Abstract
The liver is the primary hematopoietic organ of the mammalian body during the fetal stage. The postnatal liver retains immunologically important functions and contains a substantial population of immunologically active cells, including T and B lymphocytes, Kupffer cells, liver-adapted natural killer (NK) cells (pit cells), natural killer cells expressing T cell receptor (NKT cells), stellate cells, and dendritic cells. The liver is the major site of production of the acute phase proteins that are associated with acute inflammatory reactions. Kupffer cells have an important role in the nonspecific phagocytosis that comprises a major component of the barrier to invasion of pathogenic organisms from the intestine. Hepatic NK and NKT cells are important in the nonspecific cell killing that is important in resistance to tumor cell invasion. The liver has a major role in deletion of activated T cells and induction of tolerance to ingested and self-antigens. Disposal of waste molecules generated through inflammatory, immunologic, or general homeostatic processes is accomplished via the action of specific endocytic receptors on sinusoidal endothelial cells of the liver. Age-related changes in sinusoids (pseudocapillarization), autophagy, and functions of various hepatic cell populations result in substantial alterations in many of these immunologically important functions.
Introduction
Functions of the liver in normal immunobiology were previously reviewed (Parker and Picut 2005). The present treatise addresses alteration in liver immune functions that are related to age and other life events.
Overview of Immunological Functions of Liver
The liver has a number of major functions in both innate and adaptive immunity, which may be outlined as follows:
Innate (nonspecific) immunity involvement:
production of acute phase proteins
nonspecific phagocytosis
nonspecific cell killing
disposal of waste molecules of inflammation and nonspecific immunity
nonspecific immunoregulation.
Adaptive (specific or acquired) immunity involvement:
deletion of activated T cells
induction of tolerance to ingested and self antigens
extrathymic proliferation of T cells
disposal of waste molecules of specific immunity
specific immunoregulation.
Following is a brief discussion of these major functions, with emphasis on variations that result from age or other life stage influences.
Ontogeny of Hepatic Immune Functions
The immune system of rodents develops from a population of pluripotential hematopoietic stem cells (HSC) generated early in gestation from uncommitted mesenchymal stem cells in the intra-embryonic splanchnoplure surrounding the heart (Landreth 2002). HSC give rise to all circulating bone cell lineages, including cells of the innate and acquired immune system. Definitive hemopoietic progenitors have been shown to emerge in the aorta-gonad-mesonephros (AGM) region before the fetal liver develops. Progenitors committed to T, B, or myeloid lineages exist in the AGM region of mice at gestation day (GD) 10. The progenitor cell population in the AGM at GD12 is two hundred to a thousandfold increased over GD10 level, suggesting that hematopoiesis has just started in the AGM at GD10 (Ohmura et al. 1999).
Progenitor B and T cells arrive in the liver at approximately the same point in gestation, but there are differences in the relative prominence of the various developmental forms of hepatic B and T cells. Pro-T cells began to appear in the liver of mice at GD9 in a study using JORO 37-5 as a marker for pro-T lymphocytes (Palacios and Samaridis 1991). One day later, the JORO 37-5+ cells migrated to and colonized the thymus, where the population expanded vigorously during the next four to five days. B progenitor cells of mice were detectable in the embryonal body rather than yolk sac around GD9.5 (Ogawa et al. 1988). The progenitor B cell population immediately enters blood and becomes detectable in the liver and yolk sac of the GD10 embryo. At GD15-16, the B cell progenitor population enters the femur as vascularization of the femur starts. When 14.8 antibody was used as pro-B cell marker in mice, 14.8+ cells appeared in the liver on GD11, reached peak numbers in the first week after birth, and decreased thereafter (Velardi and Cooper 1984). Pre-B cells (defined as mu+) appeared in significant numbers in the liver at GD13, reached a peak shortly after birth, and disappeared from the liver by the end of the second postnatal week. Pre-B cells appeared in spleen and bone marrow on GD15 and GD19, respectively. Mature (sIgM+) B cells appeared in the liver at GD17, reached a peak at birth, and declined thereafter. Mature T cells were later acquisitions in the liver, spleen, and bone marrow.
There are major shifts in B cell populations as the newborn rodent encounters environmental antigenic stimuli. In a study of the frequency of B cell lineages in liver and spleen of mice during first ten days of life, newborn liver contained large numbers of early precursors as well as pre-B cells, but at most ages the number of mature (sIg+) B cells was low (Jyonouchi and Kincade 1983). By contrast, the spleen had early precursors and pre-B cells at birth, but these declined with age as mature (sIg+) cells increased. In this study, mature B cells were identified by the presence of surface Ig, pre-B cells had mu heavy chains but no surface Ig, and earlier precursors that did not synthesize immunoglobulin were identified by monoclonal 14.8 antibody.
Macrophages appear in the liver somewhat later than lymphocytes. In studies of fetal and neonatal Wistar rats, using macrophage markers ED1, ED2, and ED3, the first ED1-positive macrophages appeared in liver on GD15 and did not express Ia (dendritic cell marker) (van Rees et al. 1988). Mac1+ cells were present in the liver of mice on GD11, where they peaked shortly before birth and declined subsequently (Velardi and Cooper 1984). Waves of myeloid growth began in spleen and bone marrow by GD15 and GD19, respectively.
