Abstract
Assessment of the peripheral nervous system (PNS) tissues during animal toxicity studies generally is included within guiding documents issued by regulatory agencies of individual nations (eg, US Environmental Protection Agency, US Food and Drug Administration) and multinational federations (eg, European Medicines Agency) as well as international cooperative efforts (eg, International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use, Organisation for Economic Co-operation and Development). The present list of major regulatory guiding documents categorizes recommendations from around the world for sampling and processing PNS tissues (nerves and ganglia) for general animal toxicity studies (ie, where neurotoxicity is not expected) and specialized neurotoxicity studies (ie, where neurotoxicity is anticipated or known to occur). In general, regulatory guidelines call for collection of one or more sensorimotor nerves (usually the sciatic trunk and its branches), though details vary among agencies. Regulatory guiding documents represent a “starting point,” after which additional PNS samples and/or special methods may be implemented at the applicant’s discretion. Best practice recommendations for PNS sampling and processing in animal toxicity studies endorsed by multiple global societies of toxicologic pathology encompass and expand on existing regulatory guidelines.
The peripheral nervous system (PNS) consists of parallel nerve conduits serving to explore and respond to stimuli in the external (sensorimotor arm) and internal (autonomic arm, consisting of enteric, parasympathetic and sympathetic divisions) environments. Each part of the PNS comprises a series of nerves and ganglia that connect to the central nervous system (CNS), chiefly to the spinal cord but in some cases to the base of the brain. The fact that each of these PNS domains possesses unique anatomic, functional, and molecular attributes ensures that PNS neuropathologic evaluation is complex in terms of both study design and study conduct. Ideal preservation and preparation of PNS tissues both require many special techniques and highly trained, experienced technical staff.
Existing regulations for registration of new products often include safety assessment using animal toxicity studies. The design, conduct, and methodologies of such animal toxicity studies are often detailed within guiding documents formulated by regulatory agencies to assist applicants as they seek approval for their products. Such guiding documents offer insight into a regulatory agency’s current thinking on topics governed by legally enforceable regulations. For example, in the United States, regulatory guidance/guidelines produced by the US Environmental Protection Agency (US EPA) and the US Food and Drug Administration (US FDA) are guiding documents designed to help applicants interpret and achieve compliance with the legally mandated US Code of Federal Regulations (CFR). In addition to country-specific regulations and guiding documents, an applicant may also choose to adopt international guidelines developed through collaboration among representatives from regulatory agencies, industrial firms from many countries, and academia; examples of multinational partnerships include the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH; https://www.ich.org/products/guidelines/safety/article/safety-guidelines.html) and Organisation for Economic Co-operation and Development (OECD; https://www.oecd-ilibrary.org/environment/oecd-guidelines-for-the-testing-of-chemicals-section-4-health-effects_20745788). Based on a desire to market a product across many geographic regions, applicants seeking approval for their regulated product may develop multiple parallel regulatory submissions based on substantial differences in regulatory requirements among countries. Careful attention to experimental design may permit applicants to devise a series of animal toxicity studies that simultaneously address guidelines from many regulatory agencies, thereby allowing the applicant’s regulated product to be approved in many venues.
The following compilation lists current major regulatory guidance and/or guidelines (collectively termed guiding documents) that are relevant to appropriate sampling for histopathologic evaluation of the PNS in animal toxicity studies. All the documents listed are available online at no cost, and the uniform resource locators (URLs, or “web addresses”) are current as of July 1, 2019. Table 1 provides links to the most recent guidance documents that mention PNS neuropathology analysis. Requirements for PNS (nerve and ganglia) sampling among various guidelines are compared for general toxicity studies (Table 2) and specialized neurotoxicity studies (Table 3). For the purposes of this article, general toxicity studies are those where a specific toxicity is not expected, while specialized neurotoxicity studies are those where neurotoxicity is anticipated or known to occur. As for other toxicology end points related to histopathology, the applicant is tasked with selecting the appropriate PNS neuropathology guidance for a particular application. Regulatory guidance/guidelines represent a “starting point,” after which additional PNS samples and/or special methods may be implemented at an applicant’s discretion. An organization may choose to follow the basic practices set forth in relevant guidelines, to undertake a more detailed assessment than is listed in the guidelines (eg, to understand the biological basis for neurological signs that were seen during a study), or even—with justification—to disregard a portion of the guideline. In general, decisions to process and examine a set of PNS tissues that comprise a less comprehensive or alternative evaluation strategy than indicated in guidelines should be made after consultation with appropriate regulatory agencies and/or with established scientific practices (eg, references nos. 1,2 ).
