Abstract
Early deaths of young or juvenile animals (before sexual maturation is achieved) in routine regulatory safety studies present pathologists and toxicologists with the challenge of interpreting findings in the male reproductive tract. Additionally, the advent of toxicity testing regulations has resulted in a growing need for the use of juvenile animals in toxicology studies. Here, we present the reproductive toxicity findings from a 13-week inhalation toxicity study with ortho-phthalaldehyde (OPA) in male rats and mice as a case example for working through this challenging task. In this study with OPA, survival was significantly reduced in the two highest exposure concentrations of OPA tested. Early deaths and histopathological lesions in the testes and epididymides were generally also limited to these two highest exposure groups. Therefore, there was concern that peripubertal morphological features could be a confounding factor for the histopathological evaluation of exposure-related testicular and epididymal findings. Although it can be difficult to differentiate exposure-related effects from the normal morphological features defining peripubertal changes in the testes and epididymides in animals that die early in a toxicity study, the use of age-matched controls in this case study with OPA provided a reference and aided in the differentiation of these effects.
Pathologists and toxicologists are increasingly confronted with the challenging task of distinguishing potential exposure-related histologic lesions from normal maturation changes associated with puberty in juvenile animals (Remick et al. 2015). There has been an increased need for toxicity safety assessments in juvenile animals, as a result of new requirements for the safe use of pharmaceuticals in pediatric patients, stemming from the Best Pharmaceuticals for Children Act and the Pediatric Research Equity Act passed by the U.S. Federal Drug Administration (USFDA) in 2002 and 2003, respectively. The importance of juvenile animal studies for safety testing in pharmaceutical development, and the need for case-by-case study design, is also emphasized in the regulatory guidances from both the European Medicines Agency (EMA) and the USFDA (FDA 2006; EMA 2008). Additionally, the Environmental Protection Agency (EPA) Endocrine Disrupting Screening Program uses juvenile male rats in the evaluation of pubertal development and thyroid function. In these studies, the male rats are exposed from postnatal day (PND) 23 through 53 and euthanized at the end of the peripubertal window (EPA 2011). Routine regulatory safety studies often place rats on study that are 6 to 8 weeks of age (PND 42–56), which coincides with the peripubertal period in these animals (Lanning et al. 2002). Early deaths of the animals in these routine studies before sexual maturation is achieved can lead to difficulty in interpreting findings when concurrent age-matched controls are typically unavailable. This is particularly challenging in male rats, where sexual maturation occurs later than male mice.
Complexities in differentiating morphological effects stem from similarities between some common manifestations of toxicity and the normal spectrum of peripubertal changes of the testis in sexually immature animals. Toxicant exposures that affect the testis can result in the loss of one or more generations of germ cells or decreased numbers of round or elongating spermatids, which can be specific to germ cell type or seminiferous tubule stage. For example, toxicant-induced reduced testosterone levels can result in a characteristic increased level of apoptosis in round spermatids and pachytene spermatocytes, as well as progressive loss of the elongating spermatids; these lesions are present in specific spermatogenesis stages that are dependent on testosterone (stages VII and VIII; Creasy 2001). Conversely, low numbers of elongating spermatids are a normal feature in the peripubertal rat testis. Additionally, there are periods of apoptosis of germ cells in the developing testis, such as during closure of the blood–testis barrier (∼PND 15–20; Morales, Mohamed, and Cavicchia 2007) and during the first wave of spermatogenesis, which generally occurs over a period of three weeks (∼PND 10–30 in the rodent; Yan et al. 2000; Rodriguez et al. 1997). The initial inefficiency of spermatogenesis in a peripubertal male rat, where there is a greater incidence of abnormal and degenerating cells in the testis, can easily be mistaken as a toxicological response if the testis evaluation in peripubertal animals is being done in comparison to more mature controls (Russell, Alger, and Nequin 1987).
Rodent toxicity studies are often initiated prior to sexual maturity. While male mice are considered sexually mature earlier, at >7 weeks of age (PND 49), male rats are considered sexually mature later, at >9 weeks of age (PND 63; Lanning et al. 2002). In male rats, the peripubertal stage ranges from PND 33 to 55 (Picut and Remick 2017; Picut et al. 2015). Puberty in male rats is a midpoint of this period, as defined microscopically by the appearance of mature step 19 spermatids at the lumen of stage VII seminiferous tubules around PND 46 (with preputial separation occurring at approximately PND 43; Picut et al. 2015; Whitney 2012; Marty, Crissman, and Carney 2001). At this time, all stages of tubules should be present, but the seminiferous tubules will still vary from those of mature rats. Lower cell numbers, fewer elongating spermatids, and smaller tubular diameters are still typically present in the peripubertal testis as compared to the adult rat (Picut et al. 2015). Spermatozoa reach the epididymis around PND 52 in the rat (Picut et al. 2015; Campion et al. 2013). However, there is continued and substantial growth in the sizes of the testis and epididymis and increased numbers and density of elongating spermatids in the testis and sperm in the epididymis in the late pubertal period, PND 56 to 70, until establishment of the weight and morphological features that are typical of a sexually mature male rat around PND 70 (10 weeks; Campion et al. 2013; Picut et al. 2015). For histologic changes during testis development in rats, the reader is referred to Picut and Remick (2017), Picut et al. (2015), and Whitney (2012). Challenges in examination of the testes and epididymides in sexually immature animals include distinguishing test article–related effects from normal age-appropriate physiological changes, as well as the lack of concurrent age-matched controls in the case of unscheduled deaths.
These challenges were highlighted during the review of the pathology data from the 13-week inhalation toxicity studies with ortho-phthalaldehyde (OPA) in Harlan Sprague-Dawley (SD) rats and B6C3F1/N mice (Catlin et al. 2017; National Toxicology Program [NTP] 2018). OPA is primarily used as a chemical sterilization agent for heat-sensitive medical and dental equipment including endoscopes and thermometers (Rutala and Weber 2001). In these studies, inhalation exposure to OPA resulted in a spectrum of significant respiratory tract lesions and early deaths in many of the animals in the top two exposure concentrations within the first two weeks of the studies. In the male rats, there were changes in reproductive organ weights and sperm parameters in the lower exposure concentrations that had no histopathologic correlates, whereas at the higher exposure concentrations, where morbidity and mortality were observed, there were histopathologic alterations in the testis and epididymis. The differentiation of the toxicological response to OPA from the normal peripubertal state of the OPA exposed rats is presented here as a case study to provide future investigators with an example for working through this complex task.
Materials and Methods
Chemicals
OPA was obtained from MP Biomedicals, LLC (Solon, OH) in a single lot. The chemical was identified as OPA using infrared spectroscopy and Fourier transform infrared and proton nuclear magnetic resonance spectroscopy, and the purity (99.7%, relative to the commercial standard), was determined based on a chromatography profile of major components obtained by gas chromatography (GC) with flame-ionized detection (FID) or mass spectrometry (MS).
