Abstract
Selective chemonucleolytic effects of condoliase, a glycosaminoglycan degrading enzyme, was investigated histopathologically in cynomolgus monkeys. Condoliase was administered once into the lumber intervertebral disc (IVD), and as a comparative control, chymopapain, a proteolytic enzyme, was administered in a similar manner. Histopathological changes of the IVD and the adjacent vertebral body (VB) were examined at 1 to 26 weeks after administration. Major changes induced by condoliase in the IVD were degenerative and necrotic changes in the nucleus pulposus, annulus fibrosus, cartilaginous endplate (CEP), and epiphyseal growth plate (EGP); focal disappearance of the EGP; and neovascularization and ossification of the CEP. Decreased/necrosis of bone marrow cells with new bone formation was observed in the VB. Cellular regeneration in the IVD was observed as a recovery changes on and after week 4. The changes in the IVD and VB subsided at week 26. Chymopapain induced qualitatively similar but more widely extended changes. The degrees of the changes in the IVD and VB were more severe than those of condoliase, and the changes were exacerbated even at week 26. These results indicated that histopathological changes caused by condoliase were less severe and more selective than those by chymopapain.
Introduction
Chemonucleolysis induces an enzymatic degradation of the nucleus pulposus (NP) in the intervertebral disc (IVD) to reduce intradiscal pressure on the nerve root and improve lumbar disc herniation (LDH) symptoms. 1 This treatment is a less invasive technique and provides an intermediate treatment of LDH between conservative and surgical treatments. 2
Chymopapain, a proteolytic enzyme, had been used as a chemonucleolytic agent in Europe, North America, and Australia, and the efficacy of the agent has been reported in the literature. 3 Although complications such as anaphylaxis, infections, hemorrhage, neurologic, and miscellaneous reactions have been reported, the complication rate of chemonucleolysis with chymopapain is less than that reported for lumbar discectomy. 3 –5 Furthermore, no information was found in any of the literature and postmarketing reports that indicated withdrawal reasons for chymopapain were due to lack of efficacy or safety concerns. 6 However, chymopapain was withdrawn from the market in 2002 for nonscientific commercial reasons. 3,6
As a new chemonucleolysis agent, condoliase (a generic name of chondroitinase ABC) has been developed for the United States and Japan. Condoliase is a mucopolysaccharidase purified from the gram-negative rod proteus vulgaris. 7 Condoliase lacks protease activity and causes a selective degradation of chondroitin sulfate and hyaluronic acid, both of which are main components of glycosaminoglycan (GAG) chains on proteoglycan (PG) in the NP. 8 Because of its high substrate selectivity, condoliase is considered to be suitable for selective chemonucleolysis. A phase III clinical trial for patients with LDH was completed in Japan, demonstrating that condoliase was effective in improving leg pain comparable to lumbar discectomy and well tolerated without any clinical relevant adverse events. 9 –12 Condoliase has been approved by the Japanese health authority in March 2018 for the treatment of subligamentous extruded LDH resistant to conservative treatment. 13
In nonclinical studies, the selective effects of chondroitinase ABC have been reported in several animal species. 14 –23 Chondroitinase ABC did not affect tissues surrounding the IVD such as spinal nerve tissues, ligamentum flavum, and blood vessels. 14 –17 In the IVD, histopathological changes in the NP and annulus fibrosus (AF) caused by the enzyme were more localized and less severe than those caused by both chymopapain and lumbar discectomy. 18 –23 On the other hand, there are few reports evaluating the selective effects of condoliase in the cartilaginous endplate (CEP), epiphyseal growth plate (EGP), and vertebral body (VB), 23 and detailed histopathological changes in the area have not been clarified. Furthermore, there are many species differences among animals such as the IVD structure and mechanical stress on the disc, 24,25 leading to difficulty in attempting to predict the effects of condoliase in humans from the results of nonclinical studies.
Nonhuman primate such as cynomolgus monkey has IVDs similar to those of humans in terms of the absence of notochordal cells in adult life, having a thick CEP and epiphyseal ring, exposure to mechanical stress, and extent and frequency of spontaneous disc degeneration. 24 –28 These similarities would be relevant to predict the histopathological changes in humans. On the development of condoliase, we conducted some single intradiscal dose studies as routine toxicity studies in cynomolgus monkeys and obtained histopathological data in the IVD and adjacent VB. At the same time, we conducted a similar study using chymopapain as the only similar chemonucleolytic agent to examine whether there are differences of histopathology between condoliase and chymopapain. In this article, we demonstrate selective chemonucleolytic effects of condoliase, comparing the detailed process of the histopathological changes caused by both enzymes.
Materials and Methods
In this study, to evaluate the selective effects of condoliase, the data obtained from independent experiments (experimental chemonucleolysis with condoliase and chymopapain) were compared. Healthy cynomolgus monkeys of Chinese origin with weighing 3 to 5 kg and 3 to 5 years old, which are commonly used in toxicity studies, were selected and used 63 cynomolgus monkeys (57 males and 6 females). Cynomolgus monkeys in this range of age have a full structure of the CEP, EGP, and VB without closure of the EGP and are considered to be suitable to evaluate possible toxic effects of chemonucleolytic agents on these parts.
No abnormalities in the lumbar vertebrae including the IVDs were noted radiographically in any animal before the experiments. The animals were randomly allotted into the groups in each study (Table 1), based on the body weight at the day of grouping, so that the mean body weight of each group was approximately equal. Animals were individually housed in stainless steel cages (70 × 78 × 65 cm in depth, height, and width) in environmentally controlled room with a temperature range of 20 °C to 28 °C, a relative humidity of 30% to 80%, a ventilation rate of 10 to 16/hour, and a light: dark cycle of 12:12 hours. PS-A pelleted feed, standard breeding feed for monkeys (Oriental Yeast Co, Ltd) was provided daily, and tap water was available ad libitum. The experimental protocols were approved by the Institutional Animal Care and Use Committees of Seikagaku Corporation and Cmic Bioresearch Center Co, Ltd.
Group Composition.
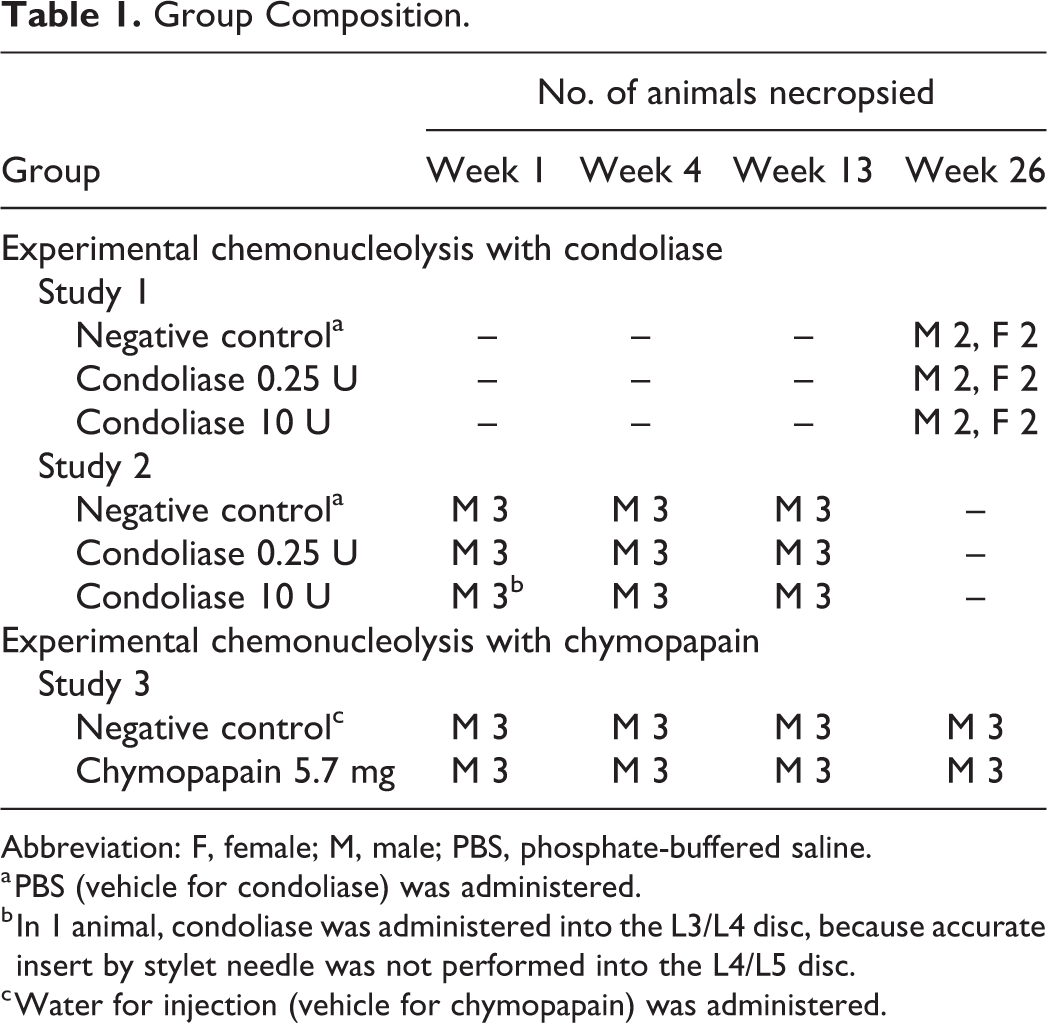
Abbreviation: F, female; M, male; PBS, phosphate-buffered saline.
a PBS (vehicle for condoliase) was administered.
b In 1 animal, condoliase was administered into the L3/L4 disc, because accurate insert by stylet needle was not performed into the L4/L5 disc.
c Water for injection (vehicle for chymopapain) was administered.
