In the brain of this rat, there was unilateral cyst-like cavitation of a portion of the parietal cortex and adjacent thalamus. The cavity communicated with the lateral ventricle and the subarachnoid space. The presence of dystrophic calcification and rare hemosiderin-laden macrophages at the edge of the cavity suggested prior parenchymal injury. The voting choices and results were as follows: atrophy (2%), developmental malformation (51%), dysplasia (4%), hydrocephalus (24%), mineral (9%), necrosis (5%), and other (5%). The majority vote for this case agreed with the study pathologist’s diagnosis of developmental malformation. The changes in this case were postulated to have resulted from in utero or perinatal necrosis with subsequent cystic cavitation, consistent with porencephaly. 29 Porencephaly may result from a perinatal viral infection, nutritional deficiency, vascular, or traumatic insults 29- 31 and can be experimentally induced in rodents. 30,31
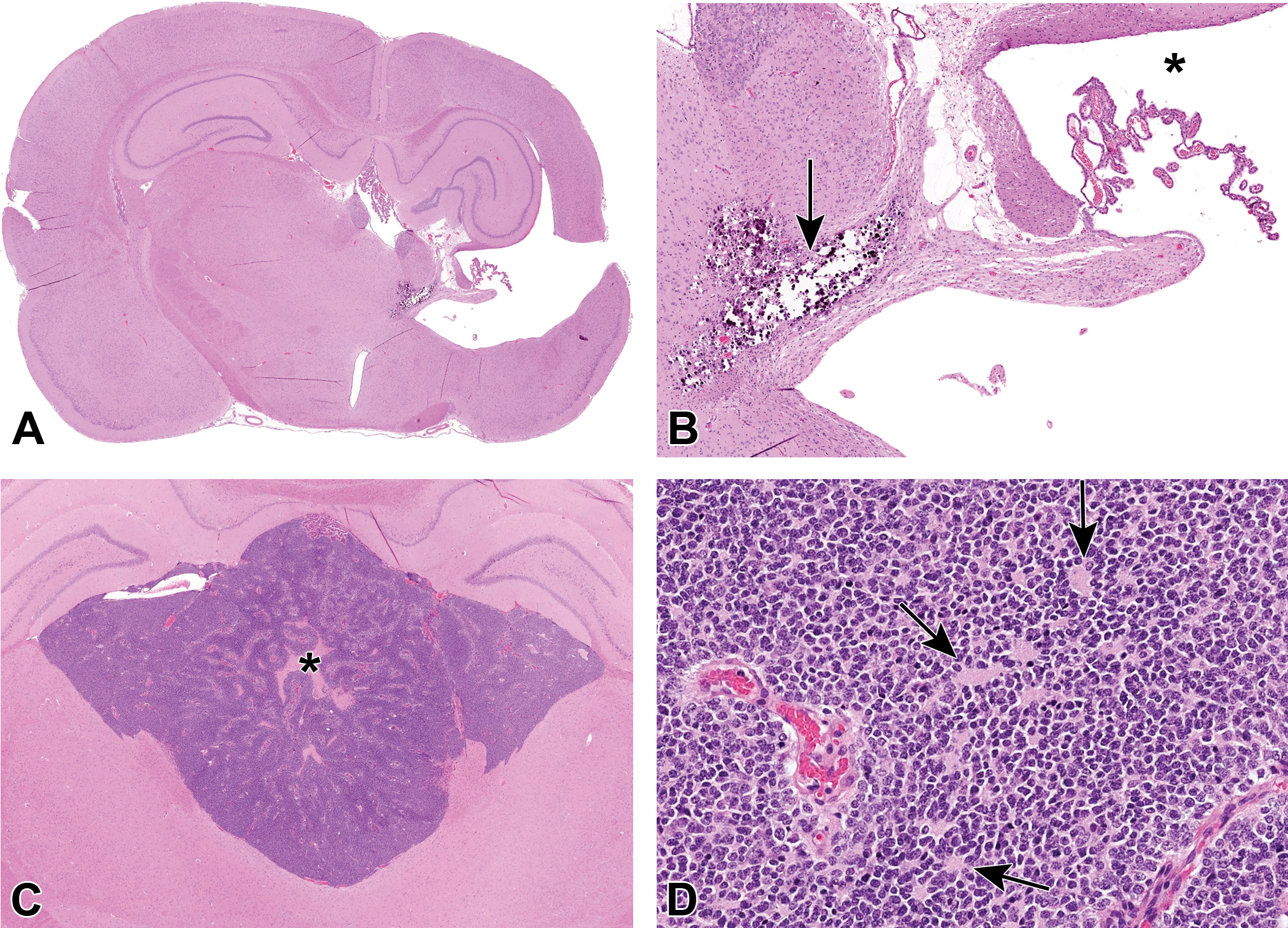
Brain from an adult Sprague Dawley [Crl: CD(SD)] rat from the vehicle control group of a National Toxicology Program (NTP) 26-week bioassay (A-B) and brain from an adult F344/N rat from the vehicle control group of an NTP 2-year carcinogenicity bioassay (C-D). A, There is partial cavitation of the cerebral cortex and thalamus unilaterally. B, The cavity communicates with the lateral ventricle (asterisk) and there is mineralization in the adjacent parenchyma (arrow). C, A densely cellular neoplasm expands the dorsal third ventricle and interventricular foramina. There is central necrosis (asterisk). D, The neoplastic population is relatively uniform with indistinct borders, moderate amounts of eosinophilic cytoplasm, round nuclei, and frequent rosette formation (arrows; hematoxylin and eosin [H&E]).
Case 2 (Figure 3C and D) was from a male Fischer 344 (F344)/N rat from a 2-year chronic toxicity/carcinogenicity bioassay with oral gavage administration. A densely cellular neoplasm expanded the dorsal third ventricle and interventricular foramina. There were central areas of serpiginous necrosis. Neoplastic cells were arranged in sheets, rosettes, and pseudorosettes. They had round nuclei and moderate amounts of eosinophilic cytoplasm with indistinct cell borders. Mitotic count was 1 of 10 high-power fields (hpf; 2.37 mm2). The voting choices and results were as follows: benign choroid plexus tumor (5%), malignant choroid plexus tumor (8%), benign ependymoma (23%), malignant ependymoma (27%), malignant glioma (3%), lymphoma (6%), primitive neuroectodermal tumor (PNET; 28%), and other (0%). The original diagnosis used for this case was benign ependymoma. Dr Tokarz described the histologic features of ependymoma that were evident in this tumor, which included ventricular location, rosette and pseudorosette formation, and cell morphology (polygonal cells, round hyperchromatic nuclei, indistinct borders). 32 Although these features are also evident in PNETs, another diagnosis favored by the audience, PNETs typically feature a more infiltrative growth pattern and most often arise from the cerebellum where they are referred to as medulloblastomas. 32 Criteria for malignancy in ependymoma were often lacking in older literature. 33,34 The INHAND criteria for malignancy in ependymoma include invasion of the neuropil, and other typical features of malignancy including pronounced cellular atypia or pleomorphism, tumor necrosis, and presence of multinucleated giant cells. 32 The original diagnosis was benign ependymoma, although some audience members favored a malignant diagnosis based on features such as necrosis and infiltration seen in the images presented.
Both of the presented cases were taken from a survey of findings in the nervous system (brain, spinal cord, nerves, ganglia) of control mice and rats taken from published NTP/NIEHS studies with report or peer review dates between 2008 and 2019. Dr Tokarz briefly summarized the results of this data survey. During the queried time period, there were 100 applicable studies in mice, all utilizing the B6C3F1 strain. Study lengths included 14-day (n = 4), 28-day (n = 2), 90-day (n = 51), and 2-year (n = 43). There were no findings recorded in the nervous tissue for controls in any study <90 days for mice. During the queried time period, there were 119 applicable studies in rats, which included the following 6 strains: F344/N, F344/NCTR, F344/NTac, Hsd: Sprague Dawley SD, Crl: CD (SD), and Crl: WI(Han). The majority, 75 studies, utilized the F344/N strain. Study lengths included 14-day (n = 10), 28-day (n = 10), 7-week (n = 1), 90-day (n = 51), 18-week (n = 1), 26-week (n = 1), and 2-year (n = 45). There were no findings recorded in nervous tissue for 14-day, 28-day, or 18-week studies. Because the vast majority of findings were recorded in control mice and rats from 2-year studies, Dr Tokarz summarized the nervous system findings from these controls.
The incidence rates of the most common spontaneous findings in the brain of control mice and rats from 2-year studies are shown in Table 2. The most common finding recorded in the brain of control mice was mineral. This is a common finding in the thalamus in B6C3F1 and CD-1 mice. 35,36 In contrast, the most common finding recorded in the brain across all rat strains was neoplasia, with an incidence rate near 4%. Incidence rates of nervous system neoplasia are generally higher in rats than mice. 37,38 The most common neoplasm recorded in the brain differed by rat strain. Among the F344 strains, mononuclear leukemia was recorded most often, while lymphoma and benign granular cell tumor were most common in Sprague Dawley and Wistar Han rats, respectively.
Incidence of Select Findings in Brain of Control Mice and Ratsa From NTP 2-Year Bioassays.
