Abstract
Furan, a possible human carcinogen, is a product of incomplete combustion and is present in cigarette smoke, engine exhaust, and processed food. Oral administration induces liver toxicity and carcinogenesis in F344 rats and B6C3F1 mice. To assess possible adverse effects from inhalation, A/J mice were nose-only exposed for 3 hours to furan (0, 30, 75, 150, 300, or 600 ppmv) and euthanized after 24 hours, 48 hours, or 1 week. Histopathology evaluation revealed bronchiolar club cell necrosis (diffuse, marked) with airway denudation following exposure to 300 and 600 ppmv furan with evidence of club cell regeneration and partial repair after 1 week. Initial signs of hepatotoxicity were observed in the 150 ppmv furan-exposed group. Acute necrosis and mineralization were observed in livers at 24 and 48 hours with hepatocyte regeneration by 1-week postexposure in mice exposed to 300 and 600 ppmv furan; the 300 ppmv exposed group had multifocal mineralization that evoked a mild granulomatous response. Measurement of urinary furan metabolites confirmed that the mice metabolized furan to the toxic intermediate, cis-2-butene-1,4-dial. These observations indicate that inhaled furan is toxic to lungs with club cells as the target as well as liver.
Introduction
Furan has been detected in many foods and beverages. 1 –3 In addition, furan is an important industrial chemical that is also present in the environment in smog, engine exhaust, wood smoke, and cigarette smoke. 4,5 While the toxicity of oral administration of furan has been well studied, few studies have addressed the adverse effects that may arise from inhalation exposure. This shortcoming is particularly disconcerting, because human exposure to inhaled furan may be significant, particularly in smokers. It is both hepatotoxic and carcinogenic after oral administration in mice and rats. 6,7 Based on these results and the large potential for human exposure, furan has been listed as a possible human carcinogen by the National Toxicology Program and the International Agency for Research on Cancer. 5,8
The liver is the primary target organ when furan is given orally. 9 The extent of furan-induced toxicities reported at extrahepatic locations is limited to the kidney, forestomach, and adrenal gland. 6 The explanation for this highly selective target organ toxicity is that furan toxicity requires cytochrome P-450-catalyzed oxidation to the reactive metabolite, cis-2-butene-1,4-dial (BDA; Figure 1), which occurs at a high rate in the liver. 10 –15 Consequently, there is an extensive first-pass effect that results in the liver becoming the primary target with very little furan leaving the liver to reach other organ sites. 10,16
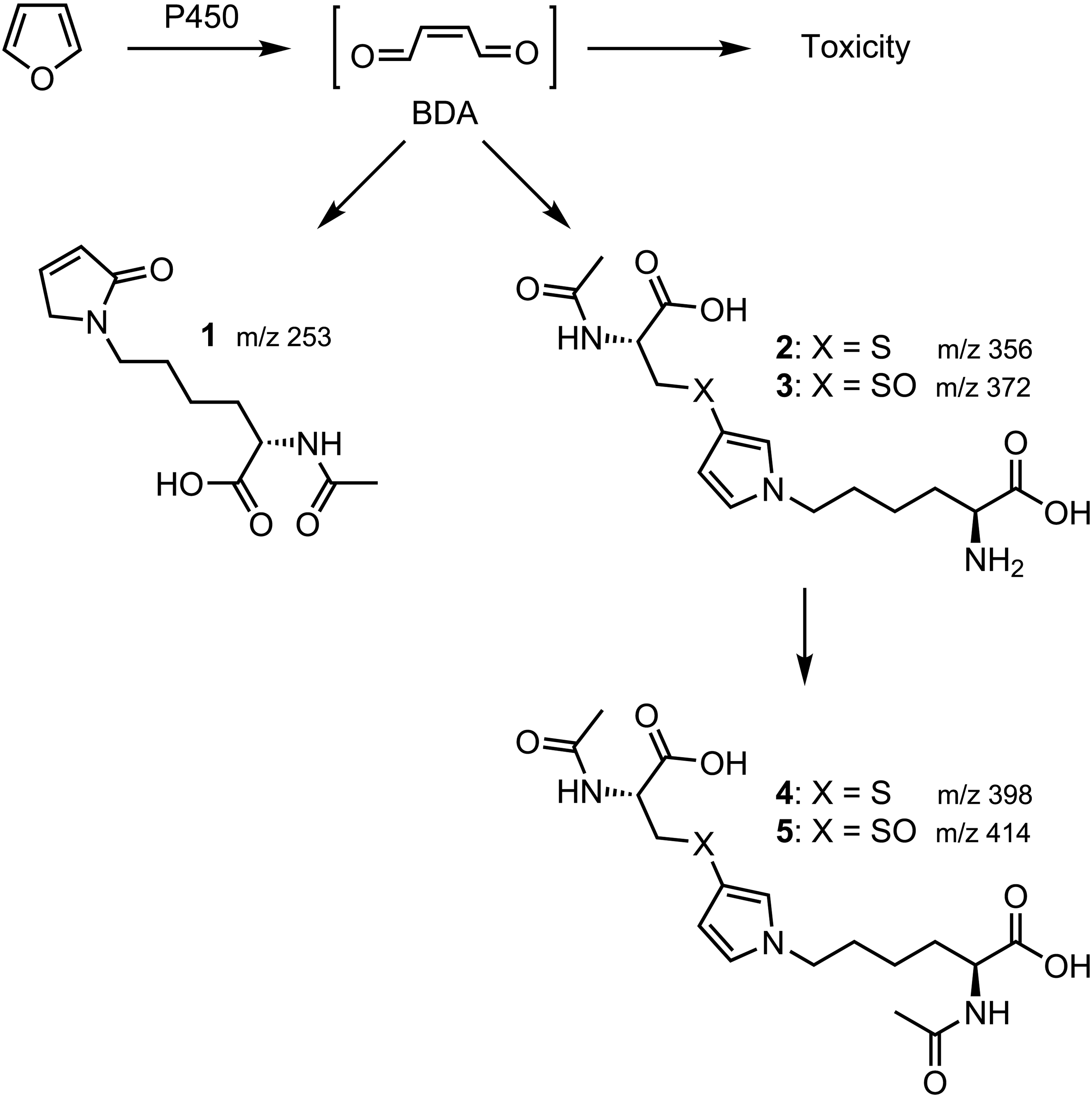
Metabolism of furan to the toxic intermediate and the urinary metabolites of furan detected by liquid chromatography with tandem mass spectrometry (LC-MS/MS) analysis.
