Abstract
Daprodustat (GSK1278863) is a hypoxia-inducible factor (HIF)-prolyl hydroxylase (PHD) inhibitor in development for treatment of anemia of chronic kidney disease. Daprodustat’s biological activity simulates components of the natural response to hypoxia; inhibition of PHDs results in HIF stabilization and modulation of HIF-controlled gene products, including erythropoietin. The carcinogenic potential of daprodustat was evaluated in 2-year carcinogenicity studies in Sprague-Dawley rats and CD-1 mice, where once-daily doses were administered. The mouse study also included evaluation of daprodustat’s 3 major circulating human metabolites. There were no neoplastic findings that were considered treatment related in either study. Exaggerated pharmacology resulted in significantly increased red cell mass and subsequent multiorgan congestion and secondary non-neoplastic effects in both species, similar to those observed in chronic toxicity studies. In rats, these included aortic thrombosis and an exacerbation of spontaneous rodent cardiomyopathy, which contributed to a statistically significant decrease in survival in high-dose males (group terminated in week 94). Survival was not impacted in mice at any dose. Systemic exposures (area under the plasma concentration–time curve) to daprodustat at the high doses in rats and mice exceed predicted maximal human clinical exposure by ≥143-fold. These results suggest that daprodustat and metabolites do not pose a carcinogenic risk at clinical doses.
Introduction
Daprodustat (GSK1278863), 2-(1,3-dicyclohexyl-6-hydroxy-2,4-dioxo-1,2,3,4-tetrahydropyrimidine-5-carboxamido) acetic acid, is a novel oral small molecule in development for the treatment of anemia of chronic kidney disease (CKD). Daprodustat stimulates erythropoiesis through potent inhibition of hypoxia-inducible factor (HIF)-prolyl hydroxylase enzymes (PHDs: PHD1, PHD2, and PHD3). 1 This biological activity simulates components of the natural response to hypoxia; inhibition of PHDs results in HIF stabilization and modulation of HIF-controlled gene products, including erythropoietin (EPO). Daprodustat has demonstrated the ability to correct anemia in completed global phase 2 clinical trials and phase 3 clinical trials conducted in Japan and is currently being assessed in global phase 3 studies, utilizing both once-daily and 3-times weekly dosing paradigms.
Since being discovered as the hypoxia-induced complex responsible for regulation of EPO gene expression, HIF has been shown to recognize hypoxia response elements in many diverse genes. 2 -6 The HIF family includes 3 hypoxia responsive proteins: HIF1α, HIF2α, and HIF3α. 7 -9 The HIFα proteins form a heterodimer with HIFβ, resulting in a transcriptionally active DNA-binding complex. 10 There is a large diversity of genes regulated by HIFα, with many exhibiting cell type and/or environment specificity in gene regulation. 5,11 -14
During normoxia, hydroxylated proline residues in HIFα promote interaction with the von Hippel-Lindau (VHL) tumor suppressor protein/E3 ubiquitin ligase, resulting in continual ubiquitination and subsequent proteasomal degradation of HIFα. 15,16 Hydroxylation of these conserved proline residues in HIFα is mediated by a family of oxygen-sensing HIF-PHDs. Hydroxylated proline residues on HIFα provide the essential recognition element for the HIFα–VHL interaction. 17 -19 The PHDs are members of the iron and α-ketoglutarate-dependent dioxygenase superfamily and are found in overlapping but distinct tissue expression patterns with differences in subcellular localization. 20
Since inhibition of PHDs results in HIFα stabilization and modulation of HIF-controlled gene products (including EPO), PHD inhibition represents a promising therapeutic approach for inducing the production of physiologic levels of endogenous EPO and alleviating anemia. 21
While the potential benefits of PHD inhibition for treatment of anemia in patients with CKD are under evaluation, some investigators have suggested that this mechanism of action could also potentially increase the risk of carcinogenesis and/or contribute to tumor progression due to upregulation of HIF/hypoxia-regulated gene products that have activities associated with cancer biology, such as vascular endothelial growth factor (VEGF). This view is based on findings in experimental xenograft models suggesting a role of HIF in tumor progression, and clinical data associating increased HIF with angiogenesis, metastasis, and poor prognosis. 22 Discussions regarding the potential benefit–risk profile of therapies that increase HIF foster scientific debate, especially given the complexity of HIF-regulated pathways as well as experimental evidence demonstrating anticancer effects of HIF stabilization in certain contexts. 23
Daprodustat is extensively metabolized in humans mainly to products of di- and trioxygenation. Additional minor oxidative products have been detected in human plasma. All notable human metabolites are also detected in rabbit and monkey plasma, but not in mouse, rat, or dog plasma following administration of daprodustat. Based on clinical data in anemic patients with CKD, 3 of these human circulating metabolites, GSK2391220 (M2), GSK2506104 (M3), and GSK2531401 (M13), are considered major, as circulating levels of each exceed 10% of total drug-related exposure. 24 Structures of daprodustat and its human metabolites and the proposed metabolism scheme have been previously published. 25 In comparison to daprodustat, the major human metabolites exhibit similar inhibitory potency against the PHDs and similar selectivity against closely related enzymes, collagen prolyl hydroxylase (CP4H) and factor-inhibiting HIF (FIH). Despite differing metabolite profiles among the species studied in repeat-dose toxicity studies (mice, rats, dogs, and monkeys), toxicologic profiles have been similar across species and consistent with expected and/or exaggerated pharmacology, suggesting that the metabolites do not contribute effects distinct from those evoked by daprodustat.
Evidence from completed genotoxicity studies of daprodustat and the 3 major circulating human metabolites (M2, M3, and M13), including Ames tests and other assays, indicates that these chemical entities do not represent genotoxic hazards to humans (data not presented). However, since these 3 major human circulating metabolites are not produced by rodents following administration of daprodustat, they were directly administered (in a cocktail formulation via once-daily subcutaneous injection) in combination with daprodustat (once daily via oral gavage) in the mouse carcinogenicity study as agreed with Food and Drug Administration (FDA) Carcinogenicity Assessment Committee (CAC) via special protocol assessment.
Recently, Beck et al 26,27 reported on the absence of carcinogenic effects of the PHD inhibitors FG-2216 and roxadustat (FG-4592) in 2-year rat and mouse oral carcinogenicity studies. In these studies, FG-2216 and roxadustat were dosed 3 times weekly to mimic the clinical dosing regimens for these agents. The objective of the work presented herein was to assess the carcinogenic potential of daprodustat in support of the nonclinical development program to support marketing authorization. The carcinogenicity studies of daprodustat were conducted using once-daily dosing as the most conservative approach for supporting both the 3 times weekly and once-daily clinical dosing regimens that are being assessed in phase 3 studies.
Materials and Methods
Study Conduct
All portions of studies were conducted at Charles River Laboratories Montreal ULC (Sherbrooke and Senneville sites) with the following exceptions: toxicokinetic sample bioanalysis (PPD, Middleton, Wisconsin), toxicokinetic data analysis (mouse study: GlaxoSmithKline, King of Prussia, Pennsylvania; rat study: Covance Laboratories Inc, Madison, Wisconsin), and transcriptomic analyses (GlaxoSmithKline).
Studies were designed in accordance with International Conference on Harmonization guidelines, to fulfill the requirements of regulatory authorities, for the toxicity testing of new drugs and were generally conducted in accordance with the OECD Principles of Good Laboratory Practice and as accepted by Regulatory Authorities throughout the European Union, USA, and Japan.
Animals
Sprague-Dawley rats, Crl: CD(SD), and CD1 mice, Crl: CD1 (ICR), were obtained from Charles River Laboratories (Quebec, Canada). Prior to randomization into study groups, animals were evaluated for suitability based on detailed clinical examinations, body weights, and ophthalmoscopy evaluations. Animals were acclimated for 15 to 20 days preceding dosing initiation. At onset of studies, rats were approximately 7 weeks old (males weighed between 219 and 307 g and females weighed between 170 and 231 g), and mice were approximately 6 weeks old (males weighed 27.9-40.5 g and females weighed 20.7-30.9 g). Animals were group housed (up to 3 animals of the same sex and same dosing group together) in solid bottom cages with appropriate bedding and equipped with an automatic watering valve. Animals were offered water and PMI Nutrition International Certified Rodent Chow No. 5CR4 (14% protein) ad libitum throughout the duration of the studies except during designated study procedures. Environmental controls were set to maintain temperature within the range 19 to 25°C and relative humidity within the range 30% to 70%, with an approximate 12-hour light/12-hour dark cycle, except during designated procedures. Animals were socially housed for psychological/environmental enrichment and were provided with items such as a retreat, a chewing object, and nesting material.
These studies were conducted in accordance with the Guide for the Care and Use of Laboratory Animals 28 and the GlaxoSmithKline Policy on the Care, Welfare and Treatment of Laboratory Animals, and were reviewed by the Institutional Animal Care and Use Committee ethical review process at the institutions where the work was performed.
Test Articles
Daprodustat (GSK1278863) was manufactured and supplied by GSK and was kept at 21°C, protected from light and moisture. Suspensions at concentrations of 0.01, 0.08, 0.4, and 0.7 mg/mL in 1% (wt/vol) methylcellulose (400 cP at 2%), 0.2% Tween 80 (vol/vol), and 5 mM NaOH in Ultra-Pure Water, pH 6 (±0.2) were used for the rat study. Suspensions at 0.02, 0.08, and 0.3 mg/mL in this vehicle were used for the mouse study. Formulations were prepared at least weekly and stored at 4°C.
For the mouse study, M2, M3, and M13 were manufactured by Wuxi Apptec (Shanghai, China), supplied by GSK Global API Chemistry, and were stored in a refrigerator set to maintain 4°C, protected from light and moisture. A metabolite cocktail solution containing 2.0 mg/mL M2, 2.5 mg/mL M3, and 1.3 mg/mL M13 (all concentrations as active moieties) in aqueous 25 mM phosphate buffer, pH 7.4 (±0.2), was prepared at least once weekly and stored at 4°C.
Study Designs
High doses of daprodustat were selected based on evidence of pharmacologic effects (statistically significant increases in hematocrit [Hct]) and were considered to represent the maximum tolerated doses (MTDs) for 2 years of administration, based on morbidity/mortality from exaggerated pharmacologic effects at higher doses in previous chronic or subchronic oral toxicity studies. Doses for both studies, and route of administration of metabolites for the mouse study, were agreed upon via special protocol assessment by FDA CAC.
For rats, high doses of 4 mg/kg/d and 7 mg/kg/d were selected for males and females, respectively, and were associated with clear pharmacodynamic effects (statistically significant increase in mean Hct) in prior studies. In a 26-week study, male rats did not tolerate 10 mg/kg/d, and while this dose was tolerated by females, the marked Hct increases (1.23X to 1.43X control) observed were considered unsustainable for more prolonged administration based on mortality at comparable Hct values in males. Hence, daprodustat was given once daily to rats (70/sex/group) by oral gavage at 0, 0.02, 0.1, 0.8, or 4 mg/kg/d to males for at least 90 weeks and at 0, 0.02, 0.1, 0.8, or 7 mg/kg/d to females for 99 weeks. Satellite animals were added at each dose level for toxicokinetics (weeks 4 and 26) or transcriptomics (week 26). The dosing volume was 10 mL/kg for all groups and adjusted for weight, with exception of the 0.02 mg/kg/day group (2 mL/kg dose volume of 0.01 mg/mL formulation was administered; 0.01 mg/mL is the lowest concentration that demonstrated reproducibly acceptable formulation stability).
