Abstract
The National Toxicology Program (NTP) now uses an extended longitudinal sectioning protocol for the uterus to better evaluate female rodent reproductive tract toxicity for all developmental and reproductive toxicology and 2-year toxicity and carcinogenicity bioassays. The previous protocol for toxicity/carcinogenicity studies involved 1 cross section midway through each uterine horn and collection of uterine cervix and vagina only if gross lesions were present. Here we compare the histological findings of the original cross sections with the additional longitudinal sections of residual uterine tissues of 7 chronic NTP rat bioassays. The goal of this study was to determine whether there might be any advantages to examining additional uterine tissue. The longitudinal protocol allowed for 10 to 20 times more uterine tissue for evaluation. Results indicate that the potential advantages of a more complete evaluation of female reproductive tract tissue include increased detection of reproductive targets, increased detection of neoplastic and nonneoplastic lesions, improved detection of tissue origin of neoplasms, less reliance on gross identification of lesions, improved accuracy in the application of severity grades, and increased detection of preneoplastic or subtle lesions.
Keywords
Introduction
Since 1978, the National Toxicology Program (NTP) has evaluated agents of public health concern by developing and applying tools of modern toxicology and molecular biology, always striving to remain at the cutting edge when developing and applying new technologies. Two distinct trimming protocols have been used in NTP rodent bioassays, depending on study type: toxicity and carcinogenicity or developmental and reproductive toxicology (DART) studies.
National Toxicology Program specifications 1 for toxicity/carcinogenicity studies previously indicated that a transverse section through each uterine horn, approximately 0.5 cm from the uterine cervix/body, was required (Figure 1). The remaining uterine horns, body, cervix, and vagina were not evaluated histologically unless there were gross lesions identified during necropsy or wet tissue audit.
Example of transverse sections from the uterine horns that were evaluated in the original review. Hematoxylin and eosin.
National Toxicology Program specifications (NTP 2011) for DART studies 2 require a more thorough evaluation of the female reproductive tract where the uterus, uterine cervix, vagina, and ovaries are removed en masse, and the uterus/uterine cervix/vagina (together) are mounted on cardboard prior to fixation in 10% neutral buffered formalin (NBF). 2 Transverse sections through each uterine horn are collected, fixed, embedded, and sectioned. In addition, the remaining 2 distal free sections of uterine horn, 2 proximal sections of uterine horn attached to the uterine body, and the uterine cervix/vagina are also collected, fixed, embedded, and sectioned longitudinally (Figure 2).
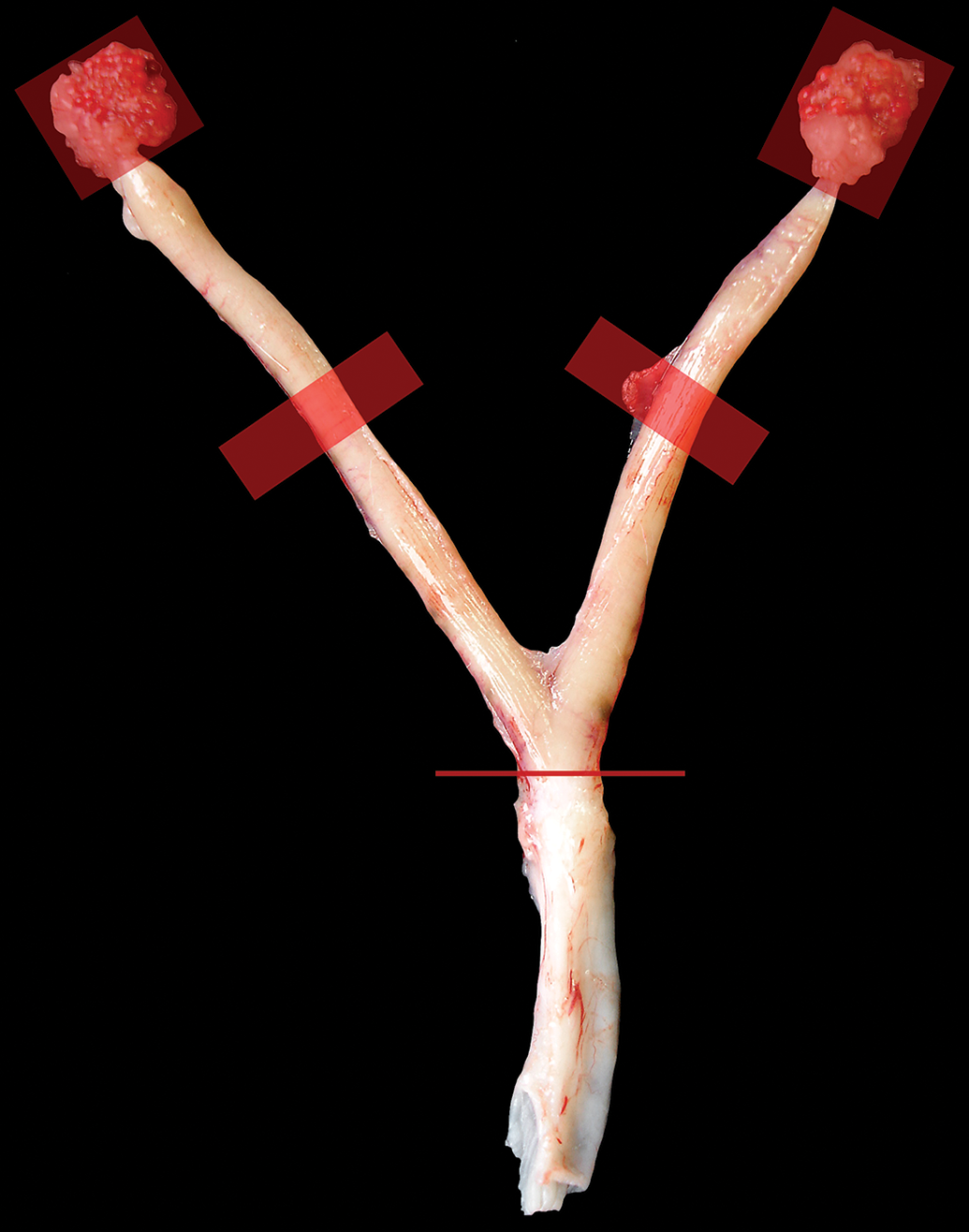
Gross image of normal female rat reproductive organs. This image illustrates what has been done for NTP DART studies as well as the current protocol for all NTP toxicity and carcinogenicity studies. The ovaries (red boxes) are attached in this image but are removed and embedded whole separately. The thick red horizontal lines show where a transverse cut is made through the midpoint of the uterine horns for the original review of NTP toxicity and carcinogenicity studies and is also a section that is evaluated for DART studies. The thin red line shows where the cut is made so that the cervix and vagina are collected as 1 piece. Hematoxylin and eosin. DART indicates developmental and reproductive toxicology; NTP, National Toxicology Program.
