Abstract
Ginkgo biloba extract (GBE) is a popular herbal supplement that is used to improve circulation and brain function. In spite of widespread human exposure to relatively high doses over potentially long periods of time, there is a paucity of data from animal studies regarding the toxicity and carcinogenicity associated with GBE. In order to fill this knowledge gap, 3-month and 2-year toxicity and carcinogenicity studies with GBE administered by oral gavage to B6C3F1/N mice and F344/N rats were performed as part of the National Toxicology Program’s Dietary Supplements and Herbal Medicines Initiative. The targets of GBE treatment were the liver, thyroid, and nose. These targets were consistent across exposure period, sex, and species, albeit with varying degrees of effect observed among studies. Key findings included a notably high incidence of hepatoblastomas in male and female mice and evidence of carcinogenic potential in the thyroid gland of both mice and rats. Various nonneoplastic lesions were observed beyond control levels in the liver, thyroid gland, and nose of rats and mice administered GBE. Although these results cannot be directly extrapolated to humans, the findings fill an important data gap in assessing risk associated with GBE use.
Introduction
Ginkgo biloba extract (GBE) is an alcohol extract of leaves from the Ginkgo biloba tree. The extract is generally dried to a powder and formulated into pills, tablets, or capsules for ingestion. Standardized GBE is manufactured to contain specified amounts of key components (i.e., 24% flavonol glycosides, 6% terpene lactones, and ≤5 ppm ginkgolic acids) with the aim of enriching the purported active ingredients (i.e., flavonol glycosides and terpene lactones) and limiting potentially toxic components (ginkgolic acids; van Beek and Montoro 2009). However, manufacturing processes vary across the industry, and surveys of commercially available GBE products have described a lack of conformity to specifications (Fransen et al. 2010; Kressmann, Muller, and Blume 2002). Although the suggested health benefits of GBE are generally attributed to the complex interplay of mixture constituents, favorable biological activities have been linked to 2 major constituent classes: free radical scavenging and antioxidant activity with flavonol glycosides (e.g., quercetin, kaempferol, isorhamnetin; Ding et al. 2009) and antagonism of platelet-activating factor (PAF) receptor (Braquet 1987), glycine receptor (Heads et al. 2008; Kondratskaya et al. 2002), and γ aminobutyric acid (GABAA) receptor (Huang et al. 2004) with terpene lactones (e.g., bilobalide and ginkgolides A, B, C, and J). Quercetin, a flavonol glycoside that is not unique to GBE, but is found in many plants, was previously assessed in 2-year feed studies in F344 rats, resulting in a conclusion of no evidence of carcinogenicity in females and some evidence of carcinogenicity in males based on renal tubule cell adenomas (National Toxicology Program [NTP] 1992). Ginkgolic acids have been associated with allergenic (Koch, Jaggy, and Chatterjee 2000), cytotoxic (Hecker et al. 2002), and mutagenic effects (Westendorf and Regan 2000).
GBE is an herbal supplement marketed for improving circulation and brain function. According to data from the 2007 National Health Interview Survey, approximately 3 million people in the United States are estimated to use dietary supplements labeled as Ginkgo biloba (Wu, Wang, and Kennedy 2011). However, in the largest, randomized, double-blind, placebo-controlled clinical trial of GBE (Ginkgo Evaluation of Memory Study) involving 3,069 participants aged 75 or greater, a dosage of 240 mg per day (recommended doses are generally 120–240 mg per day) was found to be ineffective in reducing the development of dementia or Alzheimer’s disease (DeKosky et al. 2008), decreasing cognitive decline (Snitz et al. 2009), reducing blood pressure or preventing hypertension (Brinkley et al. 2010), or decreasing cardiovascular disease events (Kuller et al. 2010), while there was some evidence that GBE treatment may have decreased the number of peripheral arterial disease events (Kuller et al. 2010). A secondary analysis for effects of GBE on cancer development provided mixed results, finding GBE-associated increases in incidences of breast and colorectal cancer and a decrease in prostate cancer (Biggs et al. 2010).
Although GBE is widely used, there are knowledge gaps regarding its potential to elicit toxicity and carcinogenicity. The majority of animal studies with GBE have focused on identifying and characterizing its therapeutic effects with little attention to toxicity. For example, many studies have explored the antioxidant and anticancer potential of GBE (Mahadevan and Park 2008). The few subchronic or chronic (27 weeks and 2 years) studies of the toxicity of GBE describe a general lack of effect, but provide little to no details regarding study design or results (Salvador 1995; Woerdenbag and De Smet 2000). A suspected health concern with GBE use in humans is related to its purported antagonism of PAF activity. However, there has been conflicting evidence regarding the risk of spontaneous bleeding with GBE use (Bent et al. 2005; Kellermann and Kloft 2011).
The National Toxicology Program (NTP) evaluated GBE for toxicity and carcinogenicity in 3-month and 2-year studies in F344/N rats and B6C3F1/N mice. A complete presentation of the findings from these studies can be found in the NTP (2013) technical report.
Materials and Methods
Test Article
GBE was procured from Shanghai Xing Ling Science and Technology Pharmaceutical Company (Shanghai, China) in 2 lots (lot GBE-50-001003 was used for analytical method development and lot 020703 was used for in vivo studies). Chemical characterization of the material was performed using high-performance liquid chromatography (HPLC)/ultraviolet (UV) detection and HPLC/evaporative light scattering (ELS). The α-glycosides (quercetin, isorhamnetin, and kaempferol) were quantified against the quercetin dihydrate standard in hydrolized extract using HPLC/UV, while the terpenoids (bilobalide and ginkgolides A, B, and C) were quantified against the bilobalide standard using HPLC/ELS. HPLC/mass spectrometry (MS) was used to detect colchicine and quantify the concentration of ginkgolic acids. Stability of the test article was assessed using the same HPLC methods used for characterization.
Dose formulations were prepared in National Formulary grade corn oil (multiple lots; Spectrum Chemicals and Laboratory Products; Gardena, CA) by mixing. Stability of the corn oil was periodically assessed using potentiometric titration to measure peroxide concentration (rejection level ≥ 3 mEq/kg).
Animals
All studies were conducted at the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility of Battelle (Columbus, OH) and approved by Battelle’s Institutional Animal Care and Use Committee. Male and female 4- to 5-week-old F344/N rats and B6C3F1/N mice were obtained from Taconic Farms, Inc. (Germantown, NY). Rats and mice were held for 11 to 15 days before being randomly assigned to treatment groups to achieve approximately equal mean body weights among groups. Animals were housed in polycarbonate cages with irradiated Sani-Chips hardwood chip bedding (P. J. Murphy Forest Products Corp., Montville, NJ) with 2 to 3 (2-year core study male rats) per cage or 5 (all female mice and rats and all other male rats) per cage; male mice were housed individually. Animals were identified by tail tattoo. NTP-2000 wafer feed (Zeigler Brothers, Gardners, PA) and municipal water (Columbus, OH) were provided ad libitum. The animal room was set to maintain a temperature of 72° ± 3°F and a relative humidity of 50% ± 15%.
Study Design
In all studies, animals were dosed by oral gavage 5 days per week with corn oil vehicle at dosing volumes of 2.5 ml/kg for rats and 5 ml/kg for mice. In the 3-month studies, groups of 10 males and 10 females per treatment were administered GBE at doses of 0, 62.5, 125, 250, 500, and 1,000 mg/kg for rats and 0, 125, 250, 500, 1,000, or 2,000 mg/kg for mice. In the 2-year studies, groups of 50 males and 50 females per treatment received GBE at doses of 0, 100, 300, and 1,000 mg/kg (rats) or doses of 0, 200, 400, and 2000 mg/kg (mice). A special study designed to assess thyroid hormone concentrations at 2 time points (day 22 and week 14) consisted of groups of 10 male and 10 female rats per treatment per time point and received the same doses as in the 2-year rat study for up to 14 weeks.
All animals were observed twice daily throughout the study period. Animals were weighed initially, weekly (through 13 weeks) or monthly (after week 13 of the 2-year studies), and at study termination. In the 3-month studies in rats and mice, clinical findings were recorded initially, weekly, and at the end of the study. In the 2-year studies in rats and mice, clinical findings were recorded at week 5, monthly thereafter, and at the end of the study.
Clinical chemistry (rats only) and hematology parameters were assessed during the 3-month studies. Additionally, thyroid hormones including thyroid-stimulating hormone (TSH), total triiodothyronine (T3), and total thyroxine (T4) were measured in special study male and female rats at day 22 and week 14 during the 2-year study. In all studies, blood was collected from the retroorbital plexus of rats or retroorbital sinus of mice following anesthesia with a mixture of 70% carbon dioxide to 30% oxygen and placed into tubes containing ethylenediaminetetraacetic acid (EDTA) for hematology determination (rats and mice) and into serum separator tubes for clinical chemistry and thyroid hormone determination (rats only). Hematological parameters measured were hemoglobin, hematocrit, red blood cell count, reticulocytes, mean cell volume, mean cell hemoglobin, mean cell hemoglobin concentration, platelets, white blood cell count, and white blood cell differential. Clinical chemistry parameters measured were urea nitrogen, creatinine, glucose, total protein, albumin, alanine transferase, alkaline phosphatase, creatine kinase, sorbitol dehydrogenase, and bile salts.
Animals were euthanized by carbon dioxide (100%) followed by necropsy. In the 3-month study, weights were recorded for the heart, right kidney, liver, lung, right testis, and thymus and complete histopathological examinations were performed on all vehicle control and high-dose rats (1,000 mg/kg) and mice (2,000 mg/kg). For histopathological examination, tissues were fixed and preserved in 10% neutral buffered formalin with the exception of eyes, which were first fixed in Davidson’s solution. Tissues were then processed, trimmed, and embedded in paraffin, sectioned (4–6 μm), and stained with hematoxylin and eosin. In the 2-year study, complete histopathological examinations were performed on all rats and mice, except the special study rats for which the liver and thyroid were examined exclusively.
