Abstract
The 2022 annual National Toxicology Program Satellite Symposium, entitled “Pathology Potpourri,” was held in Austin, Texas at the Society of Toxicologic Pathology’s 40th annual meeting during a half-day session on Sunday, June 19. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers’ talks along with select images that were used by the audience for voting and discussion. Various lesions and topics covered during the symposium included induced and spontaneous neoplastic and nonneoplastic lesions in the mouse lung, spontaneous lesions in the reproductive tract of a female cynomolgus macaque, induced vascular lesions in a mouse asthma model and interesting case studies in a rhesus macaque, dog and genetically engineered mouse model.
Keywords
Mouse Lung Tumors: As Easy as A/B, C!
Dr Erin M. Quist (Charles River Laboratories, Inc., [CRL] Durham, North Carolina) presented several interesting cases recently reviewed by a Pathology Working Group (PWG) conducted by the National Toxicology Program/National Institute of Environmental Health Sciences (NTP/NIEHS). The presented cases featured lung lesions from four different B6C3F1/N mice that were part of a 2-year toxicity/carcinogenesis study in which Dr Mike Ryan (Battelle Columbus) was the study pathologist and Dr Quist was the quality assurance pathologist and PWG coordinator. Thanks to Drs Mark Cesta, Susan Elmore and Michelle Cora for their assistance and Emily Singletary, Maureen Puccini and Beth Mahler for photographic editing and support.
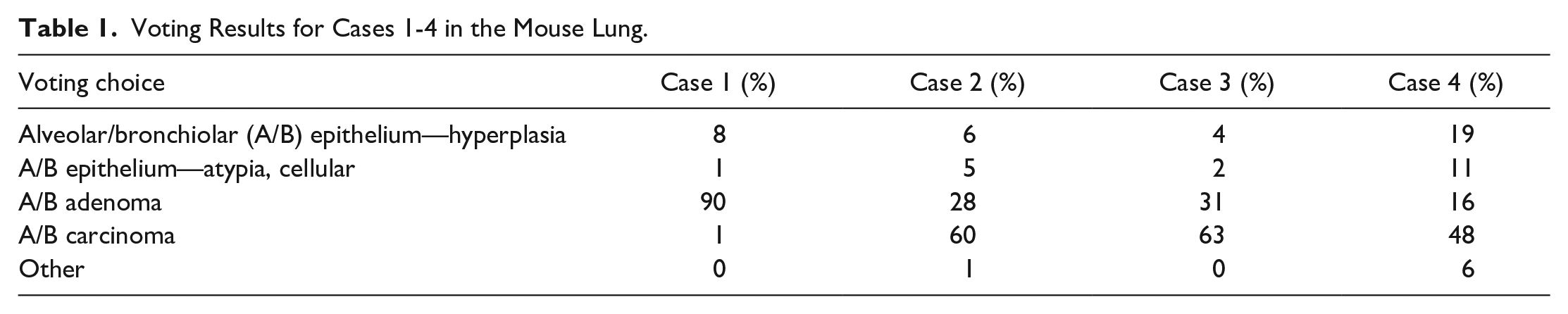
For the 4 cases, a series of photomicrographs were presented to the audience that included both low-power and high-power magnifications of select sections of lung from the 2-year study (Figure 1). Dr Quist informed the audience that the diagnostic choices would be the same for all four cases; the voting choices and results are presented in Table 1.
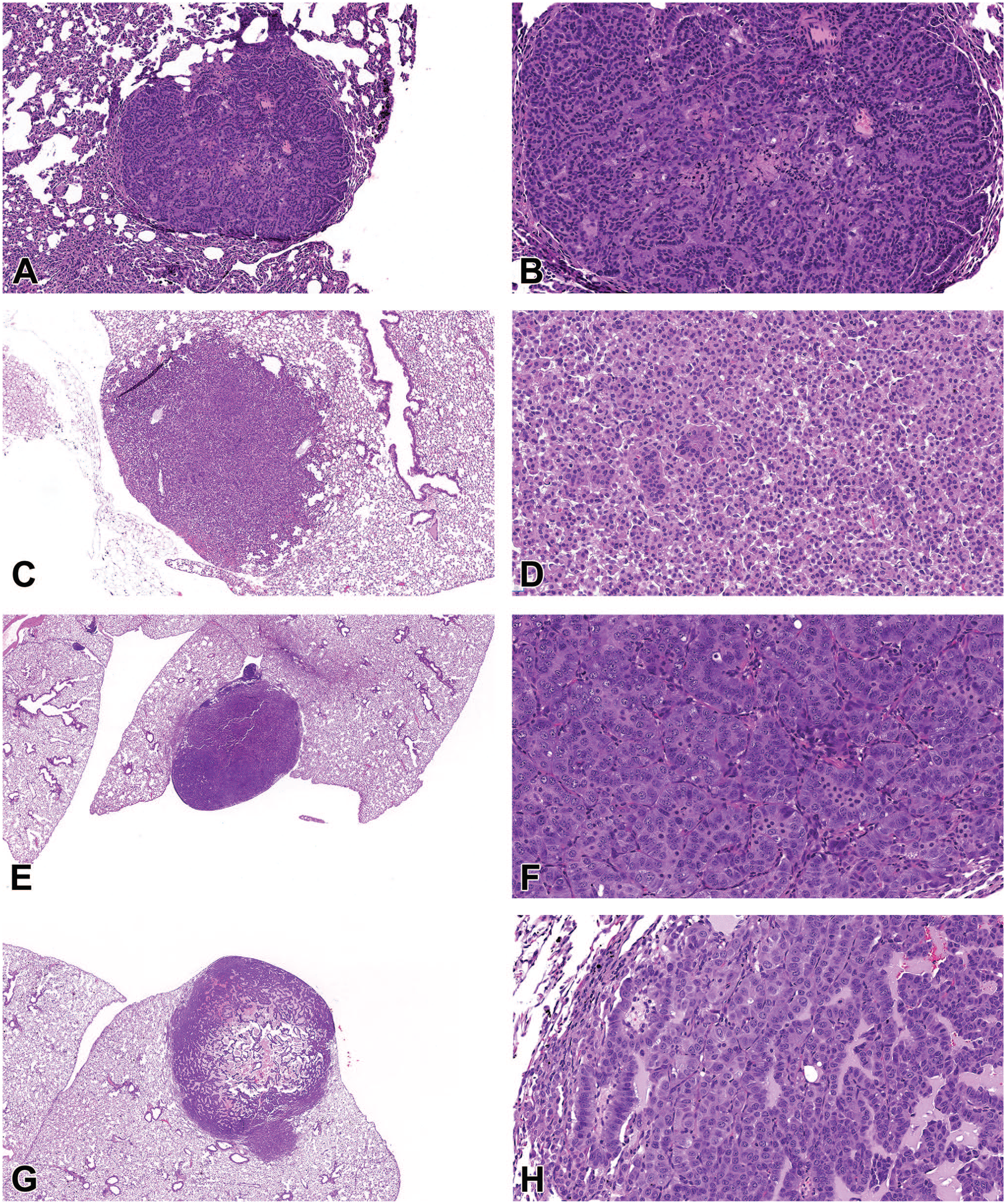
Mouse lung tumors, B6C3F1/N mouse. (A-D) Alveolar/bronchiolar adenoma. (A) A discrete, well-demarcated neoplasm is within the peripheral lung causing minimal compression of the surrounding lung tissue. H&E, 2X. (B) On higher magnification, deeply basophilic neoplastic cells are arranged in delicate papillary structures and are uniform in appearance with no apparent mitotic activity. H&E, 20X. (C) A discrete, well-demarcated neoplasm is within the peripheral lung causing minimal compression of the surrounding lung tissue. H&E, 4X. (D) On higher magnification, the neoplasm is composed of epithelial cells forming solid nests, supported by a fine fibrovascular stroma that completely obliterates the alveolar spaces. H&E, 20X. (E-H) Alveolar/bronchiolar carcinoma. (E) A fairly well-demarcated, solid, neoplasm is within the peripheral lung. H&E, 2X. (F) On higher magnification, the neoplasm is composed of pleomorphic epithelial cells arranged in papillary structures exhibiting increased mitotic activity and areas of single-cell necrosis. H&E, 20X. (G) A fairly well-demarcated neoplasm is within the peripheral lung with evidence of infiltration within the surrounding tissue and a necrotic center infiltrated by large numbers of macrophages. H&E, 2X. (H) On higher magnification, the neoplasm is composed of pleomorphic, epithelial cells arranged in papillary structures and supported by a fine, fibrovascular stroma. H&E, 20X. H&E indicates hematoxylin and eosin.
Voting Results for Cases 1-4 in the Mouse Lung.
Cases 1 and 4 provided examples of lung—alveolar/bronchiolar (A/B) adenoma (Figure 1A-1D) both papillary (Figure 1A and 1B) and solid (Figure 1C and 1D) types. Each example featured well-circumscribed, densely cellular neoplasms located within the lung periphery (Figure 1A and 1C) that exhibited a highly convex border with compression of the surrounding lung tissue. On higher magnification, neoplastic epithelial cells were uniform in appearance and arranged in delicate papillary structures (Figure 1B) or solid nests (Figure 1D) with little to no mitotic activity. Cases 2 and 3 featured examples of lung—A/B carcinoma (Figure 1E-1H). Each example featured moderately well-circumscribed neoplasms within the lung periphery with some evidence of invasion of the surrounding lung tissue (Figure 1E and 1G). On higher magnification, carcinomas were composed of pleomorphic cuboidal to columnar epithelial cells arranged in papillary structures that exhibited increased mitotic activity and cellular atypia (Figure 1F and 1H). For cases 1 through 3, the audience voting results (Table 1) agreed with the NTP diagnosis; however, 48% of audience members selected the diagnosis of A/B carcinoma rather than an A/B adenoma for case 4, demonstrating the difficulty of distinguishing between these tumor types in the mouse lung.
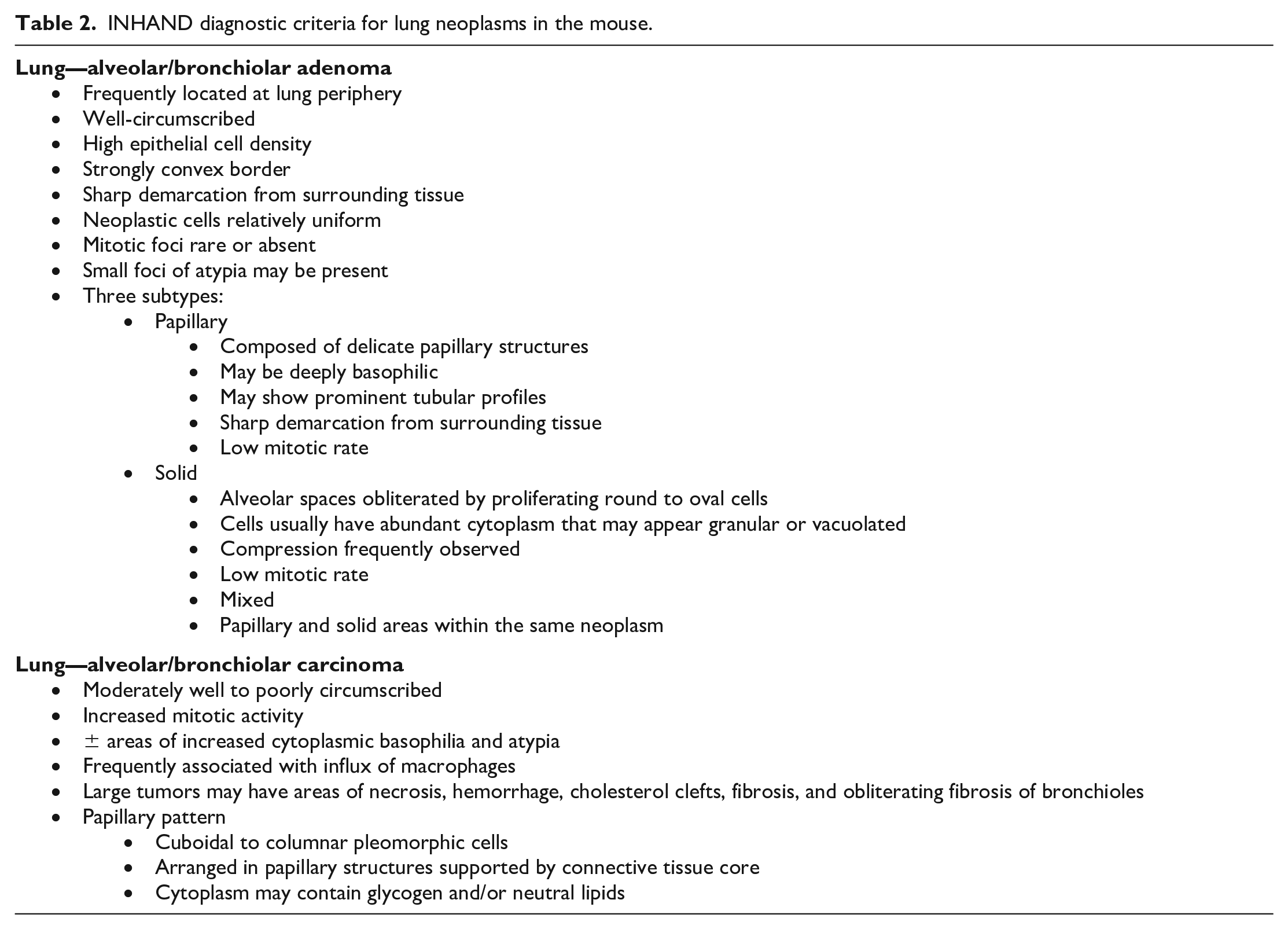
After voting was complete, Dr Quist then reviewed key diagnostic features of A/B neoplasms in the mouse lung as summarized in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guidance document (Table 2). 1 She then added her own creative twist to tie in the title of the presentation with a pneumonic that captured important points and considerations for lung tumor diagnosis in the mouse:
INHAND diagnostic criteria for lung neoplasms in the mouse.
