Abstract
We describe and characterize unilateral renal aplasia in a cynomolgus monkey (Macaca fascicularis) from a chronic toxicology study adding to the limited histopathology reports of congenital renal anomalies in macaques. In the current case, the affected kidney was macroscopically small and characterized microscopically by a thin cortex with an underdeveloped medulla and an absent papilla. The remnant medulla lacked a corticomedullary junction and contained only a few irregular collecting duct-like structures. The cortex had extensive interstitial mature collagen deposition with fibromuscular collar formation around Bowman’s capsules. Due to parenchymal collapse, mature glomeruli were condensed together with occasional atrophic and sclerotic glomeruli. The majority of the cortical tubules were poorly differentiated with only small islands of fully developed cortical tubules present. Histochemical and immunohistochemical stains were utilized to demonstrate key diagnostic features of this congenital defect, to assist with differentiating it from renal dysplasia, and to provide potential mechanistic pathways. Immunostaining (S100, paired box gene 2 [PAX2], aquaporins) of the medulla was compatible with incomplete maturation associated with aplasia, while the immunostaining profile for the cortex (vimentin, calbindin, PAX2-positive cortical tubules, and smooth muscle actin–positive fibromuscular collars) was most compatible with dedifferentiation secondary to degenerative changes.
Introduction
Normal development of the kidney requires appropriate initiation of the pronephros–mesonephros–metanephros sequence that involves growth and advancement of the ureteric bud from the mesonephric duct and proper interactions with the metanephric blastema. 1 -5 This sequence of development depends on the orderly temporal and spatial expression of multiple genes in the urinary tract. 5,6 Derangement in gene expression induced by genetic mutations or environmental factors results in congenital abnormalities of the kidney. 7
In humans, congenital anomalies of the kidney and urinary tract (CAKUT) comprise a broad spectrum of renal and lower urinary tract structural and functional malformations that include renal agenesis, aplasia, hypoplasia, dysplasia, supernumerary kidneys, ectopic or fused kidneys, duplication, ureteropelvic junction obstruction, primary megaureter or ureterovesical junction obstruction, vesicoureteral reflux, ureterocele, and posterior urethral valves. 8 Renal dysplasia, hypoplasia, and aplasia/agenesis comprise 15.8% of the primary diagnoses in pediatric kidney transplant patients, and without surgery treatment is palliative. 9 Hypoplasia refers to small kidneys with a decreased number of nephrons, but existing nephrons are mature and normal cortical medullary organization is preserved. 8 Aplasia represents incompletely or defectively developed kidneys, whereas agenesis indicates the absence of renal tissue. 10 Dysplastic kidneys have an abnormal architectural organization with asynchronous differentiation resulting in abnormal or aberrant tissues, such as bone, cartilage, or embryonic structures. 8,11 Morphologic criteria have been utilized to diagnose the various renal defects, but extreme cases of dysplasia and aplasia can ultimately present as agenesis due to involution. 10,12,13 In addition, secondary degenerative, cystic, and/or atrophic changes can mask the primary renal malformation, resulting in misinterpretation of a spontaneous congenital lesion in a toxicology study. 14 Xenobiotic treatment during neonatal and juvenile toxicity studies may result in altered nephrogenesis and/or maturation resulting in abnormal renal morphology and/or function. 1,15 Differentiating between the various renal developmental anomalies is important not simply for academic reasons of proper terminology, but it may also provide an understanding of the pathologic mechanism responsible for negatively affecting renal development, which may be critical for potential translation to neonatal and pediatric risk.
Spontaneous unilateral renal aplasia has not been reported in macaques but has been reported in other nonhuman primate species, 16,17 humans, 10,12,18 domestic animals, 19,20 and laboratory animals. 21 One report of unilateral aplasia in a rhesus (Macaca mulatta) macaque was actually renal agenesis due to the absence of one kidney. 22 Although the usage of partial aplasia is not conventional terminology in human pathology, unilateral partial renal aplasia was described in a howler monkey (Aloutatta caraya). 17 In addition, unilateral renal aplasia was reported in a wild-caught African baboon (Papio sp.) 16 and in a baboon (Papio anubis) utilized in a toxicity study, but the latter monkey had unilateral absence of renal tissue and ureter which was more consistent with renal agenesis. 23
Other spontaneous congenital anomalies have been reported in cynomolgus (Macaca fascicularis) and/or rhesus (Macaca mulatta) macaques but at a low frequency: a few cases of spontaneous renal dysplasia, 22,24,25 several cases of spontaneous congenital polycystic kidney disease, 17,26 -28 and single cases of hypoplasia, double ureter, renal ectasia, and unilateral cystic kidney. 22,29 Spontaneous renal dysplasia has been reported relatively frequently in many species 8,11,14,30,31,32,33,34,35 -37,38,39,40 -42,43,44 and is known to be familial/inherited in some species. 33,34,37,19,44 Multiple genetic syndromes have been associated with renal malformations in humans, but the specific gene defect is only known for some conditions, whereas the genetic mutations responsible for the majority of human renal dysplasia cases remain unknown. 45,46 In addition to genetic defects, other known underlying causes of renal dysplasia have been identified and include lower urinary tract obstruction and teratogens in humans 11,47 and fetal viral infections, gestational hypovitaminosis A, renal amyloidosis, neoplasia, and lower urinary tract obstruction in animals. 19,48,49
We describe and characterize the features of spontaneous unilateral renal aplasia in a cynomolgus monkey noted at the termination of a chronic toxicology study. The diagnosis and differentiation from dysplasia were based on histochemical and immunohistochemical features, and the data provide some insight into potential molecular pathogenesis.
Materials and Methods
Animals
The 2 animals, one control female (6.6 years old) and one treated female (6.5 years old) cynomolgus monkey (Macaca fascicularis) of Asian origin, were from the 16-week off dose phase that followed a 26-week toxicity and toxicokinetic study for a biotherapeutic drug. This standard chronic toxicity study included daily clinical observations, hematology, serum chemistry, coagulation, urinalysis, urine chemistry, terminal organ weights for selected organs, macroscopic observations, and microscopic evaluation of a full set of tissues stained with hematoxylin and eosin (H&E). The study was conducted in an American Association for the Accreditation of Laboratory Animal Care (AALAC)-accredited facility at Covance Madison, Wisconsin, in accordance with Covance policies on the care, welfare, and treatment of laboratory animals, and they were reviewed and approved by Covance’s Institutional Animal Care and Use Committee as appropriate.
Histochemical and Immunohistochemical Stains
The following histochemical stains were used to characterize brush border and basement membranes (periodic acid Schiff [PAS]) to detect fibrosis (Masson’s trichrome [MTC]) or persistent mesenchyme (Alcian blue [AB]) and to identify amyloid (Congo red [CR]) or lipofuscin (Schmorl’s method [SM]). The following immunohistochemical stains were selected to identify nephron segments: aquaporin 1 (AQP1) for proximal tubules and descending thin limbs; aquaporin 2 (AQP2) and S100 for connecting and collecting ducts; uromodulin (UMOD) for thick ascending limbs; uroplakin II (UPII) for pelvic urothelium; and calbindin-D28 k (CALB) for distal tubules, connecting ducts, and collecting ducts. Structural components were identified using the following immunohistochemical stains: α smooth muscle actin (α-SMA) for vascular smooth muscle, myofibroblasts, interstitial cells; vimentin (VMTN) for tissues of mesenchymal origin; and pancytokeratin (panCK) for cells of epithelial origin. In order to characterize features associated with renal development, the following immunostains were used: PAX2 for ureteric bud and mesenchyme development, dedifferentiation, and regeneration; Wilms’ tumor 1 (WT1) for metanephric mesenchyme development and podocyte differentiation; and β-catenin (CTNB) for nephrogenic induction, ureteric morphogenesis, and cell-to-cell adhesion. The following immunohistochemical stains were used to characterize the response to injury: ki67 for proliferation; CALB for a protective response from injury; and VMTN for the epithelial–mesenchymal transition.
Formalin-fixed, paraffin-embedded kidneys were sectioned 5-micron thick, mounted on AutoFrost IHC hydrophilic slides (Cancer Diagnostics), and stained histochemically for MTC, PAS reaction, AB, SM, or CR. Poly Scientific R&D Corp staining kits were used according to the manufacturer’s instructions.
Formalin-fixed, paraffin-embedded kidneys and additional positive control tissues (uterus, colon, and urinary bladder) were sectioned 5-microns thick, mounted on AutoFrost IHC hydrophilic slides (Cancer Diagnostics), and immunostained on the Roche Ventana Discovery Ultra autostainer using primary antibodies specific for α-SMA, AQP1, UMOD, CALB, AQP2, PAX2, VMTN, panCK, S100, CTNB, WT1, Ki67, and UPII (Table 1). Briefly, sections were deparaffinized, subjected to heat-induced epitope retrieval (and subsequent protease retrieval for WT1), blocked to reduce nonspecific staining, incubated for 32 minutes with a primary antibody, followed by Roche secondary antibodies (and Jackson Lab rabbit anti-sheep secondary antibody for UPII) and amplification polymers (DAB chromogen), and counterstained with hematoxylin. Specific immunostaining was assessed by examining standard control slides, that is, the substitution of the primary antibody with species and concentration-matched isotype control immunoglobulins. Positive and negative tissue controls were used to assess each staining run. Whole slides were scanned using a Leica Aperio AT2 whole digital slide scanner (Leica) at the 20× objective setting, and images were saved in .svs format. Representative images of histopathology and staining results (Figures 1 -6) were digitally captured using Leica Aperio ImageScope software (v11.2.0.780) at original virtual magnifications of 1×, 5×, 10×, or 20×.
