Abstract
A factor limiting widespread use of the transgenic rasH2 mouse model for carcinogenicity testing of pharmaceuticals is the paucity of published data on actual drug candidates in rasH2 mice. This report addresses this gap by highlighting rasH2 mouse study data for 10 pharmaceutical candidates. These results were compared with findings in the 2-year studies in Sprague-Dawley rats for the same 10 compounds. In the 6-month rasH2 studies, only 2 of the 10 compounds tested positive for carcinogenicity and these correlated with positive findings in the companion 2-year rat studies. One compound, sunitinib, produced gastroduodenal carcinoma in both sexes and increased hemangiosarcoma in spleen and uterus in female rasH2 mice; in rats it produced gastroduodenal carcinoma and increased pheochromocytoma (males only). The second compound, bazedoxifene, produced ovarian granulosa cell neoplasms in rasH2 mice and rats, and renal tubular neoplasms associated with increased chronic progressive nephropathy only in rats. The higher percentage of carcinogenicity positive rat bioassays could be attributed to rat-specific phenomena with little or low relevance to man. Thus, this article confirms previous reports that rasH2 mice develop rodent-specific neoplasms less frequently than rats and positive findings, when present, are accompanied by similar positive results in the rat.
Introduction
The value of 2-year rodent bioassays in carcinogenicity risk assessment has been questioned for 2 decades (Alden et al. 2011; Friedrich and Olejniczak 2011; Sistare et al. 2011). Research on alternative models of carcinogenicity risk assessment has yielded valuable short- or medium-term mouse models such as the p53+/− mouse and rasH2 mouse. These models, having gained scientific and regulatory acceptance, are slowly gaining industry acceptance as potential replacements for the standard lifetime carcinogenicity mouse bioassay (Anonymous 1998; Boverhof et al. 2011; Friedrich and Olejniczak 2011). Advantages of these alternative models include comparable prediction of human carcinogens compared to lifetime exposure rodent bioassays, fewer irrelevant positive results, flexible resourcing, reduced animals use, shorter testing duration, and reduced costs (Alden, Smith, and Morton 2002).
Among the alternate shorter term mouse models, the rasH2 model is now more commonly used because it is the only alternative model readily accepted for testing of both genotoxic and nongenotoxic chemicals administered by the oral or parenteral routes (Long et al. 2010; MacDonald et al. 2004). The transgenic rasH2 mouse model was initially developed in Japan to evaluate the role of ras over expression in tumorigenesis. The rasH2 mouse, officially known as CByB6F1-Tg(HRAS)2Jic, is a hemizygous transgenic mouse carrying the human c-Ha-ras gene with its own promoter/enhancer elements and harboring a single point mutation in the 3′-end (in the last intron) which induces high enhancer activity (Morton et al. 2002; Suemizu et al. 2002; Tamaoki 2001). This prototypic human transgene, which encodes for the p21ras protein, is inserted in tandem (3–6 copies) in the murine chromosome 15E3 with approximately 2- to 3-fold the p21ras protein levels found in wild type mice. The hemizygous transgenic mice are produced by breeding transgenic male C57BL/6J mice with wild type BALB/CByJ female mice.
The issue of insufficient historical control data in rasH2 mice, a theme that may have limited widespread use of this model in the pharmaceutical industry, has been addressed recently (Nambiar, Turnquist, and Morton 2012). This retrospective analysis demonstrated little variation in the spontaneous tumor incidences in control rasH2 mice in 6-month studies. Moreover, the spontaneous tumor incidences reported in these studies conducted over the last 10 years were similar to reports published over a decade ago (Nambiar, Turnquist, and Morton 2012).
Despite these advances in the past decade, an important gap has been the limited data in the public domain for pharmaceutical-mediated cancer outcomes in this model. The ILSI HESI ACT (Alternatives to Carcinogenicity Testing Committee of the Health and Environment Sciences Institute within the International Life Sciences Institute) consortium, during the validation of the rasH2 model, published extensively on the effects of a limited set of xenobiotics (confirmed human carcinogens, rodent-specific carcinogens, and rodent noncarcinogens) in rasH2 mice [see reviews and individual reports in Toxicol. Pathol. 29 (1 suppl), 2001]. In the present report, treatment-related neoplastic and associated findings from 6-month rasH2 mouse studies (n = 10 pharmaceutical candidates) are compared to similar findings from companion chronic studies conducted in Sprague-Dawley (SD) rats and nonrodents.
Materials and Methods
Toxicology Report Mining
Pfizer compounds (n = 10) for which 6-month carcinogenicity studies were conducted in rasH2 mice were selected. For each compound, toxicology reports from the 1-month dose range finding studies and 6-month rasH2 mouse studies were compiled along with chronic rat and large animal studies. In this article, only neoplasms and findings that were considered histologic risk factors for tumors (hyperplasia, cellular hypertrophy, and/or atypical cellular foci) are reported (Table 1). Furthermore, for each Pfizer drug, toxicology reports from chronic repeat dose companion SD rat (6 month and 2 year) and nonrodent (dog or nonhuman primate [NHP]) studies were reviewed with emphasis on treatment-related neoplastic and proliferative findings (Table 1). All the original interpretations from the toxicology reports were maintained without any changes, reanalyses, or reinterpretation. Notably, all 10 Pfizer drugs were nongenotoxic via both in vitro and in vivo studies (unpublished results).
Repeat dose toxicology studies for Pfizer compounds that were mined for test article–related neoplastic and proliferative findings.
