Abstract
The 6-month Tg.rasH2 mouse carcinogenicity model provides an acceptable alternative to the 2-year carcinogenicity study in CD-1 mice. However, key questions related to the use of this model for testing antisense oligonucleotides (ASOs) include the similarity in the biologic response between mouse strains and the feasibility of using data from the CD-1 mouse to set doses and dose schedules for a Tg.rasH2 carcinogenicity study. To evaluate the potential strain differences, four distinct 2′-O-(2-methoxyethyl) ASOs were administered to CByB6F1 (wild type), Tg.rasH2 (hemizygous), and CD-1 mice. There were no meaningful differences in clinical signs, body weight, food consumption, or serum chemistry and hematology parameters. Histopathology evaluation indicated little to no difference in the spectrum or magnitude of changes present. The cytokine/chemokine response was also not appreciably different between the strains. This was consistent with the similarity in ASO concentration in the liver between the mouse strains tested. As the class effects of the ASOs were not meaningfully different between CD-1, CByB6F1, or Tg.rasH2 mice, data from nonclinical studies in CD-1 mice can be used for dose selection and expectation of effect in the Tg.rasH2 mouse.
A carcinogenicity study provides essential data for the completion of the nonclinical toxicology program for a drug candidate and is of considerable importance for the product label. However, the cost and duration of traditional 2-year rodent carcinogenicity studies have significant impact on the overall drug development process. Another challenge to the interpretation of 2-year rodent carcinogenicity studies is the high background incidence of certain tumors in aged animals. These, along with questions about the relevance of carcinogenicity testing in standard 2-year rodent studies to determine actual risk to humans, are prime reasons for the exploration of transgenic mouse models that involve shorter study durations, lower costs, and offer a further benefit of reducing animal numbers, that is, directly addressing the 3Rs principles (Replacement, Reduction and Refinement) of animal use (Alden, Smith, and Morton 2002; Cohen, Robinson, and MacDonald 2001; MacDonald et al. 2004; Nambiar and Morton 2013).
Of the alternative models evaluated to date, the 6-month study in the hemizygous CByB6F1 -Tg(HRAS)2Jic mouse (hereafter referred to as Tg.rasH2) has emerged as the leading model for the evaluation of drug candidates with the potential to investigate diverse routes of administration to address genotoxic or nongenotoxic mechanisms of action (Eastmond et al. 2013; Usui et al. 2001). However, relevance of the model hinges on the similarity in the biologic (pharmacological and/or toxicological) response of the Tg.rasH2 mouse to standard mouse strains commonly used in drug development.
Tg.rasH2 mice are produced by breeding male transgenic C57BL/6J mice with wild-type Balb/CByJ female mice (Nambiar and Morton 2013). The desired offspring is hemizygous (tg/wt) for the human c-Ha-ras gene. Genotypic analysis is required to confirm the hemizygous status, as breeding also produces CByB6F1 -Tg(HRAS)2Jic wild-type (wt/wt) (hereafter referred to as CByB6F1) mice. In the hemizygous mouse, the c-Ha-ras gene is incorporated along with its promoter/enhancer elements and a single-point mutation in the final intron of the 3’-end (Maruyama et al. 2001; Machida et al. 2008; Urano, Tamaoki, and Nomura 2012). This human transgene encodes for the p21ras protein, a protein that has been implicated in tumorigenesis, including that for humans (Abbas and Dutta 2009). Expression of the p21ras protein in Tg.rasH2 mice occurs in normal healthy tissues and is increased approximately 2-fold in endogenously occurring tumors (Maruyama et al. 2001). The transgenic human c-Ha-ras gene, and subsequent p21ras protein, is proposed to operate in conjunction with endogenous mouse ras genes to increase the carcinogenic potential of a drug (or chemical).
The founder strains for Tg.rasH2 and CByB6F1 mice, C57BL/6J and Balb/C mice, have an extensive history in research and development; nevertheless, the transgenic status represents a novel strain, and the potential differences in pharmacological and/or toxicological profiles must be taken into consideration for a drug development program. The CD-1 mouse strain is often used in nonclinical studies for early and late phase drug development, and the introduction of a second (or possible multiple) strain(s) into the development program can be a concern. CD-1 mice have been the preferred strain of mice in toxicology studies with antisense oligonucleotides (ASOs). The pharmacokinetics, pharmacodynamics, and toxicology of ASOs have been well described in the literature (Henry et al. 2001; Geary et al. 2007; Henry et al. 2008; Zanardi et al. 2012; Crooke et al. 2016; Shemesh et al. 2016). Long-term toxicology studies in CD-1 mice have been reported for several ASOs (Zanardi et al. 2018) and 2-year rodent carcinogenicity studies have been completed for selected ASOs in CD-1 mice and Sprague-Dawley rats. These studies provide essential information in the evaluation of ASOs as a drug class and for the interpretation of results for specific ASOs.
The Tg.rasH2 model is an attractive alternative to the traditional 2-year rodent carcinogenicity study, but only a limited number of drug and chemical classes have been tested and are reported in the literature; none of which are published reports featuring ASOs or other oligonucleotide-based drugs. To this end, a series of studies were initiated to compare and contrast the results from four prototypical 2′-O-(2-methoxyethyl) (2′-MOE) ASOs previously evaluated within development programs. This included ASO sequences identified as pro-inflammatory during routine screening in CD-1 mice. Studies reported herein involved the Tg.rasH2 and the CD-1, as well as the CByB6F1 mice. The latter was included since dose range finding studies of 5 or 28 days duration are often conducted in the CByB6F1 mice (in lieu of using hemizygous) to reduce overall animal costs and to adhere to the 3Rs principle. Also, data collected in standard mouse strains within the toxicology program are relevant for designing studies in Tg.rasH2 mice, and conversely, data in the transgenic mouse can be used with confidence for the overall evaluation of a drug candidate. Thus, the ultimate goal of the present studies was to characterize the biologic response of Tg.rasH2 and CByB6F1 mice to 2′-MOE ASOs and to determine whether the doses used in long-term repeated dose studies in CD-1 mice provide the necessary data for selecting doses in carcinogenicity studies using Tg.rasH2 mice.
Materials and Methods
Animals
All in-life activities involving animals were conducted in accordance with MPI Research, Inc. (Mattawan, MI) Standard Operating Procedures and were approved by the facility Institutional Animal Care and Use Committee. Studies were based on current International Conference on Harmonization (ICH) Harmonized Tripartite Guidelines and generally accepted procedures for the testing of pharmaceutical compounds (i.e., ICH M3(R2) 2010; ICHS6(R1) 2009) and the Guide for the Care and Use of Laboratory Animals, Institute of Laboratory Animal Resources (National Academy Press, Washington, DC, 2011).
In study 1, male and female CByB6F1 mice, approximately 9 weeks of age at receipt, were obtained from Taconic Biosciences (Germantown, NY). Male and female Crl: CD1®(ICR) mice, approximately 8 weeks of age at receipt, were received from Charles River Laboratories (Raleigh, NC). All animals were allowed an approximate 2-week acclimation period prior to initiation of study events.
In study 2, male and female Tg.rasH2 mice, approximately 4 weeks of age at receipt, were obtained from Taconic Biosciences (Hudson, NY). Animals had an approximate 5-week acclimation period prior to initiation of study events. Male and female Crl: CD1®(ICR) mice, approximately 10 weeks of age at receipt, were obtained from Charles River Laboratories (Stone Ridge, NY). Animals were allowed an approximate 1-week acclimation period prior to initiation of study events.
