Abstract
The majority of the tumors in the gastrointestinal (GI) tract of rats and mice, with spindle cell morphology, are diagnosed as smooth muscle tumors (SMTs). Similarly, several decades ago human GI tumors with spindle cell morphology were also diagnosed as SMTs. However, later investigations identified most of these tumors in humans as gastrointestinal stromal tumors (GISTs). The GISTs are considered to arise from the interstitial cells of Cajal located throughout the GI tract. Positive immunohistochemical staining with CKIT antibody is a well-accepted diagnostic marker for GISTs in humans. Since there is a considerable overlap between the histomorphology of SMTs and GISTs, it is not possible to distinguish them on hematoxylin and eosin stained sections. As a result, GISTs are not routinely diagnosed in toxicological studies. The current study was designed to evaluate the tumors diagnosed as leiomyoma or leiomyosarcoma in the National Toxicology Program’s 2-year bioassays using CKIT, smooth muscle actin, and desmin immunohistochemistry. The results demonstrate that most of the mouse SMTs diagnosed as leiomyoma or leiomyosarcoma are likely GISTs, whereas in rats the tumors are likely SMTs and not GISTs.
Keywords
Introduction
In toxicological studies, the majority of the tumors in the gastrointestinal (GI) tract of rats and mice with a spindle cell morphology are diagnosed as leiomyoma or leiomyosarcoma. 1 In the 1930s, human GI tumors with spindle cell morphology were also diagnosed as leiomyoma and leiomyosarcoma. 2 –4 The diagnosis was not challenged until some of the ultrastructural observations questioned the accuracy of the leiomyoma/leiomyosarcoma diagnosis. 3 –6 Later, certain immunohistochemical observations further strengthened the doubts on smooth muscle cell origin of these tumors. With these developments, the term “stromal tumor” was introduced in 1983 for human tumors. 4,5 The diagnosis of gastrointestinal stromal tumors (GISTs) gained more importance after the demonstration of gain of function mutation in the CKIT gene in mesenchymal tumors of the human GI tract. 7 Now, it is accepted that GISTs arise from the interstitial cells of Cajal (ICC), pacemaker cells in the intestinal wall, and CKIT, a protooncogene, has been recognized as a reliable marker of GISTs. 8,9 Based on the CKIT positivity, the majority of human GI tumors with spindle cell morphology are now classified as GISTs and not leiomyoma or leiomyosarcoma.
Histologically, GISTs have a spindle cell morphology similar to smooth muscle tumors (SMTs) and hence it is difficult to distinguish between the 2 lesions based on hematoxylin and eosin (H&E) stained sections. In humans, clinical behavior of GISTs is variable and therefore, they are categorized for risk of malignant behavior based on the size and mitotic index. Although accurate incidence rate for the human GIST in the United States is not available (Gastrointestinal Stromal Tumors Treatment (PDQ®)–Health Professional Version, the National Cancer Institute. https://www.cancer.gov/types/soft-tissue-sarcoma/hp/gist-treatment-pdq#cit/section_1.3; accessed on 01 11 2019) it is estimated to be ∼6.8 cases/million/year. 10 In chronic rat and mouse toxicity/carcinogenicity bioassays conducted by the National Toxicology Program (NTP), GIST is not routinely diagnosed. In addition, no treatment-related mesenchymal GI tumors have been reported in the NTP’s rat and mouse toxicity/carcinogenicity bioassays. Knowledge of GIST’s existence in rats and mice will help in understanding the relevance of rodent GI tumors with spindle cell histomorphology to human health, especially if the tumors are treatment-induced. Our current study was designed to address this data gap by evaluation of rat and mouse GI tumors diagnosed as leiomyomas or leiomyosarcomas in the NTP’s toxicity/carcinogenicity bioassays.
Materials and Methods
The NTP’s 2-year toxicity/carcinogenicity bioassay database (from 1982 to 2013) was searched for the diagnosis of leiomyoma and leiomyosarcoma in the stomach, duodenum, jejunum, ileum, cecum, colon, and rectum of rats and mice. Routinely, in the NTP bioassays, tissues are fixed in formalin for up to 6 months. Formalin-fixed paraffin-embedded (FFPE) blocks (rats, n = 54; mice, n = 32) were retrieved from the NTP archives. The sample selection was based on the availability of the FFPE blocks, ability to obtain sufficient sections from the block, and the extent of tissue autolysis/necrosis. Five-micron-thick sections were collected on charged slides. One slide from each block was stained with H&E stain, and the rest of the slides were used for immunohistochemistry (IHC).
