Abstract
The recent development of mouse cages equipped with monitoring wireless technology raised questions on the potential effects on animals induced by electromagnetic fields (EMFs) generated by electronic boards positioned underneath the cages. The aims of this study were to characterize the EMF produced by digitally ventilated cages (DVC) and perform a clinicopathological study on mice maintained in DVC for up to 1 year. The EMFs were measured in empty individually ventilated cages (IVC) and DVC. Male (n = 160) and female (n = 160) C57BL/6NCrl mice were randomly housed in IVC and DVC in a single rack, 4 mice per cage. Body weight and food and water consumption were recorded at 14-day intervals. At sacrifice (days 60, 120, 180, and 365), body and testes weight was measured, and necropsy, hematology, bone marrow cytology, histology, and immunohistochemistry for cleaved-caspase 3 on the testes were performed. Digitally ventilated cages produced extremely low-intensity electric fields ranging from 5 Hz to 3 GHz. No exposure-related clinical signs and mortality occurred. Occasional statistical differences in body weight, food and water consumption, hematology, bone marrow, and histopathology were recorded, but considered without biological or clinical relevance. In conclusion, long-term maintenance in DVC had no definite effects on C57BL/6NCrl mice.
Introduction
Electric fields are associated with the presence of electric charges, while magnetic fields are the result of the physical movement of an electric charge (electric current). 1 Exposure to electromagnetic fields (EMFs) from human-made sources have increased steadily over the past century due to the increased use of electricity and new technologies. In the past decades, there has been much debate about the potential detrimental effects produced on humans and animals from exposure to EMF, 2 as well as more recently about their potential therapeutic use (eg, pulsed EMF, tumor-treating fields), 3 –5 indicating that EMF can produce biological effects, regardless of the adverse or favorable outcomes. The mechanisms implicated in the generation of biological effects by EMF are so far not fully understood, but involve nonthermal effects, resulting from ions fluxes (ie, increased intracellular Ca2+), oxidative stress, free radicals, and melatonin suppression. 6 –8
Several epidemiological studies in humans pointed toward an association between long-term exposure to EMF of different frequency ranges (including extremely low frequency [ELF]-EMF ranging from 0 to 300 Hz, and radiofrequency [RF] ranging from 100 kHz to 300 GHz) and increased cancer risk, 9,10 but when all the studies are considered together, the association that results is weak, leading to the conclusion by different panels of experts of an overall limited or inadequate evidence of causal relationship between exposure to EMF and incidence of some tumors (ie, leukemia, brain, head, and breast cancers). 2,11 –13 Similarly, neurobehavior, neuroendocrine, and reproductive effects following exposure to EMF were inconsistently reported in epidemiological studies; in particular, no effects were detected on hemopoietic and germinal cells. 2
From the animal model point of view, studies performed on laboratory rodents on the impact of chronic exposure to EMF mostly failed to demonstrate an effect on tumor development, 14 –20 with only one study reporting the induction of lymphoproliferative diseases in 3 successive generations of CPW mice exposed to very strong (25 mT) 60-Hz EMF for up to 408 days, 21 and one study reporting a slight increase in risk for myeloid leukemia in female B6C3F1 mice after 15.5 months of exposure to 50 μT 50 Hz EMF. 22 Although a conclusive role of EMF on cancer was not yet demonstrated, evidence of biological effects of EMF on rodents was occasionally reported, in particular on nervous and neuroendocrine systems, 23 –25 testes (including sperm and testicular weight abnormalities, and increased germ cell apoptosis), 26 –29 and hematological parameters, 30 –32 even though other studies failed to replicate these effects. 33,34
Recently, Digitally ventilated cages (DVC; Tecniplast, Buguggiate, Italy) equipped with wireless technologies that use extremely low-intensity EMF were developed to enable continuous automated intracage monitoring and data capture of the animal activity and cage microenvironment. 35 In this context, the introduction of the DVC raises concerns about the potential effects on mice that could be induced by the EMF generated by the electronic boards positioned underneath the cages. Before introducing any change that may influence the animal welfare or the scientific outcomes of experiments, it is essential to assess the impact of this new technology on the animals. The aim of the study was to characterize the EMF produced by the DVC and perform a long-term trial (up to 1 year) on C57BL/6NCrl mice of both sexes to evaluate the effects of the exposure to this recently commercialized intracage wireless data collection technology.
Materials and Methods
Animals and Husbandry
The study was conducted in a distinct unit of the animal facility of the M. Negri Institute (Milan, Italy), not previously populated by animals and fumigated with 30% hydrogen peroxide before the beginning of the study. Specific pathogen-free C57BL/6NCrl mice (Charles River, Calco, Italy), aged 3 weeks at arrival, for a total of 160 male and 160 female mice were used in the first (main) experiment, and 40 female mice used in the second experiment. Upon arrival, mice were weighed and randomly divided into control and exposed groups on a single rack (DVC rack, Tecniplast), 4 mice per cage. Control mice were housed in standard Individually Ventilated Cages (IVC; GM500, Tecniplast), while exposed mice were housed in standard IVC equipped with DVC boards positioned underneath the cage and fixed to the rack. The chessboard distribution of IVC and DVC on the rack is shown in Figure 1. In the main experiment, groups were formed at the beginning of the study according to the scheduled time point of sacrifice (days 60, 120, 180, and 365). Each group included 20 male and 20 female control mice, and 20 male and 20 female exposed mice. Mice were allowed to acclimate for 1 week prior to the beginning of exposure (ie, when DVC boards were switched on).

Chessboard distribution of standard IVC (white) and DVC (gray) on the rack. Cages included in the central area delimited by the bold black line underwent radiate electromagnetic field measurements.
Individually ventilated cages ventilation was set up in positive pressure at 75 air changes per hour. Room environmental conditions were controlled with a temperature of 22°C (2°C) and a 55% (10%) relative humidity. The animal room had a controlled photoperiod of 12-h:12-h (light:dark) cycle. Mice were maintained on autoclaved Corn Cob bedding (Follador, Treviso, Italy), 150 g per cage. Routine cage changing occurred every 14 days. At cage change, random cage rotation was applied for both control and exposed groups. Mice were provided filtered autoclaved water and autoclaved diet (2014 S Envigo/Teklad global diet rodent maintenance, 14% protein) ad libitum. Water bottles and diet were weighed at cage change for the assessment of water and food consumption. Clinical signs were assessed daily. Body weights were recorded at cage change and before the sacrifice. Body weight gain in each group was calculated as follows: (mean body weight per cage at day of sacrifice − mean body weight per cage at randomization)/mean body weight per cage at randomization × 100.
Microbiological monitoring was performed on sampling dates (at days 60, 120, 180, and 365). Fur, mouth swabs, and fecal pellets were sampled from one control and one exposed cage, and a swab was taken from the exhaust prefilter of the Air Handling Unit (AHU). The prefilter was changed at each time point after swabbing. All samples were submitted to Charles River Laboratories (Wilmington, Massachusetts) for infectious disease polymerase chain reaction testing. Intracage samples were analyzed according to the Mouse Surveillance PRIA panel (at day 60) or the Mouse FELASA Complete PRIA panel (at 120, 180, and 365 days). Prefilters were analyzed according to the Environmental (EAD) Mouse Surveillance Plus PRIA panel (at days 60, 120, 180, and 365).
Mice were euthanized by carbon dioxide inhalation using a gradual 20% (vol/min) displacement rate 36 at days 60, 120, 180, and 365 after the beginning of the experiment. At sacrifice, blood was drawn from the heart and immediately placed in tubes containing ethylenediaminetetraacetic acid (EDTA), stored at room temperature, and transported to the laboratory, when they were employed to perform routine hematology, as described below. Bone marrow was collected from the femur using a 24G syringe needle and immediately smeared on glass slides and air dried. Then, mice underwent complete necropsy, as specified below.
The second experiment was carried out on 20 control and 20 exposed female mice, maintained for 60 days in the same conditions as aforedescribed, to verify the effects on neutrophils observed during the main experiment. At sacrifice, blood and bone marrow were collected and processed as described above.
Animals were maintained according to the guidelines set out in Commission Recommendation 2007/526/EC of June 18, 2007, for the accommodation and care of animals used for experimental and other scientific purposes, and were used in accordance with the Italian laws (D.L 26/2014), which enforces the Council Directive 2010/63/UE, on the approximation of laws, regulations, and administrative provisions of the member states regarding the protection of animals used for experimental and other scientific purposes. The study was approved by M. Negri Institute Ethical Committee. The Ethical Committee of M. Negri Institute uses the COST Action B-24 suggestions on Laboratory Animal science and welfare to evaluate any study involving laboratory animals.
Radiate EMF and Temperature Measurements
The DVC board is an electronic board with an array of 12 electrodes that are multiplexed by a central proximity sensor. The working principle is based on charging and discharging 4 times/s each electrode to measure electrical capacitance of all the materials (eg, plastic of the cage, bedding, animals) that are immersed into the generated low-intensity EMF. The entire DVC board power consumption is approximately 250 mW (powered at 5.4 VDC and absorbing 50 mA) distributed on a total surface of approximately 500 cm2.
To assess the intensity of EMF produced by the DVC board, radiate EMF measurements were performed in 10 IVC and 10 DVC with the board on, positioned in the central region of the rack, as shown in Figure 1. An electric and magnetic field strength analyzer (EHP-50, Narda STS, Italy) was used to measure EMF in the frequency range 0 Hz to 100 kHz, and an EMF strength meter (8053 2004/40 + ES330 Probe, Narda STS) was used to measure EMF in the frequency range 100 kHz to 3 GHz. The EMF were measured close to the floor of the cage, and results were reported as root mean square (V/m; electric field strength) and μT (magnetic field flux).
To assess the local heating produced by the DVC board, temperature measurements were performed in 4 IVC and 5 DVC positioned in the central region of the rack, as shown in Online Figure S1. Data loggers for temperature (174H mini temperature and humidity data logger, Testo Inc, West Chester, Pennsylvania) were placed on the floor of the cages and measured the temperature every 3 minutes for 2 hours. One additional data logger was placed on the top of the rack to measure the environmental temperature. Temperatures were measured when DVC was switched off and on.
