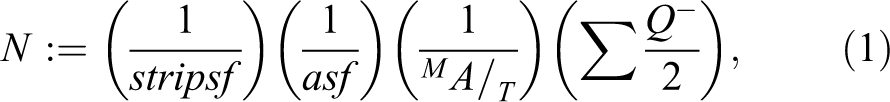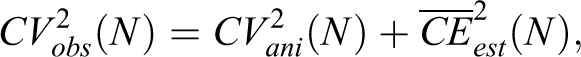Abstract
The physical fractionator is a convenient and practical solution for estimation of total cell number in a regulatory toxicology setting because it is insensitive to shrinkage allowing for paraffin processing/embedding and does not require measurement of the reference or organ volume. The principle involves sampling a known fraction of an organ in one or more steps and counting the total number of cells present in the final sample, physical disector section pairs. The total cell number in the organ is estimated by multiplying the cell count in the final fraction by the inverse of the sampling fraction(s). The key feature of the design is that tissue shrinkage due to paraffin processing occurs before the organ is uniformly sampled. Another requirement is that thermal expansion or contraction is avoided during the preparation of disector sections from the individual embedded subsamples, which ensures that the disector sections represent a known constant fraction. This vertical physical fractionator with subsampling is a simple and fast estimator to obtain precise and robust estimates of total cell number in large flat or hollow organs that do not prolong routine necropsy procedures. It is compatible with paraffin processing, avoids exhaustive sectioning, and allows for the collection of routine histopathology sections.
Keywords
Introduction
Determination of possible test article–related effects on cell number is the most common situation in toxicologic pathology where quantitative estimates are required—particularly when changes in number are subtle and cannot be appreciated qualitatively by the pathologist (from inspection of ordinary 2-dimensional sections). When the magnitude of these changes is small but maybe toxicologically significant, sensitive, accurate, and precise quantitative methods are needed. Although surrogate estimates of cell numbers such as cell profile counts in routine sections collected for histopathology have been widely used, these indirect or assumption-based estimates cannot conclusively identify small changes. Design-based stereology provides the necessary sensitive tools to obtain accurate and precise estimates of total cell numbers in an organ.
Stereological approaches for cell number estimation have not been routinely used in toxicologic pathology for many practical reasons. Historically, these methods were labor-intensive and somewhat time-consuming, lacking the throughput necessary to deal with large numbers of animals typical for a toxicology study in a time-effective manner. The estimation of total cell number was usually done in a two-step process. Numerical density (number of cells per unit volume) was first estimated from statistically valid tissue samples then multiplied by the reference or organ volume. This required estimation of organ volume and meticulous attention to tissue processing volume changes, for example, shrinkage, which substantially impacts numerical density estimates. Because of shrinkage concerns, plastic, frozen, or vibratome sections were typically required instead of paraffin embedding.
Cell number estimation was greatly simplified with the description of the fractionator principle that avoids the problems of tissue processing-induced shrinkage and the requirement for estimation of the reference or organ volume. 1 -3 For general concepts of stereology including the fractionator principle the readers are referred to several review papers and book chapters in the reference section. 4 -6 The fractionator is a sampling principle where a known fraction of the entire organ is sampled in one or more steps and cell number is estimated in the final fraction. The total number is estimated by multiplying the number in the final fraction by the inverse of the sampling fraction(s). In the final fraction, cells must be counted using a 3-dimensional sampling probe, the disector. 7 The physical disector consists of a set of adjacent sections in which cells are counted that are present in one section but not the other. When combined with an unbiased sampling frame, this principle ensures that cells are only counted once and proportionally sampled to their number independent of their size and shape. 2,3 When the simple fractionator is combined with either optical or physical disectors, the estimator is known as optical or physical fractionator, respectively. 1
One great benefit of the classical physical fractionator is that paraffin sections can be used. A form of the classical physical fractionator principle, known as the simple physical fractionator, is ideal for the toxicologic pathology setting. This design is specifically targeted at the estimation of cell numbers in small organs or regions of interest, such as a brain nucleus that is amenable to embedding in toto in a paraffin block. A known fraction of all sections is collected during exhaustive sectioning consisting of a uniform random set of adjacent thin serial sections (a disector section pair). Because processing-induced shrinkage occurs before the fractionator sample is collected, the paraffin disector sections can be used. Although the design meets a key need for the toxicologic pathology setting because routine sections for histopathology can also be collected during sectioning, it is labor-intensive because exhaustive sectioning is required.
Widespread application of classical physical fractionator designs has been limited for two reasons. First, to count cells in physical disectors, systematic sampled fields in two serial sections must be spatially matched at high magnification. This is technically challenging and time-consuming. However, advancement in stereology software (AutoDisector Visiopharm, Hørsholm, DK’) now automates the capture and registration of matched high magnification fields. Secondly, exhaustive sectioning is required to collect the final fractionator sample. Although this cannot be avoided for small organs where subsampling is not possible, novel fractionator designs can now obviate this requirement for larger organs.