The A, B, Cs of Mouse Lung Tumors
During the discussion, Dr Quist reminded the audience that A/B neoplasms in the mouse generally originate from alveolar type II cells; however, current literature indicates that A/B neoplasms may rarely originate from club cells (CCs).
1
Immunohistochemistry (IHC) can be a helpful tool to determine cell of origin as alveolar type II cells will stain positively for surfactant protein C (SP-C) and CCs will stain positively with CC-10.1,2 Dr Quist went on to conclude that it is important to remember that proliferative lesions in the mouse lung are part of a disease spectrum in which epithelial hyperplasia is considered a preneoplastic change that may progress to A/B adenoma and then A/B carcinoma, over time. It is well-understood that A/B neoplasm pathogenesis typically involves early activation/mutation of the
Dr Quist briefly discussed the concepts of “tumor plasticity” and “tumor heterogeneity” as they relate to lung neoplasms. In simple terms, tumor plasticity represents the tumor cells’ ability to reactivate developmental programs and pathways that provide “stem cell properties,” such as the ability to differentiate and self-renew. “Tumor heterogeneity” refers to (1) how tumors of the same type differ in different patients, (2) how tumor cells differ within a single neoplasm, and (3) how tumor cells differ between a primary and secondary neoplasm. 3 Dr Quist concluded that as we learn more about the molecular pathogenesis of mouse lung tumors, tumor plasticity and heterogeneity may become more important indicators of tumor behavior than morphology, as high tumor plasticity and tumor heterogeneity have been associated with increased drug resistance. 3
During the audience discussion, one member asked how we are able to conclude that most A/B neoplasms in the mouse lung arise from alveolar type II cells. Before providing an answer, Dr Quist acknowledged that she was not an expert but that the evidence within the current literature supported that most A/B neoplasms were of alveolar type II origin. She added that in the mouse lung, epithelial hyperplasia, a precursor of A/B neoplasia, typically involves alveolar type II cells, so that, it makes sense that any adenomas or carcinomas that arise from this proliferative change would also be of alveolar type II origin. Another audience member asked about when it would be appropriate to use the term “hyperplasia with atypia” as opposed to diagnosing adenoma in the mouse lung. Dr Quist explained that this term would be used when atypical cells were present within the hyperplastic lesion, but other features of adenoma were not observed, that is, well-demarcated, discrete with sharp convex border, solid, or papillary pattern, and so on. The last question was about what changes Dr Quist would look for to diagnose adenoma versus epithelial hyperplasia. Dr Quist said that more solid, discrete proliferations with compression of the lung tissue and cells forming papillary patterns would usually push her toward a diagnosis of adenoma.
Badly Behaved Glands: Spontaneous Reproductive Tract Lesions in a Female Cynomolgus Macaque
Dr Shambhunath Choudhary (Pfizer Inc., Pearl River, New York), presented some spontaneous lesions in the reproductive tract of a female cynomolgus macaque. An intact female macaque, aged approximately 6 years old, was part of an investigative study. Clinical findings included a large, firm, immobile mass (4 × 6 cm) palpated in the caudoventral abdomen and a small, less firm, slightly less mobile, pea-sized nodule in the left cranial aspect of the abdomen. An abdominal ultrasound revealed a thickened myometrium and a large fluid-filled structure to the right of the uterus (suspected ovarian cyst). At necropsy, the uterus was grossly enlarged and firm with a thickened wall and markedly and diffusely thickened mucosa (endometrium). The right ovary also had a large cystic fluid-filled structure.
Histopathologic evaluation of the uterus revealed characteristic uterine (Figure 2A and 2C) and ovarian lesions (Figure 2D and 2E), which correlated with clinical and gross pathology findings in the uterus and ovary, respectively.
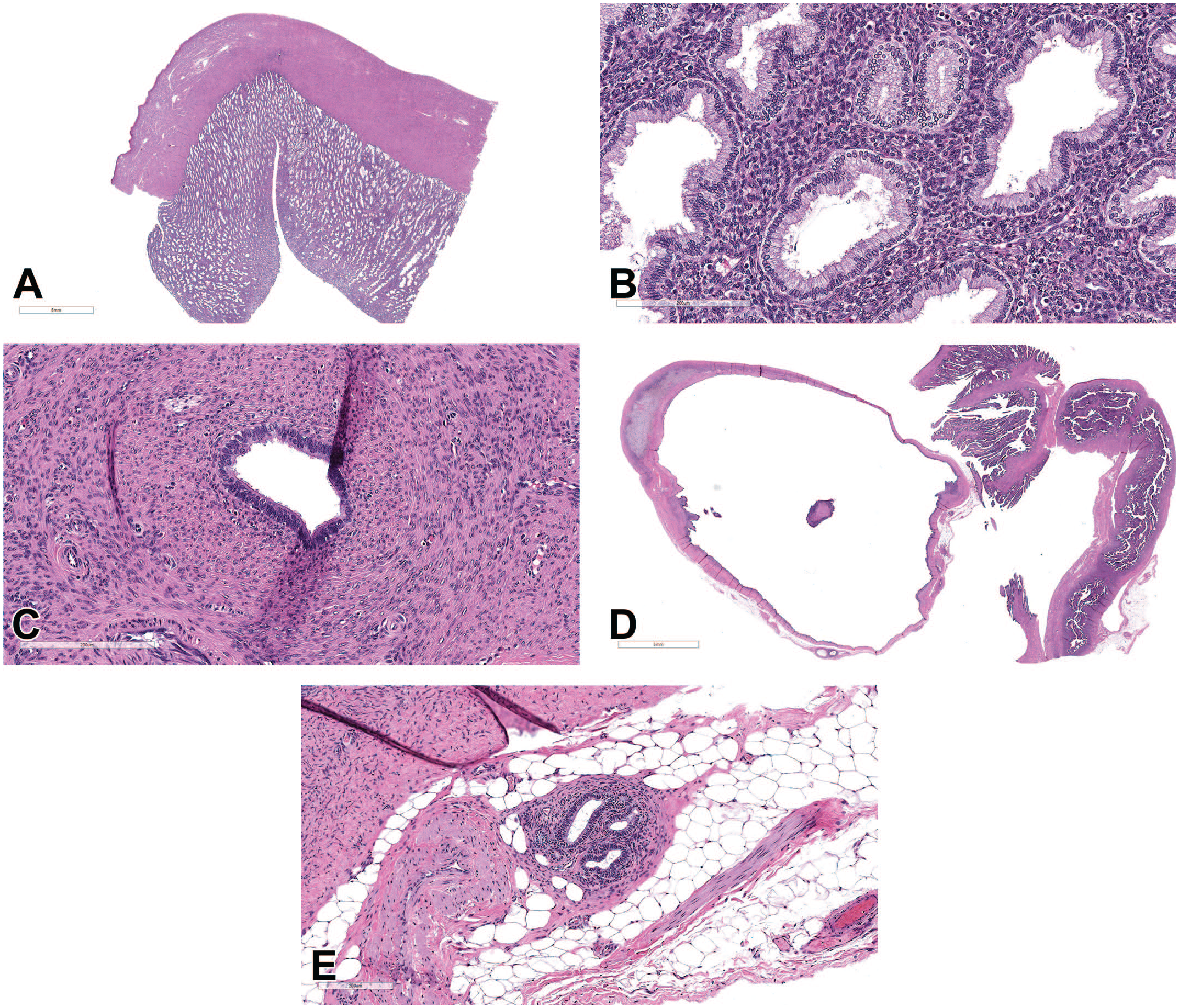
(A) Low-magnification image of transverse section of uterus from a cynomolgus monkey showing thickened endometrium. (B) Higher magnification of (A) showing proliferating endometrial glands and stroma within the endometrium. (C) High magnification showing a well-differentiated endometrial gland deep within the myometrium. (D) Low magnification of ovary containing a large cyst and multifocal aggregates of glands and stroma outside the ovary. (E) Higher magnification of (D) showing glands and stroma within the adipose tissue outside the ovary (H&E). H&E indicates hematoxylin and eosin.
Low-magnification and high-magnification photomicrographs of hematoxylin and eosin (H&E)-stained uterine sections of representative lesions observed in the animal were shown to the audience for voting (Figure 2A-2C). The most prominent cellular feature included diffusely thickened endometrium with extensive proliferation of endometrial glands and stroma (Figure 2A and 2B). Endometrial glands were lined with a single layer of ciliated columnar epithelium; glandular lumina were often empty and glandular crowding was not observed (Figure 2B). Occasionally, few, well-differentiated endometrial glands were observed deep within the myometrium (Figure 2C). The audience was polled for their preferred diagnosis to best capture the histologic changes. Voting choices and audience responses were: (1) endometrial polyps (1%), (2) endometriosis (5%), (3) diffuse endometrial hyperplasia (12%), (4) endometrial carcinoma (2%), (5) adenomyosis (5%), (6) endometrial adenoma (0%), (7) 1, 3, and 5 (6%), (8) 3 and 5 (64%), and (9) 1 and 5 (4%). Both Dr Choudhary and the audience favored choice 8, which included diffuse endometrial hyperplasia and adenomyosis.
Low-magnification and high-magnification photomicrographs of H&E-stained ovarian sections of representative lesions were then shown to the audience for voting (Figure 2D and 2E). The most prominent cellular feature included a large cyst lined by one to several layers of flattened cuboidal to low columnar epithelium (Figure 2D). Multifocally, within the adipose tissue outside the ovary, there were aggregates of gland-like structures surrounded by stromal cells; glands were lined with a single layer of ciliated columnar epithelium (Figure 2E). The audience was polled for their preferred diagnosis to best capture these histologic changes. Voting choices and audience responses were: (1) adenomyosis (5%), (2) ovary, endometriosis (9%), (3) embryonic duct-like remnants (2%), (4) ovarian cyst (2%), (5) uterine adenocarcinoma (1%), (6) 2 or 3 and 4 (73%), (7) 4 and 5 (7%), and (8) retroperitoneal fibromatosis (0%). Dr Choudhary, again, favored the most popular diagnosis (choice 6) in agreement with the audience. This set the stage for a quick review and discussion on spontaneous reproductive tract lesions in nonhuman primates (NHPs).