Statistical Analysis
Mean body and organ weights were compared among groups using Dunnett’s or Williams’s test depending on whether a dose-related trend was observed using Jonckheere’s trend test. Clinical chemistry and thyroid hormone data, which are typically not normally distributed, were compared among groups using Dunn’s or Shirley’s test, depending on whether a dose-related trend was observed. Incidences of neoplastic and nonneoplastic lesions are presented as the number of animals affected with each lesion along with the total number of animals examined for that lesion. Dose-related trends of lesion incidences and pairwise comparisons of each dose group with the control group were assessed using the Cochran–Armitage trend test and Fisher’s exact test for the 3-month studies and the Poly-3 test (Bailer and Portier 1988; Piegorsch and Bailer 1997; Portier and Bailer 1989) for the 2-year studies in which survival needed to be taken into account. p values are one-sided and are considered significant if less than .05.
Results
3-Month Studies in Rats
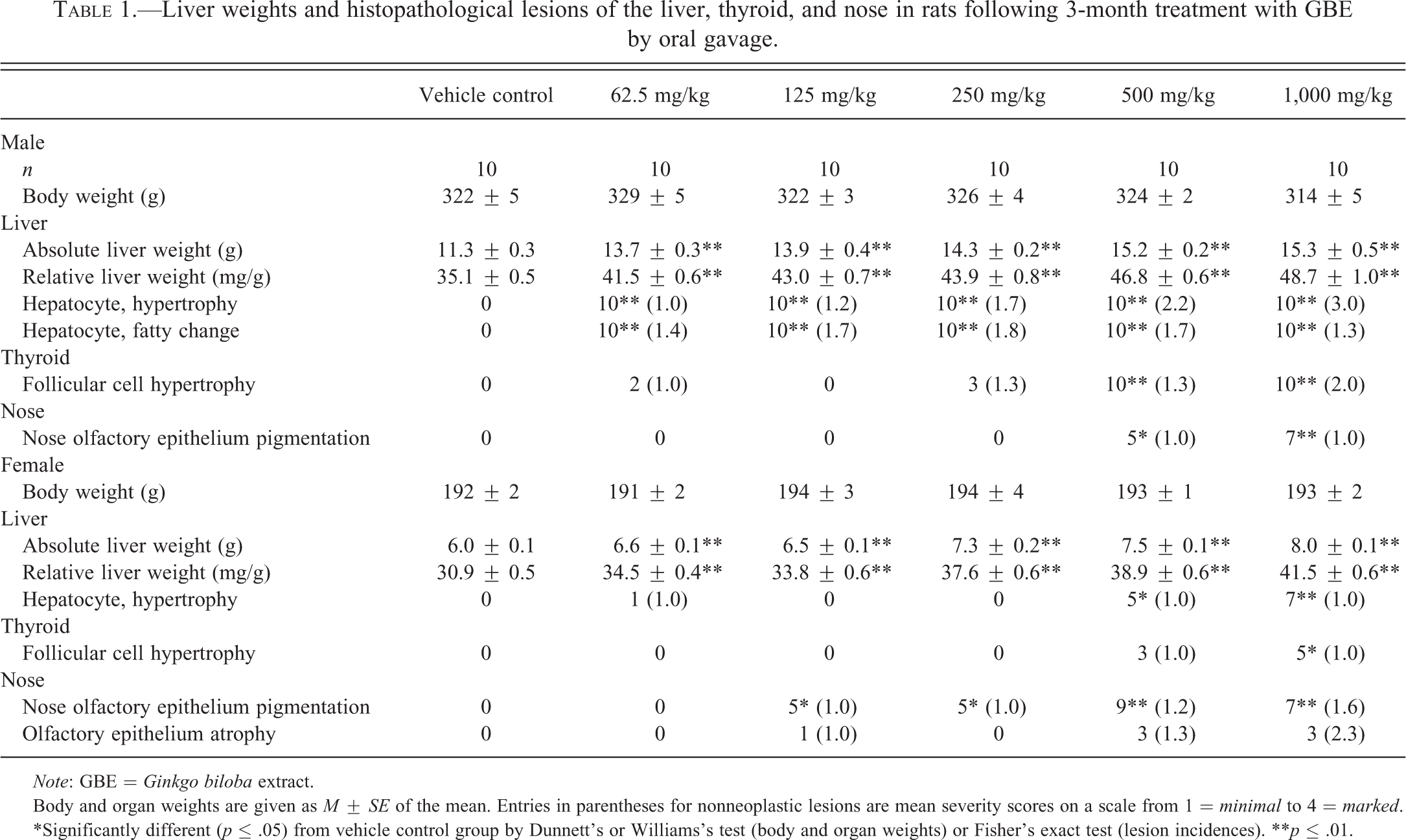
GBE treatment up to 1,000 mg/kg did not affect mortality or body weight in male or female rats. Absolute and relative liver weights were significantly increased in a generally dose-related manner compared to controls at all doses of GBE in the 3-month study (Table 1). There were some statistically significant changes in clinical chemistry parameters, including a decrease in alanine aminotransferase, alkaline phosphatase, and bile salts at week 14 in males and females, as well as increases in total protein and albumin in male rats only (data not shown). Hepatocyte hypertrophy was observed in all treated male rats and the incidence increased in a dose-related manner in female rats (Table 1). The hypertrophy began in centrilobular areas and expanded to the midzonal areas. There was a dose-related increase in severity from minimal to moderate in males (Table 1). Hepatocytes were enlarged up to approximately 1½ times the normal size with increased cytoplasm and slightly enlarged nuclei. Hepatocyte fatty change was observed in all treated male rats (Table 1). Fatty change consisted of small numbers of scattered midzonal hepatocytes having small to a few large, clear, discrete intracytoplasmic vacuoles that had the typical appearance of lipid droplets filling most or all of the cytoplasm.
Liver weights and histopathological lesions of the liver, thyroid, and nose in rats following 3-month treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Body and organ weights are given as M ± SE of the mean. Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
*Significantly different (p ≤ .05) from vehicle control group by Dunnett’s or Williams’s test (body and organ weights) or Fisher’s exact test (lesion incidences). **p ≤ .01.
In both sexes at higher dose levels, there was a dose-related increase in incidences of thyroid gland follicular cell hypertrophy, which was of minimal to mild severity in males and minimal severity in females (Table 1). The hypertrophy was characterized by an increase in size of follicular cells with the affected cells assuming a tall cuboidal to low columnar shape. Enlarged follicular cells occasionally formed small protrusions into the lumen. An increase in clear vacuolation (resorption vacuoles) was observed in the follicular cell cytoplasm due to increased resorptions of colloid. Peripheral follicles in the affected thyroid glands were usually lined by large cuboidal cells indicating activity, as compared with the dilated, flattened epithelial cells lining the peripheral follicles of control thyroid glands.
There was a dose-related increase in pigmentation of the olfactory epithelium of the nose in males (≥500 mg/kg) and females (≥125 mg/kg; Table 1). Pigment accumulation appeared as golden brown pigment within macrophages scattered throughout the basal aspect of the olfactory epithelium. Although it did not reach the .05 level of significance, there was a slight increase in the incidence and severity of atrophy in the olfactory epithelium of the nose in female rats.
GBE doses were selected for the 2-year rat studies based on results from the 3-month studies described above. A high dose of 1,000-mg/kg GBE was selected based on a lack of effect of the high dose on survival or body weights combined with the determination that the hypertrophy observed in histopathologic assessment of the livers and thyroid glands of rats in the 3-month studies was not considered to be life threatening; and the dose spacing was increased to half-log intervals in order to capture a range of effect levels.