Compilation of Current Online Regulatory Guidelines for Neuropathology Assessment During Animal Toxicity Studies.

Abbreviations: EMA, European Medicines Agency; EPA, US Environmental Protection Agency; FDA, US Food and Drug Administration; OECD, Organisation for Economic Co-operation and Development; PNS, peripheral nervous system; TSCA, Toxic Substances Control Act.
aIn reorganizing the US EPA to create the Office of Chemical Safety and Pollution Prevention (OCSPP), guidance offered in the TSCA Part 798 and Part 799 guidelines was superseded by the US EPA “Series 870—Harmonized Health Effects Test Guidelines”. The TSCA guidelines are included here for completeness (since they remain unaltered in the Code of Federal Regulations), and to communicate their “retired” status.
bThe OECD is not a regulatory agency. Instead, OECD guidelines are designed as harmonized (ie, globally accepted) guidance for common problems encountered in studies being conducted at multiple study sites in more than one country.
Regulatory Guidance for Peripheral Nervous System (PNS) Sample Collection in General Toxicity Studies in Animals.
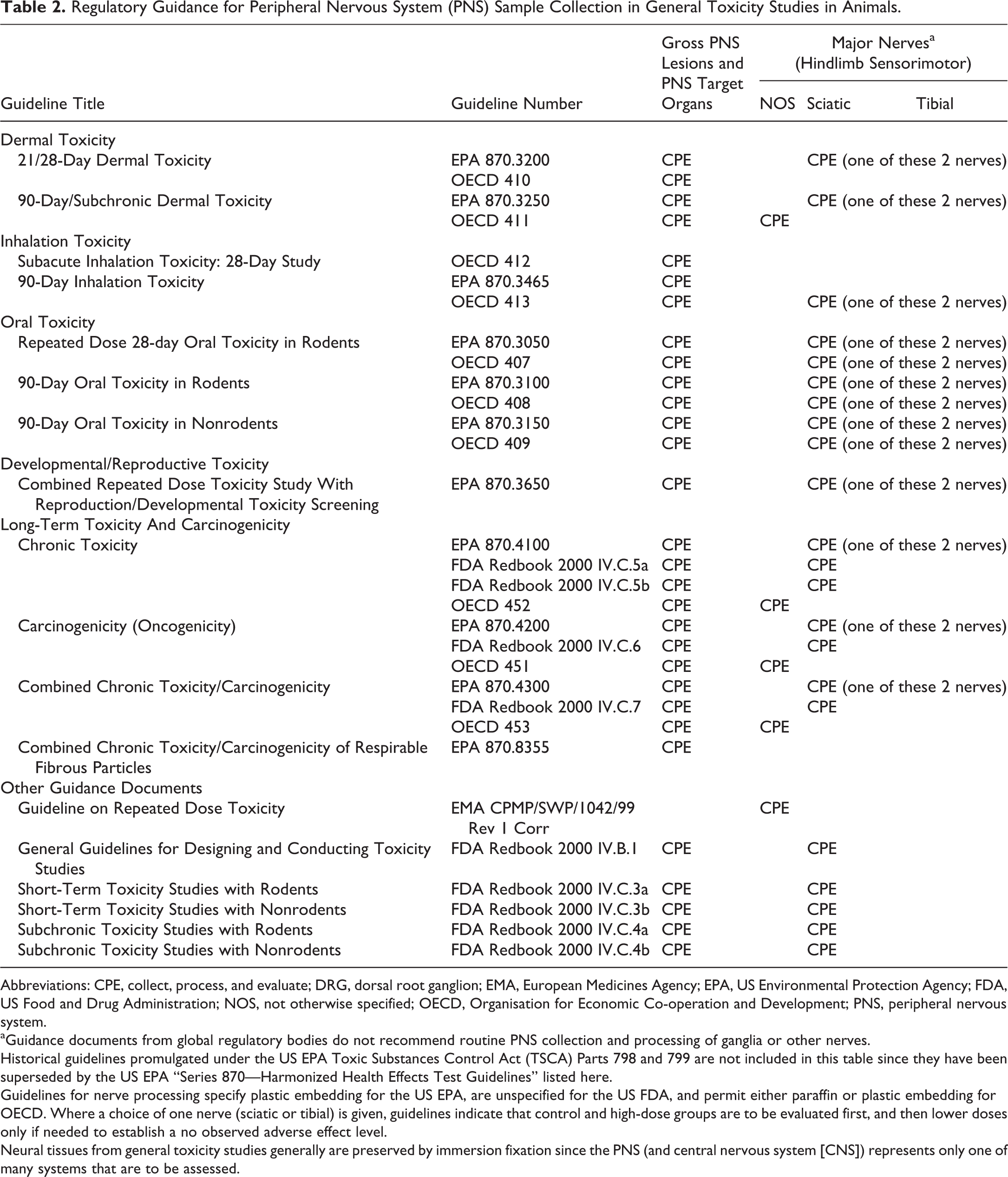
Abbreviations: CPE, collect, process, and evaluate; DRG, dorsal root ganglion; EMA, European Medicines Agency; EPA, US Environmental Protection Agency; FDA, US Food and Drug Administration; NOS, not otherwise specified; OECD, Organisation for Economic Co-operation and Development; PNS, peripheral nervous system.
