Abstract
Delgocitinib ointment 0.5% is the world’s first topical Janus kinase inhibitor product and was approved for treatment of atopic dermatitis (AD) in Japan. Although topical corticosteroids (TCSs) have been the mainstay of pharmacotherapy in AD over the past decades, long-term use of TCSs causes skin atrophy and alteration of the epidermal tight junction (TJ) leading to epidermal barrier dysfunction. In this study, delgocitinib ointment 0.5% or representative TCSs of different potencies were applied dermally once daily to the ear pinna of normal ICR mice for 14 days, and ear pinna thickness, histopathology, and immunohistochemistry for epidermal TJ proteins claudin-1 and -4 were evaluated. All the TCSs caused decreases in ear pinna thickness with epidermal thinning, sebaceous gland atrophy, and atrophy/decreased number of the subcutaneous adipocytes and decreased immunohistochemical staining intensity for epidermal claudins. In contrast, delgocitinib ointment 0.5% did not cause any of those changes. In conclusion, once daily topical delgocitinib ointment 0.5% for 14 days did not cause skin atrophy or decreased immunohistochemical staining of epidermal claudins, which are common safety concerns associated with TCSs. These characteristics suggest that delgocitinib ointment 0.5% has an improved safety profile over currently available TCS therapies particular for the long-term AD treatment.
Keywords
Introduction
Delgocitinib is a Janus kinase (JAK) inhibitor. delgocitinib ointment 0.5% is a nonsteroidal and the world’s first approved topical JAK inhibitor product that improves atopic dermatitis (AD) through suppressing the overactivation of the immune response by inhibiting the function of JAKs, which play a key role in immune activation signaling in immune cells and keratinocytes. In the phase III comparative clinical study in patients with AD (aged ≥16) for delgocitinib ointment 0.5%, conducted in Japan, the primary end point of efficacy, the percentage change of the modified Eczema Area and Severity Index score from the baseline, has met superiority to the placebo. 1 Furthermore, delgocitinib ointment 0.5% demonstrated a favorable safety profile in long-term treatment. 1
Although topical corticosteroids (TCSs) have been the mainstay of pharmacotherapy in AD over the past decades, long-term use of TCSs causes specific adverse reactions including steroid flushes and skin atrophy. 2 –4 In addition, suppression of tight junction (TJ) protein expression may be one of the mechanisms to explain the TCS-induced skin barrier dysfunction including impairment of the permeability and antimicrobial barrier function of the skin. 5 The effects of TCSs on the skin barrier are unfavorable in the treatment of AD because epidermal barrier dysfunction in the skin is the initial step in the development of AD 6,7 and TJ proteins, claudin (Cldn)-1 and -4 play a crucial role to maintain the mammalian epidermal barrier. 8 Claudin-1 downregulation, which is seen in AD lesions and has been linked to AD pathogenesis, results in impairment of TJ and epidermal barrier function and leads to inflammation in the human epidermis. 9 Therefore, in the treatment of AD, drug products, which have a safety profile without concerns for skin atrophy and impairment of the epidermal barrier, are highly desired.
We investigated the effects of delgocitinib ointment 0.5% on normal mouse skin by using measurements of ear pinna thickness and histological and immunohistochemical approaches to compare effects on skin atrophogenicity and alteration in epidermal TJ with those of representative TCSs in clinical use in Japan.
Materials and Methods
Chemicals
Delgocitinib, 3-[(3S,4R)-3-methyl-7-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1,7-diazaspiro[3.4]octan-1-yl]-3-oxopropanenitrile, was synthesized and its clinically approved preparation (delgocitinib ointment 0.5%) and its respective placebo (composed of white petrolatum, paraffin, and squalane) were prepared in the Central Pharmaceutical Research Institute, Japan Tobacco Inc. Three clinically approved and commercially available TCS products of different potency according to the 5-level clinical efficacy ranking system in Japan (strongest, very strong, strong, mild, and weak) were chosen for the present study: prednisolone valerate acetate ointment 0.3% (PV) of medium potency, betamethasone valerate ointment 0.12% (BV) of strong potency, and difluprednate ointment 0.05% (DF) of very strong potency. These TCSs were purchased from Kowa Company, Ltd, Shionogi & Co., Ltd, and Mitsubishi Tanabe Pharma Corporation, respectively.
Animals
Six-week-old female Crl: CD1(ICR) mice were purchased from Charles River Laboratories Japan, Inc. The animals were housed individually in plastic cages with bedding (Paper-Clean, Japan SLC Inc) in an environmentally controlled room (temperature of 23 °C ± 3 °C, relative humidity of 55% ± 15%, and a 12-hour light/dark cycle) and were allowed free access to a commercial diet (CRF 1, Oriental Yeast Co) and tap water. This study was conducted in accordance with the Japanese Law for the Humane Treatment and Management of Animals (Law No. 105, as revised in 2013, issued on October 1, 1973).
Experimental Design
The animals (7 weeks old at initial dose, 10 females per group) received applications of delgocitinib ointment 0.5%, delgocitinib ointment placebo as a control drug, or PV, BV, or DF as reference TCSs used clinically in Japan, dermally at a dosing volume of 20 μL/animal once daily to the 2 sides (10 µL each on the external and internal surfaces) of the left ear pinna for 14 days. A nontreated group was also included. The dosing volume and dosing period were determined based on the results of a preliminary study, which demonstrated that 20 µL was sufficient to homogenously cover the whole application area of the pinna, and a 14-day dosing period was well tolerated with significant decrease in ear pinna thickness. Animals were allocated to each groups based on the ear pinna thickness values on the day before the initiation of the dosing so that the mean values of the ear pinna thickness were almost the same between each group.