3-Month Studies in Mice
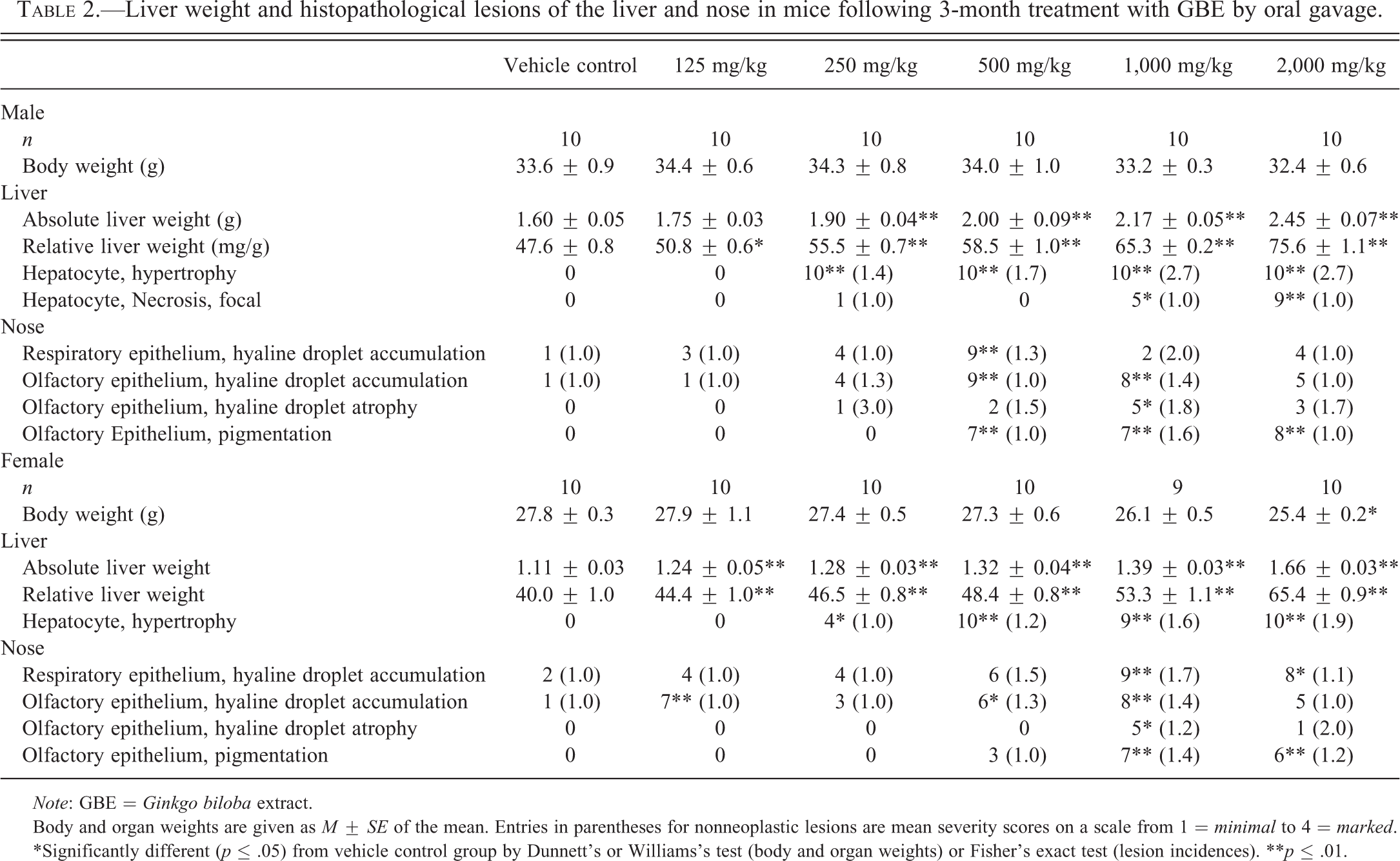
GBE treatment up to 2,000 mg/kg did not affect mortality in male or female mice or body weight in male mice. In female mice, body weight was significantly lower in the 2,000-mg/kg group than in the control group (Table 2). There was a significant, dose-related increase in absolute and relative liver weights in all dose groups of males and females compared to controls, with the exception of absolute liver weights in 125-mg/kg males (Table 2). There was also a dose-related increase in incidences and severity of hepatocyte hypertrophy in males and females (≥250 mg/kg; Table 2). The presentation of hepatocyte hypertrophy in mice was similar to that described above in rats. A significant increase in focal hepatic necrosis was observed in the top 2 doses of GBE in male mice (Table 2). Focal hepatocytic necrosis was minimal in male mice and presented as occasional, minute foci of hepatocytes with brightly eosinophilic cytoplasm and pyknotic or fragmented nuclei.
Liver weight and histopathological lesions of the liver and nose in mice following 3-month treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Body and organ weights are given as M ± SE of the mean. Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
*Significantly different (p ≤ .05) from vehicle control group by Dunnett’s or Williams’s test (body and organ weights) or Fisher’s exact test (lesion incidences). **p ≤ .01.
Incidences of nonneoplastic nasal lesions in male and female mice generally increased in GBE-treated animals (Table 2). Histopathological lesions observed in the nose of male and female mice included hyaline droplet accumulation in the respiratory and olfactory epithelium, hyaline droplet atrophy in the olfactory epithelium, and pigmentation in the olfactory epithelium (Table 2).
GBE doses were selected for the 2-year mice studies based on results from the 3-month studies described above. Body and organ weight changes and liver hypertrophy observed in the 3-month studies were not considered to be life threatening; therefore, a high dose of 2,000-mg/kg GBE was selected and the dose spacing was increased to half-log intervals in order to capture a range of effect levels.
2-Year Studies in Rats
There was a significant 58% decrease in survival in the 1,000-mg/kg male rats as compared to controls; survival in dosed groups of females was similar to controls (data not shown). Most deaths occurred late in the study (after day 90) and the most frequent cause of early death in male rats in the 1,000-mg/kg group was mononuclear cell leukemia. Incidences of mononuclear cell leukemia in male rats were 9 (18%), 12 (24%), 22 (44%), and 21 (42%) of the 50 in the vehicle control, 100-mg/kg group, 300-mg/kg group, and 1,000-mg/kg group, respectively. The incidences in the male 300-mg/kg and 1,000-mg/kg GBE-treated groups were significantly greater than in the vehicle control and exceeded the historical control range for corn oil gavage studies (8–28%) but not for all routes of exposure (8–58%). Mean body weights of 300- and 1,000-mg/kg male and female rats were less (at least 10% lower) than those of controls starting at week 93 and 89, respectively.
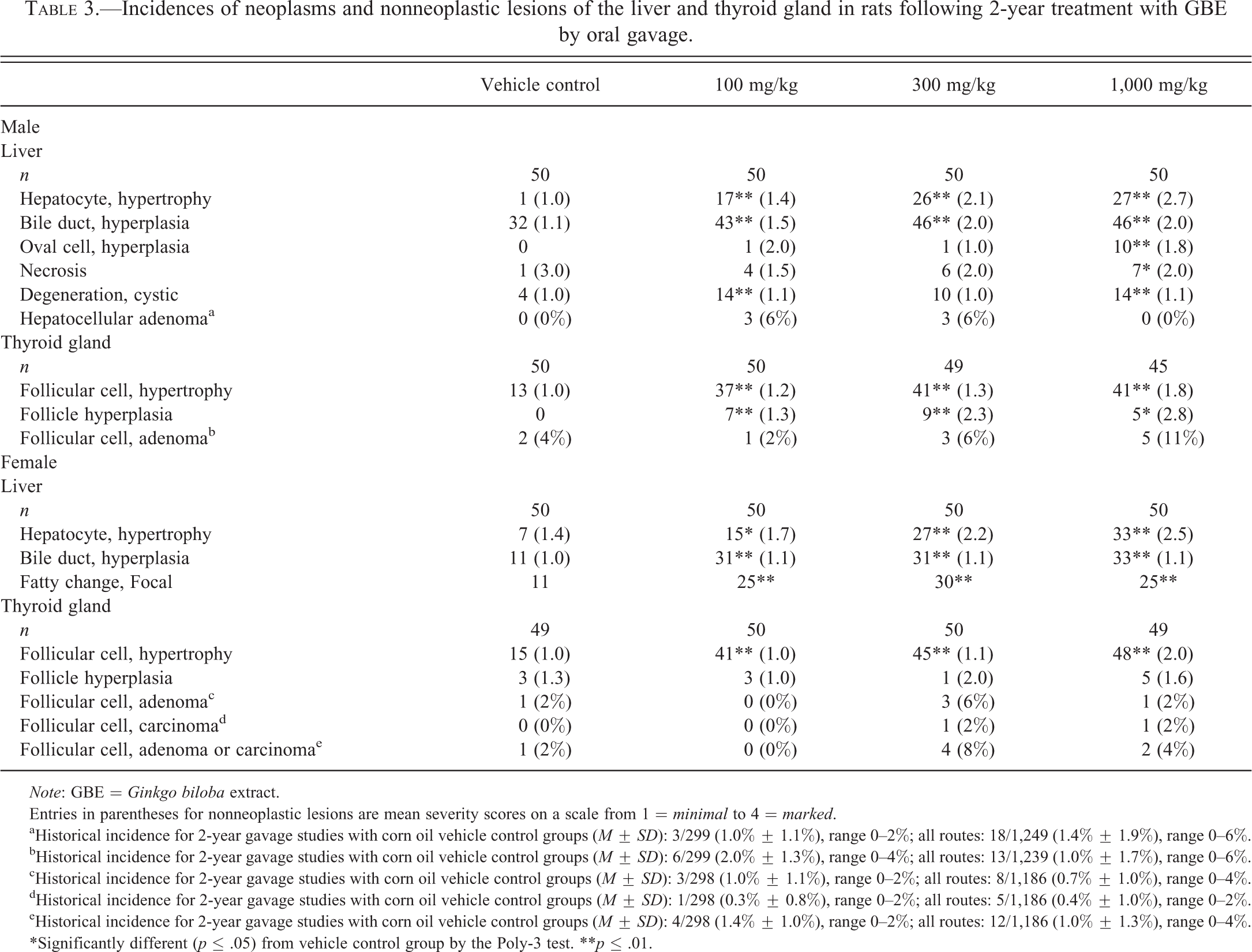
Incidences of hepatocellular adenoma in the 100- and 300-mg/kg males were not significantly different from controls, but exceeded the historical control range for corn oil gavage studies (Table 3). The hepatocellular adenomas were generally well circumscribed, composed of well-differentiated hepatocytes that varied in size and characteristics (i.e., eosinophilic, basophilic, clear, vacuolated, or an admixture). Histopathological lesions significantly increased in the livers of GBE-treated rats included hepatocyte hypertrophy and bile duct hyperplasia in males and females, oval cell hyperplasia, necrosis, and cystic degeneration in males, as well as focal fatty change in females (Table 3 and Figure 1).
