Abstract
Neuropathology analyses as end points during nonclinical efficacy and toxicity studies are challenging and require trained personnel and particular equipment to achieve optimal results. Accordingly, many regulatory agencies have produced explicit guidelines for designing and performing neuropathology assessments for nonclinical studies. This compilation of international regulatory guidance for toxicologic neuropathology end points represents a set of criteria recommended for general toxicity studies and specialized neurotoxicity studies that should facilitate the efforts of individuals who plan, perform, analyze, and report neuropathology evaluations in nonclinical toxicity studies.
Keywords
The anatomic, functional, molecular, and neurochemical complexities of various nervous system domains make neuropathologic investigations complicated in terms of both their design and their conduct. The experimental requirements needed to achieve optimal preservation and preparation of neural tissues dictate that personnel involved in such studies have a substantial degree of specialized training in neurohistology and neuropathologic evaluation. However, such technical expertise does not automatically result in an equivalent degree of familiarity with nation-specific regulatory considerations that usually drive the planning and performance of such studies.
This compilation of Internet links to current regulatory guidelines for nervous system sampling during general toxicity and specialized neurotoxicity nonclinical studies (Table 1) is intended as a resource for professionals engaged in neuropathologic assessment of tissues collected from nonclinical efficacy and toxicity studies. Tabular comparisons of the requirements for neuropathology assessment among these guidelines are given for both specialized neurotoxicity studies (Table 2) and general toxicity studies (Table 3). If evidence acquired during the course of a general toxicity study (e.g., neurological signs develop during the in-life phase, gross lesions are evident in neural organs by noninvasive imaging or at necropsy), the sponsor may choose to perform a more extensive neuropathology analysis than that which is strictly required in the guidance documents for general toxicity studies. Additional neuropathologic end points for inclusion in such situations may be selected following examination of the recommendations established in guidelines for specialized neurotoxicity studies.
