Abstract
Development of intravitreal drugs presents several challenges due to the delicate ocular environment and volume constraints of what can be safely administered in the eye. Formulation development of intravitreally administered drugs may necessitate the use of nonphysiological pH in order to accommodate manufacturing processes or achieve favorable drug properties. Clinical and nonclinical data show that intravitreal drugs formulated in the pH 5.5 to 7.4 range are well tolerated. The aim of this study was to provide ocular toxicity data for formulations in the pH 4.0 to 5.5 range following intravitreal administration in New Zealand White rabbits. This range was evaluated as part of formulation development for an intravitreal drug that necessitated the use of pH outside the available tolerability data for formulations. Toxicity was assessed by ophthalmic examinations, intraocular pressure (IOP) measurement, clinical observations, body weights, and microscopic analysis of ocular tissue. Histidine chloride pH 5.0 to 5.5 and acetate chloride pH 4.0 to 5.0 solutions were well tolerated, and no test article-related ocular inflammation, IOP changes, or gross or microscopic findings were observed in any eye. The data presented here add to the knowledge of pH ranges that can be explored for intravitreal drug formulation development.
Introduction
Intravitreal administration remains the preferred route to achieve high drug concentrations in the posterior segment of the eye while maintaining low systemic exposures. The eye is a very sensitive organ, and maintenance of the delicate intraocular homeostasis is key to the preservation of visual function. Formulation development of intravitreally administered drugs presents several challenges due to physiological and anatomical constraints that are unique to the eye. Compounds and formulations that are suitable for systemic administration may not be well tolerated in the ocular environment. More importantly, as new drug candidates and ocular drug delivery technologies are developed, there is a need to increase our understanding of the tolerability of formulation conditions that are safe for intravitreal administration while maximizing favorable drug properties.
The current nonclinical and clinical data suggest that pH 5.5 to 7.4 is an acceptable range for intravitreally administered drugs. 1,2 The pH of the drug product is an important manufacturability parameter as it generally affects drug properties as well as shelf life. It is desirable to avoid extreme pH conditions and formulate drugs close to the physiological vitreous pH in order to limit the disruption of the ocular environment. 3 However, the specific properties of the drug, such as solubility and stability considerations, may necessitate formulating at nonphysiological pH conditions. Furthermore, new therapeutic modalities and delivery platforms for intravitreal administration may require formulations with pH outside the established tolerability range.
The aim of this study was to evaluate the tolerability of pH 4.0 to 5.5 solutions over a 9-day observation period after a single intravitreal administration in both eyes of New Zealand White rabbits. This pH range was evaluated as part of formulation development for an intravitreal drug that necessitated the use of pH outside the existing tolerability data for formulation. Two different buffer systems were used: acetate chloride buffer for pH 4.0 to 5.0 solutions and histidine chloride buffer for pH 5.0 and 5.5 solutions due to their ability to buffer their respective pH range. We included histidine chloride pH 5.5 buffer as a control since clinical tolerability data can be cross-referenced for this formulation. 2 Here we show that all buffers in the pH 4.0 to 5.5 range were well tolerated and did not cause inflammation, intraocular pressure (IOP) changes, or gross or microscopic findings in any eye.
Materials and Methods
Animals
The study was conducted at Absorption Systems, Inc (ASI). The ASI is accredited by the Association for Accreditation and Assessment of Laboratory Animal Care, International. During the study, the care and use of animals was conducted in accordance with the regulations of the US Department of Agriculture Animal Welfare Act (ie, relevant sections of Section 9, Parts 1, 2, and 3, of the Code of Federal Regulations) and in compliance with ASI’s Animal Welfare Assurance (A4282-01) filed with the National Institutes of Health. Treatment of the animals was in accordance with ASI SOPs and the conditions specified in the Guide for Care and Use of Laboratory Animals (NRC, 2011, National Academy Press)
Twelve male New Zealand White rabbits, approximately 18 weeks of age, weights ranging from 3.8 to 4.4 kg, were used in the study. Rabbits were housed individually in cages, within the same temperature and humidity-controlled room. The rabbits had ad libitum access to species-specific chow and water, with additional dietary enrichment provided.