Clinical Pathology (Hematology and Bone Marrow Evaluation)
The complete cell blood count (CBC) was performed on whole blood collected in EDTA using a laser-based cell counter (Sysmex XT-2000iV, Kungsbacka, Sweden) validated in mice. 37 Samples with evident clots were excluded from the hematological analysis. Differential leukocyte counts provided by the instrument were verified through microscopic evaluation on May Grünwald Giemsa-stained smears, allowing the differentiation between mature (segmented) neutrophils and immature (band) neutrophils. During microscopic evaluation, particular attention was paid to any possible morphological abnormality of erythrocytes, leukocytes, and platelets. The following parameters of the CBC were recorded: erythrocyte number (RBC), hemoglobin concentration (HGB), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelet number (PLT), number of total leukocytes (WBC), and of the different leukocyte populations: neutrophils, lymphocytes, monocytes, eosinophils, and basophils.
Bone marrow smears were stained with May Grünwald Giemsa and microscopically analyzed to perform a 500 nucleated cell count, in order to estimate the myeloid:erythroid (M:E) ratio. Within each cell population, the number of cells belonging to the proliferative (P) pool (composed by blasts able to divide) and the maturation (M) pool was recorded. Based on these numbers, the P:M ratio and the percentage of precursors on the total number of cells belonging to each cell lineage (%PE = percentage of erythroid precursors and %PM = percentage of myeloid precursors) were calculated. The percentage of lymphocytes (%L) and percentage of plasma cells (%PL) on the total number of counted cells were also recorded. Any possible abnormal morphology of cells of both proliferative and maturation pool was also recorded.
Gross Examination and Histopathology
At sacrifice, mice underwent complete necropsy, and any gross change was recorded. The testes weight was measured. For histological examination, the spleen, kidneys, adrenal glands, liver, small intestine (duodenum and jejunum), testes, mammary gland, brain (only at day 365), and organs with gross lesions were fixed in 10% neutral buffered formalin for at least 48 hours at room temperature, routinely processed for paraffin embedding, sectioned at 4 μm thickness, stained with hematoxylin-eosin, and evaluated under a light microscope. Grading of histopathological lesions detected in the liver, spleen, and kidneys was performed according to the grading system reported in Online Table S1.
Immunohistochemistry
Immunohistochemistry was performed on a single section of both testes from all examined mice to assess germ cell apoptosis. Four micrometer sections underwent deparaffinization and heat induced epitope retrieval for 40 minutes at 96°C (Dewax and HIER Buffer H, Thermo Scientific, Runcorn, UK, cat. No. TA-999-DHBH). Endogenous peroxidase activity was blocked by incubating sections in 3% H2O2 for 10 minutes. Slides were rinsed, incubated with phosphate-buffered saline containing 10% normal goat serum for 30 minutes at room temperature to reduce nonspecific background staining and then incubated for 1 hour at room temperature with a rabbit polyclonal anti–cleaved-caspase 3 antibody (clone Asp175, Cell Signalling, Leiden, The Netherlands, cat. no. 9661). Sections were then incubated with a biotinylated secondary antibody (goat anti-rabbit, Vector Laboratories, Orton Southgate, UK cat. No. VC-BA-1000-MM15), labeled by the avidin–biotin–peroxidase procedure (VECTASTAIN Elite ABC-Peroxidase Kit Standard, Vector Laboratories, cat. No. VC-PK-6100-KI01). The immunoreaction was visualized with 3,3′-diaminobenzidine (DAB, Peroxidase DAB Substrate Kit, Vector Laboratories, cat. No. VC-SK-4100-KI01) substrate and sections were counterstained with Mayer’s hematoxylin. Known positive control sections were included in each immunolabeling assay. For the evaluation of germ cell apoptosis, the percentage of the number of cleaved-caspase 3–positive cells/number of seminiferous tubules per section of both testes was calculated.
Statistical Analysis
Hematological, bone marrow, gross, histopathological, and immunohistochemical evaluations were performed in a blinded manner, that is, without knowledge of exposed and control groups. Data were analyzed using GraphPad Prism version 7.00 (GraphPad Software, La Jolla, California, www.graphpad.com). The results of EMF measurements in IVC and DVC, and the results obtained in control and exposed groups, within each sex, regarding body weight, food and water consumption, hematological parameters, bone marrow cells counts, histopathological grade, and germ cell apoptosis were compared to each other using Mann-Whitney U test. Prism 7.00 handles 2 identical values assigning them to the same rank and computing an exact P value. Fisher exact test was used to compare the prevalence of mortality, gross, and histopathological findings between control and exposed groups within each sex. P values <.05 were considered statistically significant.
Results
Radiate EMF and Temperature Measurements
Results of radiate EMF measurements are reported in Tables 1 and 2. Overall, the detected EMF values were extremely low in intensity in empty IVC and DVC. In DVC, there was a significant increase in the electric field values as compared to adjacent IVC, at all the frequency ranges examined. No significant differences between DVC and IVC were observed in the induction of magnetic fields.
Results of Electric Field Measurements in Empty IVC and DVC.a
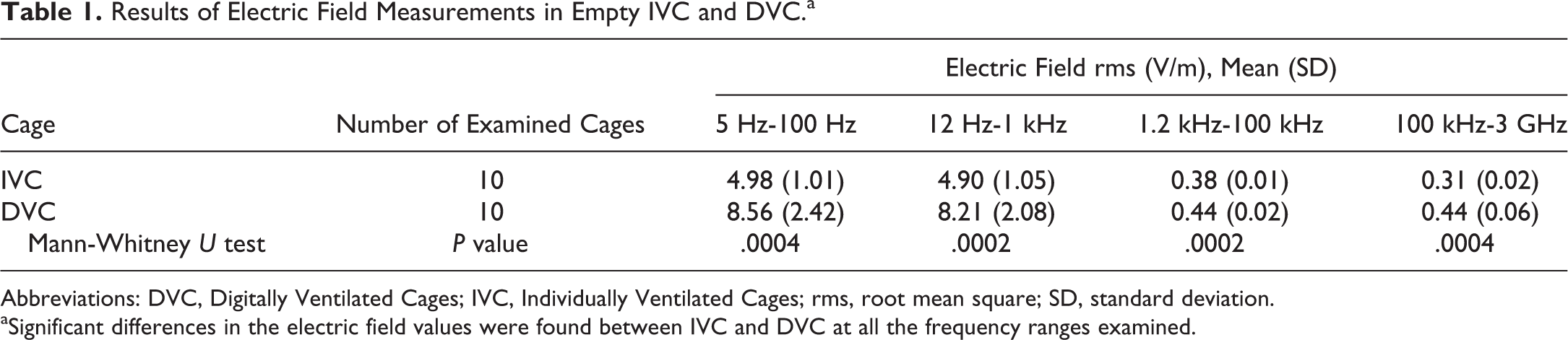
Abbreviations: DVC, Digitally Ventilated Cages; IVC, Individually Ventilated Cages; rms, root mean square; SD, standard deviation.
aSignificant differences in the electric field values were found between IVC and DVC at all the frequency ranges examined.
Results of Magnetic Field Measurements in Empty IVC and DVC.a
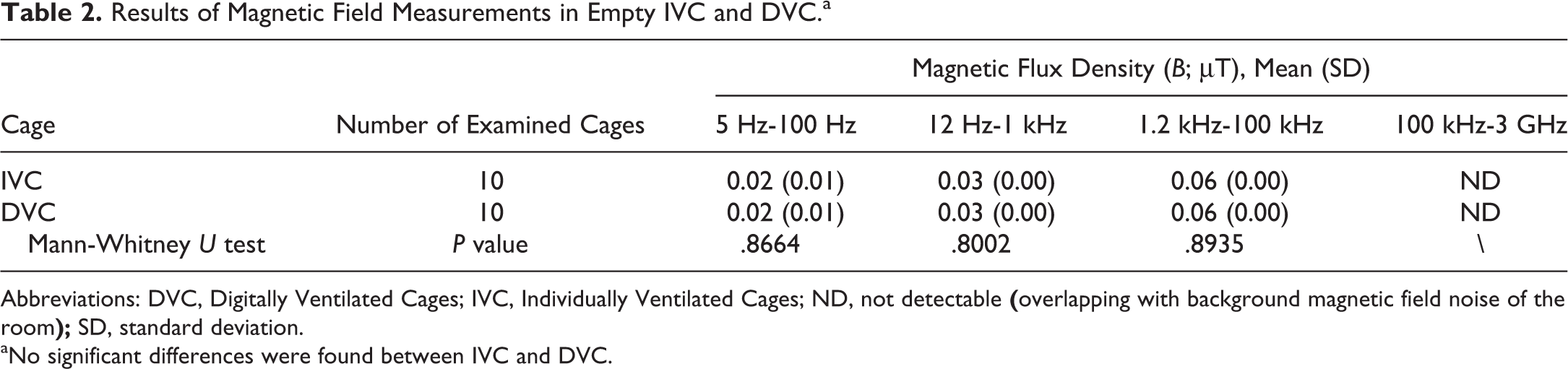
Abbreviations: DVC, Digitally Ventilated Cages; IVC, Individually Ventilated Cages; ND, not detectable
aNo significant differences were found between IVC and DVC.
Results of temperatures measured in IVC and DVC are reported in Table 3. The temperature at the bottom of the DVC was not significantly different from that at the bottom of the IVC when the DVC was switched on (Mann-Whitney U test, P = .7857).
