Abstract
The 2018 annual National Toxicology Program Satellite Symposium, entitled “Pathology Potpourri,” was held in Indianapolis, Indiana, at the Society of Toxicologic Pathology’s 37th annual meeting. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers’ talks along with select images that were used by the audience for voting and discussion. Various lesions and other topics covered during the symposium included seminiferous tubule dysgenesis in rats, ameloblast and odontoblast degeneration/necrosis in a Sprague Dawley rat, intestinal leiomyositis in a beagle dog, gallbladder mucinous hyperplasia, focus of hepatocellular alteration and bile duct alteration in otters, renal tubule cytoplasmic vacuolation with basophilic granules in mice treated swith antisense oligonucleotide therapy, a uterine choriocarcinoma in a rhesus macaque, and rete ovarii proliferative ovarian lesions in various aged rat strains. One particularly provocative lesion was a malignant neoplastic proliferation in the renal pelvic region of a cynomolgus macaque from a 21-day study. Additional challenging lesions included thyroid proliferative lesions in zebra fish and gross findings in fish larvae during routine chemical screening. The Rabbit and Minipig International Harmonization of Nomenclature and Diagnostic Criteria Organ Working Groups also presented a series of challenging lesions.
Keywords
The 2018 annual National Toxicology Program (NTP) Satellite Symposium, entitled “Pathology Potpourri,” was held in Indianapolis, Indiana, at the Society of Toxicologic Pathology’s (STP) 37th annual meeting. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article summarizes individual speaker presentations along with select images that were used by the audience for voting and discussion. Various lesions and other topics covered during the symposium included seminiferous tubule dysgenesis in Hsd:Sprague Dawley (SD) rats, ameloblast and odontoblast degeneration/necrosis with dentin matrix alteration in an SD rat, intestinal leiomyositis in a beagle dog, gallbladder mucinous hyperplasia, focus of hepatocellular alteration and bile duct alteration in otters (Lutra lutra), renal tubule cytoplasmic vacuolation with basophilic granules in CRL:CD1(ICR) mice treated with antisense oligonucleotide therapy, a uterine choriocarcinoma in a rhesus macaque (Macaca mulatta), and rete ovarii proliferative ovarian lesions in various aged rat strains. One particularly provocative lesion was a malignant neoplastic proliferation in the renal pelvic region of a cynomolgus macaque (Macaca fascicularis) from a 21-day oral gavage study. Additional challenging lesions included proliferative thyroid lesions in zebra fish (Danio rerio) and gross findings in Japanese medaka (Oryzias latipes) larvae during routine chemical screening. The Rabbit and Minipig International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) Organ Working Groups also presented an interesting series of challenging lesions.
Testicular Dysgenesis
Dr. Cynthia J. Willson (Integrated Laboratory Systems, Inc., Research Triangle Park, NC) started the day by presenting two cases highlighting unusual findings in the testis of Hsd:SD rats.
The first case was a testis from a 2-year-old rat from an NTP 2-year carcinogenicity bioassay with perinatal exposure (continuous dosing beginning on gestation day 6, then via lactation, and then dosed feed) to an agent that is currently in peer review. There were other changes present in the testis, but the audience was asked to pay attention to a focus near the middle of the section. Two serial sections of the same focus were shown, one stained with hematoxylin and eosin (H&E) and one stained with periodic acid-Schiff/hematoxylin (PAS/H; Figure 1A–D). The choices and voting results were seminiferous tubule hypoplasia (3%), seminiferous tubule atrophy (3%), seminiferous tubule dysgenesis (38%), Leydig cell adenoma (2%), Leydig cell hyperplasia (5%), atypical Leydig cell hyperplasia (10%), Leydig cell adenoma and seminiferous tubule atrophy (8%), Leydig cell hyperplasia and seminiferous tubule atrophy (24%), or other (6%).
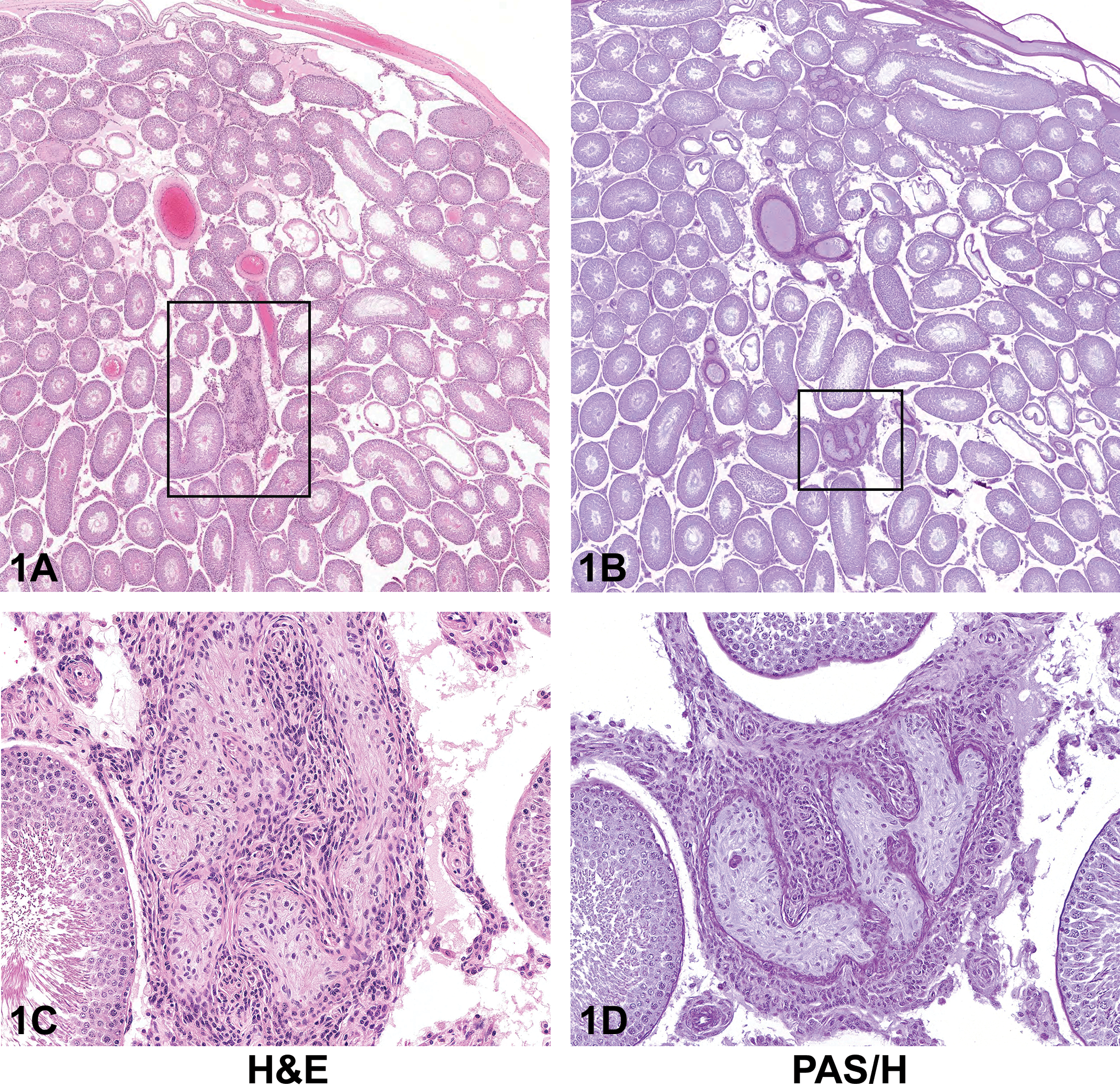
(A–D) Seminiferous tubule dysgenesis from a 2-year-old male rat from a National Toxicology Program (NTP) perinatal chronic bioassay presented as case 1. The sections are stained with H&E (A and C) and periodic acid-Schiff/hematoxylin (PAS/H; B and D). At low magnification stained with H&E (A), there are numerous Sertoli cell-only tubules within the section and a focus of seminiferous tubule dysgenesis within the box. At higher magnification (C), the focus of seminiferous tubule dysgenesis is seen as an anastomotic network of abnormally formed seminiferous tubules, surrounded by an aggregation of poorly differentiated, spindle-shaped Leydig cells. The aberrant seminiferous tubules contain poorly differentiated Sertoli cells. When stained with PAS/H (B and D), the same focus (boxed in B) in a serial section of the testis shows that the basement membrane is thickened and convoluted when compared to the surrounding seminiferous tubules. Spermatogenesis is not present in the dysgenetic tubules. In serial sections (compare A to B and C to D), the focus of dysgenesis changes in appearance due to the convoluted nature of the abnormally formed tubules.
The second case was a testis from a 10-week-old, F1 generation rat from an NTP Reproductive Assessment by Continuous Breeding (RACB) study that is currently in peer review. The animal was exposed continuously throughout gestation, lactation, and then via dosed feed. Two serial sections of the same focus were shown, one stained with H&E and one stained with PAS/H (Figure 1E–H). The choices and voting results were no remarkable lesion (0%), seminiferous tubule degeneration (20%), seminiferous tubule dysgenesis (49%), seminiferous tubule hypoplasia (9%), seminiferous tubule atrophy (16%), atypical seminiferous tubule (6%), or other (2%).
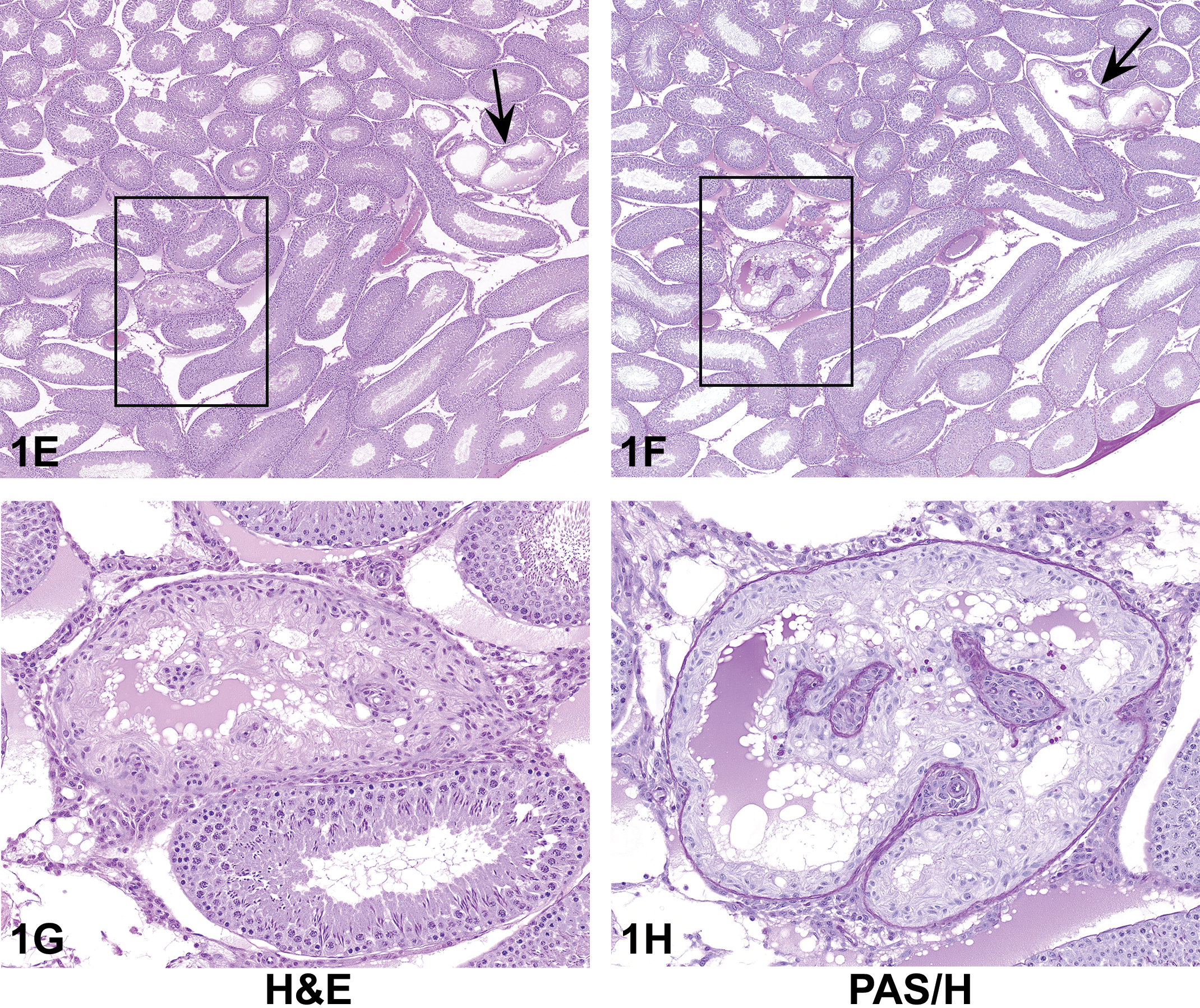
(E–H) Seminiferous tubule dysgenesis from a 10-week-old male rat from the F1 generation of an NTP Reproductive Assessment by Continuous Breeding study presented as case 2. The sections are stained with H&E (E and G) and PAS/H (F and H). Unlike the testis in case 1 (A–D), abnormal aggregation of Leydig cells is not a prominent feature in the focus of dysgenesis in case 2 (E–H). At low magnification (E and F), a focus with an abnormally shaped seminiferous tubule(s) (dysgenesis) is present within the box, as well as in the upper right of the image (arrows); the latter focus has prominent luminal dilation. At higher magnification (G and H), the aberrant seminiferous tubule(s) contains immature-appearing Sertoli cells and lacks spermatogenesis. Like in case 1, the same focus in serial sections in case 2 differs in appearance (compare E to F and G to H). A serial section from the same testis, when stained with PAS/H, makes the convolutions of the thickened basement membrane more evident (compare G to H). The invaginations of the thickened basement membrane are more readily apparent with PAS/H staining, such that it appears that there are “islands” of Leydig cells and vessels within the focus of dysgenesis (H). H&E = hematoxylin and eosin.
Both cases were of “dysgenesis” of the seminiferous tubules of the testis. This lesion has been described in the literature after in utero exposure to various phthalates (Barlow, McIntyre, and Foster 2004; Fisher et al. 2003; Mahood et al. 2005; van den Driesche et al. 2017). It is not yet defined in the online NTP Non-Neoplastic Lesion Atlas (https://ntp.niehs.nih.gov/nnl/, last accessed July 12, 2018) nor in the INHAND guidelines for the male reproductive system (Creasy et al. 2012). Seminiferous tubule dysgenesis is characterized in experimental studies as a developmental malformation seen microscopically as aberrant or misshapen seminiferous tubules. This lesion is associated with immature Sertoli cells, focal Leydig cell aggregates, and intratubular Leydig cells. It may occur as small foci (1–3 per testis) or occupy the entire testis, with no lumens or dilated lumens. The malformed tubules may appear to form anastomotic networks. The dysgenetic tubules contain poorly differentiated Sertoli cells, with small, elongated, and sometimes cleaved nuclei and less prominent nucleoli than the typical, prominent, tripartite nucleoli seen in mature Sertoli cells. Spermatogenesis is absent in these foci of dysgenesis, but it may be present elsewhere in the testis, as seen in both cases presented (Figure 1A–H; Fisher et al. 2003). Dysgenetic seminiferous tubules often have thickened basement membranes, which are more readily apparent with PAS/H than with H&E. Atrophic tubules can also appear to have thickened basement membranes. The thickened basement membranes of dysgenetic tubules, however, may be convoluted, discontinuous, or have invaginations, such that it appears that there are “islands” of interstitial cells that may contain vessels in the “center” of tubules, as seen in case 2 (Figure 1E–H).
Foci of tubular dysgenesis may be quite small, such as in case 2 (Figure 1E–H), making sampling an important consideration. Foci of dysgenesis tend to be located toward the center of the testis and often are present within a section that also contains the rete testis (Barlow, McIntyre, and Foster 2004). This has generally been true for the cases of tubular dysgenesis seen in three NTP studies currently under review. Foci of dysgenesis may be present in one or both testes and may be more severe in cryptorchid than in scrotal testes (Fisher et al. 2003; van den Driesche et al. 2017). Multiple sections of testis, especially containing the rete testis, may increase the likelihood of detecting this unusual lesion.
Another feature that has been demonstrated in seminiferous tubule dysgenesis is the presence of ectopic cells. In rats exposed in utero to phthalates, immunohistochemical markers have been used to demonstrate Sertoli cells and gonocytes within the interstitium and Leydig cells inside the malformed seminiferous tubules (Fisher et al. 2003; Mahood et al. 2005; Lara et al. 2017; van den Driesche et al. 2017). Lara and colleagues (2017) reported that rats exposed in utero to dibutyl phthalate showed normal formation of seminiferous cords between embryonic days (E)13.5-14.5, then abnormal fetal Leydig cell aggregation at E17.5. Then at E18.5, with the use of triple staining for Sertoli cells, gonocytes, and peritubular myoid cells, the seminiferous cords in exposed rats appeared to rupture, releasing their contents (i.e., Sertoli cells and gonocytes), admixed with scattered peritubular myoid cells, into the interstitium. The cords appeared normally formed at one end, with a normal smooth muscle actin (SMA)-positive peritubular myoid layer, and the other end appeared “open” at the point of rupture, with loss of SMA staining (Lara et al. 2017). The actual histologic lesion of dysgenesis, malformed/misshapen seminiferous tubules, is not evident in embryonic testes and only becomes evident in the first week of life in rats (Fisher et al. 2003). The ectopic Sertoli cells and gonocytes appear in late gestation and disappear early in postnatal life, but the ectopic intratubular Leydig cells can persist throughout postnatal life.
Dr. Willson explained why Leydig cell hyperplasia or adenoma were not the preferred diagnoses. Although dysgenetic tubules often are accompanied by aggregations or clusters of Leydig cells, Leydig cells are not necessarily a prominent feature of the dysgenetic foci, as demonstrated by the second case (Figure 1E–H). Regarding location, unlike Leydig cell hyperplasia or adenoma, foci of dysgenesis tend to be located in the same anatomical position in the testis, as previously mentioned (Barlow, McIntyre, and Foster 2004). Others have shown that the Leydig cells in rat embryos exposed to phthalates aggregate and form clusters, without an increase in the actual number of Leydig cells (Fisher et al. 2003; Mahood et al. 2005). Furthermore, the Leydig cells within foci of dysgenesis differ morphologically from those in hyperplasia or adenoma. The Leydig cells in these foci appear to be poorly differentiated, are spindle shaped like embryonic Leydig cells, and do not have the abundant eosinophilic or vacuolated cytoplasm often apparent in hyperplasia or adenoma (Barlow, McIntyre, and Foster 2004). Similarly, the Leydig cell aggregates in dysgenesis, while they can form nodular mass-like lesions, are generally not expansile or compressive. Finally, while Leydig cell adenomas can contain entrapped, atrophic seminiferous tubules, Leydig cell clusters within foci of dysgenesis contain seminiferous tubules that may also contain only Sertoli cells, but these tubules are aberrant and malformed rather than simply atrophic (Barlow, McIntyre, and Foster 2004). It is the presence of malformed seminiferous tubules, often bizarrely shaped or anastomotic, that is important in identifying the lesion of dysgenesis.