Antibody Source, Concentration, and Specific Methodology Used for Immunohistochemistry Procedures.
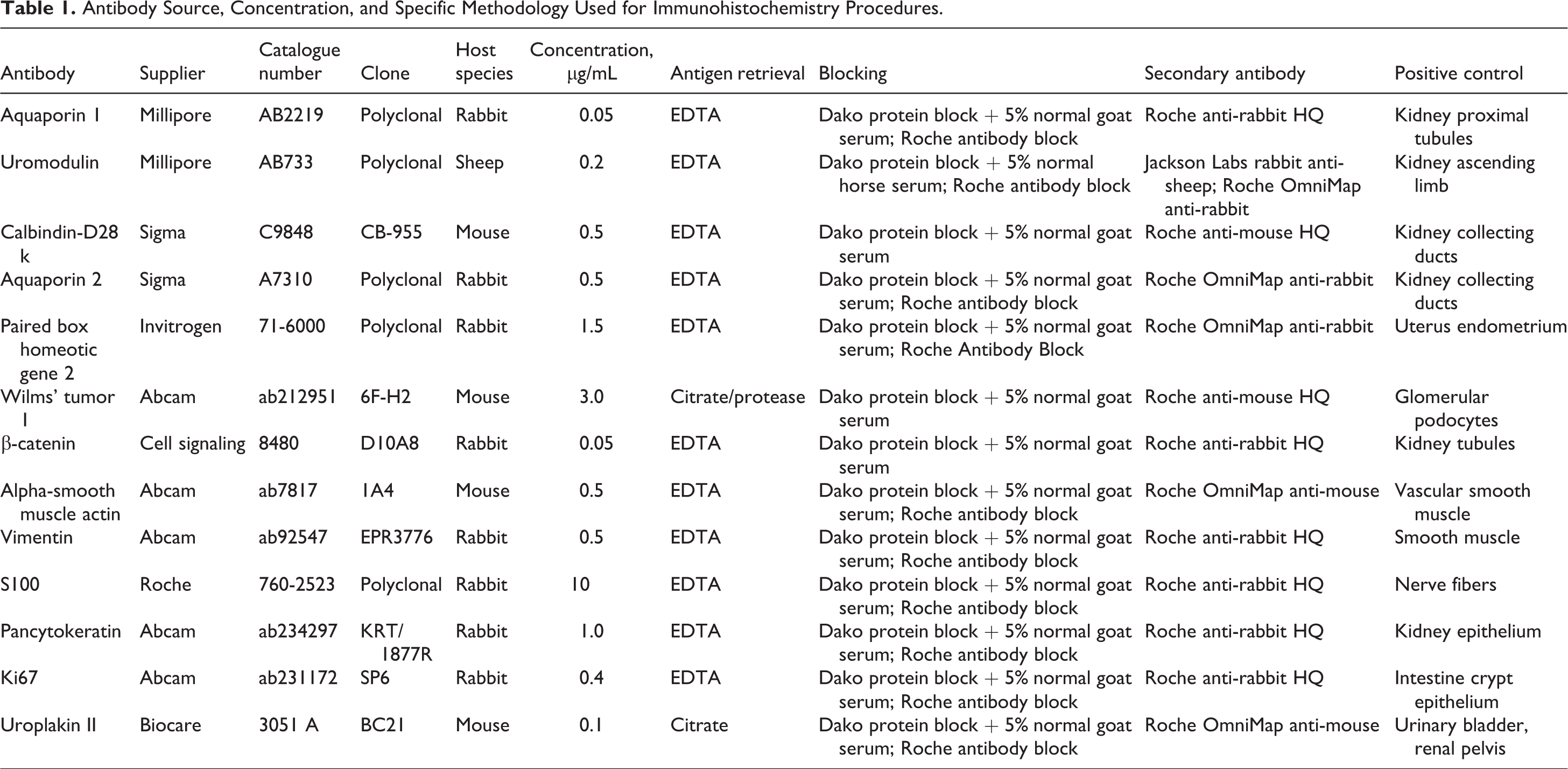
Hematoxylin & Eosin (H&E). (A) The aplastic kidney is small with a thin cortex and absent corticomedullary junction and papilla, ×1 original magnification (mag). (B) Normal control kidney, ×1 mag. (C) The aplastic kidney cortex has a paucity of mature cortical tubules with increased interstitial connective tissue and inflammation, ×5 mag. (D) The aplastic kidney cortex has variably sclerotic and/or atrophic glomeruli that are in close proximity due to decreased mature cortical tubules and parenchymal collapse. The interstitium has increased connective tissue and inflammation. Poorly differentiated tubules are scattered between the glomeruli, ×10 mag. (E) The aplastic kidney cortex has rare small islands of mature cortical tubules present among the poorly differentiated tubules and variably sclerotic and atrophic glomeruli. Peri-Bowman’s capsule connective tissue collarettes encircle glomeruli. ×20 mag. (F) Several collecting duct-like tubules are present in the underdeveloped medulla/papilla region with increased interstitial connective tissue and inflammation, ×20 mag.
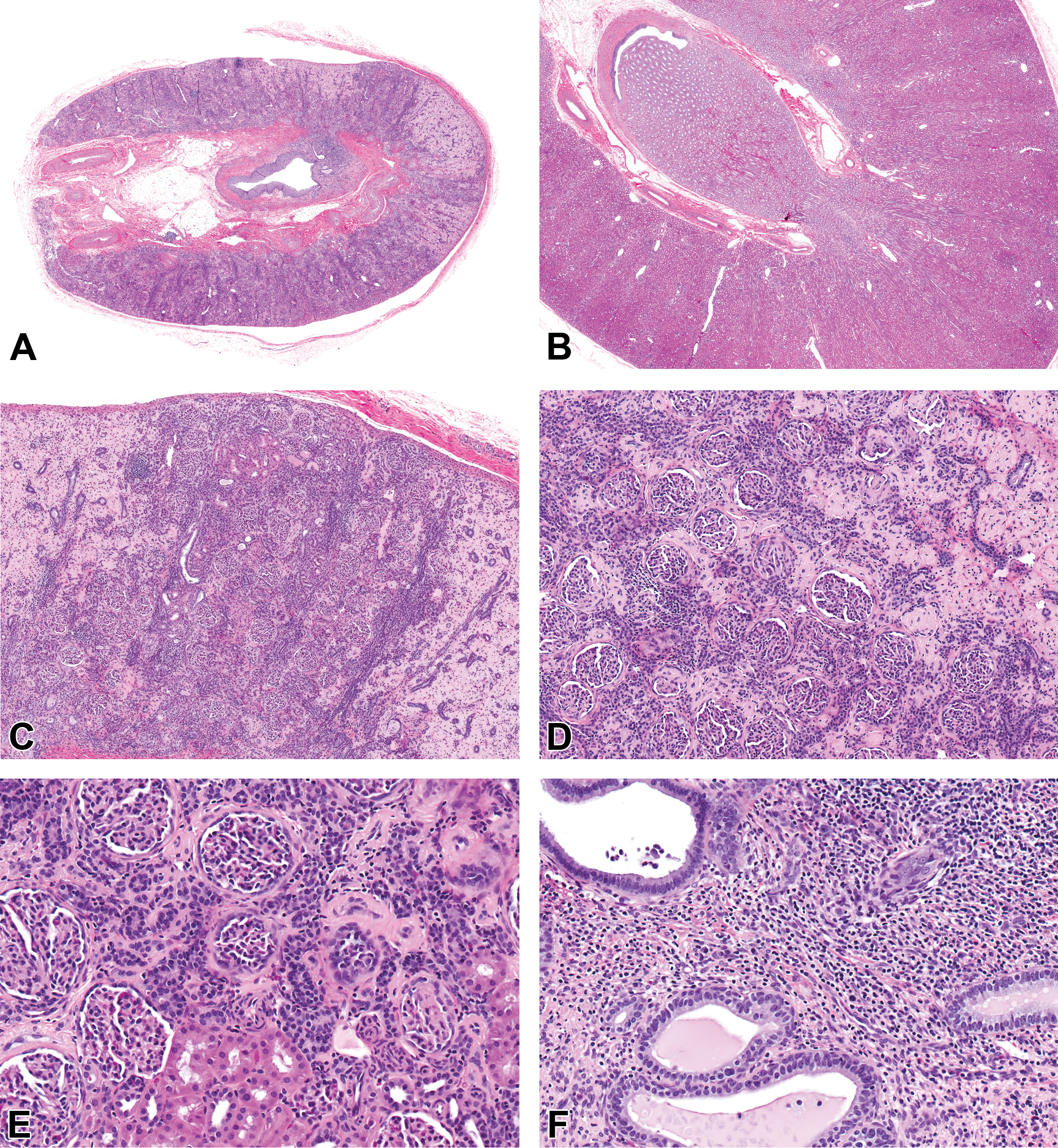
Alcian blue (AB; A and B): The mesenchymal collarettes encircling Bowman’s capsule in the aplastic kidney stain AB+ (2A), but the normal kidney did not have any peri-Bowman’s capsule AB staining (A, inset). The expanded interstitium in the medulla region of the aplastic kidney has decreased AB staining (B) compared to the normal kidney (B, inset). Masson’s trichrome (MTC; C and D): The mesenchymal collarettes encircling Bowman’s capsule and the cortical interstitial connective tissue are MTC+ and are increased compared to the normal kidney (C, inset). The expanded interstitium in the medulla region of the aplastic kidney was MTC+ and was increased compared to the normal kidney (D, inset). Periodic acid Schiff (PAS; E and F): The cortical interstitial connective tissue and sclerotic glomeruli are PAS+ in the affected kidney. The cortical interstitium and glomeruli are negative in the normal kidney (E, inset). The expanded interstitium in the medulla region of the aplastic kidney has decreased PAS+ staining compared to the normal kidney (F, inset). PAS+ homogenous material was also present within the lumen of the collecting duct-like structures in the aplastic kidney (F). ×20 original magnification.
