Abstract
Rats repeatedly exposed to high airborne concentrations of ethylene develop eosinophilic rhinitis and mucous cell hyperplasia/hypertrophy (MCH) in nasal respiratory epithelium. Mechanisms underlying these lesions are not well understood to inform occupational exposure guidelines. In this study, we determined (1) the nasal histopathology in rats episodically exposed to ethylene, (2) the ethylene-induced nasal histopathology in similarly exposed mice, and (3) how innate lymphoid cells (ILCs) play a role in ethylene-induced MCH. Animals were exposed to 0 or 10,000 ppm ethylene, 6 h/d, 5 d/wk, for 2 weeks and sacrificed 1 day or 2 weeks postexposure. Others received three 2-week exposure blocks separated by 2-week intervals of no exposure. Episodic exposure was chosen to aid in distinguishing irritant from immune responses. Mucous cell hyperplasia/hypertrophy was induced by ethylene in both species. Rats developed a mild, but transient, eosinophilic rhinitis. Mucous cell hyperplasia/hypertrophy was transient in mice, but persistent in rats. Increases in epithelial mucosubstances after 2 weeks of exposure were only present in ILC-sufficient mice, but not in ILC-deficient mice suggesting that ILCs play a role in MCH and overexpression of genes associated with mucus production/secretion. These findings in animals suggest that inhaled ethylene does not act as a sensitizing agent and will not induce allergen-like nasal airway disease.
Keywords
Introduction
Ethylene is a colorless, volatile, unsaturated hydrocarbon
In a subchronic whole-body inhalation study, Sprague-Dawley rats were exposed to 0, 300, 1000, 3000, or 10,000 ppm ethylene, 6 h/d, 5 d/wk for 14 weeks. 5 In this study, there was no exposure-related mortality or evidence of systemic toxicity. In a later chronic whole-body inhalation/carcinogenicity study, F344/DuCrl rats were exposed 6 h/d, 5 d/wk for 106 weeks to 0, 300, 1000, or 3000 ppm ethylene. No chronic toxicity or carcinogenic effects were observed, 6 and the no-observed-adverse-effect level was determined to be 3000 ppm.
In 2005, the American Conference of Governmental Industrial Hygienists recommended an 8-hour, time-weighted average threshold limit value of 200 ppm for airborne ethylene, based on all the available data at that time. 7
More recently, we have reported that repeated inhalation exposures of F344/DuCrl rats to ethylene (6 h/d, 5 d/wk for up to 4 weeks; continuous exposure) caused a dose- and time-dependent mucous cell hyperplasia/hypertrophy (MCH) in nasal respiratory epithelium along with a focal eosinophilic rhinitis, both of which were associated with type 2 immunity.
8
There was a significant increase in MCH after 5 days of exposure to 10,000 ppm ethylene and after 20 days of exposure to 10 ppm ethylene. Ethylene-induced MCH was, however, reversible after cessation of exposure. In addition, nasal histopathology induced by inhaled ethylene was concurrent with enhanced gene expression for type 2 inflammatory/immune cytokines (eg,
Enhancement of type 2 inflammatory cytokines (eg,
Laboratory findings that rats repeatedly exposed to high concentrations of ethylene develop eosinophilic rhinitis and MCH in nasal respiratory epithelium (both key pathologic features of allergic rhinitis and asthma), raises safety concerns about inhalation exposure in the workplace and the development or exacerbation of allergic (or nonallergic) airway disease. Cellular and molecular mechanisms underlying ethylene-induced nasal lesions in rodents are not well understood. Further elucidation of the pathogenesis of these exposure-induced upper airway alterations may impact occupational safety protocols for this high production volume organic chemical.
In the present study, we further explored the pathogenesis of ethylene-induced nasal pathology in both rats and mice. Our specific aims were to determine (1) the progression and persistence of inflammatory and epithelial lesions in the nasal airways of rats that were episodically, rather than continuously, exposed to ethylene, (2) the nasal pathology of mice repeatedly exposed to ethylene, and (3) the roles Th2s and ILCs play in the pathogenesis of ethylene-induced MCH. Episodic exposure was chosen to aid in distinguishing irritant from immune responses. We hypothesized that (1) eosinophilic rhinitis and increased epithelial mucosubstances in rats progress with episodic exposures to ethylene and persist postexposure, (2) mice have similar ethylene-induced nasal lesions as rats, and (3) ILCs (particularly group 2 ILCs—ILC2s), but not Th2s, play a role in the pathogenesis of ethylene-induced nasal lesions.
Material and Methods
Animals
Male F344/DuCrl rats were obtained from Charles River Laboratories, Inc, at the age of 8 weeks. Male C57BL/6 mice, 6 to 8 weeks of age on arrival, male Rag2−/− mice, 6 to 8 weeks on arrival, and male Rag2−/−/IL2rg −/− mice, 5 to 7 weeks on arrival, were purchased from Taconic. Rats and mice were housed in solid floor stainless steel cages with corncob bedding and shredded aspen for enrichment and were provided LabDiet Certified Rodent Diet #5002 (PMI Nutrition International) and water ad libitum except during inhalation exposures. This experiment was reviewed and approved by a veterinarian and the Institutional Animal Care and Use Committee. The laboratory is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Controlled Inhalation Exposures
Rats and mice, 6 to 9 weeks of age, were exposed to filtered air (control) or 10,000 ppm ethylene gas in 4 m 3 stainless steel and glass Rochester-style whole-body inhalation exposure chambers. Chamber airflow was maintained at approximately 900 L/min, which was sufficient to provide 12 to 15 air changes per hour. The ethylene exposure chambers were operated at a slight negative pressure relative to the surrounding area, whereas the control (0 ppm) chambers were operated at a slight positive pressure to minimize the potential for accidental exposure of control animals to ethylene. Both rats and mice were singly housed to minimize crowding during the exposure.
Chamber airflow data were collected using Setra Differential Pressure Transducers (Setra Systems, Inc). The differential pressure transducers were calibrated with a gas meter (Singer Aluminum Diaphragm Meter, Model AL-2300, American Meter Division) prior to the start of the study. Chamber temperature and relative humidity were measured with a resistance temperature device and a humidity sensor (Omega Engineering Inc), respectively, approximately once each hour. The temperature and relative humidity in all chambers maintained at was maintained at approximately 22 °C ± 2 °C and 40% to 60%, respectively, throughout the study. Airflow, temperature, relative humidity, and ethylene concentration were monitored by a CAMILE TG Data Acquisition and Control System (Biotage AB), displayed, logged once per hour, and printed for inclusion in the study file.
Ethylene exposure atmospheres were generated by mixing ethylene gas with high efficiency particle air (HEPA)-filtered breathing air as it entered the whole-body exposure chambers. Target chamber concentrations were maintained by adjusting the amount of ethylene gas delivered to each chamber using calibrated mass-flow controllers.
Prior to exposure of animals to ethylene gas, the homogeneity of ethylene within the breathing zone of the animals was determined. In addition, the position of each animal’s exposure cage was altered daily to ensure uniform exposure of all animals in the chamber.
A nominal concentration for the exposure chambers was calculated from the mass of ethylene passing through the mass flow controller for each exposure chamber divided by the total chamber airflow.
Ethylene oxide is a volatile reactive metabolite of inhaled ethylene. Each chamber atmosphere was analyzed for the presence of ethylene oxide once during exposure to determine whether toxicologically significant levels of ethylene oxide were present during ethylene exposures. No ethylene oxide was detected in any chamber at or above the lowest level quantified of 10 ppm. Sample analysis was performed by gas chromatography (GC) with flame ionization detection on samples from all of the ethylene control and exposure chambers. Samples were withdrawn from each chamber by gastight syringe and transferred to 10-L tedlar bags. Aliquots were then withdrawn by gastight syringe and manually injected into the GC injection port. The GC was calibrated with known ethylene oxide gas standards ranging in concentration from 10 to 50 ppm.