The tissue sections were deparaffinized in xylene and rehydrated through decreasing grades of alcohol followed by Tris-buffered saline automation wash buffer (Biocare Medical, Pacheco, California). Endogenous peroxidase was quenched by incubating the tissue sections with 3% hydrogen peroxide. Endogenous biotin and nonspecific binding of avidin/biotin were blocked using an avidin-biotin blocking kit (Vector Laboratories, Burlingame, California) after which the sections were incubated with the primary antibody (Table 1). For the negative controls, tissue sections were incubated with either normal rabbit serum or normal rabbit IgG. The concentration of the negative control serum was normalized to that of primary antibody. Subsequently, the sections were incubated with a secondary antibody (Table 1) followed by Vectastain Elite ABC kit (Vector Laboratories, Burlingame, California) or Rabbit on Rodent Horseradish Peroxidase (HRP)-Polymer detection system (Biocare Medical, Pacheco, California) for 30 minutes. The antigen-antibody complex was visualized using 3,3′-diaminobenzidine (DAB) chromogen. Finally, the slides were counterstained with hematoxylin, dehydrated through increasing grades of alcohol, cleared in xylene, and coverslipped.
Details of Immunohistochemistry.

Abbreviations: DAB, 3,3′-diaminobenzidine; GI, gastrointestinal.
Results
The combined incidence of leiomyoma and leiomyosarcoma of the GI tract in F344 rats from 203 two-year toxicity/carcinogenicity bioassays of NTP was determined using the data available in the NTP database. The combined incidence was 118/42, 037 (0.28%) in males and 67/39, 629 (0.16%) in females. The tumors were present in stomach, duodenum, jejunum, ileum, cecum, colon, and rectum. The majority of tumors were present in stomach and jejunum in males, and in jejunum and duodenum in females. The distribution of tumors in various tissues is shown in Figure 1.
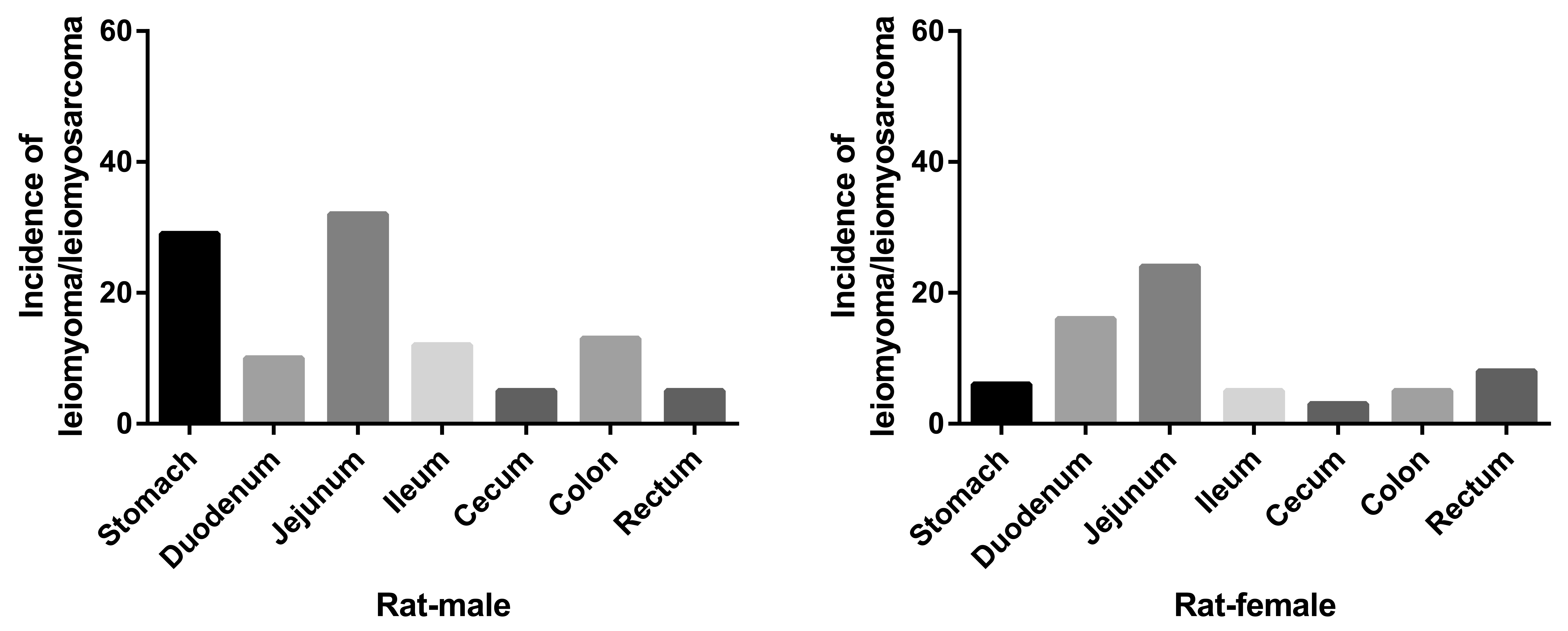
Incidence of leiomyoma/leiomyosarcoma in rats in the National Toxicology Program’s 2-year bioassays. The graphs represent the total incidences of leiomyoma/leiomyosarcoma found in 42,037 males and 39,629 females across 203 two-year toxicity/carcinogenicity bioassays.