For mice, a high dose of 3 mg/kg/d was selected based on adverse ischemia-related histopathology findings and/or markedly increased Hct at ≥6 mg/kg/d in 13-week studies; these included a study of once-daily daprodustat given alone for 13 weeks, and a study of once-daily daprodustat plus once-daily subcutaneous injection of a cocktail formulation containing the 3 major human circulating metabolites. Doses of metabolites (2.0 mg/kg/d M2, 2.5 mg/kg/d M3, and 1.3 mg/kg/d M13) selected for administration to each daprodustat dose group were the same as those included in the 13-week study, since systemic exposures (area under the plasma concentration-time curve [AUC] ) achieved in that study were comparable to or slightly exceeded respective metabolite exposure in patients at the maximal human dose. In addition, an extra mid-dose daprodustat-only group (0.8 mg/kg/d) was included to help differentiate causality (parent vs metabolites) if potential tumor responses had been observed. The subcutaneous route was selected to allow evaluation of the 3 major human circulating metabolites (M2, M3, and M13), since these metabolites are not produced by rodents and have very low oral bioavailability (≤3%). The dose groups (60/sex/group) in mice were therefore 0, 0.2, 0.8, or 3 mg/kg/d once daily by oral gavage for 104 weeks. Given the added study design complexity of also administering the 3 major metabolites of daprodustat, a summary of the mouse study design is presented in Table 1. As soon as practical after receiving their oral dose, all 3 daprodustat dose groups received once-daily subcutaneous injection of a cocktail formulation containing the 3 metabolites or the metabolite vehicle (aqueous 25 mM phosphate buffer, pH 7.4 ±0.2) for the control group. An additional dose group received 0.8 mg/kg/d daprodustat by once-daily oral gavage and the metabolite vehicle by once-daily subcutaneous injection (no metabolites). Satellite animals were added at each dose level for toxicokinetics (weeks 4 and 26) or transcriptomics (week 26). The daily subcutaneous injections were rotated daily across 4 sites (left scapular, right scapular, left dorsal thoracic, and right dorsal thoracic regions). The oral dose volume was 10 mL/kg, and the subcutaneous injection dose volume was 1 mL/kg for all groups.
Summary of Mouse Carcinogenicity Study Design.

a Received once-daily oral gavage of daprodustat’s vehicle; 1% (wt/vol) methyl cellulose (400 cP at 2%), 0.2% Tween 80 (vol/vol), 5 mM NaOH in Ultra-Pure Water, pH 6.0 (± 0.2).
b Received once-daily subcutaneous injection of the metabolite cocktail vehicle; aqueous 25 mM phosphate buffer, pH 7.4 (± 0.2).
In Life
Viability checks were performed near the start and end of each working day. Necropsies were performed on animals that died or were euthanized during the dosing period. Once daily, main study animals were examined upon return to the home cage after dosing. Detailed examinations, including for palpable masses, were performed on all main study animals weekly starting during the last week of the prestudy period and on the day of necropsy. Main study and toxicokinetic study animals were weighed at randomization, on day 1, weekly for the first 16 weeks of dosing, then monthly for the remainder of the studies, and on the day of necropsy. Food consumption (per cage measurement) was quantitatively measured the last week preceding dosing initiation and monthly thereafter, during the dosing period.
Toxicokinetics
For toxicokinetic analysis, blood samples were collected from rats (jugular vein at approximately 0.5, 1, 2, 4, 8, and 24 hours after dosing) and mice (terminally via abdominal aorta after isoflurane anesthesia at approximately 0.083, 0.25, 0.5, 1, 2, 4, 8, and 24 hours after dosing) during weeks 4 and 26. Toxicokinetic bioanalysis for daprodustat has been previously described. 25 Briefly, plasma samples were separated and transferred to uniquely labeled clear polypropylene tubes containing citric acid (a ratio of 1:40 of 0.48 g/mL citric acid to the volume of plasma was maintained) and frozen immediately over solid carbon dioxide (dry ice) and then stored in a freezer set to maintain −20°C. Concentrations of daprodustat (rats and mice) and metabolites (mice) were determined using validated bioanalytical methods based on solid phase extraction followed by ultra-high pressure liquid chromatography tandem mass spectrometry. Toxicokinetic analysis was performed by noncompartmental pharmacokinetic analysis using WinNonlin, version 6.2 (rats) and version 6.3 (mice). All computations utilized the nominal sampling times. The systemic exposure to daprodustat and metabolites was determined by calculating the AUC from the start of dosing to the last quantifiable time point (AUC0-t) using a linear up/log down version of the linear-log trapezoidal method. Concentrations at 24 hours were used as predose values in calculating the AUC in weeks 4 and 26, and maximum observed peak plasma concentrations (Cmax) and the times observed (Tmax) were determined.
Ophthalmoscopy
The eyes of all animals were examined by a board-certified veterinary ophthalmologist by indirect ophthalmoscopy and biomicroscopic examinations prior to initiation of dosing (all animals) and again during weeks 13, 26 (rats only), 52, 78, 98 (rats only), and 104 (mice only) of the dosing period (main study surviving animals). Prior to examination, a mydriatic (rats: 1% tropicamide, mice: 0.5% atropine sulfate) was instilled into each eye.
Hematology
Blood was collected from the jugular vein of main study rats (first 10 surviving rats/sex/group) of groups 1 (0 mg/kg/d), 4 (0.8 mg/kg/d), and 5 (4/7 mg/kg/d for M/F) for hematology assessment at weeks 26, 39, and 52. Blood samples from mice were collected from the saphenous vein of main study animals (first 10/sex in group 1 [controls] and group 5 [3 mg/kg/d + metabolites]) into capillary tubes for Hct determination at week 26, 39, and 52.
Terminal Procedures
All main study animals, including found dead and animals euthanized in extremis, and toxicokinetic animals found dead or euthanized for humane reasons were subjected to a complete necropsy examination. Following detailed external and internal examinations, tissues listed in Table 2 were taken from each main study animal and preserved in 10% neutral-buffered formalin except as noted. Samples of these tissues, unless indicated otherwise, were processed to paraffin wax, sectioned, stained with hematoxylin and eosin, and examined microscopically by a board-certified veterinary pathologist with subsequent peer review.
Tissues Collected at Necropsy for Histopathologic Examination.

a Eyes and optic nerves were fixed in Davidson’s transferred to 70% alcohol, trimmed and processed.
b Collected from mice only.
c Also collected from n = 6/sex from toxicokinetic animals (rats: groups 1, 3, and 5; mice: groups 1, 2, 4, and 5) in week 26 for transcriptomics analysis for erythropoietin and vascular endothelial growth factor.
d Testes were fixed in Modified Davidson’s fluid, rinsed with water, transferred to 10% neutral buffered formalin, and then trimmed and processed.
e Collected from rats only.
Transcriptomics
Immediately following exsanguination of designated toxicokinetic mice and rats in week 26, kidney, liver, lung, spleen, and mesenteric lymph node were removed. Tissues were placed on a clean, nonporous surface that had been previously wiped with RNase AWAY (Molecular Bio Products, San Diego, CA) and then RNase-free water. Two to 4 representative portions of each tissue were collected and trimmed to size placed into aluminum foil and submerged into liquid nitrogen and once frozen transferred into dry ice and stored at −70°C to −80°C until shipment and further analysis as described subsequently.
Approximately 10 to 40 mg of each sample and an appropriate amount of RNA lysis buffer containing β-mercaptoethanol were added to each sample for homogenization. For mouse samples, RNA was isolated using MagMAX mirVana Total RNA Isolation Kit (Applied Biosystems by Thermo Fisher Scientific, Foster City, CA). For rat samples, RNA was isolated using the Absolutely RNA Miniprep Kit (Agilent Technologies, Wilmington, DE). RNA was quantified using the NanoDrop UV Spectrophotometer (NanoDrop Products, Wilmington, DE). Approximately equal amounts of RNA from each tissue type were loaded into complementary DNA (cDNA) synthesis reactions, and cDNA was generated using the SuperScript VILO cDNA Synthesis Kit (Life Technologies, Carlsbad, CA).
Quantitative polymerase chain reaction (qPCR) primers and hydrolysis probes for GAPDH (reference gene for mouse analyses), 18S ribosomal RNA (rRNA; reference gene for rat analyses), and HIF-related genes (EPO and VEGF A) were purchased from Life Technologies. Quantitative PCR was performed using Taqman Fast Gene Expression Master mix (Life Technologies); data were collected using a 7900HT Fast Real-Time PCR System (Applied Biosystems by ThermoFisher Scientific, Foster City, CA). Files were uploaded into Expression Suite Software version 1.0.3 (Life Technologies), then the threshold cycle (CT) values were exported to Microsoft Excel, and the ΔΔCT method was used as described in Applied Biosystems User Bulletin 2 (Applied Biosystems, Foster City, California). Reference gene CT data (GAPDH and 18S rRNA) were evaluated to ensure low variability among samples (calculated using percentage of standard deviation). Data of EPO and VEGF for each tissue were analyzed separately. The ΔCT values were obtained by subtracting the applicable reference gene (GAPDH or 18S) from each gene of interest for each sample. Values of ΔΔCT were obtained by subtracting vehicle group average ΔCT value from each of the ΔCT values for each sample. Input values were generated by using the formula (2−(ΔΔCT)) for each sample for each gene (messenger RNA [mRNA]). Group average input values were calculated and then individual input values were calibrated (individual input values divided by average input) to the control group average input to obtain relative quantity (RQ) for each gene for each sample. A gene was considered as “changed” if the group average RQ value had an absolute fold change of ≥2 (noted as ≥2 or ≤0.5 when compared to control average; RQ set at 1).
Study Statistics
Numerical data of interest were submitted for statistical analysis per the following, where all inferential statistical tests were performed at the 5% significance level. Levene’s test was first used to check the homogeneity of group variances. Whenever the results of this test were not found to be significant, the overall group effect was assessed using an analysis of variance F-test; otherwise results were compared using a Kruskal-Wallis test. The pairwise comparisons between the reference item group and each of the test article–treated groups were conducted whenever the overall group effect was found to be significant. Depending on the retained approach, parametric or nonparametric, these comparisons were done via Dunnett’s or Dunn’s test, respectively.
The statistical analyses of rat and mouse main study mortality and tumor data were performed separately for each sex, via SAS (version 9.2 for the rat study, version 9.4 for the mouse study; SAS Institute Inc., Cary, NC). 29 For the mortality data, the survival function of each group was estimated using the Kaplan-Meier product-limit method applied on daily intervals, and graphs were produced. Any animal with accidental injury that caused its death or its unscheduled euthanasia or still alive at the day following the last day of the experimental period were censored in the estimation. Using dose-level scores, the significance of an overall dose-related trend in mortality and pairwise comparisons between the reference item group and each test article–treated group was evaluated via a Peto’s 2-sided test. 30 For male rats, doses were adjusted due to early termination of dosing. For mice, the overall trend test was performed across the reference item (group 1) and the 3 test article–treated groups that included the metabolites (groups 2, 4, and 5).