Vapor Generation and Monitoring
Generation and monitoring of OPA vapor was performed as described previously (Catlin et al. 2017; NTP 2018). Briefly, OPA vapor was generated from the liquid in a heated vaporization flask. The vapor was purged from the flask with heated nitrogen, mixed with heated dilution air, and then transported to a distribution manifold. Vapor concentration was controlled by the reservoir temperature, nitrogen flow rate, and dilution airflow rate. Due to the high boiling point of OPA, vapor transport lines were heated to prevent condensation. A heated Teflon®-line carried the vapor from the distribution manifold to the chamber inlets, where it was further diluted with conditioned (High Efficiency Particulate Air [HEPA] and charcoal filtered, humidity- and temperature controlled) chamber air to achieve the desired concentration in each Hazelton 2000 exposure chamber (Harford Systems Division of Lab Products, Inc., Aberdeen, MD). The chamber air was monitored with a small particle detector (Model 3022A; TSI Inc., St. Paul, MN) to ensure that OPA vapor, and not aerosol, was produced. Concentrations of OPA in the exposure chambers were monitored by an online GC/FID system. Chamber concentrations were within 5% of the target concentrations throughout the study.
Study Design
Groups of 10 randomly assigned male rats and mice were exposed to OPA via whole-body inhalation at concentrations of 0, 0.44, 0.88, 1.75, 3.5, or 7.0 ppm, 6 hr plus T90 (the time required to reach 90% of the target concentration; 17 min) per day, 5 days per week for 14 weeks. Selection of the highest exposure concentration was based on evaluations of the maximum achievable concentration without aerosolization under normal chamber environmental specifications. The lowest concentration was close to the experimental limit of quantification for the online monitor used in these studies. Although a lower limit of quantification may have been achievable using the online monitor or available offline methods, exposure of animals to lower concentrations was not feasible under the conditions of these studies, due to reactivity of the aldehyde moieties of OPA with amines resulting from the presence of animals.
Animals
Male Sprague-Dawley (Hsd: Sprague Dawley® SD®) rats were obtained from Harlan Laboratories, Inc. (Livermore, CA), and B6C3F1/N mice were obtained from Taconic Biosciences, Inc. (formerly Taconic Farms, Germantown, NY) at 4 to 5 weeks of age (PND 28–35), and were tail tattooed for identification. Following a quarantine period, exposure was initiated when the rats and mice were approximately 6 weeks old (PND 42). The animals were housed individually and provided NTP-2000 irradiated wafer diet (Zeigler Brothers, Inc., Gardners, PA) ad libitum, except during exposure periods. Tap water was available ad libitum. A 12-hr light/dark cycle was maintained in the animal room, with at least 13 room air changes per hour. The animal room environment was maintained at 75°F ± 3°F with relative humidity at 55% ± 15%. Animal care and use were in accordance with the Public Health Service Policy on Humane Care and Use of Animals. All animal studies were conducted in an animal facility accredited by the Association for the AAALAC International. Studies were approved by the Battelle Toxicology Northwest Animal Care and Use Committee and conducted in accordance with all relevant National Institutes of Health and NTP animal care and use policies and applicable federal, state, and local regulations and guidelines. Additionally, studies were conducted according to federal good laboratory practice regulations and specifications defined by the NTP. The animals were monitored twice daily for signs of moribundity and mortality. Body weight and clinical findings were recorded initially, weekly, and at the end of the studies. Following the last day of exposure, animals were euthanized via carbon dioxide asphyxiation and necropsied. Following the study, two additional rats were obtained from Harlan Laboratories, Inc., at 6 weeks of age (PND 42) confirmed by specific birth dates and necropsied on PND 45 to serve as age-matched controls.
Histology
At necropsy, the left cauda epididymis, left epididymis, and left testis from the rats and mice were weighed. Right testes and epididymides for microscopic evaluation were first fixed in modified Davidson’s solution for up to 72 hr and then placed in 10% neutral-buffered formalin. Following fixation, tissues were processed and trimmed, embedded in paraffin, sectioned to a thickness of 4 to 6 μm, and stained with hematoxylin and eosin. The testes were examined using a stage aware evaluation. As a comparator, the testis with epididymis from two untreated 45-day-old rats, obtained from the same vendor after study completion, were also processed and microscopically examined with the same methods. The testes and epididymides underwent additional pathology peer review, and the final diagnoses represent a consensus between the reviewing pathologist and NTP pathologists.
Sperm Count and Motility Assessments
At the end of the study, samples for sperm count and motility assessments were collected from the rats and mice exposed to 0, 0.44, 0.88, or 1.75 ppm. Due to excessive toxicity and early animal removal, these assessments were not made for the two highest exposure groups, 3.5 and 7.0 ppm. The left testis and left epididymis were isolated and weighed. The cauda epididymis was then removed from the corpus epididymis and weighed. Test yolk (rats) or modified Tyrode’s buffer (mice) was applied to slides, and a small incision was made at the distal border of the cauda epididymis. The sperm effluxing from the incision were dispersed in the buffer on the slides, and the numbers of motile and nonmotile spermatozoa were counted under a microscope for five fields per slide by two observers. Following completion of sperm motility assessments, each left cauda epididymis was placed in buffered saline solution. Caudae were finely minced, and the tissue was incubated in the saline solution and then heat fixed at 65°C. Sperm density was then determined microscopically with the aid of a hemocytometer. To quantify spermatogenesis, the testicular spermatid head count was determined by removing the tunica albuginea and homogenizing the left testis in phosphate-buffered saline containing 10% dimethyl sulfoxide. Homogenization-resistant spermatid nuclei were counted with a Hamilton Thorne Bioscience Integrated Visual Optical System following homogenization in buffer using a Brinkman Polytron with generator.
Statistics
For calculation and analysis of lesion incidences, the Fisher exact test (Gart, Chu, and Tarone 1979) was used to determine significance between exposed and chamber control animals. Organ and body weight data, which are typically normally distributed, were analyzed with the parametric multiple comparison procedures of Dunnett (1955) and Williams (1971, 1972). Sperm parameter data, which are typically normally distributed, were analyzed with the nonparametric multiple comparison procedures of Dunn (1964) and Shirley (1977), as modified by Williams (1986). Jonckheere’s (1954) test was used to assess the significance of the exposure-related trends and to determine whether a trend-sensitive test (Williams’s or Shirley’s test) was more appropriate for pairwise comparisons than a test that does not assume a monotonic exposure-related trend (Dunnett’s or Dunn’s test). Implausible values were eliminated prior to statistical analysis, with identification by the outlier test of Dixon and Massey (1957). p Values less than .05 were considered statistically significant.
Results
Survival and Body Weights
Survival was greatly reduced in the rats and mice exposed to 3.5 and 7.0 ppm OPA. All of the male rats and mice exposed to 7.0 ppm OPA died by the end of weeks 2 and 1 (∼PNDs 49 and 46), respectively. In the 3.5 ppm exposure group, four male rats died by day 10 (∼PND 52) of the study, and three male rats died by days 39 to 47 of the study (∼PND 81–89). Five male mice died by the end of week 6 (∼PND 42) of the study. In both rats and mice, exposure-related clinical observations increased in a concentration-dependent manner and included abnormal breathing, sneezing, and thinness. One or more of these findings were present in animals that were found dead or euthanized prior to study completion. However, of the animals that were found dead, there were no adverse clinical findings that warranted their removal from the study. The probable cause of death for both rats and mice was undetermined; however, it was noted at necropsy that the nasal cavity in the mice could not be flushed with formalin, potentially due to suppurative inflammation observed histologically.
Exposure to OPA resulted in exposure-dependent lower final body weights relative to controls in both rats and mice (Figure 1 and Table 1). The final body weights of surviving male rats in the 0.44, 0.88, 1.75, and 3.5 ppm groups were 8%, 14%, 24%, and 46%, respectively, lower than those of controls. Similarly, body weights of surviving exposed male mice in the 0.44, 0.88, 1.75, and 3.5 ppm groups were 17%, 24%, 27%, and 39%, respectively, lower than those of controls.
