Abstract
GlaxoSmithKline has recently made significant organizational changes to its nonclinical safety, drug metabolism and pharmacokinetic, and laboratory animal science/veterinary functions, with the goal to increase our focus on scientific partnership with the discovery part of the organization. One specific change was bringing together pathologists and comparative medicine veterinarians and scientists into a single functional unit. We describe our early activities (assessing our capabilities and gaps, external benchmarking, listening to our discovery partners, redesigning some of our working practices) aimed at implementing these changes. In addition, early on we held a Discovery Engagement Workshop attended by all pathologists and comparative medicine veterinarians and scientists, as well as selected discovery scientists. The purpose of this workshop was to share learnings from the above activities and devise plans aimed at achieving our overall goal of functional integration: driving pathobiology expertise into drug discovery and increasing the human (translational) relevance of experimental data. This review describes the new organizational structure, the workshop activities, and implementation plans; updates our progress; and considers the opportunity for a pan-industry network of discovery-focused pathologists and comparative medicine veterinarians and scientists.
The idea of applying the full range of pathology expertise to questions other than preclinical toxicology is not new (see Alden et al. 1999; Arp 1999; Bhoumik et al. 2017; Boorman et al. 2002; Burkhardt et al. 1999; Burkhardt, Ryan, and Germann 2002; Car and Robertson 1999; Cockerell, Aaron, and Moe 1999; Cockerell, Burkhardt, and Ryan 1999; Ettlin 2013; Everitt 2015; Fielden and Kolaja 2008; Kramer, Sagartz, and Morris 2007; MacDonald and Robertson 2009; Ramaiah and Walker 2016; Rudmann and Durham 1999; Theodosiou, Amir-Aslani, and Megarbane 2014; Tomlinson et al. 2013; van Tongeren et al. 2011), and several companies have well-developed efforts in this arena. Building on our own successful local site-based or individual discovery pathology engagements, and by competitor awareness, in 2016 a major reorganization of the nonclinical safety, laboratory animal science, and drug metabolism–pharmacokinetics (DMPK) functions within GlaxoSmithKline (GSK) also created an explicit expectation that our skills and expertise should be used from early target identification through clinical trials and postmarketing surveillance, leading to major changes in the way we work and our developing expertise. The comparative medicine veterinarians and scientists who have had a major focus on the clinical care of animals, as well as providing surgical support and comparative medicine expertise, are now aligned with the pathology, histotechnology, and clinical pathology groups in a single department, Translational Medicine and Comparative Pathobiology (TMCP) and with an increased focus on animal model selection, validation, and translation, as well as the traditional roles of replacement, reduction, and refinement of animal use. This review describes ongoing collective efforts to broadly achieve fully active involvement of our pathologists, comparative medicine veterinarians and scientists, clinical pathologists, and histologists in support of the aspirations of TMCP.
As part of the push to achieve this, we undertook several initiatives: Creation of several workstreams looking at all aspects of our work and how this might be improved to provide greater benefit to our discovery partners. A benchmarking survey of our veterinary and pathologist colleagues in 22 other companies to assess how they were addressing similar issues. A worldwide face-to-face meeting of GSK pathologists, comparative medicine veterinarians and scientists, clinical pathologists, senior histologists, and discovery and other partner representatives to discuss the output of the above and to advance proposals for best practice.
While this transformation is a work in progress, we provide a summary of these efforts, and our progress so far, as well as highlighting some of the challenges.
Background—A New Organizational Structure
In 2016 GSK’s nonclinical safety, DMPK and laboratory animal science functions were combined into a single global organizational structure to promote cross-disciplinary achievement potentially hindered by the prior structure and to better apply comparative pathobiology and medicine approaches to improve translational relevance of efficacy, safety, and biomarker end points. One potentially unique approach is the pairing of scientists with a pathobiology (pathologists) and comparative medicine (research-focused laboratory animal veterinarians and in vivo biologists/scientists) backgrounds with each discovery therapeutic area group. In addition, the comparative medicine veterinarians and scientists have the objective to connect the therapeutic area groups with other internal in vivo and in vitro expertise such as immunotoxicology, safety pharmacology, reproductive toxicology, and complex in vitro models (CIVM). The senior leadership of TMCP currently consists of pathologists and veterinarians.
