Abstract
Mucosa-associated lymphoid tissue (MALT) of special senses is poorly described and can be confused with nonspecific mononuclear cell infiltrates and tertiary lymphoid structures (TLS). In the eye, MALT consists mostly of conjunctiva-associated lymphoid tissue (CALT) and lacrimal drainage-associated lymphoid tissue (LDALT). In humans, CALT and LDALT are important components of the normal eye-associated lymphoid tissue (EALT), but EALT is less frequently described in ocular tissues of animals. The EALT are acquired postnatally in preferential mucosal sites, expand with antigenic exposure, form well-developed lymphoid follicles, and are reported to senesce. Lymphoid follicles that are induced concurrently with chronic inflammation are more appropriately considered TLS but must be differentiated from inflammation in MALT. Less understood is the etiology for formation of lymphoid tissue aggregates in the ciliary body, limbus, or choroid of healthy eyes in animals and humans. In the healthy eustachian tube and middle ear of animals and humans, MALT may be present but is infrequently described. Concurrent with otitis media, lymphoid follicles in the eustachian tube are probably expanded MALT, but lymphoid follicles in the middle ear may be TLS. The purpose of this comparative review is to familiarize toxicologic pathologists with MALT in the special senses and to provide considerations for differentiating and reporting eye and ear MALT from immune or inflammatory cell infiltrates or inflammation in nonclinical studies, and the circumstances for reporting TLS in compartments of the eye and ear.
Keywords
Introduction
Toxicologic pathologists frequently encounter microscopic clusters of mononuclear cells or mixed inflammatory cells in multiple tissues outside primary and secondary lymphoid tissues. The primary challenge is to determine whether these aggregates are inflammatory or noninflammatory and then to determine the significance of the presence of these cell aggregates in the tissue. Immune cells in association with congestion, hyperemia, hemorrhage, edema, necrosis, fibrosis, and foreign material or microorganisms would be considered inflammation. In the absence of indicators of inflammation, the term often applied to immune cell clusters is “infiltrates,” usually modified by the dominant cell type (eg, infiltrate, mononuclear). Other diagnostic terms reported for immune cell aggregates in tissues include nonproliferative and proliferative changes such as increased cellularity, hyperplasia, extramedullary hematopoiesis, neoplasia, mucosa-associated lymphoid tissue (MALT), ectopic lymphoid tissue, and tertiary lymphoid structures (TLS). Increased cellularity would include an increase in normal cells with normal maturation in normal tissues with normal architecture (usually in lymphoid organs), hyperplasia as an expansion of normal maturing cells in hematolymphoid organs, extramedullary hematopoiesis consists of mature and immature hematopoietic cells responding to a systemic condition, and hematopoietic neoplasia is typified by a homogeneous population of lymphoid, myeloid, or erythroid cells that efface tissue architecture. 1,2 In mucosal tissues, loose clusters of lymphocytes and discrete lymphoid follicles may also be normal acquired MALT. Another consideration is TLS, also reported less preferentially as ectopic lymphoid tissue, which consists of lymphocyte-specific microdomains in nonlymphoid tissues under chronic inflammatory conditions. 2 These terms suggest that the hematolymphoid cells are a local response to an antigenic stimulus or pathophysiological state.
Mononuclear or mixed cell infiltrates within normal tissues are widely recorded as a morphological diagnosis in toxicologic pathology reports (see all INHAND guides; https://www.toxpath.org/inhand.asp#pubg). The etiology of these infiltrates is implied to be a focus of active inflammation if composed of mixed cell populations and a chronic or recovering focus of local inflammation when mononuclear cells, especially lymphocytes and plasma cells predominate. However, the lack of damage or changes to adjacent tissues would suggest otherwise. In particular, immune cell aggregates and lymphoid follicles that occur routinely in specific or predetermined anatomic locations are associated with mucosa and epithelial-lined drainage ducts, are more likely acquired MALT, and are part of the normal immune surveillance system of these tissues. 2 Aggregates of lymphocytes in specific stromal locations, near but not immediately adjacent to epithelial surfaces, may not only be part of the tissue immune surveillance system but could also be TLS.
A few mucosal sites contain MALT at birth (Peyer patches, lung, and nasopharynx), but many more mucosal tissues will acquire MALT with postnatal antigen exposure. Even in familiar sites such as the small intestine and lung, MALT can be difficult to study as these lymphoid aggregates may be represented histologically by small aggregates of immune cells in lamina propria or as a single lymphoid follicle that is usually not visible macroscopically. Identification is particularly difficult for sites of acquired MALT in the head and around the special senses, as these MALT locations are not routinely collected in toxicology studies, and purposeful collection requires knowledge of the potential anatomic location for the MALT and often the use of decalcified bone sections. It is important for pathologists to histologically recognize normal acquired MALT structures versus the implied abnormal nonspecific infiltrates or potentially adverse inflammation of a mucosal site. Normal MALT might only be recorded as a comment (“MALT present”), whereas infiltrates and inflammation would be recorded and categorized as an abnormal and potentially adverse finding. Sometimes, the distinction between MALT and nonspecific infiltrates may be difficult, but an understanding of the expected localization and histological characteristics of MALT is required to make the proper categorization. Normal MALT may also be subject to nonproliferative and proliferative changes, which should be recorded in nonclinical toxicology study as changes occurring within lymphoid tissue rather than within mucosal tissues. 3 The purpose of this introductory review is to familiarize toxicologic pathologists with the preferred sites of acquired MALT in and around the eye and ear of laboratory animals and to discuss these findings in the context of standardized terminology for reporting spontaneous and therapeutic-induced pathology of the special senses.