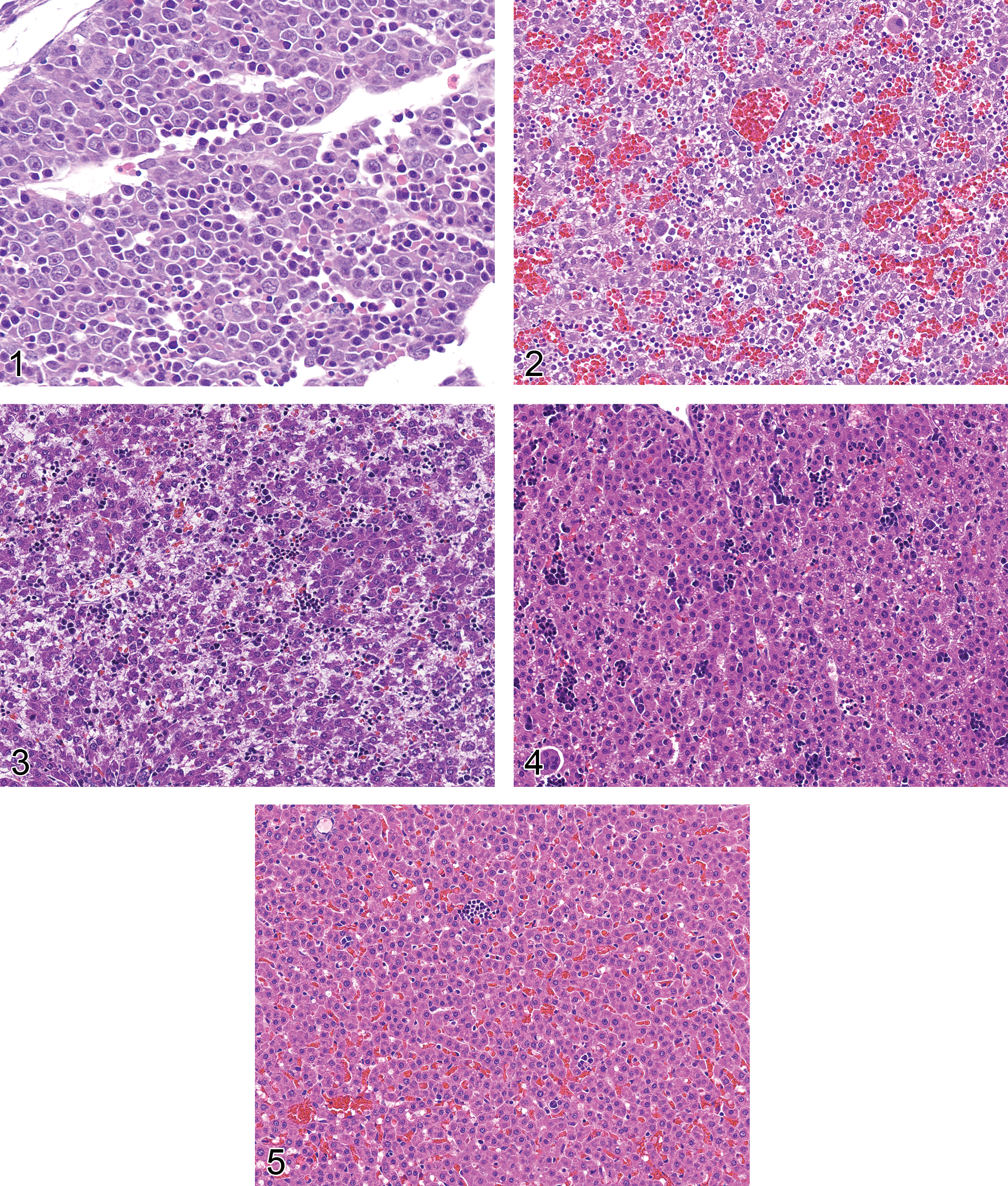
Observations by the authors (Burns-Naas et al. 2008; Weinstock et al. 2010) are consistent with the liver being the primary hematopoietic organ of the rat at GD15 (Fig. 1). Hematopoietic activity in the liver is somewhat diminished by GD20 (Fig. 2), though the liver retains a high level of hematopoietic activity through postnatal day (PND) 0 (Fig. 3). Hematopoietic activity in the liver becomes progressively less prominent through PND3 (Fig. 4) and PND10 (Fig. 5), and by PND21 is essentially at adult level. However, the liver has a well-known ability to return to hematopoietic activity in the face of a significant demand on hematopoietic reserves.
Figure 1 Rat liver, GD15. A diffuse population of hematopoietic cells is interspersed with disorganized, primitive-appearing hepatocytes. GD = gestation day; H&E stain, 400×.
Figure 2.— Rat liver, GD20. Large numbers of densely stained hematopoietic cells are interspersed with hepatocytes, the latter containing a variable number of clear lipid vacuoles. GD = gestation day; H&E stain, 200×.
Figure 3.— Rat liver, PND0 (day of birth). Numerous darkly stained clusters of hematopoietic cells are interspersed with immature-appearing hepatocytes. PND = postnatal day; H&E stain, 200×.
Figure 4.— Rat liver, PND3. Darkly stained clusters of hematopoietic cells are interspersed with hepatocellular cords. PND = postnatal day; H&E stain, 200×.
Figure 5.— Rat liver, PND10. Widely scatter clusters of hematopoietic cells are scattered through hepatic parenchymal elements. PND = postnatal day; H&E stain, 200×.
Characteristics of Hepatic Lymphoid Cell Populations
Hepatic T cells are phenotypically different from T cells in blood, lymph nodes, or spleen (Norris et al. 1998):
CD4:CD8 ratio of hepatic T cells is reversed (1:3.5 for liver vs 2:1 for blood lymphocytes); Higher percentage of CD3+CD4+CD8+ (double positive) and CD3+CD4–CD8– (double negative) T cells in the liver; TCRγδ T cells are five times more abundant in liver than in blood (15% vs 2.7%).
