Abstract
Testicular fibrous hypoplasia is an incidental lesion characterized by replacement of the testicular parenchyma by mature collagen. A retrospective survey of hematoxylin and eosin–stained testicular sections from 722 purpose-bred Asian and 90 Mauritian cynomolgus monkeys from 56 safety assessment studies conducted between 1999 and 2011 was performed. The incidence of the lesion increased markedly over time. No cases occurred between 1999 and 2004. Between 2005 and 2009, the incidence ranged between 8.1% and 11.0% of the monkeys examined and then rose to 26.1% in 2010 and 30.9% in 2011. Overall, the lesion was identified in 10.94% of Asian monkeys with the highest incidence in animals originating from China and Vietnam; severity ranged from minimal to severe and it occurred unilaterally (38.5%) and bilaterally (61.5%). In Mauritian monkeys, the lesion was predominantly minimal in severity, bilateral in distribution, and affected 6.6% of the animals examined. The lesion occurred regardless of sexual maturation status but when present in mature monkeys was often associated with cystic tubular atrophy of the seminiferous epithelium. Based on the morphological characteristics of the lesion and the unilateral/bilateral distribution, the lesion is considered to be a congenital or developmental abnormality.
Cynomolgus monkey (Macaca fascicularis) is the most commonly used nonhuman primate species for nonclinical safety evaluation of pharmaceuticals. It is also used commonly in other areas of biomedical research. Evaluation of induced pathologic changes relies on a detailed knowledge of the normal background, incidental pathology in the species under investigation. Despite the availability of some literature in primate pathology, relatively little has been published about disorders of the male reproductive system in nonhuman primates. In 2011, we described an unusual lesion of the testis of cynomologus monkeys, characterized by replacement of varying amounts of the testicular parenchyma by mature collagen (Pereira and Creasy 2011). We suggested the term “fibrous hypoplasia” for this lesion on the basis of increased amounts of collagen replacing seminiferous tubules and interstitial tissue and proposed a congenital or developmental origin for the lesion. The same or similar lesion has since been reported by a number of other investigators (D. Creasy 2012; Sato et al. 2012; D. M. Creasy and Chapin 2013; Patrick and Rebellato 2015; Kozlosky et al. 2015). In all cases, the lesion was described in immature animals. Based on the current authors’ experience, this fibrotic lesion appears to be increasing in incidence over time and occurs in sexually mature animals as well as immature monkeys. The current retrospective study of 812 cynomolgus monkeys was conducted to investigate the detailed morphological features of the lesion, its occurrence over time (1999–2011), the maturity status of the affected monkeys, and whether the lesion was unilateral or bilateral in its distribution.
Material and Method
A retrospective survey was conducted in 722 male Asian (Indochinese) cynomolgus monkeys and 90 male Mauritian cynomolgus monkeys from control and test article–treated groups from 56 toxicity studies (51 studies with Asian and 5 with Mauritian monkeys). The studies were conducted between 1999 and 2011.
Source and Country of Origin of Animal Supply
The animals were purpose-bred for laboratory use. The Asian cynomolgus monkeys were obtained from Covance Research Products (Alice, TX or Denver, PA), Primate Products (Miami, FL), R. C. Harlust Tiilburg (the Netherlands), Rhenos LLC (Newton, MA), Buckshire Corporation (Perkasie, PA), Bristol–Myers Squibb (Somerville, NJ), or Labs of Virginia (Yemassee, SC). The country of supply of the Asian monkeys included China, Cambodia, Indonesia, and Vietnam. In a few cases, the precise country of supply was unknown because the cohort was sourced from two or more Asian countries. The Mauritian cynomolgus monkeys were supplied by Covance Research Products (Alice, TX).
Study Details
The survey included both testes from all monkeys (control and test article–treated animals) from 56 studies performed at Huntingdon Life Sciences (now Envigo, Inc.), East Millstone, NJ, between 1999 and 2011. There were no test article–related testicular findings identified in any of the studies examined. All procedures and care of animals for each study were conducted in accordance with the principles for humane care outlined by the Institute of Laboratory Animal Research Guide for the Care and Use of Laboratory Animals, the United States Department of Agriculture (USDA) Animal Welfare Act, and were reviewed and approved by the Huntingdon Life Sciences Institutional Animal Care and Use Committee. The monkeys were euthanized by exsanguination following intravenous injection of sodium pentobarbital. The testes were weighed and fixed in modified Davidson’s solution, for a minimum of 48 hr prior to storage in 10% neutral buffered formalin. Transverse sections of both testes were taken slightly cranial to the midline to ensure sampling of the rete testis, processed and embedded in paraffin wax, mounted on glass slides, and stained with hematoxylin and eosin stain using standard procedures. For developing and mature monkey testes, samples always included the mediastinal testis and the rete, but the edges of the testes were trimmed to allow sections to fit on the microscope slide. The trimming, processing, and sectioning procedures were all performed at the same facility in accordance with the Standard Operating Procedures of Huntingdon Life Sciences. Additional sections from 1 fibrotic testis and 1 normal testis, each from a different Asian monkey, were stained with Masson’s trichrome stain for the demonstration of connective tissue. All sections of testes were reexamined microscopically for the presence and severity of fibrosis, whether unilateral or bilateral and for the level of sexual maturation based on the degree of tubular spermatogenesis (M.P.B. or V.V.). The presence of the lesion in one or both testes was then correlated with the year of the study and the country of origin of the monkey.
Diagnostic Criteria and Severity Grading
The presence and severity grade of the testicular lesion was recorded based on the following 5-grade system:
Minimal: Focal; small; excessive amounts of collagen within the rete testis, septum, and/or mediastinum; and <10% of the testicular section was affected.
Mild: Multifocal; small; excessive amounts of collagen within the rete testis, septum, and/or mediastinum; between 10% and 30% of the testicular section was affected; and seminiferous tubule numbers reduced due to replacement by collagen.
Moderate: Multifocal to coalescing; large amounts of collagen within the rete, septum, and/or mediastinum; between 30% and 50% of the testicular section was affected; capsule sometimes appeared thickened; and seminiferous tubule numbers reduced due to replacement by collagen.
Marked: Multifocal to coalescing; large amounts of collagen within the rete testis, septum, and mediastinum; between 50% and 75% the testicular section was affected; capsule often appeared thickened; and seminiferous tubule numbers markedly reduced due to replacement by collagen.