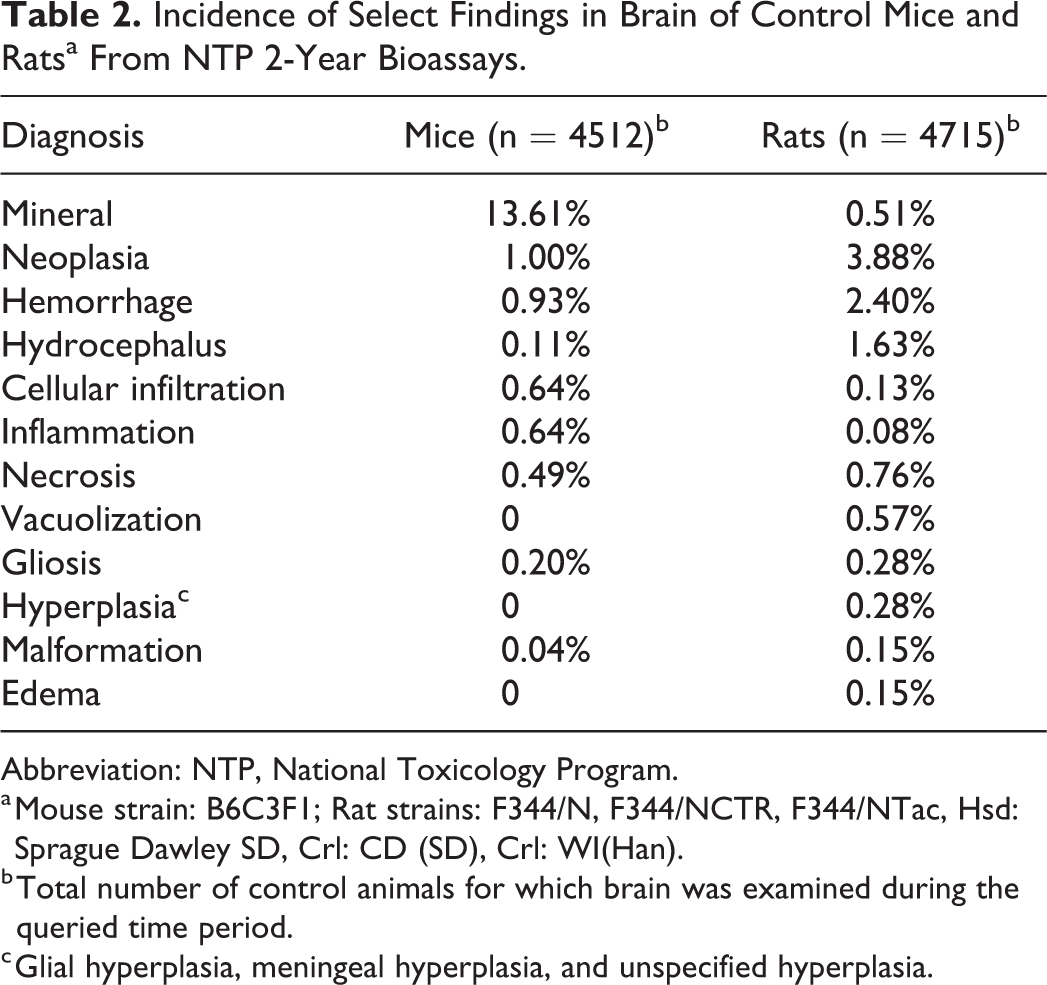
Abbreviation: NTP, National Toxicology Program.
a Mouse strain: B6C3F1; Rat strains: F344/N, F344/NCTR, F344/NTac, Hsd: Sprague Dawley SD, Crl: CD (SD), Crl: WI(Han).
b Total number of control animals for which brain was examined during the queried time period.
c Glial hyperplasia, meningeal hyperplasia, and unspecified hyperplasia.
The incidence rates of spontaneous findings in the spinal cord of control mice and rats from 2-year studies are shown in Table 3. Degeneration was by far the most frequently recorded finding in both species. As noted by audience participants, this may be seen in the spinal cord and spinal nerve roots at all levels in aged rats of various strains, although exact incidence rates are not always reported. 39 -41 Degeneration in the spinal cord occurs in aging mice as well, though at a lower frequency than in rats, 36 as seen in this data cohort (Table 3). In mice, degeneration was most frequently recorded in the thoracic spinal cord during the queried period. In the rats, degeneration was recorded at all levels of the spinal cord in the F344 and Sprague Dawley strains, although there was an opposite distribution in frequency. In the F344 strains, degeneration was recorded most frequently at the cervical segment, with a 37% incidence rate and decreased in caudal segments. In Sprague Dawley rats, however, the frequency of axon degeneration increased caudally with an incidence rate reaching 90% in the lumbar segment.
Incidence of Findings in Spinal Cord of Control Mice and Ratsa From NTP 2-Year Bioassays.
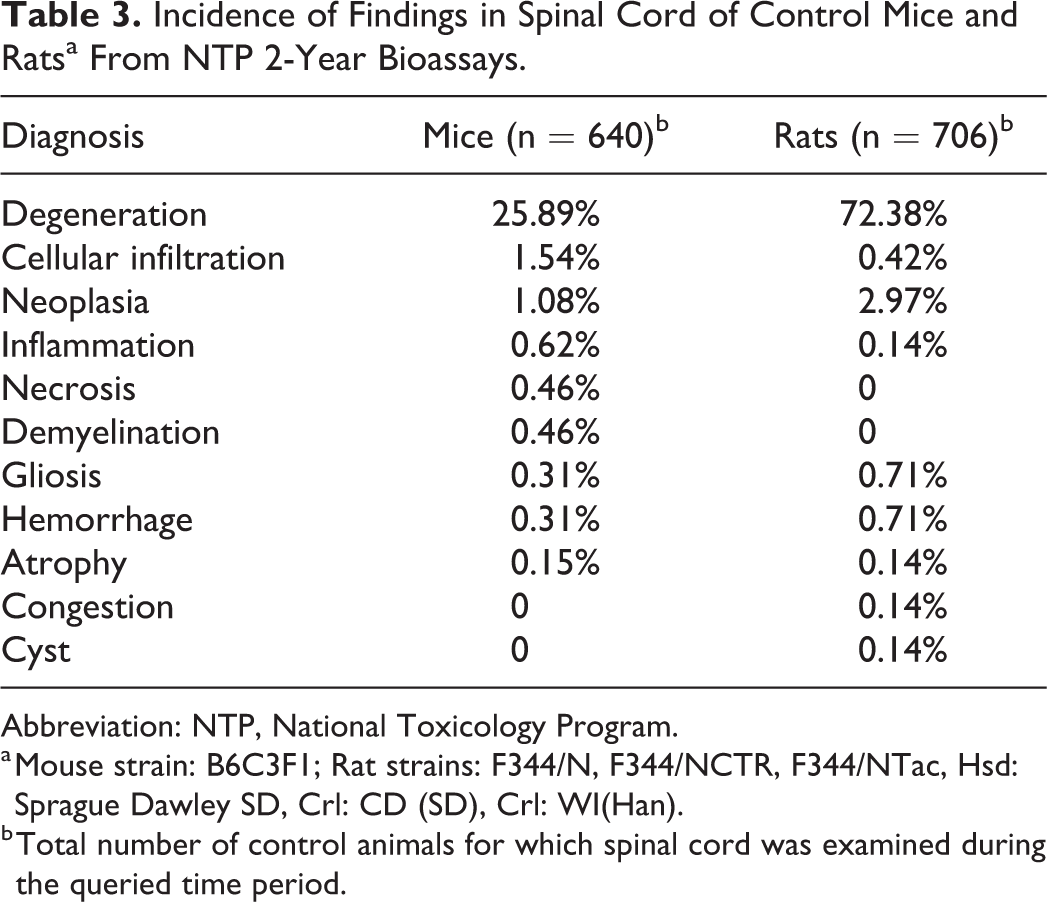
Abbreviation: NTP, National Toxicology Program.
a Mouse strain: B6C3F1; Rat strains: F344/N, F344/NCTR, F344/NTac, Hsd: Sprague Dawley SD, Crl: CD (SD), Crl: WI(Han).
b Total number of control animals for which spinal cord was examined during the queried time period.
The incidence rates of spontaneous findings in the peripheral nerves of control mice and rats from 2-year studies are shown in Table 4. Again, degeneration was the most common finding recorded in both mice and rats. This is also a reported aging change in both species, although it is typically considered to occur at much less frequency in mice. 42
Incidence of Findings in Peripheral Nerve of Control Mice and Ratsa From NTP 2-Year Bioassays.
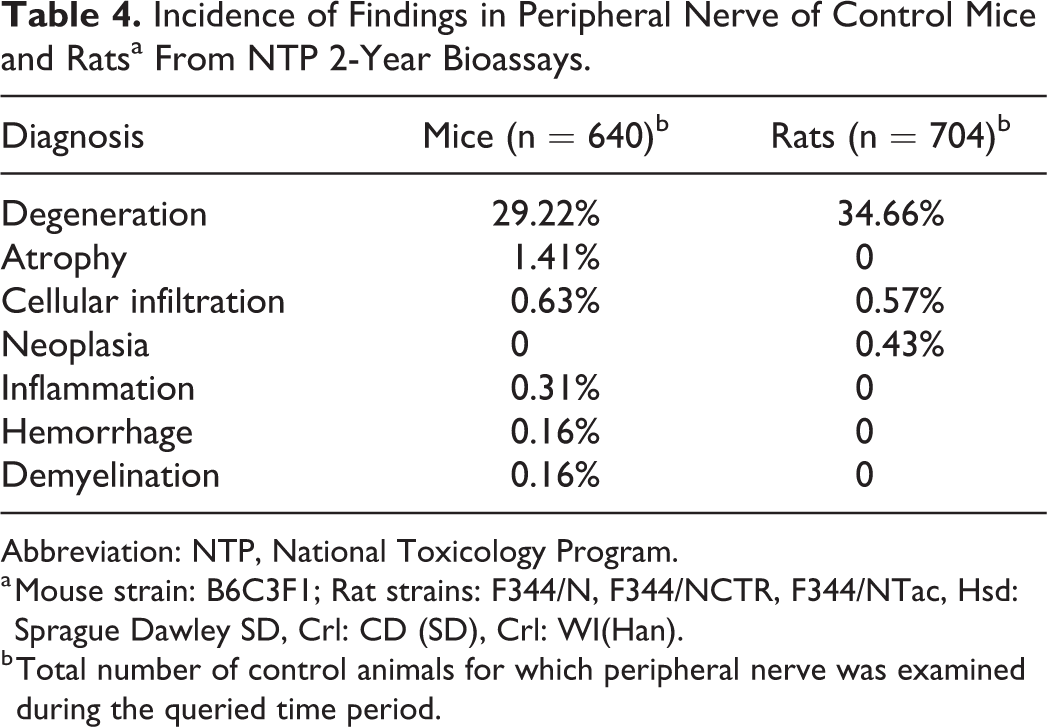
Abbreviation: NTP, National Toxicology Program.
a Mouse strain: B6C3F1; Rat strains: F344/N, F344/NCTR, F344/NTac, Hsd: Sprague Dawley SD, Crl: CD (SD), Crl: WI(Han).
b Total number of control animals for which peripheral nerve was examined during the queried time period.
Trigeminal ganglion was only evaluated in one 2-year study, using Sprague Dawley rats, during the queried time period. The only finding recorded in these control rats was degeneration, which was recorded in 27% of male and 37% of female Sprague Dawley rats in that study (data not shown in text table).
Kidney Lesions Caused by the Drug Vehicle Cyclodextrin
Dr Shambhunath Choudhary of Pfizer Inc presented vehicle-related renal lesions in cynomolgus monkeys from a non-good laboratory practice toxicity study.
Male and female monkeys, aged approximately 2.5-year-old, were part of a 14-day repeated-dose oral toxicity study. At necropsy, there were no significant gross pathology observations. Histopathologic evaluation revealed characteristic renal lesions (Figure 4A-C) in mid- and high-dose animals which correlated with clinical pathology findings of increases in blood urea nitrogen and creatinine concentrations.
