Abstract
Vertebral metastases of non-small cell lung cancer (NSCLC) are frequently diagnosed in the metastatic setting and are commonly identified in the thoracic vertebrae in patients. Treatment of NSCLC bone metastases, which are often multiple, is palliative, and the median survival times are 3 to 6 months. We have characterized spontaneous vertebral metastases in a brain metastases model of NSCLC and correlated these findings with epithelial–mesenchymal transition (EMT). Brain metastases were established in athymic nude mice following intracardiac injection of brain-seeking adenocarcinoma NSCLC cells. Thirty-nine percent of mice (14/36) developed spontaneous vertebral metastases, spinal cord compression, and hind-limb paralysis. Vertebral metastases consisted of an adenocarcinoma phenotype with neoplastic epithelial cells arranged in cords or acini and a mesenchymal phenotype with spindloid neoplastic cells arranged in bundles and streams. Quantitative and qualitative immunohistochemical and immunofluorescence assays demonstrated an increase in vimentin expression compared to cytokeratin expression in vertebral metastases. A correlation with EMT was supported by an increase in CD44 in vertebral metastases and parenchymal metastases. These data demonstrate a translational lung cancer metastasis model with spontaneous vertebral metastasis. The mesenchymal and epithelial phenotype of these spontaneous metastases coupled with EMT provide a conduit to improve drug delivery and overall patient survival.
Keywords
Vertebral metastases are a common sequel to carcinomas, sarcomas, melanomas, and lymphomas. They lead to epidural compression of the spinal cord and are associated with a poor prognosis (Coleman 2006; Confavreux et al. 2018; Hatzoglou et al. 2014; Kawamoto et al. 2016; Prasanna et al. 2018; Pruitt 2017; Roodman 2004). Skeletal-related events (SREs) of vertebral metastasis are frequently reported and include hypercalcemia, bone pain, pathologic fractures, compression of the spinal cord, and paralysis (Confavreux et al. 2018; Zhou et al. 2017). SREs occur with increased frequency with multibone involvement and decreased therapeutic options (Coleman 2006). Primary clinical end points in vertebral metastasis clinical trials include ambulation, urinary continence, regained functional status, and overall survival (Patchell 2003).
In lung cancer patients, particularly non-small cell lung cancer (NSCLC), bone is a common site of metastasis, along with brain and liver (Riihimäki et al. 2014). NSCLC metastases to bone and brain are associated with the highest mortality rates (Dolgushin, Kornienko, and Pronin 2018; Oliveira, Mello, and Paschoal 2016; Riihimäki et al. 2014; Siegel, Miller, and Jemal 2018; Ulas et al. 2016). The adenocarcinoma subtype of NSCLC metastases is frequently identified in multiple bones, most commonly the ribs, followed by the thoracic and lumbar spine (Wang, Shen, and Zhu 2015; Zhou et al. 2017). In two retrospective studies, bone metastases were reported in 62% (n = 2,021) and 69% (n = 211) of NSCLC patients (Kong et al. 2017; Zhou et al. 2017). Furthermore, in the retrospective study by Zhou et al., 53% (n = 2,021) of patients developed bone metastases in thoracic vertebrae. Predictive biomarkers in patients for bone metastatic carcinomas, such as NSCLC, include alkaline phosphatase, which is correlated with osteoblastic activity particularly in disease, and N-telopeptide type I collagen (NTX), which has a positive correlation with osteoclastic bone resorption, SREs, and death (Brown et al. 2005; Zhou et al. 2017). Existing experimental models of vertebral metastasis of NSCLC have demonstrated tumor cell invasion and proliferation in parallel with SREs (Cai et al. 2015; Tatsui et al. 2009); however, a comprehensive analysis of the cellular phenotype of these metastases has, to our knowledge, not been reported.
This study was focused on understanding the pathologic paradigms of spontaneous vertebral metastases following intracardiac inoculation of NSCLC adenocarcinoma brain-seeking tumor cells. The primary goal of this study was to characterize the pathologic features of NSCLC metastases in bone and spinal cord. Second, we aimed to characterize the mesenchymal, epithelial, and cancer stem cell phenotypes of the NSCLC vertebral metastases. Finally, we sought to identify a correlation of the neoplastic cell phenotype with epithelial–mesenchymal transition (EMT), a phenomenon well documented in metastatic cells. Therefore, we hypothesized that following inoculation of NSCLC tumor cells in our mouse model, metastases will develop in parenchymal organs and bone and exhibit phenotypic characteristics of EMT.
Based on the pathologic features, expression of epithelial, mesenchymal, and cancer stem cell proteins, we identified a pathologic correlation with EMT in our model system. Our approach provides a model for brain metastases and vertebral metastases in the adenocarcinoma type of NSCLC. We have created and characterized aspects of the pathology of an NSCLC brain metastasis mouse model that reproducibly results in a high incidence of vertebral metastasis. These metastases exhibit features of EMT and should be useful for studies of this phenomena.
Materials and Methods
Cell Lines
A brain-seeking variant of the adenocarcinoma type of the NSCLC cell line A549 (A549-Br) was generously donated by Dr. Patricia Steeg, National Cancer Institute. The primary long-term objective in our laboratory is to evaluate changes in the blood–brain barrier to the blood–tumor barrier in the presence of brain metastases. A549-Br cells were maintained in RPMI 1640 media enriched with 5% fetal bovine serum and 0.5% penicillin/streptomycin antibiotic. Cells were dissociated using 0.05% trypsin and maintained in a 37°C incubator at 5% CO2. The A549-Br cell line was authenticated at American Type Culture Collection (Manassas, VA) using Short Tandem Repeat analysis.
Metastasis Experimental Model
Thirty-six 6-week-old 25 g, Hsd:Athymic Nude-Foxn1nu mice (18 males, 18 females), were anesthetized with 2% to 3% isoflurane, monitored for anesthetic depth, and placed in dorsal recumbency on a heated imaging stage. A Vevo2100 small animal ultrasound system (FUJIFILM VisualSonics; 22–55 MHz transducer) and an automated image-guided precision microinjection system (FUJIFILM VisualSonics) were used to guide and confirm the inoculation of 1 × 106 A549-Br cells into the left ventricular lumen of the heart.
Animals were monitored daily for alterations in posture, locomotion, and awareness of surroundings. Body weight was measured and recorded weekly. Animals were housed in a specific pathogen free (SPF) facility in autoclaved and individually ventilated rectangular plastic cages in same-sex groups of 4 with crinkle cut paper bedding. Water from autoclaved water bottles and Hsd:Athymic Nude-Foxn1nu, (2011) sterilized dry pelleted feed were available ad lib. Animals were housed in a 12:12-hr light:dark cycle in a controlled temperature and humidity room (between 21.1°C and 23.8°C, 30% to 70% humidity). Mice were euthanized using CO2 asphyxiation following development of hind-limb paralysis at approximately four to six weeks post-ICI. All animal procedures were reviewed and approved by Purdue Animal Care and Use Committee (ACUC). Purdue program of animal care and use is accredited by Assessment and Accreditation of Laboratory Animal Care (AALAC) international and holds an animal welfare assurance with National Institutes of Health (NIH)/Office of Laboratory Animal Welfare (OLAW).
Histopathology
The vertebral column, including spinal cord, cervical, thoracic, and lumbar vertebrae, were removed en bloc. Tissues were placed in room temperature 10% neutral-buffered formalin for 24 hr prior to decalcification. Sections were decalcified with 25% formic acid and 10% sodium citrate for 4 days at room temperature on an orbital shaker. Following decalcification, tissues were washed in tap water and placed in 65% ethanol for 24 to 48 hrs. Tissues were processed over 10 hrs using a Sakura Tissue-Tek VIP6 tissue processor. Tissues were processed sequentially in 70%, 80%, 95%, and 100% ethanol followed by xylene and paraffin and were embedded in Surgipath Paraplast Plus (Leica Biosystems, Wetzlar, Germany, Cat. #39602004). Tissue sections, 4-µm thickness, were placed on charged slides, stained with hematoxylin and eosin, and cover-slipped using a Leica ST5010-CV5030 integrated workstation.