For tumor data, the statistical analysis was limited to subcutis and hemolymphoreticular tissue using all study animals, to all nonsecondary neoplastic lesions found in study plan-required tissues/sites and to the combination of hemangiosarcoma findings across whole body. Palpable neoplastic lesions found under study plan-required glands were statistically analyzed in a “mortality-independent” context according to Peto’s onset rate using all study animals. Whereas, nonpalpable neoplastic findings were statistically analyzed in a “mortality-dependent” context according to Peto’s prevalence and death rate methods. For each data set of interest, Peto’s test, using integer dose-level scores, was conducted to assess significance of tumor rate increase across all considered groups and in each test article–treated group when compared to the reference item group. For male rats, due to early termination of dosing in groups, doses were adjusted and used in 2 separate overall trend tests accordingly. For mice, the overall trend test was performed across the reference item (group 1) and the 3 test article–treated groups that included the metabolites (groups 2, 4, and 5). As per Lin and Rahman, 31 the discrete permutation distribution was used for each statistical test involving a data set with 10 or less tumor occurrences. Significance of statistical results was per FDA recommendation, 32 according to tumor prevalence classification (common or rare).
Results
Survival
Between 40 and 55 rats/sex/group died or were euthanized prior to study termination. Survival data are summarized in Table 3. Rat survival curves are depicted in Figure 1. A statistically significant decrease in survival was seen in males at 4 mg/kg/d when compared to controls (pairwise groups comparisons, P value = .0131). Because of poor survival in these high-dose males (number of remaining males had reached 15 on day 650), and poor survival in male and female control groups (number of survivors reached 20 on days 675 and 695, respectively), the study was terminated prior to target week 104. Terminal necropsies were initiated on day 653 (week 94) for 4 mg/kg/d males, day 679 (week 97) for all other male groups, and day 698 (week 100) for all female groups. Group/study terminations were communicated to, and endorsed by, FDA eCAC.
Survival Summary.
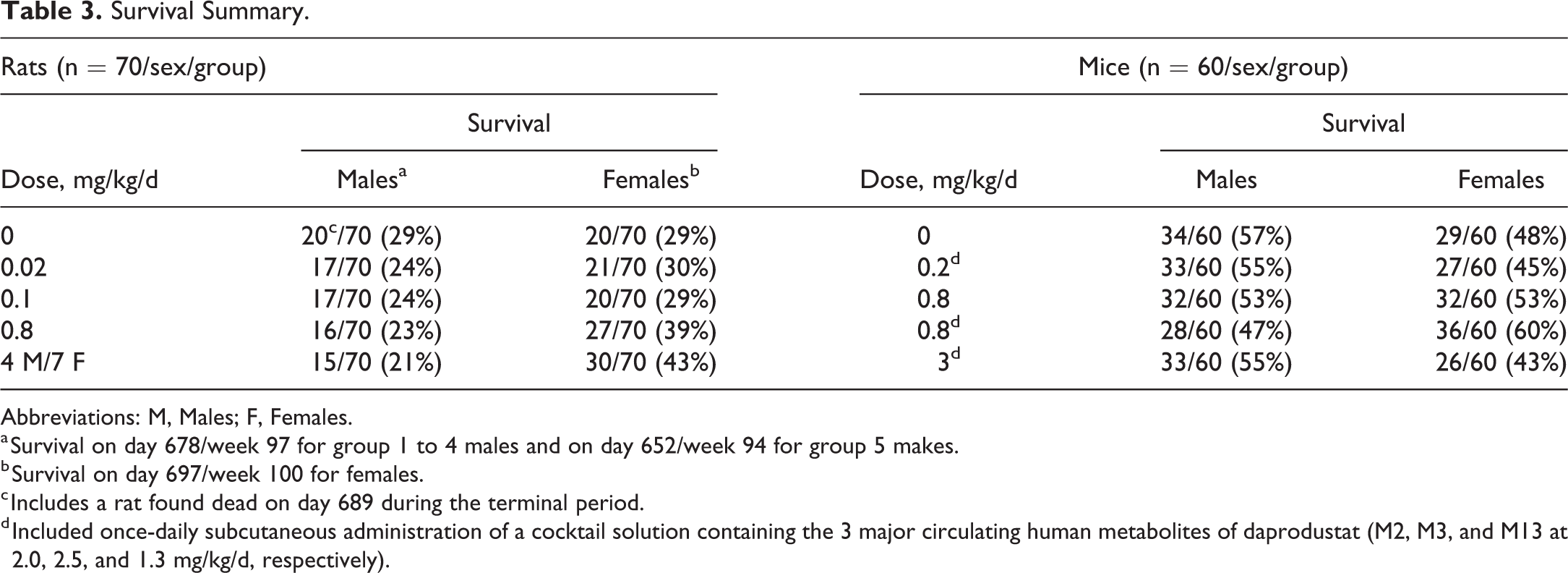
Abbreviations: M, Males; F, Females.
a Survival on day 678/week 97 for group 1 to 4 males and on day 652/week 94 for group 5 makes.
b Survival on day 697/week 100 for females.
c Includes a rat found dead on day 689 during the terminal period.
d Included once-daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively).
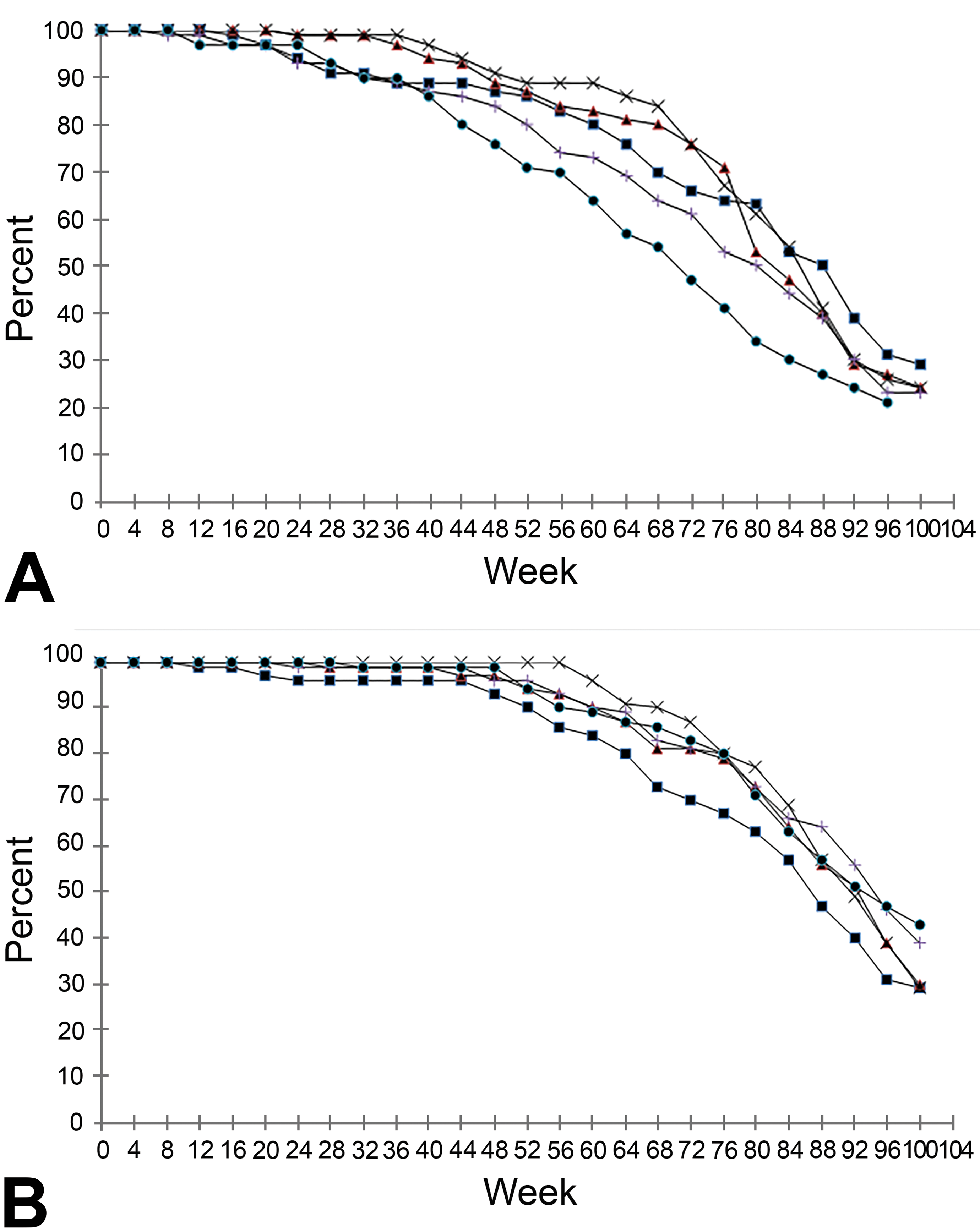
Survival data for (A) male and (B) female Sprague-Dawley rats administered vehicle control or daprodustat once daily by oral gavage during a 2-year carcinogenicity study. For males, end of study for groups 1 to 4 (day 678/week 97) and for group 5 (day 652/week 94). -▪- Vehicle control (0 mg/kg/d); -▴- 0.02 mg/kg/d daprodustat; -x- 0.1 mg/kg/d daprodustat; -+- 0.8 mg/kg/d daprodustat; -•- 4 (males) or 7 (females) mg/kg/d daprodustat.
For each preterminal decedent rat, the most probable cause of death was determined. Tumor-related causes of mortality are captured in Table 4. Cause of death could not be determined for a proportion of rats in all groups (males ranging from 9 to 21/group and females ranging from 1 to 3/group). Common tumors in aged Sprague-Dawley rats, pituitary and mammary gland tumors, were responsible for numerous mortalities. However, the total incidence of tumors contributing to death was comparable between daprodustat-treated rats and control rats. Commonly encountered background lesions in aged rats, progressive cardiomyopathy and atrial thrombosis, were the most common non-neoplastic causes of death in males given 4 mg/kg/d and likely contributed to the daprodustat-related and statistically significant increase in mortality in males at this dose.
Tumor-Related Causes of Mortality in Sprague-Dawley Rats.
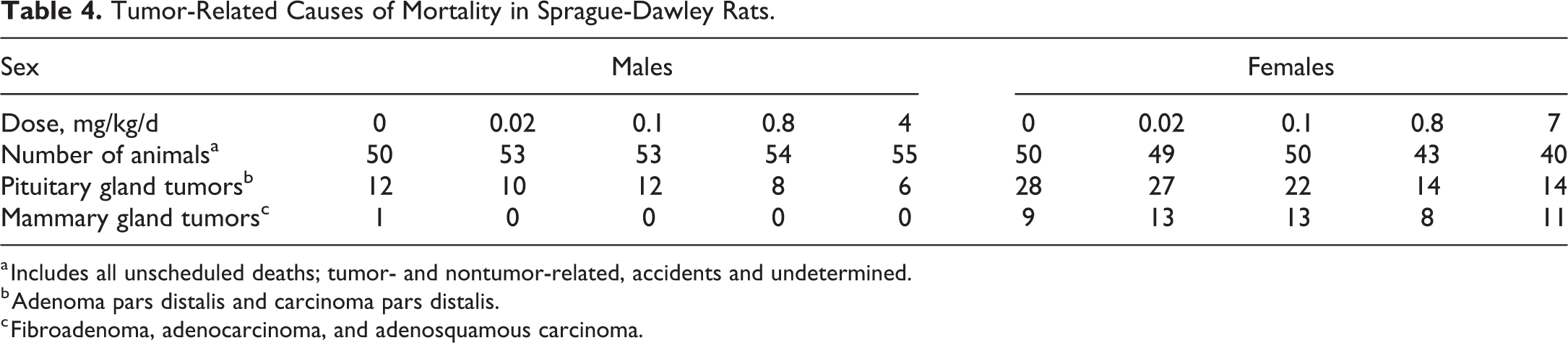
a Includes all unscheduled deaths; tumor- and nontumor-related, accidents and undetermined.
b Adenoma pars distalis and carcinoma pars distalis.
c Fibroadenoma, adenocarcinoma, and adenosquamous carcinoma.