Scope of the New Organization
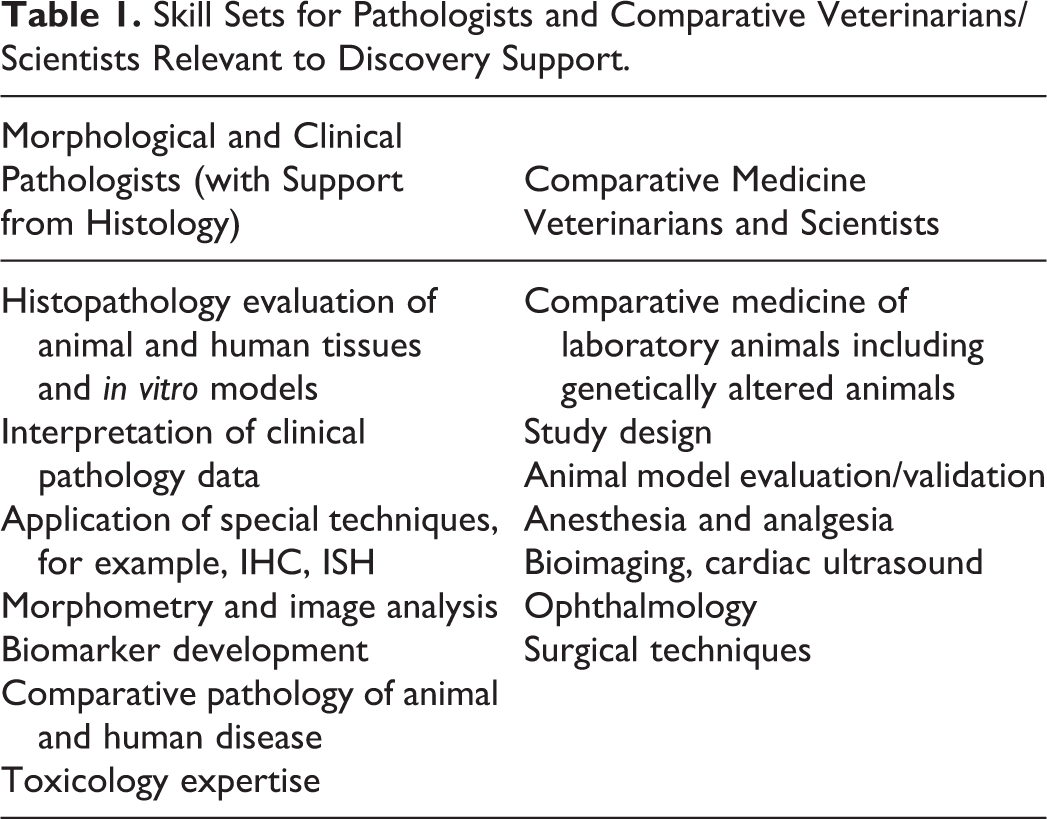
The senior leadership of TMCP consists of pathologists and veterinarians who share a bold vision to transform how GSK research and development (R&D) applies animal models and non-animal alternatives (e.g., human tissue/cell-derived in vitro models), emphasizing multiple discipline data integration and line of sight to the clinic, with the overall goal of reducing late stage project attrition. Specifically, the roles of comparative pathobiologists and comparative medicine veterinarians and scientists were broadened, and individuals in these roles were given the freedom to work with increased scientific continuity across preclinical discovery and development stages increasing their contribution to target validation, efficacy modeling, and translational biomarker studies. Achieving this requires a close working relationship with our discovery partners, creating a sense of combined ownership of early stage programs, collaboratively defining the critical translational questions, and the most relevant complex biology models. Key enablers include our comparative pathobiology and comparative medicine expertise, drug development and regulatory expertise, effective application of technologies to answer critical questions with commitments to animal care and welfare, and quality and data integrity principles as well as leveraging the depth of expertise that currently exists across discovery groups. Most recently, the larger department that TMCP reports into was organizationally transferred to Research, and we will universally share Discovery goals going forward which should further support our vision. The specific skills that we considered relevant to these aims are listed in Table 1.
