Abstract
One strategy employed to prolong the ocular half-life of large molecule therapeutics is via covalent attachment to a carrier, resulting in an increase in size thereby slowing their clearance from the eye. Rabbit antigen-binding fragment conjugated to nanolipoprotein (RabFab-NLP) is a novel conjugate intended to prolong ocular half-life through an increase in hydrodynamic radius compared to Fab alone (∼12 vs ∼3 nm). Nanolipoproteins are mimetics of endogenous high-density lipoproteins and consist of lipids and apolipoproteins (ApoE422k), both biologically derived materials. The objective of this study was to evaluate the ocular toxicity and toxicokinetics of RabFab-NLP after a single intravitreal administration in New Zealand White rabbits. Serum toxicokinetic data suggested a significant increase in ocular residence time of RabFab-NLP compared to RabFab alone. Ophthalmic examinations showed that RabFab-NLP caused vitreous and lens opacities as early as day 3 and day 8 postdose, respectively, which persisted for the entire study duration to day 30. The RabFab-NLP-related microscopic findings were present in the lens, vitreous cavity, and/or optic nerve head. Based on the observed ocular toxicity, a single intravitreal dose of 1.3 mg/eye RabFab-NLP was not tolerated and caused vitreous opacity and cataracts in rabbit eyes.
Introduction
Intravitreal injection ensures bioavailability of therapeutics to the posterior segment of the eye and is the preferred route for administering treatments for diseases such as macular degeneration and diabetic retinopathy. Although intravitreal injection is a common and well-established procedure, there may be certain risks with frequent administration such as endophthalmitis, retinal detachment, and vitreous hemorrhage. 1,2 Thus, there is a need for long-acting drug delivery strategies to prolong the ocular residence of drugs and reduce the frequency of intravitreal injections. One such strategy is increasing the hydrodynamic radius (R h) by conjugating the active drug to a carrier, for example, polymers such as polyethylene glycol and hyaluronic acid. 3 A proof-of-concept rabbit intravitreal study has provided evidence that increasing the hydrodynamic size is an effective strategy to prolong ocular half-life. 4
One promising platform to improve R h are nanolipoprotein particles (NLPs), also referred to as nanodiscs or reconstituted high-density lipoproteins. Nanolipoprotein particles have been successfully developed for in vivo delivery of a wide variety of cargo, including therapeutic drugs, 5 –7 diagnostic imaging agents, 8 and vaccine and immunomodulation therapeutics. 9 Nanolipoprotein particles are mimetics of endogenous high-density lipoproteins (HDLs) and consist of lipids and apolipoproteins, both biologically derived materials. Nanolipoprotein particles are formed through a self-assembly process and adopt a discoidal lipid bilayer shape, where the hydrophobic periphery of the disc is stabilized by binding to the apolipoprotein. 10 Nanolipoprotein particles may offer several distinct advantages over other nanoparticle-based delivery technologies, including low toxicity, low immunogenicity, and diversity in compatible cargo. 11 Nanolipoprotein particle self-assembly protocols have been optimized for a variety of applications and drug cargo 12 –14 and are amenable to chemical conjugation to various moieties such as proteins/peptides, oligonucleotides, carbohydrates, and small molecules. More recently, antigen-binding fragment (Fab) conjugation to the NLP platform was reported. 15 In this study, Fab conjugation density could be tailored with a maximum Fab loading of ∼30 Fab/NLP (Figure 1). Interestingly, Fab conjugation was shown to significantly enhance NLP stability in a biological matrix, 15 which has implications for ocular delivery. More importantly, the average R h for the Fab-NLP conjugate was ∼12 nm, which is expected to prolong ocular half-life by approximately 2-fold.
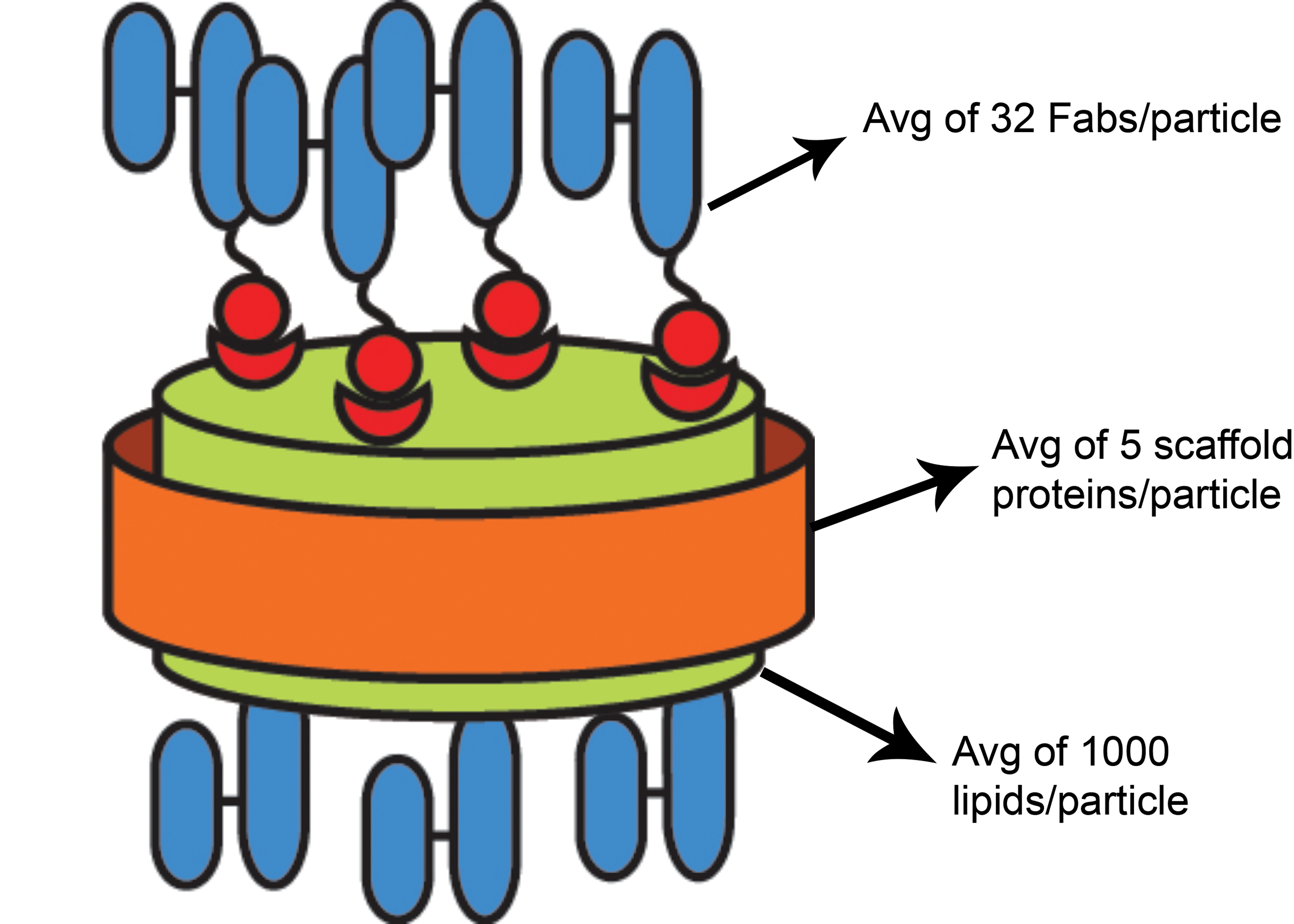
RabFab-NLP. A schematic of RabFab-NLP with quantification of each component (Fab, apolipoprotein, lipids) determined by reverse phase HPLC-evaporative light scattering detection (RP-ELSD). HPLC indicates high-performance liquid chromatography; RabFab-NLP, rabbit antigen-binding fragment conjugated to nanolipoprotein; RP-ELSD, reverse phase HPLC-evaporative light scattering detection.