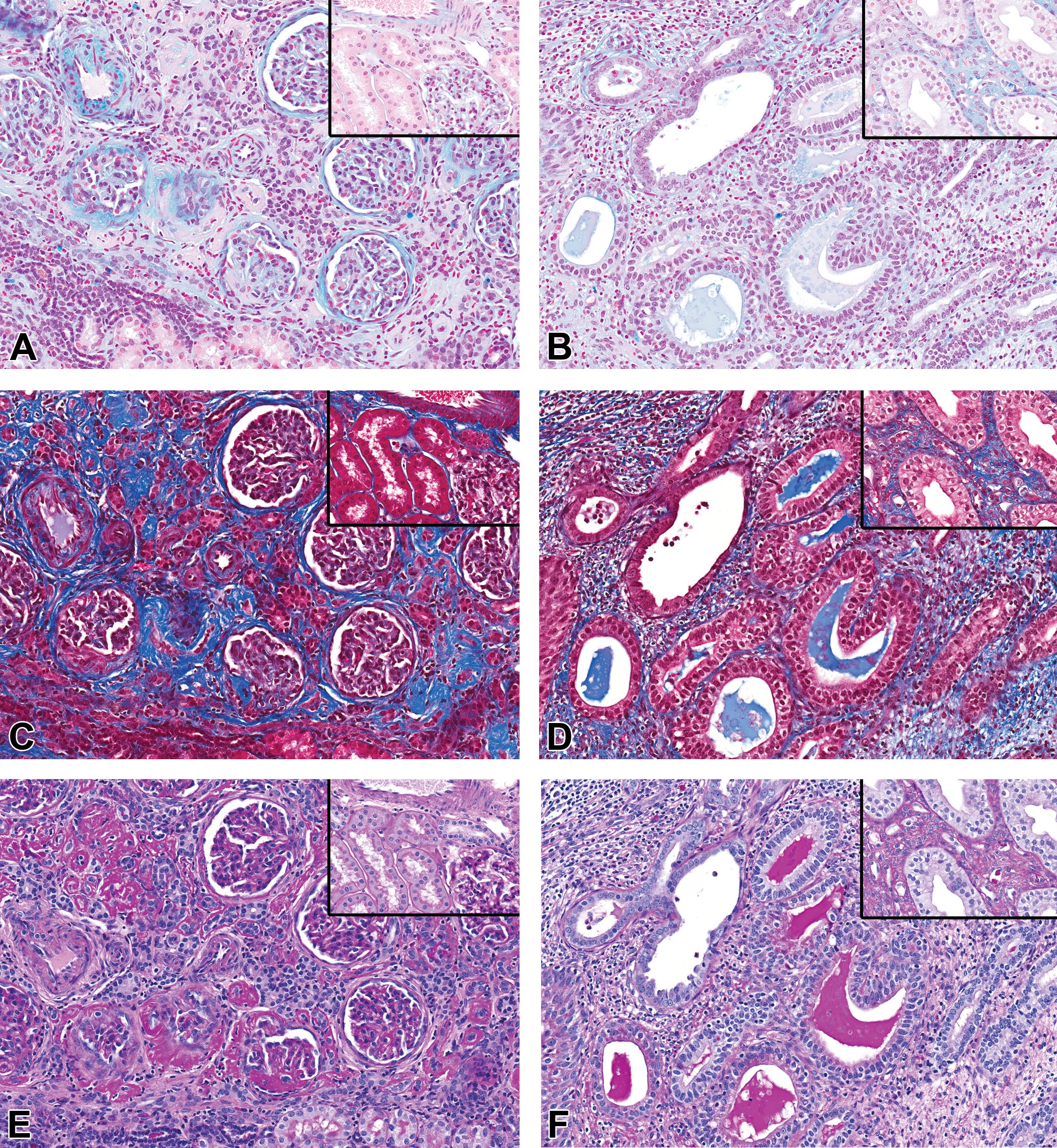
α-Smooth muscle actin (α-SMA, A and B): The mesenchymal collarettes encircling Bowman’s capsule and vascular smooth muscle are α-SMA+ in the aplastic kidney (A) but no peri-Bowman’s capsule staining is present in the control kidney (A, inset). Similar to the normal kidney medulla (A and B, insets), interstitial cells and vascular smooth muscle are α-SMA+ in the medulla region of the aplastic kidney, but α-SMA+ interstitial cells are increased (B). Vimentin (VMTN; C and D): The mesenchymal collarettes encircling Bowman’s capsule in the aplastic kidney and the increased interstitial connective tissue are VMTN+, as well as, the vascular and glomerular structures, leukocytes, and collagen similar to the normal kidney (C, inset). The increased interstitial matrix and some of the collecting duct-like structures (*) are VMTN+, whereas collecting ducts in the normal kidney are negative (D, inset). Wilms’ tumor 1 (WT1; E and F): Podocytes in the aplastic kidney (E) have nuclear WT1+ staining, but Bowman’s capsule collarettes and the increased cortical (E) and medulla region (F) interstitial connective tissues are negative paralleling the normal kidney (E and F, insets). ×20 original magnification.
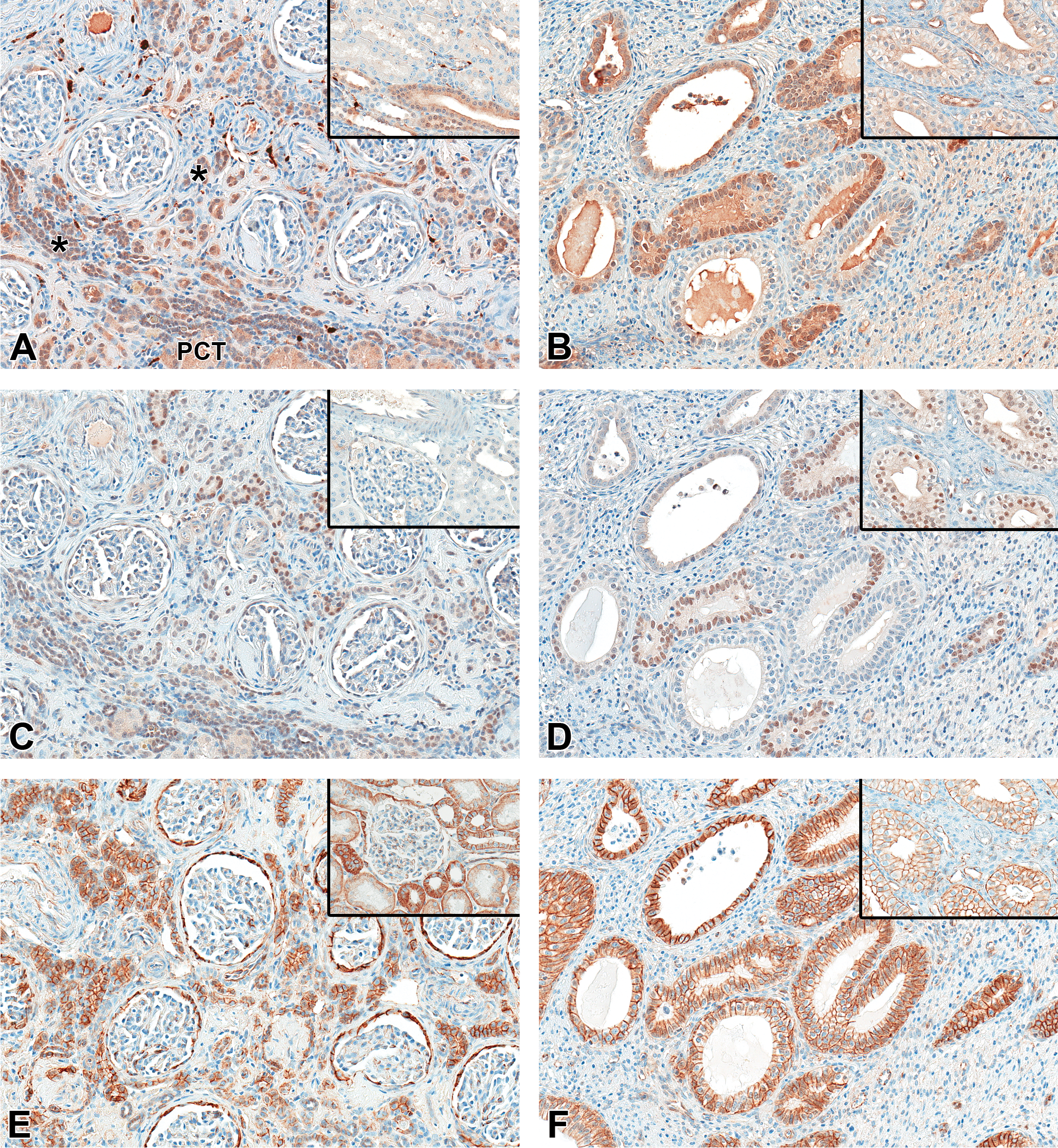
S100 (A and B): Distal tubules, thin limbs, thick ascending limbs, cortical connecting and collecting ducts, medullary collecting ducts, and nerves are S100+ in both the normal kidney cortex (A, inset) and medulla (B, inset) and in the same unaffected structures of the aplastic kidney cortex (A). Mature proximal convoluted tubules and poorly differentiated cortical tubules (*) are also S100+ in the aplastic kidney (A) and some of the smaller collecting duct-like structures have increased S100+ immunostaining (B) compared the normal kidney (B, inset). Paired box homeotic gene 2 (PAX2; C and D): In the aplastic kidney cortex (C), the poorly differentiated tubules have nuclear PAX2+ staining and the distal tubules and collecting/connecting ducts have increased PAX2+ nuclear labeling compared to the control kidney (C, inset), whereas in the medulla region of the aplastic kidney (D), some of the collecting duct-like structures are variably PAX2+ compared to the normal kidney medullary collecting ducts which have more consistent nuclear PAX2+ staining (D, inset). β-Catenin (CTNB, E and F): In the aplastic kidney, CTNB+ labeling of the remnant normal structures parallels that seen in the normal kidney cortex (E, inset) and medulla (F, inset), but Bowman’s capsule epithelium (E), poorly differentiated cortical tubule epithelium (E), and medullary collecting duct-like epithelium (F) have increased cytoplasmic and/or membrane immunostaining intensity compared to the normal structures. ×20 original magnification.