Morphology and Immunobiology of MALT
Microscopically, MALT typically consists of a single or multiple discrete but unencapsulated B-cell-dominant follicles with a subepithelial dome and parafollicular T cells. By immunohistochemistry, these mucosal lymphoid tissues contain typical lymphoid cells including CD3+, CD4+, and CD8+; and major histocompatibility complex class II+ cells; and antigen-presenting (APC) macrophages and dendritic cells (DCs). B cells can expand and show immunoglobulin A (IgA) class switching and release secretory IgA (SIgA), and T cells are activated in the face of antigen exposure. 4,5 B-lymphocyte activation by antigens or vaccination in one site of MALT can extend systemically, resulting in SIgA in distant MALT, but MALT can also act independent of the systemic immune response. 6 Immunocompetent cells in MALT may also be lost during local and systemic therapeutic immunomodulation and chemical exposure. 3,7 The MALT follicles may become secondary follicles with well-developed germinal centers that elevate the epithelium. The specialized follicle-associated epithelium (FAE) overlying the follicles is a flattened epithelium that contains microfold cells with weak intercellular junctions that allow intraepithelial leukocytes, mostly T cells, to cross the mucosa to sample antigens. Translocation of these CD8+ T cells bearing γδ T-cell receptors is critical to SIgA responses. The FAE has few to no goblet cells and no microvilli. In addition to these features, MALT lacks afferent lymphatics, but specialized high endothelial venules occur deep within or in immediate parafollicular locations. 4,8 –10 Recent reviews should be consulted for additional details on morphology, immunophenotyping, and functional activity of MALT. 5,6,11
The above is a description of typical MALT, but in routine histologic sections, lymphocytes may obscure the FAE; lamina propria may multifocally separate the follicle and the FAE; follicles may be small, lack germinal centers, and have little to no overlying FAE; or subepithelial lymphocytes may form a diffuse layer rather than a focal aggregate or follicle. All of these profiles are possible depending on the state of acquisition or activation of the MALT and whether the tissue section captures MALT at the edge of the follicle or in the center. Serial sections may be required to clearly demonstrate typical follicles and other characteristic features of MALT. 12 Small clusters or scattered submucosal lymphocytes are also part of the effector mucosal immune system in tissues, and it is not clear how frequently these loose aggregates become organized follicles or MALT, or whether they always remain as isolated collections of lymphocytes. Referring to these cells as part of MALT rather than submucosal infiltrates or inflammation is more consistent with the presumed general immunosurveillance activity of these cells.
Immunosurveillance of the Eye and Ear
The immunology of the special senses continues to be explored, particularly related to cellular and secretory antibody activity and therapeutic interventional pathways for inflammatory and autoimmune diseases of the eye and ear. That the eye is an immune-privileged site is often misunderstood to mean that this tissue lacks an active immunologic surveillance system. 13 –16 The ocular surface is known to have an active immune system that includes MALT and glandular and immunoregulatory secretions that form the tear film. The CALT and LDALT, as well as the lacrimal gland, are considered part of the eye-associated lymphoid tissue (EALT). In human medicine, the concept of EALT is widely accepted, and the location and incidence of the predetermined postnatally acquired subepithelial lymphoid tissues has been studied in normal and some disease conditions. 17 Less effort has been directed toward defining EALT in laboratory animals. 3,4 Consequently, MALT may go unrecognized or be inappropriately reported as nonproliferative or proliferative findings, particularly as nonspecific mononuclear infiltrates in pathology reports.
Less is known about the immunology or immune surveillance system of the ear, 18 –22 and the majority of the information is related to otitis media (OM). 20,23 The external ear immune responses reflect those found in other cutaneous sites. Immune responses occur in the middle and inner ear, but there appear to be no immune-privileged sites in the ear. 19,20,24 The eustachian tube is the connection between the nasopharynx and the middle ear, the terminus of which is anatomically similar to the nasolacrimal duct (NLD) ostium (NLDO) or opening in the nasal cavity. There is a variety of small and large MALT tissue in the naso-oro-pharyngeal area that makes up the tonsillar or pharyngeal ring (PR). 25 As the eustachian tube and middle ear are connected to this area, it should be expected that this region may develop MALT. Ear-associated lymphoid tissue has been described in the middle ear and eustachian tube of humans, but rarely in laboratory animals. 20 The ear is generally only sampled in toxicology studies directed toward ear-specific therapeutics or when evaluating therapeutics in a class with known ototoxicity. Therefore, most toxicologic pathologists will not be familiar with the normal anatomy of this organ under either normal or abnormal conditions or the expected locations of MALT.