Despite the fact that inhalation is a major route of furan exposure in humans, the inhalation toxicity of furan in rodents has not been well studied, with only 2 studies reported in the literature: one in Swiss mice indicated that furan’s lethal concentration (LC50) was 0.12 µg/mL (43 ppmv). 17 This study has been questioned as there is a concern that the exposure chamber design resulted in nonspecific asphyxiation. 18 The other involved exposure of Sprague Dawley rats to 1014, 2851, or 4049 ppmv for 1 hour in a dynamic inhalation chamber followed by a 2-week observation period. 19 The animals were reported to have shown signs of toxicity such as weight loss, respiratory distress, and death. Gross postmortem evaluation indicated that there were no treatment-related lesions. Histopathological analysis was not performed.
Given the adverse effects with oral exposure, including carcinogenicity, the high levels of furan vapor in the environment, particularly in cigarette smoke, and the lack of detailed studies, we wanted to identify the vapor concentration at which furan toxicity occurred and to assess the nature of the adverse events following inhalation exposure. We exposed A/J mice to escalating furan vapor levels and assessed the resulting toxicity to lungs and livers through histopathological analysis. We also evaluated the ability of the mice to metabolize furan to the reactive metabolite BDA by measuring the urinary levels of BDA-derived metabolites.
Materials and Methods
Chemicals
Furan was obtained from Sigma-Aldrich (St Louis, Missouri). [2H6]Acetic anhydride was purchased from Cambridge Isotope Laboratories, Inc (Andover, Massachusetts). Isotopically labeled standards for furan urinary metabolites were prepared as previously described. 20
Animals
This study was accepted by the University of Minnesota Institutional Animal Care and Use Committee. Female A/J mice, 7 weeks of age, were obtained from Jackson Laboratories (Bar Harbor, Maine) and housed 5 mice per cage under standard conditions and sustained on American Institute of Nutrition-76 pellet diet (Research Diets Inc, New Brunswick, New Jersey). They were randomly divided into treatment groups and acclimated to the facility for 1 week. Acclimatization to the nose-only, flow past cylindrical stainless steel inhalation chamber (In-Tox Products, LLC, Moriarty, New Mexico) was achieved by placing mice into the chamber for 3 consecutive days, where they were exposed to compressed air for 1.5 hours each day. Body weight was measured weekly with the exception of the 600 ppmv furan-exposed group which was weighed 24 hours following the exposure. Clinical signs were monitored twice daily.
Furan Treatment
Groups of 6 female A/J mice were exposed to 3 hours of 0, 30, 75, 150, 300, or 600 ppmv furan (6 mice/group) in the inhalation chamber. The furan vapor was produced by bubbling a controlled flow rate (Omega FL-3007SA flow meter, Stamford, Connecticut) of air through furan liquid, held in a test tube that was placed in a glycerol/water/dry ice bath. The saturated air was then diluted with additional air at a controlled flow rate (Omega FL-3096SA) and directed into the chamber.
Furan concentration was measured in the chamber, using one of the animal ports, every 1 minute with a VOC-TRAQ II USB photoionization detector (MOCON, Lyons, Colorado), with the acquired data stored on a laptop computer. The detector was calibrated before each exposure by measuring the signal of a sealed Erlenmeyer flask to which known volumes of liquid furan had been added and allowed to vaporize. The calibration curve was based on 3 sets of 3 injections and blanks that encompassed the target chamber concentration.
Two animals from each group were euthanized at 24 hours, 48 hours, and 1-week postexposure. Urine and blood were collected from most animals at the time of euthanasia. Urine was collected by sacrificing each mouse alone in a clean cage without bedding. The urine was collected from the bottom of the cage with a clean syringe. Blood samples from each time point were pooled prior to centrifugation at 10 000 g for 10 minutes. The serum was stored at −20°C. Alanine aminotransferase (ALT) levels were determined by Marshfield Labs (Marshfield, Wisconsin) on a Beckman Coulter AU5800 Clinical Chemistry System (Brea, California). Lungs, livers, heart, and kidneys were collected for histopathological analysis. The lungs were slightly inflated by slowly infusing 0.5 mL of 10% formalin in phosphate buffer through the trachea into the lungs with a syringe prior to placement in 10% formalin in phosphate buffer. All tissues were immediately placed in 10% phosphate-buffered formalin (Fisher Scientific, Pittsburg, Pennsylvania) at room temperature. After 24 hours, fixed tissues were transferred to 70% ethanol.
Histopathology
Tissue processing and histopathological analyses were performed by the Comparative Pathology Shared Resource, University of Minnesota Masonic Cancer Center. Formalin-fixed sections of the lungs (all lobes) and livers (all lobes) were processed into paraffin blocks using standard histology techniques, sectioned to 4-µm thickness, and stained with hematoxylin-eosin (H&E) and von Kossa stain. Histology slides were evaluated with light microscopy, and diagnoses were verified by 2 American College of Veterinary Pathologists board-certified pathologists (A.-F.T. and M.G.O’S.). When histopathology severity modifiers are used, they are based, in general, on a previously described grading scheme using the following criteria: minimal (when lesions involved <10% of the tissue section), mild (when lesions involved 11% and 20% of the tissue section), moderate (when lesions involved 21%-40% of the tissue section), and marked (when lesions involved 41%-100% of the tissue section). 21
Immunohistochemistry
For immunohistochemical detection of the bronchiolar club (formerly referred to as Clara) cells, the lung tissue was incubated with goat polyclonal anti-club-cell-specific 10 kDa protein/club cell secretory protein antibody (CC10, clone T-18, catalog no. SC-9772; Santa Cruz Biotechnology, Dallas, Texas). Briefly, 4-µm-thick histological sections of paraffin-embedded pulmonary tissue were deparaffinized, rehydrated, and after endogenous peroxidase blocking were incubated for 30 minutes with the anti-CC10 antibody in a 1:500 dilution. An horseradish peroxidase (HRP) polymer-based detection system (Goat-on-Rodent HRP-Polymer; Biocare, Concord, California) was used with 3,3-diaminobenzidine as chromogen, and slides were counterstained with hematoxylin.