The objective of this study was to assess the ocular toxicity of a species-matched, nonbinding rabbit Fab conjugated to NLP (RabFab-NLP) following a single intravitreal administration in New Zealand White rabbits. A species-matched Fab was used to reduce the potential for an immunogenic response and mitigate the ocular inflammation risk to enable assessment of the NLP platform. The serum toxicokinetic (TK) behavior of RabFab-NLP was consistent with the increase in R h; however, RabFab-NLP was not tolerated in the eye and caused lens and vitreous opacity. Data from the rabbit intravitreal study and poststudy investigation of vitreous samples are presented here.
Materials and Methods
Animals
This study was approved by the Institutional Animal Care and Use Committee (Approval No. IACUC 148-070) and was conducted in accordance with the animal welfare bylaws of Shin Nippon Biomedical Labratories Ltd., Drug Safety Research Labratories (SNBL DSR), which is accredited by Association for Assessment and Accreditation of Laboratory Animal Care International. Six New Zealand White male rabbits, 4 to 6 months of age and weighing 1.8 to 3.5 kg at the initiation of quarantine and acclimation, were used in the study. Rabbits were quarantined and acclimated for a total of 12 days before study initiation.
Protein Production and Purification of apoE422k and Fab
ApoE422k was produced in Escherichia coli cells under shake flask conditions using established expression plasmids and methods as described previously. 15 Briefly, apoE422k was purified over a nickel-nitrilotriacetic acid (Ni-NTA) column (XK16/20 3 mL), followed by size exclusion chromatograph (SEC; Superdex 75 16/60), and reverse phase high-performance liquid chromatography (RP-HPLC). The Ni-NTA column was washed and protein bound in 50 mM phosphate buffer, 200 mM sodium chloride, and 10 mM imidazole, pH 8. The protein was washed (20 column volumes) with 0.05% Triton X114 + 0.05% Triton X100 and eluted with 50 mM phosphate buffer, 200 mM sodium chloride, and 400 mM imidazole, pH 8. The pooled fractions were filtered and concentrated using 3 kDa molecular weight (MW) cutoff spin concentrators. The histidine (His) tag was then removed through Tobacco Etch Virus nuclear-inclusion-a endopeptidase (TEV protease) digestion (TEV tag was added on N-terminus between the His tag and protein sequence) as described previously. 15 The cleaved protein was purified from the TEV protease, which contained a His tag, over the same Ni-NTA column described above. The pooled protein fractions were concentrated and run on SEC in phosphate-buffered saline (PBS). Fractions were analyzed for identity using mass spectrometry and for aggregation by SEC. Pooled SEC fractions were then run on RP-HPLC to further remove endotoxin. The high-performance liquid chromatography (HPLC) fractions were collected and lyophilized to remove organic solvent. The apoE422k was resuspended in water and stored at −80 °C until used. Protein concentration was determined by absorbance at 280 nm.
The RabFab constructs were designed with a C-terminal cysteine to enable site-specific conjugation to NLP. Production and purification of RabFab were performed as previously described. 4 Removal of cysteine adducts from the C-terminus was accomplished by treating RabFab with 20 mM dithiothreitol followed by 6.5 mM glutathione (GSH). Samples were then buffer exchanged into 200 mM arginine succinate pH 5 to minimize C-terminal reactivity during storage.
Nanolipoprotein Assembly and RabFab-NLP Conjugation and Purification
Nanolipoproteins were assembled as described previously. 11 Briefly, NLPs were assembled with a combination of 80 mol% 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) and 20 mol% 1,2-dioleoyl-sn-glycero-3-phospho ethanolamine-N-[4-(p-maleimidomethyl) cyclohexanecarboxamide (DOPE-MCC) lipids at a total lipid to apoE422k molar ratio of 80:1. The lipids were either prepared or obtained in chloroform and aliquoted into glass vials. Chloroform was removed under a stream of N2 with agitation to form a thin lipid film. Lipids were solubilized in 50 mM sodium phosphate buffer, pH 6.0, 150 mM sodium chloride using 80 mM sodium cholate. ApoE422k was then added (150 µM in final assembly volume), and samples were incubated at 22 °C for at least 1 hour. The samples were incubated with detergent removing biobeads (Sigma-Aldrich) for 2 hours with rocking in 500 µL costar 0.22 spin filters. The samples were then centrifuged at 200 g for 5 minutes, and the filtrate containing the NLPs was collected. After cholate removal, the apoE422k concentration was measured by A280 absorbance, and the NLPs were incubated with RabFab in 50 mM sodium phosphate buffer, pH 6.0, 150 mM sodium chloride at an RabFab: NLP molar ratio of 60. The samples were placed on a rocker and incubated for 2 to 4 hours. After the 2- to 4-hour reaction incubation period, n-acetylcysteine was added at a 2-fold molar excess over DOPE-MCC to quench any unreacted maleimides. The RabFab-NLP conjugates were then purified over a S200 10/300 increase column using an AKTA Avant system. Each fraction across the RabFab-NLP peak was analyzed by size exclusion chromatography—multiangle static light scattering and fractions were pooled based on MW and R h analysis to yield the most homogeneous RabFab-NLP sample as described previously. 15
Endotoxin removal was performed by loading the purified RabFab-NLP pool over a cation exchange column (5 mL HiTrap SPHP, GE Healthcare) and performing a 19-hour wash step using 0.05% Triton X100, 0.05% Triton X114 spiked into 20 mM sodium acetate, pH 5.0. Once all traces of Triton were washed away, RabFab-NLP was eluted using a gradient between 0% and 30% sodium chloride before being concentrated to 26 mg/mL in previously sanitized centrifuge concentrators with a MW cutoff of 50 kDa. Finally, the material was passed through a 0.22 µm filter under aseptic conditions.