Experimental Chemonucleolysis With Condoliase
We firstly conducted a single intradiscal dose general toxicity study of condoliase in male and female cynomolgus monkeys with a 26-week chronic observation period (study 1) to confirm systemic toxicity as well as local tolerance. The doses of condoliase were set at 0.25 and 10 U/disc that were equivalent to doses of 12- and 494-fold the clinical dose, converted from the ratio of the magnetic resonance image (MRI)–measured NP volume (Supplemental Table 1). Although no systemic toxicities related to condoliase were noted in any animals in the study (data not shown), chronic changes caused by condoliase occurred locally in the IVD. In the following study, morphological changes over time caused by condoliase were examined in cynomolgus monkeys with 1 to 13-week subchronic observation periods (study 2). Since there were no sex difference of IVD histopathology in the previous chronic study, only males were used in this subchronic study. Group composition in both studies is shown in Table 1.
Condoliase was once administered into the IVD in each animal as follows: Animals were anesthetized with an intramuscular injection of a mixture of atropine sulfate at 0.01 mg/kg (Atropine Sulfate Injection, Mitsubishi Tanabe Pharma Corp), xylazine at 0.9 mg/kg (Selactal 2% Injection, Bayer Yakuhin Ltd), and ketamine hydrochloride at 4.5 mg/kg (Katalal, Daiichi Sankyo Co, Ltd). An anesthetized animal was immobilized on a side position on a radiography table (X-ray system for small animals VPX-20; Toshiba Medical Supply Co, Ltd). Under fluoroscopic monitoring, the tip of a 23G spinal needle was inserted into L4/L5 disc. The stylet was removed, and a 31G needle attached to a micro syringe was inserted into the spinal needle. The tip of the 31G needle introduced into the center of NP was confirmed under fluoroscopic image, and then condoliase (Seikagaku Corp) prepared at a concentration of 25 or 1000 U/mL was once administered in a volume of 10 μL/disc (0.25 or 10 U/disc). As a negative control, 10 mmol/L of phosphate buffered saline (Thermo Fisher Scientific K.K; vehicle for condoliase) was administered in a volume of 10 μL/disc.
From the lateral and dorsolateral directions, the lumbar vertebrae including the treated IVD were radiographically monitored at 1, 5, 9, 11, 13 17, 21, and 26 weeks (study 1) or 1, 2, 4, 8, and 13 weeks (study 2) after administration. The radiograms were captured as electronic image data by a transmission scanner, and the IVD height was measured, using an image analysis software (Image-Pro Express, version 5.1, Nippon Roper K.K.). The rate of changes in disc height index (DHI; %) was calculated by the formula as shown in Figure 1. In both studies, clinical observations and body weight measurements were performed, and animals were necropsied at 1, 4, 13 (study 2) and 26 (study 1) weeks after administration. They were euthanized by bleeding from the axillary artery under anesthesia with an intravenous administration of pentobarbital sodium at 25 mg/kg (Somnopentyl, Kyoritsu Seiyaku Corp). The spinal column was examined macroscopically, and the lumbar vertebrae including the treated IVD were removed for evaluation of the administration site.
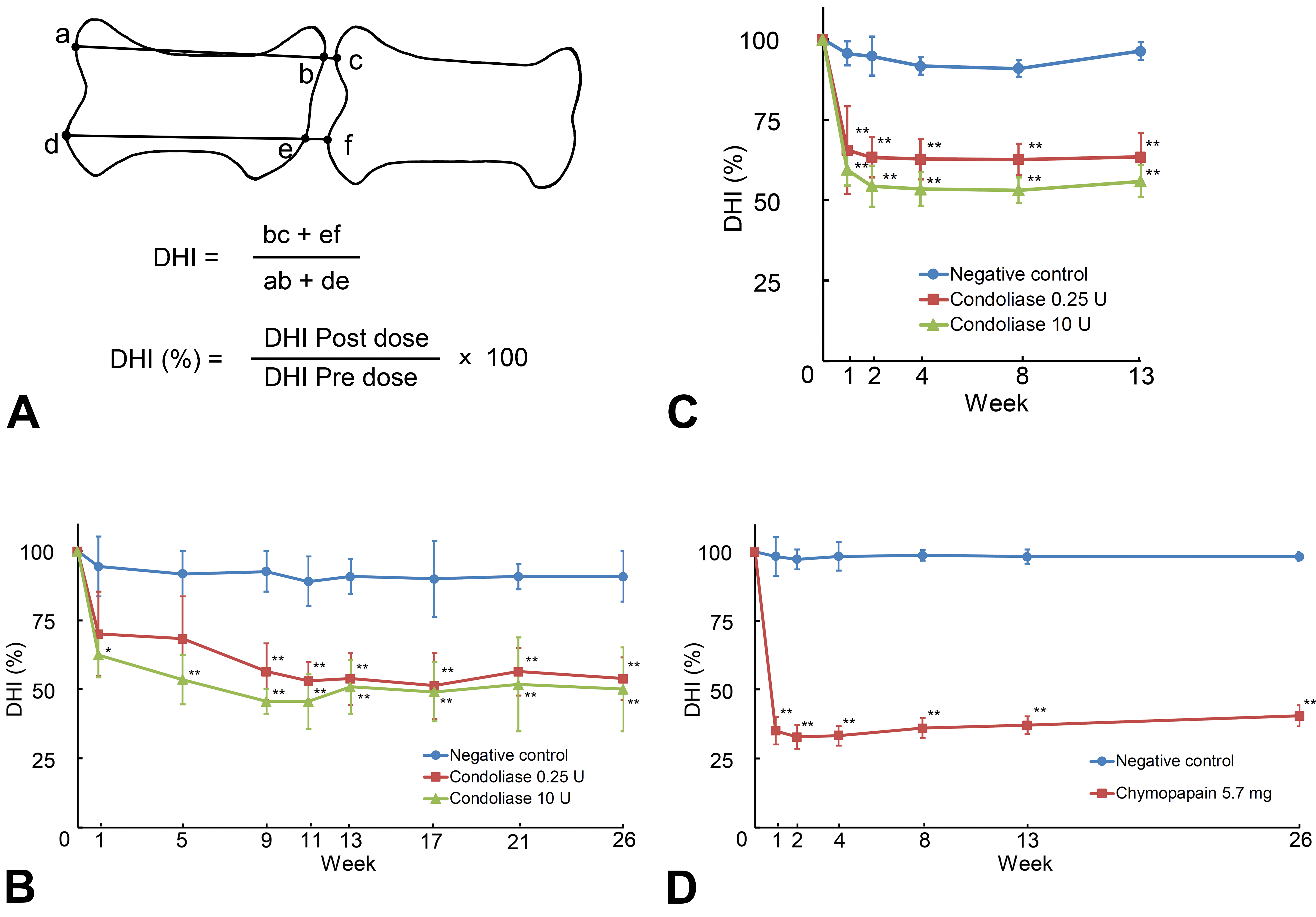
Changes of disc height index (DHI). The DHI (%) was calculated from the length of straight line (ab, de, bc, and ef) and the formula shown in A. Data are the mean ± standard deviations of 4 animals in the experimental chemonucleolysis with condoliase in study 1 (B), 3 animals in the study 2 (C), and study 3 (D). * P < .05, ** P < .01, significantly different from negative control by Dunnett test.