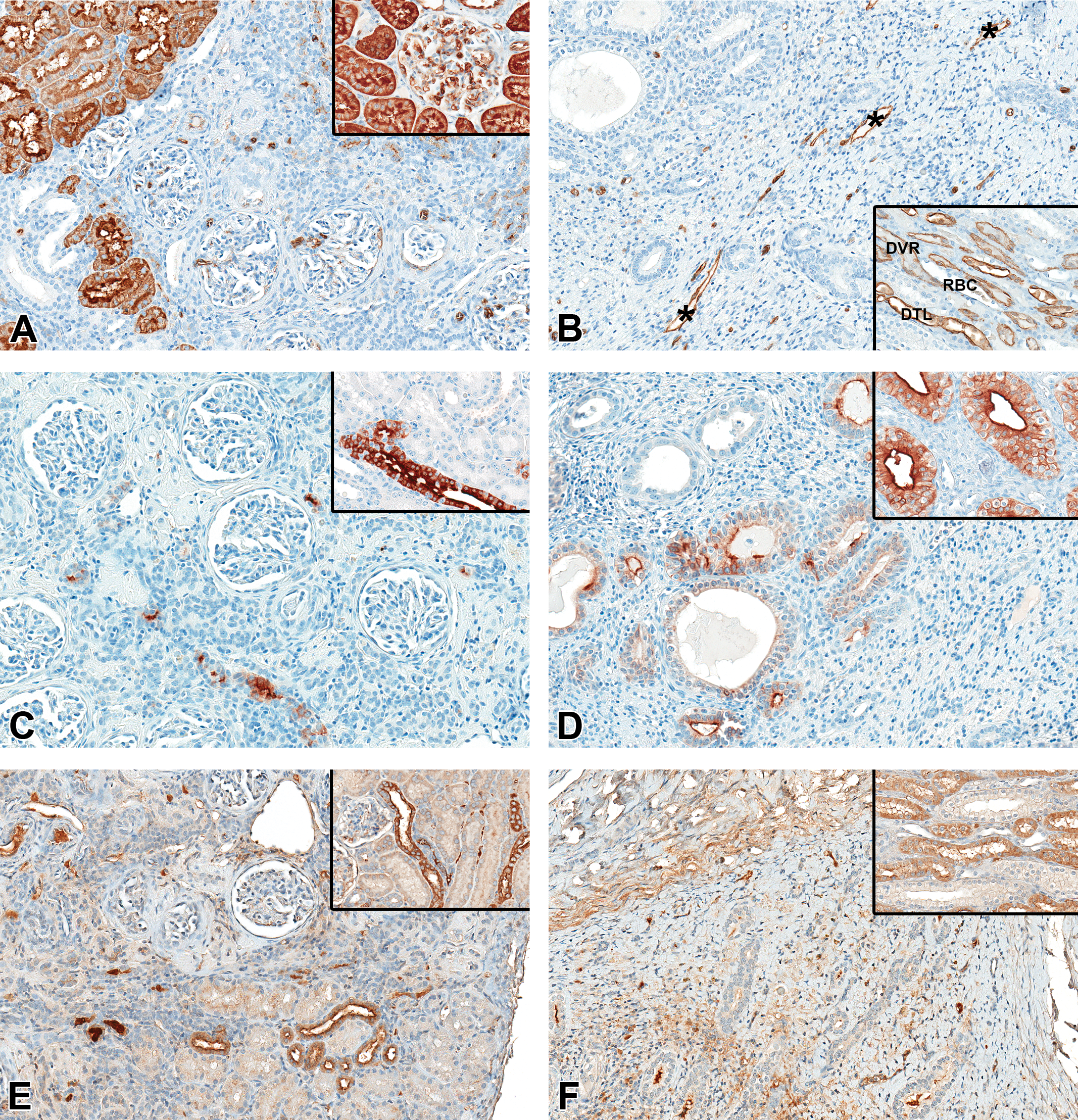
Aquaporin 1 (AQP1; A and B): In the normal kidney cortex (A, inset), proximal tubules, vascular endothelium, and RBCs have AQP1+ cytoplasmic and membrane immunostaining. The aplastic kidney (A) has similar endothelial and RBCs AQP1+ immunostaining, but there are only scattered islands of AQP1+ mature proximal tubules present. The poorly differentiated cortical tubules are variably positive, but most are negative. In the normal kidney medulla (B, inset), descending thin limbs of long looped nephrons (DTL), descending vasa recta (DVR), and RBCs are AQP1+. The undeveloped medulla in the affected kidney (B) has no AQP1+ descending thin limbs and no distinct vasa recta present; however, some AQP1+ vessels (*) are present. Aquaporin 2 (AQP2; C and D): In the normal kidney cortex (C, inset) and medulla (D, inset), cortical connecting and collecting ducts and medullary collecting ducts have AQP2+ cytoplasmic and apical membrane immunostaining that is more intense in the principle cells compared to intercalated cells. The aplastic kidney has lesser numbers of AQP2+ connecting and collecting ducts in the cortex (C) with only a few AQP2+ collecting ducts in the underdeveloped medulla (D). The majority of the medullary collecting duct-like structures are negative and/or have variable apical membrane AQP2+ immunostaining (D) which is less intense than the normal kidney (D, inset). Uromodulin (UMOD, E and F): In the normal kidney cortex (E, inset) and medulla (F, inset), thick ascending limbs have UMOD+ cytoplasmic immunostaining in both the cortex and the outer medullary stripe. In the aplastic kidney, there are lesser numbers of UMOD+ thick ascending limbs in the cortex (E), and no UMOD+ tubules are present in the underdeveloped outer medulla (F). ×20 original magnification.