To determine whether there might be any advantages to examining additional female reproductive tract tissue for NTP toxicity and carcinogenicity studies, a tissue sectioning protocol similar to the one used for the DART studies was used for 7 chemicals that were on study: pentabromodiphenyl oxide mixture (DE-71), 3 green tea extract (GTE), 4 tetrabromobisphenol A (TBBPA), 5 CIMSTAR 3800 (metalworking fluid), 6 2,3 butanedione (diacetyl), 7 indole-3-carbinol (I3C), 8 and perfluorooctanoic acid (PFOA). 9 The most interesting and important findings were in the additional uterine horns, rather than in the uterine cervices or vaginas, so they are emphasized here. In these extended evaluations, additional neoplastic and nonneoplastic lesions were discovered in the uterine residual tissues and here we focus on these lesions for comparison: adenoma, adenocarcinoma, malignant mixed Müllerian tumor (MMMT), stromal polyp, stromal sarcoma, leiomyosarcoma, malignant schwannoma, atypical hyperplasia, and cystic endometrial hyperplasia (CEH).
Materials and Methods
These data come from seven 2-year toxicity and carcinogenicity bioassays conducted by the NTP. The rats were either Wistar Han [Crl: WI (Han)] or Hsd: Sprague Dawley SD strains; the route of administration was either gavage, inhalation, or feed; and the vehicle control was either corn oil, deionized water, or air (Table 1). Female rats were obtained from either Charles River Laboratories, [Crl: WI (Han)], or from Harlan Sprague-Dawley, Inc (Hsd: Sprague Dawley SD). The DE-71 and PFOA studies included a perinatal exposure (gestation day 6 through lactation) such that exposure began in utero and continued throughout the rest of the study for the F1 animals that were evaluated. Animal care and use was in accordance with the Public Health Service Policy on Humane Care and Use of Animals. All animal studies were conducted in an animal facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. Animals were fed NTP-2000 diet ad libitum with the exception of DE-71 and PFOA animals, which were fed the NIH-07 diet (standard prenatal diet) before postnatal day 20 and NTP-2000 diet thereafter.
Chemicals, Rat Strains, Routes of Administration, and Vehicle Controls.
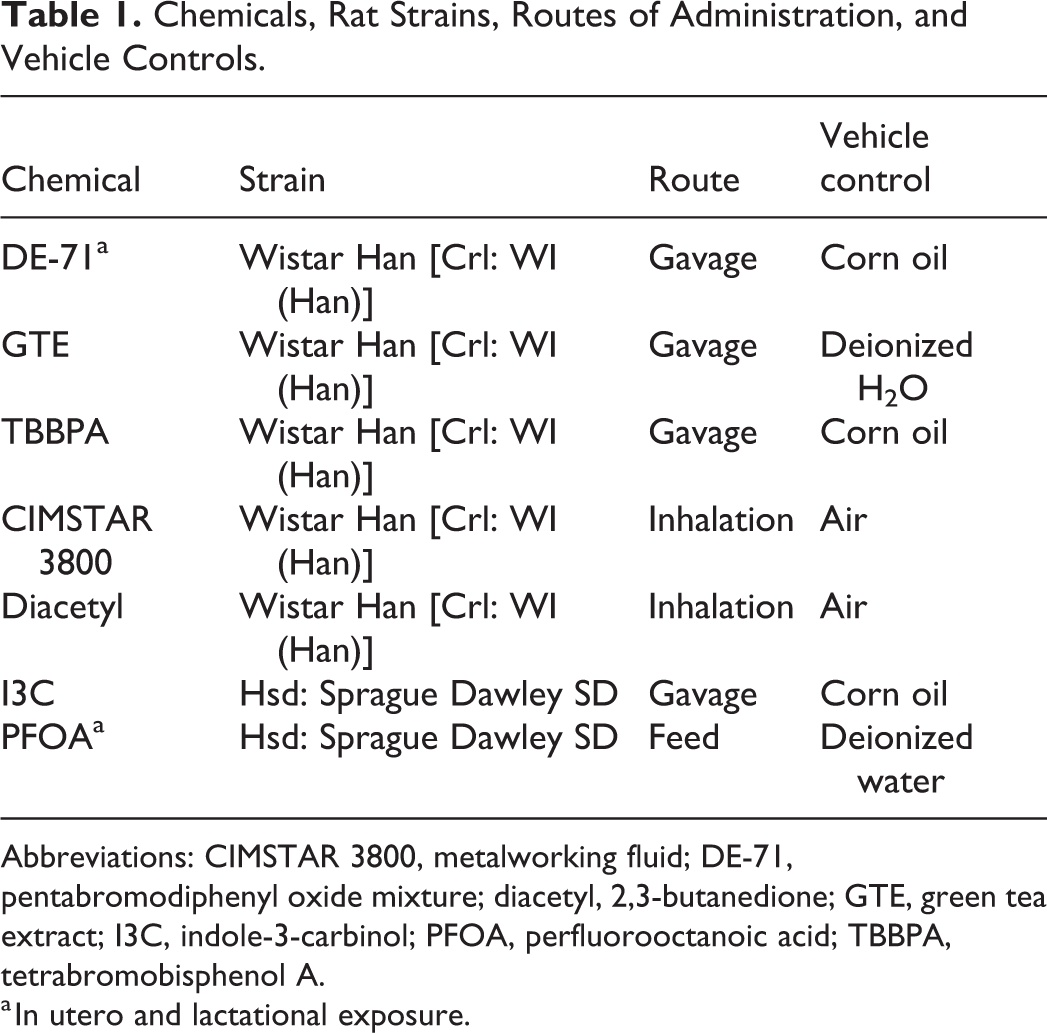
Abbreviations: CIMSTAR 3800, metalworking fluid; DE-71, pentabromodiphenyl oxide mixture; diacetyl, 2,3-butanedione; GTE, green tea extract; I3C, indole-3-carbinol; PFOA, perfluorooctanoic acid; TBBPA, tetrabromobisphenol A.
a In utero and lactational exposure.