Seminiferous tubule dysgenesis is a unique, treatment-induced developmental lesion that has been described with in utero exposures to various phthalates that include the “masculinization programming window” of E15.5-18.5 (van den Driesche et al. 2017). Developmentally, the first sign of dysgenesis is the abnormal aggregation of fetal Leydig cells in late gestation (E17.5; Mahood et al. 2005; Lara et al. 2017). These abnormal Leydig cell aggregations progress in severity to E21.5 and persist after birth and throughout life (Mahood et al. 2005; Lara et al. 2017). Another sign of dysgenesis is the presence of binucleated/multinucleated gonocytes within the tubular lumens in late gestation/early postnatal life, with subsequent decreased numbers of spermatogonia and, consequently, spermatocytes (Barlow and Foster 2003; Clewell et al. 2013; Mylchreest et al. 2002; Parks et al. 2000). As with the ectopic Sertoli cells and gonocytes in the interstitium, the abnormal multinucleated gonocytes within tubules also disappear in early postnatal life. Finally, the actual lesion of seminiferous tubule dysgenesis, with malformed tubules, becomes evident in the first week of life. Dr. Willson also presented images of seminiferous tubule dysgenesis from two 3-day-old rats from an NTP bioassay that were exposed in utero to a third chemical (related to the chemicals from the first two cases), demonstrating that these abnormally formed seminiferous tubules can be found in postnatal life from 3 days to 2 years (case 1).
Dr. Willson ended with a discussion of the potential human relevance of the lesion of seminiferous tubule dysgenesis. The “testicular dysgenesis syndrome” (TDS) hypothesis states that four common reproductive disorders of the newborn (cryptorchidism, hypospadias) or young adult human male (decreased spermatogenesis/low sperm count, testicular germ cell cancer) may all have a common origin of impaired development of the fetal testis (Skakkebaek, Rajpert-De Meyts, and Main 2001). Genetics, maternal lifestyle factors, and environmental exposures have been implicated in the impairment of embryonic testis development in TDS disorders. Animal models of TDS have been based on in utero exposures (inclusive of the masculinization programming window) to dibutyl and other phthalates. In utero phthalate exposure in rats causes the TDS disorders of cryptorchidism, hypospadias, and decreased sperm counts (Fisher et al. 2003) but does not induce testicular germ cell cancer. However, in utero phthalate exposure does cause a disorder of embryonic germ cells that manifests as multinucleated gonocytes in the neonatal rat. In addition, and germane to the two cases presented, in utero phthalate exposure in rats causes the lesion of focal histologic testicular dysgenesis, with its malformed seminiferous tubules, such as in cases 1 and 2, with immature-appearing Sertoli cell-only tubules, and with or without abnormal Leydig cell clustering and intratubular Leydig cells (Barlow and Foster 2003; Fisher et al. 2003; Mahood et al. 2005; Hutchison et al. 2008). Interestingly, the lesion of focal histological testicular dysgenesis is also reported in testicular biopsies from patients with TDS disorders, including infertile men and men with testicular germ cell cancer (Sharpe 2006; Sharpe and Skakkebaek 2008).
White Rat Tooth
Dr. Bhanu P. Singh (Janssen Research & Development, Spring House, PA) presented an incisor lesion from a rat. Collaborators on this project included Janssen colleagues Drs. Junguo Zhou, Srinivas Mamidi (study sponsors), and Katie Turner (study monitor) as well as Claudine Tremblay (Charles River Laboratories, Inc.), who was the original study pathologist.
Dr. Singh’s case focused on gross and microscopic findings in the teeth of an SD rat from a proprietary 9-week oral gavage study. At the end of study, a male rat (about 18 weeks old) from a treated group had focal white discoloration on the labial surface of multiple incisors (Figure 2A). The incisors in control rats were yellow/orange, which is the expected color for these teeth in rats. Microscopically, white discoloration of the incisor correlated with a focal lesion, primarily affecting the odontoblasts and ameloblasts, along with changes in the dentin. There were focal regions of thinning and discontinuation of dentin, altered matrix staining, and disruption and thinning of the ameloblast and odontoblast layer (Figure 2B). Focally, there was degeneration and necrosis of ameloblasts with squamous cell metaplasia of adjacent ameloblasts and deposition of eosinophilic material (likely keratin; Figure 2C). Changes in the odontoblast layer were observed as loss of odontoblastic cell height and vacuolated odontoblasts associated with irregular foci of poorly mineralized, amphophilic dentin and altered orientation of dentin tubules. Occasionally, within the region of altered dentin matrix, there were inclusions of cellular debris, which were likely necrotic or degenerated odontoblasts shed into the newly formed dentin (Figure 2D). The outer layer of dentin (the oldest dentin) appeared normal, whereas the inner layer was thinned or irregular in thickness.
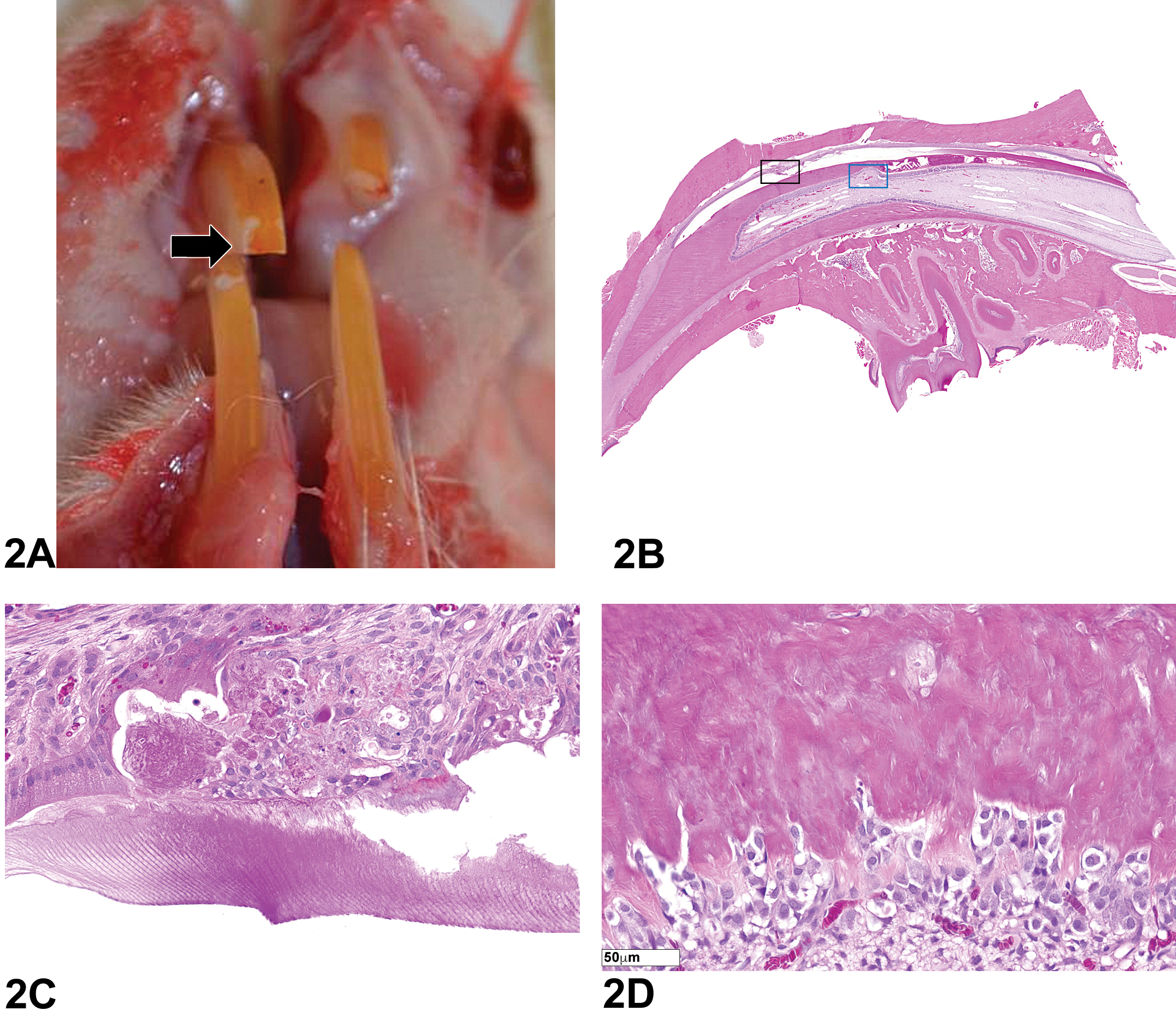
(A–D) Focal lesion in the incisor of a treated male Sprague Dawley rat. Focal white discoloration (A, arrow) on the labial surface of multiple incisors. Longitudinal section of incisor tooth (B) showing focal regions of thinning and discontinuation of the ameloblast (black box) and odontoblasts (blue box) along with changes in the dentin. Higher magnification (C) of black boxed region in (B) showing degeneration and necrosis of ameloblasts with squamous cell metaplasia of adjacent ameloblasts and deposition of eosinophilic material (likely keratin). Higher magnification (D) of blue boxed region in (B) showing degenerated and vacuolated odontoblasts associated with dentin matrix alteration.
A series of images illustrating microscopic changes in ameloblasts and odontoblasts were presented and the audience was asked to vote for their preferred diagnostic term. The voting choices and results were 1) ameloblast degeneration/necrosis (9%); 2) odontoblast degeneration/necrosis (5%); 3) dentin matrix alteration (4%); 4) dentin dysplasia (5%); 1, 2 and 3 (37%); and 1, 2 and 4 (40%). The presenter favored the second most popular diagnosis (choices 1, 2 and 3) of the audience.
A quick overview of rodent tooth histology was discussed. Rat incisors are prominent teeth exposed by a cleft lip. These are four long, sharp front teeth, two on the top (maxillary incisors) and two on the bottom (mandibular incisors). A film of iron is deposited between the dentin and enamel giving rodent incisors a yellow/orange appearance. Histologically, longitudinal sections of rat incisor can be divided mainly into two sides: labial (convex side), which is covered by a layer of enamel, and lingual (concave side), which is enamel-free but does have a very thin layer of cementum into which fibers of the periodontal ligament are embedded. The apical foramen, at the end of each tooth, allows for passage of blood vessels and nerves. The dental pulp is contained within the center of the tooth (pulp cavity) and is composed of delicate connective tissue interspersed with small blood vessels, sensory nerves, lymphatics, and primitive connective tissue cells. Ameloblasts are columnar epithelial cells responsible for producing enamel. Enamel is usually completely removed during decalcification leaving a clear vacant area in its place (enamel space). Odontoblasts are columnar mesenchymal cells that line the perimeter of the pulp cavity and are responsible for dentin production (Leininger and Schutten 2018).
Audience members generally agreed to use the term degeneration/necrosis for the ameloblast and odontoblast lesions. The discussion among audience members mainly focused on what term should be used to describe the changes in the dentin: dentin dysplasia or dentin matrix alteration. As per the INHAND skeletal tissues guidance (Fossey et al. 2016), dentin matrix alteration is characterized as abnormal dentin (dentin tubules arranged in disorderly fashion) with trapped cells or cellular inclusions. Dentin matrix alteration is considered a secondary response to odontoblast degeneration. Dentin dysplasia refers to aberrant development of odontogenic tissues, characterized by an irregular mass of dentin-like material surrounded by fragments of the original tooth and small islands of bone. Both terms communicate a different pathogenesis for a tooth lesion. Based on the mode of action of the compound in the present case, odontoblast degeneration/necrosis was considered a primary change; therefore, a diagnosis of dentin matrix alteration was preferred for changes in the dentin. There was also discussion about whether a single term can be used to lump all of the microscopic features. The presenter noted that the lumping approach, using a generic term, will mask the findings in specific cell populations of tooth and therefore should not be used.
Rodent incisors have no roots and grow continuously for most of a rat’s life. It takes about 40 to 50 days for a new tooth generated at the base to reach the tip. The entire tooth is, therefore, never more than 40 to 50 days old (Schour and Massler 1949). During the proliferation stage, tooth formation proceeds through increased mitotic activity, leading to the development of ameloblasts and odontoblasts, which produce enamel prism (tightly packed mass of hydroxyapatite crystals in an organized pattern) and dentin (hard, dense bony tissue forming the bulk of a tooth beneath the enamel). Odontoblasts differentiate from mesenchymal cells around the apical end of the incisor, start to secrete dentin, and move in the incisal direction as they differentiate (Kuijpers, van de Kooij, and Slootweg 1996). Differentiating cells of the odontoblast series are more sensitive to the action of chemical and physical agents than fully differentiated or completely undifferentiated odontoblasts and cells of the ameloblast series (Adachi et al. 1957). The morphology and distribution of incisor lesions in the present case are consistent with injury to a select population of odontoblasts and ameloblasts. Dentin matrix alteration was likely a secondary change due to loss of dentin producing odontoblasts and not due to a disturbance in the calcification process of dentin (no associated changes in serum calcium or phosphorus were observed). Similar microscopic changes in rodent incisor teeth affecting odontoblasts and/or ameloblasts are known to be induced by a variety of cytotoxic agents such as vincristine (Stene 1979), vinblastine (Mikkelsen 1978), colchicine (Nogueira Tde, Stene, and Koppang 1981), and 5-fluorouracil (Kim 1982). In most of these studies, alterations in dentin matrix were described as a reparative response secondary to the cytotoxic effect to preodontoblasts, the most susceptible population of odontoblasts.
Lastly, the presenter discussed the significance of rodent tooth findings for human health risk. Incisor findings in the rat likely reflect specificity to growing teeth in the rats. There was no evidence of effects on nongrowing, molar teeth in the rodents. Similar findings were not observed in the incisors of nonrodent species (two weeks and one-month repeated dose toxicity study). Unlike in the rat, odontogenic tissues do not remain functional during adult life in humans; hence, rat teeth findings are not relevant to the adult human population. Although the mature dentin in humans ceases to grow, in children the dentition is in a growth phase that starts in utero and lasts into the second decade (around 13 years) of their life. Therefore, the significance of rodent incisor findings should be carefully evaluated for the pediatric patient population. A few marketed drugs such as Vincristine and Vinblastine have been approved for treatment of pediatric malignancy and are associated with degeneration and necrosis of odontoblasts/ameloblasts in the rodent incisors. Abnormalities associated with dental development such as dental disturbances, delayed tooth eruptions, and craniofacial effects have been observed in surviving pediatric patients treated for acute lymphoblastic leukemia. These children required aggressive dental follow-up. Careful surveillance (clinical and radiographic) is recommended to reduce the morbidity associated with dental sequelae of chemotherapy and radiation (Kaste et al. 1997).
In summary, careful clinical, gross and microscopic examinations of teeth are important for toxicity studies. The teeth should be examined in longitudinal section for focal lesions. Considering the dynamic nature of ameloblasts and odontoblasts, appropriate recovery times (6 to 8 weeks) should be used to study the reversibility of the rodent incisor findings.
Proliferative Thyroid Lesions in Fish
Dr. Shannon M. Wallace (Experimental Pathology Laboratories, Inc., Sterling, VA) presented 4 cases representative of varying degrees of proliferative thyroid lesions in bony fish. Because the majority of symposium attendees do not typically evaluate fish histopathology, this exercise was meant to be novel as well as provide a comparative review to mammalian proliferative thyroid lesions. The author would like to thank Katrina Murray (Zebrafish International Resource Center) and Dr. Jeff Wolf (Experimental Pathology Laboratories, Inc., Sterling, VA) for granting permission to present these cases.
All cases were zebra fish (wild type, mutant, and transgenic) that presented from the same facility. Within this facility, visible masses were observed on or near the jaw. Approximately 10% of 220 tanks were affected, with 25% of fish affected per tank. Fish were not exposed to any known xenobiotic and there was no apparent sex or tank predilection.
For each case, a series of low- to high-magnification images were shown to the audience. Only high magnification images are included here, as they depict the most distinguishing features of each described thyroid lesion.
The first case for voting was a parasagittal section of a zebra fish with a moderately cellular mass anterior to the eye, dorsal to the jaw, and protruding from the normal body contour. The mass was well demarcated and composed of colloid-filled follicles lined by basophilic cuboidal to columnar epithelial cells with oval basophilic nuclei (Figure 3A). The voting choices and results were as follows: normal (16%), simple follicular hyperplasia (15%), nodular follicular hyperplasia (23%), ectopic follicular hyperplasia (29%), adenoma (12%), carcinoma (3%), and other (0%). The audience majority was correct in their voting, as this case was representative of ectopic thyroid follicular cell hyperplasia.
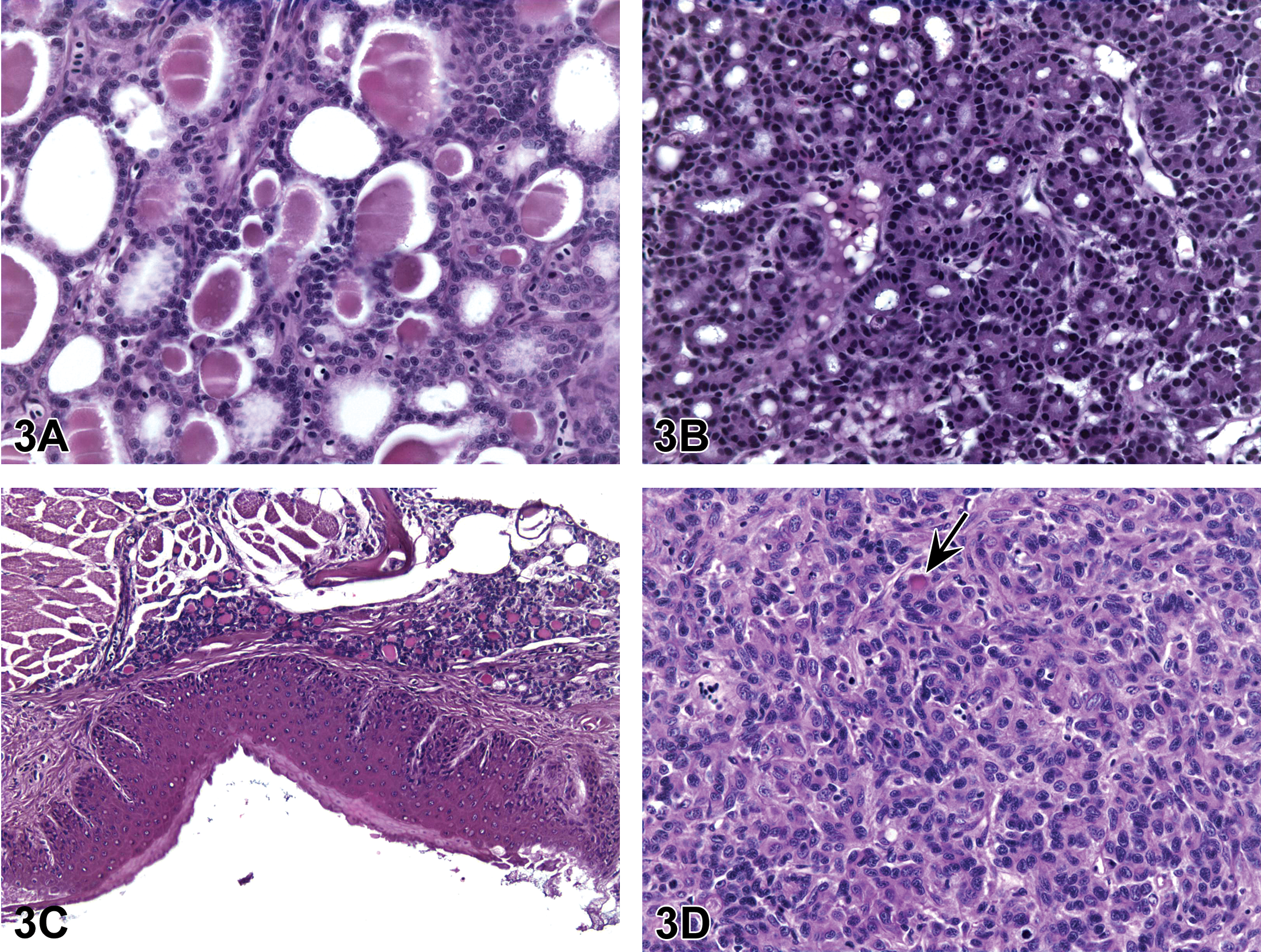
(A–D) Microscopic features of various proliferative thyroid lesions in bony fish. (A) is an image of the thyroid lesion presented as case 1. This is representative of ectopic follicular hyperplasia, with increased numbers of well-differentiated follicles. (B) is an image of the thyroid lesion presented as case 2. This lesion is representative of an adenoma, with more cellular atypia than the lesion in (A), but still identifiable as thyroid. Images of normal thyroid follicles within the muscle and connective tissue surrounding the esophagus (C) were presented as case 3. (D) is an image of the thyroid lesion presented as case 4. This is representative of carcinoma with cellular pleomorphism, mitotic figures, and rare indicators of tissue of origin; there is one visible colloid-filled follicle (arrow) in the image. H&E = hematoxylin and eosin.