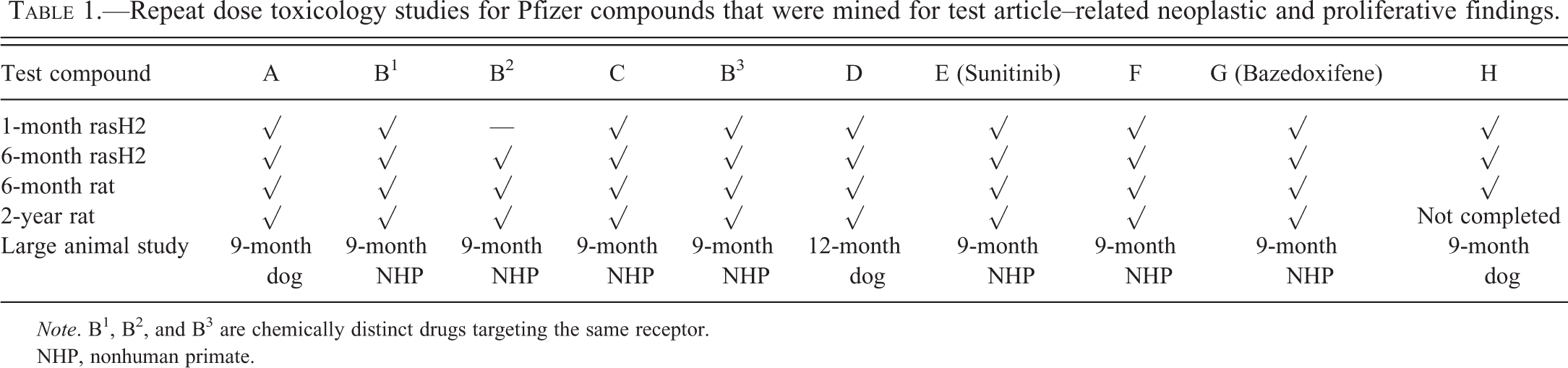
Note. B1, B2, and B3 are chemically distinct drugs targeting the same receptor.
NHP, nonhuman primate.
General rasH2 Mouse Study Design for 6-month Carcinogenicity Studies
Data reported in this article include test article–related findings (neoplastic and related non-neoplastic findings) from 10 carcinogenicity studies of 6-month dosing duration conducted between March 2004 and July 2009 under good laboratory practice (GLP) conditions (Table 1). These were sponsored or conducted by Pfizer. All studies and animal procedures were conducted in accordance with animal use protocols approved by the animal care and use committee in compliance with The Guide for Care and Use of Laboratory Animals and all applicable regulations. CByB6F1-Tg(HRAS)2Jic (Taconic model 001178) male and female mice were purchased from Taconic (Germantown, NY), at 6 weeks of age and housed individually in suspended polycarbonate or stainless steel wire cages in environmentally controlled rooms (temperature 18–26°C, relative humidity 30–70%, and controlled 12-hr light cycle) and provided ad libitum access to water and a certified rodent diet. Each mouse was genotyped before leaving the vendor’s facility. The mice were typically allowed to acclimate for 2 weeks before study start, and weighed between 17.6 and 25.4 g at study initiation. The total number of study animals in each study was 25/group/sex. The number of dose groups for most studies was three along with a concurrent vehicle control group. The alkylating carcinogen, N-methyl-N-nitrosourea (MNU), was used as a positive control in 8 of the 10 studies. A single dose of MNU pH 4.0 in citrate buffer was given as a positive control agent at 75 mg/kg intraperitoneal (IP; n = 7 studies) or at 58 mg/kg IP (n = 1 study). For 2 studies (compounds G and H), urethane was used as a positive control at 1,000 mg/kg (3 IP injections on days 1, 3, and 5 of the first week). Test article was administered either in a certified rodent diet (n = 4 diet admix studies) or by oral gavage (n = 6 studies). The vehicles in most gavage studies were 0.5% methylcellulose ± Tween or sterile water. An exception was compound H which had 7.5% vitamin E d-alpha tocopheryl polyethylene glycol 1000 succinate in water as vehicle.
Companion Chronic Studies
Test article–related neoplastic and related non-neoplastic findings (defined by Sistare et al. 2011 as histologic risk factors for tumors including hyperplasia, cellular hypertrophy, and/or atypical cellular foci) for each of the 10 compounds were obtained from companion chronic or dose range finder studies (Sistare et al. 2011). These include 1-month rasH2 mouse, 6-month rat, and dog/NHP chronic studies. These studies were conducted under GLP conditions and were of standard design and study design details are not discussed.
Non-Pfizer 6-month rasH2 Mouse Studies
In addition to the aforementioned Pfizer compounds (Table 1), test article–related tumor data from five additional approved compounds (aliskiren, olmesartan, telbivudine, indacaterol, and palifermin) were obtained from the respective drug labels and/or the literature (Aliskiren-label; Friedrich and Olejniczak 2011; Indacaterol-label; Olmesartan-label; Palifermin-label).
Results
Test Article–related Effects on Survival and Body Weight Gain in 6-month rasH2 Mouse Studies
Test article–related increase in mortality in rasH2 mice was observed in 2 of the 10 compounds that were tested (unpublished results). The increased mortality in these 2 compounds, compounds B3 and sunitinib, was attributed to test article–related non-neoplastic findings (inflammation of the urinary tract) and neoplastic findings (gastroduodenal carcinoma and hemangiosarcoma), respectively. In few studies (sunitinib and compound D), there was treatment-related evidence of moribundity (hunched posture, reduced activity, rough hair coat, etc.) and reduced body weight (generally between 5 and 10% decrease from control mean).
Test Article–related Neoplastic and Proliferative Findings: 6-month rasH2 Mouse Studies
Neoplastic Findings
Test article–related tumors were observed in 2 of the 10 compounds [compounds E (sunitinib) and G (bazedoxifene)] that were tested in rasH2 mice (Figure 1 and Tables 2 and 3). For sunitinib, there were test article–related gastroduodenal carcinoma (often involving Brunner’s gland) and an increase in incidence of hemangiosarcomas in females only (≥7× human area under the curve [AUC])). For bazedoxifene, there were test article–related benign ovarian granulosa cell tumors at 19× human AUC, while the no observed effect level was 10× human AUC.