For all 7 studies, transverse sections of both uterine horns were collected for evaluation in the original study. The remaining uterine horns and bodies, cervices, and vaginas were collected from the residual tissues stored in 10% NBF. The uterine horns distal to the transverse sections and the uterine horns/bodies proximal to the transverse sections were collected (Figure 3). The uterine body, cervices, and vaginas were separated from adjacent tissues and collected separately (Figures 4 and 5). Any residual tissues that were “curled” or otherwise distorted were processed with a sponge in the cassette to help with flattening. During paraffin embedding, tissues were further flattened when adhered to the base of the paraffin mold. Tissues were sectioned at 5 microns in a longitudinal (dorsoventral) manner and stained with hematoxylin and eosin. Microscopic evaluations were performed, and the results were peer-reviewed by 9 to 11 board certified veterinary anatomic pathologists.
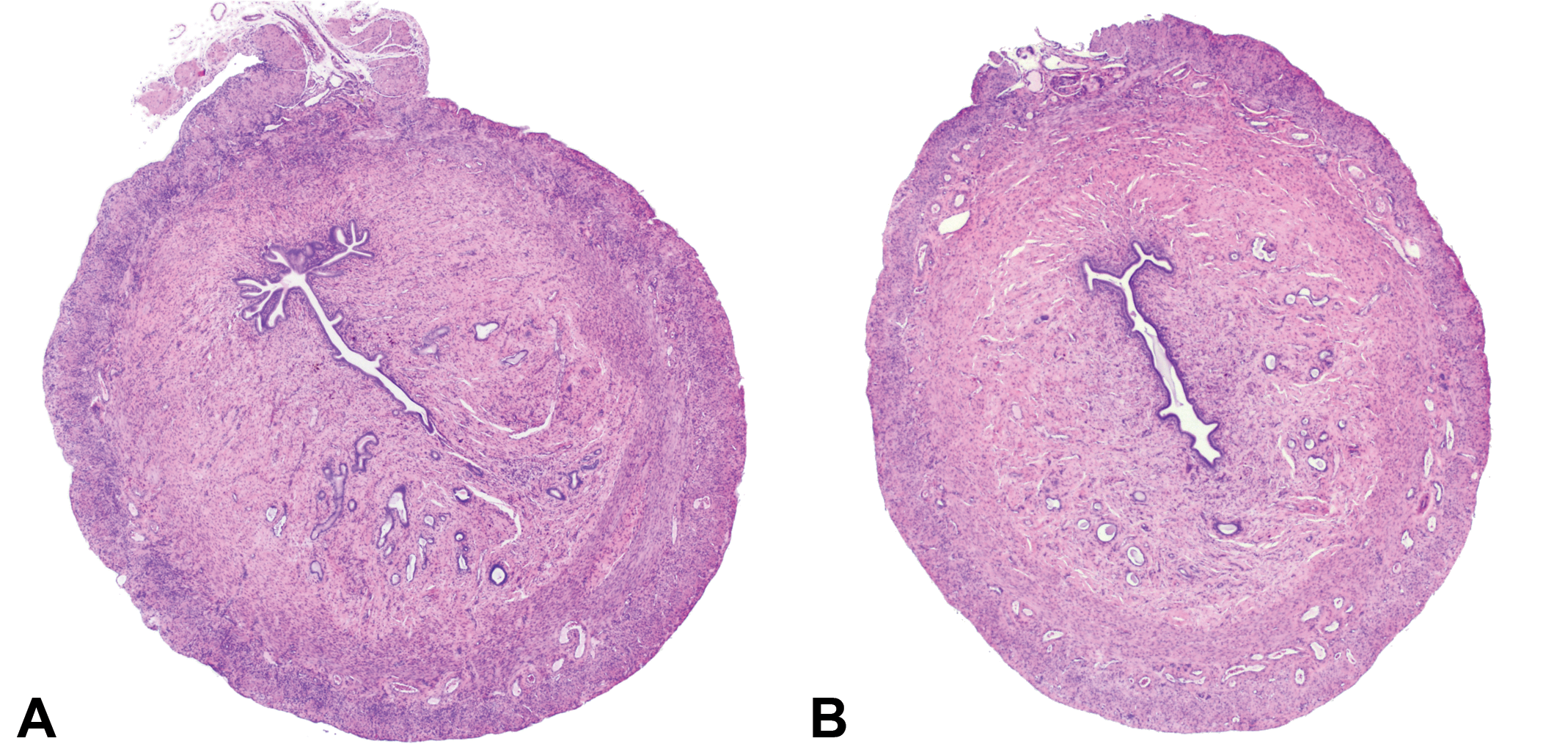
An example of uterine horn residual tissue that was sectioned longitudinally. The left and right uterine horns attached to the uterine body (left side of image) and the corresponding 2 free portions of uterine horns (right side of image) were collected and sectioned longitudinally and evaluated in the extended review. A transverse section is included in this image for comparison of additional amount of tissue that the pathologist has to evaluate. Hematoxylin and eosin.
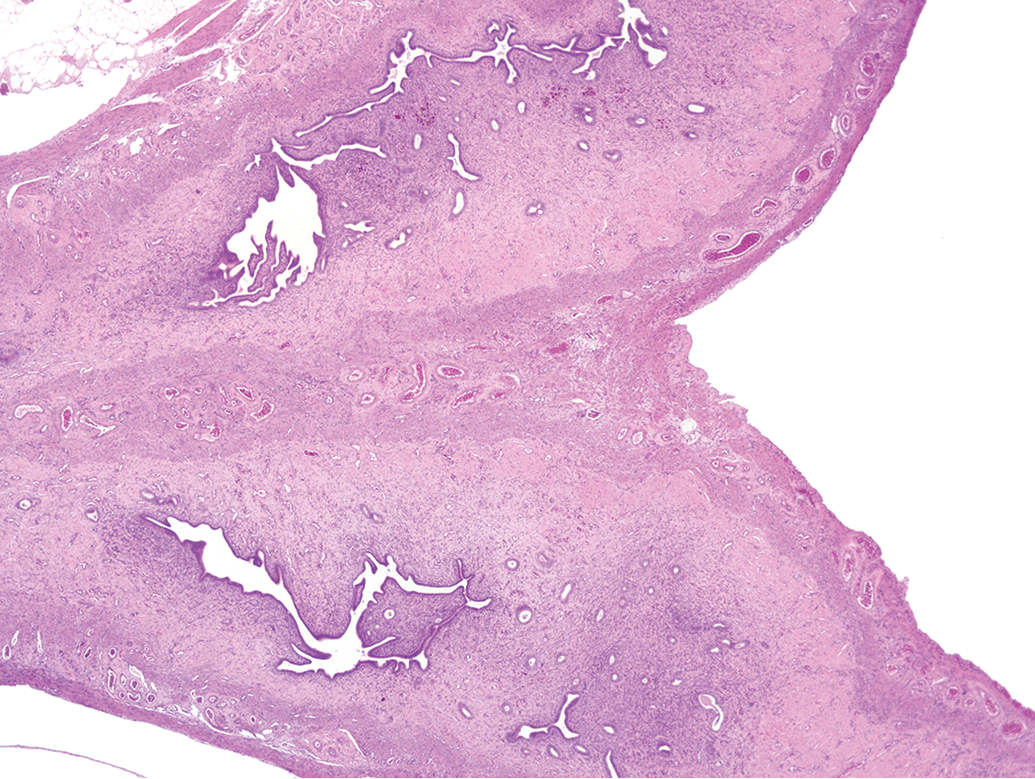
In addition to the uterine horns, the entire uterine body was collected and sectioned longitudinally in the extended review. Hematoxylin and eosin.