Analysis of Urinary Metabolites of Furan
The levels of furan metabolites were quantified by a modification of a previously described liquid chromatography with tandem mass spectrometry (LC-MS/MS) assay. 20 Briefly, urine (1 or 10 µL) was spiked with isotopically labeled internal standards, acetylated with [2H6]acetic anhydride, then cleaned up by 2-stage solid-phase extraction (SPE): first with Waters Oasis SPE cartridges, then with Waters Sep-pak SPE cartridges. Extracts were analyzed by LC-MS/MS in negative mode on Phenomenex Synergi Hydro RP 4 µm 250 × 0.5 mm with 15 mM ammonium acetate and methanol at 10 µL/min.
Results
Our inhalation study was designed to identify the vapor concentration at which toxicity occurred and to assess the nature of the adverse events. Female A/J mice were evaluated in this study since our long-term goal is to evaluate the carcinogenic activity of furan in this animal model; this model has been extensively employed in short-term bioassays to determine the carcinogenic activity of tobacco chemicals including inhaled compounds. 22 –25 The initial concentration was selected based on the previously reported mouse study. 17 We exposed the initial group of mice to 30 ppmv furan, which was below the IC50 determined in that study. The exposure dose was then elevated in each subsequent groups until external signs of toxicity were observed. This occurred at 600 ppmv furan. Serum ALT levels were elevated starting at 150 ppmv. Microscopic evidence of toxicity was observed in the lungs at exposure levels of 300 and 600 ppmv and in the liver at exposure levels of 150, 300, and 600 ppmv.
Clinical Signs
External signs of toxicity were only observed in the mice exposed to 600 ppmv for 3 hours. These signs included an unkempt appearance and changes in whisker and ear position, indicating that the mice were experiencing some pain. 26 The morning following the exposure, the mice had somewhat labored breathing and appeared to be continuously trembling. Trembling ceased by that afternoon and breathing appeared normal. These mice experienced a 6% loss in weight at 24 hours and 18% loss at 1 week; none of the other furan exposures affected body weight. Consistent with the hepatic damage described below, a limited analysis of the 24-hour time point serum ALT levels showed modest increases (700 U/L) in the 150 ppmv group and substantial elevations ≥ 4000 U/L) in the 300 and 600 ppmv groups compared to control levels of less than 70 U/L.
Histopathology
Dose-related histopathological changes were observed in the lungs. No furan-dependent changes were detected in the lungs of mice exposed to 30, 75, or 150 ppmv furan. The 300 and 600 ppmv exposure levels caused club cell degeneration (cell swelling and cytoplasmic vacuolation) and necrosis, with widespread cell sloughing and multifocal bronchiolar denudation affecting all terminal bronchioles at 24 and 48 hours (Figure 2). For both 300 and 600 ppmv exposure levels, only a few CC10-positive club cells were still lining the bronchiolar walls at 24 hours (Figure 2E and F). At 48 hours, the bronchioles and the alveolar ducts contained abundant cell debris (comprised mainly of CC10-positive club cells), occasionally occluding the lumen of the lower respiratory tract (Figure 2H and K). At this time point, the bronchiolar wall was multifocally denuded and lined by attenuated, squamoid-flat to cuboidal-ciliated epithelium, immunonegative for CC10 (Figure 2H and K). Multifocal mild alveolar wall necrosis was occasionally present in the 600 ppmv exposure group. The inflammatory reaction was comprised of bronchiolar wall infiltration by low numbers of neutrophils and lymphocytes. One week following the 300 and 600 ppmv exposures, there was incomplete terminal bronchiolar repair with the denudated bronchiolar areas now partially covered by CC10-positive cuboidal cells interspersed with CC10 immunonegative flat to cuboidal cells (Figure 2I and L). Cyto- and karyomegaly and piling of CC10-positive cells were occasionally observed in terminal bronchioles (Figure 2I).