In both studies, animals were randomized into groups using body weight as the randomization factor. Each study design is outlined in Table 1.
Study Designs.
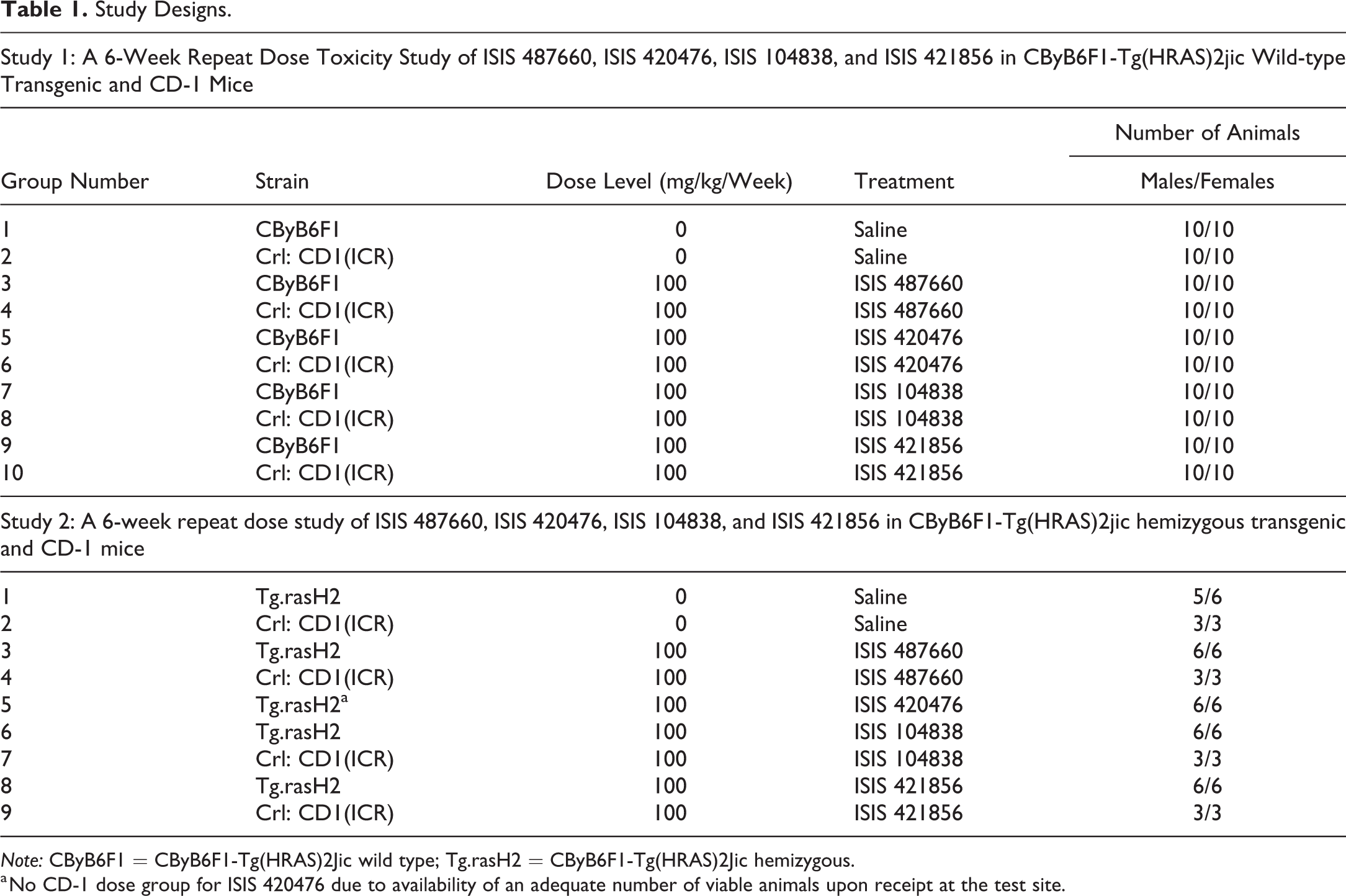
Note: CByB6F1 = CByB6F1-Tg(HRAS)2Jic wild type; Tg.rasH2 = CByB6F1-Tg(HRAS)2Jic hemizygous.
a No CD-1 dose group for ISIS 420476 due to availability of an adequate number of viable animals upon receipt at the test site.
Sequence, Target, and Cross-Species Activity of the Antisense Oligonucleotides (ASOs).
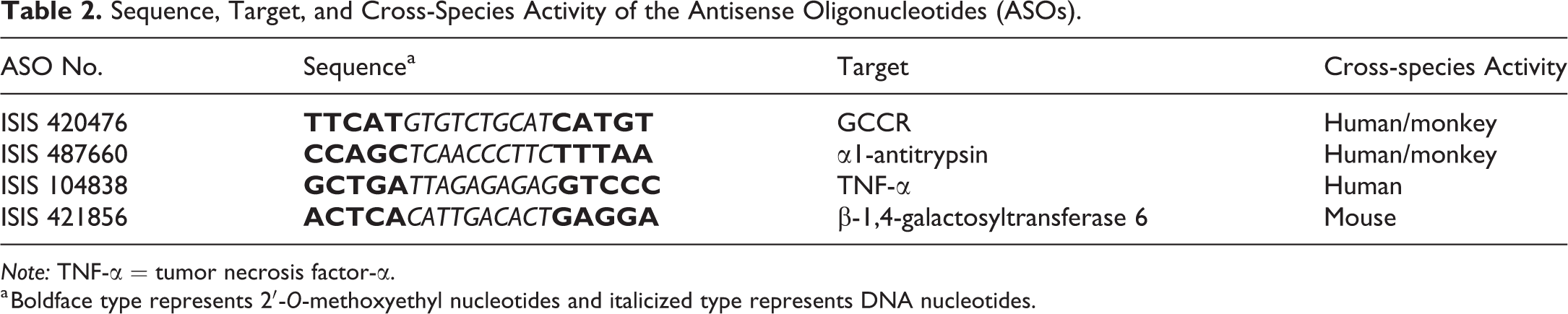
Note: TNF-α = tumor necrosis factor-α.
a Boldface type represents 2′-O-methoxyethyl nucleotides and italicized type represents DNA nucleotides.
ASOs
The sequence, modification pattern, targeted gene, and cross-species activity for each of the ASOs (ISIS 487660, ISIS 420476, ISIS 104838, and ISIS 421856) are provided in Table 2. Each ASO can be described as a “5-10-5 MOE gapmer” in which 2′-MOE nucleotides comprise the first five positions in the 5′ and 3′ termini (indicated in boldface text) with deoxy nucleotides in the central region. Cytosines are 5-methyl cytosine and a phosphorothioate backbone occurs throughout the entire sequence.
Experimental Design
Administration
Saline (control) and 2′-MOE ASOs were administered via subcutaneous injection. A loading dose followed by a maintenance dose schedule was followed in which doses were administered on days 1, 3, 5, and 7 and then once weekly after that (days 14, 21, 28, 35, and 42). Two sites (intrascapular and a region proximal to the tail) were designated for injection, with dose administration systematically rotated between the two sites.