The second case for voting was a parasagittal section of a zebra fish with a densely cellular, well-demarcated expansile mass ventral to the eye and caudal to the jaw that protruded from the normal contour of the ventral midline. The mass was composed of numerous follicle-like structures that were devoid of colloid and lined by cuboidal epithelial cells with basophilic oval nuclei (Figure 3B). The voting choices and results were as follows: normal (1%), simple follicular hyperplasia (19%), nodular follicular hyperplasia (11%), ectopic follicular hyperplasia (21%), adenoma (39%), carcinoma (6%), and other (1%). Again, the audience majority was correct in their selection, as this case was representative of a thyroid follicular cell adenoma.
The third case for voting was a parasagittal section of a zebra fish with loosely arranged and dispersed, often distended, colloid-filled follicles lined by flattened basophilic columnar epithelium that was situated between adipose tissue, muscle fibers, and intestinal submucosa (Figure 3C). The voting choices and results were as follows: normal (21%), simple follicular hyperplasia (3%), nodular follicular hyperplasia (3%), ectopic follicular hyperplasia (44%), adenoma (2%), carcinoma (25%), and other (0%). The audience majority was incorrect in its selection for this case, as this was representative of normal thyroid tissue in bony fish.
The fourth case for voting was a parasagittal section of a zebra fish with a densely cellular, well-demarcated mass that was located ventral to the eye and caudal to the jaw and protruded from the normal contour of the ventral midline. The mass was poorly differentiated, composed of sheets and packets of pleomorphic basophilic cells that had basophilic atypical nuclei and frequent mitoses, and rarely formed rudimentary follicle-like structures (Figure 3D). The voting choices and results were as follows: normal (1%), simple follicular hyperplasia (1%), nodular follicular hyperplasia (1%), ectopic follicular hyperplasia (1%), adenoma (3%), carcinoma (93%), and other (0%). The audience majority was overwhelmingly correct in its diagnosis of thyroid follicular cell carcinoma.
The presentation then focused on features of thyroid tissue in fish, progressing from normal anatomy to proliferative lesions that included hyperplasia, adenoma, and carcinoma. This was followed by a discussion on the causes of tissue proliferation, during which comparisons were made to other fish and mammalian species. In many commonly studied teleost (bony fish) species, thyroid follicles are typically located in the pharyngeal region near the ventral aorta. However, cells capable of forming thyroid tissue are widespread anatomically. Not only have thyroid follicles been found in nearby muscle, cartilage, bone, and gill, in certain species and under certain conditions, they have also been observed in areas such as the ocular choroid, kidney, spleen, intestine, liver, and heart. This distribution was demonstrated in case 3 (Figure 3C and D), in which thyroid follicles surrounded the esophagus (Figure 3C), intestine, and liver. There are additional anatomical and functional differences between mammals and fish; for example, the thyroid gland in bony fish is an unpaired and unencapsulated gland, and the lack of a capsule may contribute to indiscriminate distribution of tissue that is capable of forming thyroid. Cartilaginous fishes, such as skates, rays, and sharks, are exceptions to this rule as they have an encapsulated thyroid gland (Crow, Luer, and Harshbarger 2001). Additionally, fish thyroids lack parafollicular cells (C-cell function is instead relegated to cells of the piscine ultimobranchial gland). Histological similarities between fish and mammalian thyroids include the general pattern of round to oval colloid-filled follicles that are lined by a single layer of squamous to cuboidal epithelium, the height of which is dependent on the level of thyroid-stimulating hormone (TSH) activity. Some fish species may have a different follicular epithelial appearance based on gender; for instance, the follicles of laboratory-reared male Japanese medaka (Oryzias latipes) are often lined by tall cuboidal to columnar epithelial cells, unlike the flattened epithelium of their female counterparts (Fournie et al. 2005).
Fournie and colleagues (2005) examined 97 archived cases of proliferative thyroid lesions in teleosts and proposed diagnostic criteria for such lesions; those criteria form the basis for the following discussion. Follicular cell hyperplasia presents as an increase in the number of follicles, some or all of which usually contain colloid, without surrounding tissue compression. Follicular cell hyperplasia is categorized as simple, nodular, or ectopic. Simple hyperplasia involves a proliferation of thyroid tissue at or near the anatomic site where it normally occurs (i.e., the pharyngeal region). Hyperplastic thyroid follicles are lined by variably basophilic cuboidal to columnar epithelial cells, are well differentiated, and do not form nodules or masses. Nodular hyperplasia is histologically similar to simple hyperplasia (i.e., follicles lined by a single layer of flattened to cuboidal cells) but occurs within a discretely circumscribed area and can be a single structure or multinodular. Ectopic hyperplasia can occur in a variety of extrapharyngeal sites. Proliferation may be extensive, and follicular tissue can occupy large areas of an organ, but the follicles are still well-differentiated. Gross and histologic examples of nodular hyperplasia in a trout were shown to the audience (Fournie et al. 2005).
Follicular cell adenomas occur as well-demarcated expansile masses that often arise within areas of follicular cell hyperplasia. Large adenomas can have a “pseudocapsule” of compressed stroma and collapsed follicles of adjacent normal thyroid. Compared to hyperplastic lesions, adenomas are more structurally intricate and demonstrate a moderate amount of cellular atypia. They can have papillary or solid growth patterns, which are features not present in hyperplastic thyroids. Adenomas with solid growth patterns may be comprised of cells that are tinctorially different from those of the surrounding normal thyroid. Examples of solid and papillary adenomas were shown to the audience (Fournie et al. 2005).
Follicular cell carcinomas are characterized by cellular pleomorphism, nuclear atypia, and/or a high mitotic index and often exhibit a disorganized growth pattern. Atypical features in piscine carcinomas can include piling up of multiple cell layers with the appearance of stacked nuclei (Fournie et al. 2005). Some may be observed as discrete masses, but most have indistinct boundaries. Although local invasion and/or distant metastasis can occur, this is not required for the diagnosis of carcinoma in fish. In fact, because thyroid tissue may become hyperplastic or adenomatous in ectopic locations, invasion and metastases are not reliable criteria for malignancy. Consequently, diagnoses of malignancy are dependent on the degrees of cellular atypia and disorganized growth. Categories of carcinomas include well differentiated and poorly differentiated. Well-differentiated carcinomas have atypical features but are still clearly recognizable as thyroid tissue, whereas poorly differentiated carcinomas have few characteristic features of thyroid tissue (e.g., they may contain few or no identifiable follicles) and can be difficult to differentiate from other tumor types.
Thyroid proliferation can occur due to a variety of reasons, including iodine deficiency, poor water quality, seasonal and environmental influences, and genetic susceptibility (Fournie et al. 2005). Iodine deficiency was the inciting cause in the initial cases that were presented. Upon correction of the deficiency, lesions in the affected population regressed to hyperplastic and normal thyroid tissue over time. Poor water quality can contribute to thyroid lesions in animals through increased nitrate levels. High doses of nitrate have caused goiter and depressed serum thyroxine in rats and sheep (Hinther et al. 2012). Nitrate (and nitrite) competes with iodine uptake, transport, and retention in the thyroid gland, impairing thyroid hormone synthesis which results in increased TSH and enlarged and/or proliferating follicles. Nitrate elevation can occur in aquatic animal enclosure systems due to fish waste, rotting food, and decomposing plant matter, while environmentally, increases can occur as a result of fertilizer usage, animal production waste, and waste water treatment discharges (Hinther et al. 2012). Seasonal and environmental factors can also impact the thyroid gland in fish. Studies have shown that, in grouper in the Suez Gulf, Egypt, water temperature can affect epithelial height and colloid content in fish (Abbas et al. 2012). The authors found that the follicular epithelium was tall in warm months, at which point follicles contained abundant colloid, while the opposite was true in colder months. Additionally, T3 and T4 levels were increased in summer months compared to decreased levels from October through December. Exposure to goitrogenic substances can affect the thyroid via inhibition of iodine metabolism (thiocyanates, perchlorates) or through suppression of thyroid hormone production or secretion (thiouracil and imidazole derivatives), resulting in increased TSH and subsequent hypertrophy and hyperplasia of follicular cells (Todd 1986).
The general incidence and behavior of thyroid proliferative lesions in fish were then discussed. Review of the literature suggests that the incidence of spontaneous thyroid proliferative lesions in fish is likely low and varies by species. The true incidence of such lesions is difficult to gauge because lesions are typically investigated when a problem is observed and in many instances may remain subclinical. Often, an inciting cause (e.g., goitrogen exposure) is suspected but this is not always easily confirmed. In fish, thyroid proliferations are moderately easy to induce and hyperplastic lesions tend to be more common than tumors. The historical literature may overestimate the number of tumors due to the misinterpretation of ectopic follicular cell hyperplasia as metastasis (Fournie et al. 2005). The functional secretory capacity of thyroid lesions in fish is not often investigated as it is in mammalian species, nor is molecular testing routinely performed. Theoretically, immunohistochemial or molecular testing might help to confirm that thyroid proliferative lesions diagnosed in fish as malignant based on morphologic features are actually true carcinomas that possess multiple genetic changes associated with cancer disease progression and capability of sustainable autonomous growth. Although such diagnostic markers for thyroid tumor malignancy have been identified in humans (Saggiorato et al. 2001; Wiseman et al. 2007), this is still a developing area of research for fish tumors (Mione and Trede 2010).
The presentation then transitioned to discussions of comparative pathology between fish and mammals including diagnostic criteria for ectopic thyroid tissue, hyperplasia, adenoma, and carcinoma. Ectopic thyroid tissue also occurs in mammals in various locations among different species. Example locations include the tongues of cats (Patnaik, Peterson, and Hidgon 2000), anywhere between the tongue and cranial mediastinum in dogs (Stephens, Saunders, and Jaenke 1982), and in the duodenal submucosa and adrenal gland in humans (Noussios et al. 2011). Histologically, hyperplasia in fish and mammals is generally similar. As in fish, adenomas in mammals can present as single or multiple masses that compress adjacent tissue and have nuclear crowding and papillary patterns. However, in contrast to bony fishes, partial to complete capsule formation tends to occur in mammalian adenomas. Adenomas and adenomatous hyperplasia occur commonly in older cats, and such lesions are often capable of secreting elevated levels of thyroid hormone. Adenomatous hyperplasia in cats can be multinodular, but hyperplastic nodules are not encapsulated and do not compress adjacent thyroid tissue. Carcinomas occur more commonly in dogs than cats. Adenocarcinomas in mammals are characterized by follicular or papillary patterns, or some combination thereof, and feature cellular pleomorphism. Hallmarks of thyroid carcinomas in mammals include capsular and adjacent tissue and/or blood vessel invasion. Again, this differs from the situation in fish in which the primary criterion for malignancy is cellular atypia rather than invasiveness.
At the conclusion of the presentation, an audience member questioned why it is important to specifically categorize lesions that occur distant from the pharyngeal cavity as ectopic, when it is recognized that thyroid tissue in fish can be found in extrapharyngeal locations. In response, the designation of ectopic is used because, diagnostically, it helps to better distinguish benign proliferative lesions that develop in situ in various locations from carcinomatous metastases, which probably occur extremely rarely, if at all.
Dr. Wallace concluded her presentation with helpful take-home points. In bony fish, the diagnosis of carcinoma is based on cellular atypia and disorganized growth rather than invasion or metastasis, as in mammals. However, in cartilaginous fishes, capsular invasion may be a valid criterion for malignancy due to the presence of a thyroid capsule.
Intestinal Pseudo-obstruction in a Dog
Dr. Ashley Talley (Charles River Laboratories, Inc., Durham, NC) presented a companion animal case submitted to the surgical pathology service at North Carolina State University (NCSU). Multiple full thickness biopsies of small intestine were submitted from a 5-year-old, spayed, female beagle dog with several days history of anorexia, abdominal distension, and loose stool. Pertinent clinical pathology findings included mild hypoalbuminemia with corresponding low total calcium (normal ionized calcium), mild hypocobalaminemia, and low serum trypsin-like immunoreactivity. By abdominal radiography and upon exploratory laparotomy, the small intestine was diffusely, severely dilated with no evidence of mechanical obstruction.
Small intestinal biopsies were obtained. Low and high magnification photomicrographs were presented (Figure 4A–D), which featured a cellular infiltrate, primarily within the inner circular layer of the tunica muscularis (Figure 4A), with no significant histologic lesions within the mucosa (Figure 4B). The mononuclear cells dissected between degenerative myofibers of the inner circular muscle layer (Figure 4C). The cellular infiltrate was mixed mononuclear, and the affected myofibers were atrophied with vacuolated or hypereosinophilic sarcoplasm (Figure 4D). The voting choices and results were as follows: lymphoma (2.86%), dysautonomia (5.71%), intestinal leiomyositis (60.95%), inflammatory bowel disease (11.43%), lymphangiectasia and lymphangitis (14.29%), sarcoidosis (2.86%), and other (1.90%). Dr. Talley agreed with the majority of the participants that intestinal leiomyositis was the favored diagnosis.
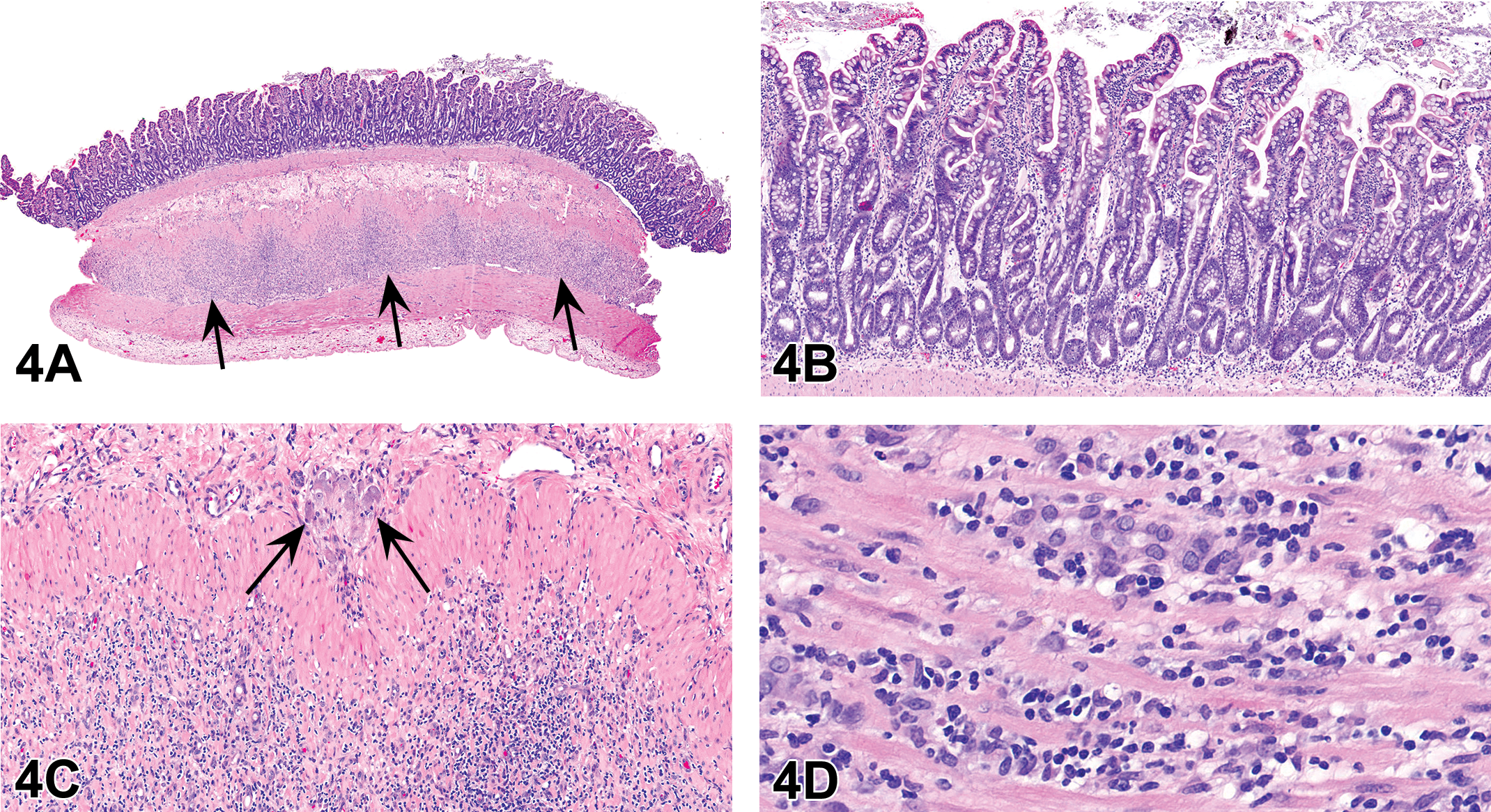
(A–D) Jejunal biopsy from a 5-year-old, spayed, female beagle dog with severe intestinal hypomotility. At low magnification (A), the inner circular layer of the tunica muscularis contains a dense cellular infiltrate (arrows) that extends minimally into the outer longitudinal layer of the muscularis. In the mucosa (B), there are no significant histologic changes. At higher magnification (C), mononuclear cells dissect between degenerative myofibers of the inner circular muscle sparing enteric ganglia (arrows) and a thin band of smooth muscle deep to the submucosa. (D) shows that the cellular infiltrate within the tunica muscularis is composed of mixed mononuclear cells and the affected myofibers are atrophied with vacuolated or hypereosinophilic cytoplasm. (E–H) Immunohistochemical characterization of mononuclear cells (E–G) and smooth muscle (H) within the tunica muscularis. Mononuclear cells are primarily CD3-positive T lymphocytes (E) and Iba1-positive histiocytes (G) with fewer CD20-positive B lymphocytes (F). (H) highlights the loss of α smooth muscle actin staining in the inner circular muscle (top) compared to the outer longitudinal muscle (bottom). H&E = hematoxylin and eosin.