A complete necropsy examination was performed on each animal (n = 14). No significant gross lesions were seen in the oral cavity, skull, esophagus, trachea, lungs, heart, spleen, stomach, small intestines, large intestines, pancreas, liver, kidneys, sciatic nerve, or urinary bladder.
All brain and vertebral columns examined contained metastases (n = 14). Representative samples of heart, lung, liver, kidneys, spleen, and pancreas were examined histologically (n = 5). Representative sections of adrenal glands which were unilaterally or bilaterally enlarged (n = 3) were examined histologically. Representative sections of ovaries which were unilaterally or bilaterally enlarged (n = 3) grossly were evaluated histologically. Peritoneal metastases (n = 2) observed grossly were examined histologically.
Immunohistochemistry (IHC) and Digital Pathology Analysis
Tissue sections were deparaffinized and rehydrated on the Leica ST5010-CV5030 Autostainer. Rodent Decloaker (Biocare, RD913M) was applied at 95°C for 20 min for antigen retrieval. Slides were cooled to room temperature and washed in Tris-Tween buffer. Slides were placed in a Dako Cytomation Autostainer Plus and quenched in hydrogen peroxide for 5 min. Slides were washed in Tris-buffered saline–Tween 20 (TBS-T), blocked in 2.5% goat serum (Vector, MP-7451) for 20 min, and incubated in antibody solution for 30 min. Cytokeratin (1:100, Abcam, ab9377), vimentin (1:1,000, Abcam, ab45939), and CD44 (1:2,000, Abcam, ab157107) were diluted in RTU Animal-Free Diluent (Vector Laboratories, SP-5035). Sections were washed in TBS-T; incubated with the secondary antibody, goat anti-rabbit ImmPRESS HRP (Vector Laboratories, MP-7451), for 30 min; washed in TBS-T; and developed with 3,3′-Diaminobenzidine (DAB, ImmPACT DAB, Vector Laboratories, SK-4105) for 5 min. Slides were washed, placed on the Leica-integrated workstation, and counterstained with Gill’s II hematoxylin. Immunoglobulin G (IgG) isotype controls were used as negative controls for each antibody (Janardhan et al. 2018).
IHC slides were digitized at 40× magnification using the Aperio VERSA 8-slide scanner. Digital pathology images were analyzed using Aperio ImageScope 12.3 (Leica Biosystems). DAB staining was accurately quantified and separated from hematoxylin stain using a color deconvolution algorithm in annotated tumor regions of vertebral bones (n = 4), brains (n = 2), adrenal glands (n = 4), peritoneal (n = 3), and ovarian metastases (n = 2). Strong positive expression was characterized as the highest percentage of DAB positive pixels at a strong positive threshold.
Immunofluorescence Analysis and Imaging
Tissue sections were deparaffinized and rehydrated on the Leica ST5010-CV5030 Autostainer. Rodent Decloaker (Biocare, RD913M) was applied at 95°C for 20 min for antigen retrieval. Slides were cooled to room temperature and washed in TBS and TBS-T and placed in a Dako Cytomation Autostainer Plus. Cytokeratin (1:100, Abcam, ab9377) and vimentin (1:1,000, Abcam, ab45939) were diluted in RTU Animal-Free Diluent (Vector Laboratories, SP-5035). Slides were blocked in 2.5% goat serum (Vector, MP-7451) for 20 min and incubated with vimentin (Abcam, ab45939) primary antibody for 30 min in the absence of light. Slides were rinsed with TBS-T and anti-rabbit IgG (Vector Labs, AI-1000) was applied for 30 min. Following two more rinses with TBS-T buffer, 594 horse anti-goat IgG secondary antibody (Vector Labs, DI-3788) was applied for 30 min in the absence of light. Slides were rinsed with TBS-T buffer and blocked with 2.5% normal horse serum (Vector Labs, DI-3788). Cytokeratin primary antibody was applied to slides for 30 min in the absence of light, followed by TBS-T buffer rinses, and then 488 horse anti-rabbit IgG secondary antibody was applied for 30 min (Vector Labs, DI-1788) in the absence of light. Slides were rinsed and cover slipped. Digital images were acquired via Zeiss Axio Scope A2 at 20× and exposure times for each channel were set to avoid pixel saturation.
Statistical Analysis
Weekly body weights were averaged and normalized to reference animal weights in Hsd:Athymic Nude-Foxn1nu mice of the same age and sex (Hsd:Athymic Nude-Foxn1nu, 2011). Statistical significance (p < 0.05) of cytokeratin and vimentin expression was confirmed using an unpaired t-test with commercially available Prism version 7 software.
Results
Clinical Presentation
Fourteen out of thirty-six mice (39%) inoculated with A549-Br cells developed vertebral metastases; clinical signs were identified within 4 to 6 weeks post-ICI. Within four weeks of cellular colonization, nine 10-week-old female mice developed hind-limb paralysis. Three 11-week-old mice (2 males, 1 female) and two 12-week-old mice (all females) developed hind-limb paralysis 5 and 6 weeks post-ICI, respectively. Hind-limb paralysis was characterized by dragging of rigidly extended hind limbs laterally or caudally, absence of pinched toe reflex, dragging of the tail, and cold, pale digits. There was no significant difference in body weight in paralyzed male mice compared to published controls. After six weeks of cellular colonization, 12-week-old female mice (n = 2) weighed 18g (mean) compared to 23.1g of age-matched published controls, a 22% decrease in body weight (Hsd:Athymic Nude-Foxn1nu, 2011).
Gross Lesions
The peritoneum of the cranial abdomen, caudal to the greater curvature of the stomach, contained numerous 0.5 to 1 mm diameter nodules (n = 3). The nodules were pale tan, multifocal to coalescing, round, firm, glistening, and arranged in a cobblestone-like pattern. Single ovaries from two mice were enlarged to 6 mm in diameter. Approximately 90% of the ovary was effaced by pale tan, polycystic, irregularly shaped, firm masses. Gross lesions were not identified in parenchymal organs, testes, long bones, vertebrae, spinal cord, or brain.
Histopathology
Vertebral metastases
NSCLC metastases in the mouse model were identified throughout the vertebral column but were most common in thoracic vertebrae and in the associated spinal cord or dorsal root ganglia. In cervical, thoracic, and lumbar vertebrae, multiple 389 to 1,300 µm metastases infiltrated the marrow cavity. These metastases were roughly spherical to multilobular and densely cellular. For example, a single metastasis, measuring 1.3 mm, infiltrated a thoracic vertebra, extended into the spinal canal and compressed the overlying spinal cord (Figure 1A and B). Osteoclastic bone resorption was occasionally identified with scalloping of trabecular and occasionally cortical bone. The dorsal root ganglia, and rarely spinal cord, were infiltrated by unencapsulated clusters of neoplastic epithelial cells ranging in diameter from 12 to 15 µm (Figure 1C).
