Abstract
Congenital uterine wall cysts arising from paramesonephric (Müllerian) and mesonephric (Wolffian) ducts are typically incidental findings in most species. We used immunohistochemistry to characterize and determine the origin of uterine cysts in Sprague-Dawley (SD) rats from multigeneration studies conducted by the National Toxicology Program. Subserosal uterine cysts were observed in 20 of the 2,400 SD rats evaluated in five studies, and 10 cysts were characterized for this study. Single cysts were unilocular, fluid-filled, and occurred throughout the uterus. Microscopically, all cysts had a well-developed smooth muscle wall, lined by flattened to cuboidal, sometimes ciliated, epithelium that stained intensely positive for cytokeratin 18 and paired box protein 8 (PAX8). Most cyst epithelia displayed weak to moderate positivity for progesterone receptor (PR) and/or estrogen receptor α (ER-α), as well as were negative for GATA binding protein 3 (GATA3). Cyst lumens contained basophilic flocculent material. The cysts appeared to be developmental anomalies arising from paramesonephric tissue based on positive PAX8 and ER-α and/or PR staining. Additionally, 70% of the cysts lacked GATA3 expression. Taken together, the subserosal uterine cysts observed in adult rats in these studies most likely arose from the paramesonephric duct.
Various uterine cysts have been reported in humans as well as in most domesticated and laboratory animal species. Cysts are commonly associated with cystic degeneration of leiomyomas, adenomyosis, serosal inclusion cysts, and cystic endometrial hyperplasia (Matalliotakis, Kourtis, and Panidis 2003; Mutter et al. 2007; Protopapas et al. 2008). In the rat, uterine cysts are predominately of endometrial epithelial origin and are a common age-related change. Development of spontaneous cystic endometrial hyperplasia has occurred in rats, exhibiting persistent estrus, hormonal imbalances, and excess estrogen secretion (Greaves 2012; Dixon et al. 2014).
Although uncommon, congenital uterine cysts may arise from embryonic (Wolffian or Müllerian) ducts (Sherrick and Vega 1962). The ovaries, uterus, and upper vagina develop from the paramesonephric (Müllerian) duct (Goldman and Cooper 2013). The male reproductive tract originates from the mesonephric (Wolffian) duct that lies parallel to the paramesonephric duct during embryonal development. Traditionally, it has been proposed that it regresses in females in the absence of the production of anti-Müllerian hormone by Sertoli cells and testosterone by interstitial cells of the embryonic testes (Kobayashi and Behringer 2003; Jacob, Yusuf, Jacob 2012; Dixon et al. 2018). However, recently, it has been suggested that elimination of the Wolffian duct in female mouse embryos is androgen-independent; and regression is actively promoted by the transcription factor, chicken ovalbumin upstream promoter transcription factor II (Zhao et al. 2017). In women, congenital cysts of the uterus characteristically have variably thick fibromuscular walls lined by ciliated to nonciliated cuboidal to columnar epithelium; lack association with the uterine cavity, endometrial glands, and stroma; and are located within the uterine myometrium (Sherrick and Vega 1962).
Distinguishing between benign and neoplastic tissue derived from paramesonephric and mesonephric ducts is difficult, and recent research has focused on identifying immunohistochemical markers specific to each duct. Paired box 8 (PAX8) is a transcription factor involved in the organogenesis of paramesonephric-derived organs (Mittag et al. 2007). GATA binding protein 3 (GATA3) transcription factor is involved in the organ differentiation of the urogenital tract originating from the mesonephric duct (Grote et al. 2006). PAX8 and GATA3 have been identified as immunomarkers to differentiate paramesonephric and mesonephric neoplasms, respectively, in humans (Tong et al. 2011; Howitt et al. 2015). Based on the histological appearance of the uterine wall cysts identified in Sprague-Dawley (SD; Hsd: Sprague Dawley SD) rats in our study, we hypothesized that these cysts are remnants of either the paramesonephric or the mesonephric ducts. We used routine light microscopy and histochemical and immunohistochemical staining to identify the origin of these congenital cysts.
Materials and Method
Tissue Collection and Histological Stains
Subserosal uterine cysts were observed grossly and/or microscopically in 20 of the 2,400 SD rats evaluated in 5 multigeneration studies conducted by the National Toxicology Program (NTP). All animals were handled, cared for, and used in accordance with the NRC Guide for the Care and Use of Laboratory Animals (U.S. FDA 2018; NRC 2011). These studies were conducted in American Association of Laboratory Animal Care (AALAC)-approved and Public Health Service (PHS)-assured facilities. Uterine tissue from routine necropsies was collected according to the study protocol and fixed in 10% neutral-buffered formalin in accordance with U.S. FDA Good Laboratory Practice (GLP) Regulations (U.S. FDA 2018).
The female reproductive organs were trimmed according to the specifications for the conduct of studies to evaluate reproductive and developmental toxicity of chemical, biological, and physical agents in laboratory animals for the National Toxicology Program (2011). Briefly, the cervix and vagina were freed en masse from the uterine body to achieve sagittal sections for evaluation. Cross sections of the midpoint of both uterine horns and longitudinal sections of remaining uterine tissue were taken for assessment. Fixed tissues were embedded in paraffin, sectioned at 5 µ, and stained with hematoxylin and eosin. Ten of the 20 animals diagnosed with cysts had adequate cystic tissue available for further evaluation with Masson’s trichrome and immunohistochemical staining. Uterine, cervical, and vaginal tissue sections were examined histologically to determine phase of estrous cycle as outlined in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) female reproductive tract document (Dixon et al. 2014).
Immunohistochemical Stains
A total of 9 to 10 uterine cysts were immunohistochemically stained for α smooth muscle actin (SMA, smooth muscle marker), cytokeratin 18 (CK-18, epithelial cell marker), estrogen receptor α (ER-α, steroid receptor marker), progesterone receptor (PR, steroid receptor marker), PAX8 (paramesonephric tissue differentiation marker), and GATA3 (mesonephric tissue differentiation marker) as outlined in Table 1. Briefly, formalin-fixed, paraffin-embedded tissue sections were deparaffinized in xylene and rehydrated through graded alcohols. Endogenous peroxidase activity was blocked using 3% hydrogen peroxide before or following heat-induced antigen retrieval using a Decloaker® pressure chamber (Biocare Medical, Concord, CA). Nonspecific sites were blocked using normal horse, donkey, or goat serum, respective to the secondary antibody used, (Jackson Immunoresearch Laboratories, Inc., West Grove, PA) prior to or followed by incubation with an avidin/biotin blocking kit (Vector, Burlingame, CA). All tissues were incubated with the primary antibodies listed in Table 1 for 1 hr at room temperature, followed by incubation with the appropriate secondary antibody for 30 min at room temperature. Negative controls were incubated with normal rabbit IgG (Abcam, Cambridge, MA) or normal mouse IgG1 (BD Biosciences, San Jose, CA) diluted to the same concentration as the primary antibody. Antigen–antibody complexes were labeled using a Streptavidin SS Label (Biogenex Laboratories, San Ramon, CA) or avidin–biotin affinity system (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA) and visualized with 3, 3’-diaminobenzidine chromogen (DakoCytomation, Carpenteria, CA). Tissue sections were counterstained with hematoxylin, dehydrated through graded ethanol, cleared in xylene, and cover-slipped. Positive controls included rat kidney for PAX8, rat mammary tissue for GATA3, and the uterus, which also served as an internal control for CK18, ER-α, PR, and SMA.