We report here a novel metric physical fractionator design that is ideal for estimation of total cell number of interest in flat or hollow organs using paraffin-embedded tissue that includes collection of sections from a preferred location for standard histopathology. 8 The pivotal feature is that paraffin-embedded subsamples (strips) are taken after complete processing (dehydration, paraffin infiltration, and block cooling) of the organ. Urinary bladders from male and female cynomolgus monkeys are used as a model system. The urinary bladder can be a potential target organ with certain drug classes where subtle increases in cell numbers are impossible to appreciate qualitatively due to variable organ contraction.
Materials and Methods
Animals
All studies were conducted in accordance with the GlaxoSmithKline Policy on the Care, Welfare and Treatment of Laboratory Animals, and were reviewed the Institutional Animal Care and Use Committee either at GlaxoSmithKline or by the ethical review process at the institution where the work was performed.
Cynomolgus monkeys (4/sex) aged 2 to 5 years were utilized in this study. Before necropsy, each monkey was pretreated with a subcutaneous injection of ketamine HCl, anesthetized with an intravenous injection of sodium pentobarbital, and then exsanguinated. Urinary bladders were voided of urine and inflated with approximately 300 mL of Modified Davidson’s fixative. Bladders were tied with a suture at the base of the bladder, ventral side marked with India ink, entire bladder removed, and immersion fixed for 18 to 24 hours in the same fixative.
Tissue Sampling
Bladders were cut into quadrants to allow flattening of the tissue before sampling. Quadrants were processed to paraffin using a Vacuum Infiltration Processor (VIP; Sakura FineTek Torrence, CA) to allow for global shrinkage to occur prior to sampling of the organ.