Functional Integration of the Lymphoid Tissues of the Head
As a site of antigen uptake from the nasal cavity, oral cavity, and ocular surface, the head is endowed with multiple secondary lymphoid tissues (Figure 1). Immune responses in the nasal cavity involve the NALT in rabbits and smaller mammals. 3,4 Similarly, the nasopharyngeal tonsil (NPT) or less prominent nasopharyngeal lymphoid tissue in large animals (adenoid in humans) respond to respiratory tract antigens. 26 These nasal-associated lymphoid structures are immune-inductive sites present at birth. 9,27 Inhaled antigens land primarily by impaction and gravitational sedimentation on these nasal-associated lymphoid structures, 28 and subsequent regional inflammation may lead to lymphoid proliferation with obstruction at the NLDO. This may lead to retrograde dacryocystitis, conjunctivitis, and dacryoadenitis; stasis of tears; and induction and proliferation of lymphoid follicles throughout the lacrimal drainage system. 29 Nasal-associated lymphoid tissue and the ocular immune system are interconnected by the lacrimal system. 6,9,17,30,31 An abundance of lymphatics in the NALT and around the NLDO in the nasal cavity of mice appears to connect the immune responses of the nose and eye and the regional draining lymph nodes. 11,31 Surgical occlusion at the lacrimal (conjunctival) punctum in the rabbit does not alter the conjunctival response but enhances the systemic response after local ocular immunization. After intranasal immunization with lacrimal system occlusion, the ocular, but not the systemic response, was decreased. 32 Nasal cavity infections may also inflame and obstruct the eustachian tube, contributing to retrograde middle and inner ear inflammation, 33,34 as antigens usually do not gain access from the outer ear unless the tympanic membrane is breached. Although there are only a few tracing studies, the parotid lymph node appears to receive lymphatic drainage from the conjunctiva and periocular tissues, and the middle and inner ear, with subsequent drainage to the retropharyngeal and mandibular lymph nodes. 35 –37 The oral cavity is less well endowed with lymphoid tissue than the nasal cavity, and there are marked species differences 25 with no oral lymphoid tissue in rodents but various combinations of palatine tonsils and PR tonsils in larger mammals (see Figure 1). Inflammation of the PR tubal tonsils near the pharyngeal opening of the eustachian tube may contribute to eustachian tube dysfunction and OM in children. 38 Submucosal lymphocyte increases in the middle ear of mice can be induced after intranasal immunization, indicating a possible relationship of immune responses in NALT and the ear. 27
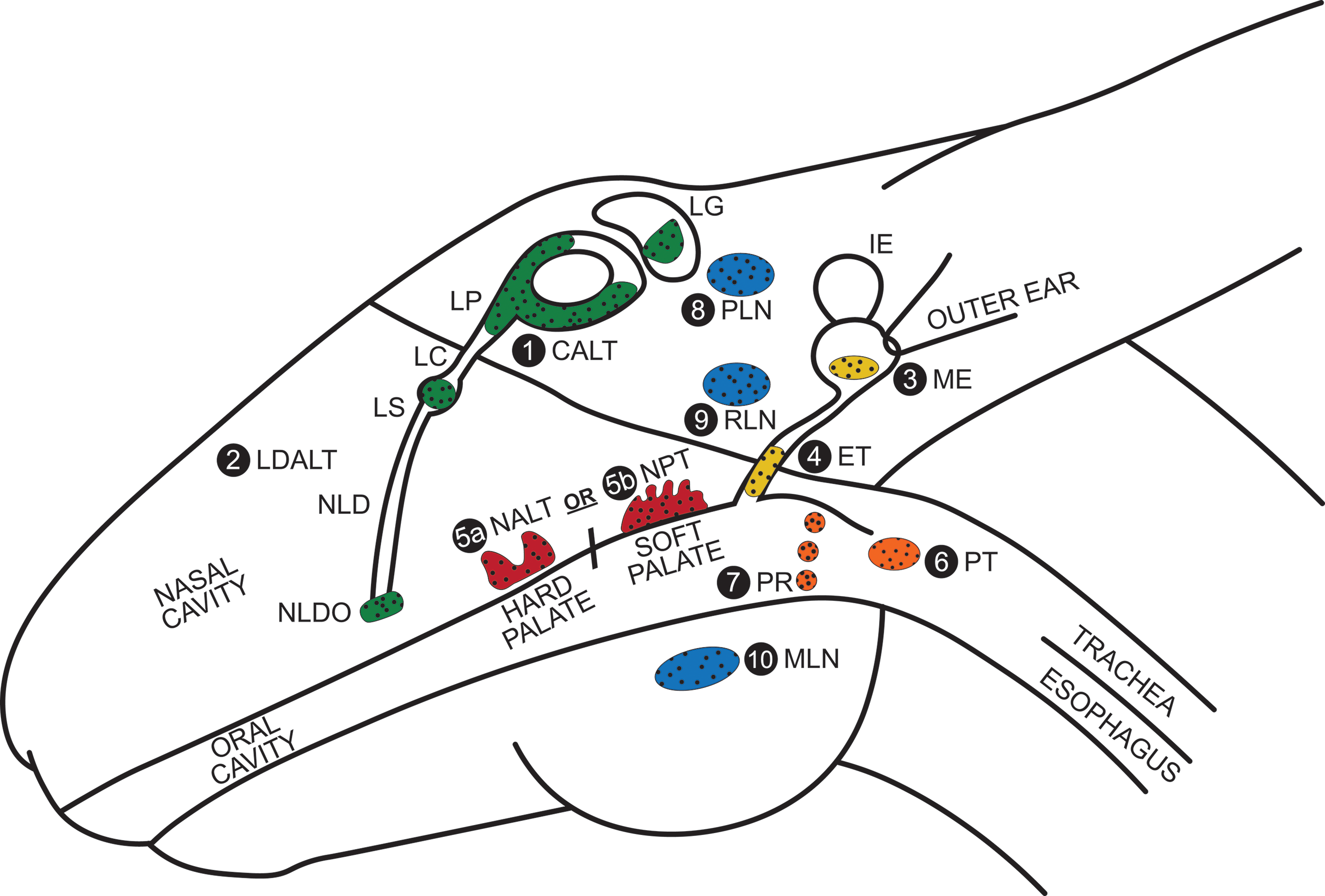
Using the rabbit head as an example, the approximate location and physical relationship of secondary lymphoid tissues in the head including the nasal (red) and oropharyngeal (orange) cavities and lymph nodes (blue), to lymphoid tissues of the eye (green) and ear (yellow) are shown. Not all of these lymphoid structures are present or may not yet have been described in all species. See the text for functional interconnections. The lymphoid tissues of the eye include the conjunctiva-associated lymphoid tissue (CALT; 1) and the components of the lacrimal drainage-associated lymphoid tissue (LDALT; 2), which includes the lacrimal gland (LG), lacrimal punctum (LP), lacrimal canaliculus (LC) between the LP and lacrimal sac (LS), and nasolacrimal duct (NLD) that drains into the nasal cavity at the NLD ostium (NLDO). In the ear, lymphoid tissue may be present in the middle ear (ME; 3) and eustachian tube (ET; 4). In the nasal cavity, nasal-associated lymphoid tissue (NALT; 5a) is present in rabbits and smaller species, but the nasopharyngeal tonsil/lymphoid tissue (NPT; 5b) is present in larger mammals (adenoid in humans). Rodents lack oral lymphoid tissue. The rabbit, dog, pig, nonhuman primates, and humans have a palatine tonsil (PT; 6) and pharyngeal ring (PR; 7). Lymphoid tissue is absent in the rabbit, consists of a lingual tonsil in the dog and 5 small tonsils in the pig, including tubal tonsils near the ET. Nonhuman primates and humans have a PT and up to 6 PR tonsils. The eye and ear drain into the parotid lymph nodes (PLN; 8) and then into the retropharyngeal lymph node (RLN; 9) and mandibular lymph node (MLN; 10).