Immunohistochemical Stains.
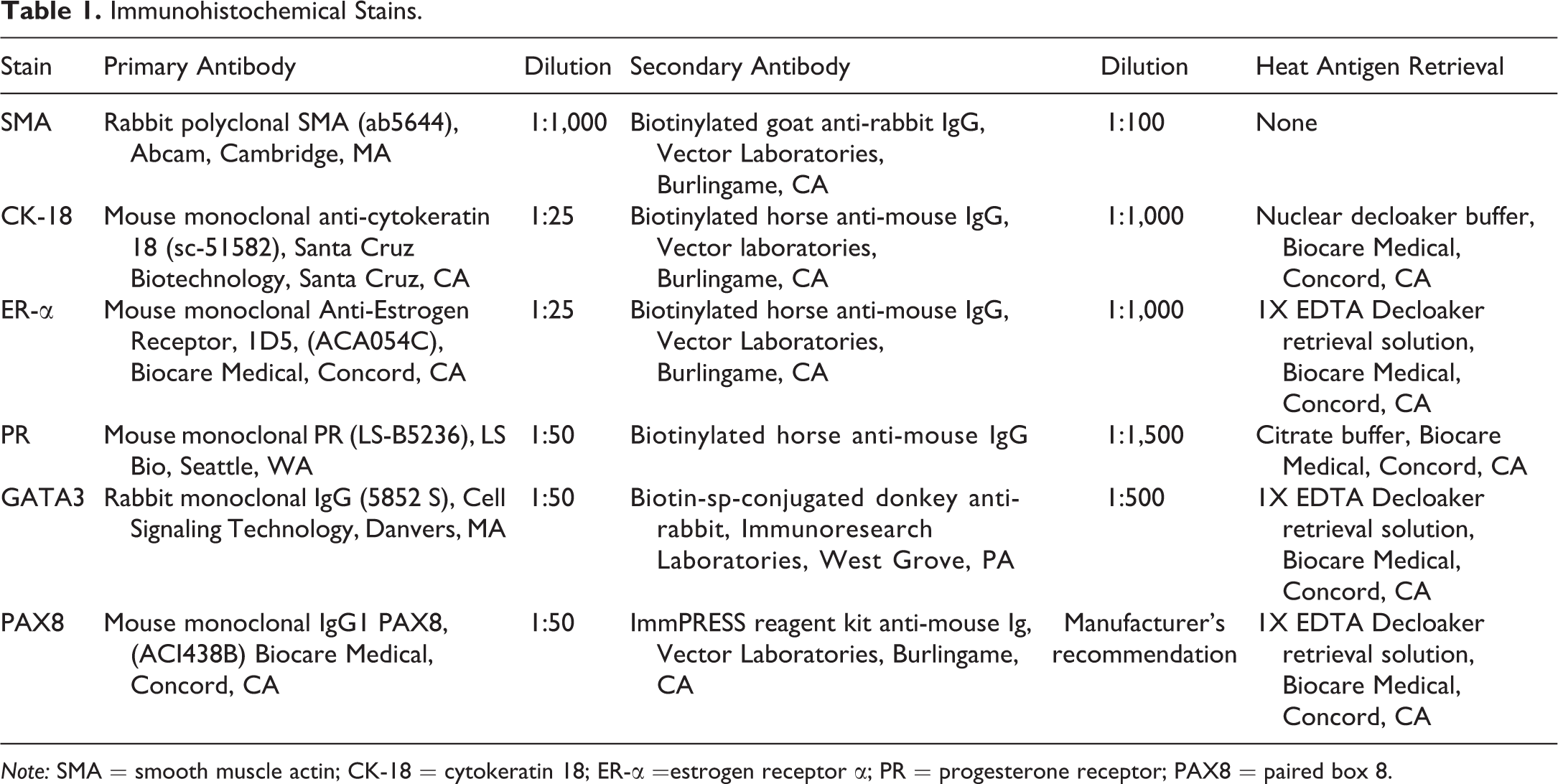
Note: SMA = smooth muscle actin; CK-18 = cytokeratin 18; ER-α =estrogen receptor α; PR = progesterone receptor; PAX8 = paired box 8.
Microscopic Evaluation and Scoring
All uterine tissues were evaluated by routine light microscopy. Immunohistochemical stains were assessed, and a semiquantitative scoring method, which combines the overall percentage of immunopositive cells and average staining intensity, was used to determine a multiplicative quickscore for the wall or epithelium of each cyst at an original magnification of 20X (Detre, Saclani Jotti, and Dowsett 1995; Dixon, He, and Haseman 2000). Briefly, a numerical value was generated for the percentage of the cyst showing positive immunostaining (0–4% = 1, 5–19% = 2, 20–39% = 3, 40–59% = 4, 60–79% = 5, and 80–100% = 6). This number was multiplied by a numerical value assigned to the intensity of immunostaining in each cyst (negative = 0, weak = 1, moderate = 2, or intense = 3) to obtain a quickscore number ranging from 0 to 18. The quickscore values were used to assign each cyst to an immunostaining category of negative immunostaining = 0 to 1 (−), weak = 2 to 6 (+), moderate = 7 to 12 (++), and intense = 13 to 18 (+++).
Results
Animal Information
Uterine subserosal cysts were examined in 20 adult female SD rats (n = 13 rats at 91–93 days of age; n = 7 rats at 266 days of age) from five multigeneration studies conducted by the NTP. Similar appearing subserosal cysts occurred in treated and control rats and were independent of mating status (Table 2).
Animal Information.