Endotoxin Detection strategy
All endotoxin measurements were performed using a kinetic limulus amebocyte lysate (LAL) assay with a 4-point standard curve (0.005-5.0 EU/mL). Reagent kits were obtained from Charles River Laboratories, and data were analyzed using WinKQCL software. For all measurements, LAL Reagent Water (Lonza) was used to achieve the maximum valid dilution. Since apoE422k is known to interact with endotoxin, 16,17 and since nanoparticle formulations have been known to trap endotoxin and also interfere with standard endotoxin detection assays, 18 it was necessary to develop a strategy to validate the measured endotoxin level of assembled RabFab-NLP. Unbound protein components (apoE422k and RabFab), NLP prior to Fab conjugation, and RabFab-NLP were measured for endotoxin burden under native conditions. Next, to test whether endotoxin was trapped inside the particles, lipids were extracted from RabFab-NLP, and endotoxin burden of the proteins-only mixture was measured. Independently, RabFab-NLP was also treated with an EndoPrep Kit (BioDtech Inc) to digest the proteins, and endotoxin of the lipids-only component was measured. Together, these results supported the conclusion that endotoxin was not trapped within the particle nor bound to apoE422k.
Intravitreal Administration
A single 50 μL injection of RabFab-NLP (26.1 mg/mL), RabFab (23.8 mg/mL), and vehicle control (PBS) was administered bilaterally in each rabbit by intravitreal injection (Table 1). Prior to dosing, a mydriatic (Mydrin-P ophthalmic solution, Santen Pharmaceutical Co, Ltd) was instilled after which animals were anesthetized by intramuscular injection of ∼1.2 mL/kg of a 1:4 (volume ratio) mixture of xylazine (20 mg/mL, Celactal, Bayer Yakuhin Ltd) and ketamine hydrochloride (50 mg/mL, Supriya Lifescience Ltd or Arevipharma GmbH) or ketamine hydrochloride (ketalar for intramuscular injection, 500 mg, Daiichi Sankyo Propharma Co, Ltd, 50 mg/mL). After the animal was anesthetized, each eyelid was disinfected with cotton swabs permeated with iodine-polyvinyl alchohol (PA-IODO) Ophthalmic and Eye Washing Solution (Nihon Tenganyaku Kenkyusho Co, Ltd) diluted 4-fold with physiological saline (Otsuka Pharmaceutical Factory, Inc).
Study Design of the Rabbit Intravitreal Study.a
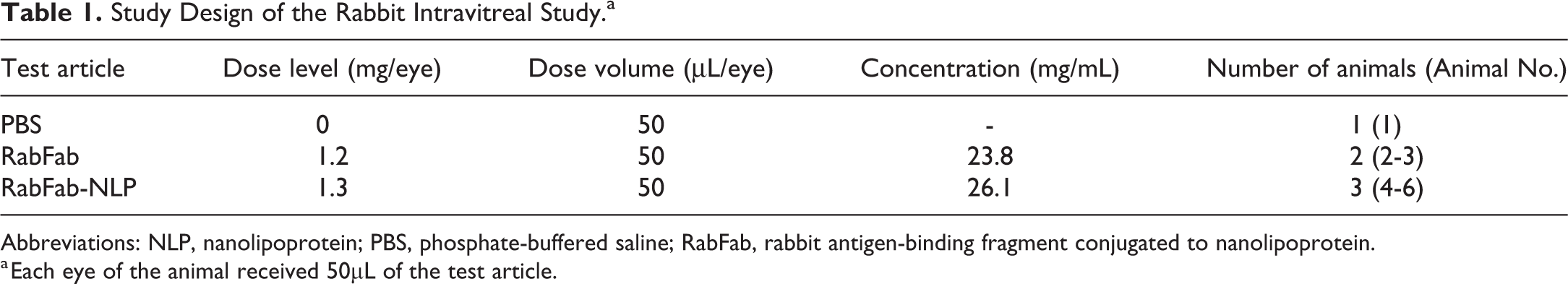
Abbreviations: NLP, nanolipoprotein; PBS, phosphate-buffered saline; RabFab, rabbit antigen-binding fragment conjugated to nanolipoprotein.
a Each eye of the animal received 50µL of the test article.
Intravitreal injections were performed from the inferotemporal position of the conjunctiva at approximately 2.0 to 3.0 mm posterior from the limbus (5 and 7 o’clock positions for the left and right eyes, respectively). A 1 mL insulin syringe with a fixed 27 gauge, 0.5-inch needle was used for the injection. The 50 μL was injected over a period of ∼6 seconds.
Ophthalmic Examinations and Intraocular Pressure
Ophthalmic examinations (OEs) were scheduled predose (day −2), day 1 (immediately after dosing), and 3, 8, 15, 22, 29, 36, and 42. Ophthalmic examinations scheduled on day 36 and 42 did not take place since the study was terminated earlier, on day 30. Ophthalmic examinations were performed by a Diplomate of the Japanese College of Fundamental Ophthalmologist and consisted of a gross observation and pupillary light reflex examination using a penlight or portable slit lamp biomicroscope (SL-15 or SL-17, Kowa Company, Ltd). After instillation of a mydriatic, the anterior ocular segment and optic media were examined using a portable slit lamp biomicroscope. The fundi were examined using an indirect ophthalmoscope (ID-10, Topcon Corporation or IO-α Small Pupil, Neitz Instruments Co, Ltd). Whenever abnormalities were observed, photographs were taken with a hand held digital fundus camera (GENESIS-Df, Kowa Company, Ltd). Ophthalmic evaluations were conducted according to the criteria by the McDonald-Shadduck method. 19
Intraocular pressure (IOP) was measured on the same occasion as OEs. Intraocular pressure was measured using a TonoVet tonometer (TV01, Tiolat Oy) after instillation of an ophthalmic surface anesthetic (Benoxil 0.4% solution, Santen Pharmaceutical Co, Ltd).
Clinical Observations and Body weight
Animals were observed for clinical signs at least once daily during quarantine and acclimation period and at least twice daily (prior to dosing and 2-4 hours after dosing) and twice daily on nondosing days. Animal weights were monitored once on days −12, −6, and −1 before dosing, once on the day of dosing, and once weekly on nondosing days.
