Abstract
A 24-month oral carcinogenicity study of permethrin was conducted by feeding male and female CD-1 mice diets containing concentrations of 0, 20, 500, and 2,000 ppm of permethrin (males) or 0, 20, 2,500, and 5,000 ppm of permethrin (females). After approximately two years on study, surviving mice were sacrificed for the evaluation of chronic toxicity and/or carcinogenicity. An expert panel of pathologists was convened as a Pathology Working Group (PWG) to review coded liver histology sections from male and female mice and to classify all liver neoplasms according to current nomenclature and diagnostic criteria guidelines. The PWG results indicate that permethrin induced a significant dose-dependent increase in the incidence of hepatocellular neoplasms in treated female mice (p < .01) as well as a nonstatistically significant increase in the incidence of hepatocellular tumors in treated male mice. Given the continuum of the diagnoses of adenoma and carcinoma, and the difficulty in distinguishing some of the lesions, it is appropriate to consider only the combined incidences of hepatocellular tumors (adenoma and/or carcinoma) for biological significance and risk assessment.
Keywords
Permethrin [(3-phenoxyphenyl)methyl 3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane carboxylate] is a broad-spectrum, synthetic pyrethroid used widely as an insecticide, acaricide, and insect repellent (Boffetta and Desai 2018; United States Environmental Protection Agency [U.S. EPA] 2009; Yamada et al. 2017). First registered in the United States in 1979 for use on cotton, the U.S. EPA classified permethrin as a weak carcinogen, “likely to be carcinogenic to humans” by the oral route. Permethrin is also highly toxic to fish and aquatic invertebrates and is currently classified as a restricted use pesticide for crop and wide area applications, with the exception of wide area mosquito adulticide use (U.S. EPA 2009). U.S. EPA data reflect that each year approximately 2 million pounds (907,184.74 kg) of permethrin are applied to agricultural (e.g., food/feed crops, livestock housing, and food-handling establishments), residential (e.g., outdoor and indoor spaces; pets and clothing) and public health uses sites (e.g., Public Health Mosquito abatement programs). The majority of permethrin (70%) is used and applied in nonagricultural settings and, due to its low cost, high efficacy, and low incidence of pest resistance, permethrin remains the most widely used mosquito adulticide in the United States (U.S. EPA 2009).
Although previous studies have not shown permethrin-induced carcinogenicity in the rat, an increased incidence of lung and liver tumors have been reported in mice exposed to high levels of permethrin in the two-year bioassay (Ishmael and Litchfield 1988; U.S. EPA 2000). However, in these two reports, both lung (Ishmael and Litchfield 1988; U.S. EPA 2000) and liver (U.S. EPA 2000) neoplasms appear to arise through modes of action that may not be applicable to humans and, therefore, are likely irrelevant for determining human risk and disease development. Nonneoplastic lesions in the liver, such as centrilobular hypertrophy, karyomegaly, and proliferation of the smooth endoplasmic reticulum (SER), have also been documented in the rodent model. Yet, these changes are considered representative of reversible and adaptive hepatocellular alterations rather than an adverse response (Ishmael and Litchfield 1988; U.S. EPA 2000).
A 24-month mouse oral carcinogenicity study of FMC 33297 (permethrin) was conducted in 1979 at Bio/Dynamics, Inc., in East Millstone, NJ (currently Envigo, Inc.). For this study, groups of 75 male and 75 female Charles River, CD-1 mice were exposed to permethrin at dietary concentrations of 0, 20, 500, and 2,000 ppm (males) or 0, 20, 2,500, and 5,000 ppm (females). After approximately two years on study, surviving mice were humanely sacrificed for the evaluation of toxicity and carcinogenicity associated with chronic permethrin exposure. As part of a voluntary review in accordance with U.S. EPA Pesticide Regulation (PR) Notice 94-5: Requests of Reconsideration of Carcinogenicity Peer Review Decisions Based on Changes in Pathology Diagnoses (U.S. EPA 1994), a Pathology Working Group (PWG) was conducted to reexamine all liver slides from this study.
The present PWG was composed of independent consulting pathologists with expertise in rodent toxicity and carcinogenicity. The purpose of the PWG was to classify all liver neoplasms following current nomenclature guidelines as stated in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) publication for the hepatobiliary system (Thoolen et al. 2010). This article provides the results, discussion, and conclusions of the PWG review of the microscopic slides.