Between 26 and 36 mice/sex/group survived until the end of the study. Survival data are summarized in Table 3. There were no test article–related effects on mortality in mice (mouse survival curves are depicted in Supplemental Figure 1). For each preterminal decedent mouse, the most probable cause of death or reason for moribundity was determined. Cause of death or early euthanasia could not be determined for a proportion of mice in all groups (1 to 4 mice/group for males and females). Tumor-related causes of mortality are captured in Table 5. There was no test article-related effect on the distribution of neoplastic lesions contributory to cause of death.
Tumor-Related Causes of Mortality in CD1 Mice.
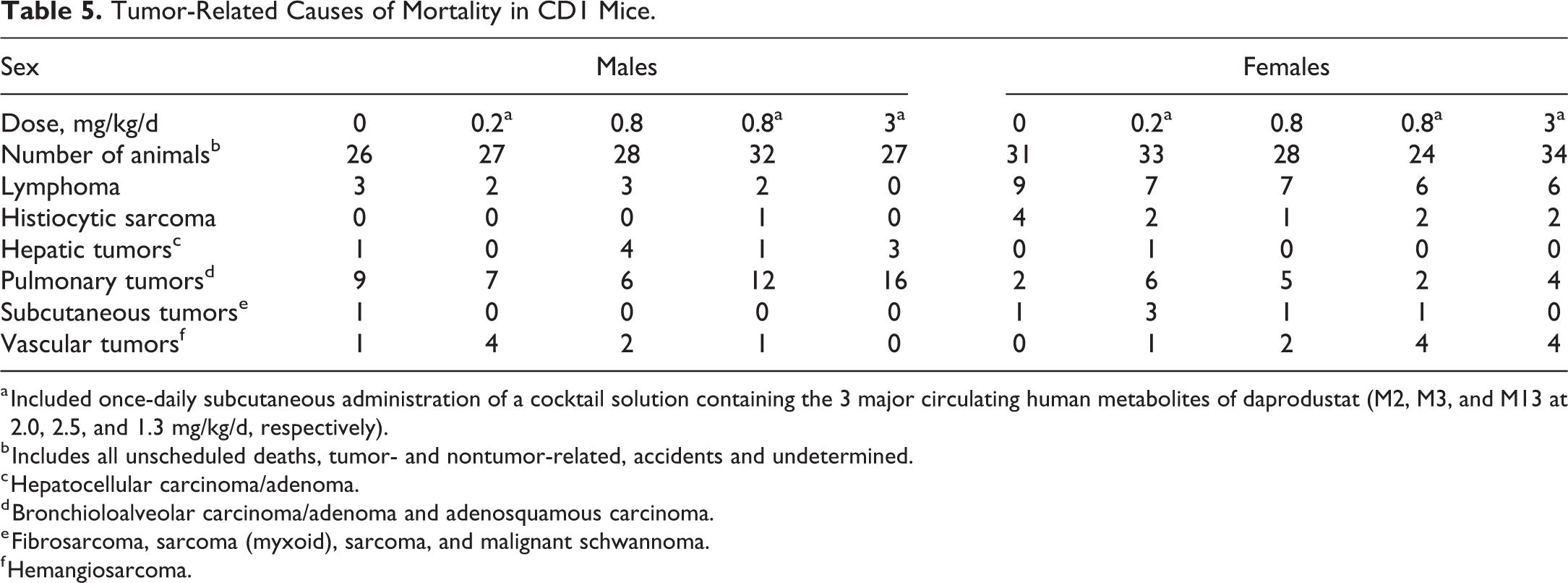
a Included once-daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively).
b Includes all unscheduled deaths, tumor- and nontumor-related, accidents and undetermined.
c Hepatocellular carcinoma/adenoma.
d Bronchioloalveolar carcinoma/adenoma and adenosquamous carcinoma.
e Fibrosarcoma, sarcoma (myxoid), sarcoma, and malignant schwannoma.
f Hemangiosarcoma.
In Life (Body Weight, Food Consumption, Ophthalmoscopy, and Hematology)
There were no test article–related effects on body weight, body weight gain, food consumption, or ophthalmoscopy in either study (data not presented). On all occasions assessed in rats (weeks 26, 39, and 52), pharmacologically mediated, daprodustat-related increases in red cell mass parameters (red blood cell count, hemoglobin, and Hct) were observed for male rats at 4 mg/kg/d (up to 1.35X control) and female rats at 7 mg/kg/d (up to 1.26X control). In female rats, increased red cell mass parameters were associated with increases in absolute reticulocyte counts (1.31X to 1.52X control) in weeks 26, 39, and 52.
Similarly, on all occasions assessed in mice (Weeks 26, 39, and 52), increases in Hct were observed in mice given 3 mg/kg/d + metabolites (up to 1.25X and 1.56X control values in males and females, respectively). Mean Hct values from each study are presented in Figures 2 (rats) and 3 (mice).
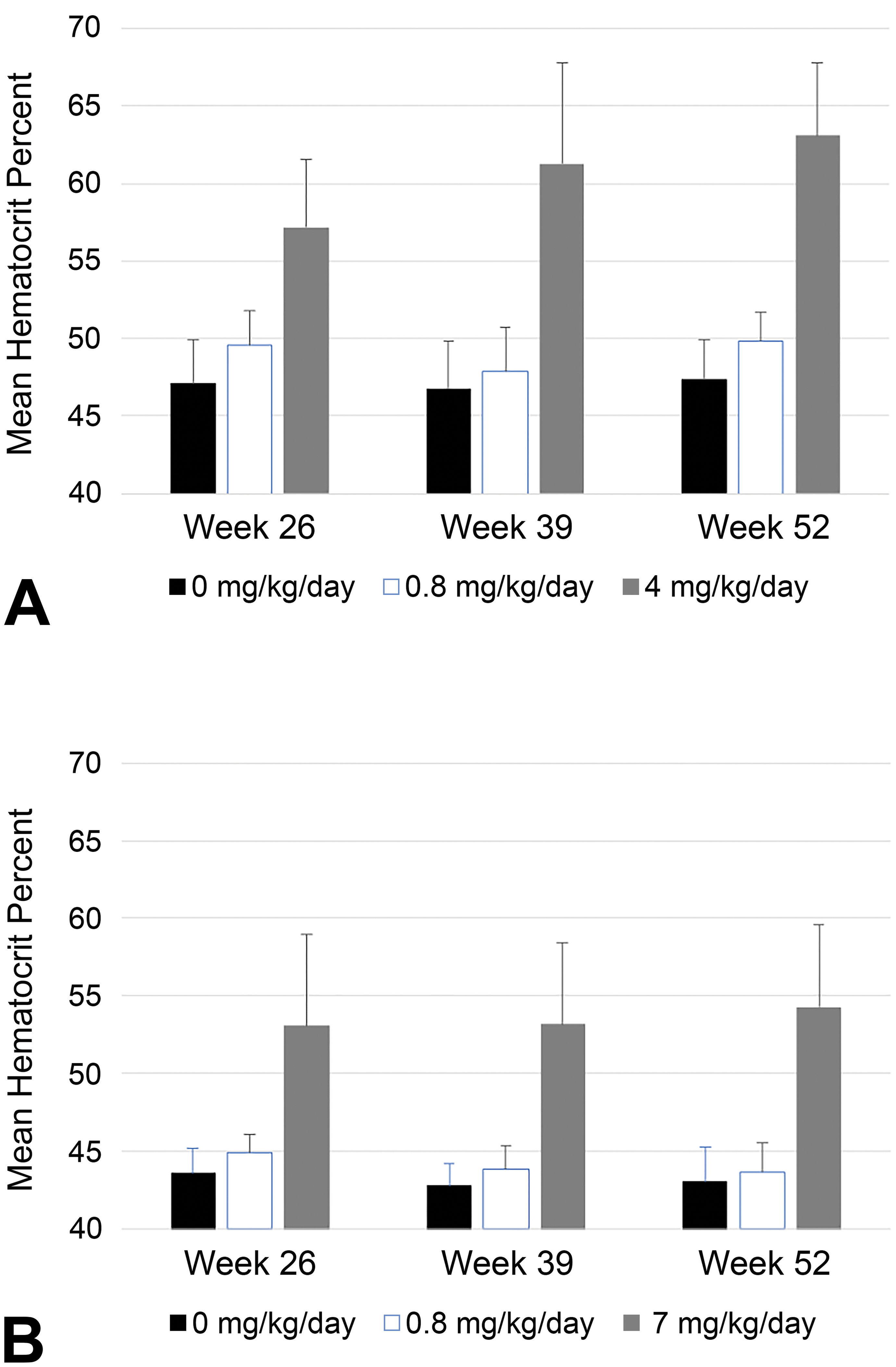
Hematocrit (Hct) data (mean + SD, n = 10/sex/group) from (A) male and (B) female Sprague-Dawley rats administered vehicle control or daprodustat once daily by oral gavage during a 2-year carcinogenicity study.