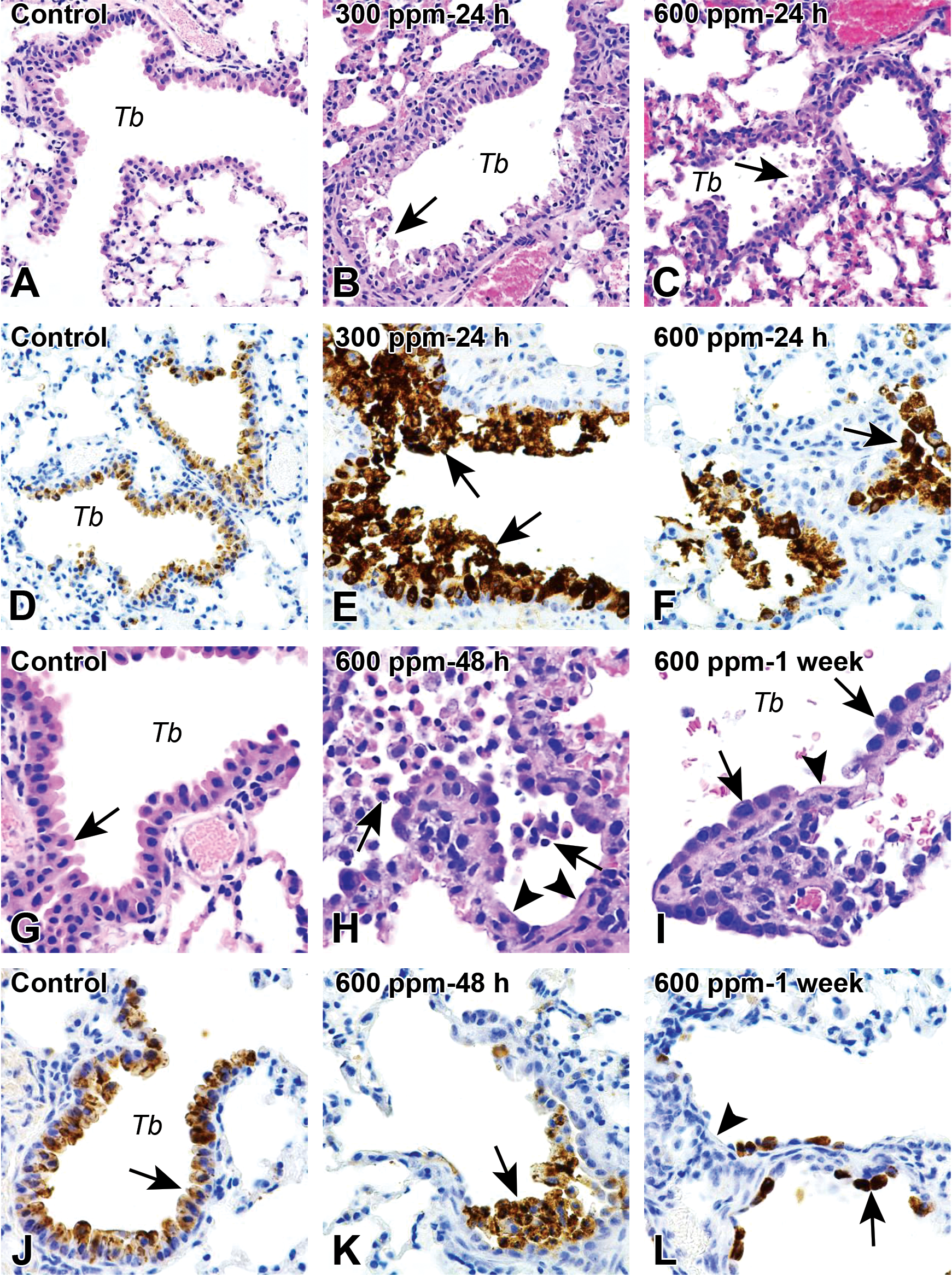
Photomicrographs of Hematoxylin-eosin (H&E) histology (A-C and G-I) and immunohistochemistry for club cell secretory protein antibody/CC10 (D-F and J-L) of lung following 3-hour inhalation exposure to furan (0, 300, or 600 ppmv). (A) and (D) (24 hours) and (G) and (J) (48 hours), Control with terminal bronchiole lined mainly by dome-shaped club cells with eosinophilic (H&E) cytoplasm and intense cytoplasmic CC10 immunolabeling. (B) and (E), Furan at 300 ppmv, 24 hours and (C) and (F), Furan at 600 ppmv, 24 hours; Multifocal degeneration, necrosis, and sloughing of the club cells (arrows) of the bronchioles. (H) and (K), Furan at 600 ppmv, 48 hours, Widespread necrosis and sloughing of club cells (arrows), with locally extensive denudation of the bronchiolar wall (arrowheads) and the bronchioles are partially filled with necrotic club cells. (I) and (L), Furan at 600 ppmv, 1 week; Partial regeneration of the bronchiolar epithelium, which is lined by flat to cuboidal CC10 immunopositive club cells (arrows) interspersed with CC10-immunonegative squamoid to cuboidal cells. Some denuded areas are still present (arrowheads). Tb, terminal bronchiole. (A) to (D): ×20 original objective; (E) to (L) ×60 original objective.
No significant changes were observed within the trachea and bronchi upon microscopic examination of H&E-stained sections. Nevertheless, depletion of CC10-positive club cells within the trachea and bronchi was observed at 48 hours, followed by partial restoration at 1 week following exposure to furan.
The systemic effects of furan were present only in the liver; no lesions were observed in kidneys and heart. The hepatotoxic effects were first observed at 150 ppmv (Figure 3) and presented as a specific midzonal lesion comprised of hepatocyte degeneration and necrosis, minimal, sometimes mild in extent (Figure 3B and D). This gave a very distinct reticulated pattern to the liver at 24 and 48 hours when examined under low magnification. The degenerate hepatocytes were swollen, with amphophilic to homogenous, occasionally vacuolated cytoplasm and sporadic intracytoplasmatic eosinophilic glassy granules (hyaline bodies). At 1 week following the 150 ppmv exposure, the hepatic changes were largely absent, which was interpreted as parenchymal regeneration (Figure 3G).