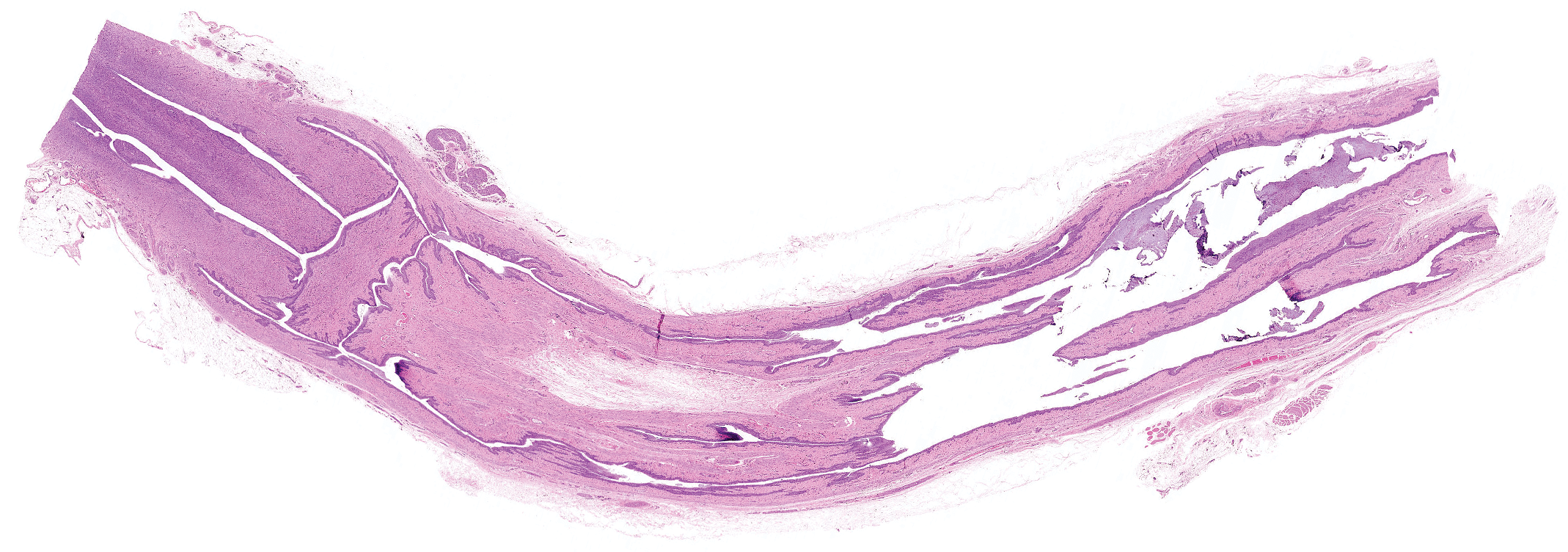
Example of cervix and vagina embedded as 1 piece and sectioned longitudinally. Hematoxylin and eosin.
Results
The results of the original and extended evaluations of uterine tissues from all 7 chemicals are presented in Table 2, a comparison of the uterine stromal sarcomas from the original and extended evaluations of all 7 chemicals is provided in Table 3, and the summary of the carcinogenic activity level of evidence conclusion from each study 10 after the extended evaluation is provided in Table 4. For additional information on animals per group, see Supplemental Tables 1-3.