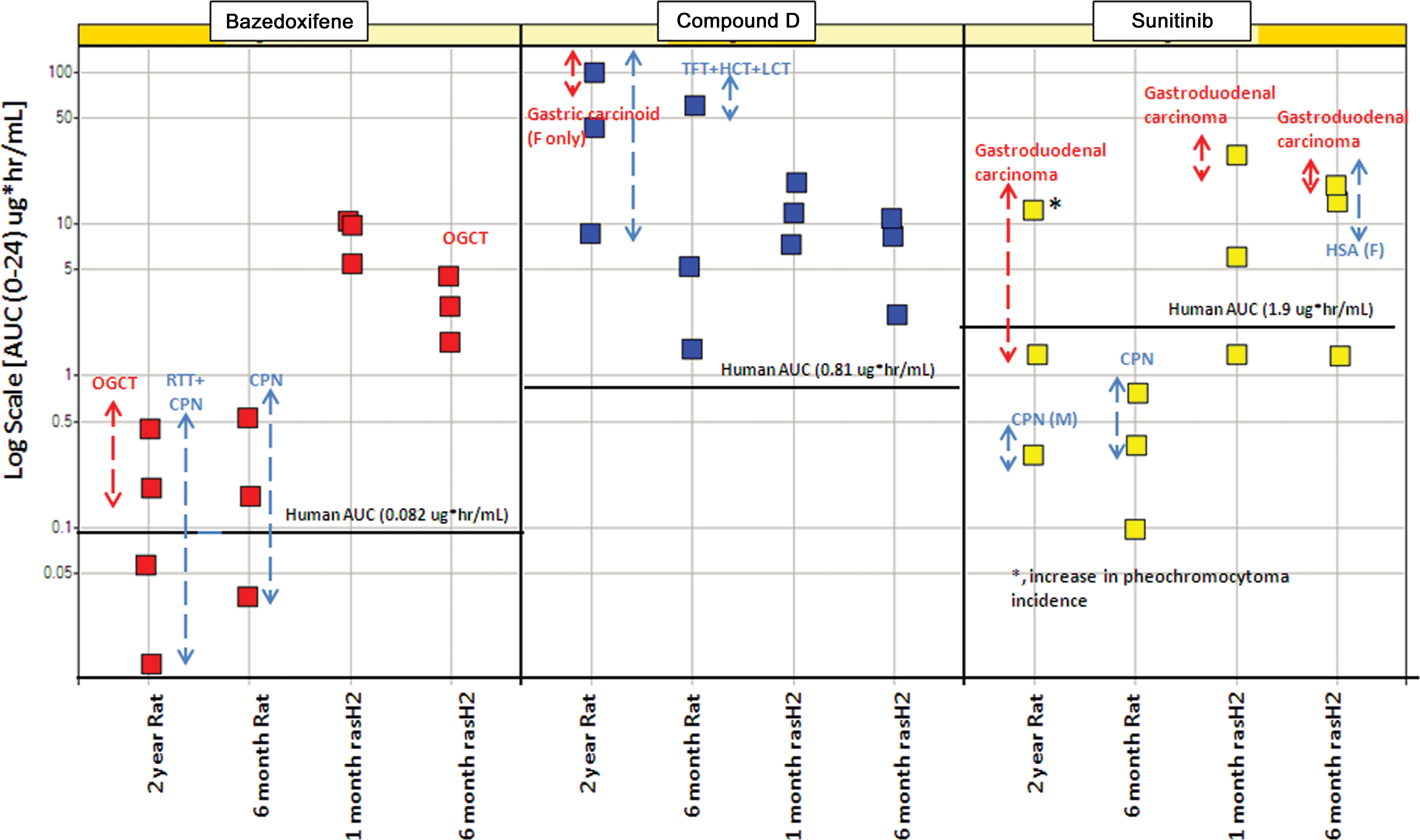
Interstudy comparison of tumor and tumor-related findings with bazedoxifene (compound G), compound B, and sunitinib (compound E) in chronic mouse and rat studies. Each square represents a dose exposure in a toxicology study. The color-coded vertical lines highlight the dose exposures that correspond to a specific finding. The solid horizontal line depicts human AUC (0–24) at a clinical dose for the drug (OGCT = ovarian granulosa cell tumors, RTT = renal tubular tumors, CPN = chronic progressive nephropathy, TFT = thyroid follicular tumors, HCT = hepatocellular tumors, LCT = Leydig cell tumor, HSA = hemangiosarcoma, AUC = area under the curve).
Comparison of data from different species for 10 compounds evaluated in the rasH2 mouse carcinogenicity model.
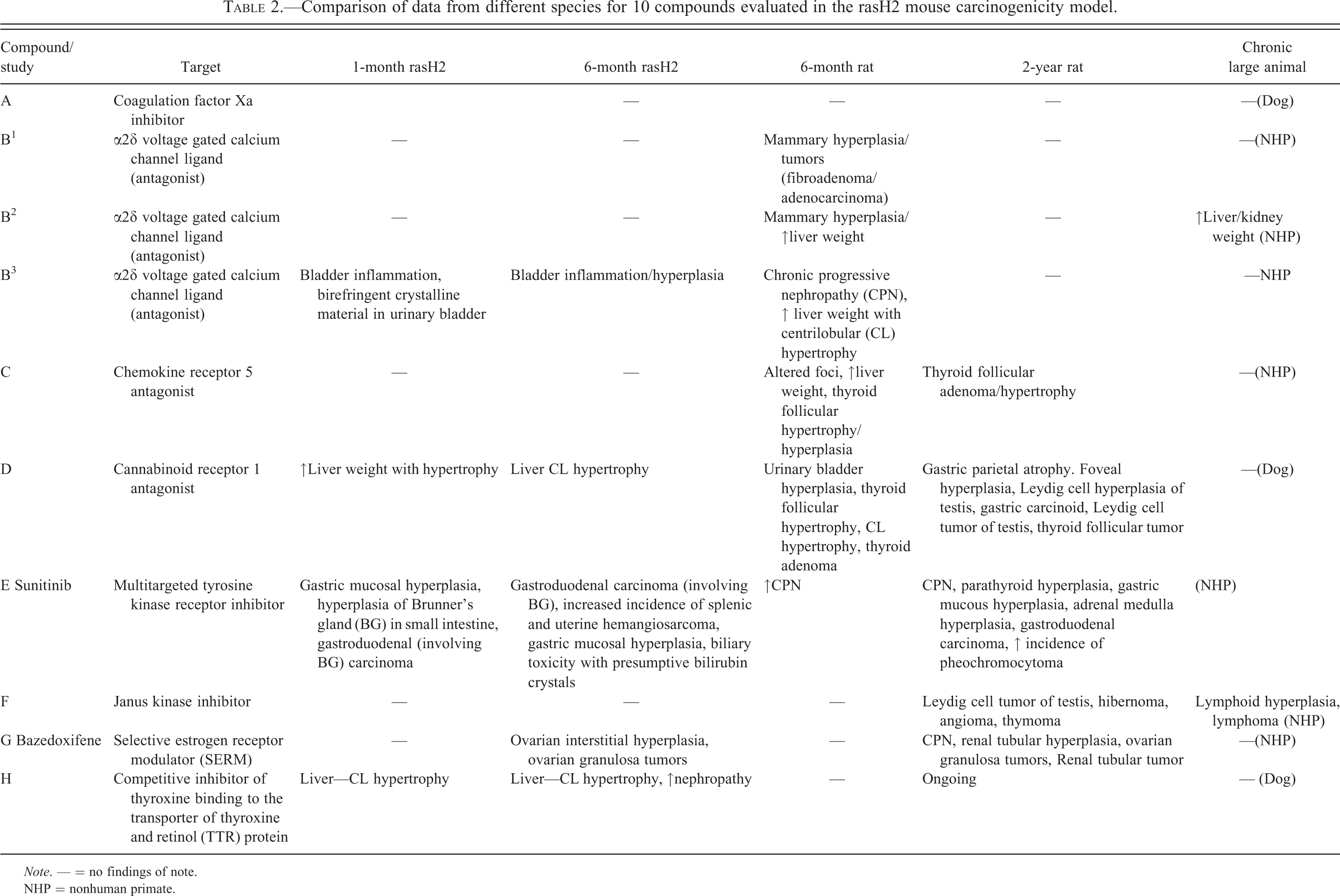
Note. — = no findings of note.