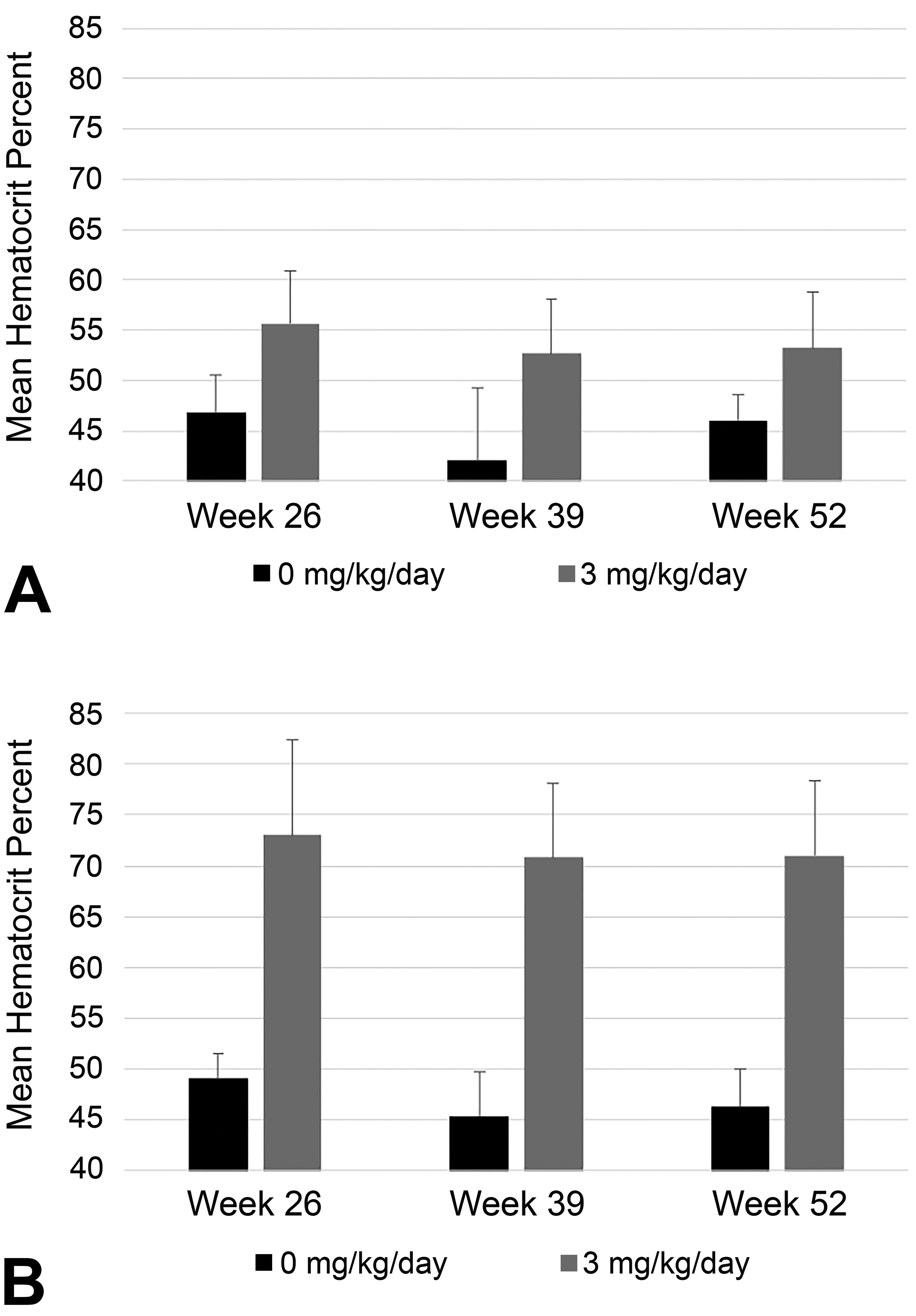
Hematocrit (Hct) data (mean + SD, n = 10/sex/group) from (A) male and (B) female CD1 mice administered vehicle control or 3 mg/kg/d daprodustat once daily by oral gavage along with once-daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively) during a 2-year carcinogenicity study.
Toxicokinetics
A summary of toxicokinetics of daprodustat in rats and mice are captured in Table 6. A summary of the toxicokinetics of the 3 major metabolites of daprodustat are captured in Supplemental Table 1.
Daprodustat Exposure From Rat and Mouse Carcinogenicity Studies.
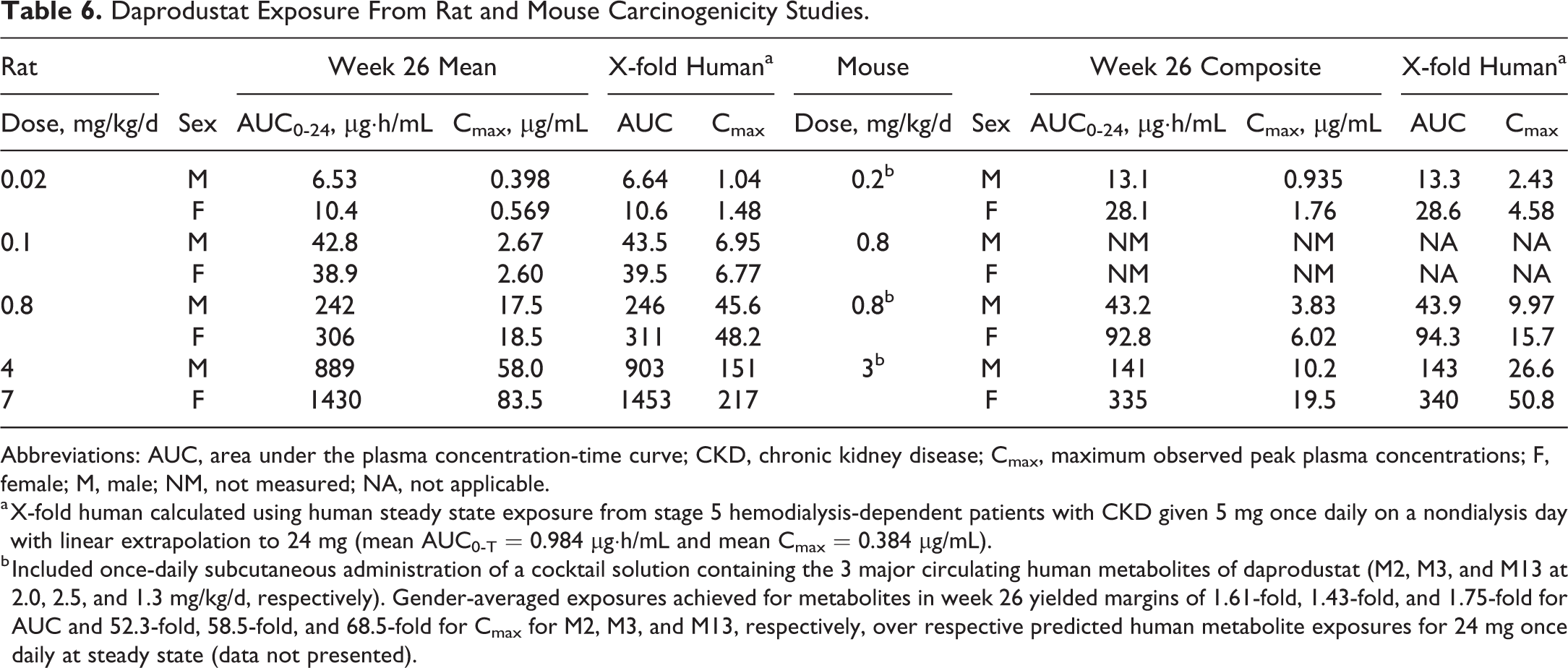
Abbreviations: AUC, area under the plasma concentration-time curve; CKD, chronic kidney disease; Cmax, maximum observed peak plasma concentrations; F, female; M, male; NM, not measured; NA, not applicable.
a X-fold human calculated using human steady state exposure from stage 5 hemodialysis-dependent patients with CKD given 5 mg once daily on a nondialysis day with linear extrapolation to 24 mg (mean AUC0-Τ = 0.984 µg·h/mL and mean Cmax = 0.384 µg/mL).
b Included once-daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively). Gender-averaged exposures achieved for metabolites in week 26 yielded margins of 1.61-fold, 1.43-fold, and 1.75-fold for AUC and 52.3-fold, 58.5-fold, and 68.5-fold for Cmax for M2, M3, and M13, respectively, over respective predicted human metabolite exposures for 24 mg once daily at steady state (data not presented).
In rats, daprodustat was quantifiable in plasma through the entire 24-hour sampling period. Values of Tmax ranged from 0.5 to 2 hours on weeks 4 and 26. There were no marked (>2-fold) differences in mean Cmax and AUC0-t values between males and females on weeks 4 and 26 for the dose levels of 0.02, 0.1, and 0.8 mg/kg/d. Systemic exposure (gender-averaged Cmax and AUC0-t) was generally dose proportional from 0.02 to 0.8 mg/kg/d, increasing from 32.3 to 37.2-fold for a 40-fold increase in dose. Increases in systemic exposure were also dose proportional from 0.8 to 4 mg/kg/d (males) or from 0.8 to 7 mg/kg/d (females), ranging from 3.3 to 4.6-fold for a 5-fold increase in dose and 4.5 to 6.0-fold for an 8.8-fold increase in dose, respectively. There were no marked differences in systemic exposure between weeks 4 and 26 at any dose.
Following oral administration of daprodustat to mice at doses of 0.2, 0.8, and 3 mg/kg/d with concomitant subcutaneous administration of metabolites M2, M3, and M13, daprodustat plasma concentrations were quantifiable during the entire 24-hour sampling period on weeks 4 and 26. The metabolite plasma concentrations were generally quantifiable up to 8 hours on weeks 4 and 26 with some exceptions for M13. Maximum plasma concentrations of daprodustat were observed between 0.25 and 8 hours on weeks 4 and 26. Maximum plasma concentrations of the metabolites were observed at 0.083 or 0.25 hours after dosing on weeks 4 and 26. Generally, there were no marked (>2-fold) differences in systemic exposure (defined as AUC0-t and Cmax) between males and females for daprodustat or the metabolites at any dose, with exception of daprodustat AUC0-t values on week 26 which were 2.1- to 2.4-fold higher in females. With dose increasing 15-fold from 0.2 to 3 mg/kg/d, systemic exposure to daprodustat increased proportionally; gender-averaged AUC0-t and Cmax values increased from 11.0 to 11.7-fold on weeks 4 and 26. With the same doses of metabolites given to mice administered 0.2, 0.8, and 3 mg/kg/d daprodustat, systemic exposure of metabolites was similar across the 3 groups. There were no marked differences in systemic exposure of daprodustat or metabolites from weeks 4 to 26 at any dose.
Transcriptomics
Expression of EPO was not observed in lung, mesenteric lymph node, or spleen in rats or mice. A slight dose-dependent decrease in EPO expression was observed in kidney samples from rats and mice. Group average EPO expression was increased in liver samples from rats given ≥4 mg/kg/d and in liver samples from mice given ≥0.2 mg/kg/d + metabolites. It should be noted that the average CT values for EPO expression in liver samples from rats and mice given vehicle were approximately 40, indicating marginal to no EPO mRNA expression. Fold-change calculations were performed using methods described in Thermo Fisher User Bulletin No. 2 (http://tools.thermofisher.com/content/sfs/manuals/cms_040980.pdf); however, since it is clear from the raw CT values that EPO expression in liver samples from vehicle-treated rats was marginal to absent, the conclusion to be drawn is simply that expression was increased in liver samples from rats and mice given high doses of daprodustat compared to liver samples from vehicle-treated animals. Relative EPO mRNA expression was calculated using respective tissues from vehicle-treated groups as comparators. Average ΔCT values (CT values normalized to a housekeeping/reference gene) may be used to infer estimated expression across tissue types. Comparison of normalized EPO mRNA expression (represented by ΔCT values, data not shown) suggests higher EPO mRNA expression in kidneys from vehicle-treated rats and mice when compared to EPO expression in liver samples from rats and mice given any dose of daprodustat. The maximal expression of EPO mRNA observed in livers from daprodustat-treated rats and mice compared to EPO mRNA expression in kidneys from vehicle-treated rats and mice was approximately 2- to 5-fold higher, respectively.
There were no test article–related changes in VEGF expression in any tissue evaluated in rats. An increase in mean VEGF expression was observed in lungs from mice that received 0.2 or 0.8 mg/kg/d + metabolites. However, as there was a high degree of variability within the data, and as no test article–related histopathologic changes were seen in lungs or any other tissue, the biologic significance of this result is questionable, especially as there was no apparent consequence to tumorigenesis. Additionally, VEGF mRNA expression was decreased in mesenteric lymph node from mice given ≥0.8 mg/kg/d + metabolites and in spleen from mice given 3 mg/kg/d + metabolites, indicating lack of a consistent response across the tissues evaluated. Summaries of group mean mRNA data are captured in Tables 7 (rats) and 8 (mice).