Severe: >75% of the testicular parenchyma was replaced by collagen affecting rete testis, septum, mediastinum, and capsule and seminiferous tubule numbers markedly reduced due to replacement by collagen.
Diagnostic Criteria for Sexual Maturation Status
Using morphological criteria similar to the criteria used by Lawrence and Saladino (2009) and Luetjens and Weinbauer (2012), the maturation status of the testes was recorded on the following 3-grade system:
Immature: Incomplete spermatogenesis in all seminiferous tubules, presence of only Sertoli cells and undifferentiated spermatogonia, cross section of seminiferous cords with small diameter, and no lumen.
Maturing: Tubular lumen present, mixture of seminiferous tubules with complete and incomplete spermatogenesis, and approximately 25–75% of tubules contain elongated/maturation phase spermatids.
Mature: Approximately >75% of the seminiferous tubules with complete spermatogenesis including elongating and maturation phase spermatids
Results
Incidence of the Lesion in Asian Monkeys
Of the 722 Indochinese monkeys, 464 (64.3%) were sexually immature, 68 (9.4%) were maturing, and 190 (26.3%) were sexually mature. Of the 722 monkeys, 79 had fibrous hypoplasia in one or both testes (10.9%). The lesion occurred regardless of maturational status, but, overall, it was more prevalent in immature or maturing monkeys than in mature monkeys. It was present in 14.0% of immature, 14.7% of maturing, and 2.2% of mature monkeys (Figure 1). Out of the 79 affected animals, 65 (82.2%) were sexually immature, 10 (12.6%) were maturing, and 4 (5.0%) were sexually mature. Based on the country of supply, the lesion was most commonly seen in monkeys sourced from China and Vietnam (19.8% and 14.4%, respectively). It was much less common in monkeys from Cambodia and Indonesia (4.4% and 0.9%, respectively; Figure 2). There was no correlation between the incidence of the lesion and the commercial supplier of the monkeys.
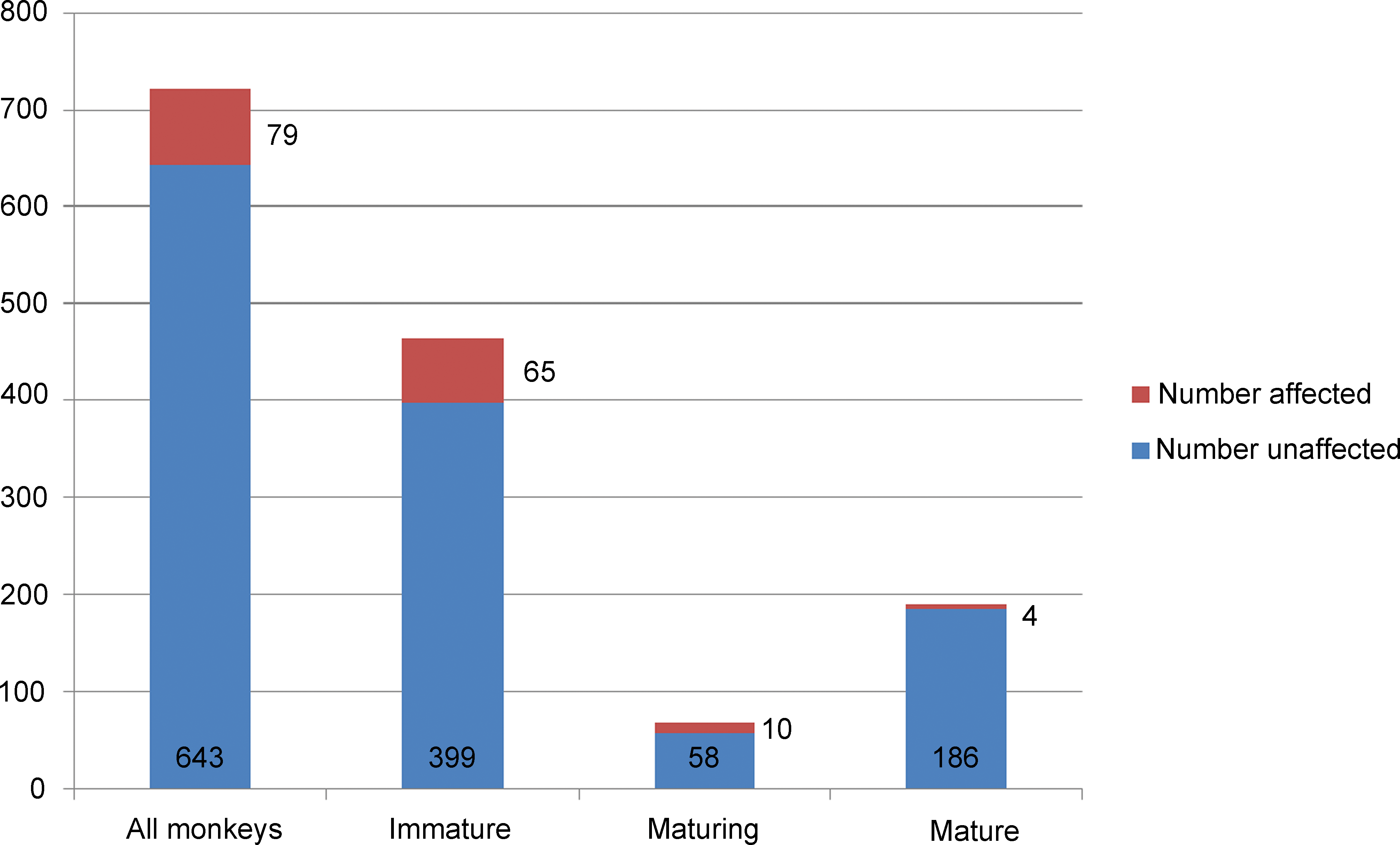
Proportion of immature, maturing, and mature Asian monkeys affected by fibrous hypoplasia from studies performed between 1999 and 2011.