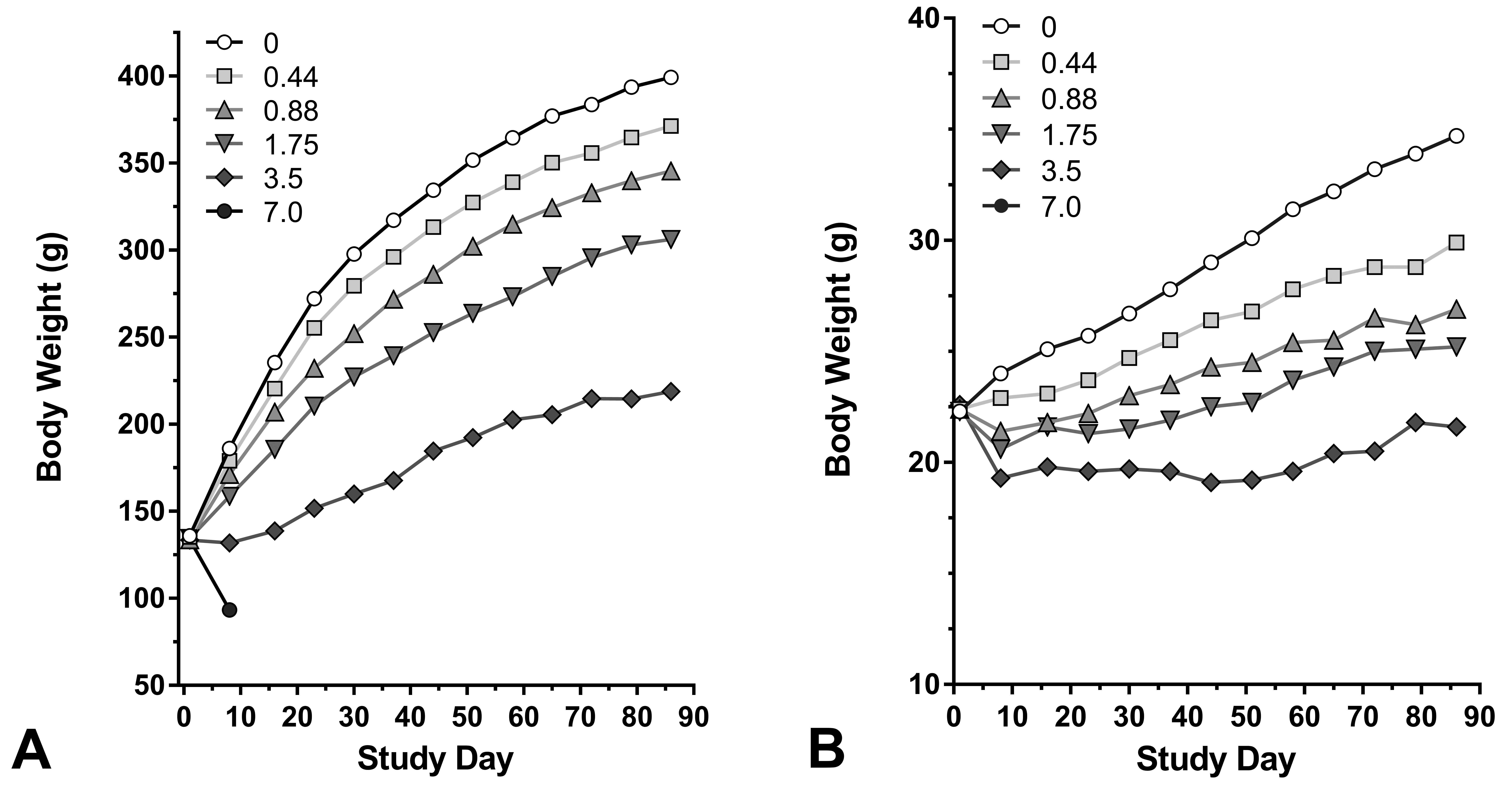
Mean body weight changes in grams (g) for each exposure level (0, 0.44, 0.88, 1.75, 3.5, or 7.0 ppm) for rats (A) and mice (B) exposed to ortho-phthalaldehyde for 3 months.
Reproductive Organ Weights and Sperm Parameters in Male Rats Exposed to Ortho-phthalaldehyde for 3 Months.
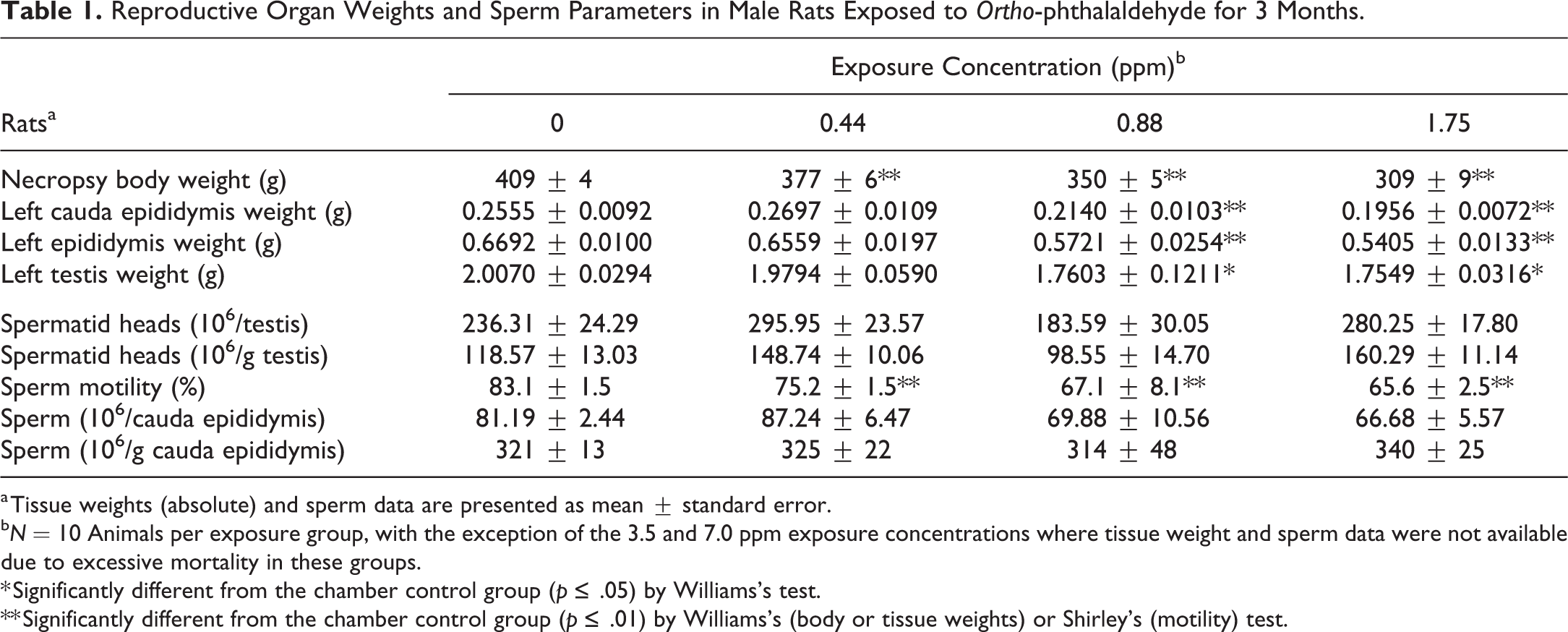
a Tissue weights (absolute) and sperm data are presented as mean ± standard error.
b N = 10 Animals per exposure group, with the exception of the 3.5 and 7.0 ppm exposure concentrations where tissue weight and sperm data were not available due to excessive mortality in these groups.