Uterine Findings From Original and Extended Evaluations.a
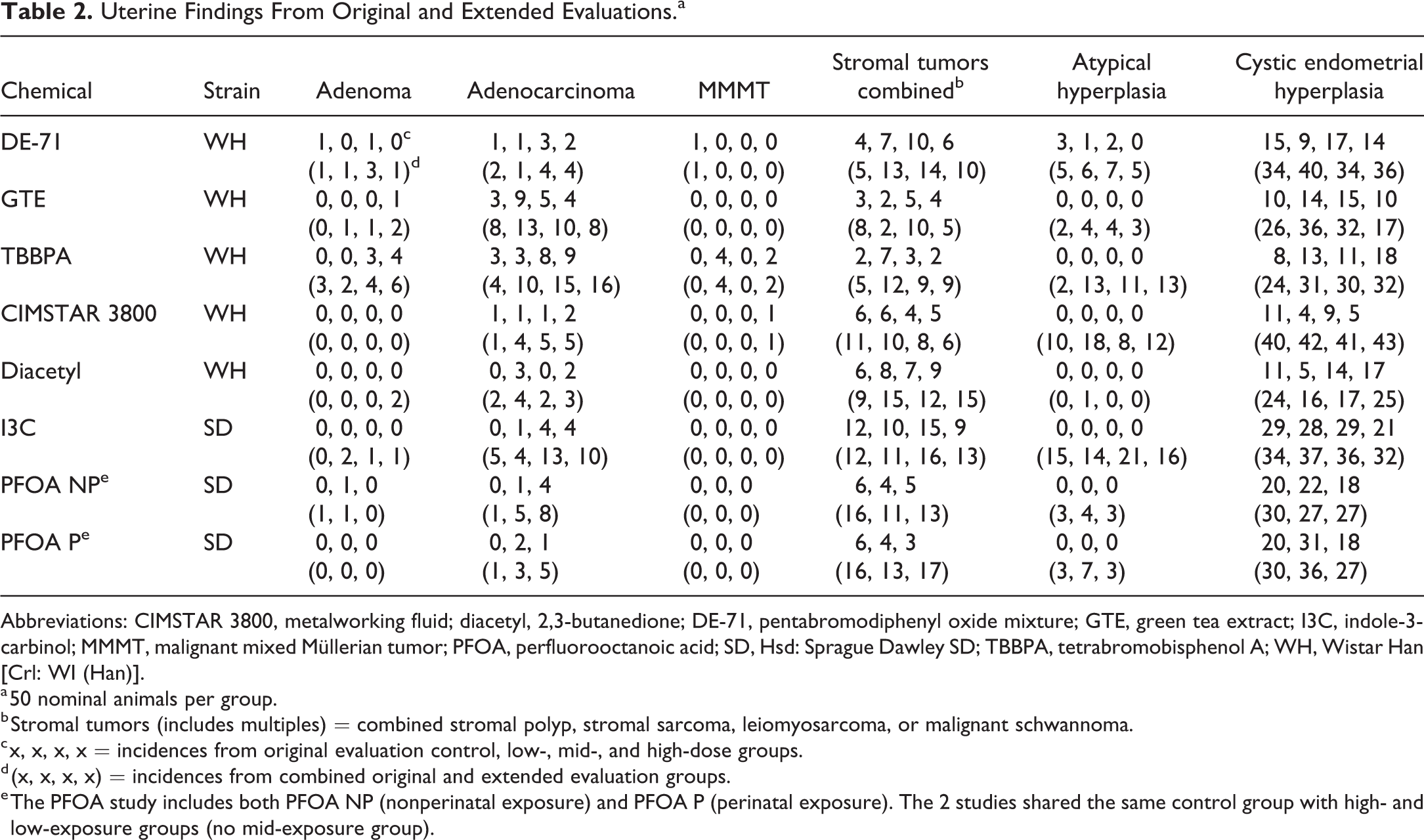
Abbreviations: CIMSTAR 3800, metalworking fluid; diacetyl, 2,3-butanedione; DE-71, pentabromodiphenyl oxide mixture; GTE, green tea extract; I3C, indole-3-carbinol; MMMT, malignant mixed Müllerian tumor; PFOA, perfluorooctanoic acid; SD, Hsd: Sprague Dawley SD; TBBPA, tetrabromobisphenol A; WH, Wistar Han [Crl: WI (Han)].
a 50 nominal animals per group.
b Stromal tumors (includes multiples) = combined stromal polyp, stromal sarcoma, leiomyosarcoma, or malignant schwannoma.
c x, x, x, x = incidences from original evaluation control, low-, mid-, and high-dose groups.
d (x, x, x, x) = incidences from combined original and extended evaluation groups.
e The PFOA study includes both PFOA NP (nonperinatal exposure) and PFOA P (perinatal exposure). The 2 studies shared the same control group with high- and low-exposure groups (no mid-exposure group).
Original and Extended Evaluations of Uterine Stromal Tumors.a

Abbreviations: CIMSTAR 3800, metalworking fluid; diacetyl, 2,3-butanedione; DE-71, pentabromodiphenyl oxide mixture; GTE, green tea extract; I3C, indole-3-carbinol; PFOA, perfluorooctanoic acid; SD, Hsd: Sprague Dawley SD; TBBPA, tetrabromobisphenol A; WH, Wistar Han [Crl: WI (Han)].
a 50 animals per group.
b Includes multiples.
c x, x, x, x = incidences from original evaluation control, low-, mid-, and high-dose groups.
d (x, x, x, x) = incidences from combined original and extended evaluation groups.
e Leiomyoma.
f The PFOA study includes both PFOA NP (non-perinatal exposure) and PFOA P (perinatal exposure). The two studies shared the same control group with high and low exposure groups (no mid exposure group).
Summary of Results After Uterine Longitudinal Sectioning Protocol.

Abbreviations: CIMSTAR 3800, metalworking fluid; diacetyl, 2,3-butanedione; DE-71, pentabromodiphenyl oxide mixture; GTE, green tea extract; I3C, indole-3-carbinol; MMMT, malignant mixed Müllerian tumor; NP, non-perinatal exposure; P, perinatal exposure; PFOA, perfluorooctanoic acid; TBBPA, tetrabromobisphenol A.
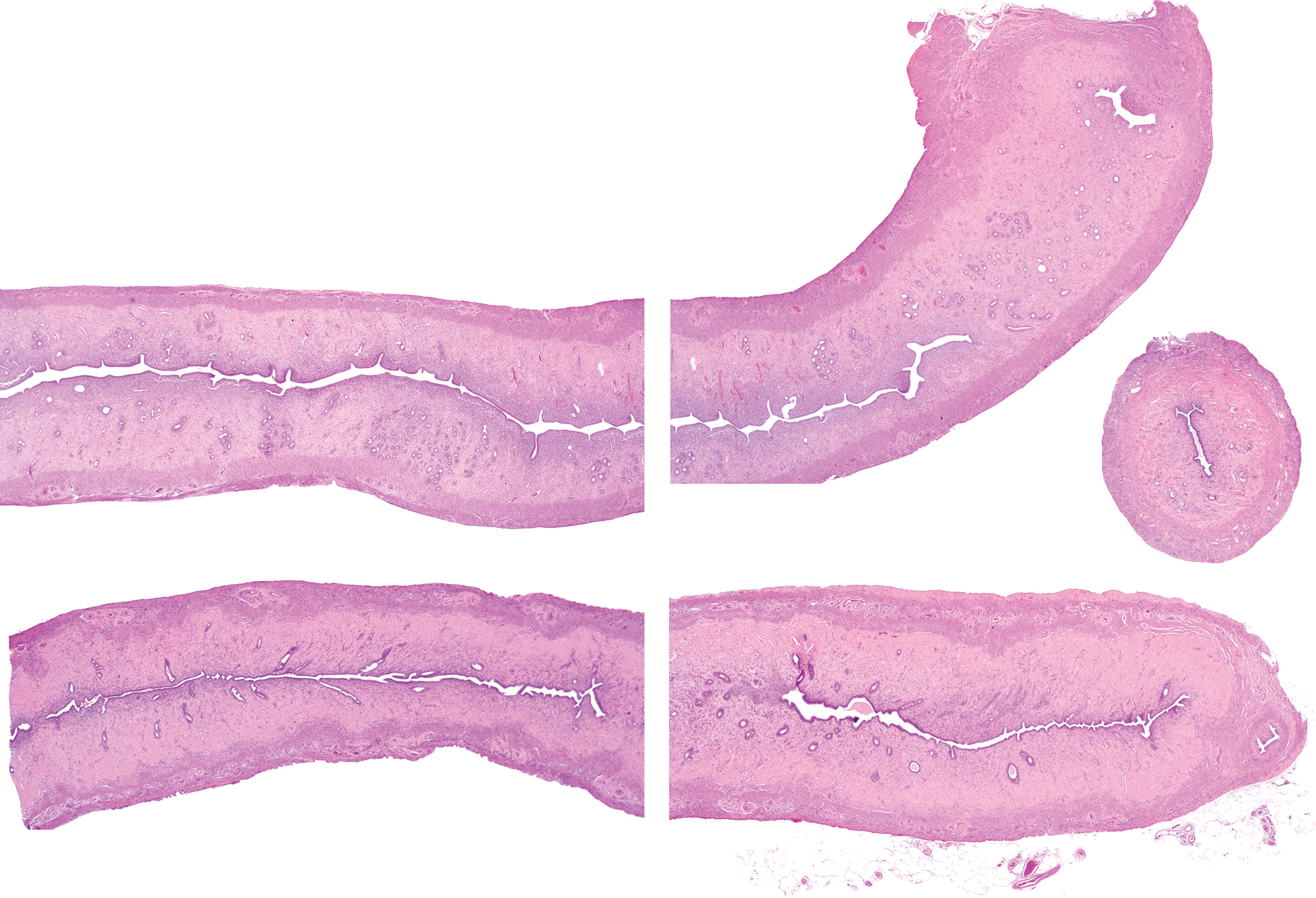