The lumbar vertebrae were fixed in neutral buffered 10% formalin and then decalcified with 10% formic acid formalin. The treated IVD and adjacent vertebrae were cut off on a midline and trimmed as the longitudinal section and embedded in paraffin, thin-sectioned. The specimens were stained with hematoxylin–eosin, and the treated IVD and adjacent VB were examined microscopically. Histopathological findings in the treated IVD and adjacent VB were confirmed and graded to slight, moderate, and severe by 2 expert pathologists through the peer-review process based on Organisation for Economic Co-operation and Development guidance. 29
Experimental Chemonucleolysis With Chymopapain
Morphological changes caused by chymopapain were examined in male cynomolgus monkeys (study 3) to compare with those caused by condoliase at a dose of 0.25 U/disc (studies 1 and 2). The dose of chymopapain was set at 5.7 mg/disc that were equivalent to doses of 12-fold the clinical dose (Supplemental Table 1). The animals were grouped as follows: chymopapain 5.7 mg group and negative control (water for injection, vehicle for chymopapain) group (Table 1). In the same manner of studies 1 and 2, chymopapain (Sigma-Aldrich Corp) prepared at a concentration of 570 mg/mL was administered in a volume of 10 μL/disc (5.7 mg/disc). As a negative control, water for injection (Fuso Pharmaceutical Industries, Ltd) was administered in a volume of 10 μL/disc. Animals were subjected to clinical observations and body weight measurements, and the lumbar vertebrae were radiographically monitored at 1, 2, 4, 8, 13, and 26 weeks after administration. Animals were necropsied at 1, 4, 13, and 26 weeks after administration, followed by gross pathology on the spinal column and histopathology on the treated IVD and adjacent VB.
Results
Radiographs
Representative radiographs are shown in Figure 2. Narrowing of the IVD height was noted from week 1 in both enzyme groups. In 1 animal in the chymopapain 5.7 mg group, radiolucency of VBs increased in the area adjacent to the administration site from week 2, and decreased in the outside area surrounding the increased area from week 8. The DHIs decreased at week 1 from baseline level to 65.6% in the condoliase 0.25 U group, to 59.5% in the condoliase 10 U group, and to 35.1% in the chymopapain 5.7 mg group, and then they remained at similar levels until week 26 (Figure 1).
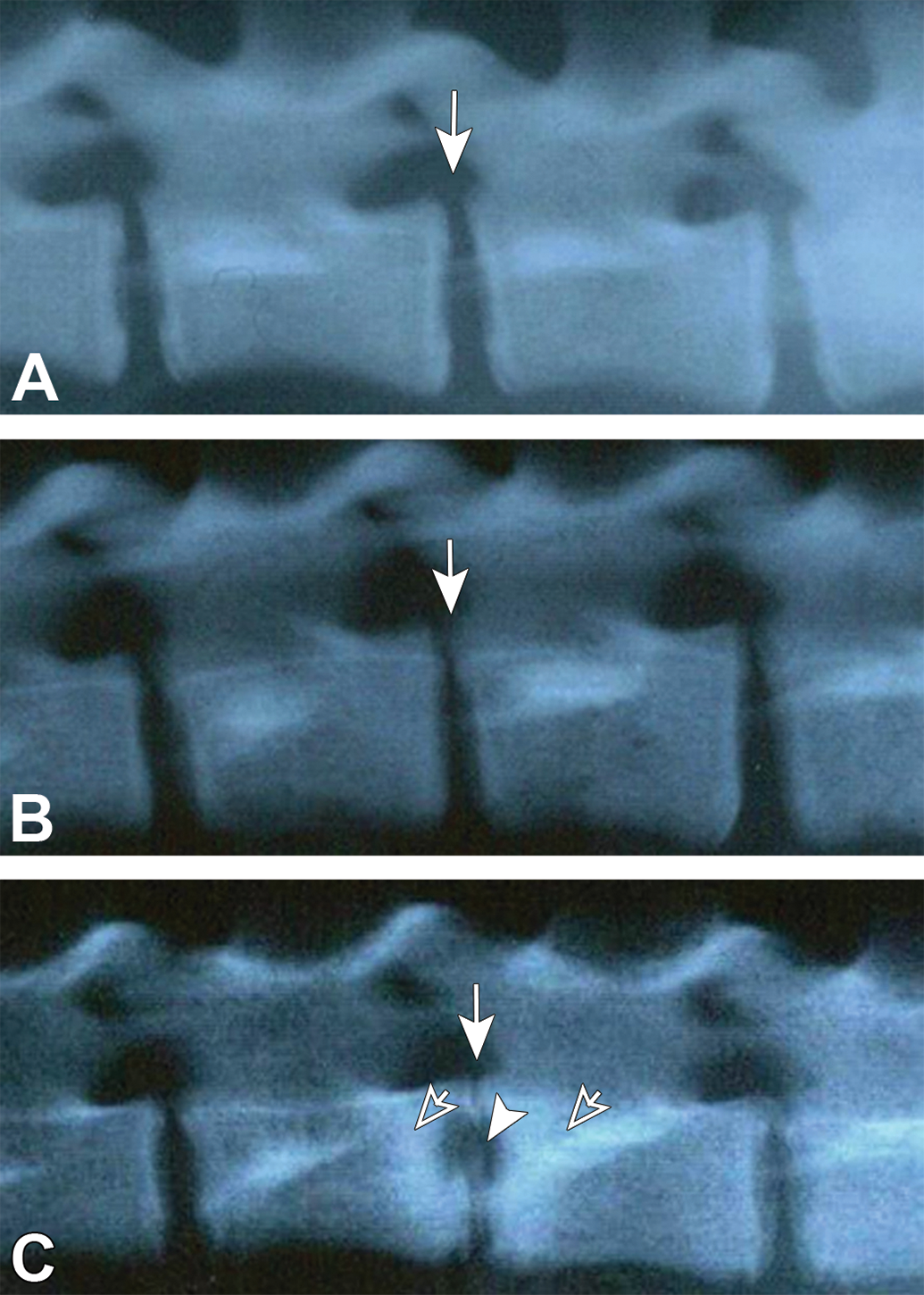
Representative radiographic photographs. No noteworthy findings were noted in the treated disc or vertebrae of the negative control groups at week 13 (A). Narrowing of the disc height was noted in the treated disc of the condoliase 0.25 U group at week 13 (B). Remarkable narrowing of the disc height and increased radiolucency in the adjacent vertebrae were noted in the treated disc of the chymopapain 5.7 mg group at week 13 (C). Arrows indicate the treated disc; arrowhead, increase in radiolucency; and open arrows, decrease in radiolucency.
General Conditions and Gross Pathology
No complications or discomfort were noted in relation to clinical signs and body weight changes in any animal in any group during the observation period. Representative macroscopic photographs are shown in Figure 3. Narrowing of the IVD was noted accompanied with reduced transparency and luster, and a grayish white coloration of the NP in both enzyme groups at all necropsy time points. In 2 animals in the chymopapain 5.7 mg group, the loss of the NP accompanied with cavity formation was noted at week 1, and effects of chymopapain spread to the VB from week 4. From week 13 in the chymopapain 5.7 mg group, 2 animals showed loss of the NP leading to the IVD collapse where either an irregularity of the CEP or a close location of the CEPs in the both sides of the disc was noted.
