Abstract
The Tg.rasH2 model was accepted by regulatory agencies worldwide for 26-week carcinogenicity assays as an alternative to the standard 2-year assays in conventional mice in 2003. Several references documenting spontaneous nonneoplastic findings and incidences of spontaneous tumors in the Tg.rasH2 mice have been published. The purpose of this publication is to add adrenal gland subcapsular hyperplasia to the database pertaining to spontaneous lesions noted in Tg.rasH2 mice, review physiology related to this finding, and discuss its significance. The incidence of spontaneous subcapsular adrenal gland hyperplasia was determined in control Tg.rasH2 mice from nine 26-week carcinogenicity studies carried out within the last 5 years at two contract research organizations. Incidence of this finding ranged from 56% to 79% in males and 88% to 100% in females, with an incidence average of 62% in males and 93% in females. Adrenal gland subcapsular hyperplasia is a common finding in male and female Tg.rasH2 mice that did not progress to neoplasia in Tg.rasH2 mice. In general, it tends to be more frequent and severe in females in comparison to males.
Keywords
For over a decade, the Tg.rasH2 model has been accepted by regulatory agencies worldwide for use in 26-week carcinogenicity assays as an alternative to the standard 2-year assays in conventional mice (Paranjpe et al. 2013). Continuous updating of prepublished references documenting spontaneous background findings and incidences of spontaneous tumors in the Tg.rasH2 mice is necessary to ensure robustness of historical control data in this relatively contemporary strain. Adrenal gland subcapsular hyperplasia has been noted in our studies as a common lesion in control Tg.rasH2 mice.
Adrenal glands are bilateral endocrine organs located rostral to the anterior poles of the kidneys. The right adrenal gland is in closer proximity to the kidney than the left. The adrenal gland is composed of a cortex and medulla surrounded by a fibrous connective tissue capsule (Frith 1996; Sass 1996). Female adrenal glands are approximately 25% larger than males (Figures 1 and 2), at least in part due to greater lipid content in females (Frith and Dunn 1994; Yarrington 1996). The adrenal gland secretes hormones that affect numerous physiologic processes, most notably stress responses, immune function, and cardiovascular regulation. Growth and function of the adrenal glands are markedly influenced by gender and age (Bielohuby et al. 2007).
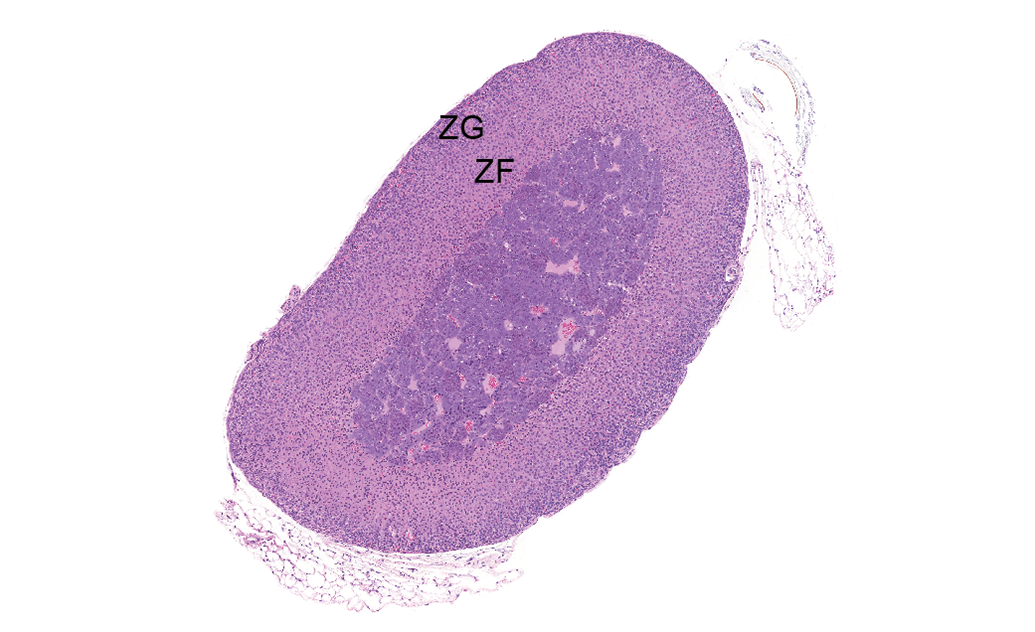
Control male adrenal gland. Mice do not have a perceptible zona reticularis, and the zona glomerulosa (ZG) and zona fasciculata (ZF) do not have a distinct separation between them, but are labeled for general location. Hematoxylin & eosin stain; original magnification 3×.