Similar to women, endometrial hyperplasia can be easily induced in macaques by treatment with estrogens.4,5 Proliferation of endometrial glands can be observed in both normal (follicular phase endometrium) and pathologic conditions (simple and complex hyperplasia). Normal follicular phase endometrium has orderly glandular proliferation. Whereas, simple endometrial hyperplasia is characterized by disorganized proliferation of glands with rare epithelial atypia (loss of cell polarity, increased nucleus-to-cytoplasm ratio, nuclear clearing with clumping of chromatin at the membrane, and a round to irregular nuclear contour) and occasional cystic changes in the glandular lumen. Glandular crowding (back-to-back crowding of glands) is the main feature of complex endometrial hyperplasia. Cystic changes and epithelial atypia are also commonly observed in complex endometrial hyperplasia. In women, complex endometrial hyperplasia with atypia has the strongest association with endometrial cancer risk. 4
Adenomyosis and endometriosis are commonly reported reproductive tract lesions in macaques that may occur simultaneously.4,6 Histologically, adenomyosis is characterized by the presence of well-differentiated glands ± evidence of stroma deep within the myometrium but without the presence of epithelial atypia and lack of scirrhous reaction. There should be enough distance between the lower border of the endometrium and the adenomyotic lesion to avoid misclassifying a normal histologic finding as adenomyosis. Endometriosis is characterized by the presence of “ectopic” endometrial tissue outside of the uterus. The ovary is the most common location for endometriosis in macaques; peritoneum/mesometrium, colon, and urinary bladder are other common locations for this lesion. Grossly, endometriosis appears as an adhesion or nodular lesion (“chocolate cyst”) in the caudal abdomen. 7 Histologically, an adhesion or nodular endometriosis lesion consists of glandular epithelium resembling that of the endometrium, surrounded by endometrial stroma, and evidence of hemorrhage with hemosiderin-laden macrophages. Endometriosis is a common reproductive tract lesion in macaques and more than 30% of sexually mature rhesus and cynomolgus macaques are affected in some colonies. 4 There are statistically significant risk factors for developing endometriosis in rhesus macaques, including hysterotomy (10-fold risk), long-term estrogen treatment (6-fold risk), and whole-body irradiation. 4
Embryonic duct-like remnants or mesonephric duct remnants in females are tubular, glandular or cyst-like structures lined by epithelium. 8 In males, under the influence of testosterone, the mesonephric duct develops into the epididymis, vas deferens and seminal vesicle. In females, the absence of testosterone causes mesonephric duct regression; however, this may be incomplete, and inclusions/remnants may persist. Some ductular/duct-like remnants may appear as “out of place” glands and may also resemble endometrial glands.
Dr Choudhary then briefly explained why a differential diagnosis of embryonic duct-like remnants was included with endometriosis to characterize the glandular structures observed outside the ovary. Although the lesion was observed in an adult cynomolgus macaque (endometriosis is commonly reported), the animal was comparatively young with no known statistical risk factors, such as history of hysterotomy, irradiation, or estrogen treatment. Characteristic gross lesions (adhesion or “chocolate cyst”) were lacking, and hemorrhage/hemosiderin-laden macrophages were not observed microscopically.
The main purpose of this presentation was to raise awareness among students, resident/trainees, and toxicologic pathologists about spontaneous reproductive tract lesions in NHPs. Use of the term hyperplasia in the uterus can be interpreted as complex hyperplasia with atypia by regulators when this may, in fact, not be the case. Hopefully, practicing pathologists will now be able to carefully chose the most appropriate terminology (normal endometrium, follicular phase, simple hyperplasia, and complex hyperplasia) when evaluating proliferative changes in the uterus. Pathologists should also be paying attention to reproductive tract changes specific to monkeys and other laboratory animals. For example, although endometrial hyperplasia can be readily induced in monkeys and there is a strong association between endometrial hyperplasia and endometrial cancer risk in humans, adenocarcinomas appear to be exceedingly rare as a spontaneous lesion in monkeys. 4
A Marrow Escape
Dr Richard Lang (Wake Forest University [WFU], Winston-Salem, North Carolina) presented a case of acute myeloid leukemia (AML) in a rhesus macaque (
The case presentation featured a 13-year, 3-month-old male rhesus macaque that received a single dose of 6.5 gray whole-body irradiation at 9 years of age and was part of a cohort studying the long-term effects of radiation exposure. Two months prior to necropsy, the animal developed a progressive and severe thrombocytopenia (from 192 to 20 K/µL), generalized pallor and anemia (hematocrit [HCT] decreasing from 39 to 11.8%), and lethargy. Treatments for the anemia included a blood transfusion and iron/vitamin B supplementation, and four days prior to necropsy dexamethasone injections were initiated for suspected immune-mediated thrombocytopenia. One day prior to necropsy the animal developed hematuria and supportive care was continued; however, the animal was found dead the following morning.
Prior to the clinical history, the audience was shown a gross photo of petechial hemorrhages on the facial skin (Figure 3A). Voting choices and results were as follows: vesicular dermatitis: macacine herpesvirus 1 (“herpes B virus”) (3%), vesicular dermatitis: simian varicella virus (8%), petechial hemorrhage: disseminated intravascular coagulation (DIC) (10%), maculopapular rash: measles virus (10%), petechial hemorrhage: thrombocytopenia (62%), and normal sex skin (7%). After voting, gross photos of all voting options were shown, with focus on the petechial hemorrhages on the serosal surfaces of the epicardium and urinary bladder (Figure 3B and 3C). “Sex skin,” the cyclical changes to the skin around the perineum in various species of female NHPs, was included as a confounder for the audience. Dr Lang explained that while sex skin typically presents in females as a change in the skin of the perineum and caudal half of the body, the facial skin can also be affected 9 and could, therefore, be confused with petechial hemorrhage. The audience was then shown a subgross image and a higher-magnification photomicrograph of the liver with round cell infiltration of a portal tract (Figure 3D and 3E). The voting choices and results were as follows: chronic hepatitis (4%), myeloid sarcoma (4%), extramedullary hematopoiesis (EMH) (53%), histiocytic sarcoma (9%), AML (19%), and lymphoma (10%). The audience was then shown sequentially magnified photomicrographs of the humeral bone marrow (Figure 3F and 3G). The voting choices and results were as follows: normal bone marrow (0%), myeloid hyperplasia (19%), AML (44%), histiocytic sarcoma (13%), myelodysplastic syndrome (17%), and myelitis (immune-mediated thrombocytopenia) (7%). Before revealing the answer, the audience was again polled, this time to determine which IHC panel would be most appropriate; voting choices and results were as follows: CD3/CD20/pan-cytokeratin/vimentin/chromogranin A (7%), CD56/CD3/CD4/CD8/MUM1/CD20 (28%), CD34/CD3/CD20/MPO/CD61/Ham56 (59%), and CD31/MUM1/CD3/CD20/pan-cytokeratin (5%). IHC staining of the bone marrow was shown (Figure 3H), and the audience was once again asked for their final morphologic diagnosis for the bone marrow lesion. The voting choices and results were as follows: normal bone marrow (0%), myeloid hyperplasia (12%), AML (62%), histiocytic sarcoma (8%), myelodysplastic syndrome (11%), and myelitis (immune-mediated thrombocytopenia) (7%).
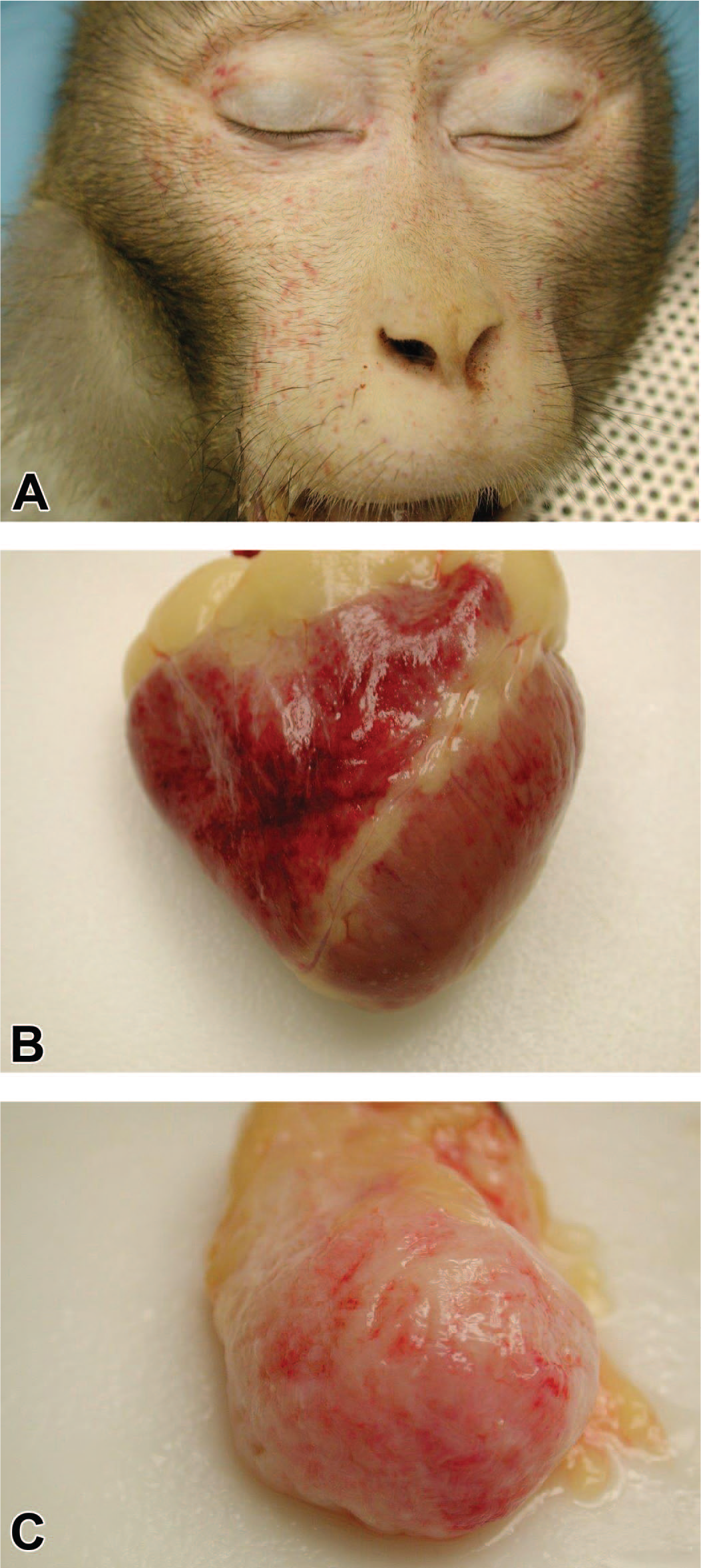
(A-C) Acute myeloid leukemia in a 13-year-old male rhesus macaque that received 6.5 Gy whole-body irradiation and had a 2-month history of progressive thrombocytopenia. A gross photograph (A) demonstrates petechial hemorrhages around the face, which are also present on the epicardium (B) and serosal surface of the urinary bladder (C). A low-magnification H&E-stained photomicrograph of the liver
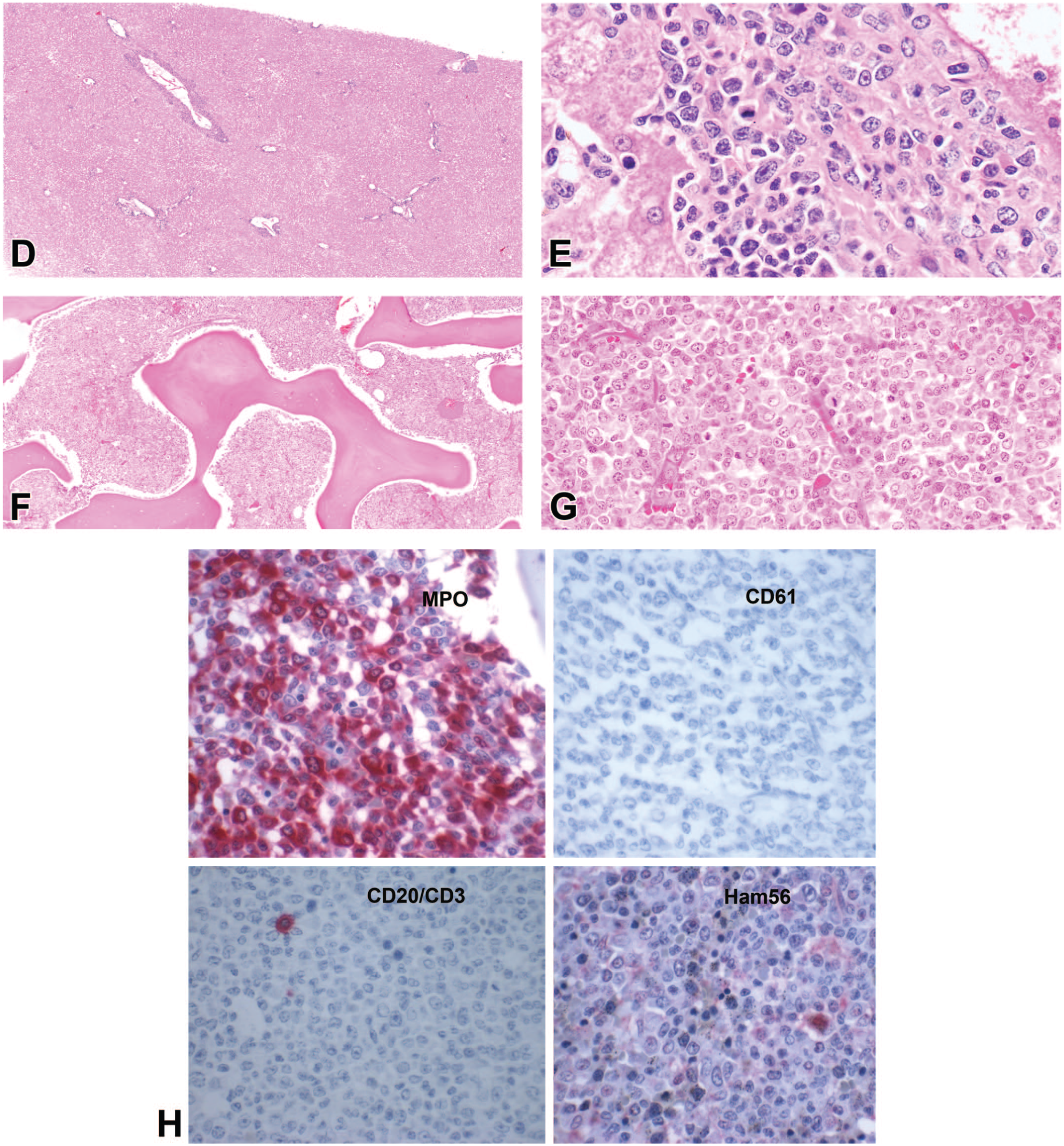
(D) demonstrates the infiltration and expansion of a portal tract by round cells, which at higher magnification (E) predominantly have round nuclei with vesiculate chromatin and prominent nuclei, admixed with rare mitotic figures and cells with segmented nuclei. A low-magnification H&E-stained photomicrograph of the humeral bone marrow (F) is markedly hypercellular, and higher magnification (G) demonstrates effacement of the marrow by a similar population to that observed in the portal tract. Four immunohistochemistry-stained panels of humeral bone marrow (H) demonstrate diffuse strong cytoplasmic reactivity to myeloperoxidase (MPO), and negative staining for CD61, CD3/CD20, and Ham56. H&E indicates hematoxylin and eosin.
