Abstract
Acute kidney injury (AKI) is encountered in high-risk hospital settings where patients often suffer systemic illnesses, undergo surgery, or receive medications toxic to the kidneys. It is associated with high morbidity and mortality. Despite advances in understanding the pathophysiologic mechanisms involved in AKI, our ability to detect AKI early is limited, and outcomes remain unchanged. Once AKI is identified by the traditional markers of urine output and serum creatinine, the main therapeutic intervention is usually supportive care and/or renal replacement therapy until renal injury resolves. Urine biomarkers provide an optimistic future, offering the ability to identify patients at high risk for AKI such that preventative measures can be introduced. In this article, the etiologies of AKI are discussed, new biomarkers are assessed, and areas for further investigation are suggested.
Acute kidney injury (AKI) is defined as a rapid decline in glomerular filtration rate (GFR) that occurs over hours to weeks and results in an acute increase in serum creatinine (SCr) or blood urea nitrogen levels and/or a decline in urine output (Figure 1; Sreedharan, Devajaran, and van Why 2009). The Kidney Disease Improving Global Outcomes (KDIGO) network separates AKI into three stages of increasing severity (Table 1). These clinical changes in kidney function are often not evident until well after the kidney has actually been injured. SCr is affected by an individual’s muscle mass, protein metabolism, tubular creatinine secretion, and reserve kidney capacity, so correlation with GFR is nonlinear (Kellumand and Devarajan 2014). The kidneys may sustain significant damage without a detectable change in SCr (Thomas et al. 2015).
KDIGO AKI Stages.
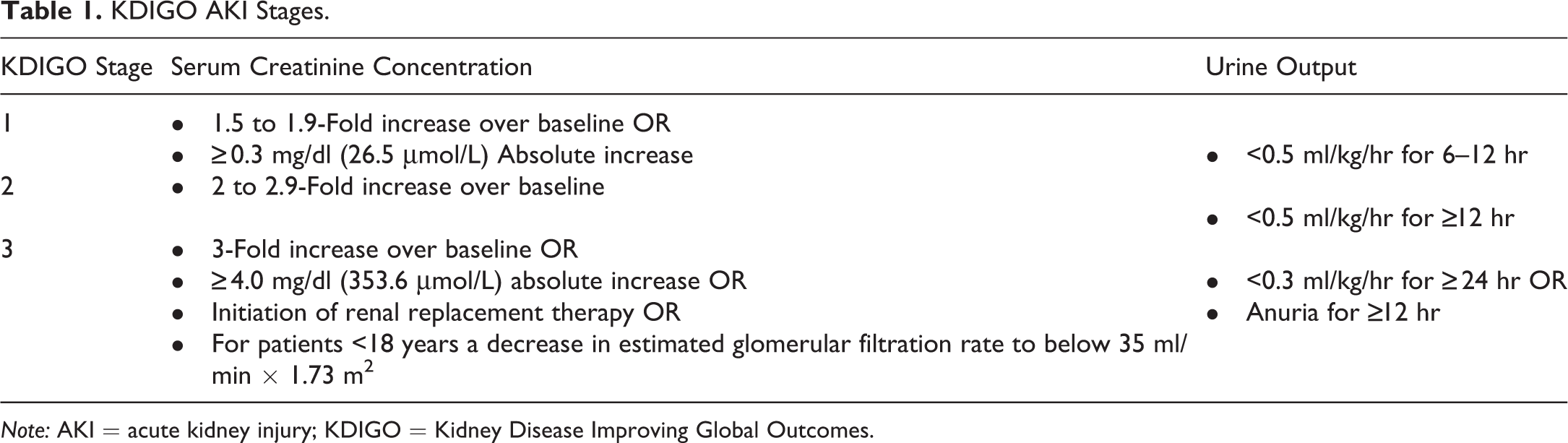
Note: AKI = acute kidney injury; KDIGO = Kidney Disease Improving Global Outcomes.

The definition of acute kidney injury from the Kidney Disease Improving Global Outcomes network.
AKI is a significant clinical problem that is becoming increasingly common, especially in hospitalized patients (Bonventre and Yang 2011). A recent systematic review estimated the rate of AKI in adults to be as high as 21.6%, most prominently in critical care and cardiothoracic surgery settings (Susantitaphong et al. 2013). Long-term kidney survival decreases and mortality increases with each KDIGO AKI severity stage (Khwaja 2012). In studies using the KDIGO-based AKI definitions, pooled AKI mortality rate increased with each stage of severity whether in-hospital, at 90 days or at 6 months, and were highest in those requiring renal replacement therapy (Ali et al. 2007).