Gross Appearance and Location of Uterine Cysts
Grossly identified single cysts were located along the uterine horn or body and were approximately 2 to 3 mm in diameter, spherical, and appeared fluid-filled (Figure 1A; Table 3). Fifteen of the 20 cysts were observed grossly, while 5 cysts were only identified microscopically. All cysts were unilocular and adjacent to the serosa and the outer myometrial layer of the uterus (Figure 1B). The uterus, cervix, and vagina from ten of the rats with adequate cystic tissue available were further examined for this study.
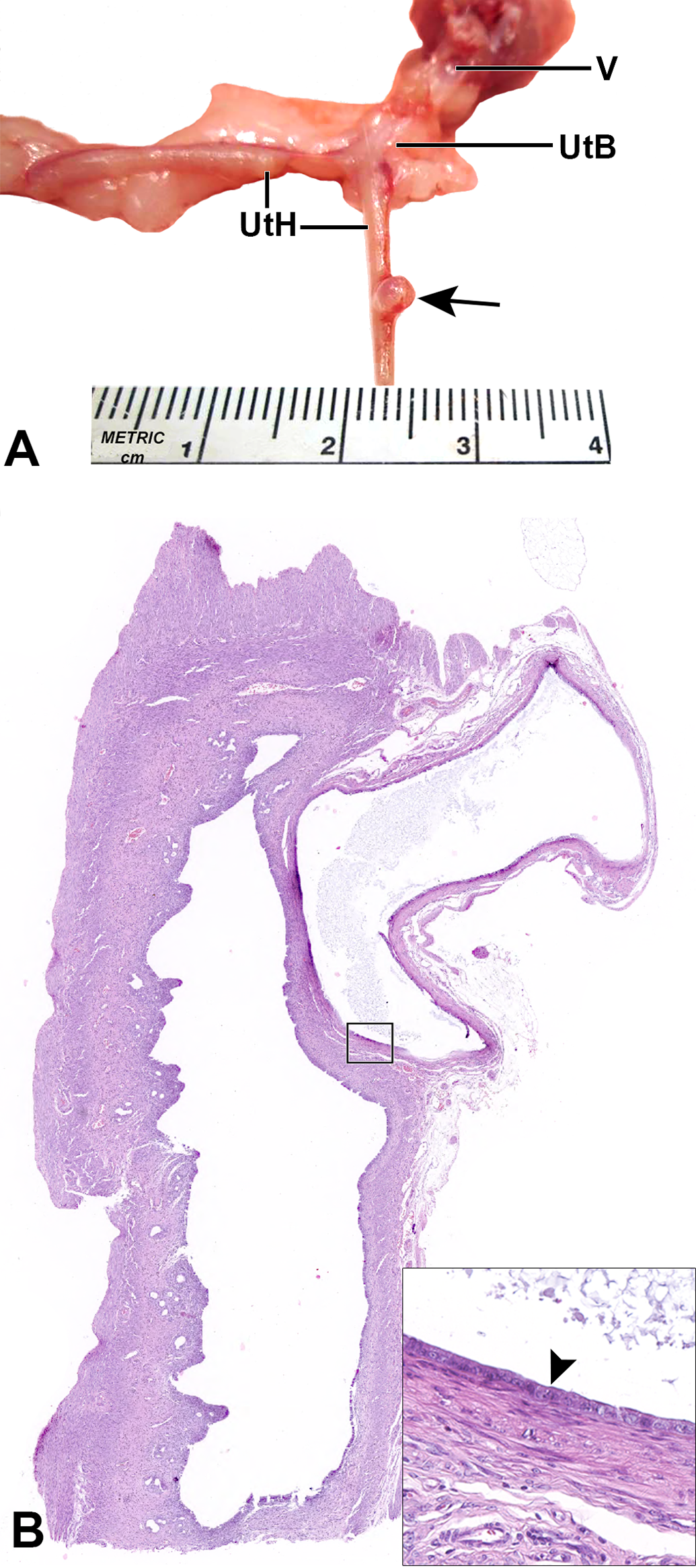
Uterine subserosal cyst from a SD rat. A shows a 3-mm raised, fluid-filled cyst (arrow) on the left uterine horn (UtH). B is a low-magnification image of the cyst that is present between the serosa and the outer myometrial layer and not associated with uterine cavity. The inset shows the cyst lined by a single layer of flattened cuboidal epithelium (arrowhead). Note, lightly basophilic staining flocculent intraluminal material. SD = Sprague-Dawley, UtB = fused uterus and cervix, V = vagina.
Gross Appearance and Localization of Uterine Cysts in Sprague-Dawley Rats.
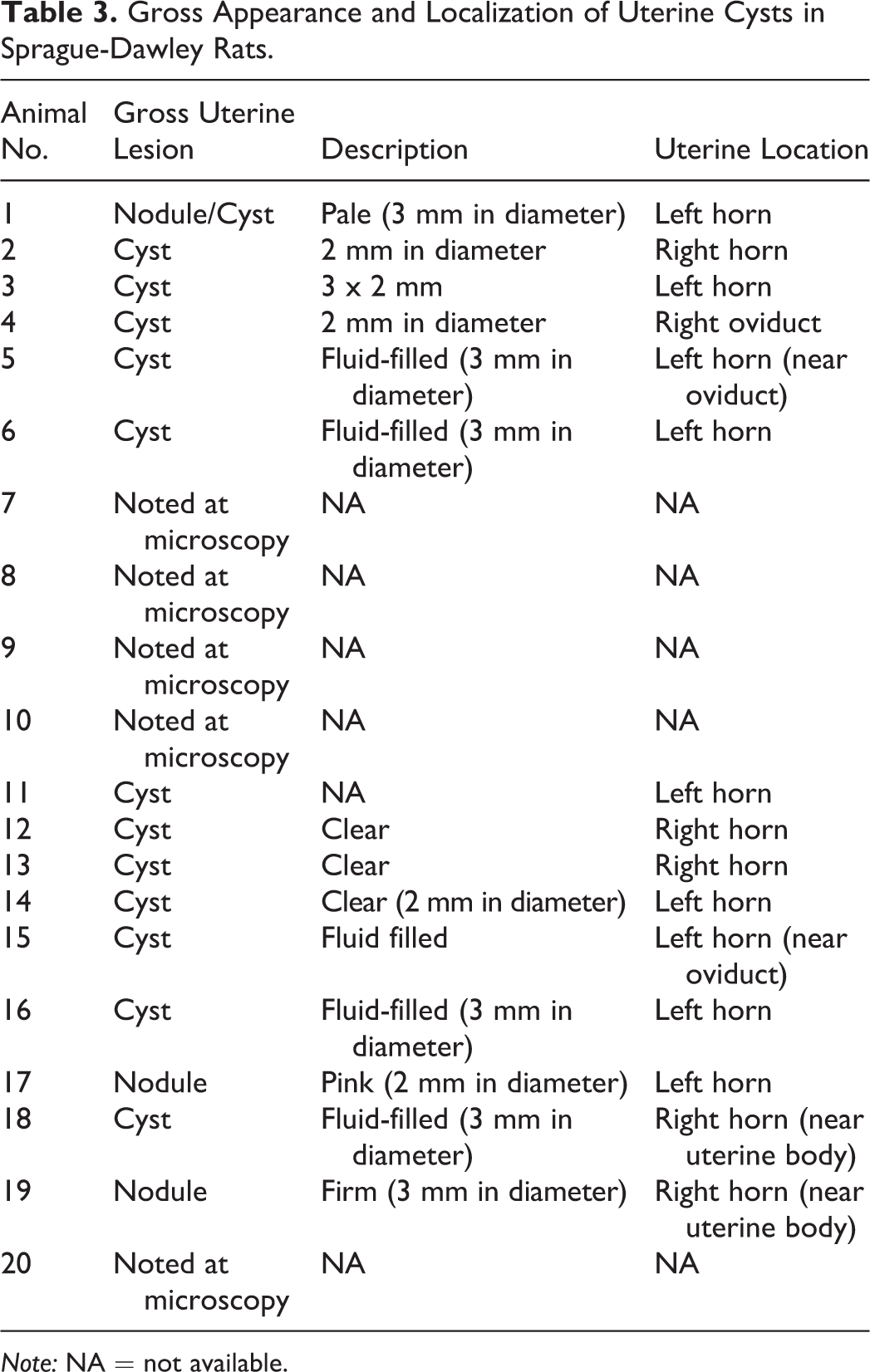
Note: NA = not available.