Bladder quadrants were removed from the tissue processor and placed on the cold plate of an embedding center (Thermo Shandon, Carlsbad, CA), mucosal side up, and allowed to cool to ∼4°C. The quadrants were randomly rotated around an axis oriented perpendicular to the mucosal surface (designated the “vertical axis”; Figure 2) and a tissue slicer consisting of thin metal cutting (razor) blades uniformly separated by 4-mm spacers was placed over the quadrant and 4-mm thick tissue strips were prepared (Figure 1). Only strips with a surface cut by the slicer on their “left” edge were included; hence the left-most strip did not participate in the estimate.
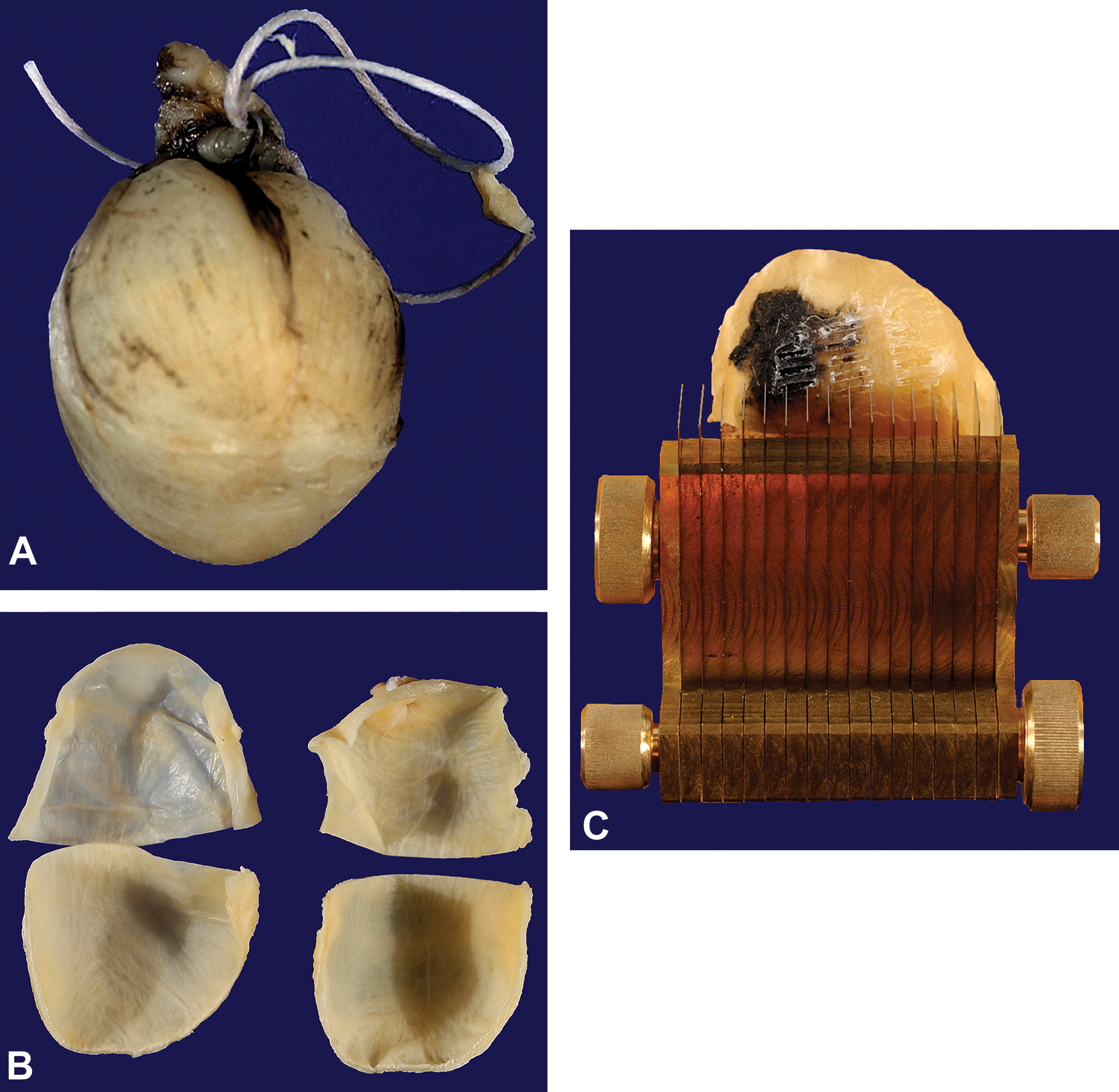
Fixed and inflated primate urinary bladder (A). First, bladders were divided into four quadrants and processed to paraffin (B). Cooled bladder quadrants were randomly rotated and sliced using a tissue slicer with blades fixed a constant distance apart (4 mm) and placed over each bladder quadrant (C).
To reduce the number of strips for analysis, strips from the 4 quadrants were pooled and a known fraction, ⅓ to ½, was sampled using the smooth fractionator sampling principle illustrated in Figure 2. 9 This fraction is known as the tissue strip sampling fraction, stripsf. The total number of strips from each bladder was targeted at 10 to 12 to yield 10 to 12 disector pairs for analysis. The smooth fractionator sampling principle is a form of systematic uniform random sampling that ensures all strips are given equal probability of being sampled that greatly reduces the variance of sample sets from an individual. The final prechilled paraffin-infiltrated tissue strips were placed “left” cut edge down in chilled metal embedding mold; the temperature of the mold base was maintained at 4°C on a temperature-controlled embedding plate. While holding the tissue strip in place, the molds were then filled with melted paraffin, placed immediately on the cold plate, and topped with the embedding cassette to form the paraffin block. This embedding procedure was used to eliminate thermal deformation of the front cut surface of each tissue strip, from which disector pairs were to be collected. The embedding orientation provided sections of mucosa in a standard cross-sectional presentation.
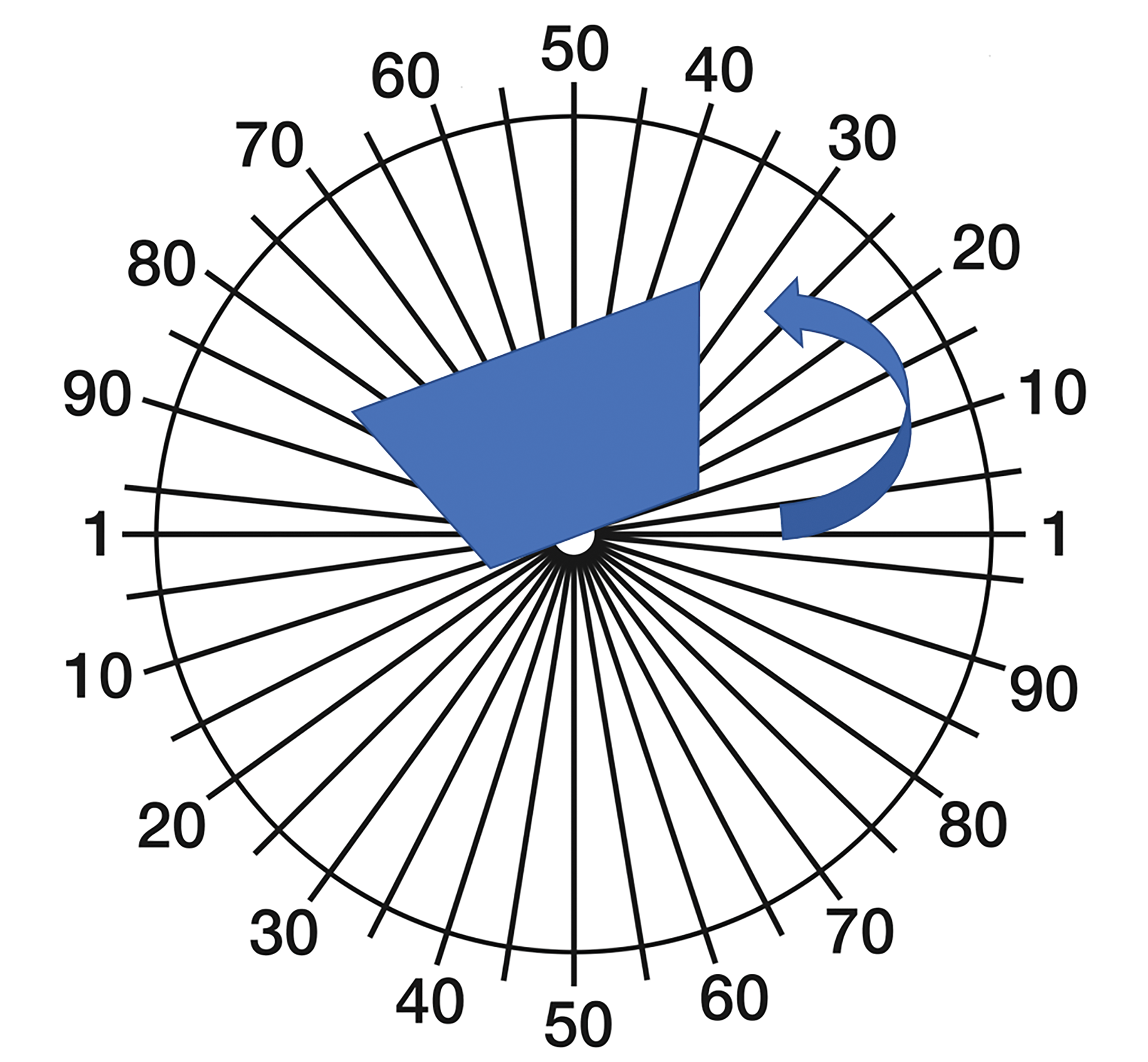