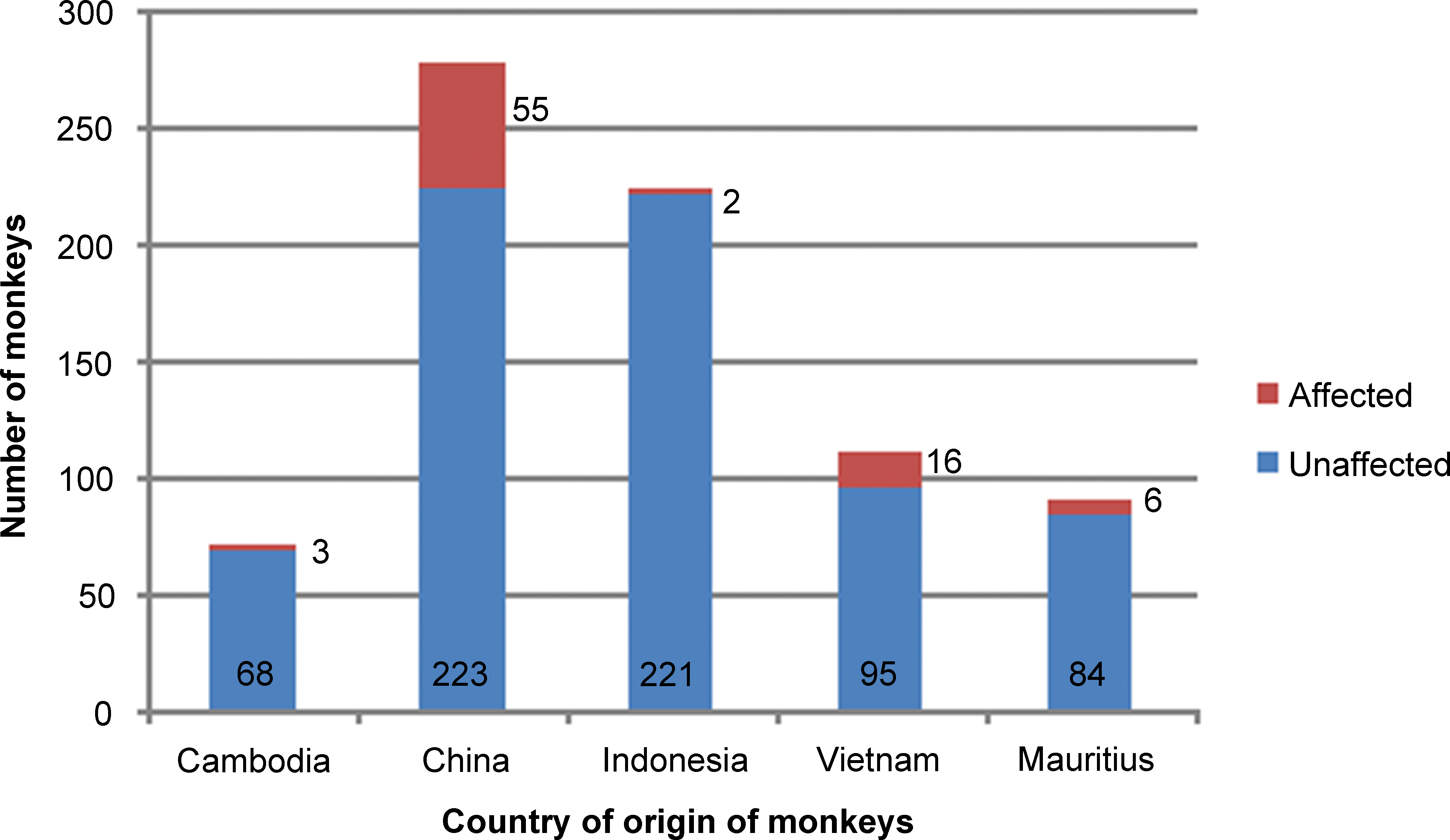
Country of origin of monkeys with testicular fibrous hypoplasia.
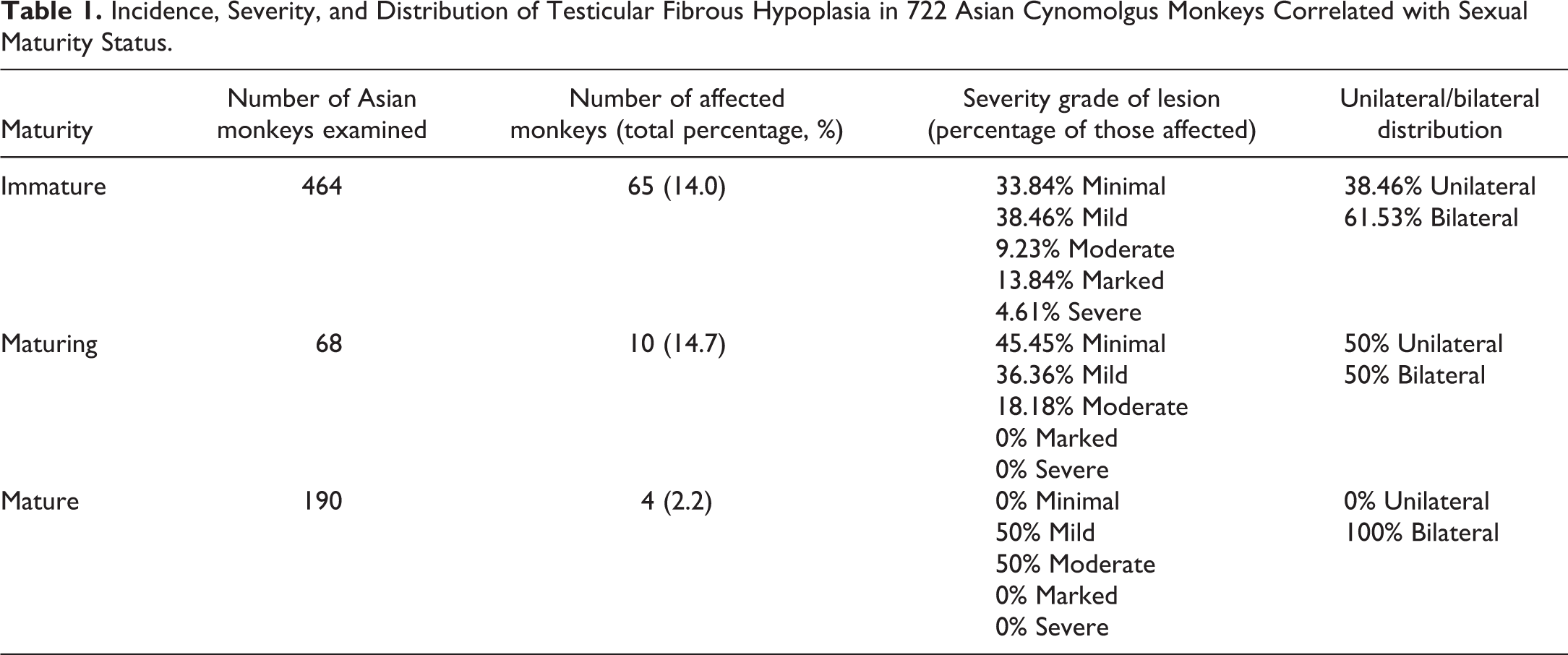
Rete/mediastinal and septal fibrosis occurred bilaterally in 61.5% of cases and unilaterally in the remaining 38.5% of cases. The severity grade of the fibrosis ranged from minimal to severe; but the majority of animals had only minimal or mild severity (Table 1).