Histopathology and Histochemical Staining of Subserosal Uterine Cysts
Uterine subserosal cysts had well-defined walls of variable thickness with an average measurement of 91 μm. The walls were not associated with the endometrial cavity. The majority of cysts were lined by a single layer of flattened to cuboidal epithelial cells with random, focal cilia, moderate eosinophilic cytoplasm, and large oval nuclei containing 1 to 2 prominent nucleoli. One cyst was lined by more endometrium-like epithelium characterized by pseudostratified tall cuboidal cells with focal cilia and moderate to abundant foamy cytoplasm. Cysts often contained lightly basophilic staining flocculent intraluminal material (Figure 2A and C). Staining with Masson’s trichrome showed scant to moderate collagen fibers (blue) and abundant smooth muscle (red) present in the cyst walls (Figure 2D).
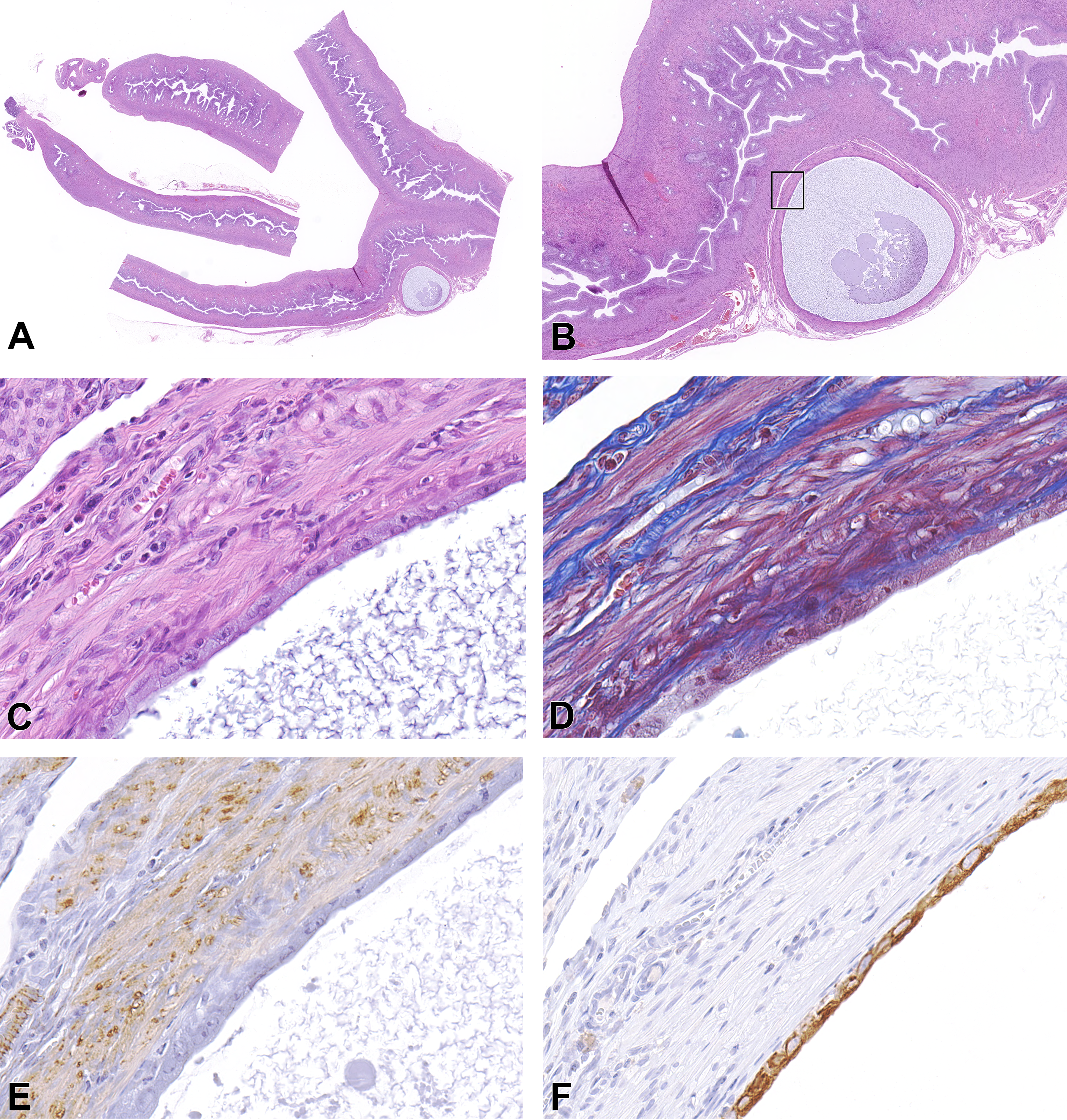
Uterine cyst from a SD rat. Low-magnification images of a second cyst filled with basophilic flocculent material located beneath the serosa and compressing the outer myometrial layer (A and B). C to F are higher magnifications of the cyst that is lined by flattened to cuboidal epithelium that has moderate eosinophilic cytoplasm and large oval nuclei with 1 to 2 prominent nucleoli. Endometrial cavity is not associated with the cyst (C, H&E). The wall is composed of scant to moderate blue staining of collagen and abundant red staining of muscle (D, Masson’s trichrome). E and F show positive immunostaining for SMA with moderate brown staining of muscle fibers (E) and intense brown cytoplasmic staining for CK-18 in cyst epithelial cells (F). CK-18 = cytokeratin 18, H&E = hematoxylin and eosin, SD = Sprague-Dawley, SMA = smooth muscle actin.