Clinical Observations and Measurements of Body Weights
Clinical observations of the animals were conducted once daily during the 14-day dosing period. All the animals were observed for abnormalities in physical appearance, nutrition, body posture, behavior, and excrement. Body weights were measured on days 3, 7, and 14 of the dosing period.
Measurements of Ear Pinna Thickness
Thickness of the left ear pinna was measured using a Digimatic Micrometer (Mitsutoyo) under isoflurane anesthesia on days 3, 7, and 14 of the dosing period.
Collection of Ear Pinna Samples and Histopathology
On the day after the last dosing of the 14-day dosing period, the animals were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia. After euthanasia, the left ear pinna of each animal was collected, fixed in 10% neutral buffered formalin solution, embedded in paraffin, sectioned at 3 μm, and stained with hematoxylin and eosin for histopathological evaluation.
Immunohistochemistry for Epidermal TJ Proteins (Cldn-1 and -4)
Immunohistochemistry using rabbit polyclonal antibodies for Cldn-1 (1:3000 dilution, LS-B6327, LifeSpan BioSciences, Inc) and Cldn-4 (1:800 dilution, LS-B8382, LifeSpan BioSciences, Inc) was conducted on the 3 µm paraffin sections of the ear pinna. The sections were boiled for 15 minutes in 0.01 M sodium citrate buffer (pH6.0) for antigen retrieval and incubated with the primary antibodies overnight at 4 °C. Histofine Simple Stain MAX-PO (Nichirei) was employed for the detection of the primary antibodies.
Statistical Analysis
The mean values and standard errors (SE) in each group were calculated, and statistical analyses were conducted for the body weights and the ear pinna thickness. Shapiro-Wilk tests were used to confirm normality of ear thickness measurements for nontreated and delgocitinib ointment placebo groups. A Student t test for comparison of the parameters between the nontreated and delgocitinib ointment placebo groups was conducted in order to investigate the potential effects of treatment with placebo and treatment procedures. In the treated groups, a Dunnett test for comparison of the parameters between the delgocitinib ointment placebo and delgocitinib ointment 0.5%, PV, BV, and DF groups was conducted in order to investigate the effects of delgocitinib and TCSs. The levels of significance were set at 5% and 1% (2-tailed).