Method
PWG
A PWG was conducted to review all liver slides from a 24-month oral carcinogenicity study of permethrin (study no. FMC 33297) in CD-1 mice (Table 1). The goal of the PWG was to classify all liver neoplasms in accordance with the current INHAND guidelines (Thoolen et al. 2010). The PWG review followed the procedures recommended in the U.S. EPA PR Notice 94-5 (U.S. EPA 1994). As part of the PWG process, a peer review of all liver sections was conducted by a board-certified anatomic veterinary pathologist prior to the PWG meeting. The PWG chairperson organized and presented the material to a PWG panel composed of five independent consulting pathologists with expertise in rodent toxicity and carcinogenicity studies. The PWG chairperson was a nonvoting member during the PWG review. As required by U.S. EPA PR Notice 94-5, the PWG participants examined all slides containing sections of liver for which there were differing diagnoses involving proliferative lesions (neoplasia or hyperplasia) as recorded by the study and reviewing pathologists. In addition, the PWG examined all sections of liver that had a diagnosis of hepatocellular adenoma, hepatocellular carcinoma, or hepatocholangiocarcinomas that were originally reported by the study pathologist or by the reviewing pathologist.
Experimental Design: A 24-month Oral Carcinogenicity Study with Permethrin (FMC Study Number 33297).
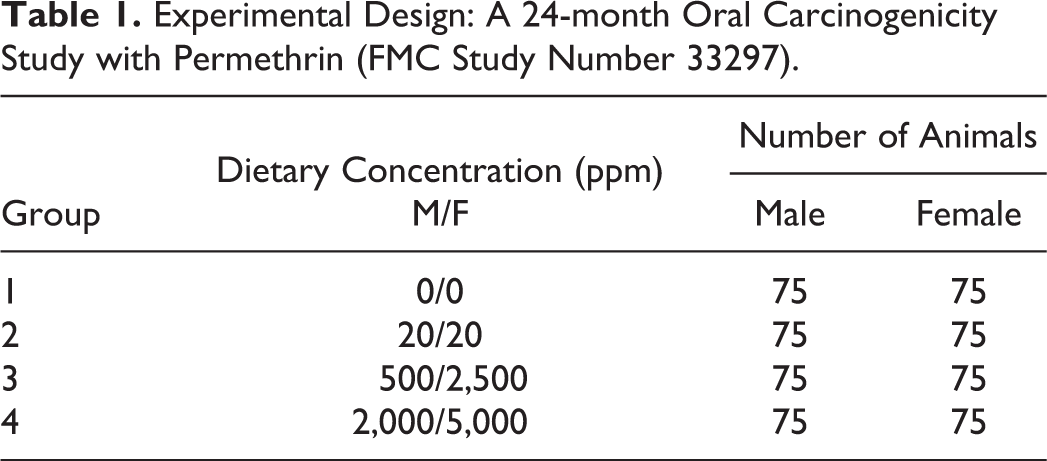
Liver slides from male groups I, II, III, and IV (0, 20, 500, and 2,000 ppm) and female groups I, II, III, and IV (0, 20, 2,500, and 5,000 ppm) were presented to the PWG as coded slides, so that the PWG participants were blinded to the treatment group and previous diagnoses for each animal. Each PWG participant recorded their diagnoses and comments on worksheets that were prepared by the PWG chairperson. The PWG only evaluated the liver for proliferative hepatocellular changes and did not consider other lesions that may have been present. Each lesion was discussed by the panel, reexamined if necessary, and final opinions were recorded by the PWG chairperson. The consensus diagnosis of the PWG was achieved when the majority of the PWG participants were in agreement.
After the PWG completed the slide review and the consensus diagnoses were recorded by the PWG chairperson, the slides were decoded, and the microscopic findings were tabulated by treatment group for both male and female mice. No changes were made to the consensus diagnoses after the slides were decoded by treatment group.
Statistical Analysis
Tumors were statistically analyzed using the poly-3 statistical analysis as first described by Bailer and Portier (1988) and modified by Bieler and Williams (1993). Analysis of malignant tumors, benign tumors, or benign and malignant tumors combined was performed. The following comparisons were made (1) each test article group versus control (3 comparisons) and (2) trend test across all 4 groups.
For each tumor “X,” tumor-bearing animals were assigned a weighted at-risk score = 1. Likewise, non-tumor-bearing animals that lived the full study period (733 days in this study for males and 734 for females) were assigned a weighted at-risk score = 1. Non-tumor-bearing animals that died prior to the end of the full study period were assigned a weighted at-risk score, based on the time of death, according to the following formula: (day of death/full study period)3. The weighted number of animals at risk (Nw) in each group was calculated for each tumor individually and defined as the sum of these weighted at-risk scores across a treatment group.