Illustration of generating isotropic uniform random (IUR) vertical slices using a cosine weighted orientator (cutting guide). Bladder quadrants were placed and rotated on top of the cosine weighted cutting guide (arrow). The quadrant was rotated aligned with a preassigned random start (angle). The tissue slicer was placed on the rotated quadrant to generate the strips. This sampling procedure was performed on each bladder quadrant to acquire the tissue strips in figure 3.
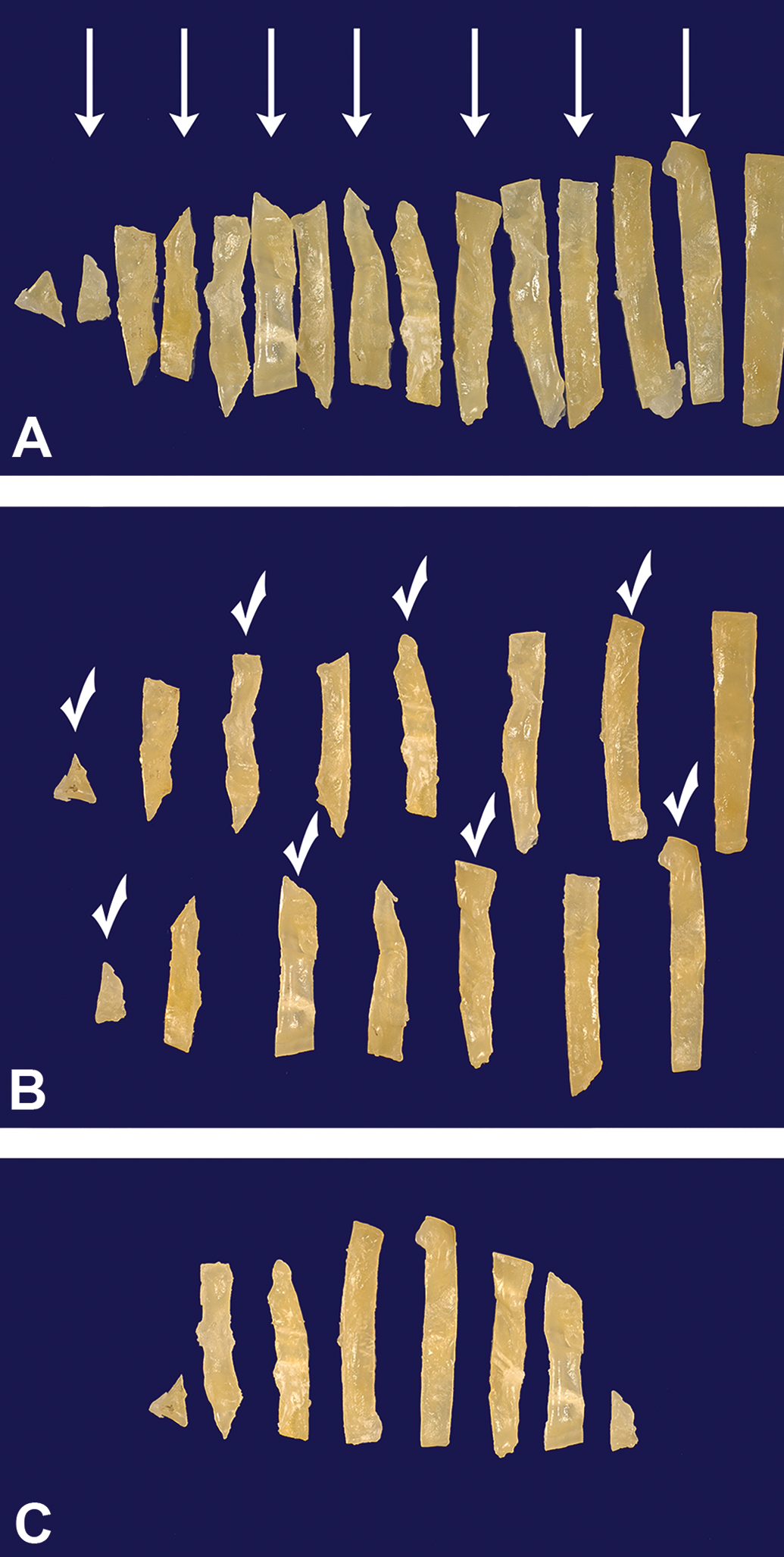
Example of the smooth fractionator sampling design for subsampling of bladder tissue strips. All tissue strips except the left-most in the cutting sequence are arranged according to size and starting with the second smallest, every second strip is pushed down to form a row below the existing one (A, arrows). With a random start, sample selection starts in the upper row (left to right) followed by the second row, but in the opposite direction (B). In this example, the random start is 1 with every other strip selected (check). The final strip sample with a sampling fraction, stripsf, of ½ (C).
Tissue Sectioning
An accurate measurement of the microtome advance (MA) is required in this design. Microtome advance determines the disector height and this height must represent a known and constant fraction of the T = 4 mm width of the paraffin-infiltrated strips, in which all processing-induced shrinkage has occurred, to obtain unbiased estimates using the metric fractionator principle as applied in this design. 8 Section thickness is determined by the MA at a given micrometer setting, and micrometer settings often do not accurately reflect the true block advance. 10 Consequently, the calibration of the microtome being used should be checked and the calculated microtome advance (MA) must be determined.
Briefly, the thickness of a paraffin block was determined by several measurements at each corner using a Vernier caliper. At a micrometer setting of 2 µm, 100 sections were removed, and the block thickness was measured again. This was performed three times. The average MA was then calculated. An automated microtome (Microm H355S) fitted with Tissue-Tek low profile disposable blades was used to generate the sections. For this dedicated microtome, MA was determined to be 2.03 µm at a