Based on the morphology, extent of marrow effacement (≥20% neoplastic blast cells), 10 and IHC staining, the neoplasm was diagnosed by contributors from WFU as AML. More specifically, it was considered to be most consistent with “AML, not otherwise specified (NOS): AML with minimal maturation” in accordance with the updated World Health Organization (WHO) classification system. 10 Most categories of AML in humans are now characterized based on the underlying chromosomal translocations (i.e., AML with t(8;21)(q22; q22)), 10 but these specific chromosomal abnormalities have not yet been elucidated in nonhuman primates. Interestingly, in this case, the neoplastic myeloblasts in the bone marrow and infiltrated organs (liver and kidney) did not stain with CD34, which is a common marker of hematopoietic stem cells. 11 Approximately, 30% of human AML cases lack CD34 expression, 12 presumably due to loss during malignant transformation or being derived from the small subset of CD34− hematopoietic stem cells in bone marrow. 12 The prognostic implications of CD34 staining are controversial in human AML, 11 but in general, CD34+ neoplasms are associated with a poorer prognosis.11,12
Spontaneous hematopoietic malignances seem to be rare in NHPs compared to humans and rodents.13,14 AML is a common malignancy associated with irradiation in humans and mice,15-19 with a specific-dedicated category of “therapy-related myeloid neoplasms” in the latest WHO classification. 10 Irradiation is associated with a higher incidence of neoplasia, particularly sarcomas in macaques 19 ; however, AML is infrequent in irradiated animals with only 3 reported cases.20-22 Future studies are needed to explore the reasons for this discrepancy, which could provide further insight in the prevention and management of post-irradiation neoplasms. Despite this apparent difference in delayed irradiation effect, the acute changes in macaques are very similar to humans and provide an important research model. 23
Extramedullary infiltration (EMI) and myeloid sarcoma (discrete masses outside the bone marrow) are common in human AML, 24 and particularly in CD34− subtypes. 12 It can be difficult to differentiate between EMH and EMI when they occur concurrently in AML cases, especially as hematopoietic stem cell markers like CD34 are variably positive in both EMH and EMI.12,25 Further complicating the issue is that mass-forming EMH has been reported in humans without other hematologic disorders,26,27 and myeloid sarcoma may arise prior to bone marrow involvement in AML and is often misdiagnosed (as EMH, inflammation, melanoma, or lymphoma). 27
In the final portion of the presentation, Dr Lang inquired if the audience had experience with this rare neoplasm of NHP and/or thoughts on distinguishing between EMH and EMI in cases of AML in any species. One audience member asked about evidence of platelet sequestration/concurrent immune-mediated thrombocytopenia in the spleen and mentioned that in her experience the distribution and specific pattern of hematopoietic cells in organs like the liver have helped in distinguishing EMH and EMI. Another question arose from a different audience member as to whether simian varicella virus had been conclusively ruled out, as it has been associated with thrombocytopenia and may have contributed. Dr Lang replied that to his knowledge the animal did not have a history of lesions or positive serologic tests, but could not completely rule out an infectious contribution. Another audience member asked about the CD34 staining and controls, to which Dr Lang mentioned that the initial CD34 staining was performed on decalcified sections which can lead to false negatives, but that the staining in the liver (no decalcification) was similarly negative and also had a positive internal control (endothelium). Another question was posed about the use of Ham56, to which Dr Lang responded that at the time of necropsy it was the best marker of monocytes available for the species. Another participant asked if vincristine had been given for the thrombocytopenia as it has been associated histologic artifacts. Dr Lang replied that it had not been administered but discussed how the dexamethasone may have induced lymphocyte apoptosis and cells reminiscent of nucleated red blood cell precursors, complicating the distinction between EMH and EMI in the case (especially in the kidney).
Dark Matter in (Alveolar) Space: Pulmonary Findings in Mice and Rats From an Inhaled Particulate Study
Dr Debra Tokarz (Experimental Pathology Laboratories, Inc. [EPL], Durham, North Carolina) presented four cases highlighting findings in the lung from companion 2-year chronic toxicity/carcinogenicity studies in mouse and rat conducted by the National Toxicology Program at the National Institute of Environmental Health Sciences (NTP/NIEHS), in which the route of administration was whole-body inhalation. Dr Tokarz acknowledges contributions from Drs Mark Cesta (NTP/NIEHS), Erin Quist (CRL), Daphne Vasconcelos and Mike Ryan (Battelle) and imaging help from Emily Singletary, Maureen Puccini, and Michael Carden (EPL).
Case 1 was lung from a 2-year-old female B6C3F1/N mouse. In the lung, coarse granules and clumps of black material were present within the cytoplasm of alveolar macrophages in the alveolar spaces and within the alveolar and perivascular interstitium (Figure 4A). The voting choices and results were as follows: foreign body (0%), foreign material (27%), pigment (65%), dark matter (8%), and other (0%). As a follow-up question, audience members were polled on whether to include a subsite for the foreign material diagnosis in this study. The voting choices and results were as follows: yes—alveolus (21%), yes—interstitium (3%), yes—macrophage (66%), and no—subsite (10%). The black granular material in this case was consistent with the administered test article, based on the known properties of the test article and the occurrence in only test article–exposed animals. “Foreign material” is the NTP preferred terminology for inhaled known/presumed test material, 28 and was the term applied to this finding in this study. The severity of foreign material accumulation in this study increased with dose. For NTP studies, “pigment” is reserved for endogenous pigments, such as hemosiderin, and “foreign body” for environmental or other aberrant material, such as bedding or hair shafts. 28 Most audience respondents favored a diagnosis of pigment. Some audience members preferred this term because it is more descriptive, in keeping with the general trend for INHAND terminology, and avoids presumption or interpretation of the source of the material. Another suggestion offered by the audience was to use a slash term of “foreign material/pigment” or use of a modifier to the “foreign material” term, that is, “foreign material, pigmented,” as a way of conveying some description of the material in summary tables. “Foreign material/pigment” is the current INHAND recommended term for endogenous or exogenous pigmented material in the lung. 29
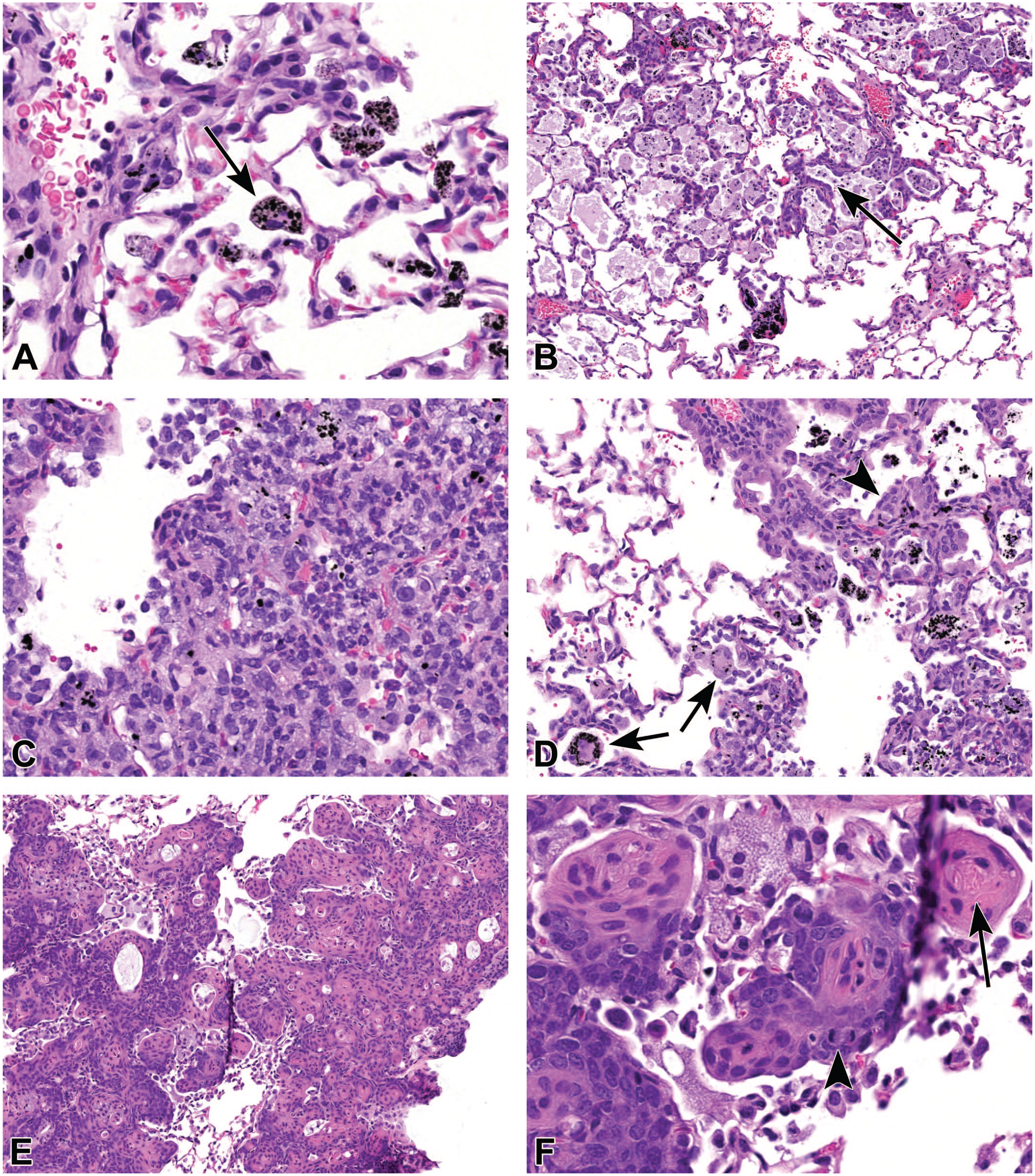
Examples of lung findings from B6C3F1/N mice (A, C-F) and Sprague Dawley rats (B) from a National Toxicology Program chronic whole-body inhalation study. (A) Coarse granules and clumps of black material are present within alveolar macrophages in alveolar spaces and within the interstitium (arrow). (B) In the rat, alveolar spaces contain individual to aggregated macrophages with abundant foamy cytoplasm, sometimes with intracellular black granular material. Alveoli are multifocally lined by a single layer of cuboidal pneumocytes (arrow). (C and D) In mice, alveolar spaces are obscured by consolidated foci of inflammation composed primarily of macrophages admixed with neutrophils and lymphocytes (C). Affected foci contain multinucleated giant cells with intracellular black granular material (D, arrows) and alveoli lined by a single layer of cuboidal pneumocytes (D, arrowhead). (E and F) The alveolar parenchyma is obscured by a poorly circumscribed mass composed of solid nests (E). Higher magnification (F) shows neoplastic cells making up the nests progress in a concentric but disorderly manner from basophilic and plump to flattened and eosinophilic and contain central keratin (F, arrow). There are frequent mitotic figures (F, arrowhead).