Results of Temperature Evaluation in the Environment, and Within IVC and DVC, Measured at the Bottom of the Cage.a
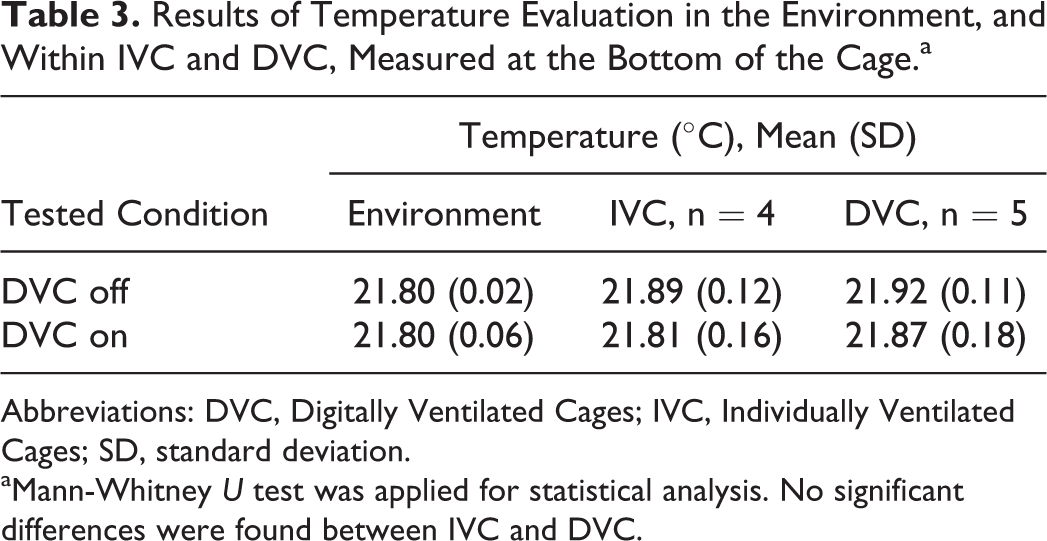
Abbreviations: DVC, Digitally Ventilated Cages; IVC, Individually Ventilated Cages; SD, standard deviation.
aMann-Whitney U test was applied for statistical analysis. No significant differences were found between IVC and DVC.
Clinical Signs, Body Weights, Food and Water Consumption, Mortality, and Microbiological Monitoring
No clinical signs were noted during the whole period of the experiment in control or exposed mice of either sex. The body weight evaluation resulted in no significant differences between control and exposed groups of female mice at all time points and male mice at days 60, 120, and 365. At day 180, exposed male mice had a significantly reduced body weight compared to control male mice (control: 39.5 [3.2] g; exposed: 34.3 [3.9] g, Mann-Whitney U test: P = .0002; Figure 2). Despite the difference in the body weight, there was no significant difference in body weight gain between control and exposed male mice at day 180 (control: +136.12%; exposed +117.62%; P = .0556; Table 4), and there was no significant difference in the body weight of male mice maintained up to 365 days when measured at day 180 (control: 37.4 [3.3] g; exposed: 36.2 [4.4] g, Mann-Whitney U test: P = 0.2110). Significant differences between control and exposed animals were found for water consumption in females at day 180 (P = .0256) and day 365 (P = .0055), and in males at day 365 (P = .0296; Figure 3) and for food consumption in females at day 365 (P < .0001) and in males at day 180 (P < .0001; Figure 4).
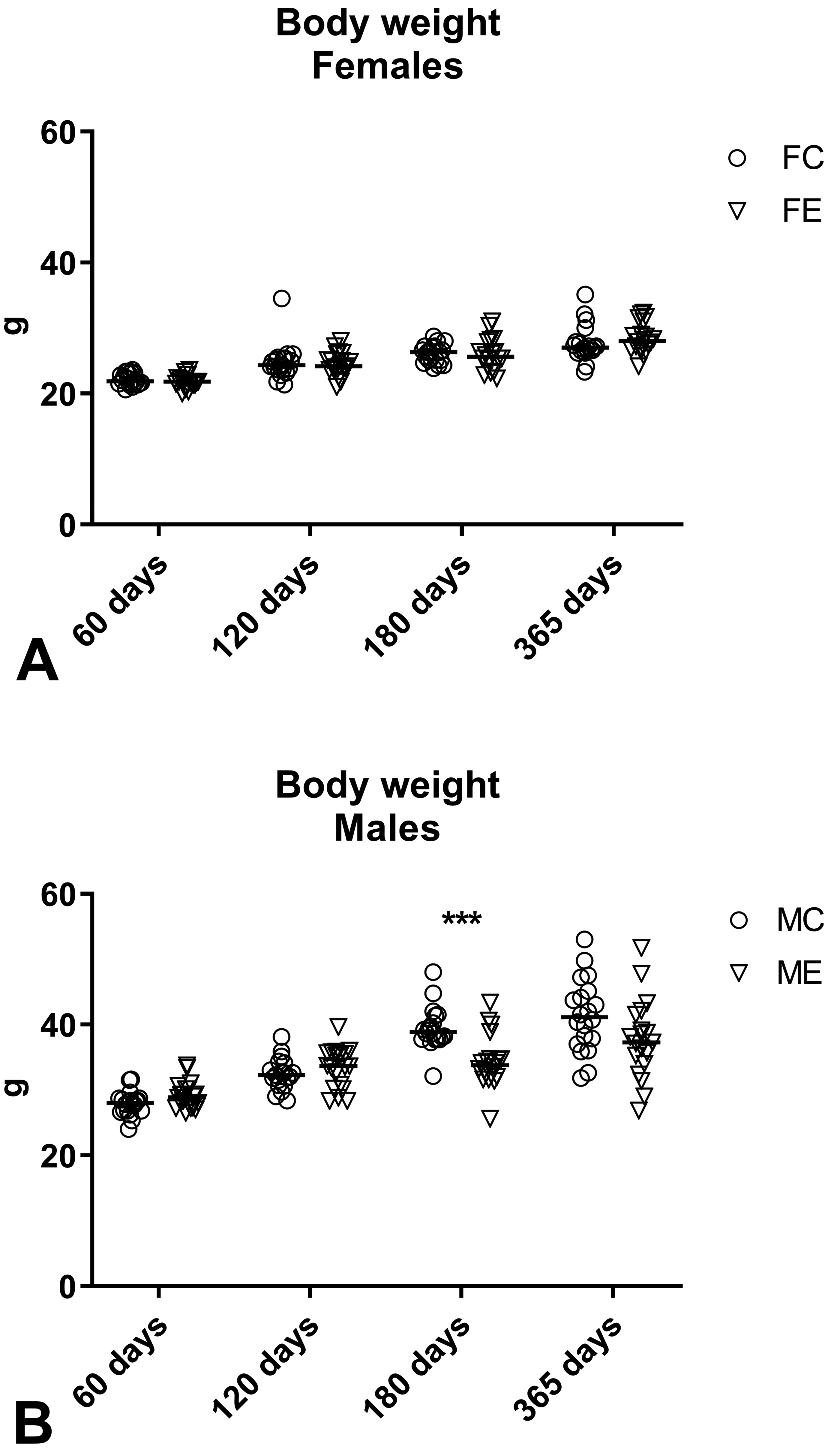
Body weight of female (A) and male (B) mice measured at days 60, 120, 180, and 365 after the beginning of the exposure. Data are reported as individual values and median. Mann-Whitney U test was applied for statistical analysis. ***P = .0002.
Body Weight Gain in Female and Male Mice at Days 60, 120, 180, and 365.a.
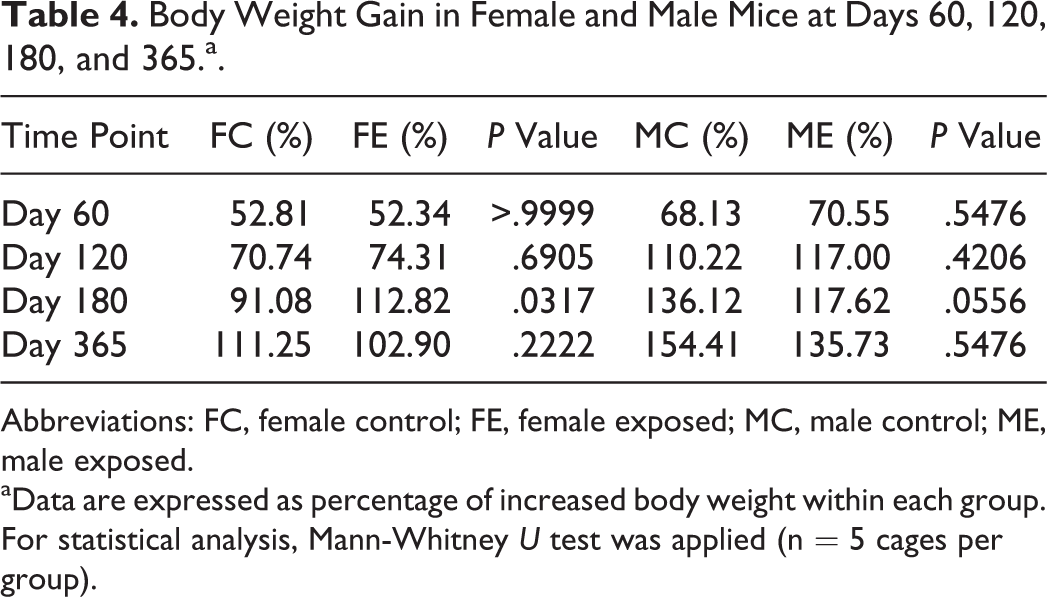
Abbreviations: FC, female control; FE, female exposed; MC, male control; ME, male exposed.
aData are expressed as percentage of increased body weight within each group. For statistical analysis, Mann-Whitney U test was applied (n = 5 cages per group).