Toxicokinetics and Anti-drug Antibody
For assessment of serum toxicokinetics, approximately 0.5 mL of blood was drawn from the auricular vein on day 1 (6 hours after dosing) and 2, 5, 8, 12, 15, 18, 24, and 30. The serum concentration of RabFab molecules was determined by enzyme-linked immunosorbent assay (ELISA). Anti-RabFab ID antibody #9 (Genentech) was diluted to 2 µg/mL in coating buffer (50 mM sodium bicarbonate, pH 9.6) and coated onto 384-well MaxiSorp plates (Thermo Scientific) overnight at 4 °C. Plates were washed with washing buffer (PBS 0.05% Tween-20) and blocked with blocking buffer (0.5% bovine serum albumin [BSA], 15 ppm Proclin in PBS) for 1 to 2 hours. The blocking incubation and all subsequent incubations were performed at room temperature with gentle agitation. Standards were made from the source material of each different rabbit Fab molecule. The standards and samples were diluted in the standard/sample dilution buffer (PBS, 0.5% BSA, 15 ppm Proclin, 0.05% Tween 20, 0.25% CHAPS, 5 mM EDTA, 0.35 M NaCl [pH 7.4]) with 10% normal rabbit serum and added to the blocked plate for 2 hours. The standard range was 0.078 to 5 ng/mL. Serum samples had an initial dilution of 1:100. The plate bound RabFab molecules were detected with biotinylated anti-rabbit Fab ID antibody #6 (Genentech) diluted to 1 µg/mL in assay buffer (PBS, 0.5% BSA, 15 ppm Proclin 0.05% Tween 20) for a 1-hour incubation. The plate was washed and followed by incubation with streptavidin poly-horseradish peroxidase (HRP; Fitzgerald) diluted to 25 ng/mL in assay buffer for a 1-hour incubation. After a final wash, the plate was developed with tetramethyl benzidine (TMB) peroxidase substrate system (KPL), and the reaction was stopped with 1 M phosphoric acid. The plates were read at 450 nm with a 620 nm reference. The concentrations of the rabbit Fab molecules were calculated from a 4-parameter fit of the respective standard curve. Pharmacokinetic parameters were determined by noncompartmental analysis using the extravascular input model (201, WinNonlin, version 5.2.1, Pharsight).
For anti-drug antibody (ADA) analysis, approximately 0.5 mL of blood was drawn from the auricular vein on day −5, 5, 8, 15, 18, 24, and 30. Samples were analyzed with an ELISA assay to qualify the presence of ADAs to RabFab in rabbit serum. RabFab was diluted to 1 μg/mL in coating buffer and incubated on 384-well MaxiSorp plates overnight at 4 °C. Plates were washed and then blocked for 2 hours. This and all subsequent steps were performed at room temperature with gentle agitation. Naive rabbit serum samples were employed as a negative control. Serum samples were diluted to a minimum of 1:100 and serially diluted 1:3 in sample buffer. The controls and samples were incubated on the blocked plates for 2 hours. Bound ADAs were detected using HRP-conjugated goat-anti-rabbit Fc antibody (Jackson ImmunoResearch) at 40 ng/mL in assay buffer for 1 hour. After a final wash, TMB was added, color developed, and the reaction was stopped with 1 M phosphoric acid. The plates were read at 450 nm with a 620 nm reference using a microplate reader. A cut point (3 times the negative control) was used to determine ADA-positive and ADA-negative samples. Samples with a signal above the cut point were considered ADA positive and samples with a signal below the cut point were considered ADA negative. Titer was defined as log10 of the last dilution of serum that has an oprical density (OD) above the cut point. The lowest titer was 2 since the minimum dilution was 1:100.
Histopathology
The animals were euthanized earlier than scheduled (changed from day 42 to day 30) due to adverse ocular observations of lens and vitreous opacity. At necropsy, the external appearance and internal organs and tissues were examined macroscopically. Only the eyes were examined by histopathology. Four of 6 eyes (1 eye of animal #4, 2 eyes of animal #5, and 1 eye of animal #6) were processed for microscopic assessment, and the other 2 eyes, 1 eye each of animal #4 and #6, were used for follow-up investigative work. For microscopic assessment, the eyes and optic nerves were fixed by submerging in a mixture of 3% glutaraldehyde and 2.5% formalin for at least 24 hours. A lateral, medial, and central superioinferior calotte were trimmed and routinely processed to paraffin block. Three 5 µm sections from each calotte were cut at 100 µm intervals and stained with hematoxylin and eosin.
Poststudy Investigation
To investigate the mechanism behind vitreous opacity that was observed in all eyes administered RabFab-NLP, vitreous body was collected from 2 eyes (1 eye each of animals #4 and #6) and submitted to liquid chromatography-mass spectrometry (LC/MS) and reverse phase HPLC-evaporative light scattering detection (RP-ELSD) analysis as follows. Briefly, the vitreous was centrifuged to separate the soluble and the insoluble fractions, following which the insoluble fraction was resuspended in dimethylformamide (DMF) to solubilize lipids or guanidine hydrochloride (GuHCl) to solubilize proteins. The soluble fraction and extracts of the insoluble fraction were analyzed by LC/MS for identification and RP-ELSD for quantification. The LC/MS and RP-HPLC analyses were performed using an Agilent 6230 ESI-TOF LC/MS and Agilent 1290 Infinity Bio-inert HPLC, respectively. A Kinetex 2.6 µm XB-C18 column (Phenomenex), heated to 80 °C, was used to analyze the injected vitreous samples. The solvent was run as a gradient from a mixture of 30% methanol (MeOH) and 70% water to 100% 2-propanol. All solvents had 0.05% trifluoroacetic acid.
To assess whether the particulates observed on OEs were due to the fragmentation of the RabFab-NLP in the vitreous, the stability of RabFab-NLP was monitored at 37 °C for over 1 month. For these experiments, RabFab-NLP was spiked in PBS or rabbit vitreous and heated to 37 °C using different methods (H20 bath, bead bath, metal, and air/convection). The samples were extracted in MeOH, DMF, and GuHCl and analyzed by LC/MS and RP-ELSD as described above.
Results
There were no RabFab and RabFab-NLP-related changes in mortality, clinical signs, body weights, IOPs, or macroscopic findings in any animal. All animals underwent necropsy ahead of schedule (changed from day 42 to day 30) because of adverse cataracts or vitreous opacities in RabFab-NLP dosed animals. There were no notable in-life or microscopic findings in animals administered PBS or RabFab.
Ophthalmic Examination Findings
There were no ophthalmic changes noted in RabFab or PBS dosed eyes. The RabFab-NLP dosed animals developed vitreous particulate opacities as early as 3 days postdose in all eyes, which persisted and increased in severity over time (Figure 2A). Furthermore, lens opacity (posterior cortical, posterior subcapsular, and posterior capsular) was observed in 3 of 6 eyes (both eyes of animals #4 and 1 eye of animal #5) on day 8 OEs and worsened over the course of the study (Figure 2B). All 3 types of cataracts were observed in 1 eye of animal #5 and both eyes of animal #4. Animal #5 had cataracts only in 1 eye. Marked lens opacity in both eyes of animal #4 precluded assessment of the vitreous body in this animal starting day 8. No lens opacity was present in either eye of animal #6 (Table 2).