Case 2 was from a 2-year-old male Sprague-Dawley rat. The lung finding in this case consisted of accumulation of individual to aggregated macrophages with abundant foamy cytoplasm within alveolar spaces (Figure 4B). Alveoli within affected areas were multifocally lined by a single layer of cuboidal pneumocytes. The voting choices and results were as follows: infiltration, cellular, histiocyte (7%); inflammation, chronic (4%); inflammation, granulomatous (6%); hyperplasia, alveolar epithelium (3%); infiltration, cellular, histiocyte and hyperplasia, alveolar epithelium (59%); inflammation, chronic and hyperplasia, alveolar epithelium (6%); inflammation, granulomatous and hyperplasia, alveolar epithelium (10%); and other (4%). The diagnoses used for this finding in the rat study were infiltration, cellular, histiocyte, and hyperplasia, alveolar epithelium. Histiocyte infiltration, rather than inflammation terminology, was used because it occurred in both control and test article-dosed groups in this study and is similar to histiocytic infiltrates commonly observed in aged rats. 30 This finding has also been referred to as alveolar histiocytosis. Compared to controls, there was a test article, dose-related exacerbation of the finding in both males and females. Foreign material, as described in case 1, was typically present in macrophages within foci in the test article-dosed animals but was absent in histiocytic infiltrates in controls. A small amount of alveolar epithelial hyperplasia was sometimes associated with the histiocytic infiltrates in controls; this minimum level of hyperplasia was described but not separately diagnosed. However, when hyperplasia exceeded this expected amount, as in this case, a separate diagnosis of alveolar epithelium hyperplasia was given. During the discussion, audience members noted that among the histiocytic infiltrate in the lung there appeared to be extracellular foamy amphophilic material within alveolar spaces. Potential considerations discussed included cellular debris, surfactant, or vehicle, and it was suggested that it may be useful to further characterize this material to understand potential adversity of these changes.
Case 3 was from a 2-year-old female B6C3F1/N mouse. In the lung, there were consolidated foci of foamy macrophages and multinucleated giant cells, mixed with neutrophils and lymphocytes (Figure 4C). Alveoli within affected areas were multifocally lined by a single layer of cuboidal pneumocytes (Figure 4D). Foci of necrotic debris occurred in some foci. The voting choices and results were as follows: infiltration, cellular, histiocyte (5%); inflammation, chronic (5%); inflammation, granulomatous (6%); hyperplasia, alveolar epithelium (0%); infiltration, cellular, histiocyte and hyperplasia, alveolar epithelium (19%); inflammation, chronic and hyperplasia, alveolar epithelium (38%); inflammation, granulomatous and hyperplasia, alveolar epithelium (24%); and other (3%). The diagnoses used for this finding in the mouse study were inflammation, granulomatous and hyperplasia, and alveolar epithelium. Compared to the tissue reaction associated with the test article in the rats, as demonstrated in case 2, the tissue reaction associated with the test article in mice was more mixed inflammatory, but dominated by macrophages and multinucleated giant cells, hence the designation of granulomatous. These foci of granulomatous inflammation only occurred in test article–exposed mice.
Case 4 was from a 2-year-old male B6C3F1/N mouse. This lung contained a poorly circumscribed mass composed of solid nests of cells that progressed in a concentric but disorderly manner from basophilic and plump to flattened and eosinophilic (Figure 4E and 4F). Some nests contained central keratin. There was mild anisocytosis and anisokaryosis, and frequent mitotic figures and individual necrotic cells. Foamy macrophages, which sometimes contained scant foreign material, were intermingled with the neoplastic nests. The voting choices and results were as follows: metaplasia, squamous (29%); epithelioma, cystic keratinizing (6%); carcinoma, adenosquamous (45%); carcinoma, squamous cell (19%); and other (0%). The study pathologist, reviewing pathologist, and PWG participants favored a diagnosis of squamous cell carcinoma for this case. Squamous cell carcinoma in the rodent lung is similar in appearance to squamous cell carcinoma arising elsewhere. It is characterized by clusters and nests of cells with distinct intercellular bridges and/or central keratinization. The concentrically arranged cells have a disorganized appearance and cellular atypia that differentiates carcinoma from other benign squamous proliferations. Mitotic figures are often frequent. These tumors may show marked pleomorphism with giant cells, invasion of the adjacent parenchyma, and/or a scirrhous response. 1 Spontaneous primary pulmonary squamous cell carcinomas appear to be very rare in mice. 31 The other diagnostic options were squamous metaplasia, which is characterized by replacement of airway epithelium by stratified squamous epithelium with orderly maturation, with or without keratinization. Areas of squamous metaplasia generally maintain normal pulmonary architecture and this finding may occur as a response to chronic irritation. 1 Cystic keratinizing epitheliomas are irregularly shaped cysts lined by squamous epithelium with central keratinization. Foci of disorganized maturation occur within the squamous epithelial lining, distinguishing these from simple squamous cysts. Cystic keratinizing epitheliomas are reported only in rats. Adenosquamous carcinomas are composed of significant amounts of both adenocarcinomatous (i.e., resemblance to alveolar-bronchiolar carcinoma or acinar carcinoma) and malignant squamous neoplastic cells. The squamous component may undergo keratinization. In this case, the tumor lacked the adenocarcinomatous component.
Dr Tokarz discussed common test article-related findings in NTP/NIEHS studies involving inhaled particulate matter. Common potential target tissues for inhaled particulates include nose, larynx, lung, and the lymph nodes draining the lung, including mediastinal and tracheobronchial lymph nodes.32-35 Common findings in the lung include accumulation of foreign material (presumed/known test-article) in alveolar macrophages, air spaces, interstitium, and bronchial-associated lymphoid tissue. Alveolar epithelial hyperplasia, alveolar histiocyte infiltration/inflammation, and interstitial fibrosis are other commonly reported findings. Some inhaled particulates have been associated with increased incidence of alveolar-bronchiolar neoplasms in rodent carcinogenicity studies.34,35 Although not seen in this study, inhaled particulate matter can frequently cause changes in the upper respiratory system. Common findings in the nasal cavity have included inflammation and hyaline droplet accumulation in respiratory epithelium. Common findings in the larynx have included inflammation and squamous metaplasia. The most frequent finding in bronchial and mediastinal lymph nodes has been infiltration of macrophages containing the test material, but other findings, such as fibrosis and necrosis have been seen in some particulate studies.
Let’s Get Ready to Stumble: Degenerative Myelopathy and Neuropathy in Xenografted NSG Mice
Dr Mark Hoenerhoff (University of Michigan Medical School) presented a case of degenerative myelopathy and neuropathy in a 12-week-old, female, nonobese diabetic (NOD).Cg.-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mouse, which was part of a cohort of 30 mice receiving patient-derived xenografts (PDX) of human breast carcinoma. Several animals developed progressive, unilateral to bilateral, hindlimb paresis, and/or paralysis, sometimes progressing to forelimb involvement, over 3-4 weeks following xenograftment. Dr Hoenerhoff provided a video that demonstrated the typical clinical presentation of afflicted animals, illustrating a hunched and scruffy appearance, weight loss, and hindlimb paralysis with forelimb paresis in an affected mouse.
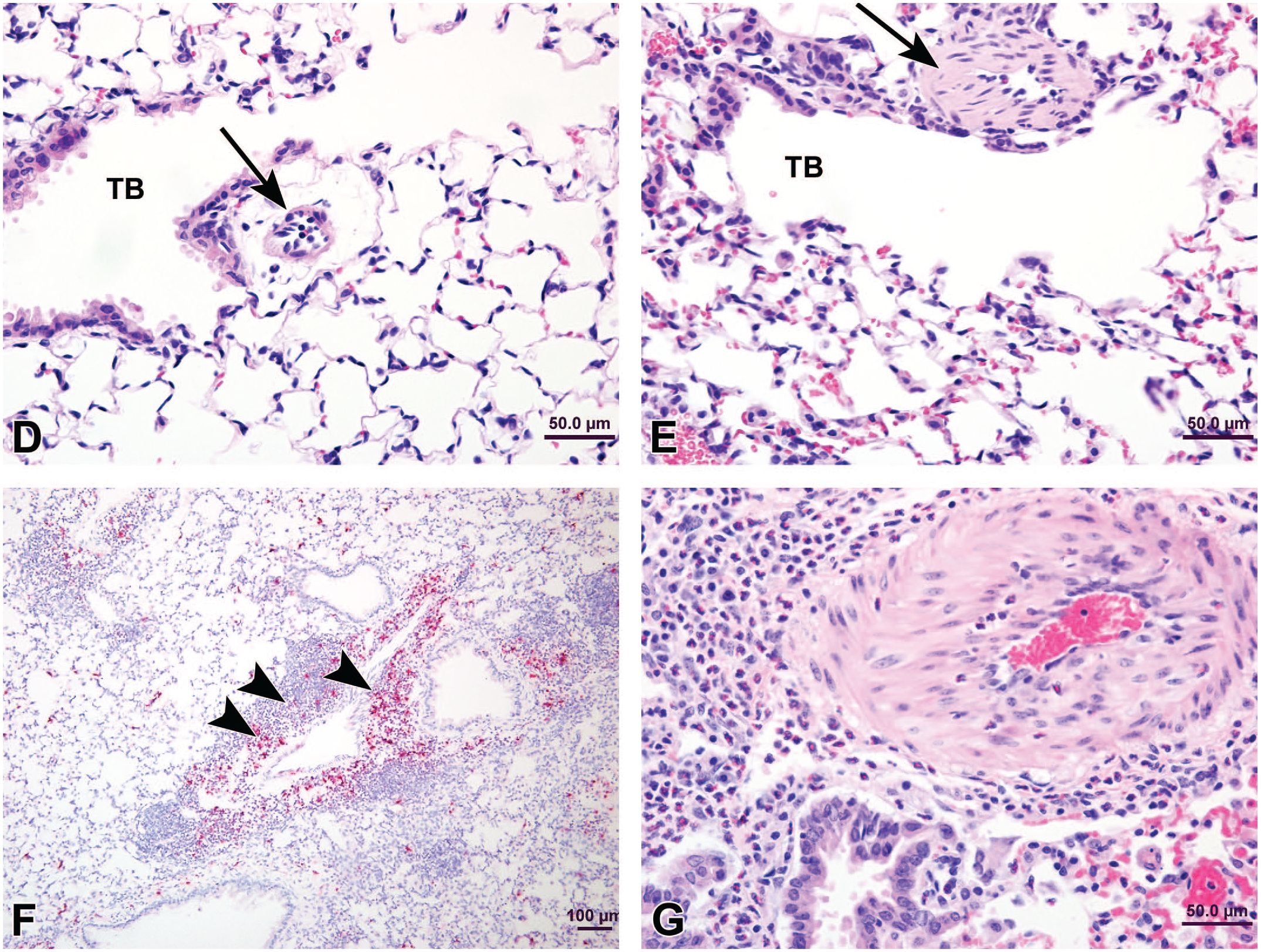
Representative histologic images of spinal column were presented to the audience (Figure 5A-5E), including cross-sections of vertebral segments, spinal cord, and spinal nerve roots and ganglia. Sections of associated peripheral nerve (sciatic nerve) were also provided for microscopic evaluation (Figure 5F). Spinal cord lesions were illustrated using segments of thoracolumbar spinal cord, and were characterized by vacuolation, dilation of myelin sheaths, presence of digestion chambers containing gitter cells, and swollen axons (spheroids) within the ventral funiculi of the spinal cord, ventral spinal nerve roots, and ganglia. Similar findings occurred in sections of sciatic nerve.