Incidence, Severity, and Distribution of Testicular Fibrous Hypoplasia in 722 Asian Cynomolgus Monkeys Correlated with Sexual Maturity Status.
Incidence of the Lesion in Mauritian Monkeys
Out of the 90 Mauritian monkeys, 46 (51.1%) were sexually immature, 14 (15.5%) were maturing, and 30 (33.3%) were sexually mature. Minimal fibrosis of the mediastinum and/or septum of one or both testes was present in 6 out of the 90 monkeys (6.6%). Of the 3 affected animals, 1 was immature (unilateral fibrosis), 1 maturing (bilateral fibrosis), and 1 was sexually mature (bilateral fibrosis).
Correlation of Fibrous Hypoplasia with Year of Necropsy
The incidence of the lesion in Asian monkeys was plotted against the year of necropsy of the affected monkeys. There was an obvious increase in the frequency of the lesion with time (Figure 3). No cases were seen in studies terminating between 1999 and 2004. The first case was seen in an immature monkey in 2005 and then the incidence rose to 8.1 to 11.0% in the years between 2006 and 2009. In 2010, the incidence more than doubled to 26.1% and rose again in 2011 to 30.9% of monkeys examined.
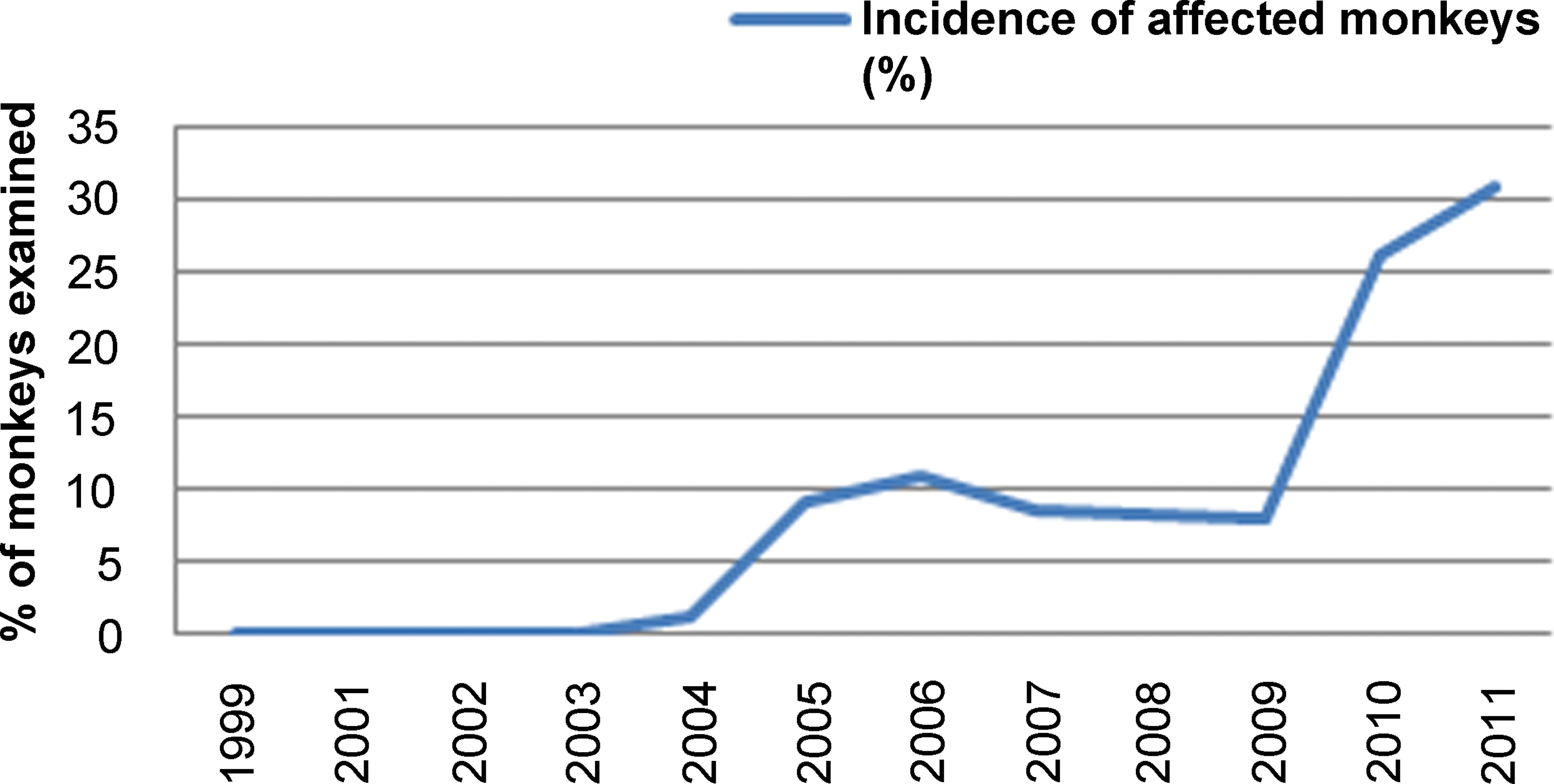
Increasing incidence of monkeys with testicular fibrous hypoplasia over time.