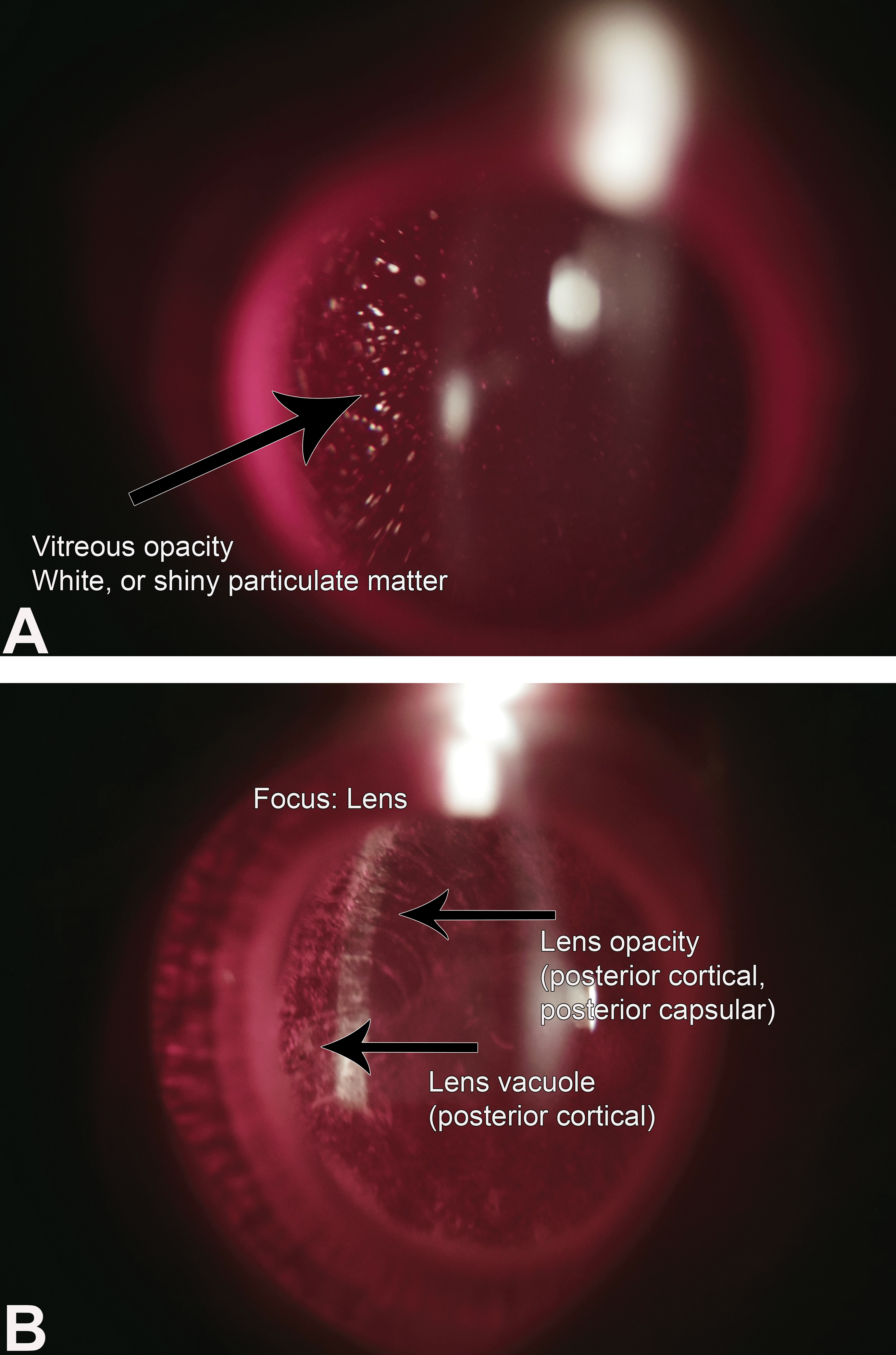
A, Representative picture of vitreous particulate opacity observed in RabFab-NLP dosed eyes. Slit-lamp examination of right eye of animal #5 on day 29. B, Slit-lamp examination of the left eye of animal #4 on day 29 showing lens opacity and vacuolation. RabFab-NLP indicates rabbit antigen-binding fragment conjugated to nanolipoprotein.
Lens and Vitreous Opacity Observations on Ophthalmic Examinations in RabFab-NLP Dosed Eyes.a
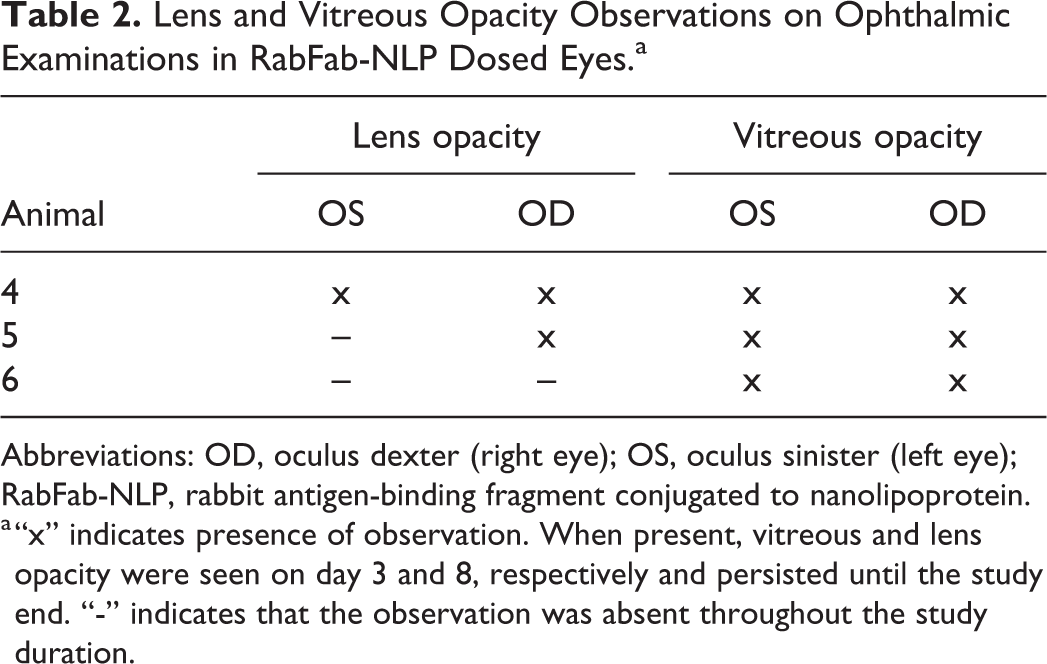
Abbreviations: OD, oculus dexter (right eye); OS, oculus sinister (left eye); RabFab-NLP, rabbit antigen-binding fragment conjugated to nanolipoprotein.
a “x” indicates presence of observation. When present, vitreous and lens opacity were seen on day 3 and 8, respectively and persisted until the study end. “-” indicates that the observation was absent throughout the study duration.
No anterior chamber inflammation was noted in any RabFab or RabFab-NLP dosed eye. However, presence of inflammatory cells in the vitreous chamber was difficult to assess due to particulate opacity in the vitreous of RabFab-NLP dosed eyes.