(A) Low magnification showing vacuolation of renal tubular epithelium. (B) Higher magnification showing prominent intracytoplasmic vacuolation within renal tubular epithelial cells. (C) High magnification showing basophilic tubules with high nuclear density indicative of regenerative changes of damaged tubules; lumen contains necrotic tubular epithelial cells. Note the inflammatory cell infiltration within the interstitium surrounding the basophilic tubules (hematoxylin and eosin [H&E]).
Low and high-magnification photomicrographs of H&E-stained kidney sections with representative lesions observed in both sexes were shown to the audience for voting (Figure 4A-C). The most prominent cellular feature included vacuolated tubular epithelium in both cortical and medullary regions (Figure 4A and B). Multifocally, tubular epithelium exhibited degenerative and regenerative changes with scattered individual cell necrosis; these changes were occasionally associated with minimal interstitial inflammation (Figure 4C).
Voting choices and audience responses were (1) vacuolation, tubular epithelium (8%); (2) accumulation, glycogen, tubular epithelium (0%); (3) single cell necrosis, tubular epithelium (0%); (4) tubular degeneration/regeneration & inflammation (8%); (5) CPN (4%); (6) 1, 3, and 4 (66%); (7) 1 and 5 (10%); and (8) other (4%). Both Dr Choudhary and the audience favored choice 6, which included vacuolation, tubular epithelium; single cell necrosis, tubular epithelium; and tubular degeneration/regeneration and inflammation.
At this point, Dr Choudhary revealed that these histologic renal changes were vehicle or excipient related. A drug is rarely administered alone and always is a part of the combination that contains active pharmaceutical ingredient and vehicle/excipient. 43 Although the terms vehicle and excipient are used interchangeably in the field of toxicologic pathology, a vehicle can contain one or more excipients. As per European Pharmacopoeia “A vehicle is the carrier, composed of one or more excipients, for the active substance(s) in a liquid preparation.” For semisolid and solid drug preparation, the term basis is used instead of vehicle. Excipients protect, support, or enhance stability, bioavailability, or patient acceptability of the drug in use. They also assist in the effectiveness and/or delivery of the drug in use and assist in maintaining the integrity of the drug product during storage. 43 A list of commonly used vehicles/excipients in pharmaceutical drug products was then presented and the audience was asked to vote for their preferred vehicle associated with previously shown microscopic renal lesions. The voting choices and results were (1) dimethylacetamide (2%), (2) glycerine (0%), (3) cyclodextrin (CD; 46%), (4) ethanol (0%), (5) dimethylsulfoxide (6%), (6) liposomes (2%), (7) 1& 6 (26%), and (8) I don’t know (18%). Dr Choudhary again favored the most popular diagnosis (choices 3) of the audience. This set the stage for a quick review and discussion on CDs.
Cyclodextrins are a family of cyclic oligosaccharides made up of 6 to 8 dextrose units (α-, β-, and γ-CDs, respectively) and have mainly been used in the pharmaceutical industry as complexing agents to increase the aqueous solubility of poorly soluble drugs, and to increase their bioavailability and stability. 44 Among various derivatives of α-, β-, and γ-CDs, 2 modified β-CDs, sulphobutylether-β-CD (or Captisol) and hydroxypropyl-β-CD (HP-β-CD) have undergone extensive safety studies and are currently used in many Food and Drug Administration (FDA)–approved drugs. Since CDs are bulky and hydrophilic, only insignificant amounts are absorbed from the intact gastrointestinal (GI) tract on oral administration. Systemic absorption of CDs happens upon parenteral administration. Although CDs distribute to various tissues including the kidney, urinary bladder, liver, adrenal gland, and others, the kidney has the highest level of accumulated CDs and highest retention period of all tissues (several hours to a couple of days). Most CDs disappear from the tissue dramatically within the first several hours of administration and are renally excreted. 44,45
Oral administration of CDs is generally safe in animal studies, but GI tract effects including soft feces or diarrhea and cecal enlargement have been observed in a few rat and dog studies. These changes, however, are reversible and thought to represent physiologically adaptive responses to a large load of poorly digestible carbohydrates and other osmotically active nutrients, and hence, the relevance to humans is minimal. 45 Upon parenteral administration or following systemic absorption of CDs, the kidney is mostly affected due to the highest level of CD accumulation and retention in this organ. In the kidney, the most common observation is vacuolation of proximal tubular epithelium with a low dose of systemically absorbed CDs. This change is reversible following withdrawal of treatment and, if present alone, suggests physiological adaptation. However, with a higher dose of CDs or prolong treatment period, vacuolation may progress to degeneration, cell injury/cell death, and eventual alterations of renal functions. The length of period for complete recovery of renal lesions may depend on the rapidity of cell turnover, severity of lesions, and period and dosage of treatment. 45 In addition to the renal findings, vacuolation of hepatocytes and umbrella cells in urothelium (urinary bladder) and foamy macrophages in the liver, lung, and lymph nodes have been observed with very high doses or prolong period of CD exposure when administered parenterally. In addition, mild increases in hepatic transaminases without any histopathologic correlates have been observed. Cyclodextrin-related toxic effect has been mainly observed in rodent and dog studies; published data are limited among monkeys. 46
Characteristic microscopic lesion (vacuolation of renal tubular epithelium) along with knowledge of CD as a vehicle in drug preparation is the most useful diagnostic criteria for CD-related renal toxicity. Electron microscopy can be used to visualize CD-specific changes in the renal tubular epithelium; electron microscopy studies showed that the vacuoles were membrane bound and contained finely granular and some electron-dense membranous profiles, indicating lysosomal origin. 45 Cytoplasmic vacuolation of renal tubular epithelium can also be seen with phospholipidosis-inducing chemicals (eg, cationic amphiphilic drugs), vehicles (eg, polyethylene glycol–based vehicles and dextran) or it can be related to autolysis or poor fixation (artifactual vacuolation). 47
Carcinogenic potential of CDs has been evaluated in rats and exocrine pancreatic tumors have been observed following HP-β-CD oral administration; however, numerous studies suggest that acinar tumor of rats may be irrelevant to human risk. 45 Recently, subcutaneous sarcomas related to chronic subcutaneous administration of Captisol in rats have been reported. 48
Dr Choudhary then briefly presented the study design including the details of vehicle used in this study (20% [wt/vol] sulfobutyl ether β-CD). He then explained why these renal lesions were considered CD-related and were observed only in mid- and high-dose animals. The appearance and distribution of renal lesions (renal tubular vacuolation) in this study were typical of CD-related renal findings as described in published literature. In addition, these renal lesions were not observed with other studies that used a different vehicle. Mid- and high-dosed animals in this study had severe test-article-related ulceration/erosion in the GI tract which led to greater absorption of CD and eventual accumulation in the kidney. Additionally, these animals were dehydrated (secondary to diarrhea and emesis) that caused a reduced glomerular filtration rate leading to a higher accumulation of CD in the kidney. Both these factors (ulceration/erosion of GI tract and dehydration) contributed to toxic levels of CD accumulation in the kidneys. Since insignificant amounts of CDs are absorbed from the intact GI tract, vehicle control and low-dose animals in this study didn’t accumulate significant CDs in the kidney and hence no toxic changes were observed.
Dr Choudhary’s presentation ended with a lively audience discussion. Many attendees mentioned that they have observed CD-related lesions in the kidney of rodents, dogs, and monkeys. Some attendees remembered seeing toxic effects in the lungs (alveolar macrophage vacuolation) and liver (hepatocyte vacuolation) as well.
The main purpose of this presentation was to raise awareness among toxicologic pathologists about toxic changes specifically related to vehicles used in regulatory toxicity studies. It is hoped that practicing pathologists will now be able to differentiate CD-related changes described here from direct test-article-related changes. Pathologists should also pay attention to factors that can enhance/contribute to vehicle bioavailability and toxicity. For example, test-article-related changes or overall body condition may influence the overall vehicle concentration within the body fluids or tissues.
Uterine Neoplasms in Rats From a 2-Year Chronic Bioassay
Quinci Plumlee (CRL) presented 2 cases that highlighted uterine neoplasms in Hsd: Sprague Dawley SD rats from an NTP 2-year, oral gavage, and carcinogenicity bioassay. Dr Plumlee acknowledged Drs Amy Brix (EPL), Anthony Skowronek (Battelle), and Ashley Talley (Rollins Animal Disease Diagnostic Laboratory) for their contribution to this study and/or presentation.