The combined incidence of leiomyoma/leiomyosarcoma in B6C3F1/N mice was determined from 195 two-year NTP bioassays. The combined incidence was 25/40, 226 (0.06%) in males and 106/40, 229 (0.26%) in females. The majority of tumors were present in cecum in both males and females. Distribution of tumors in various tissues is shown in Figure 2.
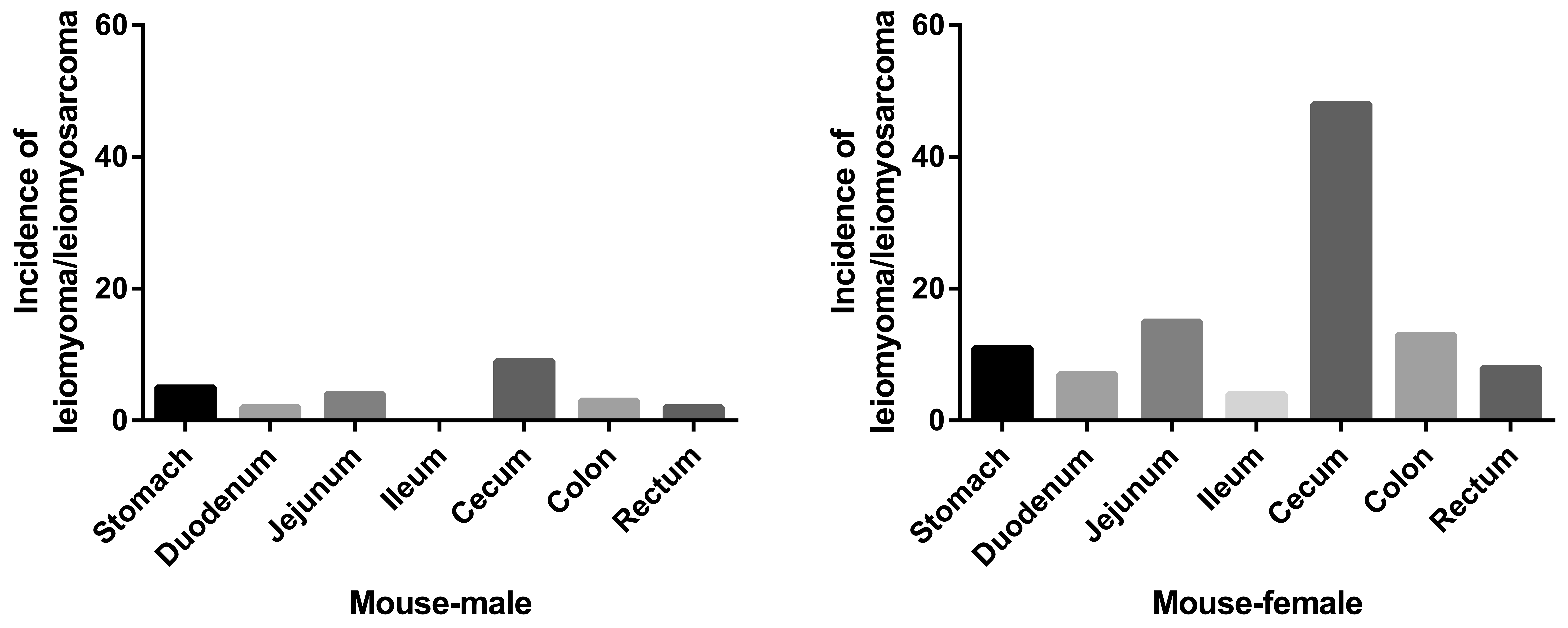
Incidence of leiomyoma/leiomyosarcoma in the mice in the National Toxicology Program’s 2-year bioassays. The graphs represent the total incidences leiomyoma/leiomyosarcoma found in 40 226 males and 40 229 females across 195 two-year toxicity/carcinogenicity bioassays.