These observations suggest local control of function and/or differentiation of hepatic T lymphocyte populations.
Experimental data suggest T cell–mediated immunity declines with age (Orme 1987). Young (three to eighteen months) mice were fully resistant to tuberculosis (TB), but progressive mortality was observed in old (twenty-four to twenty-eight months) mice. Death of the older mice was associated with inability to contain or eliminate TB infection in the spleen and liver and inability to prevent spread of TB to lungs, due to inability of aged mice to generate protective T cells. By contrast, there was no apparent decrease in function of macrophages in aged mice. There is experimental evidence that vitamin E reverses some of the senescence effects in T cells (Wu and Meydani 2008).
Microarray data from liver tissue of mice have shown an increase in inflammation-associated genes with age (Amador-Noguez et al. 2004). Flow cytometry studies in mice have shown an age-related decrease in TCRαβ T cells and concurrent increase in TCRγδ T cells in the liver (Tsukahara et al. 1997). Immunohistochemistry studies performed on the liver of aged mice revealed clusters or foci of immune cells, preferentially located near perivascular regions (Singh et al. 2008). These observations suggest the aging liver exhibits a pronounced inflammatory or immunoreactive status accompanied by increased immune cell infiltration, with a shift from TCRαβ to TCRγδ T cells.
It is generally assumed that TCRγδ T cells participate in protection against intracellular microbial pathogens, and there is experimental data to suggest there are age-related variations in these functions. In one study, fourteen-day-old mice lacking TCRγδ T cells were far more susceptible to Listeria monocytogenes than wild-type mice of the same age. It was concluded that TCRγδ T cell gamma-delta cells play a critical role in protection against Listeria in early life of mice, probably because the TCRαβ T cell population is not fully competent at that age (Emoto et al. 2001).
CD8+CD122+ (activated) T cells of mice increase with age and may influence the progression of pathologic processes. In a study based on interleukin-12 (IL-12) priming and subsequent lipopolysaccharide (LPS) challenge in mice that were four to fifty weeks old, mortality due to a generalized Schwartzman reaction increased with age (Sato et al. 2005). All mice ≥ twenty weeks of age died within forty-eight hours of LPS challenge. Young mice transferred with CD8+CD122+ T cells from aged B6 nude mice showed an enhanced Schwartzman reaction, thus confirming the increased susceptibility to LPS was associated with the CD8+CD122+ T cells rather than some other aspect of aging.
Deletion of Activated T Cells by the Liver
After an immune reaction subsides, there remains a population of immunologically active cells and molecules that must be neutralized or eliminated. The liver has a major role in eliminating these signaling and effector molecules from the circulation (see the following) and has a specific role in removal of T cells that were activated at sites distant from the liver.
A basic understanding of leukocyte emigration from blood vessels into inflamed tissue is required for discussion of activated T cell clearance by the liver. Leukocyte emigration from postcapillary venules into inflamed tissue is a two-step process that involves an initial low-avidity, selectin-mediated rolling process followed by a high-avidity, integrin-mediated adhesion of leukocytes to the luminal surface of endothelial cells. The initial rolling step serves to slow the movement of leukocytes to a rate where they make effective contact with the integrin molecules expressed on the luminal surface of activated endothelial cells in the area of inflammation. The combination of selectin- and integrin-mediated adhesion serves to localize leukocyte emigration to the site of inflammation.
Leukocyte emigration in the liver occurs in sinusoids rather than post-capillary venules and does not require the initial selectin-mediated rolling step (Wong et al. 1997). Selectin-mediated rolling is not required because hemodynamic and other factors serve to slow the rate of blood flow through liver sinusoids. These alterations in blood flow vary in different regions of the hepatic acinus and vary between species (MacPhee, Schmidt, and Groom 1995). The end result is that leukocytes in hepatic sinusoids have extensive, “slow-motion” exposure to sinusoidal endothelial cells without a preliminary rolling step.
Endothelial cells in vessels at sites of inflammation have cytokine-mediated expression of adhesion molecules such as intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1). This local expression of ICAM-1 and VCAM-1 on the luminal surface of endothelial cells of post-capillary venules serves to localize emigration of leukocytes to the site of inflammation. In contrast to typical endothelial cells, sinusoidal endothelial cells (SEC) constitutively express ICAM-1 and VCAM, thus facilitating high-avidity, integrin-mediated adhesion of activated T cells in the absence of a local inflammatory reaction.
Hepatic trapping of activated T cells is dependent on presence of ICAM-1 and/or VCAM-1 as well as an activated status of the T cells. Intrahepatic accumulation of murine CD8+ T cells was significantly reduced when either ICAM-1 or VCAM-1 was blocked by specific antibody, suggesting these adhesion molecules are responsible for the majority of trapping of activated CD8+ T cells in the mouse liver (John and Crispe 2004). Perfusion of the liver with lymphocyte mixtures revealed selective hepatic retention of activated CD8+ T cells, but not resting T cells or T cells that were actively involved in apoptosis (Kuniyasu et al. 2004). Following trapping there was a temporary expansion of the population of activated T cells in the liver, but the massive proliferation of T cells that is seen at sites of inflammation did not occur in the liver. The transient expansion of the T cell population was followed by apoptosis and a reduction in the intra-hepatic population of CD8+ T cells over the next four days.