Results
Clinical Signs and Body Weights
During the dosing period, no clinical abnormalities were observed in any group. When comparing the delgocitinib ointment placebo and nontreated groups, body weights in the delgocitinib ointment placebo group were significantly lower than those in the nontreated group on days 14 and 15 (Table 1 and Figure 1; P < .05 or P < .01). Although there were no differences in body weights between the delgocitinib ointment placebo group and delgocitinib ointment 0.5%, PV, or BV group throughout the dosing period, body weights in the DF group were significantly lower than those in the delgocitinib ointment placebo group on days 14 and 15 (Table 1 and Figure 1; P < .05).
Body Weights.a,b

Abbreviations: BV, betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%; SE, standard error.
a Each value shows mean ± SE.
b P values and 95% CIs (the figure in the parentheses) reflect comparisons between delgocitinib ointment placebo and nontreated, and comparisons between delgocitinib ointment 0.5%, PV, BV, and DF with delgocitinib ointment placebo.
c Significantly different from the nontreated group (P < .05 by Student t test).
d Significantly different from the nontreated group (P < .01 by Student t test).
e Significantly different from the delgocitinib ointment placebo group (P < .05 by Dunnett test).
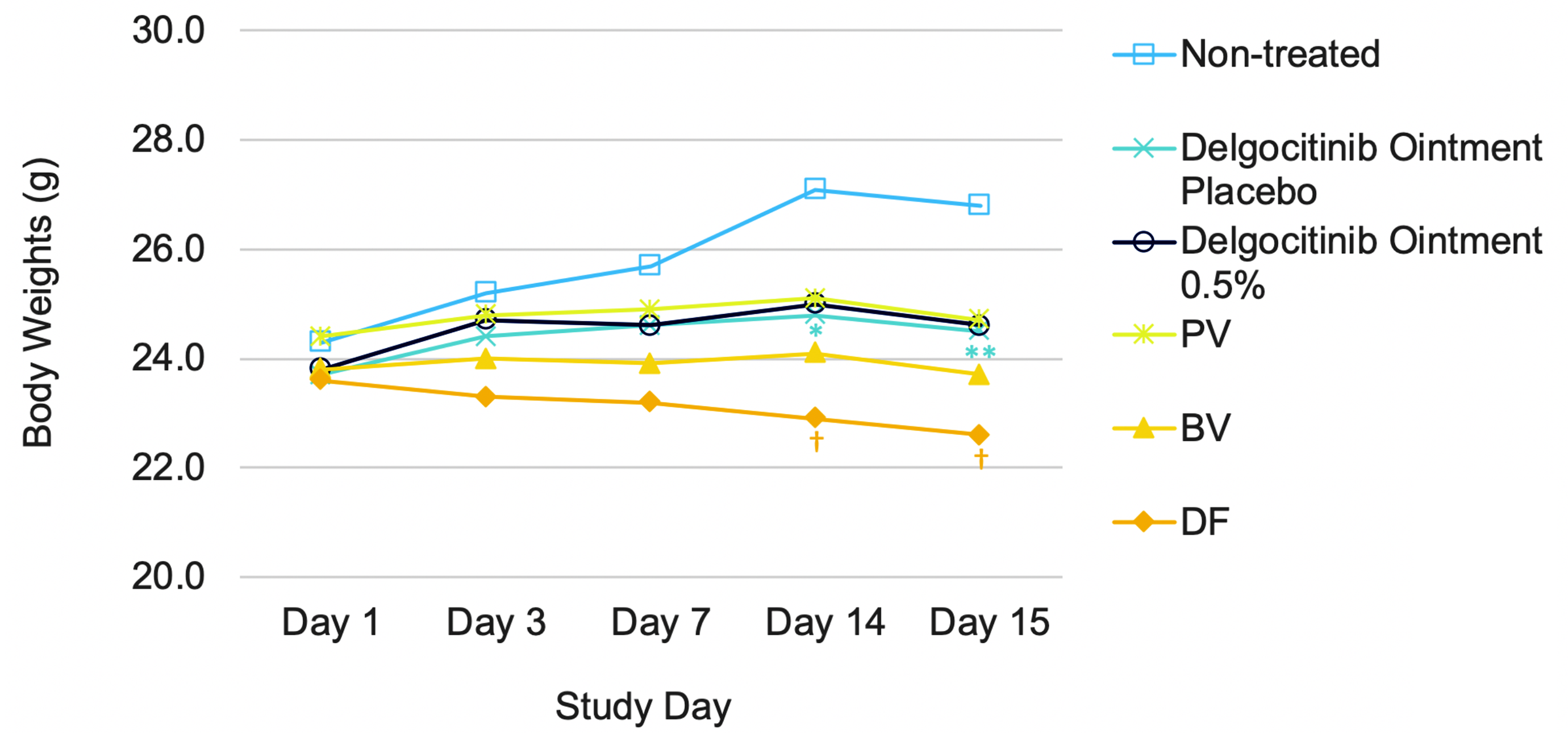
Body weights during the study period. Significantly different from the nontreated group (*P < .05, **P < .01 by the Student t test). Significantly different from the delgocitinib ointment placebo group († P < .05 by the Dunnett test). BV indicates betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%.
Ear Pinna Thickness
Ear pinna thickness values in the delgocitinib ointment placebo and delgocitinib ointment 0.5% groups were similar to those in the nontreated group throughout the dosing period (Table 2 and Figure 2). All the TCS groups showed significantly lower ear pinna thickness values than those in the nontreated, delgocitinib ointment placebo or delgocitinib ointment 0.5% groups on day 3 onward (Table 2 and Figure 2; P < .01), indicating that the effects of TCSs on the skin had occurred by day 3.
Ear Pinna Thickness.a,b
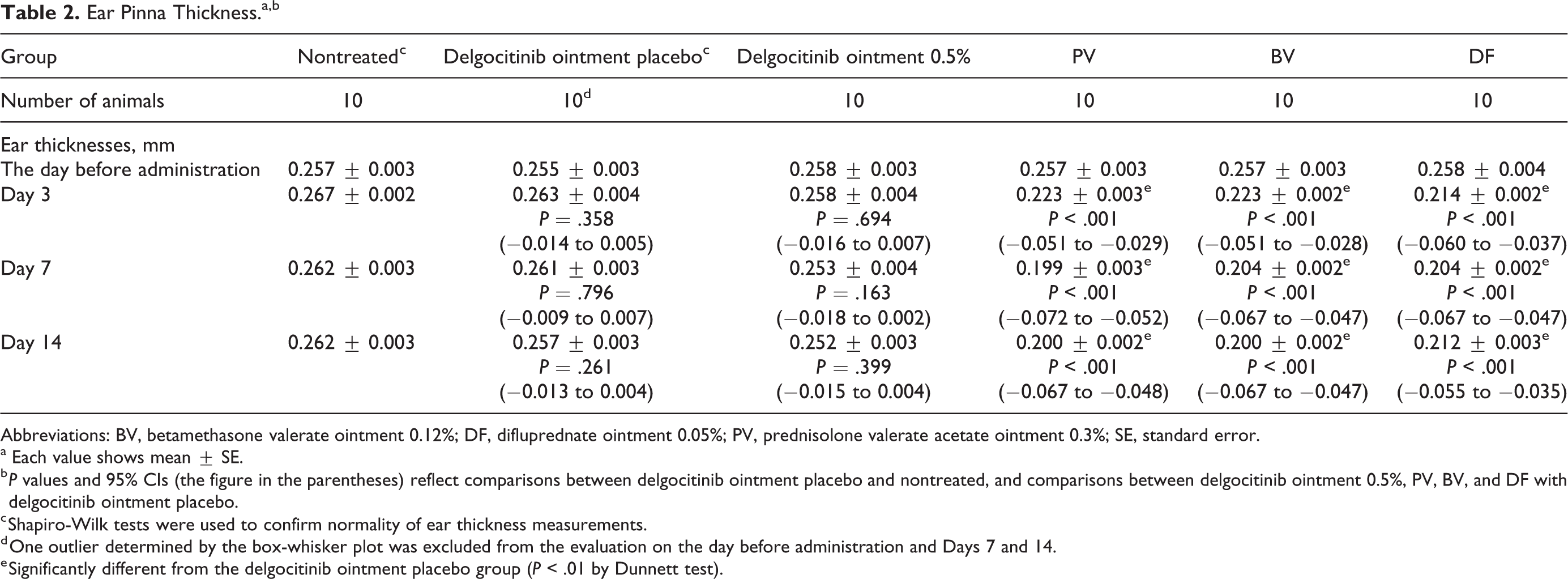
Abbreviations: BV, betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%; SE, standard error.