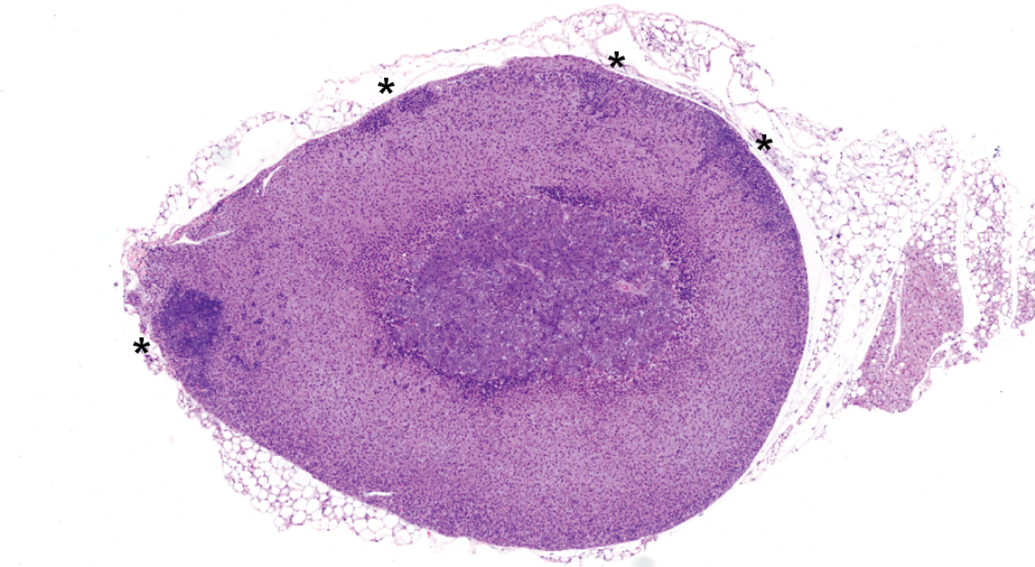
Control female adrenal gland (with multiple foci of subcapsular hyperplasia, asterisks). Note larger size of female adrenal gland compared to male in Figure 1. Hematoxylin & eosin stain; original magnification 3×.
Subcapsular hyperplasia in the cortex has been recognized as a frequent finding in inbred mice, first occurring around 4 months of age and reaching incidences of 59% in males and 91% in females by 19 months (Yoshida, Maita, and Shurasu 1986; Taylor 2011). Histologically, proliferation is surface-oriented; often extends toward the medulla; and may be focal, multifocal, or circumferential in distribution, with frequent wedge-shape distribution. With increasing severity, there may be minimal compression of the underlying cortex or slight bulging of the adrenal surface without capsular invasion (Nyska and Maronpot 1999; Capen et al. 2001). Subcapsular hyperplasia originates from glomerulosa cortical cells and can present with phenotypic variability (Nyska and Maronpot 1999). Type A cells are small, basophilic spindloid cells with scant cytoplasm and lacking lipid vacuoles. Type B cells are lipid-laden, larger, and polyhedral. Mixed lesions are those in which there is approximately an even mixture of type A and type B cells (Goodman 1996).
Of course, the recognition of background findings with respect to species, strain, sex, and age is crucial for proper interpretation of xenobiotic-induced lesions. The aim of this compilation is to provide the range and incidence of spontaneous adrenal subcapsular hyperplasia in Tg.rasH2 mice from 26-week carcinogenicity studies carried out at Envigo and BioReliance. This report presents quantitative data across 9 studies and can serve as a historical control reference when submitting nonclinical data to, and corresponding with regulatory authorities, as well as guidance for the study and peer review pathologists in diagnosing and interpreting this finding.