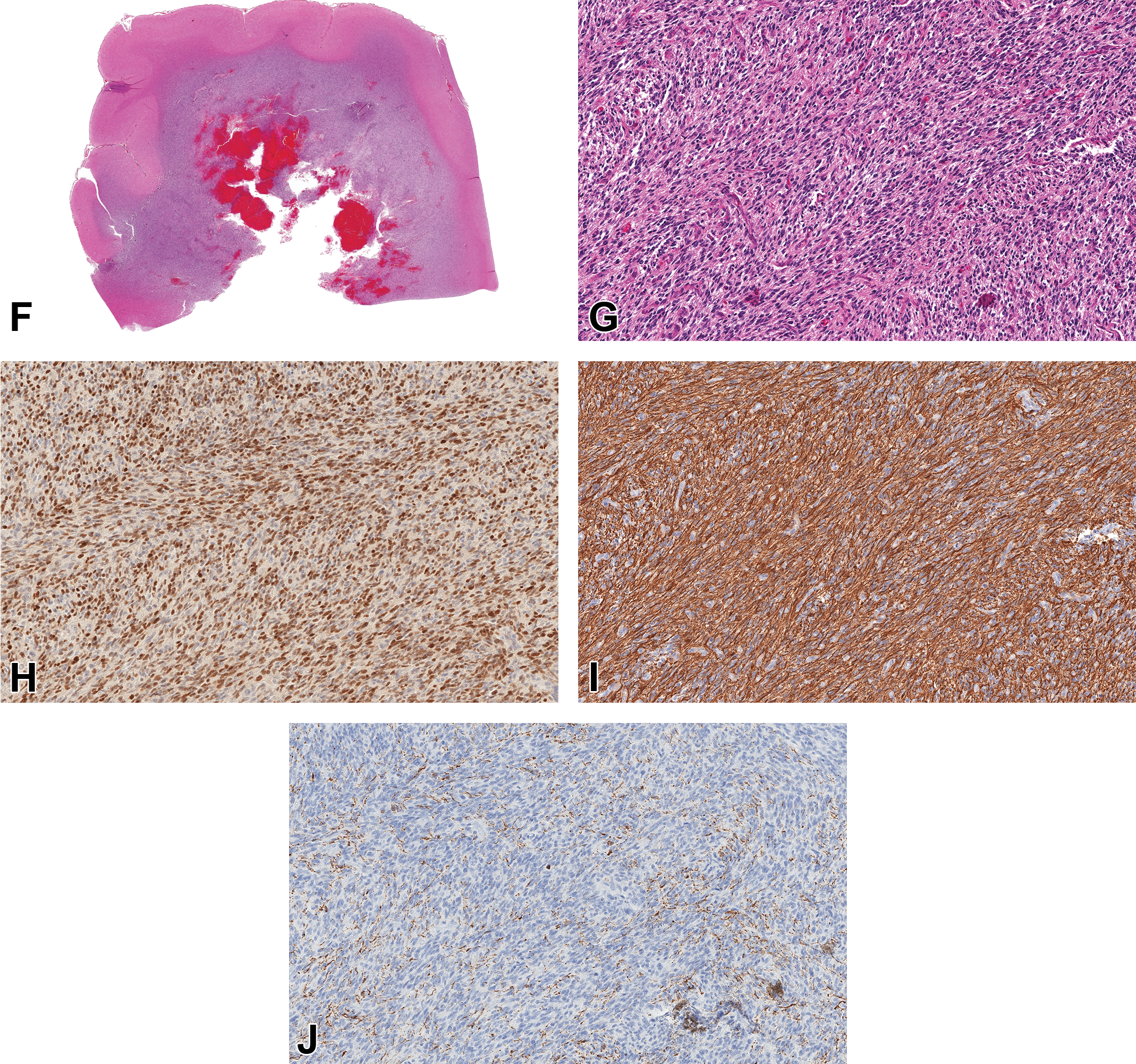
The first case was a uterus from a high-dose female who survived until study termination. Microscopically, 2 adjacent exophytic masses arose from the mucosal surface of a markedly thinned uterus (Figure 5A-D). The left exophytic mass was composed of well-differentiated columnar epithelium with prominent goblet cells and scattered Paneth cells, arranged in simple or branching tubuloglandular structures, and supported by a fine fibrovascular stroma. Mitoses were approximately 0 to 1/hpf. In contrast, the right exophytic mass was composed of pleomorphic cells arranged in poorly organized papillary structures supported by minimal fibrovascular stroma. Focally, the pleomorphic cells appeared to invade beyond the basement membrane, and multiple dilated glands underlying the masses were lined by similar atypical cells. Atypia, anisocytosis, and anisokaryosis were marked. Mitoses were approximately 2 to 3/hpf with bizarre mitoses present. Low numbers of mixed inflammatory cells admixed with minimal hemorrhage infiltrated the masses and underlying endometrium.
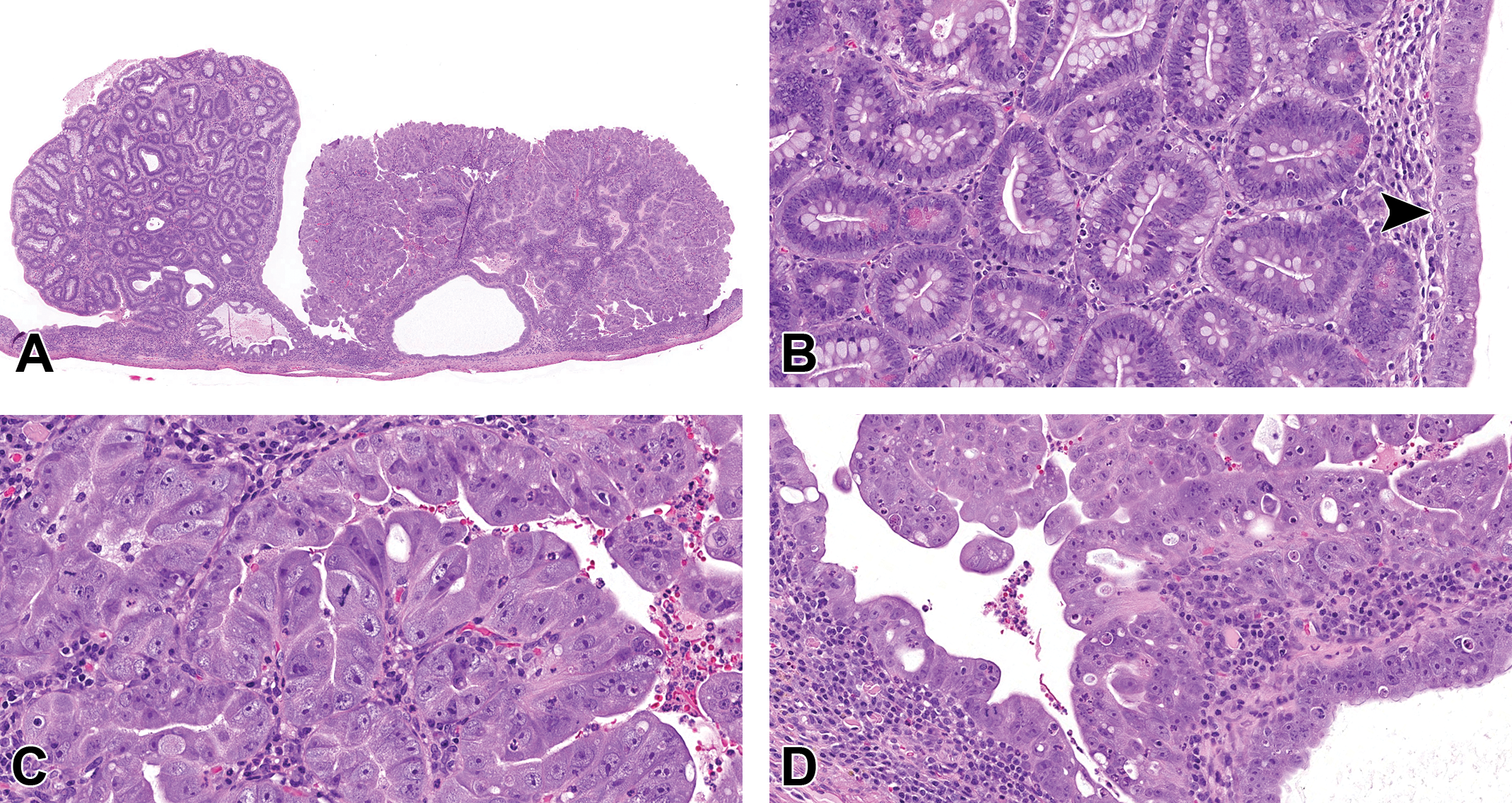
(A-D) Endometrial adenocarcinoma from a 2-year-old female rat from a National Toxicology Program (NTP) chronic carcinogenicity bioassay presented as case 1. A, Two adjacent exophytic masses arise from a markedly dilated and thinned uterus. B, The left mass is composed of well-differentiated enterocyte-like epithelial cells arranged in tubuloglandular structures and supported by a fine fibrovascular stroma. Goblet cells and scattered Paneth cells are throughout the mass with approximately 0 to 1 mitoses per high-power field. C, The right mass is composed of pleomorphic cells arranged in poorly organized papillary structures supported by minimal fibrovascular stroma. D, The pleomorphic cells appear to focally breach the basement membrane, line multiple dilated glands underlying the masses, extend to cover the superficial surface of the left mass (arrowhead in B). Atypia, anisocytosis, and anisokaryosis are marked. Mitoses are approximately 2 to 3 per high-power field, and bizarre mitoses are present (hematoxylin and eosin [H&E]).
Because there was little remaining normal uterine architecture and the unusual morphology of the masses, the voters were asked to identify both the organ and the lesion(s). The organ choices and voting results were urinary bladder (8%), gallbladder (7%), intestine (48%), uterus (38%), and other (0%). The diagnosis choices and voting results were glandular polyp (5%), adenoma (0%), adenocarcinoma (14%), glandular polyp and adenoma (26%), glandular polyp and adenocarcinoma (44%), adenoma and adenocarcinoma (11%), and other (0%). Dr Plumlee informed the audience that there was much discussion among the pathology working group (PWG) of diagnostic nuances related to this case, and ultimately, the preferred diagnosis by the PWG for this case was uterus, endometrium adenocarcinoma.
Three main topics of the discussion among the PWG for this case included the microscopic features and criteria of malignancy; if the lesion represented a single neoplasm or 2 neoplasms; and the unusual intestinal morphology within the neoplasm. The first and arguably most important diagnostic challenge was determining if this lesion met the criteria of malignancy. As stated above, the PWG discussed the significant atypia of the right mass and the presence or absence of invasion. The PWG decided that while the right mass was predominantly exophytic there appeared to be enough evidence (including focal disruption of the basement membrane and involvement of underlying glands by atypical cells) that invasion could not be definitively ruled out. Therefore, the PWG decided the criteria of malignancy were met for the lesion, and adenocarcinoma was preferred. Second, the PWG discussed if the lesion should be classified as 1 mass or 2. The more atypical cells extended from the right mass and lined the left mass and the transition between the 2 was quite subtle. Therefore, the PWG decided to make a single diagnosis for this lesion. Finally, the PWG discussed the unusual intestinal differentiation including the presence of goblet cells and Paneth cells of the left mass. Additionally, Dr Plumlee informed the audience there were additional distinct masses in this uterus (not shown in presentation) that demonstrated invasion and/or atypia, in the uterus of this female, and the final diagnosis for the uterus was endometrium, adenocarcinoma, multiple.