Conceptually, Nw estimates the weighted number of animals at risk based on the cumulative time on study of all animals in a group. If all non-tumor-bearing animals in a group survived until the scheduled terminal sacrifice, then Nw = N (the weighted number of animals at risk = the original number of animals in the group). If at least one non-tumor-bearing animal died prior to the scheduled terminal sacrifice, then Nw < N. Thus, Nw reflects group mortality in that early deaths of non-tumor-bearing animals yield a smaller Nw relative to N.
The weighted number of animals at risk and an adjusted variance estimate described by Bieler and Williams (1993) were used in a modified 1-sided Cochran–Armitage test looking for increased incidence of tumors in the test article as compared to control. Asymptotic p values are reported in the summary tables.
According to the Haseman (1983) rule for the statistical evaluation of common and rare tumors in the two-year rodent bioassay, p values less than .01 (p < .01) and .05 (p < .05) should be used to determine the statistically significant differences for common and rare tumors, respectively (Haseman 1983; Lin and Ali 1994). For a two-year bioassay, in which both sexes from two different animal species are used, a revised decision rule of p < .025 and p < .005 is used to evaluate trend tests for rare and common tumors, respectively (U.S. Food and Drug Administration 2001; Organization for Economic Cooperation and Development 2010). Common tumors are defined as tumors with a spontaneous incidence of >1%. Therefore, since hepatocellular adenoma and carcinoma are considered common tumors in the mouse model with an incidence rate >1%, the significance level for this study was set at p < .01.