Materials and Method
Studies and Experimental Design
These data were assembled based on samples from a total of 450 (225 males, 225 females) Tg.rasH2 mice from negative control groups (untreated or vehicle-treated) by oral gavage across nine 26-week oral gavage carcinogenicity studies conducted within 5 years at Envigo (Princeton Research Center, Somerset, NJ) and BioReliance (Rockville, MD). On the first day of treatment, animals were 6 to 10 weeks old and weighed 15 to 26 grams.
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the Envigo or BioReliance Institutional Animal Care and Use Committees. Research was conducted in compliance with the Animal Welfare Act, and other federal statutes and regulations relating to animals as stated in the Guide for Care and Use of Laboratory Animals). Envigo CRS, Inc. and BioReliance are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The studies were conducted in compliance with principles of Good Laboratory Practice Standards as set forth in the U.S. Food and Drug Administration’s Good Laboratory Practice Regulations (Part 58 of 21 CFR-Code of Federal Regulations).
Animals
CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) mice, obtained from Taconic Farms (Germantown, NY), were used in all studies. The knock-in Tg element (human prototype c-Ha-ras gene with its own promoter/enhancer) is injected into C57BL/6 × BALB/c F2 zygotes, which are crossed back to C57BL/6 J forming C57BL/6 Jic-Tg(HRAS)2Jic. The CByB6 F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mouse with the BALB/cByJJic female mouse. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study.
Housing and Environmental Conditions
Animals were either single- or group-housed in solid bottom cages with cellulose-based contact bedding or hardwood bedding chips. Up to five animals of the same sex and group were housed in each cage. An enrichment device was provided in each cage at all times, as well as shelters. Temperature and humidity were monitored in accordance with Testing Facilities’ SOPs (standard operating procedures).
Animals had free access to food (Certified Rodent Diet, No. 2016C [Envigo Teklad, Madison, WI or Harlan TEKLAD Global Diet, Madison, WI]) and water ad libitum.
Study Termination and Pathological Examination
Animals were humanely euthanized using isoflurane followed by exsanguination, or CO2 overdose. Comprehensive necropsy was performed on all animals at study termination or on the day of death for animals found dead or euthanized in moribund condition prior to study termination. Tissues were fixed by immersion in 10% neutral-buffered formalin, embedded in paraffin wax, sectioned to a thickness of 4 to 5 micrometers, mounted onto glass slides, stained with hematoxylin and eosin, and coverslipped. Microscopic data from all studies were recorded by direct computer entry by the study pathologist. All studies underwent pathology peer review, and all data were reviewed by the Quality Assurance Department at Envigo or BioReliance prior to the release of the final pathology report. Data presented here are from untreated control groups only.
Results
Incidence of spontaneous adrenal subcapsular hyperplasia in 225 male and 225 female Tg.rasH2 mice is presented in Table 1. Lesion severity was graded microscopically using a 4- or 5-point scale (minimal to marked or minimal to severe, respectively). Since the severity of subcapsular hyperplasia never exceeded moderate, only the incidence of the lesion has been provided in Table 1. Lesions of moderate severity were diagnosed least frequently and usually in females. Severity and incidence was consistently higher in females.
Incidence of SCH in Tg.rasH2 Mice Used in 26-week Carcinogenicity Studies.