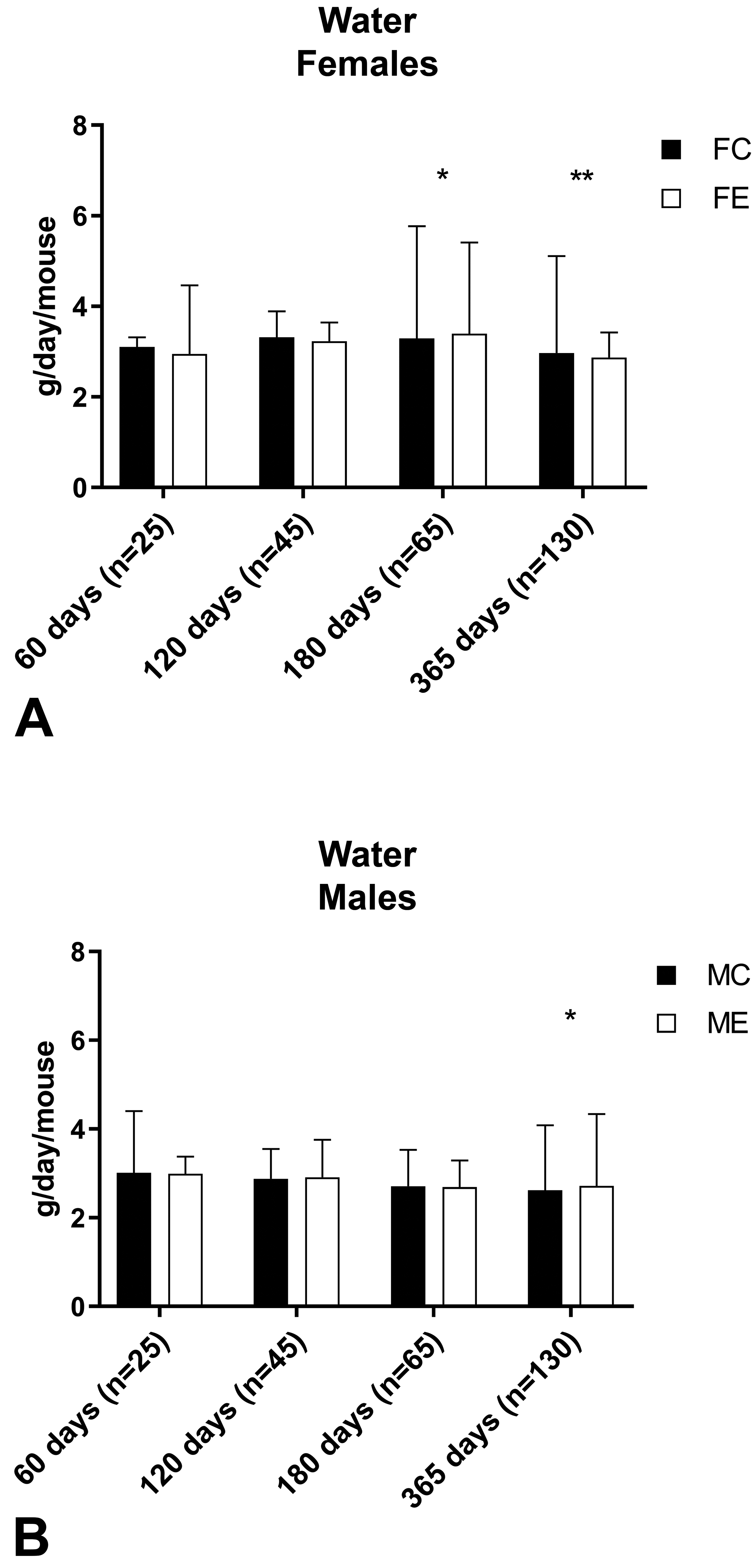
Water consumption in female (A) and male (B) mice measured at days 60, 120, 180, and 365. Data are reported as median value and range. Mann-Whitney U test was applied for statistical analysis (n = number of cage changes). *P < .05; **P < .01.
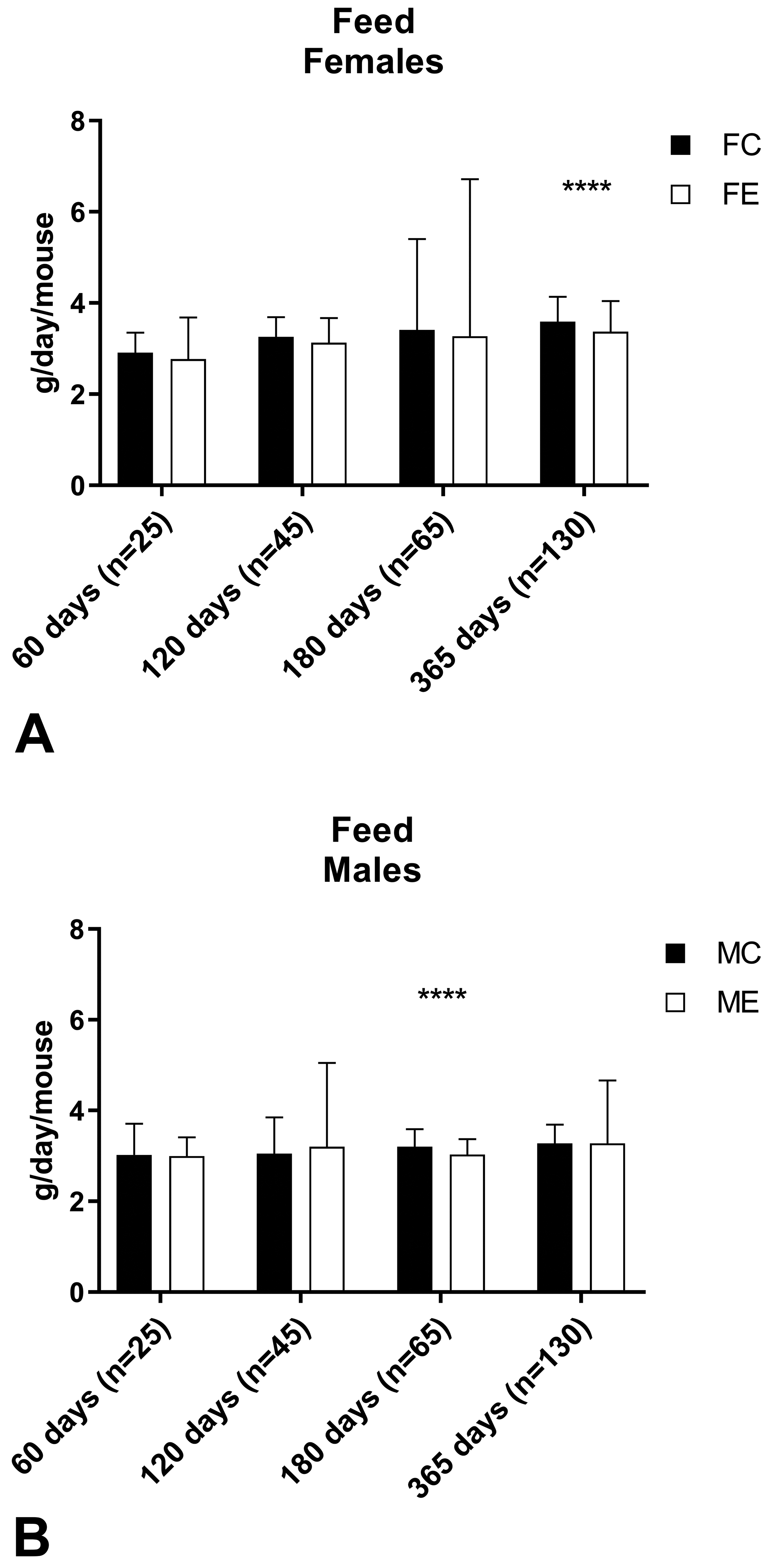
Feed consumption in female (A) and male (B) mice measured at days 60, 120, 180, and 365. Data are reported as median value and range. Mann-Whitney U test was applied for statistical analysis (n = number of cage changes). ****P < .0001.
During the study, 6 mice died (Table 5). In 3 cases, it was not possible to assess the cause of death due to advanced postmortal changes or cannibalism. Two cases were related to husbandry issues (eg, bottle obstruction), and 1 was a case of malocclusion. Regardless of the (determined/undetermined) cause of death, no association of mortality with treatment was found.
Prevalence of Mortality in Female and Male Mice at Days 60, 120, 180, and 365.a
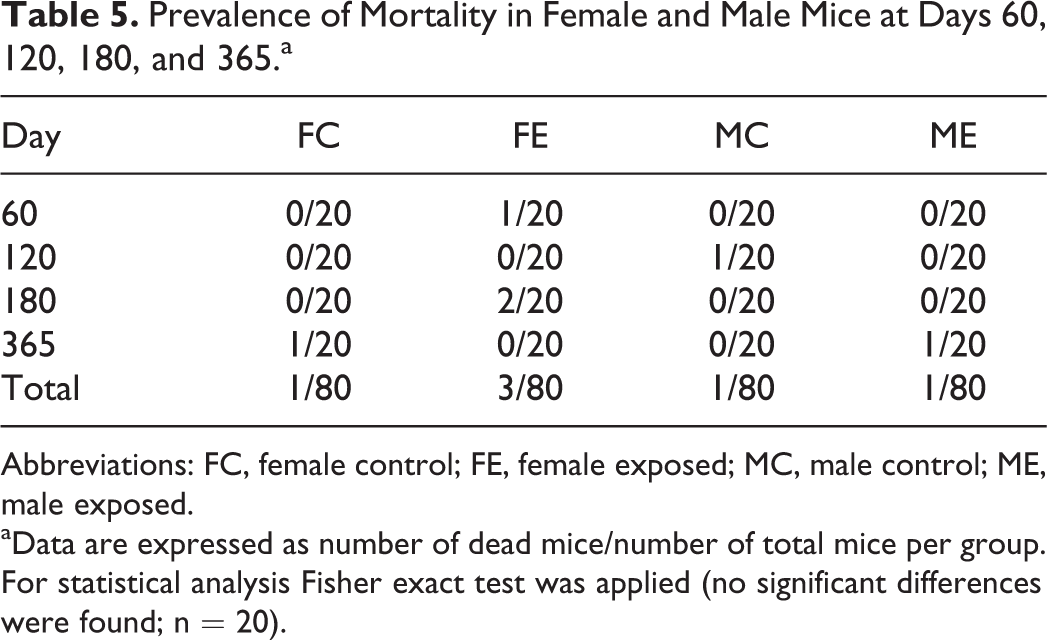
Abbreviations: FC, female control; FE, female exposed; MC, male control; ME, male exposed.
aData are expressed as number of dead mice/number of total mice per group. For statistical analysis Fisher exact test was applied (no significant differences were found; n = 20).
Health monitoring revealed the presence of Staphylococcus aureus and Klebsiella oxytoca from intracage sampling at day 60 (no more tested at the following time points) and in the prefilters of the AHU (S aureus at days 60, 120, and 180; K oxytoca at days 60 and 180; Online Table S2).
Hematology and Bone Marrow Evaluation
Complete hematology and bone marrow results are reported in Online Tables S3-S6. Statistical analysis revealed some significant differences between control and exposed mice, summarized in Table 6.