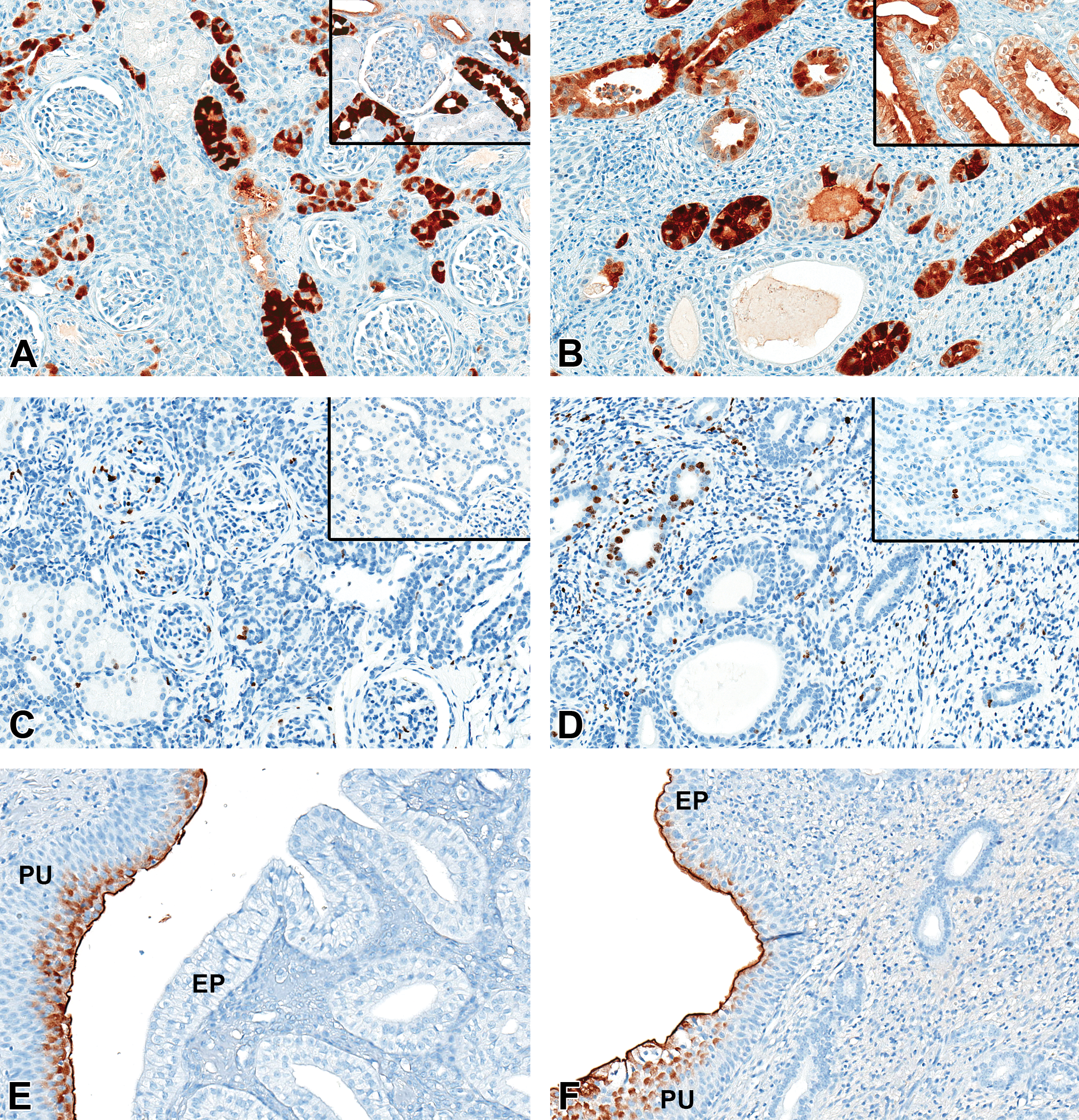
Calbindin-D28 k (CALB, A and B): In the normal kidney, distal tubules, macula densa, cortical thick ascending limb, cortical connecting and collecting ducts (A, inset), and medullary collecting ducts (B, inset) have CALB+ cytoplasmic and apical membrane immunostaining with more intense CALB+ immunostaining observed in principal cells of the cortex than in the medulla. The aplastic kidney has lesser numbers of CALB+ cortical connecting and collecting ducts (A) and has only a few CALB+ collecting duct-like structures in the aplastic medulla region (B) which stain more intensely than the normal medullary collecting ducts (B, inset). Some of the larger medullary collecting duct-like structures are negative and/or have variable CALB+ immunostaining. In addition, some of the poorly differentiated cortical tubules are variably CALB+ (A). ×20 original magnification. Ki67 (C and D): In the normal kidney, rare ki67+ nuclear staining is noted in all components of the nephron (C and D, insets). In the affected kidney, increased ki67+ immunostaining is observed in the glomerulus (C) and in a few of the medullary collecting duct-like structures along with a few cells in the interstitium (D). Uroplakin II (UPII, E and F): In the normal kidney, the superficial layers of the pelvic urothelium (PU) have UPII+ membrane and cytoplasmic immunostaining and the epithelium of the renal papilla (EP) is negative. In the affected kidney, the epithelium covering the remnant papilla-like structure (EP) immunostained UPII+ as well as the pelvic urothelium (PU). ×20 original magnification.
Histochemical- and immunohistochemical-stained whole slide sections were evaluated by a veterinary pathologist (L.A.O.). Presence or absence of staining was qualitatively scored as positive (+) or negative (−) for each of the nephron segments and/or tissue components comparing the affected kidney to the normal control kidney and indicating increased (↑) or decreased (↓) staining as appropriate. The scoring results are summarized in Table 2.
Histochemical and Immunohistochemical Staining Results.a
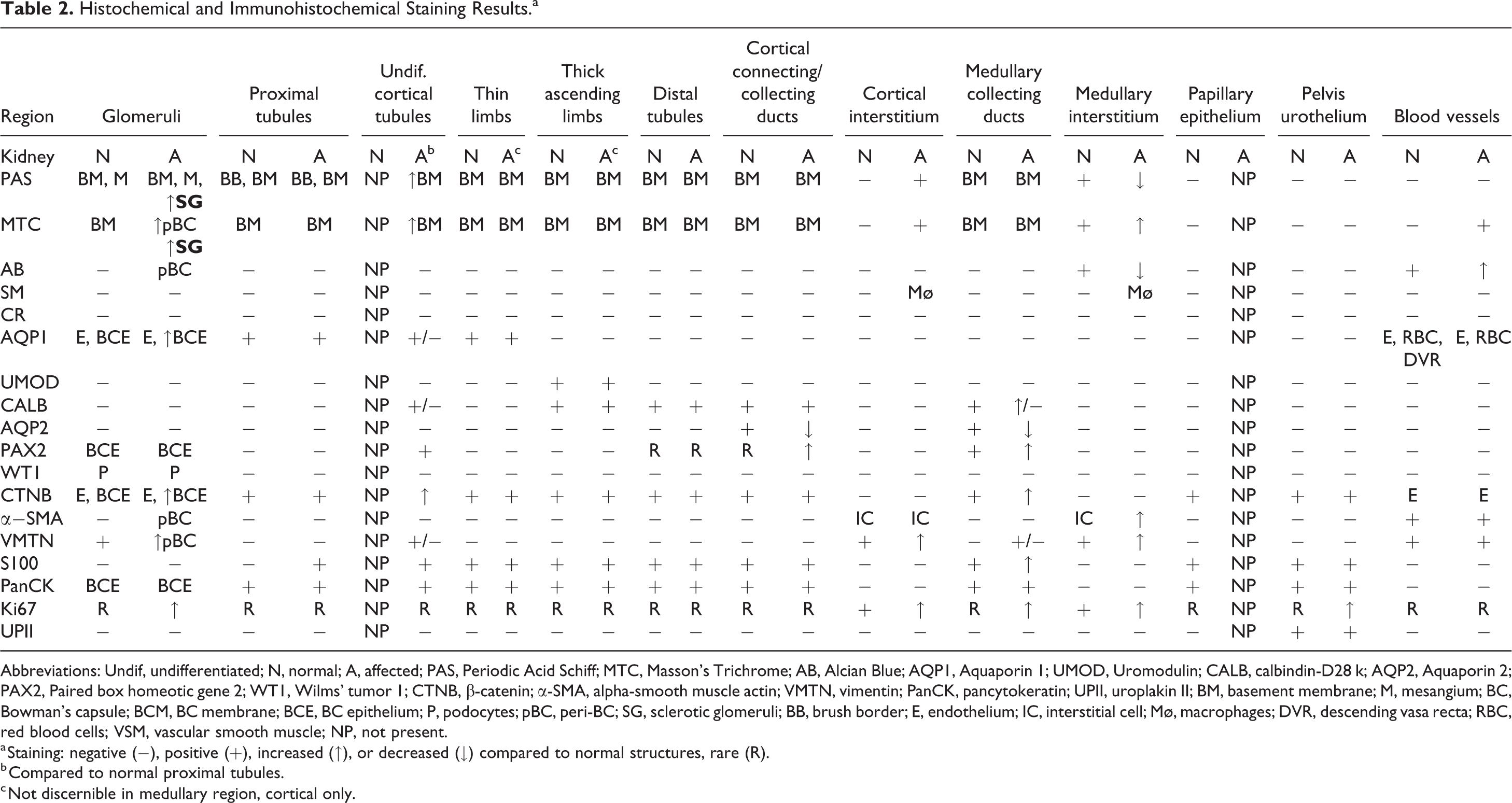
Abbreviations: Undif, undifferentiated; N, normal; A, affected; PAS, Periodic Acid Schiff; MTC, Masson’s Trichrome; AB, Alcian Blue; AQP1, Aquaporin 1; UMOD, Uromodulin; CALB, calbindin-D28 k; AQP2, Aquaporin 2; PAX2, Paired box homeotic gene 2; WT1, Wilms’ tumor 1; CTNB, β-catenin; α-SMA, alpha-smooth muscle actin; VMTN, vimentin; PanCK, pancytokeratin; UPII, uroplakin II; BM, basement membrane; M, mesangium; BC, Bowman’s capsule; BCM, BC membrane; BCE, BC epithelium; P, podocytes; pBC, peri-BC; SG, sclerotic glomeruli; BB, brush border; E, endothelium; IC, interstitial cell; Mø, macrophages; DVR, descending vasa recta; RBC, red blood cells; VSM, vascular smooth muscle; NP, not present.
a Staining: negative (−), positive (+), increased (↑), or decreased (↓) compared to normal structures, rare (R).
b Compared to normal proximal tubules.
c Not discernible in medullary region, cortical only.