Morphologic Characterization of the Lesion
The morphologic characteristics of the lesion varied depending on the severity of the fibrosis and the maturational status of the testis. In immature testes, the fibrotic lesion appeared to originate in the mediastinal tissue surrounding the rete testis with variable involvement of the interlobular septa and the testicular capsule (Figure 4A–D). In normal testes, the rete comprises a series of epithelial-lined ducts embedded in a relatively narrow band of collagen fibers, which form the mediastinum while the interlobular septa comprise very thin strands of collagen (Figure 4A). In testes with minimal to mild fibrous hypoplasia (Figure 4B), the amount of collagen surrounding the rete ducts and between the lobules was increased, resulting in an overall expansion of the mediastinum testis. In moderate to severe cases, the anatomy of the rete testis appeared disrupted and dispersed by the excess mature collagen bundles between the ducts (Figures 4–6). As severity increased, the collagen deposition replaced varying amounts of the testicular parenchyma (seminiferous tubules, interstitial tissue, and blood and lymphatic vessels). This was sometimes accompanied by an increase in the thickness of the capsule (Figures 5 and 6). The abnormal fibrous tissue resembled the connective tissue of the tunica albuginea and was made up of excessive amounts of mature collagen (Figure 5B). The collagen bundles in the affected testes were more dense, with a higher cytoplasmic to nuclear ratio than the sparse collagen fibers that form the normal interstitial connective tissue matrix. There was never any evidence of necrosis, inflammatory cell infiltrate, or granulation tissue. This indicates that the excess collagen deposition was not a response to prior injury, but instead was probably laid down during development, resulting in substitution of seminiferous tubules by collagen, hence our proposal to use the term fibrous hypoplasia to describe the lesion. In cases of unilateral moderate or severe lesions, the fibrotic testis was often of smaller diameter than its normal contralateral partner (Figure 6A). However, unilateral testicular hypoplasia (reduced diameter of the testis with decreased number of tubular profiles) was occasionally observed in testes that had only minimal levels of excess collagen (Figure 6B). In such cases, the reduction in testicular size and tubule numbers could not be explained by the deposition of excess collagen. This may suggest two different developmental disturbances, but since we did not directly measure testis diameter or tubule numbers in this retrospective survey, we have insufficient data to propose an explanation.
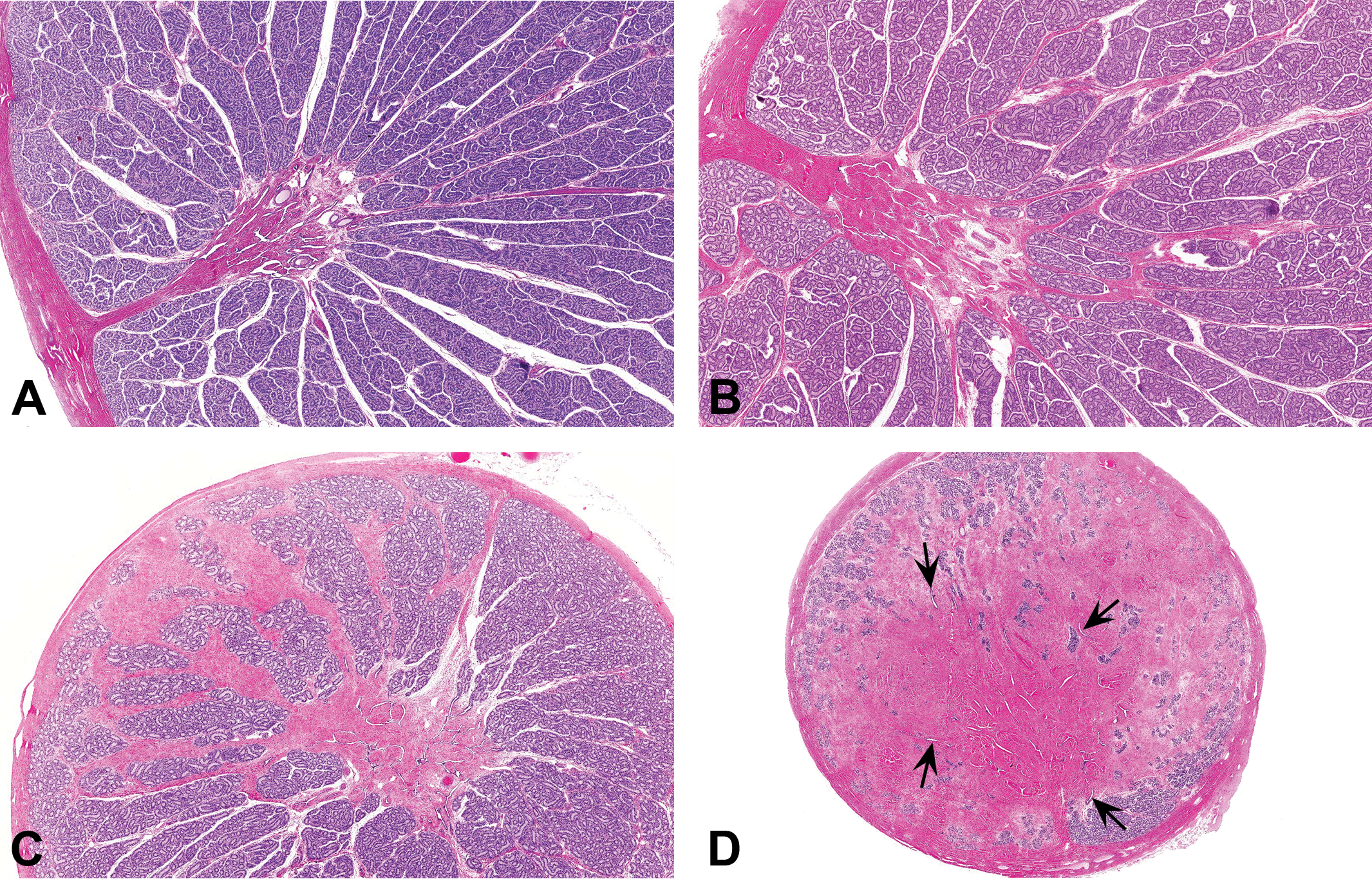
(A) Immature testis. Normal mediastinum testis containing rete ducts. Note the amount of collagen surrounding the ducts of the rete testis and the small amount of collagen in the interlobular septae. Hematoxylin and eosin stain (H&E). (B) Immature testis with minimal fibrous hypoplasia. Excessive collagen expanding the mediastinum testis surrounding the ducts of the rete testis. Collagen is also increased in some of the interlobular septae. H&E. (C) Immature testis with mild fibrous hypoplasia. Excessive collagen surrounds the rete testis and extends as tracts of collagen bundles expanding the interlobular septa. H&E. (D) Immature testis with marked fibrous hypoplasia. Approximately 75% of the testicular parenchyma has been substituted with collagen. The ducts of the rete testis are dispersed over a much wider area than normal (arrows) and are surrounded by thick tracts of mature collagen. H&E.