Summary of Results of Hematological and Bone Marrow Examination at Days 60, 120, 180, and 365 of Exposure That Resulted in a Significant Difference Between Control and Exposed Mice.a
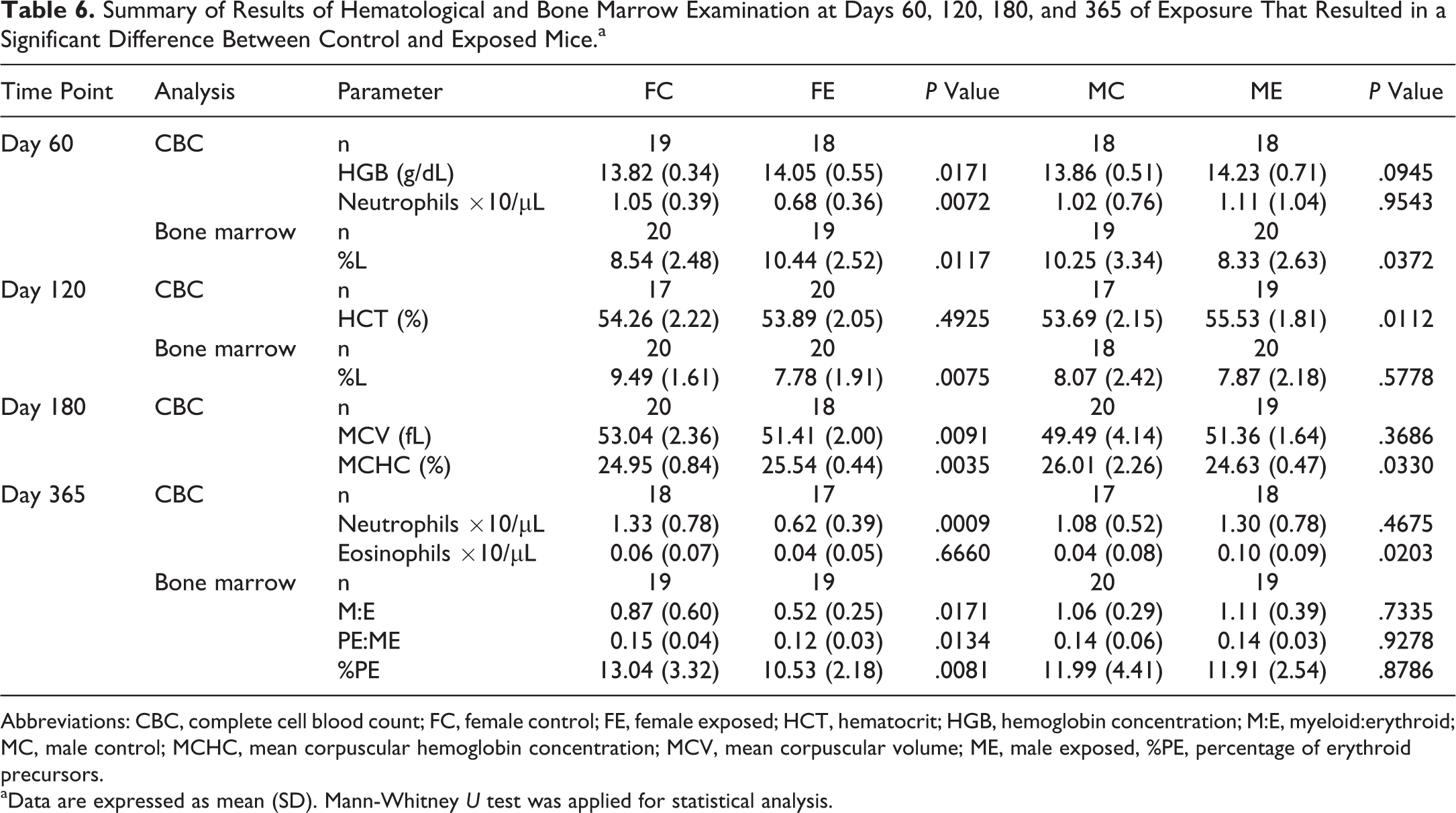
Abbreviations: CBC, complete cell blood count; FC, female control; FE, female exposed; HCT, hematocrit; HGB, hemoglobin concentration; M:E, myeloid:erythroid; MC, male control; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; ME, male exposed, %PE, percentage of erythroid precursors.
aData are expressed as mean (SD). Mann-Whitney U test was applied for statistical analysis.
In a second replicate experiment carried out by exposing a new additional group of female mice for 60 days, no significant difference in the neutrophil count was detected between control and exposed animals (Figure 5).
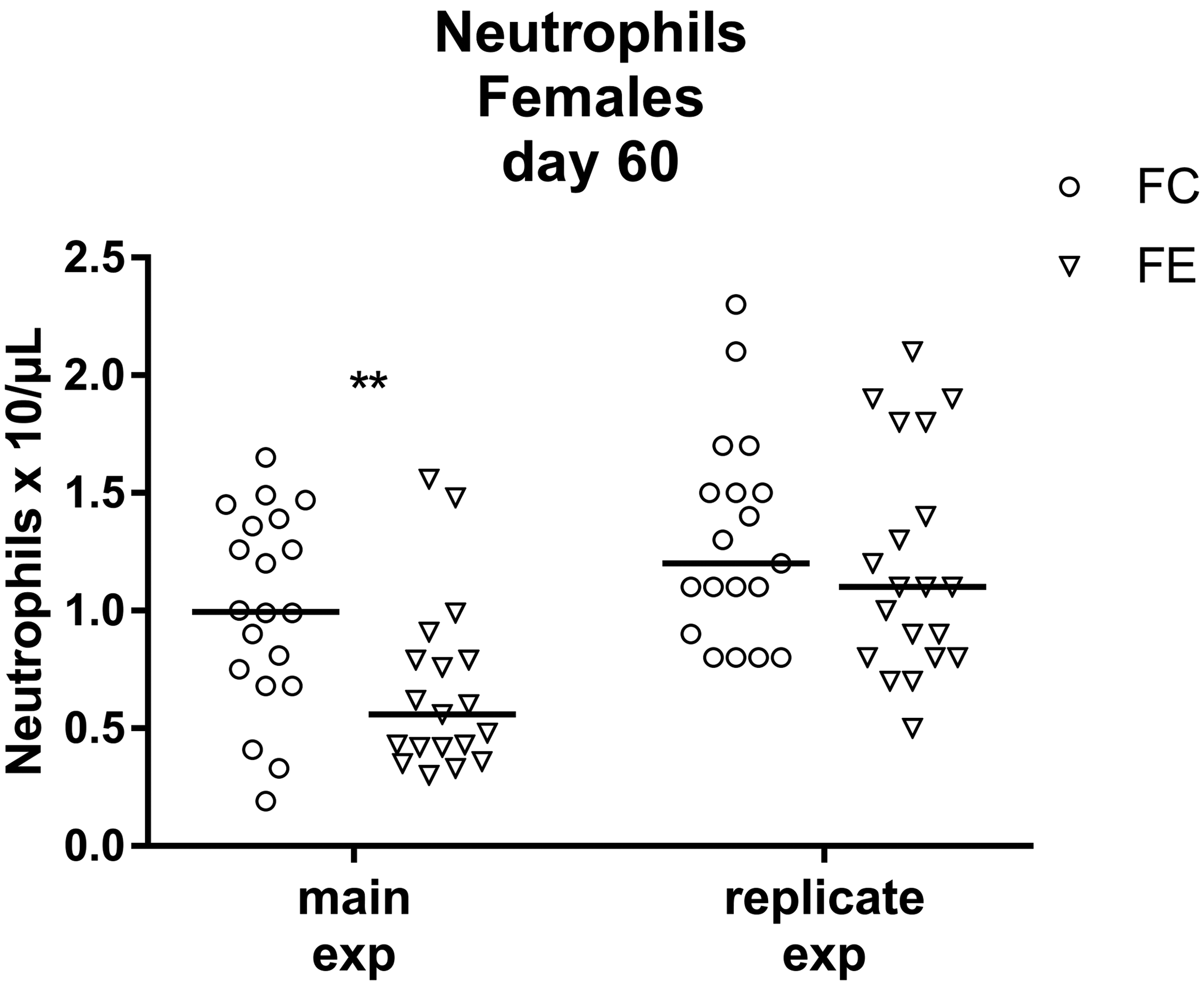
Neutrophils were significantly reduced in exposed female mice at day 60 of exposure in the main experiment (**P = .0072), but in a replicate experiment no significant difference was observed (P = .2594). Data are reported as individual values and median. Mann-Whitney U test was applied for statistical analysis (n = 18-20).
Pathology
Gross lesions
At necropsy, gross changes were only occasionally observed at all time points (Table 7) and included splenic melanosis, splenomegaly, unilateral hydronephrosis, uterus enlargement, preputial gland adenitis, submandibular lymph nodes, and salivary glands enlargement, and a single case of a retroauricular mass. Barbering lesions in both aged male and female mice and kyphosis in aged female mice were also seen. There was no significant effect of exposure to DVC on the prevalence of the observed gross lesions in both sexes.
Prevalence of Gross Lesions at Days 60, 120, 180, and 365 of Exposure.a
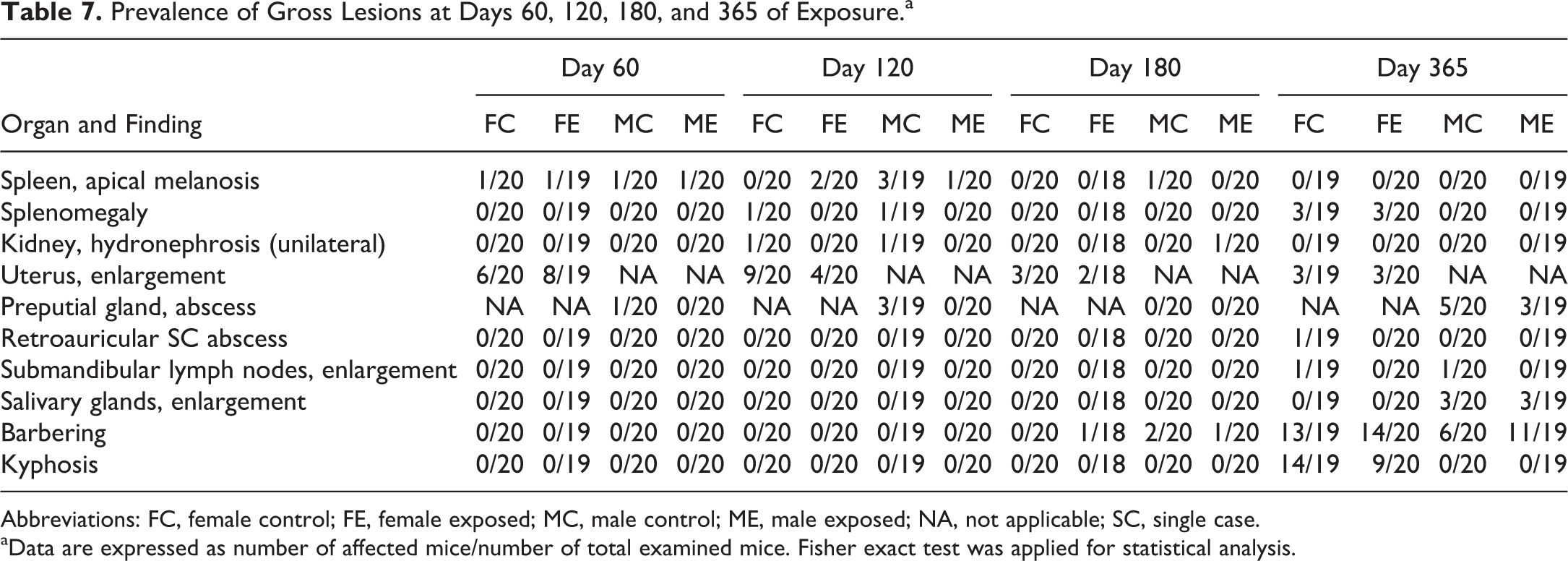
Abbreviations: FC, female control; FE, female exposed; MC, male control; ME, male exposed; NA, not applicable; SC, single case.