Ocular MALT
Conjunctiva-Associated Lymphoid Tissue
The conjunctival mucous membrane includes the bulbar conjunctiva from the corneal limbus back to the fornix of the conjunctival sac, which folds forward to the palpebral conjunctiva consisting of orbital, tarsal, and marginal segments, 17 on the inner surface of the upper and lower eyelids. 39 –41 Conjunctival epithelium is stratified polyhedral to columnar nonkeratinizing epithelium with randomly distributed goblet cells in the nonhuman primate, dog, rabbit, and mouse, but stratified squamous with clusters of goblet cells in the rat. 39,40,42 Goblet cell density is highest in the fornix and bulbar regions. Near the cornea, goblet cells disappear and dendritic Langerhans cells predominate. 39,40
The CALT is the most well-described MALT around the eye, 8,10,12,43 –56 and CALT is more frequently found and reported in large laboratory animals 53 than in rodents. 57,58 In aged rabbits and postadolescent humans, the large number of CALT follicles will senesce. 10,48,50,59 The CALT can also consist of a loose and diffuse lymphoid layer in some species, 8,49,52,60 rather than the more common single to multiple discrete follicles, with or without secondary follicles containing germinal centers that are considered classic CALT. Discrete lymphoid follicles are located in the lamina propria of the palpebral conjunctiva and are less frequent near the fornix and bulbar conjunctiva in most species (Table 1.) 48,55
Reported Anatomic Location of Conjunctiva- and Lacrimal Drainage-Associated Lymphoid Tissue in Animals and Humans.
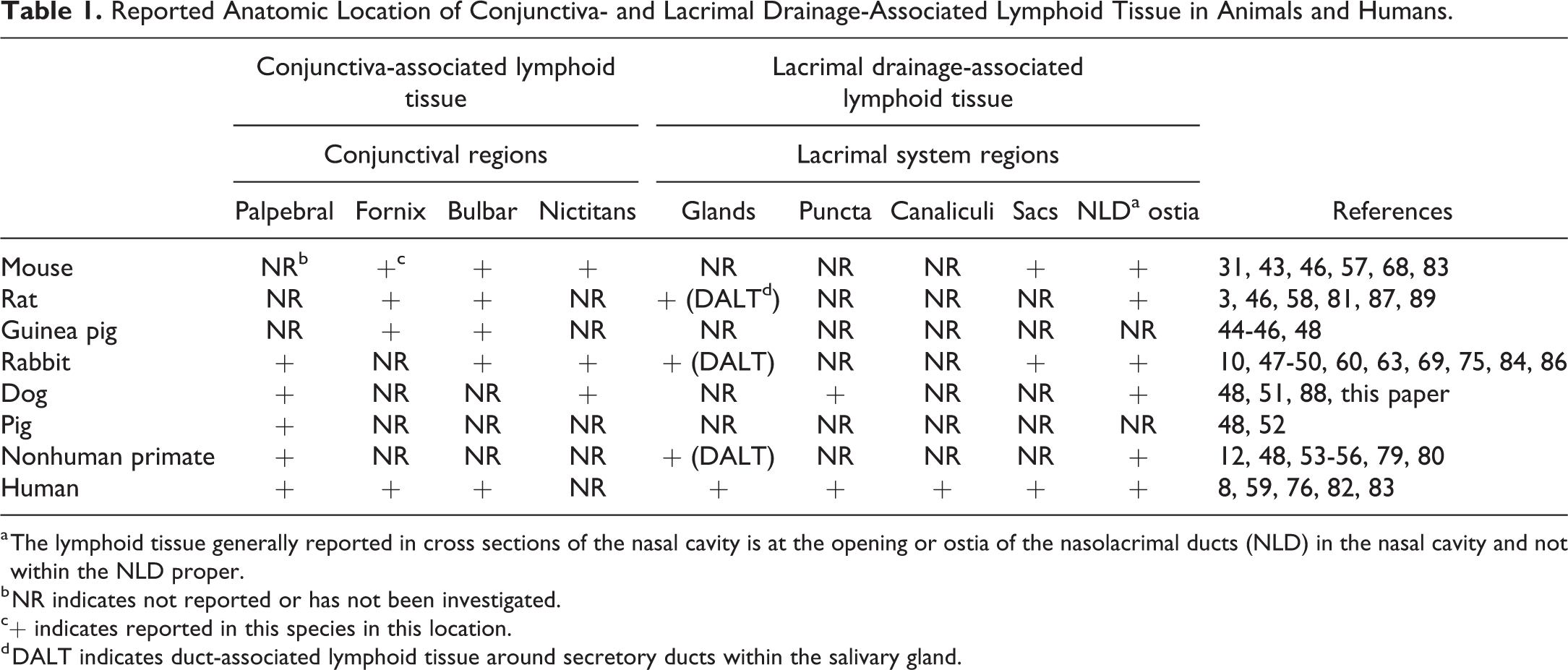
a The lymphoid tissue generally reported in cross sections of the nasal cavity is at the opening or ostia of the nasolacrimal ducts (NLD) in the nasal cavity and not within the NLD proper.
b NR indicates not reported or has not been investigated.
c+ indicates reported in this species in this location.
d DALT indicates duct-associated lymphoid tissue around secretory ducts within the salivary gland.
