Abstract
The toxicologic pathologist plays a vital role in the scientific community, using their unique blend of diagnostic and investigative skills to advance biomedical research, public health, drug discovery, or regulatory practices. But what exactly do toxicologic pathologists contribute? Where do these specialized professionals work? How can toxicologic pathologists maximize their efficiency and potential? To enlighten students and trainees, as well as early- or mid-career toxicologic pathologists, or even those approaching retirement, the Career Development and Outreach Committee of the Society of Toxicologic Pathology (STP) sponsored a career development workshop entitled “Practical Strategies for Navigating Toxicologic Pathology in One’s Early Career…and Beyond!” in conjunction with the STP 37th annual symposium. The workshop featured toxicologic pathologists from contract research organizations and the pharmaceutical industry, who provided their perspectives on career preparation, evolving veterinary pathologist roles within various sectors of toxicologic pathology, the fundamentals of safety assessment, logistics of projects involving good laboratory practices, tools for effective interpretation and communication of anatomic and clinical pathology results, and a recap of scientific resources available to support the toxicologic pathologist in his or her journey. This article provides brief summaries of the talks presented during this career development workshop.
Toxicologic pathology is the subspecialty of pathology that bridges and applies the principles of multiple related disciplines, including toxicology, pharmacology, pharmacokinetics, and clinical and anatomic pathology, to assess the toxicity of an agent in complex biological systems. The training track that culminates in a specialization in toxicologic pathology typically involves general medical training in veterinary medicine followed by postgraduate training and certification in (clinical and/or anatomic) pathology and/or toxicology, with or without a research graduate degree (masters and/or PhD) in related disciplines (Turner et al. 2015; Bolon et al. 2010; Ettlin et al. 2009; van Tongeren et al. 2011). Notwithstanding the decade or more of focused training, a substantial amount of on-the-job training separates the entry-level and independent full-fledged toxicologic pathologists in this field. The job description for toxicologic pathologists is ever-evolving to incrementally include activities beyond histological slide or clinical pathology data interpretation. Therefore, a comprehensive understanding of experimental research, chemical or pharmaceutical product development, and safety assessment enables the toxicologic pathology professional to apply his or her skills to influence decisions and drive scientific progress. In this paradigm, a strategy empowered by the awareness of the required credentials and experiences for distinct roles that toxicologic pathologists may play (in academia, contract research organization [CRO], pharmaceutical, chemical/agrochemical, and government) is strongly related to one’s career success.
The Career Development and Outreach Committee sponsored the career development workshop entitled “Practical Strategies for Navigating Toxicologic Pathology in One’s Early Career…and Beyond!” as part of the activities at the 2018 Society of Toxicologic Pathology (STP) annual symposium in Indianapolis, Indiana. Based on an informal audience poll, there were students/trainees; early-, mid-, and late-career colleagues; consultants; and retirees in attendance for the session. Keith Nelson, DVM, PhD, DACVP, opened the workshop with an overview of the training and career development milestones that lie ahead of a trainee in his or her trajectory toward becoming a toxicologic pathologist. Stacey Fossey, DVM, PhD, DACVP, DABT, followed with an overview of the basics of pharmaceutical product discovery and development and evaluation of safety in toxicology studies. Bevin Zimmerman, DVM, PhD, DACVP, reviewed the logistics of evaluating toxicity studies in a good laboratory practice (GLP) setting. Lila Ramaiah, DVM, PhD, DACVP, provided an update on current and future paradigms for data visualization and analysis and discussed approaches for effective communication of findings and management of scientific disagreements. Ken Schafer, DVM, PhD, DACVP, ended the workshop with an extensive overview of the various resources available to support the toxicologic pathologist, including important textbooks, reference articles, and websites. The workshop concluded with a roundtable discussion involving all five presenters engaged by an active audience participation.
Becoming the Toxicologic Pathologist You Never Knew You Wanted to Be
Dr. Keith Nelson (MPI Research), an experienced toxicologic pathologist and mentor (STP Mentor of the Year, 2017), addressed the gap in the education of veterinary students and graduate trainees as it relates to the field of toxicologic pathology. As Dr. Nelson pointed out, most pathology training is focused on diagnostics even though veterinary pathologists, particularly anatomic pathologists, are equally employed in academia and industry. Dr. Nelson discussed how mentorship plays a critical role in closing this information gap and provided valuable insights on how to become an effective mentor or mentee for future generations of toxicologic pathologists. The narrative below is provided by Dr. Nelson as a part of his presentation.
The subdiscipline of toxicologic pathology is practiced by many veterinary pathologists, in approximately equal numbers as seen in academic positions, some of which may also have some toxicologic pathology aspect (34% in industry vs. 38% in academic positions, of respondents in a 2012 American College of Veterinary Pathologists [ACVP] survey retrieved from https://cdn.ymaws.com/www.acvp.org/resource/resmgr/News_Resources/Newsletters/2013/july13.pdf, last accessed August 27, 2018), and greater numbers than are employed in purely diagnostic positions. This is particularly true for anatomic pathologists, while clinical pathology has lagged in representation in the field but is now becoming more commonly represented in toxicologic pathology. Despite this strong representation in the overall veterinary pathology field, toxicologic pathology as a subdiscipline is often not formally taught beyond laboratory animal pathology courses in many training programs. Occasionally, there are programs that do have additional training or opportunities for gaining experience in the practice of toxicologic pathology, but potential practitioners should be proactive in managing their own early training, both prior to entering the profession and in their early career. This includes identifying and utilizing mentors within the field and continuing aspects of their early training in this more specialized subdiscipline. The earliest career development often occurs within veterinary school where trainees may focus on the overall discipline of veterinary pathology, utilizing didactic classwork, externships, informal interactions with peers through the ACVP-sponsored pathology clubs, and formal and informal interactions with residents and faculty mentors. These interactions are predominantly aimed, as they should be, at developing the core skills underlying the discipline of general pathology.
Following veterinary school, those interested in pathology will almost universally continue into residency positions where focused didactic coursework and apprenticeship-style training with pathology faculty are the primary methods of training and acquiring the basic skills of pathology (Kidney, Dial, and Christopher 2009; Christopher et al. 2003; Munson et al. 2010; Sharkey et al. 2006). Rarely do residents at this stage recognize the potential for a career in toxicologic pathology. Much of the time, residency training in pathology is focused on companion and food animal diagnostic pathology, which represents most of the casework seen at training institutions. Additional training opportunities in toxicologic pathology or laboratory animal pathology are available to a limited extent but are generally driven by individual interests and consist of an array of formal and informal training including: STP modular courses, externships, individual research projects, attendance at STP or other association meetings, and formal and informal interactions with mentors in the field. During this phase, there may be additional research-focused training or entry into a graduate research program.
As trainees complete their initial residency training and enter the profession, they are rarely fully prepared for toxicologic pathology. The experiential portion of their training does not often include extensive experience with toxicologic pathology or laboratory animal pathology, although this may vary with different training programs. A variable amount of additional training and mentoring is required in most positions and arguably forms an essential part of professional maturation for many toxicologic pathologists over the first 2 to 5 years of their postboards professional career. Additionally, this period would be where specialized skills and abilities would be focused, developed, and integrated based on the needs of the employee and employer. Opportunities for self-development and learning can be taken advantage of at this point in the career, if only for providing improved knowledge of the field. Directed development of a specialized area of interest or expertise often starts at this time and continues throughout one’s career. At this point, training generally consists of self-directed study of core materials and texts, corporate training in standing operating procedures, or of various forms of mentorship, with rare or sporadic didactic training in toxicologic pathology or associated disciplines.
