Abstract
The “exposome” is an individual’s lifetime spectrum of chemical exposures beginning at conception. An exposome includes general external influences such as pollution and weather; external individual-specific factors (diet, infections, self-selected chemical intake); and internal individual-specific constituents (metabolic byproducts, microbiome derivatives, inflammatory mediators, stress hormones, etc). The exposome paradigm is inherent in animal toxicity testing because laboratory studies are designed so that subjects share a common exposure history encompassing not only exposure(s)/treatment(s) but also other chemical sources (eg, air, bedding, food, water). Toxicologic pathologists should remember that some differences in responsiveness to a test article may reflect subtle differences in individual exposomes of seemingly equivalent test animals. Translation of toxicity data obtained in tests of genetically inbred animals maintained under controlled environmental conditions to produce quasi-identical exposomes at best offers only approximate guidance regarding potential responses in genetically heterogeneous human populations who live in many environmental settings and thus have divergent, complex exposomes.
Recent advances in understanding the genetic bases of diseases have shown that genetic damage explains only a portion of disease risk. Instead, the environment and gene–environment interactions account for most human disease. The “exposome” paradigm was proposed in 2005 in recognition that the genetic underpinnings of disease often are influenced profoundly by an individual’s cumulative experience of myriad environmental influences. 1 This paradigm is a direct acknowledgment that nongenetic factors play a major role in modulating health and disease. 2 –5 The exposome, both conceptually and practically, provides a holistic view of an individual’s health and disease. It includes exposures from diet, lifestyle, and personal behaviors as well as encompassing how the body responds to these challenges. This mini-review explores the relevance of the exposome concept for the modern practice of toxicologic pathology as applied to toxicity studies used for product discovery and development.
The environmental influences included within an individual’s exposome may be divided conceptually among 3 broad categories (Figure 1). 7 The first category is the general external environment, which includes communally experienced geographic factors such as climactic and local weather patterns as well as air and water pollution. Many people and animals within the region will be exposed to these elements, though likely to different degrees depending on individual choices (time spent indoors vs outdoors, proximity to highly traveled urban streets vs remote rural areas, etc). The second category is the specific external environment, which comprises uniquely experienced extrinsic factors such as an individual’s lifestyle (environmental microflora, indoor air quality, noise, nutritional status, occupation, physical activity, etc) and chemical consumption (alcohol, caffeine, food preservatives, medications, “recreational” drugs, smoking, etc). 7,8 The final category is an individual’s specific internal environment, which consists of one’s personal chemical makeup as dictated by the intermingled byproducts of one’s hormonal balance (especially sex- and stress-related), metabolism, microbiome (particularly, but not confined to, gastrointestinal commensals and pathogens), and immune responses, among many others. 9,10 The overall chemical composition resulting from these biological processes has been defined as an individual’s “exposotype,” or exposure phenotype. 11 The combinations of external and internal exposures are both complex and numerous, and when superimposed on the heterogeneous genetic backgrounds characteristic of human populations, the implication is that most people are likely to possess a unique exposotype.