* Significantly different from the chamber control group (p ≤ .05) by Williams’s test.
** Significantly different from the chamber control group (p ≤ .01) by Williams’s (body or tissue weights) or Shirley’s (motility) test.
Inhalation exposure to OPA resulted in a spectrum of lesions at sites of contact within the respiratory tract (nose, larynx, trachea, and lung), skin, and eye that were generally consistent with an irritant effect (Catlin et al. 2017; NTP 2018). In general, exposure of rats and mice to OPA resulted in lesions throughout the respiratory tract that included necrosis, inflammation, regeneration, hyperplasia, and metaplasia, ranging from minimal to moderate in severity. These histologic findings were generally present in all exposure groups and occurred at deeper sites within the respiratory tract with increasing exposure concentration. These respiratory tract findings are provided for context on the body weight decreases and decreased survival of the animals in these studies and further description of these findings have been published previously (Catlin et al. 2017; NTP 2018).
Male Reproductive Organ Weights and Sperm Parameter Data
As a result of the excessive mortality of the rats and mice in the 3.5 and 7.0 ppm exposure groups, organ weight and sperm parameter data could not be obtained for these animals. However, reproductive organ weights were significantly lower in the male rats in the 0.88 and 1.75 ppm exposure groups (Table 1). Rats exposed to 1.75 ppm had 23%, 19%, and 13% lower absolute cauda epididymis, epididymis, and testis weights, respectively, compared to the controls. Similarly, cauda epididymis, epididymis, and testis weights were lower in the 0.88 ppm exposure group (16%, 15%, and 12%, respectively), compared to controls. In the evaluation of epididymal spermatozoal measurements in the rats, sperm motility was 10% to 21% lower in all the OPA exposed groups that were evaluated. Additionally, there was a negative trend in the number of sperm per cauda, which was 14% and 18% lower in the 0.88 and 1.75 ppm exposure groups, respectively. There was no statistically significant effect in any of the other sperm parameters assessed in the rats in these studies. In the male mice, there were no significant alterations in reproductive organ weights, nor were there any significant changes in sperm parameters, with the exception of sperm motility, which was 10% to 21% lower in all exposure groups evaluated (data not shown).
Age-matched Control Testis and Epididymis Histopathology
Two age-matched untreated rats that were 6 weeks of age (PND 42), confirmed by specific birth dates, were necropsied on PND 45. The testes of these animals displayed incomplete spermatogenesis, characterized by reduced number of elongating spermatids, especially in stages VII and VIII tubules (compare the peripubertal testis in Figure 2A to the adult testis in Figure 2B). In comparison to mature rats (Figure 2B), the tubular lumen diameters were relatively smaller in size (Figure 2A). Additionally, the epididymides contained no or negligible amounts of sperm and the diameter of the ducts of the distal corpus were smaller (Figure 2C) than those from the mature controls (Figure 2D). Low amounts of sloughed germ cells and debris were also scattered throughout the epididymis (Figure 2C). These findings are consistent with what has been reported for 45-day-old-rats (Picut et al. 2015) and indicate that rats at 6 weeks of age are peripubertal.
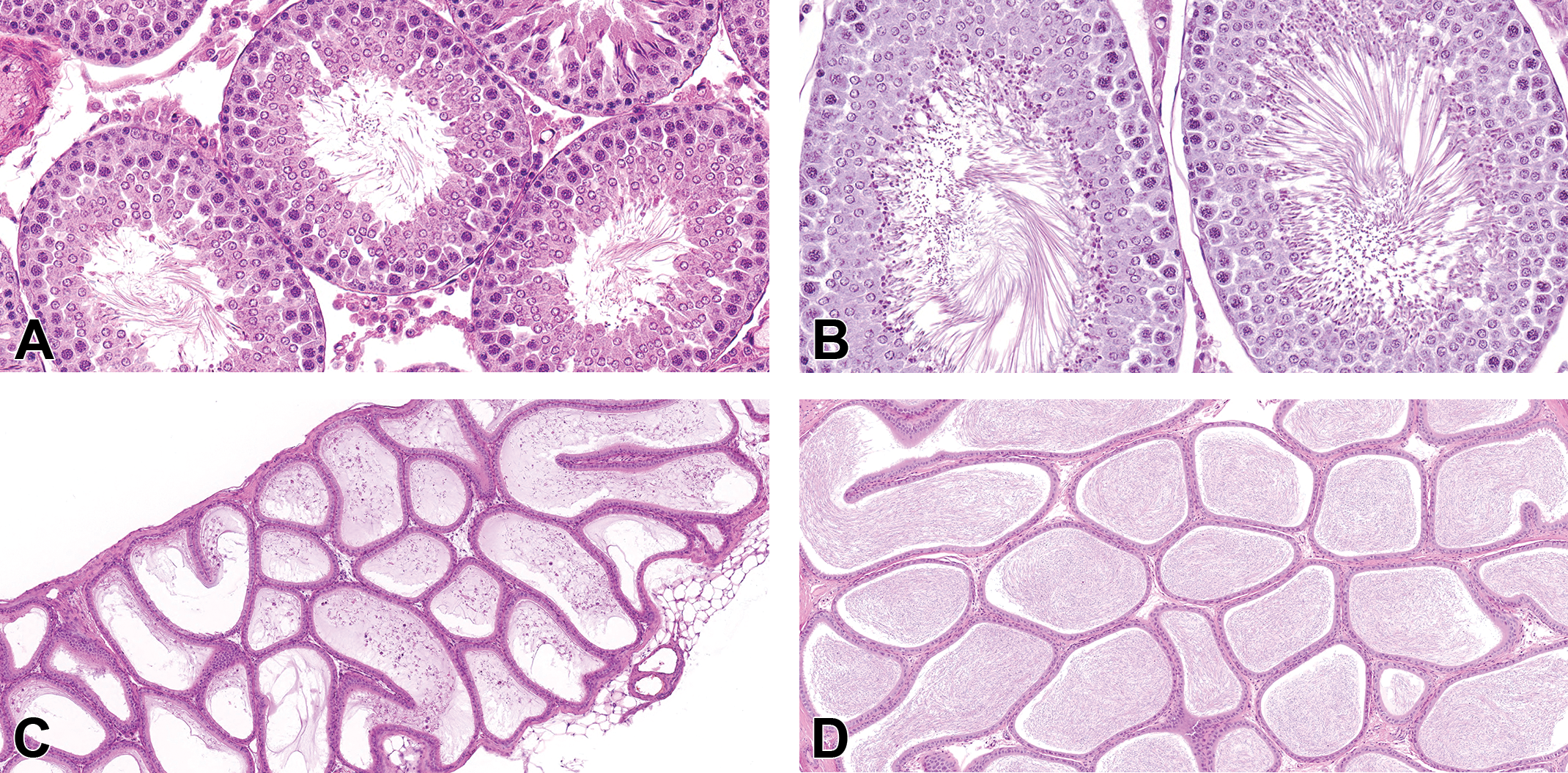
Control testes and epididymides from peripubertal (A, C) and adult (B, D) untreated rats. (A) The testis from an untreated 6 week and 3-day-old (postnatal day 45) rat showed incomplete spermatogenesis characteristic of the stage of peripuberty seen as reduced numbers/density of elongating spermatids, especially in midstage (VII and VIII) tubules, as compared to the numbers/density of elongating tubules seen in the concurrent control adult rat in (B). (B) Elongating spermatids showed typical numbers/density for an adult rat in a stage VIII (left) and stage VII (right) tubule. Also, seminiferous tubule lumen diameters of the peripubertal untreated rat (A) were relatively smaller than those of the adult control rat (B) at the same magnification (original objective 40×). (C) In the peripubertal rat corpus epididymis, there was no or negligible sperm in the duct lumen and the diameter of the duct profiles in the distal corpus and cauda were smaller than in the adult concurrent control (D) at the same magnification (original objective 10×). Sloughed germ cell debris is scattered throughout the peripubertal epididymis (C) but is rare in the adult control rat epididymis (D). Hematoxylin and eosin stain.