Study Design and Inhalation Exposures
Male F344/DuCrl rats, C57BL/6 (wild-type) mice, and lymphoid cell deficient Rag2−/− (single knockout [KO]) and Rag2−/−Il2rg−/− (double KO) mice were contemporaneously exposed to 0 or 10,000 ppm in whole-body inhalation exposure chambers. The Rag2−/−Il2rg−/− (double KO) mouse is a cross of the common cytokine receptor gamma chain (Il2rg) KO mice with Rag2−/− (recombinase activating gene 2) KO mice. The resultant double KO mice lack T cells, B cells, NK cells, and ILCs. The Rag2−/− (single KO) mice contain a disruption of the recombinase activating gene 2 leading to a total inability to initiate V(D)J rearrangement and a failure to generate mature T or B lymphocytes; however, they do have a functional complement of ILCs, including type 2 ILCs (ILC2). Therefore, if ethylene-induced nasal lesions were observed in wild-type C57BL/6 mice but not in the Rag2−/− (single KO) mice that would provide evidence that B and T lymphoid cells were involved in the development of the ethylene-induced nasal pathology. If nasal lesions were present in ethylene-exposed Rag2−/− (single KO) mice but not in the Rag2−/−/IL2rg−/− (double KO) mice this will provide evidence that the ethylene-induced nasal lesion is mediated by ILCs.
Mice and rats were exposed 6 h/d, 5 d/wk for 2 weeks (10 exposure days) followed by 2 weeks postexposure (
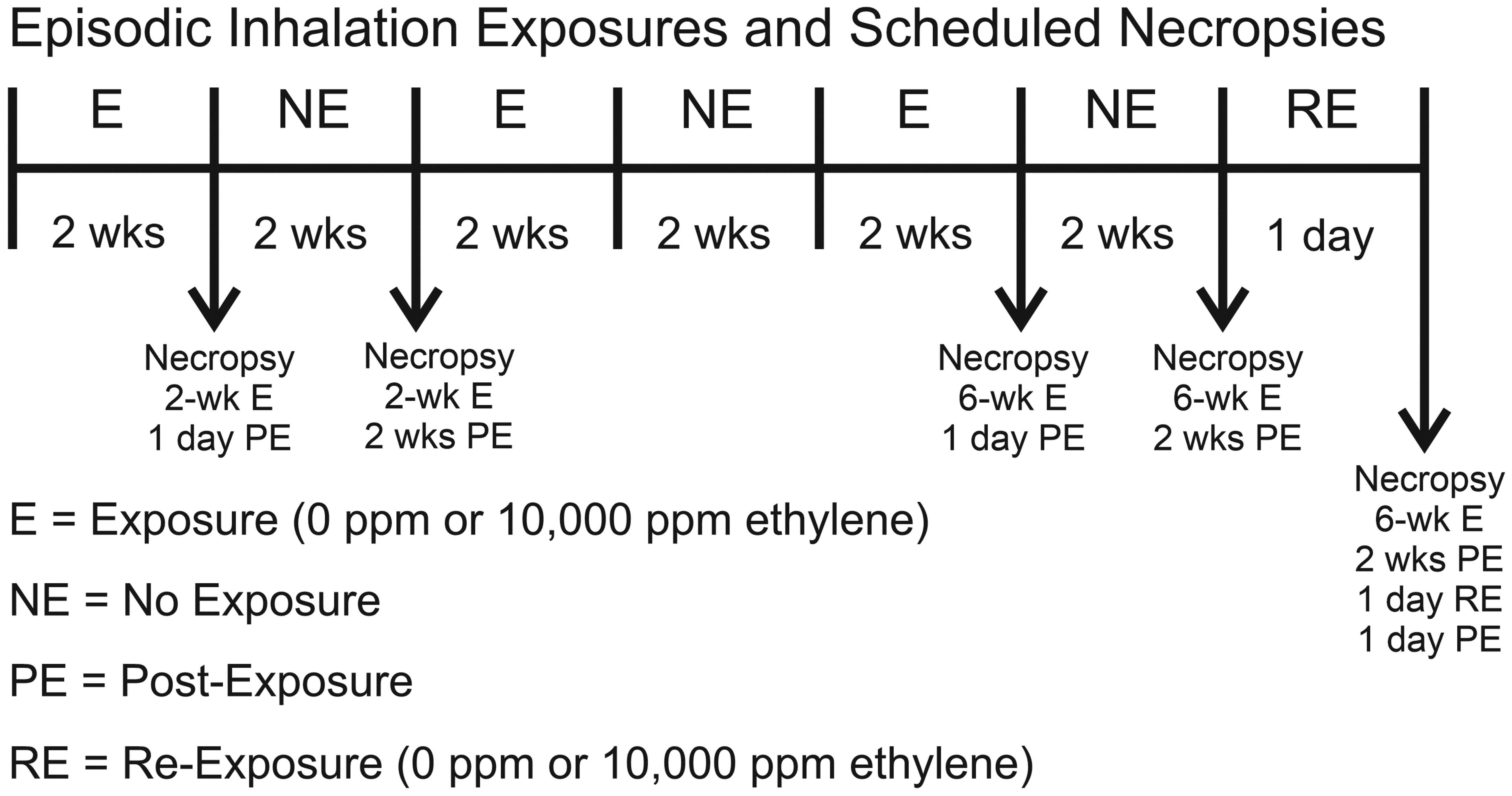
Episodic inhalation exposure regimen and scheduled necropsies. Rats and mice were contemporaneously exposed to 0 or 10,000 ppm in whole-body inhalation exposure chambers for 2 weeks (6 h/d, 5 d/wk) or three 2-week exposures (E) separated by 2-week no exposure intervals (NE). Animals were necropsied 1 day or 2 weeks postexposure (PE). Some 6-week E/2-week PE animals were necropsied 1 day after a 1 day reexposure (RE).
Animal Necropsies and Nasal Tissue Selection and Processing
Two-week exposure and 6-week episodic exposure groups of rats and mice were sacrificed 1 day or 2 weeks postexposure. Rats and mice in the 6-week episodic exposure/2-week postexposure/1-day reexposure groups were sacrificed 1 day after the reexposure. At necropsy, animals were anesthetized by inhalation of isoflurane/O2 and euthanized by exsanguination via the abdominal aorta.
Heads from all rats and mice designated for light microscopic examination were removed from the body at necropsy, fixed in neutral buffered formalin and decalcified using 13% formic acid, as previously described in detail. 18 After decalcification, 4 transverse nasal tissue blocks were taken from specific sites, defined by dental and palatine landmarks, of the nasal cavity (proximal T1 – distal T4; Figure 2) by methods previously reported. 21,22 Tissue blocks were embedded in paraffin and sectioned at a thickness of 5 µm. Tissue sections were stained with either Alcian blue (pH 2.5)/periodic acid-Schiff (AB/PAS) to detect acidic and neutral mucosubstances in nasal airway epithelium or hematoxylin and eosin for routine histopathology. Additional nasal sections were immunohistochemically stained with an antibody specific for rat or mouse major basic protein (MBP; mice, clone MT-14.7, Mayo Clinic; rats, clone BMK-13, BioRad) to identify eosinophils infiltrating the nasal mucosa. 19 To ensure consistency of histochemical AB/PAS staining among tissue sections prior to morphometric analysis (described below), all nasal tissue sections within each study were stained as a single batch. Rodent intestinal tissue was used as a positive control for epithelial mucosubstance staining. The pH of AB stock solution was measured to ensure a pH of 2.5 prior to batch staining. Quality control of tissue staining was performed by a certified histotechnologist who restained any individual slides that were deemed improperly stained.