Toxicokinetics and ADA
Clearance from the vitreous humor is expected to be the rate-limiting step in elimination of intravitreally administered test articles, resulting in flip-flop kinetics in serum where terminal half-life in serum reflects vitreal elimination half-life. The observed TK behavior was consistent with a prolonged ocular half-life of RabFab-NLP (serum t 1/2 = 5.5 days) compared to RabFab (serum t 1/2 = 3.1 days), and exposure was maintained for the full duration of the study (Table 3 and Figure 3A). Observed TK behavior for RabFab was consistent with historical data (t 1/2 = 3.3 ± 0.4 days, n = 4 studies; data not shown). The ∼1.75-fold prolongation in half-life of RabFab-NLP compared to RabFab alone is consistent with the expected ∼2-fold extension based on the increase in R h. However, the observed TK data were highly variable between individual animals before and after the appearance of anti-RabFab ADA in RabFab-NLP treated animals (Figure 3B). Thus, the TK parameters presented here are not considered quantitatively reliable in the absence of confirmatory data in vitreous humor. Serum ADAs were detected in all RabFab-NLP treated animals; at day 15 and beyond for animal #6 and day 24 and beyond for animals #4 and #5 (Table 4). No ADAs were detected in RabFab-treated animals. The serum ADA assay only measured reactivity against RabFab, not NLP, so it is unknown whether ADAs reactive to NLP were present. In addition, ADAs against RabFab were not characterized to determine whether they may neutralize the binding of anti-idiotype antibodies that were used for capture or detection in the TK assays.
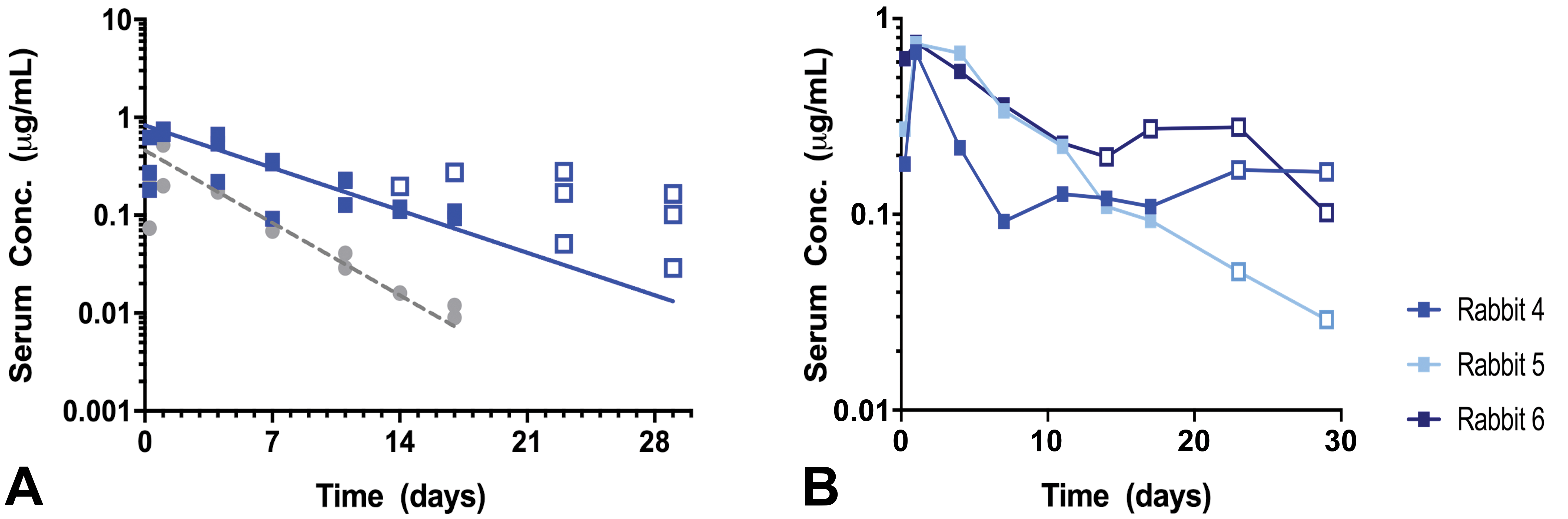
A, Concentration of RabFab and RabFab-NLP in serum following bilateral intravitreal injection of 1.2 and 1.3 mg/eye, respectively. Symbols indicate measured concentrations of RabFab (grey) or RabFab-NLP (blue); open symbols indicate samples which were positive for ADA; lines indicate non-compartmental analysis (NCA) fits to ADA negative mean data. Y-axis: serum concentration (μg/mL) and X-axis: time (days). B, Individual concentrations of RabFab-NLP in serum following bilateral injection of 1.3 mg/eye in NZW rabbits. Symbols indicate measured concentrations of RabFab-NLP; open symbols indicate samples which were positive for ADA. Y-axis: serum concentration (μg/mL) and x-axis: time (days). ADA indicates anti-drug antibody; NZW, New Zealand White; RabFab-NLP, rabbit antigen-binding fragment conjugated to nanolipoprotein.
Noncompartmental Toxicokinetic Parameters of RabFab and RabFab-NLP in Serum.a

Abbreviations: AUCall, area under the serum concentration versus time curve from time = 0 to time of the last measurable concentration calculated using the log linear trapezoidal rule; AUCinf = area under the serum concentration versus time curve extrapolated to infinity (AUClast + C last/λz); CL/F, clearance = nominal dose (mg/kg)/AUC0-inf; C max, maximum observed concentration; T max, time of maximum observed concentration; t 1/2λz, half-life associated with the terminal phase = ln(2)/λz; V z/F, volume of distribution based on the terminal phase = Dose/(λz*AUC0-inf).
a Note that marked interindividual variability in toxicokinetic behavior for RabFab-NLP limits the quantitative reliability of the toxicokinetic parameters, as described in the text.
Serum ADA Titers of RabFab and RabFab-NLP Intravitrally Dosed Animals.a

Abbreviations: ADA, antidrug-antibody; RabFab-NLP, rabbit antigen-binding fragment conjugated to nanolipoprotein.
a Titer is defined as log10 of the last dilution of serum that has an OD. Above the cut point in the assay which is 3× the OD of normal rabbit serum.
b Samples with ADA.
Histopathology
No PBS or RabFab-related microscopic findings were observed in any region of the eye, whereas RabFab-NLP-related microscopic findings were present in the lens, vitreous cavity, and/or optic nerve. The primary microscopic finding was minimal to moderate lens degeneration characterized by lens fiber swelling, cortical liquefaction and vacuolation, and posterior migration of lens epithelium nuclei (Figure 4A and B). The posterior aspect of the lens was most severely affected, and in 2 eyes almost 100% of the posterior surface and the equators of the lens were affected. These findings correlated to the lens opacity noted during OE in animals #4 and #5 and were not apparent in animal #6. Additional findings included minimal mononuclear infiltrates surrounding blood vessels at the optic nerve head (2 of 4 eyes) and extending into the vitreous chamber in all eyes (Figure 4C). Pale eosinophilic fibrillar material was also noted in the vitreous chamber of all eyes. These findings are most consistent with very slight inflammation, which was not considered sufficiently severe to result in the vitreous opacities noted during slit-lamp examination. Therefore, there was no definitive microscopic correlate for the observed in-life vitreous opacities. Due to the expected dehydration and condensation of the vitreous during fixation and the relatively small area of the vitreous chamber assessed in histologic sections, it is possible that the cause of the vitreous opacities was simply not in section.