NHP = nonhuman primate.
Incidence of key test article–related findings in the “positive” 6-month rasH2 studies.
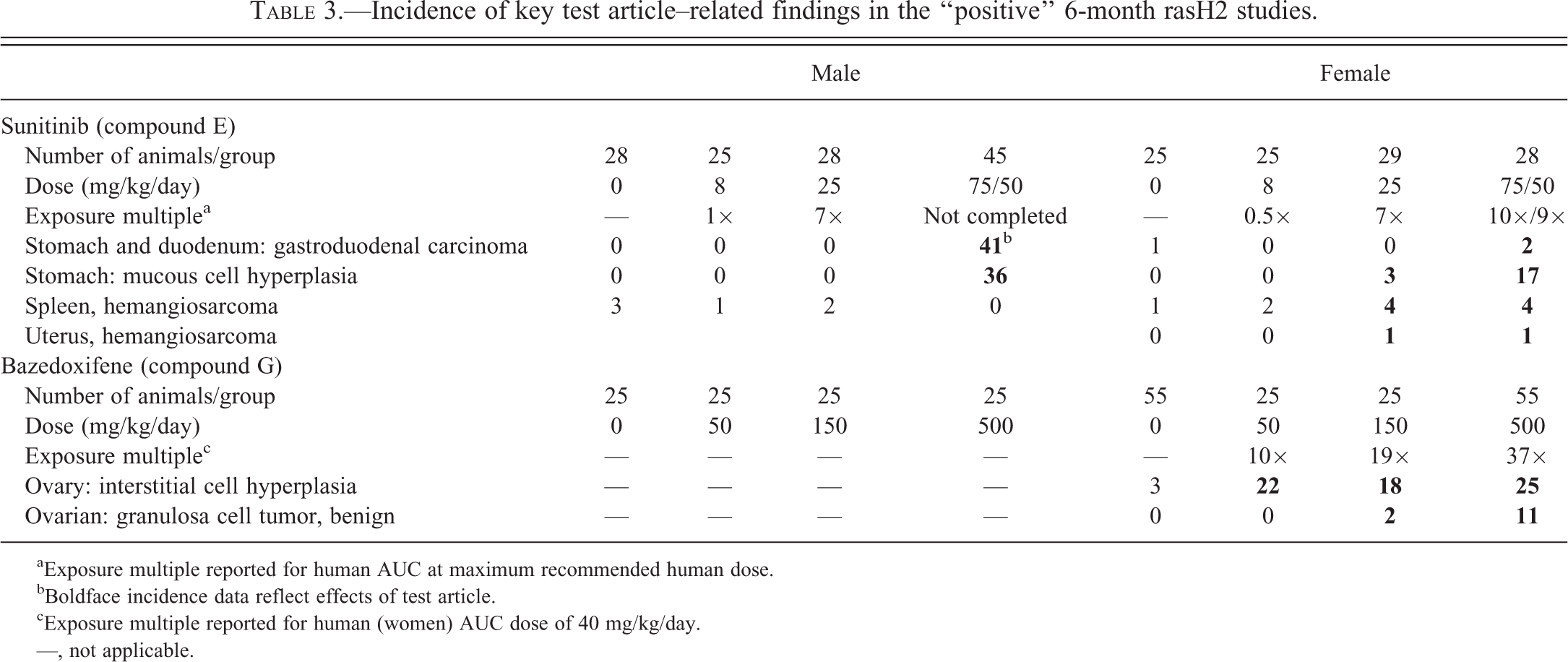
aExposure multiple reported for human AUC at maximum recommended human dose.
bBoldface incidence data reflect effects of test article.
cExposure multiple reported for human (women) AUC dose of 40 mg/kg/day.
—, not applicable.
Proliferative Findings
Test article–related proliferative findings likely associated with the aforementioned tumors were observed with sunitinib and bazedoxifene (Table 2). For sunitinib, gastric foveolar mucosal hyperplasia was observed in both the 6-month (≥7× total human AUC) and the shorter 1-month rasH2 mouse studies (at higher exposure). In addition, hyperplasia of the Brunner’s glands was noted in the 1-month rasH2 mouse study (at higher exposure). For bazedoxifene (a selective estrogen receptor modulator [SERM]), ovarian interstitial hyperplasia was observed in the 6-month rasH2 mouse study at all tested doses (10–37× human AUC). However, the relationship of ovarian interstitial hyperplasia to the observed ovarian granulosa cell tumor is unclear. There were no compound G-related proliferative findings in the shorter 1-month rasH2 mouse study.
Test Article–related Proliferative and Related Findings (without Neoplasia): 6-month rasH2 Mouse Studies
Apart from the proliferative findings described above for sunitinib and bazedoxifene, there were three compounds (compounds B3, D, and H) that had test article–related proliferative findings in the 6-month rasH2 mouse studies without evidence of neoplastic treatment effects (Table 2).