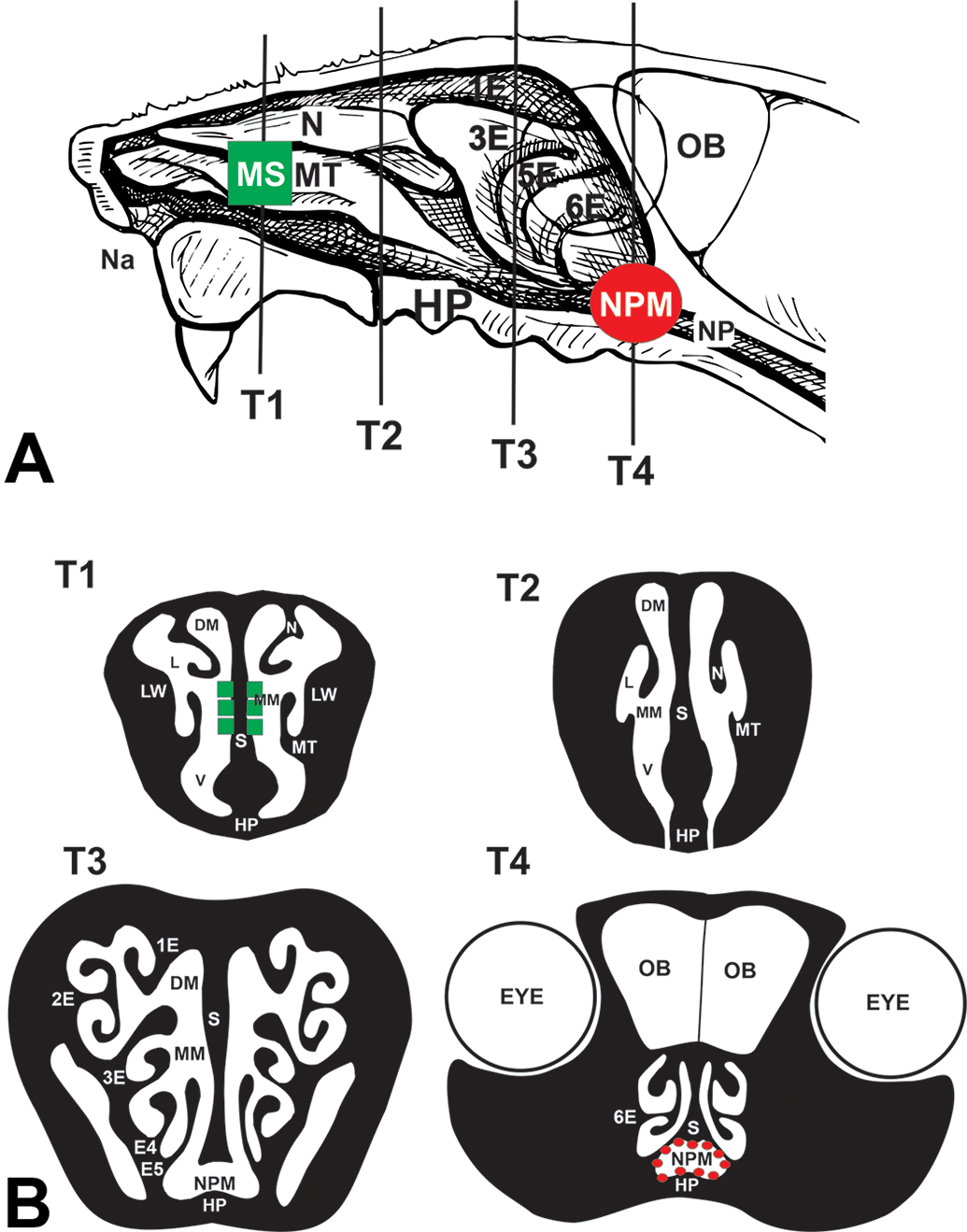
Diagrammatic representation of the rodent nose with species-specific sites of nasal epithelial lesions in ethylene exposed rats and mice. A, Right nasal passage with the septum removed and exposing the nasoturbinate (N), maxilloturbinate (MT), and ethmoid turbinates (1-E, 3-E, 5-E, 6-E) and (B) transverse sections of the nasal cavity (T1-T4; proximal to distal tissue sections containing both nasal passages) that were microscopically examined by light microscopy and morphometrically analyzed. Principal sites of ethylene-induced mucous cell hyperplasia/hypertrophy were in the respiratory epithelium lining the proximal mid-septum (MS; highlighted green squares) of mice but not rats, and in the distal nasopharyngeal meatus (NPM, highlighted with red spheres) of rats but not mice. DM indicates dorsal medial meatus; HP, hard palate; L, lateral meatus; LW, lateral wall; MM, medial meatus; Na, naris; OB, olfactory bulb; S, septum; v, ventral meatus; ethmoid turbinates (1E-6E).
Nasal Morphometry
Glass slides of nasal tissue sections were digitized with a slide scanner (VS110, Olympus America). For quantification of AB/PAS-positive, intraepithelial mucosubstances, digital images of the respiratory nasal epthelium lining the proximal mid-septum (MS) in both nasal passages (T1 section) and the distal nasopharyngeal meatus (NPM; T4 section) were evaluated via morphometric methods using the newCAST software (VisioPharm) that has been previously described in detail.
8
,
19
For quantification of AB/PAS-positive, intraepithelial mucosubstances, the entire respiratory epithelium lining the MS in the T1 section and the NPM in the T4 section were selected and captured at ×400 magnification (one tissue section per animal). A point intercept grid was placed over the sampled images to estimate density of mucosubstances per basal lamina. The number of points hitting AB/PAS-positive mucosubstances (Pm) was counted. The density of AB/PAS-positive mucosubstances
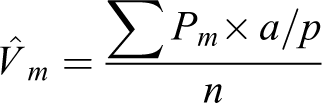
The surface density of the basal lamina
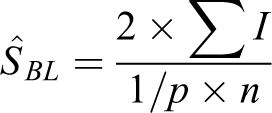
The positive density per basal lamina of the respiratory epithelium was then estimated by dividing
In addition, morphometry was used to determine the density of MBP-laden eosinophils in the mucosa lining the proximal lateral wall (T1 section), by methods previously reported in detail. 17,18 Briefly, the number of points hitting areas positive for MBP antibody was counted with a point grid in the sampled images of each mouse. The number of points on the reference space (ie, mucosal surface epithelium and lamina propria) was also counted with a point grid. The total number of points was multiplied by the area/point (a/p) for the eosinophil or reference space to calculate each density. Finally, the percentage of eosinophil density per the reference space was calculated and expressed as the percentage of eosinophilic influx in the mucosal tissue.
Gene Expression Analysis
An additional group of rats exposed episodically for 6 weeks to ethylene rats were designated for analysis of specific gene transcripts (messenger RNA [mRNA] expression; reverse transcription–polymerase chain reaction (RT-PCR) group; n = 6/strain/concentration/sacrifice) in nasal mucosal tissue (Table 1). At the time of necropsy, the entire nasal cavity from these rats designated for mRNA analyses was preserved in RNAlater (Qiagen) and then stored at −20 °C until nasal mucosal tissues lining the NPM and proximal nasopharynx of each rat were microdissected and excised from the nasal cavity for RNA extraction. Total RNA from the nasopharynx was extracted using the RNeasy Plus Mini Kit according to the manufacturer’s instructions (Qiagen). Eluted RNA was quantified using a Nanodrop 1000 Spectrophotometer (Nanodrop Technologies). Reverse transcription reactions were conducted using High Capacity cDNA Reverse Transcription Kit reagents (Applied Biosciences) and a GeneAmp PCR System 9700 Thermocycler PE (Applied Biosystems) according to the manufacturer’s protocol.
Ethylene-Induced Gene Expression in the Mucosa Lining the Nasopharyngeal Meatus of Rats Relative to 0 ppm Air Controls.