Intestinal leiomyositis is a rare cause of chronic intestinal pseudo-obstruction (CIPO), which is characterized by a lack of normal intestinal peristalsis (ileus) in the absence of mechanical obstruction (Jain 2009; Washabau 2013). CIPO is subdivided into myopathic and neuropathic forms and those affecting interstitial cells of Cajal or neurohormonal peptides (Jain 2009). The myopathic group includes both spontaneous and familial forms, which are primarily noninflammatory with the exception of intestinal leiomyositis (Jain 2009). In dogs, the clinical features of intestinal leiomyositis include vomiting, regurgitation, diarrhea, anorexia, and weight loss (Zacuto et al. 2016). The most common clinical pathology findings include hypoalbuminemia, hypocholesterolemia, and neutrophilia (Zacuto et al. 2016). Histopathologically, there is infiltration of the tunica muscularis by mononuclear cells along with myofiber degeneration and variable fibrosis (Zacuto et al. 2016). The small intestine is most commonly affected; however, involvement of the stomach and colon is also reported (Couraud et al. 2006; Johnson et al. 2007; Zacuto et al. 2016). In affected patients, similar lesions have also been described at distant sites, such as the urinary bladder and smooth muscle of the eyelid (Johnson et al. 2007; Zacuto et al. 2016). Prokinetic and immunomodulatory drugs are the mainstay of treatment, although successful management is rare (Zacuto et al. 2016). Common comorbidities include small intestinal bacterial overgrowth and aspiration pneumonia secondary to vomiting and regurgitation. The pathogenesis of intestinal leiomyositis is unknown, although autoimmunity is suspected (Ruuska et al. 2002; Zacuto et al. 2016).
The salient histologic features of the intestinal biopsies were discussed (Figure 4A–D). The mononuclear inflammatory cell infiltrate was primarily composed of CD3-positive T lymphocytes and Iba1-positive histiocytes with fewer CD20-positive B lymphocytes (Figure 4E–G). These findings are similar to human and canine cases, which are characterized by a predominance of T lymphocytes (Ruuska et al. 2002; Zacuto et al. 2016). In addition, α-SMA immunohistochemistry highlighted the loss of myofibers among the inflammatory cell infiltrate (Figure 4H). No microorganisms were identified with histochemical stains (Grocott’s methenamine silver, Gram stain, Ziehl–Neelsen acid fast; data now shown). Primary differentials included lymphoma and neuropathic forms of CIPO, such as dysautonomia. Lymphoma was excluded due to the mixed cell population, apparent targeting of myofibers, and absence of mucosal involvement. Intact enteric ganglia precluded the diagnosis of neuropathic CIPO. Systemic disease, such as muscular dystrophy or systemic lupus erythematosis, was also considered but was thought to be unlikely given the lack of supporting clinicopathologic findings.
Discussion ensued regarding the preferred diagnosis of this lesion in a regulatory setting. Since “-itis” terms are not generally used in this context, the preferred diagnosis was “inflammation, mononuclear, smooth muscle, muscularis.” Given the uniqueness of the lesion, attendees encouraged a thorough histologic description when reporting these findings. An inquiry was made regarding circulating anti-smooth muscle antibodies, which have been detected in humans but were not evaluated in this case or previous canine cases (Ruuska et al. 2002).
Following diagnosis and continued clinical decline, the dog presented here was euthanized and no postmortem examination was performed. Dr. Talley concluded with the following take-home points: (1) intestinal leiomyositis is a rare cause of CIPO, (2) full thickness intestinal biopsies are necessary for antemortem diagnosis, and (3) the pathogenesis is unknown, although autoimmunity is suspected.
Common Gross Findings in Fish Larvae during Chemical Screening
Dr. Debabrata Mahapatra (Integrated Laboratory Systems, Inc. [ILS], Research Triangle Park, NC) presented 2 cases in different species of fish larvae. These cases were selected for representative gross morphological lesions that are routinely encountered during developmental toxicity screening assays in fish models. Commonly used models include Japanese medaka (Oryzias latipes) and zebra fish (Danio rerio) larvae. Dr. Mahapatra acknowledged Dr. Seth W. Kullman (Prof. of Toxicology at NCSU), Megan Knuth, and Atlee Watson (Kullman Lab members) for assistance with fish embryo collection and image acquisition. The author also acknowledged the ILS pathology team members for their support.
The presentation began with a general introduction to toxicity screen assays, utility of fish models, and a brief methodology. Zebra fish, and more recently Japanese medaka, have become popular fish models for assessing developmental toxicity as an alternative to the use of rats and mice. The ease of detection of morphological anomalies through transparent embryos, feasibility of genetic manipulation, and well-characterized developmental stages are just some notable advantages that both models offer compared to their rodent counterparts (Carney et al. 2006; Hanno, Oda, and Mitani 2010). Husbandry practices and assay protocols differ considerably depending on the fish model being used and study design parameters. However, basic steps usually involve spawning and egg collection, egg cleaning, staging, and dosing, preferably in the blastula stage (4–6 hr postfertilization; Figure 5A). Thereafter, embryos are allowed to hatch (48–96 hr postfertilization) into larvae at which time morphological assessment is performed as a routine end point. Behavioral alterations and/or changes in gene expression could be used as additional end points for a given assay along with morphology.
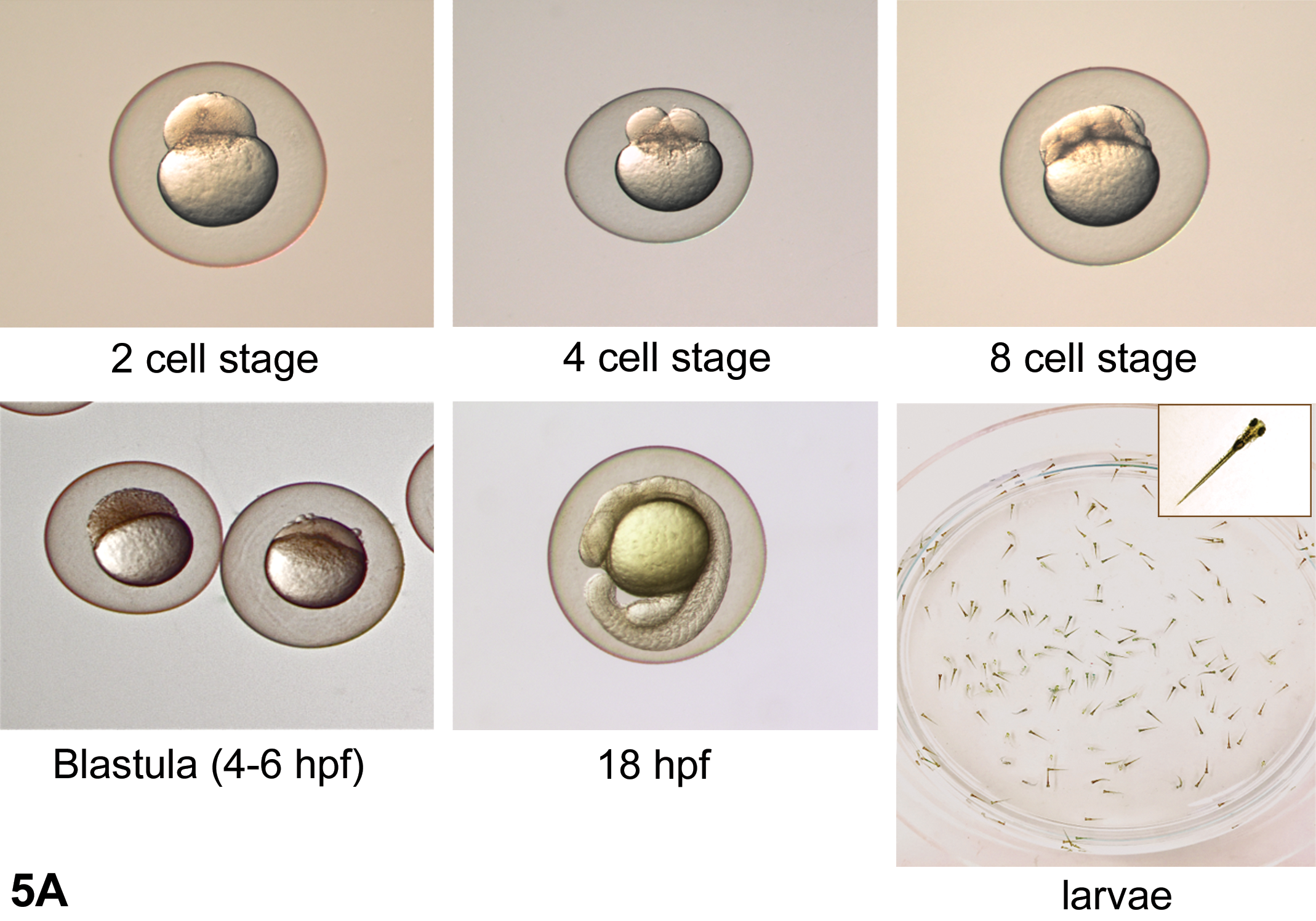
(A) Brief overview of the developmental stages from the 2-cell stage embryo to larvae in zebra fish. Embryos in the blastula stage, at 4 to 6 hr postfertilization (hpf), are commonly used for dosing in chemical screening assays. Morphologic assessment for gross anomalies are usually conducted post hatching (48–72 hpf) in the larvae, but this may vary depending on assay requirements. (B and C) Gross morphological anomalies in a 12- to 14-day-old Japanese medaka larva (B) after treatment with a farsenoid X receptor antagonist, presented as case 1. Compared to the control larva, the treated larva shows stunted growth, axial skeletal and craniofacial deformities, and underdeveloped swim bladder. Gross morphological anomalies in a 5-day-old zebra fish larva (C) after treatment with a vitamin D receptor antagonist, presented as case 2. Compared to the control larva, the treated larva shows pericardial edema, yolk sac edema, underdeveloped swim bladder, and axial skeletal and craniofacial deformities.
The first case was from a 12- to 14-day-old Japanese medaka larva that had been treated with a farsenoid X receptor (FXR) antagonist. The photographs presented to the audience consisted of a control and a treated larva (Figure 5B). The voting choices and results were (1) delayed organogenesis, deformed yolk sac, eye abnormalities, and pigmentation defects (2%); (2) stunted growth, axial skeletal deformities, craniofacial deformities, and underdeveloped swim bladder (32%); (3) both 1 and 2 (66%), and (4) none of the above (0%). A majority of the audience voted for choice (3) and Dr. Mahapatra explained why choice (2) was the correct diagnosis. While delayed organogenesis and deformed yolk sac were possibilities, a confirmatory diagnosis required follow-up histopathology, specialized staining techniques, or gene expression studies. Additionally, there were no apparent gross eye abnormalities or pigmentation defects. An audience member asked whether or not there were functional assays to test neuronal defects. In response, Dr. Mahapatra mentioned that using immunohistochemistry or in situ hybridization techniques are common useful tools for identification and verification of neuronal defects. Also, a variety of functional assays are available that utilize behavior as an end point to identify treatment-related alterations in basic motor functions, learning, and memory in live embryos or adult fish (Tierney 2011).
The second case was from a 5-day old zebra fish larva that had been treated with a vitamin D receptor (VDR) antagonist. The photographs presented to the audience consisted of a control and a treated larva (Figure 5C). The voting choices and results were (1) pericardial edema, craniofacial deformities, yolk sac edema, axial skeletal deformities, and underdeveloped swim bladder (49%); (2) eye abnormalities, pigmentation defects, delayed organogenesis, and neuronal defects (3%); (3) both 1 and 2 (46%); and (4) none of the above (2%). A majority of the audience opted for choice (1) which was the presenter’s preferred diagnosis.
Discussion for both cases ensued with the question of whether the audience thought that these lesions were specific for their respective test article. Both cases were different in terms of usage of fish model, larval age-groups, test article used, and their respective target nuclear receptors. Despite these differences, there were findings that were common to both cases. These included craniofacial deformities, axial skeletal deformities, and underdeveloped swim bladder. Thereafter, photomicrographs were presented to the audience that included two zebra fish larvae treated with different classes of aryl hydrocarbon receptor (AHR) agonists. These lesions included craniofacial and skeletal deformities, as well as pericardial and yolk sac edema. This provided evidence for the nonspecificity of lesions presented earlier in cases 1 and 2 with respect to receptor involvement (i.e., FXR or VDR). Moreover, larval lesions in fish specific for alteration of either FXR or VDR from chemical exposures have not been reported. This further brought up the question as to whether these lesions were specific for another nuclear receptor. However, these lesions closely mimic those caused by the induction of AHR, indicating that they are not receptor-specific lesions.
AHR is a crucial transcription factor involved in a variety of integral cellular activities such as cell cycle regulation, adhesion, migration, cell pluripotency, and stemness. AHR has a widespread tissue expression that is conserved across phyla and its biological activity is under strict transcriptional control. Disruption of this controlled signaling contributes to system-wide adverse effects in vertebrates and the underlying mechanisms are still poorly understood (Carney et al. 2006; Stockinger et al. 2014; Mulero-Navarro and Fernandez-Salguero 2016). A variety of exogenous ligands including halogenated aromatic hydrocarbons, polycyclic aromatic hydrocarbons, polychlorinated biphenyls, and polychlorinated dibenzo-p-dioxins are persistent environmental contaminants. These compounds pose a risk to human and environmental health and toxic effects are mediated through their ability to bind and induce AHR in a variety of mammals, birds, and fish (Henry et al. 1997; Carney et al. 2006; Billiard et al. 2006). Induction of AHR (AHR2 and AHR2a in zebra fish and medaka, respectively) by 2,3,7,8-tetrachlorodibenzo-p-dioxin has been shown to affect the cardiovascular system causing pericardial edema, hemorrhage, and slow heart rate in fish larvae (Henry et al. 1997; Wisk and Cooper 1992). Lesions also manifest in the skeletal system and are associated with craniofacial and axial skeletal deformities. Additionally, overall growth retardation, arrested swim bladder development, and pigmentation defects have been reported in fish larvae (Henry et al. 1997; Prasch et al. 2003). Exposure of zebra fish to dioxin during early embryonic life or during sexual development has also been reported to cause malformations and increased mortality in subsequent generations in adults besides cranial and axial skeletal defects (Baker, Peterson, and Heideman 2013), thus demonstrating transgenerational effects of the toxicant. Induction of AHR results in upregulation of cytochrome p450 phase I metabolizing enzymes CYP1A and CYP1B1 in zebra fish and CYP1A1_ORYLA in Japanese medaka, respectively (Carney et al. 2006; Hanno, Oda, and Mitani 2010). Hence, activation of CYP enzymes can act as biomarkers for exposure to environmental toxicants, capable of inducing AHR in fish and mammals alike.
A series of questions from the audience included cataloging individual defects in larvae versus lumping of terms, characterization of craniofacial defects, and spontaneity of lesions in larvae. In response, the speaker briefly outlined the protocol for morphological assessment and stressed the importance of reporting individual lesions versus lumping them together since not all lesions discussed above are manifested in affected larvae at the same time. Also, incidences and degrees of severity that occur in these larvae depend on variations in genetic susceptibilities among individuals. Characterization of morphological lesions, such as craniofacial defects, is not routinely performed unless specified by the study protocol. These lesions are treatment related and not spontaneous in nature.
The overarching goal of this presentation was to make the audience aware of these commonly observed lesions in chemical screening assays in fish models. These lesions are specific for AHR induction and must not be misinterpreted as lesions specific for other nuclear receptors or spontaneously occurring lesions. It is advisable to confirm the involvement of AHR receptor induction by performing downstream assays such as messenger RNA (mRNA) expression of one or more of the CYP enzymes (CYP1A, CYP1B).
What Findings in Otters (Lutra lutra) Can Tell Us for Nonrodent Tox Path
In his talk “A Mucinous and a Clear Matter,” Matthias Rinke (retired director of pathology and clinical pathology at Bayer AG, Germany) presented cases from two adult male Eurasian Otters (Lutra lutra) with the intention to link these to nonrodent toxicopathology. After asking for the identification of the altered organ in the first slide, only 40% of the responders recognized it as the gallbladder while others thought it to be seminal vesicles (25%) or thyroid gland (11%). After knowing the organ, 65% of the voters chose the right answer of cystic mucinous hyperplasia of the gallbladder (Figure 6A–C). Others chose the proposed more descriptive terms of mucinous hypertrophy/hyperplasia of the gallbladder (17%) or hypertrophy of gallbladder epithelium with cystic dilatation of the glands, inflammation, and debris (12%).