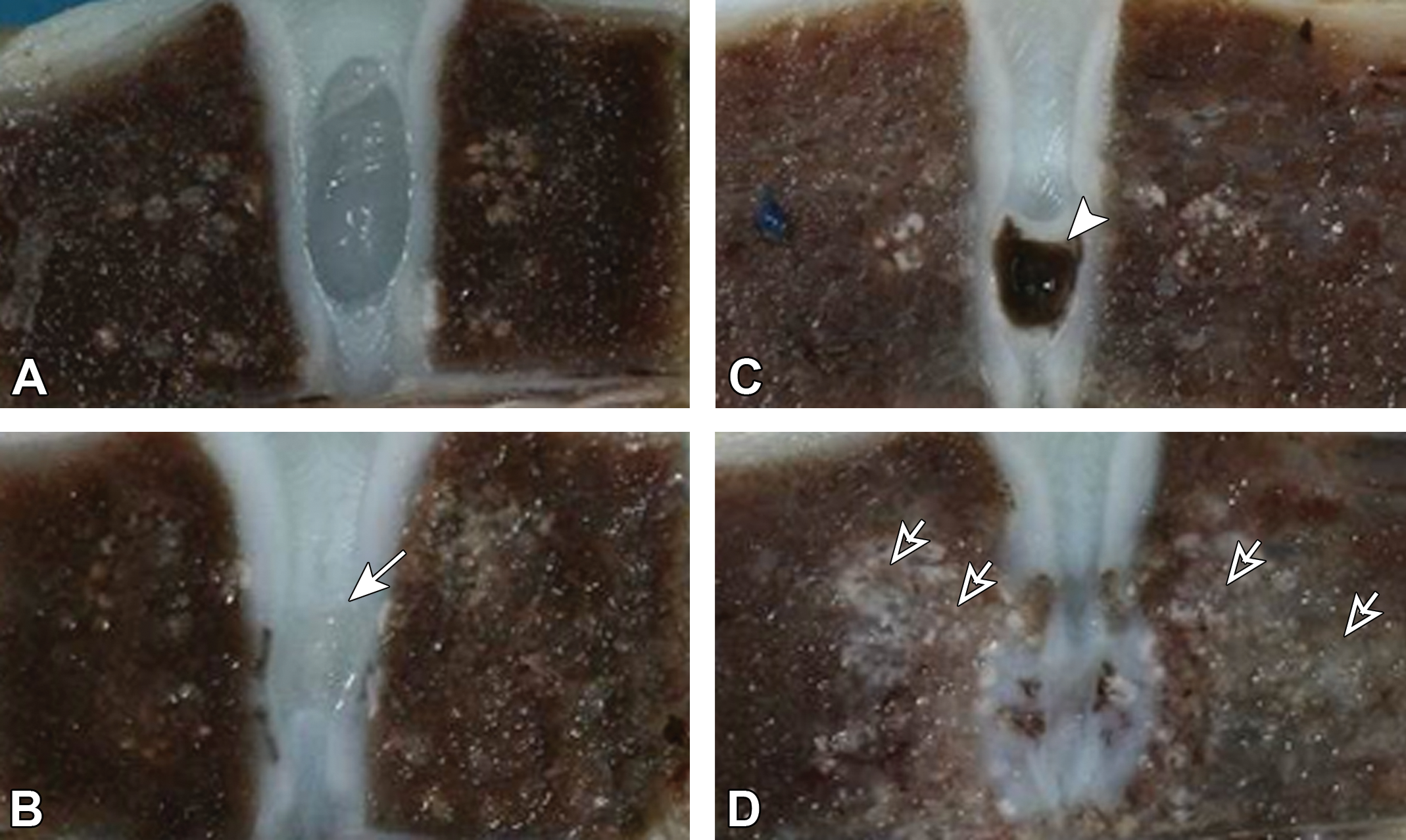
Representative macroscopic photographs. No noteworthy findings were noted in the treated disc or vertebrae of the negative control groups at week 13 (A). Reduced transparency and luster, and grayish white coloration of the nucleus pulposus were noted in the condoliase 0.25 U group at week 13 (B, arrow). Cavity formation in the nucleus pulposus was noted in the chymopapain 5.7 mg group at week 1 (C, arrowhead), and the effects of chymopapain were also seen in the vertebrae as grayish white coloration at week 13 (D, open arrows).
Histopathology
Figure 4 shows overview of histopathological changes. No noteworthy findings were noted in any animal in the negative control groups at any observation time point (total 25 animals), except for 2 animals with slight changes in the NP (decreased in size, degeneration/necrosis of NP cells) and AF (bulging toward the NP) caused by administration procedure. The histopathological findings in both enzyme groups were summarized in Table 2 for the NP and AF, and in Table 3 for the CEP to VB. In animals treated with condoliase, at week 1, the size of the NP decreased with changes including degeneration/necrosis of NP cells, and the concentration of intercellular matrix. In the AF adjacent to the NP, focal degeneration/necrosis of the AF and bulging of the AF toward the NP occurred. No cleft, tear and fissure formations were noted in the NP and AF in any animals treated with condoliase. Changes seen at the CEP to the VB included degeneration/necrosis of chondrocytes, decreased staining in both the CEP and EGP (Figure 5), and decreased/necrosis of bone marrow cells with new bone formation in a limited part (just beneath the EGP) of the VB. At week 4, in addition to the above findings, necrosis of osteocytes and bone marrow cells in the epiphyseal ring observable in the CEP of some animals, and focal disappearance of the EGP (bone island within cartilage) were noted. At the same time, neovascularization and ossification of the CEP and cellular regeneration in the NP, AF, and EGP occurred as recovery changes. The changes in the VB deteriorated. At week 13, regeneration of chondrocytes also occurred in the CEP, and decreased staining and degeneration/necrosis