Compilation of Current On-line Regulatory Guidelines for Neuropathology Assessment during Nonclinical Toxicity Studies


Comparison of recommendations for neural tissue collection in current regulatory guidelines for specialized neurotoxicity studies. Brain (listed in order from rostral to caudal)
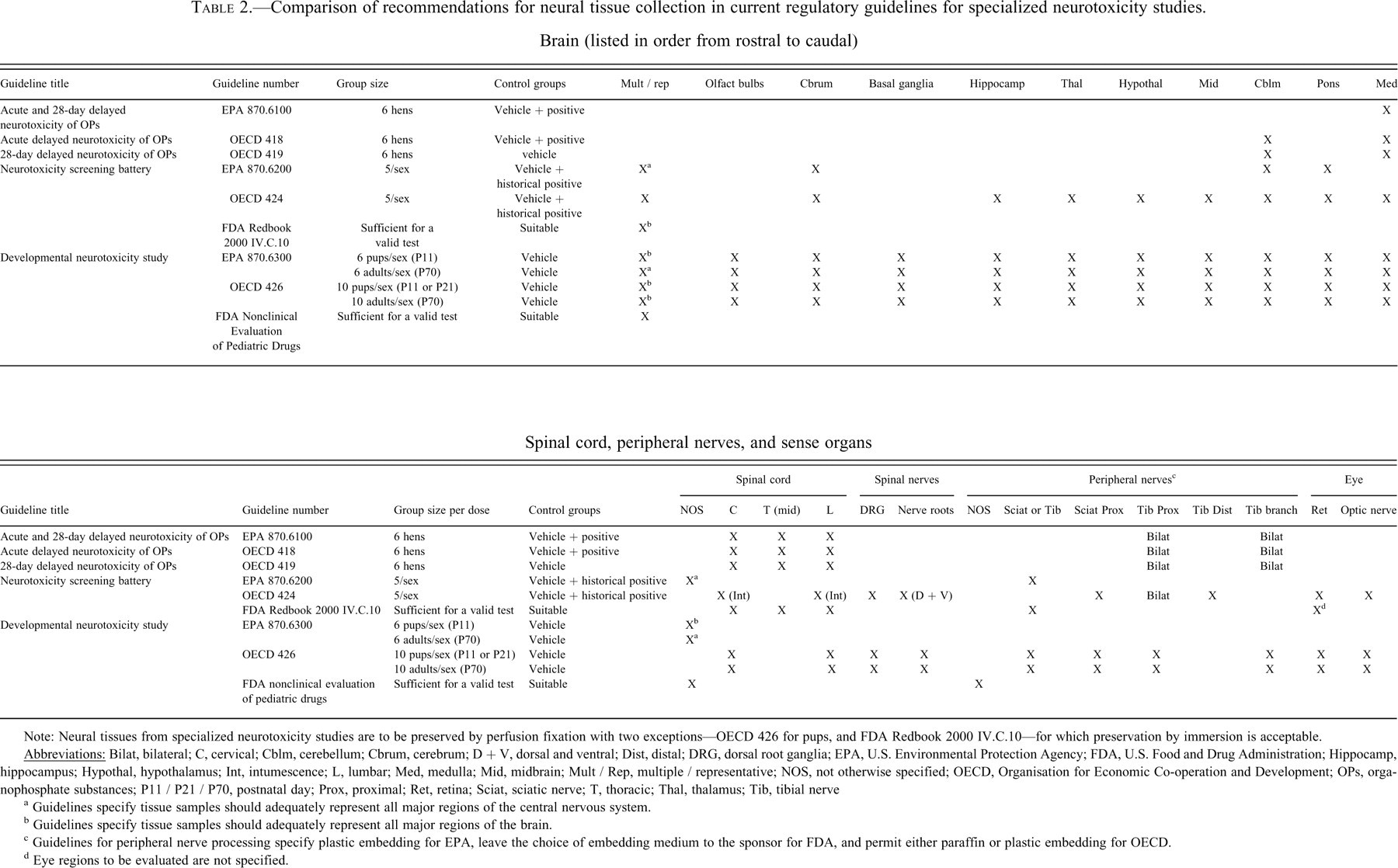

Note: Neural tissues from specialized neurotoxicity studies are to be preserved by perfusion fixation with two exceptions—OECD 426 for pups, and FDA Redbook 2000 IV.C.10—for which preservation by immersion is acceptable.
a Guidelines specify tissue samples should adequately represent all major regions of the central nervous system.
b Guidelines specify tissue samples should adequately represent all major regions of the brain.
c Guidelines for peripheral nerve processing specify plastic embedding for EPA, leave the choice of embedding medium to the sponsor for FDA, and permit either paraffin or plastic embedding for OECD.
d Eye regions to be evaluated are not specified.
Comparison of recommendations for neural tissue collection in current regulatory guidelines for general toxicity studies.
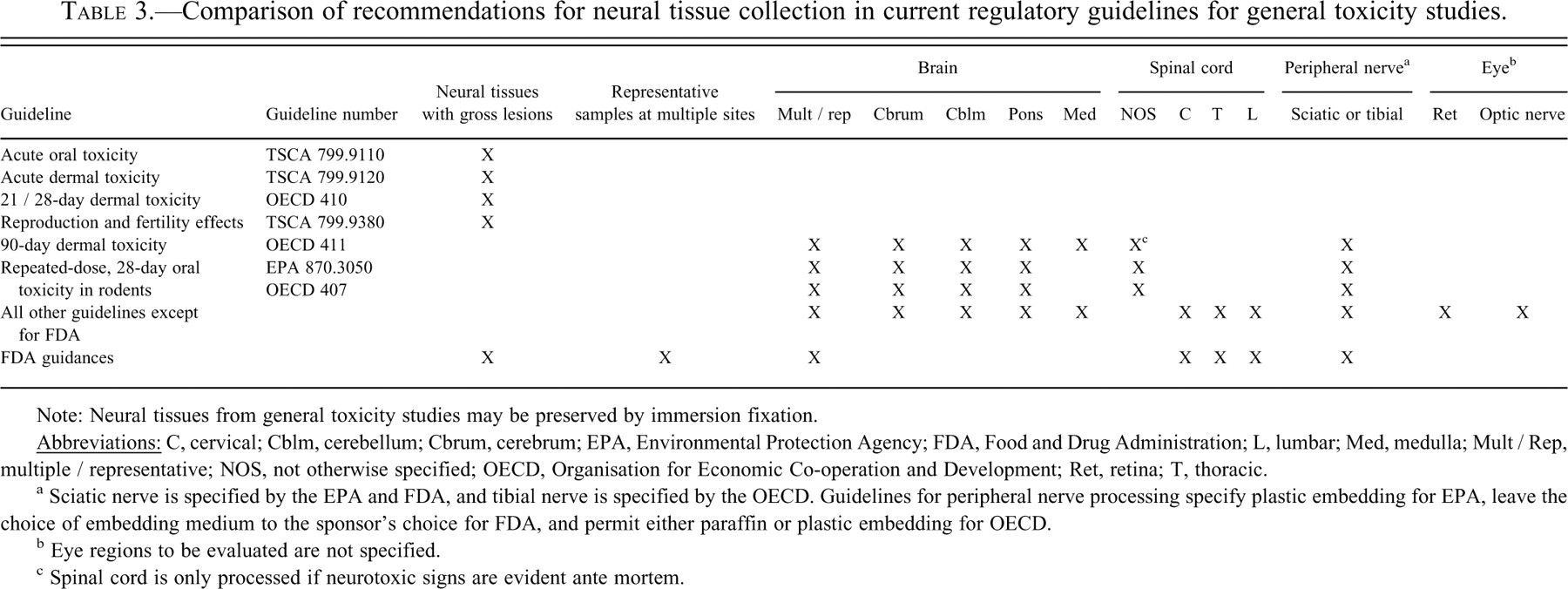
Note: Neural tissues from general toxicity studies may be preserved by immersion fixation.
a Sciatic nerve is specified by the EPA and FDA, and tibial nerve is specified by the OECD. Guidelines for peripheral nerve processing specify plastic embedding for EPA, leave the choice of embedding medium to the sponsor's choice for FDA, and permit either paraffin or plastic embedding for OECD.
b Eye regions to be evaluated are not specified.
c Spinal cord is only processed if neurotoxic signs are evident ante mortem.
Neuropathologists and neurotoxicologists are expected to be the primary users of this resource. However, it should also prove beneficial to other pathologists, toxicologists, and research scientists who design and/or perform neuropathology examinations as part of nonclinical toxicity studies. All guidance documents listed are available on-line at no cost, and they are current as of July 1, 2010.