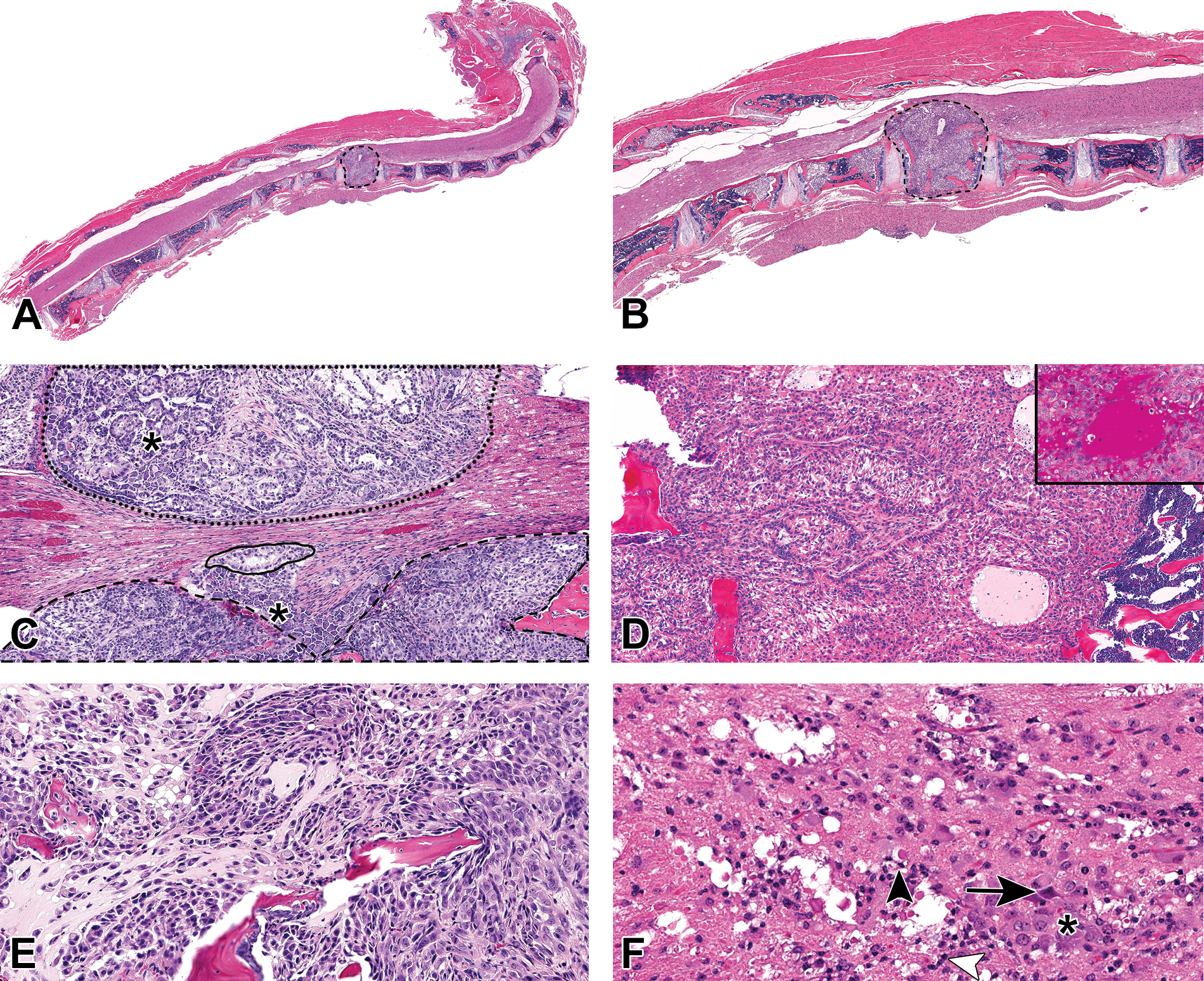
Histology of vertebral metastases in an experimental model of non-small cell lung cancer (NSCLC). (A) A 1.3-mm diameter, spherical metastasis effaced the entire vertebral body of T2 and compressed the overlying spinal cord (original objective 1.25×). (B) Higher magnification of the vertebral metastasis in A, which compressed the overlying spinal cord (original objective 4×). Neoplastic cells were present in the marrow cavities of adjacent vertebrae (T1–T4). (C) In the lumbar region (original objective 10×), an 8.2-mm diameter metastasis effaced the vertebral bone (narrow black dashed line). Neoplastic cells rarely infiltrated the spinal cord (solid black line). Another 9.8-mm diameter metastasis replaced the vertebral bone (wide black dashed line) and compressed the overlying ganglion (*). (D) Neoplastic cells in the dorsal vertebral body had typical adenocarcinoma features, arranged in palisading cords and sheets, surrounded by eosinophilic periodic acid-Schiff-positive (inset) mucinous material. (E) Mesenchymal neoplastic cells formed streams of spindle-shaped cells with eosinophilic cytoplasm and loss of cohesion. (F) The spinal cord was compressed by a NSCLC vertebral metastasis (original objective 40×) resulting in white matter rarefication with numerous spheroids (arrowhead) and neuronal necrosis (arrow) in the gray matter. The spinal cord was infiltrated by microglia (white arrowhead) and gitter cells (*).
Neoplastic cells within the same tumor exhibited both an epithelial and a mesenchymal phenotype in vertebral columns. NSCLC metastases in nude mice with an epithelial phenotype were arranged in dense cords, acini, or palisading bundles (Figure 1D). These tumor cells were 8 to 16 µm in diameter with round to ovoid nuclei, 1 to 2 nucleoli, and vacuolated eosinophilic cytoplasm. The cytoplasm of neoplastic cells was often distended by faintly eosinophilic periodic acid–Schiff (PAS) positive material. Cilia were occasionally present on the apical border of neoplastic cells, reminiscent of bronchiolar epithelium. Neoplastic cells were associated with faintly eosinophilic, vacuolated, PAS-positive mucinous material (Figure 1D, inset). In contrast, neoplastic cells with a mesenchymal phenotype were arranged in dense streams and short bundles. Tumor cells were 6 to 15 µm in diameter, roughly ovoid or spindle shaped with round nuclei, 1 to 3 nucleoli, and abundant faintly eosinophilic cytoplasm (Figure 1E). Atypical features of neoplastic cells included absence of basement membrane polarity, multinucleation, and karyomegaly. Thirty-one mitotic figures were identified in ten 40× fields (FN22 mm) in one animal; necrosis was absent.
The spinal cord of all affected mice was compressed by the vertebral metastases, particularly in the thoracic region. In some animals, the white matter of the compressed spinal cord was rarefied. Within the injured white matter, axons were swollen and formed spheroids. Few neurons were necrotic or swollen with central chromatolysis. The surrounding neuroparenchyma was expanded by gliosis, gitter cells, and hemorrhage (Figure 1F).
Brain metastases
After 4 to 6 weeks of A549-Br cellular colonization in the mouse model, the neuroparenchyma was multifocally effaced by 400 to 1,500 µm roughly spherical, unencapsulated NSCLC brain metastases (Figure 2A). These lesions were particularly evident at the gray and white matter interface, suggesting a hematogenous route of metastasis formation. Neoplastic cells exhibited moderate anisocytosis and anisokaryosis, were 10 to 20 µm in diameter, and polygonal with abundant eosinophilic cytoplasm. These cells contained a single ovoid nucleus, 1 to 3 nucleoli, and PAS-positive cytoplasmic eosinophilic material with few vacuoles. The brain metastases were surrounded by PAS-positive pale basophilic material (Figure 2B). Mitotic figures were infrequent, with three F figures in ten 400× fields; necrosis was absent.