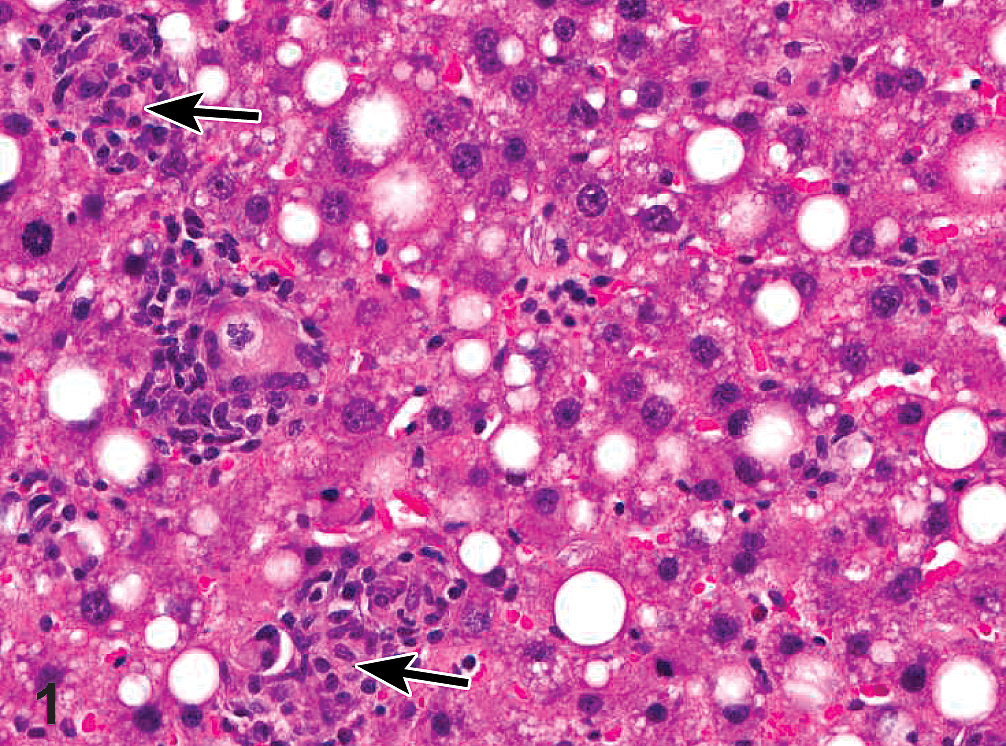
Photomicrograph of H&E-stained liver section showing focal fatty change in the liver of a female F344/N rat administered 1,000-mg/kg GBE by gavage for 2 years. Note hepatocytes displaying microvesicular and macrovesicular fatty change associated with microgranulomas (arrows) scattered throughout the lesion and composed predominantly of macrophages with fewer lymphocytes, plasma cells, and occasional neutrophils. The macrophages often contained fine, acicular clefts (cholesterol clefts).
Incidences of neoplasms and nonneoplastic lesions of the liver and thyroid gland in rats following 2-year treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
aHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 3/299 (1.0% ± 1.1%), range 0–2%; all routes: 18/1,249 (1.4% ± 1.9%), range 0–6%.
bHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 6/299 (2.0% ± 1.3%), range 0–4%; all routes: 13/1,239 (1.0% ± 1.7%), range 0–6%.
cHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 3/298 (1.0% ± 1.1%), range 0–2%; all routes: 8/1,186 (0.7% ± 1.0%), range 0–4%.
dHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 1/298 (0.3% ± 0.8%), range 0–2%; all routes: 5/1,186 (0.4% ± 1.0%), range 0–2%.
eHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 4/298 (1.4% ± 1.0%), range 0–2%; all routes: 12/1,186 (1.0% ± 1.3%), range 0–4%.
*Significantly different (p ≤ .05) from vehicle control group by the Poly-3 test. **p ≤ .01.
Although not significant, the incidence of follicular cell adenoma of the thyroid gland was increased in 1,000-mg/kg males and 300-mg/kg females, and there was a single incidence of follicular cell carcinoma in each of the 300- and 1,000-mg/kg female groups (Table 3). The incidences of follicular cell adenoma in 1,000-mg/kg male rats and follicular cell adenoma and carcinoma in 300-mg/kg female rats exceeded the historical control range for all routes of administration (Table 3). In addition to these neoplasms, incidences of follicular cell hyperplasia were increased in all groups of GBE-treated males and incidences of follicular cell hypertrophy were increased in all groups of GBE-treated males and females (Table 3; Figure 2). Follicular cell hyperplasia was characterized by follicles lined by a crowded cuboidal to low columnar single-cell layer epithelium. Occasional follicles were enlarged with increased colloid, while other hyperplastic follicles were small with decreased colloid. In some follicles, the epithelium formed papillary projections that protruded into the follicular lumen. Hyperplasia was usually focal to multifocal and only rarely involved the majority of follicles. In no cases were all of the follicles in a gland hyperplastic (diffuse). Hyperplastic foci were not encapsulated and there was no to minimal compression of the adjacent normal thyroid follicles. Atypia and mitotic figures were not noted. Thyroid follicular cell adenomas were well circumscribed, nonencapsulated masses formed by the proliferation of follicular cells within the thyroid gland. They were small and generally there was minimal compression of the surrounding follicles. The neoplastic cells were generally hyperchromatic and formed colloid-containing follicles of variable size. Neoplastic cells formed papillary projections with complex branching protruding into lumens of larger cystically dilated follicles. The neoplastic cells varied from cuboidal to columnar in shape and there was often an increase in the nuclear to cytoplasm ratio. Follicular cell carcinomas were typically large lesions that were highly cellular, although small follicles with pale colloid were typically present throughout the neoplasms. Invasion was present in some neoplasms. Mitotic activity and cellular atypia were present in many carcinomas.
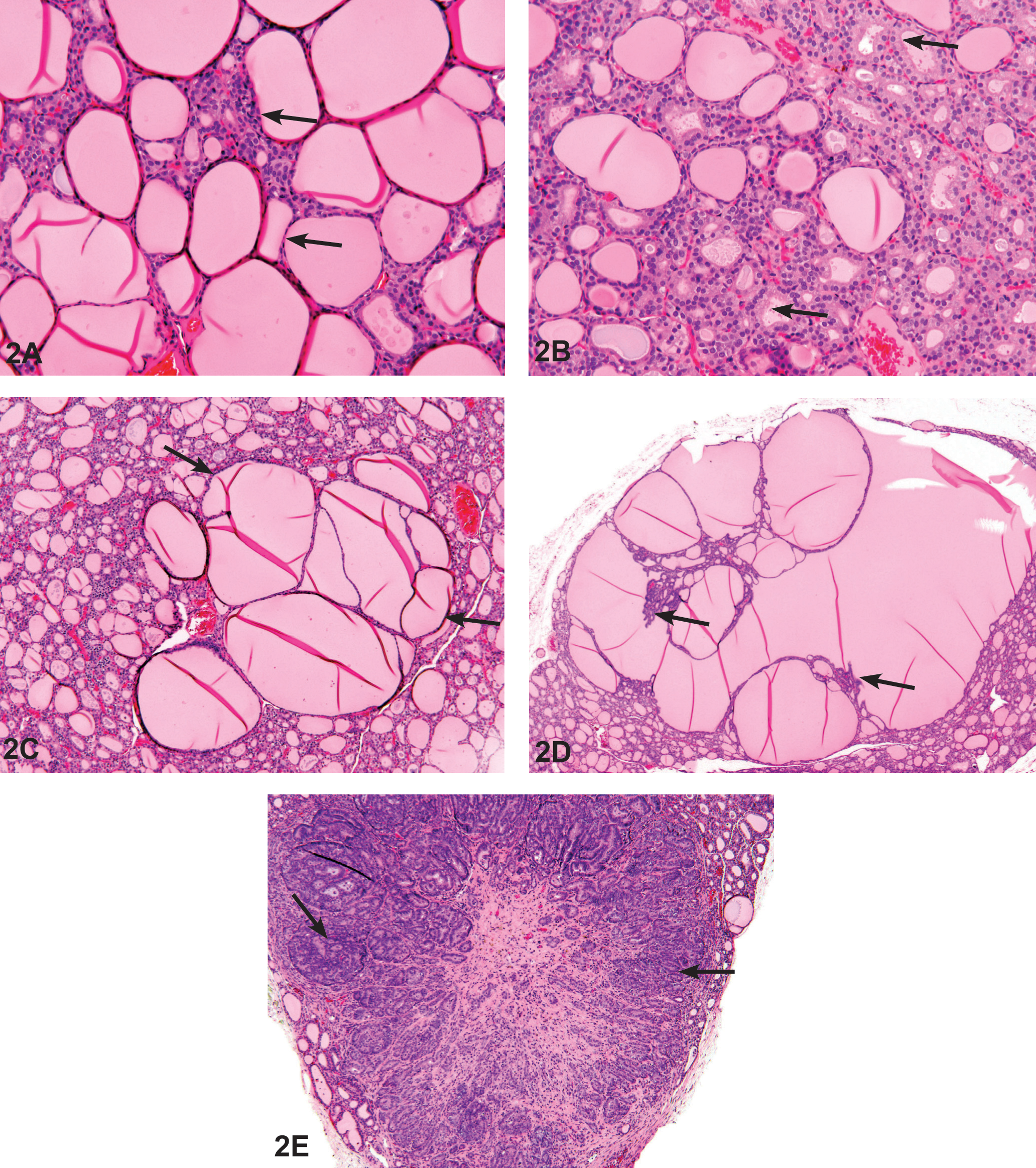
Photomicrograph of H&E-stained thyroid gland sections taken from 2-year F344/N rat study of GBE. (A) Normal aspect of thyroid gland follicles from a male vehicle control F344/N rat. Most of the follicles are lined by flattened epithelium (arrows), and the follicles are distended with homogeneous colloid. (B) Thyroid gland follicular cell hypertrophy in a female F344/N rat administered 1,000-mg/kg GBE. Most of the follicles are lined by cuboidal epithelium (arrows), and there is a decreased amount of colloid as compared to (A). (C) Thyroid gland follicular cell hyperplasia in a male F344/N rat administered 1,000-mg/kg GBE. Note the focal enlargement of follicles that typically compress the surrounding parenchyma (arrows). The follicles are lined by hyperplastic follicular epithelium and epithelial lined septae, and papillary projections frequently project into the follicular colloid. (D) Thyroid gland follicular cell adenoma in a male F344/N rat administered 1,000-mg/kg GBE. The adenoma tends to be larger with more compression of the adjacent parenchyma and more complex epithelial infoldings than in hyperplasia (arrows). (E) Thyroid gland follicular cell carcinoma in a female F344/N rat administered 300-mg/kg GBE. The neoplasm is highly cellular (arrows) with local invasion. GBE = Ginkgo biloba extract.