A variety of stimuli induces and expands CALT. Confocal microscopy of CALT in rabbit has shown that pro-inflammatory stimulation results in increased intraepithelial M cells, goblet cells, polymorphonuclear cells (PMNs), and intrafollicular accumulations of PMNs and macrophages. 61 In the rat, corneal allograft transplantation resulted in development of CALT within the palpebral conjunctiva. A Th1 phenotype mixed cell infiltrate with trailing to the limbus suggested a point of leukocyte trafficking into the conjunctiva. Along with increased lymphocyte cellularity and other MALT-like changes, a small number of granulocytes were also present in the foci starting 3 days post-transplantation. 58 Similarly, CALT was induced in BALB/c mice repeatedly challenged with topical Chlamydia trachomatis. 57 In a toxicity study, rabbits topically administered antiglaucoma prostaglandin analogs, with and without benzalkonium chloride (BAK) preservative, developed inflammation in CALT related to the BAK concentration. 62 The CALT in humans increases in size and number in response to multiple ocular diseases. 59 Conversely, the immunosuppressant cyclosporin has been shown to suppress multiple immunocompetent cell subtypes in the CALT of rabbits. 63 Lymphoma of CALT (MALToma) has been reported in humans, 8 where this tumor has been associated with dysbiosis. 64
The CALT in animals appears to develop similarly to that in humans. In the mouse, and other altricial species, CALT develops postnatally with antigen exposure after the eyelids open. 8,10,53 In the rabbit, CALT has been identified as early as 11 days of age. 10,50 In the guinea pig, CALT has been reported as small accumulations of lymphocytes in the fornix of fetal and newborn animals, with no difference between specific pathogen-free (SPF) and conventionally housed animals. Adult animals in this study had concurrent inflammation of the conjunctiva, and no other study has identified constitutive prenatal or perinatal CALT. This suggests that these older data from the guinea pig need to be reevaluated. 44 Although studies in mice and guinea pigs suggest that there is no difference in the development of CALT under SPF versus conventional barrier housing, current husbandry practices of strict barrier housing generally exclude infectious and many environmental antigens. Barrier housed adult mice lack primed mucosal memory T cells and are immunologically similar to neonatal mice, indicating that barrier housing represents a near SPF status. 65 Dust and particulates from bedding and food material present within the cage microenvironment may be sufficient stimulus for the development of some CALT even in SPF animals. The reason that CALT and other MALT sites are larger and more frequently reported in larger laboratory animals compared to rodents may be sampling errors. The ability to sometimes see CALT nodules macroscopically in larger animals, the preference for rabbits and larger animals for ocular toxicology studies, and generally limited sampling of conjunctiva including the nictitating membrane in routine toxicology studies limits the opportunity to observe the true incidence of CALT in all laboratory animals.
Nictitating Membrane CALT