Failure of continued expansion of the trapped CD8+ T cell population may be explained by the cytokine microenvironment of the liver. To survive and numerically expand, activated T cells must receive a co-stimulatory signal from macrophages or helper T cells. Absence of co-stimulatory and helper molecules in the liver microenvironment results in apoptosis of sequestered activated T cells (“death-by-neglect”). As a result of these processes, the liver is regarded as a “sink” for activated T cells (Mehal, Juedes, and Crispe 1999).
In addition to the death-by-neglect mechanism previously mentioned, there is evidence for direct apoptosis induction in trapped CD8+ T cells. Galectin-1 is a beta-galactoside binding protein that is produced by endothelial cells and induces apoptosis in bound activated T cell (Perillo et al. 1995; Rabinovich et al. 1998; Lotan et al. 1994).
Intrasinusoidal binding of activated T cells may explain the accumulation of T cells in the liver of mice with defects in apoptosis mechanisms, such as the lpr/lpr mutant (Huang et al. 1994). In this situation, T cells are bound in sinusoids but lack the necessary intracellular machinery to complete the apoptotic process (Cohen and Eisenberg 1991).
Experimental data indicate that T cells from aged mice are resistant to activation-induced cell death in vitro. When severe combined immunodeficiency (SCID) mice were reconstituted with T cells from aged or young mice with the D(b) H-Y TCR (herein called Tg71) transgenic marker, recipients of T cells from aged mice exhibited a threefold increase in autoreactive T cells when compared with recipients of T cells from young mice (Hsu et al. 2001). By day twenty-one posttransfer, the recipients of T cells from aged mice but not young mice exhibited infiltration of CD3+ cells into nonlymphoid organs. This feature of T cells in aged animals, coupled with the effects of extrathymic T cell production and altered self-antigen presentation in the liver (discussed in the following), offer possible explanations for the increased incidence of autoreactive immunologic phenomena in aged populations.
Liver Involvement in Extrathymic T Cell Production
T cell development and differentiation occur primarily in the thymus, but not exclusively so. Extrathymic pathways of T cell differentiation exist in the intestine and liver (Sato et al. 1995), where T cell populations may arise from their own preexisting precursor cells rather than thymus (Sugahara et al. 1999). Extrathymic T cells are also present in the uterus and exocrine glands, for example, salivary gland (Abo 2001). Extrathymic T cells have unusual features: large proportion of TCRγδ T cells, double-negative (CD4–CD8–) T cells and self-reactive oligoclones, constitutive expression of the IL-2Rβ chain, expression of the αα homodimer of CD8 in CD8+ cells, expression of TCR(int), and presence of self-reactive clones (Abo 1993; Ohteki et al. 1992). Evidence suggests extrathymic pathways comprise a primitive and fundamental immune system and play a pivotal role in immune reactions with aging, bacterial infections, malignancies, autoimmune diseases, and pregnancy (Abo 1993).
Extrathymic T cells increase with age, apparently in synchrony with thymic involution. In youth, extrathymic T cells are increased in number and function by stress, in autoimmune diseases, and during pregnancy. Acute thymic atrophy typically accompanies this increase in extrathymic T cell activity (Abo 2001).
A study of hepatic mononuclear cell populations in mice revealed the overall hepatic mononuclear cell population increased with age, whereas the number of mature thymocytes decreased (Ohteki et al. 1992). This effect on hepatic mononuclear cell populations was more prominent in conventionally housed mice than in specific pathogen free (SPF) housed mice. TCR(int) cells increased in frequency in aged mice. BrdU labeling showed extensive proliferation of cells in hepatic sinusoids of aged mice. These observations suggested extrathymic T cells may differentiate predominantly in the liver in older mice.
Multiple observations suggest a gender difference in immunologic responses in the liver. There is a synchronous expansion of TCR(int) cells in the liver and uterus during pregnancy (Kimura et al. 1995). TCR(int) cells are more common in the liver and other organs of female mice than male mice, and estrogen administration activates extrathymic T cells in mice (Kimura et al. 1994). Ovariectomy of female mice results in a reduction in the population of TCR(int) cells in the liver, and the effect is reversed by administration of physiologic doses of estrogen. This combination of observations suggests estrogen has a role in the known predominance of autoimmune diseases in females of several mammalian species (Okuyama et al. 1992).
Production of Acute Phase Proteins
Acute inflammation in mammals is associated with a transient increase in a group of circulating proteins that are collectively known as acute phase proteins (APP). This includes serum amyloid A protein (SAA), fibrinogen, C-reactive protein (CRP), complement factors C3 and C9, haptoglobin, hemopexin, ceruloplasmin, α2-macroglobulin, CD14, α1-antichymotrypsin (ACT), α1-cysteine proteinase inhibitor (α1CPI), α1-antitrypsin (AAT), and lipopolysaccharide binding protein (LBP) (Fey et al. 1994; Fan et al. 2002). Circulating acute phase proteins are responsible for many of the systemic effects of inflammation, which are largely aimed at preparing the body for resistance to systemic invasion, facilitating local resistance to pathogens, and limiting the potential for bystander damage by effector molecules and products of inflammation.
The acute phase response is generated when injury at an extrahepatic site prompts local macrophages to release a first wave of cytokines that includes IL-1, tumor necrosis factor alpha (TNFα), and a small amount of IL-6. The first wave of cytokines causes surrounding immunologically active cells to release a second wave of cytokines, which includes a large amount of IL-6 that promotes massive production of acute phase proteins by hepatocytes. IL-6 is the prototype signaling molecule in the induction of the acute phase response, but IL-22, which is produced by activated T cells, also has the ability to up-regulate production of acute phase proteins (Nagalakshmi et al. 2004). Production of acute phase proteins has historically been considered a function of hepatocytes, but more recent evidence indicates a similar spectrum of proteins is produced during involution of the mammary gland and uterus (Stein et al. 2004) .