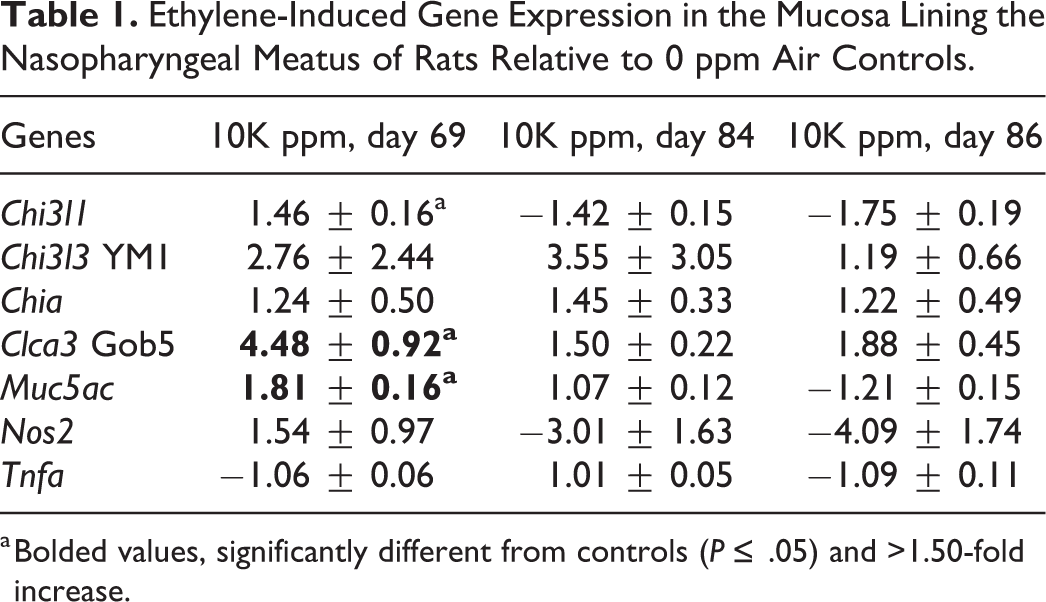
a Bolded values, significantly different from controls (
Quantitative gene expression analyses were performed at Michigan State University’s Genomic Technology Support Facility using Taqman Gene Expression Assay reagents and Taqman Gene expression master mix (Applied Biosystems) on the Wafergen SmartChip Real-Time PCR System (Takara Bio USA). The cycling parameters were 95 °C for 10 minutes, and 40 cycles of 95 °C for 10 seconds followed by 60 °C for 1 minute. Gene expression levels were reported as fold-change of mRNA in tissues from ethylene-exposed rats compared to control tissues from rats exposed to filtered air. Real-time PCR amplifications were relatively quantified using the ΔΔCt method,
23
by normalizing the Ct of target gene to its endogenous reference (
Statistics
All morphometric and gene expression data were analyzed with SigmaStat 11.0 (Jandel Scientific) with the criterion for significance set at
Results
Rats Episodically Exposed to Ethylene
Nasal histopathology and morphometry
No exposure-related histopathology (inflammatory or epithelial) was present in the nasal airways (T1-T4 sections) of filtered air-exposed (FA) control rats (0 ppm ethylene) at any examined time point. In contrast, rats exposed to ethylene for a single 2-week exposure block had MCH only in the respiratory epithelium (RE) lining the NPM (T4 section; Figures 3 and 4A and B). Figure 2 illustrates the intranasal location of this ethylene-induced change in RE of the distal nasal airway.
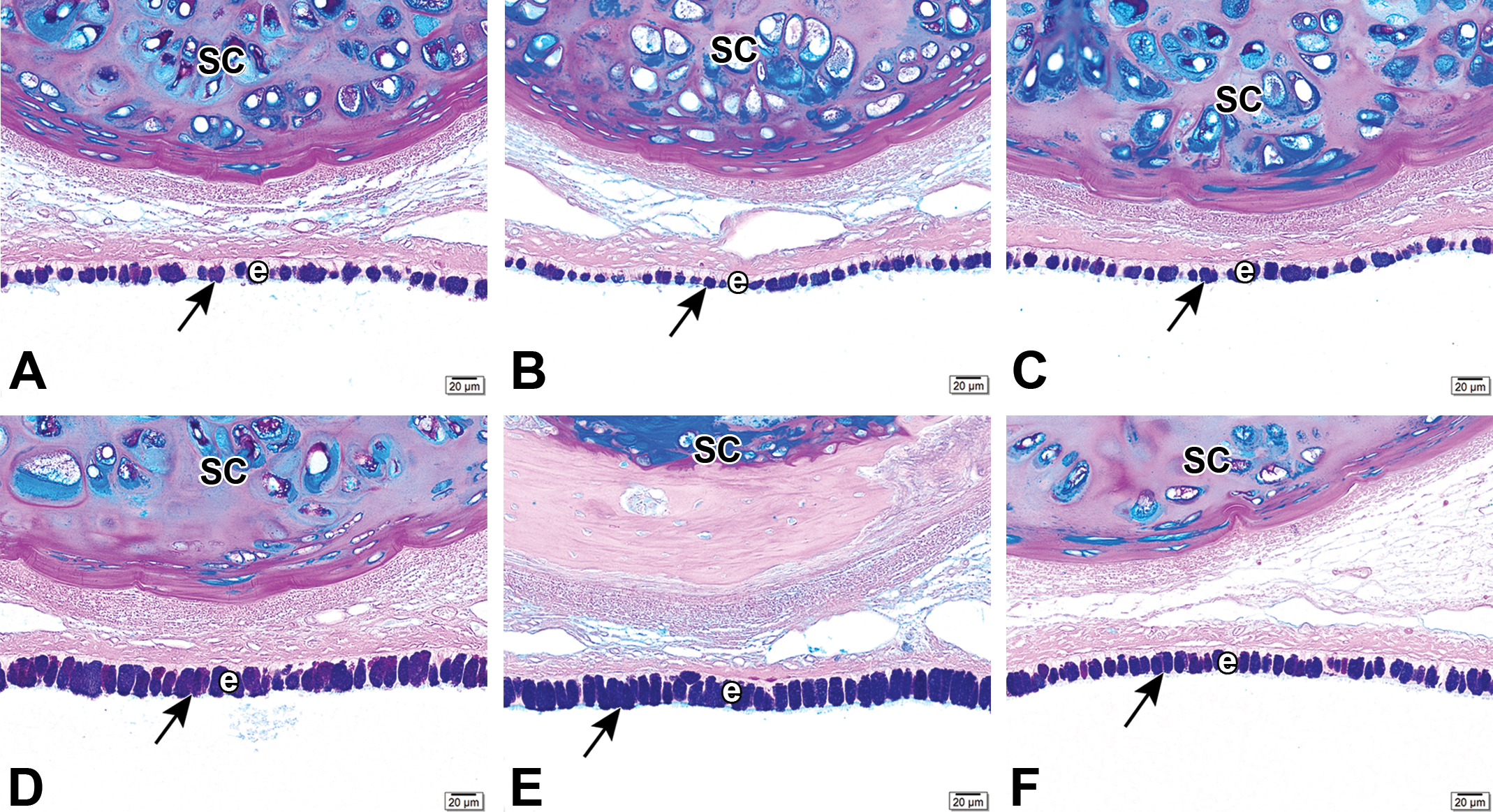
Light photomicrographs of Alcian blue/periodic acid-Schiff (AB/PAS)–stained mucosubstances (arrows) in the nasal respiratory epithelium (e) lining the nasopharyngeal meatus (NPM; T4 section) of rats after 2-week exposure (A, D) and 6-week episodic exposures (B, C, E, F) to air (A, B, C) or ethylene (D, E, F). Rats were examined 1 day (A, B, D, E) or 2 weeks postexposure (C, D).