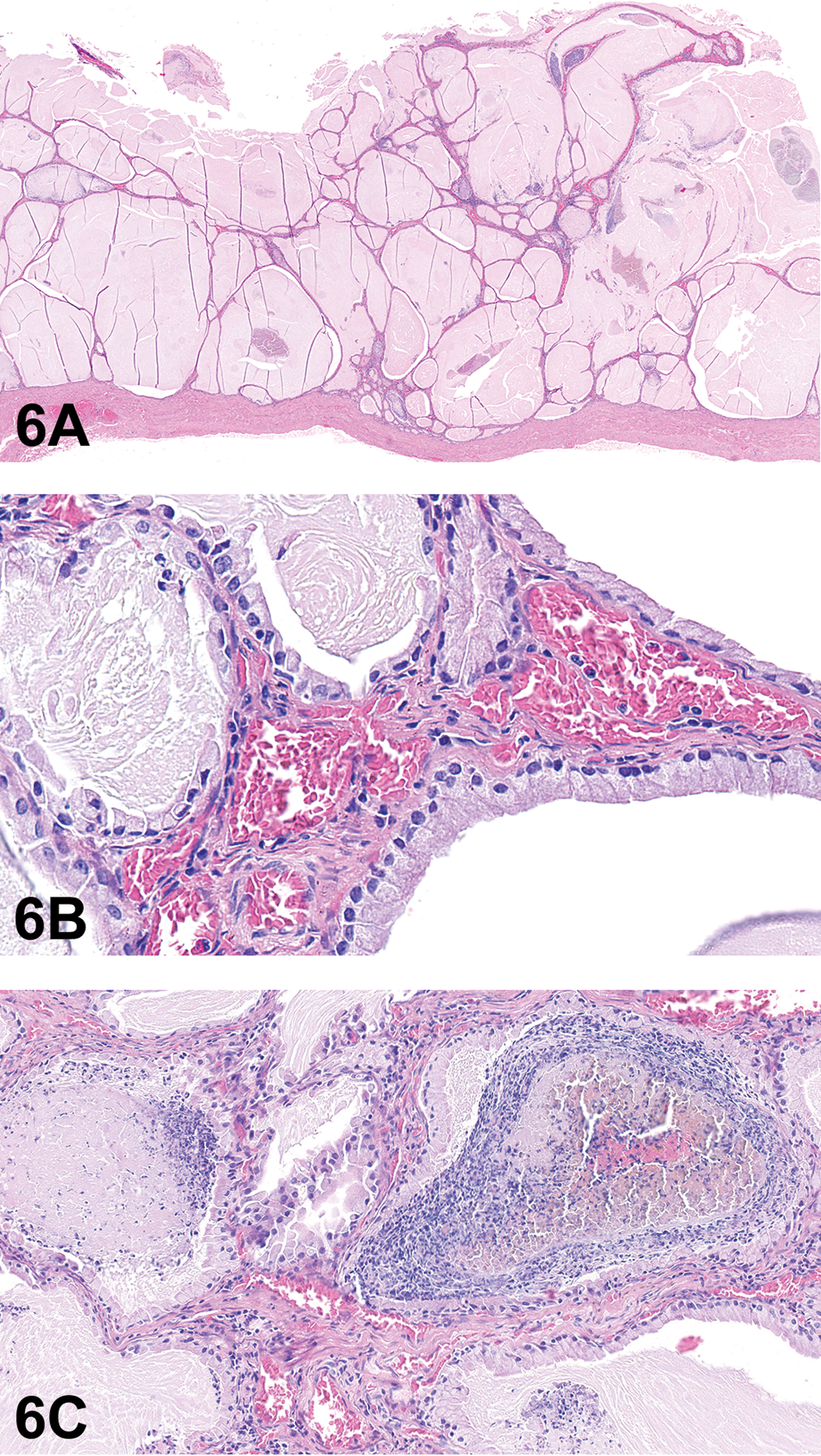
(A) shows the gallbladder of an adult male Eurasian otter presented as case 1, revealing marked cystic mucinous hyperplasia. (B) is the higher magnification of (A) showing mucus production and mainly hypertrophic epithelial cells. Inflammatory reaction and occasional debris can be seen in the lesion (C). (D–G) Edge of a large hepatocellular focus (D and E), clear cell type, in the liver from an adult male Eurasian otter presented as case 2. The lesion is composed of glycogen containing cells and shows only little compression to the surrounding tissue (D). Normal architecture is maintained, and the portal triads are normal in appearance (E). (F), presented as case 3, shows a portal area of an adult male Eurasian otter with slight bile duct hyperplasia and inflammation/fibrosis. There is also a small clear cell focus in the adjacent hepatic tissue. Bile duct hyperplasia with pronounced fibrosis and inflammatory response (G), presumably caused by the bile fluke Pseudamphistomum truncatum, was presented as an additional example. This sample originated from an adult male Eurasian otter found in the wild. H&E = hematoxylin and eosin.
Cystic mucinous hyperplasia is observed in dogs, especially small breeds, including the beagle dog and is characterized by increased numbers of hypertrophic cells with pale cytoplasm and abundant PAS-positive mucus production with areas of chronic inflammatory response and intraluminal debris. This lesion has to be distinguished from hyperplasia of the mucosal epithelium, which is a rare finding with neither copious mucin production nor cystic change (Sato et al. 2012). A case of cystic mucinous hyperplasia of similar appearance has also been described in a ferret, which is a mustelid species like the otter (Reindel and Evans 1987). Ferrets are also used in toxicological studies. Moreover, this lesion is also known to occur in female sheep (Fell, Robinson, and Watson 1983). The question “Which species has this lesion been described in?” was answered correctly by approximately 50% of the voters. A question from the audience about treatment-induced lesions was answered by Dr. Rinke. There is an old literature reference that refers to the administration of progestational compounds to beagle dogs (Mawdesley-Thomas and Noel 1967), which may possibly explain why it is seen in female rather than male sheep.
For his second case, Dr. Rinke presented a 2.5-cm pale nodule in the liver that was regarded as “nodular hyperplasia” or “hyperplasia, nodular, clear cell” by 42% of the voters. Thirty-four percent chose focus of (hepato-)cellular alteration, clear cell, or clear cell focus, while 21% of the votes were for hepatocellular adenoma. The cellular features of the lesion included an increased number of PAS-positive, vacuolated, pale, sometimes hypertrophic cells with “empty” cytoplasm. The nuclei had no specific location within the cells and thus could be clearly differentiated from fat, which has peripheralized nuclei. Due to its size, the focus caused some compression to the surrounding normal tissue, but the portal triads were without any abnormality (Figure 6D and E). Since the hepatic cords were still regularly arranged and showed no right angular distortion or any other signs of autonomous growth, like an increase in mitotic figures, the criteria for adenoma were not fulfilled. According to the current INHAND nomenclature for rodents (Thoolen et al. 2010), this finding would have been classified as “focus of hepatocellular alteration, clear cell type” while diagnostic veterinary pathology would probably call it nodular hyperplasia. The INHAND dog group might have to consider this discrepancy for their final terminology.
For the third case, Dr. Rinke asked “How many diagnoses would you make for SEND?” The lesion showed an area of rarefaction of hepatocyte cytoplasm and a slight hyperplasia of the bile ducts with little fibrosis and inflammatory reaction (Figure 6F). Fifty-three percent of the voters chose the Standardization for Exchange of Nonclinical Data (SEND) terms “focus of hepatocellular alteration, clear cell with hyperplasia, bile duct, focal and with infiltrate/fibrosis, periportal,” and this was the preferred diagnosis. For a small lesion like this, accumulation of glycogen was proposed by Sato and colleagues (2012) for dogs but is not considered to be an appropriate SEND term for the liver. Bile duct hyperplasia represents a common finding in otters and is frequently caused by the biliary fluke Pseudamphistomum truncatum (Simpson et al. 2005; Hildebrand et al. 2011). Infection with this trematode results in marked fibrosis and inflammatory reaction and resembles chronic changes caused by the coccidium Eimeria stiedae in rabbits (Figure 6G).
For the take-home message, Dr. Rinke discussed how liver and gallbladder findings of otters can be similar to those in dogs and other mustelids like the ferret, which is used as a nonrodent laboratory animal species, and that foci of cellular alteration in the liver may be larger in size in nonrodents compared to rodents and should be not misinterpreted as neoplasms.
Select Cases from the Rabbit INHAND Working Group
Dr. Lyn Wancket (NAMSA, Northwood, OH) presented on behalf of the INHAND Rabbit Working Group and acknowledges the Working Group Members and INHAND manuscript coauthors for their review of the presentation and for providing images: Alys Bradley, Chair (Charles River Laboratories, Inc.), Franck Jacques-Antoine Chanut (Sanofi), Begonya Garcia (Charles River Laboratories, Inc.), Margarita Gruebbel (Experimental Pathology Laboratories Inc.), Kate Hughes (University of Cambridge), Osamu Katsuta (Santen Pharmaceutical Co., Ltd.), Elizabeth McInnes (Syngenta), Keith Nelson (Charles River Laboratories, Inc.), Matthias Rinke (Bayer AG, retired), Brett Saladino (Seventh Wave Laboratories), Kenneth Schafer (Vet Path Services, Inc.), and Keisuke Uchida (Asahi Kasei Pharma Corporation).
INHAND is a joint initiative of toxicologic pathology societies from North America, Europe, Great Britain, and Japan. INHAND provides a standardized nomenclature system for diagnostic terms in rats and mice (available online at www.goreni.org, last accessed July 12, 2018). Recently, the focus was expanded to include other common nonclinical species (dog, fish, minipig, nonhuman primate, and rabbit). Nonrodent manuscripts will use the rodent terms as the foundational nomenclature and will then emphasize key species-specific differences in anatomy and lesions. Rabbits (Oryctolagus cuniculus) are often used in vaccine, ocular, medical device, and reproduction nonclinical studies (reproduction studies may not always include histopathology). Commonly used strains include New Zealand White (NZW), New Zealand Red (NZR), NZW × NZR F1 crosses, Dutch Belted (for ocular studies requiring a pigmented iris), and Watanabe (atherosclerosis model).
Dr. Wancket presented two cases from rabbits. Case 1 was tissue with abundant lymphocytes from a young adult female NZW rabbit on a two-month repeat dose vaccine tolerance study (Figure 7A and B). The vast majority of attendees chose “within normal limits” (79%), which was also the preferred diagnosis of the INHAND rabbit coauthors. The tissue was sacculus rotundus (also called the cecal tonsil) and was shown to emphasize the abundant lymphoid tissue in rabbit gastrointestinal tract. Other options receiving votes included lymphoid hyperplasia (14%), lymphoproliferative disease (5%), and lymphosarcoma (2%).
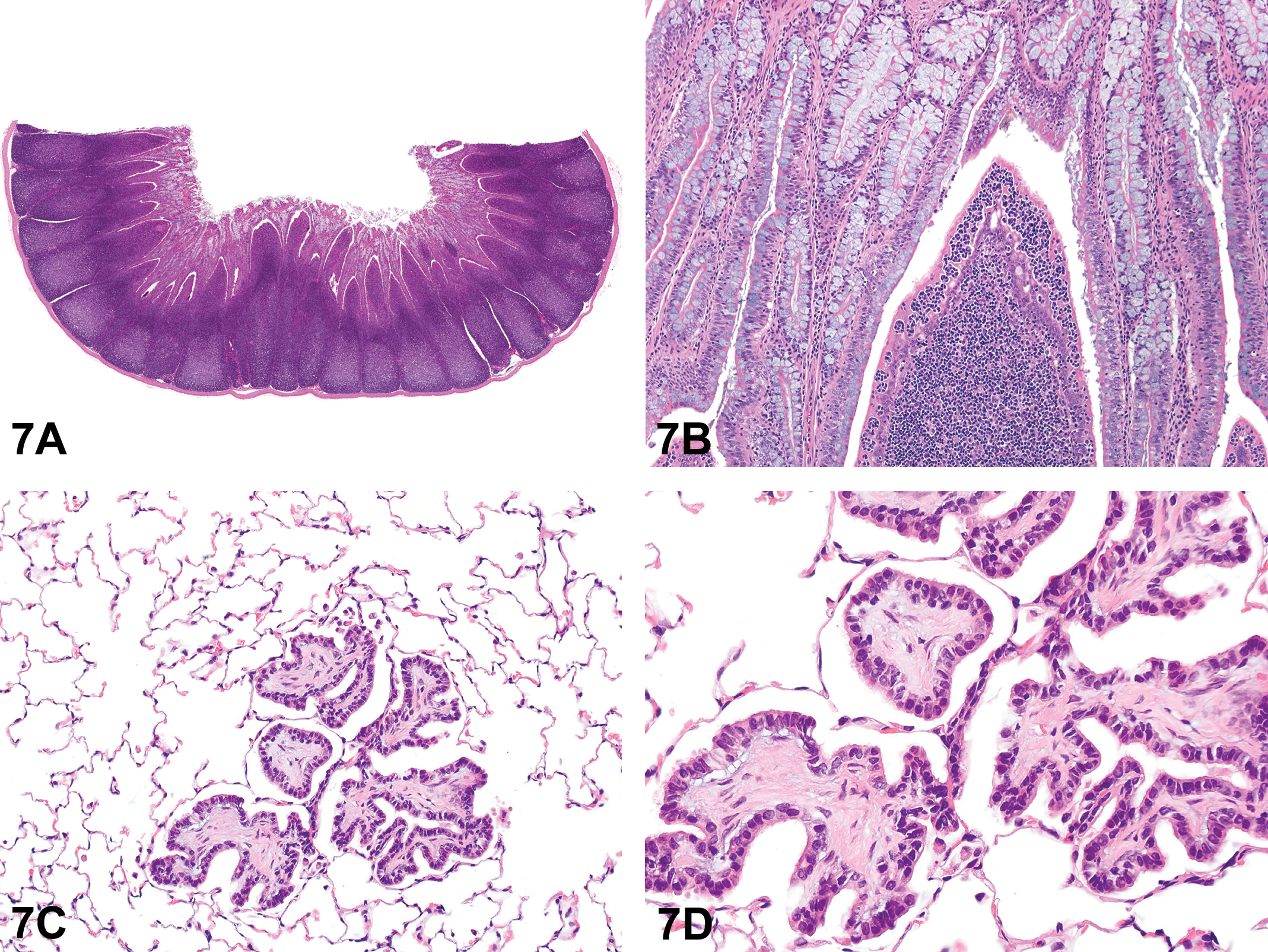
(A–D) Examples of common findings in rabbits from the Rabbit International Harmonization of Nomenclature and Diagnostic Criteria Working Group. Low (A) and high (B) magnifications of the sacculus rotundus from a young adult female New Zealand White (NZW) rabbit on a two month repeat dose vaccine tolerance study presented as case 1. The higher magnification (B) shows intraepithelial lymphocytes common in this location. Low (C) and high (D) magnifications of a section of lung from a young adult female NZW rabbit on a topical ocular study presented as case 2. The tissue contains an expansile connective tissue mass covered by a single layer of cuboidal to short columnar epithelial cells. H&E = hematoxylin and eosin.
Case 2 was a section of lung from a young adult female NZW rabbit on a topical ocular study (Figure 7C and D). The majority of voters selected “hyperplasia, bronchiolo-alveolar” (54%), which was not a preferred diagnosis for a change with a prominent connective tissue core covered with a single layer of epithelial cells. The INHAND rodent nomenclature uses “hyperplasia, bronchiolo-alveolar” to describe areas where epithelial cells are prevalent and are the main component of this type of hyperplasia (as illustrated in Renne et al. 2009). The next most popular choice, fibropapilloma (13%), was also not a preferred diagnosis of the INHAND rabbit coauthors. While similar lesions have been described as fibropapillomas in the lungs of aging rabbits (Cooper et al. 2017), the rabbit INHAND coauthors do not see evidence that this is a neoplastic process. During the open discussion at the end of the talk, there was general agreement that this was a common, nonspecific response to pulmonary injury and should not be labeled as a neoplasm. The third most common option was osseous metaplasia (11% of voters chose either osseous metaplasia or osseous metaplasia, early), which was the preferred diagnosis for the INHAND rabbit coauthors. To support this diagnosis, Dr. Wancket presented several images of osseous metaplasia at various stages of maturation. The choice of osseous metaplasia generated robust discussion. The opinion of multiple attendees was that, while the lesion could eventually undergo osseous metaplasia, it was currently not bone and therefore needed a different diagnosis. The INHAND rabbit coauthors in attendance agreed with the audience that since INHAND terms should be descriptive terms, a term other than osseous metaplasia should be used in the rabbit manuscript. The remaining votes were split between within normal limits (10%), alveolar fibrosis, focal (5%), chronic inflammatory polyp (3%), other (2%), and interstitial fibrosis, focal (1%).
After the cases were presented, Dr. Wancket briefly reviewed unique anatomy, potential pitfalls in anatomy and organ weight interpretations, and common spontaneous findings for rabbits. Rabbit periocular Harderian glands contain white and pink components that are named based on the macroscopic color of each section. In contrast to the macroscopic color of the gland, in H&E-stained sections, the pink gland is clear staining and the white gland is more eosinophilic. Rabbits also have a scent gland on the ventral chin called the mental gland. In the INHAND rabbit coauthors’ experience, this gland is sometimes mistaken for a tumor. Additionally, rabbits have an abundant ovarian interstitial gland. While rabbits are induced ovulators, corpora lutea can develop in animals that have not been bred; this may occur in medical device studies where an intravaginal device is tested.
Attendees were also cautioned about citations that describe a nonglandular portion in the cardiac region of the rabbit stomach. It is the opinion of the INHAND rabbit coauthors that the tissue described is actually tangential sections through the esophageal-gastric junction. One of the members of the INHAND rabbit group serially sectioned the stomach every 1 cm and, upon microscopic evaluation, found that all tissue examined was glandular. Additionally, caution was urged when interpreting certain organ weight necropsy results. Fat infiltration into the thymus occurs even at an early age and pulmonary alveolar edema may skew organ weights.
Several common spontaneous findings in rabbits are caused by their stress response and urinary physiology. As a prey species, rabbits are highly susceptible to stress. The stress response is visible clinically as diarrhea (Bradley 2012) and can manifest in cardiac inflammation/necrosis (Sellers et al. 2017). Additionally, mineralization within the renal pelvis is common due to high urine CaCO3 content, leading to precipitate (“sludge”), uroliths, and sometimes secondary urothelial hyperplasia. Rabbits also develop microscopic changes (similar to rodent chronic progressive nephropathy) that may mask nephrotoxic effects. Changes can include basophilic tubules, thickened basement membranes, and mesangial proliferation in glomeruli. In discussions within the INHAND group, it appears that the incidence and severity of these microscopic changes may vary widely between individual laboratories.
Dr. Wancket ended the talk with a request for images and feedback on the INHAND rabbit manuscript draft when it is available for review to members of the STPs. Dr. Wancket also emphasized the work of the previous organ-specific rodent manuscripts and terms, which are currently available on the STP website (https://toxpath.org/index.asp, last accessed July 12, 2018).
Reproductive Lesion in a Rhesus Macaque
Dr. Caralyn Labriola (Wake Forest University, Winston-Salem, NC) presented a reproductive lesion in a rhesus macaque (Macaca mulatta). The animal was a 9 year, 8-month-old female rhesus macaque of Chinese origin, which had been imported from China at 5 years of age and received 4 gray whole-body irradiation at 6 years of age. The animal presented for menorrhagia with resultant anemia, and therefore, a hysterectomy was performed. Grossly, the entirety of the endometrium at the apex of the uterus was irregularly thickened up to 1.5 mm by a papillary mass (Figure 8A). The reproductive history prior to importation was unknown, and the animal was never pregnant while at Wake Forest University; however, prior pregnancy is presumed due to vascular changes within the myometrium consisting of large profiles of tortuous blood vessels with thick surrounding zones of loose connective tissue. At the time of this publication, the hysterectomy appeared to have been curative for the clinical signs, and the animal is alive without development of other neoplasms.