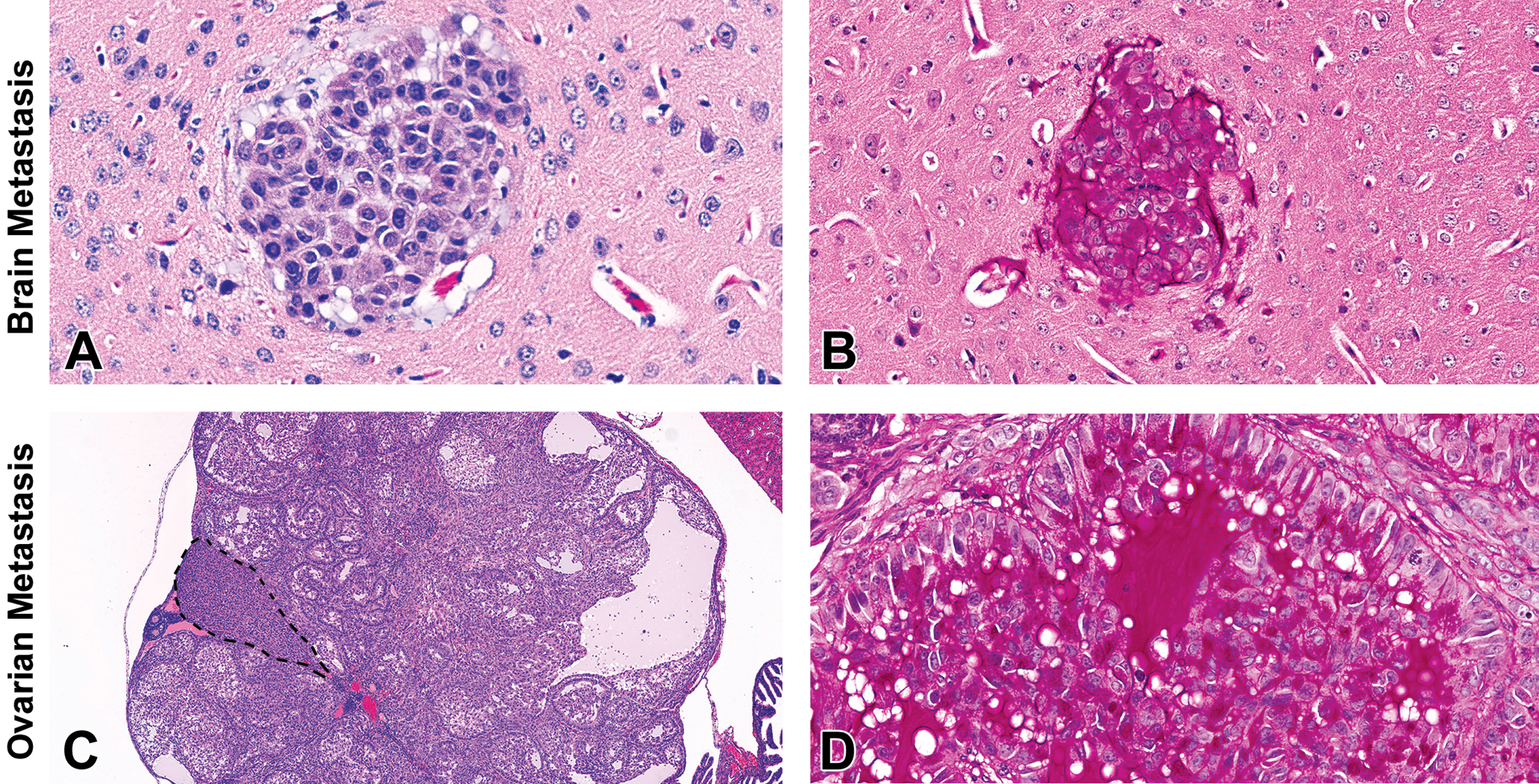
Hematoxylin and eosin stain and periodic acid–Schiff (PAS) of brain and parenchymal metastases. (A) A 1.6-mm brain metastasis was present at the gray and white matter cerebral interface (original objective 40×). Neoplastic cells were arranged in sheets with occasional cytoplasmic vacuoles. (B) PAS-positive material was present in the brain metastasis from A (original objective 40×). (C) A 2.6-mm cystic metastasis effaced the ovarian architecture and compressed the remaining parenchyma (black dashed line; original objective 2×). (D) Neoplastic cells were arranged in prominent acini surrounding PAS-positive mucin.
Parenchymal metastases
Metastases were present in the cranial abdominal peritoneum, ovaries, and adrenal glands. Peritoneal metastases were ovoid and replaced the underlying adipose tissue. Metastases effaced approximately 80% of the adrenal gland and 90% of the ovarian architecture (Figure 2C). In contrast to the dual phenotype of vertebral metastases, all neoplastic cells in visceral metastases were well-differentiated, polygonal to cuboidal, and often arranged in prominent acini and cords. These findings were consistent with the adenocarcinoma phenotype. Neoplastic cells were 8 to 25 µm in diameter, with abundant eosinophilic cytoplasm, a single ovoid nucleus with clumped chromatin and 1 to 3 nucleoli. Neoplastic cells were often centered on pools of pale basophilic, PAS-positive, mucin-containing vacuoles (Figure 2D). Mucin was identified intracellularly and extracellularly and was consistent with the PAS-positive material identified in both brain and vertebral metastases. There was no evidence of necrosis. Sixteen mitotic figures were identified in ten 400× fields.
Neoplastic cells were also identified in the heart and lungs. In the lungs, rafts of neoplastic cells were in pulmonary arteries. Neoplastic cells were identified in few alveolar spaces admixed with hemorrhage. Clusters of these cells were in the myocardium.
IHC
IHC analysis of vimentin and cytokeratin protein expression highlighted the mesenchymal and epithelial phenotype of the NSCLC metastases in the model. In vertebral metastases, neoplastic foci contained both epithelial and mesenchymal phenotypes (Figure 3A). Epithelial neoplastic cells had faint or intense vacuolated cytokeratin expression (Figure 3B). More intense cytokeratin expression was identified in vertebral metastatic cells that maintained a polygonal, palisading, or acinar shape. Mesenchymal neoplastic cells had diffuse and intense cytoplasmic expression of vimentin with a granular pattern (Figure 3C). Cytokeratin expression was present in 8% of neoplastic cells compared to 23% of neoplastic cells expressing vimentin (p < 0.05, n = 4, Figure 3D).