All statistical analyses were conducted with the SAS® software system (SAS® Proprietary Software; Version 9.2, SAS Institute Inc., Cary, NC, 2002-2008).
Results
The PWG results indicated that, in females, permethrin induced a dose-dependent increase in typical appearing hepatocellular adenomas without any features of carcinoma in the mid-dose (2,500 ppm) and high-dose (5,000 ppm) groups, compared to controls (p < .01). This effect was not observed in the livers from female mice in the low-dose (20 ppm) group (Table 2). Trend analysis further supported the statistically significant increases in hepatocellular adenoma and adenoma/carcinoma combined in the high-dose (5,000 ppm) females (p < .0001, Table 2). In male mice, a higher incidence of hepatocellular adenomas was observed across all treatment groups (20, 500, and 2,000 ppm), compared to controls (p < .05). However, this apparent increase in tumor incidence was not statistically significant at the suggested significance level of p < .01 for common hepatocellular neoplasms in the CD-1 mouse strain (Haseman 1983). There was no increase in the incidence of hepatocellular carcinoma in male or female mice treated with permethrin. The incidence of hepatocellular adenoma or adenoma/carcinoma combined was increased in the mid-dose (500 ppm) and high-dose (2,000 ppm) males, but only with a significance level of p < .05; this increase was not associated with a positive trend test (Table 3).
Pathology Working Group Incidence of Primary Hepatocellular Neoplasms in Female Mice.
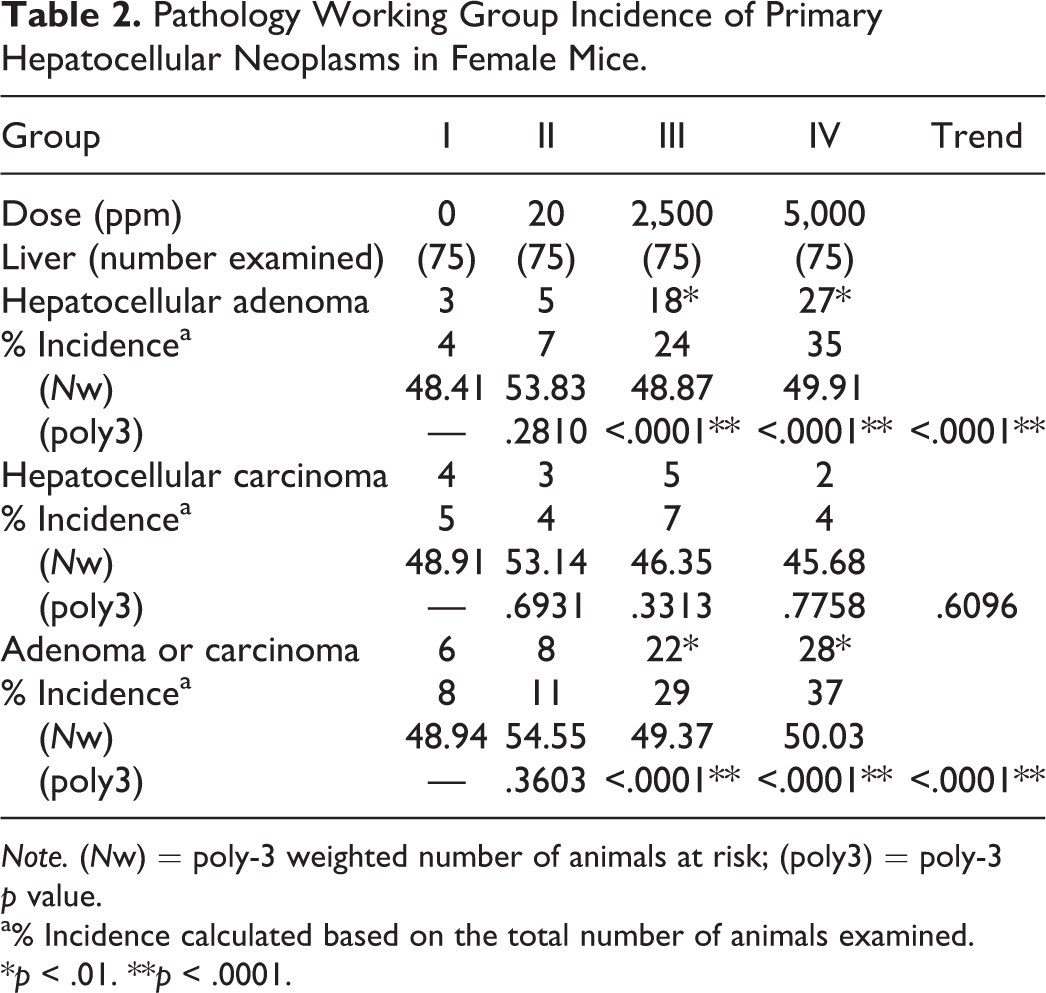
Note. (Nw) = poly-3 weighted number of animals at risk; (poly3) = poly-3 p value.