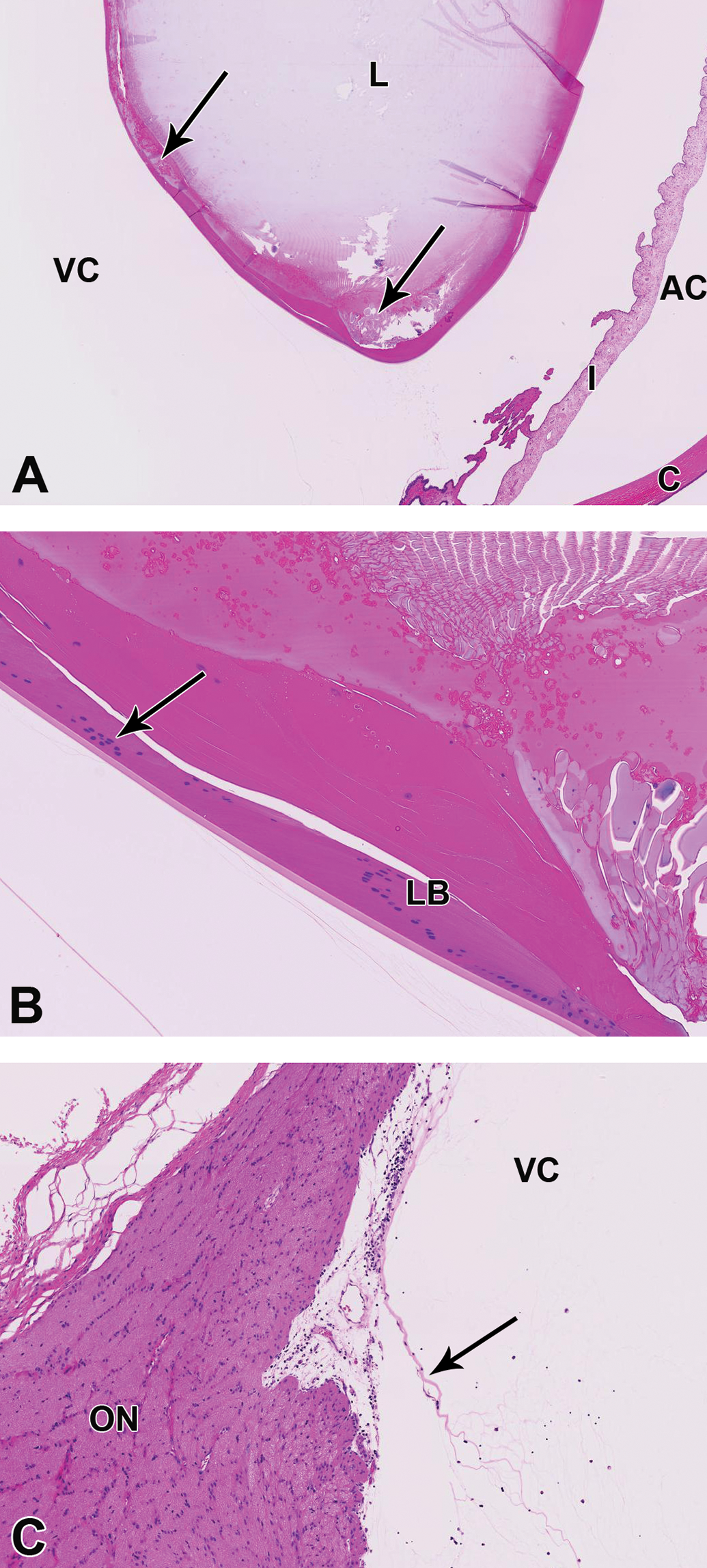
A, The posterior aspect of the lens and the equator exhibit multifocal swelling of lens fibers, vacuolation, and regions of liquefaction (arrows). B, High power image of the regions of lens fiber swelling and vacuolation. Note the migration of lens epithelium nuclei (arrow) posterior to the lens bow (LB). C, Vessels at the optic nerve head are multifocally cuffed by mononuclear cells and similar cells extend into the vitreous chamber. Pale eosinophilic fibrillar material (arrow) is present in the vitreous chamber. AC, anterior chamber; C, cornea; I, iris; L, lens; ON, optic nerve; VC, vitreous chamber.
Investigative Studies
The LC/MS and RP-ELSD analyses of the vitreous from RabFab-NLP dosed eyes (1 eye of #4 and 1 eye of #6) were conducted after separating the soluble and insoluble fractions and extracting the insoluble fraction with DMF and GuHCl to solubilize lipids and proteins, respectively. No apoE422k or RabFab were detected in any of the fractions. There was a small amount of protein, small amounts of unidentified lipids not originally present in the NLP, and trace levels of DOPC detected in analyzed samples (Figure 5A and B). Thus, there was very little remaining test article present at the time of necropsy on day 30. The LC/MS method used here was mainly done to identify peaks seen by RP-ELSD and was not sensitive enough to quantify small amounts of test article in the vitreous, and thus does not exclude the presence of test article at the time of necropsy.