aGuidance documents from global regulatory bodies do not recommend routine PNS collection and processing of ganglia or other nerves.
Historical guidelines promulgated under the US EPA Toxic Substances Control Act (TSCA) Parts 798 and 799 are not included in this table since they have been superseded by the US EPA “Series 870—Harmonized Health Effects Test Guidelines” listed here.
Guidelines for nerve processing specify plastic embedding for the US EPA, are unspecified for the US FDA, and permit either paraffin or plastic embedding for OECD. Where a choice of one nerve (sciatic or tibial) is given, guidelines indicate that control and high-dose groups are to be evaluated first, and then lower doses only if needed to establish a no observed adverse effect level.
Neural tissues from general toxicity studies generally are preserved by immersion fixation since the PNS (and central nervous system [CNS]) represents only one of many systems that are to be assessed.
Regulatory Guidance for Peripheral Nervous System (PNS) Sample Collection in Dedicated Neurotoxicity Studies in Animals.
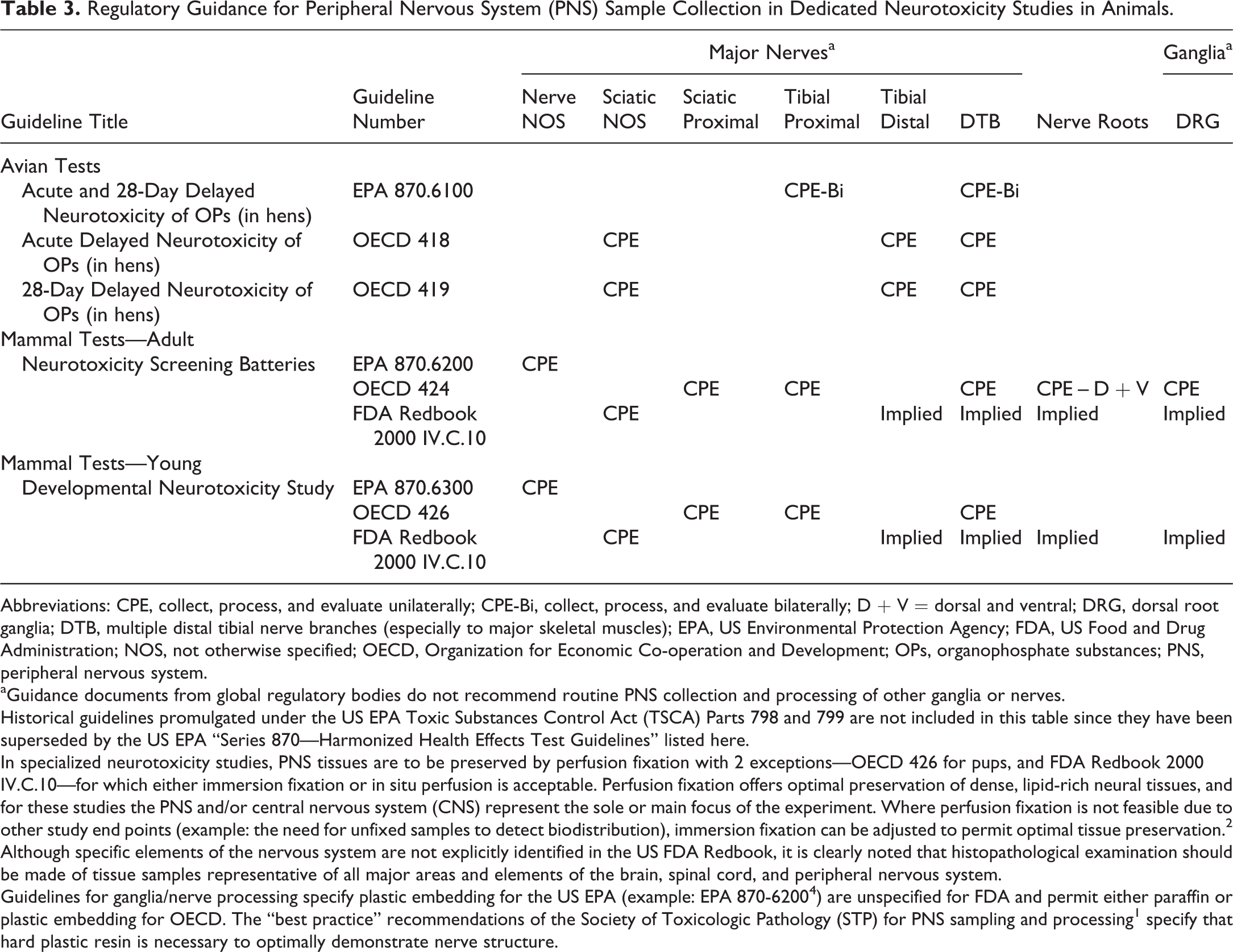
Abbreviations: CPE, collect, process, and evaluate unilaterally; CPE-Bi, collect, process, and evaluate bilaterally; D + V = dorsal and ventral; DRG, dorsal root ganglia; DTB, multiple distal tibial nerve branches (especially to major skeletal muscles); EPA, US Environmental Protection Agency; FDA, US Food and Drug Administration; NOS, not otherwise specified; OECD, Organization for Economic Co-operation and Development; OPs, organophosphate substances; PNS, peripheral nervous system.
aGuidance documents from global regulatory bodies do not recommend routine PNS collection and processing of other ganglia or nerves.
Historical guidelines promulgated under the US EPA Toxic Substances Control Act (TSCA) Parts 798 and 799 are not included in this table since they have been superseded by the US EPA “Series 870—Harmonized Health Effects Test Guidelines” listed here.