Mentorship is often one of the most utilized forms of training in the discipline of veterinary pathology and subdiscipline of toxicologic pathology. This may take on many forms, but the basic format of mentoring system remains; be it as informal or formalized mentorship, peer-based or instructor-student based, and short-term or lifelong. Thus, it would behoove those interested in furthering their knowledge base and careers to have a less abstracted knowledge of mentorship. There are several forms of mentorship, and they may be used differently at different points during training and early career development as a toxicologic pathologist. An active involvement of both mentor and mentee is crucial in the process. Mentorship goal here is not to have a single individual as the sole deliverer of all the accumulated wisdom and knowledge that a mentee receives. A village of mentors may be the best approach for an individual rather than one specific person. This will particularly hold true when trying to gain a broad knowledge base. Mentorship works best when the mentor understands not only the needs of the mentee but also his own capabilities. While one may be a formal or informal mentor to a trainee, this does not preclude the use of other mentors as valuable resources to expand the knowledge base or progress the career of the mentee. Serial or parallel mentors, short-term situational mentors, and peer mentors are all ways in which mentorship can be beneficial to an individual. Having an array of resources to draw on, including experiential, didactic, and professional relationships, will make a mentor more valuable and strengthen the relationship between the mentor and mentee. An ideal mentor strives for teaching a mentee more about how to learn rather than telling them what to learn.
Mentorship is also an aspect of the professional growth of any experienced pathologist. Certain philosophical tenets are useful in mentoring others, such as the teaching tool of “See one…Do one…Teach one”, as it encapsulates the experience of working alongside a mentor, followed by the freedom to learn on one’s own, and ultimately providing the learning experience of mentoring to others. Similarly, the Japanese concept of Shu ha ri, as taught in traditional martial arts and craft apprenticeships (Klens-Bigman 2002a, 2002b; Takamura and Okura 1986; Yokota, Narita, and Hamada 2014). In this analogy, the stages of learning are outlined, with Shu, the first, being one of emulating the mentor, learning the correct outlines and forms of the discipline. Ha, the second, is a period of learning in which the mentee breaks free of the constraints, expanding their knowledge base, finding other mentors, and seeking to understand the discipline beyond rote practice. Ri, the third stage, is where the student has fully internalized the discipline and become capable of leading and mentoring others, bringing a deeper and more profound knowledge to the process. The pinnacle of success in mentorship is for mentees to achieve the Ri stage. Engaging in active and thoughtful mentorship is mutually rewarding for both mentor and mentees.
Within the field of toxicologic pathology, there are many opportunities for mentorship. The basic technical knowledge and abilities need to be honed, both in reading slides, writing reports, managing research, leading projects, and other aspects of the career. As these differ greatly between companies and job types, there will be specific aspects that will need to be emphasized, de-emphasized, or even retrained, depending on the specific employment circumstances. Navigating within a corporate structure, GLP environment, departmental guidelines, research group, or those of corporate sponsors or partners, to name a few, requires another degree of mentorship. Expanding on an individual’s existing abilities and specializing within the field is another opportunity for providing mentorship. Even involvement in professional organizations is a prime opportunity for mentorship and promoting active engagement and networking. Mentorship is best achieved when the rules and expectations of the mentor–mentee relationship are clearly defined up front. For the mentor, it is important to realize that each mentee is a unique individual whom may need a unique approach. An effective mentor should allow the mentee to learn by seeing, through experience, by modeling, and leading him or her through learning opportunities. The mentor should also introduce the mentee to new avenues, new concepts, and new relationships within the profession, enabling the mentee to stand on their own. The mentor must provide the mentee with the tools and resources to excel, encouraging independence and self-reliance. A mentor should encourage the mentee to expand his or her horizons, to find new mentors, and to also pass knowledge on to mentees of their own. As a mentee, there are ways to be more engaged and more productive as well. Mentees should actively listen to and engage with their mentor’s suggestions, although this does not mean slavishly following everything that the mentor tells them. Responsible mentees will independently follow through on the mentor’s guidance, without seeking help from mentors constantly throughout the process. This will improve the mentee’s learning and progress. Realize that each mentor is a unique individual who may need a unique approach but also has something unique to teach. The mentee’s improvement is reliant on his or her own efforts as well as those of the mentor. The interaction and relationship between the mentor and mentee are what creates a successful mentorship and personal growth.
The Basics of Evaluating Safety in Toxicology Studies—Why, How, When, Who, and Where
Dr. Stacey Fossey (AbbVie) provided an overview of the fundamentals of safety assessment in preclinical toxicology studies. Dr. Fossey, whose expertise stems from a career dedicated to the pharmaceutical industry, also discussed various career paths and job opportunities available in toxicologic pathology via shared perspectives collected from colleagues working in the agrochemical industry, biotech companies, the public health sector (government), academic institutions, and CRO or consultant settings. The following narrative is provided by Dr. Fossey as a part of her presentation.
Scientists trained in toxicology and pathology support the study of chemicals to inform human safety in a variety of venues. In fact, the effort to ensure the safety of medicines has been in existence throughout history. In the United States, the Food, Drug, and Cosmetic Act was enacted in 1938 and mandated the preclinical and clinical testing of new medicines. The U.S. Food and Drug Administration (U.S. FDA), as well as international agencies, regulates pharmaceuticals through guidelines that facilitate bringing safe drugs to market. Regulatory documents on diverse types of preclinical studies and compounds such as those from the International Conference on Harmonization (ICH) provide more information on these requirements. In the United States, the Code of Federal Regulations Title 21, Chapter 1, contains requirements governing the development of new drugs to ensure human safety. One aspect of this document, the U.S. FDA’s Animal Rule, dictates that for new drugs where administration to humans in clinical trials to determine efficacy would be unethical (e.g., treatment of conditions due to lethal or debilitating biological, radiological, or chemical substances), approval may be based on animal safety and efficacy studies (https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=314&showFR=1&subpartNode=21:5.0.1.1.4.9, last accessed August 27, 2018). GLP standards cover various aspects of preclinical safety study conduct including personnel responsibilities, test and control article characterization, standard operating procedures (SOPs), study performance, reporting, audit oversight, and archiving of data; more detail on GLPs were provided in the next presentation.
Toxicologists and pathologists support the design, conduct, and interpretation of data on preclinical safety studies along the drug development pipeline. As a compound progresses along this time line, different safety assessments are required prior to administering the drug in people. Early studies include in vitro safety pharmacology and genetic toxicology studies with progression to short duration exploratory in vivo toxicology studies and eventually to studies of longer duration and focus depending on the type of molecule, disease indication, and intended duration of dosing. The Investigational New Drug application is filed with the FDA prior to dosing of a drug in clinical trials. A New Drug Application is submitted to the FDA by a sponsor to gain approval for a drug to be marketed in the United States. These preclinical safety studies are designed to inform risk for a candidate compound. The benefit versus risk for a compound is an important consideration that is informed by factors such as the intended patient population, disease severity and prevalence, type of safety risk and its probability, and alternate treatments.