Compound B3 was associated with urinary bladder inflammation and transitional cell hyperplasia in the 6-month rasH2 mouse study. The same compound had evidence of gritty calculi (presumed to be precipitated drug or metabolite) in the urinary bladder in a shorter term 1-month rasH2 mouse study. Notably, these findings were not associated with tumors in the urinary bladder or elsewhere in the urogenital tract in rasH2 mice. Compounds D and H had evidence in the 6-month rasH2 mouse studies of an adaptive response (i.e., centrilobular hepatocellular hypertrophy) that is commonly associated with hepatic enzyme induction (Table 2). Furthermore, compound H was associated with more severe nephropathy in rasH2 mice (a condition common in aging rodents). However, there was no concurrent evidence of renal tumors in the 6-month rasH2 mouse study for compound H.
Correlation of Test Article–related Neoplastic Findings between 6-month rasH2 Mouse and 2-year SD Rat Studies
To assess the degree of concordance or lack thereof between the results of the 6-month rasH2 mouse and the 2-year rat studies, toxicology reports were evaluated from 2-year rat bioassays for all 10 compounds. Of the 10 compounds, 5 (compounds C, D, sunitinib, F, and bazedoxifene) had test article–related neoplasia in the 2-year rat bioassays (Table 2 and Figure 2). Of these 5 compounds, sunitinib and bazedoxifene had a tumor spectrum that was comparable to that observed in the 6-month rasH2 mouse studies (i.e., gastroduodenal carcinoma and ovarian granulosa cell tumor, respectively). In addition, sunitinib produced renal tubular neoplasia in rats associated with increased incidence and severity of chronic progressive nephropathy, while bazedoxifene produced an increase in pheochromocytomas in male rats.
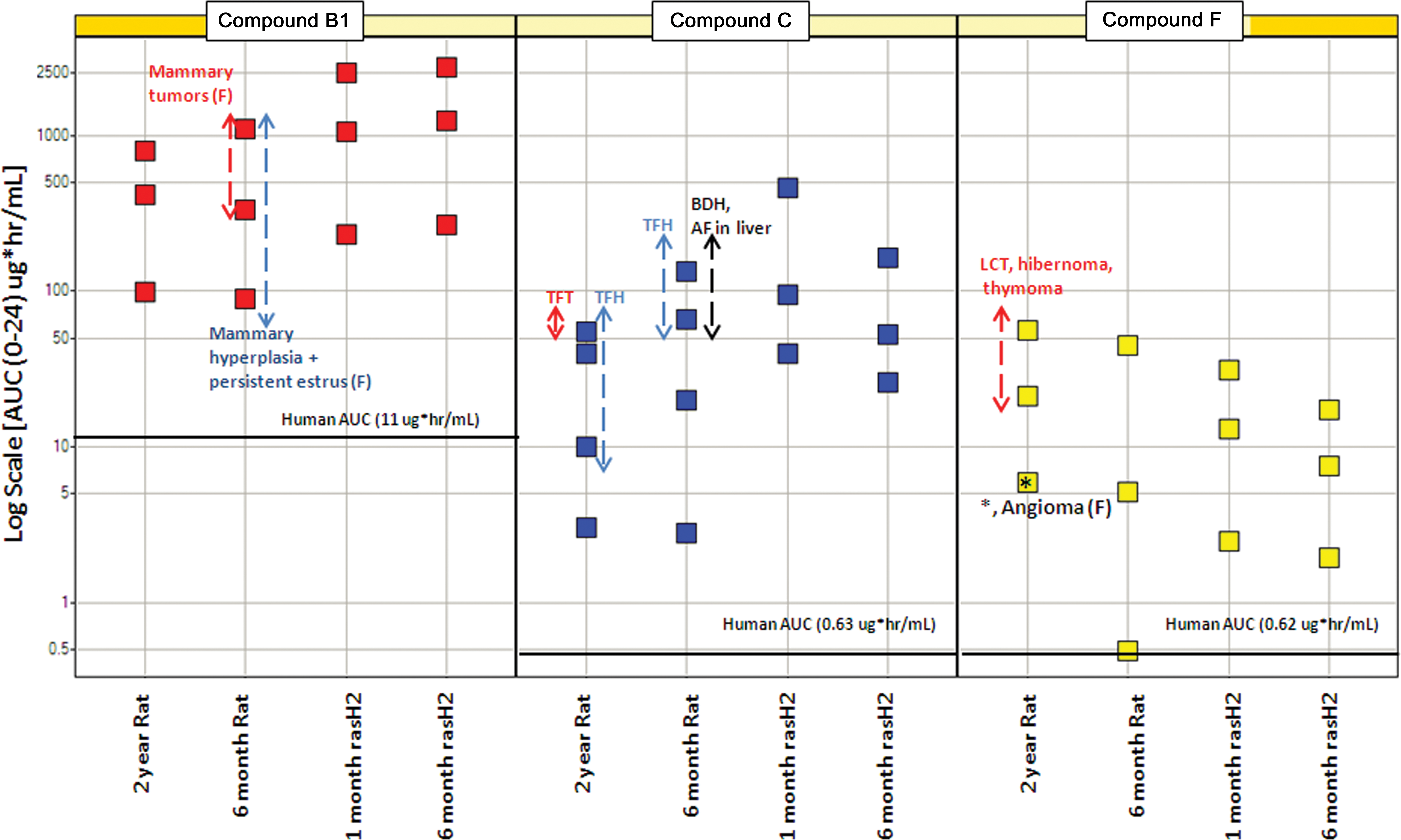
Interstudy comparison of tumor and tumor-related findings with compounds B1, C, and F in chronic mouse and rat studies. Each square represents a dose exposure in a toxicology study. The color-coded vertical lines highlight the dose exposures that correspond to a specific finding. The solid horizontal line depicts human AUC (0–24) at a clinical dose for the drug (TFT = thyroid follicular tumors, TFH = thyroid follicular hyperplasia, BDH = bile duct hyperplasia, AF = altered foci, LCT = Leydig cell tumor, AUC = area under the curve).