Results
Necropsy and Macroscopic Examination
Both animals were clinically normal and weighed 3.2 kg (control) and 4.6 kg (treated, unilateral renal finding) at postmortem examination. For both monkeys, there were no abnormalities in hematology, coagulation, clinical chemistry, urinalysis, or urine chemistry test results. At necropsy, the right kidney in the treated monkey was described macroscopically as small and measured approximately 2 × 1 × 1 cm in the hilar transverse section, whereas the contralateral left kidney was of normal size and measured approximately 4 × 2.5 × 2.5 cm. Size measurements were not obtained for the control monkey kidney. The combined absolute kidney weight (9.7 g) and kidney relative to body weight (0.21%) were less than the control monkey’s values (11.9 g and 0.37%, respectively), which correlated with the reduced mass of the small right kidney.
Histopathologic Evaluation
Microscopically, the renal cortex of the affected right kidney was thin, the medulla was underdeveloped with no corticomedullary junction and only a few collecting duct-like structures present near the pelvic region, and the papilla was absent (Figure 1A). The majority of the cortical tissue consisted of attenuated poorly differentiated tubules, eosinophilic collagenous matrix that expanded the interstitium and encircled glomeruli, and moderate interstitial mononuclear cell infiltrates including pigment-laden macrophages (Figure 1C-E). The attenuated tubules were lined by cuboidal epithelial cells with minimal basophilic cytoplasm and indistinct brush borders and had crowded hyperchromatic nuclei resembling immature undifferentiated tubules or possibly dedifferentiated tubules. Only small clusters of mature proximal tubules were present within the cortex.
The glomeruli appeared closer together due to parenchymal collapse and the lack of fully developed cortical tubules. Most glomerular Bowman’s capsules were encircled by a connective tissue/matrix collar. Occasional small atrophic glomeruli and variably sclerotic small glomeruli were also present. Ghosts of obsolescent small glomeruli were evident within the regions of increased cortical connective tissue. Medullary rays contained reduced numbers of collecting ducts and thick ascending limbs. The corticomedullary junction was absent. Interstitial mononuclear infiltrates extended into the “medullary” area surrounding the collecting duct-like structures. The ducts had varying epithelial differentiation from low cuboidal to tall columnar with rare mitotic figures, otherwise resembled normal collecting ducts (Figure 1F). The interstitium was expanded by connective tissue stroma/matrix in the area occupied by these collecting ducts and in particular, the smaller ducts. Eosinophilic homogenous material and/or cellular debris was present within the lumen of some of these ducts. The renal pelvic urothelium was hyperplastic and extended over the surface of the rudimentary medulla instead of what should have been the epithelium of the renal papilla. Thus, the underdeveloped medulla appeared to be directly connected to the renal pelvis without the formation of the papilla. In addition, arterial and arteriolar hypertrophy was present with the spiraling of the smaller vessels. The contralateral kidney did not exhibit compensatory tubular hypertrophy (data not shown). There were no noteworthy microscopic findings in the control animal’s kidneys (Figure 1B). Also, no other urogenital abnormalities were present in the affected monkey.
Histochemical and Immunohistochemical Stains
A summary of the histochemical and immunohistochemical staining results for the normal control cynomolgus monkey kidney and the abnormal kidney are presented in Table 2 and are briefly described.
Alcian Blue
The interstitial matrix in the medulla of the normal kidney stained AB+ as expected, but the interstitial matrix in the medullary region of the affected kidney had decreased staining. The matrix collars around the glomerular Bowman’s capsules and the intima and media of blood vessels had increased AB+ staining in the affected kidney (Figure 2A and B).
Masson’s Trichrome
Bowman’s capsule and tubular basement membranes stained MTC+ (blue) as expected in both the normal kidney and the unaffected structures of the affected kidney. However, in the affected kidney, increased blue MTC+ staining was observed in sclerotic glomeruli, the increased cortical and medullary interstitial matrix, the basement membranes encompassing the poorly differentiated cortical tubules, matrix collars around Bowman’s capsules, and within the intima and media of blood vessels (Figure 2C and D).
Periodic Acid Schiff
Glomerular and tubular basement membranes, mesangium, and proximal tubule brush border stained PAS+ (dark pink) as expected in both the normal kidney and the unaffected structures of the affected kidney. However, increased PAS+ basement membrane staining was observed encompassing the poorly differentiated cortical tubules and within sclerotic glomeruli in the affected kidney. The medullary interstitium in the normal kidney and the increased cortical and medullary interstitial matrix in the affected kidney also stained PAS+, but the medullary interstitial matrix in the affected kidney had less PAS+ staining compared to the normal kidney. Some of the medullary collecting duct-like structures had PAS+ homogenous material within their lumens (Figure 2E and F).
Schmorl’s Method
No SM+ staining was observed in the normal kidney. Multifocal SM+ staining macrophages were seen within the cortical and medullary interstitium of the affected kidney indicating the presence of intracytoplasmic lipofuscin pigment (data not shown).
Congo Red
No CR+ staining/birefringence was observed in the normal or affected kidney indicating that amyloid was not present (data not shown).
Alpha-Smooth muscle Actin
The vascular smooth muscle and interstitial cells had cytoplasmic α-SMA+ immunostaining in the normal and affected kidney as expected, but there were increased numbers of α-SMA+ interstitial cells in the interstitium of the medulla region in the affected kidney. In the cortex, the matrix collars surrounding Bowman’s capsules stained α-SMA+ in the affected kidney (Figure 3A and B).
Vimentin
In the normal kidney, cytoplasmic VMTN+ immunostaining was observed in the glomerulus, blood vessels, capillaries, immune cells, and connective and adipose tissues. However, VMTN+ immunostaining was increased in the affected kidney including the matrix collars around Bowman’s capsules, the expanded cortical and medullary interstitial matrix, many of the poorly differentiated cortical tubules, and some of the smaller medullary collecting duct-like structures (Figure 3C and D).
Wilms’ Tumor
In the normal kidney and in the affected kidney, nuclear WT1+ immunostaining was observed in glomerular podocytes. 14,50,51 All other segments of the nephron were negative in both the normal and the affected kidneys (Figures 3E and F).
Pancytokeratin
Bowman’s capsule epithelium, all nephron tubular segments, papillary epithelium, and pelvis urothelium stained panCK+ as expected in both the normal kidney and the unaffected structures of the affected kidney. However, in the affected kidney, immunostaining was also observed in the poorly differentiated cortical tubules and in the medullary collecting duct-like structures. Thin limbs and thick ascending limbs in the medullary region were not discernible with this immunostain as well with the other immunostains (data not shown).
S100
Distal tubules, thin limbs and thick ascending limbs, cortical connecting and collecting ducts, medullary collecting ducts, papillary epithelium, pelvis urothelium, and nerves had cytoplasmic S100+ immunostaining in both the normal kidney and the unaffected structures of the affected kidney. 41 However, the affected kidney had S100+ immunostaining in the mature proximal tubules and in the poorly differentiated cortical tubules and had increased S100+ immunostaining in some of the smaller medullary collecting duct-like structures (Figure 4A and B).
Paired Box Homeotic Gene 2
In the normal kidney, there was consistent nuclear PAX2+ staining observed in the medullary collecting ducts, and only rare nuclear PAX2+ staining in the cortex. 14,50,52 In the affected kidney, there was an increased number and/or intensity of nuclear PAX2+ immunostaining in the poorly differentiated tubules, cortical connecting and collecting ducts, and in the medullary collecting duct-like structures (Figure 4C and D). However, a few of the larger medullary collecting duct-like structures were either negative or were only partially PAX2+ (Figure 4D).
Beta-Catenin
In the normal kidney, predominantly basolateral membrane and lesser cytoplasmic CTNB+ immunostaining was observed in all epithelium and endothelium throughout the kidney, but more intense cytoplasmic staining was observed in the distal convoluted tubules 53 (Figure 4E and F). In the affected kidney, CTNB+ immunostaining of the remnant normal structures paralleled that of the control kidney, but Bowman’s capsule epithelium, poorly differentiated cortical tubule epithelium, and medullary collecting duct-like epithelium had increased cytoplasmic and membrane immunostaining compared to similar structures in the control kidney (Figure 4E and F).
Aquaporin 1
In the normal kidney, proximal tubules, rare Bowman’s capsule epithelium, descending thin limbs of long looped nephrons, descending vasa recta, glomerular and vascular endothelium, and red blood cells had AQP1+ cytoplasmic and membrane immunostaining. 54 -56 In the affected kidney, there were only a few small poorly differentiated cortical tubules that stained AQP1+, although nests of morphologically mature proximal tubules stained positive. Also, occasional Bowman’s capsule epithelium had increased AQP1+ immunostaining (not shown). There were also lesser numbers of AQP1+ descending thin limbs in the cortex and none in the underdeveloped medulla. Endothelium throughout the affected kidney was AQP1+ including some vessels in the underdeveloped medulla, but distinct distal vasa recta were not present (Figure 5A and B).