SMA, CK-18, PR, ER-α, GATA3, and PAX8 Immunohistochemistry
Uterine subserosal cysts were evaluated for immunopositive staining and given a quickscore for each marker. All cyst walls expressed moderate cytoplasmic immunoreactivity for SMA in muscle fibers, and epithelium of all cysts had diffuse, intense cytoplasmic staining for CK-18 (Figures 2E and 2F). A cyst located at the junction of the uterine cervix and uterine horn showed typical histological characteristics of the other observed uterine subserosal cysts (Figure 3A and B). Evaluation of cysts for hormone receptors showed PR was present in the epithelium and expressed as multifocal weak to moderate nuclear and weak cytoplasmic staining (Figure 3C; Table 4); this staining pattern was observed in of 6 of the 10 cysts. Also, nuclear staining for PR was observed in the smooth muscle of the cyst wall and adjacent endometrial stroma. Additionally, 9 of the 10 cysts had weak to intense nuclear staining for ER-α (Figure 3D, Table 4). Each cyst was immunostained for the transcription factors PAX8 and GATA3 to determine paramesonephric or mesonephric origin. We found diffuse, moderate to intense nuclear staining of epithelia for PAX8 in all cysts. The majority of cyst epithelia were negative for GATA3, with the exception of 3 cysts with multifocal, weak nuclear staining in less than 20% of epithelial cells (Figure 3E and F, Table 4). The endometrial epithelial cells of known paramesonephric origin also stained positive for PAX8 and were negative for the mesonephric marker GATA3. Based on immunohistochemistry quickscores for each marker, characteristically, cyst walls expressed moderate positive staining for SMA; and epithelia stained strongly positively for CK-18 and PAX8, moderately for ER-α, and weakly for PR. Furthermore, with a mean quickscore of 1, GATA3 staining was considered negative for cyst epithelia (Figure 4).
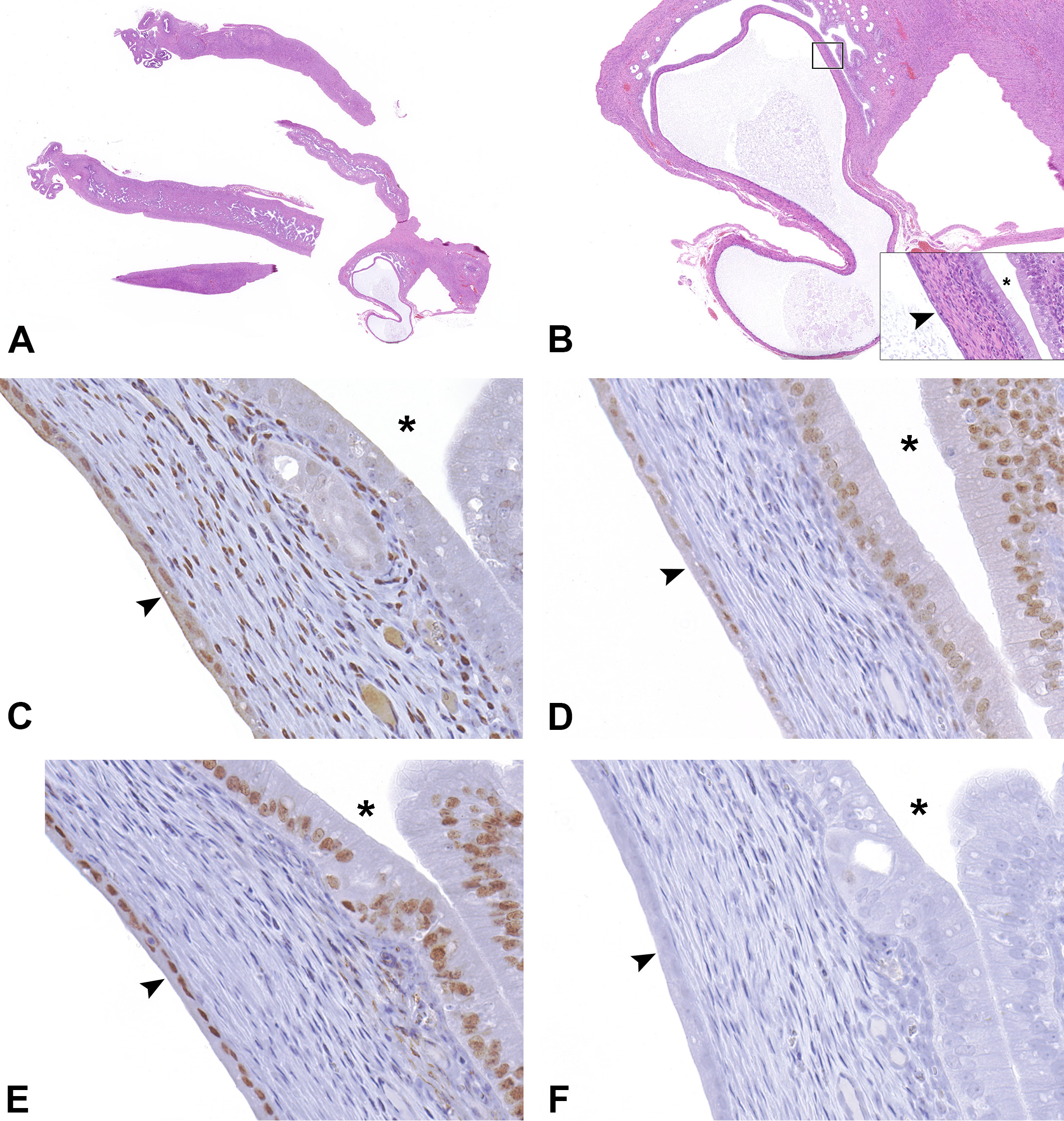
Uterine cyst in SD rat. Low-magnification images of a third cyst show similar features of the previous cysts in Figures 1 and 2 (A and B). The higher magnification inset in B demonstrates the differences of the tall columnar endometrial epithelium (adjacent to asterisk) and the flattened cuboidal cyst epithelium (arrowhead). C to F are high-magnification images of the cyst and are representative of immunohistochemical staining patterns of the cyst. Brown staining indicates positive reaction to the indicated antibody. Cyst epithelium (arrowhead) has multifocal weak to moderate nuclear and weak cytoplasmic staining for PR, while endometrial epithelium (adjacent to asterisk) has negative to weak staining (C). Nuclear staining for ER-α was weak to intense in both cyst and endometrial epithelium (D). There is diffuse moderate to intense nuclear staining in the epithelium and endometrium for PAX8 and negative staining for GATA3 (E and F, respectively). ER-α = estrogen receptor α, PAX8 = paired box 8, PR = progesterone receptor, SD = Sprague-Dawley.