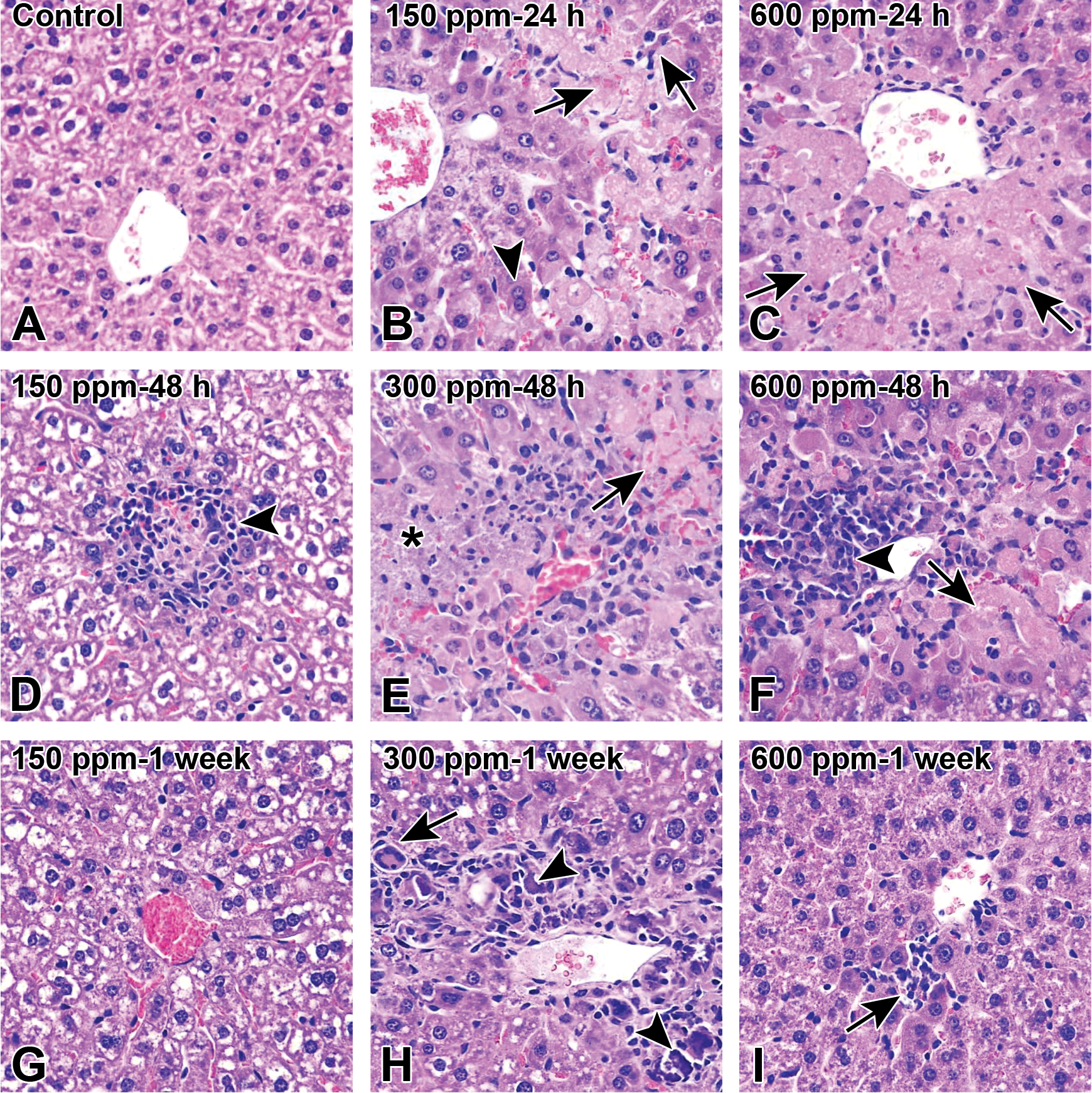
Histopathological images of liver following 3-hour inhalation exposure to furan (0, 150, 300, or 600 ppmv). (A), Control liver. (B), Furan at 150 ppmv, 24 hours; Midzonal hepatocyte necrosis (arrows) and mild degeneration (cytoplasm homogenization and amphophilia) (arrowhead). (C), Furan at 600 ppmv, 24 hours; Centrilobular hepatocyte necrosis (arrows) and sinusoidal collapse. (D), Furan at 150 ppmv, 48 hours; Focal hepatocyte necrosis, with marked, mainly neutrophilic infiltration (arrowhead). (E), Furan at 300 ppmv, 48 hours; Centrilobular and midzonal necrosis (arrows), with central vein collapse, hemorrhage, focal leukocyte infiltration (mainly neutrophils), and mineralization (asterisk). The area of necrosis is bordered by a thin rim of degenerate hepatocytes (cytoplasm homogenization and amphophilia). Furan at 600 ppmv, 48 hours; Centrilobular and midzonal hepatocyte acute degeneration and necrosis (arrow) and central vein collapse, associated with focal, marked, lymphohistiocytic infiltration; few apoptotic hepatocytes are also present. (G), Furan at 150 ppmv, 1 week; Liver is similar to control. Furan at 300 ppmv, 1 week; Replacement of the centrilobular zone by histiocytes, foreign body multinucleate giant cells (MGC) (arrow), and lymphocytes separated by fibroblasts/myofibroblasts and collagen; MGCs are encircling mineralized debris (arrowheads); Furan at 600 ppmv, 1 week; Few lymphocytes, histiocytes, and few pigment-laden macrophages are infiltrating the centrilobular areas (arrow); hematoxylin-eosin staining, ×60 original objective.
Exposure to 300 and 600 ppmv furan clearly induced acute hepatocyte necrosis (mild to moderate) at 24 and 48 hours, that, consistent with ALT levels, was substantially more extensive than the toxicity observed with 150 ppmv with zonal, centrilobular to centro-central and centro-portal bridging necrosis (Figure 3C, E, and F). Degenerate hepatocytes resembling those described for the 150 ppmv exposure group were also present, especially organized as a border between the necrotic areas and the normal parenchyma. The necrotic zones, including the collapsed centrilobular vein, were variably infiltrated by neutrophils, lymphocytes, and histiocytes (occasionally pigment-laden). In the livers from the 300 ppmv group, the infiltration was moderate and primarily neutrophilic, whereas the infiltration was marked and mainly lymphohistiocytic in mice exposed to 600 ppmv furan.
Marked mineralization (confirmed by van Kossa staining) of both degenerate hepatocytes and necrotic debris was observed at 48 hours in mice exposed to 300 and 600 ppmv furan (Figure 3E). At 1 week, the mineralized areas in the 300 ppmv exposed group were overlaid with multifocal-coalescing granulomas, which contained many histiocytes and foreign-body-type giant cells that encircled foci of mineralized necrotic debris. Many lymphocytes and occasional fibroblasts/myofibroblasts were separated by collagenous extracellular matrix (Figure 3E). This unexpected change was interpreted to be a foreign-body reaction to the marked mineralization observed at 48 hours following furan inhalation. Surprisingly, the extent of mineralization was minimal in the 600 ppmv exposure group at 1 week; the liver was essentially normal with minimal hepatic lesions comprising focal infiltration with lymphocytes and histiocytes (sometimes pigment laden), occasionally admixed with cell debris, mainly within the centrilobular areas of the lobules (Figure 3I).
In the highest 3 exposure groups (150, 300, and 600 ppmv), and consistent with hepatocyte regeneration, frequent hepatocyte mitoses (up to 6/high-power field), with occasional mitotic atypia (multipolar divisions) are present at 48 hours, mainly in the area of hepatic parenchyma adjacent to the necrotic cells.