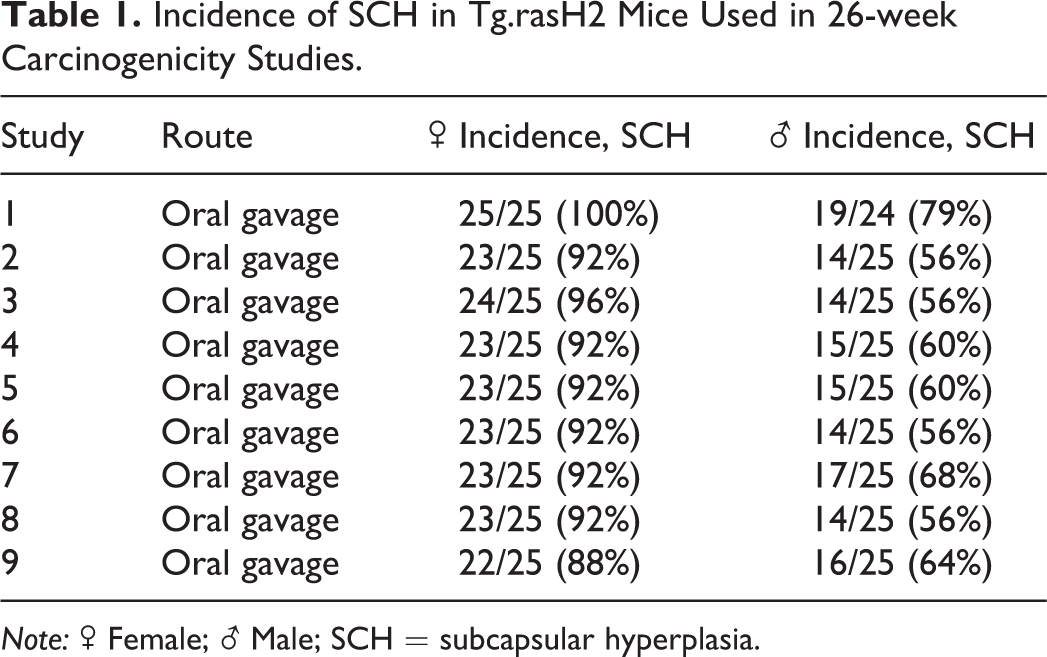
Note: ♂ Female; ♀ Male; SCH = subcapsular hyperplasia.
Incidences of subcapsular hyperplasia ranged from 56% to 79% in males and 88% to 100% in females, with the overall average incidence of 62% in males and 93% in females. No subcapsular neoplasia nor cortical adenomas or carcinomas were noted in any control animals in our studies, and there have been no reported incidences of these tumors in Tg.rasH2 mice to date. This suggests that subcapsular hyperplasia is unlikely to undergo neoplastic transformation in Tg.rasH2 mice up to 36 weeks old (maximum age at study termination). Furthermore, chemically induced proliferative lesions have been reported as uncommon in the adrenal cortex of nonclinical species (Rosol et al. 2001).
The high incidence of subcapsular hyperplasia in both sexes and greater severity in females in our analysis were consistent with the previous literature pertaining to more aged inbred mice (Hoenerhoff et al. 2014). Hyperplasia of type A (spindle) cells was the predominant phenotype. Early lesions (Figure 3) were subtle and characterized by small aggregates arranged subjacent and parallel and/or perpendicular to the capsule. With further proliferation, there was vertical and horizontal expansion of spindle cells between cords of the zona fasciculata (Figure 4). Larger hyperplastic foci (Figures 5 and 6) were composed of fusiform cells with rare polygonal cells (type B) and arranged in nests, which sometimes caused slight bulging of the capsule.
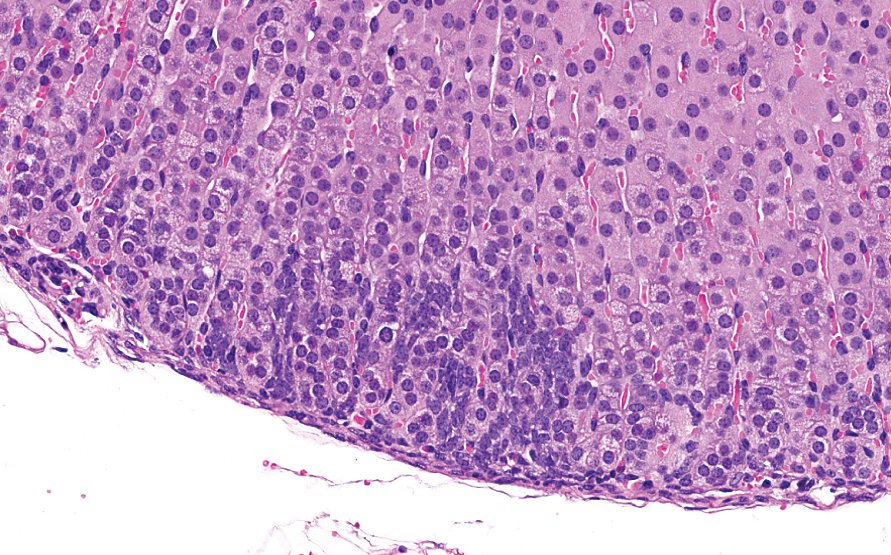
Minimal subcapsular hyperplasia, best appreciated on higher magnification, characterized by small aggregates of (type A) spindle cells arranged subjacent to the capsule. Hematoxylin & eosin stain; original magnification 20.4×.