Test Article Preparation
For all buffer systems, sodium chloride was included to achieve isotonicity in the range of 310 to 317 mOsm/kg of water and endotoxin was limited to ≤0.05 EU/mL. pH targets were confirmed using a calibrated pH meter. Solutions were sterile filtered under aseptic conditions and filled to deliver 1 dose (2× 50 µL) per sterile glass vial. Buffers were formulated as follows: pH 4.0, 20 mM acetate, 150 mM sodium chloride pH 4.5, 20 mM acetate, 150 mM sodium chloride pH 5.0, 20 mM acetate, 150 mM sodium chloride pH 5.0, 20 mM histidine hydrochloride, 150 mM sodium chloride pH 5.5, 20 mM histidine hydrochloride, 150 mM sodium chloride
Intravitreal Dose Administration
Rabbit eyes (groups 1-5) were dosed on Day 1 with test articles, as indicated in Table 1. Prior to intravitreal injection, animals were anesthetized with an intramuscular injection of ketamine hydrochloride (20 mg/kg) and xylazine (5 mg/kg). Test article vials were brought to room temperature prior to syringe filling inside a biosafety hood under sterile conditions. Doses were administered intravitreally in both eyes of each animal as 2× 50 µL injections, 10 to 20 minutes apart, into the inferior temporal region.
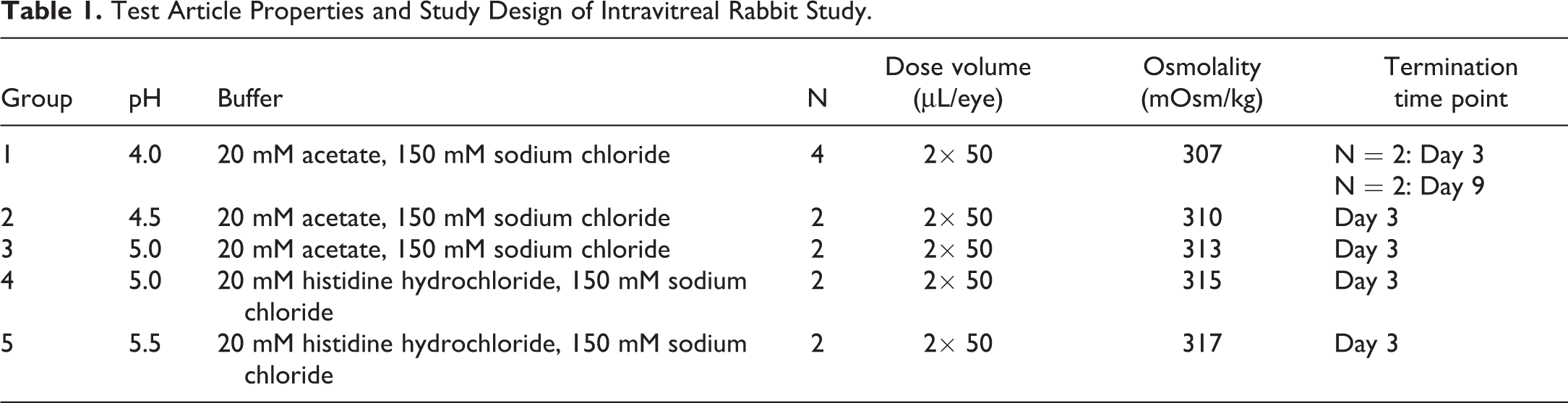
Test Article Properties and Study Design of Intravitreal Rabbit Study.
The dosing procedure was as follows. The eyelids and skin surrounding the eye were cleaned with 5% Betadine solution and rinsed with balanced salt solution, followed by application of 1 to 2 drops of topical proparacaine hydrochloride anesthetic (0.5%). The eye was draped, and a sterile wire speculum was placed to retract the eyelids. Injections were made 3 to 4 mm away from the limbus using a 0.3-mL insulin syringe with a ⅝″ 31-G needle. Injections in the right eye were alternated such that the first injection was made approximately at the 7-o’clock position and the second injection approximately at the 8-o’clock position. Injections in the left eye were alternated such that the first one was made approximately at the 4-o’clock position and the second one approximately at the 5-o’clock position. Intraocular pressure was measured 10 minutes after the first injection as described below. The second injection was administered after IOP had returned to the approximate baseline value. In cases where the value had not returned to baseline, IOP was measured again after 5 additional minutes, at which time IOP had returned to the approximate baseline value and the second injection of the vehicle was administered.