The second half of the presentation provided more details on the personnel and venues where preclinical safety studies are conducted. These studies are typically supported by a large team of scientists, technical staff, and facilities management personnel. Pathologists have roles in the identification, interpretation, and communication of findings in toxicology studies as well as delving into exploring mechanisms of toxicity, animal model characterization, and development of new investigative modalities. Toxicologists are integral members of this safety team through their roles as study directors, study monitors, as well as providing expertise on regulatory filing strategy. These scientists provide this support in a variety of venues, to include pharmaceutical companies, CROs, biotech, chemical industry, government, academia (veterinary/medical schools), and various consulting roles. Activities common to many of these venues are the conduct, analysis, and reporting of preclinical toxicology studies. Depending on the path that appeals to an individual and the point that he or she is at in his or her career, there can also be opportunities to engage with drug discovery and development project teams (pharmaceutical industry, biotech, academia), interact with a wide variety of sponsors with different compound types (CROs, academia), step outside of your comfort zone into disciplines other than pathology or toxicology (biotech), and engage in more independent pursuits such as building and growing one’s own business (consulting).
Finally, it was emphasized that individuals should consider broadening their knowledge through on the job training opportunities, continuing education, networking, and, most importantly, through mentorship via identification of mentors for your own career development and by serving as a mentor to others.
Logistics of Reading GLP Studies
Dr. Bevin Zimmerman (Charles River Laboratories) presented the logistics (or “joys”) of reading toxicologic pathology studies that abide by GLP guidelines. Her talk provided helpful advice to trainees and early-career toxicologic pathologists on how to approach reading a GLP study, the importance of communicating with one’s scientific colleagues throughout the process and general “dos and don’ts” to consider when writing the pathology narrative. The following narrative is provided by Dr. Zimmerman as a part of her presentation.
GLPs are guiding principles for the conduct of nonclinical safety testing to ensure the integrity and reproducibility of a study. GLP studies generally take place after a test article has been developed in a discovery setting and its desirable characteristics have been optimized and its liabilities limited. In vitro assays, proof of concept animal models, and dose range finding studies often precede the GLP pathology studies. The size and scope of the GLP studies are based on the intended indication and are outlined in the guidance documents set forth by ICH (http://www.ich.org/products/guidelines.html, last accessed July 17, 2018).
Histopathology of GLP studies involves far more than reading glass slides. The pathologist must be familiar with the protocol, amendments, and all applicable data. Time lines are critical in GLP work and require the pathologist to be cognizant of their schedule and the amount of time it will take to complete the study. Once reading the slides, choosing diagnostic terminology that is descriptive rather than interpretive removes some of the possibility for misinterpretation of the findings. Consistency is a key in GLP studies. This is true within the study, between studies with the same test article, and with historical control databases. The diagnoses chosen by the pathologist will stay with the test article, so consideration about the actual meaning and the intended meaning of the diagnostic terminology needs to be weighed heavily. International Harmonization of Nomenclature and Diagnostic Criteria (INHAND; https://www.toxpath.org/inhand.asp, last accessed July 17, 2018) and Global Open Registry Nomenclature Information System (https://www.goreni.org/, last accessed July 17, 2018) are valuable tools for this. The use of INHAND terminology has become even more important with the implementation of Standardization for Exchange of Nonclinical Data, an initiative for submission of data to the FDA that utilizes INHAND terminology as the basis for submission of microscopic changes (https://www.cdisc.org/standards/foundational/send, last accessed August 27, 2018). After all the slides have been read, the toxicologic pathologist crafts a narrative. GLP report requirements are outlined in Morton et al (2006). A clear concise narrative that allows the reader (often not a pathologist) to understand which findings are test article related, which findings are critical, and why the pathologist has developed the conclusions that he or she has stated is required. The pathology narrative is generally not a place for speculation or hypothesis. It is, however, the place to highlight critical findings and to define or refine the lesions and grading used. It’s the place to draw correlates and reference literature that supports the pathologist’s overall interpretations. Revisions to the narrative as it travels through the organization are not uncommon, but it is ultimately the pathologist’s signature on the report. Once completed, the narrative and data are available for peer review. Peer review is currently not a GLP requirement. Best practices for peer review are available on the STP website (https://www.toxpath.org/best-practices.asp, last accessed July 17, 2018) and are also guided by the institutional SOPs. The peer review is an opportunity for the study pathologist to confer with a peer review pathologist, to refine diagnoses, and to prevent diagnostic drift. The peer review pathologist will issue a memo that consensus has been reached. Generally, this is done after the final pathology report has been signed by the study pathologist.
As with any professional setting, communication is critical in the success of the GLP pathologist. Whether working for an agrochemical or pharmaceutical company, or a CRO, the pathologist’s results are critical to the progress of the test article. Clarity and direct proactive communication with the study director and the team are essential for success. The study director, the single point of control for the study, may rely heavily on the study pathologist to pull together the data and help to assign the no-observed-adverse-effect level (NOAEL) for the study. The last critical component of a successful GLP pathology study is the interaction with the quality assurance (QA) group. The QA group is an independent group charged with ensuring the overall integrity of the study. Minimally, they check that the protocol and all SOPs were followed. Many auditors are not comfortable with pathology data as it is not machine generated. The raw data for pathology are the signed final pathology report and that can be a challenging concept for the newer auditor. The role of the GLP pathologist involves responding to the QA questions and audits. However, QA is not a scientific check and accepting all the audits without question is unnecessary.
Anatomic and Clinical Pathology Data Visualization, Interpretation, and Presentation
Dr. Lila Ramaiah (Bristol-Myers Squibb), a clinical pathologist in the pharmaceutical industry, addressed the crucial topic of how pathologists interpret and communicate scientific data in toxicology studies, emphasizing the differences between interpreting and reporting findings in addition to providing an overview of the available software tools pathologists can use to organize, visualize, and present study results to their scientific colleagues and the community at large. Dr. Ramaiah stressed the importance of effective communication in science and briefly outlined the types of communication skills commonly used in toxicologic pathology including active listening, brevity, clarity, accuracy, and coherency. The following narrative is provided by Dr. Ramaiah as a part of her presentation.
The fundamental work of the toxicologic (anatomic and clinical) pathologist includes pathology data generation, synthesis, interpretation, and communication. Interpretation involves determination of test article relationship and potential adversity, often by assessing trends among data points to evaluate dose effects, consistency over time or between animals, and pathophysiologically relevant patterns of change. For example, results in test article–treated animals may be compared to those prior to treatment or in vehicle controls. Interpretations and conclusions are communicated in the pathologist contributor report. However, excelling as a toxicologic pathologist and deriving elevated levels of job satisfaction require more than an accurate report. A highly effective pathologist has a holistic understanding of all study data, effectively conveys meaning, and works with stakeholders to achieve common goals. He or she may use data visualization tools to more efficiently interrogate and synthesize large data sets and communicate findings. The toxicologic pathologist must also manage challenging situations using soft skills such as teamwork, adaptability, resourcefulness, communication, perspective taking, and willingness to offer solutions.