Estrous Cycle Stage and Immunohistochemical Staining Intensity of Uterine Cysts in Sprague-Dawley rats.
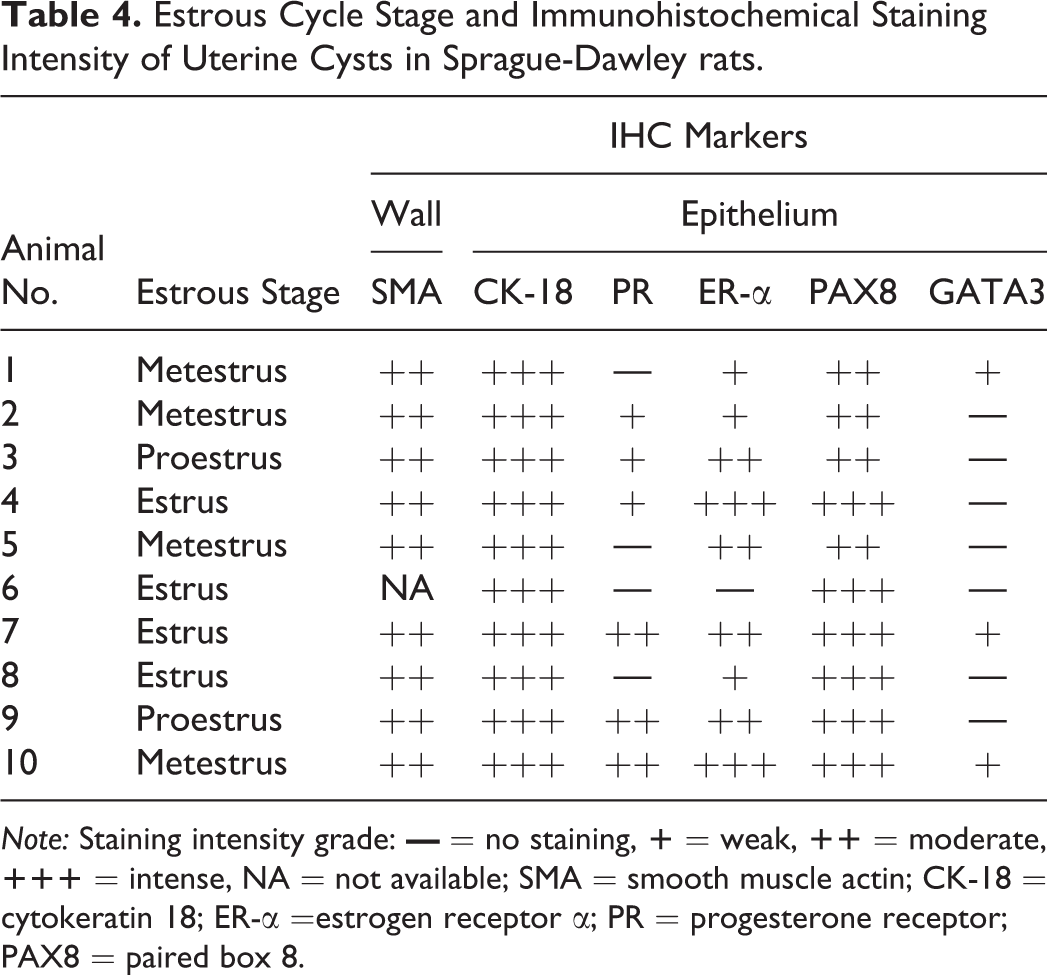
Note: Staining intensity grade:
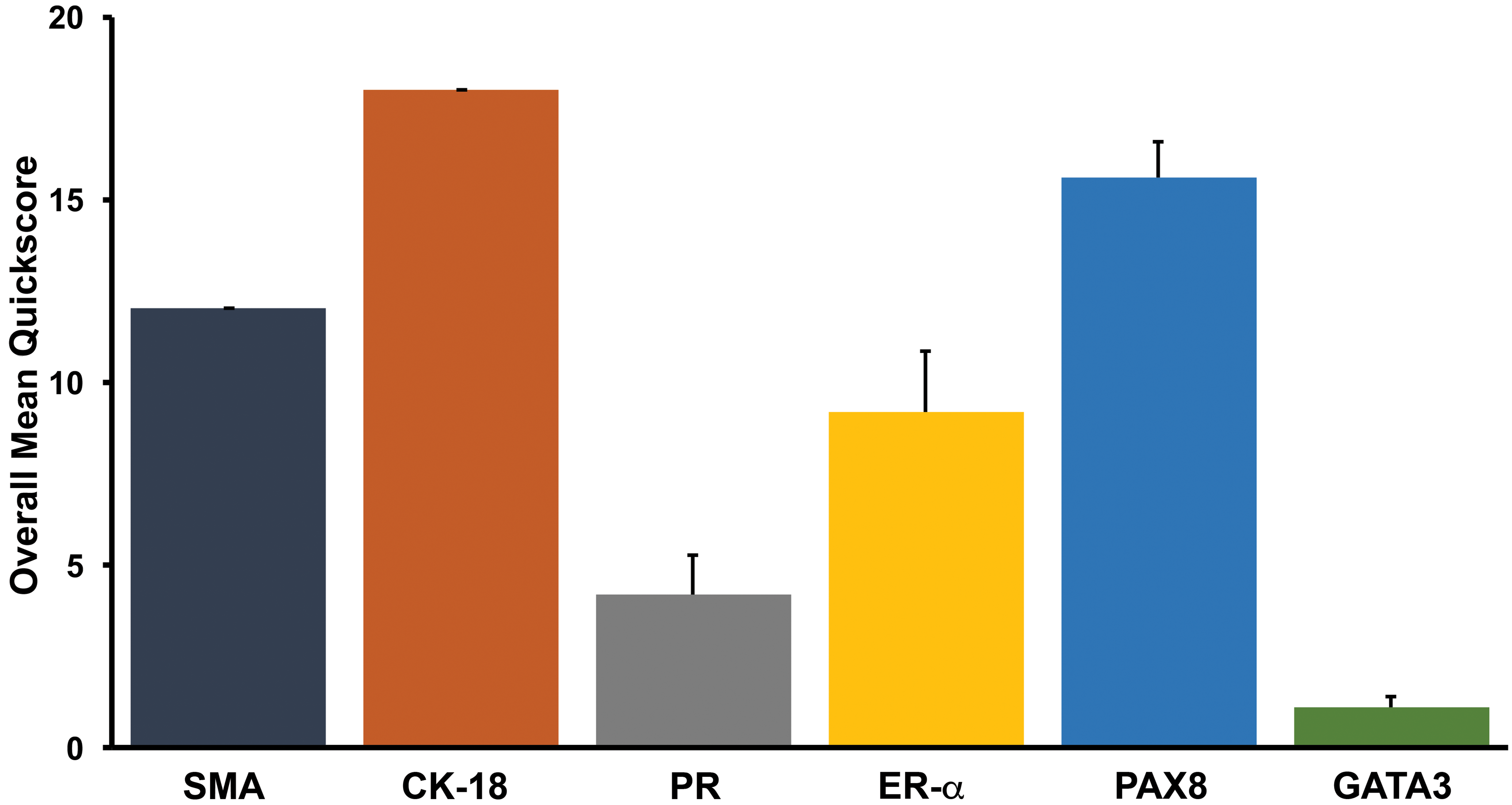
The overall average quickscores of immunohistochemical markers evaluated in uterine subserosal cyst epithelium or wall were categorized as 0 to 1 = negative, 2 to 6 = weak, 7 to 12 = moderate, and 13 to 18 = strong. Overall, cyst walls displayed moderate positive staining for SMA; and epithelia were strongly positive for CK-18 and PAX8, moderate for ER-α, weak for PR, and negative for GATA3. Data are expressed as mean + standard error of mean. CK-18 = cytokeratin 18, ER-α = estrogen receptor α, PAX8 = paired box 8; PR = progesterone receptor, SMA = smooth muscle actin.
Estrous Cycle and Cyst Immunophenotype
The uterus, cervix, and vagina were examined to determine the stage of estrous cycle within the SD rats. Animals presented in various stages of the estrous cycle. Two rats were in proestrus, and the remaining animals were equally divided into estrus and metestrus phases. There was no correlation between estrous cycle stage and expression of the various immunomarkers in the 10 rats evaluated (Table 4).
Discussion
Subserosal, unilocular cysts present along the uterine horns and uterine body from SD rats used in NTP multigeneration studies were evaluated to determine their origin. In the uterus, cysts are classified as acquired or congenital. There are several causes of acquired cystic lesions. Degeneration within leiomyomas can result in cytic cavities that lack an epithelial lining and are definitively dissimilar to the cysts characterized in this study. Other causes of cyst-like lesions include adenomyosis, which is characterized by the presence of endometrial glands and associated endometrial stroma located within the myometrium and the absence of a smooth muscle wall (Neri and Eckerling 1966; Parrott et al. 2001). Adenomyosis is not consistent with the cysts reported herein due to the lack of endometrial stroma directly associated with the cyst epithelium and the presence of a muscular wall underlying the cyst epithelia. The observed cysts are also unlike serosal inclusion cysts that develop from the invagination of the serosa into the adjacent connective tissue and are associated with aged, pluriparous animals (McEntee 1990b, Arnold et al. 1996; Godfrey and Silkstone 1998). Histologically, uterine serosal inclusion cysts are thin walled, lined by flattened cuboidal mesothelial cells, and contain clear fluid. These cysts may appear similar to those described here; however, the cysts that we characterized did not display the typical mesothelial lining cells or thin walls of serosal inclusion cysts and were also present in young nulliparous rats.