Aquaporin 2
In the normal kidney, cortical connecting and collecting ducts and medullary collecting ducts had AQP2+ cytoplasmic and apical membrane immunostaining; more intense AQP2+ immunostaining was observed in principal cells compared to intercalated cells. 55 In the affected kidney, there were lesser numbers of AQP2+ connecting and collecting ducts in the cortex, with only a few AQP2+ collecting duct-like structures in the underdeveloped medulla. The majority of the medullary collecting duct-like structures in the affected kidney were negative and/or had variable apical membrane AQP2+ immunostaining, which was less intense than in the normal kidney medullary collecting ducts (Figure 5C and D).
Uromodulin
In the normal kidney, thick ascending limbs including macula densa had UMOD+ cytoplasmic immunostaining. 54,55 In the affected kidney, there were lesser numbers of UMOD+ thick ascending limbs in the cortex. There was an absence of the outer medullary stripe region which normally is densely populated with thick ascending limbs in addition to thin limbs and collecting ducts. There were no UMOD+ tubules in the underdeveloped medulla (Figure 5E and F).
Calbindin-D28 k
In the normal kidney, distal tubules, macula densa, cortical thick ascending limb, cortical connecting and collecting ducts, and medullary collecting ducts had CALB+ cytoplasmic and apical membrane immunostaining; more intense CALB+ immunostaining was observed in the distal tubules and the principal cells of the cortical connecting and collecting ducts than in the medullary collecting ducts, thick ascending limbs, and the macula densa. 54,55 In the affected kidney, CALB+ immunostaining paralleled the staining in the normal kidney, but there were lesser numbers of CALB+ connecting and collecting ducts in the cortex and only a few CALB+ collecting duct-like structures in the underdeveloped medulla. The medullary collecting duct-like structures immunostained for CALB+ more intensely than the normal kidney’s medullary collecting ducts, and some of the larger medullary collecting duct-like structures were negative and/or had variable CALB+ immunostaining. In addition, some of the poorly differentiated cortical tubules were variably CALB+ (Figure 6A and B).
Ki67
Rare ki67+ nuclear staining was noted in all components of the nephron in the normal kidney and in the unaffected structures of the affected kidney. 57 In the affected kidney, increased ki67+ immunostaining was observed in only a few of the medullary collecting duct-like structures in the hyperplastic pelvic urothelium, glomerulus, and in the cortical and medullary interstitium, which was most often associated with inflammatory cell infiltrates (Figure 6C and D).
Uroplakin II
In the normal kidney, the superficial layers of the pelvic urothelium had UPII+ membrane and cytoplasmic immunostaining, and the epithelium of the renal papilla was negative. 58,59 In the affected kidney, the epithelium covering the remnant papilla-like structure immunostained UPII+ as well as the pelvic urothelium (Figure 6E and F).
Discussion
This manuscript describes the histopathologic findings of a spontaneously occurring renal aplasia in a cynomolgus monkey, adding to the limited published literature characterizing renal congenital abnormalities in this species. Renal aplasia is described as an incompletely or defectively developed kidney, and microscopically the affected structures may be absent or only partially developed. 10 Aplasia may result from early regression of the ureteric bud, altered metanephric differentiation, or defects in communication between the branching ureteric duct and undifferentiated metanephric blastema. 12 In contrast, renal dysplasia is characterized by disorganized development of the renal parenchyma due to asynchronous differentiation caused by imperfect inductive interaction between the mesonephric duct (Wolffian duct) and the metanephric blastema. Histologically, the dysplastic kidney has the persistence of abnormal structures consisting of undifferentiated mesenchyme, metanephric ducts, primitive glomeruli, immature tubules, anomalous interstitial fibrous connective, bone or cartilage, and abnormal patterning of cortical and medullary tissues. 19,43,47,60 Due to the morphologic similarities between aplasia and dysplasia, microscopic diagnosis is often challenging. Thus, careful histopathologic examination aided by histochemical and immunohistochemical staining was necessary to ensure the correct interpretation of the macaque renal lesion.
In the few published reports of renal dysplasia in macaques, features included a small unilaterally affected kidney with microscopic evidence of interstitial fibrosis, fibromuscular collars around glomeruli and tubules, immature nephrons, persistent mesenchyme, primitive tubules, atypical tubular epithelium, and/or hypertrophic arteries with spiraling. 22,24,25 However, there are numerous publications characterizing the histopathologic attributes of spontaneous and/or familial renal dysplasia in dogs and include the presence of fetal or immature glomeruli, fetal or immature tubules, persistent metanephric ducts surrounded by primitive mesenchyme, bone or cartilage in the parenchyma, and anomalous presence of interstitial fibrous connective tissue. 43 However, asynchronous differentiation of nephrons (presence of immature glomeruli, or tubules, or both) could not be detected in all canine renal dysplasia cases. 43 Unlike human renal dysplasia, cartilaginous or osseous metaplasia is rarely present in the dysplastic kidneys of animals. 19,43
In regard to this macaque kidney, a diagnosis of renal aplasia was preferred over dysplasia for the following reasons: (1) incomplete development of the renal medulla and papilla rather than disorganized development/abnormal patterning noted with renal dysplasia; (2) the absence of key histologic features (primitive tubules, undifferentiated mesenchyme, primitive glomeruli, metanephric ducts, and/or anomalous tissues such as bone or cartilage) of dysplasia; and (3) absence of characteristic nuclear immunostaining for WT1 and β-catenin which define renal dysplasia in human cases, while cytoplasmic and/or membrane staining was retained.
Morphologically, the aplastic kidney was macroscopically small and microscopically characterized by a thin cortex, undeveloped medulla with no papilla, a paucity of mature cortical and medullary tubules, extensive deposition of interstitial connective tissue, fibromuscular collar formation around Bowman’s capsules, small atrophic and sclerotic glomeruli, and interstitial inflammation. Although there was a paucity of mature cortical and medullary tubules, small islands of fully developed AQP1+ cortical tubules were occasionally present, as well as some AQP2+ cortical connecting and collecting ducts. The lack of normal medullary collecting duct development was characterized by an absence of AQP1+ immunostaining in descending thin limbs and an absence of URMOD+ immunostaining in thick ascending limbs in the outer medulla. Only rare AQP1+ vessels (vasa recta) were noted in the medulla. The absence of epithelium of the renal papilla with only hyperplastic UPII+ urothelium confirmed the lack of development of the papilla. Unlike the normal kidney’s medullary interstitium which stained positive for PAS, AB, MTC, and VMTN indicating a matrix composed of connective tissue, proteoglycans, and glycosaminoglycans, 61 the affected kidney’s medullary interstitial matrix had decreased PAS and AB staining, supporting a lack of development of the normal interstitial matrix constituents.
The numerous small poorly differentiated cortical tubules without lumens, brush borders, or other distinguishing features stained positive for epithelial (panCK and S100) and mesenchymal/dedifferentiation (PAX2 and VMTN) markers characteristic of cells undergoing epithelial to mesenchymal transition (EMT). 50,62 -64 In fetal human kidneys, the co-expression of cytokeratins and VMTN was detected in the visceral and parietal epithelium of the glomerulus, the proximal tubules, the thin loops of Henle, and the collecting ducts, whereas, in the normal adult kidney, the presence of VMTN and cytokeratins was nearly always mutually exclusive. 65 However, both acute and chronic damage to renal tubular epithelium has been followed by the co-expression of VMTN and cytokeratin. 66 Likewise, VMTN-positive regenerative or dedifferentiated renal tubules have been described in rodents, 62 bovine, 67 canine, 68 and humans 63,69 following injury and/or inflammation. Kusaba et al 70 found that fully differentiated proximal tubule cells that survive injury undergo a process of reversible dedifferentiation and proliferation during repair which is characterized by transient induction of both injury markers and developmental proteins that are subsequently downregulated after redifferentiation is complete. Recent work by Lazzeri et al 71 have shown that a small subset of Pax2+ tubular progenitors have higher stress resistance and undergo limited clonal expansion and mitosis to regenerate necrotic tubule segments following acute kidney injury (AKI) resulting in a net loss of tubular epithelial cells. Instead, renal functional recovery upon AKI predominantly involves remnant tubular cell hypertrophy via endoreplication cycles (alternative cell cycles without cell division leading to polyploid cells with a single nucleus) to handle the sudden increase in filtration load in the uninjured nephrons. However, this hypersecretory state is associated with persistent secretion of profibrotic mediators driving fibrogenesis, cell senescence, and further injury. 72 In the current case, the majority of the poorly differentiated cortical tubules were PAX2+ with only a few islands of remnant normal tubules present. The continued renal compromise in the affected kidney over the lifetime of this monkey resulted in the loss of normal as well as potential polyploid tubule epithelial cells with survival and expansion of the PAX2+ regenerative cells. Unexpectedly, some of the poorly differentiated cortical tubules immunostained CALB+. Proximal tubule expression of CALB has been reported in a diabetic mouse model, and the authors speculated that calbindin played a cytoprotective role in preventing apoptotic tubular cell death induced by diabetes. 73 Studies in other cell types have shown that calbindin protects against cell death by different mechanisms including calcium buffering. 74,75 Thus, the CALB+ immunostaining in the poorly differentiated cortical tubules could represent a protective response to injury rather than distal tubule/collecting duct phenotype. Additionally, the presence of immature/primitive tubules often closely associated with and/or encircled by primitive mesenchyme have been described as a feature of renal dysplasia in multiple species including rodents, 40 domestic animals, 30,31,36,39,43 nonhuman primates, 25 and humans. 14 However, in the current case, the poorly differentiated cortical tubules were not associated with primitive mesenchyme and most likely represent dedifferentiated tubules in response to chronic secondary injury and inflammation in this anomalous kidney.