Rat Study: Summary Gene Expression Data.a

Abbreviations: CT, cycle threshold; EPO, erythropoietin; F, females; M, males; RQ, relative quantity; SD, standard deviation; VEGF, vascular endothelial growth factor.
a Expression among tissue types should not be compared.
b Individual input values were calibrated to the control group average input to obtain relative quantity (RQ) for each gene for each sample then the average input for each gene was calculated for each group.
c CT values were undetectable for most rats in this group. However, to generate relative fold changes, a value was assigned using the max CT value of 40.
d Since the control group was assigned a normalized average input value (or RQ) of “1”in the absence of expression, no true values can be assigned or referenced, the only certainty is that expression is induced and has been estimated using the information available.
Mouse Study: Summary Gene Expression Data.a
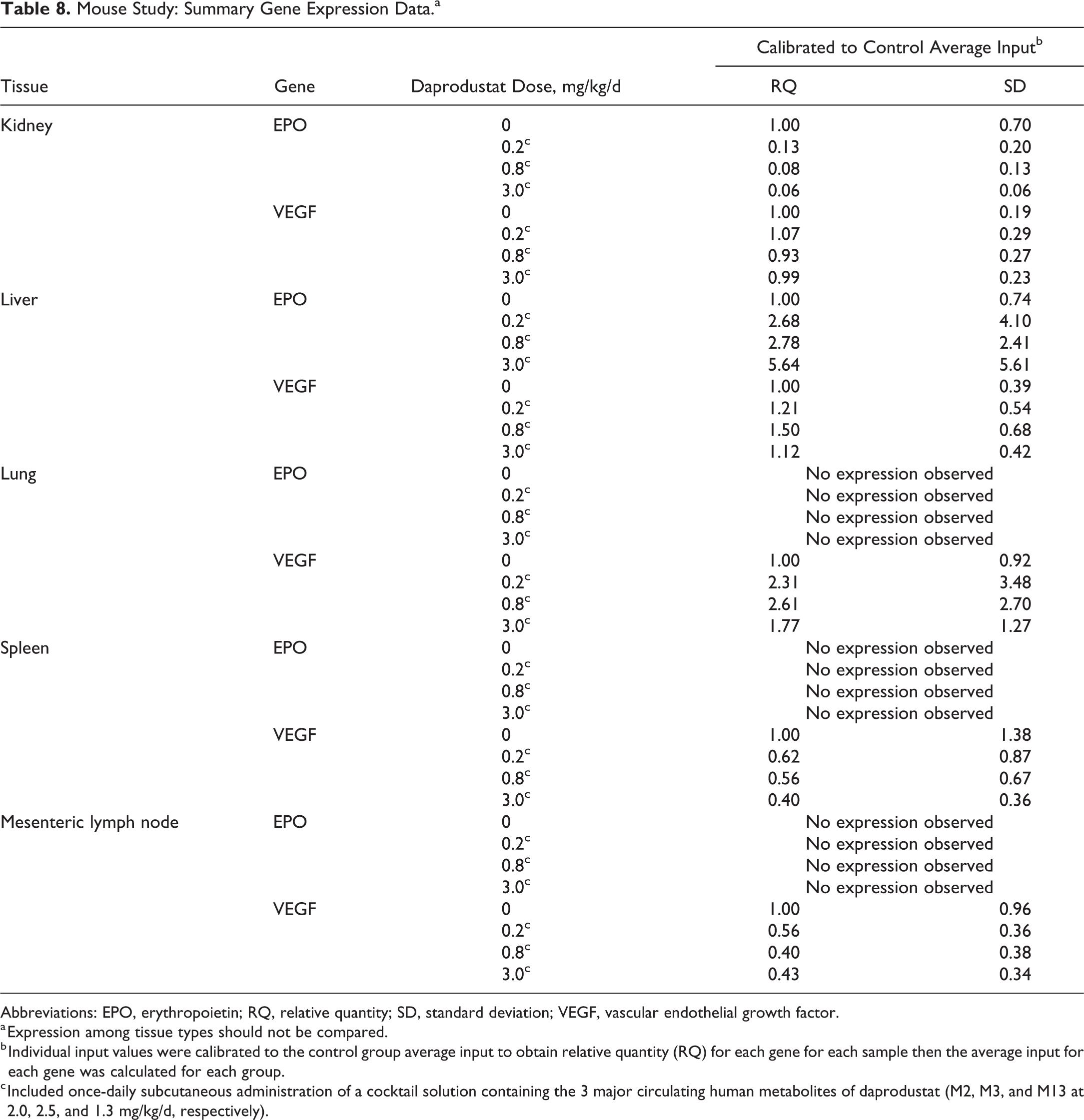
Abbreviations: EPO, erythropoietin; RQ, relative quantity; SD, standard deviation; VEGF, vascular endothelial growth factor.
a Expression among tissue types should not be compared.
b Individual input values were calibrated to the control group average input to obtain relative quantity (RQ) for each gene for each sample then the average input for each gene was calculated for each group.
c Included once-daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively).
Histopathology (Neoplastic Findings)
There were no treatment-related neoplastic findings in either study. Additionally, there were no treatment-related preneoplastic findings, including increased hyperplasia or metaplasia, in either study. Neoplastic histopathology findings from each study are summarized below by species.
Histopathology (Neoplastic Findings)—Rat Study
Neoplastic lesions observed were generally typical of those commonly encountered in Sprague-Dawley rats of this age and were not considered daprodustat related. In the mammary gland, there was a slight apparent increase of benign fibroadenomas in females given 7 mg/kg/d (54/70; incidence of 77.1%) and an increased incidence of the same tumor in males given 4 mg/kg/d (8/63; incidence of 12.7%; Table 9). Statistical analyses revealed an overall trend in the increase of fibroadenomas across dose groups only in males (P value <.0001) and a statistically significant increased incidence in males at 4 mg/kg/d when compared to control males (pairwise groups comparisons, P value = .0021). This tumor has been reported as the most common mammary tumor in aged female Sprague-Dawley rats as evidenced by high incidences in the literature historical control data ranging from 32.3% to 72.2% 33 or 30.8% to 68.3% at the test facility (Charles River Montreal; 2010-2012, 730 controls/sex in 12 studies total). In males, when the mammary gland can be evaluated, this tumor is observed less frequently but has been reported as high as 7.1% and 11.1% in the literature historical control data 33,34 and up to 3.3% at the test facility. In this study, the increase in mammary gland fibroadenomas was not considered an effect of daprodustat administration in either sex. In females, the increased incidence was only marginally outside historical or published control ranges, was not dose responsive, and was not associated with increases in other benign or malignant mammary tumors. Further, there was a greater proportion of females at 7 mg/kg/d that survived until week 100 (ie, greater proportion of aged females) compared to the control females (30 at 7 mg/kg/d vs 20 controls). Mammary fibroadenomas increase in incidence with age. In males, the increased incidence (12.7%) of fibroadenoma was also limited to the high dose and only slightly exceeded upper ranges reported in historical literature data (up to 11.1%). 34 Aside from fibroadenomas, only a single mammary adenoma was noted in the male high-dose group, with no other mammary tumor types, resulting in a total mammary tumor burden (excluding lymphoreticular tumors) in males of 9 (14.3%) of 63 in the 4 mg/kg/d group versus 4 (6.2%) of 65 in the controls (Table 9). Additionally, there were 3 mammary hyperplastic lesions found in control males and none in high-dose males. Thus, when combining mammary gland hyperplasia with fibroadenomas and adenomas, the incidences are comparable (7/63 or 11.1% incidence of mammary lesions in male controls vs 9/63 or 14.3% incidence in males given 4 mg/kg/day). Mammary fibroadenomas in male rats are generally considered to have a hormonal component to their pathogenesis, 35 but there is no evidence that daprodustat causes endocrine effects in either gender, as there were no non-neoplastic effects on the reproductive organs nor were there any treatment-related neoplastic or preneoplastic effects on the pituitary gland of either sex. Additionally, there were no increases in any mammary tumors in the accompanying mouse study (see below). Considering the lack of any significant effect in female rats and the weight of evidence, the investigators considered this small increase in male mammary fibroadenomas at this single dose to most likely represent a spurious and spontaneous change rather than a drug effect.
Mammary Gland and Pituitary Gland Tumors in Sprague-Dawley Rats—Incidence (Percent Incidence).
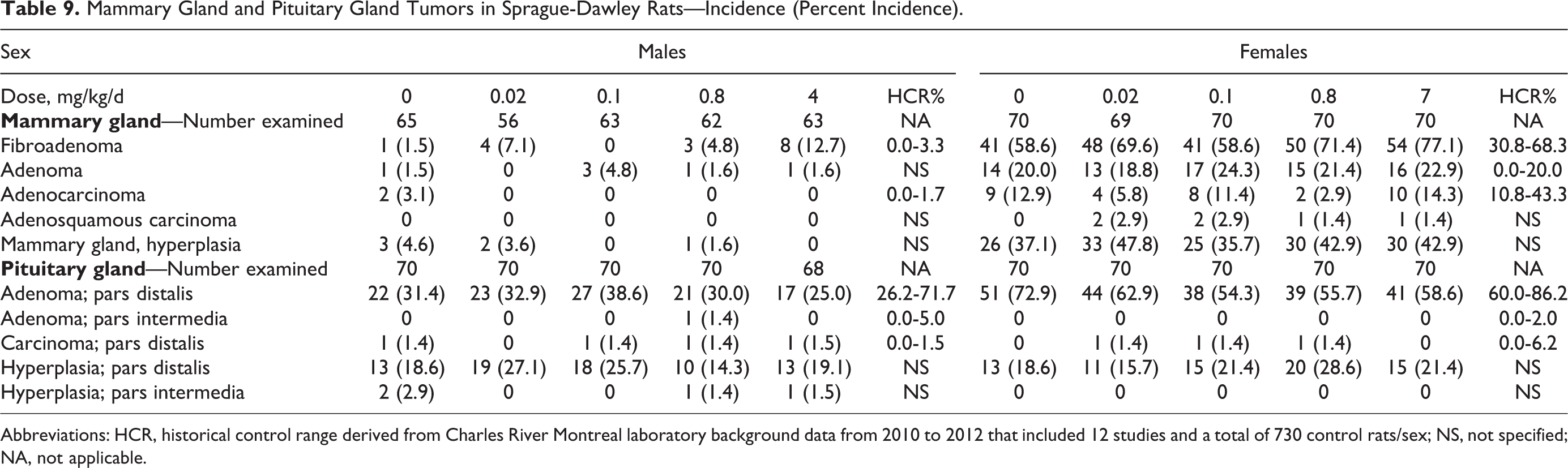
Abbreviations: HCR, historical control range derived from Charles River Montreal laboratory background data from 2010 to 2012 that included 12 studies and a total of 730 control rats/sex; NS, not specified; NA, not applicable.