The pattern of plasma protein concentrations, including acute phase proteins, in neonatal rats differs from that seen in adult animals. In early postnatal rats the concentration of α2-macroglobulin was nearly two hundred times higher than the level in adult animals and the concentration of α1-acid glycoprotein was substantially lower than the level in adult animals (Thomas and Schreiber 1985). The concentration of prealbumin was low in the immediate postnatal period, but increased concurrently with increases in thyroxin and corticosterone. Positive and negative acute phase responses to turpentine injection were blunted in neonatal rats, reaching adult levels by PND7-19 (Schwarzenberg, Potter, and Berry 1991).
Aging is associated with modulation of the acute phase response. Induction of haptoglobin, α-1-acid, and T kininogen glycoprotein in rats was reduced or retarded with aging (Gomez et al. 2008). The initial induction of IL-6 in aged rats was comparable with that seen in young rats, but the subsequent IL-6 response was increased relative to that seen in young rats. Lower levels of APP despite higher IL-6 signal generated by local immunocytes suggest a hepatocellular deficit in production of the acute phase proteins. During experimentally induced acute phase response in aged rats, there was an increase in the time required for acute phase protein mRNA to be produced and a decrease in the maximum level of mRNA produced (Carter, Post, and Papaconstantinou 1991). Plasma fibrinogen levels in turpentine-treated young rats were elevated sevenfold over untreated control levels, while plasma fibrinogen levels of similarly treated aged rats were elevated only fourfold over control levels (La Porta, Franchi, and Comolli 1998). All these observations suggest the immune system continues to generate adequate signals in aging animals, but the liver of aged animals has declining ability to respond to those signals.
High levels of α2-macroglobulin or fibrinogen, or low levels of albumin, have been associated with a higher risk of mortality in aged rats (Mayot et al. 2007). This seems intuitively correct, as it implies a higher level of inflammation is associated with higher risk of mortality. It should not be construed to indicate the acute phase proteins per se have an adverse impact on survival.
The acute phase response of offspring is influenced by maternal nutrition during pregnancy. A low protein diet (12, 9, and 6 g casein/100g diet, vs 18 g/100g for controls) fed before conception and during pregnancy resulted in a trend to blunted innate immune responses in the offspring (Langley et al. 1994). A more thorough presentation of the immunological implications of pregnancy and lactation are presented elsewhere in this issue (see Kuper).
Nonspecific Phagocytosis
Nonspecific phagocytosis in the liver is mediated primarily by Kupffer cells (KC). KC are present throughout the liver, but there is variation in the population density, cytologic characteristics, and physiologic functions of KC cells in different zones of the hepatic acinus/lobule (Sleyster and Knook 1982). KC are the largest population of tissue macrophages in the body (Naito et al. 2004). They constitute approximately 30% of the sinusoidal cells in rat liver, or 14-20 × 106 Kupffer cells per gram of tissue. Pulse labeling with latex particles produced a population of latex-labeled cells that did not change appreciably over a three-month period, suggesting a long lifespan for these resident macrophages. The KC population has a low mitotic rate even when accentuated by a six-hour arrest by vinblastin and has a low rate of labeling by 3Hthymidine (Bouwens et al. 1986).
The liver has been shown to be the primary site for removal of experimentally administered antigen and immune complexes. Soluble immunoglobulin G (IgG) complexes are eliminated from the circulation mainly by the liver, predominantly by KC and, to a lesser degree, endothelial cells. KC recognition of the Fc domain of immunoglobulins results in nonspecific phagocytosis of immune complexes as well as antibody-coated particles such as microorganisms and eukaryotic cells. Presence of the Fc receptor allows KC to have a significant role in control of inflammatory and immunologic processes (Ravetch 1994).
In addition to Fc receptors for binding and phagocytosis of erythrocytes coated with immunoglobulins, KC also have complement receptors for binding and phagocytosis of erythrocytes coated with complement fragments (human C3b or mouse inactivated C3b) (Smedsrod et al. 1985). The avid binding of immunoglobulin- or complement-coated erythrocytes allows KC to have a major role in removal of erythrocytes from the circulation, resulting in KC accumulation of iron-positive materials in disease processes that involve intravascular erythrolysis or erythrocyte sequestration.
Bacterial endotoxin (lipopolysaccharides) derived from the gastrointestinal tract and endotoxin injected intravenously in animals are cleared principally by the liver and taken up primarily by KC (Naito et al. 2004). Within the liver, LPS binds to LPS-binding protein (LBP), which then facilitates the transfer of LPS to CD14 on the surface of KC. Signaling of LPS through CD14 is mediated by the downstream Toll-like receptor 4, resulting in activation of KC and direct involvement of the innate immune system (Su 2002).
Aged rats are more susceptible to the lethal effects of endotoxin than young rats (Durham et al. 1990). In a study that involved electron microscopy and biochemical changes in young (six months) vs aged (twenty-four months) rats exposed to endotoxin, aged rats given endotoxin had more neutrophils in sinusoids at thirty minutes postexposure and thereafter as compared with young rats. KC of endotoxin-treated aged rats frequently had phagocytosed platelets and neutrophils, which was rare in young rats. Hepatocellular morphologic injury was more pronounced in aged rats, and serum biochemical indicators of hepatic damage were higher in aged rats.