Endometrial neoplasms in rodents are categorized into glandular polyps, adenomas, and adenocarcinomas. 49,50 Glandular polyps are benign polypoid masses that protrude into the lumen with well-defined endometrial stroma and hyperplastic to cystic endometrial glands. Adenomas are well-delineated, solitary, generally exophytic and papillary masses with little to no stroma and lack invasion and atypia. Adenocarcinomas are less well-circumscribed, form nests, cords, papillary, or acinar structures, have more cellular pleomorphism and atypia, and may invade the myometrium or metastasize to distant sites. Differentiation between adenoma and adenocarcinoma may rely upon cellular pleomorphism and atypia, cribriform pattern, and/or evidence of invasion or metastasis. 49,50 Focal glandular hyperplasia (adenomatous hyperplasia or atypical hyperplasia) of the endometrium, which also displays endometrial epithelial and/or glandular proliferation, possible cellular atypia and papillary projection formation, can be distinguished from endometrial neoplasia by the absence of compression and lack of a distinct mass. 49
Endometrial adenomas and adenocarcinomas are uncommon in rats except in Donryu, 51 DA/Han, BDII/Han, and aged virgin Wistar/Han, 52 and following exposure to certain compounds. 50 In rats, adenomas and adenocarcinoma are typically only described as poorly or well differentiated. Additionally, intestinal differentiation, as observed in this case, has not previously been described in rodents. In contrast, endometrial neoplasia is common in women, with greater than 200,000 cases in the United States and United Kingdom combined per year. 53 Endometrial neoplasms, however, are biologically, morphologically, genetically, and clinically heterogeneous, and classification and grading systems recommend utilizing histomorphology, clinical/epidemiologic features, and genetic alterations to differentiate these tumors. 53 Intestinal differentiation of endometrial neoplasms was first described by Berger et al 54 and is reported to rarely occur in women. Histologically, intestinal differentiation is comprised of enterocyte-like cells with the potential presence of brush border, goblet cells, and/or Paneth cells. Reports describe it as a stand-alone metaplastic change in the endometrium to metaplasia/differentiation of several pathologic processes including endometriosis, 55 uterine polyps, and uterine carcinomas. 56,57 When observed in carcinomas it is considered potentially indicative of more aggressive behavior. 56,58
Case 2 was a uterus from a mid-dose female that was humanely euthanized on study day 644. Microscopically, within a markedly dilated uterus was an exophytic mass composed of papillary projections that progressed to an area of invasive, solid growth (Figure 5E-H). Papillary projections were composed of neoplastic stratified squamous epithelium with a prominent stratum corneum with premature keratizination/dyskeratosis and mitotic activity primarily in the basal layer. Mitoses were 0 to 1/hpf in this region. Papillary projections abruptly transitioned to areas of invasive irregular islands and nests of polygonal cells into the underlying endometrium. In other areas, neoplastic cells were poorly differentiated with significant anisocytosis, anisokaryosis, pleomorphism, and loss of distinct cell borders. Individual cells, however, contained intracellular keratin filaments and small keratin pearls were scattered throughout. The mitoses were 1 to 3/hpf in this region. Neoplastic cells had variable faint to intense cytoplasmic staining with pan-cytokeratin IHC stain.
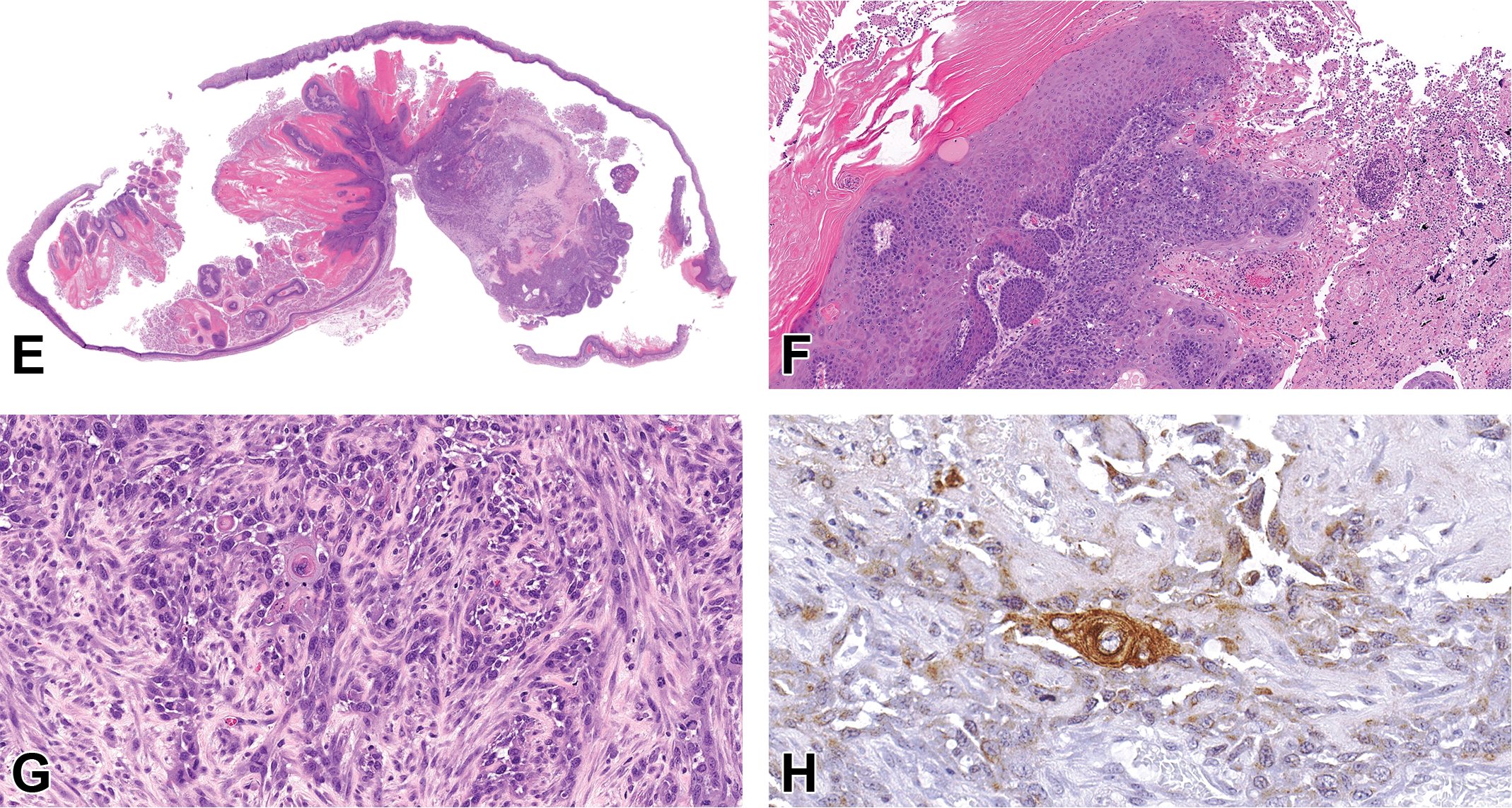
(Continued). E-H, Squamous cell carcinoma arising within a squamous papilloma of the uterus from an approximately 2-year-old female rat from an NTP chronic carcinogenicity bioassay presented as case 2. E-G, Stained with H&E and (H) is stained with pan-cytokeratin immunohistochemical (IHC) stain. E, Arising within a markedly dilated uterus is an exophytic mass composed of papillary projections that progresses to an area of invasive, solid growth. The papillary projections are composed of neoplastic stratified squamous epithelium with a prominent stratum corneum. Mitoses are generally confined to the basal layer and are 0 to 1 per high-power field. F, Papillary projections abruptly transition to areas of invasive, moderately well-differentiated to poorly differentiated, polygonal cells forming irregular islands and nests into the underlying endometrium and myometrium. G, In areas of poor differentiation, neoplastic cells are arranged haphazardly without forming clear islands or nests and have marked atypia, anisocytosis, and anisokaryosis with loss of distinct cell borders. Occasional atypical, neoplastic cells contain intracellular keratin filaments and/or form small keratin pearls. Mitoses are 1 to 3 per high field in this region and are frequently bizarre. H, Neoplastic cells throughout the mass, including the poorly differentiated areas, had variable, faint to strong, cytoplastic staining with pan-cytokeratin IHC.
Low- and high-power images of the areas of interest were shown to the audience, and voters were asked to identify the lesion. The choices and voting results were squamous papilloma (3%), squamous cell carcinoma (SCC; 69%), carcinosarcoma (16%), mixed Mullerian tumor (9%), endometrial stromal sarcoma (3%), teratoma (0%), and other (0%). Dr Plumlee informed the audience the PWG’s preferred diagnosis for this case was a SCC arising within a squamous papilloma, and that there were several uterine squamous findings in this study including squamous metaplasia, squamous papillomas, and SCC.
In rats, endometrial squamous metaplasia is common and well documented, but squamous neoplasia is uncommon. Squamous metaplasia is typically localized within endometrial glands (rarely a diffuse change) that develops under estrogen influence and can be a reactive change with inflammation. Brix and colleagues reported a 44% incidence in Sprague Dawley rats in a series of NTP studies. 59 Squamous differentiation of endometrial tumors is also quite common. If the squamous component of an endometrial tumor is highly atypical, the lesions are classified as adenosquamous carcinomas. In contrast, squamous neoplasms of uterine origin are rare spontaneous tumors and more commonly arise from the cervix and/or vagina within the female reproductive tract. 50 Squamous cell carcinomas have been reported to be induced in rats with topical application of chemical-impregnated threads. 60
In women, primary SCC of the endometrium is also extremely rare, but aggressive and associated with a poor prognosis. 61 Primary SCC is defined by a lack of coexistence of endometrial adenocarcinoma present, no connection to cervical or vaginal squamous epithelium, and no coexistence of SCC of cervix. 62 Case reports describe similar moderately differentiated neoplastic cells as observed in this case. 61
During the audience discussion, audience members described similar experiences in chronic/carcinogenicity studies with occasional endometrial adenocarcinomas and rare uterine SCCs in aged female rats. Additionally, the importance of establishing the potential involvement of the cervix with all squamous findings within the uterus was reiterated by audience members and Dr Plumlee alike. In conclusion, there were multiple and varied proliferative uterine findings in this study. Intestinal differentiation was observed in an endometrial adenocarcinoma that has not previously been reported in rats, and malignant transformation of a uterine squamous papilloma was observed.
A Discussion About Terminology for Degenerative Foci in the Aging Rat Adrenal Gland
Dr Andrew Suttie (Labcorp Early Development Laboratories, Inc) gave a presentation on degenerative foci in the adrenal cortex of aging Sprague Dawley rats. It was noted that publications (eg, INHAND) and textbooks typically present clear-cut examples of lesions with a single pathological process only. 63 Examples of these focal lesions include hyperplasia, hypertrophy, vacuolation, vascular ectasia, and cystic degeneration. The published literature also includes examples of diffuse changes in the adrenal cortex resulting from compound administration and physiological changes. Examples of compound-induced changes include lipid vacuolation from the administration of imidazole antimycotic drugs, hyperplasia from clenbuterol administration, 64 and hypertrophy from angiotensin II (restricted to the zona glomerulosa). 65 An example of physiological effects on the adrenal cortex includes diffuse hyperplasia in pregnant rats. 66
Focal degenerative lesions are common in the adrenal cortex of aging rats, notably in the Sprague Dawley rat. These lesions are usually spontaneous although there are examples of compounds that induce focal regenerative hyperplasia. 64 Additionally, it was noted that reduced numbers of these lesions are observed in rats that have undergone dietary restriction, either by experimental design, as a result of tumors in other organs, or from the administration of compounds that affect food consumption.
Dr Suttie pointed out that the focal degenerative lesions noted in the adrenal cortex of aging rats generally include 2 or more processes, which leads to questions as to how to report these lesions. Some approaches to reporting these lesions are using the terminology of the predominant process, reporting each aspect of the focus separately with the different terms, and using terms that combine the processes. Standardization for Exchange of Nonclincal Data (SEND) criteria allow for the use of only one forward slash in a term; therefore, a single term that includes numerous processes (eg, hypertrophy/hyperplasia/cystic degeneration) is not practicable.