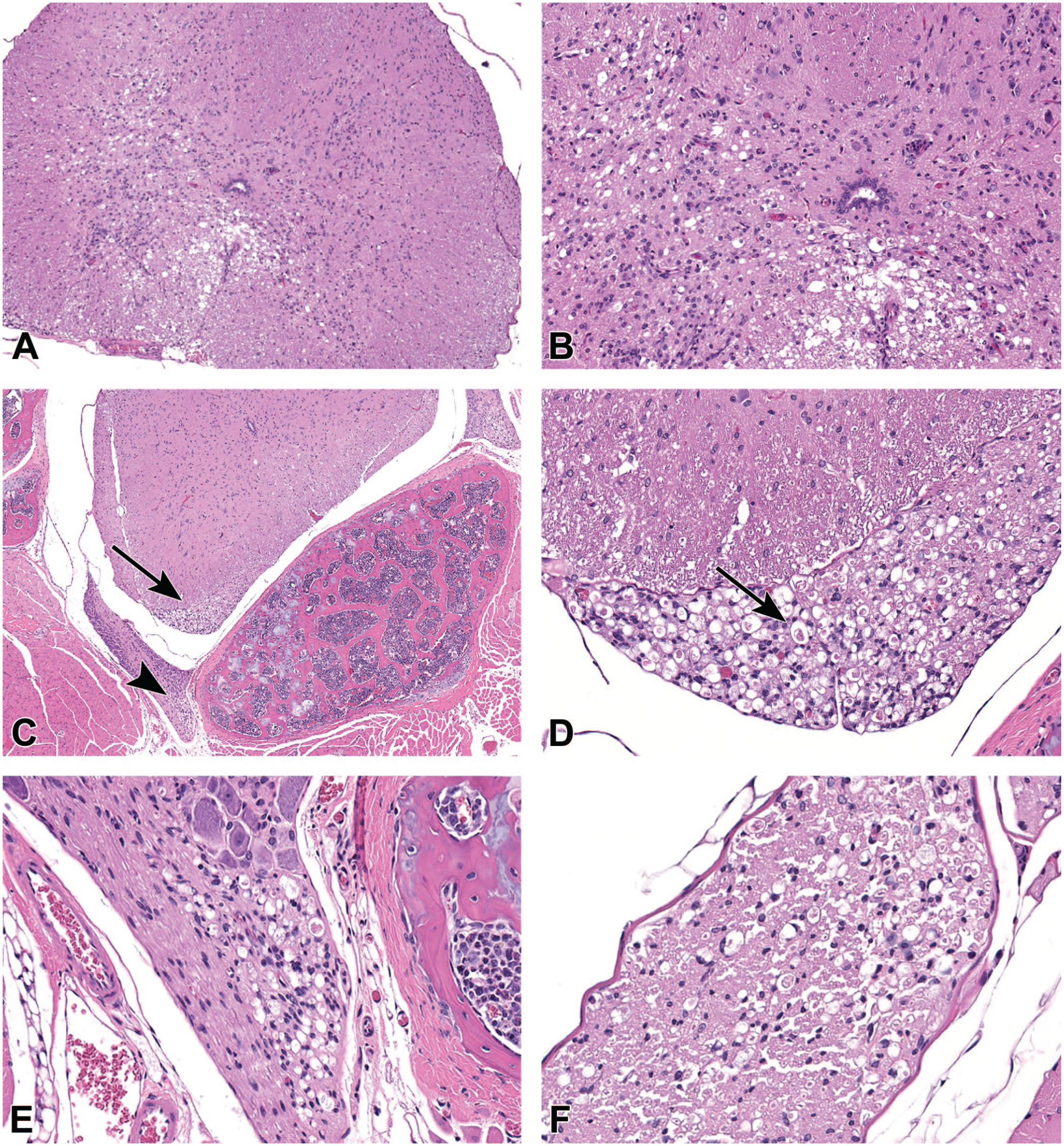
Degenerative myelopathy, thoracolumbar spinal cord, NSG mouse. (A) There is locally extensive vacuolation present ventrally and laterally within the white matter of the spinal cord. H&E, 10X. (B) Vacuolation is accompanied by digestion chambers containing gitter cells, swollen axons (spheroids) and mild gliosis. H&E, 20X. (C) There is vacuolation extending within the ventral spinal nerve root (arrow) and spinal nerve (arrowhead). H&E, 4X. (D) Higher magnification of the lesion at arrow in Figure B, showing dilation of myelin sheaths, vacuolation, and axonal swelling (spheroids, arrowhead) in the ventral nerve root. H&E, 20X. (E) Degenerative neuropathy, thoracolumbar spinal cord, NSG mouse. Higher magnification of lesion at arrowhead in (B), demonstrating vacuolation, spheroid formation, and dilation of myelin sheaths within the ventral spinal nerve. H&E, 20X. (F) Dilation of myelin sheaths and spheroid formation in the associated sciatic nerve. H&E, 20X. H&E indicates hematoxylin and eosin.
The audience was asked to vote for their preferred diagnostic term, with voting choices and results as follows: (1) vacuolation, neuronal (3%); (2) demyelination (11%); (3) degeneration, axonal (51%); (4) radiculoneuropathy (8%); (5) degeneration, nerve fiber (26%); and (6) intramyelinic edema (1%). Although the majority of respondents opted for the diagnosis of “degeneration, axonal,” the preferred diagnosis of the presenter was “degeneration, nerve fiber.” Dr Hoenerhoff proceeded to discuss each of the diagnostic terms and the rationale for the choice of “degeneration, nerve fiber” as the preferred choice. According to the newly revised INHAND manuscript on Proliferative and Nonproliferative Lesions of the Rat and Mouse Central and Peripheral Nervous Systems: New and Revised INHAND Terms, 36 the catch-all diagnosis of “degeneration, nerve fiber” is the preferred term since the affected site is the nerve fiber, but it is uncertain whether the axon (degeneration, axonal) or the myelin sheath (demyelination) is the target of injury. To differentiate between axonal or myelin damage as the primary lesion, additional histochemical and/or immunohistochemical labeling is necessary. To document axonal damage, Bielschowsky’s silver histochemical staining or antineurofilament protein IHC would be warranted, and to identify the primary lesion as demyelination, Luxol fast blue staining or antimyelin basic protein labeling would be used. Dr Hoenerhoff then showed examples of Bielschowsky’s silver staining demonstrating axonal staining and Luxol fast blue staining of myelin, with an example of axonal swelling and degeneration using Bielschowsky’s silver staining (image care of Dr Brad Bolon).
Dr Hoenerhoff then discussed the clinical differential diagnostic list for the clinical presentation of affected animals on this study. Since several animals were becoming clinically affected over a short period of time, necessitating euthanasia, steps to rapidly determine the cause of disease was necessary while tissue processing for histology was being performed. General overarching diagnostic differentials discussed with the clinical veterinary staff at the University of Michigan included (1) factors related to the experimental manipulation (surgical, anesthetic complications), (2) toxicity (test-article effect of potential study compound or therapeutic if administered), (3) tumor effect (cancer cachexia, tumor metastasis), and (4) infectious disease. Factors related to the experimental manipulation were ruled out as a cause, since multiple mice were affected, with similar clinical signs, the surgical procedure was not technically difficult (subcutaneous implantation), and the surgeon performing the procedure was very experienced, having previously performed numerous similar surgeries. No test-article or therapeutic was being tested as this point in the experimental protocol, so that, a toxicity was not considered as a potential cause. Since cancer cachexia presents as overall systemic debility, and tumor metastasis in these models are very rare unless specifically using an intracardiac route of tumor cell engraftment, factors related to a potential tumor effect were also ruled out. In addition, there was no evidence of gross metastasis to the spine or other organs, and histology ultimately defined the lesion process. This left the primary diagnostic differential of infectious disease as a likely cause of clinical signs in affected animals.
Based on the clinical history, experimental manipulation, and histologic findings, Dr Hoenerhoff posed the question of which infectious agent would be most likely to cause such lesions and clinical disease in immunosuppressed mice. Dr Hoenerhoff provided a list of differentials for paralysis in immunodeficient mice, including (1) neurotrophic mouse hepatitis virus (MHV, mouse coronavirus), (2) Theiler’s murine encephalomyelitis virus (TMEV), 3) mouse polyomavirus, (4) lactate dehydrogenase-elevating virus (LDV), (5) reovirus serotype 3, and (6) lymphocytic choriomeningitis virus (LCMV), and asked the audience for their top differential. Top choices from audience polling included TMEV (37%), LCMV (28%), mouse polyomavirus (16%), and LDV (13%), with minority votes for reovirus (5%) and MHV (1%). Dr Hoenerhoff then described how the etiologic diagnosis was made, and details for each diagnostic rule out.
Each of the differentials for hindlimb paralysis in immunodeficient mice listed above also involve brain or brainstem lesions as well as spinal cord lesions, except for LDV, which was the ultimate etiologic diagnosis in this case. MHV is associated with necrotizing encephalomyelitis with demyelination, and TMEV causes neuronal necrosis, inflammation, and demyelination in the ventral horn of the spinal cord and various portions of the brain (but generally, only reproducible through experimental intracranial injection). Lesions caused by polyomavirus are generally only inducible experimentally and include brainstem lesions. Reovirus serotype 3 causes paralysis secondary to encephalomyelitis, as well as diarrhea and runting in neonatal mice; however, adult mice are resistant to natural infection and develop paralysis only with experimental infection. Finally, LCMV is associated with a meningoencephalitis in addition to spinal cord involvement. 37
Having discussed this, Dr Hoenerhoff mentioned the caveat that the NSG mouse is exquisitely immunosuppressed, so that, making this conclusion based on traditional presentation of disease would be unreliable, and that the observed disease process could have actually been a result of infection by any of the above viruses in reality, rather than restricted to LDV based on distribution or severity of lesions. Dr Hoenerhoff revealed that in fact, the ultimate etiology was determined based on extensive fecal PCR Rodent Infectious Agent (PRIA) screening, PCR testing of xenograft tumors, and environmental screening of pooled plenum swabs of animal caging racks. All other viral pathogens were ruled out on PRIA screening of fecal pellets, and all xenograft tumors from affected animals tested positive for LDV by PCR. Furthermore, pooled plenum swabs of racks housing affected animals were positive for LDV and negative for other pathogens, while pooled swabs on racks housing unaffected animals were negative for all pathogens.
Dr Hoenerhoff then provided background information on the pathogenesis of LDV in mice, and factors influencing LDV infection and disease progression. LDV is an arterivirus, related to Porcine Reproductive and Respiratory Syndrome (PRRS) virus, equine arteritis virus, and simian hemorrhagic fever virus. 38 Transmission of the virus in the natural setting is predominantly through animal-to-animal contact (fighting, bite wounds),39,40 but in the laboratory setting, the most common mode of transmission is through use of contaminated biologics of mouse origin, including basement membrane matrix, cell lines, and transplanted tumors. 41 There are two predominant forms of the virus, a nonneuropathic (LDV-P) and two neuropathic (LDV-V and LDV-vx) strains. Following infection, LDV causes a life-long viremia in most mice, with animals generally remaining asymptomatic, making it difficult to recognize the presence of this virus in animal facilities.
LDV was first recognized as a cause of transient increase in serum lactate dehydrogenase (LDH) in affected animals. 42 This increase in serum LDH is due to the fact that the virus replicates in, and causes cytolysis of, a subset of macrophages responsible for clearance of this enzyme from serum. 41 Other than a transient elevation of this serum enzyme, mice remain generally asymptomatic. However, in susceptible mouse strains and immunodeficient mice, animals infected with neuropathic strains of LDV can develop a progressive, ascending paresis and paralysis, also known historically as age-dependent poliomyelitis (ADPM).43-45
Development of ADPM is predominantly dependent on three factors in animals infected with neuropathic LDV: (1) presence of endogenous murine retrovirus, such as murine leukemia virus (MuLV), (2) strain genetics, and (3) concurrent immunosuppression, whether age-related (physiologic) or induced (chemically or genetically induced). In terms of strain genetics, for example, C58 and AKR mouse strains are susceptible to development of ADPM. This is because they are (1) homozygous for the permissive allele for N-tropic retroviral replication (
In terms of immune function, immunosuppression plays a critical role in disease progression, whether age-related or induced. Traditionally, in LDV susceptible immunocompetent strains (C58, AKR), defects in immune function related to advanced age are responsible for the onset of poliomyelitis. Defects in cell-mediated immunity allow for the replication and expression of MuLV, and a weakened or defective anti-LDV antibody response results in replication of neuropathic LDV in the spinal cord.41,46 However, Dr Hoenerhoff noted, it is important to understand that neurologic disease can occur without the presence of retrovirus in severely immunocompromised mice (NOD-SCID, FoxN1, NSGs). Due to severe immune system defects in these models, development of neurologic disease is accelerated, affecting younger animals, occurs without the influence of retrovirus, and occurs without a significant inflammatory response. Therefore, the disease in immunosuppressed animals presents as a degenerative myelopathy and neuropathy, rather than poliomyelitis as seen in immunocompetent strains, such as C58 and AKR. 40
Dr Hoenerhoff then went on to describe the potential impact of this virus in biomedical research. Outside of a transient increase in serum LDH, this virus has been shown to be immunomodulatory in immunocompetent mice. LDV can induce nonspecific polyclonal activation of B cells, immunoglobulin secretion, acute lymphopenia, chronic lymphocytosis, and abnormal interferon and natural killer (NK) cell response.39-43,47,48 Therefore, the presence of this virus in animal studies, particularly those investigating pathophysiology of the immune system, can be a significant confounder in immunology research. In addition, LDV has been shown to interfere with normal disease mechanisms in several other models, including increased susceptibility to lipopolysaccharide (LPS)-induced septic shock, suppression of immune-mediated islet cell injury in type I diabetes in NOD mouse models, and antibody-mediated thrombocytopenia.49-52
Furthermore, LDV infection has been reported to impact xenograft studies. With the extensive use of immunosuppressed mouse models, this is particularly relevant for cancer researchers. Infection with LDV in immunocompromised mice used for tumor transplant studies can result in alteration in the efficiency of xenograftment causing transplant failure, and can alter the growth and progression of xenograft tumors in recipient mice.53,54 Therefore, the presence of this virus in tumor transplant studies can significantly impact cancer studies in biomedical research. Therefore, it is clear that the effects of this virus are far more reaching and significant than merely transient elevation in serum LDH.