Skill Sets for Pathologists and Comparative Veterinarians/Scientists Relevant to Discovery Support.
Challenges and Approaches
An advantage of the relatively narrow but different areas of focus of the previously existing safety assessment, laboratory animal sciences, and DMPK departments was that their expertise was utilized in a relatively discrete aspect of the drug discovery and development process with clear expectations and accountabilities. As these existed historically, GSK therapeutic areas understood the available resources and when to request them, often without leveraging the full range of expertise and technology existing in these groups. In addition, this traditional structure favored therapeutic areas directing information flow and specific questions to individual departments, but often at the expense of information being siloed and early opportunities to positively intervene being missed. This compartmentalized structure also promoted a service provider relationship rather than an engaged scientific partnership. Some important areas of scientific need were unmet and need for increased translational relevance may not have been appropriately recognized by some discovery scientists.
While frequently represented as specific and linear processes, drug discovery and development are better served by addressing scientific challenges in a more holistic manner. Therefore, valuable collaboration by pathologists and comparative medicine veterinarians and scientists should occur at any stage of a developing program. For that opportunity to be realized, this new organization requires earlier and more frequent communication around therapeutic area program directions, needs, and research. Finding the appropriate processes to share timely information while balancing the need for individuals to use their time efficiently and effectively is one of the challenges we encountered.
As this new organization becomes more successful at increasing our involvement throughout the drug discovery and development processes, prioritization of our resources across therapeutic area programs will become a more frequent concern. While questions on prioritization of efforts within a given therapeutic area can be directed to therapeutic area leadership, prioritization across GSK R&D requires input from more senior leaders, as portfolio interests and stage of development are considered.
As we began to explore and establish this new working paradigm, several challenges and questions related to our new approach were articulated: What capability gaps can we identify and address? Where are the opportunities to engage with discovery at its earliest stages and what is needed to deliver? Focusing on successful existing discovery interactions, what makes those interactions successful and what can we do to facilitate interactions in the absence of a preexisting relationship? How can we benefit from understanding how other pharmaceutical companies are currently working (both organizationally and technically) with discovery and what can we learn from our external colleagues approaches to help strengthen our ways of working? Which technologies and approaches benefit most from a centralized approach and what are the compromises? How can we create an infrastructure that we consider valuable while maintaining simplicity and agility for our research and development collaborators? Are there opportunities to improve the effectiveness of lead optimization by applying an integrated understanding of pathophysiology? With a discovery focus, how do we apply our expertise to important efforts and capabilities such as biomarkers, clinical pathology, in vivo and in vitro models, and the use of human samples? How do we use this new department to enable and promote strategic R&D initiatives? How do we best capture our discovery-focused efforts and impacts of these efforts? While working toward all the above, how do we maintain leading edge expertise and knowledge and provide opportunities for learning?
Implementation Activities
Although organizational changes had already been made to promote an increased focus on, and integration with, discovery, these alone were clearly not sufficient to fulfill our vision. We recognized the need to transform our department from the traditional roles toward new and better ways of working with discovery. A Discovery Engagement Workshop (DEW) was therefore organized with the objective of increasing our understanding of discovery and reaching global consensus on the best approaches to address specific issues and challenges.