Histologically, the majority of rat tumors involved tunica muscularis externa and interna. In some instances, tumors also occupied the submucosa. Approximately 50% of the tumors were well circumscribed. Mitotic figures were rare except for 25% of the cases, which had frequent mitotic figures (∼1-3/40× field). In 85% of the cases, the neoplasm was composed of interlacing bundles or whorls of spindle-shaped cells and in the remaining cases the neoplasm was composed of closely packed round to oval cells. One tumor had an epithelioid morphology admixed with spindle cells. Inflammatory cell infiltration was a common feature; eosinophilic-, lymphocytic- and mast cell infiltrations were present in 85%, 15%, and 20% of the cases, respectively. In a small number of cases, inflammatory cell infiltration was not a feature. A moderate amount of collagen was present between the neoplastic cells in 40% of the cases.
Similar to rats, the majority of the tumors in mice involved both tunica muscularis interna and externa. Approximately 63% of the tumors were well circumscribed. Mitotic figures were occasional to rare in approximately 53% of the cases while the rest had frequent mitotic figures (∼1-3/40× field). In 83% of the cases, the neoplasm was composed of interlacing bundles or whorls of spindle-shaped cells. In the rest of the cases (4/32), the neoplasm was composed of closely packed epithelioid or a mix of epithelioid and spindle cells. Unlike tumors of the rat, inflammatory cell infiltration was not a feature in mouse GI spindle cell tumors. Two of the tumors were very different from the rest in having a large amount of collagen.
Since CKIT IHC was used to distinguish the GIST from SMTs, normal intestine was stained to demonstrate the specificity of the antibody. In both rat and mouse intestine, the positively stained cells located within the tunica muscularis were consistent with the phenotype of the ICCs and the CKIT antibody did not stain the smooth muscle cells (Figure 3).
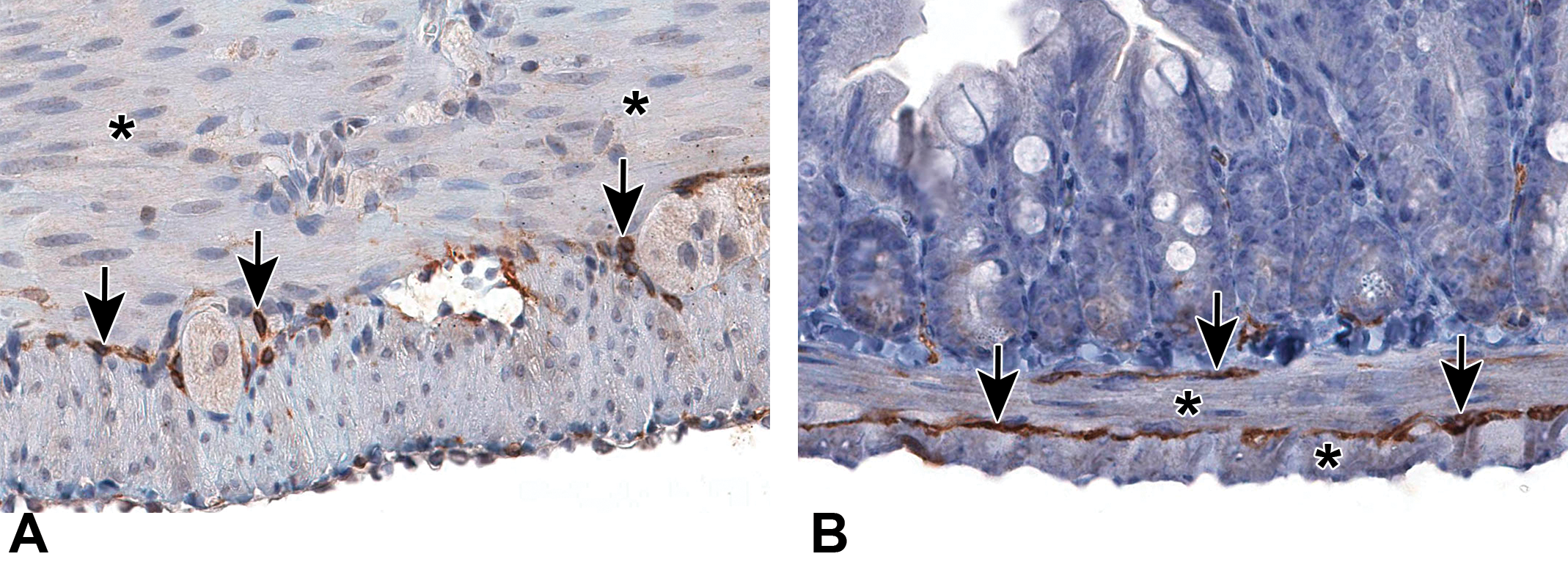
Demonstration of CKIT staining in normal colon of a rat (A) and normal duodenum of a mouse (B). The cells staining positive (arrows) are present between the muscle layers and in submucosa, locations consistent with the presence of interstitial cells of Cajal. There is no staining in the smooth muscle cells (asterisks). 3,3′-diaminobenzidine (DAB) chromogen and hematoxylin counterstain.