aData are expressed as number of affected mice/number of total examined mice. Fisher exact test was applied for statistical analysis.
Histopathology
Prevalence of histopathological findings in examined organs is reported in Table 8.
Prevalence of Histopathological Findings at Days 60, 120, 180, and 365 of Exposure.a
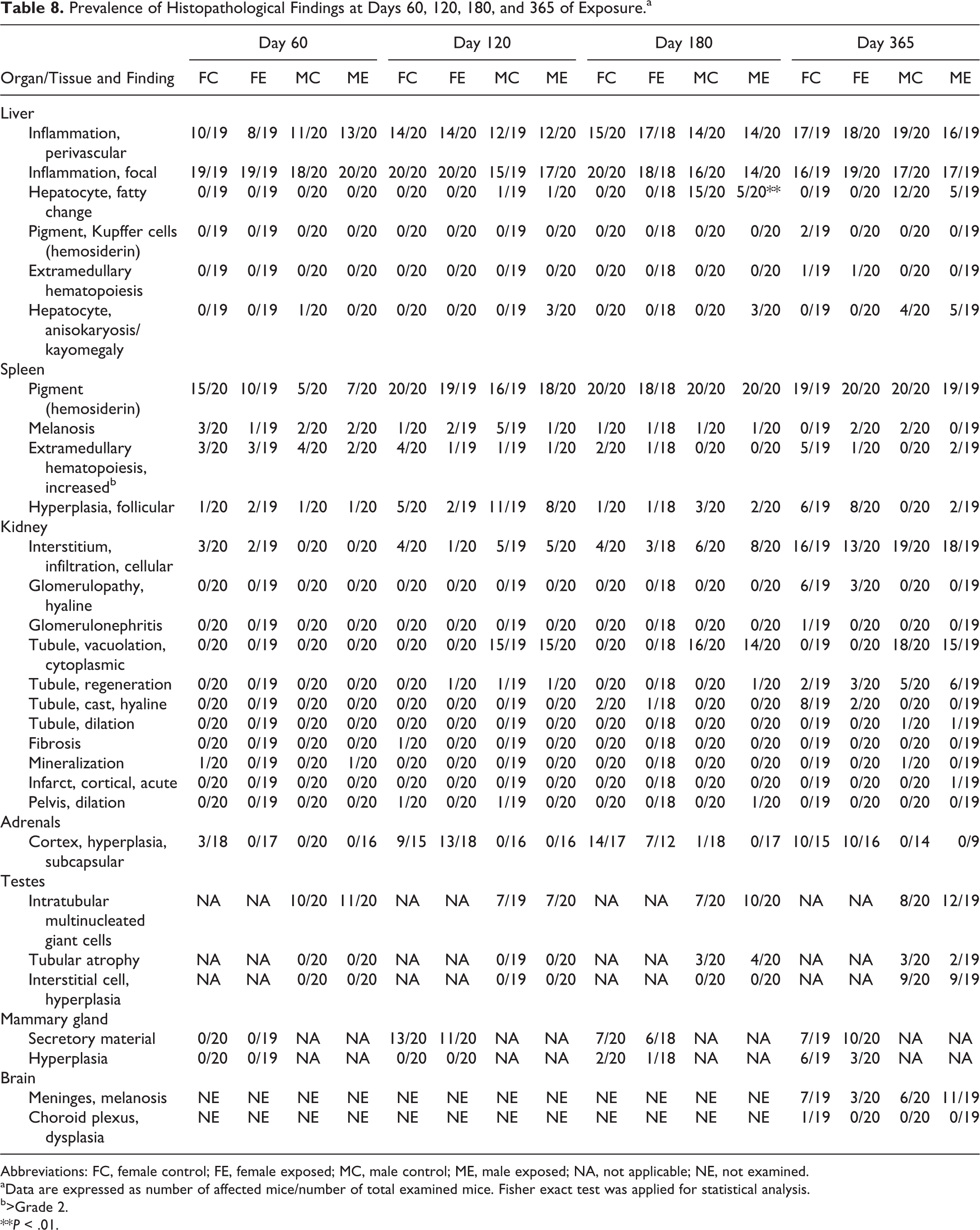
Abbreviations: FC, female control; FE, female exposed; MC, male control; ME, male exposed; NA, not applicable; NE, not examined.
aData are expressed as number of affected mice/number of total examined mice. Fisher exact test was applied for statistical analysis.
b>Grade 2.
**P < .01.
In the liver, the most common finding was the presence of inflammatory cell infiltrates, either perivascular or randomly distributed throughout the liver parenchyma, the latter variably associated with single-cell hepatocellular necrosis. The prevalence of these findings was similar in control and exposed groups at all examined time points. Although the prevalence of mice affected by perivascular inflammatory cell infiltration was similar regardless of the age and sex, the degree of severity increased with age, and at day 365 the infiltrates were so well organized to be consistent with tertiary lymphoid structures, but no exposure-related effects on the grading of these perivascular infiltrates was found (Online Table S7). Fatty change was observed only in male mice after day 120, and at day 180, there was a significant difference in its prevalence (control: 15/20; exposed: 5/20; Fisher exact test: P = .0038) and severity (control: median grade = 1; exposed: median grade = 0; Mann-Whitney U test: P = .0023). Other findings (pigment [hemosiderin] accumulation within Kupffer cells, hepatic extramedullary hematopoiesis, hepatocyte karyomegaly) were only occasionally present after day 180 of exposure, and exposure had no effect on the prevalence of these findings.
In the kidneys, tubular vacuolation was observed only in male mice after day 120, and the presence of hyaline casts and hyaline glomerulopathy was found only in female mice after days 180 and 365, respectively. At day 365, there was a significant reduction of the prevalence of intratubular hyaline casts in exposed female mice as compared to control ones (control: 8/19; exposed: 2/20; Fisher exact test: P = .0310). Interstitial inflammatory cell infiltrates, mainly composed of lymphocytes and plasma cells, were found in the cortex and around the pelvis. They increased in prevalence with age and were more frequent in male mice than in female mice, but no exposure-related effects on the grading of these infiltrates were found.
In the spleen, the prevalence of histopathological findings was similar in control and exposed groups at all examined time points (Table 7). The presence of macrophages with pigment accumulation (consistent with hemosiderin) was the most common finding, increased in severity with age, and was more abundant in female than in male mice. No exposure-related effect was observed in the grade of severity of follicular hyperplasia, pigment (hemosiderin) accumulation, and melanosis, while at day 120, splenic extramedullary hematopoiesis resulted significantly higher in control female mice than in exposed female mice despite the same median value (control: median grade = 2; exposed: median grade = 2; Mann-Whitney U test: P = .0162; Online Table S7).
In the adrenals, testes, and mammary glands, the prevalence of histopathological findings was similar in control and exposed groups at all examined time points (Table 7). No relevant histopathological findings were observed in the small intestine of examined mice.
In the brain of animals at day 365, the most common finding was meningeal melanosis with similar prevalence in control and exposed groups. Only a single case of focal choroid plexus dysplasia was observed in the lateral ventricle of a control female mouse.
In a single exposed female mouse at day 365, a histiocytic sarcoma involving the spleen and renal lymph node was found, associated with hepatic extramedullary hematopoiesis and presence of atypical mononuclear cells detected by bone marrow cytology.
The examination of unscheduled grossly affected organs revealed the presence of sporadic cases of preputial gland suppurative adenitis, and occasional cases of uterine mucometra and cystic endometrial hyperplasia, depending on the age of examination. The single case of retroauricular mass was an abscess with intralesional unidentified foreign body material. Submandibular lymph nodes enlargement was consistent with nodal reactive follicular hyperplasia, and salivary glands enlargement with the presence of perivascular/interstitial chronic inflammatory cell infiltrates.
Testes
No significant differences were found in the testes relative weight between control and exposed male mice at all examined time points (Figure 6), while testes absolute weight was significantly reduced in exposed mice at day 180. Histologically, the most frequent finding at all time points was the presence of rare intratubular multinucleated giant cells (1-2 per section). Tubular atrophy and interstitial cell hyperplasia were observed after days 180 and 365, respectively. Prevalence of testicular histopathological findings was similar between control and exposed animals (Table 7).