micrometer setting of 2 µm. With this design, T = 4 mm, the width of the strips, and MA = 2.03 µm, the block advance or disector height, the section sampling fraction, ssf =(MA/T) was therefore 2.03 µm/4 mm or 1/1970. Note that the section sampling fraction (and the disector height) is independent of subsequent changes in section thickness due to floating and mounting on glass slides.
Blocks were mounted and carefully faced until a complete section was obtained. While in the block holder, the surface was chilled with a gauze cloth soaked in ice water and an ice cube to allow for the section sampling to occur at approximately the same temperature as the organ sampling. Two consecutive 2-µm (nominal) sections (2-µm physical disector) were collected from each block and immunohistochemically stained for β-catenin to delineate cell boundaries and facilitate unequivocal identification of individual transitional epithelial cells (TECs). The best practice rule for determining the optimal disector height is to target ¼ - ⅓ of the height of the counting feature.
Immunohistochemistry
Slides were stained on the Ventana Discovery System and all reagents, with the exceptions noted below, were obtained from Ventana (Ventana Medical Systems, Inc, Tucson, Arizona). Briefly, sections were deparaffinized, rehydrated, and subjected to heat antigen retrieval (CC1 citrate buffer Ventana Medical Systems, Inc, Tucson, Arizona). Nonspecific staining was blocked with 3% hydrogen peroxide and protein block (DAKO, Carpinteria, California) with 5% normal goat serum. Monoclonal rabbit IgG anti-β catenin antibody (Epitomics Abcam. Cambridge, MA) was applied manually at a concentration of 5 μg/mL for 1 hour. Controls included omission of primary antibody and substituting nonimmune rabbit IgG for primary antibody (SouthernBiotech, Birmingham, Alabama) matched for concentration. Anti-β catenin antibody was detected with anti-rabbit IgG HRP labeled polymer (Ultra Map, Ventana Medical Systems, Tuscon, Arizona) and visualized with 3,3-diaminobenzidine (DAB) chromogen. Sections were counterstained with hematoxylin, dehydrated, cleared, and coverslipped with a permanent mounting media.