a Each value shows mean ± SE.
b P values and 95% CIs (the figure in the parentheses) reflect comparisons between delgocitinib ointment placebo and nontreated, and comparisons between delgocitinib ointment 0.5%, PV, BV, and DF with delgocitinib ointment placebo.
c Shapiro-Wilk tests were used to confirm normality of ear thickness measurements.
d One outlier determined by the box-whisker plot was excluded from the evaluation on the day before administration and Days 7 and 14.
e Significantly different from the delgocitinib ointment placebo group (P < .01 by Dunnett test).
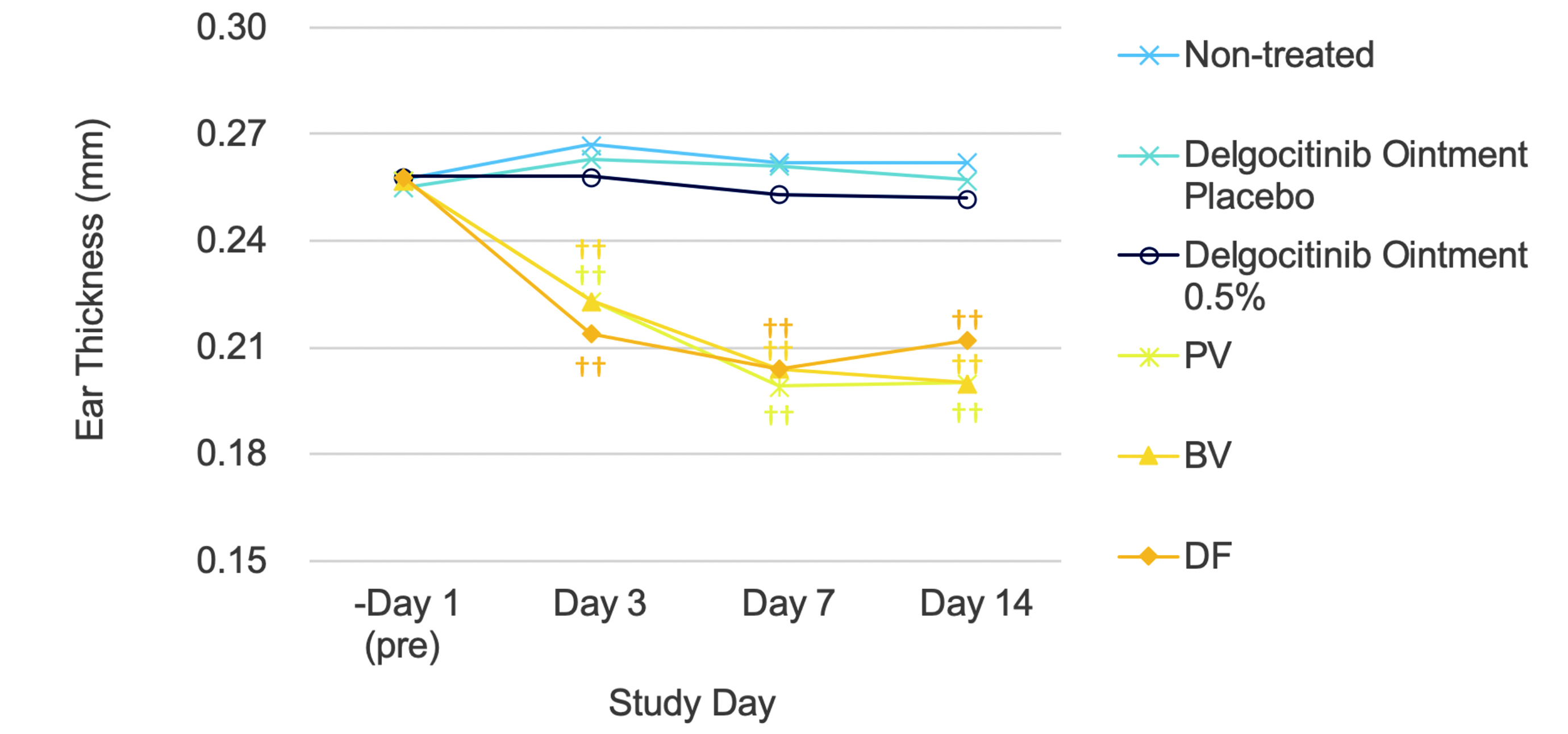
Ear pinna thickness during the study period. Significantly different from the delgocitinib ointment placebo group (†† P < .01 by the Dunnett test). BV indicates betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%.
Histopathology of the Ear Pinna
There were no histopathological changes in the ear skin in any animals in the delgocitinib ointment placebo, delgocitinib ointment 0.5%, or nontreated groups (Table 3 and Figure 3). On the other hand, epidermal thinning of the ear skin, atrophy of the sebaceous glands, and/or atrophy/decreased number of the subcutaneous adipocytes were observed in all the animals in all the TCS groups (Table 3 and Figure 3).