DEW and Outcome
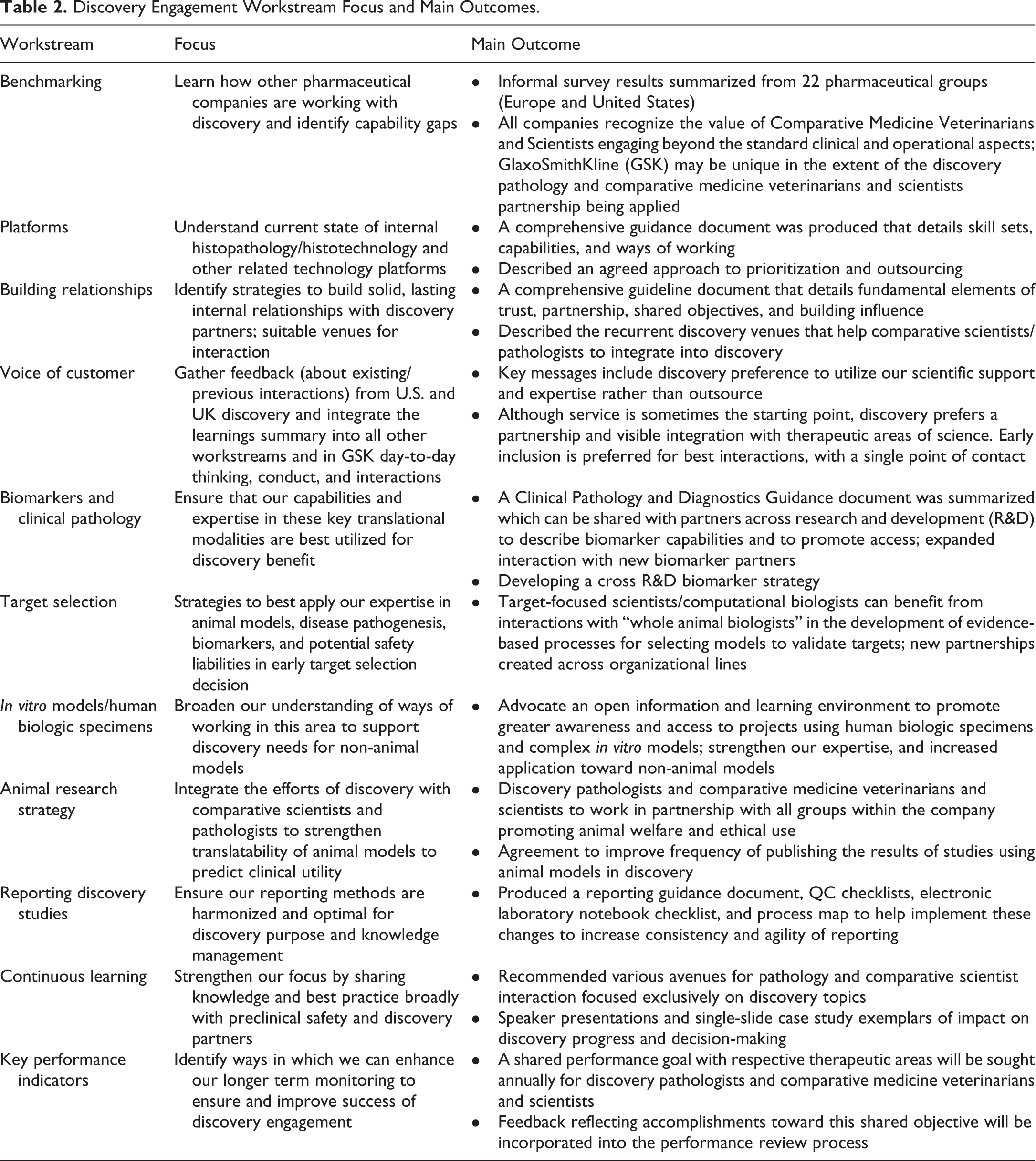
Eleven workstreams were commissioned in advance of the DEW. These workstreams identified a desired future state, articulating benefits and key activities and output (guidance documents, agreements, recommendations) needed to achieve that state. The workstreams, their goals, and outcomes are summarized (Table 2), and selected workstreams are further discussed in the subsequent section.
Discovery Engagement Workstream Focus and Main Outcomes.
Benchmarking
The benchmarking workstream surveyed 22 pharmaceutical/biotech companies in the United States and Europe, by e-mail and then telephone follow-up, to learn how these companies approach discovery pathology support and application of expertise in comparative medicine and laboratory animal science. An informal questionnaire was used to guide these discussions. Questions included: How have you used “brand” development (what expertise and efforts are you or are you trying to be known for; such as early safety, efficacy, target validation, model validation) to improve discovery-focused interactions and impacts? Are there any individual contributor-level actions that you have found helpful (or unhelpful)? How about management level actions? Are there specific processes/tools that you have used to incorporate comparative scientists in early project stages; including embedding of pathologists/comparative scientists within therapeutic areas; specific group of pathologists focused on discovery pathology (or just part of wider workload)? How do you approach special techniques request from discovery groups (external vs. internal, wet lab work, image analysis)? Are there specific roles for comparative scientists? How do you handle time lines and prioritization (needs of one therapeutic area vs. another, regulatory efforts vs. discovery, therapeutic area focused vs. clinical/programmatic/regulatory)? Do you have reporting guidelines for capturing these discovery-focused efforts and impacts?