The nictitating membrane, nictitans or third eyelid, consists of a T-shaped cartilaginous support covered by a fold of conjunctiva. The nictitans acts as a mobile and protective glandular structure that contributes to the tear film content and spreads the tear film. In laboratory animals, the nictitans membrane is well-developed in rabbits, dogs, and pigs, but is vestigial in rodents, guinea pig, chinchilla, and nonhuman primates, 52,66 and in humans is a remnant semilunar fold. 67 As the nictitans is covered by conjunctiva, lymphoid structures in the nictitans should be considered CALT. The CALT has been described in the vestigial nictitating membrane of young mice, along with CALT formation in the conjunctival fornix associated with antigenic challenge. 43,57 In SPF versus conventionally housed BALB/c mice, CALT development is similar with slightly more follicles in mice under conventional housing at 16 weeks of age, but more follicles in SPF mice at 24 weeks of age. Topical antigen challenge results in development of nictitans CALT or to an increase in nictitans follicles, and immunophenotypic changes are similar in SPF and conventionally housed mice. Interestingly, unilateral cervical lymphadenectomy results in development of increased CALT in both eyes, depending on the antigen tested, but splenectomy significantly increased the number of secondary follicles. 68 The occurrence of CALT in rodents is controversial with few published accounts. However, the general lack of screening for conjunctival CALT including in the vestigial nictitans and the general assumption that CALT does not exist in rodents may be limiting our knowledge on CALT in these species. The CALT in the nictitans occurs in the rabbit, 69 but this CALT has also been reported as lymphoid hyperplasia. 49 Older dogs frequently have well-developed CALT with multiple secondary follicles in the nictitans, 51 and they may also have loose sheets of lymphocytes in the adjacent palpebral conjunctiva (Figure 2B and C). In the dog, lymphoma of the nictitating membrane MALT (MALToma) has been reported. 70
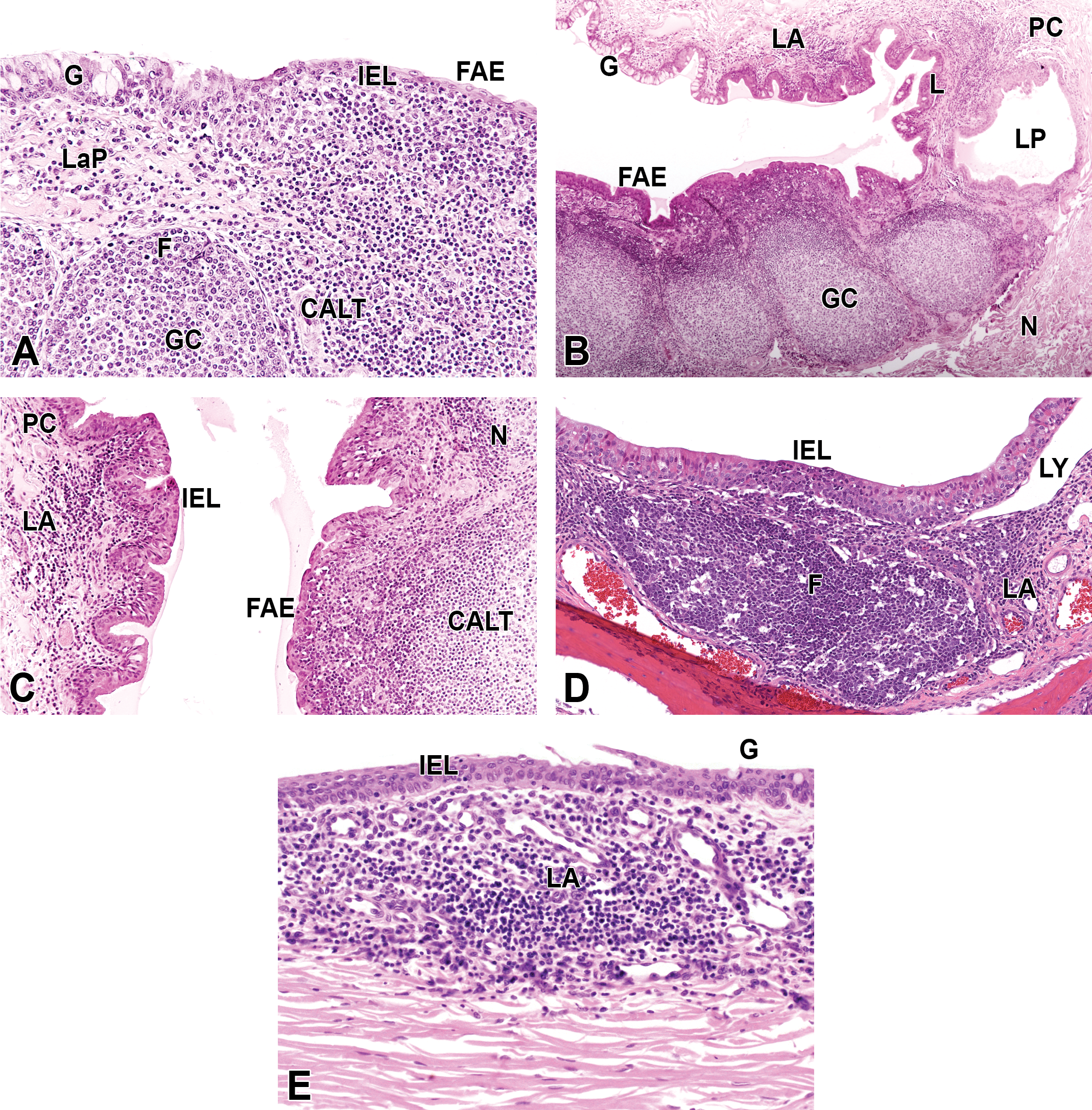
Representative variations on the histologic appearance of conjunctiva-associated lymphoid tissue (CALT) and periocular limbal lymphoid tissue in control animals from small molecule and medical device ocular studies. A, Palpebral CALT in a New Zealand (NZ) White rabbit. Follicle-associated epithelium (FAE) with intraepithelial lymphocytes (IELs). The CALT under the slightly domed FAE is densely packed but does not contain a delineated follicle. Multiple follicles (F) with a small dome and early germinal centers (GC) are overlain by lamina propria (LaP) containing numerous loosely arranged lymphocytes and normal conjunctival epithelium with goblet cells (G). B, CALT in the nictitans of a purpose-bred hound dog. Multiple large secondary follicles with GC segmentally elevate the epithelium. The lacrimal punctum (LP) is also partially surrounded by a layer of lymphocytes (L), and a loosely compact lymphoid aggregate (LA) is present in the palpebral conjunctiva (PC) opposite the nictitans (N). C, Higher magnification of N in (B). In the PC opposite to the N, FAE is not obvious, but the area lacks G and a few IEL are present. D, MALT around the nasolacrimal duct ostium in the nasal cavity of a Dutch-belted rabbit. The slightly raised FAE contains IEL directly above a large discrete F. There is an LA adjacent to the F, and the lymphatics (LY) are dilated. Courtesy of Covance Laboratories, Inc. E, The limbus of an NZ White rabbit contains an LA, with occasional plasma cells. The overlying limbal epithelium contains a few IEL and G. Numerous vascular profiles (spaces) are part of the limbal venous plexus.