Justification of dose level
Four 2′-MOE ASOs were selected specifically to cover the range of known ASO-mediated pro-inflammatory effects in mice. The dose of 100 mg/kg was selected based on data generated from previously conducted studies where this dose was shown to be markedly pro-inflammatory in CD-1 mice administered ISIS 421856 and minimally to mildly pro-inflammatory in mice administered ISIS 104838, ISIS 420476, or ISIS 487660.
In-life examinations
All animals were observed for morbidity, mortality, and injury twice daily. Body weights and food consumption were recorded weekly. Detailed clinical observations were conducted on days 1, 3, 5, and 7 and then weekly (to coincide with dose administration). In addition to evaluation of the dose site, examination involved visual assessment of all external surfaces and appendages, as well as observations for any neurological effects such as tremors, convulsions, reactivity to handling, or any unexpected behaviors.
Terminal procedures and postmortem evaluations
For study 1, hematology (first 5 animals/sex/group) and serum chemistry (last 5 animals/sex/group) were evaluated prior to necropsy on day 44. The order of bleeding was alternated among the dose groups to minimize potential time bias. Postmortem evaluations included the collection of representative tissues (adrenal glands, aorta, bone marrow [femur and sternum], gallbladder, heart, injection sites, kidneys, liver, lungs, mandibular and mesenteric lymph nodes, ovaries, pancreas, prostate, seminal vesicles, spleen, testes, thymus, uterus with cervix, and vagina) for histopathologic examination. To determine tissue concentrations of ASO, liver samples were collected from CD-1 and CByB6F1 mice administered ISIS 421856.
For study 2, blood (3–6 animals/sex/group) was collected prior to necropsy (day 44) and processed for serum cytokine/chemokine analysis. Selected organs (bone marrow [femur and sternum], injection sites, kidneys, liver, lungs, mandibular and mesenteric lymph nodes, spleen, and thymus) were collected for histopathologic examination. To determine tissue concentrations of ASO in liver, samples were collected from CD-1 and Tg.rasH2 mice administered ISIS 487660, ISIS 420476, ISIS 104838, or ISIS 421856.
Statistical analysis for in-life, terminal, and postmortem data
In study 1, the appropriate control group for each strain was compared to the respective treatment group for each strain. Raw data were tabulated, and the mean and standard deviation (SD) were calculated for each endpoint by sex and group. Body weight, food consumption, and organ weights (absolute and relative to body or brain weight) were analyzed by group pairwise comparison (Levene’s/analysis of variance [ANOVA]-Dunnett’s/Welch’s). Hematology (except leukocyte count) and serum chemistry were analyzed by group pairwise comparison (Levene’s/ANOVA-Dunnett’s/Welch’s), and leukocyte counts (total and differential) were analyzed by log transformation/group pairwise comparisons.
Determination of cytokine/chemokine levels
In study 2, a Mouse Cytokine/Chemokine Magnetic Bead Panel Milliplex Map Kit (BioPlex 200; BioRad, Hercules, CA) was used to determine the concentrations of 9 cytokines in serum: interleukin-1β, interleukin-6, interferon-γ, keratinocyte chemoattractant (KC), monocyte chemoattractant protein-1 (MCP-1), macrophage inflammatory protein-1α (MIP 1α), macrophage inflammatory protein-1β (MIP 1β), regulated on activation, normal T cell expressed and secreted (RANTES), and tumor necrosis factor-α. Samples were processed according to the manufacturer’s instructions. This involved washing and incubation steps prior to addition of premixed beads for magnetic separation steps and addition of detection antibodies and streptavidin-phycoerythrin for final quantification on a BioPlex 200 Instrument with BioPlex Manager, version 6.1 software (BioRad, Hercules, CA). No statistical analysis was conducted on these data. Individual concentrations for each analyte in serum samples were tabulated using Microsoft Excel and the mean and SD were calculated for each strain and dose group.
Determination of ASO concentrations in mouse liver
Liver samples from CD-1 (studies 1 and 2), CByB6F1 (study 1), or Tg.rasH2 (study 2) mice were analyzed for ASO concentration using an high performance liquid chromatography-UV (HPLC-UV) method as previously described (Yu et al. 2016). The quantification range of the assay was 3.6 to 1,445.3 µg/g for study 1 (ISIS 421856). For study 2, the ranges of the assay were 7.1 to 1,426.1 µg/g for ISIS 487660, 7.2 to 1,438.2 µg/g for ISIS 420476, 7.3 to 1,459.9 µg/g for ISIS 104838, and 7.2 to 1,455.3 µg/g for ISIS 421856. No statistical analysis was conducted on these data. Raw data were tabulated, and the mean and SD were calculated for each strain and dose group.
Results
Clinical Signs, Body Weight, and Food Consumption
There were no clinical findings in study 1 or study 2 that were considered related to the administration of ISIS 487660, ISIS 420476, ISIS 104838, or ISIS 421856. While several statistically significant changes in body weight were noted in one or both sexes across all 2′-MOE ASOs with the exception of ISIS 420476, the variability in these findings (and across studies) suggested this effect was not biologically meaningful for differences relative to saline controls for each strain of mice (Figures 1A and B). No changes in food consumption were noted for either Study 1 or Study 2 (data not shown).
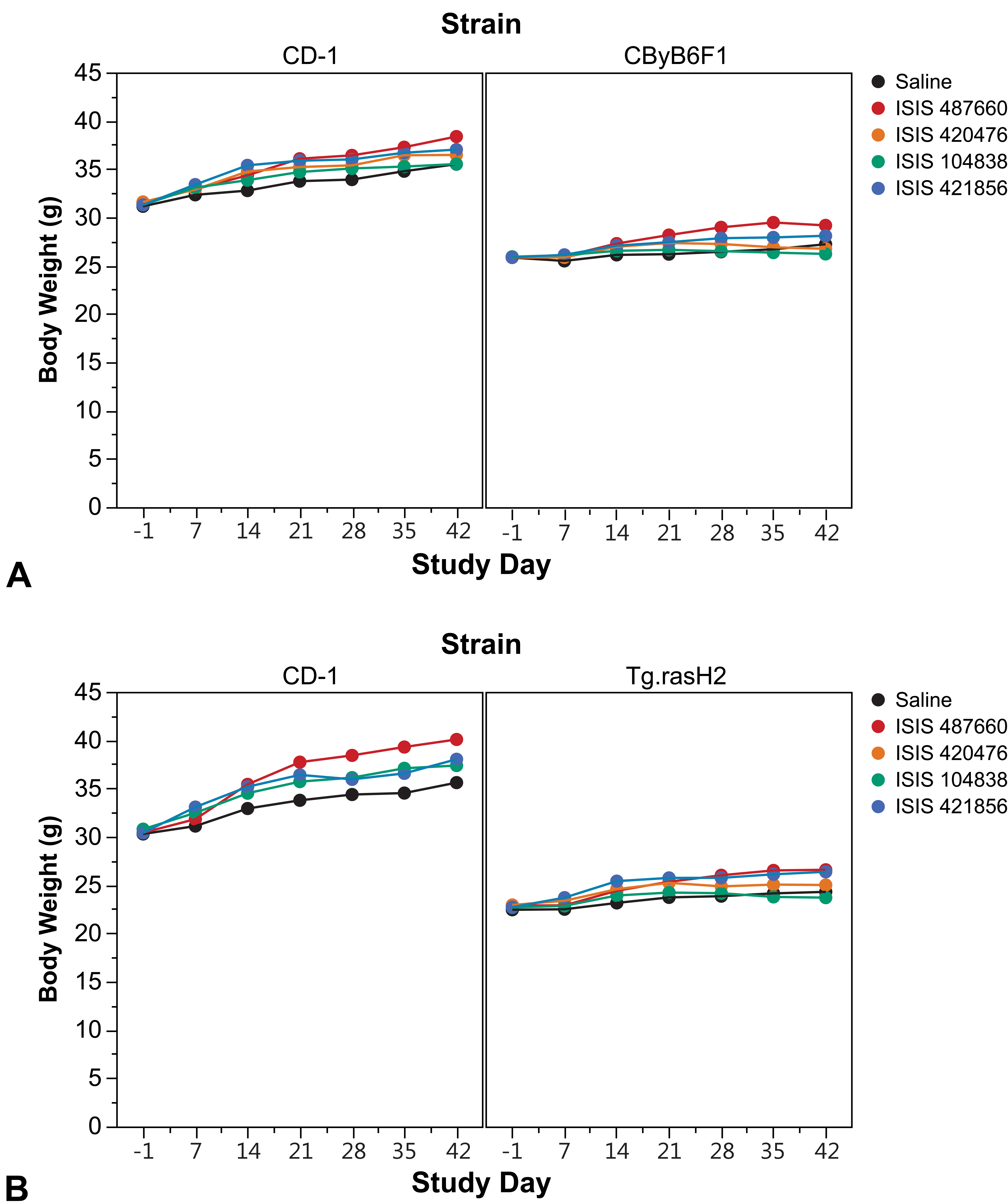
Mean body weight changes in CByB6F1 and CD-1 mice (study 1, A) and Tg.rasH2 and CD-1 mice (study 2, B).