Histopathological Findings of the Ear Skin.
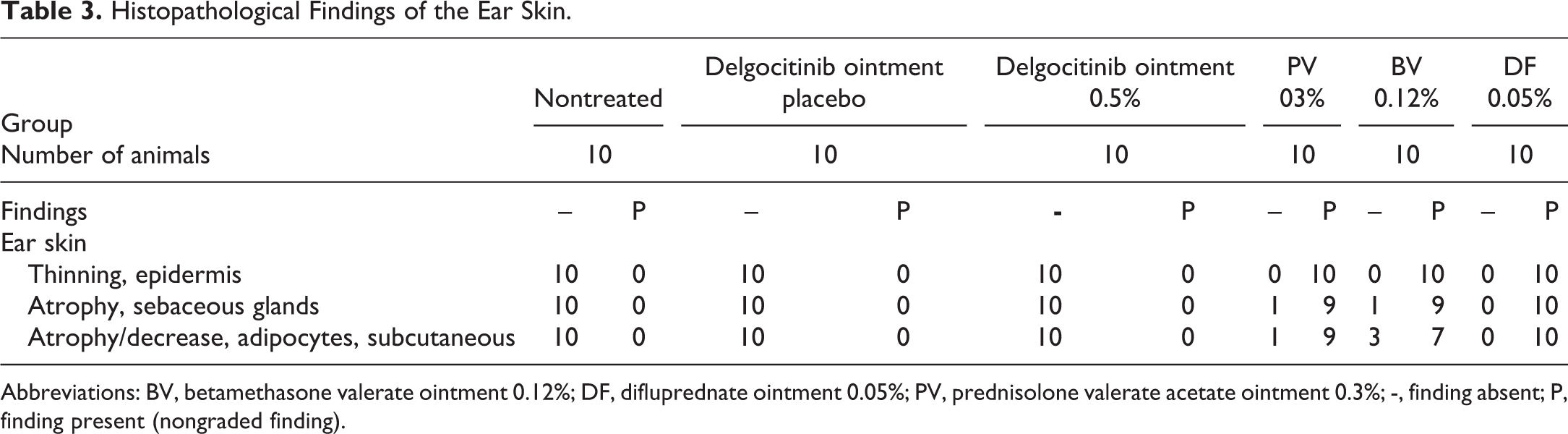
Abbreviations: BV, betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%; -, finding absent; P, finding present (nongraded finding).
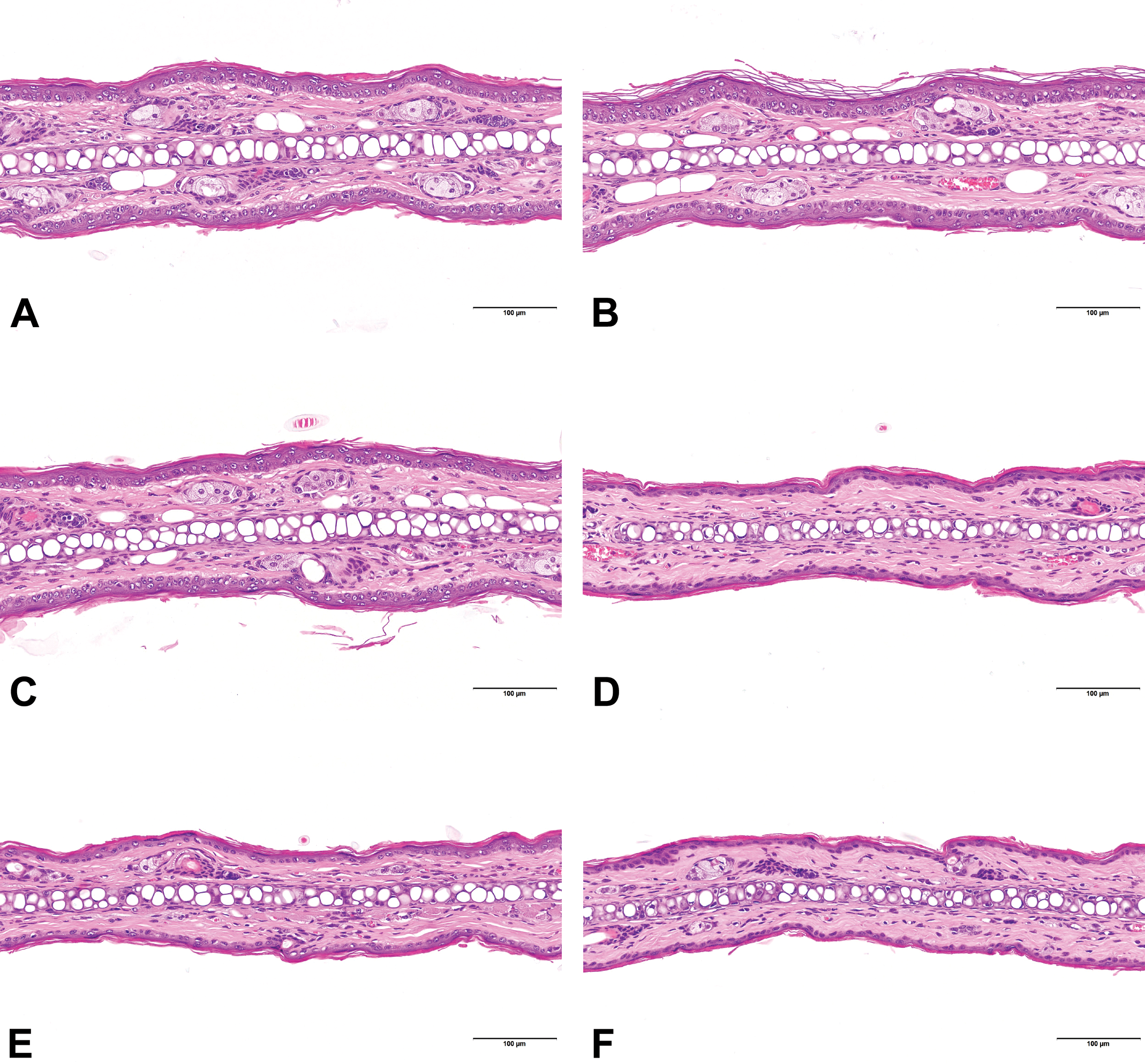
Histological findings of the ear skin, HE staining. Nontreated (A), treated with delgocitinib ointment placebo (B), delgocitinib ointment 0.5% (C), PV (D), BV (E) and DF (F). No histopathological changes were seen in the ear skin in any animal in the delgocitinib ointment placebo, delgocitinib ointment 0.5%, and nontreated groups (A-C). Thinning of the ear epidermis due to flattened keratinocytes with shrunken nuclei, atrophy of the sebaceous glands, and/or atrophy/decreased number of the subcutaneous adipocytes were observed in all animals in all TCS groups (D-F). BV indicates betamethasone valerate ointment 0.12%; DF, difluprednate ointment 0.05%; PV, prednisolone valerate acetate ointment 0.3%; TCS, topical corticosteroids.
Immunohistochemistry for Cldn-1 and -4
In the normal (nontreated) epidermis, Cldn-1 positivity was seen in the suprabasal layer (Figure 4A) and Cldn-4 positivity was restricted to the stratum granulosum (Figure 5B). There