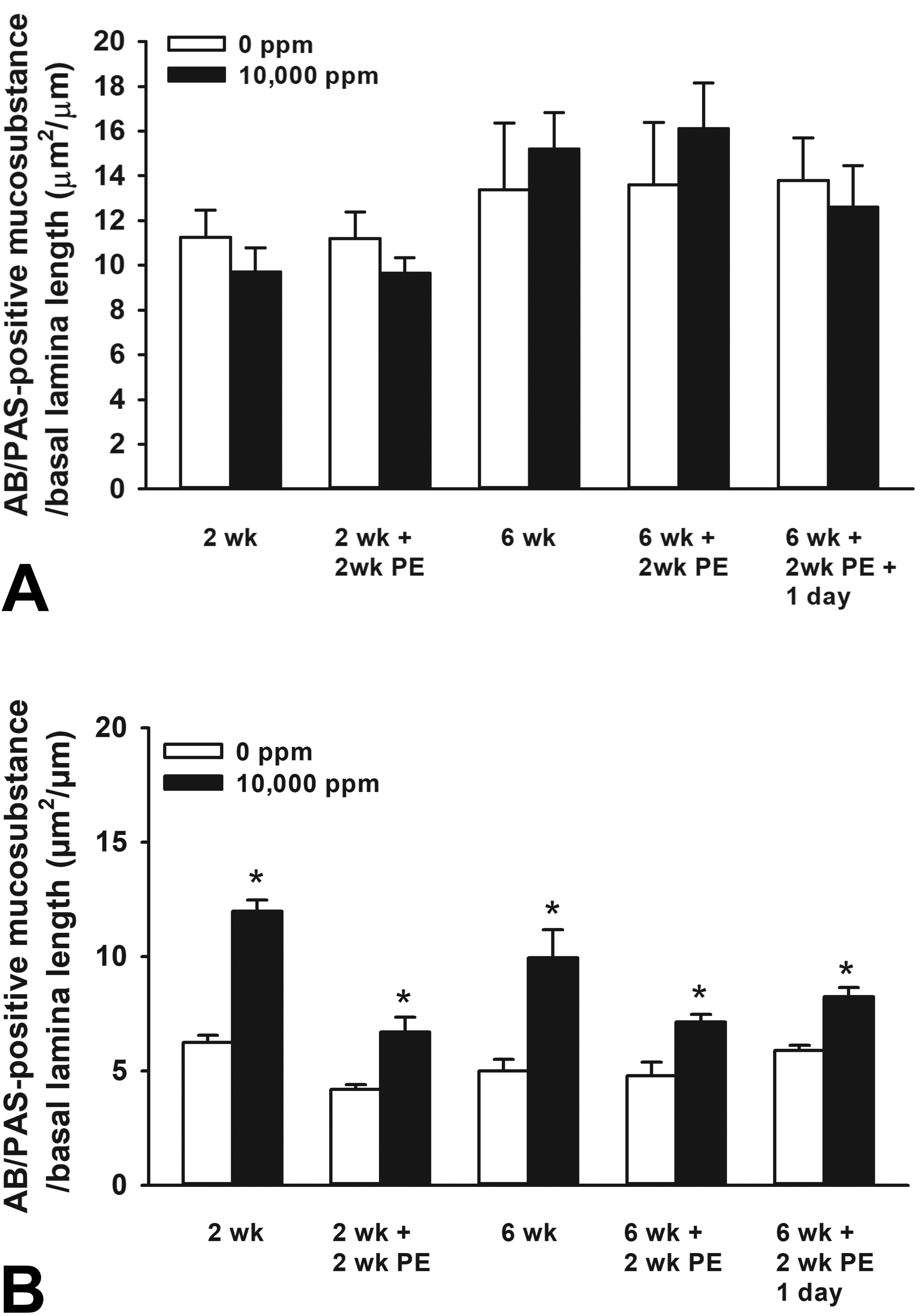
Graphical representation of the morphometrically determined amount of intraepithelial Alcian blue/periodic acid-Schiff (AB/PAS)–stained mucosubstances in the (A) proximal mid-septum (MS; T1 section) and (B) distal nasopharyngeal meatus (NPM; T4 section) of rats episodically exposed to 0 ppm (air control) or 10,000 ppm ethylene and examined at various scheduled times after designated repeat exposures. Bars, group means ± standard error of the means (SEM). PE indicates postexposure; asterisk, statistically greater than representative air controls, P ≤ .05.
Morphometrically determined amounts of AB/PAS-stained mucosubstances in the RE lining the NPM (T4 section) were approximately 100% greater in ethylene-exposed rats as compared to FA control rats (Figure 4B). By 2 weeks postexposure, this ethylene-induced increase in intraepithelial mucosubstances in the RE was attenuated by about 50%, but still significantly increased above that of air controls (Figure 4).
Rats exposed to ethylene for three 2-week exposure/no exposure blocks (6-week episodic ethylene exposure) had a similar, but slightly less severe MCH (as compared to a single 2-week exposure to ethylene) in the RE lining the distal NPM (T4 section; Figure 4B). No MCH was present in the RE lining the proximal MS of rats that received a 6-week episodic ethylene exposure (Figure 4A and B). Two weeks after this 6-week episodic exposure, the amount of intraepithelial mucosubstances in the RE lining the NPM had markedly attenuated and did not increase further with a 1-day reexposure to ethylene (Figure 4B).
A very mild bilateral eosinophilic rhinitis, influx of eosinophils in the nasal mucosa lining the lateral wall of the proximal nasal airways (T1 section; only 0.1% of the nasal mucosa) was present in rats that received the 6-week episodic ethylene exposure, but not in those that received the 2-week exposure (Figure 5). There was an approximately 150% greater density of MBP-positive eosinophils in the lamina propria of the nasal mucosa of 6-week ethylene-exposed rats as compared to FA controls. No significant difference in mucosal eosinophil density between FA control and 6-week ethylene-exposed rats was morphometrically determined after 2 weeks postexposure, indicating full resolution of this inflammatory response. There was also no ethylene-induced eosinophilic influx after a 1-day reexposure in rats previously exposed ethylene (Figure 5C).
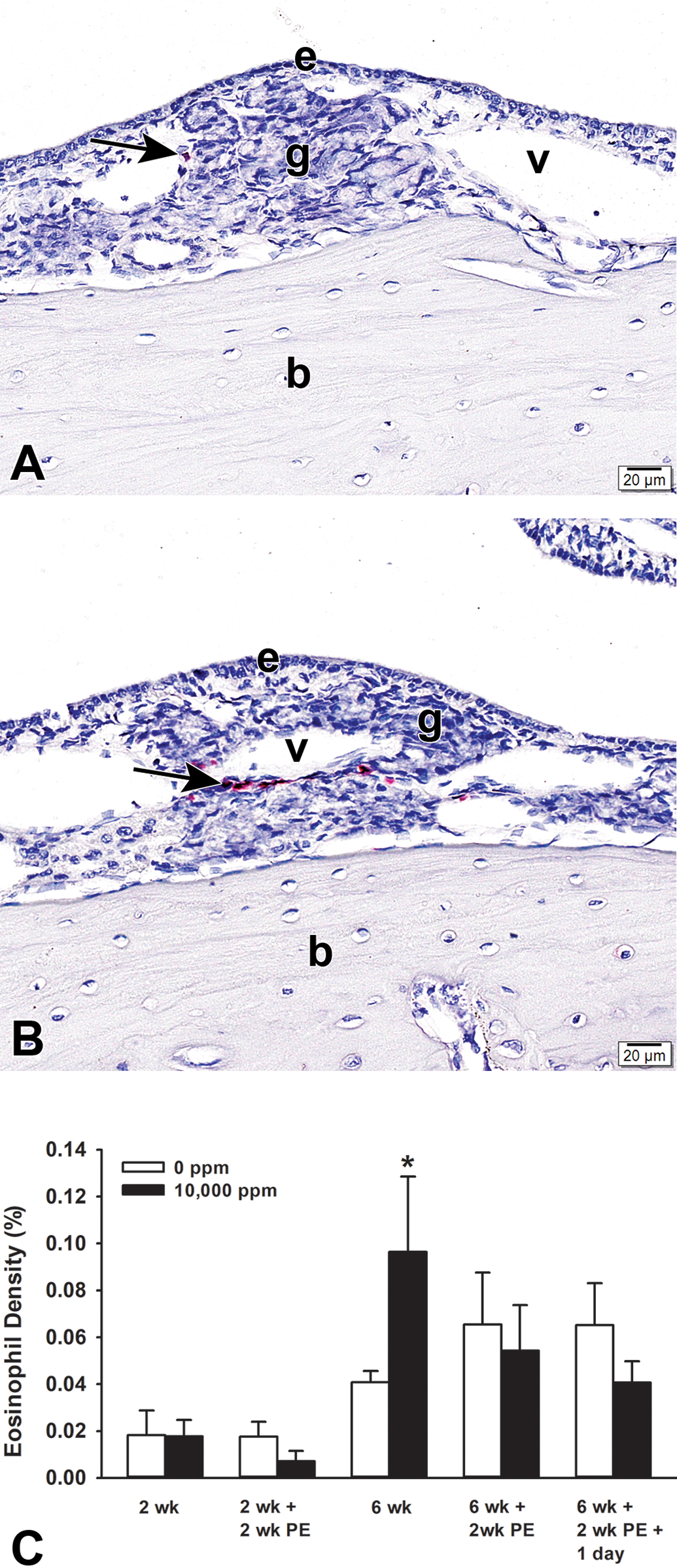
A and B, Light photomicrographs of perivascular eosinophils immunohistochemically stained for major basic protein (arrows, red chromagen) in the nasal mucosa lining the proximal lateral wall (T1 section) of rats exposed to 0 (A) or 10,000 (B) ppm ethylene for 6 weeks. Tissues were counterstained with hematoxylin. C, Graphical representation of morphometrically measured densities of eosinophils in the nasal mucosa after air or ethylene exposures and postexposures (PE). Asterisk, statistically greater than representative air controls, P ≤ .05. b indicates bone; e, nasal epithelium; g, gland in lamina propria; v, blood vessel in lamina propria.