Hematology/Serum Chemistry
Table 3 summarizes the percentage change, relative to saline controls for each strain, of various hematological parameters among the four groups treated with 2’-MOE ASOs in study 1. Red blood cell (RBC) count, hemoglobin (Hgb), and hematocrit were generally decreased, as were mean corpuscular volume and mean corpuscular Hgb. Increased red cell distribution width was likely reflective of the release of newly generated RBCs in response to decreased RBC count. The trend was similar among the mice strains that were dosed with the four 2′-MOE ASOs, although the magnitude of change differed slightly, as the least effect was observed with ISIS 487660 and the greatest effect observed with ISIS 421856. The trend and magnitude of these changes were similar between CByB6F1 and CD-1 mice for each of the four 2′-MOE ASOs-treated groups. Total white blood cell (WBC) counts, as well as the individual cell types, were increased in association with 2′-MOE ASO treatment. As noted for RBCs, the trend was similar among the four groups treated with 2′-MOE ASOs, but with greater variability, so there were no clear differences in magnitude. In consideration of strain, the magnitude of effect and the number of cell types affected was modestly greater in CD-1 mice as compared to CByB6F1 mice.
Hematology: Percentage Change among Treated Mice Compared to the Saline Control Group of the Same Strain on Day 44 in Study 1 (Combined Data from Male and Female Mice).
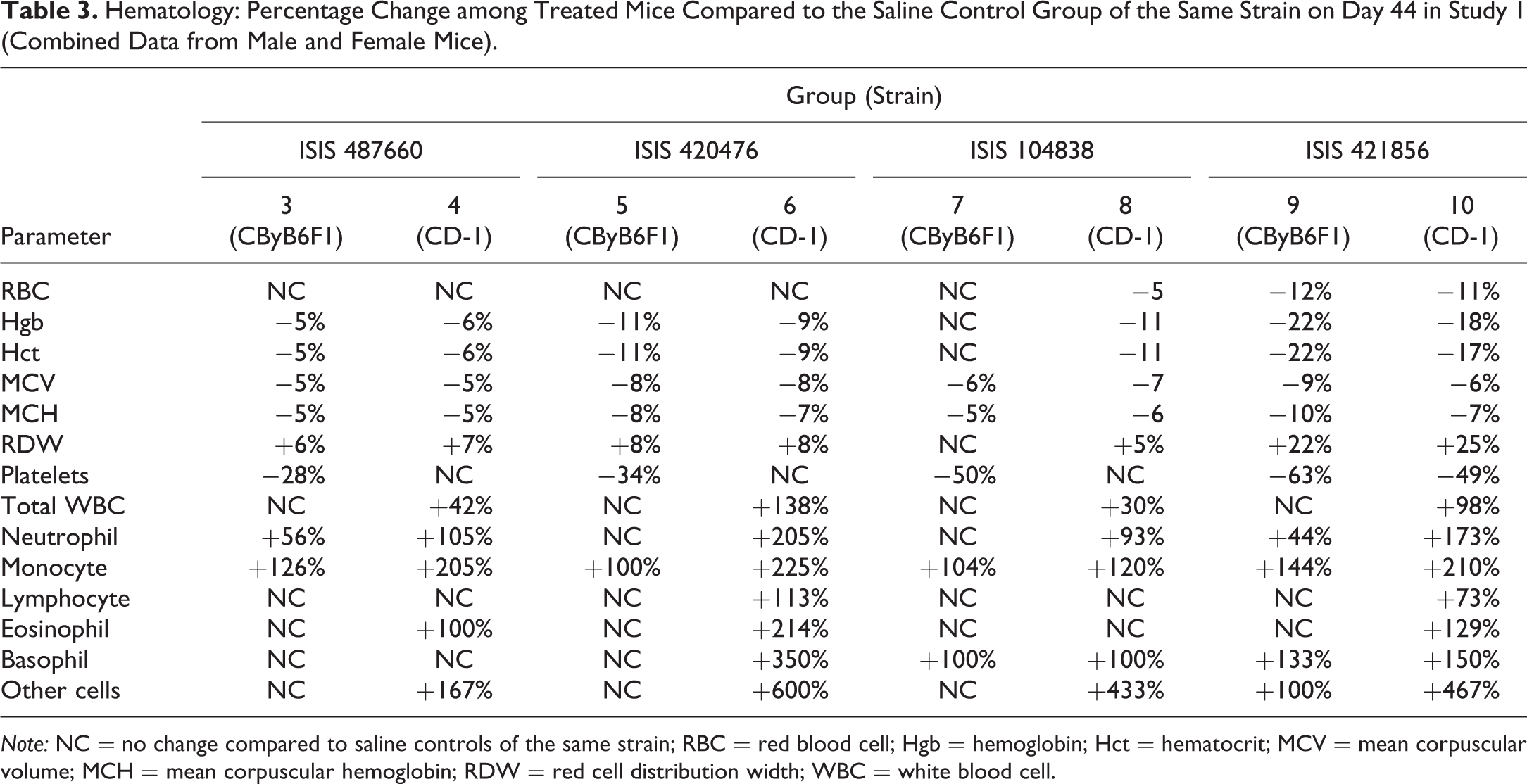
Note: NC = no change compared to saline controls of the same strain; RBC = red blood cell; Hgb = hemoglobin; Hct = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; RDW = red cell distribution width; WBC = white blood cell.
Serum chemistry parameters for study 1 are summarized in Table 4. Increases in aspartate transaminase, alanine transaminase, and alkaline phosphatase (ranging from 1.4- to 5-fold compared to control) and minimal decreases in albumin, total protein, cholesterol, and triglycerides were noted, with no clear differences in the magnitude of change between CByB6F1 and CD-1 mice or in either strain with ASO treatment. A modest increase (1.3-fold compared to control) in blood urea nitrogen was noted only for CD-1 mice administered ISIS 421856.