Ophthalmic Examinations and IOP Measurement
Ophthalmic examination (OE) and IOP evaluations were conducted for both eyes of each animal at baseline (during week −1), on Day 1, Day 3 (n = 2 group 1 animals and all group 2-5 animals), and Day 9 (n = 2 group 1 animals). Day 1 examinations were performed after completion of dosing. Day 3 and Day 9 examinations were performed prior to euthanasia of the respective animals.
Eyes were examined with a slit-lamp biomicroscope and indirect ophthalmoscope. Prior to examination with the indirect ophthalmoscope, pupils were dilated with topical application of 1 drop each of 10% phenylephrine and 1% tropicamide on each cornea. The adnexa and anterior portion of both eyes were examined using a slit-lamp biomicroscope. The fundus of both eyes was examined using an indirect ophthalmoscope. Ocular findings were scored according to a modified McDonald-Shadduck scoring system. 4
Intraocular pressure measurements were performed using a pneumatometerfor both eyes of each animal in conjunction with OEs. The IOP measurements were performed on conscious animals with the exception of Day 1, when animals were under anesthesia for dosing procedures. Day 1 IOP measurements were performed prior to the first injection, prior to the second injection, and after completion of dosing. Day 3 and Day 9 measurements were performed prior to euthanasia of the respective animals.
In-Life Observations
General health observations, body weight records, and detailed clinical observations were made for each animal throughout the duration of the study. Each animal was observed for changes in general appearance and behavior, and any abnormal observations were reported.
Tissue Collection and Histopathology
Animals were euthanized on Day 3 or Day 9 via pentobarbital overdose (150 mg/kg, intravenously) followed by exsanguination. The euthanasia procedure was performed in compliance with the American Veterinary Medical Association Guidelines on Euthanasia. Immediately following euthanasia, the eyes (whole globes and optic nerves) were collected from all animals and placed into modified Davidson fixative at room temperature. After approximately 24 hours in modified Davidson fixative, tissues were transferred into 10% neutral-buffered formalin. Each eye was sectioned into 3 calottes (nasal, central, and temporal) and each calotte was routinely processed and embedded in paraffin. Three sections of each calotte, 100 µm apart, were cut, mounted on glass slides, and stained with hematoxylin and eosin. The resulting tissue sections (9 per eye) were examined by a board-certified veterinary pathologist (S.T.L.).
Results
Tolerability assessments were made in all group 1 to 5 animals up to Day 3 and in 2 group 1 animals up to day 9 (pH 4.0, acetate chloride). All animals on the study survived until their scheduled termination time point. During the in-life phase, there were no test article-related clinical observations or changes in body weight and food consumption. Conjunctival swelling and conjunctival hyperemia were noted in clinical observations for most animals up to Day 3. These findings were mild to moderate and were considered related to the procedure. No abnormal health findings were noted past day 3 in the animals that survived beyond this time point.
No test article-related effects were observed on ophthalmic evaluations throughout the 9-day period, and all observed changes were consistent with the injection procedure. During Day 1 ophthalmic evaluations, mild conjunctival swelling was noted in all eyes, except 1 animal that exhibited no swelling and another animal that had swelling only in the right eye, both in group 1 (pH 4.0). Other observations included mild focal hyperemia at the injection sites of both eyes of 1 group 1 animal (pH 4.0) and increased retinal vascularization of both eyes of 1 group 1 (pH 4.0) and 1 group 2 animal (pH 4.5). Day 3 OEs revealed mild conjunctival swelling and focal hyperemia at injection sites in 1 or both eyes of at least 1 animal from each group 1 to 5. These findings were mild in all cases, except in 1 group 2 (pH 4.5) animal that exhibited moderate focal hyperemia. Day 9 OEs of the remaining 2 group 1 (pH 4.0) animals showed increased retinal vascularization near injection sites in both eyes and mild retinal hemorrhages in 1 eye of each animal. Overall, no test article-related effects on clinical OE were observed. Mild, transient symptoms of ocular irritation (conjunctival swelling, hyperemia, and/or discharge) during the initial days of the study, as well as focal, mild abnormalities (increased retinal vascularization and/or retinal hemorrhage) at or near some of the injection sites, were consistent with injection procedure-related changes.