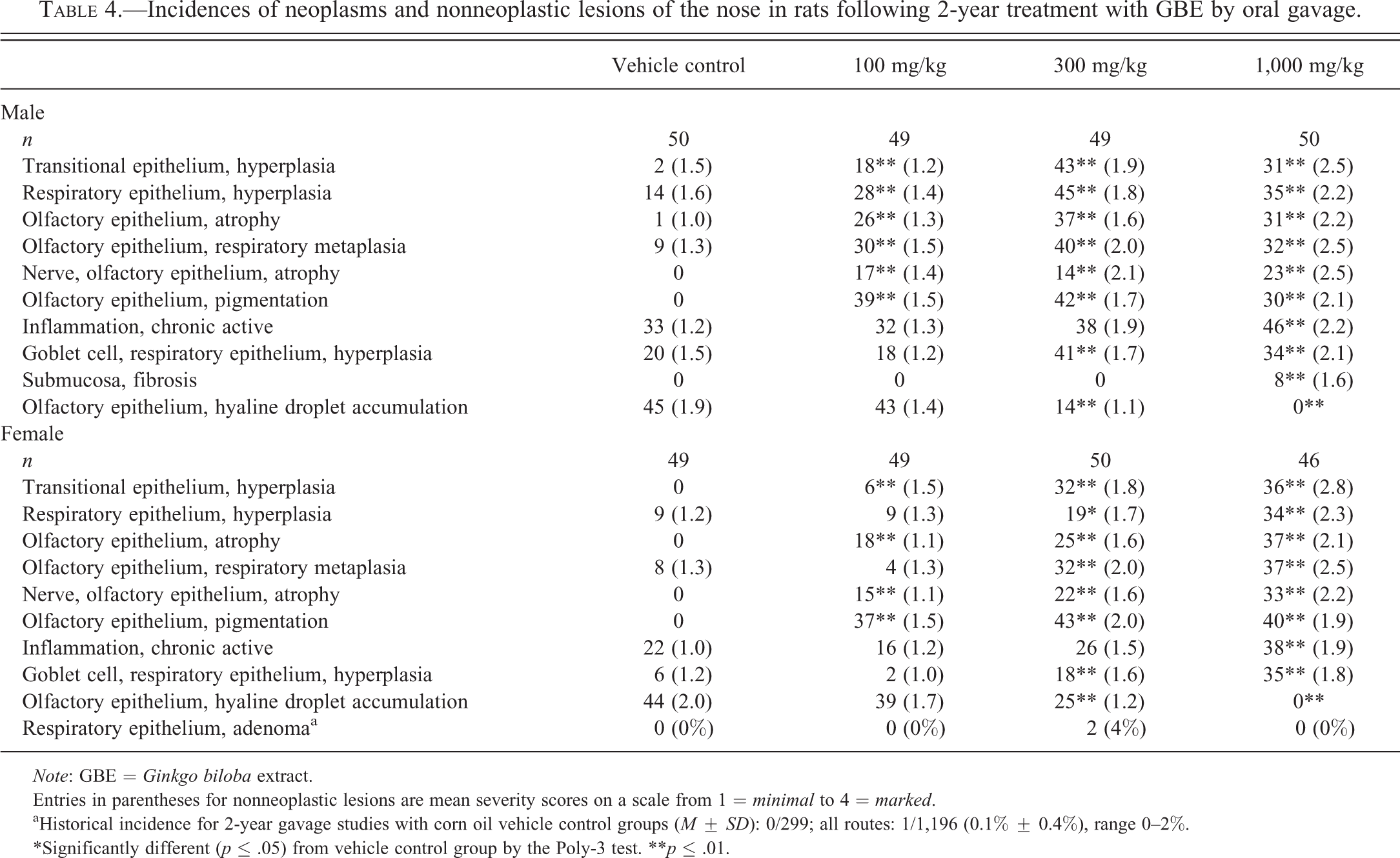
Multiple nonneoplastic lesions were observed in the nose of male and female rats exposed to GBE for 2 years (Table 4). Two 300-mg/kg females had respiratory epithelium adenomas, which is above the historical control range for all routes of administration (Table 4). Dose-related increases of the following nonneoplastic lesions were observed: hyperplasia in transitional and respiratory epithelium, atrophy and metaplasia in the olfactory epithelium, atrophy of nerves in the olfactory epithelium, pigmentation of the olfactory epithelium, chronic active inflammation, and hyperplasia of goblet cells in the respiratory epithelium (Table 4 and Figure 3). Fibrosis in the submucosa was increased in 1,000-mg/kg males. There was a dose-related decrease in hyaline droplet accumulation in the olfactory epithelium of both males and females (Table 4).
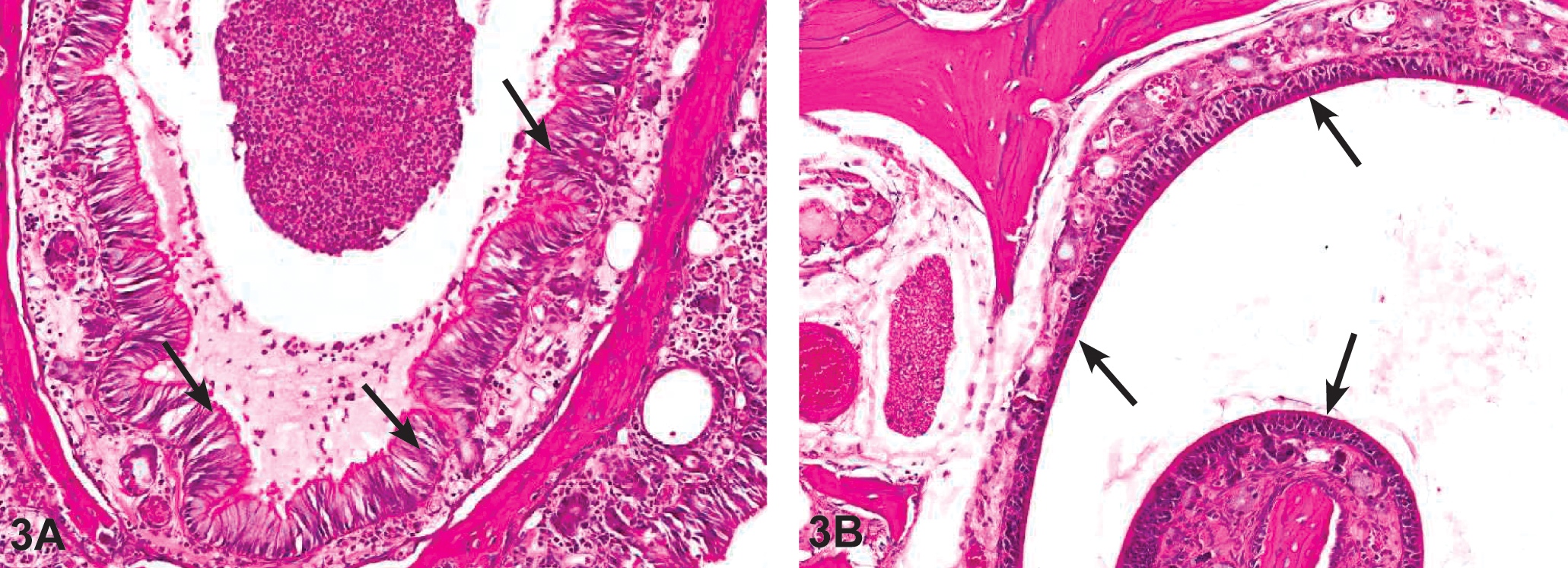
(A) Photomicrograph of H&E-stained nasal tissue sections taken from 2-year F344/N rat study of GBE. Chronic active inflammation in the nose (level III) of a female F344/N rat administered 1,000-mg/kg GBE. Note the presence of mixed inflammatory infiltrate that involves the submucosa and the lumen of the nasal cavity. There is also respiratory metaplasia of the olfactory epithelium (arrows). (B) Atrophy of the olfactory epithelium in the nose (level III) of a female F344/N rat administered 1,000-mg/kg GBE. Note the thinning and disorganization of the olfactory epithelial layer (arrows). GBE = Ginkgo biloba extract.
Incidences of neoplasms and nonneoplastic lesions of the nose in rats following 2-year treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
aHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 0/299; all routes: 1/1,196 (0.1% ± 0.4%), range 0–2%.
*Significantly different (p ≤ .05) from vehicle control group by the Poly-3 test. **p ≤ .01.