were no differences in the immunohistochemical staining intensities for Cldn-1 and -4 between the delgocitinib ointment placebo, delgocitinib ointment 0.5%, and nontreated groups (Figures 4 and 5). Staining intensities and number of cells that stained for both antigens were markedly decreased in all the TCS groups compared to those in the delgocitinib ointment placebo, delgocitinib ointment 0.5%, and nontreated groups (Figures 4 and 5).
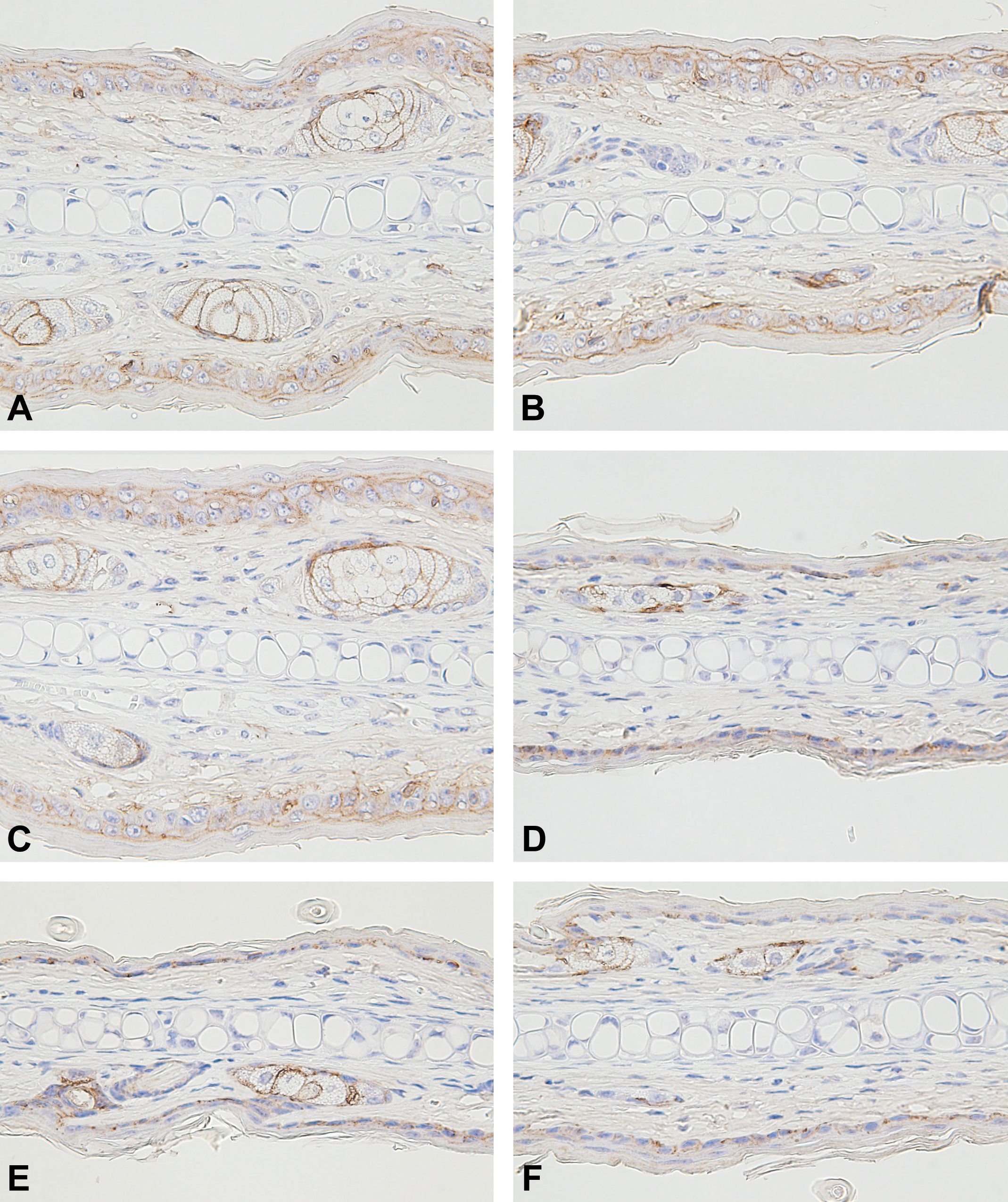
Immunohistochemistry for epidermal Cldn-1 of the ear pinna. Nontreated (A), treated with delgocitinib ointment placebo (B), delgocitinib ointment 0.5% (C), PVA (D), BV (E), and DF (F). Cldn-1 positivity was seen in the suprabasal layer in the epidermis (A). No differences were seen in the results between the delgocitinib ointment placebo, delgocitinib ointment 0.5%, and nontreated groups. Staining intensities and number of cells that stained for Cldn-1 were decreased in all TCS groups compared to those in the delgocitinib ointment and nontreated groups. BV indicates betamethasone valerate ointment 0.12%; Cldn, claudin; DF, difluprednate ointment 0.05%; PVA, prednisolone valerate acetate ointment 0.3%; TCS, topical corticosteroids.