Enumeration of TECs
Transitional epithelial cells were quantified in immunohistochemically stained disector section pairs using the Visiopharm Integrated System (VIS; Visiopharm, Hørsholm, Denmark) interfaced with a Nikon 90i Eclipse microscope equipped with a motorized stage. Low magnification overview images of the section pairs were acquired and the area of the uroepithelium was coarsely defined by outlining the region of interest using the AutoDisector module. An unbiased counting frame (25 to·25 µm) was selected and the urothelium was sampled. A predefined fraction of the delineated region of interest, known as the areal sampling fraction, asf was designated by the user. The asf is determined in a pilot study to achieve a statistically appropriate count across the entire sample (i.e 100-200 counts). This module allows for automated and unattended sampling of systematic uniform random microscopic fields of physical disector section pairs and the spatial matching and registration of high-resolution images captured with a 60× objective. Captured, registered, and matched disector fields were stored and imported into the NewCAST module (Visiopharm, Horsholm, DK) for the operator to view the match fields and manually perform the cell counts. The NewCAST system with the Autodisector™ software effectively aligns matched fields and allows the operator to refine that alignment but does require strict attention at microtomy to avoid section artifacts and section deformation in the water bath. The disector counting principle was used to enumerate cells sampled by the unbiased counting frame in these matched microscopic fields. 7,11
The image from one slide was designated the “counting” field and the image of the matched field from the other slide, the “lookup” field. Cells were only counted if their nuclear profile was sampled by the unbiased counting frame in the “counting” field but were absent in the “lookup” field. The field designation was then switched (counting field becomes look up field and vice versa) and a second count was made in the opposite “direction.” This practice of “counting in both directions” of the disector increases efficiency and reduces the number of fields that need to be sampled to obtain a predefined count (typically 100 to 200 counts) to achieve an acceptable precision (frequently a coefficient of error (CE) in the range of 10% to 15%).