There were some other isolated tumors for which occurrence reached statistical significance (Table 10) for overall trend (males: hemangiosarcoma in spleen and adenoma in stomach; females: histiocytic sarcoma in hemolymphoreticular tissue) and/or pairwise group comparisons (males: thyroid gland c-cell adenomas at 0.1 and/or 0.8 mg/kg/d; females: uterine endometrial stromal polyps at 0.1 mg/kg/d); however, the tumor incidences recorded were within published or internal historical control ranges, were generally randomly distributed in control and treated groups, and/or lacked a dose–response pattern. While hemangiosarcoma was very slightly increased in incidence in the spleen in male rats given 4 mg/kg/d (2/70 or 2.9% vs 0 in controls; maximum historical control range at the test facility of 1.7%), there was no trend for increased hemangiosarcoma in other organs (hemangiosarcoma across whole body; 2/70 or 2.9% at 4 mg/kg/day vs 3/70 or 4.3% in male controls). Thrombosis was associated with splenic hemangiosarcoma in 1 of these high dose males. Stomach adenoma was also identified in 2 (2.9%) of 70 male rats given 4 mg/kg/d, while none were found in controls. However, there were no malignant stomach tumors noted in males or females at any dose. Gastric tumors, including malignancies, can total up to 3.7% in historical background data. 33 However, in rats, these mostly represent squamous cell carcinomas. Gastric adenomas are even less common in rats with incidence usually less than 1%. 36,37 Histiocytic sarcoma was noted in 3 females given 7 mg/kg/d (4.3%) and in only 1 control. However, this incidence (4.3%) is compatible with historical control data at the test facility of up to 4%. Increased incidence of thyroid C-cell adenomas for males given 0.1 or 0.8 mg/kg/d daprodustat (8/70; 11.4% incidence for both groups) was within the historical control range at the test facility of up to 15.0%. There were several more C-cell carcinomas in the control than in other groups (Table 10), and when total benign and malignant C-cell tumors were combined, the incidence rates were similar among all dose groups. There was a slight increase in C-cell hyperplasia in males given 0.02 mg/kg/d (17/70 or 24.6% vs 8/70 or 11.4% in controls), but the increase was limited to this single dose and lacked a dose response. Finally, the incidence rate for uterine endometrial stromal polyps in female rats given 0.1 mg/kg/d (7/70; 10%) falls well within the historical control range at the test facility (up to 15.0%), and there were no uterine stromal cell sarcomas observed in any rats at this dose.
Selected Tumors in Sprague-Dawley Rats—Incidence (Percentage Incidence).
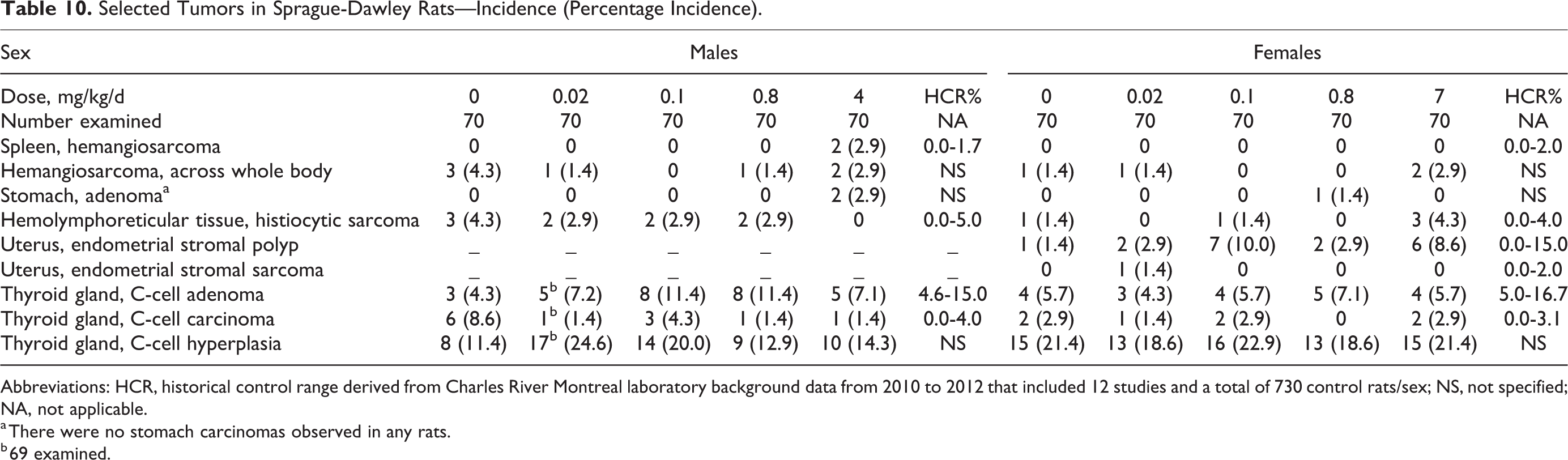
Abbreviations: HCR, historical control range derived from Charles River Montreal laboratory background data from 2010 to 2012 that included 12 studies and a total of 730 control rats/sex; NS, not specified; NA, not applicable.
a There were no stomach carcinomas observed in any rats.
b 69 examined.
Histopathology (Neoplastic Findings)—Mouse Study
Similarly, in mice, neoplastic lesions observed were generally typical of those commonly encountered in CD1 mice of this age and were not considered daprodustat related. For mice given 0.8 mg/kg/d daprodustat alone, increased rates of hepatocellular carcinomas (P value of .0377) in males and harderian gland adenomas (P value of .0430) in females were noted when compared to controls using Peto’s 1-sided test for pairwise group comparisons (Table 11). However, these results were not considered to be statistically significant (P values >.01), since these are both considered common tumors. 31 The highest incidence of hepatocellular carcinoma (12/60; 20%) occurred in the male mid-dose group given 0.8 mg/kg/d daprodustat alone and was just outside the historical range (up to 15%) at the test facility (Charles River Montreal laboratory, 2010-2012, 12 studies with 740 mice/sex). However, there was no dose response, and incidences of hepatocellular adenomas were comparable to the control group (7/60 or 11.7% incidence in controls vs 9/60 or 15.0% incidence in the 0.8 mg/kg/d group) with control incidence exceeding that observed in the other male treatment groups (Table 11). In female mice, increased incidence of harderian gland adenomas were limited to the group given 0.8 mg/kg/d daprodustat alone (12/60 or 20.0% incidence) and occurred in the absence of increases in malignant harderian gland tumors. Although exceeding the upper end of the historical control range for females at the test facility (10%) or in the published literature (up to 11.2%), 38 the increased incidence in female mice given 0.8 mg/kg/d (20%) was limited to this single-dose group with no dose response and was not associated with an increase in malignant harderian tumors. In fact, the female high-dose group given 3 mg/kg/d had a very low incidence (2/60 or 3.3%). Therefore, neither liver or harderian tumors were considered treatment related, given the lack of statistical significance, lack of dose response, limit to 1 dose group and gender, and lack of effect on other tumors in the same organ.
Selected Tumors in CD1 Mice—Incidence (Percent Incidence).
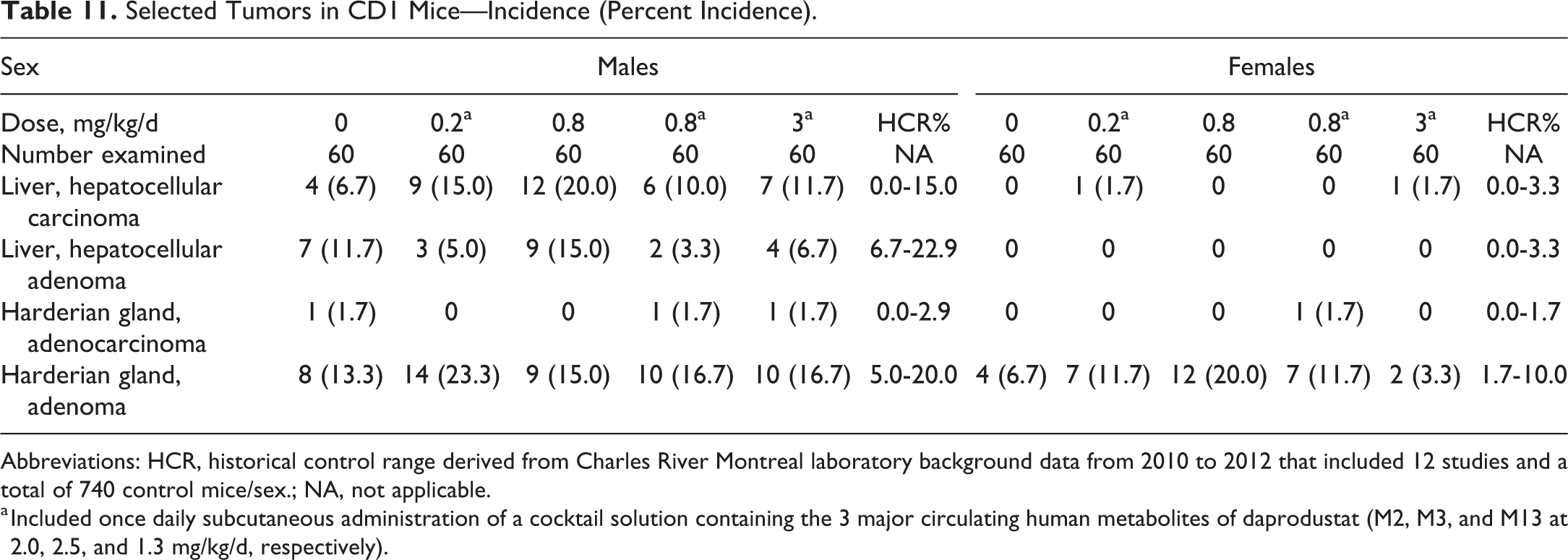
Abbreviations: HCR, historical control range derived from Charles River Montreal laboratory background data from 2010 to 2012 that included 12 studies and a total of 740 control mice/sex.; NA, not applicable.
a Included once daily subcutaneous administration of a cocktail solution containing the 3 major circulating human metabolites of daprodustat (M2, M3, and M13 at 2.0, 2.5, and 1.3 mg/kg/d, respectively).
Histopathology (Non-Neoplastic Findings)
The majority of non-neoplastic microscopic findings observed in these studies were considered incidental, of the nature commonly observed in aged Sprague-Dawley rats or CD-1 mice, and/or were of similar incidence in control and treated animals, and/or lacked a dose–response pattern and were considered unrelated to daprodustat. Exceptions are discussed below.
Histopathology (Non-Neoplastic Findings)—Rat Study
In rats, minimal to moderate erosion or ulceration of the glandular stomach was noted with increased incidence and severity in males and females at 4 and 7 mg/kg/d, respectively. In the heart, there was a dose-related increase in incidence and/or severity of progressive cardiomyopathy in males and females at ≥0.8 mg/kg/d. Although incidence was within the historical control ranges at the test facility (55.0%-100% in males; 18.3%-80.0% in females), this finding was considered exacerbated by daprodustat. Additionally, minimal to marked atrial thrombosis was seen in males at 4 mg/kg/d. Progressive cardiomyopathy and atrial thrombosis were considered as the cause of death in several preterminal males at ≥0.8 mg/kg/d and contributed to the statistically significant increase in mortality in males at 4 mg/kg/d. Also, thrombosis with or without an infarct or segmental necrosis occurred in various tissues (ileum, cecum, rectum, kidneys, spleen, and nasal cavities) with an increased incidence and/or severity in males at 4 mg/kg/d. These stomach, cardiac, and thrombotic findings are considered a consequence of exaggerated pharmacologic responses of daprodustat that resulted in hyperviscosity and prothrombotic potential. 39 Additionally, stomach erosions have been associated with gastric mucosal ischemia caused by microvascular thrombosis secondary to polycythemia. 40 These findings were also observed in previous rat oral toxicity studies of daprodustat at doses that resulted in marked pharmacologically mediated increases in red cell mass and in rat toxicity studies of marketed erythropoiesis-stimulating agents (ESA). 41 -44
Histopathology (Non-Neoplastic Findings)—Mouse Study
In mice, treatment-related non-neoplastic microscopic findings were noted in spleen, liver, bone marrow, blood vessels, kidney, thymus, sciatic nerve, and injection sites. Congestion was noted throughout the whole animal at necropsy. Most treatment-related microscopic changes observed were more pronounced in females. For findings that were observed in animals receiving 0.8 mg/kg/d with or without metabolites, microscopic findings were often more pronounced in those animals also receiving the metabolites than in animals receiving daprodustat alone.