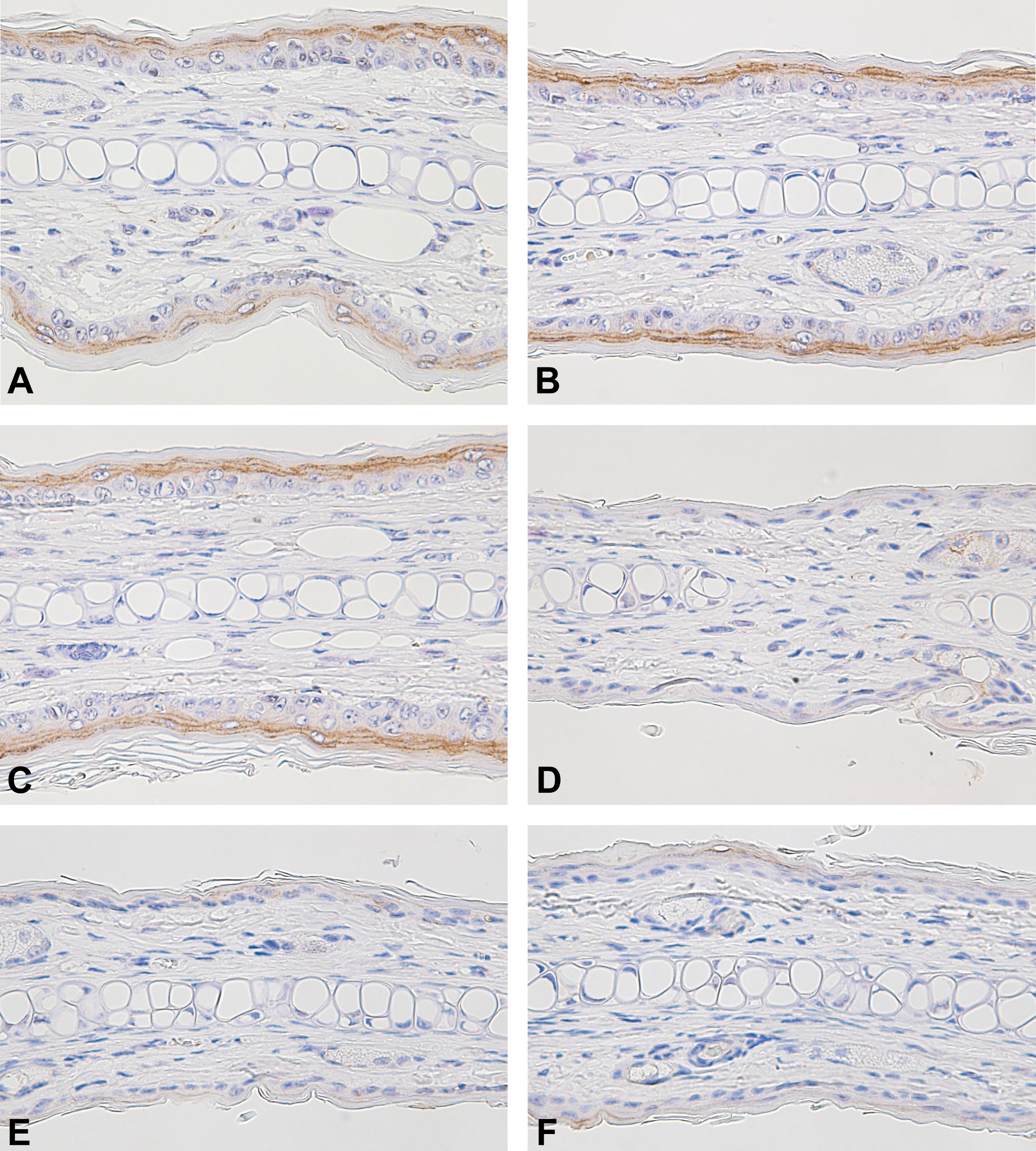
Immunohistochemistry for epidermal Cldn-4 of the ear pinna. Nontreated (A), treated with delgocitinib ointment placebo (B), delgocitinib ointment 0.5% (C), PVA (D), BV (E), and DF (F). Cldn-4 positivity was restricted to the stratum granulosum in the epidermis (A). No differences were seen in the results between the delgocitinib ointment placebo, delgocitinib ointment 0.5%, and nontreated groups. Staining intensities and number of cells that stained for Cldn-4 were decreased in all TCS groups compared to those in the delgocitinib ointment and nontreated groups. BV indicates betamethasone valerate ointment 0.12%; Cldn, claudin; DF, difluprednate ointment 0.05%; PVA, prednisolone valerate acetate ointment 0.3%; TCS, topical corticosteroids.