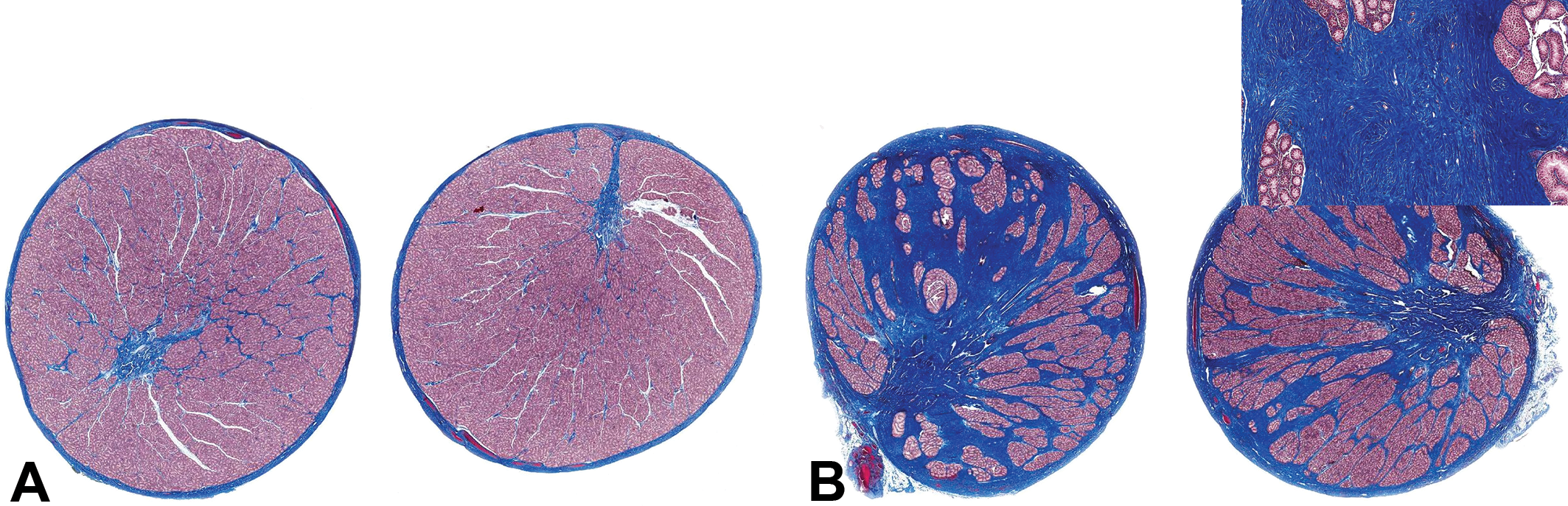
(A) Normal immature testes with normal amounts of collagen (stained blue) surrounding the rete and forming the capsule and interlobular septae. Martius Scarlet Blue stain (MSB). (B) Immature testis with moderate fibrous hypoplasia showing excess collagen (stained blue) around the rete and radiating into the interlobular septa and thickened capsule. Inset shows tracts of mature collagen replacing the testicular parenchyma. MSB stain.
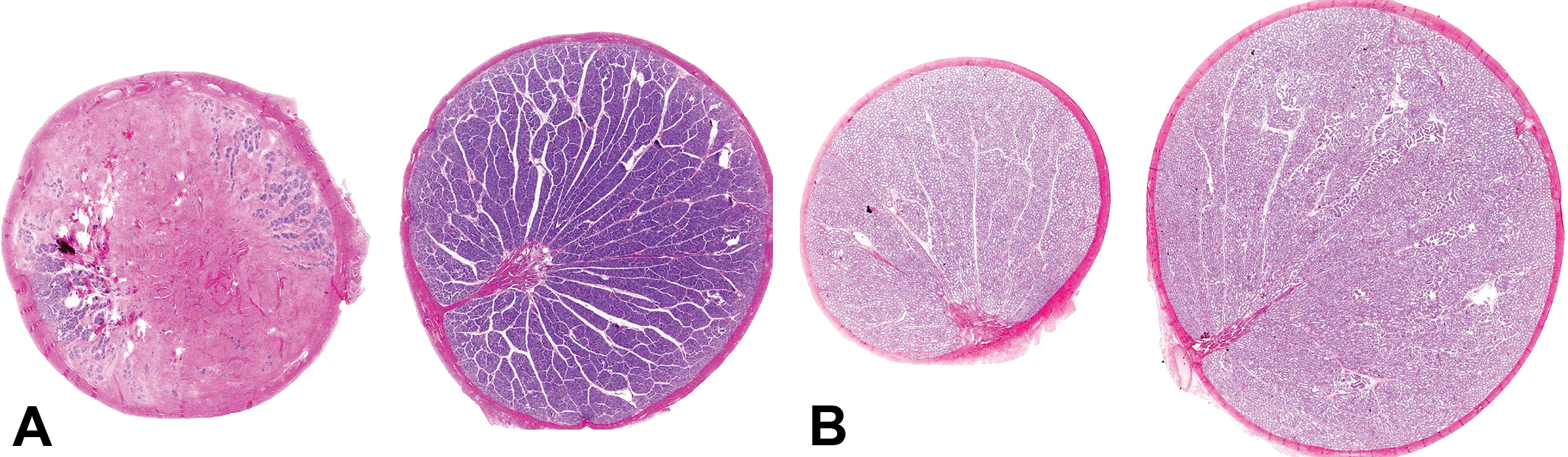
(A) Severe unilateral fibrous hypoplasia in an immature monkey. Note the thickening of the testicular capsule and slightly reduced size of the fibrotic testis. Testis on the right is the contralateral testis from the same animal. Hematoxylin and eosin stain (H&E). (B) Maturing testes with unilateral minimal fibrous hypoplasia (left) surrounding the rete testis. Note the marked reduction in size of the left testis. Testis on the right is the contralateral testis from the same animal. H&E.
In sexually mature monkeys, the ducts of the rete testis are normally expanded due to their role in transporting the sperm and seminiferous tubule fluid from the mature seminiferous tubules to the efferent ducts. Small amounts of mature collagen surround the rete ducts and separate the lobules of the normal testis (Figure 7A and 8A). In testes with fibrous hypoplasia, increased amounts of collagen expanded the mediastinum testis and the interlobular septa (Figure 7B–D). In a number of affected maturing or mature testes, the fibrotic rete was associated with cystic dilation and pressure atrophy of the seminiferous tubules, suggesting obstruction of the tubular outflow into the rete (Figures 7D and 8B). This sometimes displayed a lobular distribution, indicating the involvement of individual seminiferous tubules. In more severe cases of diffuse tubular dilatation, the tubules were cystic and the seminiferous epithelium was atrophic due to pressure atrophy.