Urinary Furan Metabolites
The urinary metabolites of furan result from the reaction of BDA with lysine (BDA-Nα-acetyllysine 1) and from cysteine-BDA-lysine cross-links (N-acetylcysteine-BDA-lysine 2, N-acetylcysteine-BDA-Nα-acetyllysine 4, and their sulfoxides 3 and 4; Figure 1). 27 –29 The levels of these metabolites were measured in mouse urine using an established LC-MS/MS assay. 20 All 5 metabolites were elevated in the mice exposed to furan, with the 2 most abundant metabolites at all dose levels being N-acetylcysteine-BDA-lysine 2 and its sulfoxide 3 (Table 1). The concentrations of metabolite 2 were linear with dose at both 24 and 48 hours (Figure 4). None of the other metabolites were linear with dose except BDA-Nα-acetyllysine 1; concentrations of this metabolite were linear with furan dose at 48 hours, but not at the other 2 time points. The levels of BDA-Nα-acetyllysine 1 were 10 to 40 times less than the sum of the 4 metabolites derived from cysteine-BDA-lysine cross-links, indicating that the formation of cysteine-BDA-lysine crosslinks was more dominant than the direct reaction of BDA with lysine in this mouse model (Figure 5).
Levels of Furan Metabolites Detected in Urine of A/J Mice Exposed to Furan for 3 Hours in a Nose-Only Inhalation Chamber.
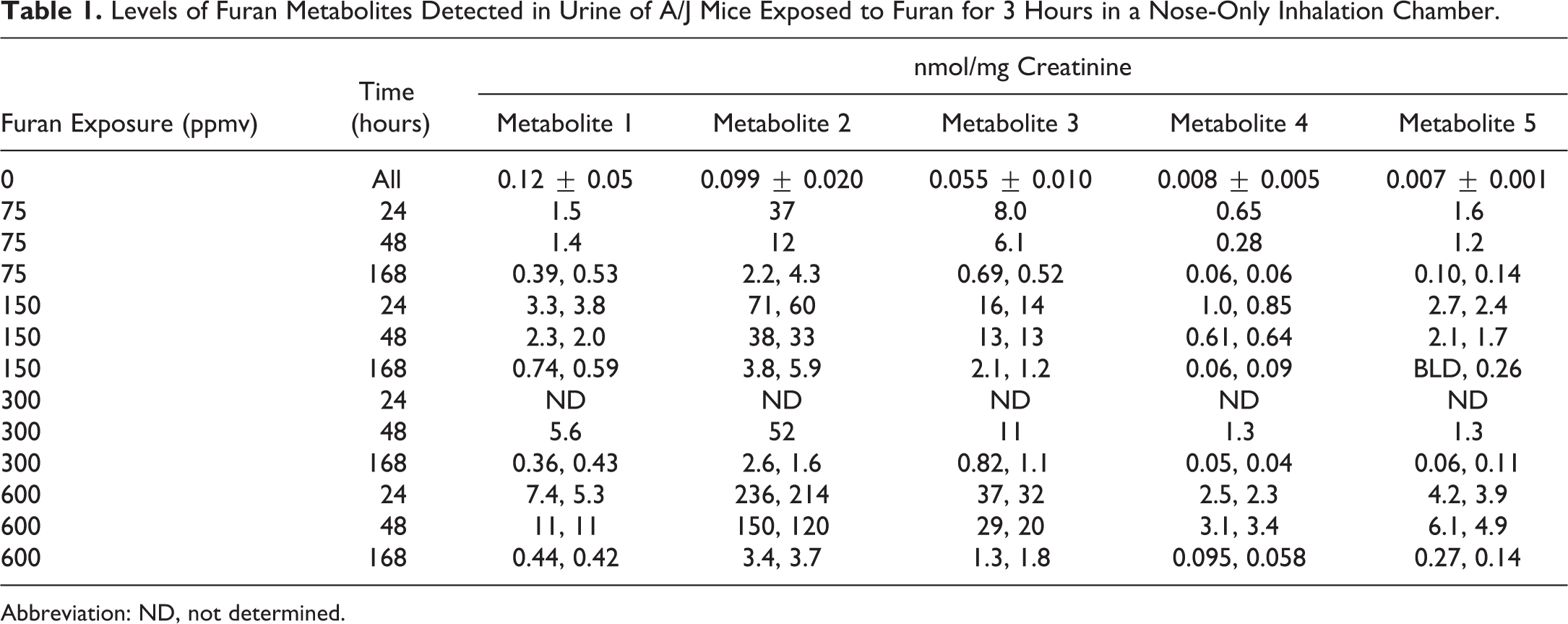
Abbreviation: ND, not determined.