All eyes had IOP in the normal physiological range throughout the study, and none of the test articles caused a sustained IOP increase other than the expected transient spike right after dosing (Figure 1). The IOP measurements were conducted after the first injection to ensure that IOP had returned to normal physiological range before administering the second injection. The IOP spike was transient, and all eyes were able to receive the second injection. The IOP values on Day 3 and Day 9 were slightly lower than baseline and Day 1 values in most animals. This difference was minimal in most cases but pronounced in group 3 (pH 5.0) animals. This pattern may have been due to variation in circadian effects, as baseline and Day 1 IOP measurements in these animals were performed in the afternoon, while Day 3 measurements were performed in the later morning. 5
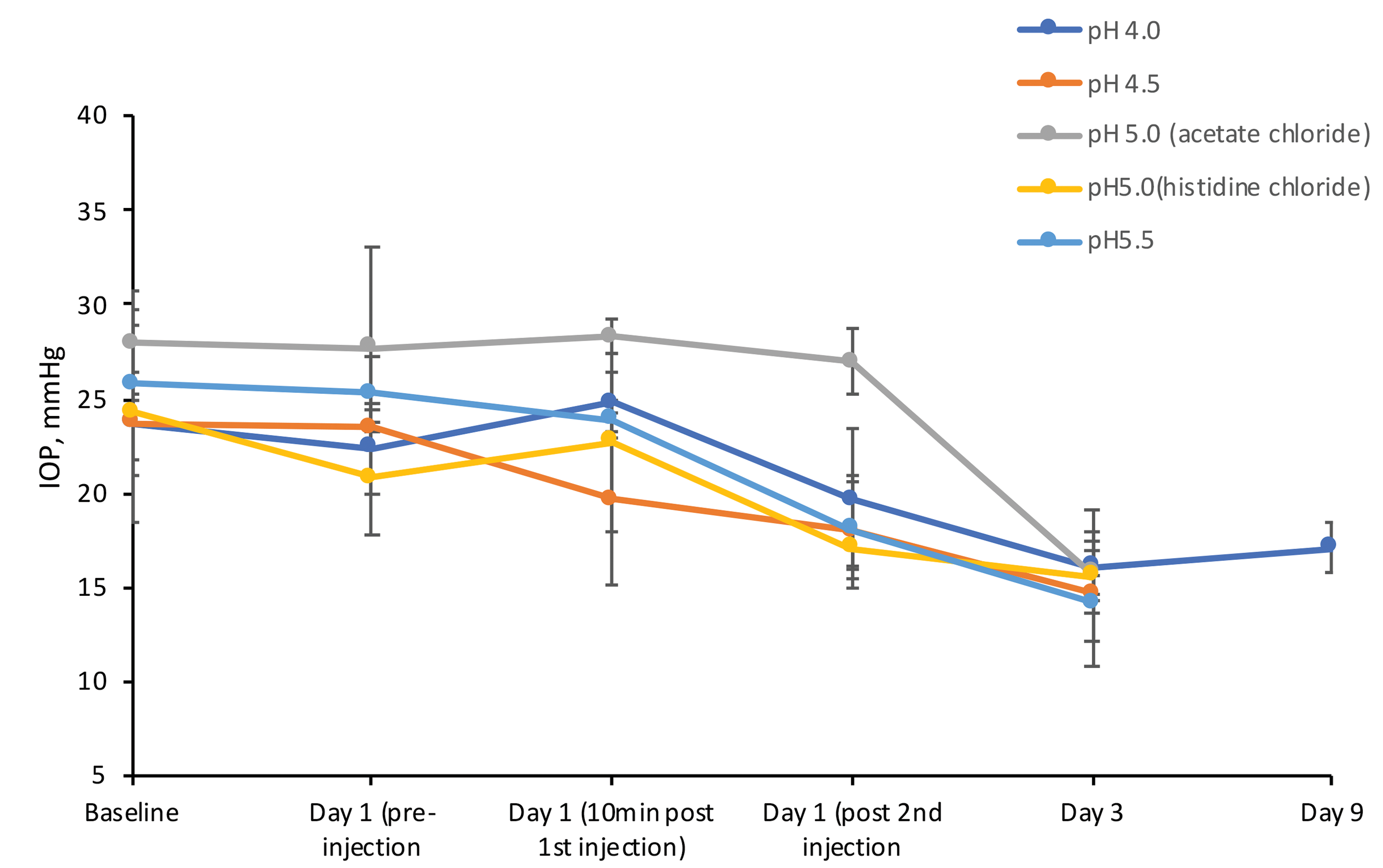
Mean intraocular pressure (IOP) values. In all animals in all groups, IOP values remained within the normal physiological range, and no systematic effects of acetate chloride pH 4.0, 4.5, or 5.0 or histidine chloride pH 5.0 or 5.5 solutions on IOP values were observed for the duration of the study. Group 1 (pH 4.0): n = 4 (n = 2/time point) and groups 2 to 5: n = 2/group.