KC number and activity level changes with age and may vary between rodent species. Isolated perfused livers of young, middle-aged, and aged F344 rats (four to six months, thirteen to fourteen months, or twenty-four to twenty-six months, respectively) had increased KC numbers and phagocytic activity in old age (Hilmer, Cogger, and Le Couteur 2007). By contrast, isolated perfused liver from aged (twenty-two months) vs young (three months) mice had 35% lower carbon particle phagocytosis and 65% lower carbon-induced oxygen consumption, increased GSH depletion, and enhanced protein oxidation (Videla, Tapia, and Fernandez 2001). When reviewing data of this type, one must carefully consider the known balance between phagocytic activity vs activation potential in KC and the variation in those functions that is observed in different regions of the hepatic lobule (Sleyster and Knook 1982).
Intact KC function is necessary for development of some pathologic processes, and there is documented age-related variation in the KC input. For example, the liver of aged male F344 rats is resistant to cadmium-induced hepatotoxicity (Yamano, DeCicco, and Rikans 2000). Following cadmium administration, phagocytic activity of KC was increased in young (five months) but not aged (twenty-eight months) rats. Hepatic levels of IL-1β and cytokine-induced neutrophil chemoattractant (CINC) were elevated in young rats treated with cadmium. Much lower cadmium-associated elevation in IL-1β and CINC were present in aged rats. Observations in this study indicated (1) KC activation is essential for inflammatory liver disease induced by cadmium, (2) IL-1β and CINC are important mediators of cadmium-associated liver inflammation, and (3) attenuation of cadmium-induced liver injury in senescent rats is caused by impairment in KC activation, leading to lower production of CINC and less inflammatory liver injury.
Nonspecific Cell Killing
Effector cells involved in nonspecific, intrahepatic cell killing include natural killer (NK) cells and natural killer T (NKT) cells. The NK cell population includes a liver-adapted subpopulation known as pit cells. NK cells are bone marrow–derived mononuclear cells that have markers of both T lymphocytes and macrophages. NK cells have distinct azurophilic cytoplasmic granules that contain perforin and granzymes, which are involved in cell membrane attack and induction of apoptosis in target cells. As opposed to target recognition by cytotoxic T lymphocytes (CTL), recognition of target cells by NK cells does not require the major histocompatibility complex (MHC), and no immunologic memory is involved in NK-mediated cell killing.
Pit cells are intra-sinusoidal, liver-specific NK cells that are defined morphologically as large granular lymphocytes (LGLs) and functionally as liver-associated natural killer cells. Pit cells are located inside sinusoidal lumina, where they adhere to endothelial cells and Kupffer cells. Morphologic features of pit cells suggest they represent a more mature form of circulating NK cells (Nakatani et al. 2004).
NKT cells have many of the phenotypic and physiologic characteristics of NK cells, but have the additional feature of surface expression of T cell receptor (TCR). The TCR on NKT cells interacts with CD1, as opposed to the MHC-1 or MHC-2 interaction with the TCR on T lymphocytes. Antigen processing into either MHC-1 or MHC-2 context involves time-consuming steps for antigen processing and molecule transposition to the cell surface. By contrast, constitutive expression of CD1 on cell surfaces allows NKT cells to interact with target cells without delay, thus constituting a “rapid response team.” NKT cells are abundant in the liver (Nakatani et al. 2004), and it has been shown that NKT cells can develop extrathymically from precursor cells in the liver (Shimamura et al. 1997). This liver-resident, locally regenerating pool of rapid response killing cells has a significant role in defending the liver from invading tumor cells.
With increasing age there are changes in the hepatic population of NK cells. Mouse liver contains T cell populations that exhibit high (TCR(hi)) and intermediate (TCR(int) ) levels of TCR expression. The TCR(int) population, which includes NK cells, is subdivided into NK1+ and NK1– subsets based on presence or absence of the NK surface marker. Natural killer activity in the liver is associated with the NK1+ TCR(int) population, not the NK1– TCR(int) or TCR(hi) populations. The NK1+ TCR(int) cell population in the liver increases until middle age, then declines, resulting in a reduction in this critical first-line defense against invading tumor cells at an age when tumor metastasis to the liver is most likely.
In eight-week-old mice, the mononuclear cell population of the liver is approximately 25% NK cells. NK cells were detected in the liver of mice at four weeks of age, peaked at six to eight weeks, and decreased starting at approximately week nine. Appearance of increased number of liver NK cells coincided with cessation of rapid liver weight increase in mice, and hepatic NK cells were significantly cytotoxic against regenerating hepatocytes in partially hepatectomized liver, suggesting that NK cells might be involved in regulation of hepatocyte growth (Itoh et al. 1988).
There are age-related changes in NK- and NKT-mediated functions as well as cell counts. Injection of CpG oligodeoxynucleotides (CpG-ODN) to mimic bacterial infection in young (six weeks) and aged (fifty to sixty weeks) C57BL6 mice resulted in a higher level of tumor necrosis factor (TNF) production in Kupffer cells of aged mice (Kawabata et al. 2008). Multi-organ dysfunction syndrome (MODS) and lethal shock were induced in aged mice, but both these occurrences were rare in young mice. Lethal shock and MODS were greatly decreased in aged mice that were experimentally depleted/deficient in TNFα, FasL, or NKT cells. KC of aged mice had increased Toll-like receptor 9 expression. CpG-ODN challenge augmented TNF receptor and Fas-L expression in liver NKT cells. Old mice had lower capacity for generation of IFN-gamma, IFN-alpha, and perforin and reduced capacity for CpG-induced tumor cell cytotoxic activity of hepatic NK cells. In summary, CpG-ODN–related hepatic injury and MODS mediated by NKT cells via the TNF and FasL-mediated pathway increased with age, but the antitumor activity of hepatic NK cells decreased with aging.