Before presenting and discussing adrenal gland lesions that include more than one process, some examples of lesions with only one process occurring were shown to the audience. These examples included adenoma, focal hypertrophy of the zona glomerulosa, vascular ectasia, and an advanced example of cystic degeneration. Two examples of adrenal lesions with only one process were then presented to the symposium for voting. Case 1 (Figure 6A) was an example of an adrenal gland with a lesion midway between vascular ectasia and cystic degeneration. The voting choices and results were as follows: angiectasis (48%), vascular ectasia (27%), cystic degeneration (18%), within normal limits (7%), and other (0%). Case 2 (Figure 6B) was a focal lesion consisting both of vacuolation on the inner (medullary) aspect and slightly enlarged, hypereosinophilic, cells peripherally. The voting choices and results were as follows: vacuolation (55%), cystic degeneration (15%), hypertrophy (9%), hypertrophy/hyperplasia (4%), and hyperplasia (0%).
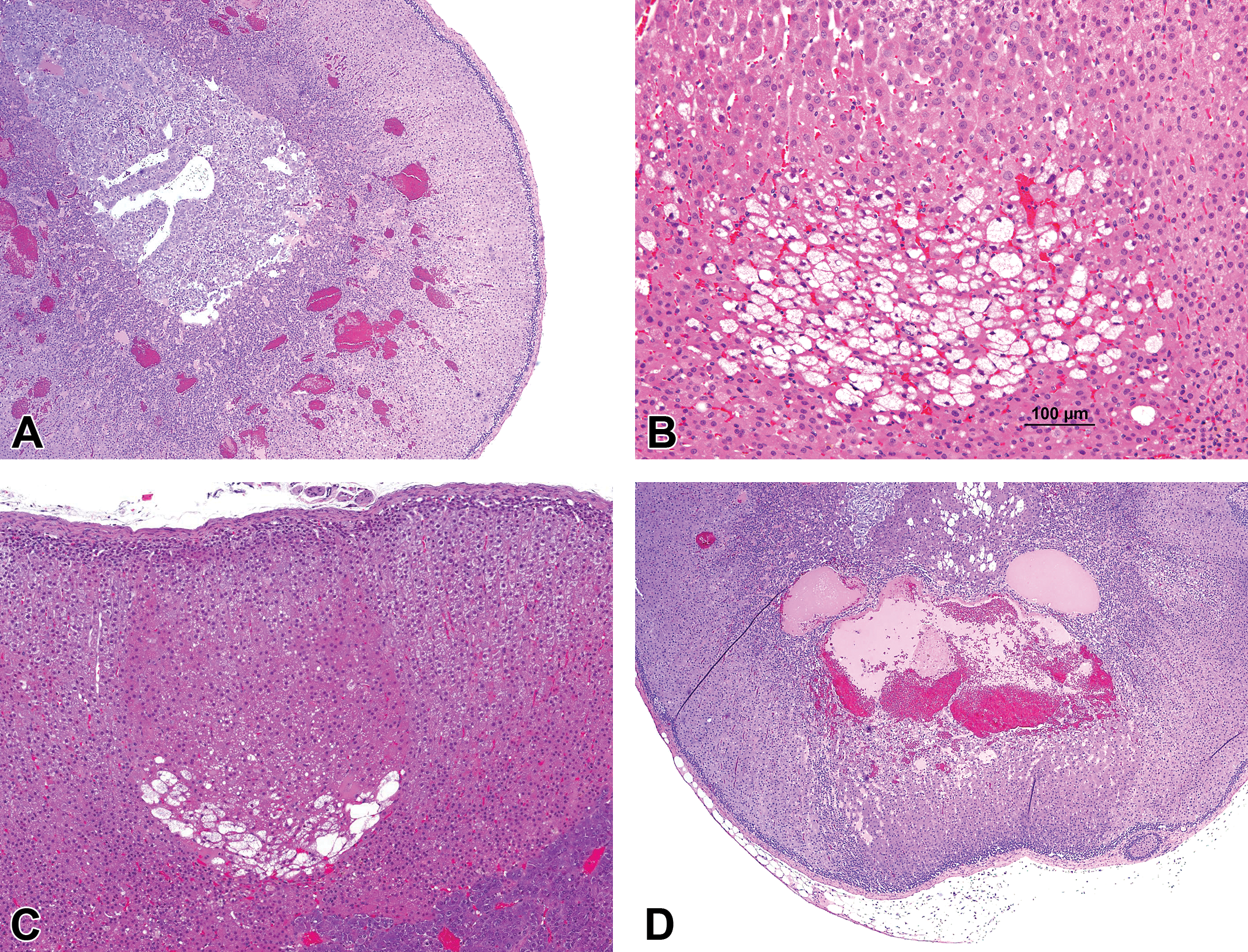
(A-D) Case 1 (A) is an adrenal cortex with dilated, blood-filled, intertrabecular spaces. Preferred diagnosis: angiectasis. Case 2 (B) is an area of adrenal cortex with slightly enlarged, hypereosinophilic cells (top) and markedly vacuolated cells (bottom), some of which are showing degeneration and dropout. Preferred diagnosis: vacuolation. Case 3 (C) is similar to case 2 but with larger empty spaces following cell degeneration dropout/cystic degeneration. Preferred diagnosis: hypertrophy/hyperplasia/vacuolation. Case 4 (D) is a complex lesion extending across the width of the adrenal cortex, with various areas of enlarged, hypereosinophilic cells, vacuolation, large spaces filled by blood or serum, and other areas with cell dropout. Preferred diagnosis: hypertrophy/hypertrophy/cystic degeneration (hematoxylin and eosin [H&E]).
Next, the audience viewed several examples of mixed focal adrenal gland lesions consisting of 2 or more of the following processes: hyperplasia, hypertrophy, vacuolation, vascular ectasia, and cystic degeneration. The audience was then presented with a third case for voting (Figure 6C), which was a relatively simple focus with slightly enlarged, hypereosinophilic cells with minimal amounts of vacuolation, and an area of early cell dropout/cystic degeneration on the aspect closest to the medulla. The majority vote (32%) was for the combined term hypertrophy/hyperplasia/vacuolation, with cystic degeneration at 26%, hypertrophy/hyperplasia/cystic degeneration at 22%, hypertrophy/hyperplasia at 12%, focal cellular alteration at 6%, and vacuolation at 2%. The final lesion considered by the symposium (Figure 6D) was more complex, with various areas of enlarged, hypereosinophilic cells, vacuolation, large spaces filled by blood or serum, and other areas with cell dropout. The majority vote (52%) was for hypertrophy/hypertrophy/cystic degeneration followed by cystic degeneration at 33%, hypertrophy/hyperplasia/vacuolation at 6%, and hypertrophy/hyperplasia and focal cellular alteration both at 2%.
In summary, focal degenerative lesions in the adrenal cortex are common in aging rats, especially in the Sprague Dawley rats and terminology to report the multiple aspects of these lesions is not well established. After reviewing several examples of these lesions, the symposium voted on examples with 2 or more concurrent processes. Overall, the preference of the symposium was to utilize either a term representing the predominant process occurring in the lesion or alternatively a combined term describing the processes, although understanding that SEND only allows for use of one forward slash in a term.
Diagnostic Challenges Associated With Proliferative Changes in the Rat Uterus
Dr Erin M. Quist (EPL) presented several interesting cases recently reviewed by both a PWG and pathology peer review (PPR) conducted at the NTP/NIEHS. The presented cases featured lesions from 3 different female, Sprague Dawley rats that were part of a 2-year toxicity/carcinogenesis study in which Dr Anthony Skowronek (Battelle Columbus) was the study pathologist and Dr Quist was the quality assurance pathologist. Thanks to Drs Arun Pandiri, Darlene Dixon, and Gabrielle Willson for their assistance and Emily Singletary for photographic editing and support.