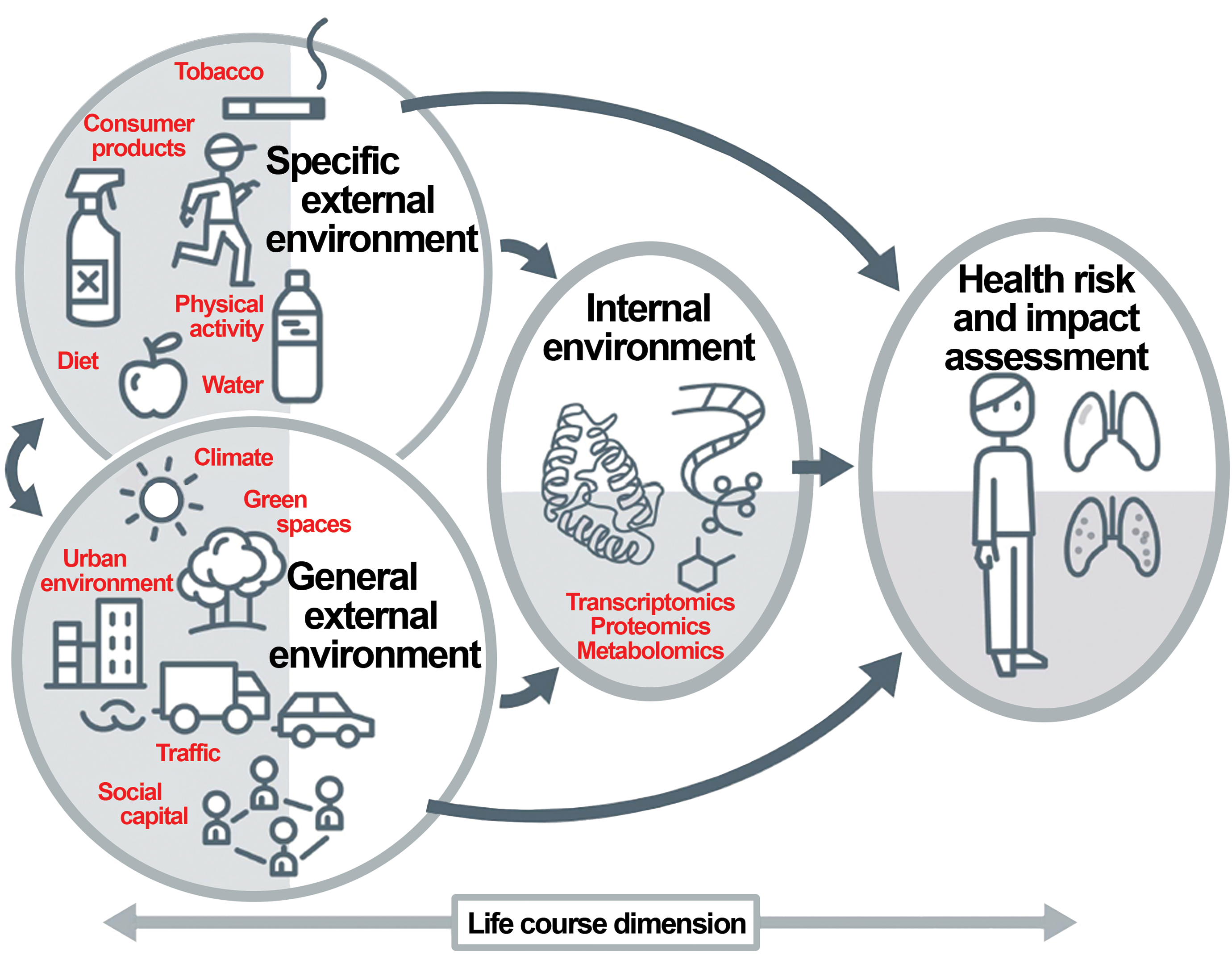
The exposome paradigm, demonstrating the interrelationship of factors in the general external environment, an individual’s specific external environment, and an organism’s specific internal environment as they apply to the health risk imparted by one’s total lifetime exposure to potential toxicants. Reproduced from Vrijheid, 6 by permission of BMJ.
The exposome reflects an individual’s cumulative exposure to various chemical influences over the entire lifespan. 1 In this context, life is judged to start at conception due to the exquisite vulnerability of developing organisms to chemical exposures. Furthermore, unlike the relatively static genome, an individual’s exposome is dynamic, reflecting shifts in external and internal chemical exposures over the course of hours, days, or years. 12 The clear implication is that assessment of an individual’s exposome (external and/or internal) at a single time point is unlikely to discover, let alone characterize, all the potential health effects that might be posed by encounters with complex environmental exposures. Therefore, the exposome experienced by animals in toxicity studies is unlikely to be limited strictly to the test article(s) being administered during the set treatment period of the study. That said, current experimental practices of including concurrent age- and sex-matched control animals (to minimize genetic variation among individuals) as well as maintaining careful control over environmental and husbandry conditions in the laboratory (substantially to standardize the “exposome” across subjects) should ensure that the test article(s) is the instigator of all or nearly all of the chemically induced health effects engendered during conventionally designed and conducted animal toxicity studies.