Serum Chemistry: Percent Change among Treated Mice Compared to the Saline Control Group of the Same Strain on Day 44 in Study 1 (Combined Data from Male and Female Mice).
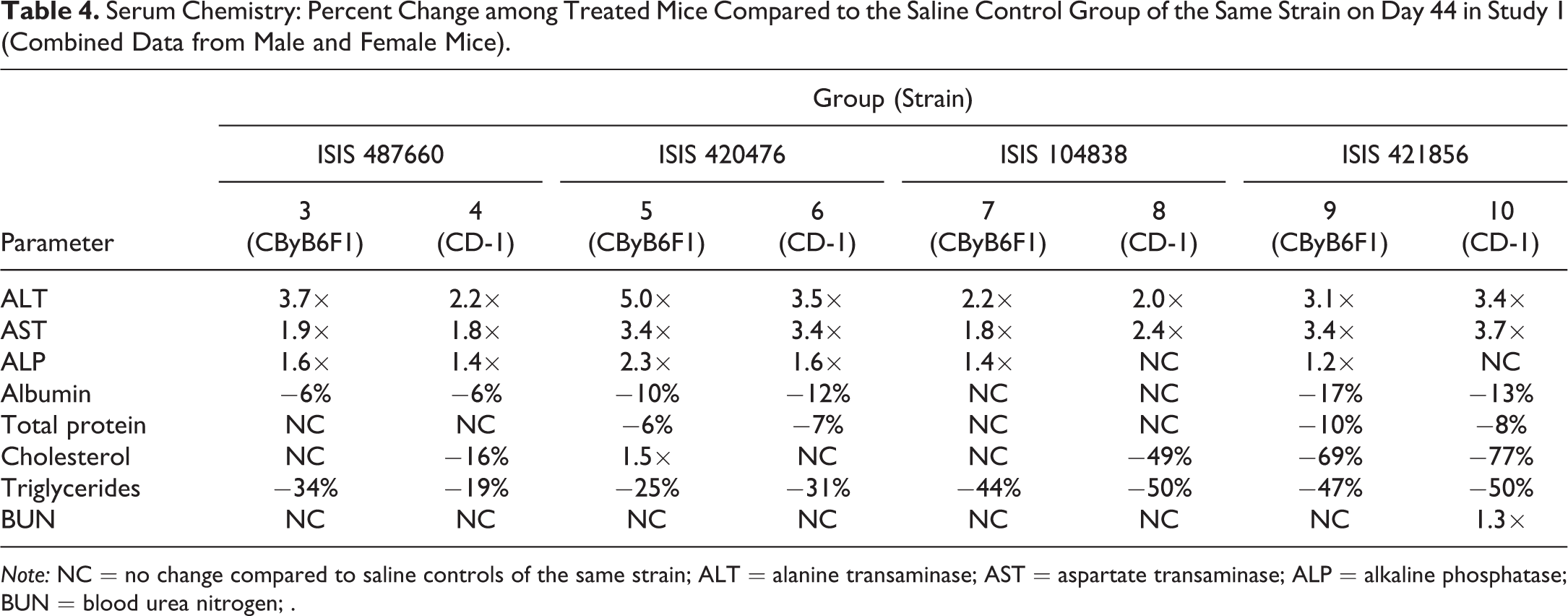
Note: NC = no change compared to saline controls of the same strain; ALT = alanine transaminase; AST = aspartate transaminase; ALP = alkaline phosphatase; BUN = blood urea nitrogen; .
Concentrations of ISIS 421856, ISIS 487660, ISIS 420476, or ISIS 104838 in Liver
Data (range and mean ± SD) for male and female mice within each strain and 2′-MOE ASO-treated group were combined for evaluation and are shown in Table 5. Concentrations of ISIS 421856 in liver from study 1 of 324 ± 117 µg/g and 632 ± 137 µg/g were noted for CD-1 and CByB6F1 mice, respectively. For study 2, mean ISIS 487660 concentrations of 583 ± 98 µg/g and 565 ± 254 µg/g in liver were noted for CD-1 and Tg.rasH2 mice, respectively. For ISIS 420476, mean liver concentrations of 594 ± 78 µg/g were noted for Tg.rasH2 mice. For ISIS 104838, mean concentrations of 632 ± 59 µg/g and 476 ± 96 µg/g in liver were noted for CD-1 and Tg.rasH2 mice, respectively. Mean ISIS 421856 concentrations of 858 ± 275 µg/g and 420 ± 77 µg/g in liver were noted for CD-1 and Tg.rasH2 mice, respectively. As demonstrated by the concentration range and the mean data displayed in Figure 2 for the three 2′-MOE ASOs and two strains from study 2, ASO exposures in tissues were slightly higher in CD-1 mice; however, these differences were not considered to be biologically meaningful (and were not statistically significant) and indicated similar tissue exposure of ASO between CD-1 mice and CByB6F1 and Tg.rasH2 mice.
Liver Concentrations of ISIS 487660, ISIS 420476, ISIS 104838, and ISIS 421856 in CByB6F1, Tg.rasH2, and CD-1 Mice (Studies 1 and 2).
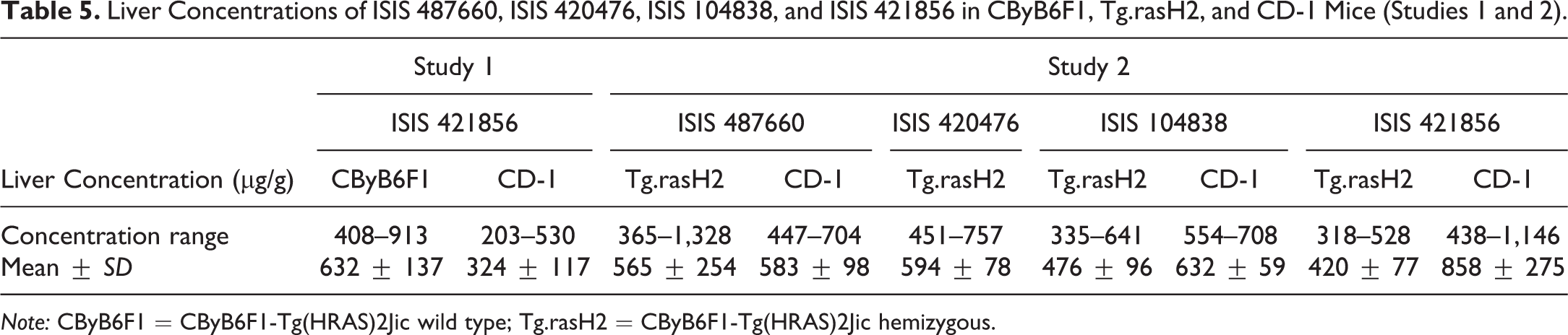
Note: CByB6F1 = CByB6F1-Tg(HRAS)2Jic wild type; Tg.rasH2 = CByB6F1-Tg(HRAS)2Jic hemizygous.