of chondrocytes recovered in the EGP. Neovascularization and ossification of the CEP became evident, and thinning of the CEP began to be noted. New bone formation in the bone marrow was not accompanied by active osteoblasts, suggesting that new bone formation was not active at this time point. Changes in the IVD and the adjacent VB subsided at week 26, and no necrotic changes were observed in the area from the CEP to VB although some changes remained such as decreased staining, thinning of the CEP, and focal disappearance of the EGP. The histopathological changes at each observation point were qualitatively comparable at all doses. The extent of the changes was more severe at 10 U/disc than 0.25 U/disc until week 13, but it was similar regardless of the dose or gender at week 26.
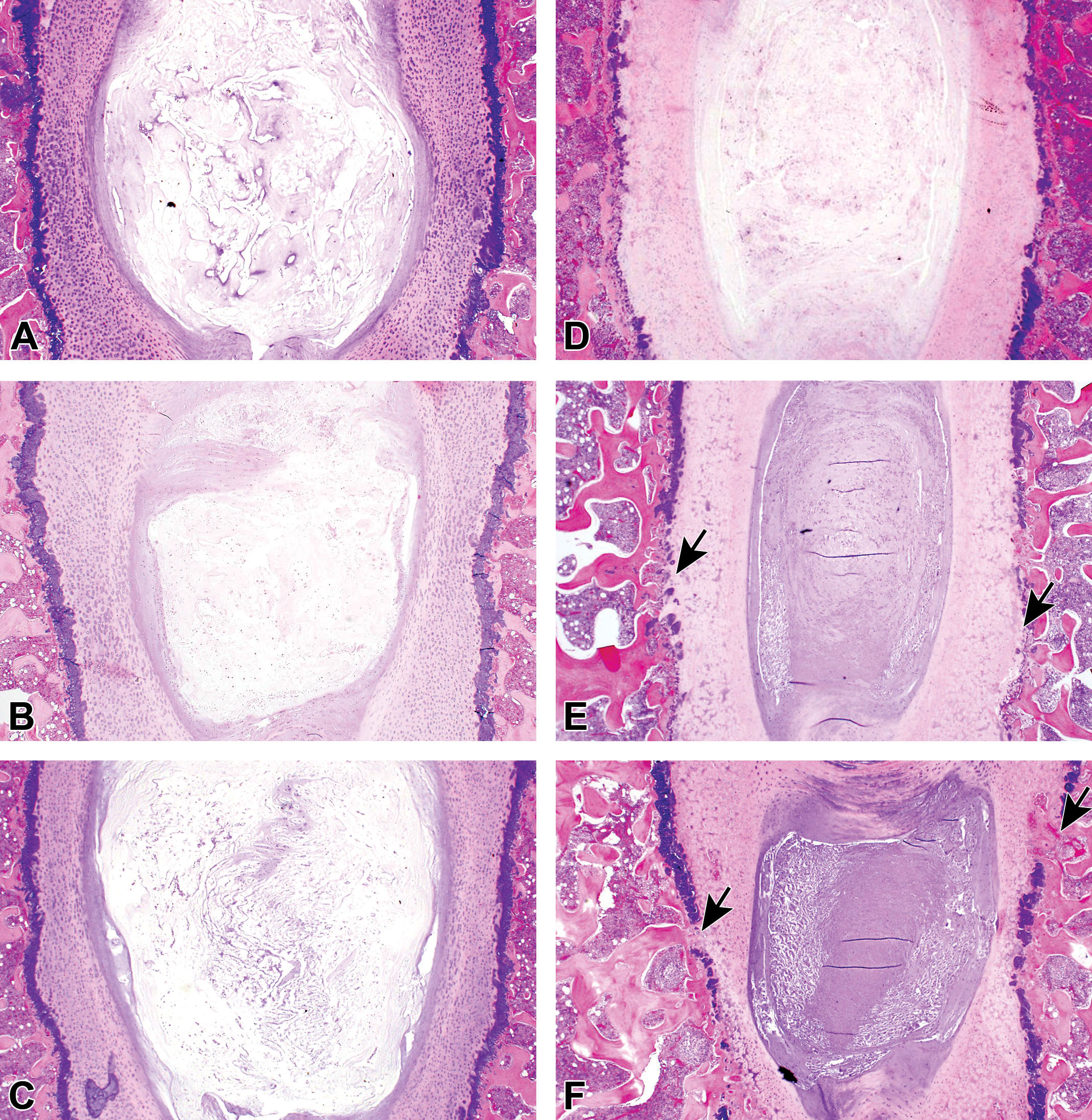
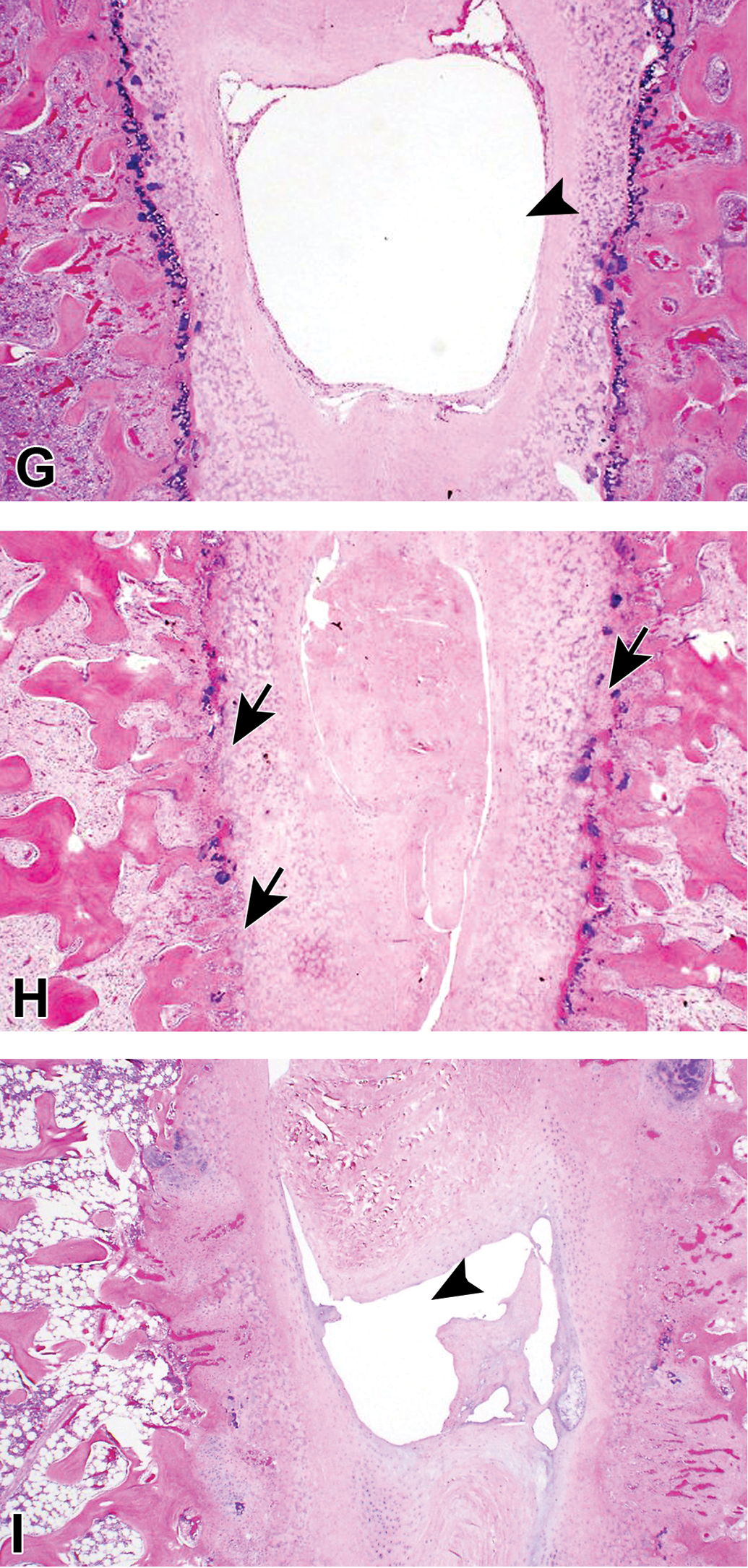
Overview of histopathological changes with low magnifications. No noteworthy findings were noted in the treated disc or vertebral body (VB) of the negative control groups at weeks 1, 4, and 13 (A, B, and C, respectively). In the condoliase 0.25 U group, histopathological changes were noted including degenerative and necrotic changes in the nucleus pulposus (NP), annulus fibrosus (AF), cartilaginous endplate (CEP), and epiphyseal growth plate (EGP), and focal disappearance of the EGP in the treated disc, and decreased bone marrow cells in the VB at weeks 1, 4, and 13 (D, E, and F, respectively). These changes were remarkable in the treated disc and VB of the chymopapain 5.7 mg group at weeks 1, 4, and 13 (G, H, and I, respectively). Arrows indicate focal disappearance of EGP; arrowheads, cavity formation.
Histopathological Findings in the Nucleus Pulposus and Annulus Fibrosus.
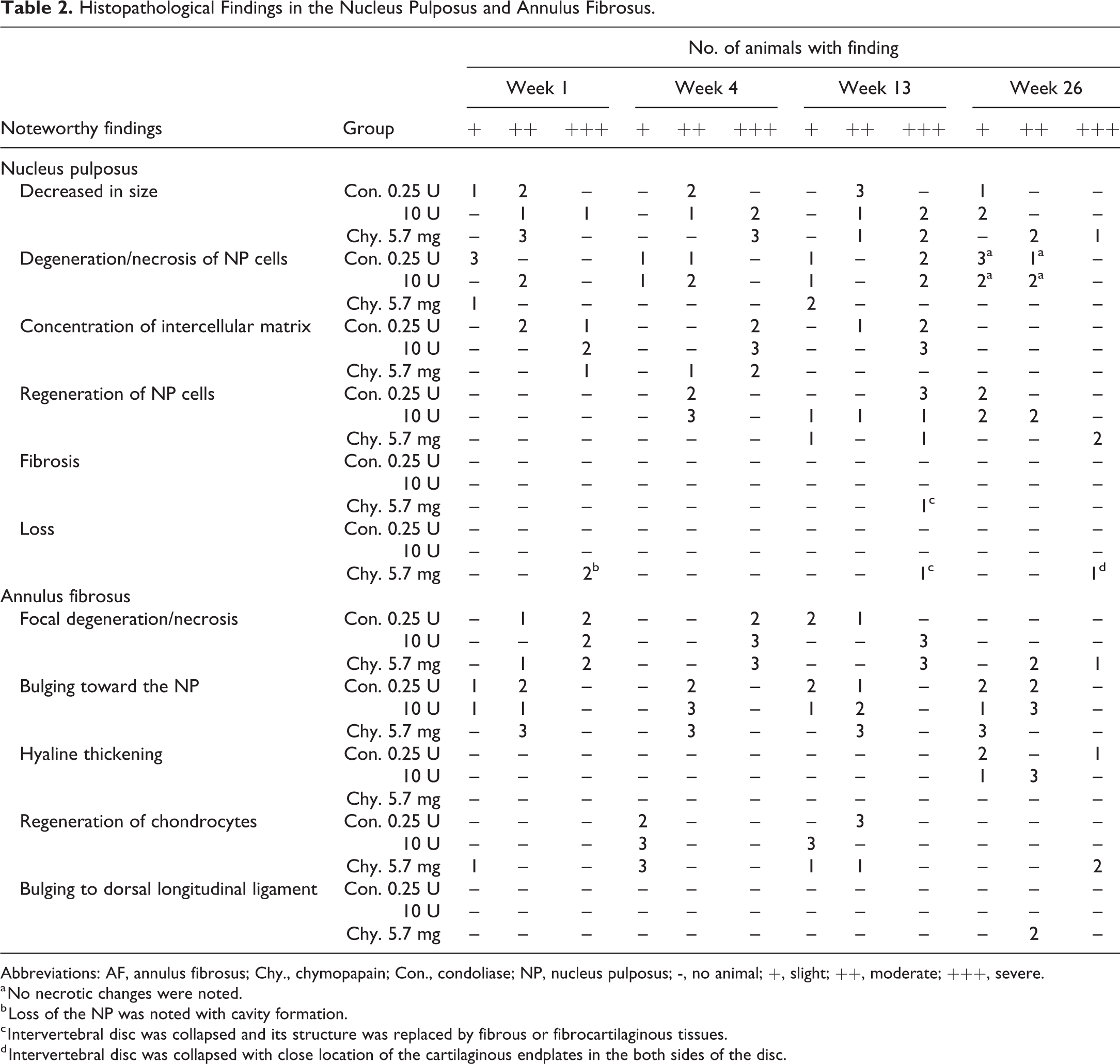
Abbreviations: AF, annulus fibrosus; Chy., chymopapain; Con., condoliase; NP, nucleus pulposus; -, no animal; +, slight; ++, moderate; +++, severe.
a No necrotic changes were noted.
b Loss of the NP was noted with cavity formation.
c Intervertebral disc was collapsed and its structure was replaced by fibrous or fibrocartilaginous tissues.
d Intervertebral disc was collapsed with close location of the cartilaginous endplates in the both sides of the disc.