Upon microscopic examination ocular tissue, no test-article related findings were observed in any eye (Figure 2). Minimal conjunctival, and in one animal administered pH 4.5 acetate chloride buffer, corneal inflammation were considered to be related to the injection and/or OE procedures. All other findings were incidental/background findings commonly noted in the eyes of New Zealand White rabbits and not related to test article administration.
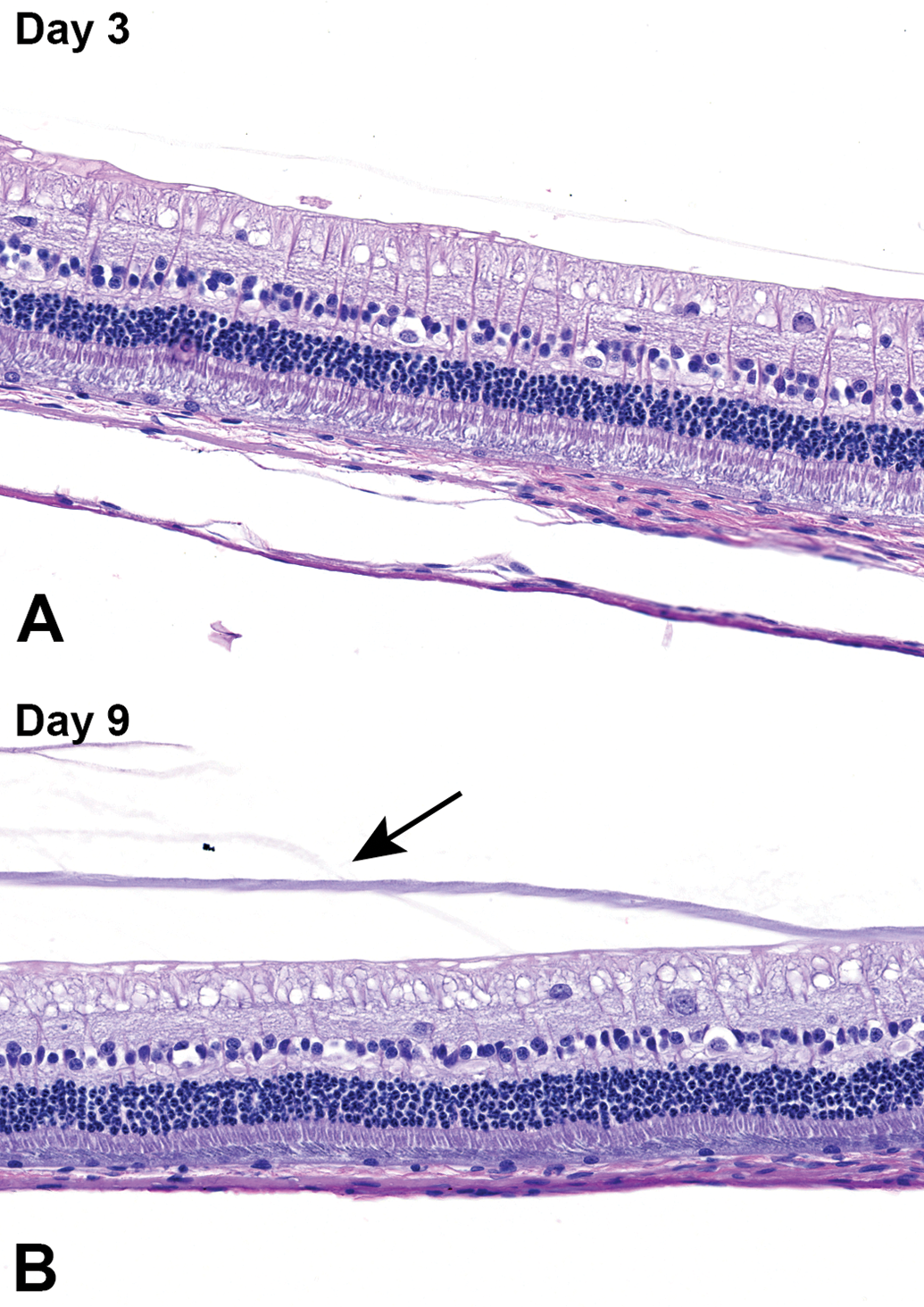
Retina and choroid from New Zealand White rabbit eyes administered acetate chloride solution, pH 4.0 (group 1), Day 3 (A) and Day 9 (B) postdose. Eyes were histologically normal at both time points. In (B), condensed vitreous proteins, a normal finding, are present as an amphophilic band in the vitreous (arrow). Hematoxylin and eosin.