Because the umbrella or cap cells, specialized cells that line the luminal surface of the bladder, can be multinucleated, the nucleus could not be used as the unique counting feature of these particular cells and the appearance of the cell “top” was used as the counting criterion. This counting rule applies for single isolated cells such as an osteoclast or cells in a single layer, such as the umbrella cells. This however would not apply to cells that have a more complex 3-dimensional spatial distribution (i.e. hepatocytes). Cell tops were identified as solid brown profiles (Figure 4), which in the lookup section had developed into a small ring.
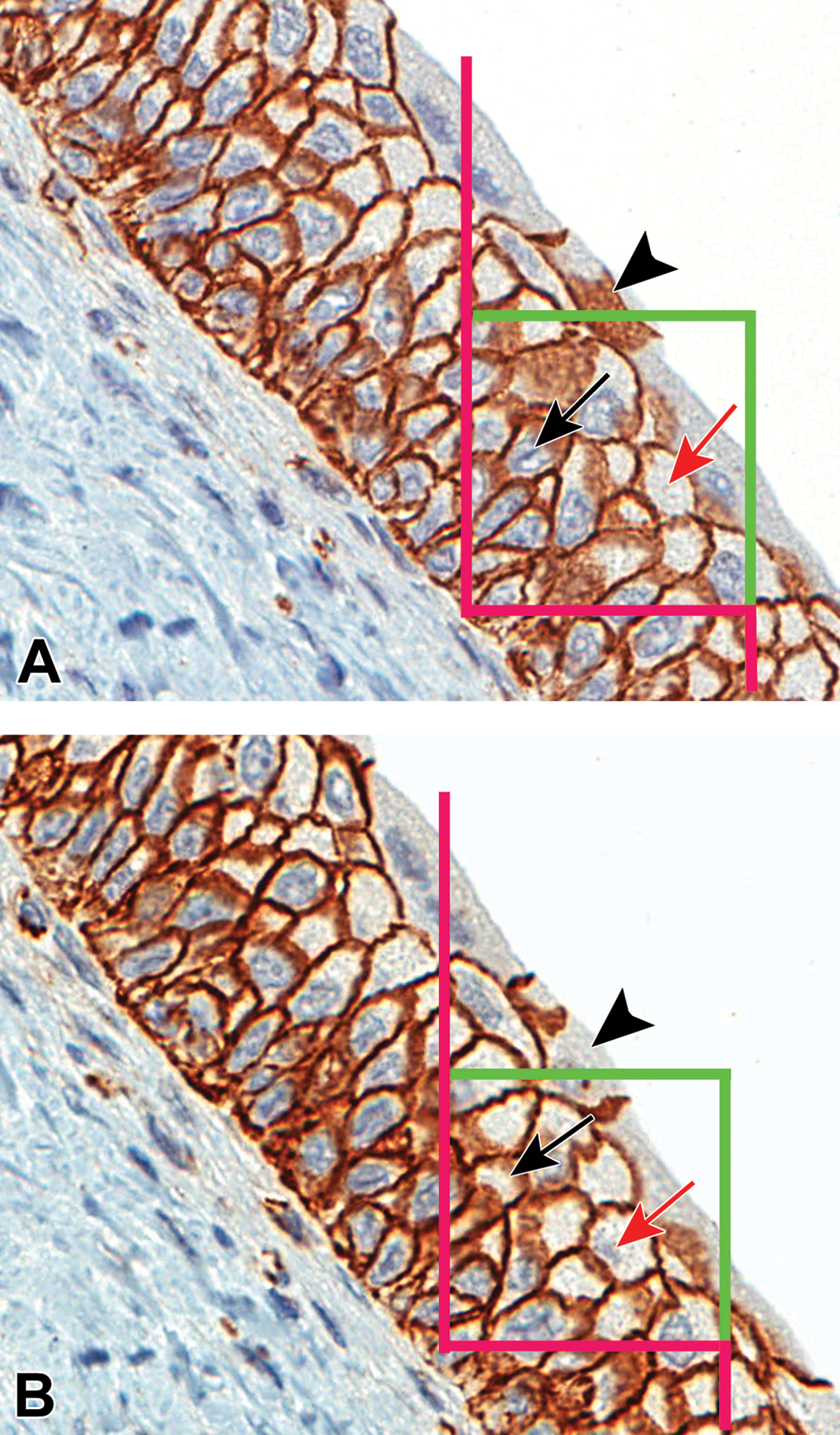
High-resolution image pairs captured at 60× with a superimposed unbiased counting frame were presented to the user for counting. The unbiased disector counting rule specifies that e.g. nuclei are sampled in the left sampling frame if they are inside the frame or touch the two green inclusion lines but are excluded if they touch the three red lines in any way. A cell is counted if the sampled nucleus is not seen in the right look-up field. The marked nuclei (below arrow in A) indicate a counting event following the disappearance of the nuclei (black arrow in B). For cap cells, the appearance of the cell “top” (arrowhead in A) and the disappearance of the cell “top” (arrow head in B) indicated a counting event as defined by the counting rule for single layer multi-nucleated cells. The red arrows illustrate a two-way sampling such that image B becomes the reference image and image A becomes the look-up image. Applying the same counting rule, the marked nuclei (red arrow in B) indicate a counting event following the disappearance of the nuclei (red arrow in A).
The nucleus was used as the unique counting feature for mononuclear basal and intermediate cells of the uroepithelium. Areal sampling fraction (asf) of uroepithelium was set at 1/100 of the total area of the uroepithelium to obtain a total count (ΣQ−) of 100 to 200 cells collected over the complete set of disector pairs for a bladder. Calculation of total TEC number was estimated based on the metric fractionator principle using the following equation:
where N is the total number of TECs, := denotes estimation, stripsf is tissue strip sampling fraction (½), asf is area sampling fraction (1/100)., T is strip width (4 mm), MA is microtome advance (2.03 µm),
The variability among animal estimates,
which states that the estimator imprecision,
Estimates of Total Transitional Epithelial Cells in Nonhuman Primate Urinary Bladder.
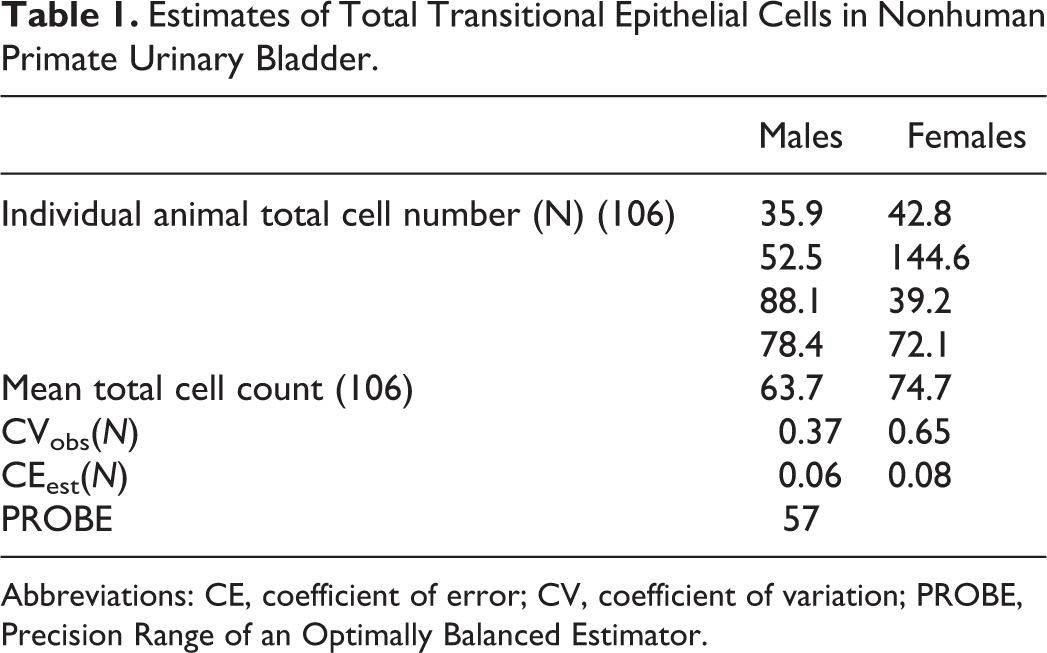
Abbreviations: CE, coefficient of error; CV, coefficient of variation; PROBE, Precision Range of an Optimally Balanced Estimator.