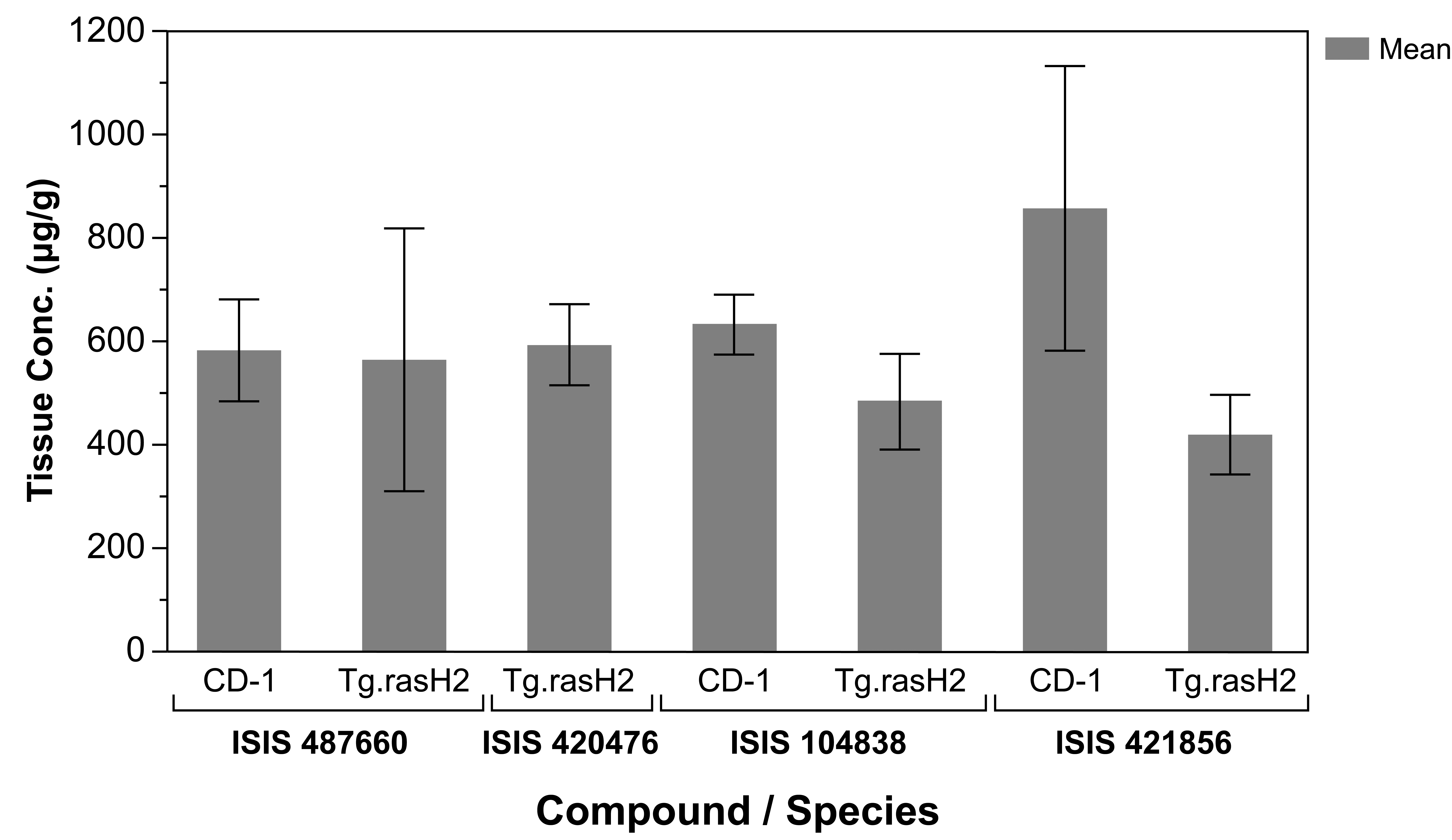
Concentrations of ISIS 487660, ISIS 420476, ISIS 104838, or ISIS 421856 in liver of Tg.rasH2 and CD-1 mice (study 2).
Cytokines/Chemokines
Group mean data (±SD) are shown in Table 6 (Study 2). Cytokine/chemokine results were similar between CD-1 and Tg.rasH2 saline control groups, indicating the baseline cytokine/chemokine levels were similar between the two strains. Each ASO-treated group was compared to the control group of the same strain and between strains. However, since ISIS 420476 was only administered to Tg.rasH2 mice, no species comparison was conducted. For ISIS 487660, as compared to Tg.rasH2-treated mice, CD-1-treated mice showed marginal increases in a majority of the analytes tested. CD-1 mice treated with ISIS 104838 showed a more moderate increase in analytes tested, specifically KC and MCP-1 levels, as compared to the ISIS 104838-treated Tg.rasH2 mice; however, the changes were comparable or within the variability of other ASO-treated groups in the same strain. The highest cytokine/chemokine levels were noted in Tg.rasH2 and CD-1 mice administered ISIS 421856, as expected, with more substantial increases in KC, MCP-1, MIP-1α, MIP-1β, and/or TNFα. It should be noted that, in general, these data were highly variable. Overall, the magnitude of changes in KC and MCP-1 levels in Tg.rasH2 and CD-1 mice, was most consistently increased among the ASO-treated groups, with other cytokine/chemokine changes observed more sporadically.
Group Mean ± SD Cytokine/Chemokine Results in Control Mice and Mice Administered ISIS 487660, ISIS 420476, ISIS 104838, or ISIS 421856 on Day 44 (Combined Data from Male and Female Mice; Study 2).
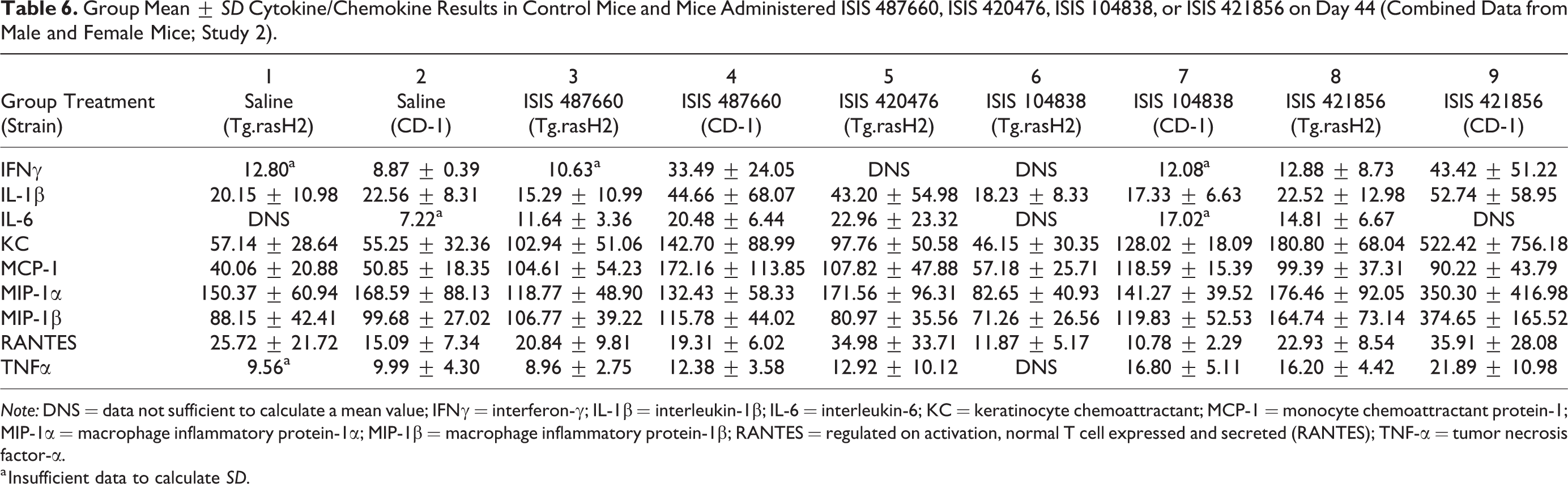
Note: DNS = data not sufficient to calculate a mean value; IFNγ = interferon-γ; IL-1β = interleukin-1β; IL-6 = interleukin-6; KC = keratinocyte chemoattractant; MCP-1 = monocyte chemoattractant protein-1; MIP-1α = macrophage inflammatory protein-1α; MIP-1β = macrophage inflammatory protein-1β; RANTES = regulated on activation, normal T cell expressed and secreted (RANTES); TNF-α = tumor necrosis factor-α.
a Insufficient data to calculate SD.