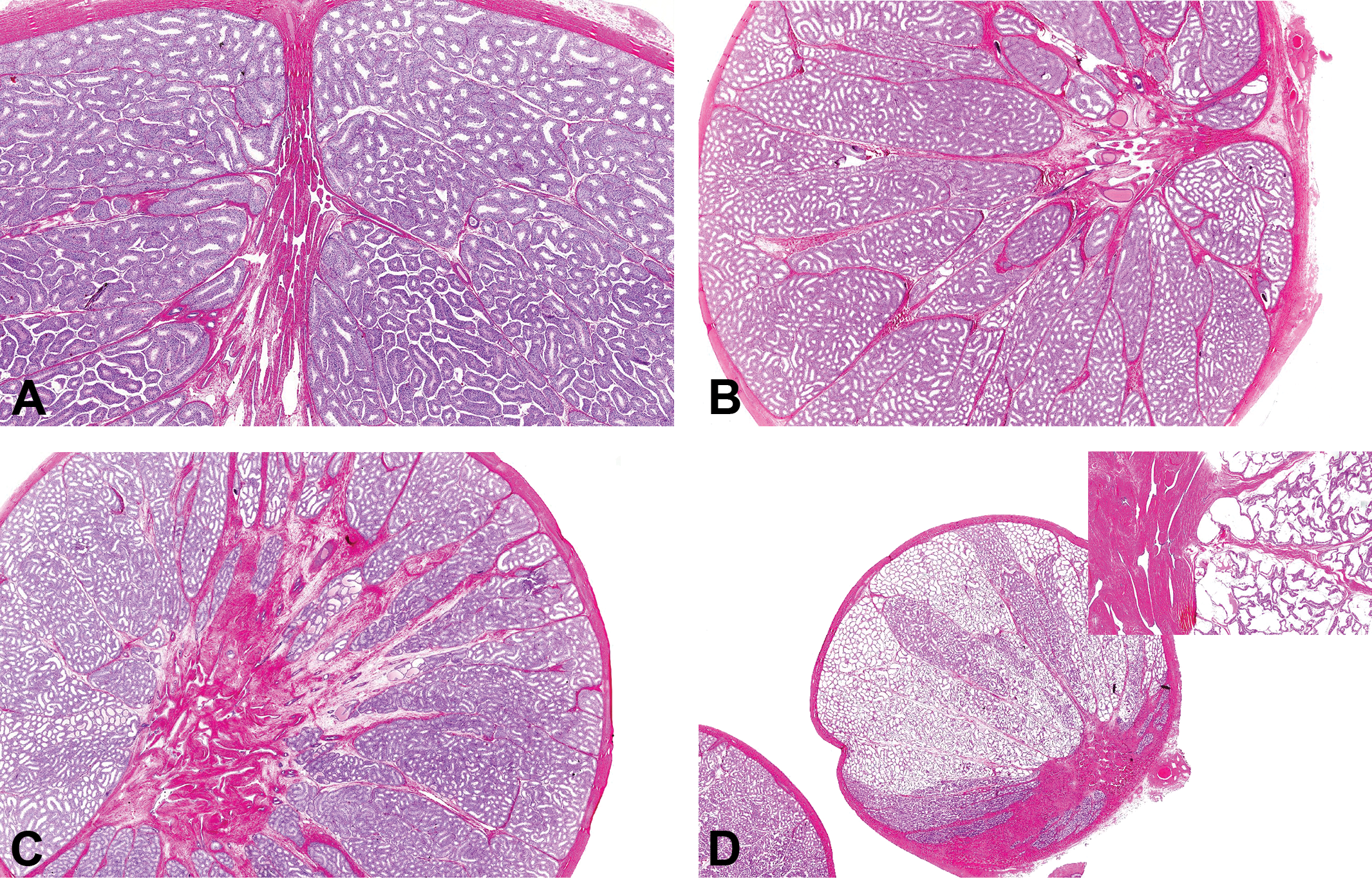
(A) Mature testis. Normal appearance of the mediastinum testis containing the dilated ducts of the rete testis. Hematoxylin and eosin stain (H&E). (B) Mature testis with minimal fibrous hypoplasia. Excessive collagen within the mediastinum testis and the interlobular septa. H&E. (C) Mature testis with mild increase in collagen expanding the area around the rete testis and interlobular septa. Note the occasional lobules of tubules that are dilated. H&E. (D) Maturing testis with mild collagen deposition around the rete and extending out to the capsule. Many lobules contain tubules with cystic dilation/atrophy (see inset). H&E.
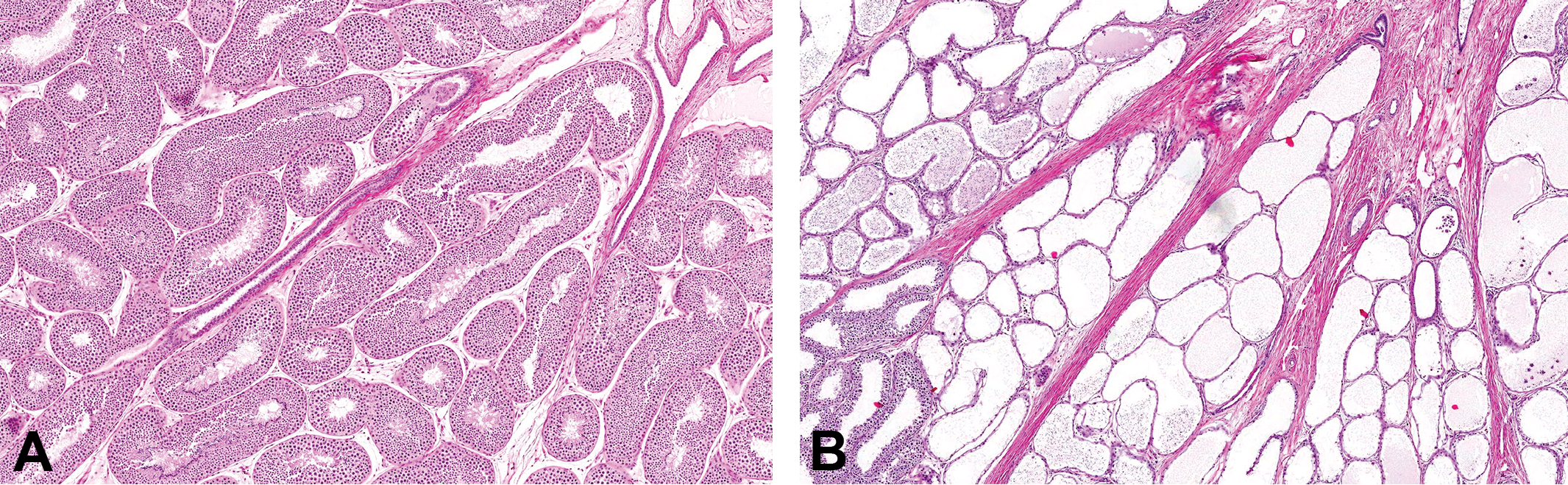
(A) Mature testis showing normal transition between seminiferous tubules and rete ducts. Hematoxylin and eosin stain (H&E). (B) Mature testis with excess collagen surrounding rete and associated with cystic dilation and pressure atrophy of the seminiferous tubules. Note the contraction and collapse of the rete ducts and fluid accumulation and sperm stasis in the dilated seminiferous tubules. H&E.