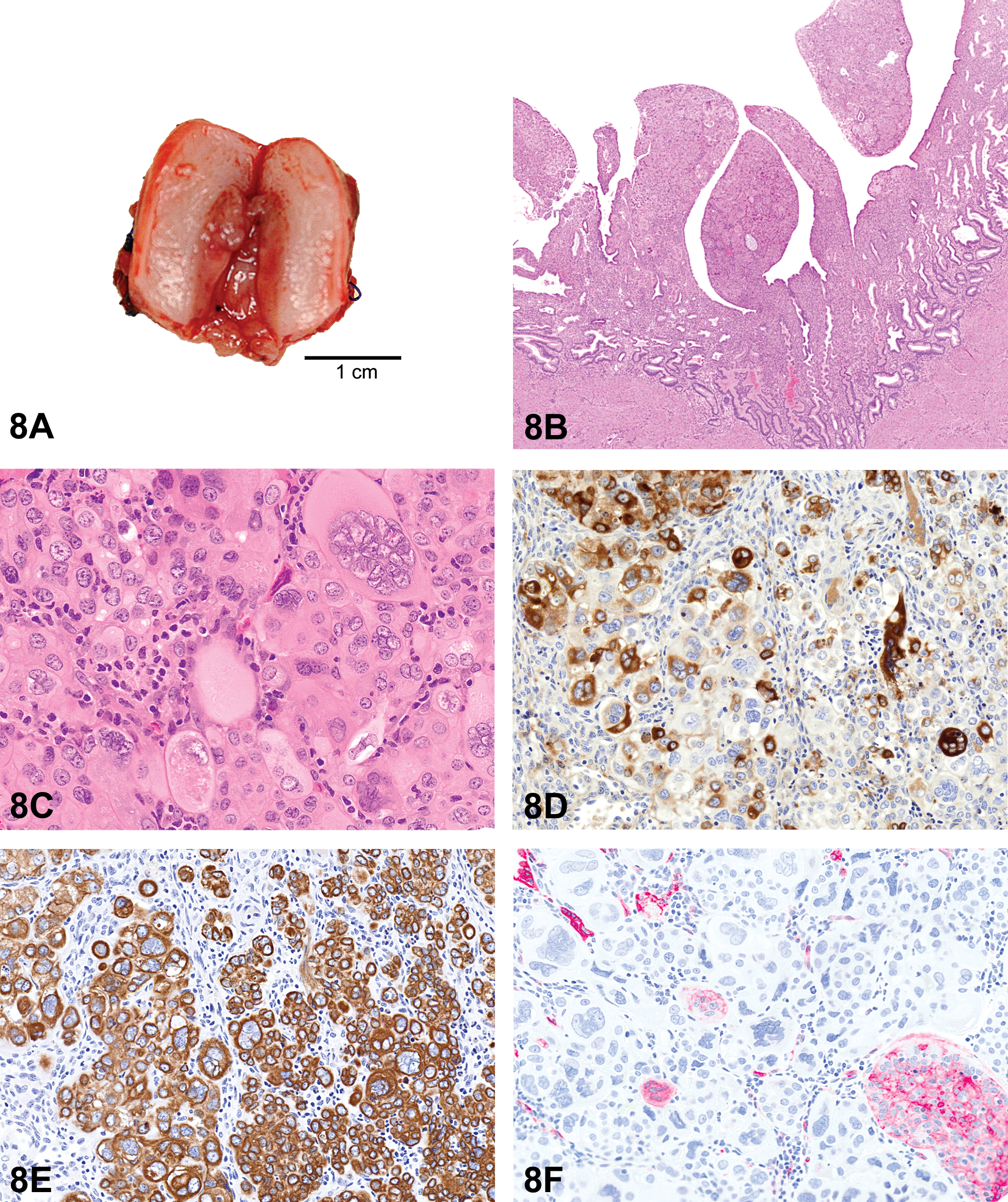
(A–F) Uterine choriocarcinoma in a rhesus macaque. A gross image (A) of the hysterectomized uterus with irregular thickening involving the entirety of the endometrium at the apex. Low magnification of histologic sections stained with H&E (B) shows an unencapsulated, poorly demarcated papillary mass expanding and effacing the endometrium. The mass is composed of sheets and packets of highly pleomorphic neoplastic cells surrounding a preexisting stroma. At high magnification (C), the neoplasm is comprised of a dimorphic population of small mononuclear and large multinucleate type cells (H&E). Immunohistochemical staining of the uterine mass with human chorionic gonadotrophin (D) shows cytoplasmic staining ranging from negative to strongly positive and pancytokeratin (E) shows diffuse, strong, cytoplasmic staining. Immunohistochemical staining with CD10 (F) was largely negative with rare, small foci of cytoplasmic positivity. H&E = hematoxylin and eosin.
The audience was shown two representative photomicrographs of H&E-stained slides of the tumor. On low power (Figure 8B), the superficial endometrium was multifocally expanded and effaced by unencapsulated, poorly demarcated sheets and packets of pleomorphic neoplastic cells with marked anisocytosis surrounding preexisting stroma. In areas, the endometrium and neoplastic cells formed projections into the lumen up to 1.5 × 0.8 mm. On high power (Figure 8C), the cells had distinct cell borders, were round to polymorphic, and comprised of two populations of small mononuclear and large multinucleate types. Both types had abundant homogeneous to finely vacuolated eosinophilic cytoplasm and the centrally located nuclei were oval with reticulate to clumped chromatin and 0 to 2 nucleoli. Mitotic figures were rare. Lymphocytes, plasma cells, and few macrophages were scattered throughout the mass. There was little hemorrhage or necrosis, and vascular invasion was not seen.
Additionally, photomicrographs were presented to the audience displaying immunohistochemical staining characteristics of the neoplasm. The cells were generally positive for human chorionic gonadotrophin (hCG) and multinucleate cells that were strongly positive (Figure 8D). Staining was diffusely positive for pancytokeratin (Figure 8E) and negative for human placental lactogen, placental alkaline phosphatase, and p63. While the majority of the lesion was negative for CD10, there were rare foci of CD10 positivity which were negative for hCG and pancytokeratin (Figure 8F). The voting choices and results were decidual reaction (34%), epithelioid trophoblastic tumor (ETT; 29%), choriocarcinoma (CC) (11%), placental site trophoblastic tumor (PSTT; 8%), placental site nodule (7%), epithelial plaque response (4%), hydatidiform mole (3%), and uterine carcinoma (3%).
Based on the histologic morphology and immunohistochemical staining, the tumor was diagnosed as a CC at Wake Forest University. This lesion falls under the umbrella of gestational trophoblastic disease (GTD), which are neoplastic and nonneoplastic lesions of the placenta. GTD in nonhuman primates consists of benign proliferations of trophoblastic tissues, malformations of the trophoblastic villi, and trophoblastic tumors (Bentley 2003). The main types of GTDs are placental-site trophoblastic tumor and ETT, CC, hydatidiform mole (molar pregnancy; complete or partial), and invasive mole. The characteristics of a range of GTD were described for the audience to assist in elucidating the diagnosis of the lesion.
Trophoblastic tumors are rare neoplasms in domestic and laboratory animal species and, in nonhuman primates, have been reported in the uterus and ovary (Chu et al. 2007; Cooper, Shih, and Gabrielson 2005; Farman et al. 2005; Giusti et al. 2005; Marbaix et al. 2008; Marr-Belvin et al. 2010; Toyosawa et al. 2000; Yokouchi et al. 2011). In humans, most occur following a normal or ectopic pregnancy, abortion, or molar pregnancy (Bentley 2003; Hui et al. 2014). These are comprised of PSTTs, ETTs, and CCs.
PSTTs are a monomorphic population of large, pleomorphic cells with abundant eosinophilic cytoplasm that infiltrate the surrounding tissue as sheets or single cells. These neoplasms are derived from implantation-type intermediate trophoblasts. Mitoses in these tumors are variable, from 0 to 6 per 10 high power fields. Necrosis and hemorrhage are focal or absent, myometrial invasion is common, and vascular invasion may occur. Although these are infiltrative, the myometrium is not effaced. Serum hCG levels in humans with PSTTs are typically low to moderate (Bentley 2003; Marbaix et al. 2008).
ETTs arise from chorionic-type intermediate trophoblasts at the chorion laeve, which is an extraembryonic fetal membrane, composed of trophoblasts lined with mesoderm. It develops villi, becomes vascularized, and forms the fetal part of the placenta (Marbaix et al. 2008; Shih and Kurman 2004). Microscopically, these tumors present as nests, cords, or solid masses of a monomorphic population of cells, which are small and round with eosinophilic or clear cytoplasm. A geographic pattern of extracellular matrix and necrosis are characteristic of ETTs (Scott et al. 2012). Mitoses are variable, from 1 to 10 per high power field, and vascular invasion is absent. They are commonly associated with normal gestation but may less frequently arise from a hydatidiform mole (a benign form of GTD). Serum hCG levels in affected humans are typically low (Bentley 2003; Giusti et al. 2005).
CCs are comprised of a dimorphic population of primitive previllous cytotrophoblast-like cells and a multinucleate population of large syncytiotrophoblast-like cells with clusters of intermediate trophoblast-like cells. The cells are irregular and highly variable with eosinophilic to amphophilic cytoplasm. Generally, CCs have extensive hemorrhage and necrosis with myometrial and vascular invasion. Mitotic rates are usually high, ranging from 2 to 22 per 10 high power fields. hCG levels are systemically high in humans with CCs (Bentley 2003; Farman et al. 2005).
Epithelial plaque responses are benign spontaneous lesions, seen exclusively in nonhuman primates, which occur at the beginning of pregnancy or are experimentally induced “pseudopregnancy” lesions. They consist of a monomorphic proliferation of trophoblastic cells. Epithelial plaques are present on both sides of the uterus, reflecting the bidiscoid placentation of primates. They are regarded as benign lesions which regress spontaneously (Cline et al. 2008; Kaspareit et al. 2004). Placental site nodules are small remnants of involuted implantation sites, which present as well circumscribed nodules of chorionic-type intermediate trophoblasts. The majority of these lesions are microscopic, and mitoses are rare or absent. These are considered to be the benign counterpart of ETTs (described below) and, though they can persist, no follow-up or treatment is necessary (Bentley 2003).
Hydatidiform moles have not been reported in macaques. In humans, they occur at the time of pregnancy and are frequently seen in conjunction with abortion or menorrhagia. These lesions are placentation with abnormal proliferation of trophoblastic villi and are called molar pregnancies. They can be categorized as complete, partial, or invasive moles. Complete moles grossly appear as clusters of grape-like structures, corresponding to villi with hydropic change, and typically there is a lack of fetal tissue. Most are chromosomally derived wholly from the sperm. Partial moles have abnormal hydropic and fibrotic villi and may or may not have associated fetal tissue (Bentley 2003). In these cases, one egg is fertilized by two sperm, or there is a duplication of paternal chromosomes (Seckl et al. 2000). Invasive hydatidiform moles are neoplastic entities that arise from either complete or partial hydatidiform moles, in which hydropic villi invade the myometrium and uterine blood vessels, or rarely are found at distant sites. Any molar pregnancy can be a predisposing factor in uterine trophoblastic tumor development, though more are seen following partial molar pregnancies (Bentley 2003; Hui et al. 2014).
Decidual reactions, also known as deciduomas, are lesions of proliferative decidual stromal cells and differ substantially between species. In rodents, decidual cells form nodular lesions including large pleomorphic cells resembling trophoblastic cells. In nonhuman primates, hypertrophic or hyperplastic decidual cells are more uniform and retain stromal morphology (Atkins et al. 2016; Cline et al. 2008).
In human gynecologic oncology, distinguishing between the trophoblastic tumors is diagnostically important due to differences in metastatic potential and responses to chemotherapy. Immunohistochemical staining is considered an important tool. A table compiled from a review of the literature, including human and nonhuman primate cases, is provided (Table 1). Both CCs and PSTTs have high rates of vascular invasion, but while CCs are highly responsive to chemotherapy, PSTTs are more resistant. PSTTs do, though, have high survival rates following surgery. Rates of vascular invasion and metastases are low with ETTs, and response to chemotherapy is largely uncharacterized as there are few reported cases (Bentley 2003).
A Summary of the Immunohistochemical Staining Properties of Different Types of Gestational Trophoblastic Disease Compared to the Presented Lesion.

Note: Select references were used to compile these data (Bentley 2003; Hui et al. 2014; Kalhour et al. 2009; Kommoss et al. 2001; Shih, Seidman, and Kurman 1999; Shih and Kurman 2004; Yokouchi et al. 2011). ETT = epithelioid trophoblastic tumor; PSTT = placental site trophoblastic tumor; CC = choriocarcinoma; PSN = placental site nodule.
Important take-home points from this lecture were that (1) trophoblastic lesions in macaques may be benign or malignant, (2) subgross histology and the presence or absence of invasion are important criteria, and (3) immunohistochemistry is required for the differentiation of trophoblastic tumors.
Considering Form, Function, and Context for a Unique Renal Lesion
Dr. Vinicius Carreira (Janssen Research & Development, La Jolla, CA) presented what some may consider a diagnostic conundrum. An unusual epithelial proliferative process occurred in the pelvic area of the kidney in two of four high-dose male cynomolgus macaques (Macaca fascicularis) of Cambodian origin. Animals were given a small molecule TGFβR1 inhibitor via oral gavage once daily for 21 days. The most severe of the two cases was presented at the symposium. Dr. Carreira acknowledged the many colleagues who have contributed scientifically to this case, including Drs. Jing Ying Ma, Sandra Snook, Eric McDuffie, Andrew Standeven, Walter Bee, William Kerns, Michael K. Jackson, Meredith Simon, Jerry Hardisty, Samuel Cohen, and Calvert Louden.
The animals on study were 3 to 5 years old at study start. Clinical observations in high-dose animals included increased incidences of abnormal urine color and increased frequencies of blood-positive urine specimens. Changes in serum chemistry parameters related to kidney toxicity included decreases in serum total protein, albumin, calcium, inorganic phosphate, and increased blood urea nitrogen and creatinine. Enlarged kidneys were noted at necropsy and correlated microscopically with tubular degeneration/regeneration, with polymorphonuclear cell infiltration, tubular casts, and proliferative epithelial lesions in the renal pelvic region.
A poorly demarcated and highly cellular epithelial proliferative process expanded, disrupted, and replaced the suburothelial pelvic stroma with multifocal evidence of vascular invasion (Figure 9A–H). The morphology and immunophenotype of the proliferative population were most consistent with a malignant epithelial neoplasm and suggested a collecting duct origin (Table 2). Most cells had patchy to absent membranous E-cadherin and β-catenin staining, with increased cytoplasmic to nuclear B-catenin and SNAI1 staining, signifying a proliferative and invasive phenotype. Furthermore, α-SMA labeling highlighted segmental displacement and disruption of the suburothelial smooth muscle layer by the epithelial proliferative process, with entrapped α-SMA-positive elongated cells amid epithelial proliferative nests. After the original pathology review, this study was peer reviewed by veterinary pathologists and a medical nephropathologist. Additionally, a veterinary pathologist examined slides for evidence of infectious diseases that could be implicated in this type of finding, such as viruses. No remarkable microscopic findings were noted to suggest an underlying etiology for this finding. Progression or recovery of these changes has not been assessed to date.
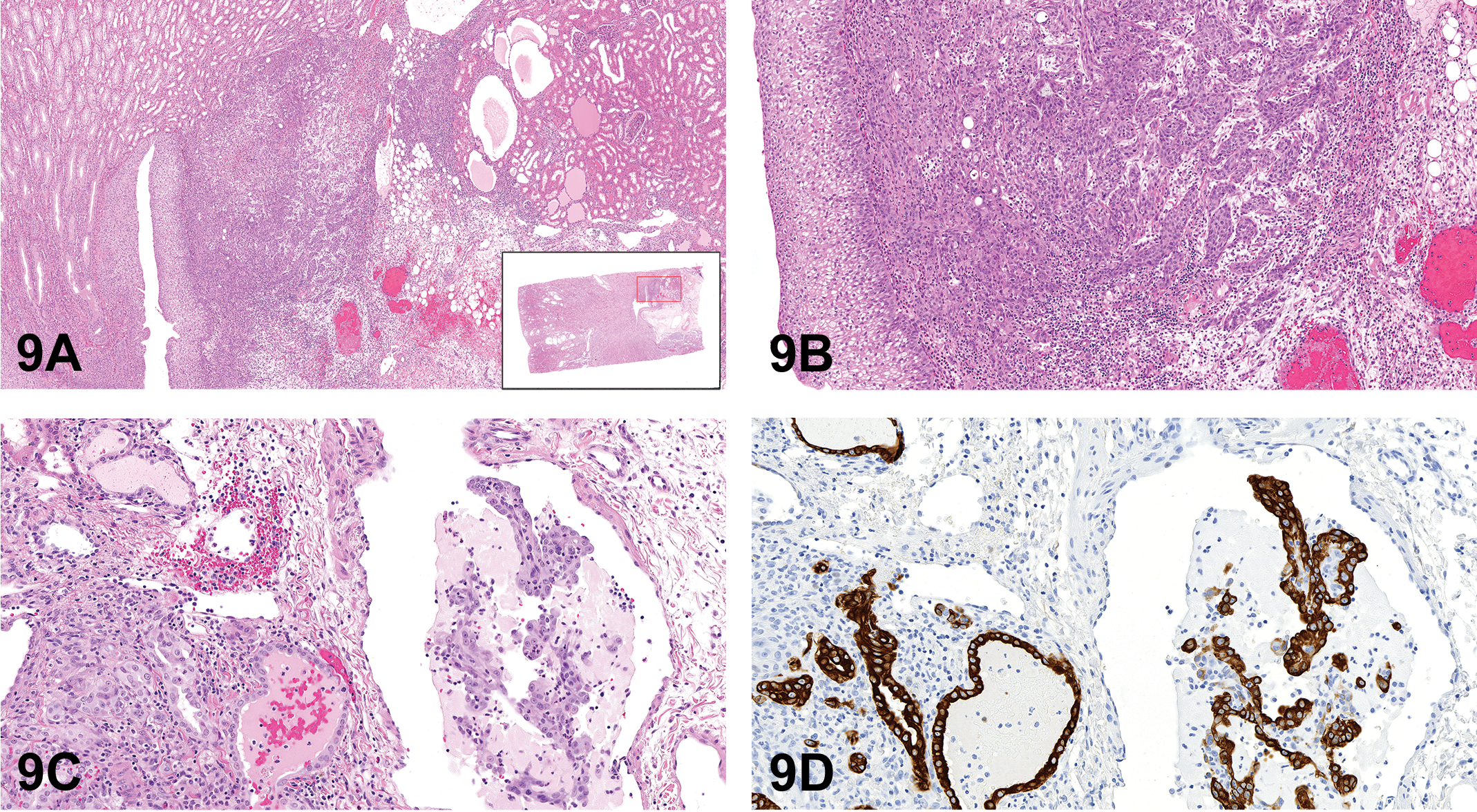
(A–D) Unusual renal lesion from a cynomolgus macaque given a small molecule TGFβR1 inhibitor. (A–C) were used for round 1 of voting. (A) shows a poorly demarcated, highly cellular process that has expanded and disrupted the suburothelial stroma, with extension into the adjacent renal cortex and hilus stroma, subjacent to the pelvic urothelium (inset). (B) shows multiple nests of proliferative epithelium infiltrate within the renal hilus fibroadipose stroma. Extensive infiltration of inflammatory cells was present in the hilus stroma, with edema, and multifocal areas of hemorrhage and necrosis. At the edge of the lesion in (C) is a longitudinal profile of a vein containing multiple intraluminal papillary arrangements of pleomorphic epithelial cells, with morphology consistent with the proliferative epithelial population of interest. (D–H) were presented for round 2 of voting. (D) shows that immunohistochemistry for Cytokeratin 19 (CK19) confirmed the epithelial nature of the emboli within the vein. (E–H) Unusual renal lesion from a cynomolgus macaque given a small molecule TGFβR1 inhibitor (continued). (E) shows epithelial proliferative cells in nests, irregular tubules, sheets, or discrete cells, with variable cell and nuclear morphology, prominent nucleoli, and abundant mitotic figures with occasional bizarre mitoses (H&E). (F–H) show that most cells stained intensely positive with CK19 (F), with a large subset also being positive for Vimentin (G) but were negative for all other markers tested (Table 2). Proliferating cell nuclear antigen (PCNA) highlighted the highly proliferative nature of the process with a markedly high density of PCNA-positive nuclei (H). H&E = hematoxylin and eosin.
Immunohistochemistry Findings.