a% Incidence calculated based on the total number of animals examined.
*p < .01. **p < .0001.
Comparison of the Study Pathologist and Pathology Working Group Incidence of Primary Hepatocellular Neoplasms in Male Mice, Including Poly-3 (Trend) Analysis.
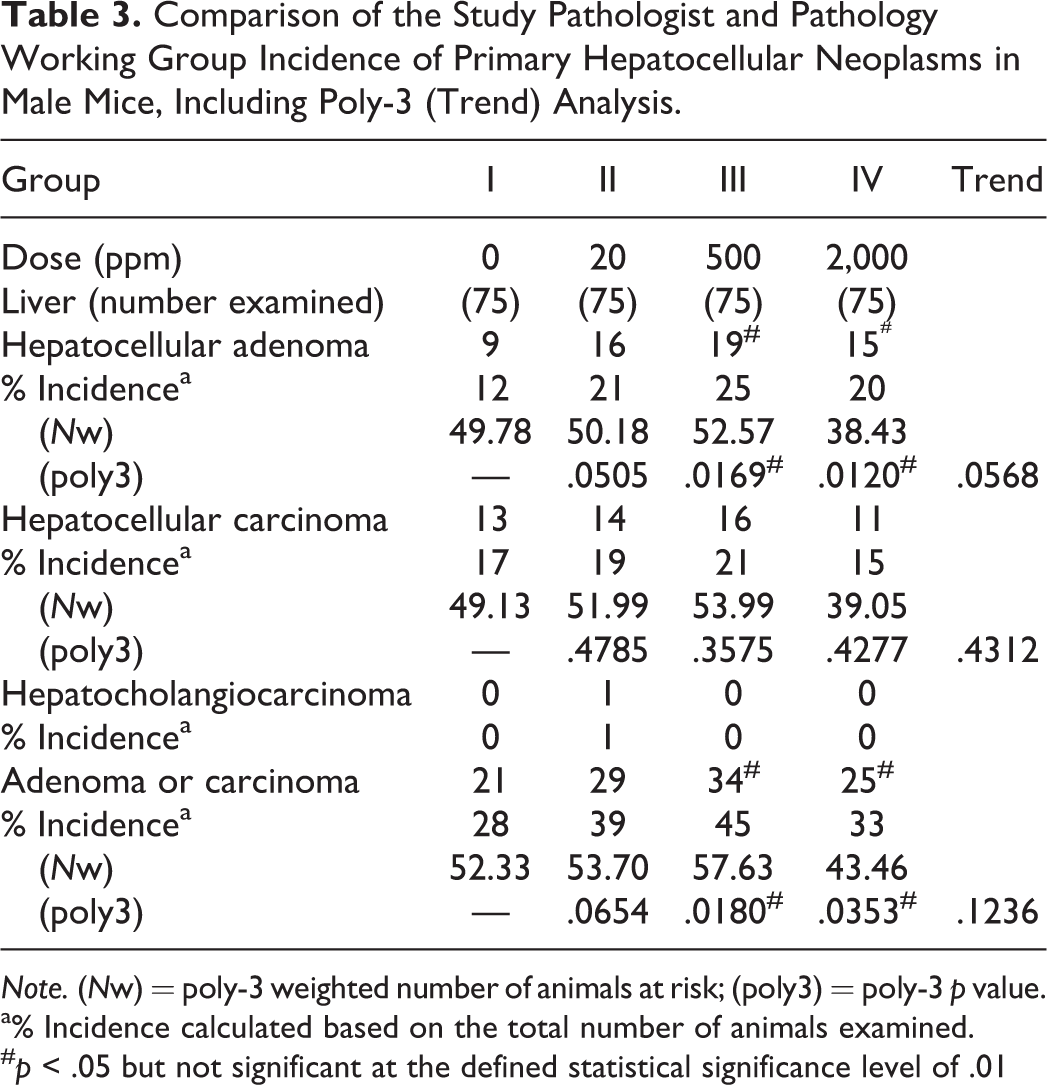
Note. (Nw) = poly-3 weighted number of animals at risk; (poly3) = poly-3 p value.
a% Incidence calculated based on the total number of animals examined.
# p < .05 but not significant at the defined statistical significance level of .01
Hepatocellular adenomas were generally well-circumscribed, nodular, and expansile neoplasms that compressed the adjacent hepatic parenchyma (Figure 1). These neoplasms were comprised of sheets of hepatocytes exhibiting an irregular growth pattern that disrupted or replaced the normal lobular architecture. Hepatocytes were variable in size and frequently exhibited increased eosinophila. Mitotic figures, cellular atypia, and necrosis were occasionally observed.
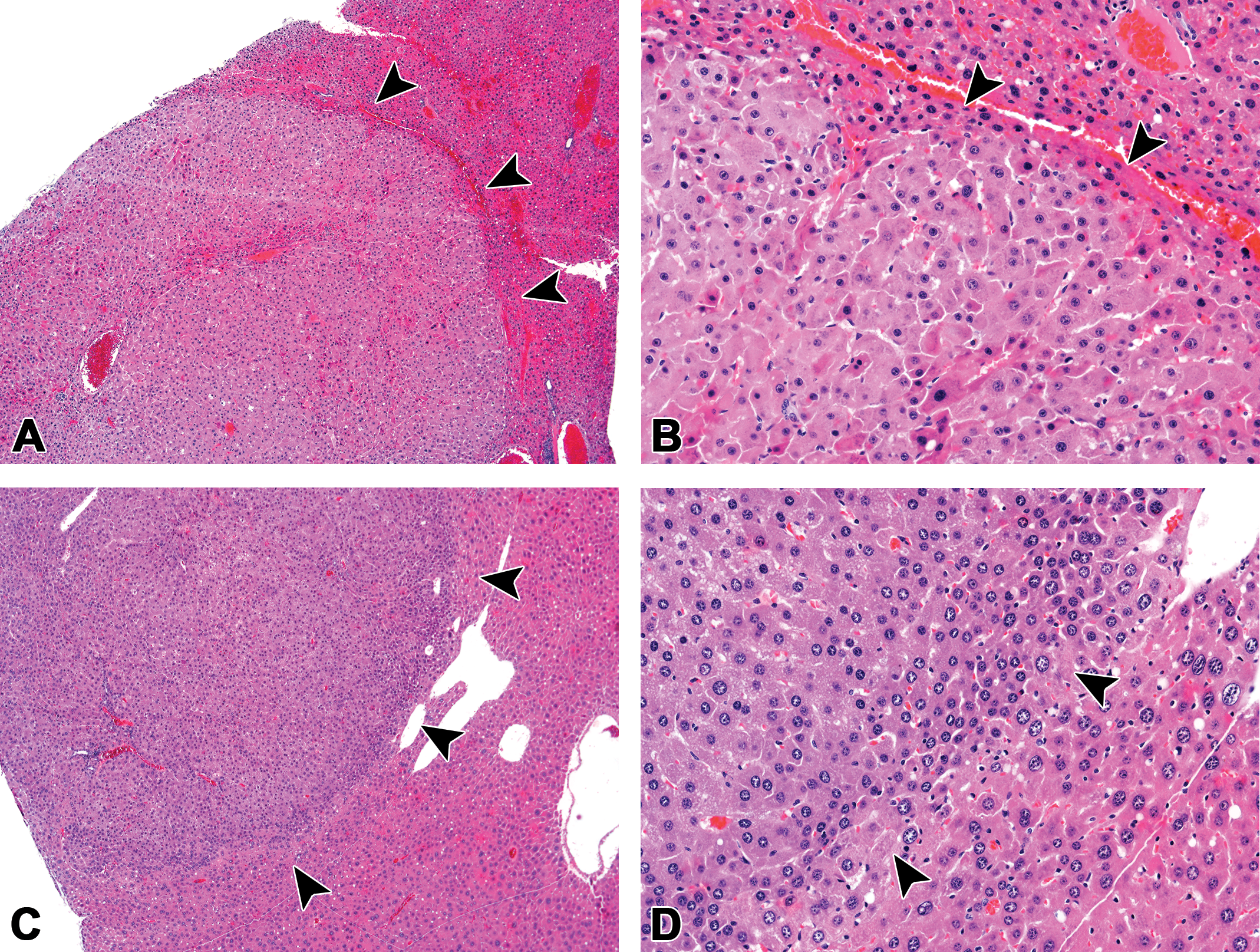
Hepatocellular adenomas in CD-1 mice chronically exposed to high dietary concentrations of permethrin. (A and B) Hepatocellular adenoma, female CD-1 mouse, high-dose (5,000 ppm permethrin) group. (C and D) Hepatocellular adenoma, male CD-1 mouse, high-dose (2,000 ppm permethrin) group. Hepatocellular adenomas were generally well-circumscribed, expansile nodules with the loss of the normal lobular architecture. Note the clear line of demarcation and compression of adjacent parenchyma (arrowheads). Original Objective 4× (A and C) and Original Objective 20× (B and D). H&E.