Special Study in Rats
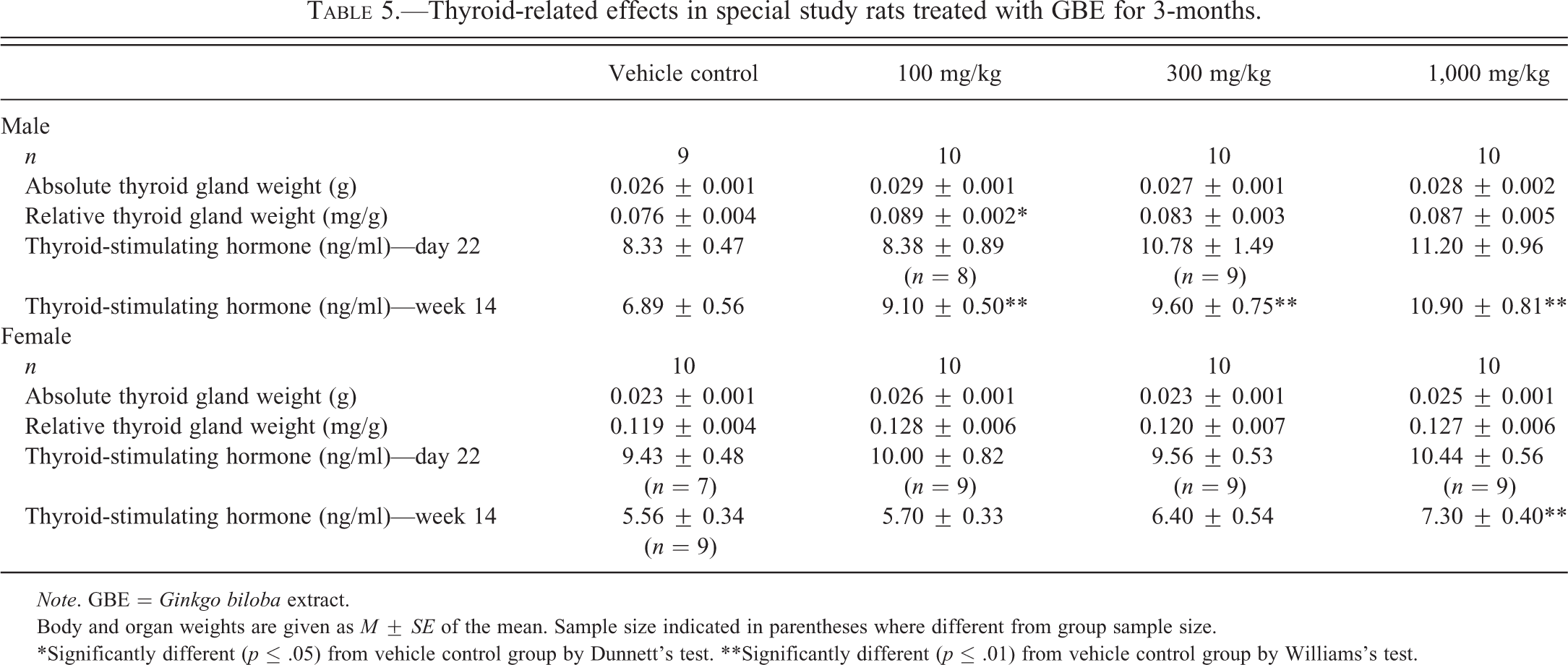
As observed in the 3-month studies, absolute and relative liver weights at week 14 in the special study were significantly greater in male and female rats treated with GBE than in the vehicle controls (data not shown). Thyroid gland weight of GBE-treated animals did not differ significantly from controls (Table 5). However, there was a dose-related increase in TSH measured at week 14 that reached statistical significance in the 100-mg/kg group and greater males and 1,000-mg/kg females (Table 5).
Thyroid-related effects in special study rats treated with GBE for 3-months.
Note. GBE = Ginkgo biloba extract.
Body and organ weights are given as M ± SE of the mean. Sample size indicated in parentheses where different from group sample size.
*Significantly different (p ≤ .05) from vehicle control group by Dunnett’s test. **Significantly different (p ≤ .01) from vehicle control group by Williams’s test.
2-Year Studies in Mice
Survival of 600- and 2,000-mg/kg males was significantly lower than controls, with the majority of deaths from liver tumors (data not shown). Survival of 600-mg/kg females was significantly higher than that of controls. Body weight of 2,000-mg/kg females was 7 to 13% less than that of controls beginning at week 20.
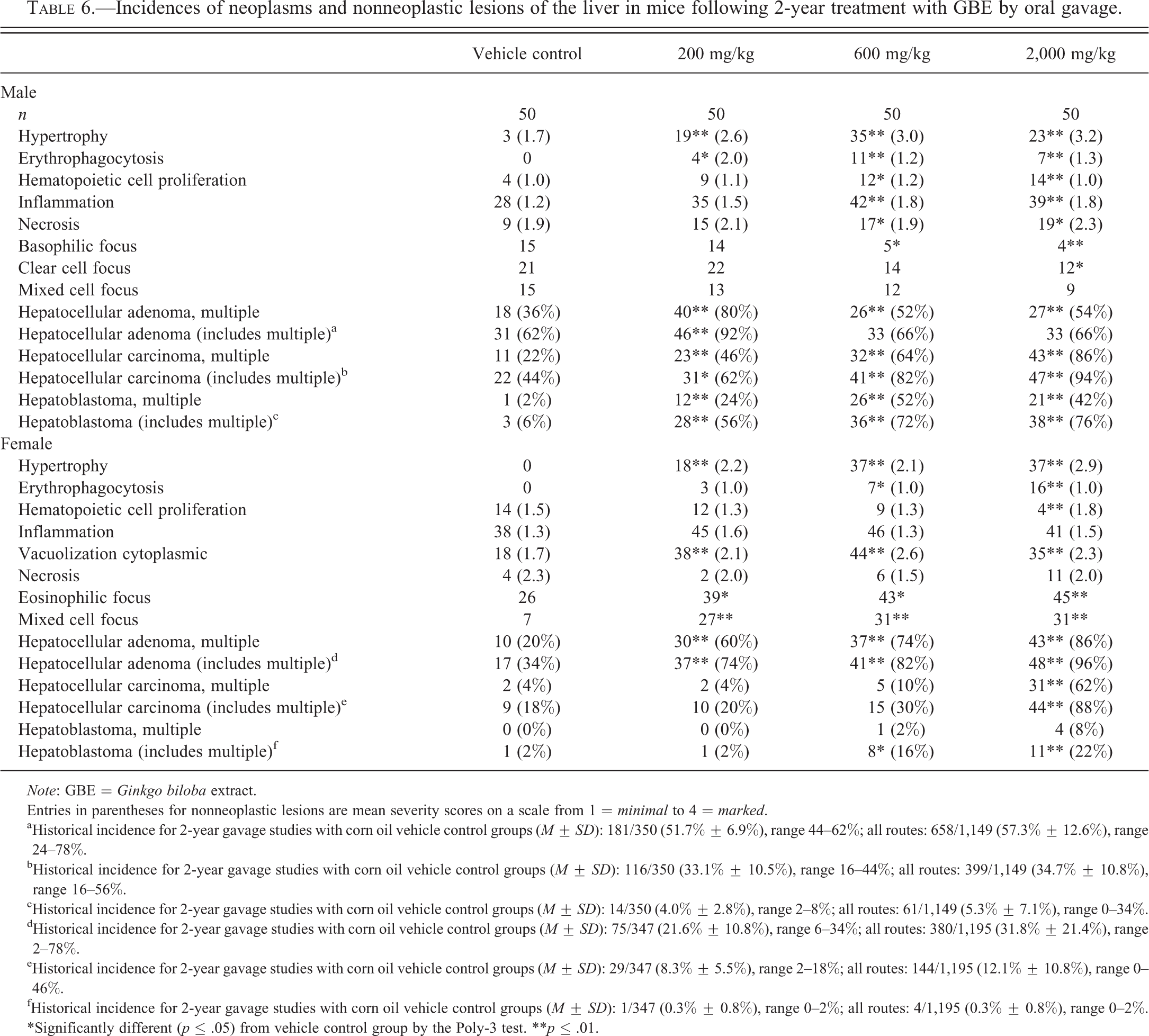
For hepatocellular adenoma, hepatocellular carcinoma, and hepatoblastoma of the liver, the incidences of animals having multiple tumors were increased in treated male and female mice. In males, the incidences of multiple hepatocellular adenoma, singular or multiple hepatocellular carcinoma, and singular or multiple hepatoblastoma (Figure 4) were significantly increased in all GBE-treated groups (Table 6). In females, incidences of multiple hepatocellular adenoma were significantly increased in all GBE-treated groups and the incidences of multiple hepatocellular carcinoma were significantly increased in the 2,000-mg/kg group (Table 6). In females, incidences of multiple hepatoblastoma, while not significantly higher than controls, increased in a dose-related manner (Table 6). Whether single or multiple in each animal, incidences of hepatocellular adenoma in all GBE-treated females, hepatocellular carcinoma in 2,000-mg/kg females, and hepatoblastoma in 600- and 2,000-mg/kg females were significantly higher than in controls. In addition to the neoplastic lesions, nonneoplastic liver lesions were observed. Nonneoplastic lesions that increased with dose of GBE included hepatocyte hypertrophy, erythrophagocytosis, hematopoietic cell proliferation (increased in males, decreased in females), inflammation (males only), cytoplasmic vacuolization (females only), and necrosis (males only; Table 6). Significant decreases in the incidences of basophilic focus and clear cell focus were observed in males, while incidences of eosinophilic focus and mixed cell focus were increased in females.