Discussion
The results of this retrospective survey of cynomolgus monkey testes have demonstrated an increasing incidence of an incidental, fibrotic, and space-occupying lesion of the mediastinum testis and interlobular septa that result in developing abnormalities of the rete testis and seminiferous tubules and is unique to cynomolgus monkeys. In the moderate to severe cases, large areas of the testicular parenchyma (seminiferous tubules and interstitial tissue) had been substituted by excessive amounts of mature collagen bundles. In the cynomolgus monkey, development of the structure of the seminiferous (medullary) cords is complete by gestation week 7 to 9 (D. M. Creasy and Chapin 2013). Postnatal development of the seminiferous tubules only involves expansion and population of the tubular loops with germ cells; it does not involve any increase in the number of tubules. Therefore, the fact that large areas of testicular parenchyma have been substituted with collagen bundles indicates that the collagen must have been laid down during gestation and that the reduced number of seminiferous tubules is a developmental abnormality that cannot recover during postnatal development. There was a lower incidence of the lesion in mature monkeys (2.2%) compared with immature (14%) or developing (14.7%). Although this could suggest that the lesion recovers, a more likely explanation is based on the fact that the lesion only began to occur in significant numbers (>11%) in immature monkeys between 2009 and 2010. Since this survey only examined monkeys up to 2011, there would not be adequate time for the developmental abnormality to have worked its way through to mature monkeys. However, an updated survey would be required to substantiate this hypothesis. Another possibility for the reduced incidence of the lesion in mature monkeys could be that the presence of the developmental abnormality resulted in delayed testicular maturation but the fact that unilateral fibrosis had no influence on maturation in the affected testis compared with the contralateral normal testis argues against this. A final possibility to explain the reduced incidence of the fibrotic lesion in mature testes could relate to sampling differences for the larger size testes. However, the size of the testis in the sexually mature males did not affect sampling of the lesion since the lesion originated in the mediastinum, and the sections were routinely taken just off the midline ensuring that even relatively small lesions around the rete would be included. The sides of the testes were trimmed to allow sections to fit on routine slides.
Based on subgross evaluation, some monkeys had unilateral hypoplasia of the testis characterized by one testis having a much smaller diameter and containing reduced numbers of seminiferous tubules compared with the contralateral testis. In many cases, this was associated with rete fibrosis; but in a few cases, the hypoplasia occurred in the absence of any significant fibrosis. This suggests that reduced seminiferous cord development and excessive collagen deposition around the rete may be two independent developmental abnormalities. It was not possible to investigate this hypothesis further since we only had access to paired organ weights and did not perform tubular counts, but it would be an interesting aspect to investigate in a follow-up study.
The localization of excessive collagen in the mediastinum testis was associated with expansion of the overall area taken up by the rete testis ducts. This was especially evident in immature testes. In maturing and mature testes, the presence of excessive collagen around the rete testis was often associated with cystic luminal dilation of seminiferous tubules with accompanying thinning or atrophy of the seminiferous epithelium. Interestingly, Kozlosky et al. (2015) also reported an unusually high incidence of seminiferous tubular dilation with associated tubular atrophy as well as substantially increased quantities of fibrous connective tissue emanating from the rete testis of Asian cynomolgus macaques, although the authors did not suggest a causal relationship between the two lesions. The most probable cause of the tubular dilation is partial or complete obstruction of fluid release from the seminiferous tubules as they empty into the fibrotic rete testis, followed by pressure atrophy of the seminiferous epithelium in the affected tubules. That this tubular dilation was only seen in maturing and mature testes is predictable because seminiferous tubule fluid only starts to be secreted once spermatogenesis progresses to the pachytene spermatocyte stage. Such dilation would not be expected in immature testes that are not secreting fluid. The tubular dilation/atrophy was generally lobular or multilobular in distribution, suggesting that single or multiple tubules were obstructed from emptying into the rete testis while others had unobstructed flow into the rete ducts. Although mild tubular dilation can sometimes be seen as a feature of pubertal testes (Haruyama et al. 2012), the severity and/or cystic nature of the tubular dilation in many of the affected monkeys was greater than that normally associated with puberty. Since tubular dilation and atrophy can be important test article–related changes, it is important for the toxicological pathologist to be aware of the possible association of this degenerative tubular lesion as a secondary consequence to the incidental, congenital/developmental fibrotic lesion around the rete.
Based on the country of origin of the monkeys, the fibrotic lesion was most frequently seen in Asian monkeys sourced from China (19.7%) and Vietnam (14.4%). The lesion was present at a much lower incidence in monkeys sourced from Cambodia (4.4%) and Indonesia (0.89%). Mauritian monkeys also had a low incidence (6.6%). Within specific geographical areas, there was no obvious difference in the incidence of the lesion based on individual suppliers. Cynomolgus monkeys used in research may come from several geographical regions that include China, Vietnam, Cambodia, Thailand, Malaysia, Indonesia, Java, Philippines, Japan, Burma, Borneo, and Mauritius (Waller et al. 2013
Footnotes
Acknowledgments
The authors would like to thank Dr. Katharine Whitney for her review of this article and her constructive comments during its preparation and Steve Van Adestine for his help with the images.
Author Contribution
All authors contributed equally to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