In mice, increased incidence/severity of minimal to severe increased hematopoiesis in the spleen at all doses and in the liver of females given 3 mg/kg/d + metabolites were observed. These findings were associated with an increased incidence and/or severity of whole-body congestion observed at all doses and are expected based on daprodustat’s pharmacologic effects on erythrocytic cell populations. There was a slight increase in the incidence of minimal to mild sciatic nerve degeneration in females given 3 mg/kg/d + metabolites. This finding was observed in previous subchronic mouse toxicity studies and is considered a result of ischemia due to compromised perfusion related to significantly increased Hct (≥70%) and/or decreased serum glucose concentrations. 45,46 Decreased serum glucose concentrations were observed in 13-week mouse toxicity studies at doses that resulted in marked increases in Hct and are considered related to greater consumptive demand from the increased red blood cell counts in these animals. 47
In mice, an increased incidence and/or severity of minimal to marked vascular/perivascular inflammation, compatible with polyarteritis, was observed in numerous tissues in males given 0.2, 0.8, and 3 mg/kg/d + metabolites and in females at all doses. This change was most prominent in the reproductive tracts and accessory glands, urinary tract, digestive tract, heart, aorta, and thymus. This finding was characterized by a mixed inflammatory cell infiltrate with degeneration or fibrinoid necrosis of the vascular wall, occasionally with dilatation or thrombosis of affected vessels, and hemorrhage in surrounding tissue. Rarely, medial degeneration was noted in the aorta without inflammation. Perivascular inflammation is a common spontaneous finding in aged mice. 48 In addition to treatment-related effects of a drug through vasoactive pharmacology, an increased incidence of this background change can be associated with a number of proinflammatory conditions. 49 -51 Hence, the comparative increased incidence of vascular and perivascular inflammation in mice given the metabolites when compared to daprodustat alone may be partially related to daily injection of the metabolites, which procedurally will induce persistent local subcutaneous irritation and inflammation. This is supported by the increased incidence of hemorrhage at injection sites at all doses given the metabolite cocktail, associated with increased pigmentation, consistent with red blood cell breakdown pigments such as hemosiderin and/or bilirubin, in splenic macrophages, hepatic Kupffer cells, and/or kidney tubules. There was also an increased incidence of lymphoid hyperplasia in the thymus of females at all doses as well as an increased incidence of increased myelopoiesis in the bone marrow of females given 3 mg/kg/d + metabolites, further suggesting immune stimulation. However, vascular lesions cannot be completely explained by proinflammatory stimuli alone, as vascular/perivascular inflammation was noted sporadically at higher doses in previous chronic rodent toxicity studies of daprodustat and was considered secondary to marked pharmacologically mediated polycythemia and increased blood viscosity at these doses. Local vascular ischemia may accompany such supraphysiologic effects on hematologic parameters and result in initiation of local degenerative changes. 39 Polyarteritis nodosa, an equivalent vascular change in rodents, is recognized to be causally related to alterations in renin–angiotensin pathways and nitrous oxide, which would be expected consequences of vascular ischemia. 52 Therefore, at high doses, daprodustat’s suprapharmacologic effects on the erythron may be augmenting ischemic vascular effects in mice. 53,54 Similar vascular lesions have been induced with recombinant human EPO in rats. 55
Discussion
The carcinogenic potential of daprodustat was assessed via once-daily oral administration in 2-year rat and mouse carcinogenicity studies. The mouse study also included once-daily subcutaneous administration of the 3 major circulating human metabolites of daprodustat (M2, M3, and M13). These studies were conducted to support clinical development and subsequent marketing application of daprodustat for the treatment of anemia of CKD. The high doses selected for each study were considered to represent the MTDs for 2 years of administration. Doses for both studies, and route of administration of metabolites for the mouse study, were agreed via special protocol assessment by FDA CAC. Pharmacologically mediated increases in red cell mass parameters were observed at the high doses in each study. Systemic exposures (AUC) to daprodustat achieved at the high doses exceed predicted exposure at the maximal human dose by 143-fold (mice) and 903-fold (rats). Systemic exposures achieved to the 3 major human circulating metabolites in the mouse study exceed predicted exposures (AUC) for each respective metabolite at the maximal human dose by approximately 1.43- to 1.75-fold.
Non-neoplastic findings were observed in each study that were predominantly consequent to pharmacologically mediated increases in erythropoiesis/red cell mass and were comparable to findings observed in previous chronic and subchronic toxicity studies of daprodustat. In rats, non-neoplastic findings of atrial thrombosis and an exacerbation of progressive rodent cardiomyopathy were observed and contributed to statistically significant increased mortality in high-dose males (4 mg/kg/d). Daprodustat administration had no impact on mortality in female rats (up to 7 mg/kg/d) or in male or female mice (up to 3 mg/kg/d + metabolites).
The absence of treatment-related neoplastic effects in these carcinogenicity studies of daprodustat are in agreement with results published for the HIF-PHDs, FG-2216, and roxadustat. 26,27 Unlike the carcinogenicity studies of FG-2216 and roxadustat, which assessed 3-times weekly administration, the carcinogenicity studies of daprodustat assessed once-daily administration. The once-daily dosing paradigm was selected as representing the worst-case scenario for supporting both 3-times weekly and once-daily clinical dosing paradigms that are being assessed in phase 3 clinical trials of daprodustat. Therefore, the results from these daprodustat carcinogenicity studies, combined with results from the FG-2216 and roxadustat studies, adds further support that chronic inhibition of the HIF-PHDs does not present a significant clinical concern for carcinogenic responses.
Our results are consistent with additional nonclinical toxicity data generated during the nonclinical development program for daprodustat. Daprodustat and its 3 major circulating human metabolites were not genotoxic based on results of a standard battery of genetic toxicity assays (data not presented). Additionally, in chronic rodent (6-month study in Sprague-Dawley rats) and nonrodent (9-month study in cynomolgus monkeys; note that cynomolgus monkeys produce the 3 major circulating human metabolites of daprodustat) oral toxicity studies, there were no preneoplastic findings, no effects on reproductive organs that would suggest hormonal perturbations, no evidence of immunosuppression, and no proliferative changes other than the expected pharmacologic effect of bone marrow erythroid hyperplasia. The toxicity profile of daprodustat across species has been consistent with effects secondary to exaggerated pharmacology and subsequent increases in red cell mass, similar to the nonclinical effects observed with marketed ESA. 41 However, some of the non-neoplastic toxicologic effects of daprodustat may have other contributory mechanisms, as it has been reported that some ESA-related toxicities are not solely dependent upon increased Hct. 56
The reduction in EPO mRNA expression observed in daprodustat-treated rat and mouse kidneys sampled at week 26 of dosing was surprising, given the dose-dependent increases observed in liver EPO mRNA expression. The mechanism behind these differing EPO expression patterns between liver and kidney are not understood. These results may simply reflect the timing of tissue sampling for these analyses; only a single sampling time point (week 26) was assessed for this end point during the course of these 2-year studies. Single-dose studies of daprodustat in mice (at 60 mg/kg) that assessed the time course of EPO mRNA expression have demonstrated transient increases in both liver and kidney mRNA levels, with differences in the time-of-peak expression (liver EPO peaked at 6 hours postdose while kidney EPO peaked at 8 hours postdose; data not presented).
The role of HIF in cancer biology is complex; there are conflicting nonclinical and clinical data associating increased HIF with tumor progression or angiogenesis, metastasis, and poor prognosis, respectively, versus experimental evidence demonstrating anticancer effects of HIF stabilization. Reviews by Semenza 57 and Pugh and Ratcliffe 58 discuss pharmacologic targeting of HIF, either HIF stabilization or HIF inhibition, in various contexts. The reader is referred to these reviews for more in-depth discussion. Additionally, Keith et al 59 provide an excellent review on the complex role of HIF in hypoxic tumor growth. In short, these authors highlight both increased or decreased tumor growth depending on the model and context and describe opposing roles for HIF1α and HIF2α, concluding that a better understanding of the unique roles of these HIF isoforms in human cancer is warranted. Additionally, given the well-documented role of VEGF in cancer biology, we included assessments of VEGF mRNA from several selected tissues from the rat and mouse carcinogenicity studies of daprodustat. The minimally increased VEGF mRNA observed in lung in the mouse study was not toxicologically significant, as there was no consequence on tumorigenesis. Thus, potential concerns regarding HIF pathway upregulation and its role in carcinogenesis were not supported, given the limited VEGF modulation by daprodustat.
In summary, there were no treatment-related neoplastic findings in 2-year rat or mouse carcinogenicity studies of daprodustat. Daprodustat was dosed once daily to MTDs in each study and elicited expected pharmacologic effects as evidenced by increased red cell mass parameters in each study. The mouse study included once-daily administration of daprodustat’s 3 major circulating human metabolites to systemic exposures (AUCs) equivalent to approximately 1.43- to 1.75-fold respective predicted human exposures at the maximum human once-daily dose (24 mg/day). Exposure (AUC) to daprodustat achieved at the high doses exceed respective predicted human exposures at the maximum human once-daily dose by 143-fold (mice) and 903-fold (rats). These data support chronic administration of daprodustat in clinical trials without increased tumorigenic risk.
Supplemental Material
Supplemental Material, gales_supplemental_fig1a-b - Carcinogenicity Assessment of Daprodustat (GSK1278863), a Hypoxia-Inducible Factor (HIF)-Prolyl Hydroxylase Inhibitor
Supplemental Material, gales_supplemental_fig1a-b for Carcinogenicity Assessment of Daprodustat (GSK1278863), a Hypoxia-Inducible Factor (HIF)-Prolyl Hydroxylase Inhibitor by David F. Adams, Mark S. Watkins, Luc Durette, Josée Laliberté, Félix Goulet, Elaine Debien, Kendall S. Frazier, Nacéra Mellal, Liangfu Chen, Wei Shi, Roberta Thomas and Erding Hu in Toxicologic Pathology
Supplemental Material
Supplemental Material, gales_supplemental_Table_1 - Carcinogenicity Assessment of Daprodustat (GSK1278863), a Hypoxia-Inducible Factor (HIF)-Prolyl Hydroxylase Inhibitor
Supplemental Material, gales_supplemental_Table_1 for Carcinogenicity Assessment of Daprodustat (GSK1278863), a Hypoxia-Inducible Factor (HIF)-Prolyl Hydroxylase Inhibitor by David F. Adams, Mark S. Watkins, Luc Durette, Josée Laliberté, Félix Goulet, Elaine Debien, Kendall S. Frazier, Nacéra Mellal, Liangfu Chen, Wei Shi, Roberta Thomas and Erding Hu in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank Tracy Gales (GlaxoSmithKline) for assistance in formatting tables and figures for submission.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David F. Adams, Mark S. Watkins, Kendall S. Frazier, Liangfu Chen, Wei Shi, Roberta Thomas, and Erding Hu are employees of GlaxoSmithKline and hold stock and/or stock options in GlaxoSmithKline.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