The major points from this subjective survey are summarized below.
All companies recognize the value of pathology support for discovery projects and either do a range of discovery support already or have plans to implement it. Where this support is well-developed, it covers all aspects of discovery using all available pathology tools. Geography (distance between “research” and “development” sites) was often a factor in why discovery support did or did not happen. All companies recognize the value of comparative medicine veterinarians and scientists engaging beyond the clinical and operational, and there is a spectrum of resource and formality for discovery engagement. We have not identified other groups which formally focus on the pathologist/comparative medicine veterinarians and scientists’ partnership, as in the group within GSK.
No matter how good work processes are, importance of personal relationships and face-to-face communication cannot be overemphasized. Pathologists and comparative scientists should feel 100% part of the team, not as part of a customer/client relationship. Staff turnover/redeployment, often because of changing focus and priorities in discovery, can be higher than in preclinical safety; meaning relationships may sometimes need to be rebuilt. Those companies considered successful in discovery support advised that we must commit to meeting agreed time lines and cannot just try to fit this discovery work in around other priorities.
Voice of the Customer (VOC) and Building Relationships
VOC feedback showed a broad appreciation for the contributions and collaboration pathologists and comparative scientists have provided toward discovery decision-making. Our support was considered valuable and essential, and this was reinforced by the preference to utilize our scientific expertise rather than outsource. Although service provision is often a starting point, discovery prefers a partnership and visible integration with discovery therapeutic areas of science, that is, “skin in the game.” Early inclusion is preferred for best interactions, such as input in the design of studies and scheduling.
Discovery partners need to understand how much and when they can rely on us, especially in terms of responsiveness, timeliness, and agility. They recognized other demands on our time and the need to retain the right to outsource technical work when necessary. We can help guide to preferred providers and enable our continued intellectual input.
Importantly, there was a universal desire to better understand our roles and capabilities as well as a simplified interface.
The relationship workstream focused on strategies to build solid, lasting internal relationships with discovery partners, creating discovery engagement meetings to encourage interaction, understanding the behaviors needed for our success in building influence and trust, identifying the key contact points with discovery decision-making, and strengthening our own team matrix interactions for best delivery. This team created a comprehensive guideline document that detailed fundamental elements of trust, partnership, shared objectives, and described the recurrent discovery venues that pathologists and comparative medicine veterinarians and scientists should seek to attend to help integrate into different therapeutic areas with emphasis on the importance of relationships and communication. The partnered approach will be reinforced and will mature over time with increased integration into each therapeutic area. Endorsement that the discovery pathologist and comparative medicine veterinarian/scientist roles should have strong therapeutic orientation, so that relationships and scientific continuity established with early discovery work will continue as the program evolves to a development project. Transformations resulting from these workstreams include integration of a specific pathologist and comparative medicine veterinarian and scientist into each therapeutic area, with single point of contact clearly identified. There is an expectation now that no piece of discovery work can be requested without input first from a discovery pathologist (and comparative scientist as needed). We have strived for more frequent personal contact and colocation (with discovery groups) wherever possible and believe this will improve effectiveness. Relationships matter, and they improve as we learn from, and about each other.
Platforms
The goal of this workstream was to understand the current state of histopathology/histotechnology and other internal technology platforms and to build consensus on the pathology tools we apply including quantitative image analysis, molecular markers (e.g., immunohistochemistry, in situ hybridization), optimal sample collection and analysis methods with a focus on common investigative and imaging technical platforms used to support discovery along with identification of additional training that pathologists and histologists may require to be proficient in selected techniques and their interpretation. A comprehensive guidance document was produced that described our unique skill sets and capabilities in the application of these technical platforms; provided a description of essential tissue-based techniques and proper methods for sample acquisition; gave a consensus position on qualitative and quantitative imaging approaches; and described an agreed approach to outsourcing as well as continuing education for histologists and pathologists.