Immunohistochemically, 54/54 rat tumors were negative for CKIT, 53/54 were positive for desmin, and 45/54 were positive for smooth muscle actin (SMA). The one tumor that was negative for desmin was positive for SMA. Staining for SMA was weak and was present in a small percentage of neoplastic cells (<10%) as opposed to staining for desmin in almost all of the neoplastic cells. Both the desmin and SMA staining was cytoplasmic. Based on the IHC pattern, all the tumors in rats were likely SMTs and not GISTs (Figure 4).
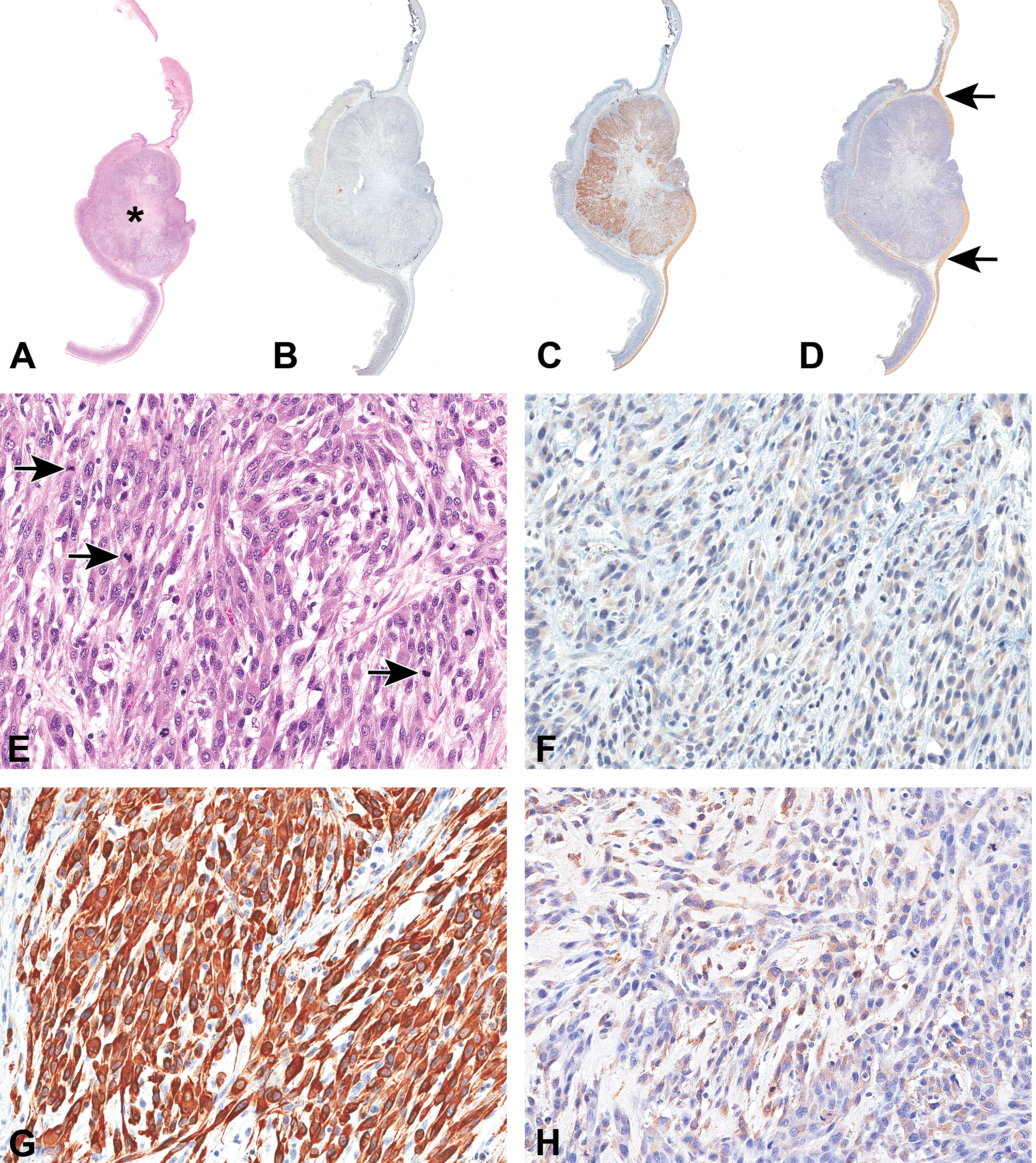
Smooth muscle tumor in the stomach of a rat. Hematoxylin and eosin stained section (A and E) shows the tumor (asterisk) involving tunica muscularis and submucosa. Spindle-shaped neoplastic cells are forming interlacing bundles. Many mitotic figures are present (arrows). The tumor is negative for CKIT (B and F) shows intense staining for desmin (C and G) and weaker staining for smooth muscle actin (D and H). Very pale brown staining seen in (F) was considered to be nonspecific staining based on the negative control slide. The smooth muscle actin staining is present in smaller percentage of cells and is not appreciable in D. However in D, positive staining in tunica muscularis (arrows; internal control) can be appreciated. 3,3′-diaminobenzidine (DAB) chromogen and hematoxylin counterstain.