When considering how to best give data meaning and convey it to others, it is helpful to consider the broader goal. In nonclinical safety assessment, the overarching goal is to inform decision-making for clinical trials. Specifically, investigators communicate hazard associated with an administered dose by identifying target organs and a NOAEL. Note that the adverse dose is being identified for a given study. Identification of adverse and nonadverse findings may be part of the process, but it is not the goal (Ramaiah et al. 2017). For a thorough and holistic evaluation of all findings (including their cause and consequence) in each animal at a given dose, one must often interrogate the data by asking specific questions. “What else happened in this animal?” “Did it have clinical signs or changes in food consumption or body weight?” “When did they first occur, peak, and stop?” “Did other animals have a similar spectrum of findings?” “How many in each group and which ones?” “How did control animals compare?” “Did the most severely affected animals have clinical signs or other correlates?” “Are these low incidence findings all occurring in the same animal or different ones?” “Did that animal have a background lesion?” The answers to such questions are generally found in the raw data, that is, extremely long, complex, and cumbersome tables/appendices in Microsoft Word® or Adobe® pdf that are designed to adhere to regulatory agency submission requirements but are not amenable to efficient interrogation of study data. As such, a thorough interrogation of individual animal study data can be extremely laborious and time-consuming. Although toxicologic pathologists adapt to the cumbersome tables and appendices, it becomes all too easy to simply review summary data and statistics to identify drug effects. This process risks missing vital details as well as turning one’s report into a “data dump” with little conveyed meaning or value to stakeholders.
Data visualization tools offer the ability to rapidly interrogate and analyze relationships in large data sets, to find meaning in the data, and to convey that meaning to stakeholders using images. With such tools, pathologists can dig deep into the individual animal detail as well as zoom out to see the big picture and effectively tell the story of what happened on a study. They can answer questions in real time instead of having go back to search through the data. They can efficiently interpret the data, investigate correlations, evaluate patterns, draft reports, communicate findings in presentations, and revisit data as additional information comes in or as new questions are asked. Due to the increasing accessibility of data in such tools, pathologists who do not use visualization tools risk being less efficient than other pathologists who do use them or risk missing details that are later caught by nonpathologists or regulatory agencies that also use them.
A thorough review of currently available data visualization tools has been recently published (Troth et al. 2018). For interpretation of single studies prior to reporting stages, Microsoft Excel® is most commonly used because it is easily obtainable, simple to learn, and has some unique capabilities (e.g., pivot charts). Excel can be used to graph, filter, and sort both quantitative data and qualitative (or semiquantitative) data. Its main disadvantages are its limited data capacity and limited controls for data integrity.
Microsoft Excel filtering and sorting can be used to manually create a heat map of changes relative to controls (Figure 1). A full panel of individual animal data can fit on a single page, allowing the reader to rapidly identify trends (incidence, magnitude, and dose relationship) for a given parameter and patterns among parameters for a given animal or dose. A full clinical pathology data set can thus fit on 6 pages. It can be given to stakeholders to facilitate their data review and can be used to quickly answer questions at meetings or months after the report was written. The filter and sorting function can also be used to rapidly identify the most severely affected animals or to interrogate qualitative data in a variety of ways (Figure 2).
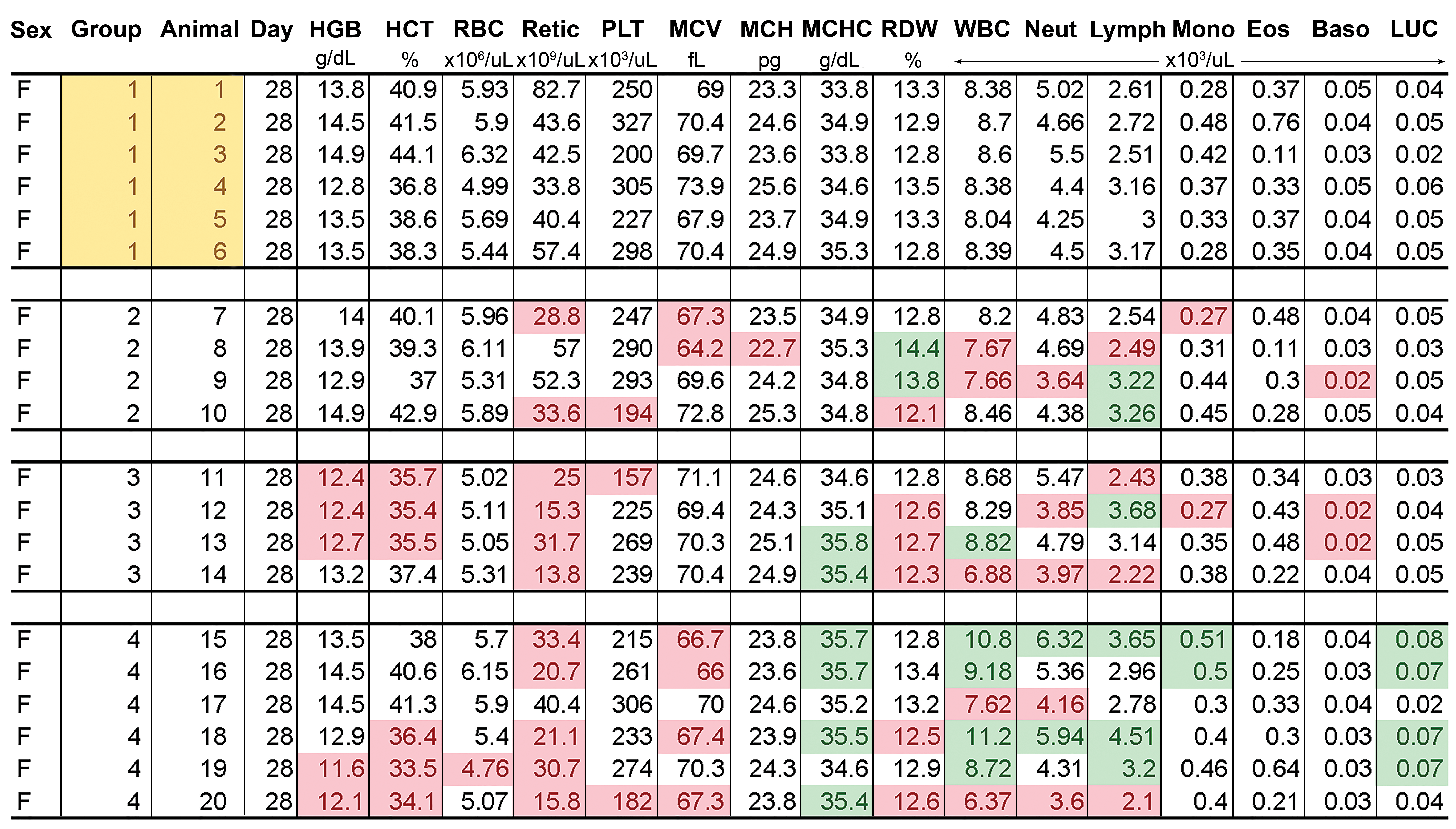
Use of Microsoft Excel® to produce a heat map of quantitative data, using sorting and formatting. Decreased reticulocytes are immediately apparent at the mid and high doses, which are variably associated with minimally decreased red cell mass. Trends across doses, animals, and parameters are readily appreciable.