Biomarkers, Clinical Pathology, In Vitro Models, and Human Biologic Specimens (HBS)
The goal of these workstreams was to ensure that our capabilities and expertise in these key translational modalities are best utilized for discovery benefit. Earlier integration of comparative scientists and discovery pathologists into animal model strategy will help promote access to biomarkers, clinical pathology, in vitro models, and HBS and to support discovery efforts. Greater collaboration with scientists in these areas, to expand utilization of those resources, will be pursued through core team memberships on established forums and participation in a program-facing consultative format. Increased opportunity to support target safety concerns will be promoted through early active engagement with, and periodic review of target safety review documents. A Clinical Pathology and Diagnostics Guidance document was developed and shared with partners across R&D to describe biomarker capabilities and to promote access.
While in vitro models and HBS is an evolving area, the workstream established a baseline of understanding among comparative veterinarian/scientists and discovery pathologists and advocated for developing an informative and learning environment to promote greater awareness and access to projects using HBS or CIVM as well as refinement of reporting guidelines for experiments using HBS. Awareness of the absence of a single internal-validated biobank, lack of a global tracking system, vendor sourcing deemed insufficiently reliable for disease verification of HBS, and the need for internal capacity to validate these samples was raised.
Target Selection
This workstream focused on how to best apply our expertise in animal models, disease pathogenesis, biomarkers, and target safety, in the interface with the GSK target sciences (TSci) group and discovery partners, to progress a coordinated, questions-based approach (QBA) to select and accelerate advancement of the best targets, to benefit patients.
The team identified several areas where pathologist and comparative veterinarian/scientist expertise in anatomic and clinical pathology, in vivo biology, critical assessment of animal models, understanding of the pathobiology of disease across species, and knowledge of drug development will all add value to ongoing target selection and validation efforts to generate human-relevant evidence of target tractability.
Target identification and hypothesis generation are frequently initiated without consideration for whether there are platforms available to functionally evaluate the target or mechanism, for example, availability of relevant animal models or suitable human biological samples. Pathologist and comparative veterinarian/scientist may bring increased understanding of the limitations of existing/published disease models and a more integrated approach for the development of evidence-based processes for selecting the most translationally relevant model. To help implement greater integration and collaboration, we have established a specific network of named contacts between comparative medicine veterinarians and scientists, discovery pathologists and TSci, organized by therapeutic area, which promotes consultancy and facilitates filling target identification and validation gaps.
Animal Research Strategy
The goal of this workstream was to help select, deliver, and optimize the most translationally relevant nonclinical models (“best model”) by integrating efforts of various groups involved in animal model selection, to strengthen translatability of animal models to predict clinical value. The DEW was used to communicate GSK’s animal research strategy, the use of QBAs to define model validity and aid in strategic model selection and the use of after action reviews and highlighted examples, with best practices, of engagement in target and animal model decisions. Discovery pathologists and comparative medicine veterinarians and scientists now serve as “ambassadors” for the translational modeling principles (target homology and tissue-specific distribution, pharmacodynamic responsiveness, similarity to human pharmacokinetics and disposition, metabolite profile, physiological and toxicological human relevance, study-specific and animal welfare considerations, ethical considerations, and historic background data) to help (i) drive these principles toward broader adoption and (ii) more formally assess the strengths and limitations of any given model. A curriculum was proposed to continue to embed animal research strategy principles and examples throughout TMCP. This is particularly important as we continue to venture into numerous and complex transgenic and chimeric mouse models.