Disposal of Waste Molecules
Hepatic sinusoidal endothelial cells (SEC) have a voracious appetite for circulating molecules, to the degree that SEC are known as “professional pinocytes.” SEC receptor-mediated endocytosis occurs primarily via four categories of surface receptors: collagen receptor, mannose receptor, scavenger/hyaluronan receptor, and Fc receptor.
Formation and turnover of collagen result in the release of large quantities of collagen components into the circulation. Collagen receptors on SEC of rat liver mediate the endocytosis of collagen alpha 1 monomers and denatured collagen (gelatin) (Smedsrod, Johansson, and Pertoft 1985), circulating C-terminal propeptide of type I procollagen (Smedsrod, Melkko, et al. 1990), and NH2-terminal propeptides of types I and III procollagen (Melkko et al. 1994).
Carbohydrates on circulating glycoproteins sometimes serve as markers for rapid clearance of the glycoproteins. The mannose receptor on SEC, which recognizes terminal mannose residues on macromolecules, is particularly important in the regulation of serum glycoprotein homeostasis. Proteomic analysis of mice that were genetically deficient in mannose receptor had elevated levels of multiple lysosomal hydrolases and additional proteins that are up-regulated during inflammation and wound healing, indicating hepatic mannose receptor function is important in the control and resolution of inflammation (Lee et al. 2002).
The scavenger receptor of SEC cells assists in the clearance of numerous physiological and foreign waste macromolecules from the blood, including polysaccharides and proteins released during turnover of the extracellular matrix, intracellular macromolecules, modified serum proteins, and bacterial and fungal proteins (Smedsrod, Pertoft, et al. 1990).
An increased level of circulating hyaluronan may result from increased connective tissue synthesis/destruction or impaired hepatic capacity for waste molecule removal, as occurs in hepatic cirrhosis. The hyaluronan receptor shares functional properties with the scavenger receptor family (McCourt et al. 1999).
The Fc receptor on SEC has specificity similar to the Fc receptor on Kupffer cells. Both cell types are capable of removal of waste immunoglobulins from the circulation, though Kupffer cells have the major activity (Kuiper et al. 1994).
In addition to Fc receptor–mediated assimilation and disposal of immunoglobulin molecules, there is accumulating evidence that the liver is a major site of assimilation of cytokines. The liver has a large number of cells with cytokine receptors and a high density of cytokine receptors per cell, thus is regarded as a “cytokine sink” (Fey et al. 1994).
The endocytotic capacity of SEC is known to be decreased in older mice (Ito et al. 2007). SEC dysfunction was seen as early as fourteen months, when there was a threefold increase in swollen SEC on transmission electron microscopy (TEM. Aging-associated reduction in the endocytic capability of SEC is particularly apparent in pericentral regions of lobule (Le Couteur et al. 2008).
Generalized Aging Changes in Hepatic Sinusoids
The hepatic sinusoid is comprised of fenestrated endothelial cells, phagocytic Kupffer cells, extraluminal stellate cells, and pit cells, the latter being liver-adapted NK cells that are attached to the luminal surface of endothelial cells (Winnock et al. 1993). This specialized endothelium is fenestrated and generally lacks a basal lamina, thus solutes and small particles have direct access to the peri-sinuoidal space that contains processes of stellate cells and microvilli of hepatocytes (McCuskey 2008). The fenestrations are true discontinuities in the endothelium, having neither a diaphragm nor an underlying basal lamina. Approximately 5-10% of the surface of the SEC is perforated by fenestrations (Le Couteur et al. 2008). The fenestrations tend to be grouped into clusters known as “sieve plates.”
Sinusoidal changes associated with aging include thickening and defenestration of SEC, deposition of collagen and basal lamina in the perisinsoidal space, and increased numbers of fat engorged, nonactivated stellate cells (Le Couteur et al. 2008). These three aging-related hepatic sinusoidal changes are collectively known as pseudocapillarization. In addition, SECs have up-regulation of von Willebrand factor and ICAM-1 as well as reduced expression of caveolin-1.
Aging of the liver is associated with multiple clinically significant changes that include impaired metabolism of drugs, adverse drug interactions, and increased susceptibility to toxins, therefore the effects of aging on hepatic sinusoids have been studied extensively. In a study of hepatic blood flow in 0.8-, 3-, 14-, and 27-month-old C57Bl/6 mice there was a 14% reduction in perfused sinusoids between 0.8 and 27 months, with associated 35% reduction in sinusoidal blood flow (Ito et al. 2007). This was accompanied by a fivefold increase in leukocyte adhesion, up-regulated ICAM-1 expression, and increases in intrahepatic macrophages in 27-month-old mice. During the interval between 0.8 and 27 months the sinusoidal diameter decreased 6-10%. Altered SEC ultrastructure, consisting primarily of cellular swelling, was seen as early as 14 months. The combination of leukocyte accumulation in sinusoids, narrowing of sinusoidal lumens due to pseudocapillarization, and structural alterations in SEC were interpreted as the cause of the reduced sinusoidal blood flow in the liver of aged mice.
Another study compared livers of young (three to four months) to aged (twenty to twenty-four months) mice via transmission electron microscopy and immunohistochemistry (IHC) (Warren et al. 2005). Thickness of sinusoidal endothelium of young vs aged mice was 154 ± 4 nm vs 244 ± 8 nm, respectively. There was reduction in fenestrations in old mice (porosity 4.1 ± 0.3% in young mice vs 2.2 ± 0.2% in old mice. Aged mice had perisinusoidal staining with Sirius red, indicating collagen deposition. Fat-engorged perisinusoidal stellate cells were observed extensively in old mice.