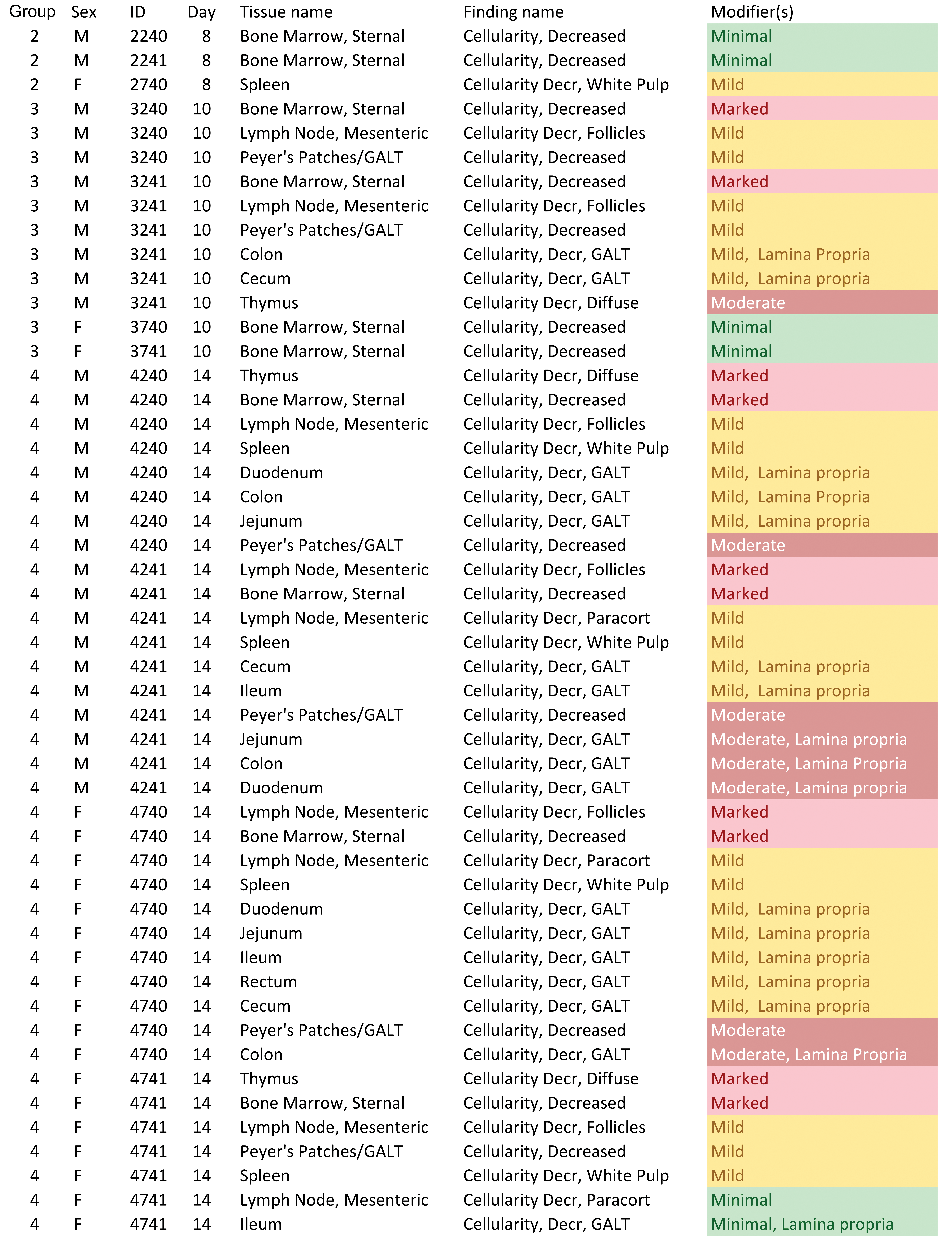
Use of Microsoft Excel® to produce a heat map of qualitative data, using filtering, sorting, and formatting. In this case, the finding name was filtered for “decreased cellularity” to enable the observer to immediately appreciate the wide variety of affected tissues, affected doses, incidence, individual affected animal numbers, and severity.
Microsoft Excel pivot charts can be used to easily create graphical representations of quantitative and qualitative (or semiquantitative) data, visualizing them in different groupings by dragging columns to different sections of a chart. For quantitative data, line pivot charts (with markers) may be used to display individual animal data (including longitudinal data; Figure 3). This allows rapid visualization of individual animal changes in the context of overall biological variability, group trends, changes over time, and procedure-related changes. Changes relative to pretest and concurrent controls may be interpreted simultaneously.
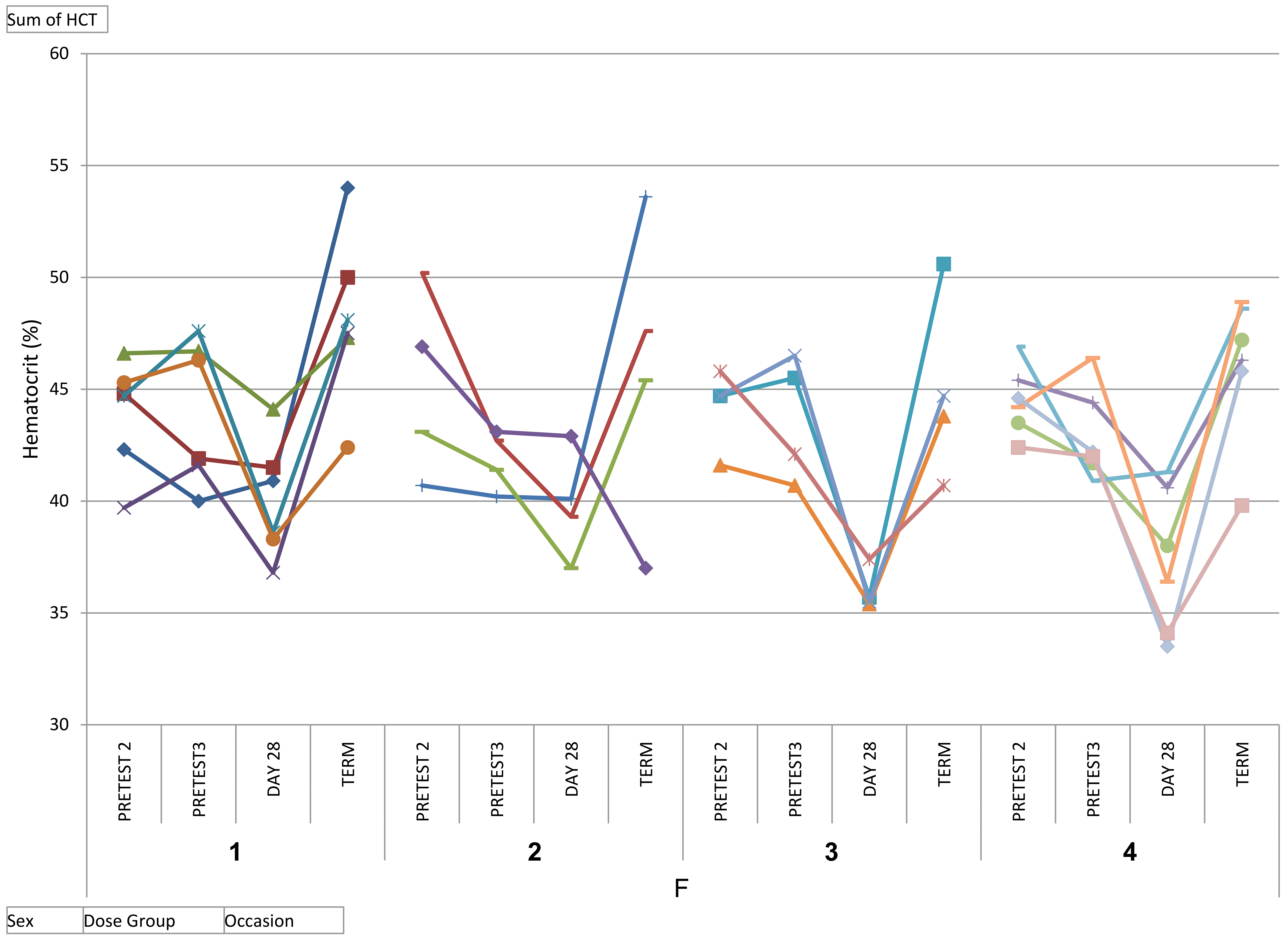
Use of Microsoft Excel® to produce a line Pivot Chart (with markers) of quantitative longitudinal data (hematocrit data from Figure 1). Y-axis: hematocrit value (%)(top left: pivot charts refer to the sum of HCT). X-axis: data stratification scheme, first by Sex (F), then by Dose Group (1-4), and then by Occasion (Pretest 2, Pretest 3, Day 28 and Term), to allow visualization of individual animal and group trends. When compared to Pretest 2, there is a clear procedure-related decrease in hematocrit in all groups (including controls), putting into question the relevance of hematocrit decreases in Group 4.