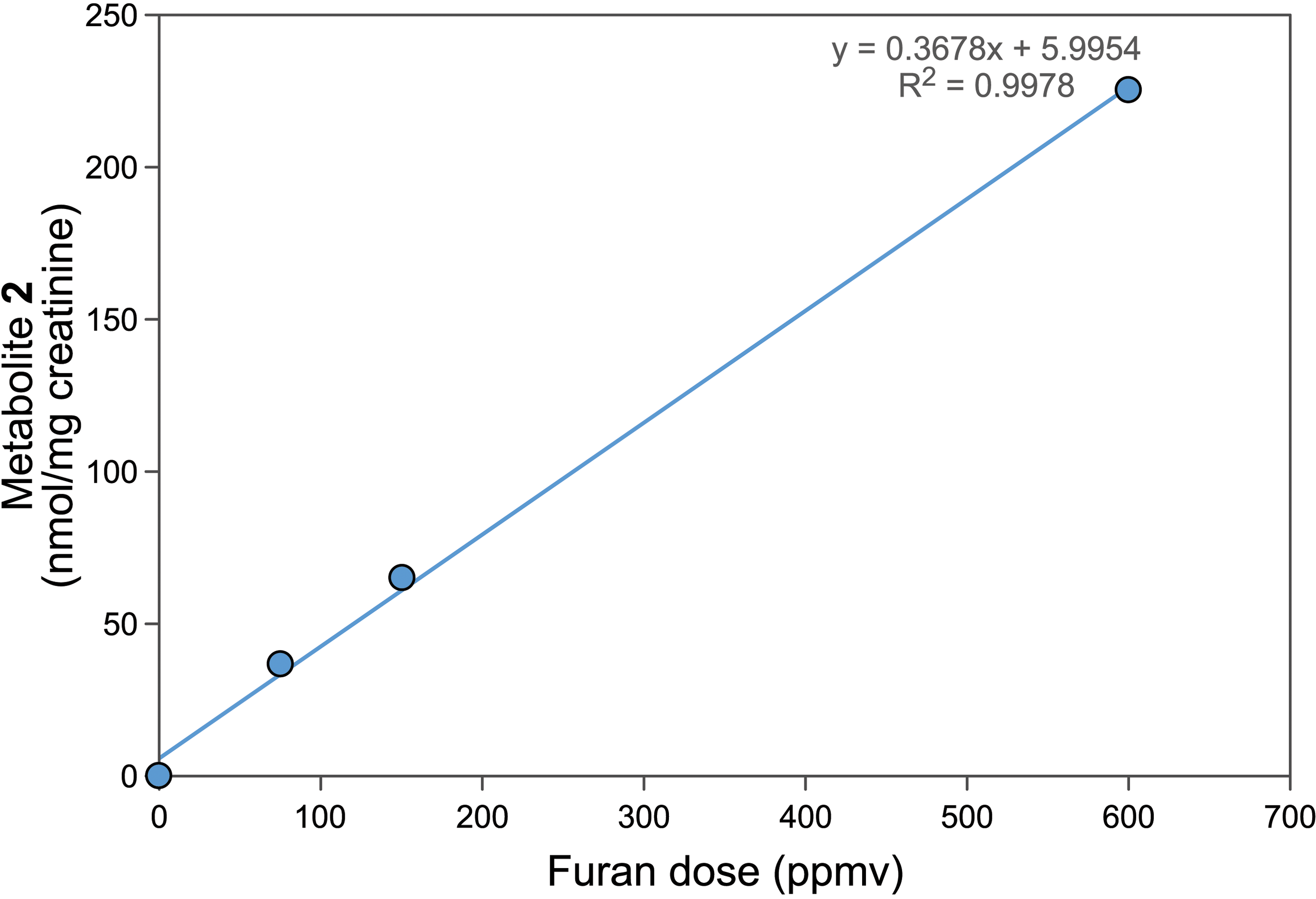
The dose dependence of N-acetylcysteine-BDA-lysine 2 concentrations in urine of A/J mice exposed to 3 hours of 0, 75, 150, or 600 ppmv furan in a nose-only exposure chamber. Data points are averages of 2 mice with the exception of the controls (n = 6) and the 75 ppmv mice (n = 1). Replicates differed by less than 10%. Linear regression was determined using Excel.
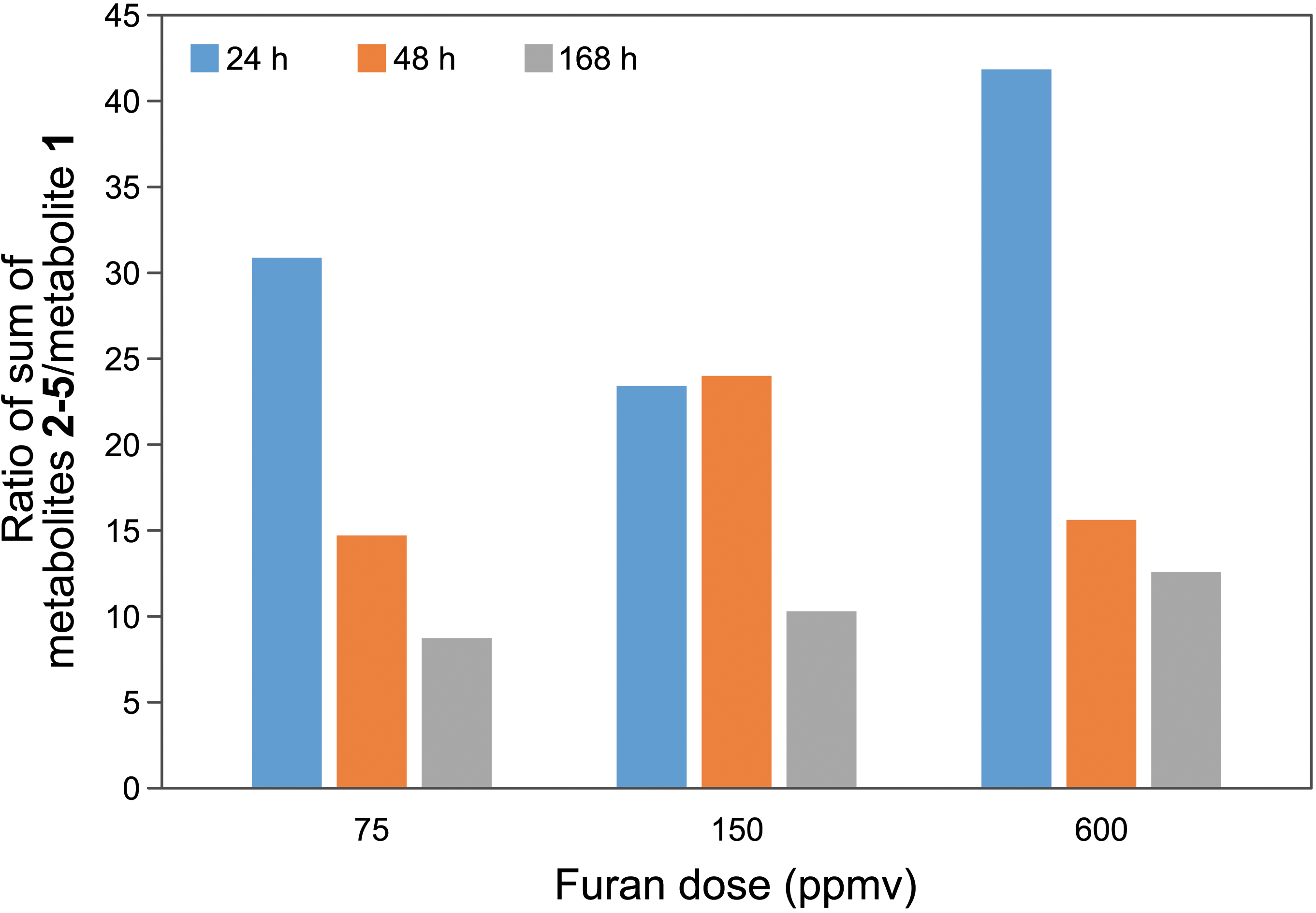
Relative levels of the cysteine-BDA-lysine cross-link metabolites (2-5) to levels of furan metabolite 2 in urine of A/J mice after exposure to 75, 150, or 600 ppmv furan for 3 hours in a nose-only exposure chamber. Data points are averages of 2 mice with the exception of the 24 and 48 hours time points for mice exposed to 75 ppmv, in which case n = 1. Replicates differed by between 1% and 22%.