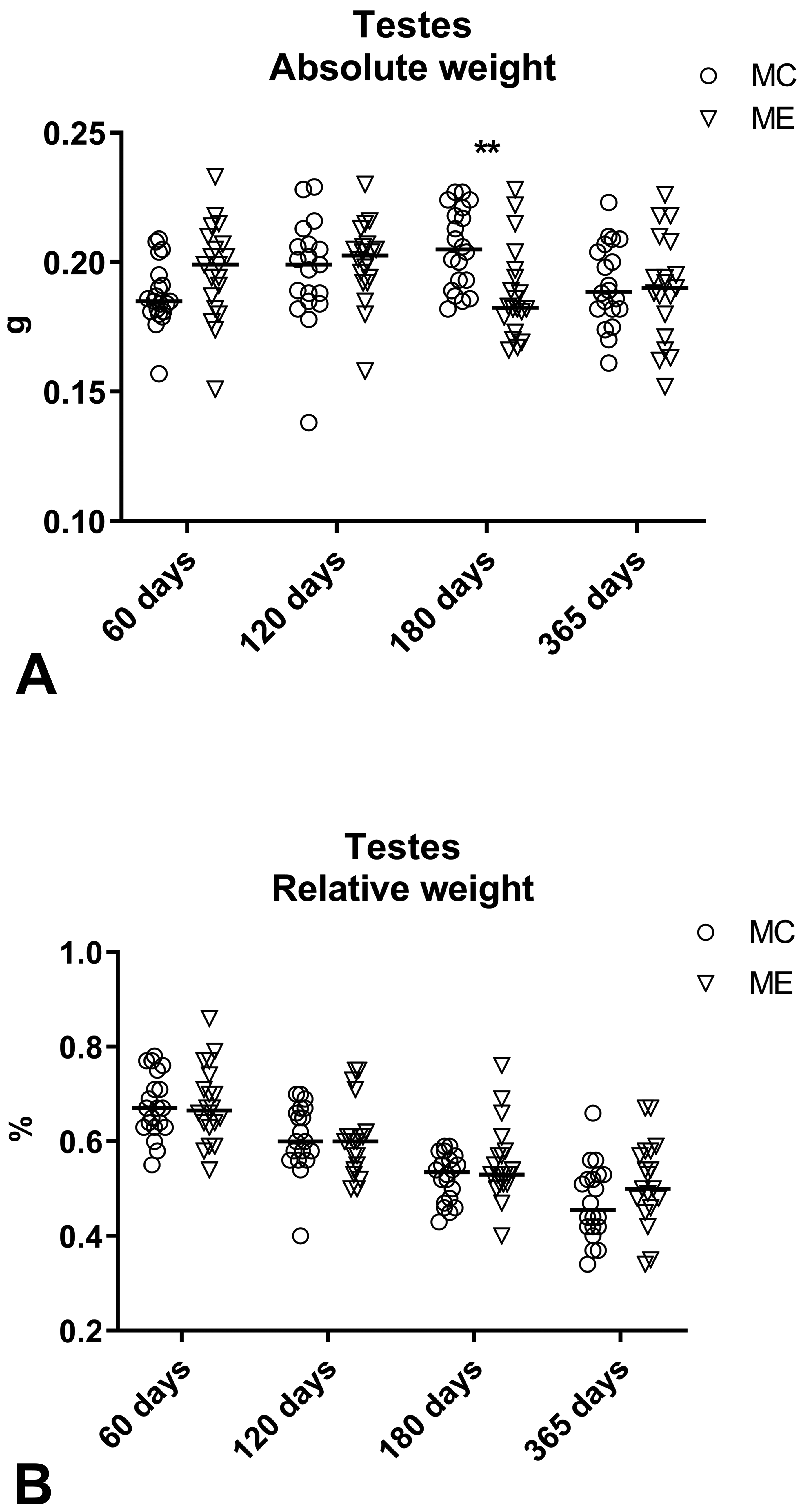
Absolute (g; A) and relative (% of body weight; B) testes weight measured at days 60, 120, 180, and 365. Testes absolute weight was significantly reduced in exposed male mice at day 180 of exposure (**P = .0015). No significant differences were found in testes relative weight. Data are reported as individual values and median. Mann-Whitney U test was applied for statistical analysis (n = 19-20).
The evaluation of apoptosis through the immunohistochemical staining for cleaved-caspase 3 revealed only rare intratubular apoptotic germ cells in both groups that were mainly located at the basal layers of the seminiferous epithelium. After quantification of cleaved-caspase 3-positive cells, no significant differences were found in the percentage of apoptotic cells per tubule between control and exposed male mice at all time points (days 60, 120, 180, and 365; Figure 7).
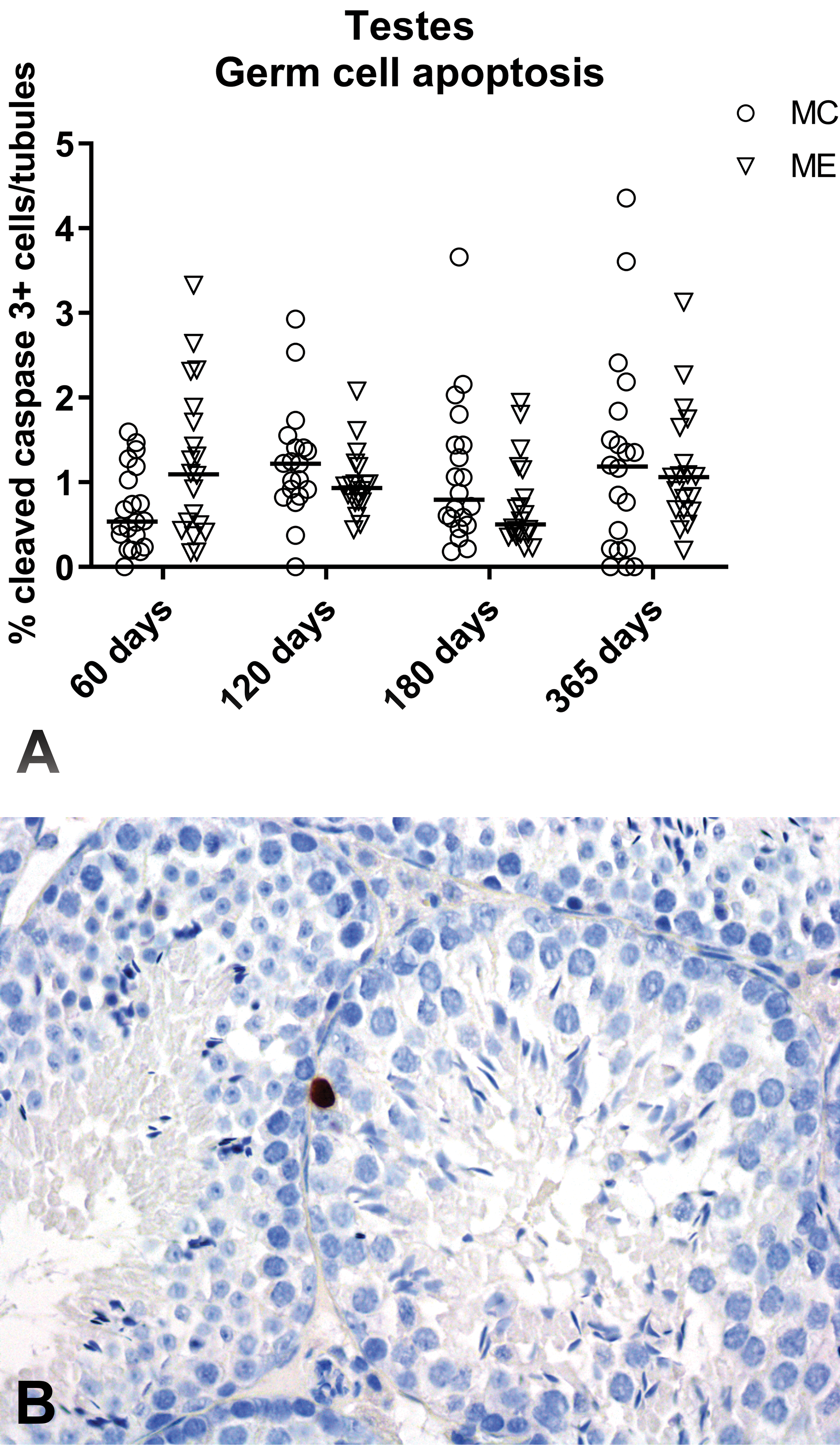
Testes, immunohistochemical evaluation of germ cell apoptosis. A, Percentage of the number of cleaved-caspase 3–positive cells per number of tubules per section of both testes, at days 60, 120, 180, and 365. Data are reported as individual values and median. Mann-Whitney U test was applied for statistical analysis (n = 19-20), no significant differences were found. B, Representative image of a cleaved-caspase 3–immunostained section: note the nuclear brown staining of a single apoptotic cell located in the basal layer of the seminiferous epithelium in an exposed mouse, at day 60 (immunoperoxidase staining, original objective ×40). Image of cleaved-caspase 3–immunostained mouse spleen section used as positive control is shown in Online Figure S2.
Discussion
The recent introduction of DVC for continuous automated intracage monitoring for data capture of the animal activity and cage microenvironment raised concerns about the potential effects on mice that could be induced by the EMF produced by the electronic board positioned underneath the cages. Thus, the aim of this study was to determine the intensity and frequency of EMF produced by DVC and whether the maintenance of mice in DVC over the long term would result in any adverse effect, as compared to mice housed in standard IVC.
The room of a facility is a complex environment from the point of view of the EMF, because there are several possible sources of EM radiations (eg, power sockets, lighting system, extensions, changing station, Wi-Fi system). Before starting the in vivo study, preliminary investigations revealed that there was a background level of electric fields in the empty room of the facility and that it varied depending on the position in the room (eg, close or not to building power sockets) and whether these sources (eg, lightening system) were switched off or on (Online Table S9). Considering the variability of the environmental electric fields, we decided to house all the animals of the study in the same room, and within the same rack, to reduce the impact of external sources of EMF that could mask the potential effects induced on mice by the EMF generated by the DVC. Since the distance of detection of the DVC was restricted to 2.55 cm above/below and 1.5 cm laterally to the DVC board (according to internal testing performed by the manufacturer, personal communication), adjacent cages could be not reached by the EMF generated by the DVC boards, and thus the checkboard arrangement of the 2 types of cage within the rack was chosen to minimize the effect on mice of the cage position within the rack.