Discussion
In the present study, delgocitinib ointment 0.5% and its placebo did not alter ear pinna thickness or cause histopathological changes in the ear pinna even after 14 days dosing. On the other hand, all the TCSs of different potencies caused statistically significant decreases in ear pinna thickness in a short dosing period (within 3-day dosing period) and the decrease in ear pinna thickness was considered to be due to the atrophic changes in the ear skin detected on histopathological examination (ie, atrophy of the sebaceous glands, epidermis, and subcutaneous adipocytes). Only female mice were used in the present study since they are easier to be handled compared to male mice and are commonly selected for this type of studies. Although female mice are reportedly more sensitive to skin atrophy induced by topical glucocorticoids, 10 TCSs induce skin atrophy regardless of gender. 11,12
Although TCSs have been the mainstay of pharmacotherapy in AD over the past decades, long-term use of TCSs can cause specific adverse reactions such as steroid flushes and skin atrophy. 2 –4 Although not life-threatening, these adverse reactions significantly affect the quality of life of the affected individuals. 13 Corticosteroids cause skin atrophy through suppression of cell proliferation, inhibition of collagen synthesis, and promotion of lipid metabolism. 3,12 Atrophic changes in the mouse skin noted in the TCSs-treated animals in the present study are also consistent with the changes observed in patients treated with TCSs. 2,14 Delgocitinib did not induce skin atrophy in any nonclinical toxicology studies with rodents or nonrodents either via the dermal route or oral route including chronic toxicology studies in rats up to 6 months (oral), in dogs up to 9 months (oral), and in minipigs up to 9 months (dermal) and 2-year carcinogenicity studies in rats (oral) and in mice (dermal). 15 Delgocitinib ointment 0.5% did not cause skin atrophy in a 52-week clinical study in adult patients with AD for the evaluation of the long-term safety and efficacy. 1 All of these observations taken together suggest that the risk of delgocitinib inducing skin atrophy in patients would be very low, contrary to TCSs.
In the present study, all the TCSs of different potencies caused decreased immunostaining intensities for the epidermal TJ proteins Cldn-1 and -4 in the ear pinna, while delgocitinib ointment 0.5% did not alter the expression of these TJ proteins. Cldn-1 and -4 play a crucial role in the maintenance of the mammalian epidermal barrier. 8 Claudin-1 downregulation, which is seen in AD lesions and has been linked to AD pathogenesis, results in the impairment of TJ and epidermal barrier function and leads to inflammation in the human epidermis. 9 Topical corticosteroids widely used in AD therapy, however, have been reported to suppress expressions of Cldn-1 and -4 and alter the structures of epidermal TJ components leading to impairment of the epidermal permeability barrier function. 5
In the present study, atrophy of sebaceous glands was also noted in the TCSs-treated animals. Skin surface lipids secreted from the sebaceous gland are also important for the skin barrier hydrating the skin, 16 and sebaceous gland atrophy could be an intervention target for skin barrier impairment. 17 This indicates that atrophy of the sebaceous glands is also involved in the corticosteroid-induced skin barrier impairment.
Epidermal barrier dysfunction in the skin is the initial step in the development of AD, 6 and therefore, in the treatment of AD, effects of TCSs on the skin barrier function are unfavorable and therapies which will not impair the epidermal barrier are highly desired. Even in terms of maintenance of skin barrier function, delgocitinib ointment 0.5% is considered to be a promising drug product in the treatment of AD.
In the present study, body weights were decreased during the dosing period only in the DF group. It has been reported that body weight loss in rodents treated with corticosteroids is caused by their systemic catabolic effects. 18 –20 Although systemic exposure to the TCSs was not measured in the present study, the body weight loss in the DF group was considered possibly related to a systemic effect of DF of very strong potency compared to PV (medium potency) and BV (strong potency). Difference in clinical efficacy/potency may explain why the body weight loss was seen only in the DF group.
In conclusion, once daily topical delgocitinib ointment 0.5% for 14 days did not cause the skin atrophy or decreased immunohistochemical staining of epidermal claudins both of which are critical safety concerns in terms of the quality of life or skin barrier maintenance in patients having AD with prolonged corticosteroid treatment. These characteristics suggest that delgocitinib ointment 0.5% has an improved safety profile over currently available TCS therapies particular for the long-term treatment of AD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Akiko Anagawa-Nakamura, Katsunori Ryoke, Yuzo Yasui, Toshiyuki Shoda, and Shoichiro Sugai are employees of Japan Tobacco Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Japan Tobacco Inc.