In mice, 22/32 (69%) tumors were positive for CKIT and hence are probably GISTs (Figure 5). The staining for CKIT was cytoplasmic and was present in almost all the neoplastic cells in 19 of the 22 tumors. In the other 3 CKIT-positive tumors, the staining was present in 70%, 30%, and 15% of the neoplastic cells. Of these 22 CKIT-positive tumors, 3 also had desmin positivity and the rest were negative for desmin. In these tumors, desmin positivity was cytoplasmic and observed only in a very small percentage of neoplastic cells (<10%). All the CKIT-positive tumors were negative for SMA. Out of the 22 CKIT-positive tumors, 16 were in cecum, 5 were in colon, and 1 was in stomach. Five (16%) of the 32 tumors were positive for either desmin and/or SMA and therefore considered to be SMTs. Staining for SMA was cytoplasmic, when present. Five of the 32 tumors were negative for all the 3 markers. Out of these 5 tumors, 4 were in small intestine (duodenum/jejunum) and 1 was in the cecum.
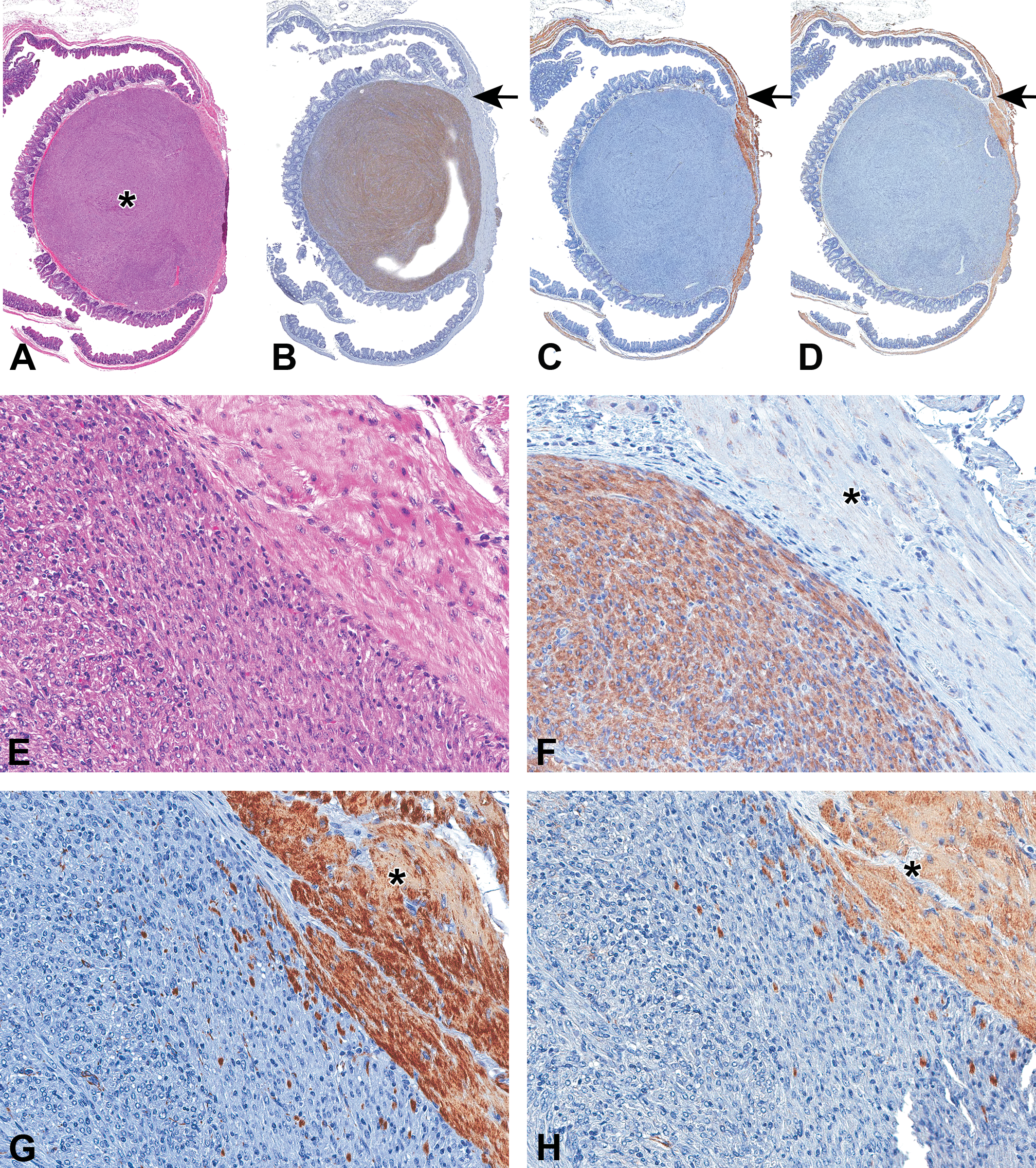
Tumor in the cecum of a mouse. Hematoxylin and eosin stained section (A and E) shows the tumor (asterisk) involving submucosa and part of tunica muscularis. The tumor is composed of bundles of spindle-shaped cells. The tumor shows staining for CKIT (B and F) and no staining for desmin (C and G) and smooth muscle actin (D and H). Smooth muscle, internal negative controls for CKIT, in B (arrow) and F (asterisk) had no staining. Smooth muscle, internal positive control for desmin and smooth muscle actin (arrow in C and D; asterisk in G and H) showed staining. 3,3′-diaminobenzidine (DAB) chromogen and hematoxylin counterstain.
Discussion
Histologically, leiomyomas in the GI tract are characterized by the presence of interlacing bundles or whorls of spindle-shaped cells with cigar-shaped or blunt-ended nuclei. In addition to the features described above, leiomyosarcomas also have pleomorphic cells, increased mitotic activity, and can commonly have necrosis, cystic change, and mineralization. 11 Gastrointestinal stromal tumors have been reported in humans and dogs. 12,13 In rats and mice, spontaneous GISTs are very rare and can be experimentally induced in rats by duodenal reflux, and in mice by Ckit mutations. 14 –17 Histologically, GISTs are characterized by the presence of interlacing bundles or whorls of spindle-shaped cells. Because of this histological resemblance to SMTs, it is often difficult to distinguish GISTs from leiomyomas/leiomyosarcomas based on H&E stained sections alone, and IHC is essential to diagnose GISTs. 11 In the current study, the histological features of GISTs were similar to what has been described for leiomyomas/leiomyosarcomas.