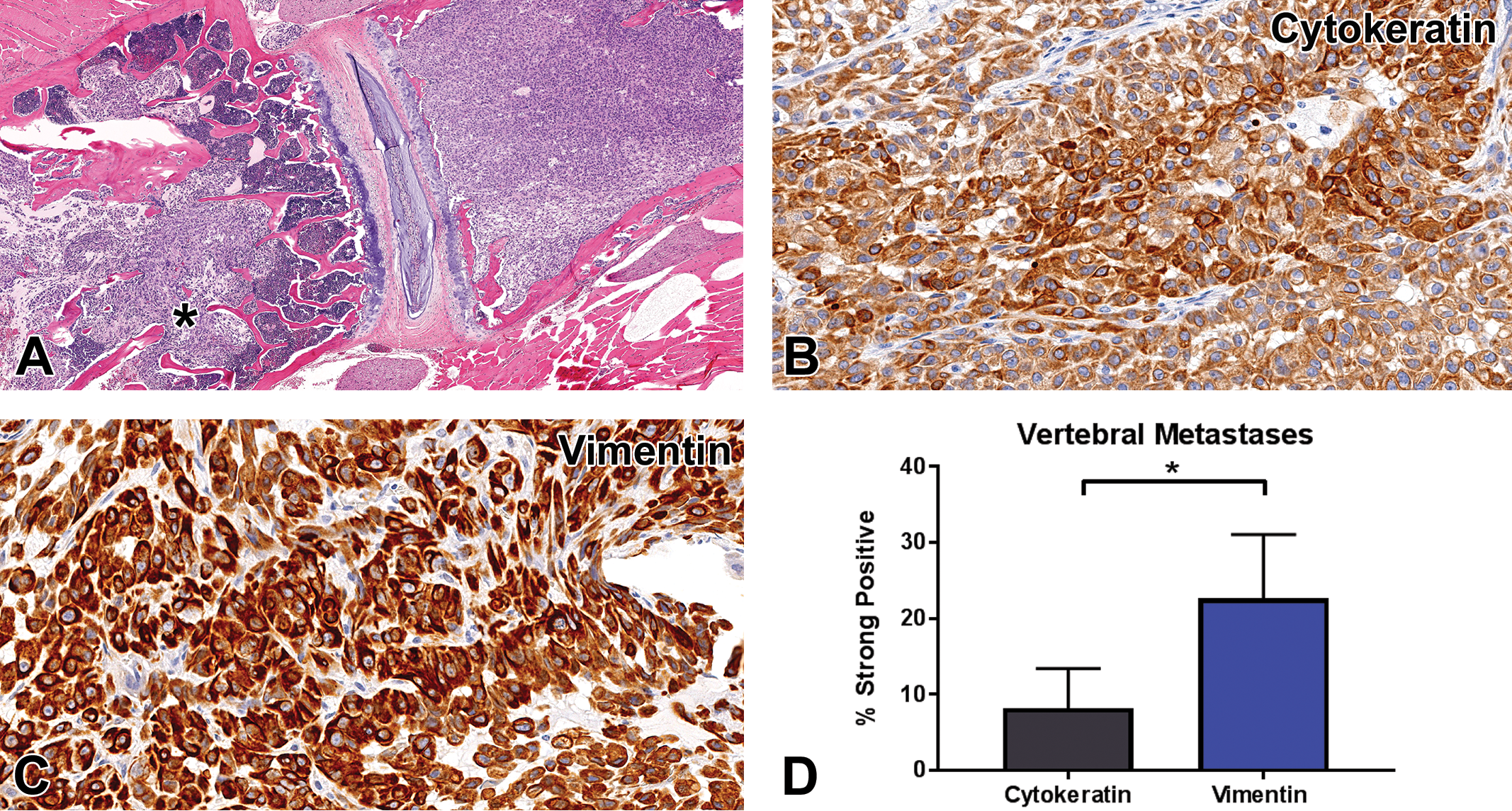
Vimentin and cytokeratin expression within non-small cell lung cancer vertebral metastases. (A) Neoplastic cells in vertebral bodies of mouse 2 had epithelial and mesenchymal features. Neoplastic cells on the right of the image were arranged in sheets, whereas those on the left (*) demonstrated loss of cohesion and were arranged in streams (original objective 1.25×). (B) Neoplastic cells within the vertebral metastasis retained moderate cytoplasmic cytokeratin expression (original objective 40×). (C) Neoplastic cells exhibited abundant vimentin expression (original objective 40×). (D) Strong positive cytokeratin expression was present in 8% of neoplastic cells compared to 23% of neoplastic cells with strong positive vimentin expression (p < 0.05, n = 4).
In NSCLC brain metastases in mice, neoplastic cells had diffuse immunohistochemical expression of cytokeratin with variable intensity (Figure 4A). The staining pattern was cytoplasmic and threadlike. Vimentin expression was diffuse and intense in all NSCLC brain metastases in the model (Figure 4B). The expression of vimentin and cytokeratin in brain metastases was characterized by a modest increase in vimentin expression compared to cytokeratin; however, this trend was not significant. Approximately 39% of cells had strong cytokeratin expression; however, 50% of neoplastic cells exhibited strong vimentin expression (Figure 4C).
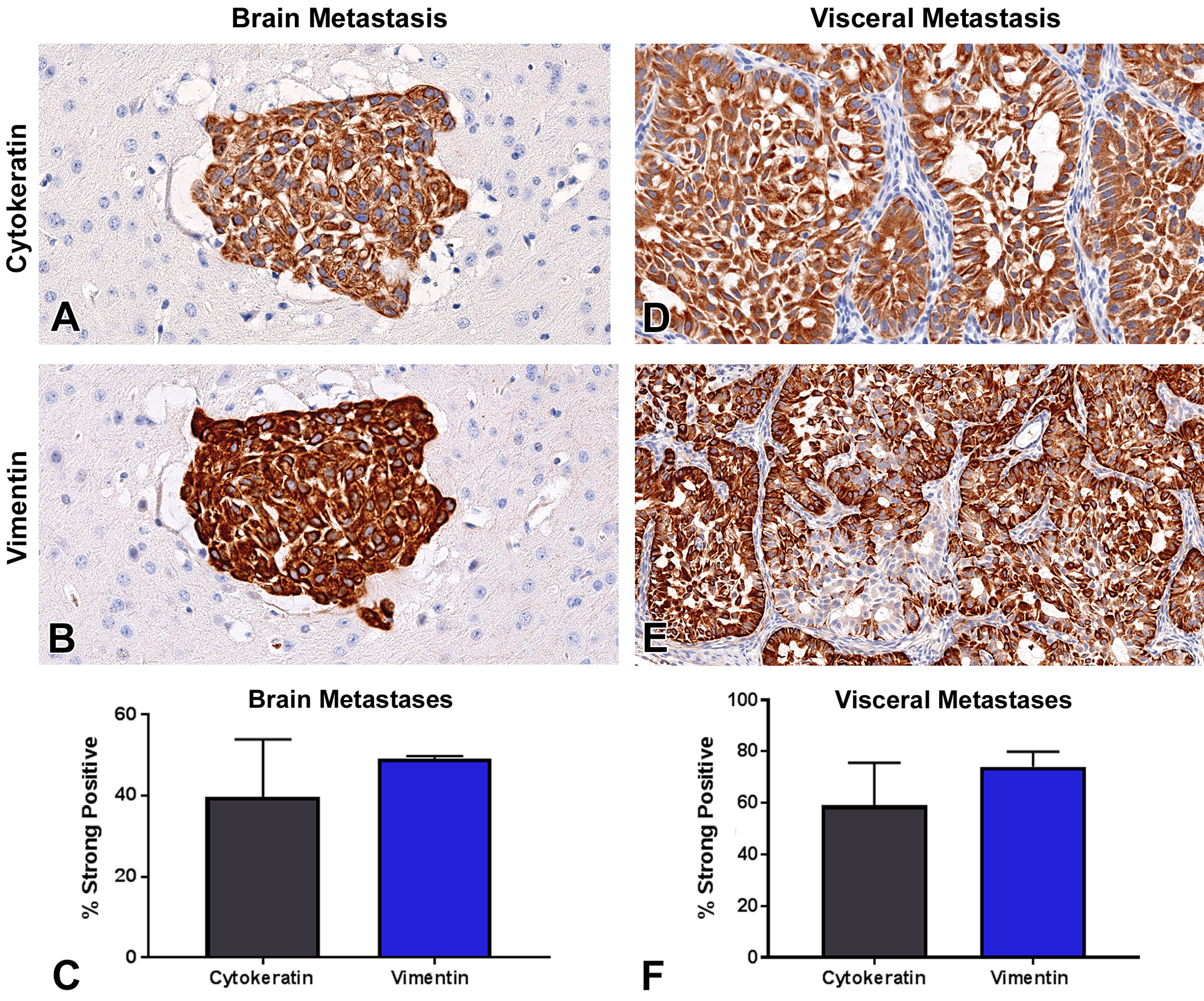
Vimentin and cytokeratin expression within brain and visceral metastases. (A) Moderate cytoplasmic cytokeratin expression in brain metastatic cells in mouse 14 (original objective 40×). (B) Strong vimentin expression was present within brain metastatic cells in the same animal (original objective 40×). (C) Strong positive cytokeratin expression was quantified in 39% of neoplastic cells and 50% of neoplastic cells exhibited strong positive vimentin expression (n = 2, ns).