The remaining three compounds (C, D, and F) were positive for neoplasia only in the 2-year rat carcinogenicity studies. Specifically, compound C was associated with thyroid follicular tumors; compound D was associated with gastric carcinoid, hepatocellular tumors, thyroid follicular tumors, and Leydig cell tumors; and compound F was associated with Leydig cell tumors, hibernoma, angioma of the mesenteric lymph node, and thymoma (Figures 1 and 2, Table 2). Compounds C and D were hepatic microsomal enzyme inducers. Compound D–related gastric carcinoid occurred at exposure multiple of 183× human therapeutic exposure (based on total AUC, Figure 1). Compound F–related hibernoma occurred at an exposure margin >10× human therapeutic exposure (based on free drug AUC, unpublished results). Compound F also had an increase in benign thymomas in females at exposure margins of approximately 90× human therapeutic exposure (based on free drug AUC) with no thymoma tumor response at 41× human therapeutic exposure (Figure 2).
Correlation of Neoplastic Findings between 6-month rasH2 mouse and 6-month SD Rat Studies
The compounds that were positive for tumors in 6-month rasH2 mouse studies (compounds E and G) were not positive for neoplasia or other proliferative lesions in the 6-month rat studies, despite generally higher exposures in the 6-month rat studies. However, several other neoplastic or proliferative findings were noted with a few compounds in the 6-month rat studies (compounds B1, C, and D). Specifically, the observations included mammary tumors (fibroadenomas and adenocarcinomas) with compound B1, altered foci in the liver and thyroid follicular hyperplasia (related to hepatic enzyme induction) with compound C, and thyroid follicular hyperplasia, hepatocellular hypertrophy (related to hepatic enzyme induction), and Leydig cell hyperplasia with compound D (Table 2). For compound B1, it is unclear why the mammary hyperplasia and tumor observed in the 6-month rat study did not lead to mammary neoplasia in the 2-year rat study despite higher exposures in the 2-year study. Similarly, compound B2–related mammary hyperplasia was observed in the 6-month rat study without any evidence of test article–related effects in the 2-year rat study.
The compounds (C and D) that had test article–related proliferative or adaptive changes in 6-month rat studies such as hepatocellular hypertrophy, altered foci in the liver, and/or thyroid follicular hyperplasia developed thyroid or hepatocellular tumors in the 2-year rat studies.
The dog or NHP studies did not have any evidence of proliferative or neoplastic findings with all 10 compounds, except one. Compound F is an immunosuppressive agent that caused malignant lymphoma in several monkeys secondary to infection with lymphocryptovirus (unpublished results). Lymphoma was not observed in either the rat or rasH2 mouse carcinogenicity studies for the same compound.
Summary of the rasH2 Mouse Carcinogenicity Data Submitted to the Food and Drug Administration (FDA)
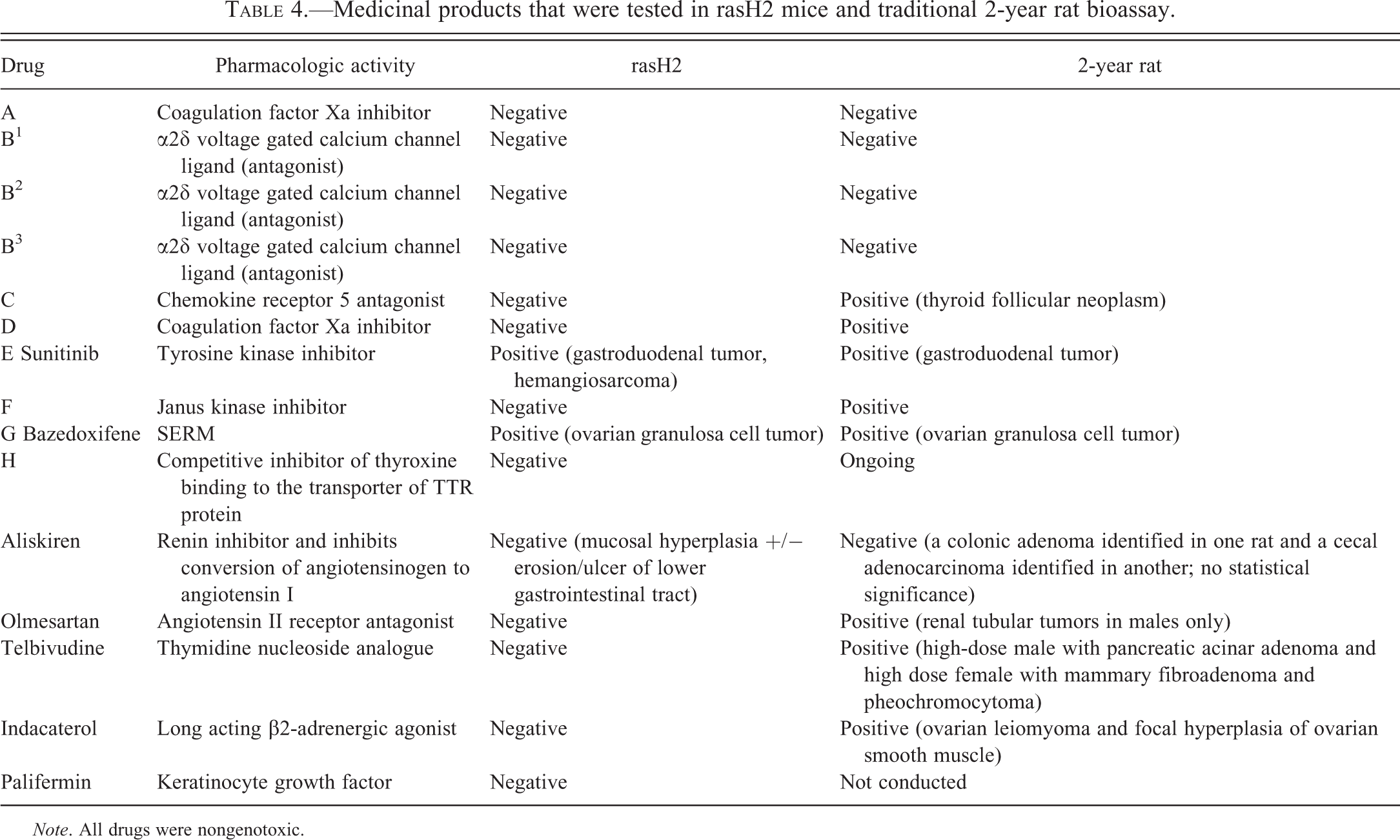
Among the 20 rasH2 mouse studies submitted to the FDA for product registration, the number of positive studies was 3, while the number of negative or inadequate studies was 17 (Boverhof et al. 2011; Table 5). These 3 positive studies include 2 of the compounds discussed above. Data were available for five additional biopharmaceuticals (aliskiren, olmesartan, telbivudine, indacaterol, and palifermin) tested in the 6-month rasH2 model (Aliskiren-label; Friedrich and Olejniczak 2011; Indacaterol-label; Olmesartan-label; Palifermin-label). There were no test article–related tumors at the end of the 6-month rasH2 mouse studies for any of these compounds. For Aliskiren, intestinal mucosal hyperplasia associated with ulcer/erosion was observed in both the 6-month rasH2 mouse study and the 2-year rat study. However, rare intestinal tumors (not statistically significant) were observed only in the 2-year rat study (Aliskiren-label). Interestingly, 3 of these compounds were positive in the 2-year rat bioassay (Table 4) and 1 (palifermin) was not tested in a rat carcinogenicity model to our knowledge (not reported in the label).