Similar pseudocapillarization has been reported in the liver of baboons (Cogger et al. 2003). In the liver of aged (21.8 ± 0.7 yrs) vs young (5.4 ± 0.5 yrs) baboons, the thickness of sinusoidal endothelium was 130 ± 8 nm vs 186 ± 9 nm, respectively. Frequency of endothelial fenestrae decreased with aging, with porosity declining from 4.2 ± 0.5% to 2.4 ± 0.4%. Expression of laminin and von Willebrands factor was more extensive in aged baboons. Perisinusoidal ring-shaped cells, consistent with fat-engorged stellate cells, were prominent in aged baboons.
The process of pseudocapillarization is thought to have far-reaching implications for hepatic function, including an impact on dyslipidemia. Caloric restriction in F344 rats has been shown to reduce the level of pseudocapillarization of hepatic sinusoids (Jamieson et al. 2007).
Induction of Tolerance to Ingested and Self-antigens
During induction of cell-mediated immunity, professional, marrow-derived antigen-presenting cells (APC) typically present antigens to CD8+ T cells in association with MHC molecules. Liver SEC cells can take up antigen, process the antigen, and present it to T cells, but, probably due to the lack of input from helper T cells, the end result is tolerance rather than immunity (Limmer et al. 2000; Rubinstein, Roska, and Lipsky 1986, 1987). This major function of sinusoidal endothelial cells prevents immunologic reaction to the wide spectrum of potentially antigenic molecules that are assimilated from the gastrointestinal tract.
Hepatocytes may also function as antigen-presenting cells. Extension of hepatocellular microvilli through intercellular junctions between SEC allows direct contact between hepatocytes and naïve intra-sinusoidal CD8+ T cells. However, T cell activation by hepatocytes leads to premature T cell death or tolerance rather than the formation of fully competent CTL. Apoptosis of hepatocyte-activated T cells is suspected to be an example of death by neglect resulting from absence of an effective co-stimulatory signal (Bertolino et al. 1995, 1999; Bertolino, Trescol-Biemont, and Rabourdin-Combe 1998).
Autophagy Involvement in Immune Functions of the Liver
Autophagy is the only known mechanism that eukaryotic cells possess to dispose of intracellular organelles and protein aggregates that are too large to be degraded by the proteasome (Levine, Mizushima, and Virgin 2011). Autophagy has been implicated in both innate and acquired immunity through sampling, digestion, and presentation of peptides from invasive cellular pathogens and also from their own cellular milieu (Levine and Deretic 2007). The autophagy pathway or autophagy proteins have a central role in controlling diverse aspects of immunity in eukaryotic organisms.
Three main types of autophagy are described in mammalian cells. (1) Macroautophagy degrades soluble proteins, organelles, and protein aggregates after they are sequestered into a membrane-bound vesicle that subsequently fuses with the enzyme-laden lysosome, resulting in proteolysis. (2) Microautophagy degrades proteins and organelles that are incorporated into small vesicles formed from invaginations of the lysosomal membrane. (3) Chaperone-mediated autophagy (CMA) results in degradation of specific soluble proteins that are guided to the lysosome by attached chaperone proteins and then transported across the lysosomal membrane by a specific transport system. The diverse steps in macroautophagy are orchestrated by a group of proteins that are generically known as Atg (autophagy-related) proteins, which are conserved throughout the phylogenetic scale. Among these Atgs, two major cytosolic kinase complexes, beclin-1/phosphatidylinositol kinase type III complex and mammalian target of rapamycin (mTOR) kinase and its auxiliary proteins, regulate the initiation of macroautophagy (Bejarano and Cuervo 2010). Microtubule-associated protein 1 light chain 3 (LC-3), a homologue of yeast Atg 8, serves as a marker for macroautophagy activity (Holt et al. 2011; Yang and Klionsky 2010).
Autophagy participates in nearly all aspects of immunity, affecting both innate and adaptive immune processes. Three principal types of involvement in immune functioning are (1) specialized autophagy immune processes that are performed by autophagic machinery at the cellular level (these include recognition, capture, and elimination of intracellular pathogens via a process termed xenophagy), (2) generic autophagy’s role in cellular homeostasis of immune cells, and (3) non-autophagic role of Atg factors (Deretic 2011). Macroautophagy has been shown to contribute to the presentation of endogenous peptides on both MHC class II (Dengjel et al. 2005) and MHC class I molecules (English et al. 2009).
There is experimental evidence of an age-related decline in autophagy activity in the liver. LC3 expression was strongly reduced in the liver of twelve- and twenty-four–month-old mice as compared with young mice (Uddin et al. 2011). Reduction in autophagic activity in the liver may result in a reduction in the presentation of self and ingested antigenic peptides on hepatocellular villi, thus reducing the ability to activate and subsequently kill potentially self-reactive T cells within sinusoids.
Summation
The liver is the primary hematopoietic organ of the mammalian fetus and has a major role in the development of the immune system. The adult liver not only retains its ability to act as a hematopoietic organ but also gains new immune functions that may shift in importance with age. Age-related changes in sinusoids (pseudocapillarization), autophagy, and functions of various hepatic cell populations result in substantial alterations in many immunologically important hepatic functions. Hepatic alterations related to xenobiotic administration have the potential to disrupt normal immune functions of the liver, and these chemically mediated hepatic alterations may be manifested as systemic alterations in immune functioning. Given the common occurrence of xenobiotic-related hepatic alterations, this potential pathway of immunomodulation warrants particular attention in safety assessment studies.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article. The author(s) received no financial support for the research and/or authorship of this article.