Measurements of radiate EMF revealed that DVC boards produced EMF at a frequency ranging from 5 Hz to 3 GHz, thus including ELF, intermediate frequency (IF), and RF EMF. Also inside IVC low levels of EMF were detected, and since EMF generated by the DVC were not able to reach adjacent cages, they were regarded as part of the background EMF present in the room due to external sources, as discussed above. Electric fields were significantly increased in DVC as compared to IVC at all ranges of frequency examined, while no significant differences between DVC and IVC were observed in the induction of magnetic fields. Although the electric fields were significantly increased in DVC, their intensity was extremely low in general terms and, in comparative terms, well below the International Commission on Non-Ionizing Radiation Protection reference levels recommended for general public exposure (that are based on established evidence regarding onset of acute effects on people; Online Table S8). 1 The evaluation of the temperature at the bottom of both types of the cages (IVC and DVC) revealed that DVC board did not generate heating, excluding a potential contributing role of heating on the induction of biological effects.
No clinical signs or increased mortality were observed in exposed mice of both sexes. Body weight of exposed male mice sacrificed at day 180 was significantly reduced compared to control male mice and likely correlated with the reduced food consumption observed in this group. The other statistically significant differences detected in food and water consumption were considered not biologically relevant because no differences were noted in the body weight of animals. Despite the difference in the body weight detected at day 180, no significant difference in the body weight gain between control and exposed male mice was observed in this group. When looking at the group of male mice that was maintained up to 365 days, no difference in body weight between control and exposed mice was present at days 180 and 365, indicating that this result was not reproducible and exposure time dependent. At randomization of the 180-day male group, there was no significant difference in body weight between control and exposed mice, but the individual body weights of the exposed male mice were overall lower (except for one animal) than those of control mice, and we can speculate that over time this initially irrelevant difference became more evident. In literature, effects of EMF on body weight were contradictory. Gradual body weight loss was reported in male Balb/c mice after continuous whole-body exposure to 50 Hz approximately 1.4 mT ELF-EMF for 30 days, 32 and in male and female B6C3F1 mice exposed to 50 Hz 50 μT ELF-EMF for 15.5 months, with male mice starting to lose weight after 6 months and female mice after 4 months. 22 On the contrary, no significant effect on the body weight was found in male and female Swiss mice exposed for 90 days to 50 Hz 25 μT ELF-EMF 38 ; in male and female B6C3F1 exposed for 2 years to 60 Hz 2 μT, 200 μT, and 1000 μT ELF-EMF 16 ; in male Sprague-Dawley rats exposed to 50 Hz 25 mT ELF-EMF for 18 consecutive weeks 39 ; in male albino rats exposed to 50 Hz 5 μT ELF-EMF for 32 weeks 33 ; in female F344/N rats exposed to 60 Hz 2, 20, 200, and 2000 μT ELF-EMF for 2 years 14 ; in male and female F344 rats exposed to 50 Hz 0.5 and 5 mT magnetic fields for 2 years 15 ; and in male and female F344/N rats exposed for 2 years to 60 Hz 2, 200, and 1000 μT ELF-EMF. 40
Statistical analysis of hematology and bone marrow parameters revealed the presence of some significant differences between control and exposed mice in both sexes. However, results in both groups were always within the published reference intervals provided by the supplier of mice (https://www.criver.com/sites/default/files/resources/C57BL6MouseModelInformationSheet.pdf) or reported in literature. 41 Despite reference intervals reported in previous studies have been likely generated in different housing conditions and using different analyzers, the consistency of data recorded in this and previous studies suggests that most of the significant differences detected in this study did not have a biological relevance. This hypothesis is also supported by the fact that the magnitude of the changes recorded in exposed mice compared with control mice was lower than the intrinsic variability (imprecision) of the methods: for example, the changes regarding HGB (day 60), HCT (day 120), and MCV and MCHC (day 180) ranged from 1.6% to 5.3%, consistent with the inherent imprecision of most erythroid parameters. 42 Similarly, the differences regarding percentage of lymphocytes (days 60 and 120) or erythroid precursors (day 365) in the bone marrow, although of great magnitude (around 20%), are consistent with the intrinsic variability of microscopical cell counts, especially of cell populations that are poorly represented in the sample. 43
The only hematological result considered biologically relevant was the decreased neutrophil count in females after 60 and 365 days of exposure, given the magnitude of the difference recorded between control and exposed animals (−35.2% at day 60, and −53.4% at day 365 in exposed female mice) and the result largely lower in the exposed groups than the reference intervals. 41 Results of previous studies provided contrasting data regarding neutrophils: Çetin et al 31 reported a decrease in neutrophil counts in male Swiss mice after 90 and 120 days of exposure to pulsed 60 Hz 3μT ELF-EMF, while Hashish et al 32 reported an increase in neutrophils in male Swiss mice after 30 days of exposure to 50 Hz approximately 1.4 mT ELF-EMF. However, this latter result was referred to neutrophil percentages and not to neutrophil count, and therefore it may be due to the corresponding decrease of lymphocytes rather than to a true increase in neutrophil counts, which should be considered unaffected by treatment.
A second experiment carried out by exposing a new additional group of female mice for 60 days, failed to confirm the previously observed difference in the neutrophil count between control and exposed female mice. The lack of reproducibility of this finding, along with the lack of bone marrow changes at day 60 and the lack of changes in neutrophils or bone marrow at days 120 and 180, suggests that the possible effect of DVC on neutrophils, if any, is transient and occasional. However, a decrease in the neutrophil count in exposed female was found again at day 365, when also the M:E ratio showed a significant decrease in great magnitude (−40.2%), suggesting that long-term exposure may depress myeloid activity in the bone marrow, resulting in a decreased number of circulating neutrophils. Interestingly, no changes in the ratio between the proliferative or maturation myeloid pools were found, indicating that this possible effect, if any, may depend on a depressed myelopoietic activity rather than on a direct effect on a specific stage of myeloid progenitor cells. Finally, it is interesting to note that either at day 60 or at day 365, the reduction of neutrophil counts was detected only in exposed female, suggesting a possible influence of gender in this effect. Despite the significant decrease in neutrophils observed at days 60 and 365, considered of biological relevance because of its magnitude, there was no increase in clinical signs or histopathological lesions indicative of infection, the main expected complication of neutropenia.
No exposure-related gross changes were observed during necropsy. Histopathological examination revealed the presence of several findings in sampled organs, but overall, all the lesions were considered spontaneous, either incidental or background lesions (strain, age, and/or sex-related), with similar prevalence and severity observed in control and exposed groups, except for the reduction of hepatic fatty accumulation observed at day 180 in exposed mice (consistent with the decreased body weight observed in this group), and a significant decrease in the prevalence of renal intratubular hyaline casts in the exposed females at day 365. The biological meaning of this latter finding is doubtful, leading to the conclusion that it could be a fortuitous finding. Since only a single case of tumor was found in this study (histiocytic sarcoma in an exposed female at day 365), no conclusions can be drawn about the effect of DVC on cancer, and longer studies should be performed to investigate this issue.
According to previous studies, testis might represents one of the potential target organs of EMF in mice, where sperm abnormalities, increased or reduced testes weight, and/or increased germ cell apoptosis were previously observed. 26 –29,44 In the current study, no significant differences were found in testes relative weight, histopathology, and germ cell apoptosis between control and exposed mice at all time points. The reduced absolute testes weight observed in exposed male mice at day 180 was correlated with the reduced body weight observed in this group, since no testicular histopathological lesions were identified. The number of intratubular apoptotic cells was overall very low (less than 1 per 100 tubules). At day 60, germ cell apoptosis was slightly increased (P = .0760 Mann-Whitney U test) in exposed mice, but this trend was no more observed at later time points, indicating that even if a potential effect might exist, it was early and transient.
Based on the results of this study, DVC electronic boards produced extremely low-intensity electric fields at a wide range of frequency, from 5 Hz to 3 GHz. Maintenance of mice in DVC up to 1-year exposure resulted in statistically significant differences in some parameters (body weight, water and food consumption, some hematology and bone marrow parameters, and some histology findings), but most of them were considered likely fortuitous, resulting from biologic variability, or related to inbuilt variability of the assay method, as similarly concluded by other authors. 20,45 The only exception considered relevant from a biological point of view, and based on the results of previous studies, was the reduced neutrophil count at days 60 and 365 in exposed female mice. This finding was, however, not time dependent, transient, not reproducible, and, most importantly, not associated with signs of infection and therefore likely not clinically relevant. Similarly to the reported controversial role of EMF on eliciting biological effects on humans and animals, also the results of this study were not completely unequivocal, and further studies are needed to unveil whether the exposure to the extremely low-intensity EMF generated by the DVC might elicit unquestionable adverse effects on mice. In conclusion, the results of this study indicate that 1-year maintenance in DVC for the purposes of intracage monitoring of the animal activity and cage microenvironment did not produce any definite clinicopathological effect in either sex of C57BL/6NCrl mice.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, DS1_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS2_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, DS2_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS1_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS1_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS2_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS2_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS3_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS3_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS4_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS4_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS5_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS5_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS6_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS6_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS7_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS7_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS8_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS8_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS9_TPX_10.1177_0192623319852353 - Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields
Supplemental Material, TS9_TPX_10.1177_0192623319852353 for Long-Term Study on the Effects of Housing C57BL/6NCrl Mice in Cages Equipped With Wireless Technology Generating Extremely Low-Intensity Electromagnetic Fields by Camilla Recordati, Marcella De Maglie, Gerardo Marsella, Gianpaolo Milite, Alessandro Rigamonti, Saverio Paltrinieri and Eugenio Scanziani in Toxicologic Pathology
Footnotes
Acknowledgments
We are grateful to S. Bianchessi for in vivo treatments, M. Ferrario for assistance during necropsies, and M. Losa for technical histological support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Fondazione Unimi (formerly Fondazione Filarete) received a payment from Tecniplast S.p.a for the execution of necropsies, hematology, bone marrow cytology, and histopathological examination. Fondazione Unimi assigned the execution of the service to the Mouse and Animal Pathology Laboratory and the Department of Veterinary Medicine. Camilla Recordati presented the preliminary results of this work at 2 meetings (AALAS 2015; Phoenix, Arizona); FELASA 2016 [Brussels, Belgium]) and received reimbursement from Tecniplast S.p.a for travel expenses. Gianpaolo Milite worked as a scientific consultant in Laboratory Animal Sciences for Tecniplast S.p.a. Marcella De Maglie, Gerardo Marsella, Alessandro Rigamonti, Saverio Paltrinieri, and Eugenio Scanziani declare that they had no direct conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Tecniplast S.p.a.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