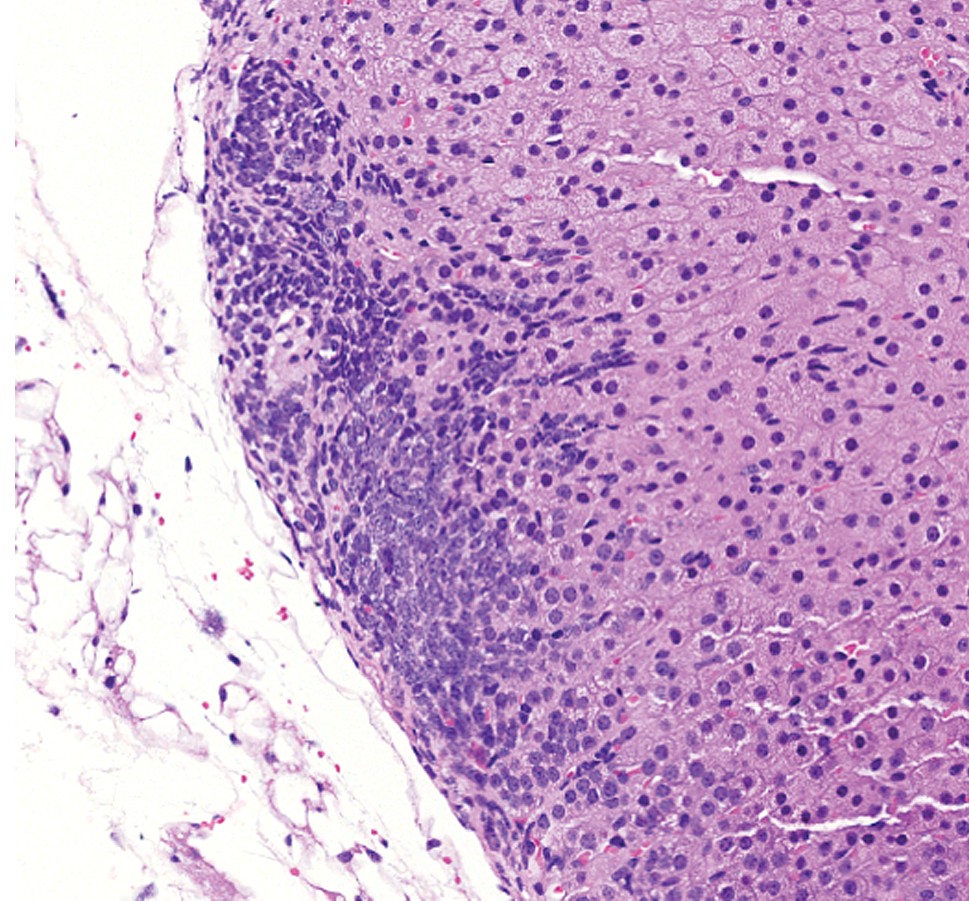
More pronounced subcapsular hyperplasia with vertical and horizontal expansion of spindle cells between cords of the zona fasciculata. Hematoxylin & eosin stain; original magnification 12.8×.