Organ Weights
Increased or decreased organ weights were noted in both studies, in each mouse strain, and among the four 2′-MOE ASOs evaluated. Liver, spleen, kidneys, thymus, heart, ovaries, seminal vesicles, and uterus were evaluated (see Supplemental Tables 7 and 8). There were ASO sequence-dependent differences in organ weight changes. These reflected the sequence-dependent variability in pro-inflammatory effects. When present, the magnitude of the pro-inflammatory effects on organ weight changes was largely the same between strains. Those sequences with relatively mild pro-inflammatory effects did not increase organ weight in CD-1 mice nor the CByB6F1 mice. Likewise, the more inflammatory sequence, ISIS 421856, produced similar organ weight increases in CD-1 and Tg.rasH2 mice.
Microscopic Examination
Histomorphologic alterations were present in the liver, kidneys, spleen, lymph nodes, bone marrow, thymus, lungs, and injection site(s). Occasional differences were noted between CD-1 and CByB6F1 mice in study 1 and between CD-1 and Tg.rasH2 mice in study 2; however, these involved either the incidence (number of mice in each group) or severity (ranging from minimal to severe) of individual observations but not the spectrum of observations. Furthermore, the observations for CD-1 mice in each of the studies were similar in organs/tissues involved.
Liver
Findings in the liver included vacuolated/granular macrophages, increased mononuclear cell infiltrates, increased hepatocellular mitotic figures, individual hepatocellular necrosis and hepatocellular cytoplasmic alteration and intranuclear inclusions (see Supplemental Tables 9 and 10). Macrophages (Kupffer cells) with abundant vacuolated cytoplasm with variable accumulation of basophilic granules consistent with ASO uptake, were noted, as were foci of extramedullary hematopoiesis. The severity of these observations typically ranged from minimal to mild within each of the groups and mouse strains evaluated, although findings were occasionally moderate to severe. In consideration of mouse strain, there were no apparent differences between CD-1 and CByB6F1 mice (study 1) or Tg.rasH2 mice (study 2). Trends were noted among the four 2′-MOE ASOs with ISIS 487660, ISIS 420476, and ISIS 104838 showing a similar pattern and severity of effect and ISIS 421856 generally exhibiting the greatest incidence and severity of proinflammatory effects, regardless of mouse strain.
Kidneys
2′-MOE ASO-related observations of basophilic granules, cytoplasmic vacuolation, and intranuclear inclusions within proximal tubular epithelial cells, interstitial mononuclear cell infiltration, and vacuolated/granular macrophages were noted (see Supplemental Tables 11 and 12). For ISIS 487660, these changes were considered to be of minimal magnitude, whereas the severity for ISIS 420476, ISIS 104838, and ISIS 421856 ranged from minimal to moderate (with an occasional severe score). There was no clear difference in the observations for kidney among the CD-1 and CByB6F1 or Tg.rasH2 mice.
Spleen/lymph nodes/bone marrow
Findings within the hematopoietic/lymphoid organs (see Supplemental Tables 13 and 14) were consistent with a pro-inflammatory effect for each of the 2′-MOE ASOs and increased hematopoiesis. In the spleen, this involved increased hematopoietic cellularity in the red pulp, generalized lymphoid hyperplasia, and increased numbers of vacuolated/granular macrophages. Examination of lymph nodes revealed a generalized lymphoid hyperplasia, vacuolated/granular macrophages, and extramedullary hematopoiesis. Bone marrow changes included increased myeloid/erythroid ratio and vacuolated/granular macrophages. Overall, the changes ranged from minimal to severe, with no apparent differences between CD-1 and CByB6F1 or Tg.rasH2 mice. Consistent with the alterations in hematology/serum chemistry, cytokine/chemokine levels, and histologic/morphologic changes in the liver, the severity of effects in the hematopoietic/lymphoid organs, was greatest for ISIS 421856.
Injection site/other tissues
Findings at the injection sites (data not shown) included the presence of vacuolated/granular macrophages and mononuclear cell infiltration consistent with uptake of ASO and cytokine activation within the dermis and subcutis. The accumulation of vacuolated/granular macrophages was generally scored as minimal to mild. The mononuclear cell infiltrate was similar to that noted for other organs/tissues and was composed primarily of macrophages and a lesser number of lymphocytes. Lymphoid depletion in the cortex of the thymus was considered to be a secondary response to stress; however, observations of minimal vacuolated/granular macrophages for all four 2′-MOE ASOs in studies 1 and 2 were considered to be associated with 2′-MOE ASO administration and exposure and were noted in all strains evaluated. Additional affected tissue/organs (data not shown) included lungs, pancreas, urinary bladder, ovaries/oviducts, uterus with cervix, and vagina, and increased mononuclear cell infiltration and vacuolated/granular macrophages were variably observed in these organs at a low magnitude. Most importantly, there were generally no significant differences between CD-1 and CByB6F1 mice (study 1) or CD-1 and Tg.rasH2 mice (study 2).
Discussion
Rodent carcinogenicity studies are typically the final nonclinical studies to be completed for a drug candidate prior to submission for regulatory approval and commercialization; however, establishing key study parameters such as dose levels, route of administration, and dose schedules are dependent on data collected from short- and long-term repeated dose studies completed earlier in a development program. In a traditional rodent carcinogenicity study, doses are typically selected from data generated during chronic administration studies, and of even greater importance, the dose levels and biologic response in the selected mouse (or rat) strain have been considered for relevance to human exposure. When considering a transgenic mouse model for carcinogenicity evaluation, initiation of study requires confidence in not only similarity in the overall biologic response but also in dose response, such that the dose range can be selected based on relevant available data. The dose of 100 mg/kg was selected based on data generated from previously conducted studies where this dose adequately differentiated the marked pro-inflammatory response in CD-1 mice administered ISIS 421856 and the minimal to mild inflammatory response in mice administered ISIS 104838, ISIS 420476, or ISIS 487660. A common approach for Tg.rasH2 mouse has been to run 5- and/or 28-day pilot studies in Tg.rasH2, CByB6F1, or a combination, for example, Tg.rasH2 for toxicology evaluation and CByB6F1 for pharmacokinetic evaluation. However, this paradigm adds time and cost to the development process, as well as an increased use of animals and experimental drug material.