Discussion
In our study, inhaled furan was toxic to lung, with club cells as the target, and liver. Club cell toxicity likely relates to bioactivation of furan by club cell cytochrome P450 2E1 (CYP2E1) to the highly toxic and reactive metabolite, BDA. This enzyme is an efficient catalyst of furan oxidation to BDA 10 –15,30,31 and it is abundantly expressed in murine club cells. 32 Club cells are frequent targets of toxicants that require metabolic activation due to their richness in mixed function oxidases. 33 The observation that the liver is the more sensitive systemic target of inhaled furan is not surprising as the levels of CYP2E1 are highest in this tissue. 34
Considering the structure of the toxic reactive metabolite and the structural similarities, furan toxicity is expected to be similar to 3-methylfuran. 3-Methylfuran was previously reported to induce massive bronchiolar necrosis targeting club cells and necrosis of olfactory epithelium and mainly centrilobular hepatocytes in mouse, rat, and hamsters, 35 –37 although some species differences were noted. 36 The reactive metabolite thought responsible for the toxic effects of 3-methylfuran is 2-methyl-cis-2-butene-1,4-dial 37 ; this metabolite is structurally related to BDA as they are both cis-enedione.
The observation of the dose-dependent formation of BDA-derived metabolites in urine of the furan-exposed mice is confirmation that the reactive metabolite is generated in vivo. The metabolite profile is similar to that reported previously for oral administration of furan to B6C3F1 mice
20
; this mouse strain is sensitive to the hepatotoxic and carcinogenic effects of orally administered furan.
6,38,39
In both cases, N-acetylcysteine-BDA-lysine
The histopathological analysis of the livers indicates that the toxic effects of furan when inhaled are similar to those observed when furan is given orally to mice. 39,40 Our observations of mainly midzonal hepatocyte degeneration (including cytoplasmic basophilia) and multifocal hepatocellular necrosis (midzonal to centrolobular) followed by reactive leukocyte infiltration were similar to those previously reported. 38,40 We did not observe hepatic-subcapsular pattern of hepatic lesions, prominent oval cell hyperplasia, or biliary tract hyperplasia, as has been reported for oral furan exposure. 38,39,41 This is likely due to the fact that the mice in our study received only a single exposure, whereas those effects were primarily observed following chronic or subchronic exposure protocols. 38 –41
The numerous hepatocyte mitoses observed at the border of hepatic lesions starting at 48 hours is considered a compensatory mechanism following the acute liver-parenchymal loss, and was described as a key toxic response following oral exposure to furan. 39,40 The atypical mitoses observed in the hepatocytes in this study are consistent with abnormal organization of the mitotic spindle and chromosomes and abnormal cell division. A similar finding was previously reported in B6C3F1 mice following oral administration of furan, 40 and is considered to be associated with furan-induced polyploidy and micronucleus formation. 42
Our study indicates that a more in-depth investigation of the toxic effects of inhaled furan is warranted. As a product of incomplete combustion, furan is present in mainstream tobacco smoke at concentrations (7-65 µg/cigarette) comparable to 1,3-butadiene (16-75 µg/cigarette) and benzene (6-73 µg/cigarette). 43 –49 Given that the daily exposure to benzene for a heavy smoker was calculated to be about 22 µg/kg/d (calculated daytime exposure of 26 ppmv), 50 exposure to furan is likely to be significant. In contrast, the average daily dietary consumption of furan by adults has been estimated to range from 0.25 to 0.78 µg/kg body weight. 51 –54 Therefore, the major source of furan exposure to a smoker will be tobacco smoke. Consistent with this premise, levels of BDA-derived metabolites are higher in smokers than in nonsmokers. 20 Our studies with human CYP2E1, liver microsomes, and hepatocytes indicate that humans convert furan to BDA at rates comparable to rats and mice, 31,55 suggesting that humans may be at risk to the harmful effects of furan. Interestingly, smoking is associated with increased severity of advanced liver fibrosis and liver cancer 56 –58 ; furan induces these effects when given orally to rodents. 6,59 In addition, genetic variants of CYP2E1 with higher transcription, increased protein levels, and increased enzyme activity are overrepresented in smokers with chronic obstructive pulmonary disease (COPD) as compared to smokers without COPD (odds ratio = 7.02, 95% confidence interval = 1.63-30.2). 60 Furan may be a critical contributor to these effects of tobacco smoke and more detailed studies would clarify furan’s role in human disease.
Footnotes
Acknowledgments
The authors thank Mr Bob Carlson for his assistance with the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Masonic Cancer Center, University of Minnesota and Minnesota Masonic Charities, and a grant from the National Cancer Institute and the Food and Drug Administration Center for Tobacco Products. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or the FDA. The Masonic Cancer Center Analytical Biochemistry and Comparative Pathology Shared Resources are funded in part by the National Cancer Institute [P30 CA-77598].