Hepatocellular carcinomas were characterized by local infiltrating growth and loss of the normal lobular architecture with no distinct line of demarcation (Figure 2). Trabecular formation with thickened cords comprised of multiple layers of hepatocytes was a key feature in this tumor type. Hepatocellular carcinomas also exhibited cellular pleomorphism with alterations in tinctorial staining patterns and increased mitoses and areas of hemorrhage and necrosis were frequently observed.
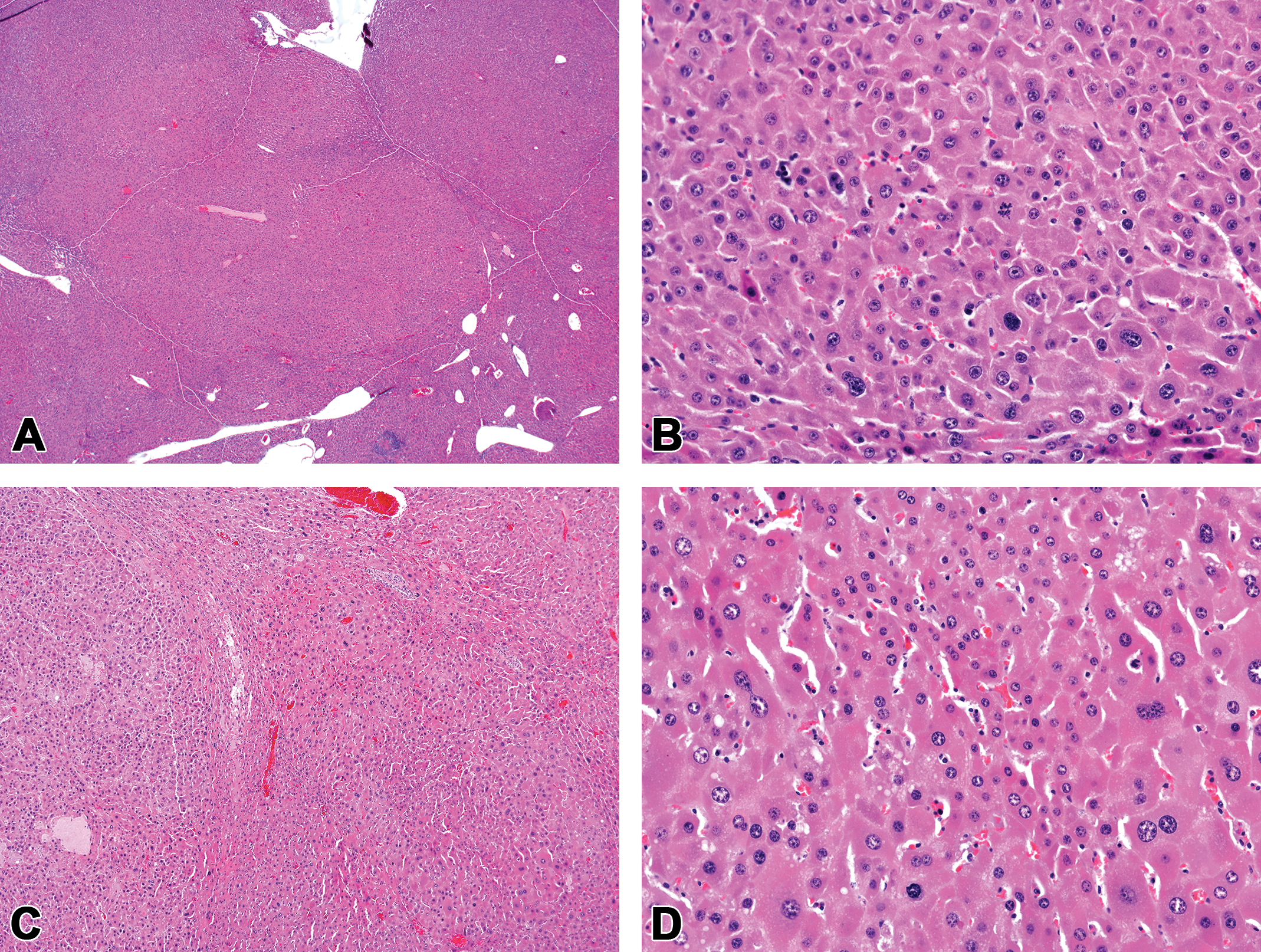
Hepatocellular carcinomas in CD-1 mice chronically exposed to high dietary concentrations of permethrin. (A and B) Hepatocellular carcinoma, female CD-1 mouse, high-dose (5,000 ppm permethrin) group. (C and D) Hepatocellular carcinoma, male CD-1 mouse, high-dose (2,000 ppm permethrin) group. Hepatocellular carcinomas were characterized by infiltrative masses with no clear line of demarcation, altered tinctorial staining patterns, and marked cellular pleomorphism. Note the thickened hepatic cords and distorted trabecular pattern. Original Objective 4× (A and C) and Original Objective 20× (B and D). H&E.
While PWG consensus concluded that the majority of proliferative lesions in the liver had features consistent with either hepatocellular adenoma or carcinoma, some adenomas, particularly in the males, contained questionable areas of atypia and/or trabecular formation that lead to considerable discussion among PWG participants. Provided that mouse adenomas may contain areas of atypia suggesting progression to malignancy (Harada et al. 1999), the PWG members concluded that hepatocellular adenomas and carcinomas in this study represented a continuum of the same disease process. As such, the PWG recommended that the incidence rates of adenomas and carcinomas be combined to more appropriately evaluate any possible effect of chronic permethrin exposure on the mouse liver.