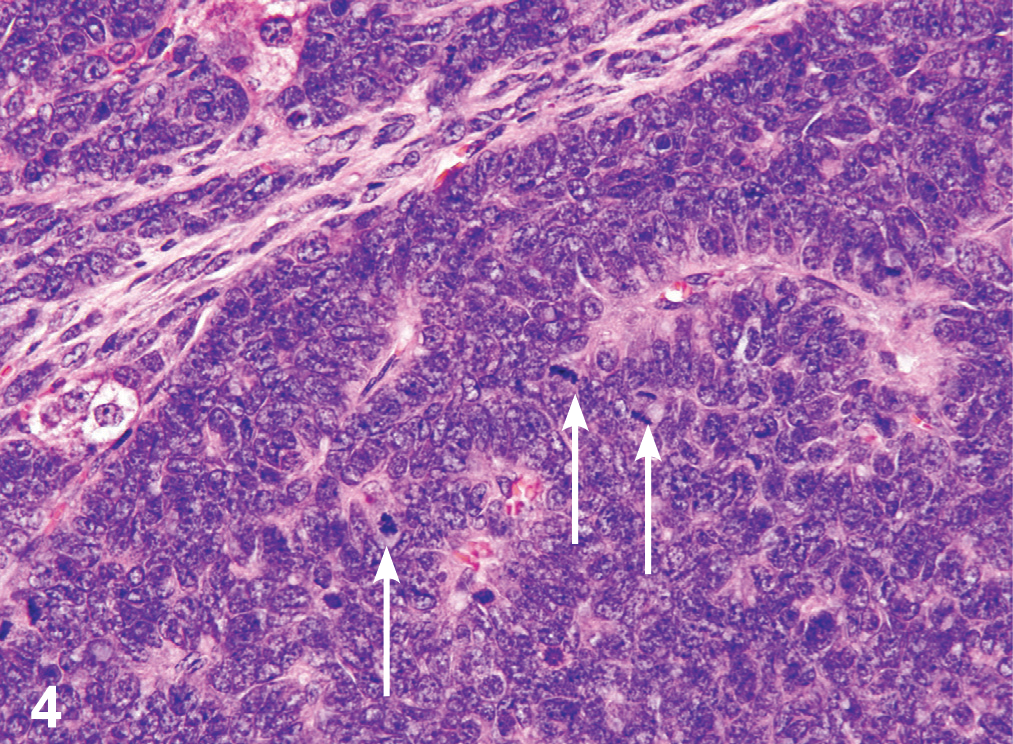
Photomicrograph of H&E-stained liver section showing a hepatoblastoma taken from a male B6C3F1/N mouse administered 200-mg/kg GBE by gavage for 2 years. Note the basophilic neoplastic cells arranged in sheets with palisading around vascular spaces. Nuclei are generally irregularly oval to round with a scant amount of basophilic cytoplasm; mitotic figures are numerous (white arrows).
Incidences of neoplasms and nonneoplastic lesions of the liver in mice following 2-year treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
aHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 181/350 (51.7% ± 6.9%), range 44–62%; all routes: 658/1,149 (57.3% ± 12.6%), range 24–78%.
bHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 116/350 (33.1% ± 10.5%), range 16–44%; all routes: 399/1,149 (34.7% ± 10.8%), range 16–56%.
cHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 14/350 (4.0% ± 2.8%), range 2–8%; all routes: 61/1,149 (5.3% ± 7.1%), range 0–34%.
dHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 75/347 (21.6% ± 10.8%), range 6–34%; all routes: 380/1,195 (31.8% ± 21.4%), range 2–78%.
eHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 29/347 (8.3% ± 5.5%), range 2–18%; all routes: 144/1,195 (12.1% ± 10.8%), range 0–46%.
fHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 1/347 (0.3% ± 0.8%), range 0–2%; all routes: 4/1,195 (0.3% ± 0.8%), range 0–2%.
*Significantly different (p ≤ .05) from vehicle control group by the Poly-3 test. **p ≤ .01.
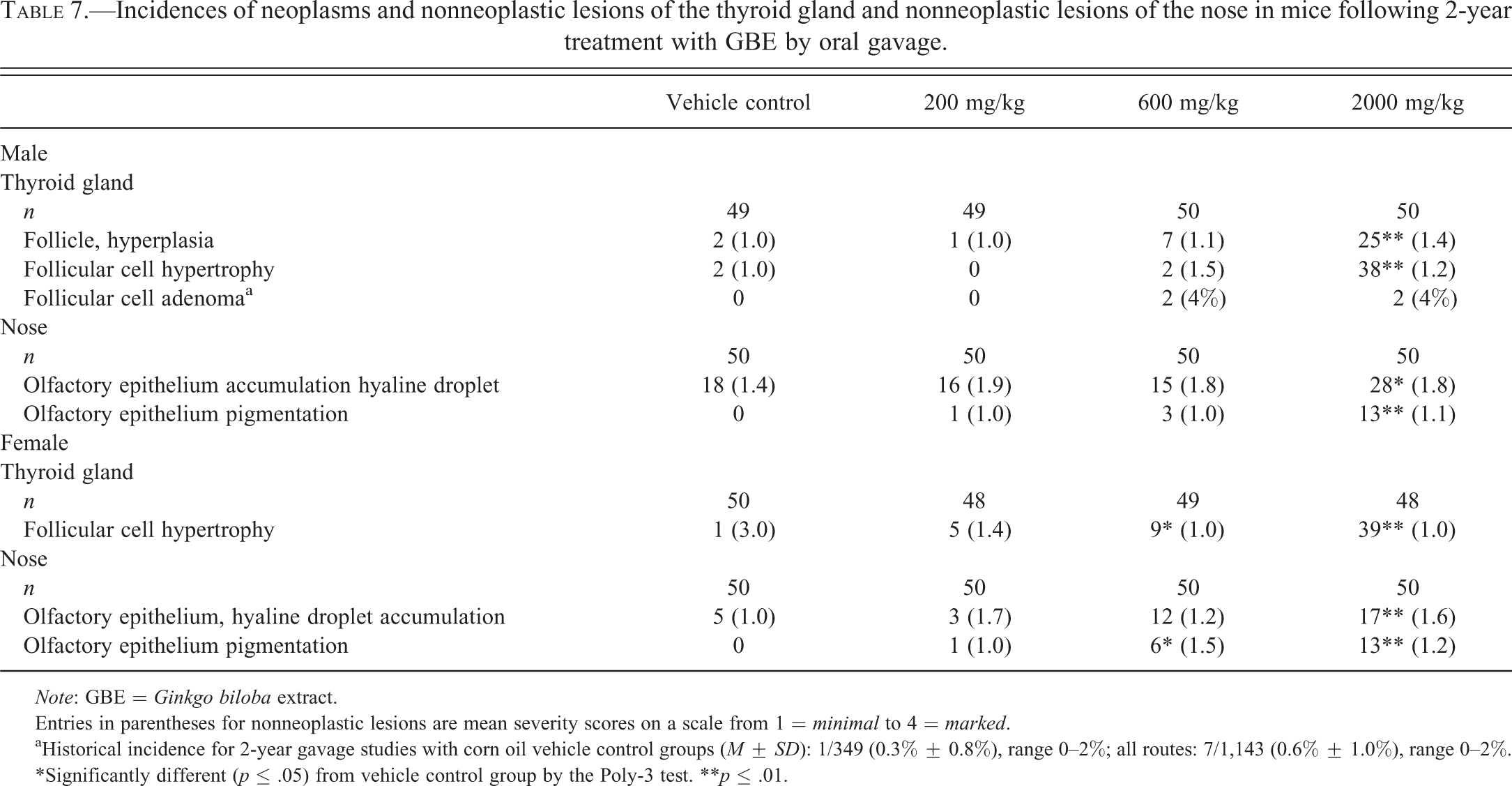
Two incidences of thyroid gland follicular cell adenoma occurred in each of the 600- and 2,000-mg/kg male groups, exceeding the historical control range for all routes of administration (Table 7). Additionally, the incidences of follicle hyperplasia and follicular cell hypertrophy were increased in the 2,000-mg/kg male group and the incidence of follicular cell hypertrophy was increased in the 600- and 2,000-mg/kg female groups (Table 7).
Incidences of neoplasms and nonneoplastic lesions of the thyroid gland and nonneoplastic lesions of the nose in mice following 2-year treatment with GBE by oral gavage.
Note: GBE = Ginkgo biloba extract.
Entries in parentheses for nonneoplastic lesions are mean severity scores on a scale from 1 = minimal to 4 = marked.
aHistorical incidence for 2-year gavage studies with corn oil vehicle control groups (M ± SD): 1/349 (0.3% ± 0.8%), range 0–2%; all routes: 7/1,143 (0.6% ± 1.0%), range 0–2%.
*Significantly different (p ≤ .05) from vehicle control group by the Poly-3 test. **p ≤ .01.
In the nose of mice, the incidence of hyaline droplet accumulation was increased in the 2,000-mg/kg males and females and the incidence of olfactory epithelium pigmentation was increased in 2,000-mg/kg males and females and in 600-mg/kg females (Table 7).
Discussion
GBE was assessed for toxicity and carcinogenicity in 3-month and 2-year oral gavage studies in F344/N rats and B6C3F1/N mice as part of the NTP’s Dietary Supplements and Herbal Medicines Initiative. Herbal supplements represent a particular challenge in terms of test article selection. They are inherently complex mixtures and can vary depending on environmental factors and manufacturing procedures. Therefore, it is difficult to select a single test article that reflects the entire market share. Considerations in selecting an appropriate test article include presence and concentrations of marker compounds or active components (if the active components are known), lack of evidence of adulteration, similar composition to other products in the same category, and availability in bulk quantity.