Histopathological Findings in the Cartilaginous Endplate, Epiphyseal Growth Plate, and Vertebral Body.
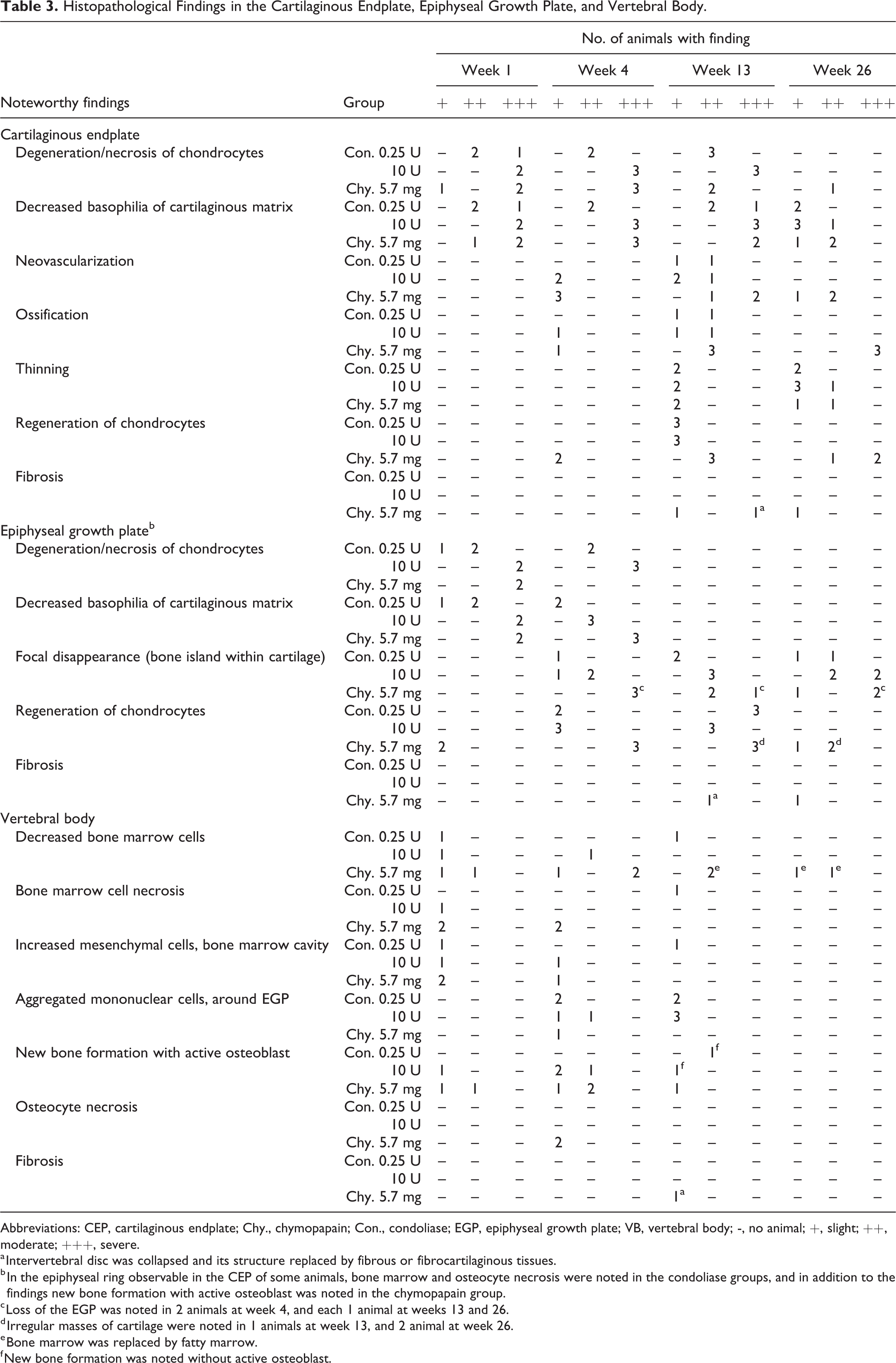
Abbreviations: CEP, cartilaginous endplate; Chy., chymopapain; Con., condoliase; EGP, epiphyseal growth plate; VB, vertebral body; -, no animal; +, slight; ++, moderate; +++, severe.
a Intervertebral disc was collapsed and its structure replaced by fibrous or fibrocartilaginous tissues.
b In the epiphyseal ring observable in the CEP of some animals, bone marrow and osteocyte necrosis were noted in the condoliase groups, and in addition to the findings new bone formation with active osteoblast was noted in the chymopapain group.
c Loss of the EGP was noted in 2 animals at week 4, and each 1 animal at weeks 13 and 26.
d Irregular masses of cartilage were noted in 1 animals at week 13, and 2 animal at week 26.
e Bone marrow was replaced by fatty marrow.
f New bone formation was noted without active osteoblast.
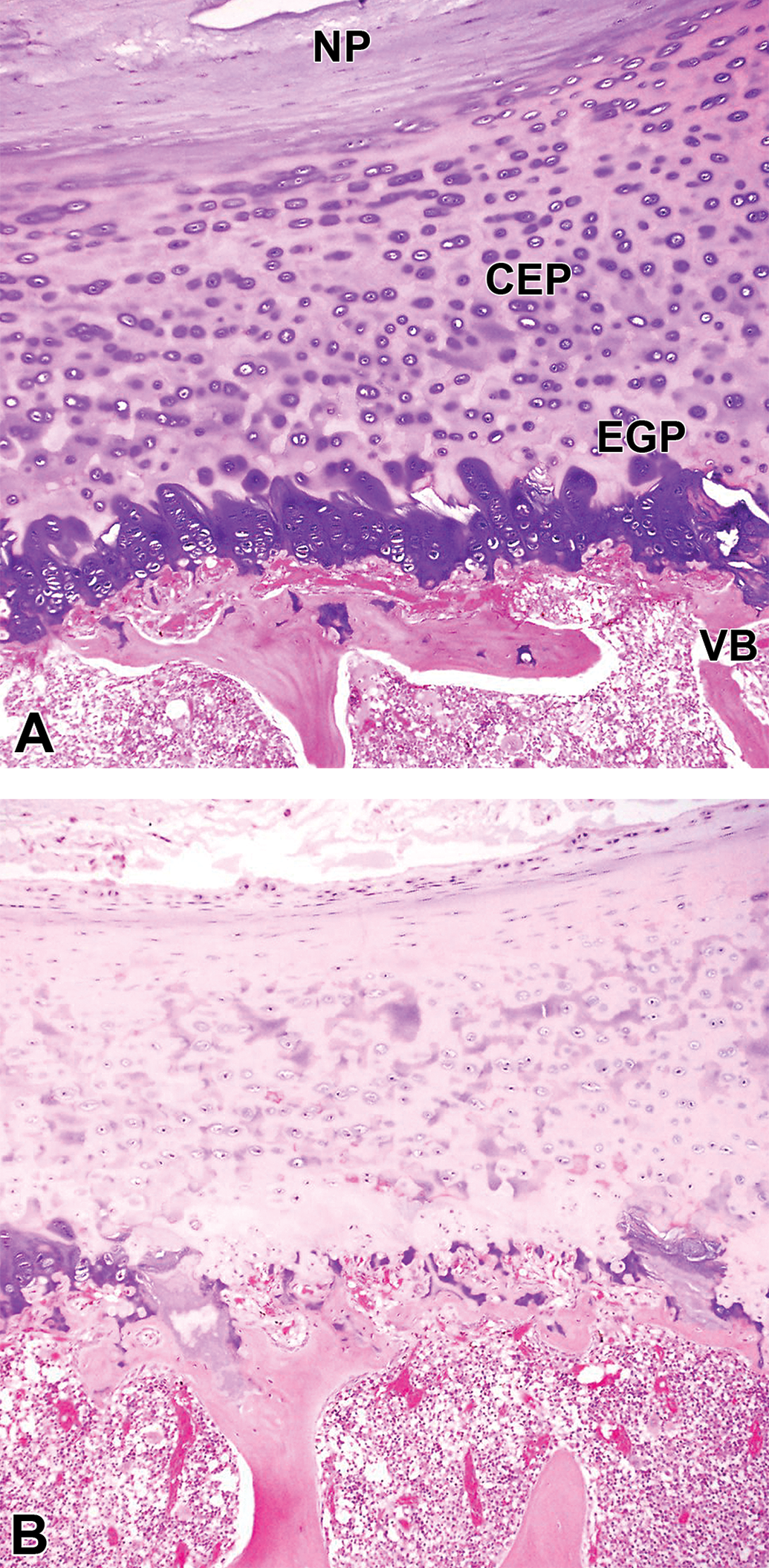
Decreased staining in the cartilaginous endplate (CEP) and epiphyseal growth plate (EGP) after administration of condoliase. The representative samples are shown in A and B sampled at week 1. The CEP and EGP were clearly stained with basophilia in the negative control-treated disc (A). In the condoliase 0.25 U group, decreased staining (basophilia) of the cartilage matrix was noted in the CEP and EGP of the condoliase-treated disc (B). NP, nucleus pulposus; VB, vertebral body.