Medicinal products that were tested in rasH2 mice and traditional 2-year rat bioassay.
Note. All drugs were nongenotoxic.
Six-month rasH2 mouse carcinogenicity studies submitted to the Food and Drug Administration (FDA).a

aStudies were submitted to the Center for Drug Evaluation and Research of the U.S. FDA as of January 2011. The data were adapted from Boverhof et al. (2011).
bStudy was considered inadequate because it did not demonstrate sufficient exposure for a major genotoxic metabolite.
Overall, among the 13 compounds for which data from 6-month rasH2 mouse and 2-year rat studies were available, 15% (2/13) of the compounds were positive in the rasH2 model while 61% (8/13) compounds were positive in the conventional 2-year rat bioassay. About 15% of compounds (2/13) were positive in both rasH2 mouse and rat bioassays, while 38% (5/13) of the compounds were negative in both rasH2 mouse and rat bioassays (Table 4).
Discussion
A 6-month rasH2 mouse carcinogenicity study, when combined with a 2-year rat carcinogenicity study, is an accepted alternative to a 2-year mouse bioassay for cancer hazard identification of genotoxic and nongenotoxic small pharmaceutical molecules. One of the major deficiencies of the 2-year rodent bioassay is that it produces positive results for many molecules that do not increase the risk of cancer in humans (Alden et al. 2011). Among the numerous advantages offered by rasH2 mice, a distinct plus is the low rate of spontaneous background findings in rasH2 mice relative to the 2-year bioassay. Furthermore, the ability of the rasH2 model to predict known human carcinogens (75–85% positive responses for human carcinogens) is roughly equivalent to the 2-year rodent bioassays. In addition, the rasH2 mouse model is found to be positive for fewer chemicals when compared to rats, when rodent carcinogens not carcinogenic in humans were evaluated (i.e., lower false positive rate with rasH2 mice; Alden et al. 2011; Morton et al. 2002; Pritchard et al. 2003). Therefore, the rasH2 mouse model may be a better model for human risk assessment than the 2-year rodent bioassay and should be considered to support registration of small molecule pharmaceuticals that require carcinogenicity studies.
However, regulatory authorities in Europe and the United States report that the rasH2 model has been used infrequently to support drug development despite acceptance of the model for over a decade (Boverhof et al. 2011; Friedrich and Olejniczak 2011). A limiting factor has been the paucity of published data profiling the effects of actual drug candidates in rasH2 mice. In this report, we attempt to address this gap by sharing the results of carcinogenicity studies conducted in rasH2 mice with several Pfizer compounds and comparing the results to those from the standard 2-year rat bioassays.
Of the 10 Pfizer compounds, only 2 were positive in the 6-month rasH2 mouse studies. The test article–related findings in these 2 studies were gastroduodenal carcinoma (sunitinib) and ovarian granulosa cell tumors (bazedoxifene). Both gastroduodenal carcinoma and ovarian granulosa cell tumors are rare (<1% background incidence) spontaneous tumors in 6-month rasH2 mouse studies (Nambiar, Turnquist, and Morton 2012). These 2 compounds are likely to represent true risks for human carcinogenicity for the following reasons: knowledge of target biology, similar treatment-related effects at the end of 2-year rat studies, and the fact that both of these tumors are uncommon spontaneous findings in 6-month rasH2 mouse studies. The relevance of the hemangiosarcomas observed in the rasH2 model with sunitinib to human risk assessment is uncertain.
The mechanism of gastroduodenal carcinoma (Brunner’s gland tumors) associated with sunitinib is not well defined. This is because these are uncommon tumors in both mice and man and their genesis is largely unknown (Akino et al. 2002). However, it is important to note that one of the pharmacological targets of sunitinib is VEGFR1 (Flt1), which is expressed in Brunner’s gland of rodents and not reported in the Brunner’s glands of humans and the commonly used dog or monkey preclinical models (Hagedorn et al. 2005; Witmer et al. 2002). Furthermore, another receptor tyrosine kinase inhibitor (epidermal growth factor inhibitor) was reported to induce proliferative lesions in the Brunner’s gland in mice (Ritland et al. 2000). Sunitinib is a broad spectrum, competitive tyrosine kinase inhibitor and has the ability at high doses to inhibit several tyrosine kinases, thus disrupting cellular homeostasis (Mendel et al. 2003). These observations in the rasH2 model and standard 2-year rat bioassay support the role of sunitinib-driven target pharmacology in the genesis of these unique neoplasms.
For bazedoxifene, the mechanism of tumorigenesis is well recognized. The test article–related benign ovarian granulosa cell tumors in female mice and rats are a class effect of SERM, related to its pharmacology in rodents when treated during their reproductive lives (Long et al. 2001; Raloxifene-label; Tucker and Patterson 1984). Specifically, the test article interferes with the estrogen feedback mechanism to the hypothalamus and pituitary resulting in loss of the preovulatory luteinizing hormone (LH) surge and sustained increase in LH levels. This leads to follicular maturation arrest, persistent proliferative follicles with cyst formation, and with prolonged gonadotropin stimulation, hyperplasia of granulosa cells leading to benign tumor formation (unpublished results). This finding is not relevant for postmenopausal women (the targeted patient group) with naturally quiescent or no ovaries as there would be no follicular development. Indeed, this conclusion is supported by the clinical experience with bazedoxifene in postmenopausal women. Specifically, there has been no evidence of any renal or reproductive organ abnormalities at the current clinical dosages used (unpublished results).