Gene expression analysis in rat nasal mucosa
Other rats that received the 6-week episodic FA or ethylene exposure were dedicated for gene expression analysis of the nasal mucosa. The heads of these rats were immersed and stored in RNAlater. Airway mucosa lining the NPM was microdissected and processed for quantitative PCR as previously described in detail. 8,19
An array with different genes associated with type 1 and type 2 immunity/inflammation and airway mucus production/secretion was performed on pooled group samples. Genes that showed a higher than 1.5-fold increase or decrease relative to FA controls were further analyzed on individual samples and statistically evaluated (Table 1).
Interestingly, only 2 genes, both related to airway mucous production/secretion proteins,
Mice Episodically Exposed to Ethylene
Nasal histopathology and morphometry
In contrast to rats, wild-type C57BL/6 mice episodically exposed to ethylene for 2 weeks had MCH in RE lining the proximal MS (T1 section) with increases in the amount of intraepithelial mucosubstances (approximately 100% more than FA controls; Figures 6 and 7). This response to ethylene, however, did not persist 2 weeks postexposure.
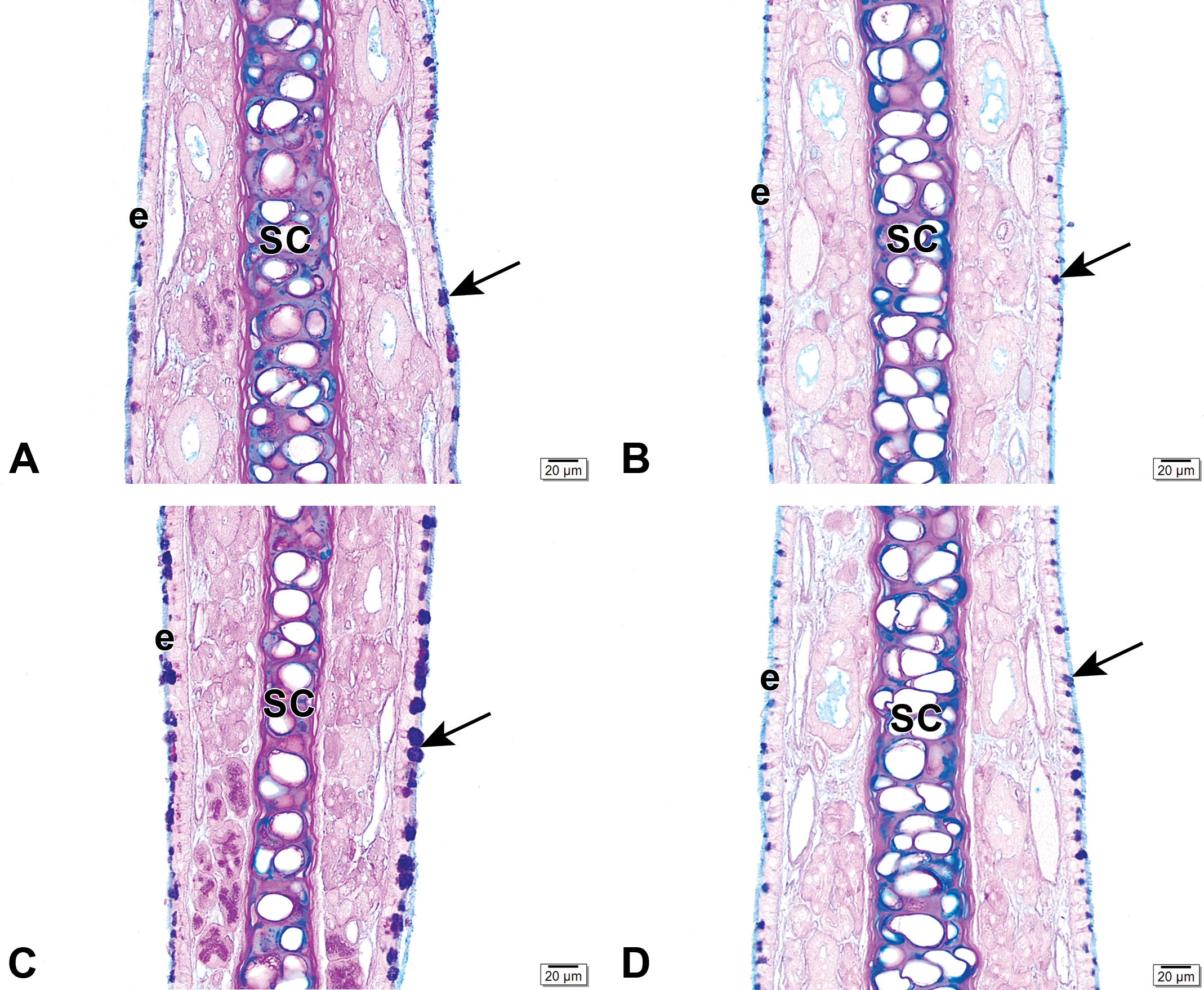
Light photomicrographs of Alcian blue/periodic acid-Schiff (AB/PAS)–stained mucosubstances (arrows) in the nasal respiratory epithelium (e) lining the proximal mid-septum (MS; T1 section) of mice exposed for 2 weeks (A, C) or 6 weeks (B, D) to air (A, B) or ethylene (C, D). SC indicates septal cartilage.