Atypical hyperplasia, also called focal atypical glandular hyperplasia, 11 is a preneoplastic lesion and was not present in the original cross section evaluations of 6 of the 7 compared studies but was observed in the extended reviews (all routes; controls range 0%-30%; Figure 6 and Table 2). These focal lesions were generally located more proximal to the uterine body than the collection site of the original cross sections. Uterine adenomas were observed with a low incidence in both the original cross section evaluations and the extended reviews (all routes; controls range 0%-6%; Table 2). Although uterine adenocarcinomas were usually present in the original cross section evaluations due to the typical large size of this tumor, higher incidences were still observed after the extended reviews (all routes; controls range 2%-16%; Table 2). Due to their typical large size, the extended evaluations did not identify any additional MMMT tumors (Table 2). The extended evaluations did occasionally identify an additional stromal sarcoma, leiomyosarcoma, or malignant schwannoma (Table 3). Additional stromal polyps were commonly identified with the extended evaluations (Table 3).
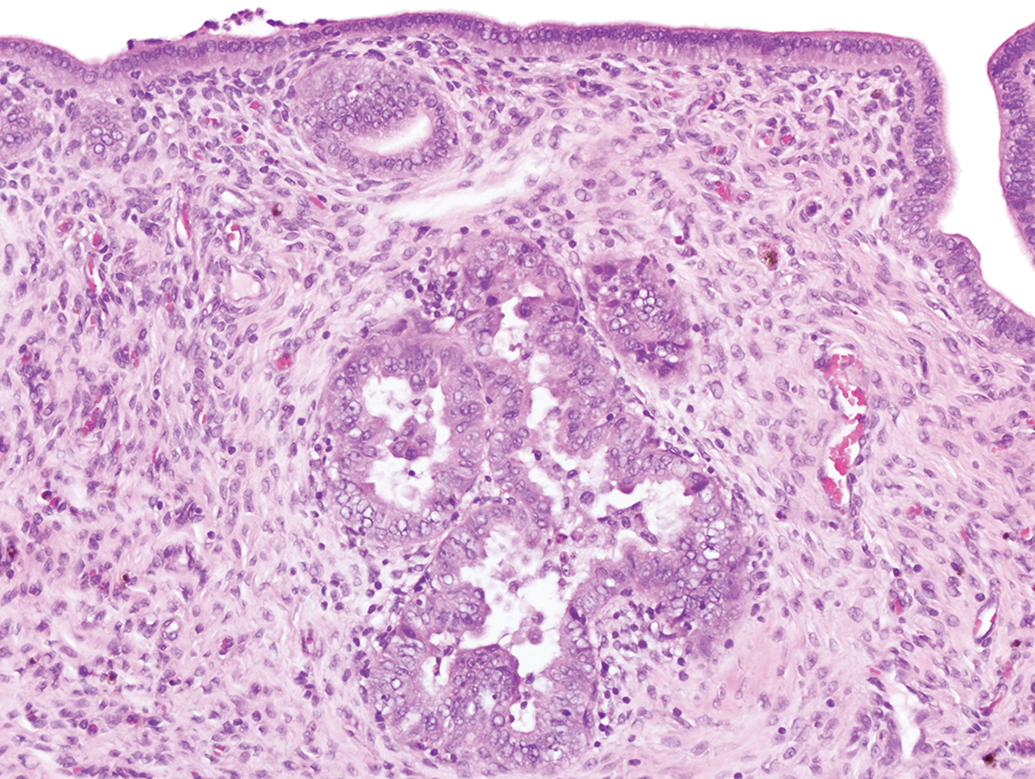
An example of atypical hyperplasia in a uterine longitudinal section. This lesion is characterized by clusters of enlarged glands separated by minimal amounts of stroma, though single glands can be affected. The epithelium is disorganized with stratification and piling up of the epithelial cells, sometimes up to 6 layers thick. Thickened epithelium often projects into the glandular lumen forming multiple infoldings and projections. Epithelial cells often display loss of nuclear polarization, karyomegaly, mitosis, and cellular pleomorphism. This lesion, when induced, is considered a precancerous lesion, in contrast to cystic glandular hyperplasia which is often seen in adult rats. Hematoxylin and eosin.
For all studies, the longitudinal evaluation of female reproductive tissues occurred before the final level of evidence calls were made. For the CIMSTAR 3800, PFOA, and I3C studies, the evaluation of additional tissues provided a clearer picture of uterine tumor incidence that the NTP used to make level of evidence calls of equivocal and some evidence of carcinogenic activity (Tables 2 -4). For TBBPA, the additional uterine adenomas, adenocarcinomas, and atypical hyperplasias added confidence to the call of clear evidence of carcinogenic activity. Moreover, the lesion of CEH had a statistically significant increase in the original review but this significant increase was not supported after the additional diagnoses from the extended review (Tables 2 -4).