Although qualitatively similar changes were noted in the IVD and the adjacent VB from week 1 after administration of chymopapain, the extent of the changes was obviously more severe than condoliase with maximum dose (Figure 6). From week 4, recovery changes as seen in the condoliase groups were noted, but focal disappearance and extensive loss of the EGP were distinct and effects on the VB were exacerbated. Tissue damage caused by chymopapain deteriorated in the IVD and effects expanded widely on the VB at week 13. At week 26, regeneration of chondrocytes progressed, but it formed irregular masses in the EGP and the bone marrow was replaced by fatty marrow in the VB. In serious cases, the IVD was collapsed and its structure was replaced by fibrous or fibrocartilaginous tissues (Figure 7).
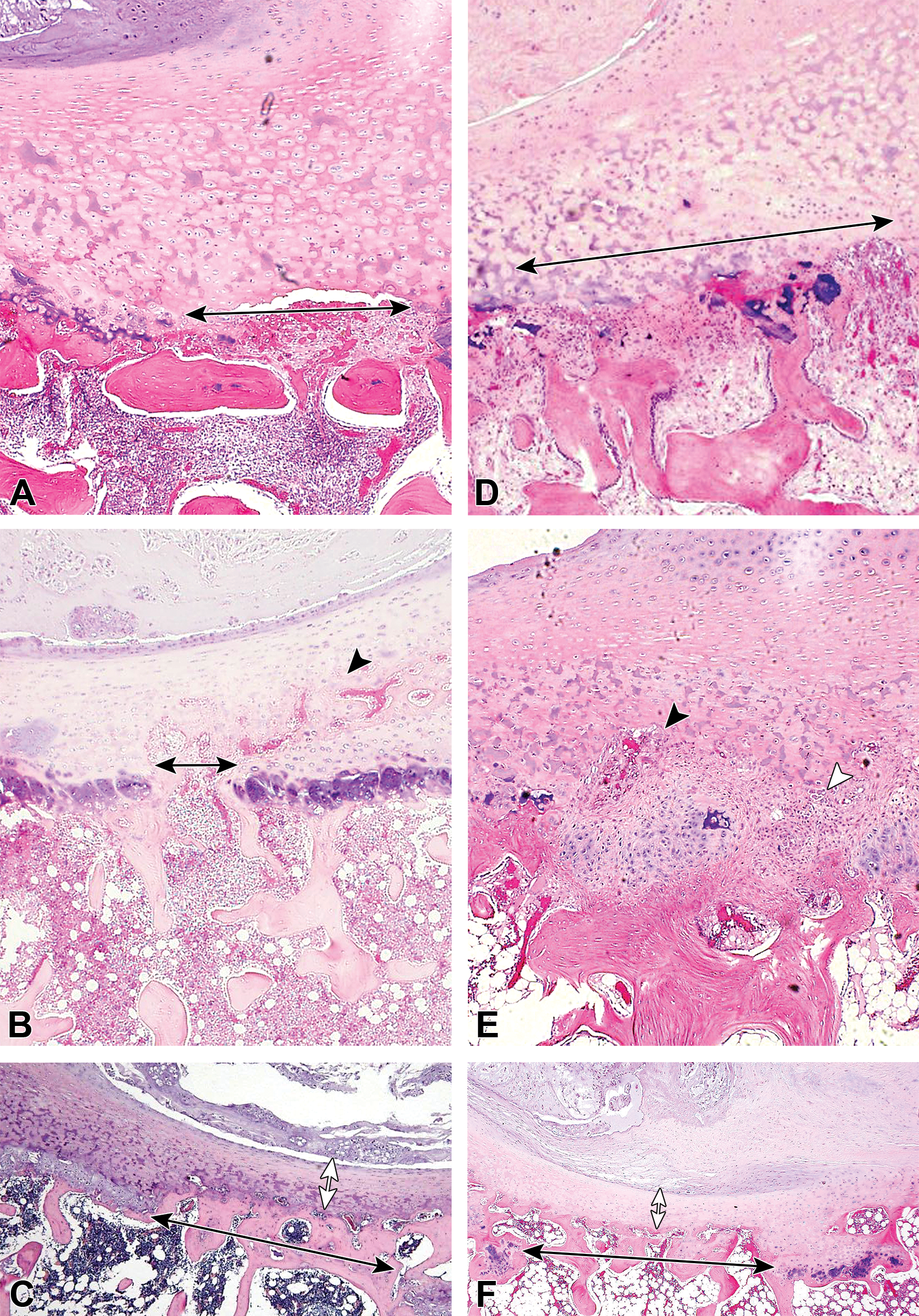
Changes in the cartilaginous endplate (CEP), epiphyseal growth plate (EGP), and vertebral body (VB) from week 4 after administration of condoliase or chymopapain. The representative samples are shown in A, B, and C for the condoliase 0.25 U group, and in D, E, and F for the chymopapain 5.7 mg group. Focal disappearance of the EGP (black double-headed arrow) and decreased bone marrow cells was began to be noted after administration of condoliase or chymopapain at week 4 (A and D), and the degrees of the change caused by chymopapain was more extent than those by condoliase. Neovascularization (black arrowhead) and ossification occurred after administration of condoliase or chymopapain in the area where cartilage of the EGP was disappeared at week 13 (B and E), leading to thinning of the CEP (white double-headed arrow) at week 26 (C and F). In the chymopapain-treated disc, fibrosis in the IVD was noted as the replacement of the damaged intervertebral disc (IVD) tissues by fibrous tissues (white arrowhead) at week 13 (E), and changes in the VB deteriorated at week 26 and fatty marrow replacement, osteocyte necrosis, and new bone formation with active osteoblasts occurred (E and F).
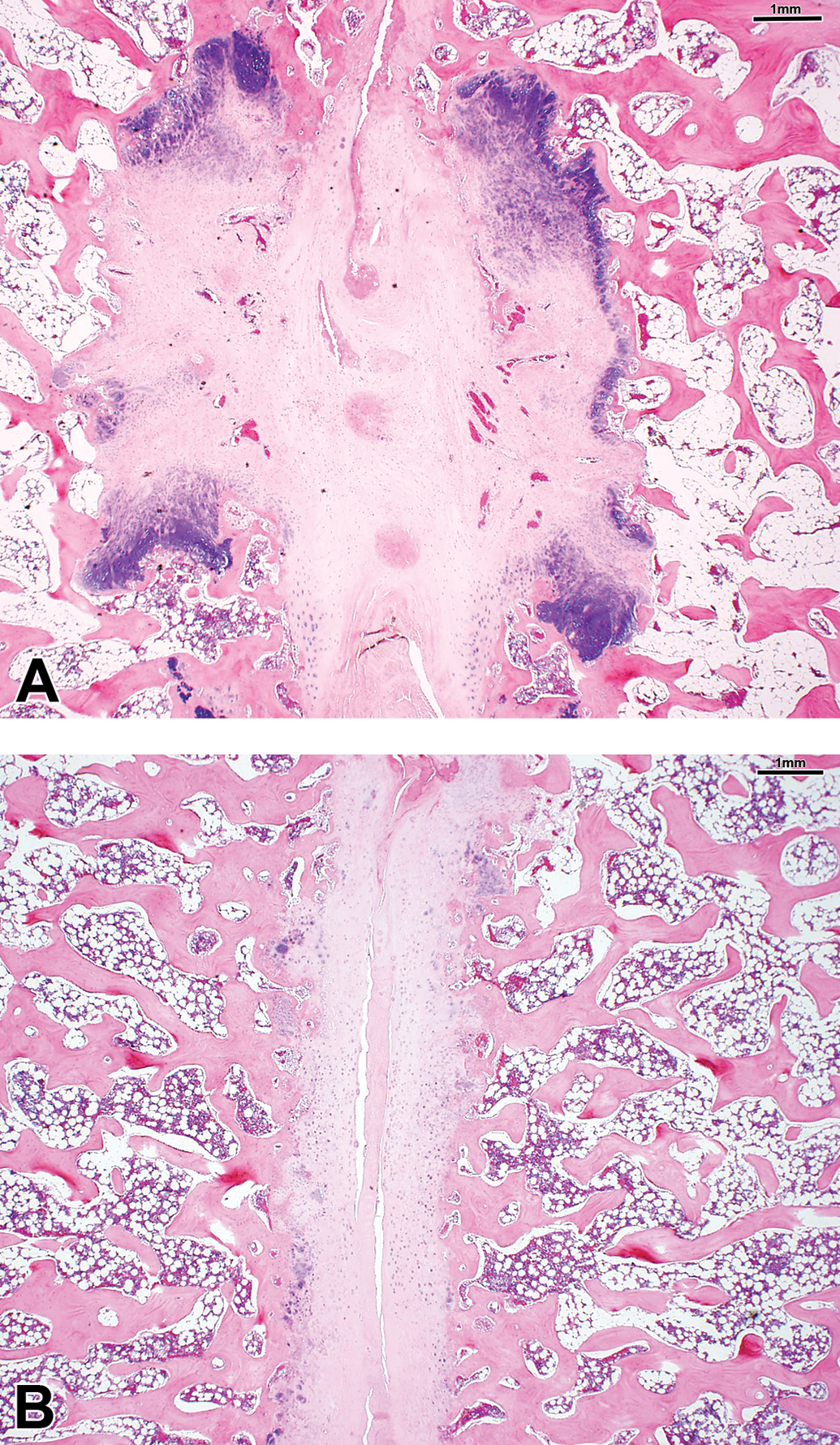
Serious cases after administration of chymopapain. The intervertebral disc was replaced by fibrosus or fibro cartilaginous tissues at week 13 (A). The cartilaginous endplates in the both sides of the disc were closely located due to the loss of the nucleus pulposus at week 26 (B).