Conclusions
Internal consultation with our discovery partners as well as external benchmarking showed overwhelming support for the value of discovery pathologists and comparative medicine veterinarians and scientists applying their varying skill sets to early discovery projects. Within GSK, we have recently implemented aggressive plans to fully integrate our scientific expertise within the various therapeutic areas in GSK. The approach taken has been fundamentally based on providing a true partnership rather than a service-client arrangement. The most successful partnerships are reflected in shared goals, full integration of scientific input, and ultimately blurred organizational lines. In addition, we have not limited our support to addressing only early safety questions. Our support has also extended across target validation, animal model and in vitro model validation, and safety assessment and efficacy end points. In doing so, individuals are expanding their knowledge of more advanced techniques, pharmacology, human pathology, and in vitro models. The strategy we have adopted has inevitably led to a significant increase in demand for our skills, and this has increased the need for prioritization and occasionally for strategic outsourcing. As a group, we are continuing to support full development projects (especially peer reviews of regulatory studies), and we maintain the flexibility to assign individual pathologists to discovery and development studies as required. Although development projects are usually considered higher priority, their needs tend to be more predictable. One challenge is that discovery groups work to even shorter time lines than that are required for regulatory toxicology studies. While data integrity is equally important for discovery studies, as it is for regulatory toxicology studies, we have found reporting such studies can be done with more agile formats.
Having recently passed the one-year anniversary of the DEW, it has been possible to reflect on the positive impact and benefits of the combined organization realignment herein described. The broad ranging output of the DEW has been realized through increased alignment, accountability and a range of new or increased activities, predicated largely on strengthened relationships, and intellectual investment in discovery-centered science. Some of the specific actions taken and their impact were: Involving representatives from the discovery part of the organizations in the DEW led to mutual raised awareness as well as agreed expectations and commitment from all parties. The restructuring of our organisations with its explicit additional focus on discovery has empowered and motivated our pathologists and comparative medicine veterinarians and scientists to be increasingly proactive in offering our skills to discovery and spending more time physically colocated in discovery laboratories and offices. Senior managers in both parts of the organization have made clear the expectation that pathologists and comparative medicine veterinarians and scientists are seen as an integral part of discovery program teams, and this has led to dramatically increased consultation and actions associated with greater input on target validation and early assessment of efficacy, more strategic selection of animal models and biomarkers (including deselection of some models based on enhanced characterization), generation of new models, crossline scientific reviews, improved study design and robustness, greater demand for and application of investigational pathology methods, earlier readout on potential liabilities of targets, and participation in after action reviews to assess the translatability of nonclinical models used for compounds which failed to achieve efficacy in clinical development.
We will continue to refine our approach and working practices internally as we learn from our experiences. Our experience with this DEW and improving integration between our therapeutic areas and GSK pathologists and comparative medicine veterinarians and scientists internally suggests that there would be value of exploring this integrated approach at a cross institution, professional society level. Since those involved in the workshop are active participants in professional organizations including Society of Toxicologic Pathology, Society of Toxicology, European Society of Toxicologic Pathology, British Society of Toxicological Pathology, American Association of Laboratory Animal Scientists, Laboratory Animal Science Association, and others, an effort to explore this further by panel discussions at relevant meetings, cross society collaborations, and other activities that would bring together individuals interested in a more integrated approach to drug discovery merits consideration by those organizations. To our knowledge, there is no formal group that draws comparative medicine veterinarians and scientists and discovery pathologists together. We welcome feedback from any of our colleagues working at different companies—either directly or through this journal.
Footnotes
Acknowledgments
The authors would like to acknowledge Chris Powell, Rich Miller, Andrea Haynes, Edith Hessel, and John Lepore who sponsored the Discovery Engagement Workshop. We would also like to thank our many colleagues in GlaxoSmithKline who provided input to the workshop (>60) and to this article; they are collectively too numerous to include everyone but we would like to specifically acknowledge Mike Kelly, Katherine Nevin, Jessica Neisen, Andrew Pearce, Mark Creighton-Gutteridge, and Jan Roger. We would also like to thank Joanne Storey and Brian Berridge for their leadership in the animal research strategy workstream. Finally, many colleagues working for other companies gave up their time to provide feedback for our benchmarking survey which was extremely useful, and we hope that we have represented their comments fairly. Since they provided their input anonymously, we cannot name them, but you know who you are!
Author Contributions
Authors (SMoh, SMag, JK, RA, RH, PC) contributed to conception or design; data acquisition, analysis, or interpretation (SMoh, SMag, JK); drafting the manuscript (SMoh, SMag, JK); and critically revising the manuscript (RA, RH, PC). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors (SMoh, SMag, JK, RA, RH, PC) are employees of GlaxoSmithKline.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The article is funded by GlaxoSmithKline.