At present, the exposome paradigm chiefly is used in epidemiological and ecotoxicological studies. 4,13 Efforts to unravel these complex scenarios where exposures occur in an uncontrolled environmental setting have focused on biomonitoring of individuals, where possible, to measure genuine exposures rather than environmental sampling to predict possible exposures. 8 Measurement of exposure is still not well developed but includes multiple approaches with heavy reliance on (1) biomarkers of exposure, such as measurements of chemical and/or metabolite levels in biological specimens, 8 or (2) biomarkers of functional/molecular impact, especially the “omics” entities (genomics, transcriptomics, proteomics, metabolomics, etc). 5 The data sets obtained during early studies in humans have shown that “omics” biomonitoring can quantify the dynamic changes in levels of multiple chemicals, genes, and gene products (especially proteins) for individuals, such as simultaneous exposures related to such well-defined factors as diet, environmental pollutants, occupational exposures, and tobacco. 14 In humans, such assessments are particularly instructive when “omics” technologies are used to quantify molecular signatures of exposure or altered biological activity in banked specimens from large population-based epidemiological studies. In theory, similar individual biomonitoring could be performed for animals in toxicity studies. However, analysis of chemicals other than the test article(s) and any relevant biomarkers of exposure and/or activity should be unnecessary during routine animal toxicity studies given the highly controlled environmental conditions in modern laboratory facilities and the relatively homogeneous genetic background of the subjects, especially for those studies that adhere to Good Laboratory Practice guidelines. That said, investigators must keep in mind that issues may arise during both the laboratory phase (eg, if animals are sourced from different vendors) and data interpretation (eg, if historical control data are used). Evaluating individual-specific exposomes may offer a useful means for selecting test subjects for future animal toxicity studies conducted in specialized populations that are rare and expensive (eg, genetically engineered animal strains, nonhuman primates). In contrast, animals in field trials are likely to have more divergent exposomes due to daily and seasonal oscillations in both environmental conditions and diet, especially in subjects that migrate during some portion of their lifespan.
The exposome paradigm is inherent in the conventional practice of toxicologic pathology, in 2 important ways. First, toxicity studies in laboratories are planned so that the test animals share a common exposure history that encompasses not only the chemical treatment(s) being tested but also other potential external sources (eg, facility, bedding, air, food, water). In this fashion, the design of toxicity studies deliberately minimizes any variations in the 3 categories of chemical exposures: general external environment, specific external environment, and specific internal environment. Second, the exposome paradigm is complementary to the adverse outcome pathways (AOP) concept, 13 which now is a common strategy in assessing potential toxic hazards induced by chemicals (agricultural, industrial, and therapeutic) and chemical mixtures. The AOP approach links a sequence of cellular and molecular events (ie, mechanistic data) to harmful health outcomes without respect to any exposure or other effect that might induce such events; nonetheless, consideration of a given AOP in the context of exposome data may serve as a means for merging in silico, in vitro, and in vivo toxicity data to quickly and accurately provide a more useful cumulative risk assessment. 15 Toxicologic pathologists are integral members of the research teams that assemble data sets for building AOP models, so in this respect toxicologic pathologists are well positioned to participate in relating mechanistic data to the exposome.
Toxicologic pathologists should remember that some differences in responsiveness to a test article may reflect subtle divergence in the individual exposomes experienced by seemingly equivalent test animals, even in a controlled laboratory setting. In most cases, any such variations will be theoretical since neither in-life nor toxicologic pathology data sets are capable of testing this premise. Moreover, the exposome paradigm highlights the fact that translation of toxicity data sets obtained in tests of genetically inbred animals maintained under highly controlled environmental conditions at best offers only approximate guidance regarding the spectrum of chemically-induced responses that might occur in genetically heterogeneous human populations who reside in and move among many different environmental settings. Despite these caveats, the exposome concept is a useful theoretical model that should be kept in mind when interpreting and extrapolating toxicologic pathology data for hazard identification and characterization as well as risk and safety assessment. In terms of human health, the exposome, when coupled with advances in genetics and medicine, will help lead to improved strategies aimed at preventing and treating many chronic diseases, especially such complex entities as Alzheimer's disease, autism, diabetes, and heart disease.
Footnotes
Acknowledgments
The authors thank Ms Beth Mahler for her assistance in optimizing the resolution of the figure and Reviewer No. 3 for several insightful comments that helped to improve the focus of the message in this piece.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