Human GISTs have been shown to harbor mutations in the CKIT gene, and the neoplastic cells consistently stain with CKIT antibody. 7 In the current study, a commercially available rabbit polyclonal antibody was used. The antibody was first tested to determine if it specifically identifies ICC in normal tissues. Interstitial cells of Cajal are located in the submucosa, between and within the circular and longitudinal muscle layer of tunica muscularis, around myenteric plexus, and submucosal plexus. 18 In the normal mouse and rat GI tract tissues tested, the antibody proved to be specific as it stained only the cells that matched the location of ICC and did not stain any smooth muscle or epithelial cells. None of the rat tumors stained with CKIT antibody but stained with SMA and desmin antibodies, indicating that the rat tumors are likely SMTs and not GISTs. Although the rat tumors are very likely to be SMTs, possibility of other tumors of myoid lineage such as rhabdomyosarcoma and inflammatory myofibroblastic tumors cannot be completely ruled-out. In contrast to the rat tumors, the majority of mouse tumors did stain with CKIT and not with SMA or desmin, indicating that many of the mouse tumors diagnosed as leiomyoma or leiomyosarcomas are probably GISTs. In humans, the majority of GISTs occur in stomach (60%) and in small intestine (25%). 4 The majority of CKIT-positive tumors of mouse in the current study occurred in cecum. Use of only IHC as a tool to evaluate the tumors is a limitation of this study. Ideally, immunohistochemical findings should be supported by other assays such as western blots, in situ hybridization, RT-PCR (Reverse transcription polymerase chain reaction), and electron microscopy. 19 However, suitable samples were not available for any of these analyses. Because of the rarity of these tumors and this being a retrospective study, the only available material was FFPE tissue blocks that could be used for IHC. Prolonged fixation (up to 6 months) affected the nucleic acid quality and therefore, in situ hybridization or RT-PCR could not be performed. Prolonged fixation in formalin is not amenable for ultrastructural examination as it yields poor morphology and definitive conclusions cannot be made. However, immunohistochemical demonstration of CKIT in the tumors is considered to be diagnostic of GISTs, 11,20,21 and in humans, CKIT IHC is approved by Food and Drug Administration for diagnostic purposes (https://www.accessdata.fda.gov/cdrh_docs/pdf4/P040011A.pdf; accessed on January 21, 2019). Therefore, it can be concluded that the majority of mouse GI tumors are probably GISTs. In 3 of the CKIT-positive tumors, there was also staining for desmin in a small percentage of neoplastic cells. In humans, a small percentage of CKIT-positive tumors can stain with desmin. 21 Five of the tumors were negative for any of the IHC markers used in this study and the internal controls stained as expected in these samples. A small percentage of human tumors have been reported to be negative for CKIT, 4 and it is possible that the tumors are CKIT-negative GISTs. In humans, other markers such as CD34 can also be used to diagnose GISTs. 12 The histomorphology of the tumors that did not stain with the 3 markers was similar to the tumors that did express the 3 markers evaluated. This suggests that the triple negative tumors may not be poorly differentiated tumors. Although the CKIT-positive tumors in mice are probably GISTs, other tumors such as mast cell tumor and melanoma with spindle cell morphology can also express CKIT and can’t be completely ruled-out.