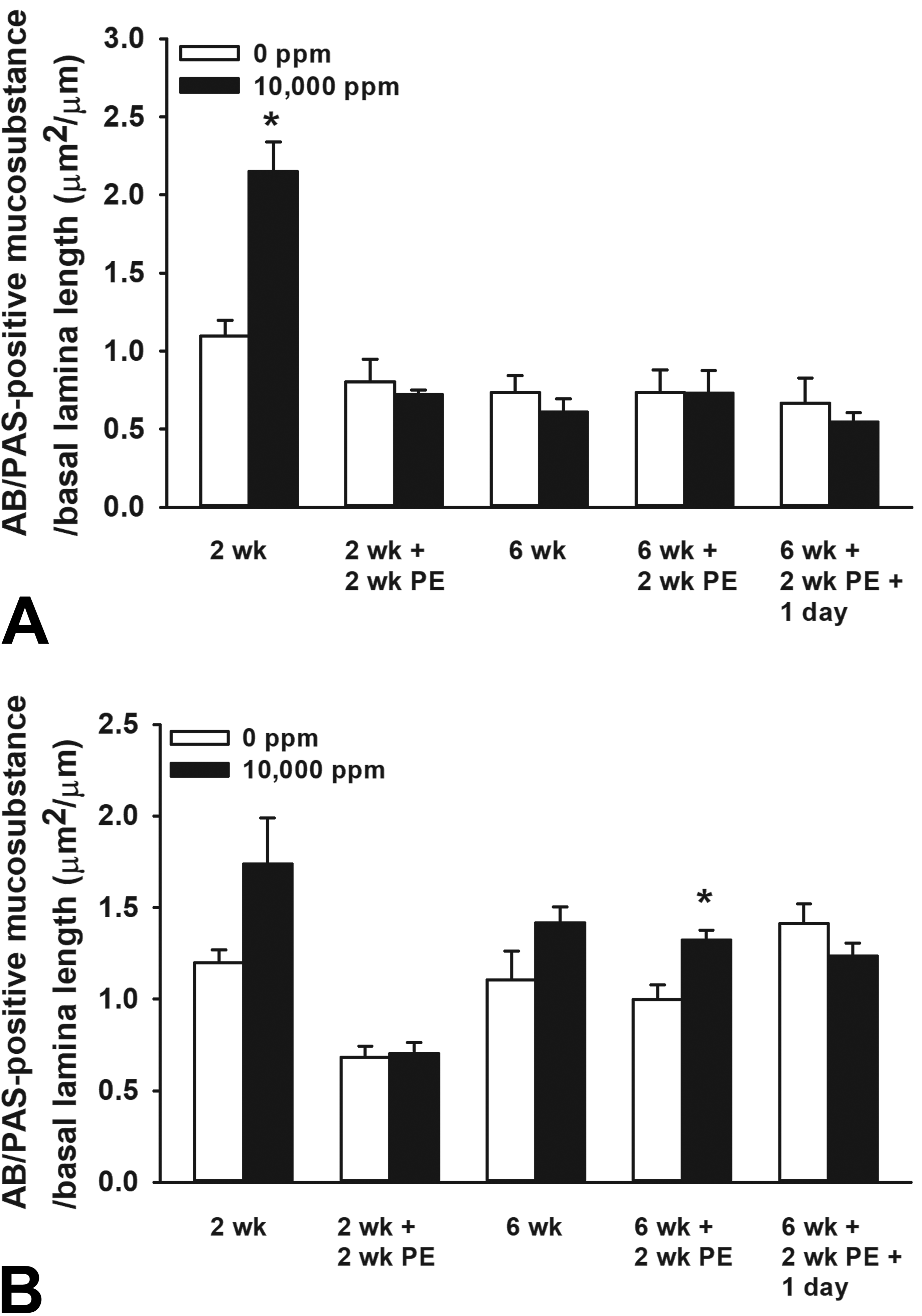
Graphical representation of the morphometrically determined amount of intraepithelial Alcian blue/periodic acid-Schiff (AB/PAS)–stained mucosubstances in the (A) proximal mid-septum (MS; T1 section) and (B) distal nasopharyngeal meatus (NPM; T4 section) of mice episodically exposed to 0 ppm (air control) or 10,000 ppm ethylene and examined at various scheduled times after designated repeat exposures. Bars, group means ± standard error of the means (SEM). Asterisk, statistically greater than representative air controls (0 ppm ethylene), P ≤ .05.
Interestingly, 2-week and 6-week ethylene-exposed wild-type C57BL/6 mice, did not develop MCH with increased mucosubtances in the RE lining the NPM (T4 section) as was observed in similarly exposed rats. Furthermore, neither mice exposed to 2-week or 6-week episodic ethylene exposures developed eosinophilic rhinitis (no mucosal influx of eosinophils) like that observed in proximal nasal airways of rats.
Role of ILCs in Ethylene-Induced MCH in Mice
Based on our findings in wild-type C57BL/6 mice, we further investigated the role of ILCs in ethylene-induced MCH by morphometrically measuring the amount of AB/PAS-stained mucosubstances in the RE lining the proximal MS (T1 section) of lymphoid cell sufficient wild-type C57BL/6 mice, T and B cell-deficient (but ILC-sufficient) Rag2−/− mice, and lymphoid cell deficient (no ILCs, T or B cells) Rag2−/− gamma c−/− mice exposed to FA or ethylene for 2 weeks.
Nasal histopathology and morphometry
Innate lymphoid cell–sufficient, C57BL/6 and Rag2−/−, mice had significant ethylene-induced MCH with increases in the amount of intraepithelial AB/PAS-stained mucosubstances in RE lining the proximal MS (Figure 8). In contrast, ILC-deficient Rag 2−/− gamma c−/− mice exposed to ethylene had no significant increases in the amount of intraepithelial mucosubstances as compared to FA controls, indicating that ethylene-induced MCH is ILC dependent.
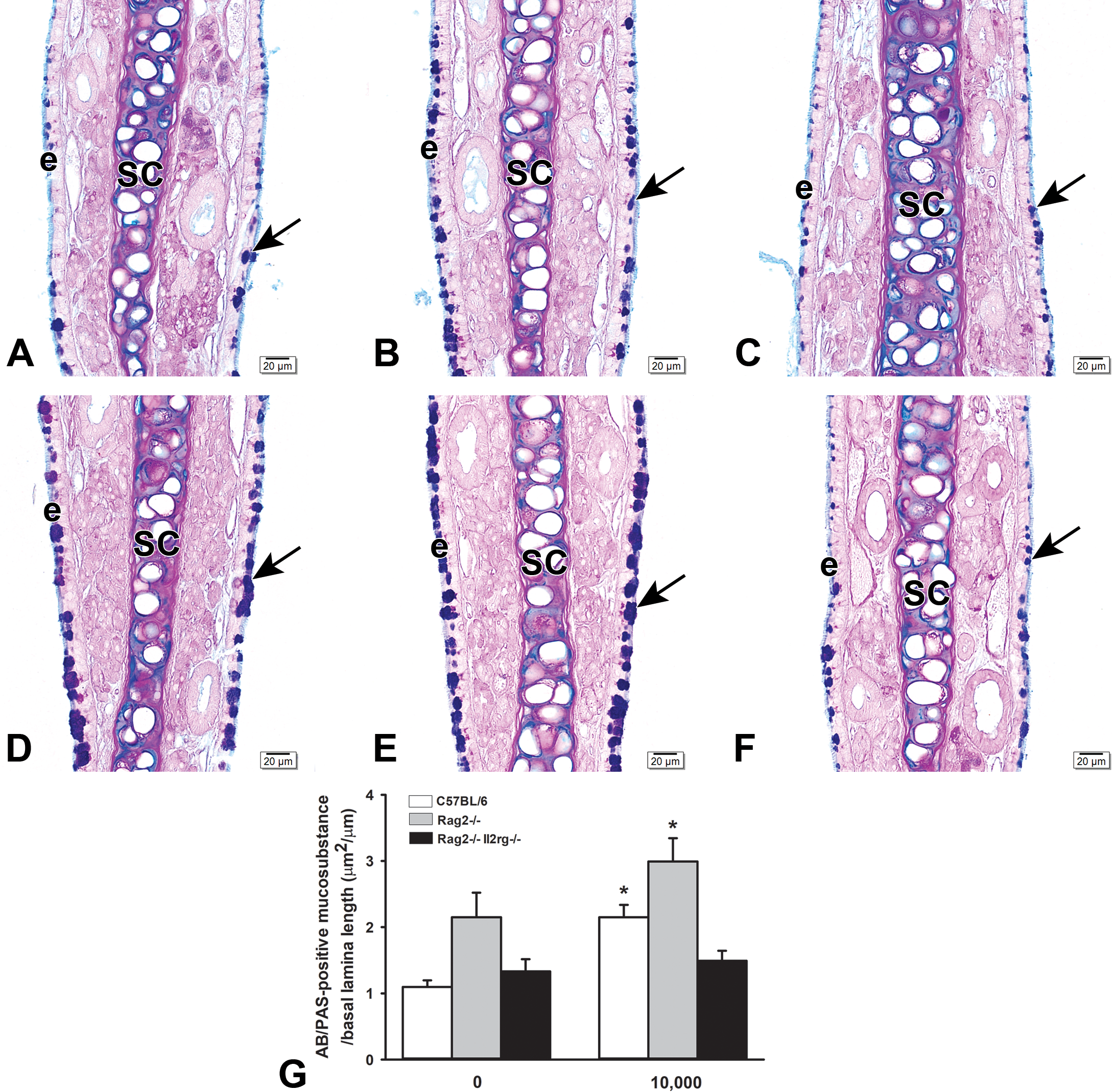
Alcian blue/periodic acid-Schiff (AB/PAS)–stained mucosubtances (arrows) in the nasal respiratory epithelium lining the mid-septum (MS; T1) of C57BL/6 (A, D), rag2−/− (B, E) and rag2−/−Il2rg−/− (C, F) mice exposed to air (A, B, C) or ethylene (D, E, F). G, Graphical representation of the morphometrically determined amounts of mucosubstances in the respiratory epithelium lining the proximal midseptum in C57BL/6, rag2−/− and rag2−/−Il2rg−/− mice exposed to 0 or 10,000 ppm ethylene. Bars, group means ± standard error of the means (SEM). Asterisks, statistically different from respective air controls (0 ppm ethylene). E indicates respiratory epithelium; sc, septal cartilage.