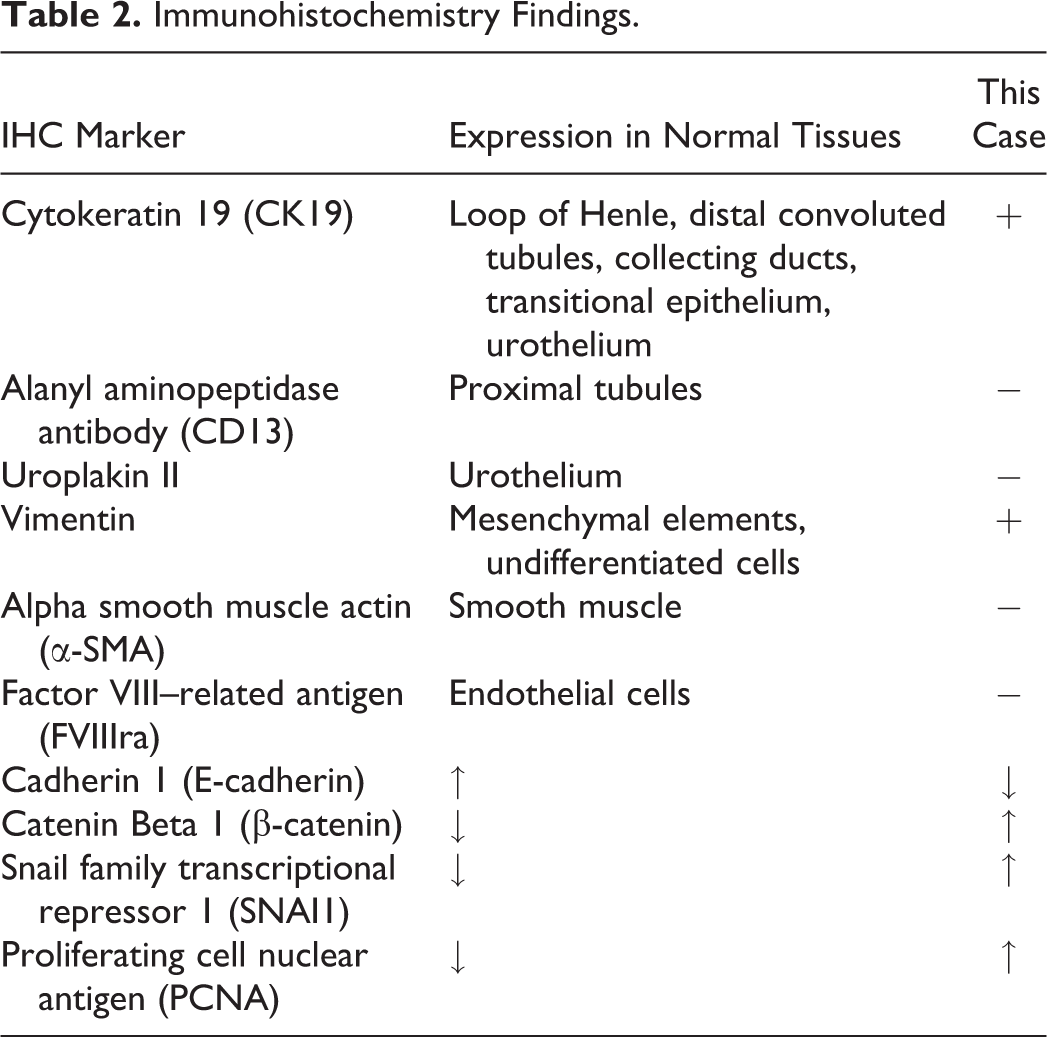
This case presentation was deliberately planned to span three rounds of voting to capture how the morphology (round 1 of voting; Figure 9A–C, H&E), the function (round 2 of voting; Figure 9D–H, immunohistochemistry), and the context (round 3 of voting; full signalment) would influence the audience’s diagnosis of such an unusual finding. Four diagnostic options were provided for voting: hyperplasia, atypical hyperplasia, benign neoplasia, and malignant neoplasia. One or more of the observed morphological features in this case overlaps with all the differentials listed in the voting options; however, invasion into the adjacent parenchyma/stroma, presence of necrosis/hemorrhage, and particularly vascular invasion/embolization are largely regarded as unique to malignant neoplasia. The audience had a majority vote for malignant neoplasia for the first two rounds (73 and 69%, respectively), with a sway toward the diagnosis of atypical hyperplasia versus neoplasia upon full signalment disclosure (split 45 vs. 42%, respectively). These data indicate that, aside from the strikingly short latency, the morphology and immunophenotype results were convincingly consistent with a malignant process. Multiple pathologists in the audience shared personal accounts of dealing with similar proliferative processes related to TGFβ biology, including hyperplasia, atypical hyperplasia, benign and malignant neoplasia, largely affecting epithelia across multiple organs, and presenting with unusually short latency periods.
TGFβR1 has been implicated in epithelial proliferative disorders (Herbertz et al. 2015; Honjo et al. 2007; Pasche et al. 2014; Stauber et al. 2014; Sun, Taguchi, and Hanash 2016; Goudie et al. 2011). The mechanism is not entirely elucidated, though it is believed to be related to known dual roles as a tumor suppressor and promoter, epithelial to mesenchymal transition biology, and extracellular matrix homeostasis. Additional microscopic findings were present in the cortex of the kidney, and in other organs of these monkeys, including florid renal tubular degeneration/regeneration and atypical tubular epithelial proliferation. The overall phenotypic properties of the proliferative epithelial cells in question overlap with hallmarks of neoplasia (Hanahan and Weinberg 2011) such as robust primary or regenerative proliferative response coupled with extracellular matrix remodeling, culminating in inappropriate and unchecked proliferation, differentiation, and migration.
Proliferative Ovarian Lesions from Aged Rats
Dr. Cynthia Shackelford (Experimental Pathology Laboratories, Inc., Research Triangle Park, NC) presented three examples of spontaneous ovarian lesions that are encountered sporadically in aged rats, and two of the three cases are presented here. All three examples were observed during a detailed review of the control animal ovaries from two-year NTP carcinogenicity studies using F344/N, Harlan SD, and Wistar Han rats. All three cases depicted a series of lower and higher magnification photographs of the lesions located at the hilus of the ovary where nerves and vessels enter.
The first case was a relatively large, focal mass composed of tubules lined by cuboidal to columnar cells with scant cytoplasm, primarily centrally located nuclei and prominent stromal proliferation with distention of an affected portion of a rete tubule. The mass showed no evidence of secretory material, extra tubular invasive growth, or cellular atypia. (Figure 10A and B). Voting choices and results were tubulostromal hyperplasia (13%), tubulostromal adenoma (24%), rete ovarii hyperplasia (35%), rete ovarii adenoma (19%), normal structure (4%), cystadenoma (1%), hamartoma (3%), and other (0%). Although the audience favored rete ovarii hyperplasia, the Pathology Working Group (PWG) diagnosis for this lesion was rete ovarii adenoma. However, the distinction between rete ovarii hyperplasia and adenoma may be arbitrary, depending on size and degree of stromal involvement. Tubulostromal adenoma is considered a differential diagnosis for this lesion.
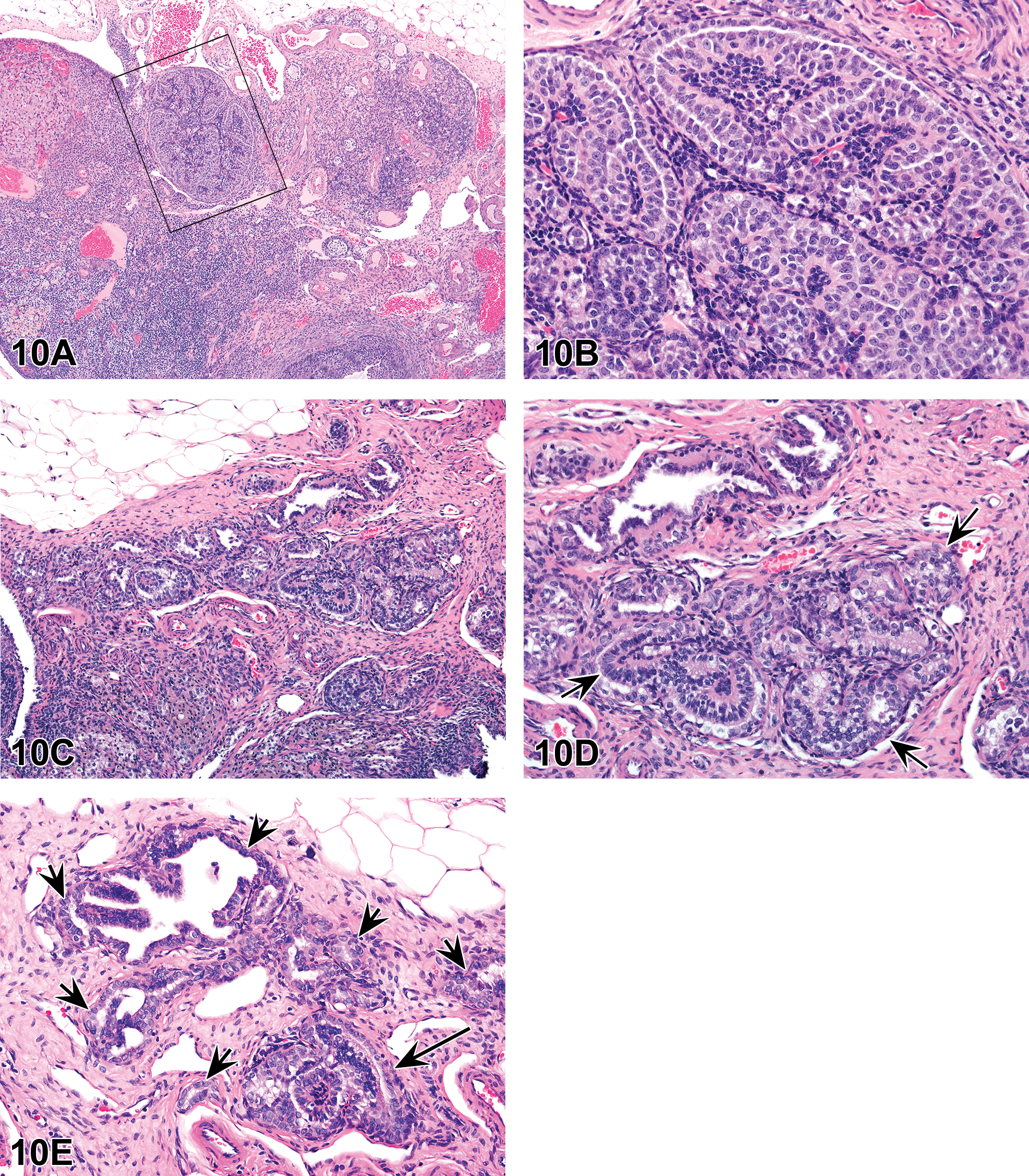
(A–E) Rete ovarii adenoma (A and B) from a control Sprague Dawley rat presented as case 1. The low magnification image (A) shows a relatively large, expansile, and slightly compressive nodule (boxed) arising from the rete ovarii located at the hilus of the ovary. Higher magnification (B) shows the mass is composed of tubules lined by one to several layers of cuboidal to columnar epithelial cells with a scant amount of pale eosinophilic to vacuolated cytoplasm, and primarily centrally located nuclei. Note the presence of a somewhat prominent stromal proliferation. Rete ovarii hyperplasia (C and D) from a control Sprague Dawley rat presented as case 2. The low magnification image (C) shows an expansile area of proliferating tubules located at the hilus of the ovary. Higher magnification (D) shows proliferating tubules (arrows) lined by hypertrophied and vacuolated epithelial cells and apically to centrally located nuclei. Note the presence of polypoid ingrowths and the absence of any stromal proliferation. Normal rete ovarii and a focus of rete ovarii hyperplasia (E) from a control Sprague Dawley rat. This low magnification image shows normal rete ovarii with tubules lined by cuboidal epithelium (short arrows) adjacent to an area of proliferating tubules with polypoid ingrowths, considered rete ovarii hyperplasia, in the lower right corner (long arrow). H&E = hematoxylin and eosin.
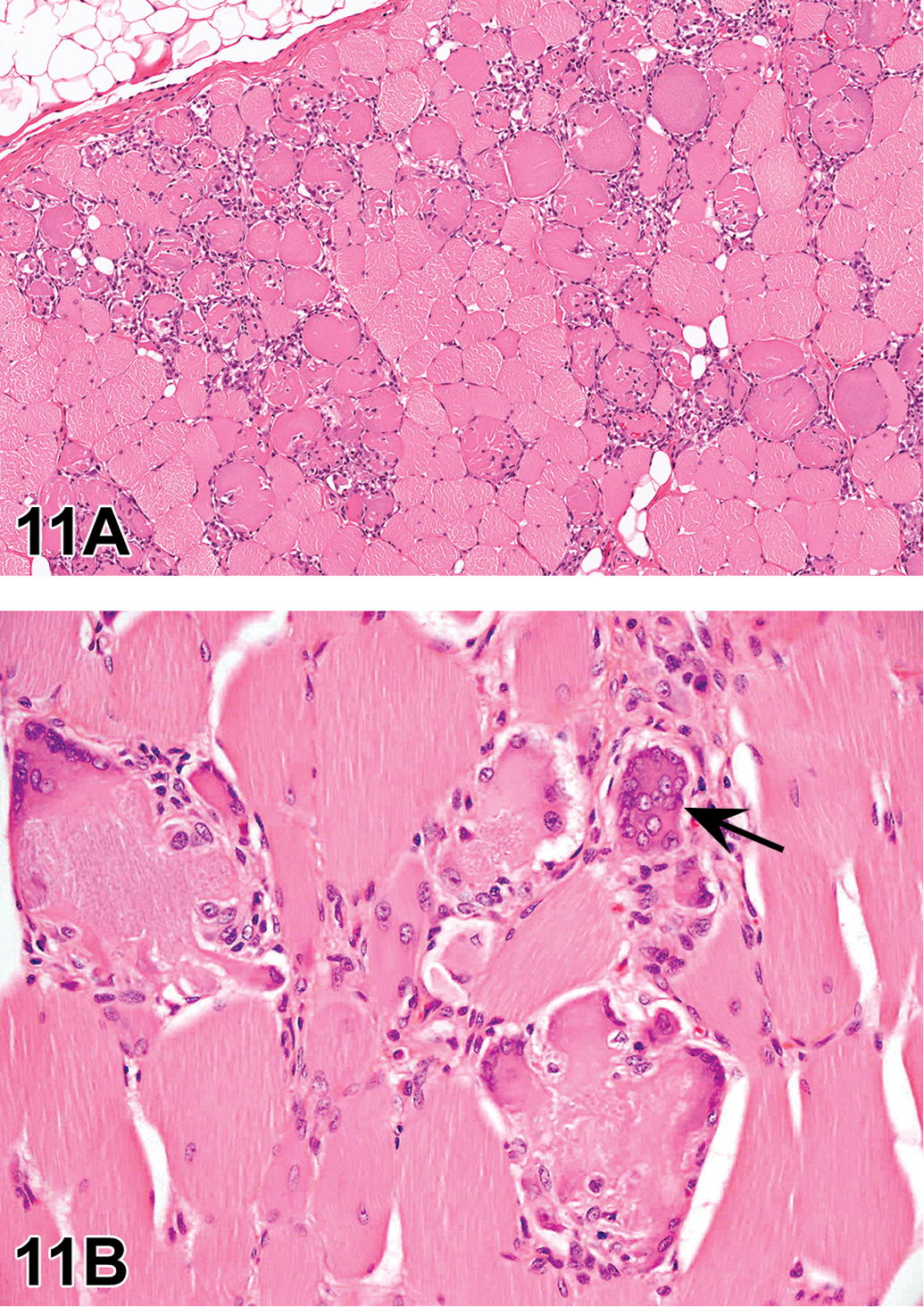
(A and B) Porcine myopathy in a Göttingen minipig, presented as case 1. (A) is a transverse section of panniculus muscle showing degenerating swollen, basophilic fibers, necrotic fibers with infiltrating mononuclear inflammatory cells, interstitial inflammation, and occasional regenerating myofibers with multiple nuclei. (B) is a transverse section of biceps femoris showing degeneration and residual mononuclear inflammatory cells and early regeneration with multiple nuclei in myofibers (arrow). (C–E) Serous atrophy in a Göttingen minipig (C–E), presented as case 2. (C) is a subgross view of the distal femur showing loss of hemopoietic cells and loss of definition of adipocytes. In (D), a few remaining hemopoietic cells are visible between degenerating adipocytes surrounded by homogenous eosinophilic material. Staining with Alcian blue at pH 2.5 (E) illustrates the mucopolysaccharide nature (predominantly hyaluronic acid) of the homogeneous eosinophilic material. (F and G) Normal Leydig cells in a Göttingen minipig, presented as case 3. Transverse sections of testes from histologically sexually mature control Göttingen minipigs on the same study, illustrating the variation in Leydig cell numbers and size that may be seen in this species. In (F), Leydig cells are large polygonal cells with dark central cytoplasm and lighter peripheral cytoplasm that fill the interstitium between seminiferous tubules. In (G), Leydig cells are considerably smaller and fewer in number, such that much of the interstitium is devoid of cells. (H–M) Chronic, necrotic cholecystitis in a Göttingen minipig (H and I) presented as case 4. (H) is a section of gallbladder illustrating irregularly thickened wall, eosinophilic contents (hemorrhage) and mucosal inflammation. (I) shows thickened wall, mucosal inflammation and necrosis, fibrosis of the muscularis, and hemorrhagic contents. Pigmented macrophages are present at the serosal surface and in the muscularis. Mesothelial hyperplasia in a Göttingen minipig (J and K), presented as case 5. (J) is a low magnification of thickened mesothelium on adipose tissue at the arch of the aorta of a Göttingen minipig. Higher magnification of this lesion shows focal thickening due to relatively uniform cuboidal cells with little stratification. This particular lesion appears quite cellular. There are few, if any mitotic figures and no cellular atypia (K). Splenic periarteriolar macrophage sheaths (PAMS) in a Göttingen minipig (L and M), presented as case 6. In (L), a periarteriolar lymphoid sheath is present on the right and a trabeculum is present on the left of the image. Multifocal, discrete, rounded structures are scattered throughout the red pulp (arrows). These PAMS consist of large, plump cells with abundant pale eosinophilic cytoplasm. (M) is a serial section stained with silver to demonstrate the reticulin network. A fine network of collagenous fibers can be seen around the periphery of the PAMS (arrows pointing to PAMS). H&E = hematoxylin and eosin.
The second case (Animal #2) contained a much smaller, but somewhat focally extensive, area of diffusely thickened rete tubular epithelium showing evidence of polypoid ingrowths (Figure 10C and D). The cells were densely packed, hypertrophied, and vacuolated. The nuclei were often present in the apical cytoplasm of the majority of cells with more centrally located nuclei when vacuolated. This focally extensive area of tubular epithelial cell proliferation was located adjacent to more normal appearing rete tubular epithelium. Voting choices and results were tubulostromal hyperplasia (13%), tubulostromal adenoma (13%), rete ovarii hyperplasia (62%), rete ovarii adenoma (13%), normal structure (3%), cystadenoma (1%), hamartoma (1%), and other (0%). The favored response by the audience, rete ovarii hyperplasia, was the correct diagnosis for this lesion.