In visceral metastases, all tumor cells had diffuse, variably intense cytokeratin expression (Figure 4D) and multifocal intense vimentin expression (Figure 4E). Fifty-nine percent of tumor cells had strong cytokeratin expression compared to 74% of tumor cells with strong vimentin expression (Figure 4F).
Expression of CD44, a cancer stem cell protein, correlated with malignancy and EMT in the experimental model (Leung et al. 2010). CD44 expression was prominent in the cellular membrane of all neoplastic cells in vertebral and ovarian metastases (Figure 5A and B). However, in brain metastases, CD44 expression was weak to moderate (Figure 5C).
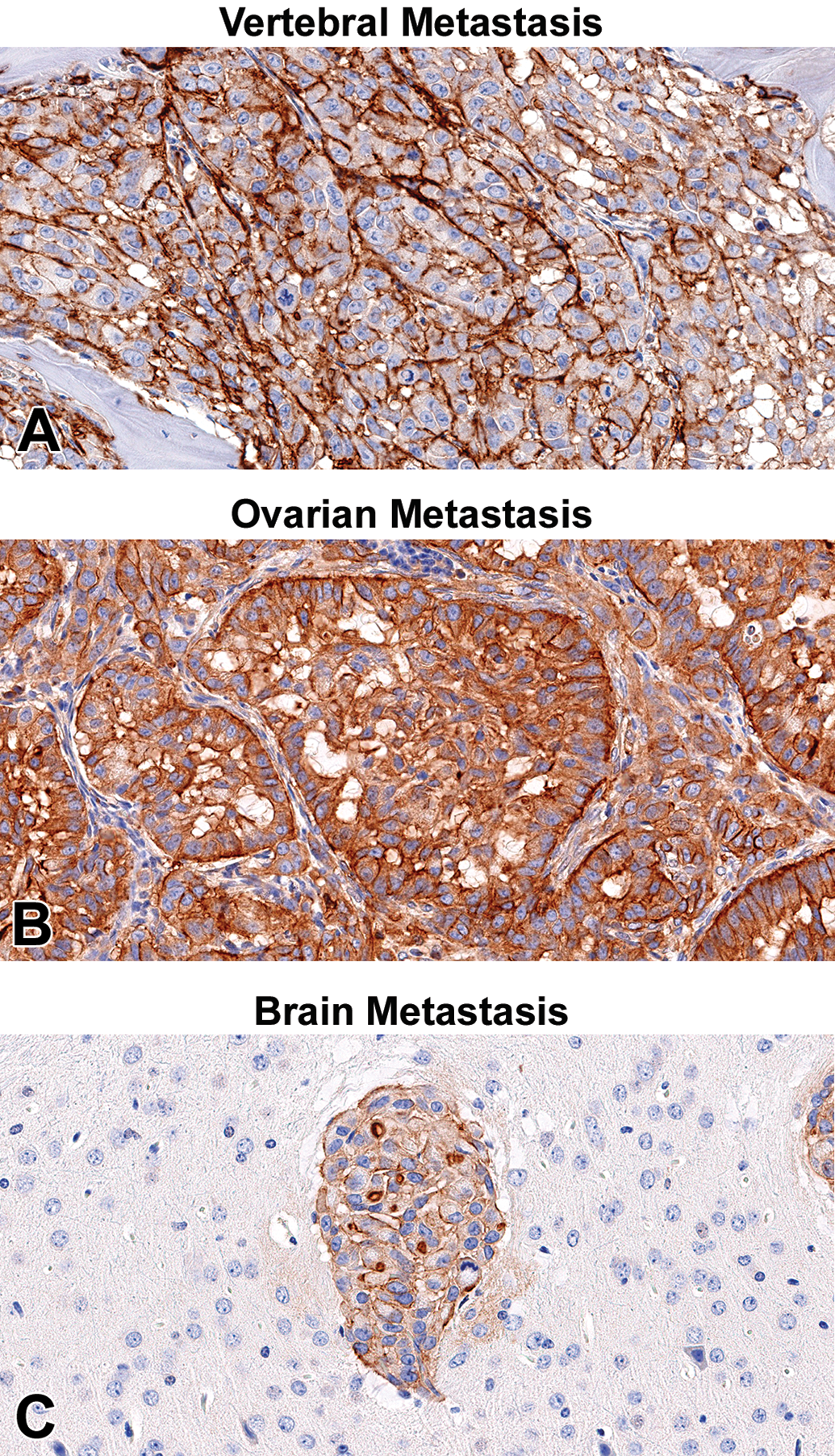
Qualitative analysis of CD44 in vertebral, brain, and ovarian metastases of non-small cell lung cancer. (A, B) Prominent CD44 positive expression was present in vertebral metastases and an ovarian metastasis (original objective 40×). (C) Scant CD44 membranous expression was present in brain metastases (original objective 40×).
Immunofluorescence
Neoplastic cells within a vertebral metastasis demonstrated granular to diffuse, intense, cytoplasmic staining for vimentin (red) and cytokeratin (green; Figure 6A and B). Costaining of vimentin and cytokeratin, identified in yellow, was present within the same neoplastic cell population (Figure 6C).
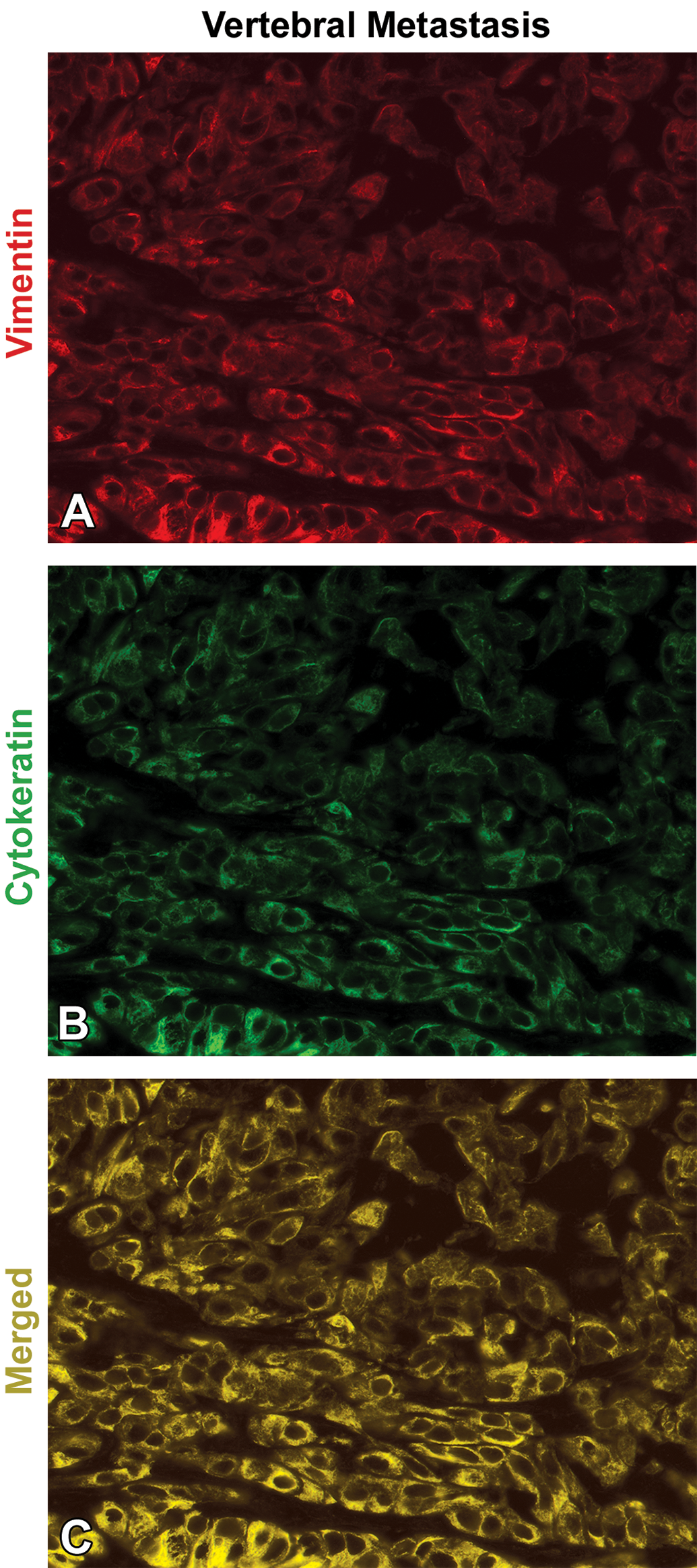
Vimentin and cytokeratin immunofluorescence within a vertebral metastasis. (A, B) Neoplastic cells in the vertebral metastases were diffusely positive for vimentin (red) and cytokeratin (green). (C) Coexpression of cytokeratin and vimentin (yellow) was identified in the vertebral metastasis (original objective 20×).