AKI is classified into three categories that reflect its pathogenesis. These are prerenal AKI, intrinsic AKI, or postrenal/obstructive AKI. Prerenal AKI is caused by a decrease in effective arterial renal perfusion and is seen in conditions of intravascular volume loss, poor cardiac function, and an imbalance between venous and arterial filling. In cases of fluid loss, prerenal AKI is quickly reversible with volume resuscitation, but in cases of poor cardiac (left ventricular) function with excess total body fluid, treatment needs to improve cardiac output, which may be difficult to achieve. Intrinsic AKI has a wide range of etiologies, including glomerular diseases, interstitial diseases, nephrotoxin exposure, vascular diseases, and ischemic tubular damage, commonly called acute tubular necrosis (ATN). In intrinsic AKI, therapy is targeted to the specific cause of injury, such as immunosuppression for glomerulonephritis or supportive care for ischemic or nephrotoxic ATN. Finally, postrenal AKI is most often related to diseases of the urinary tract that cause obstruction to urine flow at the level of the ureters or urethra (Johnson et al. 2015). Most often postrenal AKI is confirmed by finding hydronephrosis on renal imaging, and resolves with relief of the obstruction. The distribution of AKI by pathophysiologic categories is shown in Figure 2 and examples and approaches to management for these AKI phenotypes are listed in Table 2 (Praga and Gonzalez 2010).
Classification and Management of AKI.
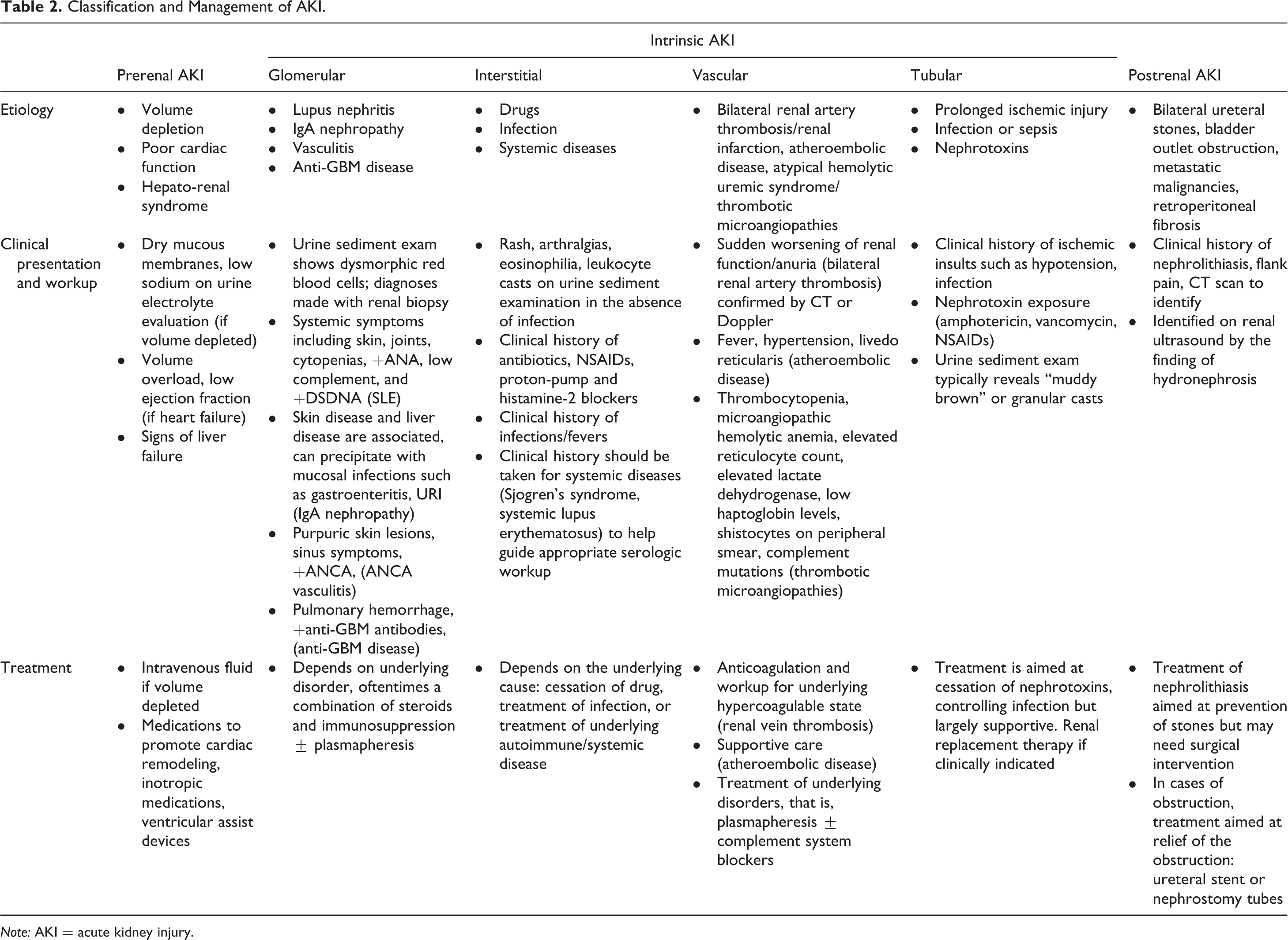
Note: AKI = acute kidney injury.