Discussion
The liver is a primary target organ for permethrin-induced toxicity in mice (U.S. EPA 2000). Hepatocellular changes associated with permethrin exposure include centrilobular hypertrophy, increased liver weights, karyomegaly, Kupffer cell hypertrophy, inflammation, and hepatocellular adenoma. Nonneoplastic lesions are generally reversible and resolve after cessation of permethrin exposure, and neoplastic lesions (e.g., adenoma and carcinoma) do not appear to increase or decrease in incidence during recovery (Ishmael and Litchfield 1988; U.S. EPA 2000). In this two-year study, permethrin induced a dose-dependent increase in hepatocellular adenoma incidence among female mice exposed to dietary concentrations of permethrin at levels of 2,500 ppm or higher (p < .01). While an increase in incidence of hepatocellular adenoma was also observed in males across treatment groups (20-, 500-, 2,000-ppm permethrin), this increase in tumor incidence was not statistically significant at a significance level of p < .01 for common hepatocellular neoplasms in the CD-1 mouse strain. There was no increase in the incidence of hepatocellular carcinoma in male or female mice treated with permethrin. However, PWG participants determined that hepatocellular adenomas and carcinomas represented a continuum of the same disease process and concluded that incidence rates of both tumor types should be combined to better assess any potential hepatocarcinogenic effects of permethrin in the mouse model.