In specialized neurotoxicity studies, PNS tissues are to be preserved by perfusion fixation with 2 exceptions—OECD 426 for pups, and FDA Redbook 2000 IV.C.10—for which either immersion fixation or in situ perfusion is acceptable. Perfusion fixation offers optimal preservation of dense, lipid-rich neural tissues, and for these studies the PNS and/or central nervous system (CNS) represent the sole or main focus of the experiment. Where perfusion fixation is not feasible due to other study end points (example: the need for unfixed samples to detect biodistribution), immersion fixation can be adjusted to permit optimal tissue preservation. 2
Although specific elements of the nervous system are not explicitly identified in the US FDA Redbook, it is clearly noted that histopathological examination should be made of tissue samples representative of all major areas and elements of the brain, spinal cord, and peripheral nervous system.
Guidelines for ganglia/nerve processing specify plastic embedding for the US EPA (example: EPA 870-6200 4 ) are unspecified for FDA and permit either paraffin or plastic embedding for OECD. The “best practice” recommendations of the Society of Toxicologic Pathology (STP) for PNS sampling and processing 1 specify that hard plastic resin is necessary to optimally demonstrate nerve structure.
A common global approach to PNS neuropathology assessment is clearly deemed to be a meaningful objective by regulatory agencies around the globe. This fact is clear based on the general agreement among the various international guiding documents that describe the PNS neuropathology examination required for studies having a similar study design (Tables 2 and 3). However, many differences, though usually negligible, remain among equivalent guiding documents (eg, the recommendations for specialized mammalian neurotoxicity studies [Table 3]). We believe that such minor variations have no or minimal scientific impact and that many possible strategies are acceptable providing that they satisfy stated study objectives, applicable regulatory guidelines, and, where applicable, provide confidence to clinicians who will conduct future clinical trials.