Testis and Epididymis Histopathology
Testis and epididymis lesions were observed in the two highest exposure concentrations (3.5 and 7.0 ppm); however, many of the animals in these exposure groups died or were euthanized within the first 10 days of exposure. In both the rats and mice, there were no histopathologic correlates identified that corresponded with the observed responses in the sperm parameters in the exposure groups up to 1.75 ppm (Table 2; data not shown for mice). Because rats are not sexually mature at 6 weeks (PND 42), which was the approximate age of the rats at the start of these studies, the rats that died within the first 10 days of the study also displayed features of sexual immaturity. This prompted additional, stage-aware histopathology peer review of the testes and epididymides in the rats in comparison to not only the concurrent controls, which were sexually mature by the end of the study, but also in comparison to two additional age-matched, untreated control animals. These age-matched untreated rats, subsequently obtained from the same vendor, were not part of the original study but were necessary to help distinguish test article–related lesions from normal peripubertal findings.
Incidences of Nonneoplastic Reproductive Tissue Lesions in Male Rats Exposed to Ortho-phthalaldehyde for 3 Months.
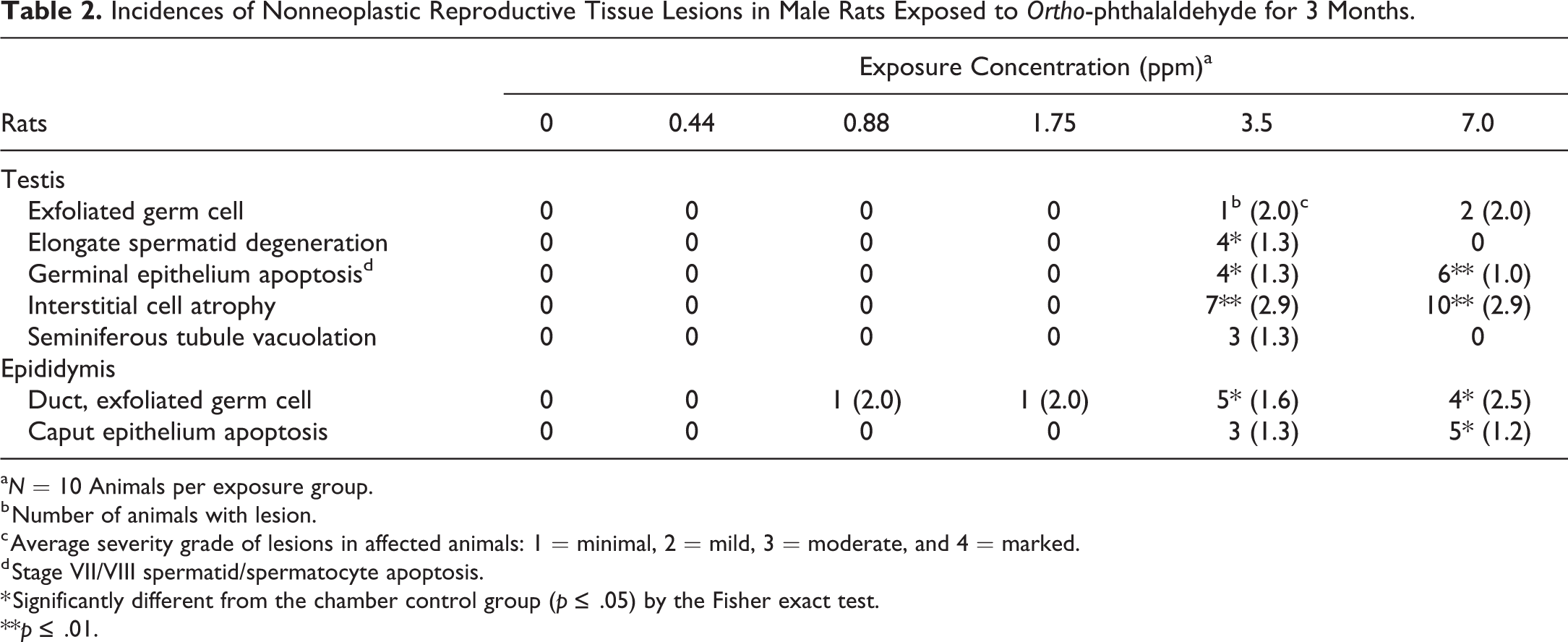
a N = 10 Animals per exposure group.
b Number of animals with lesion.
c Average severity grade of lesions in affected animals: 1 = minimal, 2 = mild, 3 = moderate, and 4 = marked.
d Stage VII/VIII spermatid/spermatocyte apoptosis.
* Significantly different from the chamber control group (p ≤ .05) by the Fisher exact test.
**p ≤ .01.
When compared with the concurrent adult controls (Figure 3C), the testes of the rat early decedents (Figure 3B) had smaller testicular cross-sectional diameters at the subgross level, smaller tubular diameters, lower density of elongating spermatids, some spermatid retention (defined as the appearance of mature step 19 spermatids in stages after stage VIII), and low numbers of sperm in the epididymis. However, when compared with the age-matched (nonstudy) unexposed rats (Figure 3A), the general appearance of the testis in the OPA-exposed early decedents was appropriate for this age of rat, and therefore these findings were attributed to peripuberty and not OPA exposure.