The certificate of analysis provided by the manufacturer of the GBE used in the present studies specified a minimum of 24% flavonol glycosides and 6% terpene lactones, along with no more than 5-ppm ginkgolic acids. These values reflect recommended target concentrations detailed in the U.S Pharmacopeia (2009), as well as the German Commission E Monographs on herbal medicine, which specifies ranges of 22 to 27% flavonol glycosides, 5 to 7% terpene lactones, and ≤5-ppm ginkgolic acids [English translation (Blumenthal 1998)]. Actual concentrations measured in the current study were 31.2% flavonol glycosides, 15.4% terpene lactones, and 10.45 ± 2.40-ppm ginkgolic acids. Although these values are higher than the specifications, the levels of flavonol glycosides and ginkgolic acids fall well within the range of concentrations measured in market surveys of GBE (Fransen et al. 2010; Gawron-Gzella et al. 2010; Kressmann, Muller, and Blume 2002), and the concentration of total terpene lactones is not uniquely high among commercial extracts (Sloley et al. 2003). Additionally, it is important to note that terpene lactones and flavonol glycosides are purportedly associated with proposed therapeutic effects of GBE, not toxicity (van Beek and Montoro 2009). Ginkgolic acids measured in the test article, which are associated with toxicity (Westendorf and Regan 2000), were on the low end of the range reported in U.S. products (<500–90,000 ppm; Kressmann, Muller, and Blume 2002).
Three-month and 2-year treatment with GBE via gavage in both sexes of two species resulted in a consistent suite of toxicity targets including liver, thyroid gland, and nose. Various lesions were observed in these target tissues. In the 2-year studies, the evidence of carcinogenicity of GBE was based primarily on thyroid gland neoplasms in male and female F344/N rats and liver neoplasms in male and female B6C3F1/N mice.
The carcinogenic effects observed in the liver of male and female mice during the 2-year studies were particularly significant. There were notable increases in incidences of multiple hepatocellular adenomas, hepatocellular carcinomas, and hepatoblastomas. Incidences of hepatoblastoma, a relatively rare tumor, were among the highest observed in NTP studies to date (Turusov et al. 2002). Microsomal enzyme–inducing compounds such as phenobarbitol—a protypical promoting agent—oxazepam, and primidone have also been found to elicit significant induction of hepatoblastomas (Bucher et al. 1994; Diwan, Henneman, and Rice 1995; NTP 2000). Some evidence indicates that hepatoblastoma development is mediated by promoting agents, rather than initiating agents (Diwan, Henneman, and Rice 1995).
Two companion studies were conducted to explore possible mechanisms of carcinogenicity of GBE using mouse liver tissue from these 2-year studies: a gene expression study in male mice (Guo et al. 2010) and a study examining gene mutations and genomic pathways in GBE-induced hepatocellular carcinomas (Hoenerhoff et al. 2012). In the gene expression study, many phase I, II, and III metabolizing enzymes exhibited altered gene expression with GBE treatment. Using Ingenuity Pathway Analysis, Guo et al. (2010) identified “metabolism of xenobiotics by cytochrome P450” and “NRF2-mediated oxidative stress response” among the top canonical pathways (Guo et al. 2010). The network that contained the largest number of differentially expressed genes was “cell cycle, cellular movement and cancer” with the proto-oncogene Myc being the central gene of the network. Myc is involved in cell growth, proliferation, differentiation, and apoptosis and has been found to cooperate with nongenotoxic hepatotoxicants to accelerate the development of liver tumors in mice (Beer et al. 2008). In the hepatocellular carcinoma study, Hoenerhoff et al. (2012) observed dose-related mutations in β-catenin (Ctnnb1) genes in GBE-treated carcinomas, while mutations occurred predominantly in H-ras genes in the control group carcinomas. These mutations are common events in both human and mouse hepatocarcinogenesis (Ctnnb1) and in spontaneous hepatocellular carcinoma tumors in mice only (H-Ras). The dose-related increase in Ctnnb1 gene mutations in GBE-treated carcinomas was accompanied by an increase in posttranslational protein modification of CTNNB1. Overexpression of gene profiles for cancer-signaling pathways, xenobiotic metabolism, and oxidative stress was also observed in GBE-induced hepatocellular carcinomas (Hoenerhoff et al. 2012). Findings in both studies are consistent with literature indicating that GBE, as well as several individual GBE constituents, can activate various receptors (e.g., constitutive androstane receptor [CAR], pregnane X receptor [PXR], aryl hydrocarbon receptor [AhR]) involved in xenobiotic metabolizing enzyme induction (Lau, Yang, Rajaraman, et al. 2012; Lau, Yang, Yap, et al. 2012; Lau et al. 2010; Li et al. 2009).
Although the carcinogenic effects of GBE in the liver were more pronounced in mice than in rats, a variety of nonneoplastic liver lesions were observed in the 3-month and 2-year studies in both mice and rats. Some of the liver changes (i.e., increased liver weight, centrilobular hepatocyte hypertrophy, minimal focal necrosis, or fatty change of hepatocytes) were consistent with a hepatic microsomal enzyme induction phenotype (Maronpot et al. 2010). Furthermore, increased liver weight and hepatocyte hypertrophy accompanied by degenerative changes, such as hepatocytic necrosis, observed in the 3-month studies have been found to be predictive of hepatocellular neoplasm development in mice and rats (Allen et al. 2004). Therefore, it is likely that the nonneoplastic lesions observed in mice and rats with GBE treatment represent earlier stages along the pathway to cancer.
The thyroid gland was also a target for GBE toxicity. In the 13-week study, follicular cell hypertrophy was observed in male and female rats. In the 2-year study, nonneoplastic effects on the thyroid included follicular cell hypertrophy and hyperplasia in male and female rats and mice. Neoplastic findings included increases in follicular cell adenomas in male rats and mice and single cases of follicular cell carcinomas in female rats in the mid- and high-dose groups. In addition to the morphological lesions, TSH levels were elevated in high-dose male rats on day 22 and male and female rats at 14 weeks. Taken together, the increased liver weights and lesions, thyroid lesions, and increased TSH levels are suggestive of a hepatic microsomal enzyme induction mechanism for carcinogenicity. Proposed thyroid carcinogenicity via this mechanism involves induction of hepatic microsomal enzymes (e.g., Uridine 5′-diphospho-glucuronosyltransferase [UDP]-glucuronosyl transferase) involved in thyroid hormone (T3 and T4) metabolism and/or clearance. Induction of these enzymes can result in a decrease in thyroid hormones followed by a subsequent increase in TSH levels, leading to proliferative lesions in the thyroid (Curran and Degroot 1991). Male rats are known to be more sensitive than female rats to this mechanism of thyroid disruption because they normally have higher TSH levels. In addition, rats are thought to be more sensitive than humans to the development of thyroid tumors via this mechanism because of more highly inducible UDP-glucuronosyl transferase and lower affinity thyroid-binding proteins leading to a notably shorter half-life of T4 in rats compared to humans (Capen 1997). In humans, only high doses of potent enzyme-inducing chemicals have been demonstrated to decrease T4 levels, and this decrease has not been accompanied by an increase in TSH (Capen 1997). However, it is important to note that there are other mechanisms for thyroid follicular cell carcinogenesis and classic enzyme inducers (e.g., phenobarbital) display thyroid hormone changes that are typically more pronounced than those seen with GBE, including larger increases in TSH accompanied by decreased T4 levels and increased thyroid gland weight (Capen 1997), which were not observed in the present study. Additionally, many enzyme inducers are nonmutagenic, while GBE was mutagenic in Salmonella typhimurium strains TA98 and TA100, as well as Escherichia coli strain WP2 uvrA/pKM101, with and without S9 metabolism (Hoenerhoff et al. 2012).
The third major target of GBE toxicity was the nose. Various nonneoplastic nose lesions were observed in male and female rats and mice in both the 13-week and 2-year studies. Neoplastic lesions were confined to 2 cases of respiratory epithelial adenomas at the middle dose of GBE in female rats in the 2-year study. This neoplasm is rare with only a 0.1% occurrence in the NTP historical controls for all routes of administration.
The pathogenesis by which administration of GBE leads to nasal lesions is not clear. Possible mechanisms include enzymatic induction following systemic exposure or reflux through the nasopharyngeal duct. Enzyme induction following systemic exposure (Sells et al. 2007) is consistent with the pathologies noted in the liver. Both the nose and liver are rich in metabolic activity. CYP450 concentration in the rat olfactory epithelium is second only to the liver (Reed 1993). Furthermore, in both studies, the olfactory epithelium was more severely affected than the respiratory epithelium, which parallels the higher levels of enzymatic activity in the olfactory epithelium (Walsh and Courtney 1998).
The alternative reflux hypothesis (Damsch et al. 2011) was proposed based on the predominant ventral and posterior distribution of nasal pathology and the presence of nasopharyngeal duct lesions in many of the GBE-treated animals. Typically, lesions induced by olfactory toxicants are severe and primarily located in the more anterior sections of the nose and along the dorsal meatus, and are generally not heavily suppurative (Renne et al. 2007). The reflux hypothesis is further supported by the dose-related increased incidence and/or severity of epithelial hyperplasia, hyperkeratosis, ulcer, and inflammation in the forestomach of treated male and female mice (data not shown). A low incidence of squamous cell papilloma was found in treated male mice, but not control. Forestomach lesions were not noted in either male or female rats.
In conclusion, GBE is a complex mixture that induces pathological changes primarily in the liver, thyroid gland, and nose of male and female mice and rats. The most notable carcinogenic responses were the increases in liver tumors in male and female mice and thyroid gland follicular cell tumors in male and female rats. Extrapolation of animal findings to humans is particularly difficult with GBE due to the complex suite of lesions, lack of a clear mechanism of action, and the inherent complexity of the material. However, the positive finding of carcinogenicity and the consistent pattern of response across males and females in 2 species should be considered in future risk assessments of GBE. Additionally, the potential engagement of the microsomal enzyme system evidenced by the pattern of effects and accompanying molecular work highlights the need for consideration of drug–herb interactions.
Footnotes
Acknowledgments
The authors would like to sincerely thank Dr. Mamta Behl and Dr. June Dunnick for reviewing this article.
This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH); however, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. government.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