The Society of Toxicologic Pathology (STP), comprised of professionals with formal training and extensive experience in evaluation of tissues from animal toxicity studies, has produced 2 “best practice” papers that offer expert technical advice regarding nervous system sampling, processing, and evaluation of Good Laboratory Practice (GLP)–compliant animal toxicity studies for safety assessment. These 2 documents were prepared by multinational panels of veterinary neuropathologists with decades of experience evaluating the nervous system. The first paper addressed the collection and evaluation of the CNS and PNS only for general toxicity studies, and advocated histopathologic analysis of the sciatic and/or tibial nerve as the minimal necessary PNS sampling. 3 A more recent paper recommends “best practices” for the morphologic evaluation of the sensorimotor and autonomic portions of the PNS in both general toxicity studies and specialized neurotoxicity studies. 1 This second paper was devised to provide investigators with the knowledge needed to meet regulatory guidance contained in all the international regulatory guidelines listed in this compilation, and it has been formally endorsed by 2 other societies of toxicologic pathology (British Society of Toxicological Pathology and Society of Toxicologic Pathology-India). The best practice recommendations for PNS sampling in this second paper (Table 4) consistently embrace but frequently exceed the baseline guidance provided in global regulatory guidelines (Tables 2 and 3). Moreover, these recommendations provide a high degree of flexibility in selecting which samples to collect and evaluate based on 4 different clinical presentations of PNS neurotoxicity: no known or anticipated toxicity (situation 1), suspected somatic PNS neurotoxicity (situation 2), suspected autonomic PNS neurotoxicity (situation 3), or known or expected somatic and/or autonomic PNS neurotoxicity (situation 4). 1 The first 3 situations reflect scenarios that occur in general toxicity studies where the PNS is one of many systems that are to be surveyed, while the last case (situation 4) covers dedicated neurotoxicity studies in which the CNS and/or PNS represent the major focus of the investigation. This distinction is key because dedicated neurotoxicity studies require more extensive tissue sampling as well as special fixation and processing conditions. That said, the rationale for routinely increasing the amount of PNS sampling as a “best practice” in all animal toxicity studies is that subjects are only available for necropsy once, and failure to obtain PNS samples at that time—even if only for archiving and not as a protocol-specified organ included in the initial histopathologic analysis—will result in their irretrievable loss, thus defeating conservation of resources and reducing the use of animals in toxicity studies. Necropsy, like death itself, is not reversible. In our experience, absence of data due to uncollected PNS tissues has required the conduct of entire new animal toxicity studies in order to answer questions raised by regulators during late-stage review of product registration applications.
Best Practice Recommendations for Peripheral Nervous System (PNS) Sampling in Animal Toxicity Studies.a
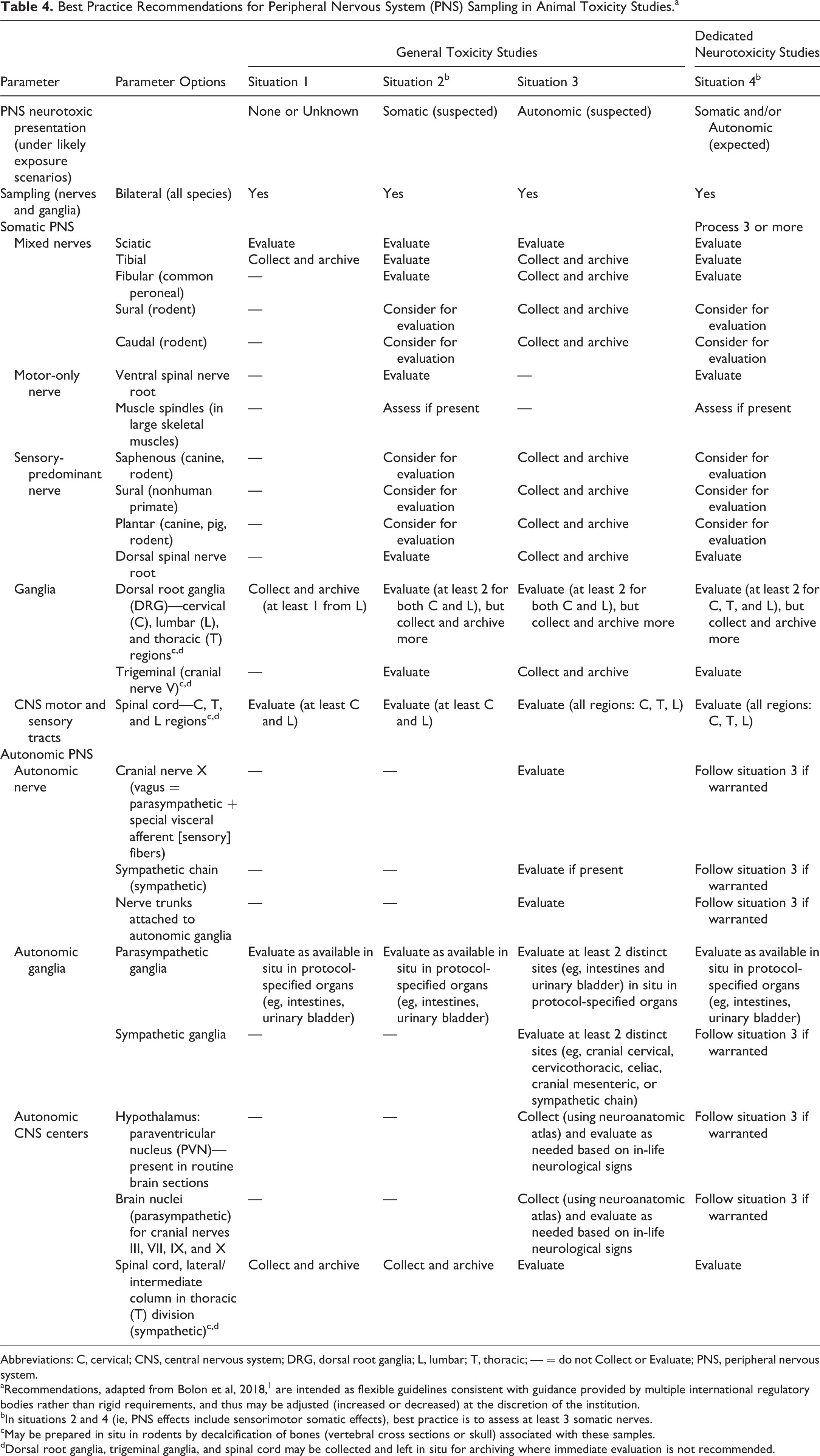