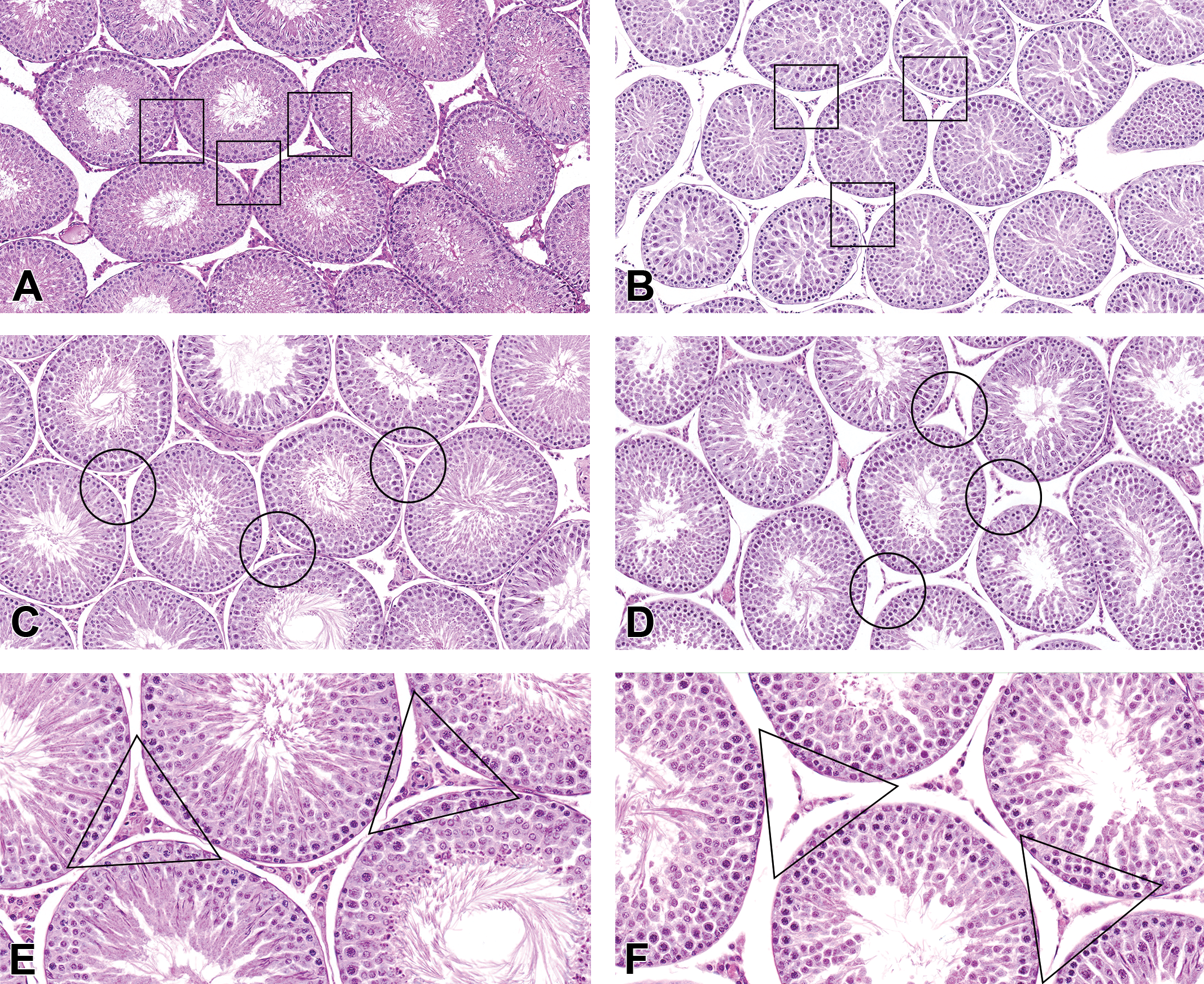
Interstitial (Leydig) cell atrophy in peripubertal (B) and adult (D and F) ortho-phthalaldehyde (OPA)-exposed rats. In both the peripubertal rat exposed to 7.0 ppm that died after 3 days on study (B) and in the adult rat exposed to 3.5 ppm that survived 39 days on study (∼81-day-old; D and F), the Leydig cells are small and contracted (atrophy), when compared to either the testis from an untreated 45-day-old rat (A) or the concurrent (adult) control (C). The small size of the Leydig cells in the early decedent peripubertal rat (B) is not attributable to peripubertal status, as the Leydig cells of the untreated peripubertal rat are plump and normal appearing (A), as compared to the adult control rat (C). Panels (E) and (F) show higher magnification of the testes from the same animals in (C) and (D), respectively, showing Leydig cell atrophy in the adult rat exposed to 3.5 ppm OPA (F) when compared to the untreated control adult rat (E; original objective 40×). Hematoxylin and eosin stain.
However, with the use of the PND 45 untreated rats as comparative controls, there were additional changes in the rat testes that could not be explained by the peripubertal status of the early death rats. There were statistically significant increased incidences of interstitial cell atrophy and germinal epithelium apoptosis in both the 3.5 and 7.0 ppm exposure groups (Table 2). Interstitial cell atrophy was noted as decreased size of the Leydig cells and was generally moderate in severity (Figure 3B, D, and F). The interstitial cell atrophy observed in early death rats (Figure 3B) could not be attributed to sexual immaturity when compared to the testes from age-matched PND 45 (nonstudy) unexposed rats (Figure 3A). The Leydig cells in the 6-week-old untreated rats (Figure 3A) were plump and prominent and generally similar in appearance to the adult concurrent control rats (Figure 3C and E). Moreover, in the 7 animals diagnosed with interstitial cell atrophy in the 3.5 ppm OPA-exposed group, 4 were early deaths (sexually immature) and 3 lived at least 39 days after study initiation (sexually mature; Figure 3D and F). Rather than atrophy, another possibility for the decreased size of the Leydig cells is that OPA may have caused delayed development of the Leydig cells during the peripubertal development phase. Germinal epithelium apoptosis was minimal to mild in severity and characterized by low numbers of round spermatids and pachytene spermatocytes with shrunken, hypereosinophilic cytoplasm and pyknotic, deeply basophilic nuclei, almost exclusively in stages VII and VIII seminiferous tubules (Figure 4A). Apoptosis was also seen in the epithelium of the caput epididymis, with statistically significant increased incidences in the 7.0-ppm-exposed males (Table 2). Severities of this lesion were minimal to mild and were characterized by single shrunken cells lining the duct of the caput epididymis with pyknotic, deeply basophilic nuclei and condensed, amphophilic to hypereosinophilic cytoplasm (Figure 4B).