For the other 3 chemicals (GTE, DE-71, diacetyl), the extended reviews did not lead to an observation of higher incidence of tumors in the exposed groups, and the lack of a statistically significant increase in uterine tumors in treated animals supported the level of evidence call of no evidence of carcinogenic activity from the original reviews (Tables 2 -4).
Discussion
The traditional NTP collection and trimming specifications for female reproductive tissues for toxicity/carcinogenicity bioassays have been in place since 1981. In this protocol, the ovaries are weighed and submitted whole and the uterine horns are bisected at their midpoint with 1 transverse section submitted from each horn. The remaining uterine horns, body, cervix, and vagina are collected but not sectioned for evaluation unless a gross lesion is present. Our primary goal was to collect all residual female reproductive tissue (uterine horns, body, cervix, vagina) from these 7 studies, section all in a longitudinal manner, and determine if there might be any advantages to examining the additional tissues in terms of identifying or clarifying treatment-related lesions.
The first study that we evaluated with this new sectioning protocol was TBBPA. In the initial review, a number of neoplastic masses were diagnosed and, in order to thoroughly characterize the response, all residual female tissues (uterus, uterine cervix, and vagina) were evaluated. Our main objective for this study was to look for additional neoplasms: additional neoplasms were identified which confirmed the conclusion of clear evidence of carcinogenic activity. Another objective was to determine the primary location for grossly identified adenocarcinomas in the uterine cervix and vagina: longitudinal review revealed uterus as the primary site in all cases. In addition, lesions of “atypical hyperplasia” (Figure 6), a preneoplastic lesion, were newly identified, which also supported the level of evidence for carcinogenic activity.
After evaluation of the TBBPA study, the NTP determined there were additional reasons to perform a more thorough evaluation of the female reproductive tract of the other 6 bioassays that were currently on study:
Lesions often arise in the epithelium and, due to the size of the cross sections, the available tissue for evaluation is limited.
Lesions not grossly apparent are not sampled: some masses in uterine horns cause no tissue distortion and are therefore not detected grossly (Figure 7).
Areas of dilatation seen in cross section may be part of the normal cycle, part of CEH, or may be due to polyps not captured in cross sections (Figure 8).
Decreased area for evaluation = decreased potential number of lesions.
To bolster confidence in the NTP level of evidence conclusion of equivocal, some or clear evidence of carcinogenic activity, or no evidence of carcinogenic activity. 10
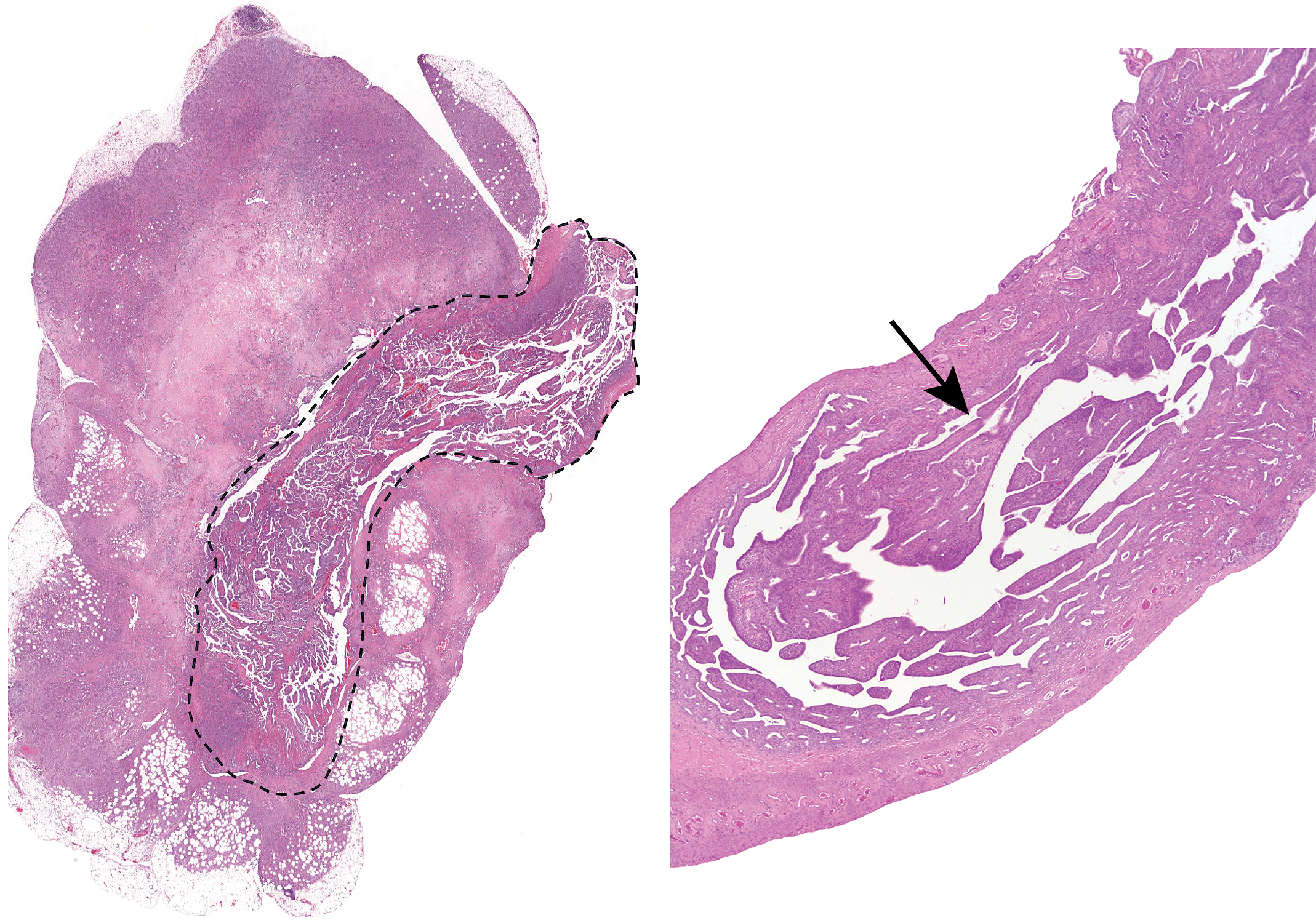
Example of a lesion that was not detected grossly. The uterus in the left panel shows a large expansive and infiltrative adenocarcinoma that was detected grossly. Dashed lines outline the uterus. The uterus in the right panel also has an adenocarcinoma (arrow), but because it did not expand the uterus, it was not detected grossly. The longitudinal sections allow for the detection of lesions, such as this, that were not seen grossly and might not have been found in the traditional sectioning method. Hematoxylin and eosin.