Dr Hoenerhoff concluded with several take home points regarding LDV infection. First, that it is critical to recognize clinical disease and pathology associated with LDV, since it can be significant cause of experimental failure due to confounding, loss of animals, data, and irreplaceable biologic samples. Particularly in xenograft studies, in which extensive serial passage of tumors is performed in several series of recipient mice, by the time clinical signs are observed, numerous animals may be affected and the source of the contamination by LDV extremely difficult, if not impossible, to determine. Furthermore, one must make certain not to confuse the paralytic effects of this virus with clinical signs related to cancer morbidity, paraneoplastic syndromes, or effects of tumor metastasis, particularly in models designed to recapitulate bone metastasis. Finally, Dr Hoenerhoff stressed that it is important to have a screening program for this virus in any institution using immunocompromised mice for xenograft studies, to detect contamination of products derived from, or passaged in rodents, to prevent compromise or loss of valuable data, animals, and research resources.
I Have a Bone to Pick: Dendriform Pulmonary Ossification and Fanconi Syndrome in a Dog
Dr Jonathan Nagel (University of North Carolina [UNC], Chapel Hill, NC; North Carolina State University [NCSU], Raleigh, NC) presented a case of dendriform pulmonary ossification and hereditary Fanconi syndrome in a 12-year-old male intact mixed breed dog (Basenji, Lhasa Apso) with homozygous mutations in
One day after a prophylactic dental procedure, the dog developed acute dyspnea, tachycardia, and intermittent collapse followed by lateral recumbency. Thoracic radiographs revealed a multifocal to coalescing alveolar pattern of mineral opacity. On blood serum chemistry evaluation, alkaline phosphatase (ALP) was 307 U/L (reference range: 20-150 U/L). The patient was euthanized three days following the onset of clinical signs due to a lack of improvement with supportive care.
Upon gross postmortem examination, the lungs contained innumerable, multifocal to coalescing, pinpoint to 2 × 5 × 1 cm, nodular to irregularly linear, hard, pale gray masses (Figure 6A). A computed tomography (CT) of a lung lobe revealed irregular multifocal to coalescing spicules of bone density throughout the parenchyma. The audience was shown low-magnification and high-magnification photomicrographs of an H&E-stained section of lung in which alveolar septa were frequently expanded by multifocal to coalescing aggregates of woven bone embedded within abundant mature fibrous stroma (Figure 6B-6E). High-magnification images highlighted bone marrow formation within the bone (Figure 6D). The audience was asked to diagnose the pulmonary lesions, and the voting choices and results were as follows: normal lung (0%), diffuse pulmonary ossification (77%), ossifying fibromas (6%), osteomas (8%), and osteosarcoma (8%). After voting, additional photomicrographs of the H&E-stained section of lung were shown to highlight type II pneumocyte hyperplasia and mild lymphohistiocytic interstitial infiltration in areas of bone formation and fibrosis (Figure 6E).
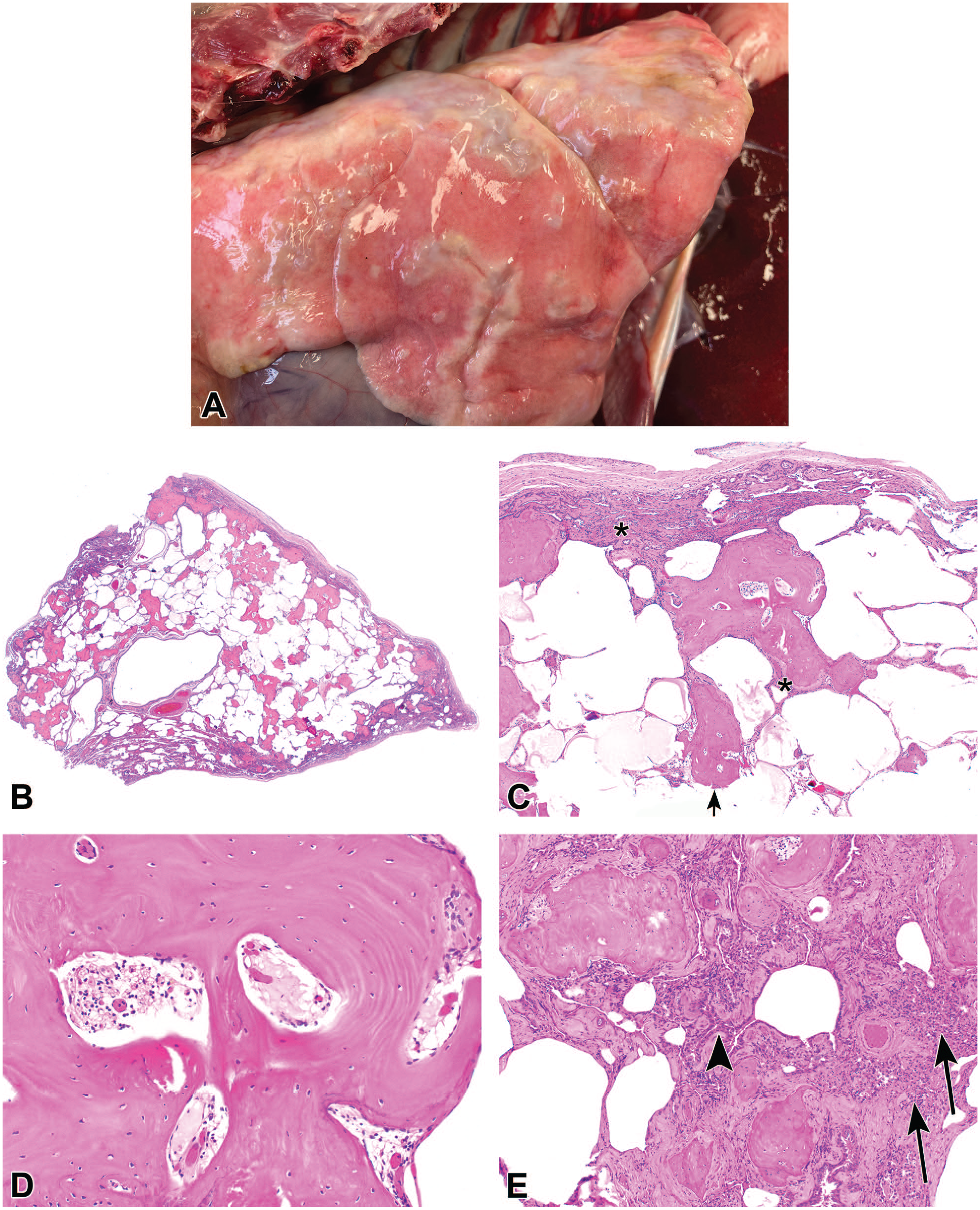
Dendriform pulmonary ossification in a 12-year-old dog (A-E). On gross examination (A), innumerable bony masses were scattered throughout the parenchyma of all lung lobes. Histologic sections of lung (B-E) are stained with H&E. At low magnification (B) there is multifocal to coalescing expansion of the pulmonary parenchyma by irregular spicules of woven bone. At higher magnification (C), the spicules are generally contained within alveolar septa with occasional extension into alveolar spaces through ruptured septa (arrow). Alveolar septa are further expanded by mature fibrous stroma (asterisk). At high magnification (D), there are occasional small bone marrow cavities within the spicules of bone containing adipose tissue admixed with erythrocyte and myeloid precursor cells. (E) Few, patchy, loose aggregates of macrophages, lymphocytes, and plasma cells (arrow) are scattered throughout areas of alveolar fibrosis and ossification. Alveoli within affected areas are often lined by cuboidal cells (arrowhead), which is consistent with type II pneumocyte hyperplasia. Renal cortex from a 12-year-old dog with homozygous
(F), the interstitium is expanded by patchy, mild fibrosis (asterisk), and tubules are often slightly dilated and lined by attenuated epithelium (arrowhead). High magnification (G-H) highlights tubular epithelial attenuation (black arrowhead) and interstitial fibrosis (asterisk). (G) Karyomegalic tubular epithelial cells (arrow) are scattered throughout the cortex. (H) Tubular epithelial cells occasionally contain acidophilic brick-shaped intranuclear inclusion bodies (yellow arrowhead). Scattered within the fibrotic interstitium (asterisk) are few lymphocytes and plasma cells (blue arrowhead). H&E indicates hematoxylin and eosin.
These findings were consistent with diffuse pulmonary ossification (DPO), a rare entity described in humans and other animals, including dogs. DPO is typically diagnosed as an incidental finding on postmortem examination, although pneumothorax may occur due to pleural rupture.55-65 Two histologic subtypes of DPO are described in humans: nodular and dendriform.
Patients with nodular pulmonary ossification have aggregates of metaplastic bone within alveolar spaces. Typically, the metaplastic bone in cases of nodular pulmonary ossification is devoid of bone marrow. These cases are often associated with mitral valve stenosis and other causes of chronic pulmonary congestion.57,58 Patients with dendriform pulmonary ossification have spicules of metaplastic bone within alveolar septa which often contain bone marrow.55-58,65 Histologic features of the presented case were most consistent with dendriform pulmonary ossification as opposed to nodular pulmonary ossification. Dendriform pulmonary ossification can be idiopathic or associated with several conditions, including usual interstitial pneumonia, chronic obstructive pulmonary disease, pneumoconiosis, chronic gastroesophageal reflux disease, end-stage renal disease, and genetic mutations.55-60,63,65 DPO has been reported in coyotes with pneumoconiosis, a dog with multiple mammary tumors, and a dog treated with exogenous cortisol.61,62,64 An overt cause of pulmonary ossification was not identified in this case. In this case, the mild inflammatory infiltrates and interstitial fibrosis may be a component of the primary etiology (e.g., idiopathic pulmonary fibrosis) and/or the widespread osseous metaplasia.