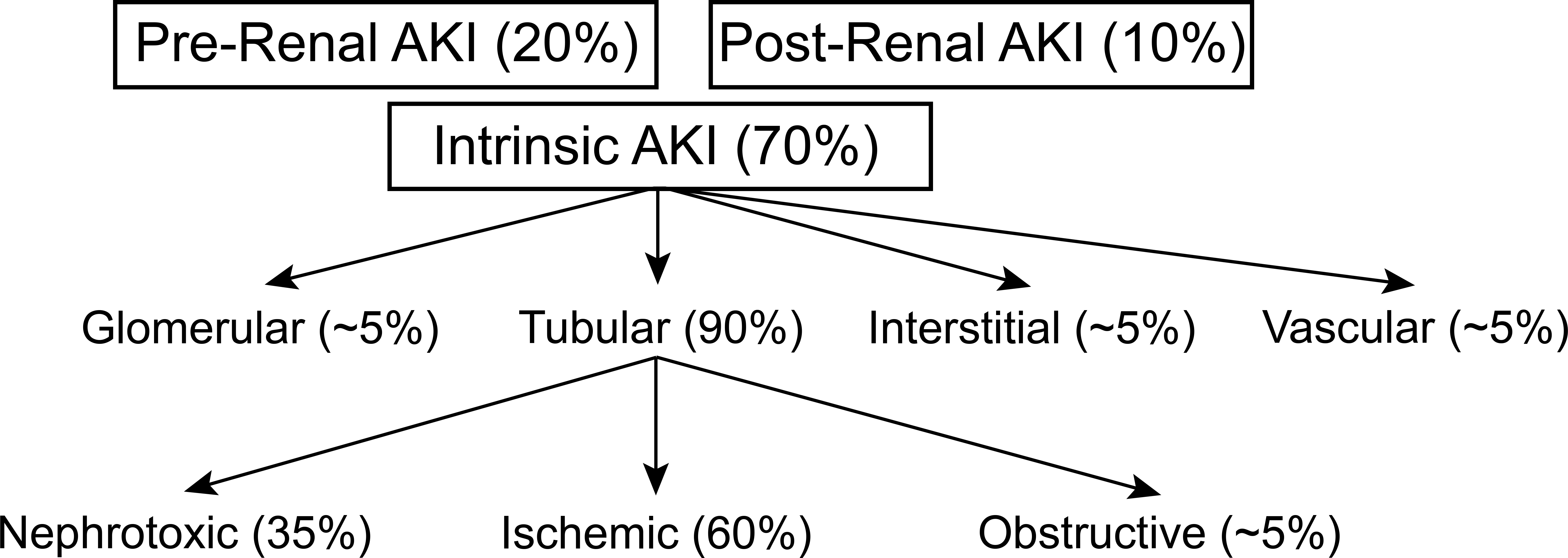
Distribution of acute kidney injury (AKI) by cause. Most AKI is intrinsic and has several etiologies and is followed in frequency by prerenal AKI and finally postrenal AKI.
The importance of AKI due to ATN cannot be understated. It is the most common type of AKI, occurs mainly in hospitalized patients, is an independent risk factor for short- and long-term morbidity and mortality, prolongs hospitalizations, and is costly to the health-care economy (Devarajan 2016). In contrast to many other types of AKI, and despite a considerable and long-standing research effort, there are currently no specific treatments for established ATN beyond supportive care and expectant waiting for the tubules to recover. This often includes initiation of acute forms of dialysis (Yap and Lee 2012). The optimal approach to management of ATN is prevention, which includes recognizing at-risk patients and the clinical situations that predispose patients to AKI in the hospital (Bellomo, Kellum, and Ronco 2012). However, given the increasing complexity and age of at-risk patients and the toxicities/complications associated with current therapies, AKI cannot be prevented in all, or possibly most cases. Therefore, early detection of AKI is critical to rapidly initiate kidney supportive measures, eliminate nephrotoxin exposure, and restore renal perfusion. These factors led to the development of the first Food and Drug Administration–approved biomarker for early detection of AKI.