Discussion
Intravitreal administration remains the preferred route to treat diseases that affect the posterior segment of the eye such as age-related macular degeneration. As new drugs and modalities are developed, more data are needed to inform the tolerability of different formulation conditions for intravitreal administration. In this study, we show that solutions formulated with acetate chloride or histidine chloride buffers in the pH 4.0 to 5.5 range were well tolerated and did not cause any inflammatory reaction, IOP changes, or any gross or microscopic findings in the rabbit eye following a single 2× 50 µL intravitreal administration. All parameters other than pH (NaCl concentration, osmolality) were kept the same in all the test articles in order to tease out the effect of pH on ocular tolerability.
Rabbits have been valuable for characterizing the preclinical safety profile of various intravitreally administered drugs such as corticosteroids, antibiotics, antiviral, and antifungal agents. 6 –9 They are also commonly used to assess direct retinal toxicities that may be associated with formulation pH and excipients. 10 –13 In various studies, direct retinal toxicities arising from intravitreal formulations are commonly observed in rabbit eyes as early as 3 days. 10 –13 In this study, all eyes were observed up to 3 days and evaluation was extended to 9 days for 2 animals that were administered the test article with the lowest pH (pH 4.0) to capture any delayed effects as this solution is expected to shift vitreal pH the most among those evaluated in this study. No test article-related gross or microscopic findings were observed in any eye at Day 3 or Day 9.
The physiological vitreal pH of New Zealand White rabbits ranges from 7.23 to 7.37 depending on the specific region of the vitreous. 3 In vivo vitreous pH measurements in rabbits with a probe inserted in 7 different regions of the vitreous cavity show that there are significant regional differences. 3 The area near the lens had the lowest pH (pH 7.23), and the superior portion of the vitreous had the highest pH (pH 7.37). 3 Measurement of pH on vitreous samples obtained under anaerobic conditions from patients undergoing vitreous surgery suggests that human vitreal pH ranges from 7.20 to 7.23. 14 It is interesting to note that vitreal pH remained constant between patients with or without diabetes mellitus even though an increase in ocular lactic acid level was observed in the former, indicating that the vitreous is able to resist pH changes. 14 Various studies show that the vitreous has the capacity to buffer pH changes and is able to equilibrate rapidly following intravitreally administered solutions 2,15 ; however, to what extent it can buffer extreme pH formulations remains unknown. Ex vivo studies using animal and human vitreous have also shown that the vitreous is able to buffer acidic and basic solutions. 2,15 In a study that evaluated the buffering capacity of ex vivo human vitreous to rituximab (pH 6.29) and 4 anti–vascular endothelial growth factor (VEGF) agents (ranibizumab, pH 5.5; bevacizumab, pH 5.91; aflibercept, pH 6.05; ziv-aflibercept, pH 6.1), it was shown that vitreal pH changes occurred only within a small range. 2 This study also demonstrated that the buffering capacity of the administered solution itself is an additional factor that determines the extent of vitreal pH changes. Drugs formulated in weak buffers had less pronounced impact on vitreal pH changes compared to strong buffers due to resisting pH change. 2
Formulation pH is one parameter that impacts ocular tolerability for intravitreally administered drugs. Ocular tolerability of various anti-VEGF and anti-inflammatory drugs has been established for pH ranges of 5.5 to 7.4. 1,2 However, more work remains to be done to establish the tolerable ranges of pH, osmolality, ionic strength, salt/sugar, excipients, and so on, that are suitable for formulating intravitreal drugs. The data generated in our study add to this knowledge and inform drug developers on the ocular tolerability of formulations within the pH 4.0 to 5.5 range. In addition, the suitability of acetate chloride buffer for intravitreal administration was also evaluated and here the data show that it was well tolerated and did not cause notable toxicities.
Footnotes
Acknowledgments
The authors thank Glenwood Glum, Stephanie Smith, Sandeep Kumar, and Thomas Patapoff for their technical assistance with study design and conduct.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