A critical question especially in the earlier stages of drug development is whether a nongenotoxic drug candidate has any tumor producing liability in humans. Although extrapolating from 2 positive studies risks over interpretation, it was interesting to note that sunitnib-related gastroduodenal carcinoma also occurred in the 1-month rasH2 mouse study (the drug exposure was only slightly higher than in the 6-month rasH2 mouse study). However, for bazedoxifene, ovarian granulosa cell tumor occurred in the 6-month rasH2 mouse study and the 2-year rat study, but not in shorter studies in either model. Bazedoxifene is a SERM and such endocrine disruptors may need longer duration of action to elicit neoplastic effects on the female reproductive tract.
Among the 2-year SD rat studies with the same 10 Pfizer compounds, the number of compounds that were positive for test article–related tumors was higher (n = 5) than in the 6-month rasH2 mouse studies (n = 2). These include the 2 compounds (E and G) that were positive both in the rasH2 mouse model and the rat 2-year studies. For the remaining three compounds that were positive in the 2-year rat bioassay, the tumors could be explained as either rat-specific and/or with little relevance to human risk assessment based on mechanism of tumorigenesis or exposure multiples in rodent assays. For example, thyroid follicular tumors (observed with compounds C and D) and hepatocellular adenomas (compound D) were attributed to hepatic microsomal enzyme induction (Greaves 2012a). Leydig cell tumor observed with compound F was linked to drug-induced disruption of prolactin signaling and was determined to be not a significant cancer risk for humans (Greaves 2012b, unpublished results). Additionally, hibernoma (observed with compound F) may be explained by increased adrenergic stimulation of rat brown fat at exposure margins >10× human therapeutic exposure (free drug AUC; unpublished results). Compound F also had an increase in benign thymomas in females at exposure margins of approximately 90× human therapeutic exposure (free drug AUC) with no thymoma tumor response at 41× human therapeutic exposure. Benign angiomas of the mesenteric lymph node have been reported to have little relevance for human risk assessment (Radi and Morton 2012). The third compound D, a cannabinoid receptor 1 (CB1) antagonist, was positive for tumors (gastric carcinoid) only in the 2-year rat study. Although compound D–related gastric carcinoid in the 2-year rat bioassay may have been related to drug-related pharmacology, relevance to human risk assessment is low for the following reasons. The compound-related observation of gastric carcinoids (noted only in high-dose females) was considered secondary to parietal cell atrophy and hyperplasia of the superficial mucosa (foveolar epithelium). The gastric atrophy may have led to reduced gastric acid secretion and subsequent compensatory enterochromaffin-like (ECL) cell hyperplasia, hypergastrinemia, and development of carcinoids. Additionally, CB1 antagonists have the potential to disrupt gastrin homeostasis with the possibility of subsequent ECL hyperplasia and carcinoids in the stomach (Adami et al. 2004; Coruzzi et al. 1999). The relevance of this finding to human cancer risk assessment is low, given higher sensitivity of rat ECL cells to neoplasia, and drug-induced carcinoids have not been observed in humans despite extensive clinical usage of drugs that induce similar effects in rats (Betton et al. 1988; Greaves 2012c; Laine et al. 2000; Larsson et al. 1988). Furthermore, the exposure margin for the gastric carcinoid was clearly irrelevant in female rats at 183× human therapeutic AUC and no similar findings were observed in the 12-month dog study (unpublished results). The negative tumor outcome in the 6-month rasH2 mouse study for compound D may have been a function of the lower exposure of parent drug (about 5-fold lower) relative to the 2-year rat study. Taken together, the positive 2-year rat study for compound D is likely an exaggerated pharmacologic effect occurring at an irrelevant exposure margin in a sensitive species, and therefore not readily translatable to man. This would mean that all three compounds (compounds C, D, and F) that were positive only in the 2-year rat studies possibly reflect rat-specific phenomena or occurred only at high exposure margins and therefore pose little risk to humans. These three compounds were also negative in the rasH2 mouse model. This further highlights that the rasH2 mouse model predicts relevant human cancer risk as well as the rat, but with fewer findings that are not relevant for human risk assessment.
The rasH2 mouse studies reported in this article amounted to approximately 50% (10/20) of the reported FDA submissions using this model (Boverhof et al. 2011). When this data set of rasH2 mouse studies was combined with that available in the public domain (i.e., via labels and approval documents), certain interesting trends were noted. The percentage of compounds that were positive in the rasH2 model, that is 2/15 (13%), was similar to the percentage of compounds (12%; 11/94) that were considered relevant to humans from a carcinogenicity risk perspective (Friedrich and Olejniczak 2011). Similarly, the six compounds that had rat findings of questionable relevance to human risk assessment without positive rasH2 mouse tumor findings constituted 75% (6/8) of the positive rat studies. This percentage was similar to the percentage of positive rodent studies within a larger series of pharmaceuticals wherein the tumors were considered irrelevant to man (73%; 63/94; Friedrich and Olejniczak 2011). All of these data suggest that the rasH2 mouse model may better predict an overall tumor outcome (positive or negative) relevant to man than the 2-year rat model.
In summary, the data presented from our series of 10 compounds in the rasH2 mouse model yielded a small subset of compounds that were positive for tumors, but these 2 positive nongenotoxic compounds were also positive in the 2-year rat studies and 1 had a pharmacologic action reflecting a class effect for human cancer risk. The relatively higher percentage of positivity in the 2-year rat studies reflects the higher rate of neoplastic findings in 2-year rodent bioassays that are not relevant for human risk assessment. This article addresses a gap in the literature on treatment-related tumor findings in 6-month rasH2 mouse studies. It is important to note that relevance of carcinogenicity findings in rodents to human cancer risk assessment must be approached by a weight of evidence of approach factoring in target biology, mechanism of tumorigenesis, and toxicokinetic parameters.
Footnotes
Acknowledgment
The authors wish to express their sincere thanks to Dr. Roy L. Kerlin for his help in reviewing this article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