For qualitative (or semiquantitative) data, stacked column pivot charts may be used to display the incidence of individual animal findings (Figure 4). This allows rapid identification of affected animals across multiple findings or tissues (not usually possible on incidence tables). The filter and sort function within pivot charts allows for multiple ways to interrogate and visualize the data according to desired criteria. For qualitative longitudinal data such as clinical observations, the charts can display changes over the course of the study.
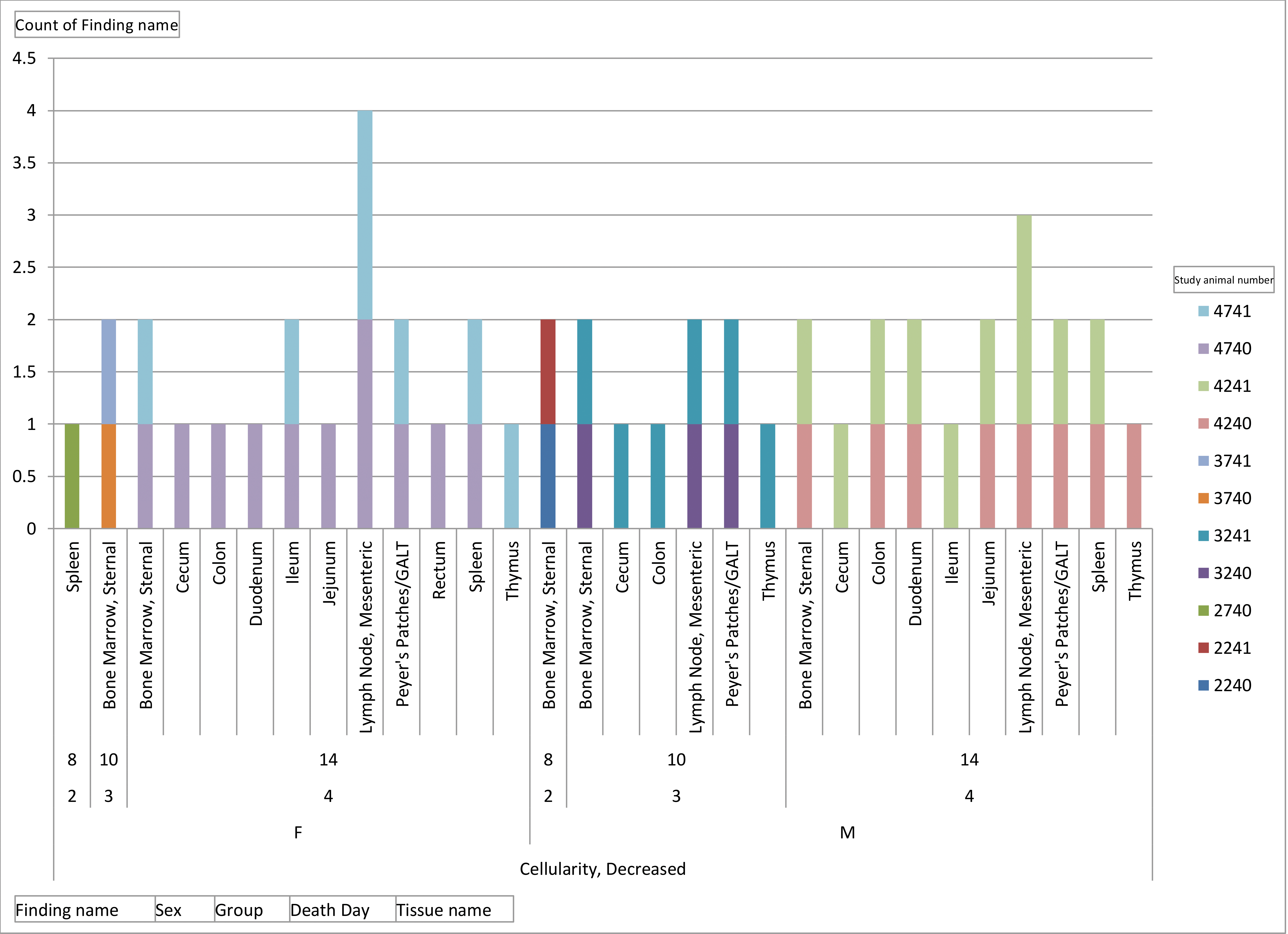
Use of Microsoft Excel® to produce a stacked column Pivot Chart of qualitative data (microscopic). Y-axis: incidence of finding (top left: pivot charts refer to the count of the finding name). X-axis: data stratification scheme, first by Finding Name (filtered for Cellularity, Decreased), then by Sex (M or F), then by Dose Group (1-4; Group 1 does not show because no Group 1 animals had decreased cellularity), then by Death Day (8, 10, or 14), and then by Tissue Name. The chart is filtered for decreased cellularity, demonstrating that decreased cellularity was present in males (M) and females (F) in Groups 2, 3, and 4 (animals that died on days 8, 10, and 14), in the tissues indicated. The incidence of the finding (regardless of tissue) can be determined using the list of affected animals to the right of the chart. The filter function of pivot tables can be used to easily and rapidly interrogate specific tissues or findings as desired.