AKI biomarkers were identified in a discovery study that utilized three independent cohorts of adult patients who had been admitted to an intensive care unit and had at least one risk factor for AKI (Kashani et al. 2013). The AKI risk factors included sepsis, hypotension, hemorrhage, radiocontrast exposure, major surgery, intravenous (IV) antibiotics, acute decompensated heart failure, or severe trauma. Over three hundred candidate biomarkers were evaluated, and urine tissue inhibitor of metalloproteinase-2 (TIMP-2) and insulin-like growth factor binding protein-7 (IGFBP-7) demonstrated a significant risk for developing KDIGO stage 2 or 3 AKI within 12 hours of sample collection. The combination of TIMP-2 and IGFBP-7 was better than either analyte alone, and measured together provided added value to the established clinical markers, such as SCr. TIMP-2/IGFBP-7 was validated in the Sapphire cohort, which was a population of critically ill adults (n = 728) admitted to the intensive care unit with either respiratory or cardiovascular failure (Kashani et al. 2013). IGFBP7 and TIMP-2 are thought to be relevant to the pathophysiology of AKI by protecting injured tubular cells during AKI through cell cycle arrest in the G1 phase (Kellum and Chawla 2016). This allows the cell to focus on repairing damage and its DNA. After repair, tubular cells can re-enter the cell cycle, but with an increased chance of survival (Price, Safirstein, and Megyesi, 2009).
The TIMP-2/IGFBP7 biomarker can now be measured in clinical laboratories using the NephroCheck™ system. This system has good sensitivity (76–92%) and modest specificity (46–51%) for predicting the risk of developing moderate to severe AKI (KDIGO Stage 2 or 3) within 12 hours. NephroCheck has a positive predictive value of 27% to 31%, but a negative predictive value of 88% to 96%, suggesting patients with a negative test are not likely to develop AKI (Ronco et al. 2017).
Despite the promising nature of urinary biomarkers, barriers to their use in practice still exist. AKI involves a variety of mechanisms and identification of biomarkers does not delineate cause or site of injury. Thus, biomarkers may not advance the therapies available, as they are nonspecific to cause of AKI, but may assist with risk stratification (Pickkers et al. 2017). Furthermore, the use of NephroCheck to detect biomarkers has a strong negative predictive value but poor positive predictive value, making a positive result difficult to interpret (Murray et al. 2014).
In practice, TIMP-2/IGFBP7 should help identify patients with tubular stress as opposed to tubular cell death, providing the opportunity for early intervention to prevent the development of AKI. This was tested in a trial of patients who underwent on-pump cardiac surgery (Meersch et al. 2017). Four hours after the bypass pump was disconnected, TIMP-2/IGFBP7 was measured and patients were stratified into a low and high risk for AKI. High-risk patients were then randomized to a control group or an intervention group. The control group received standard care consisting of maintaining mean arterial pressure >65 mm Hg and central venous pressure between 8 and 10 mm Hg. The intervention group received the “KDIGO cardiothoracic surgery bundle” which includes avoiding nephrotoxins, stopping renin-angiotensin-aldosterone system blockers for the first 48 hours after surgery, close monitoring of SCr and urine output, avoiding IV iodinated contrast, avoiding hyperglycemia in the 72 hours following surgery, and hemodynamic monitoring via a pulse contour cardiac output catheter (Meersch et al. 2017). The primary end point was AKI within the first 72 hours after cardiac surgery. The incidence of AKI and moderate to severe AKI was significantly reduced by about 15% to 16% in the intervention group (Meersch et al. 2017). This indicates that application of supportive care measures in high-risk patients, as identified by the urinary biomarkers, may reduce risk for developing AKI.
Ideally, the ability to identify patients at high risk for AKI early could lead to a more robust decline in AKI if there were effective therapeutics to add to intensive supportive care. However, despite intense investigation, such therapeutics have been elusive. For example, a recent trial failed to show that an α-melanocyte-stimulating hormone (αMSH) analog protected against AKI in patients undergoing on-pump cardiac surgery (McCullough et al. 2016). This study was based on preclinical evidence that αMSH was renoprotective, possibly due to the stimulation of melanocortin receptors in the renal medulla and collecting ducts. Additionally, αMSH can inhibit inflammatory, cytotoxic, and apoptotic pathways, which can play an additional role in the pathogenesis of AKI (McCullough et al. 2016).
In summary, AKI is a heterogeneous clinical syndrome that will most likely require a variety of therapies to target its myriad of etiologies. The development of biomarkers that allow AKI risk stratification and identify at-risk patients before significant kidney damage or cell death has accrued should facilitate the development and clinical trials of treatments that can prevent the occurrence of moderate to severe AKI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