The subserosal uterine cysts in this study are consistent with the criteria for congenital uterine cysts (Sherrick and Vega 1962). This is supported by the observations that all cysts had smooth muscle walls and were lined by cuboidal epithelium. Cyst epithelia were not associated with the endometrial cavity, glands, or stroma. All cyst walls were located immediately subjacent to or directly associated with the outer myometrium. Therefore, based on our histological evaluations, the subserosal uterine cysts observed in 10 SD rats could be characterized as congenital uterine cysts derived from mesonephric or paramesonephric ducts.
Distinguishing between mesonephric duct– and paramesonephric duct–derived tissue is difficult due to overlapping histological characteristics and anatomical locations. In humans, mesonephric cysts are described as having a prominent muscular wall lined by flattened epithelium, whereas paramesonephric cysts have less prominent smooth muscle and columnar epithelium (Sherrick and Vega 1962). In this study, only one of the 20 paramesonephric cysts observed in the rats had columnar epithelium, while the rest generally had cuboidal epithelium. This difference in epithelium in the rats could be a result of compression from intraluminal fluid accumulation or could represent a true species-related variation. In women, neoplastic and nonneoplastic tissues derived from the mesonephric duct have not shown immunoreactivity for estrogen or PRs (Devouassoux-Shisheboran, Silver, and Tavassoli 1999; Silver et al. 2001). This is not true for tissues derived from paramesonephric origin, which have been shown to express PR and ER-α (Goyal and Yang 2014; Wu et al. 2014). Both PR and ER-α are present within paramesonephric-derived tissues such as the uterus and oviduct but have been shown to have variable expression during the estrous cycle in rats (Wang, Eriksson, and Sahlin 2000). Therefore, the lack of immunostaining for steroid hormone receptors does not rule out tissue of paramesonephric duct origin. Although there was variable staining intensity for PR and ER-α, all but one cyst in this study were positive for one or both receptors, suggesting that these cysts are most likely of paramesonephric origin.