Data visualization tools are rapidly becoming essential for efficient interrogation and synthesis of increasingly large and complex data sets. However, users must be aware of their limitations and must avoid overconfidence in digital data. Not all relevant information (i.e., meaningful metadata and paper raw data) is transferred to data warehouses. Preanalytical variability should always be considered (sample comments, alternate collection site used, difficult phlebotomy, storage conditions, new necropsy technician, etc.). When necessary, pathologists should go to relevant paper records and deviations for additional context. Importantly, to truly get to the bottom of unusual data, pathologists should talk to the people who conducted the study, collected/analyzed the specimens, and made the observations.
Thorough data integration and interpretation are immaterial if they are not communicated effectively to stakeholders. Effective communication requires an understanding of the reader’s perspective and needs. Based on this understanding, the author focuses on telling stakeholders what happened on the study in a way that will inform decision-making for clinical trials. The audience ideally receives the message as intended—yet the goal is not to be heard. The author may also seek to inform, teach, clarify, or persuade—yet the goal is not to be right. For written reports, consistency, formatting, convention, and other rules (including GLPs) indicate proper processes and ways to achieve the goal, yet they are not themselves the goal. To be effective and facilitate decision-making, the narrative report should be clear, concise (brief), coherent, and accurate. This makes it easy for the audience to understand and improves chances they will agree with the stated conclusions. Excellent overviews of toxicology data reporting are available in textbook format (Wells 1996) and from the Organization for Economic Cooperation and Development online (https://www.oecd.org/about/publishing/OECD-Style-Guide-Third-Edition.pdf, last accessed July 17, 2018).
The core elements of the written contributor report include the summary/abstract, results, and discussion. An introduction, materials and methods, conclusion, and references may also be included. Text tables (fold or % difference from controls or incidence) are extremely useful because they offer the reader a way to rapidly visualize the magnitude/incidence of test article–related findings at each dose/timepoint rather than reading through long and complicated text or going to long and cumbersome data tables.
The results section may be combined with the discussion or may be separate. The results section describes test article–related findings, and the discussion provides an interpretation of the results, including correlations, causes, and adversity, as appropriate (Ramaiah et al. 2017). Style should be in plain, business English, using the active voice and simple past tense. Readability should be maximized, even at the cost of creative writing and grammatical rigor. An author should not write over the head of the reader and must avoid using distracting or complex words, jargon, literary, or foreign phrases, an everyday English equivalent should instead be used, if there is one. If English is the author’s second language, he or she should formulate a thought in English first rather than translating from his or her native language. Reading a report should not be hard mental or mathematical work.
Clarity and efficiency can be further enhanced by using the Bottom Line Up Front approach. Rather than reading like a suspense novel in which one pores through a detailed narrative to finally discover the meaning at the end, the content should be structured to place conclusions at the beginning. The narrative should discuss the most important points first; these should comprise the largest proportion of the text. The reader may then read further if more detail is desired or to learn of secondary or incidental changes (should be brief). Additional information should only be added when it helps clarify study results.
Brevity speeds decision-making by minimizing reading time. The message should be distilled into few well-chosen words (<25 words/sentence). The summary/abstract should be short (<½ page), and the conclusion should be even shorter (<2 sentences). The author should delete words when possible and choose short words over long ones. For example, “liver weights were higher” is preferred over “higher liver weights were observed,” “was higher” instead of “was found to be higher,” “can” instead of “has the ability to,” “X was higher than Y (p < .05)” instead of “X was significantly higher than Y (p < .05), and “although” instead of “despite the fact that.”
Coherent writing is logical, unified, and consistent. It is easy to follow and easy to agree with. To maximize coherency, the narrative should stay on topic and avoid tangents, inconsistency, and excessive detail. If possible, it is recommended that one sentence be used for each result or idea. It is important to use the right word to convey the intended meaning. The author must refrain from contradiction and avoid terms that may have multiple meanings (otherwise define them). For example, “decreased red cell mass” should be used instead of “anemia,” “marked increase” instead of “important increase,” “statistically significant” instead of “significant,” “not adverse” instead of “not toxicologically relevant,” and “not test article related” instead of “not biologically relevant.”
Accurate and comprehensive reports allow the reader to independently evaluate findings without referring to the original data; they save time and build credibility. Accurate interpretations are consistent with and supported by the study raw data; they avoid speculation by explicitly describing relevant supportive study correlates. Because a report is rarely perfect at its first draft, it is important for the author to wordsmith his or her work before others. The author should verify that the most appropriate word or phrase is being used to convey the intended meaning. Conclusions should be clear. Vague, ambiguous, or misleading word usage must be avoided. The author should revisit/revise/reorder the important points as additional context is added or additional study results surface and must not rely on spell-check. The report should also be quality controlled by someone else (often management) to avoid relying on QA to correct mistakes and inconsistencies.
Once written and internally reviewed, toxicologic pathology reports are reviewed by a wide variety of clients and stakeholders, each with their own needs, knowledge, experiences, perspectives, and goals. The volume and breadth of comments, questions, and demands from stakeholders can be overwhelming if not frustrating, particularly when they are communicated using a static and impersonal format. Well-intentioned questions may appear to question the pathologist’s knowledge or seem so basic (from the perspective of the pathologist) that a response feels unnecessary or tedious. The toxicologic pathologist may be faced with questions that are difficult, naive, tedious, difficult, ill-phrased, or put after inadequate self-criticism, but in most cases, questions are an indication that clarification is needed.
When faced with tough questions, it helps to put aside convention, clarify assumptions and expectations, and consider whether goals are aligned. Conflict may arise when one person’s actions make it harder for another person to achieve his or her goal. Therefore, it is important to refocus on the common goal (i.e., informing decision-making for clinical trials) and determine how an individual’s interpretations and conclusions impact the team’s ability to make decisions. Remember that in many cases, pathologist interpretations are subjective, even if well-informed, judgment calls. Adaptability may be needed in some cases, acknowledging that one may not know the answer. Pathologists should take the necessary time to investigate further, revisit, clarify, or justify interpretations and allow for “wiggle room” to find common ground when appropriate. Adaptability should also be appropriately balanced by having an opinion, standing firm, and being prepared to clarify and support one’s position using all relevant information. One should avoid defensive statements or statements that can’t be confirmed. Pathologists should confirm and reconfirm understanding and allow communication to be tracked. Active listening is essential for effective communication and conflict management. This requires being open to, respecting, and understanding the perspective of others (seeing through their lens). Active listeners are mindful, pay attention, and watch micromessaging; they avoid distractions, think before speaking, and do not anticipate, answer too quickly, or interrupt.