Discussion
We have characterized vertebral metastases in an experimental model of brain metastatic NSCLC. Our understanding of this disease is enhanced by the stem cell characteristics of these metastatic NSCLC cells and a correlation with EMT in this model. The lesions presented in this study are potential chemotherapeutic targets in NSCLC and could contribute to the development of customized therapies and improved patient survival.
Bone metastases of lung cancer are most common in metastatic NSCLC adenocarcinoma in patients with an incidence of 23.9% to 56.1% (Oliveira, Mello, and Paschoal 2016; Wang, Shen, and Zhu 2015; Zhou et al. 2017). Metastatic lung cancer patients often have multiple bone metastases, a poor prognosis, and a six-month median survival rate (Dropcho 2014; Lei et al. 2016). Common bone metastatic sites include ribs and thoracic vertebrae (Zhou et al. 2017). Metastatic epidural spinal cord compression is most commonly diagnosed in the thoracic spinal cord following metastases to the thoracic vertebral bodies (Patchell et al. 2005; Zhou et al. 2017).
Effective and long-term treatment of bone metastases of NSCLC and other primary tumor metastases continue to be an obstacle in the clinic. Treatment is mainly palliative and consists of analgesics, steroids, radiation therapy, or surgery. The median survival for NSCLC patients following radiation therapy is usually 3 to 6 months, and this time frame is shorter in patients who are unable to walk (Rades et al. 2011; Zaidat and Ruff 2002). Systemic NSCLC treatments have shown a modest improvement in metastatic patients. These treatments include chemotherapy, bisphosphonates, and radioisotopes (Harel and Angelov 2010). Bisphosphonates and Receptor activator of nuclear factor kappa-B ligand (RANKL) inhibitors have been identified as therapeutic osteoclast inhibitors (Brown et al. 2005; Garraway 2013; von Moos et al. 2018; Ulas et al. 2016). The primary clinical end point in spine metastases studies is increased ambulation, and secondary end points include urinary continence, regained functional status, and overall survival (Patchell 2003). To improve efficacy of therapies and overall survival, a reliable model system must be established.
Vertebral metastases of lung cancer have been reported in various experimental models. These metastases have been induced by direct injection of cells into the vertebrae and ICI of tumor cells selected for vertebral metastases. Experimentally induced vertebral metastases has resulted in decreased body weight and four clinical signs consistent with compression of the spinal cord: tail dragging, dorsal stepping, sweeping, and paralysis. Vertebral metastatic lung adenocarcinoma cell lines have demonstrated an increase in migration and invasion and a decreased propensity for adhesion. Histologic features of vertebral metastases of lung cancer include vertebral bone infiltration, trabecular bone destruction with spinal cord and nerve root compression (Cai et al. 2015; Tatsui et al. 2009). While these studies provided a foundation for evaluation of experimental vertebral metastases, we addressed a need for a comprehensive analysis of the cellular phenotype of these metastatic cells.
In this study, vertebral metastases were identified in 14 of 36 mice inoculated with brain-metastatic A549-Br cells. Current models for brain metastasis formation include injecting cancer cells directly into the brain parenchyma, the internal carotid artery, or left cardiac ventricle. Direct injection misses crucial steps in the complex metastatic cascade including intravascular survival, endothelial cell interactions, and extravasation and invasion of the brain parenchyma. Internal intracarotid injection is a technically challenging surgical procedure with a high complication rate in animal models (Balathasan, Beech, and Muschel 2013; Eichler et al. 2011; Fidler 2015). Furthermore, intracarotid injection often results in fatal intraventricular tumors and wider parenchymal metastases, with few brain metastases (Balathasan, Beech, and Muschel 2013).
In contrast, ICI yields a reliable intravascular distribution of cells and reproducible brain metastases. However, blind ICI is less reliable with higher rates of missed injections and a 60% survival rate related to sizable tumor burden formation at the site of injection versus 100% survival 15 days post-ICI (Balathasan, Beech, and Muschel 2013). Additionally, less than 50% of animals formed brain metastases with blind ICI (n = 13) versus 100% brain metastasis formation using ultrasonography guided ICI (n = 8). Similarly, in our results, animal survival was 100% prior to the onset of vertebral paralysis and subsequent humane euthanasia. Additionally, the tumor burden at the injection site was minimal with rare clusters of tumor cells in the subendocardium, myocardium, or pulmonary artery, which did not impact animal survival (Online Supplemental Table S2). Brain-seeking A549 cells have not been utilized in other vertebral metastases models (Moon et al. 2015); therefore, a comprehensive characterization of the pathology and associated metastatic patterns has not been described (Gupta et al. 2013; Lyle et al. 2016; Nounou et al. 2016; Yoneda et al. 2001).
Primary clinical signs of vertebral metastases included… hind-limb paralysis, dragging of the tail, and an absent toe reflex. All female mice had significant weight loss compared to published control values (Hsd:Athymic Nude-Foxn1nu, 2011). Most vertebral metastases were in the thoracic vertebrae, similar to vertebral metastases in humans (Zhou et al. 2017). In some regions, these metastases compressed the associated spinal cord resulting in white matter degeneration. Similar changes have been described in vertebral body metastases in human patients. Fifty-three percent (n = 2,021) of human patients developed bone metastases in thoracic vertebrae possibly due to the close proximity of the vertebral vein to the lungs (Zhou et al. 2017). However, in our model, which was largely devoid of lung metastases, the targeted infiltration and proliferation in the bone marrow was likely associated with high blood flow and release of adhesive and angiogenic factors within the marrow (Chirgwin and Guise 2000).
NSCLC tumors, while commonly separated into types, have been described as heterogeneous in the overall pattern within patients (Dropcho 2014). In this experimental model, primary A549 cells exhibited an adenocarcinoma phenotype in cell culture; cells proliferated in small clusters or sheets. However, striking EMT was present in vertebral body and visceral metastases. Neoplastic cells had two phenotypes, one with adenocarcinoma features with formation of acini and cords and a second with mesenchymal features organized into bundles and streams. In the adenocarcinoma phenotype, PAS-positive mucinous material was associated with cytokeratin-positive epithelial cells in vertebral bone, brain, and visceral metastases. The neoplastic epithelial cells often contained cilia and exhibited a bronchiolar pattern, mimicking that of the primary tumor. Osteoclastic bone resorption was evident in some metastases. These metastatic lung cancer cells may have exhibited an association between invasion and osteoclastic activation, which has been described in breast cancer bone metastases (Kruger et al. 2014; Papotti et al. 2006). Tumor cells in patients often stimulate osteoclasts resulting in bone resorption and release of growth factors, including transforming growth factor-β, insulin-like growth factors, and fibroblast growth factors, further stimulating metastatic invasion. Additionally, tumors with a propensity for bone metastases, including lung cancer, breast cancer, and multiple myeloma, often exhibit osteomimicry with upregulation of bone sialoprotein and osteopontin to promote tumor binding, matrix metalloproteinase (MMP) activation, and angiogenesis (Kruger et al. 2014; Papotti et al. 2006).