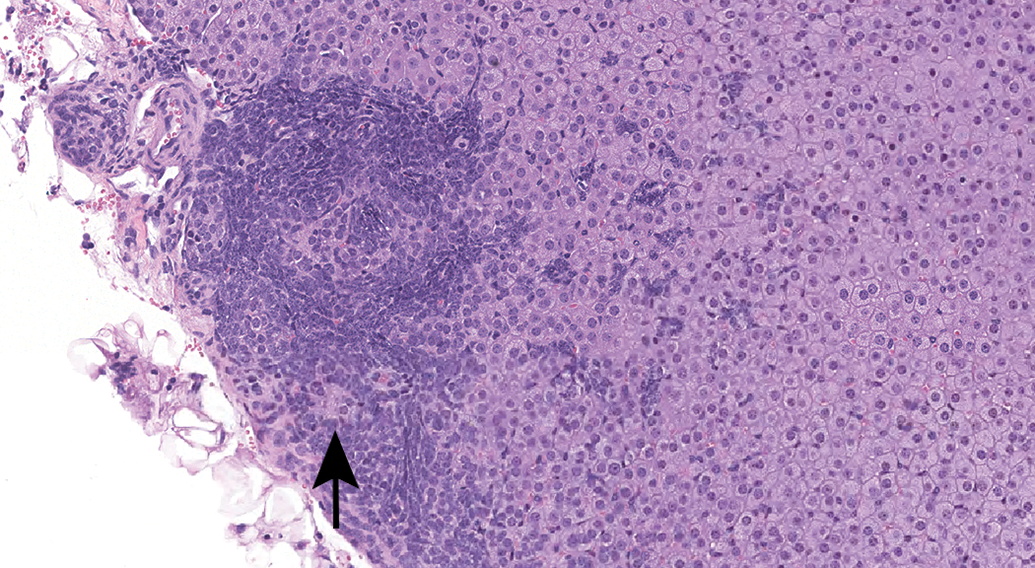
Subcapsular hyperplasia composed of spindle or fusiform cells and rare polygonal cells (type B, arrow), with arrangement in nests. Hematoxylin & eosin stain; original magnification 12.8×.
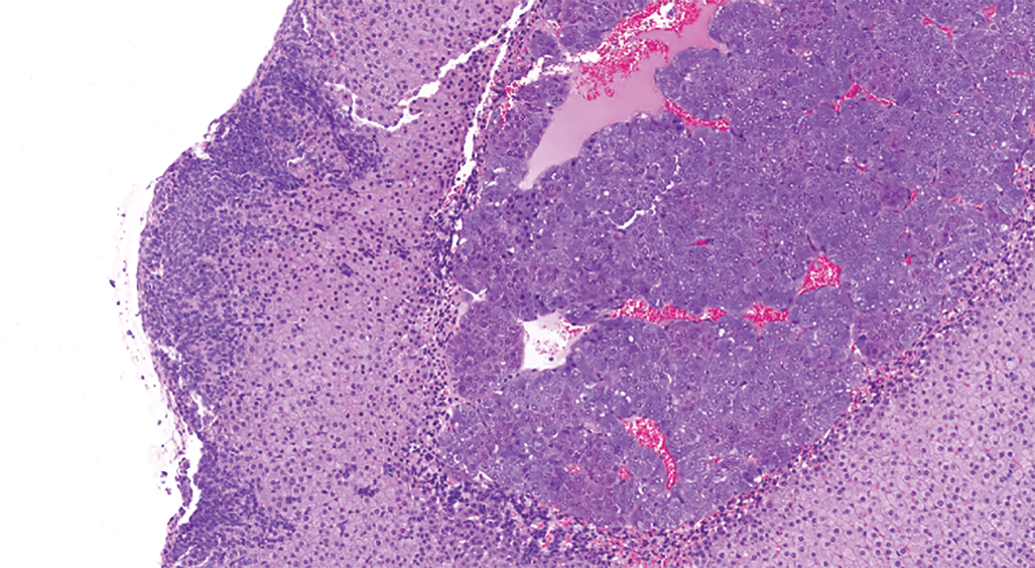
Subcapsular hyperplasia with bulging of the capsule. Hematoxylin & eosin stain; original magnification 12.4×.