The
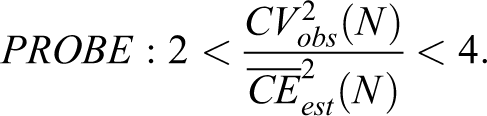
PROBE is applicable to each group of the study and useful for all stereological estimators. Once known, 3 possible outcomes are considered: if PROBE < 2, the estimator imprecision is too large for the purpose; in this instance, one should reexamine the sampling design, identify the part of the sampling that is contributing the most to the CEest(N) and intensify the sampling, if PROBE > 4, the estimator precision may be too good for the purpose; in this instance, one should consider reducing the workload where the greatest resources is being used, and if PROBE is between 2 and 4, then the precision is adequate for the purpose.
Results
The mean total number of TECs in male and female monkey bladders were 63.7 × 106 and 74.7 × 106, respectively. The average actual counts per animal were 378 (males) and 188 (females). The observed coefficient of variation (CVobs(N)) was 37% and 65% in males and females, respectively. The mean CEest(N), the error contributed by the stereological methods (sampling and counting), was 6% and 8%. Since there is no indication of a sex difference, data from both sexes were pooled resulting in mean total TEC estimate = 69.2 × 106, mean CVobs(N) = 0.53, mean CEest(N) = 0.07, and PROBE = 57. The PROBE value very clearly suggests that the precision of the estimator is way too good (ie, too much resource is being used in the sampling method) and a reduction in sample size at the organ and section level is needed. For example, adjusting the strip width to 10 mm instead of 4 mm and reducing the fields of view (FOV) will reduce the effort with essentially no impact on the precision.
Discussion
A novel metric fractionator design with subsampling was implemented in this study to estimate the total TEC number in monkey urinary bladders. 8 The key features of the design are that all shrinkage due to paraffin processing occurs before uniform sampling of the organ and before a set of disector sections is collected from each uniform sample or strip, carefully avoiding thermal expansion or contraction during the preparation of sections. Several other metric fractionators with subsampling from paraffin-embedded material have been published. 8,12
The technical challenges encountered in this pilot study were primarily associated with the requirement to process the organ quadrants to paraffin. Since sampling occurred on the paraffin-infiltrated quadrants, the most labor-intensive component of the design was slicing the paraffin-infiltrated bladder quadrants into uniform 4-mm strips and the subsequent embedding of the strips. The careful embedding of these uniform tissue strips to ensure no deformation to the tissue cutting surface required three trained personnel to perform the slicing and embedding. Estimated time for slicing and embedding the bladder strips was approximately 1 hour per bladder (which is reduced to ∼20 minutes in the more efficient design, as previously discussed). Because the design obviates the need for exhaustive sectioning in a fractionator design, sectioning time was kept to a minimum. Automated immunohistochemical staining of thin paraffin sections ensured uniform staining of the feature of interest and is critical for the execution of the subsequent identification of unique counting events.
When this work was performed, the Visiopharm system required loading the glass slides onto a motorized stage attached to a microscope and the software captured low magnification super images to enable the section registration (alignment) of the disector pairs. The systematic uniform sampling and capture of high magnification image matched disector fields was automated by AutoDisector software and was performed unattended. Cell counts were performed manually on the stored, captured images typically requiring the operator 1 hour per animal. Recent advances to the VIS allow for direct importation of high-resolution digital slide scans to capture high magnification image–matched disector fields versus performing image capture from the glass slide to allow for a more streamlined workflow within the Stereology module making the acquisition of high-quality images and quantitative data generation very efficient.
This metric fractionator design is amenable to the estimation of cell number in hollow organs that can be fixed and paraffin processed ensuring all processing-induced shrinkage has occurred prior to subsampling. Important technical issues include microtome calibration and avoiding thermal expansion of the cutting surface of the tissue strips during embedding and section preparation. If careful attention is given to these details, the estimates are unbiased because the disector height represents the predefined fraction of the tissue slice thickness.
Footnotes
Acknowledgments
The authors would like to thank Beverly Maleeff and Tracy Gales for their photography expertise in generating the photographs for this article.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.