The discussion then moved on to a review of the origin and histologic characteristics of the normal rete ovarii and proliferative and cystic lesions in rats. The rete ovarii develops from cells of mesonephric origin which migrate into the developing gonad of the ovary. It is a vestigial structure and homologue of the rete testis, the function of which is unknown; however, its presence is essential for follicular development (Davis et al. 2001; Byskov et al. 1977). The mature rete ovarii is located at the hilus of the ovaries, where nerves and vessels enter, and is defined by groups of anastomosing tubules lined by one to several layers of cuboidal or columnar epithelium, which may be ciliated. It is surrounded by a cuff of spindle cell stroma similar to the ovarian stroma and communicates with mesonephric duct tubules within the mesovarium. It is present in the mouse, rat, guinea pig, rabbit, cat, dog, deer, cow, and human (Jiang et al. 2004; Figure 10E). A schematic of the sheep rete ovarii (Wenzel, Odend'hal, and Player 1987) located within the proximal portion of the infundibulum (extraovarian rete) and extending deep into the ovary (intraovarian rete) was shown.
Rete ovarii hyperplasia is characterized by diffuse thickening of the rete tubular epithelium. The cells lining the tubules maybe vacuolated and hypertrophied or densely packed and may contain areas of ciliated cells. The hyperplastic epithelium may form polypoid ingrowths with extensive lesions showing a slight papillary pattern and may occur in cystic dilated tubules. The epithelium is immunoreactive to antibodies for intermediate filaments (cytokeratins, desmoplakins, and vimentin). Ultrastructurally, the tubular epithelial cells have abundant junctional desmosome-tonofilament complexes. There may be evidence of smooth muscle. The nuclei within the lining epithelial cells are often apical in location, but a more central to basilar location is observed in vacuolated cells. It is important to recognize evidence of origin from the rete ovarii. These foci of hyperplasia are fairly common (C. C. Shackelford, personal observation, June 16, 2018) and seem to have the potential to develop into adenoma as they enlarge and therefore may be regarded as preneoplastic lesions. Differential diagnoses for rete ovarii hyperplasia include rete ovarii adenoma, rete ovarii cyst, paraovarian cyst, and ovarian surface epithelium hyperplasia (cystic/papillary). The distinction between rete ovarii hyperplasia and rete ovarii adenoma appears to be arbitrary and would depend on size and degree of stromal involvement (Dixon et al. 2014; Kon et al. 2007; Long 2002; C. C. Shackelford, personal observation, June 16, 2018). The precursor lesions of rete ovarii hyperplasia or small early rete ovarii adenomas are not grossly detectable and begin as small, focal, focally extensive to bilaterally crowded accumulations of tubules with well-defined cytoplasm and basophilic nuclei, which are clearly delineated from the normal ovarian tissue. These precursor lesions do not overtly disrupt the overall structure and organization of the ovary.
Rete ovarii adenomas vary in size and are characterized by distention of the rete tubule by intratubular mass formation which usually has papillary structures, a central stalk, and branching fibrovascular stroma. The cells are cuboidal to columnar with scant cytoplasm and frequently central to apical nuclei. There is generally no evidence of secretory material or evidence of extratubular invasion. These tumors occur rarely in rats and mice, are most often seen in aged animals, and are generally not grossly detectable. Differential diagnoses for rete ovarii adenoma include rete ovarii hyperplasia and cystadenoma. While some authors identify the surface epithelium as the origin of epithelial tumors of the ovary (Long 2002), others indicate that the origin of cystadenomas remains to be determined (Morgan and Alison 1987).
Rete ovarii cysts arise as remnants of rete analage and are present adjacent to or within the hilus of the ovary. These cysts are lined by flattened, cuboidal, or columnar epithelium which may be ciliated and often having apically located nuclei. There may be evidence of a connection with the paraovarian structures in the mesovarium. When large, these cysts usually distort the ovarian architecture (Tan, Hurst, and Fleming 2005).
Dr. Shackelford indicated that further investigation as to the origin of proliferative lesions of the rete ovarii and surface epithelium would be investigated by the NTP. It is very important in extrapolating NTP data to human relevance and public health significance that we know the origins of tumors; therefore, it is imperative that the NTP can distinguish between a chemically induced lesion that results in a rete ovarii proliferative lesion versus one that targets the ovarian surface epithelium, with the latter being the principle cell type in 90% of cases of ovarian cancer in women. By identifying the origins of these proliferative lesions, and markers that are specific to cell types (rete vs. surface epithelium), this will allow the NTP pathologist to use tools/markers to better make definitive diagnoses for challenging chemically induced ovarian proliferative lesions and make more biologically relevant extrapolations to humans as to what cell types are actually affected.
In summary, identification of rete ovarii lesions depends on proper sectioning of the ovary (at or near the hilus; Ruehl-Fehlert et al. 2003) and familiarity with this structure and the morphology of rete ovarii hyperplasia, adenomas, and cysts (Davis et al. 2001).
Select Cases from the Minipig INHAND Working Group
The last session, presented by Dr. Sean McKeag (Covance Laboratories, Harrogate, North Yorkshire, UK) on behalf of the INHAND Non-Rodent Working Group (NRWG) for the minipig, comprised a selection of Göttingen minipig background pathology cases that either do not occur in rodents or have special relevance in the minipig. The minipig NRWG consists of Drs. Mikala Skydsgaard (Chair), Susanne Rittinghausen (GESC Liaison), Zuhal Dincer, Wanda Haschek, Kris Helke, Binod Jacob, Björn Jacobsen, Gitte Jeppesen, Atsuhiko Kato, Hiroaki Kawaguchi, Sean McKeag, Keith Nelson, Dirk Schaudien, Vimala Vemireddi, and Zbigniew Wojcinski. The NRWG for the minipig is reviewing the applicability of the INHAND rodent nomenclature for this species and adding terminology that is specific for the minipig, with particular focus on the Göttingen minipig, but also including lesions from the Yucatan, Sinclair, and Hanford breeds.
The first case was a series of multifocal to diffuse skeletal muscle lesions. Necrosis and degeneration of groups of muscle fibers, with inflammation and regeneration, were the hallmarks of these lesions. Voting choices and results were necrosis, myofiber (3%); inflammation, myofiber (2%); degeneration/regeneration, myofiber (13%); degeneration/regeneration/necrosis/inflammation myofiber (75%); myopathy (6%); implant-related change (2%); and other/I don’t know (0%).
The term chosen to describe this lesion by the INHAND group is porcine myopathy (porcine added by group after NTP symposium), with synonyms of degeneration/regeneration/necrosis/inflammation of myofibers, myositis, or myopathic change, reflecting the multiple processes that may be seen. Diagnostic features (Figure 11A and B) include signs of degeneration, including swollen fibers, vacuolation of fibers, split fibers, and hyaline changes. Myofiber necrosis may be present, with hemorrhage, edema, and inflammatory cell infiltrates. Macrophages are often a prominent feature of these infiltrates. Regenerating basophilic myofibers and mineralization may be seen in more chronic lesions. Interestingly, the majority of the audience preferred the descriptive term degeneration/regeneration/necrosis/inflammation which accurately describes the spectrum of changes that can be seen. However, due to the limitations of SEND, the use of slashes is limited to one per INHAND term. The term “myositis” was discussed; since this term only describes a part of the spectrum of changes seen, a broader term was preferred by the INHAND minipig group. However, myopathy was considered by some to be too general a term, with connotations of white muscle disease (vitamin E/selenium deficiency). While “myopathy” may be a general term, it was considered acceptable if it was well characterized in the INHAND document, and the prefix “porcine” was added, in a manner similar to “rodent progressive cardiomyopathy.”
Myopathy is considered a common incidental finding in the minipig. Pathology varies from focal minimal lesions to multifocal severe lesions. Euthanasia of severely affected animals due to adverse clinical signs may be required. This term covers the entire span of diagnostic features ranging from acute lesions dominated by necrosis, hemorrhage, edema, and mixed inflammatory cell infiltrates to more chronic lesions characterized by basophilic, multinucleated regenerating myofibers, mineralization, and, occasionally, fibrosis. Some pathologists prefer to use the term “myositis” to describe this entity—however, as the inflammatory component is not always a prominent feature, “myopathy” is considered the most appropriate term to capture the range of pathology that may be observed (Dincer and Skydsgaard 2012; McInnes 2012; Jeppesen and Skydsgaard 2015; Helke et al. 2016a, 2016b).
The second case consisted of increasing magnifications of a sagittal section of distal femur. Hemopoietic cells were absent, and adipocytes were surrounded by pale, amorphous, eosinophilic material. Voting choices and results were adipocyte atrophy (5%), decreased adipocytes (3%), cachexia (2%), gelatinous marrow transformation (23%), decreased hematopoiesis (15%), marrow necrosis (10%), and serous atrophy (41%), with the latter being the INHAND term. Gelatinous transformation is sometimes used in the literature to describe this pathology. Differential diagnoses that may be considered include adipocyte atrophy, in the absence of eosinophilic gelatinous tissue, or decreased hemopoietic cells.
This background lesion is most commonly observed in the femur and tibia of Göttingen minipigs used as control animals in toxicology studies. Serous atrophy can also be observed in the marrow of the sternum, in the adipose tissue in the coronary sulcus of the heart (groove marking the divisions between atria and ventricles), and in the subcutaneous adipose tissue; however, femur and tibia (and sternum) are the bones sampled and examined in most studies. This well-described change is seen mainly in male Göttingen minipigs but may also be observed in females (Bollen and Skydsgaard 2006; Helke et al. 2016a). The etiology of the serous atrophy seen as background pathology is thought to be nutritional, likely related to a restricted feeding regimen. It occurs as a physiological response to normal degradation of adipose tissue under conditions where the minipig needs additional energy (Bollen and Skydsgaard 2006; Glerup, Grand, and Skydsgaard 2013; Jeppesen and Skydsgaard 2015). Histopathologically (Figure 11C–E), serous atrophy of the adipose tissue in the bone marrow is seen as atrophy and/or degeneration of the adipocytes accompanied by increased interstitial acidic mucopolysaccharides which stains with Alcian Blue at pH 2.5 (Dincer and Skydsgaard 2012). Decreased cell density of the hematopoietic cells in the bone marrow often accompanies this change. The lesion usually starts in the epiphysis. If only the epiphysis is affected, it is graded as minimal or slight. With increasing severity, the metaphysis and diaphysis are affected and the assigned grade may be scored as moderate or marked. It is important to state that minipigs showing this alteration are typically clinically unaffected.
Serous atrophy is less commonly seen in minipigs in North America than in Europe (Helke et al. 2016a); however, the incidence appears to be decreasing in Europe. This could be a consequence of different feeding regimens. Serous atrophy of adipocytes in the bone marrow has not been described in Hanford or Yucatan minipigs (Helke et al. 2016a). In domestic pigs, serous atrophy of fat can be found in cases of emaciation or inanition associated with illness or malnutrition. In humans, serous atrophy is observed in patients with anorexia nervosa, alcoholism, and AIDS and patients suffering with generalized severe illness (cachexia) and is usually systemic (Bohm 2000).
The third case comprised three magnifications of testis from a histologically sexually mature minipig. Seminiferous tubules appeared to be within normal limits and were surrounded by extensive numbers of large, plump, polyhedral cells with dark eosinophilic central cytoplasm, pale eosinophilic peripheral cytoplasm, and prominent nuclei. Voting choices and results were normal minipig testis (41%), Leydig cell hyperplasia (18%), Leydig cell hypertrophy (1%), Leydig cell hypertrophy/hyperplasia (36%), Leydig cell tumor (1%), increased Leydig cell secretory product (3%), or other/I don’t know (0%). The majority of the audience chose normal minipig testis, but a large number of votes were cast for Leydig cell hypertrophy/hyperplasia. It is due to the difficulty in distinguishing Leydig cell hypertrophy/hyperplasia from normal testis that this case was selected for presentation.
Leydig (interstitial) cell density, size, and numbers may vary widely in the minipig (Figure 11F and G), both as a function of maturation status and between individual animals. Leydig cells start increasing in number at puberty (3–4 months) and numbers continue to increase during the peripubertal period (3–6 months). Leydig cells sustain intratesticular androgen levels and may be affected by direct toxic effects on steroidogenesis as well as indirectly by toxic effects on gonadotropin release. However, as there is considerable histological variation in Leydig cell numbers, size, and appearance in normal mature control boars, toxic effects on Leydig cells must be carefully evaluated, as true hypertrophy/hyperplasia is difficult to determine. Hypertrophy/hyperplasia would need to be quite pronounced, including extensive crowding of nuclei and slight compression of tubules to warrant a diagnosis (van Straaten and Wensing 1977; Allrich et al. 1983; Howroyd, Peter, and de Rijk 2016; Kangawa et al. 2016). There was some discussion around whether the variability in Leydig cell prominence should be recorded in toxicology studies, with more prominent accumulations being recorded as hypertrophy/hyperplasia. The speaker’s opinion was that the study pathologist should be familiar with the normal range of Leydig cell prominence but recognize any differences between treatment groups and controls.
The fourth case was illustrated by four images of increasing magnification showing the gallbladder from an asymptomatic control minipig with necrosis and inflammation thickening the walls, as well as hemorrhage, mineralization, and pigmented histiocytes in and around the serosa. Voting choices were inflammation (3%); necrosis (9%); hemorrhage (16%); cholecystitis, chronic, necrotic (66%); hypoplasia (0%); atrophy (2%), and other/I don’t know (4%). The chosen INHAND term for this lesion is cholecystitis, chronic, necrotic, which was the most popular choice. During the discussion on myopathy, it was pointed out that the use of “-itis” terms should be avoided where possible. Clearly that has not been done in this case, but it was argued that this is a very minipig-specific lesion with a host of well-characterized diagnostic features, so the use of the diagnostic term “cholecystitis” rather than a descriptive term was warranted to avoid having to use multiple terms to describe a well-characterized pathologic finding.
When this lesion is present, the gallbladder is macroscopically small, with thickened walls and inspissated or absent bile. Microscopically (Figure 11H and I), this lesion is typically characterized by diffuse necrosis of the mucosa and submucosa, with extensive inflammatory cell infiltration and/or granulomatous inflammation in the mucosa, submucosa, and muscle layers. There is often extensive hemorrhage. More chronic lesions are characterized by pigmented macrophages in the lumen or submucosa and muscle layers, evidence of regeneration, including fibrosis, and mineralization.
In the literature, this lesion has been called chronic necrotizing cholecystitis or chronic necrotic cholecystitis. The pathogenesis is unknown. This lesion is clinically silent, with no clinical chemistry or hematology perturbations. Intrahepatic bile ducts are within normal limits. Hypoplasia and aplasia of the gallbladder have also been recorded as background findings in the Göttingen minipig (Dincer and Skydsgaard 2012).
The next case described mesothelial changes of the pleura that covers the aorta. The audience viewed increasing magnifications of multifocal thickening of the pleural mesothelium, with plump cuboidal cells forming discrete projections, often limited to one or two cell layers. Voting choices were mesothelial hyperplasia (82%); mesothelioma, benign (11%); mesothelioma, malignant (2%); epithelioid mesothelioma, malignant (3%); locally invasive tumor (1%); metastatic tumor (0%); and other/I don’t know (1%). The INHAND NRWG decided on mesothelial hyperplasia as the term that best describes this lesion.
In the literature, this may be referred to as proliferation, mesothelial or proliferation, serosal. Hyperplasia of the mesothelial cells may be seen in the pleura, pericardium, peritoneum, or tunica vaginalis. In the Göttingen minipig, this finding is most often minimal and focal and primarily observed in relation to the arch of the aorta or the atria of the heart; however, it may be more diffuse. This lesion is characterized (Figure 11J and K) by focal thickening or villous projections covered by relatively uniform cuboidal cells with little or no stratification but may contain a fibrovascular stalk. It is usually limited to one to two cells in thickness but may appear more cellular. There are usually few, if any mitotic figures and no cellular atypia. In the minipig, fibrosis and inflammation are rarely a component of this lesion. Possible differentials include malignant mesothelioma, which would be expected to be highly cellular with cellular pleomorphism and extensive spread within the body cavities, or infiltration of adjacent tissues. Malignant epithelioid mesothelioma exhibits poorly formed glandular structures or poorly formed glands. Sarcomatoid malignant mesothelioma consists largely of spindle-shaped cells.
Focal mesothelial proliferation is a common incidental finding thought to arise due to friction at the predilection sites, mainly the aortic arch and the atria of the heart (Dincer and Skydsgaard 2012; McInnes 2012; Jeppesen and Skydsgaard 2015; Helke et al. 2016a, 2016b).
The last case examined multifocal pale eosinophilic cellular accumulations in the red pulp of the spleen (Figure 11L). Voting choices were accumulation, macrophage, multifocal (18%); hyperplasia, macrophage (5%); decreased cellularity, periartiolar lymphoid sheaths (PALS) (13%); ellipsoids (15%); Schweigger-Seidel sheaths (26%); periarteriolar macrophage sheaths (19%); none of the above (1%); and other/I don’t know (2%). The INHAND term that the working group recommends is periarteriolar macrophage sheaths; however, these may be referred to in the literature as ellipsoids or Schweigger-Seidel sheaths.
Periarteriolar macrophage sheaths are normal structures formed from phagocytic cells and reticular fibers (Figure 11M) and form generally rounded, pale eosinophilic accumulations of cells around capillaries or small arterioles in the red pulp. Periarteriolar macrophage sheaths are prominent in the Göttingen minipig and may occasionally be seen in the spleen of dogs. Their prominence varies between individual animals. Their function is to filter blood, retain circulating particulate matter, and trap immune complexes which are presented to the cells of the germinal centers of the PALS (Sørby et al. 2005; Cesta 2006; Haley 2017). No test article–related toxicity of these structures has been reported, but it is important to be aware of their variation within a study.
These cases presented in the concluding session of the NTP Symposium illustrated how rodent INHAND terminology may be interpreted differently in the larger species and how the histology/pathology of each species needs to be interpreted in the context of that species. They also showed how new terms may need to be created to describe lesions specific to a species. These discussions can be quite complex and nuanced, and the resulting terminology may not always be perfect in the eyes of all pathologists. Provided the principles and requirements of INHAND and SEND are adhered to, and terms are carefully described, they should provide a practical means of succinctly describing observed pathology to users of pathology reports. It will be interesting to read the wider pathology community’s comments when the minipig INHAND document opens for review!
Footnotes
Acknowledgements
The authors wish to thank Eli Ney of the National Institute of Environmental Health Sciences for her unique and creative artwork for the symposium. Thanks also to Beth Mahler of Experimental Pathology Laboratories, Inc. for her assistance with manuscript image preparation and to Brittany Scott (Charles River Laboratories, Inc.) for assistance with manuscript formatting. Appreciation also goes to Maureen Bayley, Tierre Miller, Matthew Price, and others at Association Innovation and Management, Inc., for their valuable help with annual advertising and meeting facilities. Also integral to the success of this meeting was the audiovisual technical expertise provided by CMI Communications. Refreshments were graciously provided by Experimental Pathology Laboratories, Inc., and Charles River Laboratories, Inc.
Author Contribution
All authors (SAE, VC, SL, DM, SM, MR, CS, BS, AT, SW, LW, CW) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the Intramural Research Program of the National Institute of Environmental Health Sciences, National Institutes of Health.