Discussion
In this study, we expanded our investigation of the nasal toxicology caused by exposure to high concentrations of ethylene (10,000 ppm) by determining (1) the epithelial and inflammatory histopathology in rat nasal airways caused by episodic, rather than continuous, inhalation exposures, (2) the ethylene-induced nasal histopathology in episodically exposed mice, as compared to rats, and (3) how ILCs may play a role in the increase of nasal epithelial mucosubstances in response to episodic ethylene exposures. The results of our study indicated that MCH was the principal nasal epithelial lesion induced by episodic ethylene exposures in both rats and mice. There was also a mild, but transient, inflammatory response (eosinophilic rhinitis) that occurred only in ethylene-exposed rats. Ethylene-induced MCH, with increases of mucosubstances in nasal respiratory epithelium, was transient in the mouse (only after initial 2-week exposure), but persistent in the rat (at all postexposure time points examined). In addition, the intranasal location of this respiratory epithelial alteration differed between the 2 rodent species (proximal nasal airways in mice and distal nasal airways in rats). Furthermore, increased amounts of epithelial mucosubstances after 2 weeks of ethylene exposure were present in ILC-sufficient C57/BL6 and Rag2−/− mice, but not in ILC-deficient Rag2−/−IL2rg−/− mice indicating that this epithelial change is ILC dependent and not T- or B-cell dependent.
Airway mucus serves an important role in maintaining the health and function of the entire respiratory tract, nose to lung. 24 It not only prevents dehydration of airway tissues but it also removes, in concert with beating cilia (mucociliary clearance), inhaled xenobiotic materials such as microbes and air pollutant particles, as well as inflammatory cells and cellular debris when airways are injured. The major macromolecular components of airway mucus are mucin glycoproteins (acidic and neutral mucosubstances) that are secreted from surface epithelial cells and subepithelial mucosal glands.
Repeated exposures to some inhaled toxicants, such as ozone, chlorine, and cigarette smoke, cause increases in the density of acidic and neutral mucosubstances in the nasal epithelium of rodents depending on the exposure dose, duration, and regimen. 20,25 –28 Repeated exposures of rats and mice to the irritating air pollutant ozone particularly cause the appearance of mucous cells in nasal transitional epithelium that is normally devoid of these mucus-secreting cells, that is, mucous cell metaplasia (MCM). 18,19,25 Although these adaptive airway epithelial changes to toxicant exposures are a part of normal tissue defense and repair processes, the progression and persistence of these lesions (epithelial remodeling) may result in excess surface mucus and a slowing of mucociliary clearance, 27 thus compromising upper airway defense from inhaled xenobiotics. Furthermore, MCH and increased epithelial mucosubstances are key histopathologic features of chronic airway diseases including allergic rhinitis, asthma, and cystic fibrosis 28 and animal models of these human respiratory diseases. 29 –31
We have previously demonstrated that F344 rats exposed to continuous weekday exposures (5, 10, or 20 days) to 10 to 10,000 ppm ethylene had significant increases in the amount of mucosubstances in the respiratory epithelium lining the distal nasal airway (NPM), but not in the proximal nasal airway (middle meatus). Animals exposed for 20 continuous weekdays (6 h/d, 5 d/wk for 4 consecutive weeks) to 10,000 ppm ethylene also had a mild influx of eosinophils in the proximal nasal airways (eosinophilic rhinitis) and overexpression of genes in the nasal mucosa that were related to type 2 immunity (ie, Ch3l3, IL-5, and IL-13) and airway mucus production/secretion (eg,
However, our hypothesis that episodic exposures to ethylene would result in a progressive increase in the amount of epithelial mucosubstances and mucosal eosinophils was not confirmed (null hypothesis). Although significant increases in epithelial mucosubstances were present in ethylene-exposed rats at all the postexposure time points, as compared to air-exposed controls (0 ppm ethylene), there were no significant differences in the amounts of mucosubstances between 2-week and 6-week episodic exposures. This finding further supports the premise that ethylene is not a chemical sensitizing agent that induces adaptive immunity and an enhanced allergen-like immune response upon reexposure of the agent (type 1 hypersensitivity reaction). This is also consistent with the finding of Brandenberger et al that 20 consecutive weekday exposures to 10,000 ppm ethylene did not cause an increase in serum levels of immunoglobulins, IgG1, IgG2a, and IgE. 8
Relative fold increases in mRNA transcripts of genes that are associated with airway mucus production and secretion (
Mucous cell hyperplasia/hypertrophy and increased production of airway mucosubstances are epithelial cell features strongly associated with the development of type 2 immunity and increased secretion of inflammatory cytokines (eg, IL-13) derived from either Th2 cells or ILC2s. 9,11,32,33 By exposing lymphoid cell-sufficient C57BL/6 mice, lymphoid cell-deficient Rag2−/−Il2rg−/−, and ILC-sufficient but T- and B-cell deficient Rag2−/− mice, we were able to explore the dependency of ethylene-induced MCH on the presence of these lymphoid cell types. Since ethylene-induced MCH with increased epithelial mucosubstances were found in the nasal respiratory epithelium of ILC-sufficient mice, but not in ILC-deficient mice, this suggests that ILCs, and most likely ILC2s, play a mediating role in the pathogenesis of these changes caused by repeated ethylene exposure in mice. It is also clear that ethylene-induced MCH in mice is not T- or B-cell dependent.
Interestingly, similarly designed studies in mice have recently found that repeated inhalation exposures to ozone induce type 2 immunity, eosinophilic inflammation, and increased airway epithelial mucosubstances, all of which are ILC2-dependent. 18 –20,34 Further studies, however, are needed to fully elucidate how ILCs mediate ethylene-induced MCH in mice and probably in rats.
Unlike the repeated ethylene exposures that induce MCH in nasal respiratory epithelium with minimal eosinophilic inflammation at high concentrations, repeated ozone exposures cause MCM in nasal transitional epithelium with marked eosinophilic inflammation at much lower concentrations (0.5-1.0 ppm; 10 times the national ambient air quality standard concentration) in the mucosa lining the lateral meatus of proximal nasal airways (T1 section). 18,19 In addition, there are no differences in the intranasal location of nasal lesions between rats and mice, as was found with ethylene-induced MCH and eosinophilic rhinitis in the present study. The biological and toxicologic reasons for these chemical- and species-dependent differences in epithelial and inflammatory responses to 2 very different chemical compounds are not yet known. The results of this and other studies, however, clearly indicate that ethylene is considerably less toxic to the rodent nasal airways than the ambient air pollutant ozone.
The findings of this animal study (and those of previous studies) strongly suggest that inhaled ethylene is not a chemical-sensitizing agent and will not induce an adaptive immune response and allergen-like nasal airway disease with repeated episodic exposures.
Footnotes
Acknowledgments
The authors thank Amy Porter and Kathy Joseph of the Michigan State University Laboratory for Investigative Histopathology for their assistance with the histotechnology.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research was funded by the American Chemistry Council, ACC-1648.