As the deviation from methods of the study, because accurate insert by stylet needle was not performed into the L4/L5 disc, condoliase was administered into L3/L4 disc in 1 animal in the condoliase 10 U group (Table 1). However, this deviation would not have a significant impact on the quality or integrity of the study because no major differences were noted in radiographs, general conditions and gross pathology, and histopathology between this animal and the others in the condoliase 10 U group.
Discussion
Experimental chemonucleolysis was conducted in cynomolgus monkeys with condoliase at 0.25 and 10 U/disc and chymopapain at 5.7 mg/disc. Converted from the ratio of the MRI-measured NP volume, 0.25 and 10 U/disc of condoliase were equivalent to doses of 12- and 494-fold the clinical dose, respectively, and 5.7 mg of chymopapain was equivalent to 12-fold clinical dose.
Narrowing of the IVD was noted in radiographs and the DHIs began to decrease in animals treated with condoliase or chymopapain from week 1. Histopathologically condoliase- and chymopapain-treated discs showed decreased size of the NP associated with bulging of the AF. These changes were more remarkable at 12-fold the clinical dose of chymopapain compared with those at 494-fold the clinical dose of condoliase. Similar tendencies were shown in previous reports of experimental chemonucleolysis with chondroitinase ABC and chymopapain, 18 –22 and differences in the extent of changes were discussed to be related to the substrate selectivity of each enzyme. Chondroitinase ABC selectively degrades GAGs but not non-GAG substances. In contrast, chymopapain degrades PG core and link proteins and leads to strong degradation of PGs.
In addition to the above changes, our study revealed the detailed histopathological changes caused by both enzymes in the area from the CEP to the VB. In the CEP and EGP, the changes occurred sequentially after administration of condoliase. Firstly, the decreased staining of cartilage and degeneration/necrosis of chondrocytes were noted in the CEP and EGP, and then focal disappearance of the EGP occurred followed by neovascularization and ossification and thinning of the CEP. The cellular degeneration/necrosis in the CEP and EGP was noted from week 1, and the extent of the changes in the condoliase-treated disc was more localized and less severe than those in the chymopapain-treated disc. Because condoliase itself does not have cytotoxicity for chondrocytes, unlike chymopapain that has cytotoxicity due to its protease activity, 30,31 these cellular changes were considered to be attributable to secondary effects such as degradation of GAGs in the extracellular matrix. The decreased staining in the CEP and EGP caused by both enzymes was noted from week 1 and considered to be related to degradation of PGs. From week 4, regenerative changes began to occur in the IVD, and cellular regeneration in the NP, AF, and EGP was noted. Neovascularization, ossification, and thinning of the CEP were suggestive of regenerative changes concomitantly with decreased staining and chondrocyte necrosis because degradation of PGs was considered to be involved in vascular invasion into the IVD and calcification of the cartilage matrix. 32,33 As the calcified cartilage layer is known to be replaced with bone leading to the discontinuous EGP cartilage in aged-cynomolgus monkeys, like humans, 33 –35 focal disappearance of the EGP induced by condoliase was considered morphologically similar to these age-related changes. The changes caused by condoliase tended to be more localized and less severe than those by chymopapain, which showed severe neovascularization and ossification of CEP, and extensive loss of the EGP. Substrate selectivity of each enzyme was therefore considered to reflect the differences in the extent of histopathological changes in the CEP and EGP, similar to in the NP and AF.
In the bone marrow and bone tissues including the epiphyseal ring and VB, osteocyte necrosis, decreased/necrosis of bone marrow cells with new bone formation were noted in animals treated with condoliase from week 1. However, the amount of GAGs in the bone marrow and bone tissues is reported to be small (bone marrow, 0.4%; bone tissues, 0.1%), 36,37 and condoliase does not have cytotoxicity for osteocytes (unpublished results, presented at the 29th Annual Meeting of the Japanese Society of Toxicologic Pathology, January 2013). Furthermore, the changes in the bone marrow and bone tissues were similar to those caused by the changes of the mechanical stress and nutrient supply associated with the histological changes in the IVD, and therefore most likely attributable to secondary effects, that is, physical effects by acute narrowing of the IVD, decrease in the diffusion rate of nutrients from the IVD, and/or transient focal ischemia that impairs the nutrient supply from blood vessels. 23,38,39 On the other hand, the changes in the bone marrow and bone tissues were more severe after administration of chymopapain, suggesting that, in addition to secondary effects, the protease activity of the enzyme could affect these tissues because proteins predominantly exist in the extracellular matrix of the bone marrow and bone tissues. 40,41
As described above, in cynomolgus monkeys which have thick CEP like humans, beyond the NP and AF, both enzymes affected primarily the adjacent CEP and EGP, and subsequently the VB. The substrate selectivity of the enzymes was considered to be related to the extent of histopathological changes, and the changes at 12- and 494-fold the clinical dose of condoliase were more localized and less severe than those at 12-fold the clinical dose of chymopapain. At week 26, changes caused by condoliase in the IVD and the adjacent VB subsided regardless of the dose. This result suggested that enzymatic reaction of condoliase no longer progressed because condoliase was saturated with substrate (GAGs) from 12-fold the clinical dose and the changes were not exacerbated even at 494-fold the clinical dose. Furthermore, the histological features caused by condoliase at week 26 were qualitatively similar to those reported in humans, who exhibit age-related thinning, ossification or disappearance of the CEP and EGP. 34,35 These results indicated that the changes caused by condoliase were within physiological ranges and not severe to affect the basic structure of the VB.
Our study has several limitations. Firstly, although LDH is a condition that predominantly occurs between the fourth and fifth decades of life in humans, 42 young adult cynomolgus monkeys were used in our study as routine toxicity studies, but not aged cynomolgus monkeys. Therefore, there would be some difficulties in extrapolating the results of our study to clinical condition, for example, age-related differences in PGs, GAGs and water content in the NP and CEP, and potency of tissue regeneration, and so on. Secondary, as methodological limitation, histopathology was only performed on decalcified tissue sections with hematoxylin–eosin staining, which is optimal to assess overall changes of cellular components and matrices. Special staining (safranin-O, alcian blue, and toluidine blue, etc) and undecalcified tissue sections may be useful to evaluate in more detail the effects of condoliase and/or chymopapain on PGs, GAGs, and bone changes.
In conclusion, we demonstrate that histopathological changes caused by condoliase were less severe and more selective than those by chymopapain, a former chemonucleolysis agent. Condoliase is therefore expected to be a new drug suitable for selective chemonucleolysis.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320928006 - Selective Chemonucleolysis With Condoliase in Cynomolgus Monkeys
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320928006 for Selective Chemonucleolysis With Condoliase in Cynomolgus Monkeys by Dai Muramatsu, Hiroaki Yamaguchi, Yuka Minamisawa and Aisuke Nii in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The investigational drug (condoliase) was produced by Seikagaku Corporation. All authors are employees of Seikagaku Corporation working in the Research & Development Division. All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest, and none were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Seikagaku Corporation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