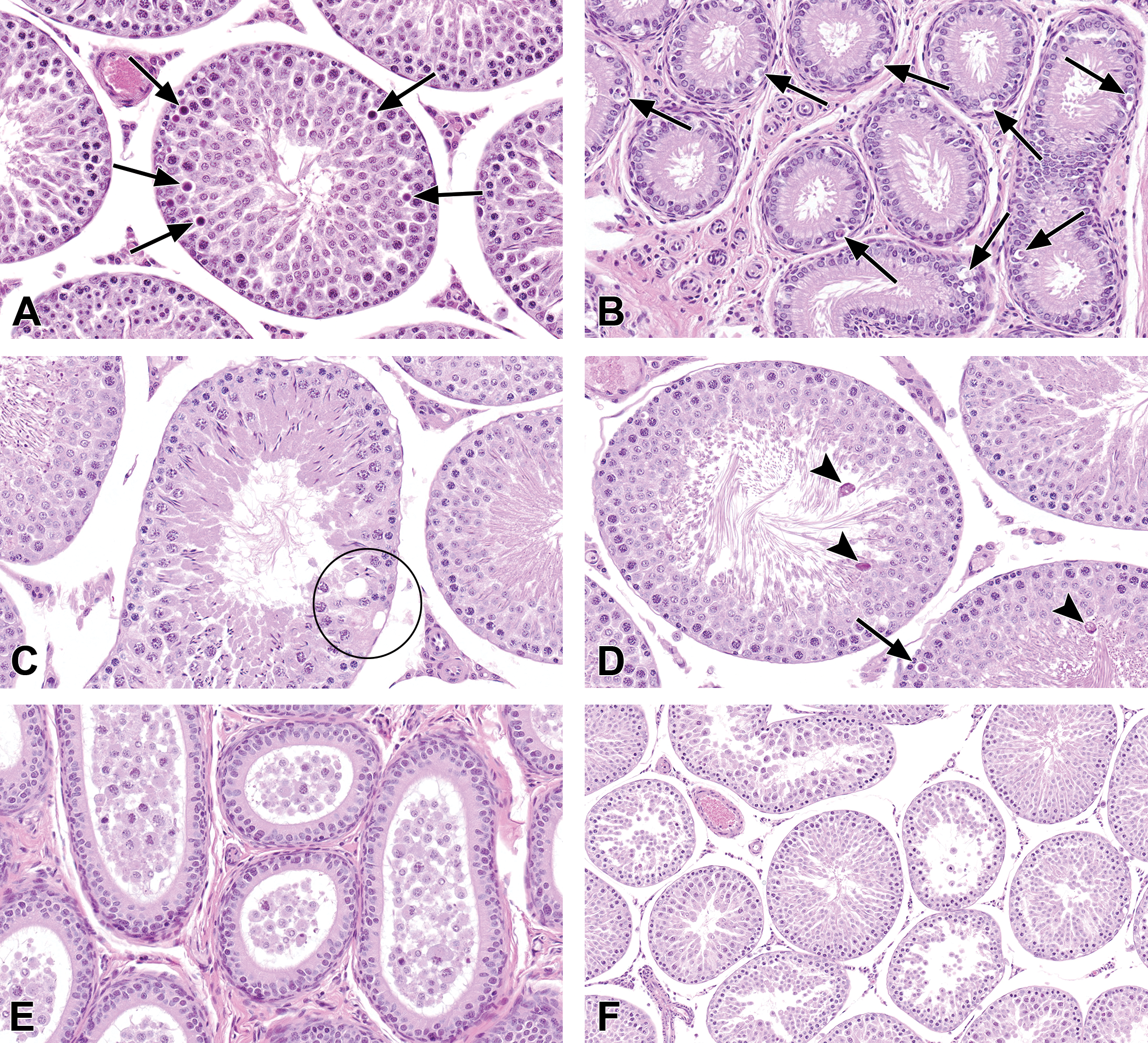
Lesions in the testes and epididymides of rats exposed to ortho-phthalaldehyde. (A) Apoptosis in a midstage tubule in a 3.5 ppm rat. There is also a decrease in the number of elongating spermatids. (B) Apoptosis in the epithelium of the caput epididymis in a 7.0 ppm rat. (C) Vacuolation and focal germ cell dropout from the Sertoli cell cytoplasm in the germinal epithelium in a tubule from a 3.5 ppm rat. (D) Elongated spermatid degeneration (arrowheads) in a 3.5 ppm rat. Degenerating elongating spermatids are seen as hypereosinophilic and irregular. Also, there is apoptosis of a pachytene spermatocyte (arrow). (E) Exfoliated germ cells and debris in the epididymal duct of a 7.0 ppm rat. The same rat had sloughing of the germinal epithelium (germ cell exfoliation) in seminiferous tubules near the rete testis (F). Hematoxylin and eosin stain.
Additionally, there were unilateral and bilateral degenerative changes in the testes of some of the six rats exposed to 3.5 ppm that survived at least 39 days after study initiation (Table 2). Three of these six sexually mature treated rats had seminiferous tubule vacuolation. Vacuolation was characterized by one or more clear spaces in the germinal epithelium, often basally located, and often present with focal germ cell loss from the Sertoli cell cytoplasm (Figure 4C). Another degenerative lesion in OPA-exposed rats that were sexually mature was elongated spermatid degeneration, which was characterized by hypereosinophilic, irregular elongated spermatids (Figure 4D), or depletion of the number of the elongating spermatids (Figures 3D, F, and 4A).
Seminiferous tubule vacuolation and degeneration of the elongating spermatids were sometimes accompanied by minimal spermatid retention and the presence of exfoliated germ cells and debris in the ductular lumen of the epididymis (Figure 4E). Although the peripubertal age-matched controls had low numbers of sloughed germ cells and cell debris scattered throughout the epididymis, rats in the 3.5 and 7.0 ppm exposure groups (including early decedents) displayed increased severity of exfoliated germ cells and cellular debris, which was particularly prominent within the ductular lumen of the caput epididymis (Table 2). Prominence of the cell debris in the caput region of the epididymis reflects recent exfoliation of germ cells from the testis. Mild to moderate amounts of debris in one of the 3.5-ppm-exposed rats and two of the 7.0-ppm-exposed rats coincided with germ cell exfoliation in the testis. In these animals, germ cell exfoliation was characterized by the presence of rounded, isolated germ cells in the lumen in a few seminiferous tubules adjacent to the rete testis (Figure 4F). However, the low incidence, focal distribution, and the frequently unilateral nature of this lesion near the rete testis make its relationship to OPA administration uncertain.
Other reproductive tissue findings in the rats exposed to OPA were of unknown toxicological significance or were considered incidental findings. Two rats in the 7.0 ppm exposure group had degeneration of the round spermatids, were noted as deeply basophilic ring-shaped nuclei, an indication of acute spermatid degeneration. Because this change was only present in two rats, both of which were unexpectedly found dead, the change may be associated with moribundity rather than OPA exposure. One rat in the 1.75 ppm exposure group had mild, nonspecific degeneration and atrophy of the germinal epithelium that was accompanied by mild sloughed germ cells and debris in the epididymis. Similarly, a 0.88-ppm-exposed rat had marked atrophy of the germinal epithelium that was associated with absent sperm in the epididymis. Based on the single occurrences of germinal epithelium degeneration or atrophy in these two exposure groups and the absence of germinal epithelium atrophy in higher exposure groups, both of these lesions were considered to be incidental findings.
In the mice, there were fewer histopathological findings in the testis and epididymis compared to the rats. Incidences of cellular depletion of the germinal epithelium were significantly increased only in the mice exposed to 3.5 ppm OPA (Table 2). Microscopically, this lesion was characterized by small numbers of seminiferous tubules with partial depletion of one or more generations of germ cells (not shown). Of the seven mice in the 3.5 ppm exposure group with cellular depletion of the germinal epithelium, four also had exfoliated germ cells present in the epididymis. Additionally, three of the mice exposed to 3.5 ppm OPA which died early had mild interstitial cell atrophy, which resembled the finding in rats but was less severe. These changes in the testis and epididymis were likely a result of the decreased body weight gain that was secondary to exposure, but their relationship to OPA cannot be completely ruled out.
Discussion
The evaluation of the male reproductive tract in sexually immature animals, whether as part of a juvenile toxicity study or due to early deaths, presents a diagnostic challenge to the pathologist. In the present study, rats and mice exposed via inhalation to OPA resulted in overt toxicity and death in the two highest exposure concentrations (Catlin et al. 2017; NTP 2018). All of the rats and mice in the 7.0 ppm exposure group died within the first two weeks of the study. Survival was also lower in the animals within the 3.5 ppm exposure group. These rats and mice had extensive respiratory lesions, including suppurative inflammation of the nose, which may have contributed to their decreased survival, especially since rats and mice are obligate nose breathing animals. Due to the age of the animals at the start of the study, which was approximately 6 weeks (∼PND 42), there was concern that normal features of sexual immaturity would be superimposed on any exposure-related lesions during the histopathological evaluation of the testis and epididymis in the early decedents.