Lacrimal Drainage-Associated Lymphoid Tissue
Ocular surface moisture and tear film secretions are provided by conjunctiva and periocular glands. Lacrimal glands consist of an exorbital gland near the ear and an infraorbital gland above the Harderian gland in rodents, a posterior lacrimal gland, Harderian gland and small glands in the nictitating membrane in rabbits, an extraocular lacrimal gland and gland of the nictitating membrane in carnivores, and a single dorsal lateral lacrimal gland and accessory lacrimal tissue in the fornical conjunctiva and eyelids in nonhuman primates. Secretions are distributed to the conjunctiva by multiple ducts, with the small glands providing continual supply to the tear film and the larger glands providing excess during irritation. In rodents and rabbits, the Harderian gland contributes lipid to tears, and in rodents, secreted porphyrin imparts a red-brown staining to fur (chromodacryorrhea). Meibomian gland at the eyelid margin contributes lipid (sebaceous secretions) in other species. 39 –42,50,71 –74 With its arborized duct system, the lacrimal gland is the upstream secretory gland of the LDALT, providing the aqueous secretions to the tear film. 75 The composition of the precorneal tear film varies between species, but secretions include water, lipids, mucins, enzymes, cytokines, chemokines, Igs, and antimicrobial peptides and proteins. Along with CALT, the lacrimal gland also provides the mucosal surfaces with antibody via SIgA. 17 The tear film secretions drain across the ocular surface to the medial canthus of the upper and lower eyelids and enter the upper and lower puncta to drain into lacrimal canaliculi that join to form a common duct. 76 In rabbits, there is a lacrimal punctum and canaliculus only in the lower eyelid. 77 Fluid then flows into the lacrimal sac, through the NLD, and exits at the NLDO into the nasal cavity. 78
Other than MALT at the NLDO, there is a lack of information on specific locations of LDALT in laboratory animals, which likely relates to the low frequency of sampling of the upper lacrimal drainage system in toxicology studies. Sites of LDALT (Table 1) have been identified in the periductal regions in lacrimal glands as probable duct-associated lymphoid tissue (DALT), 49,79 –81 around lacrimal puncta 8 (Figure 2B), in lacrimal canaliculi, 8,76 and the lacrimal sac. 8,76,82 –84 Diffuse and loose lymphocytes occur in periacinar stromal areas of the gland. 17,85 The more discrete foci of DALT probably represent periductal MALT for the glandular secretory ducts. 79,80 In all glandular tissues, DALT is probably under-recognized and is likely reported as infiltrates or inflammation. The most frequently identified site of spontaneously occurring LDALT in animals 3,31,49,60,86 –88 and humans 76,82 is at the bilateral NLDO (Figure 2D) that is visible in cross sections of the nasal cavity. 89 This lower part of the lacrimal drainage system is frequently reported as lacrimal duct or NLD, but anatomically this site of MALT surrounds the NLDO. Except near the lacrimal sac in humans, 8,76 lymphoid tissue has not been reported in the lacrimal canaliculi (ducts) or the NLDs themselves. In conventionally and SPF-housed mice, lacrimal sac MALT arises postnatally irrespective of antigenic stimulation and proliferates in response to antigens. 83 Expansion of MALT follicles has been reported in the lacrimal sac of rabbits with experimental Staphylococcus aureus dacryocystitis. 84 Increased concentrations of diffuse lymphocytes and follicles in LDALT occur when dacryocystitis, autoimmune Sjögren syndrome or any ocular surface disease is present in humans, 59 or in spontaneous 90 and experimental Sjögren in mice. 91 These lymphoid aggregates may indicate expansion of LDALT if found in anatomically appropriate sites; otherwise, these should be considered TLS due to inflammatory-based proliferation. Within the lacrimal drainage system, lymphoma and MALToma are primarily described in the lacrimal sac in humans. 82,92
Periocular Gland-Associated Lymphoid Tissue
Loose periacinar stromal lymphocytes and DALT would be expected to occur in periocular glands, particularly meibomian and Harderian glands, but there have been few investigations of any periorbital glands in normal animals 52 or humans. 93 Clusters of stromal lymphocytes and DALT have been reported in normal Harderian gland interstitium, particularly around the secretory ducts in rabbits. 49,94 Foci of lymphocytes or mixed cells not associated with the ducts or associated with glandular atrophy, necrosis, and loss of secretory granules should be considered an inflammatory or postinflammatory immune response. 49 Increased lymphoid aggregates can be found in all periocular glands of mice with experimental Sjögren syndrome, 91 and these are likely inflammatory-related TLS.
Ocular Lymphoid Tissue Aggregates Not Associated With Mucosa
The uvea or uveal tract consists of the choroid, ciliary body, and iris, and the limbus occurs at the junction of the sclera and the cornea. Immune privilege does not exclude surveillance by immune cells transiently crossing the retina or passing through the intraocular compartments to the uveal drainage system. Discrete mononuclear cell aggregates recorded as mononuclear cell infiltrates or inflammation 95 in healthy animals have been reported in the choroid of rabbits 49 and monkeys, 49,56,96 in the ciliary body in cynomolgus monkeys, 60,97 in the limbus of normal cynomolgus monkeys 56,98 and New Zealand White rabbits (Figure 2E), and in ciliary body, iris, choroid, and limbus of human eyes. 99,100 These lymphocytic aggregates are generally not expected in a normal eye, but a possible relationship of these cell aggregates to the development of CALT or general intraocular and periocular immunosurveillance needs to be investigated. Inflammatory cell aggregates have been reported in the ciliary body of monkeys treated with biologics, 97 at injection sites for intravitreal therapeutics, 101,102 and in experimental and spontaneous autoimmune uveitis. 103 –105 In the limbus of rats after corneal allograft transplantation, CALT and infiltrates of mixed leukocytes extending to the limbus were suggested as a point of entry for leukocytes trafficking into the conjunctiva. 58 The importance of the uveal tract to the immunology of the eye is demonstrated by the presence of plasma cells in uveal lymphoid foci, 98 a rich network of macrophages and DCs as APC in the iris and ciliary body, 106 and the ciliary body is also a site of immune-complex deposition. 107 Infiltrates of T and B lymphocytes at injection sites and in the vitreous, ciliary body, and choroid are also temporally associated with the development of intraocular antidrug antibodies. 101 T and B cells are also persistently increased in the uveal tract with experimental autoimmune uveitis, and uveal and sometimes retinal secondary lymphoid follicles are reported with recurrent spontaneous autoimmune uveitis in mice, 104 horses, 108 and humans. 105 When lymphocytic foci and secondary follicles develop in association with inflammation of the uveal tract and outer eye, a diagnosis of TLS may be more appropriate. 2,95,102,105,108,109