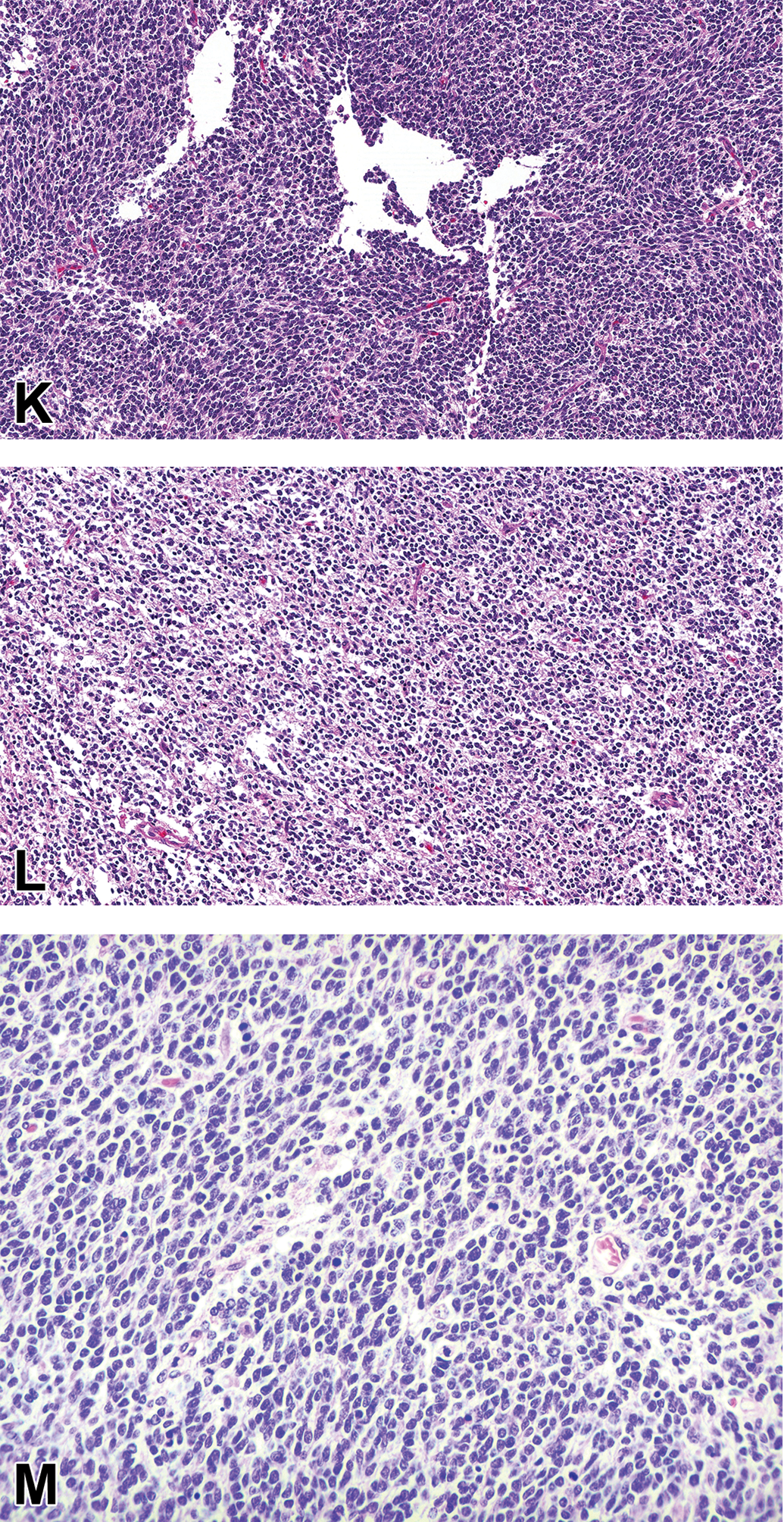
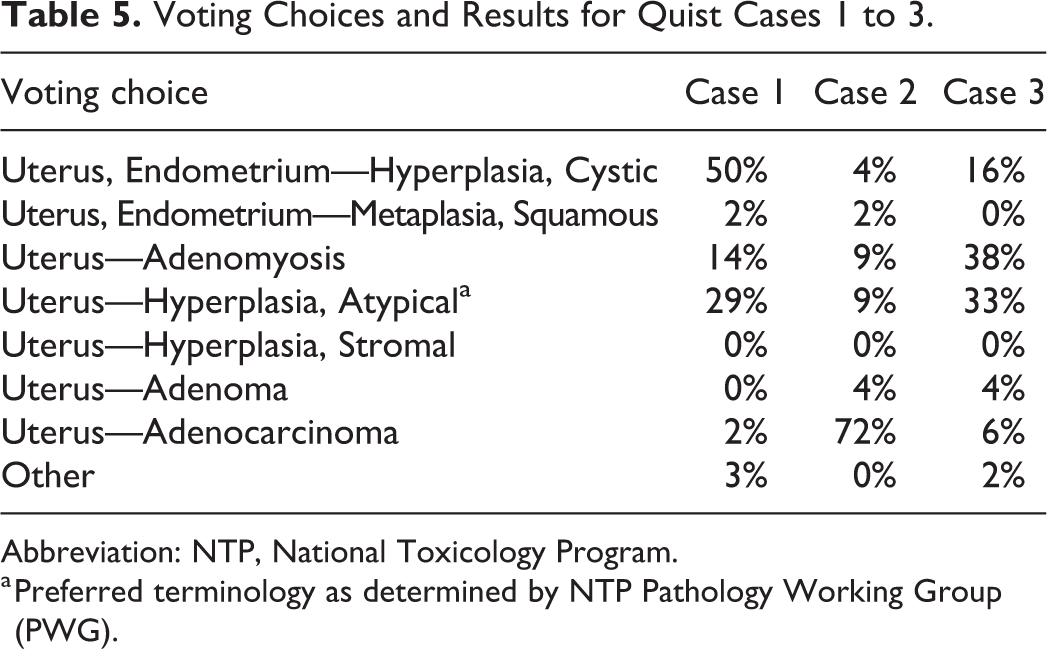
For the 3 cases, a series of photomicrographs were presented to the audience that included both low- and high-power magnifications of select longitudinal uterine sections from the 2-year study (Figure 7). Dr Quist informed the audience that the diagnostic choices would be the same for all 3 cases; the voting choices and results are presented in Table 5.
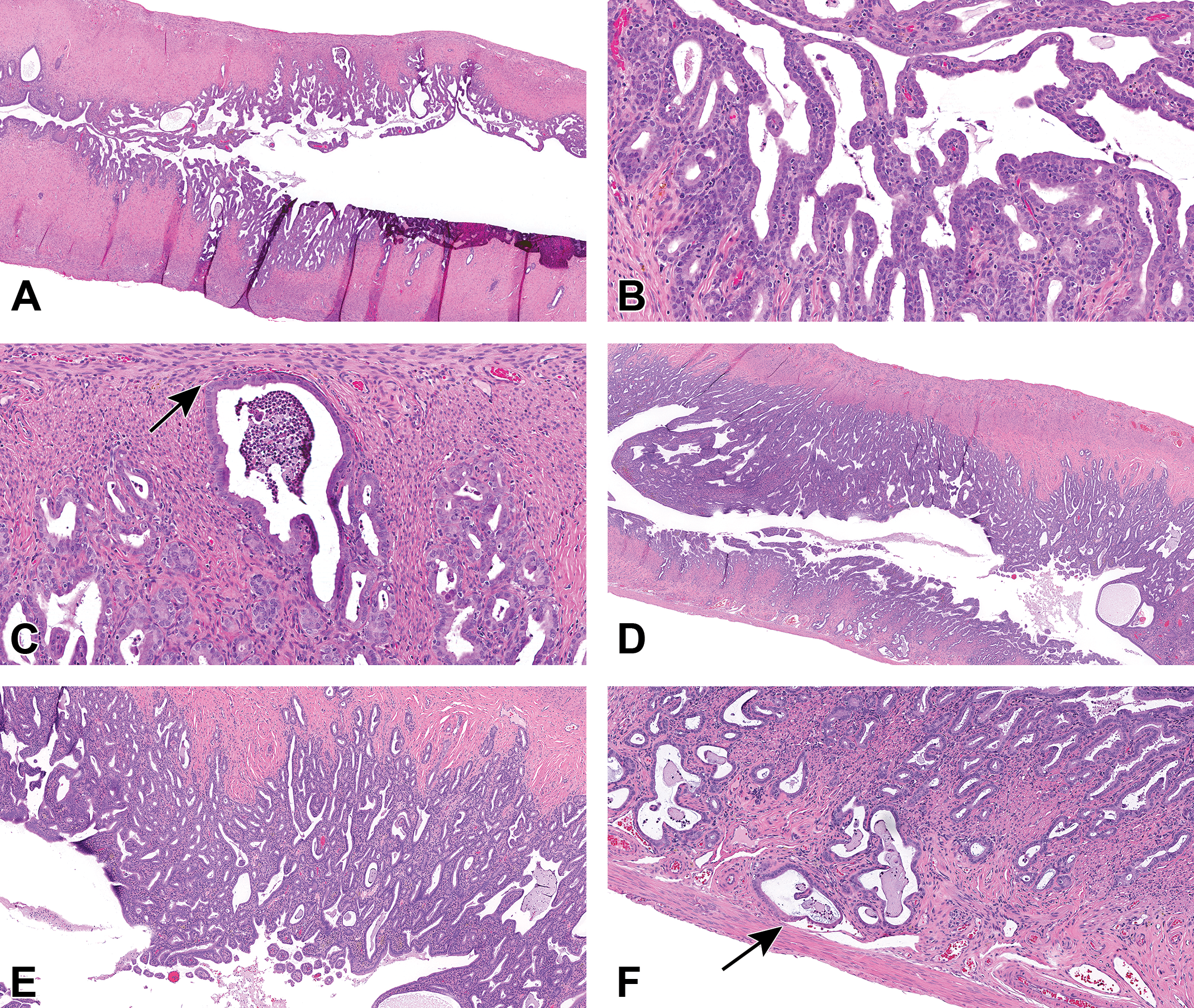
(A-F) Cases 1 and 3 are examples of atypical uterus hyperplasia from a 2-year study in Sprague Dawley rats. Case 1, atypical uterus hyperplasia, papillary type (A-C). A, The endometrium is diffusely hyperplastic with numerous small, branching papillary projections extending into the uterine lumen. Higher magnification (B) shows that papillary projections appear to arise from the surface epithelium and exhibit stratification and piling along the length of each papillary projection. In some areas (C), dilated endometrial glands appear to closely abut the endometrial–myometrial border (arrow). Case 3, Uterus–Hyperplasia, Atypical (D-F). D, The endometrium is markedly thickened and expanded by numerous hyperplastic and cystic endometrial glands. Higher magnification (E) shows that epithelial cells exhibit frequent stratification and piling. F, In some areas, several endometrial glands appear to extend into the first myometrial layer. The endometrial glands within the myometrium do not penetrate or breach the uterine wall (arrow; hematoxylin and eosin [H&E]).
Voting Choices and Results for Quist Cases 1 to 3.
Abbreviation: NTP, National Toxicology Program.
a Preferred terminology as determined by NTP Pathology Working Group (PWG).
Cases 1 and 3 (Figure 7A-F) provided examples of uterus—hyperplasia, atypical including the papillary type (Figure 7A and B). In both cases, proliferation and thickening of the endometrial epithelium was observed at low magnification (Figure 7A and D) with small, branching papillary structures projecting into the uterine lumen and arising from the surface epithelium in case 1 (Figure 7A and B). Higher magnification images depicted areas of epithelial stratification and piling within the endometrium (Figure 7B and E) with some examples of endometrial glands abutting the endometrium–myometrium border (Figure 7C and F). Case 2 was presented as an example of uterus—adenocarcinoma (Figure 7G-L).