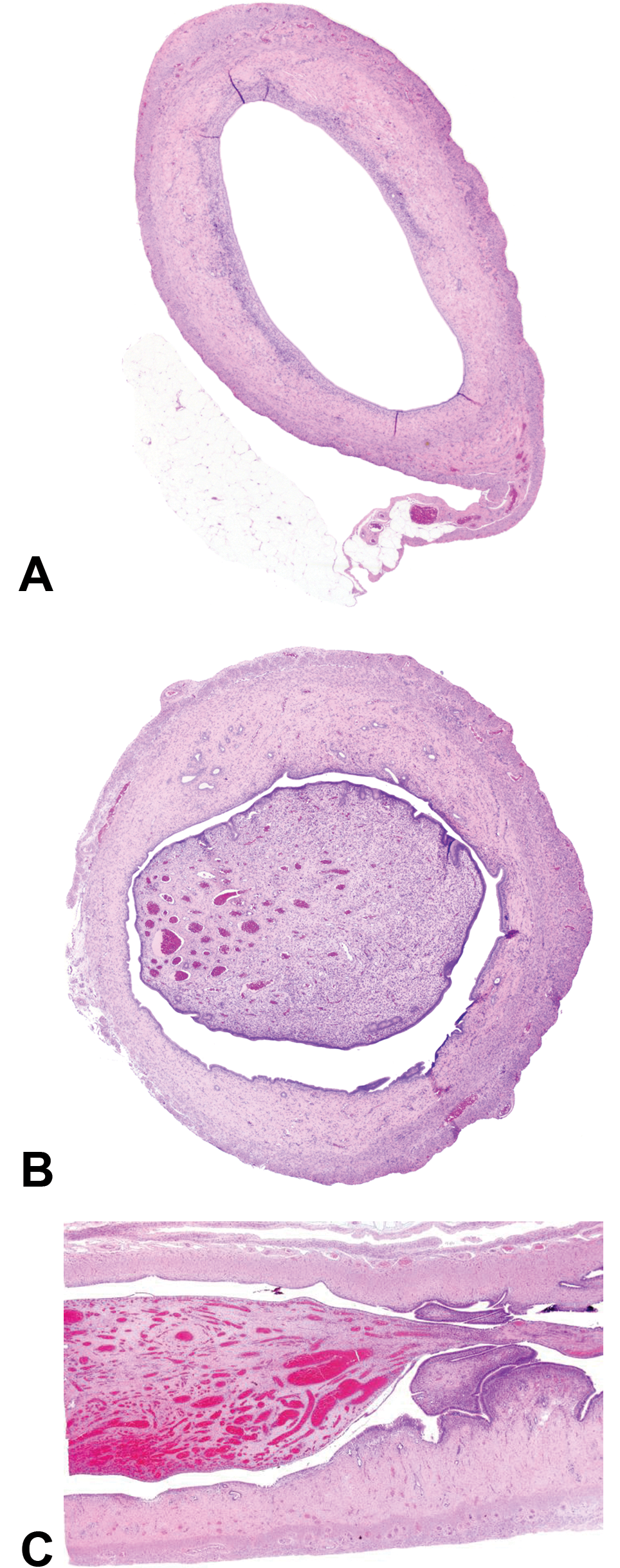
An example of uterine dilatation that might have been due to a polyp that was not detected on cross section. A, Image shows uterine dilatation with no apparent cause. B, This uterus shows uterine dilatation, but a uterine polyp is causing the dilatation. Such an unattached segment of tissue in cross section could be lost during processing. C, Longitudinal sections of uterus ensure that all lesions, such as this, are detected. Hematoxylin and eosin.
Given the advantages of the extended female reproductive tract evaluations (Table 5), all current and future NTP mouse and rat toxicity and carcinogenicity studies will be conducted with the same protocol used for current NTP DART studies (Table 6). 2 The use of this protocol allows for a more thorough evaluation of female reproductive tissues and provides a greater level of confidence for the level of evidence of carcinogenic activity. In addition, this new method of longitudinal sectioning can increase sensitivity for potential endocrine-related compounds or other concerns impacting women’s health. However, because of the potential additional costs in resources, this method might not be appropriate for the evaluations of female reproductive tissue within all institutions. Other organizations concerned with further evaluating a potential uterine response may take an ad hoc approach to determine whether longitudinal sectioning will provide additional benefit based on various criteria such as overall study goals, use of strains with a higher incidence of uterine proliferative lesions, or the signal from the initial cross section review of the uterus. This ad hoc approach is consistent with other NTP tissue evaluations such as additional sectioning of the kidney when a potential exposure response is of concern, such as an increased incidence of atypical tubular hyperplasia.
Potential Advantages of Extended Evaluation of Female Reproductive Tissues.

Current NTP Guidelines for Collection and Sectioning of Female Reproductive Organs.a

Abbreviation: NTP, National Toxicology Program.
a Specifications for the conduct of studies to evaluate the reproductive and developmental toxicity of chemical, biological, and physical agents in laboratory animals for the National Toxicology Program. 2
Some consideration toward the impact on historical control data sets should be given when considering an ad hoc or wholesale change in methods. One potential downside of an ad hoc approach would be limitations to the availability of historical control data. It should be noted that the NTP historical control database 12 has been adjusted for the newer extended evaluation. Older studies that used only the cross section evaluation are not compared to the newer studies with the longitudinal sections, and we recommend this separation of historical control data for laboratories that switch methods.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320948840 - The Assessment of Longitudinal Sections of Rat Female Reproductive Tissues for NTP 2-Year Toxicity and Carcinogenicity Studies
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320948840 for The Assessment of Longitudinal Sections of Rat Female Reproductive Tissues for NTP 2-Year Toxicity and Carcinogenicity Studies by Susan A. Elmore, Chad Blystone, Beth A. Lubeck, Shawn F. Harris and Crystal L. Johnson in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to acknowledge Ms Beth Mahler (EPL) for figure preparation. The authors would also like to acknowledge all of the NTP Study Scientists, Study Pathologists, QA Pathologists, and PWG Pathologists involved in these studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported [in part] by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences..
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