Discussion
Adrenal subcapsular hyperplasia is thought to be related to hormonal changes provoked by variations in steroidogenesis in the endocrine system and postpubertal reproductive system. The increased incidence and severity in aging females may be related to decreased ovarian function and/or the overall larger size of their adrenal glands in comparison to males. Gonadectomy has been reported to hasten and exacerbate its occurrence in both sexes, and hormone treatment has been reported to transform type A cells into type B cells (Frith 1996; Nyska and Maronpot 1999). Stressful habitation conditions, crowding or isolation, have been suggested to increase incidence, although numerous confounding factors (gastritis, markedly reduced food consumption, slightly decreased mean body weights, and a smaller variance of body weights) also occurred in densely housed animals in this frequently cited study (Chvedoff et al. 1980). Since incidences of subcapsular hyperplasia were high across all studies with increased incidence in females, individual versus group housing conditions and different bedding materials do not appear to significantly impact data. Stress in humans has been shown to induce zona fasciculata encroachment in the subcapsular area generally occupied by the zona glomerulosa (Aiba et al. 2011).
Mast cells have been seen accompanying subcapsular hyperplasia in some strains of inbred mice, most notably, A/J, BALB/c, C57BL/6, DBA/2J, and IQI/Jic mice (Kim et al. 1997), though mast cells were not noted as a conspicuous feature in Tg.rasH2 mice. Mast cells may contribute to subcapsular pathogenesis by producing proteases and a range of multifunctional cytokines, particularly stem cell factor and nerve growth factor (Kim et al. 2000). Interestingly, in humans, mast cell infiltration has been associated with aldosterone hypersecretion in a subset of cortical adenomas (Duparc et al. 2015).
Subcapsular hyperplasia is rarely seen in other species as a common background finding and has not been reported in rats (Goodman 1996). “Spindle cell hyperplasia” or “Type A cell hyperplasia” are terms that are sometimes used synonymously with subcapsular hyperplasia (Greaves 2012). Neoplasms occurring in the subcapsular location have been reported in male hamsters, rats, and goats with enhanced growth occurring after castration (Goodman 1996). Adrenal tumorigenesis in humans may also occur after disruption of the HPG (hypothalamic–pituitary–gonadal) axis, especially in postmenopausal women, and men with testicular atrophy. Such patients have significant increases in gonadotropins, which occurs similarly in gonadectomized mice (Yates et al. 2013). In studies conducted through the National Toxicology Program, subcapsular hyperplasia was considered induced by benzene, vinylcyclohexene, and nitrofurantoin, in which it was seen concurrently with ovarian atrophy and cortical adenomas and carcinomas (nitrofurantoin; NTP Technical Report No. 289 1986; NTP Technical Report No. 303 1986; NTP Technical Report No. 341 1989).
The distinction between subcapsular hyperplasia and subcapsular adenoma can be challenging and to an extent, arbitrary, as consistency in standards used to differentiate the two is of utmost importance. Both lesions originate from glomerulosa cortical cells, and while progression from subcapsular hyperplasia to adenoma does occur, hyperplasia is not a prerequisite (Nyska and Maronpot 1999). Criteria which suggest subcapsular adenoma include extent/size of the lesion (6–15 millimeters in diameter), distinct and pronounced compression of surrounding tissue, and hormonal alterations, as type B cell adenomas may exert estrogenic, androgenic, or adrenocorticoid effects (Nyska and Maronpot 1999). Cortical adenomas are rare in mice and originate in the zona fasciculata and maintain eosinophilic/amphophilic cell quality (Capen et al. 2001; Frith and Dunn 1994; Nyska and Maronpot 1999).
Due to its reliably high background incidence in healthy control animals, some pathologists choose not to report subcapsular hyperplasia in routine studies (personal communications). However, we would recommend recording the finding to establish whether or not there is a treatment-related change in incidence. It has been suggested that because adrenal subcapsular hyperplasia is unlikely to progress to neoplasia, it can be deselected for peer review if time is a major factor (Mann and Hardisty 2013).
Conclusion
To the best of our knowledge, this is the only combined study of the incidences of adrenal subcapsular hyperplasia in Tg.rasH2 mice. References to the comprehensive incidences reported here should not only assist in the distinction between background findings from induced lesions should they occur in toxicological safety studies but also guide the study pathologist in reporting these data and further investigating and interpreting its translational relevance in man.
Footnotes
Author Contributions
Authors contributed to conception or design (MB, MP, DC); data acquisition, analysis, or interpretation (MB, MP, DC); drafting the manuscript (MB); and critically revising the manuscript (MB, MP, DC). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