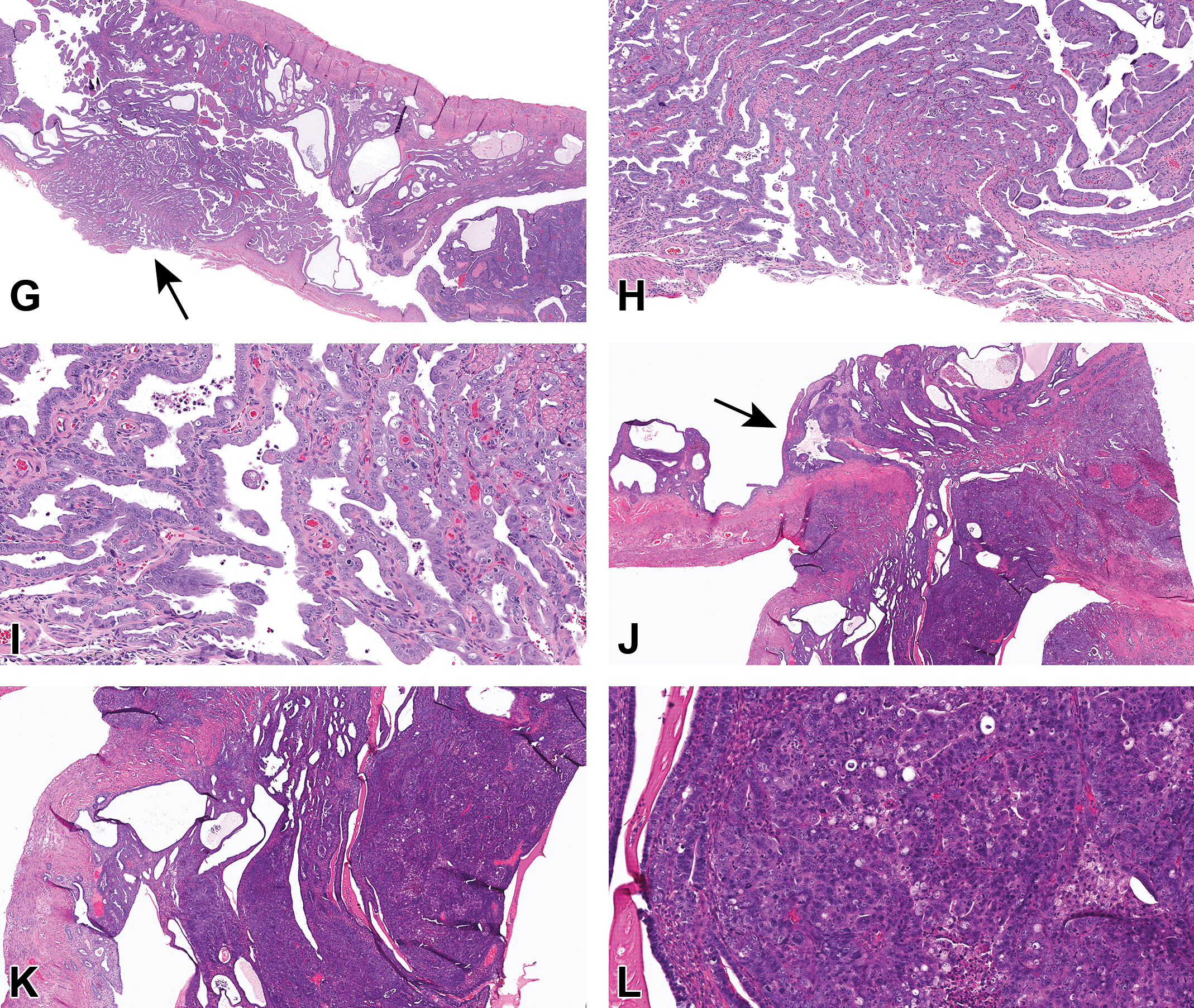
(Continued). G-L, Examples of uterus adenocarcinoma from 2-year study in Sprague Dawley rats. Case 2 (G-I) and additional example (J-L). G and J, Infiltrative, unencapsulated neoplasms diffusely disrupt the uterine architecture, invading the myometrium and breaching the uterine wall (arrows). Higher magnifications (H, I, K, L) show the neoplasms are comprised of epithelial cells exhibiting varying degrees of pleomorphism, forming solid nests and cords (H&E).
After voting was complete, Dr Quist explained that these cases had been selected for the NTP Satellite Symposium because they represented challenging diagnostic cases that had required additional review after the conclusion of the PWG for this study. Pathology working group participants noted that in many examples of uterus—hyperplasia, atypical, endometrial glands with varying degrees of cellular atypia were occasionally observed within the myometrium. After much discussion, PWG participants requested a post-PWG PPR to address the following questions: (1) Should the diagnosis of uterus—adenomyosis be added to capture this change, (2) Is this a feature of uterus—hyperplasia, atypical that should be described in the pathology narrative, or (3) Is this true invasion and thus warranting a diagnosis of uterus—adenocarcinoma?
Dr Quist then summarized the important diagnostic features of key differential diagnoses, as listed in the INHAND 49 criteria and NTP Nonneoplastic Lesion Atlas 67,68 (Table 6).
Key Differential Diagnoses of Uterus Adenomyosis, Atypical Hyperplasia, and Adenocarcinoma.

a Adenomatous hyperplasia; Hyperplasia, glandular, focal.
After reviewing the diagnostic criteria, Dr Quist revisited the challenging task of differentiating atypical hyperplasia from adenocarcinoma in longitudinal uterine sections (Figure 7M-P). Ultimately, for the cases presented during the post-PWG PPR, PPR participants determined that atypical endometrial glands could exhibit some extension into the first layer of myometrium as a feature of atypical hyperplasia (Figure 7M and N). However, atypical endometrial glands that were embedded within the second layer of the myometrium represented true invasion and were classified as uterine adenocarcinoma (Figure 7O and P).
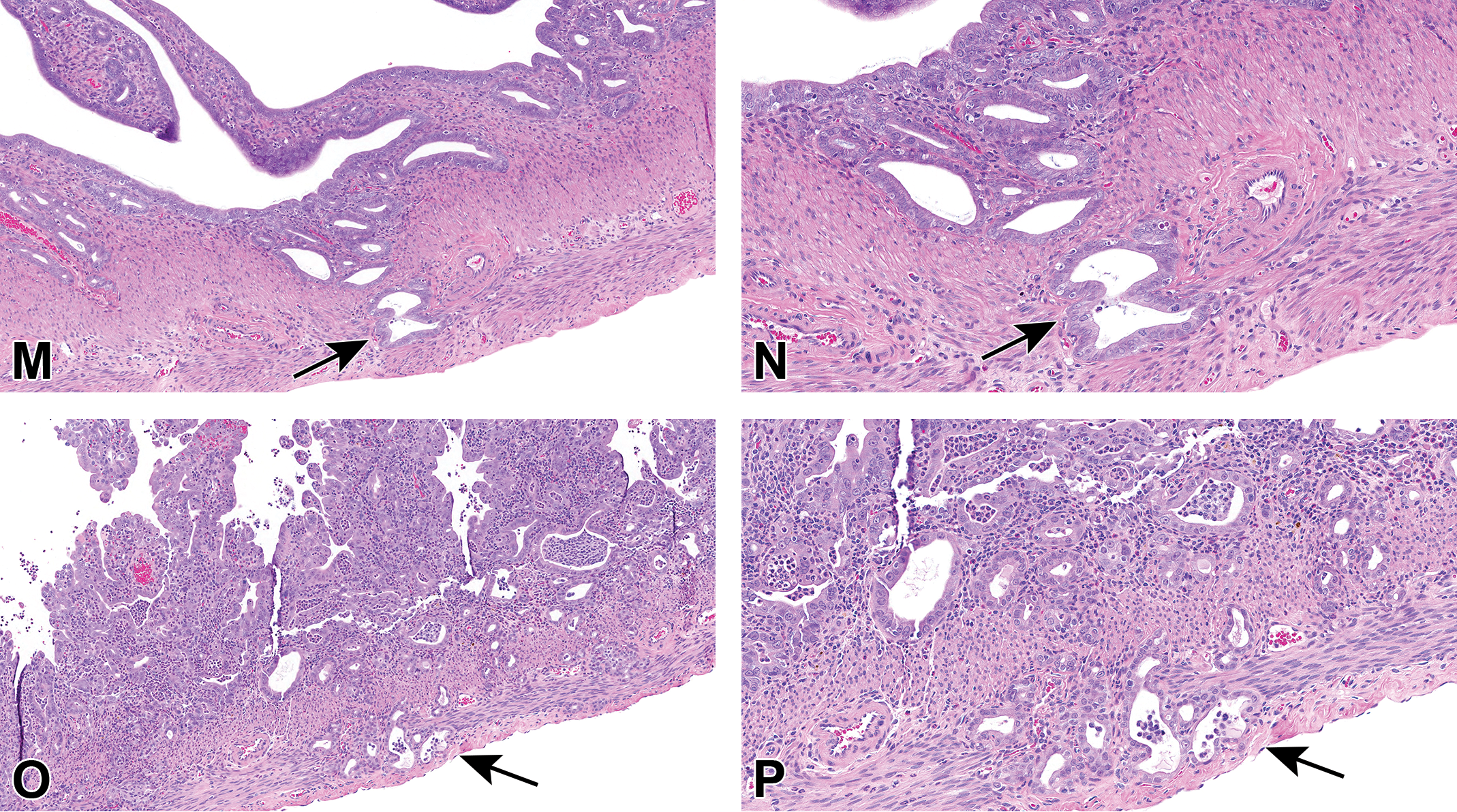
(Continued). M-P, Side-by-side comparison of atypical uterus hyperplasia (M and N) and uterus adenocarcinoma (O and P) from a 2-year study in Sprague Dawley rats. M and higher magnification N, Endometrial glands exhibiting some degree of cellular atypia, extending into the first myometrial layer (arrows) was considered by the Pathology Peer Review (PPR) to be a feature of atypical uterine hyperplasia. O and higher magnification P, Endometrial glands exhibiting features of cellular pleomorphism and/or atypia invading the second myometrial layer (arrows) were characterized as “invasive” and were consistent with the diagnostic criteria for uterine adenocarcinoma (H&E).
Dr Quist asked the audience if they thought that atypical hyperplasia could exhibit extension into the myometrium or if adenocarcinoma should always be called when any degree of invasion is present. Audience members agreed that invasion of the myometrium alone was not sufficient criteria for the diagnosis of uterine adenocarcinoma and that additional morphological features should be considered when differentiating between adenomyosis, atypical hyperplasia, and adenocarcinoma. An audience member mentioned that well-differentiated hyperplasia can penetrate the muscle, so such a feature would not be inappropriate for an atypical lesion. Another audience member added that a diagnosis of adenocarcinoma should not be made unless the epithelial cells have breached the basement membrane.
As a final take-home point, Dr Quist reminded the audience that it is important to include longitudinal sections of the uterus for all uterine evaluations. The cases presented here were from a study in which the majority of uterine lesions were only observed in the longitudinal uterine sections and were not present in the examined cross sections. One audience member asked if the NTP’s adoption of longitudinal uterine sectioning had affected the historical control data, pointing out the importance of sampling bias. Dr Quist deferred to Dr Susan Elmore for comment. Dr Elmore agreed that sampling bias was considered when the NTP adopted the updated uterine sectioning protocol. However, when comparisons are made for studies using the longitudinal sectioning method, only similarly collected control studies are used for comparison. Moreover, NTP databases are compiled and used on a rolling 5-year window.
Footnotes
Acknowledgments
The authors wish to thank Eli Ney (NIEHS) for her unique and creative artwork for the symposium, Beth Mahler (EPL) for her assistance with manuscript image preparation and Drs Beth Lubeck and Greg Travlos for critical manuscript review. Appreciation also goes to Maureen Bayley, Tierre Miller, Matthew Price, and others at Association Innovation and Management, Inc, for their valuable help with annual advertising and virtual meeting platform.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported [in part] by the NIH, National Institute of Environmental Health Sciences.