The toxicity profile of 2′-MOE ASOs in mice has been extensively reviewed (Monteith et al. 1999; Henry et al. 2001; Geary et al. 2007; Henry et al. 2008; Zanardi et al. 2012; Crooke et al. 2016; Shemesh et al. 2016). Among the typical findings, the pro-inflammatory response is characterized by elevated cytokine/chemokines, multiorgan mononuclear cell infiltrates, and activation of tissue macrophages (histiocytes). These effects have been described to vary in intensity among ASO sequences and chemical classes. The spectrum of clinical-pathologic observations in the CD-1 mouse in the present studies is consistent with the description above and with findings expected for 2′-MOE ASOs. The magnitude of the pro-inflammatory effects and/or secondary effects is sequence dependent and, as noted, it is not uncommon to have differences among individual ASO sequences or between mouse strains. As described herein, the goal was to determine whether findings observed in response to ASO administration in CD-1 mice could be considered qualitatively and quantitatively comparable to those observed in Tg.rasH2 ASO-treated mice. To that end, four distinct 2′-MOE ASOs were selected to provide an appropriate representation of the range of findings associated with 2′-MOE ASO exposure, pro-inflammatory aspects, and histopathology findings.
Clinical signs, body weight, and food consumption were generally comparable between CD-1 and Tg.rasH2 mice and among mice treated with four 2′-MOE ASOs. Body weight is a determinant for establishing the MTD (maximum tolerated dose) in pilot/dose ranging studies with CByB6F1 mice and for selecting dose levels for a 6-month carcinogenicity study in Tg.rasH2 mice. Modest variability in weight gain or weight loss was noted for CD-1, CByB6F1, and Tg.rasH2 mice and among the groups treated with the four 2′-MOE ASOs; however, overall body weight and weight gain over time remained within the expected ranges and was supportive of the health status of the CByB6F1 and Tg.rasH2 mice and similar to CD-1 mice.
Alterations in serum chemistry parameters indicated little to no difference in the spectrum or magnitude of effects for individual analytes between CByB6F1 and CD-1 mice. Platelet and RBC counts, and changes thereof, were similar, as were the RBC indices. ASO-related changes in the WBC fractions appeared to be slightly greater in magnitude for CD-1 mice as compared to CByB6F1 mice; however, considering normal variability, the difference was not likely to be biologically meaningful. In consideration of cytokines/chemokines in Tg.rasH2 and CD-1 mice, elevated levels of KC and MCP-1 levels were the most consistent among the ASO-treated groups, with other cytokine/chemokines observed as more sporadic. However, the overall cytokine/chemokine response was not appreciably different between the two strains of mice. When considered as a whole, the systemic-based endpoints were indicative of a consistent response to the various 2′-MOE ASOs between CD-1 and Tg.rasH2 mice. Organ weight changes and histomorphology/histopathology observations of basophilic granules in macrophages in multiple tissues and mononuclear cell infiltrates are expected 2′-MOE ASO class effects, with no apparent differences between CD-1 and CByB6F1 or Tg.rasH2 mice. Indications of pro-inflammatory effects, for example, multiorgan cellular infiltrates and vacuolation of macrophages, were also as expected and similar among the two strains.
As the liver is a primary target organ for ASO distribution and toxicity, ASO exposure and organ histopathology were assessed. While variable between CByB6F1 and CD-1 mice, as well as within individual mice of each strain, concentrations of ASO in the liver for each strain indicated comparable distribution to the liver. Observations of vacuolated/granular macrophages, mononuclear cell infiltrates, increased hepatocellular mitotic figures, and individual hepatocellular necrosis were consistent with the accumulation of ASO within macrophages, hepatocytes, and other cells within the liver.
Ultimately, the goal of a carcinogenicity study is to provide data for carcinogenic hazard identification and human risk assessment. Based on the data from CD-1 and CByB6F1 mice indicating a similarity in the toxicologic response between the two strains, evaluation of carcinogenic potential in Tg.rasH2 mice may allow for evaluation of generic pathways for tumor formation and the evaluation of compound-specific questions. For the general assessment, the Tg.rasH2 mouse model is reported to have a low incidence of spontaneous tumors, a low false-positive or false-negative incidence, be relevant for a genotoxic or nongenotoxic mechanism of action, and be accepted by regulatory agencies (e.g., The U.S. Food And Drug Administration, the Japanese Ministry of Health, Labour and Welfare, and the Committee for Medicinal Products for Human Use).
The ability to evaluate potential carcinogenic hazards in a model that has greater sensitivity, and of even greater importance, increased relevance to humans, is of considerable interest. In consideration of ASO evaluation, one of the potential class effects that could potentially drive increased tumorigenicity is the pro-inflammatory effects associated to varying degrees with some chemistries and sequences (Greaves 2012). Ras and the Ras super family have been implicated in tumorigenesis involving inflammation and inflammatory conditions; as such, activation of cellular pathways via cytokine, chemokine, and growth factor receptors, as well as activation of the toll-like Receptor (TLR) system for immune surveillance, can lead to further enhancement by Ras/Ras super family to impart carcinogenic effects (Cao et al. 2004; Coste et al. 2010; Dalpke and Helm 2012; Smithgall 1998; Gu et al. 2000; So and Ouchi 2010). A common action of TLR family members is recognition of pathogen-associated microbial patterns within bacterial DNA or viral RNA, which can also be present within endogenously administered nucleic acids, for example, DNA or single/double stranded RNA (Judge and MacLachlan 2008; Roberts et al. 2005). Immune stimulation associated with ASO administration via TLR activation has been reported in the mouse, and thus, Ras and the Ras super family could be involved in this response. A combination of a general ability to evaluate carcinogenic potential, and the possibility of addressing ASO-associated actions, renders the Tg.rasH2 mice as having greater relevance as a replacement for the traditional 2-year carcinogenicity study.
Selection of a transgenic model for carcinogenicity evaluation has clear advantages but also requires confidence that the transition to a different mouse strain will provide the necessary and appropriate data needed for carcinogenicity evaluation and to characterize potential human hazards. Based on the results from four distinct 2′-MOE ASOs, the overall class effects (pharmacological and toxicological) were similar between the CD-1 mouse and the CByB6F1 or Tg.rasH2 mice. This indicates the presence of the human c-Ha-ras gene does not influence, negatively or positively, the biologic response to an ASO, and therefore, the Tg.rasH2 mouse model is relevant for carcinogenicity assessment of ASOs.
It is recognized that each drug candidate must be assessed on its specific characteristics, that is, target, pharmacology, toxicology, and drug class. Nevertheless, these results indicated that the dose levels selected from general repeated-dose assessments of 2′-MOE ASOs in CD-1 mice are directly relevant for dose selection for a 6-month carcinogenicity study in Tg.rasH2 mice, as well as the potential to initiate a 6-month carcinogenicity study with limited pilot studies or, in specific cases, without prior pilot studies.
Supplemental Material
Supplemental Material, DS7_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS7_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS8_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS8_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS9_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS9_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS10_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS10_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS11_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS11_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS12_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS12_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS13_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS13_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS14_TPX_10.11770192623318813143 - Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice
Supplemental Material, DS14_TPX_10.11770192623318813143 for Comparison of the Class Effects of Antisense Oligonucleotides in CByB6F1-Tg(HRAS)2Jic and CD-1 Mice by Tae-Won Kim, Chris N. Papagiannis, Laura S. Zwick, Jeffery A. Engelhardt, Christine M. Hoffmaster, Noah M. Post, John E. Matson, Jill A. Hsiao, Sebastien A. Burel, and Scott P. Henry in Toxicologic Pathology
Footnotes
Author Contributions
Authors (TK, CP, LZ, JE, CH, NP, JM, JH, SB, SH) contributed to conception or design; data acquisition, analysis, or interpretation (TK, CP, LZ, JE, CH, NP, JM, JH, SB, SH); drafting the manuscript (TK, CH); and critically revising the manuscript (TK, CP, LZ, JE, CH, NP, JM, JH, SB, SH). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