Because the concurrent study controls were euthanized as scheduled at the end of the study and had achieved sexual maturity, it was difficult to make comparisons to the peripubertal animals that had died early on the study. Due to the earlier sexual maturity of mice (6–8 weeks, PND 42–56) than rats, the testicular changes in the mouse early decedents were not confounded by sexually immaturity. To aid in the determination of normal physiological changes associated with sexual immaturity versus exposure-related changes in the rat study, two age-matched control rats, which were 6 weeks of age (∼PND 42), were obtained from the same vendor as a reference for the rats that died early.
The early deaths in the two highest exposure groups (3.5 and 7.0 ppm) resulted in microscopic observations of features associated with peripuberty in the testis in male rats but not in male mice, because as previously mentioned, mice reach sexual maturity earlier than rats. In the OPA-exposed rats, some of the testicular microscopic observations were consistent with those that were found in the age-matched (PND 45) unexposed peripubertal rats (e.g., see Picut et al. 2015), and therefore these effects in the rat were attributed to their sexual immaturity and not related to the inhalation of OPA. These features of peripubertal maturation status included smaller tubular diameters, lower density of elongating spermatids in the seminiferous tubules (particularly stages VII and VIII) with occasional spermatid retention in the testes, accompanied by absent or decreased presence of sperm, and the presence of sloughed germ cells and debris that was predominantly located in the cauda epididymis.
The sperm from exposed animals in this study was also evaluated, with the exception of the two highest exposure groups due to early mortality. The percentage of motile sperm was significantly reduced in an exposure concentration-related manner in both rats and mice in all exposure groups evaluated in comparison to controls. Additionally, the 0.88 and 1.75 ppm OPA-exposed rats had significantly lower absolute weights of the left cauda epididymis, the left epididymis, and the left testis. However, the reproductive organ weight changes were limited to the rats as this effect was not seen in the mice. There were no microscopic changes attributed to OPA exposures of 0.44, 0.88, or 1.75 ppm, in either the testis or the epididymis of rats or mice that could explain the concentration-dependent decreases in sperm motility in both species. Either the changes were below the level of histologic detection, or there were physiological changes in the testis or the epididymis that resulted in decreased motility.
Inhalation of OPA resulted in degenerative changes in the testes of 3.5 ppm rats that survived beyond 39 days. These changes included seminiferous tubule vacuolation, focal germ cell loss from individual Sertoli cells, and degeneration and/or depletion of elongating spermatids. These changes were generally accompanied by exfoliated germ cells and/or hypospermia in the epididymis. Vacuolation is a common morphological response to toxicants and usually indicates early damage to Sertoli cells (Creasy 2001; Moffit et al. 2007). If the damage persists, the Sertoli cell vacuolation is typically followed by degeneration, disorganization, or exfoliation of the germ cells, depending on the nature of the Sertoli cell functional disturbance (Creasy 2001). The presence of exfoliated germ cells and cell debris in the epididymis of exposed mature animals in this study was particularly prominent in the caput epididymis and more severe than was seen in the 6-week age-matched controls. It reflects degeneration and sloughing of germ cells from the testis and is a sensitive indicator of germ cell injury, which accompanies most types of testicular damage, including long-term testosterone withdrawal (O’Donnell et al. 1994).
Other histopathologic alterations in the testes of rats exposed to 3.5 and 7.0 ppm OPA that could not be attributed to sexual immaturity alone included the degeneration (apoptosis) of the round spermatids and pachytene spermatocytes in the stages VII and VIII seminiferous tubules, interstitial (Leydig) cell atrophy, and apoptosis in the caput epididymal epithelium. These lesions are characteristic of low levels of testicular testosterone (Creasy 2001; Chapin and Creasy 2012). Apoptosis of spermatids and spermatocytes in stages VII and VIII seminiferous tubules has been reported following gonadotropin-releasing hormone (GnRH) antagonist treatment, but it has also been attributed to reductions in body weight gain (Hikim et al. 1995; Rehm et al. 2008). Inhibition of GnRH secretion and the resulting testosterone secretion is an effect that can be test article related or expected from decreases in body weight gain and nonspecific stress (Rehm et al. 2008; Everds et al. 2013). However, microscopic evidence of interstitial cell atrophy and apoptosis of the epididymal epithelium are generally only associated with very severe decreases in testosterone secretion and are less likely to be attributable to stress alone (Chapin and Creasy 2012; Creasy 2001; Everds et al. 2013). The age-matched controls had plump, normal appearing Leydig cells. Based on this, these three changes, which are suggestive of a reduction in testosterone secretion, are considered to be directly or indirectly (via moribundity and/or decreased body weight or food intake) related to OPA exposure.
Although many of the mice in the 3.5 and 7.0 ppm exposure groups died early, the mice that died early had complete spermatogenesis in the testis and substantial amounts of sperm in the epididymis. This difference between the rats and mice can be explained by the differential duration of spermatogenesis between the two species (8 weeks vs. 5 weeks, respectively; Adler 1996). In light of this, seven male mice in the 3.5 ppm group had small numbers of seminiferous tubules with partial depletion of one or more generations of germ cells, often accompanied by epididymal exfoliated germ cells, and three mice in this group had interstitial cell atrophy. Although these changes could be directly related to OPA exposure, mice are thought to be more sensitive than rats to the effects of body weight loss on spermatogenesis (Everds et al. 2013). The degenerative effects of stress on the testes are partially mediated through decreased GnRH, which causes reduced testosterone from the Leydig cells and germ cell exfoliation. Although these are similar histological findings to those attributed to OPA exposure in the rat, the severity of the reduced body weight gain of the mice in this group, compared with chamber controls, and the greater susceptibility of mice to stress-induced testicular changes suggest that the partial germ cell depletion and the interstitial cell atrophy are likely secondary to the stress associated with the poor condition and low body weight of the mice rather than a direct effect of OPA (Everds et al. 2013).
Although it is difficult to differentiate exposure-related morphological effects from the normal features of sexual immaturity in the testes and epididymides in rodents that are removed early in a toxicology study, it is possible to make these differentiations, as was demonstrated in this case study with OPA. In this study, age-matched, nonstudy, untreated animals from the same source were available and utilized to help make these differentiations. The addition of increased numbers of control animals should be considered in the future to serve as age- and sex-matched controls in safety toxicology studies where excessive toxicity and mortality are sometimes observed, especially where reproductive end points and juvenile animal study experimental designs are being investigated.
Footnotes
Authors’ Note
This article reflects the views of Deepa B. Rao on research conducted during her tenure at the National Toxicology Program and Integrated Laboratory Systems, Inc., and should not be construed as representing views or policies of her current employer—the U.S. Food and Drug Administration.
Acknowledgment
The authors would like to thank Vicki Sutherland and Kyathanahalli Janardhan for their critical review of this article.
Author Contributions
Authors contributed to conception or design (DR, BM, MW); data acquisition, analysis, or interpretation (NC, CW, DC, GK, MW); drafting the manuscript (NC, CW); and critically revising the manuscript (NC, CW, DC, DR, GK, BM, MW). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Research Program of the National Institutes of Health (NIH; National Institute of Environmental Health Sciences [NIEHS] Projects ZIA ES103319-01 and ZIA ES103316-01) and by NIEHS contract no. N01-ES-65406.