Abbreviations: C, cervical; CNS, central nervous system; DRG, dorsal root ganglia; L, lumbar; T, thoracic; — = do not Collect or Evaluate; PNS, peripheral nervous system.
aRecommendations, adapted from Bolon et al, 2018, 1 are intended as flexible guidelines consistent with guidance provided by multiple international regulatory bodies rather than rigid requirements, and thus may be adjusted (increased or decreased) at the discretion of the institution.
bIn situations 2 and 4 (ie, PNS effects include sensorimotor somatic effects), best practice is to assess at least 3 somatic nerves.
cMay be prepared in situ in rodents by decalcification of bones (vertebral cross sections or skull) associated with these samples.
dDorsal root ganglia, trigeminal ganglia, and spinal cord may be collected and left in situ for archiving where immediate evaluation is not recommended.
Where nerves and ganglia are to be examined, it is desirable to also evaluate white matter tracts in the spinal cord since they connect the PNS to the brain. 1,5 The various guidelines give recommendations ranging from the generic “spinal cord” or “spinal cord (3 sites)” to the more specific “spinal cord (cervical and lumbar intumescences)” or “spinal cord (cervical, thoracic, and lumbar divisions).” 6 Thus, the simplest means for addressing all global guidance documents is to routinely collect samples from the following spinal cord sites: cervical (C5-7 = cervical intumescence, the main origin for forelimb nerves); thoracic (T6-8 = middle); and lumbar (L4-5 = lumbar intumescence, the primary origin for hindlimb nerves). 3 The authors also recommend regular sampling above the cervical intumescence (eg, at C1-2) since this region of the spinal cord contains the largest fields of PNS-derived sensory axons in the dorsal funiculi. 1 Collection of these spinal cord divisions would apply to all animal species utilized for neurotoxicity testing; care will be necessary in acquiring the lumbar spinal cord since the location of the L4-5 cord segments within the vertebral column varies among species and generally occurs cranial to the L4-5 vertebrae. 3 Where autonomic dysfunction is noted, PNS assessment should also include evaluation of key brain and spinal cord autonomic centers—none of which are mentioned specifically in regulatory guidance documents, but several of which are readily available for analysis in routinely trimmed CNS specimens (Table 4). 1,3
Pathologists and toxicologists involved in animal toxicity studies are expected to be the primary users of this compilation of regulatory guidance/guidelines. However, this resource also should be beneficial to other scientists who devise, conduct, or provide regulatory review for PNS neuropathology data that have been obtained as part of translational studies in which animal-derived data are used to predict potential desirable (“efficacy”) or objectionable (“toxic”) responses in humans. 7
Footnotes
Authors’ Note
This document has been reviewed in accordance with the policies of the US Environmental Protection Agency and US Food and Drug Administration. All views reflect the opinions of the authors and should not be construed as representing views or policies of their employers, the US Environmental Protection Agency or the US Food and Drug Administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