The user must ensure that the correct neuropathology guidelines are followed for a particular application. For example, the U.S. Environmental Protection Agency (EPA) provides distinct guidance for special neuropathology testing methods, depending on the product: Series 870 harmonized test guidelines (http://www.epa.gov/oppts/pubs/frs/publications/Test_Guidelines/series870.htm) for agents regulated under the Federal Insecticide, Fungicide and Rodenticide Act or the Food Quality Protection Act, and 40 CFR Part 798 Test Rules (http://www.epa.gov/oppt/chemtest/pubs/790_799.html) for compounds considered under the Toxic Substances Control Act (TSCA). Both sets of EPA neuropathology guidelines fall under the auspices of the Office of Chemical Safety and Pollution Prevention (OCSPP, formerly the Office of Prevention, Pesticides and Toxic Substances [OPPTS]). The International Conference of Harmonization (ICH) guidelines provide internationally negotiated requirements for performing certain categories of nonclinical toxicity testing. These ICH guidances are used in the United States by the U.S. Food and Drug Administration (FDA), as well as in Europe and Japan.
A standard, global approach to neuropathologic assessment is clearly considered a worthwhile goal by regulatory agencies around the world. This fact is obvious given the harmony among the international guidance documents for neuropathologic evaluation mandated for studies having a comparable experimental design (Tables 2 and 3). However, numerous differences, albeit many relatively minor, still exist among closely related guidelines (e.g., for all the recommendations for specialized mammalian neurotoxicity studies [Table 2]). We believe such negligible differences have minimal scientific merit, and they instead serve chiefly to increase the cost and time required to conduct studies that must support simultaneous regulatory filings around the world. Accordingly, we recommend that a concerted effort be made in the near future to remove these slight variations, especially among the nearly identical guidance documents of the Organisation of Economic Co-operation and Development (OECD) and the EPA, thereby creating a truly global set of harmonized neurotoxicity testing guidelines.