Otic-Associated Lymphoid Tissue
Eustachian Tube- and Middle Ear-Associated Lymphoid Tissue
The middle ear contains ossicles that change sound from the eardrum (outer ear) into a fluid wave that is transferred to the cochlea of the inner ear to generate the neural interactions necessary to interpret sound. The eustachian tube extends from the middle ear to the nasopharynx, with a cartilaginous portion near the pharyngeal orifice side and an osseous section on the tympanic bulla orifice side. The tympanic cavity of the middle ear contains air, and swallowing and chewing motions can transiently open the normally collapsed eustachian tube to adjust air pressure in the middle ear. 42,110,111
Compared to the eye, the ontogeny, locations, and dynamic properties of MALT in and around the ear are poorly understood, infrequently reported, and mostly in reference to infectious OM. 20,112 Ear MALT is reported in the eustachian tube and occasionally in the middle ear, but has not been reported in the periauricular glands, the external ear canal, tympanic membrane, nor in the highly protected inner ear of animals or humans. Mucosa-associated lymphoid tissue has been described in the middle ear and eustachian tube of healthy children. With OM, MALT increases, and formation of germinal centers is noted in both the middle ear and the pharyngeal side of the eustachian tube, 23,113 and possibly in the tympanic side of the eustachian tube. 113 Periluminal MALT has been reported in the pharyngeal portion of the eustachian tube above the submucosal glands and adjacent to Ostmann fatty tissue, in healthy minipigs, 110,114 and after experimental infections 115 and invasive tympanic bulla surgery in chinchilla (Schuh, unpublished data). Submucosal lymphocytes, but not organized MALT, have been observed in the middle ear of minipigs. 110 Submucosal lymphoid cells increase in the middle ear when OM was induced in rats, but few lymphocytes were found in the eustachian tube. 116 The paucity of information on MALT in the middle ear and eustachian tube of animals is reflective of the lack of emphasis placed on histopathologic examination of the ear in normal animals, and even otic-specific therapeutic and medical device nonclinical studies. 117
Discussion
Mononuclear, lymphocyte/lymphocytic or mixed cell infiltrates, inflammatory cell infiltrates, and, more recently, TLS are the recommended choice of morphological diagnostic terms for aggregates of lymphocytes and leukocytes in the gastrointestinal, 1 respiratory, 89 special senses, 95 and hematolymphoid, 2 as per the INHAND guides. These guides do not list normal MALT as a possible reason for aggregates of lymphoid tissue in mucosal tissues. Differentiation of MALT versus immune cell infiltrates requires the pathologist to determine whether the mucosal tissue preferentially forms MALT in the anatomic location under examination and to determine whether 1 or more histologic features of MALT are present. Distinguishing early and poorly formed MALT or tissue sections with MALT sectioned at the periphery from nonspecific lymphocyte infiltrates is challenging. Careful examination for characteristic features of MALT and knowledge that the site is preferred for MALT should drive the decision toward MALT (eg, 1 or more discrete lymphoid aggregates, with or without secondary follicles, in conjunctiva are CALT unless proven otherwise). Familiarity with preferred sites for normal MALT is also important to correctly categorize immune cell aggregates in mucosal tissues. Similar to other lymphoid tissue, MALT may be affected by nonproliferative and proliferative changes, 2,3 including inflammation (eg, inflammation of LDALT at the NLDO), 89 that may require the use of enhanced histopathology evaluations for lymphoid tissue. 118 Inflammatory cells with tissue damage in a mucosal site that is not preferred for MALT formation is likely inflammation of the mucosa. A differential diagnosis for lymphoid follicle formation during chronic inflammation in mucosal sites not preferred by MALT would be TLS. 2 Eye and ear MALT should be recorded as a line item if these are protocol-required tissues, but at least recorded as normal lymphoid tissue in a comment, rather than categorizing these as nonspecific cellular infiltrates or inflammation. Lymphoid aggregates are also an infrequent, but possible finding in the nonmucosal sites of the healthy eye including ciliary body, limbus, and choroid. 49,56,96,97,99,119 It is recommended that these aggregates be called infiltrates. 95 However, these noninflammatory lymphoid aggregates appear to be at preferred sites of formation in healthy eyes, and these foci should be investigated for a possible relationship to the normal immunosurveillance system of the eye.
To fully characterize the effects of therapeutics or medical devices on the special senses, routine microscopic examination with regional lymphoid tissue is recommended for all toxicology studies involving the eye and ear. Examining the globe separately from the periocular tissues may be adequate, but the globe and eyelids may need to be collected and processed en bloc to facilitate the examination of topographical concentrations of CALT in palpebral, nictitans, fornix, and bulbar conjunctiva. Examination of MALT in the lacrimal drainage system (except the lacrimal puncta and NLDO) and ear is challenging due to the need for blind collection in a bony vault. The LDALT acts as a conduit for immune interactions between the NALT and the eye, 11,31,32 and collection of 1 or more of the lacrimal gland, CALT with lacrimal puncta, lacrimal canaliculi, lacrimal sac, NLD, NLDO, NALT or NPT, and regional draining lymph nodes (parotid, retropharyngeal, and mandibular) should be considered to fully evaluate the response of the eye to therapeutics, vaccines, and medical devices. The most likely site of MALT in the ear is in the cartilaginous portion of the eustachian tube near the oropharynx, but responses in this eustachian MALT may also reflect changes in the oropharynx or the middle ear, rather than just a response within the eustachian tube. Therefore, tonsils (PR and/or palatine), regional drainage lymph nodes, and NALT or NPT may also be relevant lymphoid tissues to sample to characterize responses to therapeutics and medical devices targeted to the middle ear or the eustachian tube.
There is still much to be learned about the general immunobiology of special senses and the regional MALT of the eye and ear. The CALT and LDALT (EALT) of the eye, and eustachian tube- and perhaps middle ear-associated lymphoid tissue, need to be recognized as important regional MALT of the special senses that may also be involved in toxicology of the nasal and oral cavities. Similar to the recommended pathology reporting practices for MALT in the intestine and lung, properly categorizing and recording mucosal lymphoid tissues in the eye and the ear as MALT, rather than nonspecific immune infiltrates or inflammation, will improve risk communication for ocular and otic nonclinical studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