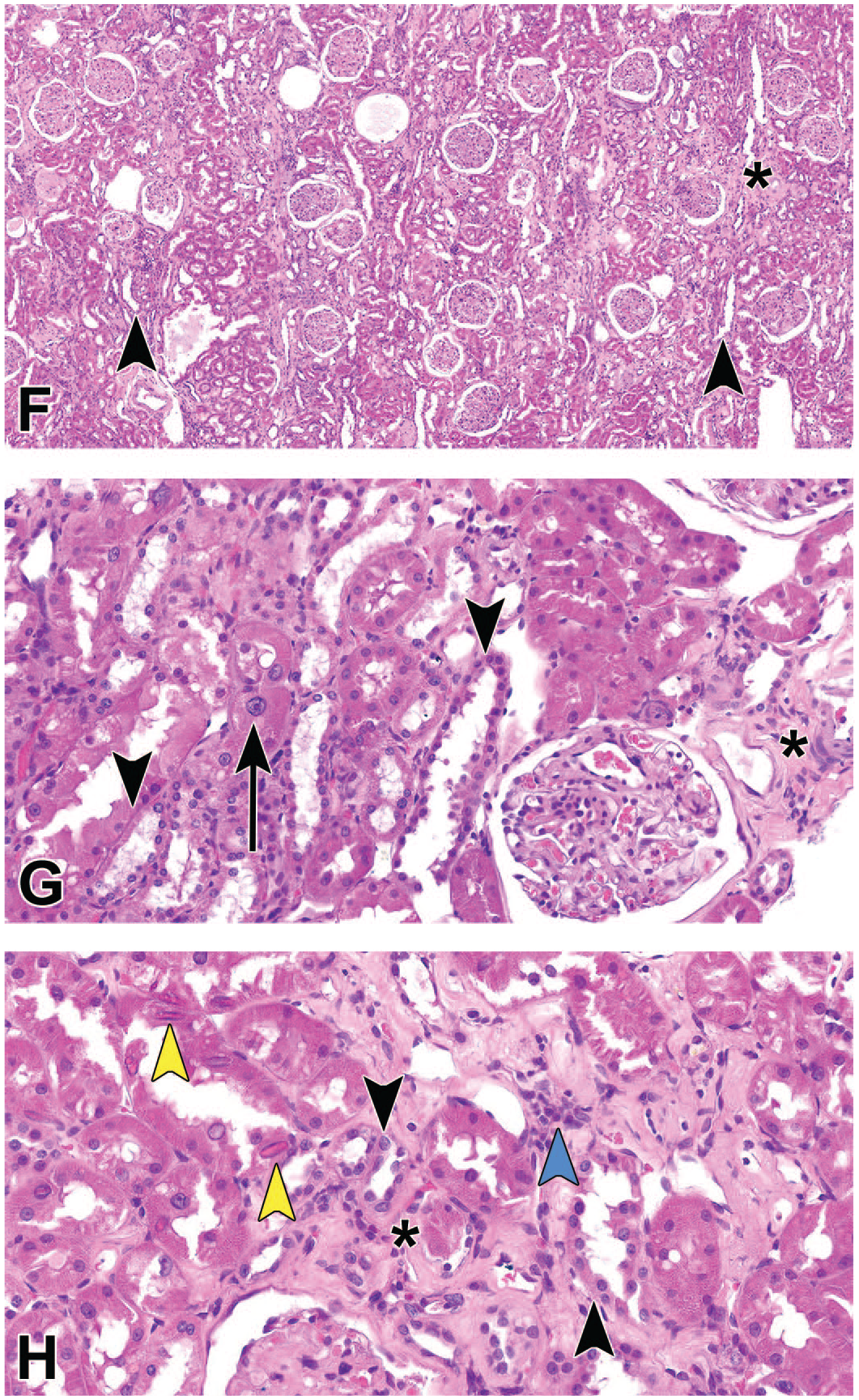
The audience was then shown low-magnification and high-magnification photomicrographs of an H&E-stained section of renal cortex from the kidney of the same dog (Figure 6F-6H). Multifocally, cortical tubular epithelium exhibited attenuation and karyomegaly and occasionally contained acidophilic brick-shaped intranuclear inclusion bodies. The cortical interstitium was expanded by patchy fibrosis and a few scattered lymphocytes and plasma cells. The audience was asked to determine the general cause of the renal lesions, and the answer choices and results were as follows: virus (3%), toxicity (15%), genetic (40%), and h(43%). Dr Nagel felt that these lesions were largely due to mutation of
Deletion of the last exon of the
Dr Nagel concluded his presentation by emphasizing the rarity of DPO and reiterating the general features of DPO and Fanconi syndrome. One audience member asked Dr Nagel if mutations in
The Devil Is in the (Vessel) Details: Pulmonary Vascular Lesions in a Murine Asthma Model
Dr Jeffrey Everitt (Department of Pathology, Duke University School of Medicine, Durham, North Carolina) presented an interesting case of pulmonary vascular lesions that arose in a mouse model of house dust mite sensitization used to model human asthma. Dr Everitt acknowledged Drs Jennifer Ingram and Mark Ihrie of Duke Pulmonary Medicine for conducting the studies and for scientific contributions to the case,
Pulmonary vascular lesions were found in the lungs of a 12-week-old male matrix metalloproteinase-19 knockout mouse (MMP-19−/−) on a C57Bl6/NCrl background used in a murine asthma model. No macroscopic lesions were noted at time of necropsy other than bilateral red-gray discoloration of the lungs. The mouse was part of a study in which animals were sensitized with 2.5 µg of house dust mite extract or saline administered intranasally three times per week for six consecutive weeks. Lesions consisted of greatly thickened and muscularized small intra-acinar arteries throughout the lungs (Figure 7A and 7C). The pulmonary vascular lesions were noted within a lung parenchymal background of severe eosinophilic pneumonitis. Eosinophilic infiltrate was marked in regions of severe perivascular lymphoid proliferation (Figure 7F and 7G). Increased numbers of intra-alveolar macrophages, lymphocytes, neutrophils, and eosinophils were present within affected lung parenchyma, along with thickened alveolar septal walls containing infiltrating inflammatory cells characteristic of hypersensitivity pneumonitis. The pulmonary vascular changes were a striking feature of the lung lesions and were unexpected as they are not part of the normal MMP-19 knockout phenotype and have not been commonly reported in murine asthma models.
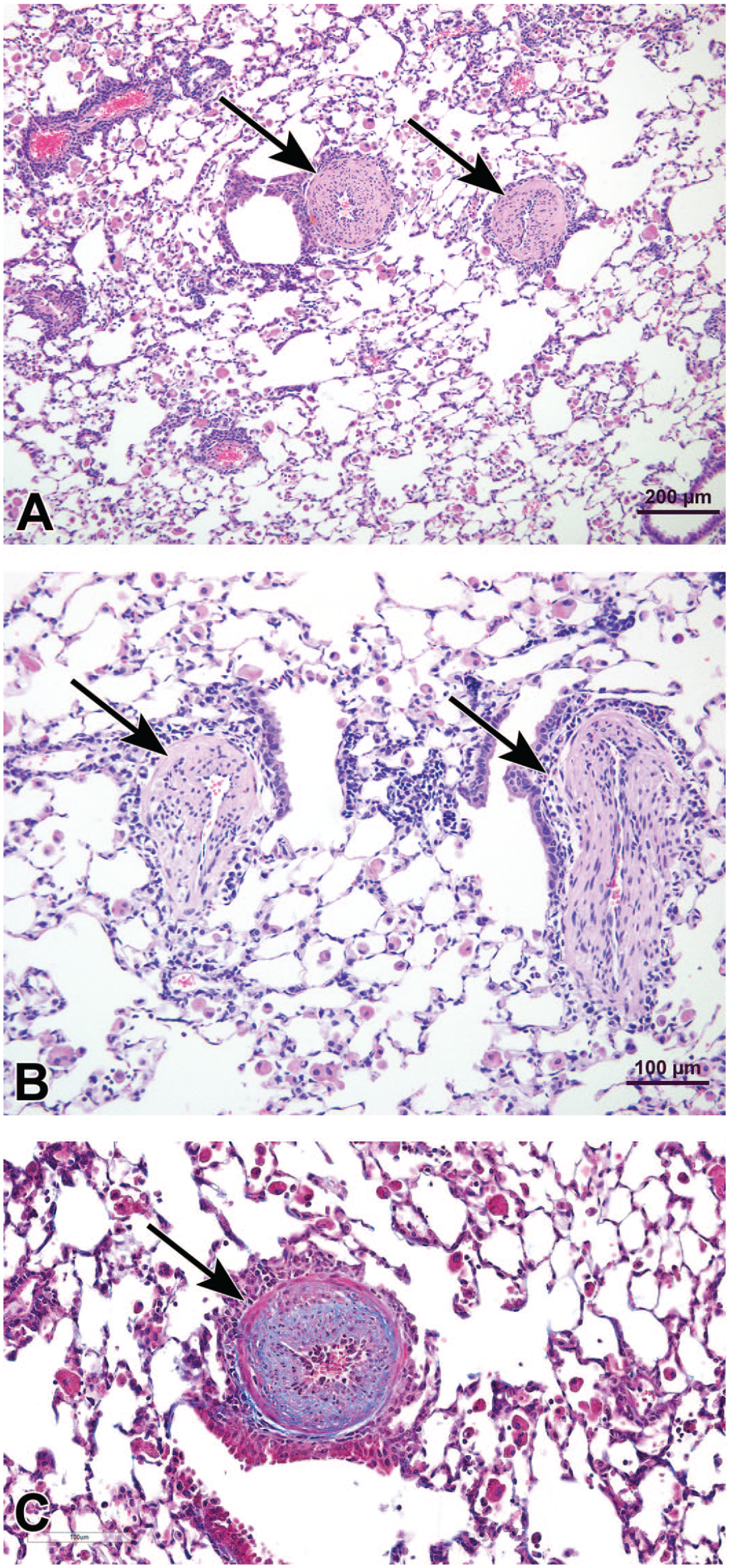
Medial hypertrophy of pulmonary arteries in mice used in murine asthma model. (A) Pulmonary arteries (arrows) in a MMP-19−/− mouse treated with house dust mite extract. H&E (B) Photomicrograph of pulmonary arteries (arrows) in a wild-type mouse similarly treated with house dust mite extract. H&E (C) Masson’s trichrome-stained section from case depicted in (A) showing increased collagen and extracellular matrix in the thickened wall of the vessel. Photomicrographs of lungs from MMP-19−/− mice treated with saline
(D) or house dust mite extract (E) for six weeks. Note markedly thickened small pulmonary artery (arrow) at TB alveolar duct junction region in animal with inflamed lung in (E) relative to the control in (D). H&E. Photomicrographs of lungs from MMP-19−/− mice treated with house dust mite extract for six weeks demonstrating numerous eosinophils in the inflammatory infiltrate. (F) Immunostain for eosinophil major basic protein showing numerous eosinophils (arrowheads) within thick perivascular cuffs of lymphoid cells as well as scattered throughout the pulmonary parenchyma. (G) Thickened pulmonary artery circumscribed by numerous eosinophils. H&E. H&E indicates hematoxylin and eosin; MMP-19, matrix metalloproteinase-19; TB, terminal bronchiole.
The audience was presented with a variety of voting options to characterize the vascular changes that were depicted. Most attendees (51%) agreed with the presenter that the best diagnosis was medial hypertrophy of pulmonary arteries. Smaller numbers of attendees thought that other choices were also appropriate, including pulmonary arteriosclerosis (15%), eosinophilic pulmonary arteritis (12%), and pulmonary hypersensitivity vasculitis (12%). Dr Everitt reviewed the published INHAND diagnostic criteria 1 for the diagnosis of medial hypertrophy of pulmonary arteries (Table 3) and reviewed why this was chosen as the most appropriate diagnosis, including type of thickening, vessels involved, maintenance of internal elastic lamina, and presence of extracellular matrix and connective tissue on trichrome-stained sections (Figure 7C).
Dr Everitt then discussed the fact that in laboratory animal species commonly used in preclinical studies there is great variation in the degree of muscular thickness in the pulmonary vasculature. 81 The audience was asked to vote on a series of different laboratory animals to state which research animal species has medial hypertrophy of pulmonary vasculature as a normal feature of lung architecture. Seventy-six percent of the attendees correctly identified the guinea pig as having particularly prominent muscularization of the pulmonary arteries. Twenty percent answered the rabbit and Dr Everitt reviewed the neointimal proliferation lesions reported in the pulmonary vessels of aged rabbits, and compared and contrasted the findings to the present case. 82
The finding of lesions of medial hypertrophy of pulmonary arteries in an MMP-19−/− mouse was unexpected as the lesion is not part of the normal phenotype of this genetically modified animal, however, matrix metalloproteinases are heavily involved in vascular remodeling processes and vascular disease through a variety of mechanisms. 83 Dr Everitt discussed the relationship of this lesion to pulmonary hypertension and reviewed studies to date in this house dust mite sensitization asthma model. He reported that wild-type animals were also found to have similar vascular lesions (Figure 7B), although less prevalent and less severe than those of the MMP-19−/−genotype. In all instances studied to date with this model, the vascular lesions are dependent on the occurrence of pulmonary eosinophilic inflammation. No saline-treated controls developed lesions and the severity of lesions correlated with the severity of the eosinophilic infiltrates.
In the discussion that ensued with the audience, several pathologists discussed their experience with the pulmonary vascular lesions acknowledging the presence of this vascular change in preclinical murine asthma models, although none had seen it with such severe manifestation. Dr Everitt discussed the significance of pulmonary hypertension in the genesis of the lesion while acknowledging that cardiac studies were underway in the MMP-19−/− model but not yet evaluated. Dr Everitt raised the point that more studies are needed to understand the role of eosinophilic pulmonary inflammation on the development of vascular disease in the lung, as there are a number of pulmonary entities in veterinary medicine characterized by eosinophils and smooth muscle changes. One such example is equine hypersensitivity pneumonitis where extensive pulmonary vasculature changes have been noted. 84
Vascular remodeling has been reported in mice in a variety of pulmonary inflammatory models, including allergic hypersensitivity models. Inhalation sensitization of mice with
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported [in part] by the NIH, National Institute of Environmental Health Sciences.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