Using the right medium of communication for the task at hand improves effectiveness. Written communication can be carefully crafted and revised to achieve optimal clarity and accuracy. It is best suited for formal communication and documentation purposes due to its static (asynchronous) nature and lack of interpersonal context. Oral communication conveys a wealth of context and additional information (body language, facial expression, eye contact, voice tone, and volume) and is faster because it allows for instant feedback (synchronous). It is best suited for building consensus, brainstorming, clarifying, or making important decisions. However, oral communication is prone to emotional responses that can’t be reversed once uttered. It reflects what is in one’s head in the heat of the moment, requires immediate mental work, and is easier to forget. E-mail and instant messaging can be documented like written communication but are faster and more synchronous. They should be used cautiously since they lack interpersonal context and are prone to emotional responses. Oral presentations should be clear, concise, and coherent and should use visualizations instead of words whenever possible. Avoid using more than 5 bullets per slide and 5 words per bullet point. Although the presentation need not be memorized, it should be well prepared and rehearsed for timing, tone, and flow. Overreliance on or reading through Microsoft PowerPoint® slides must be avoided. An oral presentation should have humanity and energy. The speaker should strive to consider the audience’s needs and perspective, to connect with the audience, and to express passion for the topic.
The role of the toxicologic pathologist is evolving as the world transitions from an industrial economy founded in factories and manual laborers to a knowledge economy founded on robots, big data, and critical thinkers. A time in which robots can rapidly scan slides and analyze images and body fluids and sophisticated analytical software can interpret data and write reports is approaching. The value of the toxicologic pathologist will increasingly be based on his or her ability to efficiently and critically integrate large data sets using specific expertise, to derive and convey meaning to others using formal written as well as informal communication, and to drive decision-making in the context of broader program goals. It is an exciting time to be a toxicologic pathologist!
Toxicologic Pathology Knowledge Tools
Dr. Ken Schafer (Vet Path Services, Inc.) reviewed some of the helpful resources available to toxicologic pathologists, expressing the need to often look beyond the internet to find the answers. His presentation highlighted the essential textbooks and references that every toxicologic pathologist should use and be aware of at any stage of their career. The following narrative is provided by Dr. Schafer as a part of his presentation.
There are an amazing number of very good and useful references available to the practicing toxicologic pathologist (Online Supplemental Tables 1–4). The discussion here relates most specifically to those of use to the toxicologic pathologist doing routine, GLP drug development work. However, many of the principles and resources also apply to a pathologist working in the discovery realm or elsewhere. A lot of what a pathologist does relies on first principles, in which everyone should be grounded. These include an understanding of regulatory guidelines and requirements as they apply to pathology. Certainly, a discovery pathologist’s output is generally exempt from such guidelines, but the principles apply and can be useful in general “Good Research Practices” and explaining to nonpathologist scientists why pathologists do what they do. Therefore, a toxicologic pathologist should be able to answer questions such as: “What is my raw data?” “Are images that I capture raw data?” “Are slides raw data?” and “What is the role of the study director?” The U.S. Code of Federal Regulations and other global regulatory documents address some of these questions and should be something with which a pathologist is at least passingly familiar (https://www.ecfr.gov/cgi-bin/text-idx?SID=3ee286332416f26a91d9e6d786a604ab&mc=true&tpl=/ecfrbrowse/Title21/21tab_02.tpl, last accessed August 27, 2018). As an adjunct to this knowledge base is a growing list of STP Best Practices, Points to Consider, and Recommendations manuscripts. These documents expand on the regulations and help guide new (and experienced) pathologists on the current art and science of toxicologic pathology. The aforementioned resources are the foundational “how to” documents of toxicologic pathology (Online Supplemental Table 1). There are also many important documents that can be accessed for the science of toxicologic pathology as well. One of the more recent being the INHAND effort that is developing guidance documents for standardized nomenclature. The forbearer to this effort was the Standardized System of Nomenclature and Diagnostic Criteria manuscripts (available on the STP website and worth a look) but which were not completed for all organ systems and needed some updating. There are also some valuable manuscripts, notably by the European Society of Toxicologic Pathology, that have proposed standard tissue trimming paradigms that are available for consideration should one need to develop necropsy and tissue trimming SOPs. In addition, the official journal of the STP, Toxicologic Pathology, and other similar journals are great resources of published literature on toxicologic pathology (Online Supplemental Table 2).
When it comes to books, it is good to be a bibliophile. When one comes across an informative book, is it worthwhile to own rather than relying on the company library as a resource? Jobs come and go, and in this dynamic world, when people leave a job, they can’t take the library with them, unless they own it. Some books go out of print and can be difficult to find. When the day comes to relocate or change jobs, it can be nice to take that reference library along. There are several books that can be helpful and useful to any toxicologic pathologist (Online Supplemental Table 3). These include generalist books that cover various organ systems and may also cover some of the topics of the “how to” of toxicologic pathology. There is also a growing library of recorded webinars that are accessible to STP members that can be launched from the STP website. Many of these were in partnerships with other pathology and toxicology organizations and can have some very useful information. Also, toxicologic pathologists should watch for any new webinars as they are advertised and broadcast. In addition to the links on the STP website, pathologists are encouraged to surf the websites of sister organizations, as they often have different links and different emphases in the resources that they consider important to list (Online Supplemental Table 4). Finally, a pathologist should consider their contacts and not be afraid to call the study director for more information. For those employed by a CRO, it is acceptable (and maybe should be expected?) for a pathologist to reach out to the sponsor to better understand what the drug is supposed to do and what they know about other toxicology studies. The sponsor may know a lot more about the toxicologic pathology attributes of the test article than anyone. In addition, fellow pathologists are some of the best toxicologic pathology resources out there. It is OK for one to lean on his or her pathology colleagues now and then.
In summary, successful careers in toxicologic pathology begin with an effective mentor–mentee relationship that can help bridge the knowledge gap in one’s pathology training. Making the decision to pursue a career in toxicologic pathology endows the ability to contribute to advancing science in a variety of fields and applications including the discovery and development of drugs and medical devices (pharmaceutical and biotech industries), agrochemical industry, public health and regulatory agencies (government), educational institutions (academia such as veterinary and medical schools), or any combination thereof (CROs and consultants). The toxicologic pathologist is often responsible for interpreting and reporting scientific findings in accordance with GLP guidelines; therefore, it is important to approach GLP studies in a consistent and methodical approach that encourages open lines of communication with all team members involved in the study including the study director (usually a toxicologist or biologist), the sponsor, administrative, technical and support staff, and, of course, the QA group. It is important that toxicologic pathologists understand how to communicate science effectively. Although listening is perhaps the most essential component of communication, there are many software tools currently available that can assist pathologists with organizing, visualizing, and presenting scientific data in a clear and concise manner. In addition, as with many professions, the learning continues throughout the toxicologic pathologist’s career. There are many resources available, beyond the Internet, that pathologists can use to enhance their knowledge and improve their efficacy in this highly rewarding and diverse field.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318805716 - Practical Strategies for Navigating Toxicologic Pathology in One’s Early Career…and Beyond!
Supplemental Material, DS1_TPX_10.1177_0192623318805716 for Practical Strategies for Navigating Toxicologic Pathology in One’s Early Career…and Beyond! by Kathleen Heinz-Taheny, Kendall Frazier, Shashi Ramaiah, Vinicius Carreira, Gopinath Palanisamy, Erin Quist, Keith Nelson, Stacey Fossey, Bevin Zimmerman, Lila Ramaiah, and Kenneth A. Schafer in Toxicologic Pathology
Footnotes
Author Contributions
All authors (VC, GP, EQ, KN, SF, BZ, LR, KS) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