Increased primitive mesenchymal matrix which is AB-positive and MTC-negative has been described as a feature of dysplastic kidneys in other species including rodents 42 and domestic animals. 30,35,39,43 In the aplastic kidney, the increased cortical and medullary interstitial matrix stained positive for MTC and VMTN but was negative to minimal for AB indicative of mature collagenous connective tissue. However, the prominent matrix collars that surrounded Bowman’s capsules in the affected kidney stained positive for a mixture of markers (MTC, AB, α-SMA, and VMTN), indicating that the matrix tissue was not fully differentiated and undergoing mesenchymal-myocyte transformation. Similar α-SMA and/or VMTN immunostaining of peri-glomerular and/or peri-ductal fibromuscular collars have been previously reported in dysplastic kidneys from monkeys 25,49 and bull 36 and in the metanephric mesenchyme of developing monkey kidneys. 50 However, in the current case, the periglomerular matrix cuffs most likely represent a feedback response from the impaired tubules to the glomerulus resulting in thickening of the Bowman’s capsular basement membrane and reactive periglomerular fibrosis and was not persistent metanephric mesenchyme as seen in dysplastic kidneys. 76 In addition, the medullary interstitial matrix had minimal AB staining, and peri-ductular α-SMA+ collars were not present surrounding the collecting duct-like structures consistent with a lack of persistent mesenchyme as has been described in dysplastic kidneys.
Small glomeruli representing immature/fetal glomeruli have been reported in the dysplastic kidney of multiple different species including rodents, 40 domestic animals, 30,36,39,43,77 nonhuman primates, 22 and humans. 60 However, the absence of fetal/immature glomeruli has also been reported in the dysplastic kidneys from dogs 78 and cats. 38 In the current case, increased PAS and MTC staining was observed in occasional small glomeruli, indicative of sclerosis of atrophic/effete glomeruli. In a previously reported case of renal dysplasia in a cynomolgus monkey, the authors considered that the glomerulosclerosis was secondary to persistent urinary flow impairment. 25 The current case did not have evidence of urinary flow impairment such as dilated tubules or Bowman’s capsules. Thus, the sclerotic glomeruli in this case were most likely a secondary event associated with primary tubular injury. 79
The medullary collecting duct-like structures stained positive for panCK, had variable CALB staining, and had decreased AQP2 immunostaining, indicating incomplete maturation and/or loss of collecting duct differentiation. 48,54,80 Vimentin-positive immunostaining and increased S100 and PAX2 immunostaining observed in most of these ducts corroborate an immature phenotype and not mesonephric duct origin, which is cytokeratin positive but S100 and VMTN negative. 36,41,50 In a rhesus model of fetal urinary tract obstruction, collecting duct intercalated cell EMT contributed to the formation of medullary α-SMA+ peritubular fibrous collars and interstitial fibrosis. In addition, the obstructed collecting duct epithelium lost E-cadherin and AQP2 and gained VMTN expression, suggesting a loss of epithelial and gain of mesenchymal characteristics. 48 Likewise, in a study of obstructed human fetal kidneys, similar features of collecting duct EMT were described, suggesting that this transition can occur in cells that are not terminally differentiated. 81 Although there was no evidence of renal obstruction in the current case, the collecting duct-like structures exhibited a similar mixed epithelial-mesenchymal phenotype which was also seen in the poorly differentiated cortical tubules.
Mouse gene-targeting studies have identified key developmental pathways and specific genes that cause CAKUT. 14,82,83 The WT1, PAX 2/8, and B-cell lymphoma 2 (BCL2) genes have been identified as being important in regulating normal branching morphogenesis and nephron differentiation during nephrogenesis. These genes have also been shown to be involved in the deregulated proliferation and apoptosis observed in the abnormal metanephric elements of the dysplastic kidney. 14,84 In addition, both the ureteric bud and the metanephric blastema progenitor cells require canonical Wnt/β-catenin signaling for normal morphogenesis, and perturbation of ureteric branching in transgenic mouse models resulted in increased β-catenin expression which contributed to the development of renal dysplasia. 85,86 Thus, the development of renal dysplasia and other CAKUT may be the final common pathway for different initiating events (genetic or environmental). 46
All glomeruli in the affected kidney including atrophic glomeruli and glomeruli undergoing sclerosis had WT1-positive nuclear immunostaining as would be expected for the maintenance of differentiated podocytes. 50,51 However, the lack of positive WT1 immunostaining in matrix tissue associated with/surrounding poorly differentiated cortical tubules, periglomerular cuffs, or the medullary tubules argues against the presence of persistent mesenchyme due to altered WT1 expression in the affected kidney. The canonical Wnt/β-catenin signaling pathway is also critical for renal development and involves cytoplasmic accumulation and translocation of the β-catenin protein into the nucleus, where it activates gene transcription; however, it is also involved in cell–cell adhesion and in epithelial–mesenchymal communication. 87,88 During kidney development, both nephrogenic induction and ureteric morphogenesis require β-catenin signaling. 88 Studies have shown that elevation of β-catenin levels during kidney development causes dysplasia 86,89 and that activated β-catenin levels are elevated in the nucleus of the ureteric epithelium, stroma, and mesenchyme in human dysplastic kidneys. 90 In the current case, although increased staining intensity of cytoplasmic and/or membrane β-catenin immunostaining was observed in Bowman’s capsule epithelium, poorly differentiated cortical tubule epithelium, and medullary collecting duct-like epithelium, no nuclear immunostaining was observed in any tubules, ducts, or stromal components arguing against elevated β-catenin expression as seen in dysplastic kidneys. Under normal conditions, the majority of β-catenin is sequestered at the cell membrane in complex with cadherin proteins, but when adherens junctions are destabilized during epithelial simplification or detachment of adjacent cells, β-catenin is released into the cytoplasm. 91 Thus, the increased staining intensity of cytoplasmic and/or membrane staining seen in the affected kidney most likely reflects the poorly differentiated state of these structures.
Important aspects of the potential molecular pathogenesis of renal aplasia in this monkey can be gleaned from both the absence of specific structures and some of the immunohistochemical data. Nephron maturation occurs in a proximal to distal hierarchy such that the distal structures form last during kidney development. 4,83,92 Delayed development of the distal structures may reflect the need to generate proximal filtration and recovery of low-molecular-weight compounds (eg, glucose and sodium) prior to the need for other renal functions. 5 Work in mice has shown that the uniformly branched ureteric bud (UB) network is generated first, followed by zone-specific elongation of interbranch UB segments and that the sixth to eighth generation of UB branches give rise to the long, unbranched outer medullary collecting ducts. 93 Renal stromal cells located adjacent to the UB branches that give rise to outer medullary collecting ducts secrete factors that control the elongation of UB segments. 94 In fact, mice with a null mutation in forkhead/winged helix transcription factor FoxD1 (BF-2), which is expressed exclusively in stromal cells in the developing kidney, lack elongated outer medullary collecting ducts. 95 In addition, Wnt7b is expressed exclusively in the non-branching collecting duct epithelium, and it is also essential for the coordinated growth of the loop of Henle. The absence of Wnt7b activity leads to medullary development failure although cortical development is normal with ureteric branching and nephrogenesis unaffected. 96 Canonical Wnt signaling molecules are expressed in the adjacent interstitial mesenchyme and removal of β-catenin also results in an absent medulla. 96 In addition, many other gene mutations in mice have resulted in defective renal medulla morphogenesis providing insight into the key genetic regulators of this process. 97 However, the mechanisms by which the inner medullary collecting ducts converge centrally to form the papilla and how collecting ducts enter the tip of the papilla remains unknown. 97 This case of renal aplasia mimics the morphologic phenotype seen in mutant mice which lack one of the many key genes involved in medullary development. Thus, some events during the latter part of gestation may have perturbed the molecular signaling during medullogenesis, resulting in the incomplete development of the renal medulla and papilla in this animal. Additionally, immunostaining for molecular markers of dedifferentiation and/or injury (VMTN, PAX2, and CALB) in the affected tubules provide evidence for secondary degenerative phenomena contributing to the pathophysiology.
Footnotes
Acknowledgments
The authors wish to thank Mark Sheffield from Covance Laboratories Inc. in Madison Wisconsin, USA, and John Kreeger from Inotiv in Gaithersburg, Maryland, for reviewing the manuscript and providing feedback.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