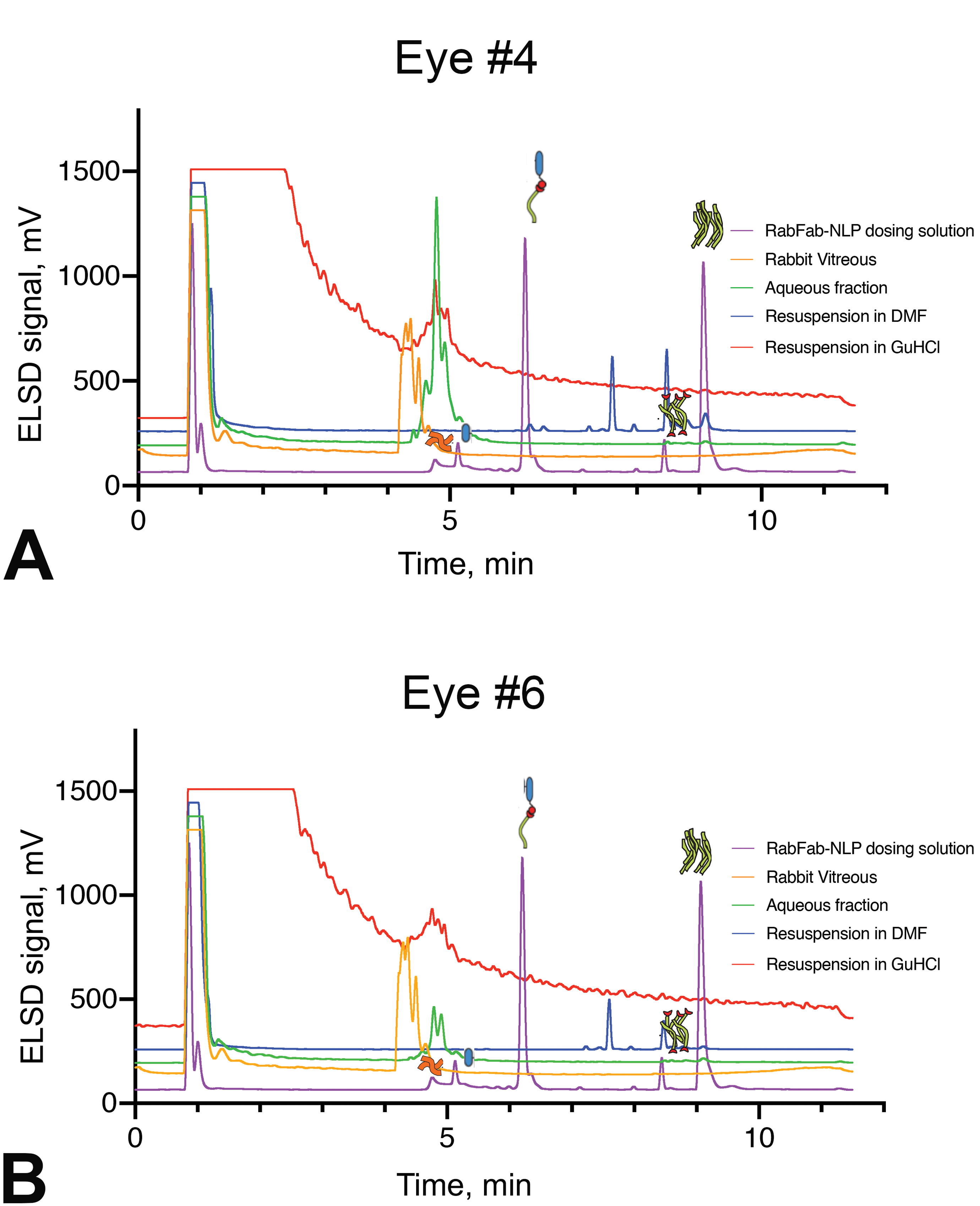
RP-ELSD analysis of vitreous samples (A) from animal #4 and (B) animal #6. All samples were run under reducing conditions. For the purple traces, the peak around 1 minute is solvent, while the peaks around 5 minutes are apoE422k and reduced RabFab heavy chain (HC). The peak around 6.5 minutes is DOPE-MCC conjugated to HC, the peak around 8.5 minutes is free DOPE-MCC and the peak at 9 minutes is DOPC. In both eye #4 and #6, no apoE422k or Fab was detected in any of the vitreous fractions, although some vitreous proteins were detected (green traces). In addition, some unidentified lipids were detected in both eyes (blue traces, around 7.5 and 8.5 minutes) and a small amount of DOPC was detected in the DMF extract of eye #4 (blue trace). The y-axis for the gold, green, blue, and red traces has been offset by 5%, 10%, 15%, and 20%, respectively, for ease of viewing. DMF indicates N, N’-dimethylformamide; DOPE-MCC, 1,2-dioleoyl-sn-glycero-3-phospho ethanolamine-N-[4-(p-maleimidomethyl) cyclohexanecarboxamide; RP-ELSD, reverse phase high-performance liquid chromatography-evaporative light scattering detection.
The particulate formation that was observed in the vitreous could not be replicated in the in vitro stability test when RabFab-NLP was spiked in either PBS or rabbit vitreous and kept for over 1 month at 37 °C. This result suggests that fragmentation of RabFab-NLP was unlikely to be a major factor in the observed particulate formation in vivo.
Discussion
In the current study, the toxicokinetics and ocular toxicity of the NLP platform were evaluated following intravitreal injection in rabbits. The serum TK data suggested approximately a 2-fold prolongation of ocular half-life of RabFab-NLP compared to RabFab, which was predicted based on R h and is consistent with published literature. 4
Rabbits are known to mount a robust immune response against intravitreally administered humanized biologics and develop ocular inflammation. 20 Here, we used a species matched Fab conjugated to NLP to circumvent the confounding effects of immunogenicity and assess the tolerability of the long-acting delivery platform. RabFab did not cause development of ADA in this or past studies, however, all RabFab-NLP dosed animals developed ADA directed against RabFab, suggesting that the conjugated molecule is likely more immunogenic than the free Fab. We have observed a similar phenomenon in another rabbit intravitreal study, where RabFab was conjugated to a different moiety (data not shown) and it is possible that this effect may be specific to rabbits. It is important to note that ADA response to NLP was not assessed in this study and it is not known whether ADAs reactive to NLP were present. The ADA reaction against RabFab was relatively low with a maximum ADA log titer of 3.4 for animal #6, while animals #4 and #5 had ADA log titers of 3 and 2.5, respectively. Anti-drug antibodies were detected at a later timepoint compared to the first observation of lens and vitreous opacity on OEs, thus, it is unlikely that these findings were related to the immunogenic response. Furthermore, animal #6 developed ADA earlier than the other 2 animals (day 15 vs day 24) and had a slightly higher ADA titer, however, cataracts were not present in this animal.
Follow-up in vitro investigative studies were not able to replicate vitreous opacities observed in vivo, and in vitro vitreous stability data suggested that the observed particulates were unlikely to arise from fragmentation of RabFab-NLP. During in life, vitreous opacity was observed on day 3 OE in all RabFab-NLP dosed eyes, which is the first timepoint following day 1 postdose assessment. The fact that the particulates were not observed on day 1 postdose OE may suggest that the cause of the opacity could potentially be related to aggregation of RabFab-NLP or interaction with vitreous components, which may require some time to form.
The toxicity observed in this study was somewhat unexpected given the reported safety profile of this nanoparticle delivery vehicle when administered systemically 11 and the fact that HDLs, a natural version of NLPs, are present at very high concentrations in the blood. 21,22 Therefore, this observed adverse event is likely due to the unique nature of the vitreous as well as the cellular composition of the lens. Unlike the blood, the vitreous contains much less HDLs 23 and it may be possible that NLPs may be less tolerated in the vitreous environment.
Several mechanisms have been proposed for the development of cataracts, including lipid-lens protein adducts, GSH depletion, and lipid peroxidation. 24- 27 It will be interesting to uncover if any of these mechanisms are also at play in NLP-induced cataracts. Given that there are several examples of drug-induced cataracts with similar mechanisms of action in rabbits and humans, 25,28,29 the findings from this study may have clinical relevance. Thus, the safety liabilities of NLPs have to be addressed prior to developing them as long-acting ocular delivery vehicles.
Footnotes
Acknowledgments
We would like to thank Julia Gray, Mandy Kwong, and Pamela Chan for support of TK and ADA assays.
Declaration of Conflicting Interests
The authors declared no real, perceived or potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