As early as 1889, Cajal, using Golgi-stained intestinal tissue of rat and rabbit, described the histological features of ICCs, from which GISTs arise. 4,7,22 Current knowledge on the distribution and morphology proven by various techniques is very much similar to what was described by Cajal more than a century ago. 22 –24 Both ICCs and GISTs express CKIT receptor. 12,25,26 Binding of the KIT ligand (stem cell factor) activates KIT receptor and helps in normal functioning of ICCs which are pacemaker cells of the GI tract. 27 –29 On the other hand, activating mutations in the Ckit oncogene can cause proliferation of ICCs leading to GISTs. 7,12,14 In humans, 75% to 80% of GISTs have CKIT mutations on one of the exons 11, 9, 13, and 17 with the majority involving exon 11. 4,9 Similar mutations on exon 11 in canine GISTs have been reported. 13,30 Limited sample size and prolonged fixation (up to 6 months) of tissues in the current project did not allow evaluation for mutations. Such evaluations, when possible, will help in understanding the molecular pathogenesis of GISTs in mice.
The majority of mouse tumors in NTP studies that were previously diagnosed as SMTs are probably GISTs. However, the incidences of these SMTs in the GI tract were sporadic and never occurred as a treatment-related finding in any of the NTP rat and mouse toxicity/carcinogenicity bioassays. Thus, the current finding will not affect the outcomes of previous NTP bioassays. In the future, any tumor in the GI tract with spindle cell morphology should be considered for immunohistochemical evaluation using SMA, desmin, and CKIT antibodies. This is essential especially when a treatment-related increase in GI tract tumors with spindle cell morphology is observed. This will also enable building a meaningful historical control database for GISTs and will allow us to determine the translational relevance of the treatment-related increase in GISTs.
In conclusion, it appears that the majority of GI tumors with spindle cell morphology diagnosed as SMTs in mice are probably GISTs. There is no evidence for the occurrence of GISTs in rats based on CKIT, desmin, and SMA IHC. Immunohistochemical evaluation of any tumor resembling SMT in the GI tract of a mouse is essential for appropriate diagnosis of tumors. The conclusions are based on the evaluation of SMTs. Additional studies are required to verify if the conclusions of this study hold true for other GI tract tumors with spindle-cell morphology such as schwannomas.
Footnotes
Acknowledgments
The authors thank the DNTP/NIEHS Pathology Support Group, histology and immunohistochemistry core laboratories for their technical assistance. They also thank Chemical Effects in Biological Systems (CEBS) database team at DNTP for their help with searching the NTP study database, Drs Mark Cesta and Daven Jackson-Humbles for the critical review of the manuscript, and the staff of the NTP Archives at EPL, Inc, for their assistance in retrieving the samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