Regulatory decisions regarding the carcinogenic potential of chemicals, compounds, and environmental contaminants are typically based on the results of long-term (2-year) rodent studies. In these carcinogenic studies, increased incidences of both benign and malignant neoplasms, arising from the same cell type, may occur in treated animals compared to controls. Yet, when analyzed separately, these increased incidences may not reach statistical significance, thus masking any possible treatment-related effect. In these instances, it is often appropriate to combine neoplasms for statistical analysis to determine the degree of a treatment-related increase in tumor incidence. Among the criteria for combining neoplasms currently used by the National Toxicology Program, neoplasms can be combined if “substantial evidence exists for progression of benign to malignant neoplasms of the same histomorphogenic type” or if the “histomorphogenesis is comparable” (McConnell et al. 1986). During this review, the PWG participants determined that there was enough similarity between areas of atypia within hepatocellular adenomas and hepatocellular carcinomas to justify combining neoplasms for statistical analysis. They concluded that hepatocellular adenoma and carcinoma likely represent a continuum of the same disease process and arise via a similar mode of action (MOA).
Cytochrome P450 induction (specifically CYP4A) via peroxisome proliferator-activated receptor alpha (PPARα) activation is the proposed MOA for permethrin-induced hepatocellular tumors in the mouse (Ishmael and Litchfield 1988; Kondo et al. 2012; U.S. EPA 2000). Mitogenicity via constitutive androstane receptor (CAR) and PPARα are considered key components of the MOA for hepatocyte tumorigenicity in the rodent model; CYP2B and CYP4A are well-documented biomarkers for CAR and PPARα activation (Corton et al. 2014; Corton, Peters, and Klaunig, 2018; Lake 2018; Yamada 2018). Female CD-1 mice exposed to feed containing 5,000 ppm (780–807 mg/kg/day) of permethrin for 52 weeks exhibited marked increases in hepatocellular microsomal enzyme activity that included a 142% to 283% increase in CYP, CYP1A, CYP2B, CYP2E1, and CYP3A2 as well as a 3-fold increase (829%) in CYP4A induction, relative to controls (U.S. EPA 2000). In a study by Kondo et al. (2012), CYP4A activity was also increased after 7 and 14 days of permethrin exposure. However, in addition to centrilobular hypertrophy and increased liver weights, proliferation of peroxisomes rather than that of SER was observed, further suggesting that PPARα may play a role in permethrin-induced tumorigenesis in the mouse liver (Kondo et al. 2012). Similar changes in CYP activity (CYP2B, CYP2E1, and CYP4A) have also been recorded in permethrin-associated lung changes, including club cell hyperplasia and bronchoalveolar adenoma (U.S. EPA 2000).
Corton et al. (2018) describe the five key events (KE) in the PPARα-dependent MOA for rodent hepatocarcinogenesis, these include PPARα activation (KE1), alterations in cell growth pathways (e.g., increased expression of cyclins or c-Myc, KE2), perturbation of hepatocyte growth and survival (e.g., hepatocyte proliferation and inhibition of apoptosis, KE3), selective clonal expansion of preneoplastic foci (i.e., initiated) cells (KE4), and, of course, increases in hepatocellular neoplasms (e.g., adenomas and carcinomas, KE5). Although there is ample scientific support for the PPARα-dependent rodent cancer MOA, overwhelming evidence exists demonstrating that humans are not responsive to the carcinogenic effects of PPARα activators (Corton et al. 2018; Felter et al. 2018). Therefore, while permethrin consistently induces proliferative changes in the mouse liver, permethrin-induced hepatocarcinogenesis appears to be specific to the rodent model with no apparent human relevance.
In summary, chronic dietary exposures to permethrin-induced increased incidences of hepatocellular adenoma in female mice at concentration levels of 2,500 ppm or higher, when compared to controls. Permethrin exposure did not increase the incidence of hepatocellular carcinoma in either male or female mice. This review enabled PWG participants to reclassify hepatic tumors using the current INHAND diagnostic criteria and nomenclature guidelines and revealed that the hepatocellular adenomas and carcinomas likely represented a continuum of the same disease process and thus should be combined for statistical analysis.
Footnotes
Acknowledgments
The authors would like to thank Samuel Cohen (University of Nebraska Medical Center), Thomas Osimitz (Science Strategies, LLC), and Tomoya Yamada (Sumitomo Chemical Co., Ltd.) for their critical review of this article. The authors also thank Ms. Nancy Harris for technical assistance and Ms. Emily Singletary and Ms. Beth Mahler for image capture, photographic editing, and support.
Author Contributions
All authors (EMQ, GAB, JMC, RRM, AKR, JAS, LF, and JFH) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Permethrin Data Group II, Washington, DC.