PAX8 is a transcription factor critical in the development of organs derived from the embryonal duct, and its expression has been found in paramesonephric-derived epithelium of the uterus and has been used to identify paramesonephric-derived tumors in humans (Laury et al. 2011; Ozcan et al. 2011; Heidarpour and Tavanafar 2014; Liang et al. 2016). However, it has been shown that mesonephric neoplasms can also be immunopositive for PAX8, suggesting that its expression is present in both paramesonephric and mesonephric derived tissues, and that the use of immunohistochemical staining panels may be most appropriate in identifying neoplastic or nonneoplastic tissue of mesonephric or paramesonepric duct origin (Roma, Goyal, and Yang 2015; Goyal and Yang 2014). In a study by Goyal and Yang (2014), the authors evaluated the expression of PAX8, p16, and ER in differentiating mesonephric proliferations and cervical adenocarcinomas and concluded that a panel of immunohistochemical stains consisting of PAX8, ER, and p16 is useful in the distinction between mesonephric proliferations and cervical adenocarcinomas. All cysts in this current study displayed intense immunoreactivity for PAX8 and as mentioned above also expressed PR and ER-α, with the latter receptors reported not to be expressed in mesonephric-derived tissues, thus strengthening support of a paramesonephric duct origin for our identified cysts.
The transcription factor GATA3 has an important role in organ development including the urogenital tract originating from the mesonephric duct (Grote et al. 2006). Developmentally, GATA3 is not expressed in endocervical and endometrial epithelium but is reported to be expressed in benign and malignant uterine mesonephric lesions in women (Roma, Goyal, and Yang 2015). Others, however, have found negative to focal weak GATA3 expression in endometrial adenocarcinomas (Miettinen et al. 2014; Howitt and Nucci 2017). Both mesonephric and paramesonephric ducts are derived from the intermediate mesoderm, and coelomic epithelium lines the developing paramesonephric duct (Mullen and Behringer 2014). Gene expression and signaling events in the intermediate mesoderm determine differentiation of both ducts. Mesenchymal expression of GATA3 regulates mesonephric duct development and migration, while Wnt signaling is required for paramesonephric duct initiation and elongation (Grote et al. 2006; Yucer et al. 2017). Additionally, GATA3 is considered an early marker of mesonephric duct differentiation (Yucer et al. 2017); therefore, it is plausible that gene expression or signaling alterations affecting the intermediate mesoderm and/or epithelial differentiation could result in GATA3 expression in addition to PAX8 expression in paramesonephric-derived epithelium. In our study, the majority of the cysts had no immunoreaction for GATA3, and only 3 cysts showed weak staining in <20% of epithelial cell nuclei, in addition to moderate to intense nuclear staining for PAX8. A recent review of mesonephric proliferations in the reproductive tract of women reported PAX8 and GATA3 expression in mesonephric remnants and cysts and concluded that mesonephric remnants/hyperplasia is immunohistochemically characterized by strong, diffuse GATA3 and negative ER/PR, as well as Cluster of differentiation 10 (CD10) apical expression (Howitt and Nucci 2017). Based on the findings of the presence of moderate to intense PAX8 and negative to weak focal GATA3 expression in only 3 cysts that also stained positive for PAX8, in addition to the expression of ER-α and PR, the cysts described in this article were considered to be most consistent with a paramesonephric phenotype.
The pathogenesis of these uterine cysts is unknown. In females, paramesonephric cysts can arise throughout the reproductive tract and are thought to be entrapments of the embryological tissue during organ differentiation (Eilber and Raz 2003). In contrast, it is thought that mesonephric cysts arise within persistent mesonephric ducts and occur in aged animals (McEntee 1990b). In this study, the subserosal uterine cysts did not appear to be an age-related change because all rats were less than one year of age. In male Cesarean Derived 1 (CD-1) mice prenatally exposed to a synthetic estrogen, diethylstilbestrol (DES), cysts occurred in the epididymis associated with estrogen-induced retention of the paramesonephric duct (Newbold, Bullock, and McLachlan 1987). Similarly, paramesonephric cysts have been reported in the spermatic cord of otters in Sweden, and authors have postulated that environmental endocrine disruptor chemical exposures associated with estrogens or estrogen-like compounds as a potential etiology (Roos and Ågren 2013). Additionally, DES was shown to cause ovarian and vaginal abnormalities including presumed mesonephric-derived paraovarian cysts, vaginal adenomyosis, and prominent mesonephric duct remnants in the cervicovaginal region in prenatally exposed female CD-1 mice (Newbold and McLachlan 1982; Haney et al. 1986). Furthermore, prominent mesonephric remnants were noted in the uteri of CD-1 mice prenatally exposed to the environmental estrogenic chemical, bisphenol A (Newbold, Jefferson, and Padilla-Banks 2009). In our retrospective study, 3 of the 10 cysts were in nontreated rats, and the remainder of the rats were exposed to various doses of 5 different chemicals. The effects of estrogenic chemicals were not addressed, and therefore, we cannot corroborate the role of estrogen in the pathogenesis of these cysts.
Congenital uterine cysts arising from mesonephric and paramesonephric ducts are rarely described in the human and veterinary literature and are often incidental findings (Sherrick and Vega 1962; Gelberg and McEntee 1986; McEntee 1990a). It is generally thought that these cysts arise primarily from remnants of the mesonephric duct. A recent study of three cases of mesonephric duct remnants in bitches found cysts lined by ectopic endometrial epithelium, glands, and stroma, which were immunopositive for cytokeratin, ER, and PR (Bartel, Berghold, and Walter 2011). In contrast, the epithelial-lined cysts reviewed in our study lacked endometrial glands and stroma. We have confirmed that subserosal uterine cysts with histomorphologic features more consistent with those previously described for mesonephric duct cysts were more phenotypically consistent with cysts derived from the paramesonephric duct based on an immunohistochemical panel of positive staining for PR and/or ER-α and PAX8 with negative staining for GATA3. It is very difficult to distinguish between cysts of paramesonephric and mesonephric duct origin without immunohistochemical staining, and therefore, it is possible that paramesonephric cysts may be misidentified as mesonephric cysts in the rodent uterus. Our findings are important because we have identified and described developmental subserosal uterine cysts in rats that are of paramesonephric duct origin, which has not been previously reported or characterized in the rat pathology literature. Additionally, we present an IHC panel that can be used in rodent tissue to identify cysts of paramesonephric duct origin.
Footnotes
Acknowledgments
The authors would like to thank Drs. Cynthia Willson and Mark Cesta for their critical review of this manuscript. The authors would also like to thank the Histology Laboratory and the Immunohistochemistry Core, NIEHS, NTP, and Mary Ellen Sutphin and Kylie Brockenfelt for their help with data acquisition. The authors greatly appreciate the technical assistance of Ms. Elizabeth Ney in compilation of image plates.
Author Contributions
Authors contributed to conception or design (DJ, JS, DD); data acquisition, analysis, or interpretation (DJ, JS, RH, DM, BM, PF, DD); drafting the manuscript (DJ, JS, DD); and critically revising the manuscript (DJ, JS, RH, DM, BM, PF, DD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