EMT is well described in the literature in A549 cells, including an association with cisplatin and paclitaxel resistance associated with cathepsin L, a lysosomal protease that contributes to tumor metastasis and cancer progression (Han et al. 2016). Transcription factors and signaling pathways contributing to EMT in A549 cells include the IL-6/JAK/STAT, hypoxia induced TGF-1β/Smad pathway and SNAIL via GRB2 resulting in e-cadherin reduction (Lin et al. 2018; R.-Y. Liu et al. 2014; Mitra et al. 2018; Sueki et al. 2014; Tirino et al. 2013) hepatocyte growth factor (HGF) via the human biliverdin reductase pathway (F. Liu et al. 2017), and upregulated Notch signaling (Yuan et al. 2014).
Features of EMT, including loss of e-cadherin and upregulation of MMPs (Pandley et al. 206), have been linked with the presence of a cancer stem cell population (Li et al. 2017), suggesting this subpopulation with the capacity for self-renewal may drive EMT and metastatic progression. Overexpression of ligands involved in increased tumor invasiveness and mobility have been identified in A549 cancer stem cells, including type II transmembrane serine protease TMPRSS4 and CXCR4 (de Aberasturi et al. 2016; Tu et al. 2017). Further, higher cancer stem cell expression in the A549 cell line has been shown to be associated with increased cellular proliferation and more aggressive biological behavior (higher T stage) in patients (Hu et al. 2018; Leung et al. 2010). Therefore, the EMT and cancer stem cells in this model contribute to the propensity for bone, brain, and visceral organ metastases.
Immunohistochemical analysis of tumor cells in vertebral bone, brain, and in the parenchymal metastases in our model system demonstrated an increase in vimentin expression compared to cytokeratin expression. The overall increase was 15% in both vertebral bone and parenchymal tissues and an 11% increase in brain. A number of cells had heterogeneous expression of vimentin and cytokeratin. This heterogeneous expression was supported by costaining of vimentin and cytokeratin in vertebral metastases using immunofluorescence microscopy. This difference in cytokeratin expression was reproducible and not impacted by decalcification. Prolonged decalcification in formic acid has been shown to have no effect on immunoreactivity in lung adenocarcinoma bone metastasis. In contrast to strong acidification techniques, a weak acid decalcification agent preserves antigen reactivity and tissue morphology with a multitude of antigens including vimentin and cytokeratin (Athanasou et al. 1987; Mukai 1986). Additionally, no difference in expression was observed in decalcified vertebral bone sections versus parenchymal and brain metastases.
A correlation between EMT and cancer stem cells of vertebral metastases was identified using immunohistochemistry in our model. EMT is characterized as the reversible shift of neoplastic epithelial cells to mesenchymal stem cells. The changes within the tumor cells are not permanent and likely are not of genetic origin but due to the tumor microenvironment (Jung, Fattet, and Yang 2015). Tumor cells involved in EMT have increased expression of mesenchymal proteins including vimentin, fibronectin, and n-cadherin and a loss of epithelial proteins such as cytokeratin, β-actin, and e-cadherin (Shi et al. 2013; Tsoukalas et al. 2017). Further, these cells exhibit loss of basement membrane polarity, loss of adhesion, and increased invasiveness (Tsoukalas et al. 2017). EMT is associated with the proliferation of cancer stem cells, tumor cell dormancy, resistance to apoptosis, and chemoresistance (Mani et al. 2008; Tsoukalas et al. 2017). EMT is critical for metastatic progression, as this is the primary mechanism that tumor cells use to degrade the extracellular matrix, migrate from the primary tumor, and traverse through the vasculature (Hao et al. 2014). EMT may not be a definitive phenotypical switch in cancer cells but may represent a hybrid or incomplete transition within the metastatic site (Christiansen and Rajasekaran 2006; Sulaiman, Yao, and Wang 2018). This phenomenon may explain the elevated vimentin expression in vertebral metastases with a sustained cytokeratin expression. In lung cancer patients, elevated vimentin expression has been associated with malignancy and poor prognosis (Tsoukalas et al. 2017). Further, in this model, visceral metastases retained a well-differentiated morphology and vimentin expression. EMT has been associated with formation of cancer stem cells along with other features of malignancy. Cancer stem cells are generally identified by expression of CD24, CD29, CD44, CD90, CD133, ALDH1, and epithelial-specific antigen (Hao et al.). The stemness of NSCLC in patients facilitates continuous self-renewal, regeneration, and differentiation (Morel et al. 2008). In our experimental model, there was diffuse expression of CD44 in vertebral metastases and ovarian metastases. This expression was less prominent in brain metastases. Our research has demonstrated elevated vimentin expression, loss of cytokeratin, and prominent CD44 expression in vertebral metastases. These data demonstrate a correlation of vertebral metastases of NSCLC with EMT in a mouse model.
Finally, numerous metastases were present within the peritoneal cavity in our model. Intraperitoneal carcinomatosis is most common in ovarian or gastrointestinal neoplasms (Segelman et al. 2012) but is rare in metastatic carcinomas, occurring in 8% of patients with NSCLC (Lu et al. 2010; Patil et al. 2016). The pathogenesis of peritoneal metastases remains unknown. However, inflammation and tumor growth in a cluster, or spheroid, have been shown to contribute to neoplastic peritoneal invasion (Erin, Hilary, and Ernst 2014; Moradi et al. 1993). EMT and cancer stem cells within the model of NSCLC may have contributed to these metastases.
This study has demonstrated the epithelial and mesenchymal phenotype of vertebral metastases of NSCLC in a mouse model. These metastases developed most commonly in the thoracic vertebrae, which correlate to the most common site of NSCLC vertebral metastases in humans. These data have provided testable hypotheses to improve drug delivery and chemotherapeutic efficacy. This work was limited by the time course of the experiment, a longer course experiment would facilitate complete evaluation of these metastases to chronicity. The primary strength of our study remains that the vertebral metastases developed spontaneously without direct intravertebral inoculation of tumor cells or injection of tumor cells selected for vertebral metastases. This research has provided a detailed characterization of vertebral metastases in NSCLC and can serve as a model for evaluating treatment of bone metastases in NSCLC.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319838491 - Epithelial–mesenchymal Transition Phenotypes in Vertebral Metastases of Lung Cancer
Supplemental Material, DS1_TPX_10.1177_0192623319838491 for Epithelial–mesenchymal Transition Phenotypes in Vertebral Metastases of Lung Cancer by Alexandra M. Dieterly, Gozde Uzunalli, Chinyere M. Kemet, Arvin H. Soepriatna, Craig J. Goergen and L. Tiffany Lyle in Toxicologic Pathology
Footnotes
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Acknowledgments
The authors thank Dr. Margaret, A. Miller, and Dr. GuangJun Zhang for article review. The authors acknowledge the assistance of the Purdue University Histology Research Laboratory, a core facility of the NIH-funded Indiana Clinical and Translational Science Institute.
Author Contributions
All authors (AD, GU, CK, AS, CG, TL) contributed to conception or design, data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was made possible, in part, with support from the Indiana Clinical and Translational Sciences Institute funded, in part, by Award Number UL1TR001108 from the National Institutes of Health, National Center for Advancing Translational Sciences, Clinical and Translational Science Award. Dr. Dieterly is supported by the Ross Graduate Fellowship and Purdue Research Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
